
Int J App Pharm, Vol 18, Issue 1, 2026, 332-339Original Article
THE EFFECT OF HAIR TONIC FROM SECRETOME MESENCHYMAL STEM CELL ON HAIR GROWTH IN HAIR LOSS CASES
MARLINA1*, NELLA NOVITA2, REGINA ANDAYANI1
1Faculty of Pharmacy, Universitas Andalas, Padang City, West Sumatra-25175, Indonesia. 2Akademi Farmasi Prayoga Padang, Kota Padang, Sumatera Barat-25129, Indonesia
*Corresponding author: Marlina; *Email: marlinaadly2015@gmail.com
Received: 16 Jun 2025, Revised and Accepted: 15 Oct 2025
ABSTRACT
Objective: This study aims to determine the optimum concentration of secretome from mesenchymal stem cells (MSC) in a hair tonic formulation and evaluate its effect on hair growth in individuals experiencing alopecia.
Methods: Tweenty volunteers were divided into five groups to test various hair tonic formulations over eight weeks. Each group received a different concentration of MSC secretome (5%, 7%, 10%) while negative controls consisted of fetal bovine serum and hair tonic base without MSC. Hair density was measured before and after the treatment period to assess the impact of each formulation.
Results: The formulations containing adipose tissue mesenchymal stem cell (ATMSCs) secretome significantly influenced hair growth, with the 10% secretome concentration showing the most substantial increase in hair density among the volunteers. Analysis revealed that the 10% formulation had higher levels of vascular endhotelial growth factor (VEGF) compared to the 5% and 7% formulations, indicating a correlation between VEGF concentration and the stimulation of hair follicle activity.
Conclusion: The MSC hair tonic formula enhanced the hair density of participants after eight weeks, indicating its potential as a treatment to stimulate hair growth in those with alopecia. The study concludes that a higher concentration of MSC secretome, specifically 10%, may yield optimal results in promoting hair regeneration.
Keywords: Stem cell, Secretome, Hair loss, Alopecia, VEGF
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.52744 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Hair serves an important function for humans, including protection from harsh environments such as cold or hot temperatures and ultraviolet rays. Furthermore, hair acts as a temperature regulator, promoting sweat evaporation, and as a sensitive sense of touch [1]. Hair serves two functions in humans: protection and appearance. Beautiful hair frequently boosts a person's self-confidence.
Hair loss, also referred to as alopecia or baldness, is the loss of hair from the scalp [2]. Alopecia is categorized into different types depending upon its pattern of diminished hair growth and its genesis [3]. In general, hair loss occurs as the anagen phase shortens, the catagen phase lengthens, and the hair on the scalp miniaturizes. Even though alopecia is not a life-threatening disease, being bald can cause emotional stress and psychological pressure, as well as be traumatic for sufferers [4, 5].
There are currently only two FDA-approved drugs, minoxidil and finasteride, but their effects are frequently unsatisfactory and transient, as well as causing a variety of side effects [6, 7]. Other treatments involve immunomodulatory mechanisms that suppress T cell proliferation and cytokine synthesis, including IL-1, IL-6, IL-12, TNF-α, and antibody production. These therapies are used for autoimmune disease such as rheumatoid arthritis and inflammatory bowel disease, including alopecia [8]. While they may induce hair regrowth, they do not alter disease progression and are unsuitable for long-term use. Many are not FDA-approved due to serious side effects and limited efficacy. Hair transplant surgery ia another option, but it is expensive and invasive [9].
Several growth factors derived from stem cells and mesenchymal stem cell (MSC) secretions have been shown in studies to promote hair growth. Hair growth and follicle size have been shown to be affected by vascular endothelial growth factor (VEGF) [9]. In this concept, VEGF is a marker of angiogenesis, stimulating hair growth by increasing follicle diameter by facilitating the supply of nutrients to hair follicles.
Shin et al. [10] discovered that after 12 w of treatment with injectable secretome or MSC conditioned medium, hair density into scalp area increased from 105.4 strands/cm2 to 122.7 strands/cm2 and hair thickness increased from 57.5 µm to 64.0 µm. This treatment had no adverse effects on any of the patients.
Adipose tissue mesenchymal stem cells (ATMSCs), which can differentiate into adipogenic, osteogenic, and chondrogenic cells, are widely used in research and clinical therapy [11–13]. There are easier to harvest via subcutaneous lipo aspiration and pose fewer ethical concerns than embryonic stem cells [13]. According to Zanzottera’s [14] research, adipose tissue is a source of multipotent stem cells with hair follicle regenerative potential.
ATMSCs are cultured in growth media containing essential nutrients, growth factors, and hormones, with environtmental conditions such as pH and temperature influencing cell proliferation [15]. The Stem cell secretome produced by stem cells that contains development factors, cytokines, and a membrane structure composed of microvesicles, exosomes, and other factors [16]. These secretomes can be derived from autologous or donor MSCs and are easier to use than live cells. They do not require donor-recipient matching and are simpler to produce, store, and distribute [17].
To date, ATMSCs secretomes have primarily been administered intradermally into the scalp [18–21]. These method requires clinical expertise, is costly, and time-consuming. However, phased studies have shown promising results in treating alopecia [18, 20, 22]. Fukuoka et al. reported no allergic reactions or infections in a study involving 1000 patients, with no persistent pain or discomfort [21].
Simpler and more practical pharmaceutical preparations would be extremely beneficial to people suffering from alopecia. Therefore, cosmetics formulations such as hair tonics containing ATMSC secretomes are being explored for their hair growth effects. Based on previous findings and in vivo research by Aldila [23], which showed increased hair growth in animal models using secretome concentrations of 5% and 10%, this study aims to investigated the hair growth effect of topical hair tonic preparations containing ATMSC secretomes in human volunteers with alopecia.
MATERIALS AND METHODS
Culture of adipose tissue mesenchymal stem cells (ATMSCs)
Subculture began by rinsing confluent ATMSCs in flask T25 (as indicated by the flask's surface, which was overgrown with 70-80% cells). The medium in the flask was discarded, and the flask was rinsed twice with PBS. The cells were then trypsinated by adding 1 ml of 0.5% trypsin-EDTA to the flask and incubating for 5 min in a 5% CO2 incubator at 37 °C to detach the cells from the flask. An inverted microscope is used to examine the cells to see if they have completely detached from the flask. 1 ml of culture medium is added once the cells have detached. The cells were then transferred to a microtube and centrifuged for 5 min at 2000 rpm. The centrifuged pellet was then resuspended in new medium and re-cultured in 2 T25 flasks with 3 ml of growth medium and incubated at 37 °C in a 5% CO2 incubator until the cells were confluent. Starting on the first day of subculture, the medium was replaced every 72 h until the cells were confluent enough to continue subculture [23–27].
Collection of secretomes
ATMSCs that are 80-90% confluent will be transferred to the new falcon. The medium from the ATMSCs culture was then taken and filtered through a 0.22 m filter to remove any contaminants left behind from the ATMSCs culture. The filtrate was collected and stored at-20 °C [23, 26, 28].
Secretomes VEGF concentration determination
Following the manufacturing procedure, the concentration of VEGF in the secretome was determined using the Elisa VEGF KIT.
Ethical clearance
Ethical clearance was secured through a rigorous review process before engaging volunteers for this research. The Ethics Commission of the Faculty of Medicine, Andalas University provided the code of ethics for this study with number 22/UN.16.2/KEP-FK/2023.
Test for irritation
This experiment involved 20 volunteers who were given hair tonic preparations. The preparation is applied behind the ear in a circle and left for 24 h, with skin irritation observed every 4 h [29]. Erythema: no erythema 0, very slight erythema 1, slight erythema 2, moderate erythema 3, very severe erythema 4. Edema: no edema 0, very slight edema 1, slight edema 2, moderate edema 3, very severe edema 4 [30].
Test for hair growth activity
The clinical trial included 20 physically and mentally healthy adults aged 30-70 y old with mild to moderate pattern hair loss (male: Norwood-Hamilton Scale classification type II, IIa, III, IIIa, or IIIvertex, IV, IVa, V; female: Ludwig classification Type I and Type II). Inclusion criteria of the patient is patients who have signed informed consent, patients aged 30-70 y old, The patient has hair loss and the exclusion criteria is people with a history of allergies, people who are taking medication to treat hair loss, people with skin disease [31]. With each preparation, 5 groups of patients (1 group with 4 patients) received hair tonic. For 8 w, patients in all five groups were instructed to apply hair tonic twice daily for 1 ml (morning and evening) and massage the scalp for 1-2 min [28]. The density of hair is measured using a hair analyzer to determine hair activity [28, 31–33].
Data analysis
Data were analyzed by the one-sample Kolmogrov-Smirnov test, followed by the homogeneity of the data using the Levene test. If the data is normal and homogeneous, the ANOVA test is performed, and the hair density data is also subjected to the ANOVA test before being subjected to the Duncan test.
RESULTS AND DISCUSSION
Culture of adipose tissue mesenchymal stem cell (ATMSCs)
Medium Dulbecco's Modified Eagle's Medium (DMEM) is the growth medium used in the subculture process. Invitrogen states that DMEM contains 4 mmol L-glutamine, 4500 mg/l glucose, 1 mmol sodium pyruvate, and 1500 mg/l sodium bicarbonate. When compared to other growth media such as Minimum Essential Medium-Alpha (MEM) and Roswell Park Memorial Institute 1640 (RPMI 1640). DMEM contains 4 times more vitamins and amino acids, 2-4 times more glucose, and additional elements of iron and phenol red [34].
Other ingredients, such as Fetal Bovine Serum (FBS), penicillin, and streptomycin, are required in the preparation of the culture medium. Because DMEM alone is insufficient to meet the nutritional needs of cells during the culture period, FBS, which contains nutrients, hormones, and growth factors to aid cell proliferation and differentiation, is added. FBS is the most commonly used supplement in culture medium. The FBS concentration used for cell culture is 20% of the total medium. At 0.1% concentrations, penicillin and streptomycin are antibacterial and antifungal [35].
The ATMSCs used have entered the fourth life phase, known as passage 4 (P4). Cells that have been subcultured three times are known as P4 cells. Subculture is the process of cultivating cells by releasing cells from cells that have been confluent (the flask has been covered by cells) 80-90% of the time, usually by using a proteolytic enzyme such as trypsin, and then dividing and growing the cells in a new medium so that they enter the living phase. The following subculture is a cell-multiplication process that can make cells more homogeneous and affect the kinetics of cell growth, increasing the potential of cells to treat degenerative diseases.
Collection of secretomes
Secretomes is a growth medium that contains a micture of growth factors, cytokines, and chemokines secreted by stem cells. Basic fibroblast growth factor (bFGF), keratinocyte growth factor, transforming growth factor (TGF), hepatocyte growth factor (HGF), and vascular endothelial growth factor (VEGF) are all secreted by ATMSCs. Secretome can come from a variety of stem cell sources, but each stem cell source produces a unique secretome. Secretomes derived from bone marrow stem cells have higher levels of VEGF and IL-6. Stem cells in the umbilical cord produce high levels of EGF and VEGF. The expression of angiogenic cytokines VEGF and EGF is higher in embryonic stem cells. VEGF, IL-8, and SDF-1 are highly expressed in amniotic fluid stem cells [17]. TGF, VEGF, bFGF, and EGF are all highly expressed in secretome derived from adipose tissue or adipose stem cells [36]. Cytokines found in secretome can promote epithelialization through proliferation, angiogenesis, fibroblast migration and angiogenesis [36].
Determination of VEGF concentration
The ELISA test was used to determine the concentration of VEGF, a growth factor that plays a role in stimulating hair growth. There are several reagents used in this test, including a standard stock solution, which is a standard solution whose optical density will later be measured to obtain a standard calibration curve to calculate the VEGF concentration of the preparation, then a buffer solution, which usually contains Phosphate Buffered Saline (PBS) and is useful for diluting the standard solution and sample according to the desired concentration, The wash solution typically contains sodium chloride (NaCl), and potassium chloride (KCl) is used to wash plates where antibodies or antigens that do not bind successfully will disappear during washing. HRP (horseradish peroxidase) conjugate 2nd Antibody, namely a secondary antibody that has been conjugated with HRP to increase the sensitivity and amplification of the signal on the binding of the primary antibody which is directly bound to the target antigen, the substrate reagent is usually TMB (3,3',5,5'-Tetramethylbenzidine), where the substrate reacts with enzymes to produce a color change, which is later measured as the amount of antibody or antigen, and finally there is a stop solution that stops the reaction, which is usually a mixture of H2SO4 and HCl solutions.
In order to prepare a hair tonic, the concentration of VEGF, a growth factor that stimulates hair growth, must be determined in each secretome concentration (table 1).
The secretome, which is the secretion of mesenchymal stem cells, contains a variety of growth factors and cytokines that can be used as an alternative therapy to stem cell-based therapies. Mesenchymal stem cells contribute to tissue regeneration by differentiating into various tissues. According to recent research, hypoxic-conditioned MSC improve their paracrine abilities. Treatment can boost the immunomodulatory effect of MSC by increasing the secretion of cytokines and soluble factors associated with immunosuppression. Hypoxic preconditioning increases the expression of VEGF, HGF, and FGF, as well as enhances interactions with endothelial cells, all of which contribute to the promotion of angiogenesis.
Table 1: The Result of the concentration determination
| Sample | Repetition | Average | Concentration (pg/ml) | SD |
| F1 5% | 0.057 | 0.056 | 6.142 | 0.894 |
| 0.056 | ||||
| 0.054 | ||||
| 0.058 | ||||
| F2 7% | 0.055 | 0.057 | 6.922 | 1.013 |
| 0.058 | ||||
| 0.061 | ||||
| F3 10% | 0.062 | 0.065 | 11.406 | 3.222 |
| 0.071 |
Alopecia is hair loss that is usually reversible in well-defined areas of the head. Alopecia treatment with growth factors demonstrates intriguing activity in promoting hair growth. Hair growth and follicle size can be controlled by VEGF-mediated angiogenesis. According to this theory, VEGF is an angiogenesis marker that promotes hair growth by improving nutrient supply to hair follicles and increasing follicle diameter [37–39].
Neutrophils, activated macrophages, and fibroblasts secrete VEGF, which appears during the proliferative phase. Through the process of angiogenesis, VEGF will stimulate endothelial cells in neovascular formation. The main factor in the process of angiogenesis, which is the formation of new blood vessel capillaries or the formation of new tissue (granulation tissue) from pre-existing vascular tissue, is the VEGF. Keratinocytes in the outer root sheath of hair produce VEGF. VEGF plays an important role in angiogenesis and influences a variety of cell functions, such as cell survival, proliferation, and the regeneration of nitric oxide (NO) and prostacyclin. Angiogenesis is composed of several stages, including the breakdown of the basement membrane by protease enzymes, endothelial cell migration, proliferation, and differentiation.
The measurement of VEGF levels in hair tonic preparations revealed that the hair tonic formula with 10% secretome contained more VEGF than the formulas with 5% and 7% secretome, implying that the higher the concentration of VEGF in the preparation, the greater the area of the hair follicles to be stimulated, which is consistent with the findings of Yano et al. [9] and Gnann et al. [40] where the researchers discovered that the higher the concentration of VEGF in the gel sample, the larger the area of the hair follicle. In animal studies, Yano et al. [9] identified VEGF as a key mediator of hair follicle growth and cycling and provided direct evidence that increased follicular vascularity correlates with increased hair growth and hair follicle size.
Furthermore, growth factors such as IGF-1/IGF binding protein-1 complex and BMP act on DPCs to rebuild and maintain hair induction capabilities [41, 42]. Indeed, IGF-1 regulates the hair development cycle and the differentiation of the hair shaft. Transgenic mice that express IGF-1 in the inner root sheath exhibit follicular proliferation, tissue remodeling, hair growth cycle, and follicular differentiation [43]. HGF, another paracrine hormone, may promote follicular growth by increasing β-catenin expression [44]. Three groups of mice received daily dorsal intradermal injections of 1 µg HGF/SF in 0.1% albumin phosphate-buffered saline for 5-7 days. They demonstrated longer and larger HF in newborn mice, as well as retention of anagen HF after 10 days, indicating a delay in the transition from anagen to catagen.
Test for irritation
One of the important parameters to consider for topical preparations, such as hair tonic, is the appearance of skin irritation if the hair tonic preparation is applied to the scalp. Irritation is a skin reaction caused by a variety of factors, such as the duration of administration, the area of administration, the degree of penetration, and the toxicity of the preparations used [45]. Skin irritation is caused by an inflammatory reaction caused by keratinocytes releasing proinflammatory cytokines. It usually happens in response to chemical stimuli that cause disruption of the skin barrier, changes in epidermal cells, and cytokine release [46]. There are two types of irritation: primary irritation, which appears immediately after the preparation reaches the scalp, and secondary irritation, which appears after several hours of use. Irritation can occur after some time of using hair tonic preparations, and it is characterized by symptoms such as dry skin that is painful to the touch and bleeding. The presence of erythema and edema characterizes the irritation that develops. Whereas erythema or redness is caused by dilation of blood vessels in the irritated area, edema [47].
An irritation test was performed to determine which hair tonic preparations were deemed safe for daily use by volunteers if they did not cause scalp irritation. Two qualitative and quantitative observations were made during the primary irritation test. The qualitative observation determined whether or not each formula caused erythema and edema. Furthermore, the skin reaction was assessed by categorizing the erythema and edema scores. The quantitative observation was then statistically analyzed by calculating the primary irritation index.
In theory, the erythema and edema scores on the skin of those who received irritant compounds were higher than those who did not receive irritant compounds. The results of the two groups were then combined to produce the Primary Irritation Index (PII), which is scored from 0 to 4 depending on the severity of the skin reaction seen (No erythema = 0, Very slight (almost imperceptible) erythema = 1, Visible erythema (diameter 25.1-30 mm) = 2, Moderate erythema (diameter 30.1-35 mm) = 3, and Severe [48–50].
The irritation test on 20 volunteers revealed no erythema or edema when using hair tonic preparations during the test. It is hoped that hair tonic preparations will not cause primary irritation during use. Primary irritation testing is necessary for all types of topical preparations to determine their ability to irritate the skin and the safety level of the preparations made [51].
Test for hair growth activity
Subjects were randomly selected depending on a doctor's diagnosis. Inclusion criteria were healthy volunteers aged 18 y or more. Exclusion criteria encompassed self-reported hair loss within the preceding 6 mo, the presence of underlying systemic illnesses, and lacking Asian ancestry, having a history of using medication or hair products that affect the hair growth cycle, a positive hair-pull test, abnormal hair or scalp observed on physical examination, and those with abnormalities. Male and female participants displayed comparable hair density patterns, with more density in the vertex and less density in the temporoparietal region. Hair density and diameter were not substantially different between male and female individuals. So, gender will not make a significant difference in findings. People with hair loss/alopecia were given hair tonic preparations in this study. Alopecia is a hair loss condition that affects both men and women. Although alopecia is not a life-threatening disease, it can cause emotional and traumatic stress in sufferers [4].
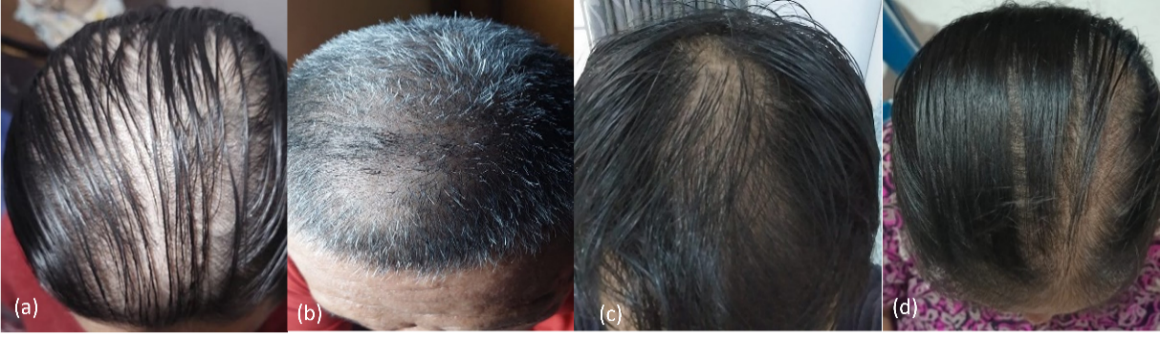
Fig. 1: (a) volunteers with the Ludwig classification type II; (b) volunteers with Norwood-Hamilton classification type II a; (c) volunteers with Norwood-Hamilton classification type III vertex; and (d) volunteers with the Ludwig classification type II
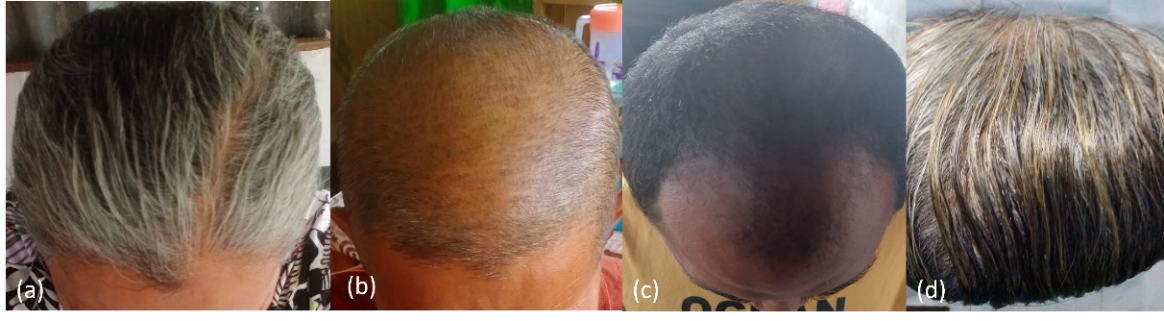
Fig. 2: (a) volunteers with the Ludwig classification type I; (b) volunteers with Norwood-Hamilton classification type V a; (c) volunteers with Norwood-Hamilton classification type IV; and (d) volunteers with the Ludwig classification type II
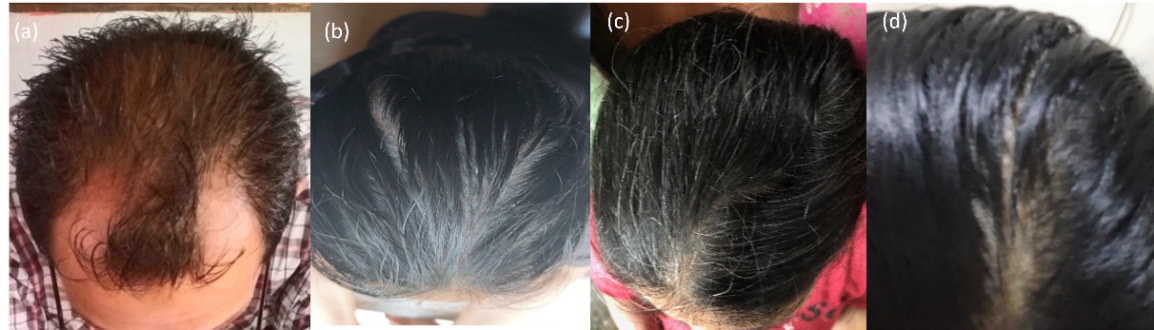
Fig. 3: (a) volunteers with the Norwood-Hamilton classification type III vertex; (b) volunteers with Ludwig classification type I; (c) volunteers with Ludwig classification type I; and (d) volunteers with the Ludwig classification type I
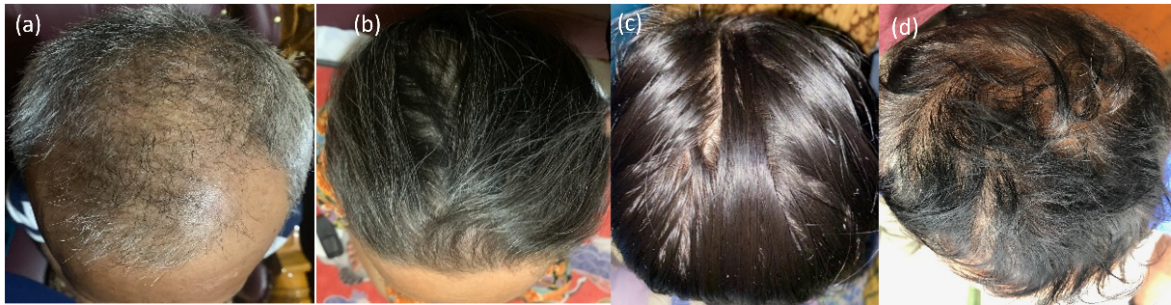
Fig. 4: (a) volunteers with the Norwood-Hamilton classification type IV a; (b) volunteers with Ludwig classification type I; (c) volunteers with Ludwig classification type I; and (d) volunteers with the Norwood-Hamilton classification type IV
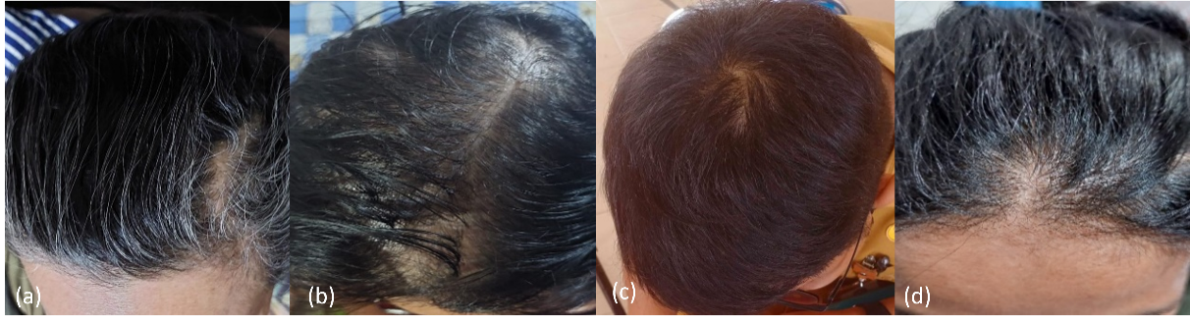
Fig. 5: (a) volunteers with the Ludwig classification type I; (b) volunteers with Ludwig classification type I; (c) volunteers with Norwood-Hamilton classification type III vertex; and (d) volunteers with the Ludwig classification type I
After one month of using hair tonic preparations, volunteers were observed because normal hair growth per day is between 0.00033 m and 0.01 m per month [52, 53]. The density of hair follicles in typical scalp hair ranges from 0.02 to 0.04 per mm2. Hair growth is normally influenced by factors such as hair growth speed and duration, as well as dilation of blood vessels in the hair that supply blood during the anagen phase [54]. The hair density of 20 volunteers who used hair tonic preparations is shown in the table 3-7.
The subject's hair growth was measured using a hair analyzer. A hair analyzer is a device that measures many aspects of hair, including hair density, which refers to the amount of hair strands in a certain area of the scalp. Measuring hair density can provide important information for a variety of purposes, including analyzing hair loss, determining the success of hair treatments, and just knowing an individual's hair health.
Hair growth each day ranges between 0.00033 m and 0.01 m every month [52, 53], hence, the analysis is performed every four weeks. The analyzer typically measures a specific area of the scalp (for example, 0.0001 m2) and counts the number of hair follicles or strands within that area.
Table 2: Hair density by providing hair tonic base
| Base | Before use | After 4 w of use | After 8 w of use |
| Volunteer 1 | 10% ±0.08 | 10% ±0.08 | 10% ±0.08 |
| Volunteer 2 | 27% ±0.08 | 27% ±0.08 | 27% ±0.08 |
| Volunteer 3 | 28% ±0.08 | 28% ±0.08 | 28% ±0.08 |
| Volunteer 4 | 17% ±0.08 | 17% ±0.08 | 17% ±0.08 |
Notes: Results presented as mean±SD (n=4)
Table 3: Hair density by providing hair tonic base+FBS
| Base+FBS | Before use | After 4 w of use | After 8 w of use |
| Volunteer 5 | 11% ±0.04 | 10% ±0.04 | 10% ±0.04 |
| Volunteer 6 | 19% ±0.04 | 19% ±0.04 | 19% ±0.04 |
| Volunteer 7 | 10% ±0.04 | 10% ±0.04 | 10% ±0.04 |
| Volunteer 8 | 14% ±0.04 | 14% ±0.04 | 14% ±0.04 |
Notes: results presented as mean±SD (n=4)
Table 4: Hair density by providing 5% secretome hair tonic
| F1 (Secretome 5%) | Before use | After 4 w of use | After 8 w of use |
| Volunteer 9 | 54%±0.18 | 68% ±0.23 | 77% ±0.23 |
| Volunteer 10 | 19%±0.18 | 23% ±0.23 | 31% ±0.23 |
| Volunteer 11 | 33%±0.18 | 39% ±0.23 | 40% ±0.23 |
| Volunteer 12 | 12%±0.18 | 15% ±0.23 | 24% ±0.23 |
Notes: Results presented as mean±SD (n=4)
Table 5: Hair tonic by providing 7% secretome hair tonic
| F2 (Secretome 7%) | before use | After 4 w of use | After 8 w of use |
| Volunteer 13 | 56% ±0.20 | 65% ±0.21 | 80% ±0.26 |
| Volunteer 14 | 16% ±0.20 | 22% ±0.21 | 29% ±0.26 |
| Volunteer 15 | 10% ±0.20 | 20% ±0.21 | 23% ±0.26 |
| Volunteer 16 | 22% ±0.20 | 26% ±0.21 | 29% ±0.26 |
Notes: Results presented as mean±SD (n=4)
Table 6: Hair density by providing 10% secretome hair tonic
| F3 (Secretome 10%) | Before use | After 4 w of use | After 8 w of use |
| Volunteer 17 | 10% ±0.30 | 25% ±0.31 | 21% ±0.32 |
| Volunteer 18 | 80% ±0.30 | 93% ±0.31 | 98% ±0.32 |
| Volunteer 19 | 41% ±0.30 | 58% ±0.31 | 64% ±0.32 |
| Volunteer 20 | 20% ±0.30 | 30% ±0.31 | 42% ±0.32 |
Notes: Results presented as mean±SD (n=4)
Hair tonic preparations are an additional option to hair cosmetic preparations. Hair tonic preparations come in the form of an easy-to-apply solution that can be spread on the scalp's surface and is not sticky like gel or other semisolid preparations. As a result, it does not leave residue that can cause scale on the scalp, allowing dandruff to be avoided [55].
The active ingredients used in the manufacture of hair tonic preparations are ATMSCs secretomes, while the preparation base is 96% ethanol, propylene glycol, phenoxy ethanol, which is also used by as a solvent, 96% ethanol is used, which also improves penetration into the skin. Propylene glycol is a humectant that can increase the solubility of the formulation's ingredients as well as the viscosity of the preparation. Phenoxy ethanol is used as a preservative because it has a broad spectrum of antimicrobial activity against a variety of Gram-negative and Gram-positive bacteria, as well as fungal infections, and its maximum use is 1% [56, 57].
The control groups received fetal bovine serum and a hair tonic base (placebo). Placebos can help assess the psychological impact of treatment. Many people may notice improvements in their hair health or look merely because they believe they are receiving excellent therapy. Genetics, food, and environmental variables can all have an impact on hair growth and overall health.
Interactions between epithelial and mesenchymal stem cells influence hair follicle development. Many genes are involved in this interaction as well as hair follicle cycling[58]. Growth factors such as IGF-1, fibroblast growth factor-7 (FGF-7), HGF, and VEGF are required during the anagen phase of hair growth.
Approximately 85-90% of all hair on the scalp is anagen [59]. In this phase, VEGF is one of the growth factors required for the process of growing the hair shaft.
Adipose tissue mesenchymal stem cell secretomes can repair the scalp, which is consistent with the findings Narita et al. [19] whereas the MSCs secretome of adipose tissue promotes hair growth via the gradual spread of paracrine effects across the scalp in the interfollicular dermis, as well as a direct paracrine effect on dermal follicular papilla cells.
It is possible to overcome hair loss by increasing blood flow around the hair follicles to supply essential nutrients to the hair roots so that hair can grow by inducing angiogenesis, which is mediated by VEGF to increase blood flow. Hair follicles require nutrients, particularly during the anagen phase, which is responsible for micro-vascularization reorganization, endothelial cell proliferation, and cell division.
Alopecia treatments based on growth factors demonstrate intriguing activity in promoting hair growth. According to this theory, VEGF stimulates hair growth by facilitating the supply of nutrients to hair follicles, increasing the diameter of the follicles, which is consistent with the findings of Gnann et al. [40], who found an increase in the number of hairs in test animals given preparations containing VEGF growth factor. VEGF expression in hair follicles is significantly lower in people with hair loss/alopecia compared to normal hair follicles [60].
The data analysis test used is a two-way ANOVA analysis, but first the data's normality and homogeneity must be tested. The data were normally distributed (p>0.05) in the Kolmogorv-Smirnov one-sample normality test, with a value of 0.200. The homogeneity test using Levene's test with homogeneous data yielded 0.593 (p>0.05). So that the requirements for performing the ANOVA test can be met. According to the SPSS ANOVA results, the effect of hair tonic preparations containing mesenchymal stem cell secretomes on hair growth was significant (Sig.) 0.000. If the p-value is less than 0.05, H0 is rejected, indicating that hair tonic preparations containing adipose tissue mesenchymal stem cell secretomes promote hair growth.
From the research that has been carried out, there are no visible side effects from using secretome as a hair growth agent. Katagiri's research [61], which included a histological evaluation after 6 mo, revealed that the experimental group had significantly higher levels of bone regeneration and angiogenesis than the control group. Apart from that, no negative consequences happened. However, further research needs to be done regarding the delayed side effects of using this hair tonic.
CONCLUSION
According to the research, hair tonic preparations containing adipose tissue mesenchymal stem cell secretome have an effect on hair growth, as evidenced by an increase in volunteer hair density after using the hair tonic, with a secretome content of 10% providing the best effect. Simpler and more practical pharmacological formulations would be extremely beneficial to persons suffering from hair loss/alopecia. As a result, it is required to develop cosmetic preparations that are simple to use, such as hair tonic preparations derived from the secretome of adipose tissue mesenchymal stem cells, which produce a hair growth impact. Because it has been shown to be beneficial in boosting the hair density of volunteers, it is hoped that the hair tonic formulation containing adipose tissue mesenchymal stem cell secretome may promote hair growth for those who suffer from hair loss/alopecia.
ACKNOWLEDGMENT
This research was supported by BIMA-Kemdikbudristek from Ministry of Research, Technology, and Higher Education of Republic Indonesia with a contract number 93/UN16.19/PT.01.03/2023. The author would like to thank LPPM (The Institute for Community Service Research) Universitas Andalas for the writing supervision workshop.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
Marlina and Regina Andayani were responsible for conceptualizing the study and supervising the manuscript development. Nella Novita contributed to the methodological design, preformed data analysis, and prepared the original draft of the manuscript.
CONFLICT OF INTERESTS
Declared none
REFERENCES
Yasir AS. Hair growth activity test of male rabbit with hair tonic preparation which contains ethanol extract of Nothopanax scutellarium L. J Farm Mahayati. 2019;2(1).
National Health Service. Hair loss overview; 2021. Available from: https://www.nhs.uk/conditions/hair-loss/. [Last accessed on 08 Nov 2025].
Bhaware P, Kore P. Systematic review on synthetic and herbal medications for the treatment of alopecia. Asian Journal of Pharmaceutical and Clinical Research. 2024;17(12):22-7. doi: 10.22159/ajpcr.2024v17i12.52284.
Patel S, Sharma V, Chauhan NS, Thakur M, Dixit VK. Hair growth: focus on herbal therapeutic agent. Curr Drug Discov Technol. 2015;12(1):21-42. doi: 10.2174/1570163812666150610115055, PMID 26058803.
Nalluri R, Harries M. Alopecia in general medicine. Clin Med (Lond). 2016;16(1):74-8. doi: 10.7861/clinmedicine.16-1-74, PMID 26833522.
Gupta AK, Charrette A. Topical minoxidil: systematic review and meta-analysis of its efficacy in androgenetic alopecia. Skinmed. 2015;13(3):185-9. PMID 26380504.
Falto Aizpurua L, Choudhary S, Tosti A. Emerging treatments in alopecia. Expert Opin Emerg Drugs. 2014;19(4):545-56. doi: 10.1517/14728214.2014.974550, PMID 25330928.
Alsantali A. Alopecia areata: a new treatment plan. Clin Cosmet Investig Dermatol. 2011;4:107-15. doi: 10.2147/CCID.S22767, PMID 21833161.
Yano K, Brown LF, Detmar M. Control of hair growth and follicle size by VEGF-mediated angiogenesis. J Clin Invest. 2001;107(4):409-17. doi: 10.1172/JCI11317, PMID 11181640.
Shin H, Ryu HH, Kwon O, Park BS, Jo SJ. Clinical use of conditioned media of adipose tissue-derived stem cells in female pattern hair loss: a retrospective case series study. Int J Dermatol. 2015;54(6):730-5. doi: 10.1111/ijd.12650, PMID 25777970.
Bunnell BA, Flaat M, Gagliardi C, Patel B, Ripoll C. Adipose-derived stem cells: isolation, expansion and differentiation. Methods. 2008;45(2):115-20. doi: 10.1016/j.ymeth.2008.03.006, PMID 18593609.
Foubert P, Gonzalez AD, Teodosescu S, Berard F, Doyle Eisele M, Yekkala K. Adipose-derived regenerative cell therapy for burn wound healing: a comparison of two delivery methods. Adv Wound Care. 2016;5(7):288-98. doi: 10.1089/wound.2015.0672, PMID 27366590.
Miana VV, Gonzalez EA. Adipose tissue stem cells in regenerative medicine. Ecancermedicalscience. 2018;12(822):822. doi: 10.3332/ecancer.2018.822, PMID 29662535.
Zanzottera F, Lavezzari E, Trovato L, Icardi A GA. Adipose derived stem cells and growth factors applied on hair transplantation. Follow-up of clinical outcome. J Cosmet Dermatol Sci Appl. 2014;4(4):268-74. doi: 10.4236/jcdsa.2014.44036.
Gibco. Cell culture basics handbook. United Kingdom: Thermo Fisher; 2015.
Marlina PR, Pradifta R, Lucida H, Sudji IR, Salsabila HN, Elida N. Formulation of mesenchymal stem cell secretome as antiaging cream. Int J App Pharm. 2023;15(1):45-50. doi: 10.22159/ijap.2023.v15s1.47506.
Pawitan JA. Prospect of stem cell conditioned medium in regenerative medicine. Bio Med Res Int. 2014;2014:965849. doi: 10.1155/2014/965849, PMID 25530971.
Fukuoka H, Suga H. Hair regeneration treatment using stem cell conditioned medium. Japan J Plast Surg. 2012;55(9):1083.
Narita K, Fukuoka H, Sekiyama T, Suga H, Harii K. Sequential scalp assessment in hair regeneration therapy using an adipose-derived stem cell–conditioned medium. Dermatol Surg. 2020;46(6):819-25. doi: 10.1097/DSS.0000000000002128, PMID 31490301.
Fukuoka H, Suga H. Hair regeneration treatment using adipose derived stem cell conditioned medium: follow-up with trichograms. Eplasty. 2015;15(10):e10. PMID 25834689.
Fukuoka H, Narita K, Suga H. Hair regeneration therapy: application of adipose-derived stem cells. Curr Stem Cell Res Ther. 2017;12(7):531-4. doi: 10.2174/1574888X12666170522114307, PMID 28530535.
Fukuoka H, Suga H, Narita K, Watanabe R, Shintani S. The latest advance in hair regeneration therapy using proteins secreted by adipose-derived stem cells. Am J Cosmet Surg. 2012;29(4):273-82. doi: 10.5992/AJCS-D-12-00015.1.
Aldila S. In vivo evaluation of hair growth effect of hair tonic formulation containing mesenchymal stem cell-conditioned medium. Universitas Andalas; 2021.
Oh HA, Kwak J, Kim BJ, Jin HJ, Park WS, Choi SJ. Migration inhibitory factor in conditioned medium from human umbilical cord blood-derived mesenchymal stromal cells stimulates hair growth. Cells. 2020;9(6):1344. doi: 10.3390/cells9061344, PMID 32481584.
Marlina M, Rahmadian R, Sudji IR, Handayana V, Safitri M, Elida N. Synovial membrane mesenchymal stem cell [SM-MSC] induced IGF-1 promote growth factor release in conditioned medium SM-MSC. IOP Conf Ser: Earth Environ Sci. 2021;741(1):012067. doi: 10.1088/1755-1315/741/1/012067.
Pradifta R. Anti-aging effect of a cream formulation containing mesenchymal stem cell-conditioned medium. Universitas Andalas; 2021.
Venitasya S. Effect of conditioned medium from adipose tissue mesenchymal stem cells (ATMSCs) on second-degree (IIA) burn wounds in mice. Universitas Andalas; 2021.
Han HS, Park KY, Ko EJ, Kim BJ. Efficacy of a hair tonic containing human umbilical cord blood mesenchymal stem cell-derived conditioned media in patients with androgenetic alopecia. Korean Journal of Dermatology. 2019;57(5):251-7.
POM D. Formularium Kosmetika. Indonesia. Vol. 86. Jakarta: Departemen Kesehatan RI; 1985. p. 206-19.
Barel AO, Paye M, Maibach HI, editors. Handbook of cosmetic science and technology. 2nd ed. New York: John Wiley & Sons, Inc; 2009. p. 626-9.
Kubanov AA, Gallyamova YA, Korableva OA. The study of growth factors in patients with androgenic alopecia. Biomed Pharmacol J. 2017;10(3):1219-28. doi: 10.13005/bpj/1224.
Chandra P. Formulation and evaluation of a water-resistant hair tonic gel containing extract and fractions of date fruit (Phoenix dactylifera L.). Universitas Sumatera Utara; 2020.
Oh HA, Kwak J, Kim BJ, Jin HJ, Park WS, Choi SJ. Migration inhibitory factor in conditioned medium from human umbilical cord blood-derived mesenchymal stromal cells stimulates hair growth. Cells. 2020;9(6):1344. doi: 10.3390/cells9061344, PMID 32481584.
Rhamayanti Y. The importance of basic teaching skills for mathematics education students in field experience practice. EKSAKTA: Journal of Mathematics & Learning. 2018;3(1):65-72.
Noverina R, Widowati W, Ayuningtyas W, Kurniawan D, Afifah E, Laksmitawati DR. Growth factors profile in conditioned medium human adipose tissue-derived mesenchymal stem cells (CM-hATMSCs). Clin Nutr Exp. 2019 Apr;24:34-44. doi: 10.1016/j.yclnex.2019.01.002.
Zhou BR, Xu Y, Guo SL, Xu Y, Wang Y, Zhu F. The effect of conditioned media of adipose-derived stem cells on wound healing after ablative fractional carbon dioxide laser resurfacing. BioMed Res Int. 2013;2013:519126. doi: 10.1155/2013/519126, PMID 24381938.
Garcia Bartels N, Jahnke I, Patzelt A, Richter H, Lademann J, Blume Peytavi U. Hair shaft abnormalities in alopecia areata evaluated by optical coherence tomography. Skin Res Technol. 2011;17(2):201-5. doi: 10.1111/j.1600-0846.2010.00484.x, PMID 21241369.
Kim MJ, Lim C, Lee JY, Im KR, Yoon KS, Song JM. Visible-to-near ir quantum dot–based hypermulticolor high content screening of herbal medicines for the efficacy monitoring of hair growth promotion and hair loss inhibition. J Biomol Screen. 2013;18(4):462-73. doi: 10.1177/1087057112464574, PMID 23190736.
Ozeki M, Tabata Y. In vivo promoted growth of mice hair follicles by the controlled release of growth factors. Biomaterials. 2003;24(13):2387-94. doi: 10.1016/S0142-9612(03)00045-0, PMID 12699676.
Gnann LA, Castro RF, Azzalis LA, Feder D, Perazzo FF, Pereira EC. Hematological and hepatic effects of vascular epidermal growth factor (VEGF) used to stimulate hair growth in an animal model. BMC Dermatol. 2013;13(15):15. doi: 10.1186/1471-5945-13-15, PMID 24168457.
Bak DH, Choi MJ, Kim SR, Lee BC, Kim JM, Jeon ES. Human umbilical cord blood mesenchymal stem cells engineered to overexpress growth factors accelerate outcomes in hair growth. Korean J Physiol Pharmacol. 2018;22(5):555-66. doi: 10.4196/kjpp.2018.22.5.555, PMID 30181702.
Rendl M, Polak L, Fuchs E. BMP signaling in dermal papilla cells is required for their hair follicle inductive properties. Genes Dev. 2008;22(4):543-57. doi: 10.1101/gad.1614408, PMID 18281466.
Weger N, Schlake T. Igf-I signalling controls the hair growth cycle and the differentiation of hair shafts. J Invest Dermatol. 2005;125(5):873-82. doi: 10.1111/j.0022-202X.2005.23946.x, PMID 16297183.
Qi Y, Li M, Xu L, Chang Z, Shu X, Zhou L. Therapeutic role of human hepatocyte growth factor (HGF) in treating hair loss. Peer J. 2016;4:e2624. doi: 10.7717/peerj.2624, PMID 27833804.
More BH, Sakhawarde SN, Tembhurne SV, Sakarkar D. Evaluation for skin irritancy testing of developed formulations containing extract of Butea monosperma for its topical application. Int J Toxicol Appl Pharmacol. 2013;3(1):10-3.
Nugraha A, Nugrahaningsih DA, Syarif RA, Yuniyanti MM, Salim IN, Sari R. Acute irritation of tetracyclic c-4-hydroxyphenylcalics [4] pyrogalolarene on skin of albino rabbits. Indones J Pharmacol Ther. 2020;1(1):15-21. doi: 10.22146/ijpther.480.
Irsan MA, Manggav E, Pakki U. Irritation test of antioxidant cream containing longan seed extract (Euphoria longana stend) on rabbit skin (Oryctolagus cuniculus). Maj Farm Dan Farmakol. 2013;17(2):55-60.
Banerjee S, Chattopadhyay P, Ghosh A, Pathak MP, Singh S, Veer V. Acute dermal irritation sensitization and acute toxicity studies of a transdermal patch for prophylaxis against ({±}) anatoxin-a poisoning. Int J Toxicol. 2013;32(4):308-13. doi: 10.1177/1091581813489996, PMID 23696561.
Dewantara IG, Jemmy IG, Nntan P, Damips A, Prabayanti NP. In vivo evaluation of erythema and edema induced by 10% sodium lauryl sulfate. J Farm Udayana. 2015;4(2):55-60.
Patrick E, Maibach HI. Comparison of the time course, dose response and mediators of chemically induced skin irritation in three species. In: Frosch P, Dooms Goossens A, Lachapelle JM, Rycroft RJ, Scheper RJ, editors. Current topics in contact dermatitis. Germany: Springer; 1989. p. 399-403. doi: 10.1007/978-3-642-74299-6_81.
Lu FC. Toksikologi dasar: asas organ sasaran dan penilaian resiko. Edisi II. Jakarta: Penerbit Universitas Indonesia; 1995. p. 428.
Mulyanti GD, Nurhayati Y, Ariska A. Evaluation of hair growth effects of hir tonic formulation from yardlong bean leaf extract (Vigna sinensis (L.) Savi ex Hassk) on male rabbits. Wellness Heal Mag. 2019;1(2):285-94.
Murphrey MB, Agarwal S, Zito PM. Anatomy hair. StatPearls. Treasure Island (FL): StatPearls Publishing; 2022.
Ittiqo DH, Jeniti P, Hati MP, Nurbaety B, Wahid AR. Uji aktivitas hair tonic madu kombinasi ekstrak daun seledri (Apium graveolens Linn) terhadap pertumbuhan rambut kelinci Jantan. Lumbung Farmasi: Jurnal Ilmu Kefarmasian. 2022;3(1):55. doi: 10.31764/lf.v3i1.7406.
Noviani V, Thauresia S, Simanjuntak P. Uji aktivitastonik rambut yang mengandung fraksi air yang mengandung flavonoid dari ekstrak etanol daun teh hijau (Camellia sinensis L.). FARM. 2019;6(1):22. doi: 10.47653/farm.v6i1.524.
Swarbrick J, Boylan JC. Encyclopedia of Pharmaceutical Technology. 2nd ed. Vol. 1. New York: Marcel Deker Inc; 2002.
Williams AC, Barry BW. Penetration enhancers. Adv Drug Deliv Rev. 2012;64:128-37. doi: 10.1016/j.addr.2012.09.032.
Cotsarelis G, Botchkarev V. Biology of hair follicles. Fitzpatricks dermatology in general medicine. 7th ed. New York: McGraw-Hill; 2008. p. 739-49.
Erdogan B. Anatomy and physiology of hair. Hair Scalp Disord. 2017;13:1-7. doi: 10.5772/67269.
Goldman CK, Tsai JC, Soroceanu L, Gillespie GY. Loss of vascular endothelial growth factor in human alopecia hair follicles. J Invest Dermatol. 1995;104(5)Suppl:18S-20S. doi: 10.1038/jid.1995.40, PMID 7738377.
Katagiri W, Watanabe J, Toyama N, Osugi M, Sakaguchi K, Hibi H. Clinical study of bone regeneration by conditioned medium from mesenchymal stem cells after maxillary sinus floor elevation. Implant Dent. 2017;26(4):607-12. doi: 10.1097/ID.0000000000000618, PMID 28727618.