
Int J App Pharm, Vol 17, Issue 3, 2025, 101-111Review Article
TRANSFORMATIVE APPLICATIONS OF AI IN HEALTHCARE AND PHARMACEUTICAL INNOVATION
P. JASMINE1, ARWA IBRAHIM1, S. SIDDIQ1, A. DHANURSHRA1, M. NIRMALA1, S. SHAKIR BASHA2*
1Department of Pharmacy, Raghavendra Institute of Pharmaceutical Education and Research, K. R. Palli Cross, Chiyyedu Post, Anantapur-51572, India. *2Department of Pharmaceutical Analysis, Raghavendra Institute of Pharmaceutical Education and Research, K. R. Palli Cross, Chiyyedu Post, Anantapur-515721, India
*Corresponding author: S. Shakir Basha; *Email: shakirbasha72@gmail.com
Received: 20 Sep 2024, Revised and Accepted: 03 Apr 2025
ABSTRACT
Artificial Intelligence (AI) is transforming the drug development and Clinical Trials by improving efficiency, accuracy, and decision-making. AI predicts Pharmacokinetic (PK) and Pharmacodynamic (PD) properties, automates compound screening and enhances clinical testing throughput. In trial design, AI optimizes patient stratification and outcome prediction by analyzing vast datasets from previous trials and electronic health records, leading to cost-effective and adaptive trials. AI also facilitates real-time data monitoring, identifying discrepancies early to ensure data integrity and regulatory compliance. By integrating diverse data sources it streamlines clinical operations, reducing human error and manual workload. However, challenges persist in data quality and integration due to varying standards across sources, necessitating advanced harmonization techniques. Regulatory frameworks often lag behind AI advancements, creating uncertainty and potential delays. Ethical concerns, including patient privacy and data security, must also be addressed for responsible AI implementation. Establishing standardized protocols and ensuring regulatory alignment are critical for AI’s successful integration into clinical research. In conclusion, AI revolutionizes drug development and clinical trials, enhancing efficiency and accuracy. However, overcoming data, regulatory, and ethical challenges is essential for its widespread adoption.
Keywords: Artificial intelligence (AI), Pharmacokinetics (PK), Pharmacodynamics (PD), Clinical trials (CT), Reinforcement learning (RL), Natural language processing (NLP), Generative adversarial networks (GANs), Variational autoencoders (VAEs)
© 2025 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2025v17i3.52719 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Introduction to AI in healthcare
AI refers to the simulation of human intelligence in machines that are designed to think and learn like humans [1]. In healthcare, AI has a broad range of applications, from diagnostics to personalized treatment plans [2]. AI-powered systems analyze vast medical datasets to identify patterns and predict health outcomes, improving diagnostic accuracy [3]. For instance, AI algorithms assist in imaging techniques like X-rays, Magnetic Resonance Imaging (MRI), and Computed Tomography (CTG) scans to detect abnormalities with high precision [4]. Predictive analytics help identify patients at risk of developing certain conditions, enabling early intervention. Natural Language Processing (NLP), another AI application, processes medical literature and patient records to extract relevant clinical information, improving decision-making [5]. Additionally, AI streamlines administrative tasks such as scheduling and billing, reducing operational costs and allowing healthcare professionals to focus on patient care [6]. AI's integration into healthcare is transforming the industry by enhancing efficiency, accuracy, and patient outcomes [7].
AI in the pharmaceutical sector
AI is revolutionizing various aspects of the pharmaceutical industry, including drug discovery, clinical trails and manufacturing [8]. In drug discovery, AI analyzes large datasets to identify potential drug candidates faster and more accurately than traditional methods [9]. Machine learning models predict drug-target interactions, significantly reducing the time required for early-stage drug development [10]. AI optimizes clinical trails` designs by selecting suitable candidates, predicting trial outcomes, and monitoring patient responses in real-time, improving efficiency and success rates [11]. AI-driven pharmacovigilance enhances drug safety by detecting adverse drug reactions through post-market surveillance data analysis [12]. Additionally, AI optimizes manufacturing processes, increasing productivity and reducing costs [13]. By incorporating AI, pharmaceutical companies can accelerate drug development, lower costs, and deliver innovative therapies more efficiently [14].
AI-driven innovation in drug development
Traditional drug development is a lengthy and costly process with high failure rates due to efficacy and safety concerns [15]. Drug candidates often fail in clinical trails, making the process inefficient [16]. AI addresses these challenges by predicting compound activity, optimizing lead compounds, and enhancing clinical trail designs [17]. AI-powered analytics refine patient recruitment, monitor responses in real-time, and facilitate adaptive trial designs, reducing costs and timelines [18]. AI also improves post-market surveillance by identifying adverse effects early, ensuring compliance with regulatory standards [19]. These advancements significantly enhance the efficiency and accuracy of drug development [20].
Challenges and future directions
Despite its potential, AI faces challenges related to data integration, standardization, regulatory frameworks, and ethical concerns [21]. Data quality issues arise due to variations in standards, necessitating advanced harmonization techniques [22]. Regulatory bodies often struggle to keep pace with AI innovations, leading to uncertainty and delays in approval processes [23]. Ethical concerns, such as patient privacy and data security, must be addressed for AI to be widely accepted in pharmaceuticals [24]. Establishing comprehensive data standards and regulatory guidelines is crucial for AI’s successful implementation in drug development [25].
In conclusion, AI is transforming healthcare and pharmaceuticals by improving diagnostics, optimizing drug development, and enhancing clinicaltrails. While challenges remain, addressing data, regulatory, and ethical concerns will ensure AI’s full potential is realized, benefiting patients and the healthcare industry [26].
AI technologies and methodologies
Machine learning and deep learning
Reinforcement learning (RL)
RL enables an agent to learn optimal decisions through trial and error, receiving rewards or penalties based on actions [27]. Inspired by behavioral psychology, RL is valuable in healthcare and pharmaceuticals for optimizing treatment protocols, personalizing patient care, and enhancing drug discovery [28]. It facilitates adaptive clinical trails by dynamically adjusting parameters to maximize outcomes [29] and accelerates drug discovery by identifying promising molecular modifications [30]. This data-driven approach improves efficiency, reduces costs, and enhances treatment success rates [31, 32].
How RL works
Trial and error learning: Like a robot navigating a maze, RL maximizes rewards by learning the best strategies over time [33].
Dynamic interaction: Unlike static models, RL continuously adapts by interacting with its environment [34].
Applications in healthcare
Personalized medicine: Adjusting treatments based on patient responses [35].
Drug discovery: Identifying novel drug candidates through chemical simulations [36].
Clinical trials: Optimizing trial parameters for better patient outcomes and resource efficiency [37].
Natural language processing (NLP)
NLP enables computers to understand and process human language [38]. In healthcare and pharmaceuticals, it extracts insights from unstructured data, enhancing research and decision-making [39]. NLP algorithms analyze scientific literature to identify relevant studies, summarize findings, and track emerging trends [40, 41]. In CT, NLP extracts patient demographics, treatment protocols, and outcomes, aiding meta-analyses and trial design improvements [42]. It also processes Electronic Health Records (EHRS) to identify patterns, predict health outcomes, and support clinical decision-making [43, 44]. Overall, NLP enhances research efficiency, clinical operations, and patient care [45].
Applications in drug discovery
NLP accelerates drug discovery by mining literature and patents to uncover gene-protein-disease relationships [46, 47]. It identifies drug targets and therapeutic approaches by analyzing vast datasets [48]. In CT, NLP extracts insights on efficacy, safety, and patient responses, optimizing study design and drug development [49]. It also examines EHRS for real-world evidence of drug effects, off-label uses, and adverse reactions [50]. Additionally, sentiment analysis of social media and patient forums provides insights into patient experiences, influencing drug development strategies [51]. These applications drive pharmaceutical advancements and improved treatments [52].
AI in image recognition
Image recognition enhances biomedical image analysis, benefiting High-Throughput Screening (HTS) and histopathology [53]. In HTS, it automates cellular image analysis to identify active compounds, assess cytotoxicity, and detect phenotypic changes, improving accuracy and speed. In histopathology, AI analyzes tissue slides to detect abnormalities like cancerous cells with high precision, assisting pathologists in diagnosis [54]. It quantifies cell morphology, count, and spatial distribution, providing critical data for research and diagnostics [55]. AI-driven image recognition streamlines biomedical analysis, accelerating drug discovery, improving diagnostics, and enhancing patient outcomes.
Generative models for drug design
Generative adversarial networks (GANs) and variational auto encoders (VAEs) in drug synthesis
Generative models create new data by learning patterns from existing datasets [56]. GANs and VAEs are widely used in drug design [57]
GANs: Work as a competition between two AI systems—
The generator creates new drug-like molecules [58].
The discriminator evaluates their authenticity by comparing them to real compounds [59].
Through continuous improvement, GANs generate highly realistic drug candidates [60, 61].
VAEs: Compress and reconstruct data, introducing slight variations to explore new molecular structures [62, 63]. In drug discovery, VAEs generate optimized molecules with enhanced properties, such as higher efficacy or lower toxicity [64, 65]. These models automate innovation, accelerating drug discovery and reducing RandD timelines [66-68].
Predicting novel molecular structures
GANs generate novel drug-like molecules by training on chemical datasets, proposing compounds with desirable properties like strong binding affinity and optimal pharmacokinetics [69-71]. VAEs explore chemical space by interpolating between known compounds optimizing molecular properties for solubility, toxicity, and bioavailability [72-74]. Together, GANs and VAEs enhance drug discovery, facilitating the rapid development of novel therapeutics [75, 76].
Applications of AI in drug discovery
Target identification and validation
AI analyzes omics data-including genomics, proteomics, transcriptomics, and metabolomics-to uncover drug targets that traditional methods might miss [77]. Machine learning and deep learning identify patterns between genes, proteins, and diseases [78], analyzing gene expression to highlight differentially expressed genes in diseased tissues [79]. AI integrates omics datasets to construct biological networks, revealing key therapeutic targets [80]. By incorporating patient samples, public databases, and literature, AI prioritizes targets based on disease relevance and druggability [81]. This approach accelerates target identification and improves clinical relevance [82], making AI a powerful tool for discovering new therapies [83].
Successful AI-based target identification
International Business Machines (IBM) Watson identified novel Amyotrophic Lateral Sclerosis (ALS)-associated genes by analyzing literature and patient data, some of which were later validated experimentally [84-86]. Insilico Medicine used AI to discover a target for Idiopathic Pulmonary Fibrosis (IPF), leading to a promising drug candidate in preclinical studies [87, 88]. Benevolent-AI and AstraZeneca leveraged AI to find new chronic kidney disease targets, now pursued in drug development [89, 90]. These successes highlight AI’s ability to expedite drug discovery and enhance treatment development [91].
Drug screening and design
AI-driven virtual screening uses machine learning and deep learning to predict the biological activity of large compound libraries. Unlike traditional molecular docking, which is computationally intensive, AI rapidly analyzes and prioritizes compounds by learning from known drug-target interactions (table 1).
Predictive modeling for drug efficacy and safety
AI-driven predictive models analyze biomedical data to assess drug interactions, side effects, and efficacy [105]. Machine learning algorithms trained on CT results, patient records, and pharmacological data predict adverse drug reactions and drug-drug interactions by identifying patterns in chemical structures and biological targets [106, 107]. These models also anticipate side effects by comparing new drugs to known compounds with adverse effects [108]. Additionally, AI correlates molecular features with clinical outcomes to evaluate drug efficacy, aiding in candidate selection for specific conditions [109]. Techniques like supervised, unsupervised, and deep learning enhance drug safety and efficacy, improving decision-making in drug development and personalized medicine [110, 111] (table 2).
Table 1: AI-driven approaches for virtual screening of compound libraries
| AI-driven approach | Description | Key benefits |
| Deep neural networks (DNNs) | Trained on datasets of compounds with known activities to predict the binding affinity of new compounds to specific targets [92]. | Rapidly identifies potential compounds for specific biological targets. |
| Reinforcement learning (RL) | Iteratively improves the screening process based on feedback from experimental results [93]. | Enhances efficiency and refines predictions over multiple iterations. |
| Quantitative structure-activity relationship (QSAR) models | Predicts the biological activity of compounds based on their chemical structures [94]. | Enables efficient identification of promising candidates from large chemical libraries. |
| General AI/ml models | Learned from large datasets of known drug-target interactions to analyze and prioritize compounds [95]. | Reduces computational time and cost compared to traditional methods. |
| Comparison to traditional methods | Traditional virtual screening, such as molecular docking, requires extensive computational resources and time [96, 97] | AI models streamline the process and reduce time-to-discovery for high-potential compounds. |
Table 2: Applications of AI in diagnostics and personalized medicine
| Application | Description | Examples | Reference |
| AI in radiology | Analyzes medical imaging data to detect abnormalities like tumors, fractures, and lesions. | Detecting breast cancer in mammograms with deep learning algorithms. | [98] |
| AI in pathology | Assists in analyzing tissue samples for cancer detection and grading. | Identifying malignant tissue in prostate and lung cancer biopsies using Convolutional Neural Networks (CNNs). | [99, 100] |
| Point-of-care devices | Portable AI-enabled tools for real-time health assessments. | Devices for early disease detection and intervention. | [101] |
| Genomic data analysis | Processes vast genomic data to identify disease-causing mutations and biomarkers. | AI models identifying genetic variants for rare diseases. | [102] |
| Precision treatment | Predicts patient responses to therapies based on genetic profiles. | Optimizing drug regimens using pharmacogenomic data. | [103, 104] |
Case studies in predictive modeling
Atomwise’s AI platform accelerates drug discovery by predicting compound interactions with target proteins [112, 113]. Novartis and IBM Watson used AI to analyze patient records and CT data, identifying previously unknown drug side effects and enhancing safety [114, 115]. Exscientia and GlaxoSmithKline (GSK) leveraged AI to identify and optimize drug candidates for Chronic Obstructive Pulmonary Disease (COPD), predicting efficacy and safety profiles, leading to preclinical testing [116, 117]. These examples highlight AI’s role in streamlining drug development, reducing costs, and increasing clinical success rates [118].
AI in preclinical and clinical trails
Preclinical testing (PCT)
AI enhances Preclinical drug testing through in silico simulations and automated assays, improving efficiency and accuracy [119]. In silico models predict drug behavior, evaluating efficacy and toxicity without extensive lab work [120]. Machine learning analyzes molecular interactions, PK, and toxicology to simulate in vivo effects, reducing time and costs in early-stage development [121, 122].
Automated assays powered by AI streamline HTS, enabling rapid testing of thousands of compounds with precision [123]. AI-driven image recognition and data analysis detect active compounds and off-target effects, enhancing reproducibility and reducing human error [124, 125]. AI also optimizes experimental conditions, ensuring reliable data collection [126]. By integrating AI, PCT accelerates drug discovery, improves candidate selection, and minimizes reliance on animal testing [127].
AI in monitoring and managing clinical trial data
AI enhances CT efficiency, accuracy, and real-time decision-making [135]. AI-powered platforms integrate data from EHRS, wearables, and patient-reported outcomes, ensuring comprehensive and up-to-date information [136]. Machine learning processes this data, identifying patterns and anomalies for timely interventions [137].
For patient safety and adherence, AI detects adverse events, protocol deviations, and safety concerns in real-time, enabling swift responses [138, 139]. It also analyzes adherence data, identifying non-compliant patients and providing targeted interventions [140].
AI improves data quality through automated validation, error reduction, and NLP-driven standardization of unstructured data [141]. Predictive analytics forecast enrollment rates, dropout risks, and trial metrics, optimizing resource management [142].
By leveraging AI, CT achieve greater accuracy, efficiency, and reliability, accelerating the development of innovative therapies [143, 144].
Table 3: AI enhances clinical trial design, patient stratification, and outcome prediction
| Aspect | How AI Helps | Benefits |
| Trial design | -Analyzes historical data, demographics, and disease characteristics. -Optimizes trial protocols (sample size, endpoints). -Simulates trial scenarios to select promising strategies [128]. |
-Reduces trial failure. -Ensures efficient trial execution [131]. |
| Patient stratification | -Analyzes genetic, phenotypic, and clinical data. -Identifies subgroups likely to benefit from treatment. -Detects biomarkers for response prediction [129]. |
-Improves personalized medicine. -Increases efficacy. -Lowers adverse effects [132]. |
| Predicting outcomes | -Forecasts success likelihood using data from past trials, real-world evidence, and preclinical studies. -Identifies safety issues. -Suggests outcome-improving modifications [130]. |
-Supports informed decisions. -Enhances trial success and safety [133, 134]. |
Challenges and limitations of AI
Data quality and integration
Ensuring high-quality, integrated data is crucial for AI in drug discovery, yet variability in clinical data poses challenges [145]. Differences in data collection, patient populations, and healthcare practices lead to inconsistencies, missing values, and errors [146]. The heterogeneity of biomedical data-spanning EHRS, CT, and omics studies—further complicates integration due to distinct formats, terminologies, and standards [147, 148]. Standardized vocabularies help harmonize data, but interoperability issues persist, along with privacy and security concerns [149]. Robust encryption and anonymization protocols are essential for AI-driven drug development to ensure reliable predictions [150].
AI-based tools for harmonizing EHRS
Onto server: Manages clinical vocabularies like Systematized Nomenclature of Medicine – Clinical Terms (SNOMED CT) and International Classification of Diseases (ICD), ensuring consistency [151].
Trinet X: Aggregates and normalizes real-world EHR data for clinical trials and research [152].
FHIR standards for interoperability
Redox: Uses Fast Healthcare Interoperability Resources (FHIR) standards to connect EHR systems, facilitating seamless data exchange [153, 154].
Regulatory and ethical considerations
The adoption of AI in drug development presents regulatory and ethical challenges [155]. The lack of standardized guidelines complicates model validation and approval, creating uncertainty for developers [156]. AI models may also introduce bias if training data lacks diversity, leading to disparities in drug efficacy and safety across populations [157-159].
Transparency and interpretability are crucial for regulatory approval, as black-box models hinder clinician trust and decision-making [160]. Privacy concerns further complicate AI integration, necessitating compliance with General Data Protection Regulation (GDPR), Health Insurance Portability and Accountability Act (HIPAA), and robust data protection measures [161]. Successfully navigating these challenges is key to responsible AI implementation in drug development [162].
Regulatory examples
Food and drug administration (FDA) framework: Focuses on Software as a Medical Device (SaMD). Approved AI tools include:
Viz. ai: Detects strokes via CT scan analysis [163].
IDx-DR: Diagnoses diabetic retinopathy [164].
European medicines agency (EMA) framework: Ensures AI tools meet safety and efficacy standards. Approved tools include:
Corti. ai: Detects cardiac arrest in emergency calls via speech pattern analysis [165].
Model interpretability and transparency
Understanding AI models is critical for trust, regulatory approval, and clinical adoption [166, 167]. Transparent models allow stakeholders to validate predictions, improving confidence in AI-driven decisions [168]. Regulators require clear insights into AI decision-making to ensure fairness, safety, and effectiveness [169, 170].
However, complex AI models, like deep neural networks, often function as black boxes, making interpretability a challenge [171, 172]. Techniques like feature importance analysis, saliency maps, Local Interpretable Model-agnostic Explanations (LIME), and SHapley Additive exPlanations (SHAP) help explain model outputs but may not fully clarify intricate algorithms [173].
There is also a trade-off between interpretability and performance. Simpler models are easier to understand but may lack the predictive power of advanced AI [174]. Balancing transparency and accuracy is an ongoing challenge for AI in drug discovery [175].
Case studies and examples
Success stories
IBM Watson for Drug Discovery leverages AI to analyze scientific literature, CT data, and patient records, accelerating drug target identification [176]. A key achievement was identifying novel genes linked to ALS, providing new therapeutic avenues [177]. This highlights Watson’s role in advancing drug discovery.
Atomwise’s AI-driven platform, AtomNet, employs deep learning to predict small molecule-protein interactions, streamlining drug design [178]. It successfully identified potential Ebola virus inhibitors, expediting drug development and reducing costs [179, 180].
Both IBM Watson and Atomwise demonstrate AI’s transformative role in drug discovery by enhancing target identification, compound design, and therapeutic development efficiency [181, 182].
Emerging trends
AI is revolutionizing personalized medicine by analyzing genomic, proteomic, and clinical data to tailor treatments [183]. It identifies biomarkers predicting patient responses optimizing therapy selection [184, 185]. AI-driven immunotherapy prediction platforms enable more targeted cancer treatments [186].
In digital therapeutics, AI-powered apps, wearables, and Virtual Reality (VR) tools provide real-time monitoring and personalized interventions [187]. For instance, AI-driven diabetes management systems track blood glucose and offer tailored lifestyle recommendations, improving adherence and outcomes.
AI is also advancing drug repurposing by analyzing drug interactions and patient outcomes, identifying new therapeutic uses for existing medications [188, 189].
These innovations enhance treatment precision, improve patient outcomes, and reshape modern healthcare [190] (fig. 1).
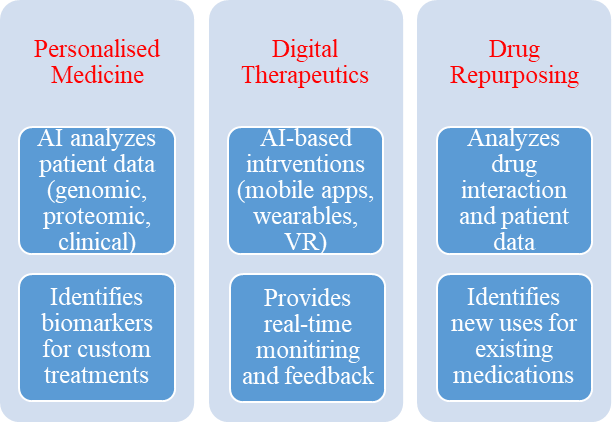
Fig. 1: The role of AI in personalized medicine and digital therapeutics
Future directions and potential
Advancements in AI technologies
Future AI developments will significantly impact the pharmaceutical sector. Federated learning enhances data privacy by training models across decentralized datasets, improving AI robustness with real-world data [191, 192]. Transfer learning enables AI models to adapt to new drug discovery tasks with limited data, increasing efficiency [193].
The integration of quantum computing with AI will revolutionize drug discovery by accelerating molecular modeling, protein folding predictions, and chemical structure optimization [194, 195]. AI-driven generative models, such as advanced GANs and VAEs, will design novel drug molecules with improved precision, optimizing potency, selectivity, and safety [196, 197].
Enhanced NLP will improve AI’s ability to analyze biomedical literature, clinical data, and patient records, facilitating faster insights and better decision-making in drug development [198, 199]. These advancements will increase efficiency, reduce costs, and improve success rates in pharmaceutical research [200].
Integration with other emerging technologies
AI's convergence with genomics, biotechnology, and blockchain is transforming drug development [201]. Genomics-powered AI enables personalized medicine by identifying disease-associated genetic variants and optimizing treatment plans based on individual responses [202, 203].
In biotechnology, AI enhances high-throughput screening, protein design, and bioprocess optimization, aiding gene editing and synthetic biology applications [204-206]. Blockchain integration ensures data security, transparency, and traceability in clinical trials, patient records, and supply chains [207-209]. AI enhances blockchain analytics, detecting anomalies and optimizing data-sharing while maintaining privacy [210]. The synergy of these technologies will drive data integrity and accelerate therapeutic advancements [211].
Vision for the future of AI in pharmaceuticals
AI will revolutionize drug discovery by digitizing the entire pipeline, enabling real-time data analysis from target identification to clinical trials [212-214]. Personalized medicine will become widespread, with AI analyzing genetics, lifestyle, and environmental factors to tailor therapies, improving efficacy and minimizing side effects [215, 216].
AI will uncover novel drug targets by analyzing complex biological data, opening therapeutic avenues for diseases lacking effective treatments, including neurodegenerative disorders and cancer [217, 218]. AI-driven drug repurposing will identify new applications for existing drugs, accelerating cost-effective treatments [219].
The integration of quantum computing, genomics, and blockchain will further optimize drug discovery. AI-driven CT optimization will improve trial design, recruitment, and monitoring, enabling adaptive, real-time adjustments for better outcomes [220].
By harnessing AI’s predictive capabilities, the pharmaceutical industry will develop safer, more effective, and personalized treatments, revolutionizing healthcare delivery and advancing medical research [221].
CONCLUSION
AI is revolutionizing the pharmaceutical industry by accelerating drug discovery, optimizing CT, and enhancing patient response predictions. Advanced models like GANs and VAEs drive innovation by designing novel molecules, navigating chemical spaces, and refining drug properties, significantly expediting therapeutic development.
The future of AI in pharmaceuticals will be further transformed by its integration with quantum computing, enabling rapid and precise molecular simulations, optimizing drug interactions, and dramatically reducing development time and costs. Additionally, decentralized clinical trials powered by AI will enhance remote participation, real-time monitoring, and data accuracy, improving patient recruitment, retention, and inclusivity in clinical research.
The combination of AI and blockchain will ensure secure, transparent, and tamper-proof data management, safeguarding patient records, clinical trial data, and supply chains while mitigating risks such as counterfeit drugs and data manipulation.
In essence, the synergy of AI, quantum computing, decentralized trials, and blockchain will redefine drug development, paving the way for personalized medicine, higher clinical trial success rates, and faster access to ground-breaking treatments, marking a new era in pharmaceutical innovation.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
P. Jasmine and Arwa contributed to the sections on AI Technology and Methodologies, Natural Language Processing (NLP), Generative Models, Applications of AI in Drug Discovery, and AI in Preclinical and Clinical Trials. Siddiq provided insights on Challenges and Limitations, while Nirmala authored the Case Studies and Examples section. Dhanursha explored Future Directions and Potential. Mr. S. Shakir Basha was responsible for revision, alignment, and final corrections, ensuring coherence and accuracy across the manuscript.
CONFLICT OF INTERESTS
Declared none
REFERENCES
Chowdhary KR. Natural language processing. In: Fundamentals of artificial intelligence. New Delhi: Springer India; 2020. p. 603-49. doi: 10.1007/978-81-322-3972-7_19.
Janowski A. Natural language processing techniques for clinical text analysis in healthcare. J Adv Anal Healthc Manag. 2023;7(1):51-76.
Bose P, Roy S, Ghosh P. A comparative NLP-based study on the current trends and future directions in COVID-19 research. IEEE Access. 2021;9:78341-55. doi: 10.1109/ACCESS.2021.3082108, PMID 34786315.
Bougioukas KI, Bouras EC, Avgerinos KI, Dardavessis T, Haidich AB. How to keep up to date with medical information using web‐based resources: a systematised review and narrative synthesis. Health Info Libr J. 2020;37(4):254-92. doi: 10.1111/hir.12318, PMID 32691960.
Mayer T, Marro S, Cabrio E, Villata S. Enhancing evidence-based medicine with natural language argumentative analysis of clinical trials. Artif Intell Med. 2021 Aug;118:102098. doi: 10.1016/j.artmed.2021.102098, PMID 34412851.
Marafino BJ, Park M, Davies JM, Thombley R, Luft HS, Sing DC. Validation of prediction models for critical care outcomes using natural language processing of electronic health record data. JAMA Netw Open. 2018;1(8):e185097. doi: 10.1001/jamanetworkopen.2018.5097, PMID 30646310.
Oladele OK. Natural language processing in healthcare: transforming electronic health records and clinical decision support; 2024.
Ahmed U, Iqbal K, Aoun M. Natural language processing for clinical decision support systems: a review of recent advances in healthcare. J Intell Connect Emerg Technol. 2023;8(2):1-16.
Jiang J, Chen L, KE L, Dou B, Zhang C, Feng H. A review of transformers in drug discovery and beyond. J Pharm Anal. 2024 Aug 30;101081. doi: 10.1016/j.jpha.2024.101081.
Zheng CJ, Han LY, Yap CW, JI ZL, Cao ZW, Chen YZ. Therapeutic targets: progress of their exploration and investigation of their characteristics. Pharmacol Rev. 2006;58(2):259-79. doi: 10.1124/pr.58.2.4, PMID 16714488.
Romano JD, Tatonetti NP. Informatics and computational methods in natural product drug discovery: a review and perspectives. Front Genet. 2019 Apr 30;10:368. doi: 10.3389/fgene.2019.00368, PMID 31114606.
RN, Khan SB, Kumar AV, TR M, Alojail M, Sangwan SR. Enhancing drug discovery and patient care through advanced analytics with the power of NLP and machine learning in pharmaceutical data interpretation. SLAS Technol. 2025;31:100238. doi: 10.1016/j.slast.2024.100238, PMID 39722407.
Domatzoglou A. An NLP approach for drug repurposing based on mental health reviews. Tilburg University; 2023. p. 1-48.
Sreejith R, Sinimole KR. User-centric evaluation of EHR software through NLP-driven investigation: implications for product development and user experience. J Open Innov Technol Mark Complexity. 2024;10(1):100206. doi: 10.1016/j.joitmc.2023.100206.
Huang D, Yang M, Wen X, Xia S, Yuan B. AI-driven drug discovery: accelerating the development of novel therapeutics in biopharmaceuticals. J Knowl Learn Sci Technol. 2024;3(3):206-24. doi: 10.60087/jklst.vol3.n3.p.206-224.
Shariff A, Kangas J, Coelho LP, Quinn S, Murphy RF. Automated image analysis for high content screening and analysis. J Biomol Screen. 2010;15(7):726-34. doi: 10.1177/1087057110370894, PMID 20488979.
Wang MH, Xing L, Pan Y, GU F, Fang J, YU X. AI-based advanced approaches and dry eye disease detection based on multi-source evidence: cases applications issues and future directions. Big Data Min Anal. 2024;7(2):445-84. doi: 10.26599/BDMA.2023.9020024.
Gurcan MN, Boucheron LE, Can A, Madabhushi A, Rajpoot NM, Yener B. Histopathological image analysis: a review. IEEE Rev Biomed Eng. 2009;2:147-71. doi: 10.1109/RBME.2009.2034865, PMID 20671804.
Cheng Y, Gong Y, Liu Y, Song B, Zou Q. Molecular design in drug discovery: a comprehensive review of deep generative models. Brief Bioinform. 2021;22(6):bbab344. doi: 10.1093/bib/bbab344, PMID 34415297.
El Kaddoury M, Mahmoudi A, Himmi MM, Editors. Deep generative models for image generation: a practical comparison between variational autoencoders and generative adversarial networks. Mobile secure and programmable networking: 5th International Conference MSPN 2019, Mohammedia Morocco; 2019. p. 5.
Miller AI. The artist in the machine: the world of AI-powered creativity: Mit Press; 2019.
Kim PT. Data driven discrimination at work. Wm & Mary L Rev. 2016;58(3):857.
Liu X, Hsieh CJ, Editors. Rob-Gan: generator discriminator and adversarial attacker. In: proceedings of the IEEE/CVF conference on computer vision and pattern recognition. New York: IEEE; 2019. p. 11226-35. doi: 10.1109/CVPR.2019.01149.
Martinelli DD. Generative machine learning for de novo drug discovery: a systematic review. Comput Biol Med. 2022;145:105403. doi: 10.1016/j.compbiomed.2022.105403, PMID 35339849.
Tadeusiewicz R. Modern computational intelligence methods for the interpretation of medical images. Springer Science Business Media; 2008.
Zhao HK, Osher S, Fedkiw R, Editors. Fast surface reconstruction using the level set method. In: Proceedings of the IEEE workshop on variational and level set methods in computer vision; 2001: IEEE.
Vogt M. Using deep neural networks to explore chemical space. Expert Opin Drug Discov. 2022;17(3):297-304. doi: 10.1080/17460441.2022.2019704, PMID 34918594.
Craik DJ, Fairlie DP, Liras S, Price D. The future of peptide based drugs. Chem Biol Drug Des. 2013;81(1):136-47. doi: 10.1111/cbdd.12055, PMID 23253135.
Thomford NE, Senthebane DA, Rowe A, Munro D, Seele P, Maroyi A. Natural products for drug discovery in the 21st century: innovations for novel drug discovery. Int J Mol Sci. 2018;19(6):1578. doi: 10.3390/ijms19061578, PMID 29799486.
Boussioux L, Lane JN, Zhang M, Jacimovic V, Lakhani KR. The crowdless future? Generative AI and creative problem solving. Organ Sci. 2024;35(5):1589-607. doi: 10.1287/orsc.2023.18430.
Khanna I. Drug discovery in pharmaceutical industry: productivity challenges and trends. Drug Discov Today. 2012;17(19-20):1088-102. doi: 10.1016/j.drudis.2012.05.007, PMID 22627006.
Romanelli V, Cerchia C, Lavecchia A. Unlocking the potential of generative artificial intelligence in drug discovery. In: Lyu Z, editor. Applications of generative AI. Cham: Springer International Publishing; 2024. p. 37-63. doi: 10.1007/978-3-031-46238-2_3.
Nguyen H. De novo molecular design using deep learning: open access te Herenga Waka-victoria University of Wellington; 2024.
Sousa T, Correia J, Pereira V, Rocha M. Generative deep learning for targeted compound design. J Chem Inf Model. 2021;61(11):5343-61. doi: 10.1021/acs.jcim.0c01496, PMID 34699719.
Gordon EM, Barrett RW, Dower WJ, Fodor SP, Gallop MA. Applications of combinatorial technologies to drug discovery. 2. Combinatorial organic synthesis library screening strategies and future directions. J Med Chem. 1994;37(10):1385-401. doi: 10.1021/jm00036a001, PMID 8182695.
XU Y, Lin K, Wang S, Wang L, Cai C, Song C. Deep learning for molecular generation. Future Med Chem. 2019;11(6):567-97. doi: 10.4155/fmc-2018-0358, PMID 30698019.
Elton DC, Boukouvalas Z, Fuge MD, Chung PW. Deep learning for molecular design a review of the state of the art. Mol Syst Des Eng. 2019;4(4):828-49. doi: 10.1039/C9ME00039A.
Zhao L, WU Q, Long Y, QU Q, QI F, Liu L. Micro RNAs: critical targets for treating rheumatoid arthritis angiogenesis. J Drug Target. 2024;32(1):1-20. doi: 10.1080/1061186X.2023.2284097, PMID 37982157.
Mariam Z, Niazi SK, Magoola M. Unlocking the future of drug development: generative AI digital twins and beyond. Bio Med Informatics. 2024;4(2):1441-56. doi: 10.3390/biomedinformatics4020079.
Biswas N, Chakrabarti S. Artificial intelligence (AI) based systems biology approaches in multi-omics data analysis of cancer. Front Oncol. 2020;10:588221. doi: 10.3389/fonc.2020.588221, PMID 33154949.
LI R, LI L, XU Y, Yang J. Machine learning meets omics: applications and perspectives. Brief Bioinform. 2022;23(1):bbab460. doi: 10.1093/bib/bbab460, PMID 34791021.
Vadapalli S, Abdelhalim H, Zeeshan S, Ahmed Z. Artificial intelligence and machine learning approaches using gene expression and variant data for personalized medicine. Brief Bioinform. 2022;23(5):bbac191. doi: 10.1093/bib/bbac191, PMID 35595537.
Chakraborty S, Sharma G, Karmakar S, Banerjee S. Multi-OMICS approaches in cancer biology: new era in cancer therapy. Biochim Biophys Acta Mol Basis Dis. 2024;1870(5):167120. doi: 10.1016/j.bbadis.2024.167120, PMID 38484941.
Dhudum R, Ganeshpurkar A, Pawar A. Revolutionizing drug discovery: a comprehensive review of AI applications. Drugs Drug Candidates. 2024;3(1):148-71. doi: 10.3390/ddc3010009.
Koutsoukas A, Simms B, Kirchmair J, Bond PJ, Whitmore AV, Zimmer S. From in silico target prediction to multi-target drug design: current databases methods and applications. J Proteomics. 2011;74(12):2554-74. doi: 10.1016/j.jprot.2011.05.011, PMID 21621023.
Pun FW, Ozerov IV, Zhavoronkov A. AI powered therapeutic target discovery. Trends Pharmacol Sci. 2023;44(9):561-72. doi: 10.1016/j.tips.2023.06.010, PMID 37479540.
Myszczynska MA. Uncovering novel drug therapies and targets for amyotrophic lateral sclerosis (ALS) using artificial intelligence (AI). University of Sheffield; 2021.
Bakkar N, Kovalik T, Lorenzini I, Spangler S, Lacoste A, Sponaugle K. Artificial intelligence in neurodegenerative disease research: use of IBM watson to identify additional RNA-binding proteins altered in amyotrophic lateral sclerosis. Acta Neuropathol. 2018;135(2):227-47. doi: 10.1007/s00401-017-1785-8, PMID 29134320.
Geraci J, Bhargava R, Qorri B, Leonchyk P, Cook D, Cook M. Machine learning hypothesis generation for patient stratification and target discovery in rare disease: our experience with open science in ALS. Front Comp Neurosci. 2023;17:1199736. doi: 10.3389/fncom.2023.1199736, PMID 38260713.
Han S, Lee JE, Kang S, SO M, Jin H, Lee JH. Standigm ASK™: knowledge graph and artificial intelligence platform applied to target discovery in idiopathic pulmonary fibrosis. Brief Bioinform. 2024;25(2):bbae035. doi: 10.1093/bib/bbae035, PMID 38349059.
Chen B, Garmire L, Calvisi DF, Chua MS, Kelley RK, Chen X. Harnessing big ‘omics’ data and AI for drug discovery in hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol. 2020;17(4):238-51. doi: 10.1038/s41575-019-0240-9, PMID 31900465.
Jones CH, Madhavan S, Natarajan K, Corbo M, True JM, Dolsten M. Rewriting the textbook for pharma: how to adapt and thrive in a digital personalized and collaborative world. Drug Discov Today. 2024;29(9):104112. doi: 10.1016/j.drudis.2024.104112, PMID 39053620.
Qureshi R, Irfan M, Gondal TM, Khan S, WU J, Hadi MU. AI in drug discovery and its clinical relevance. Heliyon. 2023;9(7):e17575. doi: 10.1016/j.heliyon.2023.e17575, PMID 37396052.
Abbas MK, Rassam A, Karamshahi F, Abunora R, Abouseada M. The role of AI in drug discovery. Chem Bio Chem. 2024;25(14):e202300816. doi: 10.1002/cbic.202300816, PMID 38735845.
Ozturk H, Ozgur A, Ozkirimli E. Deep DTA: deep drug-target binding affinity prediction. Bioinformatics. 2018;34(17):i821-9. doi: 10.1093/bioinformatics/bty593, PMID 30423097.
Hjalmarsson H, Gevers M, Gunnarsson S, Lequin O. Iterative feedback tuning: theory and applications. IEEE Control Syst. 1998;18(4):26-41. doi: 10.1109/37.710876.
Filimonov DA, Druzhilovskiy DS, Lagunin AA, Gloriozova TA, Rudik AV, Dmitriev AV. Computer-aided prediction of biological activity spectra for chemical compounds: opportunities and limitations. Biomed Chem Res Methods. 2018;1(1):e00004. doi: 10.18097/BMCRM00004.
Liu G, Singha M, PU L, Neupane P, Feinstein J, WU HC. Graph DTI: a robust deep learning predictor of drug target interactions from multiple heterogeneous data. J Cheminform. 2021;13(1):58. doi: 10.1186/s13321-021-00540-0, PMID 34380569.
Stanzione F, Giangreco I, Cole JC. Use of molecular docking computational tools in drug discovery. Prog Med Chem. 2021;60:273-343. doi: 10.1016/bs.pmch.2021.01.004, PMID 34147204.
Kampen S. Structure based virtual screening for ligands of G protein coupled receptors: design of allosteric and dual target modulators; 2022.
Shen L, Margolies LR, Rothstein JH, Fluder E, McBride R, Sieh W. Deep learning to improve breast cancer detection on screening mammography. Sci Rep. 2019;9(1):12495. doi: 10.1038/s41598-019-48995-4, PMID 31467326.
Chon A, Balachandar N, LU P. Deep convolutional neural networks for lung cancer detection. Stanford University; 2017. p. 1-9.
Horasan A, Gunes A. Advancing prostate cancer diagnosis: a deep learning approach for enhanced detection in MRI images. Diagnostics (Basel). 2024;14(17):1871. doi: 10.3390/diagnostics14171871, PMID 39272656.
Judijanto L, Anggo S, Utami TP, Anurogo D, Ningrum D. The impact of the digital revolution on health research: a bibliometric review. J World Future Med Health Nurs. 2024;2(1):51-64. doi: 10.70177/health.v2i1.704.
Brasil S, Pascoal C, Francisco R, Dos Reis Ferreira V, Videira PA, Valadao AG. Artificial intelligence (AI) in rare diseases: is the future brighter? Genes. 2019;10(12):978. doi: 10.3390/genes10120978, PMID 31783696.
Bielinski SJ, Olson JE, Pathak J, Weinshilboum RM, Wang L, Lyke KJ. Preemptive genotyping for personalized medicine: design of the right drug right dose, right time using genomic data to individualize treatment protocol. Mayo Clin Proc. 2014;89(1):25-33. doi: 10.1016/j.mayocp.2013.10.021, PMID 24388019.
Papachristos A, Patel J, Vasileiou M, Patrinos GP. Dose optimization in oncology drug development: the emerging role of pharmacogenomics pharmacokinetics and pharmacodynamics. Cancers. 2023;15(12):3233. doi: 10.3390/cancers15123233, PMID 37370844.
Zhu H. Big data and artificial intelligence modeling for drug discovery. Annu Rev Pharmacol Toxicol. 2020;60(1):573-89. doi: 10.1146/annurev-pharmtox-010919-023324, PMID 31518513.
Cheng F, Zhao Z. Machine learning-based prediction of drug interactions by integrating drug phenotypic therapeutic chemical and genomic properties. J Am Med Inform Assoc. 2014;21(e2):e278-86. doi: 10.1136/amiajnl-2013-002512, PMID 24644270.
VO TH, Nguyen NT, Kha QH, LE NQ. On the road to explainable AI in drug-drug interactions prediction: a systematic review. Comp Struct Biotechnol J. 2022 Apr 19;20:2112-23. doi: 10.1016/j.csbj.2022.04.021, PMID 35832629.
ReferencesLounkine E, Keiser MJ, Whitebread S, Mikhailov D, Hamon J, Jenkins JL. Large scale prediction and testing of drug activity on side effect targets. Nature. 2012;486(7403):361-7. doi: 10.1038/nature11159, PMID 22722194.
Gayvert KM, Madhukar NS, Elemento O. A data driven approach to predicting successes and failures of clinical trials. Cell Chem Biol. 2016;23(10):1294-301. doi: 10.1016/j.chembiol.2016.07.023, PMID 27642066.
Dara S, Dhamercherla S, Jadav SS, Babu CM, Ahsan MJ. Machine learning in drug discovery: a review. Artif Intell Rev. 2022;55(3):1947-99. doi: 10.1007/s10462-021-10058-4, PMID 34393317.
Woodcock J. The prospects for personalized medicine in drug development and drug therapy. Clin Pharmacol Ther. 2007;81(2):164-9. doi: 10.1038/sj.clpt.6100063, PMID 17259943.
Mutha RE, Bagul VS, Tade RS, Vinchurkar K. An overview of artificial intelligence (AI) in drug delivery and development. AI Innov Drug Deliv Pharm Sci Advanc Ther Technol. 2024;1.
Hemamalini V, Tyagi AK, Vennila V, Kumari S. Revolutionizing drug discovery with cutting edge technologies: issue and challenges for the next decade. In: Tripathi RK, Tiwari S, Editors. Converging pharmacy science and engineering in computational drug discovery. IGI Global; 2024. p. 76-89. doi: 10.4018/979-8-3693-2897-2.ch004.
Shah P, Kendall F, Khozin S, Goosen R, HU J, Laramie J. Artificial intelligence and machine learning in clinical development: a translational perspective. NPJ Digit Med. 2019;2(1):69. doi: 10.1038/s41746-019-0148-3, PMID 31372505.
Al Worafi YM. Technology for drug safety: current status and future developments: springer; 2023.
Batool M, Ahmad B, Choi S. A structure based drug discovery paradigm. Int J Mol Sci. 2019;20(11):2783. doi: 10.3390/ijms20112783, PMID 31174387.
Vatansever S, Schlessinger A, Wacker D, Kaniskan HU, Jin J, Zhou MM. Artificial intelligence and machine learning aided drug discovery in central nervous system diseases: state of the arts and future directions. Med Res Rev. 2021;41(3):1427-73. doi: 10.1002/med.21764, PMID 33295676.
Marques L, Costa B, Pereira M, Silva A, Santos J, Saldanha L. Advancing precision medicine: a review of innovative in silico approaches for drug development clinical pharmacology and personalized healthcare. Pharmaceutics. 2024;16(3):332. doi: 10.3390/pharmaceutics16030332, PMID 38543226.
Vattikuti MC. Improving drug discovery and development using AI: opportunities and challenges. Res Gate J. 2024;10(10).
Agamah FE, Mazandu GK, Hassan R, Bope CD, Thomford NE, Ghansah A. Computational/in silico methods in drug target and lead prediction. Brief Bioinform. 2020;21(5):1663-75. doi: 10.1093/bib/bbz103, PMID 31711157.
Mishra V. Artificial intelligence: the beginning of a new era in pharmacy profession. Asian J Pharm (AJP). 2018;12(2):72.
Wang J, Urban L. The impact of early ADME profiling on drug discovery and development strategy. DDW Drug Discov World. 2004;5(4):73-86.
Joshi C, Makwana D. Advanced high throughput screening platforms in drug discovery. In: Parikesit AA, Naderi M, Singh A, Konnur RG, Mukherjee S, editors. Genomics Driven Drug Discovery through Pharmacogenomics. IGI Global; 2024. p. 35-74. doi: 10.4018/979-8-3693-6597-7.ch002.
Cichonska A, Ravikumar B, Rahman R. AI for targeted polypharmacology: the next frontier in drug discovery. Curr Opin Struct Biol. 2024;84:102771. doi: 10.1016/j.sbi.2023.102771, PMID 38215530.
Bannach Brown A, Hair K, Bahor Z, Soliman N, Macleod M, Liao J. Technological advances in preclinical meta research. BMJ Open Sci. 2021;5(1):e100131. doi: 10.1136/bmjos-2020-100131, PMID 35047701.
Suriyaamporn P, Pamornpathomkul B, Patrojanasophon P, Ngawhirunpat T, Rojanarata T, Opanasopit P. The artificial intelligence-powered new era in pharmaceutical research and development: a review. AAPS Pharm Sci Tech. 2024;25(6):188. doi: 10.1208/s12249-024-02901-y, PMID 39147952.
Vashishat A, Patel P, Das Gupta G, Das Kurmi B. Alternatives of animal models for biomedical research: a comprehensive review of modern approaches. Stem Cell Rev Rep. 2024;20(4):881-99. doi: 10.1007/s12015-024-10701-x, PMID 38429620.
Chang M, Balser J, Roach J, Bliss R. Innovative strategies statistical solutions and simulations for modern clinical trials. Chapman & Hall/CRC; 2019. doi: 10.1201/9781351214544.
Spertus JA, Birmingham MC, Butler J, Lingvay I, Lanfear DE, Abbate A. Novel trial design: CHIEF-HF. Circ Heart Fail. 2021;14(3):e007767. doi: 10.1161/CIRCHEARTFAILURE.120.007767, PMID 33724883.
Duffy MJ, O Donovan N, Crown J. Use of molecular markers for predicting therapy response in cancer patients. Cancer Treat Rev. 2011;37(2):151-9. doi: 10.1016/j.ctrv.2010.07.004, PMID 20685042.
Manzari MT, Shamay Y, Kiguchi H, Rosen N, Scaltriti M, Heller DA. Targeted drug delivery strategies for precision medicines. Nat Rev Mater. 2021;6(4):351-70. doi: 10.1038/s41578-020-00269-6, PMID 34950512.
Roth BJ, Krilov L, Adams S, Aghajanian CA, Bach P, Braiteh F. Clinical cancer advances 2012: annual report on progress against cancer from the American society of clinical oncology. J Clin Oncol. 2013;31(1):131-61. doi: 10.1200/JCO.2012.47.1938, PMID 23213095.
Kawamoto K, Houlihan CA, Balas EA, Lobach DF. Improving clinical practice using clinical decision support systems: a systematic review of trials to identify features critical to success. BMJ. 2005;330(7494):765. doi: 10.1136/bmj.38398.500764.8F, PMID 15767266.
Sutton RT, Pincock D, Baumgart DC, Sadowski DC, Fedorak RN, Kroeker KI. An overview of clinical decision support systems: benefits risks and strategies for success. NPJ Digit Med. 2020;3(1):17. doi: 10.1038/s41746-020-0221-y, PMID 32047862.
Yin J, Ngiam KY, Teo HH. Role of artificial intelligence applications in real-life clinical practice: systematic review. J Med Internet Res. 2021;23(4):e25759. doi: 10.2196/25759, PMID 33885365.
Khatiwada P, Yang B, Lin JC, Blobel B. Patient generated health data (PGHD): understanding requirements challenges and existing techniques for data security and privacy. J Pers Med. 2024;14(3):282. doi: 10.3390/jpm14030282, PMID 38541024.
Aldoseri A, Al Khalifa KN, Hamouda AM. Re-thinking data strategy and integration for artificial intelligence: concepts opportunities and challenges. Appl Sci. 2023;13(12):7082. doi: 10.3390/app13127082.
Bates DW, Levine D, Syrowatka A, Kuznetsova M, Craig KJ, Rui A. The potential of artificial intelligence to improve patient safety: a scoping review. NPJ Digit Med. 2021;4(1):54. doi: 10.1038/s41746-021-00423-6, PMID 33742085.
Dash K. Machine learning applications for detecting anomalies and ensuring data integrity in clinical trials; 2023.
Bohlmann A, Mostafa J, Kumar M. Machine learning and medication adherence: scoping review. Med. 2021;2(4):e26993. doi: 10.2196/26993, PMID 37725549.
Kreimeyer K, Foster M, Pandey A, Arya N, Halford G, Jones SF. Natural language processing systems for capturing and standardizing unstructured clinical information: a systematic review. J Biomed Inform. 2017 Sep;73:14-29. doi: 10.1016/j.jbi.2017.07.012, PMID 28729030.
Venkateswarulu N, Pavan Kumar P, Obulesu O. Predictive analytics in healthcare. In: Singh N, Birla S, Ansari MD, Shukla NK, editors. Intelligent techniques for predictive data analytics. Chichester: John Wiley & Sons; 2024. p. 71-87. doi: 10.1002/9781394227990.ch4.
Ovy NH, Begum A. The role of AI in optimizing adaptive trials for improved clinical success and patient focus. J Multidiscip Res. 2024;10(2):152-73.
Hanna E, Remuzat C, Auquier P, Toumi M. Gene therapies development: slow progress and promising prospect. J Mark Access Health Policy. 2017;5(1):1265293. doi: 10.1080/20016689.2017.1265293, PMID 28265348.
Kumar SA, Ananda Kumar TD, Beeraka NM, Pujar GV, Singh M, Narayana Akshatha HS. Machine learning and deep learning in data driven decision making of drug discovery and challenges in high-quality data acquisition in the pharmaceutical industry. Future Med Chem. 2022;14(4):245-70. doi: 10.4155/fmc-2021-0243, PMID 34939433.
Dziura JD, Post LA, Zhao Q, FU Z, Peduzzi P. Strategies for dealing with missing data in clinical trials: from design to analysis. Yale J Biol Med. 2013;86(3):343-58. PMID 24058309.
Tong L, Shi W, Isgut M, Zhong Y, Lais P, Gloster L. Integrating multi omics data with EHR for precision medicine using advanced artificial intelligence. IEEE Rev Biomed Eng. 2024;17:80-97. doi: 10.1109/RBME.2023.3324264, PMID 37824325.
Carter AB, Abruzzo LV, Hirschhorn JW, Jones D, Jordan DC, Nassiri M. Electronic health records and genomics: perspectives from the association for molecular pathology electronic health record (EHR) interoperability for clinical genomics data working group. J Mol Diagn. 2022;24(1):1-17. doi: 10.1016/j.jmoldx.2021.09.009, PMID 34656760.
Pezoulas VC, Fotiadis DI. The pivotal role of data harmonization in revolutionizing global healthcare: a framework and a case study. Conn Health Telemed. 2024;3(2). doi: 10.20517/chatmed.2023.37.
Niazi SK, Mariam Z. Computer-aided drug design and drug discovery: a prospective analysis. Pharmaceuticals (Basel). 2023;17(1):22. doi: 10.3390/ph17010022, PMID 38256856.
Metke Jimenez A, Steel J, Hansen D, Lawley M. Ontoserver: a syndicated terminology server. J Biomed Semantics. 2018;9(1):24. doi: 10.1186/s13326-018-0191-z, PMID 30223897.
Palchuk MB, London JW, Perez Rey D, Drebert ZJ, Winer Jones JP, Thompson CN. A global federated real-world data and analytics platform for research. JAMIA Open. 2023;6(2):ooad035. doi: 10.1093/jamiaopen/ooad035, PMID 37193038.
Vorisek CN, Lehne M, Klopfenstein SA, Mayer PJ, Bartschke A, Haese T. Fast healthcare interoperability resources (FHIR) for interoperability in health research: systematic review. JMIR Med Inform. 2022;10(7):e35724. doi: 10.2196/35724, PMID 35852842.
Machireddy JR. Artificial intelligence based approach to perform monitoring and diagnostic process for a holistic environment. Int J Comput Sci Eng Res Dev (IJCSERD). 2024;14(2):71-88.
Olatunji Akinrinola, Chinwe Chinazo Okoye, Onyeka Chrisanctus Ofodile, Chinonye Esther Ugochukwu. Navigating and reviewing ethical dilemmas in AI development: strategies for transparency fairness and accountability. GSC Adv Res Rev. 2024;18(3):50-8. doi: 10.30574/gscarr.2024.18.3.0088.
Nikfal N. A development process framework for artificial intelligence/machine learning (AI/ML) based connected health informatics: purdue university graduate school; 2024.
Blanco Gonzalez A, Cabezon A, Seco Gonzalez A, Conde Torres D, Antelo Riveiro P, Pineiro A. The role of AI in drug discovery: challenges opportunities and strategies. Pharmaceuticals (Basel). 2023;16(6):891. doi: 10.3390/ph16060891, PMID 37375838.
Norori N, HU Q, Aellen FM, Faraci FD, Tzovara A. Addressing bias in big data and AI for health care: a call for open science. Patterns (NY). 2021;2(10):100347. doi: 10.1016/j.patter.2021.100347, PMID 34693373.
Hanna M, Pantanowitz L, Jackson B, Palmer O, Visweswaran S, Pantanowitz J. Ethical and bias considerations in artificial intelligence (AI)/machine learning. Mod Pathol. 2025 Mar;38(3):100686.
Barnes E, Hutson J. Navigating the complexities of AI: the critical role of interpretability and explainability in ensuring transparency and trust. Educ Res (IJMCER). 2024;6(3):248-56.
Jaime FJ, Munoz A, Rodriguez Gomez F, Jerez Calero A. Strengthening privacy and data security in biomedical microelectromechanical systems by IoT communication security and protection in smart healthcare. Sensors (Basel). 2023;23(21):8944. doi: 10.3390/s23218944, PMID 37960646.
Lescrauwaet L, Wagner H, Yoon C, Shukla S. Adaptive legal frameworks and economic dynamics in emerging technologies: navigating the intersection for responsible innovation. Law Eco. 2022;16(3):202-20. doi: 10.35335/laweco.v16i3.61.
Meyer BC, Meyer DM, ST Germain E, Pham N, Alwood BT, Van Orden K. (Stroke DOC-AI): leveraging ai tools to optimize both hub and spoke in a telestroke network. NEJM AI Sponsored; 2024.
HE M, LI Z, Liu C, Shi D, Tan Z. Deployment of artificial intelligence in real-world practice: opportunity and challenge. Asia Pac J Ophthalmol (Phila). 2020;9(4):299-307. doi: 10.1097/APO.0000000000000301, PMID 32694344.
Wang Y, Zhang H, Sun A, Meng X, editors. Cort: a new baseline for comparative opinion classification by dual prompts. Findings of the Association for Computational Linguistics: EMNLP; 2022.
Jimenez Luna J, Grisoni F, Schneider G. Drug discovery with explainable artificial intelligence. Nat Mach Intell. 2020;2(10):573-84. doi: 10.1038/s42256-020-00236-4.
Jao I, Kombe F, Mwalukore S, Bull S, Parker M, Kamuya D. Research stakeholders views on benefits and challenges for public health research data sharing in Kenya: the importance of trust and social relations. Plos One. 2015;10(9):e0135545. doi: 10.1371/journal.pone.0135545, PMID 26331716.
Soundararajan R, Shenbagaraman V. Enhancing financial decision making through explainable AI and blockchain integration: improving transparency and trust in predictive models. Educational Administration Theor Pract. 2024;30(4):9341-51.
Hassija V, Chamola V, Mahapatra A, Singal A, Goel D, Huang K. Interpreting black box models: a review on explainable artificial intelligence. Cognit Comput. 2024;16(1):45-74. doi: 10.1007/s12559-023-10179-8.
Mirakhori F, Niazi SK. Harnessing the AI/ml in drug and biological products discovery and development: the regulatory perspective. Pharmaceuticals (Basel). 2025;18(1):47. doi: 10.3390/ph18010047, PMID 39861110.
Fan FL, Xiong J, LI M, Wang G. On interpretability of artificial neural networks: a survey. IEEE Trans Radiat Plasma Med Sci. 2021;5(6):741-60. doi: 10.1109/trpms.2021.3066428, PMID 35573928.
Guidotti R, Monreale A, Ruggieri S, Turini F, Giannotti F, Pedreschi D. A survey of methods for explaining black box models. ACM Comput Surv. 2019;51(5):1-42. doi: 10.1145/3236009.
Bhattacharya A. Applied machine learning explainability techniques: make ml models explainable and trustworthy for practical applications using lime shap and more. Packt Publishing Ltd; 2022.
Najafabadi MM, Villanustre F, Khoshgoftaar TM, Seliya N, Wald R, Muharemagic E. Deep learning applications and challenges in big data analytics. J Big Data. 2015;2(1):1-21. doi: 10.1186/s40537-014-0007-7.
Linardatos P, Papastefanopoulos V, Kotsiantis S. Explainable ai: a review of machine learning interpretability methods. Entropy (Basel). 2020;23(1):18. doi: 10.3390/e23010018, PMID 33375658.
Chen Y, Elenee Argentinis JD, Weber G. IBM watson: how cognitive computing can be applied to big data challenges in life sciences research. Clin Ther. 2016;38(4):688-701. doi: 10.1016/j.clinthera.2015.12.001, PMID 27130797.
Rothstein J, Berry J, Svendsen C, Thompson L, Finkbeiner S, Van Eyk J. Answer ALS: a large scale resource for sporadic and familial ALS combining clinical data with multi-omics data from induced pluripotent cell lines; 2020.
ReferencesSingh S, Kaur N, Gehlot A. Application of artificial intelligence in drug design: a review. Comput Biol Med. 2024 Sep;179:108810. doi: 10.1016/j.compbiomed.2024.108810, PMID 38991316.
Adekunle JJ, Lawal MM, Abiodun WO, Nwose CC, Faderin E. The role of AI and ML in drug discovery and development. J Afr Innov Adv Stud; 2024.
Kiriiri GK, Njogu PM, Mwangi AN. Exploring different approaches to improve the success of drug discovery and development projects: a review. Future J Pharm Sci. 2020;6:1-12.
Uppalapati K, Dandamudi E, Ice SN, Chandra G, Bischof K, Lorson CL. A comprehensive guide to enhancing antibiotic discovery using machine learning-derived bio-computation. arXiv preprint arXiv: 241106009; 2024.
Schneider G. Automating drug discovery. Nat Rev Drug Discov. 2018;17(2):97-113. doi: 10.1038/nrd.2017.232, PMID 29242609.
Zeb S, Fnu N, Abbasi N, Fahad M. AI in healthcare: revolutionizing diagnosis and therapy. Int J Multidiscip Sci Arts. 2024;3(3):118-28. doi: 10.47709/ijmdsa.v3i3.4546.
Schork NJ. Artificial intelligence and personalized medicine. Cancer Treat Res. 2019;178:265-83. doi: 10.1007/978-3-030-16391-4_11, PMID 31209850.
Frank R, Hargreaves R. Clinical biomarkers in drug discovery and development. Nat Rev Drug Discov. 2003;2(7):566-80. doi: 10.1038/nrd1130, PMID 12838269.
Akter S. AI driven precision medicine: transforming personalized cancer treatment. J AI Powered Med Innov. 2024;2(1):10-21. doi: 10.60087/vol2iisue1.p21.
Tsiouris KM, Tsakanikas VD, Gatsios D, Fotiadis DI. A review of virtual coaching systems in healthcare: closing the loop with real-time feedback. Front Digit Health. 2020 Sep 30;2:567502. doi: 10.3389/fdgth.2020.567502, PMID 34713040.
Mohanty S, Harun AI Rashid MH, Mridul M, Mohanty C, Swayamsiddha S. Application of artificial intelligence in COVID-19 drug repurposing. Diabetes Metab Syndr. 2020;14(5):1027-31. doi: 10.1016/j.dsx.2020.06.068, PMID 32634717.
Serrano DR, Luciano FC, Anaya BJ, Ongoren B, Kara A, Molina G. Artificial intelligence (AI) applications in drug discovery and drug delivery: revolutionizing personalized medicine. Pharmaceutics. 2024;16(10):1328. doi: 10.3390/pharmaceutics16101328, PMID 39458657.
Shiwlani A, Khan M, Sherani AM, Qayyum MU, Hussain HK. Revolutionizing healthcare: the impact of artificial intelligence on patient care diagnosis and treatment. Jurihum J Inovasi Hum. 2024;1(5):779-90.
Saha S, Ahmad T. Federated transfer learning: concept and applications. Intell Artif. 2021;15(1):35-44. doi: 10.3233/IA-200075.
Beltran ET, Perez MQ, Sanchez PM, Bernal SL, Bovet G, Perez MG. Decentralized federated learning: fundamentals state of the art frameworks trends and challenges. IEEE Commun Surv Tutor. 2023.
Kalusivalingam AK, Sharma A, Patel N, Singh V. Enhancing drug discovery and repurposing through transformer models and reinforcement learning algorithms. Int J AI ML. 2021;2(3).
Kumar G, Yadav S, Mukherjee A, Hassija V, Guizani M. Recent advances in quantum computing for drug discovery and development. IEEE Access. 2024;12:64491-509. doi: 10.1109/ACCESS.2024.3376408.
Pei Z. Computer-aided drug discovery: from traditional simulation methods to language models and quantum computing. Cell Rep Phys Sci. 2024;5(12). doi: 10.1016/j.xcrp.2024.102334.
Prabhod K. Leveraging generative AI for personalized medicine: applications in drug discovery and development. J AI-Assist Sci Discov. 2023;3(1):392-434.
Cumming JG, Davis AM, Muresan S, Haeberlein M, Chen H. Chemical predictive modelling to improve compound quality. Nat Rev Drug Discov. 2013;12(12):948-62. doi: 10.1038/nrd4128, PMID 24287782.
Kaswan KS, Gaur L, Dhatterwal JS, Kumar R. AI-based natural language processing for the generation of meaningful information electronic health record (EHR) data. Advanced AI techniques and applications in bioinformatics. CRC Press; 2021. p. 41-86.
Bhatnagar R, Sardar S, Beheshti M, Podichetty JT. How can natural language processing help model informed drug development? a review. JAMIA Open. 2022;5(2):ooac043. doi: 10.1093/jamiaopen/ooac043, PMID 35702625.
Kolluri S, Lin J, Liu R, Zhang Y, Zhang W. Machine learning and artificial intelligence in pharmaceutical research and development: a review. AAPS J. 2022;24(1):19. doi: 10.1208/s12248-021-00644-3, PMID 34984579.
Garg HK. Integration of AI, ML and blockchain technology: transforming codes into life. IEHE The Quest. 2023;2(1):5-18. doi: 10.5281/zenodo.13885489.
Quazi S. Artificial intelligence and machine learning in precision and genomic medicine. Med Oncol. 2022;39(8):120. doi: 10.1007/s12032-022-01711-1, PMID 35704152.
LI Q, Lei Y, Zhang P, Liu Y, LU Q, Chang C. Future challenges and prospects for personalized epigenetics. In: Personalized epigenetics. Amsterdam: Elsevier; 2024. p. 721-44. doi: 10.1016/B978-0-443-23802-4.00019-3.
Bhambure R, Kumar K, Rathore AS. High throughput process development for biopharmaceutical drug substances. Trends Biotechnol. 2011;29(3):127-35. doi: 10.1016/j.tibtech.2010.12.001, PMID 21255855.
Maharjan R, Lee JC, Lee K, Han HK, Kim KH, Jeong SH. Recent trends and perspectives of artificial intelligence based machine learning from discovery to manufacturing in biopharmaceutical industry. J Pharm Investig. 2023;53(6):803-26. doi: 10.1007/s40005-023-00637-8.
Dara M, Dianatpour M, Azarpira N, Omidifar N. Convergence of CRISPR and artificial intelligence: a paradigm shift in biotechnology. Hum Gene. 2024 May;41:201297. doi: 10.1016/j.humgen.2024.201297.
Omidian H. Synergizing blockchain and artificial intelligence to enhance healthcare. Drug Discov Today. 2024;29(9):104111. doi: 10.1016/j.drudis.2024.104111, PMID 39034026.
Haleem A, Javaid M, Singh RP, Suman R, Rab S. Blockchain technology applications in healthcare: an overview. Int J Intell Netw. 2021;2:130-9. doi: 10.1016/j.ijin.2021.09.005.
Murgai A, Manju K, Paranjpye R, Jain K, Hati M. Integrating artificial intelligence with blockchain: a holistic examination of their combined effects on business performance across various sectors. Library of Progress Library Science Information Technology and Computer. CS H. 2024;44(3).
Tyagi AK. Privacy preservation of genomic and medical data. John Wiley & Sons; 2023.
Singh B, Kaunert C. Blockchain framework for precision medicine clinical trials and genomic biomarkers discovery: focuses on monitoring and managing data in health dimensions. In: Jermsittiparsert K, Marzo RR, editors. AI Technologies and Advancements for Psychological Well-Being and Healthcare. IGI Global; 2024. p. 255-88. doi: 10.4018/979-8-3693-9158-7.ch010.
Nwosu SN. Revolutionizing drug discovery: the impact of artificial intelligence on efficiency and accuracy. J Adv Med Pharm Sci. 2024;26(6):56-63. doi: 10.9734/jamps/2024/v26i6695.
Harrer S, Menard J, Rivers M, Green DV, Karpiak J, Jeliazkov JR. Artificial intelligence drives the digital transformation of pharma. Artificial intelligence in clinical practice. Elsevier; 2024. p. 345-72.
Tien JM. Internet of things real-time decision making and artificial intelligence. Ann Data Sci. 2017;4(2):149-78. doi: 10.1007/s40745-017-0112-5.
Subramanian M, Wojtusciszyn A, Favre L, Boughorbel S, Shan J, Letaief KB. Precision medicine in the era of artificial intelligence: implications in chronic disease management. J Transl Med. 2020;18(1):472. doi: 10.1186/s12967-020-02658-5, PMID 33298113.
Jakka S, Rossbach M. An economic perspective on personalized medicine. HUGO J. 2013;7(1):1-6. doi: 10.1186/1877-6566-7-1.
Alvarez Machancoses O, Fernandez Martinez JL. Using artificial intelligence methods to speed up drug discovery. Expert Opin Drug Discov. 2019;14(8):769-77. doi: 10.1080/17460441.2019.1621284, PMID 31140873.
Chowdhury A, Kunjiappan S, Panneerselvam T, Somasundaram B, Bhattacharjee C. Nanotechnology and nanocarrier based approaches on treatment of degenerative diseases. Int Nano Lett. 2017;7(2):91-122. doi: 10.1007/s40089-017-0208-0.
Petrova N. AI driven drug repurposing for novel therapeutic applications: utilizes ai algorithms to identify existing drugs with potential therapeutic applications in new disease areas. J Artif Intell Res Appl. 2024;4(1):182-90.
Jeyaraman N, Jeyaraman M, Yadav S, Ramasubramanian S, Balaji S. Revolutionizing healthcare: the emerging role of quantum computing in enhancing medical technology and treatment. Cureus. 2024;16(8):e67486. doi: 10.7759/cureus.67486, PMID 39310567.
Sharma P. Quantum computing in drug design: enhancing precision and efficiency in pharmaceutical development. Sage Sci Rev Appl Mach Learn. 2024;7(1):1-9.
Udegbe FC, Ebulue OR, Ebulue CC, Ekesiobi CS. Precision medicine and genomics: a comprehensive review of it-enabled approaches. IMSRJ. 2024;4(4):509-20. doi: 10.51594/imsrj.v4i4.1053.
Nahum Shani I, Smith SN, Spring BJ, Collins LM, Witkiewitz K, Tewari A. Just in time adaptive interventions (JITAIs) in mobile health: key components and design principles for ongoing health behavior support. Ann Behav Med. 2018;52(6):446-62. doi: 10.1007/s12160-016-9830-8, PMID 27663578.
Gordon M. Improving post approval risk surveillance for drugs: active post-market risk identification. Mich Telecomm Tech l Rev. 2008;15:297.
Thacharodi A, Singh P, Meenatchi R, Tawfeeq Ahmed ZH, Kumar RR, VN. Revolutionizing healthcare and medicine: the impact of modern technologies for a healthier future a comprehensive review. Health Care Sci. 2024;3(5):329-49. doi: 10.1002/hcs2.115, PMID 39479277.
Tiwari PC, Pal R, Chaudhary MJ, Nath R. Artificial intelligence revolutionizing drug development: exploring opportunities and challenges. Drug Dev Res. 2023 Dec;84(8):1652-63. doi: 10.1002/ddr.22115, PMID 37712494.
Zeb S, Fnu N, Abbasi N, Fahad M. AI in healthcare: revolutionizing diagnosis and therapy. IJMDSA. 2024;3(3):118-28. doi: 10.47709/ijmdsa.v3i3.4546.
Saha S, Ahmad T. Federated transfer learning: concept and applications. Intell Artif. 2021 Jan 1;15(1):35-44. doi: 10.3233/IA-200075.
Beltran ET, Perez MQ, Sanchez PM, Bernal SL, Bovet G, Perez MG. Decentralized federated learning: fundamentals state of the art frameworks trends and challenges. IEEE Commun Surv Tutor. 2023 Sep 15;25(6):2983-3013. doi: 10.1109/COMST.2023.3315746.
Kalusivalingam AK, Sharma A, Patel N, Singh V. Enhancing drug discovery and repurposing through transformer models and reinforcement learning algorithms. Int J Ai ML. 2021 Feb 15;2(3).
Kumar G, Yadav S, Mukherjee A, Hassija V, Guizani M. Recent advances in quantum computing for drug discovery and development. IEEE Access. 2024 Mar 11. doi: 10.1109/ACCESS.2024.3376408.
Pei T, Wang H, Chen W, Wang S, Pan D. Photovoltaic array fault detection based on a new model of series equivalent resistance. Phys Scr. 2024;99(9). doi: 10.1088/1402-4896/ad6fe1.
Prabhod KJ. Leveraging generative AI for personalized medicine: applications in drug discovery and development. J AI Assist Sci Discov. 2023 Apr 19;3(1):392-434.
Cumming JG, Davis AM, Muresan S, Haeberlein M, Chen H. Chemical predictive modelling to improve compound quality. Nat Rev Drug Discov. 2013 Dec;12(12):948-62. doi: 10.1038/nrd4128, PMID 24287782.
Kaswan KS, Gaur L, Dhatterwal JS, Kumar R. AI based natural language processing for the generation of meaningful information electronic health record (EHR) data. In: Advanced AI techniques and applications in bioinformatics. CRC Press; 2021 Oct 17. p. 41-86. doi: 10.1201/9781003126164-3.
Mitta NR. Application of AI-driven natural language processing in biomedical literature mining: developing deep learning models for automated knowledge extraction hypothesis generation and drug discovery insights. Afr J Artif Intell Sustain Dev. 2023 Oct 9;3(2):361-97.
Kolluri S, Lin J, Liu R, Zhang Y, Zhang W. Machine learning and artificial intelligence in pharmaceutical research and development: a review. AAPS J. 2022 Feb;24(1):19. doi: 10.1208/s12248-021-00644-3, PMID 34984579.
Garg HK. Integration of AI ML and blockchain technology: transforming codes into life. IEHE The Quest. 2023 Jun;2(1):5-18. doi: 10.5281/zenodo.13885489.
Quazi S. Artificial intelligence and machine learning in precision and genomic medicine. Med Oncol. 2022 Jun 15;39(8):120. doi: 10.1007/s12032-022-01711-1, PMID 35704152.
LI Q, Lei Y, Zhang P, Liu Y, LU Q, Chang C. Future challenges and prospects for personalized epigenetics. In: Pers epigenetics. Amsterdam: Elsevier; 2024 Jan 1. p. 721-44. doi: 10.1016/B978-0-443-23802-4.00019-3.
LI Q, Lei Y, Zhang P, Liu Y, LU Q, Chang C. Future challenges and prospects for personalized epigenetics. In: pers epigenetics. Amsterdam: Elsevier; 2024 Jan 1. p. 721-44. doi: 10.1016/B978-0-443-23802-4.00019-3.
Bhambure R, Kumar K, Rathore AS. High throughput process development for biopharmaceutical drug substances. Trends Biotechnol. 2011 Mar 1;29(3):127-35. doi: 10.1016/j.tibtech.2010.12.001, PMID 21255855.
Maharjan R, Lee JC, Lee K, Han HK, Kim KH, Jeong SH. Recent trends and perspectives of artificial intelligence-based machine learning from discovery to manufacturing in the biopharmaceutical industry. J Pharm Investig. 2023 Nov;53(6):803-26. doi: 10.1007/s40005-023-00637-8.
Dara M, Dianatpour M, Azarpira N, Omidifar N. Convergence of CRISPR and artificial intelligence: a paradigm shift in biotechnology. Human Gene. 2024 May 22;41:201297. doi: 10.1016/j.humgen.2024.201297.
Omidian H. Synergizing blockchain and artificial intelligence to enhance healthcare. Drug Discov Today. 2024 Jul 19;29(9):104111. doi: 10.1016/j.drudis.2024.104111, PMID 39034026.
Haleem A, Javaid M, Singh RP, Suman R, Rab S. Blockchain technology applications in healthcare: an overview. Int J Intell Netw. 2021 Jan 1;2:130-9. doi: 10.1016/j.ijin.2021.09.005.
Harshini CS, Murgai A, Manju KV, Paranjpye R, Jain K, Hati M. Integrating artificial intelligence with blockchain: a holistic examination of their combined effects on business performance across various sectors. Libr Prog. 2024 Oct 29;44(3):18587-96.
Tyagi AK, Editor. Privacy preservation of genomic and medical data. John Wiley & Sons; 2023 Nov 16.
Singh B, Kaunert C. Blockchain framework for precision medicine, clinical trials, and genomic biomarkers discovery: focuses on monitoring and managing data in health dimensions. In: Jermsittiparsert K, Marzo RR, editors. AI Technologies and Advancements for Psychological Well-Being and Healthcare. IGI Global; 2024. p. 255-88. doi: 10.4018/979-8-3693-9158-7.ch010.
Serrano DR, Luciano FC, Anaya BJ, Ongoren B, Kara A, Molina G. Artificial intelligence (AI) applications in drug discovery and drug delivery: revolutionizing personalized medicine. Pharmaceutics. 2024 Oct 14;16(10):1328. doi: 10.3390/pharmaceutics16101328, PMID 39458657.
Harrer S, Menard J, Rivers M, Green DV, Karpiak J, Jeliazkov JR. Artificial intelligence drives the digital transformation of pharma. In: Artificial intelligence in clinical practice. Elsevier; 2024 Jan 1. p. 345-72. doi: 10.1016/B978-0-443-15688-5.00049-8.
Tien JM. Internet of things real-time decision making and artificial intelligence. Ann Data Sci. 2017 Jun;4(2):149-78. doi: 10.1007/s40745-017-0112-5.
Subramanian M, Wojtusciszyn A, Favre L, Boughorbel S, Shan J, Letaief KB. Precision medicine in the era of artificial intelligence: implications in chronic disease management. J Transl Med. 2020 Dec;18(1):472. doi: 10.1186/s12967-020-02658-5, PMID 33298113.
Jakka S, Rossbach M. An economic perspective on personalized medicine. HUGO J. 2013 Dec;7(1):1-6. doi: 10.1186/1877-6566-7-1.
Vatansever S, Schlessinger A, Wacker D, Kaniskan HU, Jin J, Zhou MM. Artificial intelligence and machine learning aided drug discovery in central nervous system diseases: state of the arts and future directions. Med Res Rev. 2021 May;41(3):1427-73. doi: 10.1002/med.21764, PMID 33295676.
Chowdhury A, Kunjiappan S, Panneerselvam T, Somasundaram B, Bhattacharjee C. Nanotechnology and nanocarrier based approaches on the treatment of degenerative diseases. Int Nano Lett. 2017 Jun;7(2):91-122. doi: 10.1007/s40089-017-0208-0.
Petrova N. AI driven drug repurposing for novel therapeutic applications: utilizes AI algorithms to identify existing drugs with potential therapeutic applications in new disease areas. J Artif Intell Res Appl. 2024 Jun 7;4(1):182-90.
Jeyaraman N, Jeyaraman M, Yadav S, Ramasubramanian S, Balaji S. Revolutionizing healthcare: the emerging role of quantum computing in enhancing medical technology and treatment. Cureus. 2024 Aug 22;16(8):e67486. doi: 10.7759/cureus.67486, PMID 39310567.
Sharma P. Quantum computing in drug design: enhancing precision and efficiency in pharmaceutical development. Sage Sci Rev Appl Mach Learn. 2024 Jan 8;7(1):1-9.