
Int J App Pharm, Vol 17, Issue 2, 2025, 12-30Review Article
RECENT ADVANCES IN TREATMENT APPROACHES FOR DIABETES MELLITUS AND RELATED COMPLICATIONS: A REVIEW
SIDDHANT DHYANI1 , MANSI BUTOLA1*
, MANSI BUTOLA1* , VANSHIKA SAUTHA1
, VANSHIKA SAUTHA1 , VIKASH JAKHMOLA2
, VIKASH JAKHMOLA2
1Department of Pharmaceutics, Uttaranchal Institute of Pharmaceutical Sciences, Uttaranchal University, Dehradun, Uttarakhand-248007, India. 2Department of Pharmaceutical Chemistry, Uttaranchal Institute of Pharmaceutical Sciences, Uttaranchal University, Dehradun, Uttarakhand-248007, India
*Corresponding author: Mansi Butola; *Email: mansibutola1995@gmail.com
Received: 13 Nov 2024, Revised and Accepted: 04 Feb 2025
ABSTRACT
Diabetes Mellitus (DM) can be treated with a variety of therapeutic approaches. Patients are forced to initiate therapy with antidiabetic agents when diet and exercise are ineffective to regulate hyperglycemia. However, these drugs have several disadvantages that can influence the course of treatment. The primary drawbacks of the current oral modalities for the treatment of DM are the immediate release of the drug and the low bioavailability, which necessitates an increase in the frequency of dosing. Patient compliance to therapy decreases in conjunction with the manifestation of adverse side effects. The development of innovative delivery modalities that have the potential to improve the efficacy of anti-diabetic regimens has been a fertile area for nanotechnology in recent years. The primary objective of all attempts has been to (a) safeguard the drug by encapsulating it in a nano-carrier system and (b) release the drug in a controlled and progressive manner using effective techniques. The current review aims to compile effective nanocarriers like polymeric nanoparticles (NPs), liposomes, niosomes, dendrimers, micelles, solid lipid NPs, transfersomes, ethosomes, nanofibers, and carbon nanotubes for the treatment of diabetes mellitus, emerging treatment strategies and various complications related to this disease.
Keywords: Nanotechnology, Diabetes mellitus, Patient compliance, Hyperglycemia, Nanocarriers
© 2025 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2025v17i2.53184 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Diabetes has become one of the most significant global health and economic burdens due to its high complexity ratio in increasing number of cases. Diabetes mellitus, a prevalent chronic metabolic disorder, is represented by elevated blood glucose levels that result from neither insulin insensitivity, inadequate insulin secretion, or a combination of the two. Numerous reports have demonstrated a substantial increase in the number of individuals with diabetes, indicating that the proportion of people with the condition is continuously increasing on a global scale. Diabetes is a metabolic condition that occurs and is characterised by high glucose levels in the bloodstream. Type 1 diabetes mellitus is an autoimmune form of diabetes which frequently develops in young adults due to an insufficient insulin supply. On the other hand, type II diabetes mellitus is not insulin-dependent in responsible for approximately 90% of all diabetes cases. This form of diabetes is typically diagnosed in adults and is caused by lifestyle factors including physical activity and diet. Insulin, a hormone secreted by the β cells of the pancreas, is essential for the metabolization of glucose derived from food which is used generate energy [1]. Individuals with type I diabetes have a pancreas that either produces insufficient quantities of insulin or does not produce any insulin at all. This type of diabetes is typically detected in infants and adolescents, even though it can affect anyone at any age. Insulin injections are required daily to maintain blood glucose levels in type II diabetes mellitus, the pancreas may fail to secrete inadequate amount of insulin, or the insulin that is produced may not be effectively used by the liver, muscles, and adipose cells, resulting in a condition known as insulin resistance. As a result of insulin resistance cells require an increase the mouth of insulin to convert glucose into energy, which in turn results in hyperglycaemia and raised blood sugar levels [2]. The development of type II diabetes mellitus is frequently influenced by environmental and behavioural risk factors, genetic predisposition, and lifestyle choices. The management and treatment of type II diabetes mellitus is challenging due to the disease’s progressive nature. To lower the overall risks associated with type II diabetes mellitus, it is crucial to regularly monitor several factors, such as blood pressure, lipid profiles, and glucose numbers. By providing the required insulin replacement, insulin therapy seems to be a key part in treating all diabetic patients. Serious and even deadly health problems, including lower limb amputation, nerve damage neuropathy, eyesight impairment, various cardiovascular disorders, and renal failure, can result from poor disease management, such as poor management and monitoring of diabetes mellitus [3].
Older pharmacological treatments for diabetes, such as oral anti-diabetic medications in insulin therapy, frequently have drawbacks, such as poor efficacy from improper dosage, the first pass effect, short half-life, P-glycoprotein efflux, lack of target specificity, reduced capacity or effects from drug ingestion, and side effects. These factors can significantly impact patient compliance and the overall treatment of the illness. Additionally, the conventional therapeutic approaches, while effective in regulating blood sugar levels, fails to address the complex pathogenesis of diabetes in its complications, thereby highlighting the importance of investigating more therapeutic modalities. Nanotechnology has arisen as a promising solution to the challenges associated with conventional diabetes treatments [4]. The delivery and efficacy of anti-diabetic drugs can be enhanced by using the unique physiochemical properties of nanocarriers. The nano carriers that have been gradually developed and incorporated into nano formulations provide a variety of advantages over traditional formulations, which includes extended drug retention in the stomach, sustained drug release, increased selectivity, suppression of P-glycoprotein efflux, improved capacity, and higher bioavailability. To overcome obstacles like insulin resistance in the treatment of diabetes, the delivery of insulin using nano carriers is especially significant since it provides a more practical, safe, and non-invasive technique of delivering insulin. Furthermore, by shielding the medication from enzymatic and chemical breakdown in the gastrointestinal tract (GIT), encapsulating it in nanomaterials may increase the stability of the drug and boost the overall effectiveness of drug delivery systems. This innovative approach has the potential to transform the delivery of drugs by addressing the major problems associated with drug efficacy and patient compliance thereby, facilitating the development of personalised drugs and targeted therapies. The World Health organisation WHO estimates that over 422 million people globally are affected with diabetes mostly in low middle income countries resulting in around 1.5 million deaths per year directly related to the disease. Diabetes presents a significant public health challenge and requires immediate consideration in all aspects of prevention, management, and treatment strategies. In addition, the 2024 International Diabetes Federation (IDF) Diabetes Atlas emphasises that approximately 10.5% of adult individuals aged 20 to 79 are diagnosed with diabetes, and nearly half of them are unaware of the seriousness of their condition. Additionally, the IDF’s projection suggests that by 2045, an estimated 783 million adults, or approximately one in eight individuals, will have diabetes, reflecting a 46% increase from the current fig. [5, 6]. The current review addresses a variety of nanotechnology-based nanocarriers that serve as pathways for the administration of drugs in the treatment of the DM. Additionally, the importance of developing nanomedicine for antidiabetic agents have been highlighted as a method to address the limits of conventional approaches. Therefore, the integration of these agents within the nanocarrier matrix have been used to develop methods for the effective delivery of therapeutic drugs with the goal of precisely targeting them to improve their efficacy and safety.
All the data and information were collected from Science Direct, Springer Link, Google Scholar, Scopus, PubMed, Web of Science, EMBASE and other databases. Maximum data is collected from year 2014 to 2024. The following keywords were used to gather this data and information: solid lipid nanoparticles, dendrimer-derived nanostructure, polymeric nanoparticles, gold nanoparticles, niosomes, blood glucose, diabetes mellitus, nanocarrier, silver nanoparticles, ethosomes, carbon nanotube, nano-fibreetc.
Types of diabetes
Type 1
It is also known as juvenile-onset diabetes or insulin-dependent diabetes. It makes up between 5 and 10% of all cases of diabetes. This autoimmune condition is defined by the degeneration of pancreatic β-cells that produce insulin, leading to insulin insufficiency and eventually elevated blood sugar levels. Weight loss, frequent urination, Blurry vision, and excessive thirst are some common symptoms of type 1 diabetes. About 4% of diabetic patients also have celiac disease, and 0.5 of patients also have concurrent Addison's disease [7].
Type 2
It is also known as adult-onset diabetes or non-insulin dependent diabetes. It makes up between 90 and 95% of all cases of diabetes blood glucose levels rise because of this condition. Aging, obesity, and physical inactivity are linked to insulin resistance in people with type 2 diabetes [8]. The pancreatic islets grow larger and generate more insulin in order to combat insulin resistance [9]. After ten years of insulin resistance, the malfunction of pancreatic β cells necessitates insulin therapy for more than half of Type 2diabetes patients [10]. In those with type 2 diabetes, chronic long-term insulin resistance has a number of deleterious impacts, including microvascular problems like retinopathy, neuropathy, and nephropathy, and macrovascular problems like atherosclerosis. Increased appetite, skin discoloration, and numbness are some common symptoms of type 2 diabetes [11].
Type 3
Metabolic syndrome that may result in abnormalities associated with progressive brain insulin resistance, which in turn impairs central insulin signalling processes, accumulates neurotoxins, and induces neuronal stress, ultimately leading to a development of neurodegeneration. The brain of patient with Alzheimer’s disease (AD) displayed evidence of lower expression of neuronal insulin receptors and insulin in comparison to those of age-matched controls. This occurrence and progressively results in the complete collapse of the insulin-signalling pathway, which is characterised by insulin resistance. This, in turn, impacts cognitive functions and brain metabolism, which are the most well-documented abnormalities in AD.
Gestational diabetes mellitus (GDM)
GDM is any level of diabetes or glucose intolerance detected during pregnancy, usually in the second or third trimester, or at the start of the pregnancy. During the initial phases of pregnancy, blood sugar levels are usually lower than normal for both fasting and postprandial, but they rise by the third trimester. Nearly 90% of all cases of diabetes and associated pregnancy-related problems are caused by GDM [12]. Hormonal changes that occur during pregnancy are the cause of gestational diabetes. Cells become less sensitive to the effects of insulin when the placenta generates certain hormones [13].
Complications associated with diabetes mellitus
Diabetes is the fundamental root cause of several serious complications, including heart disease, cerebrovascular diseases, kidney failure, inflammation and immune dysfunction, and obesity. Epidemiological studies of diabetes mellitus have shown that gender, age, and cultural background are significant factors indiabetes mellitus development and associated complications. Amadori glucose adducts transform albumin into glycated albumin, which is independently associated with complications related to diabetes. Problems in insulin metabolism and dysfunctions in carbohydrate, lipid, and protein metabolism result in increased blood glucose levels, leading to long-term problems [14]. Various complications associated with diabetes mellitus are shown in fig. 1.
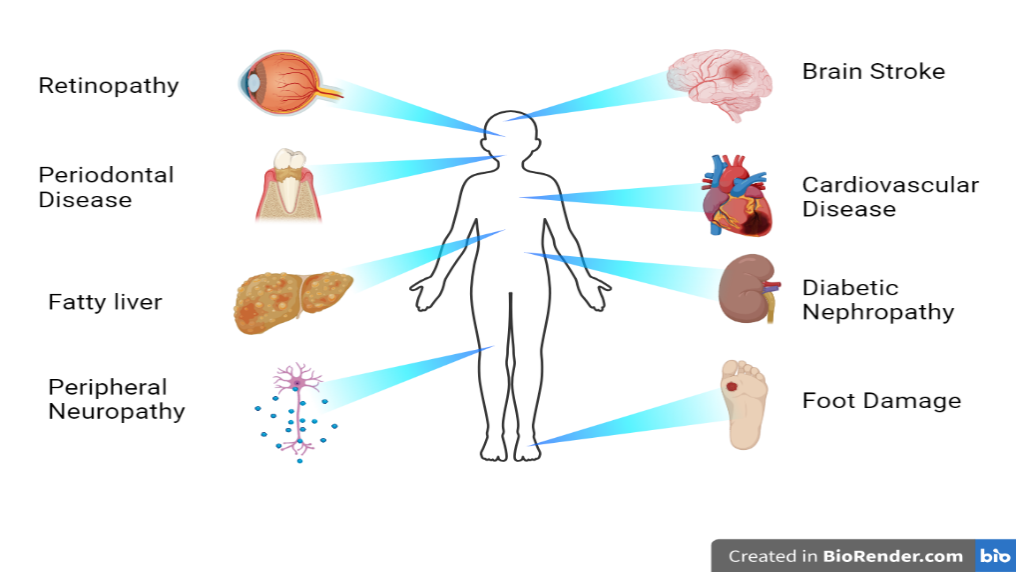
Fig. 1: Major complications of diabetes
Impact on the brain
The hyperglycaemic state may have a direct impact on neuronal cells in both type 1 and type 2 diabetes mellitus. It reduces the oxygen pressure within the cerebral cells, thereby increasing the possibility of a stroke. Furthermore, insulin resistance represents a primary factor that leads to the formation of type 2 diabetes mellitus, as it limits the ability of adipocytes, muscle cells, and hepatic cells to use insulin efficiently. The pathogenesis of Alzheimer's disease (AD) is widely recognised to be primarily influenced by the production and accumulation of amyloid-beta (Aβ). Aβ is a protein that is degraded by insulin, which exhibits an abnormal increase over time within the brain. The insulin degrading enzyme (IDE) is responsible for the degradation of insulin and amylin, as well as the Aβ peptides that are present in excess in the brains of individuals with Alzheimer's disease. The accumulation of Aβ is a defining characteristic of AD within the brain. The chronic condition of diabetes mellitus results in abnormalities in cognitive functions and is associated with anxiety, melancholy, and memory impairment, ultimately leading to a condition referred to as diabetic encephalopathy.
Diabetic retinopathy
The most common cause of blindness in individuals between the ages of 20 and 74 is diabetic retinopathy, which can be identified by a variety of retinal abnormalities. These include vascular permeability alterations, capillary microaneurysms, capillary degeneration, and the overgrowth of new blood vessels, a process known as neovascularisation. The neural retina shows dysfunction that involves the death of some cells, which ultimately changes retinal electrophysiology and leads to insufficient ability for colour discrimination [15]. Clinically, diabetic retinopathy is classified into two different stages: non-proliferative and proliferative disease. In the initial phases, hyperglycemia can result in the death of intramural pericytes and basement membrane thickening, both of which contribute to modifications in the structural integrity of retinal blood vessels. Vascular permeability and the blood-retinal barrier are eventually affected by these alterations. During the preliminary phase of non-proliferative diabetic retinopathy (NPDR), most individuals do not perceive any visual abnormalities [16].
Cardiovascular diseases
Both "type 1 diabetes mellitus" and "type 2 diabetes mellitus" contribute to the development of various cardiovascular diseases, including heart failure, arterial disease, cardiomyopathy, congenital heart defects, and coronary heart disease (CHD). People with type 2 diabetes are two to four times more likely to develop cardiovascular disorders, according to reports, and the frequency of diabetic heart disease has dramatically increased in recent years [17]. It is regarded as one of the primary contributors to the mortality rate among diabetic populations [18]. Low-density lipoprotein (LDL), very-low-density lipoprotein (VLDL), and excessive triglyceride levels are among the factors that lead to the development of cardiovascular illnesses. Previous studies have indicated that, in addition to triglycerides (TG), various other factors contribute to the increase in cardiovascular diseases (CVDs). Notably, 55% of the diabetic population also experiences heart disease due to alternative cause. It has been reported that diabetes induces modifications in the structure and function of the myocardium, specifically contributing to ischaemic heart disease and hypertension. The modification defines diabetic cardiomyopathy through its impact on cardiac energy metabolism, the diastolic dysfunction and cardiac remodeling. Other metabolic dysfunctions, including dyslipidaemia, elevated levels of free fatty acids, increased hepatic glucose production, and insulin resistance, contribute to the development of diabetic cardiomyopathy. Chronic hyperglycemia also alters cardiac cells by increasing glucose influx through polyol pathways, raising AGE levels, and activating protein kinase C (PKC) enzymes, all of which leads to cellular damage. These metabolic complications lead to the production of reactive oxygen species (ROS), which in turn reduces the activity of antioxidant enzymes, including glutathione reductase (GR), and contributes to the formation of AGEs. A reduction in the activity of antioxidative enzymatic systems results in increased oxidative stress, which subsequently induces damage to DNA and ultimately leads to the death of cardiomyocytes.
Renal disease (Nephropathy)
Kidney failure represents one of the most prevalent and severe complications experienced by individuals with diabetes. It has been reported that 40% of individuals diagnosed with diabetes experience renal failure. In patient with diabetes, the progressive deterioration of renal function is identified by an increase in proteinuria, which in turn, result from a low glomerular filtration barrier rate, leading to elevated urinary albumin excretion. Chronic hyperglycemia is well known to behave as a pro-oxidative agent, promoting the excessive production of reactive oxygen species (ROS) via the electron transport chain in the mitochondria [19]. Degradation of cell membranes and eventual organ damage or failure are caused by elevated ROS levels. Hyperglycemia may be directly or indirectly responsible for the elevated production of free radicals in the intracellular fluid, according to experimental and clinical findings. Both oxidative stress and free radicals are the primary root causes contributing to hepatorenal tissue injury. Abnormal renal function is characterised by increased concentrations of plasma creatinine, urea, and uric acid. Diabetic neuropathy (DN) arises from the complex interplay of various yet interconnected pathways induced by high glucose levels influenced by critical factors such as oxidative stress and AGEs. These factors activate various abnormal signalling pathways that includes inflammation, cellular proliferation, and the expansion of the interstitial matrix [20]. Hyperglycemia may either directly or indirectly contribute to the excessive production of free radicals in the intracellular fluid, according to experimental and clinical findings. The main causative agents of hepatorenal tissue damage are oxidative stress and free radicals. Abnormal renal function is characterised by increased concentration of plasma creatinine, urea, and uric acid. Previous investigations have indicated that the advancement of nephropathy may be regulated through the management of hyperglycemia [21].
Diabetic neuropathy
It is one of the most common complications associated with diabetes. It is characterised by the death of nerves. Neuropathy affects more than half of diabetic patients, according to previous researchers. Diabetic neuropathy is the primary risk factor for wound healing impairment, which is common in diabetic patients with type 2 diabetes. According to Obrosova et al., advanced diabetic neuropathy, which is caused by the degradation of the nerve fibre, results in a complete reduction in sensory perception among the affected individuals. Erectile dysfunction, cardiovascular dysfunction, paraesthesia, hyperalgesia, and allodynia are some of the other issues that are associated with diabetic neuropathy [22]. Clinically, neuropathy is typically defined by the development of vesicular abnormalities, such as endothelial hyperplasia and thickening of the capillary basement membrane, which ultimately result in a decrease in oxygen tension and the beginning of hypoxia. Clinically, renin-angiotensin system inhibitors and 1-antagonists increase nerve conduction velocities, which are thought to be caused by increased blood flow to the neurons. Advanced neuropathy resulting from the degeneration of nerve fibre in diabetes is characterised by a modified sensitivities to vibratory stimuli and thermal thresholds, ultimately leading to a complete loss of sensory perception. Hyperalgesia, paraesthesia, and allodynia are also observed in a subset of patients, with pain occurring in 40-50% of individuals diagnosed with diabetic neuropathy [23]. Pain is also observed in certain individuals with diabetes who do not exhibit clinical signs of neuropathy (10-20%) which may significantly affect their quality of life.
Oral antidiabetics: currently available therapy
Various classes of antidiabetic drugs are being used to control diabetes mellitus are given in fig. 2.
Biguanides
It works by increasing the body’s sensitivity to natural insulin, decreasing the amount of glucose produced by the liver, and decreasing intestinal glucose absorption [24]. It additionally boosts peripheral cell absorption of glucose [25]. Metformin (MET) is the best choice under this category. Hepatic glucose synthesis is reduced by metformin and it raises insulin sensitivity. This medication helps to control blood LDL cholesterol and triglycerides, both of which exhibit a discernible decline [26]. A typical treatment for type 2 diabetes is biguanide therapy [27]. Large dosages, frequent dosage administration requirements, poor intestinal absorption, and MET's short half-life have all considerably diminished its therapeutic efficacy. Numerous nano-drug delivery system (DDS) has been logically created for effectivedelivery of METto get over these problems [28]. By activating AMP-dependent protein kinase, biguanides prevent the degradation of fatty acids [29]. This treatment's side effects include allergic reactions, lactic acidosis, stomach discomfort, and discomfort in the chest [30].
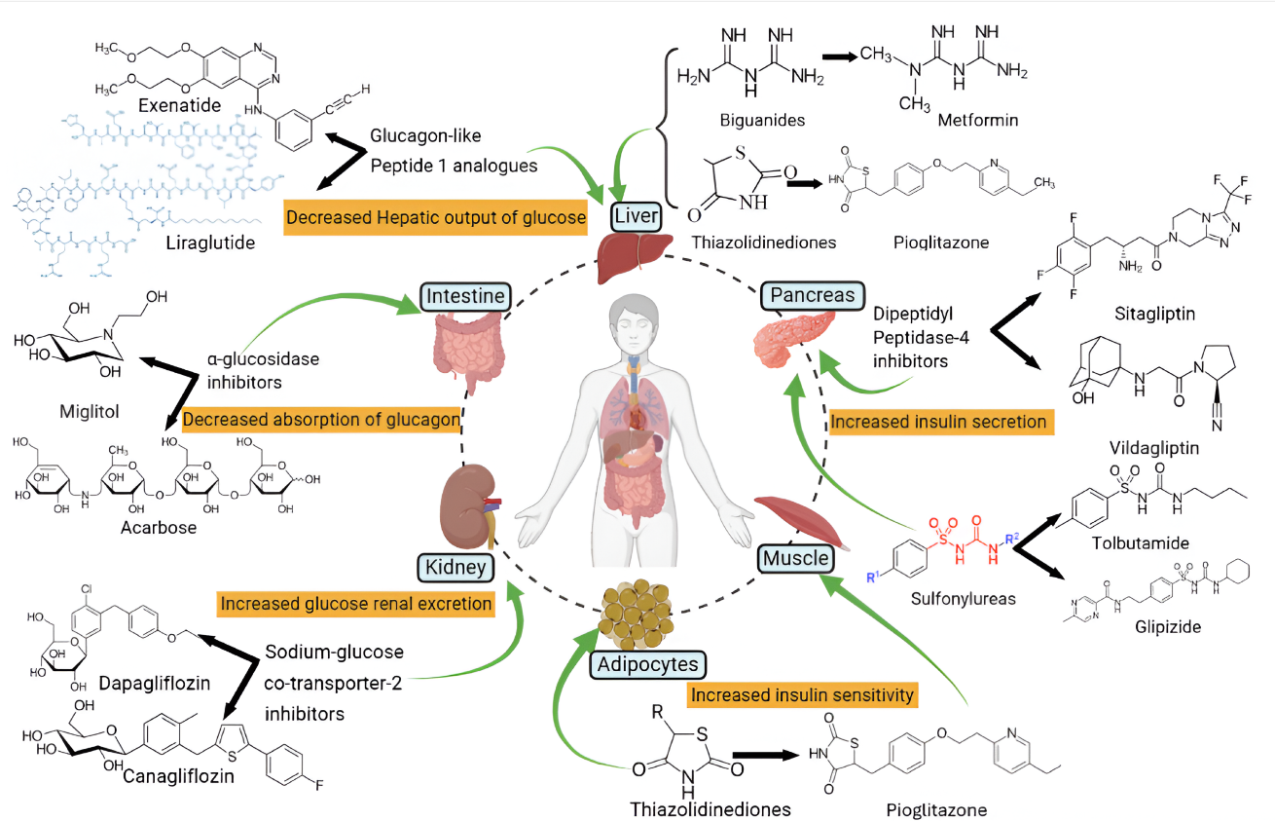
Fig. 2: Visual representation of several kinds of antidiabetic drugs and their mechanisms of action
Alpha-glucosidase inhibitors (AGIs)
These medications aid in the decrease of postprandial hyperglycemia. Acarbose was the first AGI and was obtained from Actinomyces utahensisand utilized as an enzyme alpha glucosidase's competitive moiety [31]. Miglitol and voglibose are two other AGIs used to treat type 2 diabetes [32]. Patients with inflammatory bowel disorders including Crohn's disease or ulcerative colitis, as well as those with diabetic ketoacidosis, a condition in which the body burns fat for energy instead of carbohydrates, should not use alpha-glucosidase inhibitors [33]. In individuals with liver cirrhosis, large intestinal ulcers, and pregnant women, acarbose is not advised [34].
Sulfonylureas
The sulfonylurea class of antidiabetic medications stimulates pancreatic β-cellsnatural production of insulin. In addition, sulfonylureas decrease hepatic insulin clearance, increase insulin sensitivity in peripheral tissues, and block glucagon secretion [35]. First-generationmetahexamide, tolbutamide, tolazamide, and second-generation Glimepiride, Glipizide, Glibenclamide, Glyburide, and Gliclazide are prominent instances of this type [36]. Sulfonylurea can speed up the operation of β-cells and does not have any known protective effects on β-cell action [37]. Following this, there is a first decline in blood glucose level (BGL) and a rise in HbA1c levels. While HbA1c levels drop by 1% to 2%, BGL concentrations drop by 20%. Gaining weight is an unwanted sign [38]. Its adverse effects consist of Water retention, hyponatremia, and hypoglycaemia. Drugs such as aspirin, fibrates, sulphonamides, and allopurinol are used to lessen the effects of sulfonylureas and prevent hypoglycaemia [39].
Meglitinides
These medications raise the pancreatic production of insulin; because they are glucose-dependent, they lower the risk of hypoglycaemia. Since, it acts quickly it can be given to coincide with the increase in blood sugar following a meal.
Thiazolidinediones (TZDs)
It simultaneously increases the body's sensitivity to insulin and lowers insulin resistance. The function of thiazolidinediones (TZDs), which are peroxisome proliferator-enacted receptor γ (PPR-γ) activators, is to improve insulin’s impact on heart tissueand liver adipocytes [40]. For type 2 diabetes mellitus (T2DM) patients who are insulin-resistant, these are used as a therapy regimen [41]. Increased body weight is a common adverse effect of TZD [42].
Amylin analogs
A single chain of 37 amino acids makes up the hormone amylin. It alters the brain's appetite centre to control how much food is eaten [43]. Because it delays the emptying of the stomach and prevents the release of glucagon, It keeps the blood's postprandial glucose level stable as well as the fasting state. The only drug in this class that is currently on the market is pramlintide acetate, which is sold under the Symlin® brand and is administered subcutaneously [44, 45].
Dipeptidyl peptidase-4 inhibitors (DPP4)
Gliptins, or dipeptidyl peptidase-4 (DPP4) inhibitors, are modern therapeutic medicines that work by inhibiting the DPP4 enzyme. Alogliptin, Sitagliptin, Linagliptin, Saxagliptin, Vildagliptinand are common examples in this category. Lipid levels after meals are influenced by DPP-4 inhibitors. Heart function and coronary artery perfusion have been shown to improve with sitagliptin medication. However, using metformin along with sitagliptin or sitagliptin alone results in acute pancreatitis.
Glucagon-like peptide 1 Analogue (GLP-1)
GLP-1 analogs are incretin-based therapies that decrease glucagon secretion, lower hepatic glucose generation, and increase insulin secretion in a glucose-dependent manner [46]. Reductions in blood pressure, improved lipid profiles, and delayed gastric empty time were all results of the correction of endothelial dysfunctions [47]. In this category, levaglitide and exenatide are common examples.
Sodium-glucose Co-transporter-2 inhibitors (SGLT 2)
Known by another name, gliflozins, SGLT2 inhibitors decrease the uptake of sodium, which lowers the amount of glucose that is taken up by the kidneys through the proximal tubules of the renal nephron [48]. Canagliflozin, Empagliflozin, Ipragliflozin, and Dapagliflozin are members of this class. Due to glucosuria, these medications can increase β-cell capacities, improve insulin affectability, and increase glucotoxicity. Symptoms related to volume consumption, vaginal mycotic infections, especially in women, and urinary tract infections are the noted side effects [49].
Emerging treatment strategies
Oral hypoglycaemics using nanocarrier-based therapies
Excellent uses of nanotechnology can be found in the creation of DDS. The therapeutic DDS based on nanocarriers has received more attention than conventional DDS because of their promising applications. The ratio of surface area to volume is larger in nanocarriers, which allows an increased amount of the drug surface to be in contact with the body at the same concentration. Therefore, the DDS can be more effective with the same dosage, and a lower dosage can lessen the adverse effects of medications. Nanocarriers also offer external functions for a range of therapeutic medications and targeted techniques [50]. Because of the drawbacks of pharmaceutical therapy and the benefits of NPs in drug transport and imaging, researchers are actively working with nanocarriers to treat and manage diabetes mellitus. Liposomes, inorganic NPs, and polymer-based NPs are the major nano-based drug delivery technologies in diabetes therapy [51]. Various nanocarriers used for delivering antidiabetic drugs are shown in fig. 3, and the key properties with the advantages and limitations of these nanocarriers are highlighted in table 1 which are recognised as effective drug carriers. These nanocarriers may provide benefits in several respects, including enhancing bioavailability, and protecting medications from enzymatic breakdown. They may function as an adaptive automated system that mimics endogenous insulin production and exhibit a non-linear responseto environmental stimuli, therefore reducing the risk of hypoglycaemia and enhancing patient adherence. Moreover, they showed remarkable efficacy in delivering drug delivery to specific areas and in controlling and sustaining drug release at targeted locations for an extended duration, thus minimising unwanted effects while maximising therapeutic efficacy.
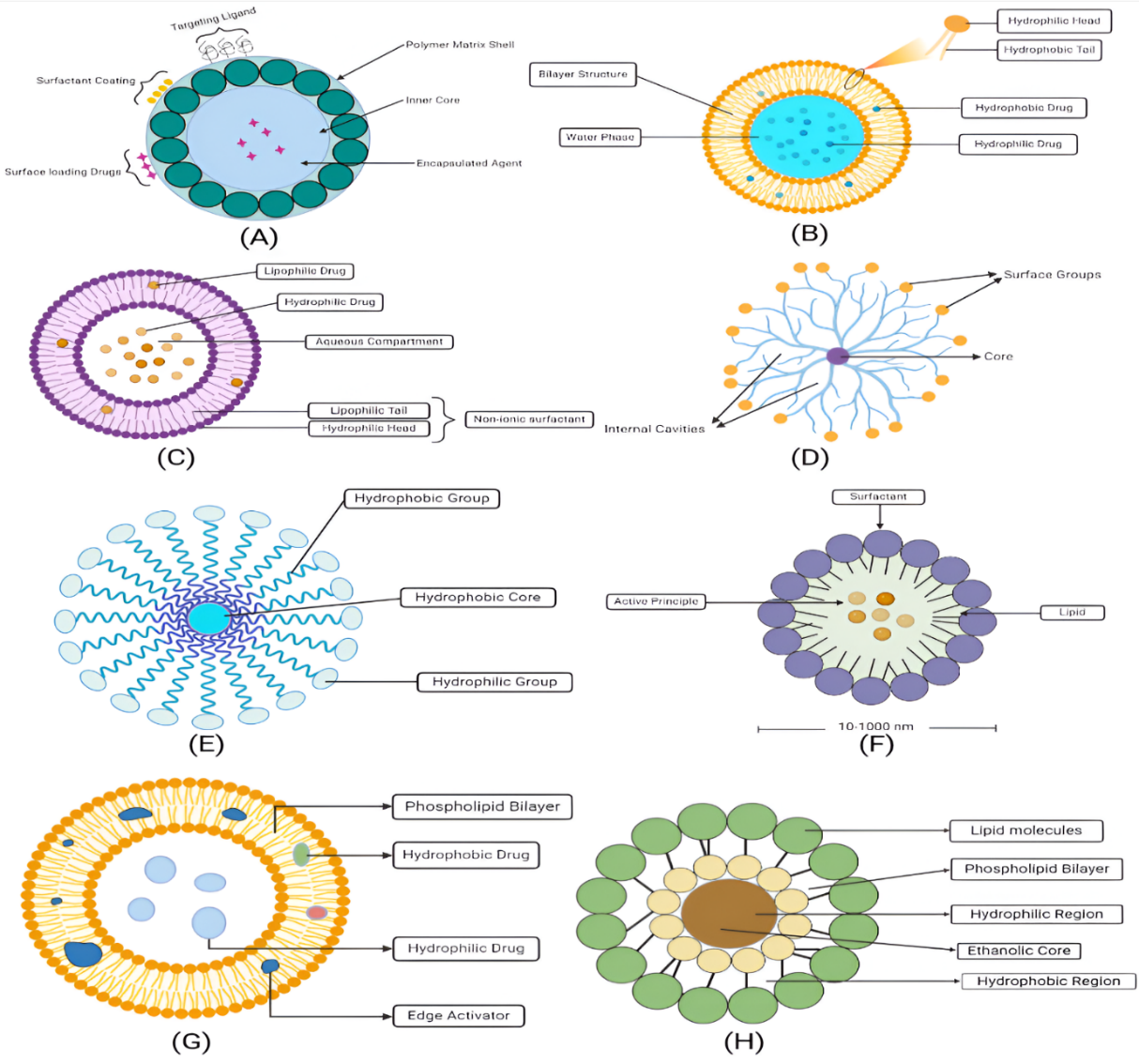
Fig. 3: Schematic representation of various nanocarriers used for delivering oral hypoglycaemic agents (a) polymeric NPs (b) liposomes (c) niosomes (d) dendrimers (e) micelles (f) solid-lipid NPs (g) transfersome (h) ethosomes
Polymeric nanoparticles (PNPs)
PNPs are colloidal drug delivery systems with a nanoscale particle size that vary from 10-1000 nanometres in diameter. These systems are extremely suitable for the delivery of a wide range of drugs due to their diverse size and physiochemical characteristics. PNPs are highly potential vehicles due to their notable in vivo and storage stability, as well as their ability to sustain drug release. Natural polymers such as chitosan (polysaccharides), human serum albumin, and sodium alginate. Through receptor-mediated endocytosis, chitosan, a cationic polymer, can enhance the cellular absorption of NPs when treated with certain ligands. Both natural and manmade polymeric materials can control the release of insulin and the pharmacological consequences that follow [52]. Insulin is transported across the intestinal mucosa more easily when intestinal epithelial cells ingest insulin-loaded nanoparticles, which are made of biodegradable polymers such polylactide-co-glycolide (PLGA), polyanhydride, poly alkyl cyanoacrylate (PACA), and PLA. Scientists have created insulin-loaded nanoparticles that the gastrointestinal system can absorb efficiently, smart, self-regulating delivery system made by advanced polymeric NPs systems that can include sensors to track blood glucose levels and release insulin appropriately. These nanoparticles are composed of a biodegradable poly-ε-caprolactone (PCL) and a non-biodegradable acrylic polymer, Eudragit RS100. PLGA and PLA are approved by the USFDA for pharmaceutical applications due to their non-immunogenic, biodegradable, biocompatible, and benign characteristics. They have shown effective uptake involving insulin, Tf, and LDL receptors [53]. The application of PNPs in diabetes management is given below.
Hadiya et al., formulated insulin-loaded NPs composed of diverse polymers with varying compositions and assessed their blood glucose-lowering efficacy both orally and subcutaneously in rats with diabetes. The NPs were well-tolerated after oral treatment in rats, as shown by the assessment of creatinine, alanine aminotransferase, albumin, aspartate aminotransferase, and urea levels. This work shown that the characteristics and delivery efficacy of nanomaterials may be regulated using various natural and synthetic polymers, as well as by precisely adjusting the ratios of these polymers [54]. Abbas et al., conducted research to improve the bioavailability of linagliptin (Lina) and create a formulation for injectable sustained-release NPs that lowers the frequency of dosage to encourage compliance of patients. In accordance with a design of experiment (DoE), all formulations were created utilizing the single emulsion solvent evaporation method with PLGA as the polymer. The chosen formula had 10% of organic solvent, 150 mg of PLGA, and a 65:35 lactide ratio, while TEM analysis revealed a spherical structure with a smooth surface. The particle measured 541.178±10.4 nm in size, with a sustained release period of seven daysand an entrapment efficacy of 67.134%. The pharmacokinetic analysis indicated a significant increase in area under curve AUC 0-∞ from 33.73 to 60.53 ng/ml. h, a significant increase of t1/2 (231.6±43.9 vs. 11.8±0.3 h), a mean residence time (MRT) of 344.1±61 vs. 3.9±1.5 h, and a Vd of 4731±393.7 vs. 161.4±87.20 ng/ml for Lina NPs and oral Lina solutions, respectively (P>0.05). The optimized formulation's pharmacodynamics study demonstrates a notable drop in levels of blood glucosecompared to oral Lina. The results indicate that PLGA Lina NPs provide an innovative approach for developing an injection administered once a week to regulate blood glucose in persons capable of managing type 2 diabetes mellitus [55]. Lari et al., developed innovative cross-linked carboxymethyl chitosan (CMCS NPs) incorporating metformin (MET) by the microfluidics (MF) approach and assessed their efficacy in diabetic treatment. Researchers exhibited a high encapsulation efficiency of 90% and facilitated drug release using CaCl2-mediated crosslinking. In diabetic rats, in vivo test revealed that MFMET-loaded CMCS nanoparticles reduced blood glucose level by 43.58% and raised body weight by 7.94% as compared to the free medication. Additionally, histological findings indicated that the pancreatic islets have an area of 2.32 m² with a cellular density of 64 cells per islet in diabetic rats administered the MF-based sample. The statistics were comparable to those obtained from the healthy rats [56]. Sharma et al., developed Pioglitazone encapsulated polymericNPs using solvent evaporation technique. This research optimised three process parameters: drug-polymer ratio (A) stirring duration (B) stirring speed (C) using a three-factor, three-level Central Composite design. With A, B, and C levels set at 1:2, 3000 rpm, and 20 min, respectively, the optimization model shows an entrapment efficiency (EE) of about 61.7%, a drug content of 12.33%, and a particle size of323 nm. The treatment of streptozotocin-induced diabetic rats with pioglitazone-loaded s markedly reduced BGL to normal levels for a maximum of six hours in contrast to the group that received native medication, during a duration of 7 days. The in vivo toxicity study of NPs in albino rats did not reveal any significant changes in haematological, biochemical, or behavioural assessments. Since Pioglitazone is used for the management of T2DM, the developed approach may provide a controlled release of the drug, potentially reducing dose frequency and enhancing patient adherence [57]. Ribeiro et al., formulated insulin-embedded chitosan (CNPs) and assessed their therapeutic effectiveness in wound healing among diabetic rats. The study postulated that the signaling pathway involved in wound healing might be activated by the addition of insulin to CNPs. The insulin alliance's productivity was 97.19%. NPs and free insulin (FI) were incorporated into a hydrogel (Sepigel®) for topical administration to the wounds of 72 diabetic rats, which were divided into four groups: empty CNPs, CNPs carrying insulin (ICNPs), F1, and Sepigel® (S, control). Three subgroups of six animals each were created to evaluate the clinical signs of the animals on days three, seven, and fourteenafter the initiation of drug. Severe fibroplasia was detected in the free or ICNP types. On the seventh day, a significant amount of blood vessels was seen in the latter. The results suggested that ICNPs and ECNPs may promote wound maturation, angiogenesis, and the proliferation of inflammatory cells [58]. EI-Dakroury et al., performed Glipizide (GPZ) encapsulated O-Carboxymethyl chitosan (O-CMC) NPs to achieve an extended antidiabetic effect and assess their impact on several T2D-related biomarkers. Optimised GPZ-O-CMC-NPs demonstrated an EE of 80.7±0.8%, a zeta potential of −14.2±2.1 mV, and a particle size of 216±2.5 nm. GPZ-O-CMC-NPs exhibited a better and extended-release profile compared to both marketed and pure GPZ. In comparison to marketed or pure GPZ, treatment with GPZ-O-CMC-NPs had a more noticeable (P<0.05) impact on blood glucose, lipid profile (three or four time), insulin, oxidative stress markers (two or three times), and inflammatory cytokines (2.5–3.5 times). These data support the possible benefits of GPZ-O-CMC-NPs in the treatment of T2D [59].
Liposomes
These structural configurations allow hydrophilic and lipophilic medications to bind to the vesicle's surface or entrap within its lipid bilayer section, which is linked to hydrophobicity, and aqueous core, enabling targeted drug administration. Liposomes fuse with cellular lipid membranes during the lipid material delivery process, which results in the release of their entire contents into the cytoplasm of the cell, where they perform their pharmacological functions. These small carriers have attracted significant interest for the purpose of encapsulating drugs that are classified as proteins or peptides. Liposomes prolong the useful life or sensitive antidiabetic medications in the body by shielding them from enzymatic or chemical breakdown, such as GLP-1 agonists or RNA-based therapies. To enhance the efficiency of insulin entrapment within the liposomal core and enhance the particle size, it is necessary to consider several factors. It is essential to maintain membrane fluidity, inhibit insulin leakage from the liposomal core, and allow the optimum number of insulin molecules to be incorporated by maintaining an appropriate phospholipid-to-cholesterol ratio [60]. The permeability and cellular absorption of antidiabetic drugs may be significantly improved using liposomal delivery methods, which also provide tailored release and enhance the effectiveness of therapeutic interventions. Non-liposomal carriers, on the other hand, often have difficulty traversing lipid bilayers, which reduce the efficiency with which they transport drugs to the target cell. Some studies on liposomes for diabetes mellitus is given below.
Amjadi et al., fabricated betanin loaded liposomes using thin layer hydration method for the treatment of diabetes, resulted in particle size of 36 nm with zeta potential of-19mV and drug loading of 26%. Drug loaded liposomes reduces the blood sugar level, and increased the serum insulin levels in diabetic rats. Histopathological study revealed that the tissue damage inpancreas, liver, and kidney caused by hypoglycaemia and oxidant stress was also reduced [61]. Ding et al., prepared dihydromyricetin (DHM) and chitosan loaded liposomes by thin film dispersion method to cure mice with diabetes mellitus that have liver damage. The drug concentration time curve in pharmacokinetic analysis increased 3.23 and 1.92 times (953.60±122.55 ng/ml/h) for the chitosan modified dihydromyricetin loaded liposomes (CDL) treated group compared to the DHM treated group (295.15±25.53 ng/ml/h) and the dihydromyricetin loaded liposomes (DL) treated group (495.31±65.21 ng/ml/h). In comparison to the DHM and DL groups, the CDL group's maximum drug concentration in blood (T max) increased 2.26 and 1.21 times, respectively. The maximal drug concentration in blood increased 1.49 and 1.31 times in the CDL group (C max) relative to the DHM and DL groups, respectively [62]. Hu et al., formulated Hyodeoxycholic acid (HDCA) modified MET liposomes for type 2 diabetes. HDCA, possessing a structure resembling cholesterol, which can lower blood glucose levels and control homeostasis of glucose. HDCA as an anti-diabetic active compound, alters liposomes to reduce the limitations of metformin and increases the hypoglycaemic effect. Using the thin-film dispersion technique, three varieties of liposomes with varying ratios of HDCA and ME were synthesised i. e., (0.5:1), (1:1). (2:1). In vivo research shown that liposomes could decrease fasting blood glucose levels, modulate oxidative stress indicators, enhance glucose tolerance, and safeguard liver tissue in type 2 diabetic mice. Histopathological studies revealed that liposomes reduced hepatic inflammation in mice and had a protective impact on the liver. Moreover, the anti-diabetic efficacy of the HDCA: ME (1:1) Liposome group was significantly greater to that of the metformin group, so confirming the synergistic effect of HDCA in the treatment of type 2 diabetic mice. The findings suggested that HDCA might serve as a viable liposome excipient for the management of type 2 diabetes, hence offering a novel pathway for its clinical use [63]. Zhang et al., Enhances the prolonged release and antidiabetic effectiveness of polydatin by developing long-circulating liposomes (PLLs) by a membrane dispersion method. According to in vivo release experiments utilizing the dialysis bag method in different buffers, the PLL encouraged a sustained release of polydatin. Furthermore, the PLL showed improved cellular absorption, extended in vivo circulation time, and bioavailability in both in vitro cellular uptake and in vivo pharmacokinetic experiments. When a high-fat diet was used to induce T2DM in mice, it was found that PLL may decrease blood glucose levels, enhance weight loss, mitigate oxidative stress, reduce blood lipids, and reduce damage to liver, spleen, and pancreatic tissues [64]. Sarhadi et al., formulated PEGylated liposomal insulin with B12 modification to enhance absorption and stability of insulin in the gastrointestinal tract. Liposomes were synthesised using the film method combined with extrusion, conjugated with B12, and characterised for zeta potential, particle size, and EE%. Simulated intestinal fluid (SIF) and simulated gastric fluid (SGF) release profiles were evaluated. The results showed that in simulated intestinal and stomach fluid, B12-targeted and PEGylated liposomes were more stable than non-functionalized PEGylated liposomes. In vitro findings demonstrated a markedly increased cellular absorption of B12-targeted PEGylated liposomes in Caco-2 cells relative to non-targeted liposomes. During this period, they exhibited no toxicity on Caco-2 cells. Accumulation of insulin in the gut and liver of BALB/c mice was enhanced by B12-targeted PEGylated liposomes. In diabetic rats, B12-targeted PEGylated liposomes exhibited superior bioavailability of insulin relative to other formulations [65].
Niosomes
Niosomes are a type of molecular cluster that were formed by the self-amalgamation of non-ionic surfactant in water-based environments. The exterior surface of this structure is characterised by a polar component, which is supported by a non-polar region internally. These amphiphilic compounds, which are referred to as surfactants, are capable of self-assembling and forming a variety of structures, such as micelles or flat lamellar bilayers, due to the presence of both hydrophobic (tail) and hydrophilic (head) segments. Sorbitan esters and their derivatives, as well as surfactants derived from sugars, polyoxyethylene, polyglycerol, or crown ethers, are surfactant that are potentially appropriate for use in niosomal drug delivery. In some cases, membrane additives such as cholesterol, or its analogues are additionally incorporated into the system. Non-ionic surfactants are the preferable option because of their limited ability to cause irritation. Niosomes are vesicular structures with special topologies that allow them to contain both hydrophilic and lipophilic molecules. While hydrophilic medications are usually contained within the internal water core, lipophilic compounds are maintained by being dispersed into the lipophilic portion of the lipid bilayer structure. Due to their capacity to transport a variety of therapeutic substances, these vesicles have been frequently used as a drug delivery vehicle to facilitate drug targeting, enhanced permeation, and regulated release. In fact, niosomes have the capacity to function as therapeutic reservoirs, permitting the controlled distribution of the drugs to enhance their bioavailability and produce long-lasting therapeutic effects. Encapsulated drugs, such as insulin, or GLP-1 agonist, are protected by niosomes against chemical and enzymatic breakdown, especially in the bloodstream or gastrointestinal tract. It provides a non-invasive substitute for injection by preventing insulin from being broken down in the stomach and promoting absorption in the intestines. Numerous studies have been conducted to evaluate niosomes' potential for aiding drug delivery, particularly for the treatment of diabetes. These studies are included below.
Samed et al. developed a niosomal formulation for encapsulating and releasing hydrophilic and hydrophobic antidiabetic drugs simultaneously. MET hydrochloride and GPZ were found to have EE of 58.72% and 67.64%, respectively. The drug release assays conducted in buffers at several pH levels (simulating blood plasma, cellular endosomal, and stomach conditions) demonstrated that the release of drugs showed a linear profile for eight to ten hours, thereafter slowed, and continued for 12–14 h. This formulation presents a novel drug delivery system for the combinatorial sustained release of antidiabetic drugs [66]. MET loaded niosomes exhibited prolonged hypoglycaemic effect for 6–8 h, when compared to the MET solution, which only reduced BGL for 2–4 h. The continuous drug release may be linked to the hydrophobic phospholipid barriers of niosomes. Prolonged drug release is caused by the interaction of the negatively charged mucosal layer with positively charged niosomes (1, 2-dioleoyl-3-trimethylammonium-propane chloride salt). The MET solution displayed a maximum fall in BGL of about 25.21% with a T max of 1 hour, whereasniosomes loaded with metformin revealed a reduction in BGL of approximately 45.89% with a T max of 4 h [67]. Plumbagin is trapped inside niosomes utilizing the quality by design (QbD) approach to ensure the efficient penetration and increased bioavailability. The α-amylase in vitro antidiabetic activity was analysed, and plumbagin-loaded niosomes (90.69±2.9%) exhibited better results in comparison to plumbagin (83.64±3.5%). similarly, the α-glucosidase assay was conducted, and the plumbagin-loaded niosomes exhibited an inhibition percentage of 88.43±0.89% while plumbagin exhibited an inhibition percentage of 81.07±1.2%. This suggests that the developed formulation has the potential to effectively treat and manage diabetes [68]. Mohsen and collaborators inserted glimepiride into niosomes composed of cholesterol sorbitan monostearate and Span 60, to enhance the drug's therapeutic effectiveness. Research conducted in vivo shown a sevenfold increase in bioavailability compared to the saline solution. The developed niosomal formulation has comparable bioavailability to Amaryl® (commercial medication) and remains in the system for a longer period. Niosomes showed continuous drug release for up to 48 h, reaching their Tmax at 6 h. On the other hand, Amaryl® and unbound drugs showed a rapid reduction in blood glucose levels within 2 h, coupled by a significant fall in plasma concentration. 10 % of the original concentration decreased after 24 h [69]. Singhal et al. formulated and assessedniosomes loaded with gymnemic acid (GA) for the treatment of streptozotocin-nicotinamide (STZ-NA)-induced diabetic nephropathy (DN) in wistar rats. Animals was given a formulation including GA-loaded niosomes, which had significantly reduced levels of antioxidants and lipids. Moreover, GA-loaded niosomes markedly reduced cytokines that promote inflammation, such as fibronectin, interleukin (IL-6), and tumor necrosis factor. The research determined the benefits of GA-loaded niosomes and described the efficacy of the formulated preparation in modulating lipid profiles, serum antioxidants, and diabetic sequelae in animals with experimentally induced DN by suppressing oxidative stress and advanced glycation end products [70]. Alam et al., synthesised embelinas niosomes, provides enhanced benefits in nano-formulations, and may be further used for therapeutic applications. The STZ induced diabetic Wistar rats were evaluated for antidiabetic efficacy. An antioxidant test (GSH) was used to measure glutathione, thiobarbituric acid reactive substances (TBARS), catalase (CAT), and superoxide dismutase (SOD). Like repaglinide, the optimized formulation had a notable hypoglycaemic effect. Furthermore, the formulation’s antioxidant efficacy was confirmed by notable increases in GSH, CAT, and SOD accompanied by a decrease in lipid peroxidation. Therefore, thenoisome formulation loaded with embelin successfully controlled the diabetes in wistar rats [71].
Dendrimers
Vogtle et al. initially conceptualised and developed the dendritic framework in 1978. Initially, these structures were referred to as "cascade molecules." Dendrimers are a unique category of artificial macromolecules that are formed by incrementally adding branches around a central multifunctional nucleus. A new "generation" is generated by each stratum of branch nodes. In large part, the final functions of dendrimers determine their characteristics. They are frequently identified as a group of soft nanoscopic compounds that are three-dimensional and possess a unique monodispersed and uniform molecular arrangement. These dendrimers, which are classified as the most modern class of polymers (starburst polymers), differentiate themselves from traditional oligomers or polymers by their high density of functional ends, extensive branching, and symmetry. The production of a wide variety of dendrimers has been influenced by the intricacy, generations, and constituent materials used. These dendrimers, such as polyamidoamine (PAMAM), polypropylene-imine (PPI), and polylysine, are particularly advantageous for the delivery of both hydrophobic and hydrophilic therapeutic agents. The versatility of dendrimers for a wide range of applications, including as drug delivery, gene delivery, antioxidant administration, peptide delivery, smart drug delivery, and biomedical imaging, has been demonstrated in numerous research. They are a promising alternative for the treatment of both type 1 and type 2 diabetes because of their capacity to deliver controlled, sustained release and minimize negative effects. Because dendrimers shield insulin from gastrointestinal tract breakdown, they can help with oral administration. Likewise, formulations based on dendrimers can be applied transdermally, offering an alternative to insulin injections that do not require a needle. Multiple therapeutic agents, including insulin and immunomodulatory drugs, can be co-delivered using dendrimers to treat type 1 diabetes's autoimmune response and hyperglycemia [72]. The application of dendrimers as a nanocarrier system in diabetes mellitus is given below.
Labieniec-Watala et al. gave PAMAM G4 dendrimers to diabetic mice via three distinct routes (subcutaneously, intragastrically, or intraperitoneally), and evaluated their hypoglycaemic effects. Both intraperitoneal and subcutaneous methods are most successful in lowering the persistent symptoms of hyperglycemia, whereas intraperitoneal injection has higher blood glucose scavenging effects. But because of the toxicity of the carriers, the intraperitoneal injection was linked to lower survival rates [73]. Zhang Et al. synthesised the Astragalosidematrix metalloproteinases (MMP 2) responsive nanocarriers, HA-pep-PAMAM, by a combination of PAMAM dendrimer conjugated with the polysaccharide hyaluronic acid (HA) via the substrate Polypeptide (Gly-PLGLAG-Cys) of matrix metalloproteinase-2 (MMP-2). Hydrogen peroxide has a dose-dependent influence on the growth of BJ and HaCaT cells, with the HA-pep-PAMAM-ASI treatment offering the maximum antioxidant capacity after MMP-2 pretreatment. Due to the considerable increase in GSH levels caused by HA-pep-PAMAM-ASI, reactive oxygen species (ROS) were reduced and antioxidant effects were produced. The MMP-2-pretreated HA-pep-PAMAM-ASI group exhibited increased cell proliferation and migratory capabilities. The HA-pepPAMAM-ASI group showed a significant in vivo therapeutic benefit, with considerably higher expression of all wound-repair-related genes compared to the ASI population. The results suggest that enzyme-responsive MMP-2-loaded PAMAM dendrimers might enhance diabetic wound healing and serve as a suitable biomaterial for diabetic treatment [72]. For diabetes patients at risk of limb amputation, developing an effective strategy to improve the wound healing process is essential. Multiple growth factors have been proposed as therapeutic alternatives; however, further research is required to validate their healing efficacy. Kwon et al. discovered a nonviral gene therapy method to enhance wound healing. Cells in the wound tissue that were actively proliferating were effectively transfected, leading to robust VEGF expression. Histological staining revealed that skin lesions in diabetic mice healed and exhibited a well-organised dermal structure after 6 days post-injection. This rapid and effective gene therapy method may serve as a significant technique for addressing diabetic foot ulcers [74]. Akhtar et al. examined whether preventing the epidermal growth factor receptor (EGFR)-ERK1/2-Rho kinase (ROCK) pathway through the continuous administration of nanosized PAMAM dendrimers could lessen vascular dysfunction brought on by diabetes. Data indicated that unmodified PAMAM dendrimers might influence EGFR cell signalling pathways in vivo, with corresponding pharmacological effects depending upon their physicochemical characteristics. PAMAM dendrimers, either by themselves or in combination with vasculoprotective drugs may contribute positively to the management of diabetes-associated vascular problems [75].
Micelles
Polymeric micelles develop by the Self-Assembly of amphiphilic copolymers into a micellar core-shell configuration after reaching their critical micellar concentration. Outer functional groups facilitate modification, whereas the inner hydrophobic core facilitates the loading of hydrophobic drugs. Because of their improved pharmacokinetics and ability to stop protein degradation, these types of systems have been utilised in medication administration [76]. Drugs like insulin or GLP-1 analogue can be released by micelles in a regulated and prolonged manner, lowering the frequency of drug administration and assisting in the long-term maintenance of stable BGL. The risk of hyperglycemia and hypoglycemia is decreased by the controlled release of insulin or other antidiabetic drugs from micelles, which have to prevent sharp increases or decreases in blood glucose. An alternative to insulin injection, micelles can release insulin from enzymatic breakdown in the stomach and promote its absorption in the intestines. Various studies have been performed for micelles as a nanocarrier system in the treatment of diabetes mellitus is given below.
Liu et al. examined polymeric micelles for insulin administration that show a dual reaction to hydrogen peroxide (H2O2) and glucose. Hydrophilic PEG formed the shell of the polymeric micelles formed by the self-assembly of poly (ethylene glycol)-block-poly (amino phenylboronic ester) (PEG-b-PAPBE), whereas the hydrophobic PAPBE provided dual sensitivity to glucose and H2O2. Glucose-responsiveinsulin release can result from the hydrolysis of the intrinsic phenylboronic ester (PBE) by H2O2 and the subsequent cleavage by glucose due to the breakdown of polymeric micelles. Glucose oxidase (Gox) co-encapsulation in the micelles significantly enhanced insulin release. The PBE was hydrolysed by the H2O2 generated by the Gox-mediated catalytic oxidation of glucose. Giving insulin/Gox-coloaded polymeric micelles to diabetic mice subcutaneously demonstrated a significantly enhanced hypoglycaemic effect in vivo compared to free insulin or micelles containing insulin only. This polymeric micelle exhibiting dual tolerance toH2O2 and glucose offers a viable approach for diabetes management [77]. Zhu et al. synthesised poly (ethylene glycol)-b-poly (3-acrylamidophenylboronic acid-co-styrene) (PEG-b-P(PBA-co-St)) to produce insulin-loaded micelles (ILM) and epidermal growth factor (EGF) further integrated into the composite hydrogels that may be rapidly gelled by the combination of succinyl chitosan (SCS) and oxidised hyaluronic acid (OHA). An in vivo wound healing investigation was conducted using a STZ-induced rat model to evaluate the wound healing efficacy of the synthesised composite hydrogels. Moreover, the synthesised composite hydrogels containing EGF and ILMexhibited remarkable efficacy in wound healing, as shown by fibroblast proliferation, preservation of tissue internal structural integrity, and deposition of myofibrils and collagen. The results suggested that the synthesised composite hydrogels containing EGF and ILM might be an appropriate choice for the applications of wound healing [78]. Bahman et al. created an oral insulin delivery system using a poly-(styrene-co-maleic acid) (SMA) micellar system to improve intestinal absorption, decrease the rate at which insulin degrades in the stomach, and supply a physiologically appropriate version of insulin that can pass into portal circulation. Animal studies indicated that orally given SMA-insulin will induce a hypoglycaemic effect lasting up to 3 h after a single dosage. The results indicated that SMA micelles may facilitate the oral delivery of bioactive molecules like insulin and may serve as useful tools in diabetes treatment [79]. Kassem et al. synthesised repaglinide-phospholipid-complex-enriched micelles (RGPLC-Ms) using the solvent evaporation technique. The data indicated that after 7 days of oral administration, the micelle formulation reduced blood glucose levels in diabetic-induced rats by 83.02% (from 558.40 to 94.80 mg/dl), as compared to a 55.40% reduction (from 543 to 242.20 mg/dl) achieved with the commercial product[80]. Kumar et al., developed orally bioavailablenano-micelles with sustained release made of the amphiphilic block copolymer of lauric acid-conjugated-F127 (LAF127). The LAF127 block copolymer was synthesised using esterification and extensively characterised prior to its use in the development of glipizide-loaded nano-micelles (GNM) using the thin-film hydration method. With a polydispersity index (PDI) of less than 0.2 and a homogeneous particle size distribution, the optimized formulation had an average particle size of 341.40±3.21 nm. The formulation exhibited a surface charge of −17.11±6.23 mV. Micelles loaded with drugs demonstrated a significant decrease in blood glucose levels in diabetic rats over a period of up to 24 h. Significantly, both the empty nano-micelles of LAF127 and the drug-loaded micelles exhibited no signs of toxicity in healthy rats. The potential of the synthesized LAF 127 block copolymer for the creation of effective oral drug delivery systems with anti-diabetic efficacy and negligible side effects is revealed by this work [81]. Singh et al., developed Quercetin-loaded Soluplus® micelles (SMs) to improve bioavailability and provide sustained release for diabetic treatment. To optimize the formulation created using the co-solvent evaporation technique, the Box-Behnken response surface methodology was employed. The physicochemical characterisation validated the nano-spherical morphology of Quercetin-loaded Soluplus® micelles (Qu-SMs), exhibiting an average particle size of 85-108 nm and an encapsulation effectiveness of 63-77%. The in vivo pharmacokinetic investigation demonstrated enhanced bioavailability with the encapsulation of the drug in SMs. Furthermore, the research conducted to assess the efficacy of diabetes therapy shown an improved anti-diabetic effect [82].
Solid lipid nanoparticles (SLNs)
Submicron particles, or SLNs, range in size from 50 to 1000 nm are made up of an aqueous dispersion with a lipid crystalline core encased in a surfactant. They are a new category of lipid emulsions with a submicron dimension, in which the solid lipid has replaced the liquid counterpart. They possess an exceptional drug-loading capacity and the ability to absorb both lipophilic and hydrophilic compounds due to their expansive surface area and distinctively small size [83]. Triglycerides (tricarpin and tripalmitin), partial glycerides (glyceryl monostearates and glyceryl palmostearate), and fatty acids (stearic acid and palmitic acid) are the main biodegradable lipids used in the production of SLNs. Insulin-dependent diabetes mellitus, often known as type I diabetes, is treated with insulin. In that instance, daily injections or an insulin pump are used to deliver insulin, which is unpleasantanduneasy. Oral insulin administration is challenging and ineffective because of the physical environment of the gastrointestinal tract (GIT), the enzymatic activity of protein digestive enzymes like trypsin, proteases, chymotrypsin, pepsins, and carboxypeptidases, as well as a particular cytosolic enzyme called insulin-degrading enzyme. The encapsulation technique can be used to protect labile proteins, such as insulin, from the harsh condition of GIT and enzymatic degradation. The process additionally facilitates a regulated release pattern [84]. The role of SLNs in diabetes management is discussed below.
Bharti Sharma et al., aims to create Tetrahydrocurcumin (THC)-loaded SLNs to increase anti-diabetic efficacy and the bioavailability of THC in streptozotocin-induced diabetic rats. Tetrahydrocurcumin-loaded solid lipid nanoparticles (THC-SLNs) were optimized using the Box Behnken Design. The modified THC-SLN showed enhanced bioavailability in an in vivo pharmacokinetics study, exhibiting a 9.47-fold increase in AUC (0-t) relative to standard THC solution. Furthermore, pharmacodynamic assessments of the optimised formulation revealed a significant reduction in blood glucose levels to 63.7% and an increase in the weight of body from 195.8±7.223 to 231.2±7.653 by the 28th day of the trial, exhibiting superior anti-diabetic efficacy compared to the plain drug solution. The stability investigations indicated that the formulation may be kept for extended durations at the room temperature [85]. Anchan et al. developedinsulin-loaded SLNs coated with chitosan for oral delivery and examined their efficacy as potential substitutes for subcutaneous injections. Following an 8-hour experiment, chitosan-coated insulin SLN administered orally to streptozotocin-induced diabetic rats produced a significant hypoglycaemic effect (p<0.05) compared to groups that received oral insulin solution or uncoated insulin-loaded SLN, which was the same as insulin administered subcutaneously. Thus, it can be concluded that chitosan-coated SLN may serve as an effective oral insulin formulation [86]. Mohseni et al., formulated resveratrol (RES)-loaded (SLN-RES) to enhance insulin resistance by upregulating the SNARE protein complex in rats with type 2 diabetes. The oral treatment of SLN-RES inhibited weight loss and demonstrated a superior hypoglycaemic effect compared to RES. SLN-RES normalised serum oxidative stress levels. Moreover, the expression of vesicle-associated membrane protein 2 (Vamp2), synaptosomal-associated protein 23 (Snap23), and syntaxin-4 (Stx4), which are key components of the SNARE protein complex, was reduced by SLN-RES to a greater extent than by RES treatment in muscle tissue. However, SLN-RES has an effect like that of RES therapy in adipose tissue. Collectively, our findings indicate that SLN-RES may serve as an emerging and promising therapeutic strategy for enhancing insulin resistance by modulating the expression of Vamp2, Stx4, and Snap23 in muscle and adipose tissues [87]. Oroojan et al. investigated the effects of myricitrin and myricitrin-infused SLNs on the reproductive system of male mice with type 2 diabetes. All dosages of SLN containing myricitrin (1, 3, and 10 mg/kg) and myricitrin (10 mg/kg) boosted antioxidant potential and SOD levels (p<0.05) while total antioxidant potential and SOD levels declined in diabetic mice. It was observed that the testes in the diabetes group had decreased in weight and volume. The diabetic group's follicle-stimulating hormone, sperm count andluteinizing hormone were all restored when a high dosage of myricitrin or all three doses of SLN containing myricitrin were administered (p<0.05). Diabetes induced vacuolation and death in testicular cells; however, myricitrin and SLN with myricitrin strengthened these cells (p<0.05). Myricitrin, or SLN containing myricitrin, cured the symptoms of reproductive problems induced by diabetes by reducing antioxidant capacity and increasing oxidative stress. Moreover, SLN with myricitrin showed superior efficacy compared to myricitrin alone [88]. Shah et al., developed linagliptin (LGP) (LGP-SLNs) using poloxamer 188 and Tween 80 as P-glycoprotein inhibitors. LGP-SLNs were synthesised using tween 80, poloxamer 188 and palmitic acid, as the co-surfactant, surfactant, and lipid, respectively, by the hot homogenisation ultrasonication technique, and optimised via a 32 complete factorial design. The pharmacokinetic and pharmacodynamic assessment was conducted in albino Wistar rats. The results showed that L12 had mean particle size, zeta potential, %EE, and polydispersity index (PDI) of 225.96±2.8 nm, -5.4±1.07 mV, 73.8±1.73%, and 0.180±0.034, respectively. A CDR of 80.96±3.13% was recorded at 24 h. In the absorptive direction, LGP-SLNs had permeability values that were 1.82, 1.76, and 1.74 times higher than those of LGP-solution (LGP-SOL) in single-pass intestinal perfusion (SPIP), everted gut sac, and Caco-2 permeability experiments, respectively. The relative bioavailability of LGP-SLNs was 300% and a superior decrease in the levels of glucose compared to LGP-SOL in rats. The improved oral bioavailability of LGP-SLNs may result from the suppression of P-glycoprotein efflux and lymphatic targeting. Enhanced bio-absorption may lead to a decrease in dosage, dose-dependent side effects, and administration frequency [89].
Transfersomes
Transfersomes are a form of carrier system defined by a lipid bilayer that coversat least one interior watery compartment, together with an edge activator. These are modified liposomes including edge activators. These vesicular systems are designed to effectively carry drugs or other therapeutic substances across biological membranes [90]. Enzymatic breakdown and inadequate absorption in the gastrointestinal tract make oral insulin difficult to use. Transfersomes improve bioavailability and make insulin therapy more convenient by enabling insulin to get around these problems and enter the bloodstream through this skin. Transferosomes can bend and compress through skin constrictions or tiny apertures much smaller than the vesicle dimensions without experiencing notable loss because of their natural elasticity. Transferosomes consist of a mixture of a phospholipid component and single-chain phosphatidylcholine, rather than the natural or synthesised phosphatidylcholine present in normal liposomes. Edge activators (EAs), functioning as membrane destabilising agents, are notably efficient in enhancing the deformability of vesicle membranes. This combination makes the transferosomes flexible and highly adaptable, boosting their penetration ability [91]. The application of transfersomes in diabetes management is given below.
Mazhar et al., synthesised Metformin hydrochloride (MF HCl) loaded transfersomes (MF-TFs) with a modified thin film hydration method. Carbopol gel with a permeation enhancer (PE) was then mixed with the prepared MF-TFsto boost medication bioavailability for the potential treatment of T2DM via the transdermal method. Skin permeation experiments showed enhanced penetration of MF-TFs gel with PE (1041.7±7.53 μg/cm²) relative to the pure drug. The glucose tolerance test indicated that MF-TFs significantly decreased blood glucose levels in test subjects relative to the pure drug present in the gel and the oral drug solution. Invivo pharmacokinetic assessment In vivo pharmacokinetic assessment revealed enhanced MF HCl bioavailability, shown by an improved AUC0-α of 42.336±1.115 ng h/ml and a Cmax of 2.195 μg/ml for the MF-TFs gel with PE, compared to the oral drug solution [92]. Chauhan et al. developed a Glimepiride formulation based on transfersomes employing sodium deoxycholate and phosphatidylcholine, subsequently transforming it into a pro-transfersome gel. A skin permeation investigation using Franz diffusion was performed on pig ear skin over a 24-hour period. The flux value of the pro-transfersomal gel was determined to be 5.129±1.24 μg/cm²/h, which exceeds that of the drug solution at 0.430 μg/cm²/h. The optimised formulation showed enhanced stability for three months. The pharmacokinetics investigation indicated that the pro-transfersomal gel of glimepiride exhibited significant release of drug in comparison to a conventional transdermal patch [93]. Abdallah et al. developed atransfersomal gel loaded with silymarin to enhance the characteristics of silymarin. Utilizing a Box-Behnken design with three levels and three factors, the transethosomal formulation was optimized, including three independent variables (duration of sonication, phospholipid concentration and surfactant concentration) and two dependent variables (EE and in vitro drug release). The drug-loaded transfersomal gel had a pH of 7.05, a reported spreadability of 55.35 mm, and a viscosity of 6.27 Pa. The silymarin-loaded transfersomal gel exhibited a transdermal flow of 92.41 μg/cm²/h, markedly surpassing that of the silymarin solution. In vivo findings shown that thetransfersomal gel loaded with silymarin considerably lowered the levels of blood glucose compared to the oral silymarin solution and silymarin gel [94]. Ramkanth et al. developed pioglitazone-based nano-transfersomes, optimised using the Box-Behnken design, and then transformed them into gel form. In a pharmacokinetic trial, the optimised nanotransfersomal gel showed superior efficacy for prolonged monitoring diabetes andhigh blood pressure in Wistar rats compared to oral nanotransfersomal formulations loaded with drugs. It was disclosed that this combinational treatment would be effective in managing diabetic hypertensive individuals [95]. Malakar et al. developed an insulin-based transfersomal gel using cholesterol, soya lecithin, tween 80, and sodium deoxycholate. The drug entrapment efficacy of the developed product was around 56.55%, with an average vesicle size ranging from 625 to 815 nm. Over the course of 24 h, the optimal transfersomal gel's in vitro penetration of insulin through pig ear skin showed zero-order kinetics. The in vivo findings of the optimised transfersomal gel in alloxan-induced diabetic rats showed a sustained hypoglycaemic impact for 24 h after transdermal administration [96].
Ethosomes
Ethosomes are flexible vesicles ranging from nanometre to micron size, mostly consisting of phospholipids, a relatively high concentration of ethanol, and water. These are innovative vesicular carriers designed for improved delivery through the skin, facilitating the penetration of drugs into the deeper skin layers and/or the systemic circulation. Despite their conceptual sophistication, ethosomal systems are defined by their simple preparation, safety, and efficacy a combination that significantly enhances their applicability. Compared to conventional liposomes, ethosomes can penetrate the skin more effectively because of their increased deformability, which is caused by their alcohol concentration. The enhanced transport of bioactive compounds over the epidermal and cellular membranes using an ethosomal carrier several challenges and possibilities for research and the development of innovative therapies [97]. The application of ethosomes in diabetes management is given below.
Aouta et al., developed a transdermal patch containing a combination of glimepiride and duloxetine using a solvent casting method, and a 32 factorial design was used to optimise the ethosomal formulation. Phospholipid and ethanol were used as independent variables in the development of nine formulations, with particle size and entrapment efficiency acting as dependent variables. Formulation F9 was shown to be optimum, demonstrating an in vitro cumulative drug release exceeding 60% within 24 h and a skin permeability more than 200 μg/cm². The findings indicated a promising method for treating diabetes and diabetic neuropathic pain by dual medication distribution through a transdermal patch that contains ethosomes [98]. Raghav et al., synthesised hybrid lecithin-chitosan (LC) Kaempferol (KMP) ethosomes (Eth) using a cold approach, and their physicochemical characteristics, as well as in vitro and in vivo studies were evaluated. Over the course of 24 h, the in vitro drug release profile demonstrated a continuous, regulated release of KMP. Approximately 80.3% of KMP was released from the LC-KMP-Eth gel, but only 46.9% of KMP was liberated from the conventional KMP-gel after 24 h. The in vivo findings on Wistar albino rat models indicated that LC-KMP-Eth gel exhibited enhanced wound closure effectiveness relative to ordinary KMP gel. The pharmacokinetic parameters of KMP from LC-KMP-Eth gel exhibited approximately a twofold increase following topical application, resulting in elevated KMP concentrations in rat plasma. This indicates a significant effect at the site of wound. Additionally, the gel maintains a moist environment and enhances adhesion of cell, thereby promoting healing in diabetic foot ulcers [99]. Bodade et al. developed repaglinide-loaded ethosomes using ethanol and dipalmitoyl phosphatidylcholine. Ethosomes loaded with repaglinide had a size range of 0.171–1.727 μm and demonstrated maximal penetration of 64–97% of the supplied dosage, in contrast to the free drug and its aqueous alcohol solution, applied to excised rat skin. The repaglinide ethosomal system’s in vivo assessment revealed extended drug release and antidiabetic infectiveness. The in vivo evaluation of the repaglinide ethosomal system demonstrated prolonged release of drug and antidiabetic efficacy [100]. Fathima et al., Developed Vildagliptin ethosomal gel for diabetic mellitus using a cold technique. A total of nine ethosome formulations were produced using varying concentrations of phospholipid (1, 2, 3% w/w) and ethanol (25, 30, 35% w/v). The optimised formulation showed an in vitro drug release of 92.06%. Carbopol 934 was used to integrate it into the gel. (1, 1.5, 2% w/w). A carbopol concentration of 1.5% w/w yields a maximal in vitro release of 94.34% and shows an ex vivo drug release of 77.46% on rat skin. The stability studies conducted at two separate temperatures, 25±2 °C/60%±5% RH and 4±2 °C/40% RH, for a duration of 3 mo revealed no significant changes [101].
Nanofibers
Nanofibers (NFs) are characterised as nanomaterials that have at least one dimension of 100 nm or less, with the length can potentially exceed diameter by a factor of 100. NFs may be produced from several natural polymers, including silk, chitosan, collagen, fibronectin, gelatin, as well as synthetic polymers such as PLA, polyglycolic acid (PGA), and PLGA [102]. NFs serve as a delivery system or reservoirs, offering a suitable matrix for the encapsulation and integration of medicinal compounds while effectively preventing degradation prior to reaching target locations, ensuring great efficiency and minimal adverse effects. These structures exhibit significant flexibility in generating diverse morphologies, contain a high drug-loading capacity (up to 60%), and show EE (up to 100%), along with the capability to evenly disperse their contents [103, 104]. Nanofibers can assist in maintaining stable blood glucose levels by delivering a continuous release of insulin, avoiding the abrupt swings that are frequently observed with conventional insulin injections. The efficacy of cell-based treatments for diabetes can be increased by encasing transplanted insulin-producing cells in nanofibers, which shields them from immune system attack. Various studies have been performed fornanofibers as a nanocarrier system in the treatment of diabetes mellitus is given below.
Panda et al., synthesised NFs using the emulsion electrospinning process with polyvinyl alcohol (PVA) both alone and in conjunction with PLGA. The optimised nanofibers have been incorporated into gelatin capsules for oral delivery. The SEM picture of the optimised formulation reveals cylindrical fibres, showing the homogenous incorporation of gliclazide inside the polymers, with an average fibre diameter of 4.357±0.83 µm. The gliclazide NFs solubility and rate of dissolutionwere markedly enhanced in comparison to pure gliclazide. The gliclazide nanofibers exhibit a biphasic drug release characteristic, which includes a quick release at first, then a gradual release phase. Oral manufactured gliclazide fibres has significant promise as a drug carrier, and different technologies for enhancing solubility, dissolution rate, minimising dosing frequency [105]. Alamer et al., fabricated a novel formulation of imeglimin encapsulated in electrospun NFs for delivery via the buccal cavity to reduce existing gastrointestinal-related side effects and to provide an easier administration route. The results showed that thedrug loading (DL) of the imeglimin NFs is 23.5±0.2 μg/mg of fibers and their diameter is 361±54 nm. The X-ray diffraction (XRD) analysis validated the solid dispersion of imeglimin, enhancing release and drug solubility, hence improving bioavailability. The disintegration rate of drug-loaded nanofibers was measured at 2±1 sec, demonstrating the fast disintegration capability of this dosage form and how well it fits for buccal distribution, with totalrelease of drug following 30 min [106].
Carbon nanotubes
Because of its unique physical characteristics, carbon nanotubes (CNTs) are inorganic nanocarriers that have been investigated as potential drug delivery vehicles. Single-walled carbon nanotubes, which have a single layer of graphene sheets, and multi-walled carbon nanotubes, which have numerous layers of graphene sheets, are the two common classifications for carbon nanotubes [107]. Insulin and other glucose-regulating substances can be transported by CNTs. They are distinct form and large surface area allow for optimal drug loading and distribution to organs, such as the liver and insulin-resistant cells enhancing therapy efficacy to create sophisticated noninvasive glucose monitoring systems, it can be integrated with biosensors. Because carbon nanotubes are conductive and are can be functionalized to detect glucose level with high sensitivity, blood glucose level can be continuously and instantly monitored. Various studies have been performed forcarbon nanotubes as a nanocarrier system in the treatment of diabetes mellitus is given below.
Zaman et al. confirmed the antidiabetic efficacy of functionalised carbon nanotubes (f-CNTs). To functionalize the multi-walled carbon nanotubes (MWCNTs), 95% concentrated nitric acid (HNO3) was refluxed. The surface functional groups of MWCNT were analysed using X-ray photoelectron spectroscopy (XPS) and Fourier transform infrared spectroscopy (FTIR). The alterations in the surface morphology of MWCNT following functionalization were directly investigated using high-resolution transmission electron microscopy (HRTEM). The f-CNTs exhibited 41.2% suppression of aldose reductase activity in vitro. The docking analysis demonstrated the carboxylic group's crucial function in inhibitory action. This research demonstrates the MWCNTs' potential for antidiabetic effectiveness [108].
Table 1: Key properties, advantages, and disadvantages of various nanocarriers
| S. No. | Nanocarrier | Key properties | Advantages | Disadvantages | Ref. |
| 1. | Polymeric nanoparticles | Biodegradable, biocompatible, high surface area | Targeted and controlled drug delivery |
Toxicity concern and complex manufacturing | [53] |
| 2. | Liposomes | Phospholipid bilayer, encapsulates both hydrophobic and hydrophilic drugs | Biocompatible, low toxicity | Instability in physiological conditions, expensive to produce | [60] |
| 3. | Niosomes | Non-ionic surfactant-based vesicles | Stable in a wide pH range and can encapsulate hydrophilic and lipophilic drugs | Limited drug release rate, some surfactants may be toxic | [66] |
| 4. | Dendrimers | Branched, tree-like polymers, highly functionalized | High drug encapsulation efficiency, precise control over size and shape | Complex synthesis, potential cytotoxicity at high doses | [72] |
| 5. | Micelles | Amphiphilic, self-assembled surfactant molecules | Efficient drug loading, low toxicity, improved solubility of hydrophobic drugs | Limited stability in biological fluids, poor drug release control | [76] |
| 6. | Solid lipid nanoparticles | Solid lipid core, high surface area | High drug loading, biocompatible and controlled release of drugs | Low encapsulation efficiency for some drugs and complex preparation methods | [83] |
| 7. | Transfersomes | Phospholipid vesicles with edge activators, elastic | Enhanced skin penetration, non-invasive drug delivery | Expensive, stability issues under certain conditions | [90] |
| 8. | Ethosomes | Phospholipid-based vesicles with higher ethanol content |
Enhanced transdermal delivery, non-invasive, improved bioavailability | Ethanol content may cause irritation, limited stability | [97] |
| 9. | Nanofibers | Ultra-thin fibers, high surface area, flexible | Controlled and sustained release, potential for wearable devices |
Difficult to scale up production, mechanical instability | [102] |
| 10. | Carbon nanotubes | Cylindrical carbon structures, excellent conductivity, high surface area | High drug loading capacity, targeted delivery, enhanced stability, and bioavailability | Potential toxicity, difficulty in functionalization, regulatory challenges |
[107] |
Insulin pump
The National Institute for Health and Clinical Excellence (NICE) Technology Appraisal Guideline TA 151 highlighted the individuals who may benefit from continuous subcutaneous insulin infusion (CSII) therapy. For children under the age of twelve, especially those under five, Continuous Subcutaneous Insulin Infusion (CSII) is recommended as the preferred therapeutic approach. Novel approaches for the delivery of insulin have shown significant improvements over conventional methods. These approaches include delivering insulin by palmary, oral, buccal, nasal, rectal, and ocular routes. The sophisticated ultra-long insulin analogue, Degludec, is also recognised as beneficial [109]. Insulin pump therapy aims to replicate physiological insulin production by constantly administering small amounts of basal insulin, supplemented by prandial boluses. More precise insulin administration is made possible by the ability to adjust boluses and basal insulin in response to daily fluctuations in personal insulin sensitivity and requirements. Insulin pumps are becoming compact and user-friendly. The fast progression of technology has resulted in the availability of "smart" pumps, which include several supplementary capabilities that enhance diabetes management. Insulin pumps provide more accurate delivery of insulin, more flexibility in daily self-management, and a reduction in the number of injections relative to multiple daily injections (MDI) or multiple dosage injection treatment. Multiple studies have shown that insulin pumps provide superior glycaemic control, resulting in a 50% decrease in the risk of cardiovascular mortality compared to MDI [110]. Insulin pumps provide rapid-acting insulin continuously in small dosages via a catheter implanted under the skin. These pumps are compact, resembling telephones, and are attached to pockets, socks, garments, or belts. Numerous pumps possess electrical memory, diverse basal metabolic rates, different bolus options, customisable menus, notifications, and electronic controls. A reservoir (insulin), a small cordless pump, and a microprocessor that controls insulin delivery make up these surface insulin pumps. This relates to a small and elastic-plastic tube, ending with a needle positioned just under the skin, next to the abdomen. This implantation set is changed in every few days. Patients position the pump to provide a continuous basal or trickle insulin dosage throughout the whole day. The pump administers a bolus dosage of insulin, which is programmed to provide a reduced amount of insulin throughout the day. Animas IR 1000, DANA Diabecare II, Cozmo, MiniMed 508, Disetronic, Amigo, and MiniMed 512 are few examples of insulin pump models [111].
Additionally, implanted pumps have been developed to provide insulin via the intraperitoneal route [112]. This pump administered a minimal daily dosage of insulin. Patients may regulate the dosage using a portable device, resulting in the insulin pump treatment delivering a predetermined amount of insulin. In two to three months, this pump was replenished. The Mini-Med Implantable Pump, including an upgraded side port catheter and a formulation of insulin with greater stability against agitation and temperature, was used in European clinics [113]. Multiple studies on individuals with insulin-dependent diabetes have shown that insulin delivered via an implanted pump is rapidly absorbed, primarily reaching the systemic circulation, hence facilitating hepatic insulin delivery. Research indicates that optimal glucose regulation may be achieved with reduced glycaemic variability and a decreased episode of hypoglycaemia [114]. Various studies have been conducted to assess the overall use of insulin pumps in recent years [115, 116]. The electronic medical report (EMR) survey data from 2011 to 2016 indicated an increase in pump usage to 61.8%, compared to earlier records and population-based data, which showed a rise from 22% in the mid-2000s to 56.3% by 2012 [117]. This matches the data collected from multi-site records in 2013 [118].
Maikawaet al., developed a stable co-formulation drug product candidate for insulin and pramlintide that may help regulate blood sugar levels and lessen patient burden. As a stabilizing agent, this formulation employs the amphiphilic acrylamide copolymer excipient acryloylmorpholine77%-N-isopropylacrylamide23% (MoNi23%) [119].
Artificial pancreas
A closed-loop artificial pancreas (AP) has shown to be a successful approach for managing diabetes [120, 121]. The AP, an effective system, autonomously monitors BGL and administers insulin or insulin combinations to patients with Type 1 Diabetes Mellitus. An emerging artificial pancreas is transformative and innovative for several type 1 Diabetes Mellitus patients [122]. On September 28, 2016, the FDA authorized Medtronic's MiniMed 670G System, the first hybrid closed-loop system. It monitors blood glucose levels and autonomously regulates basal insulin dosages in individuals with Type 1 Diabetes Mellitus. The FDA endorsed and emphasized the safety and effectiveness of this medical equipment.
Jongh et al., discovered that recipients of bioartificial pancreas (BAP) transplants may experience psychological and social comfort, which lessens their emotional burden and may even help with diabetic management. It is thought that BAP lowers healthcare expenses and improves social engagement by reducing diabetes-related problems in recipients [123].
Tissue engineering
The National Science Foundation Assembly defined tissue engineering (TE) as the application of life sciences and engineering principles to acquire fundamental knowledge regarding the function and structure of both pathological and normal mammalian tissues, as well as the development of biological substitutes that restore, enhance, or sustain tissue function. The islets of Langerhans are scattered throughout the exocrine pancreas and make up the endocrine pancreas [124]. The islets are tiny organs made up of 200–2000 endocrine cells, such as epsilon cells, alpha cells, delta cells, β-cells, and pancreatic polypeptide cells. Ghrelin, somatostatin, glucagon, pancreatic polypeptide, and insulin ghrelin are all released by these cells, respectively. Tissue engineering may address the drawbacks of the Edmonton procedure, resulting in increased lifetime of islet transplantations. Integrating cells, especially islets or β-cells, with biomaterials that offer mechanical support and a suitable extracellular environment to maintain cell viability and functionality both in vitro and in vivo is the idea behind tissue engineering for cell-based treatment [125].
A tissue engineering strategy may be essential for discovering a sustainable treatment solution for type 1 diabetes. Recent work identifies three primary components as essential for islet-based tissue engineering: (1) Functional cells capable of forming aggregates and secreting insulin in reaction to glucose; (2) extracellular matrix (ECM) components integrated into a biomaterial to promote cell viability, facilitate differentiation, and enhance the functionality of insulin-secreting cells; and (3) a biomaterial with mechanical support that is utilized as an encapsulating system or scaffold to distribute cells in vivo. A possible option for delivering insulin to patients with Type 1 Diabetes Mellitus (T1DM) is the process of transplanting separate pancreatic islets [126]. This method showed short-term efficacy in achieving insulin independence for patients; however, its long-term success was limited. A primary disadvantage of this method is the restricted availability of donor cells, necessitating xenotransplantation, which eventually causes the recipient's immune system to kill the transplanted islet cells, hence requiring lifelong immunosuppressive drug therapy. Xenotransplantation is recognised as the solution to address organ shortages. However, there exists an associated risk of graft rejection and viral transmission. Primarily, solid organ transplantation is associated with many complications. For example, patients who have undergone transplantation may develop diabetes [127, 128]. A large exposure to bacterial, viral, and parasitic diseases is a further contributor to anxiety in this transplantation. A similar mechanism may affect islet transplantation, possibly obscuring the engraftments. Certain regulatory systems and substances are linked to β-cell development (both in vivo and in vitro). The transplanted islet cells cultivated on a 3D platform serve as a barrier against the immune system while facilitating the efficient flow of nutrients, oxygen, and waste products [129–131].
Kelly et al. developed a PEG-extracellular matrix that mimics the interaction of the islet microenvironment. Thus, the hydrogel was prepared by combining PEG, fibrinogen, vitronectin, collagen type IV, and laminin with collagen type I and type IV. The PEG-ECM hydrogel was used to encapsulate the insulin secreting cells, and significant changes in the level of insulin emission and cell feasibility were observed. The primary issue associated with islet cell aggregation was hypoxia at the site, which resulted in cell mortality and necrosis due to a deficient of oxygen level [132].
Bernard et al. used a PEG hydrogel microwell to establish a culture cell framework. In comparison with single-cell cultures, the aggregated β-cell in the hydrogels showed enhanced cell functionality and viability (insulin secretion) [133].
Gene therapy
Gene therapy is a method that addresses the symptoms of an infection caused by a defective gene by the addition of an exogenous normal gene [134]. Currently, gene manipulation includes not only the insertion of a gene as well as editing and modifying genes. Gene therapy is defined as a technique for introducing or manipulating genes into a cell as a therapeutic approach for disease management [135, 136]. The introduction of gene therapies as a trend in advancing treatments is due to its potential in treating several conditions, like diabetes mellitus, autoimmune disorders, cardiovascular illnesses, and malignancies, which are challenging to control through conventional medications. Adeno-associated viral vectors have been used to introduce the gene therapy strategy for the treatment of diabetes, which is based on the gene co-expression network (GCN) of the insulin and glucokinase genes in skeletal muscles. Normal blood glucose levels may be achieved via the lasting efficacy of gene therapy, except for the need for exogenous insulin delivery [137]. Without assimilating into the host cell's genome, these vectors infect both lethargic and partitioning cells and induce a minor resistance reaction. Thus, the best option for gene therapy is an adeno-associated viral vector. The skeletal muscles of mice and dogs with STZ-induced diabetes receive the glucokinase and insulin genes via adeno-associated viral vectors. The chromosomal translocation of GLUT4 and glucokinase is improved by their co-expression, which promotes glucose absorption in the cells of muscles. In skeletal muscle, glucokinase expression prevents glucose from being phosphorylated to glucose-6-phosphate. The glucose sensor measures blood glucose levels and indicates the amount of insulin manufactured to maintain normal blood glucose levels [124, 138]. Numerous methods of gene transfer have been employed. In addition to gene transfer utilizing viral vectors, these approaches include non-viral methods like as calcium phosphate co-precipitation, lipofection, direct microinjection, electroporation, and biolistics. For the genetic alteration of pancreatic cells, calcium phosphate co-precipitation is an easy and economical method. After the introduction of calcium chloride to the buffer saline/phosphate solution containing the DNA of interest, a precipitate appears to form. Cells have the capacity to absorb the DNA-containing precipitate through the processes of endocytosis or phagocytosis. This methodology has been studied across a diverse range of cell types and can generate either transiently transfected cells or cells that can rapidly express the transgene. Unlike the in vitro calcium phosphate co-precipitation method, liposomes have been employed as extremely effective transfection agents for the cells in both in vivo and in vitro conditions. The primary advantage of in vivo lipofection is their ability to administer liposomes via injection into the circulation, making it a less invasive alternative compared to other treatments such as transplantation. Liposome encapsulating DNA exhibit a minimal positive charge, which enhances their interaction with target cells and the improves transfection efficiency [139]. The direct injection of DNA into cells constitutes an efficient approach for the transaction of cellular structure. However, since each cell must be addressed individually, this method is tedious and is not appropriate for targeting many cells. Despite the possibility of gene transfer into cells, electroporation is ineffective at integrating DNA into the host genome. Biolistic transfection produces higher transfection efficiencies than both lipofection and calcium phosphate co-precipitation techniques. Biolistics refers to the application of “gene gun” for the purpose of transfecting cells with the transgene [140–142]. Careful consideration is required while selecting an appropriate vector. To be successful, vectors must be simple to manufacture in large quantities, capable of being targeted to a specific site, capable of transducing both dividing and non-dividing cell, achieve high transduction efficiency, not cause a strong immune response, and facilitate long-term expression of the transgene. The vector must penetrate through the islet membrane and transduce the bundle of cells within to facilitate transgene delivery into islets.
The discovery of CRISPR-Cas9 has begun a novel approach in the treatment strategy for the disease, offering significant chances of complete cure at the genetic level. In recent years, gene-editing therapeutics have gained significant importance, with clustered regularly interspaced short palindromic repeats (CRISPR-Cas9) developing as an important technique. CRISPR-Cas9, widely known as a genome-editing technology, originates from an adaptive immune response developed in bacteria and archaea. However, it can also be used in eukaryotic organisms through the incorporation of single guide RNA (sgRNA), which contain complementary base sequences that correspond to the target genes.
By synthesising a specific sgRNA that is complementary to a particular gene, the RNA-guided DNA nuclease Cas9 can induce a double-strand break within that gene. Bevacqua et al. developed a CRISPR/Cas9 model aimed at targeting the PDX1 gene, which they implemented via lentiviral transduction in a culture of islet cells. Authors chose PDX1 because it encodes a tran-scription factor crucial for β-cell function and which is mutated in human diabetes. Insertions and deletions were identified in approximately 66% of the sequences analysed through polymerase chain reaction (PCR). This method demonstrated a significant reduction in the expression of the PDX1 protein within the group of cells subjected to the CRISPR system, thereby indicating that specific targeting of PDX1 was possible. Data indicate that the loss of PDX1 resulted in compromised physiological function of β-cells. similar, outcomes were observed when targeting on the KCNJ11 gene, which led to a loss of function in β-cells[143]. Wang et al. developed four different single guide RNAs (sgRNAs) aimed at regions approximately 50 to 150 base pairs (bp) upstream of the human UCP1 gene, in conjunction with a synergistic activation mediator (SAM) to enhance the efficacy of CRISPR activity. Uncoupling protein 1 (UCP1) plays a crucial role in non-shivering adaptive thermogenesis, and its expression is associated with increased energy expenditure, presenting it as a potential candidate for anti-obesity therapies. The previously mentioned CRISPR-SAM model was used on human white pre-adipocytes collected from two individuals. The results showed that the messenger RNA (mRNA) levels of UCP1 showed a 6000-fold, along with a 20-fold increase in UCP1 protein expression. Obese mice that developed human brown-like adipocytes showed a sustained improvement in glucose tolerance and insulin sensitivity [144]. Table 2 highlighted recent clinical trial data related to gene therapy.
Table 2: Clinical trial data of gene therapy
| S. No. | Trial ID | Sponsor | Title | Ref. |
| NCT04567550 | AbbVie | RGX 314 Gene Therapy Administered in the Suprachoroidal Space for Participants with Diabetic Retinopathy (DR) Without Center Involved-Diabetic Macular Edema (CI-DME) (ALTITUDE) | [145] | |
| NCT04055090 | Helixmith Co., Ltd. | Extension of Phase 3 Gene Therapy for Painful Diabetic Neuropathy | [146] | |
| NCT02561546 | Shenzhen SiBionoGeneTechCo., Ltd | p53 Gene Therapy in Treatment of Diabetes Concurrent with Hepatocellular Carcinoma | [147] | |
| NCT01002235 | Helixmith Co., Ltd. | Gene Therapy for Painful Diabetic Neuropathy | [148] | |
| NCT02427464 | Helixmith Co., Ltd. | Phase 3 Gene Therapy for Painful Diabetic Neuropathy | [149] | |
| NCT00065663 | Tissue Repair Company | Gene Therapy to Improve Wound Healing in Patients with Diabetes | [150] | |
| NCT00056290 | Losordo, Douglas, M. D. | VEGF Gene Transfer for Diabetic Neuropathy | [151] | |
| NCT04418427 | Adverum Biotechnologies, Inc. | ADVM-022 Intravitreal Gene Therapy for DME (INFINITY) | [152] |
Stem cell therapy
Traditional methods for managing diabetes mellitus fail to address the underlying causes of the condition and are associated with undesirable side effects. Consequently, there is a search for a preferable alternative therapy regimen. The cellular therapy approach now used in diabetes treatment involves pancreatic or islet-cell transplantation to restore beta cell function for insulin production. This method is limited by the shortage of donor organs. These issues prompt the investigation into the possibility of generating beta cells from stem cells. The unique regenerative capacity of stem cells may serve as a significant resource in the treatment of diabetes mellitus. The development of a renewable reservoir of islets using stem cells may reduce the current supply and demand issues in islet transplantation and provide individuals with diabetes a sustained source of beta cells for insulin production. Therefore, research on stem cells has become a successful strategy for treating diabetes mellitus [153]. The goal of stem cell diabetes treatment is to use pluripotent or multipotent stem cell to replace damaged or malfunctioning pancreatic cells. Using various techniques, this method has created surrogate beta cells or restored the physiological function of beta cells by utilizing the properties of various stem cells, including adult stem cells, induced pluripotent stem cells (iPSCs), embryonic stem cells (ESCs), and adult stem cells [154]. Technological advancements have made possible the development of stem cells from several sources of tissues, such as bone marrow, tooth pulp, adipose tissue, and umbilical cord, blood, skin, and periosteum. The pancreas is often the first organ selected in the search of potential stem cells. Using animal models in research has shown that a little quantity of pancreatic tissue, when provided, might restore the optimal mass of pancreatic beta cells [155]. Fig. 4 shows the schematic representation of steps involved in stem cell therapy.
Fig. 4: Representation of stem cell therapy for treating diabetes mellitus
Future opportunities, obstacles to clinical translation, and the clinical outlook
The advancement of nanocarriers for diabetes treatment from research to real-world clinical application offers a variety of opportunities. Lipid-based NPs, such as SLNs, are being developed to improve the oral bioavailability of antidiabetic drugs with lower solubility. Long-term drug release and enhanced pharmacokinetic characteristics have been demonstrated by these formulations, both of which are important for the effective management of diabetes. Oral insulin delivery is frequently investigated using polymeric nanoparticles [156]. Insulin has been shown to be effective in preventing gastrointestinal degradation and enhancing its absorption when protected by polymers such as chitosan, alginate, and PLGA. Research suggests that these nanoparticles significantly improve insulin bioavailability in comparison to conventional delivery methods. In addition, the potential applications of nanocarriers that are specifically engineered for gene therapy are promising [157]. Oligonucleotide delivery can offer novel approaches to the long-term treatment of type 1 diabetes by either stimulating insulin secretion or modulate immune responses. Despite the substantial potential of nanomaterials, certain challenges must be successfully addressed to ensure their successful application in antidiabetic drug delivery. Toxicology, short shelf life, pharmacokinetic characteristics, stability concerns, expensive formulations, lowering EE, and difficulties with scaling and manufacturing procedures are a few of these difficulties. To overcome these challenges and maximise the potential of nanomaterial in drug delivery system designed for antidiabetic treatment, it is essential to carry out research that is focused on the development of biocompatible, stable, and scalable nano-formulations. In that condition, the development of targeted delivery of antidiabetic drug formulation using nano-formulation and the integration of artificial intelligence and machine learning algorithms to optimize the design, production, and overall efficacy of nanotechnology-driven antidiabetic drug delivery methods are of utmost importance. Although nanomaterials have undoubtedly transformed the diabetes management landscape, there are still a variety of areas that exhibit potential for further advancements, particularly in the fields of biosensor design, medicinal nano-devices, nano-diagnostic tools, and nano-therapeutics. Nanoparticles have shown the capacity to efficiently transport proteins and genomes, thereby significantly improving therapeutic outcomes. The potential of nanomaterials in the field of gene therapy is particularly fascinating, as it presents a promising opportunity for future research and development. There is an urgent need to developed cost-effective and simplified methodologies to produce nanoparticles with the variety of functionalities and morphologies to meet the changing needs of the field. Additionally, an effective approach may be discovered by conducting research and identifying novel glucose-responsive moieties with higher association constants and materials that incorporate these binding domains. Therefore, the development of innovative diabetes treatments and the prevention of complications associated with diabetes mismanagement may be eased by ongoing research.
CONCLUSION
The use of nanotechnology has become a game-changing strategy to address the ongoing challenges associated with controlling diabetes mellitus. For several decades, many researchers have conducted detailed studies on the investigation of nanotechnology and its potential application in drug delivery. Nanomaterials have shown significant potential in the areas of glucose monitoring, glucose sensing, as well as in the administration of medications for diabetes. The development of nanotechnology-based infrastructures that incorporate polymeric nanoparticles (NPs), liposomes, niosomes, dendrimers, micelles, solid lipidNPs, transferosomes, ethosomes nanofibers and carbon nanotubes. Each provides a variety of advantages such as improved stability, targeted delivery, controlled release, enhanced bioavailability, reduced toxicity, lesser administration frequency, and doseproportionality. The previously discussed nanocarriers adaptability permits the administration of remedies via oral, parenteral, pulmonary, transdermal, and thermal routes. Therefore, patient compliance to therapies is enhanced, and the challenges associated with dissatisfying injection procedures are eliminated. In addition to the goal of enhancing diabetes patient quality of life, the contribution of insulin pumps, stem cell therapy, gene therapy, tissue engineering, and artificial pancreas is a proven asset for the control of blood sugar level.
ACKNOWLEDGMENT
The authors expressed their gratitude to Mr. Jitender Joshi, President, Prof. (Dr.) Dharam Buddhi, Vice Chancellor of Uttaranchal University Dehradun, and Prof. (Dr.) Vikash Jakhmola, Director, Uttaranchal Institute of Pharmaceutical Sciences, for their encouragement and guidance in publishing this review work.
FUNDING
None
ABBREVIATIONS
Alpha glucosidase inhibitors: AGIs, Alzheimer's disease: AD, area under curve: AUC, blood glucose level: BGL, carbon nanotubes: CNTs, cardiovascular diseases: CVD, catalase: CAT, chitosan modified dihydromyricetin loaded liposomes: CDL, chitosan nanoparticles: CNPs, coronary heart disease: CHD, cross-linked carboxymethyl chitosan nanoparticles: CMCS NPs, design of experiment: DoE, dihydromyricetin: DHM, dihydromyricetin loaded liposomes: DL, Dipeptidyl Peptidase-4 Inhibitors: DPP4, drug delivery systems: DDS, encapsulation efficiency: EE, epidermal growth factor: EGF, epidermal growth factor receptor: EGFR, Fourier transform infrared spectroscopy: FTIR, functionalised carbon nanotubes: f-CNTs, Gestational diabetes mellitus: GDM, Glucagon-like Peptide 1 Analogue: GLP-1, Glipizide: GPZ, glipizide-loaded nano-micelles: GNM, gymnemic acid: GA, hyodeoxycholic acid: HDCA, Kaempferol: KMP, large unilamellar vesicles: LUV, lauric acid-conjugated-F127: LAF127, lecithin-chitosan: LC, linagliptin: Lina, mean residence time: MRT, Metformin: MET, Metformin hydrochloride loaded transfersomes: MF-TFs, microfluidics: MF, multilamellar vesicles: MLV, multi-walled carbon nanotubes: MWCNTs, Nanofibers: NFs, O-Carboxymethyl chitosan nanoparticles: O-CMC-NPs, phenylboronic ester: PBE, poly(amidoamine): PAMAM, poly d, l-lactic-co-glycolic acid: PLGA, polydatin long circulating liposomes: PLLs, polydispersity index: PDI, polyglycolic acid: PGA, polylactic acid: PLA, Polymeric Nanoparticles: NP, quality by design: QbD, resveratrol: RES, small unilamellar vesicles: SUV, Sodium-Glucose Co-Transporter-2 Inhibitors: SGLT2, Solid lipid Nanoparticles: SLNs, streptozotocin-nicotinamide: STZ-NA, superoxide dismutase: SOD, Tetrahydrocurcumin: THC, Thiazolidinediones: TZDs, type 2 diabetes mellitus: T2DM, type 3 diabetes: T3D, X-ray diffraction: XRD, X-ray photoelectron spectroscopy: XPS.
AUTHORS CONTRIBUTIONS
Writing and data collection by SD and VS; conceptualization, editing and review of manuscript by MB and VJ.
CONFLICT OF INTERESTS
Declared none
REFERENCES
Rajendiran D, Packirisamy S, Gunasekaran K. A review on role of antioxidants in diabetes. Asian J Pharm Clin Res. 2018;11(2):48-53. doi: 10.22159/ajpcr.2018.v11i2.23241.
Cao X, Chen N, LI Y. Editorial: beta cell function and diabetes remission. Front Endocrinol (Lausanne). 2023 Dec 15;14:1298101. doi: 10.3389/fendo.2023.1298101, PMID 38161979.
Dharani B, Suba A, Sebastian S. View of understanding the benefits of stevia rebaudiana bertoni for diabetes: a comprehensive review. Int J Pharm Pharm Sci. 2024;16(11):12-6. doi 10.22159/ijpps.2024v16i11.52382.
Frimpong EK, Thembane N, Hlatshwayo S, Ngcobo M, Gqaleni N. Indigenous medicinal plants used in the management of diabetes in Africa: 5 y (2019-2024) in perspective. Plants (Basel). 2024;13(14):1898. doi: 10.3390/plants13141898, PMID 39065425.
Cho NH, Shaw JE, Karuranga S, Huang Y, DA Rocha Fernandes JD, Ohlrogge AW. IDF diabetes atlas: global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res Clin Pract. 2018;138:271-81. doi: 10.1016/j.diabres.2018.02.023, PMID 29496507.
Kahaly GJ, Hansen MP. Type 1 diabetes associated autoimmunity. Autoimmun Rev. 2016;15(7):644-8. doi: 10.1016/j.autrev.2016.02.017, PMID 26903475.
Brestoff JR, Artis D. Immune regulation of metabolic homeostasis in health and disease. Cell. 2015;161(1):146-60. doi: 10.1016/j.cell.2015.02.022, PMID 25815992.
Donath MY, Shoelson SE. Type 2 diabetes as an inflammatory disease. Nat Rev Immunol. 2011;11(2):98-107. doi: 10.1038/nri2925, PMID 21233852.
Weyer C, Bogardus C, Mott DM, Pratley RE. The natural history of insulin secretory dysfunction and insulin resistance in the pathogenesis of type 2 diabetes mellitus. J Clin Invest. 1999;104(6):787-94. doi: 10.1172/JCI7231, PMID 10491414.
Yadav NK, Mazumder R, Rani A, Kumar A. Current perspectives on using nanoparticles for diabetes management. Int J Appl Pharm. 2024;16(5):38-45. doi: 10.22159/ijap.2024v16i5.51084.
Banday MZ, Sameer AS, Nissar S. Pathophysiology of diabetes: an overview. Avicenna J Med. 2020;10(4):174-88. doi: 10.4103/ajm.ajm_53_20, PMID 33437689.
McIntyre HD, Catalano P, Zhang C, Desoye G, Mathiesen ER, Damm P. Gestational diabetes mellitus. Nat Rev Dis Primers. 2019;5(1):47. doi: 10.1038/s41572-019-0098-8, PMID 31296866.
Liu P, Zhang Z, Cai Y, LI Z, Zhou Q, Chen Q. Ferroptosis: mechanisms and role in diabetes mellitus and its complications. Ageing Res Rev. 2024 Feb;94:102201. doi: 10.1016/j.arr.2024.102201, PMID 38242213.
Hirai FE, Tielsch JM, Klein BE, Klein R. Ten year change in vision related quality of life in type 1 diabetes: wisconsin epidemiologic study of diabetic retinopathy. Ophthalmology. 2011;118(2):353-8. doi: 10.1016/j.ophtha.2010.06.022, PMID 20884058.
Ambiya V, Kumar A, Baranwal VK, Kapoor G, Arora A, Kalra N. Change in subfoveal choroidal thickness in diabetes and in various grades of diabetic retinopathy. Int J Retina Vitreous. 2018;4(1):34. doi: 10.1186/s40942-018-0136-9, PMID 30214825.
Sarwar N, Aspelund T, Eiriksdottir G, Gobin R, Seshasai SR, Forouhi NG. Markers of dysglycaemia and risk of coronary heart disease in people without diabetes: reykjavik prospective study and systematic review. Plos Med. 2010;7(5):e1000278. doi: 10.1371/journal.pmed.1000278, PMID 20520805.
Jamnitski A, Symmons D, Peters MJ, Sattar N, McInnes I, Nurmohamed MT. Cardiovascular comorbidities in patients with psoriatic arthritis: a systematic review. Ann Rheum Dis. 2013;72(2):211-6. doi: 10.1136/annrheumdis-2011-201194, PMID 22532629.
Sekiou O, Boumendjel M, Taibi F, Tichati L, Boumendjel A, Messarah M. Nephroprotective effect of Artemisia herba alba aqueous extract in alloxan-induced diabetic rats. J Tradit Complement Med. 2021;11(1):53-61. doi: 10.1016/j.jtcme.2020.01.001, PMID 33511062.
Sivakumar S, Palsamy P, Subramanian SP. Impact of d-pinitol on the attenuation of proinflammatory cytokines hyperglycemia mediated oxidative stress and protection of kidney tissue ultrastructure in streptozotocin induced diabetic rats. Chem Biol Interact. 2010;188(1):237-45. doi: 10.1016/j.cbi.2010.07.014, PMID 20643114.
Ojiako OA, Chikezie PC, Ogbuji AC. Histopathological studies of renal and hepatic tissues of hyperglycemic rats administered with traditional herbal formulations. Int J Green Pharm. 2015;9(3):184-91. doi: 10.4103/0973-8258.161237.
Obrosova IG. Diabetic painful and insensate neuropathy: pathogenesis and potential treatments. Neurotherapeutics. 2009;6(4):638-47. doi: 10.1016/j.nurt.2009.07.004, PMID 19789069.
Wessels AM, Rombouts SA, Simsek S, Kuijer JP, Kostense PJ, Barkhof F. Microvascular disease in type 1 diabetes alters brain activation: a functional magnetic resonance imaging study. Diabetes. 2006;55(2):334-40. doi: 10.2337/diabetes.55.02.06.db05-0680, PMID 16443765.
Padhi S, Nayak AK, Behera A. Type II diabetes mellitus: a review on recent drug based therapeutics. Biomed Pharmacother. 2020 Nov;131:110708. doi: 10.1016/j.biopha.2020.110708, PMID 32927252.
Simos YV, Spyrou K, Patila M, Karouta N, Stamatis H, Gournis D. Trends of nanotechnology in type 2 diabetes mellitus treatment. Asian J Pharm Sci. 2021;16(1):62-76. doi: 10.1016/j.ajps.2020.05.001, PMID 33613730.
Lin SH, Cheng PC, TU ST, Hsu SR, Cheng YC, Liu YH. Effect of metformin monotherapy on serum lipid profile in statin naive individuals with newly diagnosed type 2 diabetes mellitus: a cohort study. Peer J. 2018 Apr 12;6:e4578. doi: 10.7717/peerj.4578, PMID 29666753.
Shurrab NT, Arafa ES. Metformin: a review of its therapeutic efficacy and adverse effects. Obes Med. 2020 Mar;17:100186. doi: 10.1016/j.obmed.2020.100186.
Chen Y, Shan X, Luo C, HE Z. Emerging nanoparticulate drug delivery systems of metformin. J Pharm Investig. 2020;50(3):219-30. doi: 10.1007/s40005-020-00480-1.
Bourron O, Daval M, Hainault I, Hajduch E, Servant JM, Gautier JF. Biguanides and thiazolidinediones inhibit stimulated lipolysis in human adipocytes through activation of AMP activated protein kinase. Diabetologia. 2010;53(4):768-78. doi: 10.1007/s00125-009-1639-6, PMID 20043143.
Turner R. Effect of intensive blood glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). Lancet. 1998;352(9131):854-65. doi: 10.1016/S0140-6736(98)07037-8.
Wehmeier UF, Piepersberg W. Biotechnology and molecular biology of the α-glucosidase inhibitor acarbose. Appl Microbiol Biotechnol. 2004;63(6):613-25. doi: 10.1007/s00253-003-1477-2, PMID 14669056.
Narita T, Yokoyama H, Yamashita R, Sato T, Hosoba M, Morii T. Comparisons of the effects of 12 w administration of miglitol and voglibose on the responses of plasma incretins after a mixed meal in Japanese type 2 diabetic patients. Diabetes Obes Metab. 2012;14(3):283-7. doi: 10.1111/j.1463-1326.2011.01526.x, PMID 22051162.
Derosa G, Maffioli P. α-glucosidase inhibitors and their use in clinical practice. Arch Med Sci. 2012;8(5):899-906. doi: 10.5114/aoms.2012.31621, PMID 23185202.
Holt RI, Lambert KD. The use of oral hypoglycaemic agents in pregnancy. Diabet Med. 2014;31(3):282-91. doi: 10.1111/dme.12376, PMID 24528229.
Feingold KR. Oral and injectable (non-insulin) pharmacological agents for the treatment of type 2 diabetes. Endotext; 2024.
Thule PM, Umpierrez G. Sulfonylureas: a new look at old therapy topical collection on pharmacologic treatment of type 2 diabetes. Curr Diab Rep. 2014;14(4):1-8.
Sola D, Rossi L, Schianca GP, Maffioli P, Bigliocca M, Mella R. Sulfonylureas and their use in clinical practice. Arch Med Sci. 2015;11(4):840-8. doi: 10.5114/aoms.2015.53304, PMID 26322096.
Roumie CL, Hung AM, Greevy RA, Grijalva CG, Liu X, Murff HJ. Comparative effectiveness of sulfonylurea and metformin monotherapy on cardiovascular events in type 2 diabetes mellitus: a cohort study. Ann Intern Med. 2012;157(9):601-10. doi: 10.7326/0003-4819-157-9-201211060-00003, PMID 23128859.
Chaudhury A, Duvoor C, Reddy Dendi VS, Kraleti S, Chada A, Ravilla R. Clinical review of antidiabetic drugs: implications for type 2 diabetes mellitus management. Front Endocrinol. 2017;8:6. doi: 10.3389/fendo.2017.00006, PMID 28167928.
Eldor R, DE Fronzo RA, Abdul Ghani M. In vivo Actions of peroxisome proliferator activated receptors: glycemic control insulin sensitivity and insulin secretion. Diabetes Care. 2013;36 Suppl 2:S162-74. doi: 10.2337/dcS13-2003, PMID 23882042.
Defronzo RA, Tripathy D, Schwenke DC, Banerji MA, Bray GA, Buchanan TA. Prevention of diabetes with pioglitazone in ACT NOW: physiologic correlates. Diabetes. 2013;62(11):3920-6. doi: 10.2337/db13-0265, PMID 23863810.
Gastaldelli A, Ferrannini E, Miyazaki Y, Matsuda M, Mari A, DE Fronzo RA. Thiazolidinediones improve β-cell function in type 2 diabetic patients. Am J Physiol Endocrinol Metab. 2007;292(3):E871-83. doi: 10.1152/ajpendo.00551.2006, PMID 17106061.
Adeghate E, Kalasz H. Amylin analogues in the treatment of diabetes mellitus: medicinal chemistry and structural basis of its function. Open Med Chem J. 2011;5 Suppl 2:78-81. doi: 10.2174/1874104501105010078, PMID 21966328.
Schmitz O, Brock B, Rungby J. Amylin agonists: a novel approach in the treatment of diabetes. Diabetes. 2004;53 Suppl 3:S233-8. doi: 10.2337/diabetes.53.suppl_3.s233, PMID 15561917.
Hoogwerf BJ, Doshi KB, Diab D. Pramlintide the synthetic analogue of amylin: physiology pathophysiology and effects on glycemic control body weight and selected biomarkers of vascular risk. Vasc Health Risk Manag. 2008;4(2):355-62. doi: 10.2147/vhrm.s1978, PMID 18561511.
Bunck MC, Corner A, Eliasson B, Heine RJ, Shaginian RM, Taskinen MR. Effects of exenatide on measures of β-cell function after 3 y in metformin treated patients with type 2 diabetes. Diabetes Care. 2011;34(9):2041-7. doi: 10.2337/dc11-0291, PMID 21868779.
Klonoff DC, Buse JB, Nielsen LL, Guan X, Bowlus CL, Holcombe JH. Exenatide effects on diabetes obesity cardiovascular risk factors and hepatic biomarkers in patients with type 2 diabetes treated for at least 3 years. Curr Med Res Opin. 2008;24(1):275-86. doi: 10.1185/030079908x253870, PMID 18053320.
Kalra S. Sodium glucose Co-Transporter-2 (SGLT2) inhibitors: a review of their basic and clinical pharmacology. Diabetes Ther. 2014;5(2):355-66. doi: 10.1007/s13300-014-0089-4, PMID 25424969.
Cherney DZ, Perkins BA, Soleymanlou N, Maione M, Lai V, Lee A. Renal hemodynamic effect of sodium glucose cotransporter 2 inhibition in patients with type 1 diabetes mellitus. Circulation. 2014;129(5):587-97. doi: 10.1161/circulationaha.113.005081, PMID 24334175.
Rai VK, Mishra N, Agrawal AK, Jain S, Yadav NP. Novel drug delivery system: an immense hope for diabetics. Drug Deliv. 2016;23(7):2371-90. doi: 10.3109/10717544.2014.991001, PMID 25544604.
Wang J, HU S, Mao W, Xiang J, Zhou Z, Liu X. Assemblies of peptide cytotoxin conjugates for tumor homing chemotherapy. Adv Funct Materials. 2019;29(7):1807446. doi: 10.1002/adfm.201807446.
Mudshinge SR, Deore AB, Patil S, Bhalgat CM. Nanoparticles: emerging carriers for drug delivery. Saudi Pharm J. 2011;19(3):129-41. doi: 10.1016/j.jsps.2011.04.001, PMID 23960751.
Souto EB, Souto SB, Campos JR, Severino P, Pashirova TN, Zakharova LY. Nanoparticle delivery systems in the treatment of diabetes complications. Molecules. 2019;24(23):4209. doi: 10.3390/molecules24234209, PMID 31756981.
Hadiya S, Radwan R, Zakaria M, El Sherif T, Hamad MA, Elsabahy M. Nanoparticles integrating natural and synthetic polymers for in vivo insulin delivery. Pharm Dev Technol. 2021;26(1):30-40. doi: 10.1080/10837450.2020.1832117, PMID 33019826.
Abbas A, Eissa NG, El Bassossy HM, Ghorab MM, El Nahas HM. Polymeric linagliptin nanoparticles as a sustained release treatment for type 2 diabetes. J Drug Deliv Sci Technol. 2024 Mar;93:105438. doi: 10.1016/j.jddst.2024.105438.
Lari AS, Zahedi P, Ghourchian H, Khatibi A. Microfluidic based synthesized carboxymethyl chitosan nanoparticles containing metformin for diabetes therapy: in vitro and in vivo assessments. Carbohydr Polym. 2021 Jun 1;261:117889. doi: 10.1016/j.carbpol.2021.117889, PMID 33766375.
Sharma DK, Pattnaik G, Behera A. Development and in vitro-in vivo evaluation of pioglitazone loaded polymeric nanoparticles using central composite design surface response methodology. Open Nano. 2023 May;11:100141. doi: 10.1016/j.onano.2023.100141.
Ribeiro MC, Correa VL, DA Silva FK, Casas AA, Chagas A DE l das, Oliveira LP DE, Miguel MP, Diniz DGA, Amaral AC, Menezes LBD. Wound healing treatment using insulin within polymeric nanoparticles in the diabetes animal model. European Journal of Pharmaceutical Sciences. 2020 Jul 1;150:105330.
El Dakroury WA, Zewail MB, Amin MM. Design optimization and in vivo performance of glipizide loaded O-carboxymethyl chitosan nanoparticles in insulin resistant/type 2 diabetic rat model. J Drug Deliv Sci Technol. 2023;79:104040. doi: 10.1016/j.jddst.2022.104040.
Bulboaca AE, Boarescu PM, Bolboaca SD, Blidaru M, Festila D, Dogaru G. Comparative effect of curcumin versus liposomal curcumin on systemic pro-inflammatory cytokines profile MCP-1 and RANTES in experimental diabetes mellitus. Int J Nanomedicine. 2019 Nov 18;14:8961-72. doi: 10.2147/IJN.S226790, PMID 31819412.
Amjadi S, Mesgari Abbasi M, Shokouhi B, Ghorbani M, Hamishehkar H. Enhancement of therapeutic efficacy of betanin for diabetes treatment by liposomal nanocarriers. J Funct Foods. 2019 Aug;59:119-28. doi: 10.1016/j.jff.2019.05.015.
Ding Q, Liu X, Zhang S, Chai G, MA S, Sun S. Chitosan modified dihydromyricetin liposomes promote the repair of liver injury in mice suffering from diabetes mellitus. Int J Biol Macromol. 2024;273(2):133040. doi: 10.1016/j.ijbiomac.2024.133040, PMID 38857721.
HU M, Gou T, Chen Y, XU M, Chen R, Zhou T. A novel drug delivery system: hyodeoxycholic acid modified metformin liposomes for type 2 diabetes treatment. Molecules. 2023;28(6):2471. doi: 10.3390/molecules28062471, PMID 36985444.
Zhang K, Chen J, Raza F, Zafar H, XU Y, LI R. Advancing diabetes treatment: novel formulation of polydatin long circulating liposomes and their glucose-regulating impact on hyperlipidemia induced type 2 diabetic mice. Mater Adv. 2024;5(16):6516-34. doi: 10.1039/D4MA00020J.
Sarhadi S, Moosavian SA, Mashreghi M, Rahiman N, Golmohamadzadeh S, Tafaghodi M. B12-functionalized pegylated liposomes for the oral delivery of insulin: in vitro and in vivo studies. J Drug Deliv Sci Technol. 2022 Mar;69:103141. doi: 10.1016/j.jddst.2022.103141.
Samed N, Sharma V, Sundaramurthy A. Hydrogen bonded niosomes for encapsulation and release of hydrophilic and hydrophobic anti-diabetic drugs: an efficient system for oral anti-diabetic formulation. Appl Surf Sci. 2018 Aug 15;449:567-73. doi: 10.1016/j.apsusc.2017.11.055.
Hasan AA, Madkor H, Wageh S. Formulation and evaluation of metformin hydrochloride loaded niosomes as controlled release drug delivery system. Drug Deliv. 2013;20(3-4):120-6. doi: 10.3109/10717544.2013.779332, PMID 23651102.
Tyagi R, Waheed A, Kumar N, Ahad A, Bin Jardan YA, Mujeeb M. Formulation and evaluation of plumbagin loaded niosomes for an antidiabetic study: optimization and in vitro evaluation. Pharmaceuticals (Basel). 2023;16(8):1169. doi: 10.3390/ph16081169, PMID 37631084.
Mohsen AM, Abou Samra MM, ElShebiney SA. Enhanced oral bioavailability and sustained delivery of glimepiride via niosomal encapsulation: in vitro characterization and in vivo evaluation. Drug Dev Ind Pharm. 2017;43(8):1254-64. doi: 10.1080/03639045.2017.1310224, PMID 28330377.
Singhal T, Mujeeb M, Ahad A, Aqil M, Rahman SO, Najmi AK. Preparation optimization and biological evaluation of gymnemic acid loaded niosomes against streptozotocin nicotinamide induced diabetic nephropathy in wistar rats. J Drug Deliv Sci Technol. 2019 Dec;54:101328. doi: 10.1016/j.jddst.2019.101328.
Alam MS, Ahad A, Abidin L, Aqil M, Mir SR, Mujeeb M. Embelin loaded oral niosomes ameliorate streptozotocin induced diabetes in wistar rats. Biomed Pharmacother. 2018;97:1514-20. doi: 10.1016/j.biopha.2017.11.073, PMID 29793314.
Zhang D, Huang Q. Encapsulation of astragaloside with matrix metalloproteinase-2-responsive hyaluronic acid end-conjugated polyamidoamine dendrimers improves wound healing in diabetes. J Biomed Nanotechnol. 2020;16(8):1229-40. doi: 10.1166/jbn.2020.2971, PMID 33397553.
Labieniec Watala M, Przygodzki T, Sebekova K, Watala C. Can metabolic impairments in experimental diabetes be cured with poly (amido) amine (PAMAM) G4 dendrimers? In the search for minimizing of the adverse effects of PAMAM administration. Int J Pharm. 2014;464(1-2):152-67. doi: 10.1016/j.ijpharm.2014.01.011, PMID 24463003.
Kwon MJ, AN S, Choi S, Nam K, Jung HS, Yoon CS. Effective healing of diabetic skin wounds by using nonviral gene therapy based on minicircle vascular endothelial growth factor DNA and a cationic dendrimer. J Gene Med. 2012;14(4):272-8. doi: 10.1002/jgm.2618, PMID 22407991.
Akhtar S, Chandrasekhar B, Yousif MH, Renno W, Benter IF, El Hashim AZ. Chronic administration of nano sized pamam dendrimers in vivo inhibits EGFR-ERK1/2-ROCK signaling pathway and attenuates diabetes induced vascular remodeling and dysfunction. Nanomedicine. 2019 Jun;18:78-89. doi: 10.1016/j.nano.2019.02.012, PMID 30844576.
Kassem AA, Abd El Alim SH, Basha M, Salama A. Phospholipid complex enriched micelles: a novel drug delivery approach for promoting the antidiabetic effect of repaglinide. Eur J Pharm Sci. 2017 Mar 1;99:75-84. doi: 10.1016/j.ejps.2016.12.005, PMID 27998799.
Liu X, LI C, LV J, Huang F, AN Y, Shi L. Glucose and H2O2 dual responsive polymeric micelles for the self regulated release of insulin. ACS Appl Bio Mater. 2020;3(3):1598-606. doi: 10.1021/acsabm.9b01185, PMID 35021650.
Zhu J, Jiang G, Hong W, Zhang Y, XU B, Song G. Rapid gelation of oxidized hyaluronic acid and succinyl chitosan for integration with insulin loaded micelles and epidermal growth factor on diabetic wound healing. Mater Sci Eng C Mater Biol Appl. 2020 Dec;117:111273. doi: 10.1016/j.msec.2020.111273, PMID 32919637.
Bahman F, Taurin S, Altayeb D, Taha S, Bakhiet M, Greish K. Oral insulin delivery using poly (styrene Co-maleic acid) micelles in a diabetic mouse model. Pharmaceutics. 2020;12(11):1026. doi: 10.3390/pharmaceutics12111026, PMID 33120872.
Kassem AA, Abd El Alim SH, Basha M, Salama A. Phospholipid complex enriched micelles: a novel drug delivery approach for promoting the antidiabetic effect of repaglinide. Eur J Pharm Sci. 2017 Mar 1;99:75-84. doi: 10.1016/j.ejps.2016.12.005, PMID 27998799.
Kumar V, Poonia N, Kumar P, Kumar Verma P, Alshammari A, Albekairi NA. Amphiphilic lauric acid coupled pluronic based nano micellar system for efficient glipizide delivery. Saudi Pharm J. 2024;32(5):102046. doi: 10.1016/j.jsps.2024.102046, PMID 38577487.References
Singh J, Mittal P, Vasant Bonde G, Ajmal G, Mishra B. Design optimization characterization and in vivo evaluation of quercetin enveloped Soluplus®/P407 micelles in diabetes treatment. Artif Cells Nanomed Biotechnol. 2018;46 Suppl 3:S546-55. doi: 10.1080/21691401.2018.1501379, PMID 30322273.
Welengodage I, Katuwavila NP. Recent advancements in lipid nanoparticle technology for oral insulin delivery. University of Colombo Review. 2024;5(1):130-44. doi: 10.4038/ucr.v5i1.134.
Wang M, Wang C, Ren S, Pan J, Wang Y, Shen Y. Versatile oral insulin delivery nanosystems: from materials to nanostructures. Int J Mol Sci. 2022;23(6):3362. doi: 10.3390/ijms23063362, PMID 35328783.
Bharti Sharma J, Bhatt S, Tiwari A, Tiwari V, Kumar M, Verma R. Statistical optimization of tetrahydrocurcumin loaded solid lipid nanoparticles using box behnken design in the management of streptozotocin induced diabetes mellitus. Saudi Pharm J. 2023;31(9):101727. doi: 10.1016/j.jsps.2023.101727, PMID 37638219.
Anchan RB, Koland M. Oral insulin delivery by chitosan coated solid lipid nanoparticles: ex vivo and in vivo studies. J Young Pharm. 2021;13(1):43-8. doi: 10.5530/jyp.2021.13.10.
Mohseni R, Arab Sadeghabadi Z, Ziamajidi N, Abbasalipourkabir R, Rezaei Farimani A. Oral administration of resveratrol loaded solid lipid nanoparticle improves insulin resistance through targeting expression of SNARE proteins in adipose and muscle tissue in rats with type 2 diabetes. Nanoscale Res Lett. 2019;14(1):227. doi: 10.1186/s11671-019-3042-7, PMID 31290033.
Oroojan AA, Ahangarpour A, Paknejad B, Zareian P, Hami Z, Abtahi SR. Effects of myricitrin and solid lipid nanoparticle containing myricitrin on reproductive system disorders induced by diabetes in male mouse. World J Mens Health. 2021;39(1):147-57. doi: 10.5534/wjmh.190010, PMID 32009314.
Shah P, Chavda K, Vyas B, Patel S. Formulation development of linagliptin solid lipid nanoparticles for oral bioavailability enhancement: role of P-gp inhibition. Drug Deliv Transl Res. 2021;11(3):1166-85. doi: 10.1007/s13346-020-00839-9, PMID 32804301.
Fernandez Garcia R, Lalatsa A, Statts L, Bolas Fernandez F, Ballesteros MP, Serrano DR. Transferosomes as nanocarriers for drugs across the skin: quality by design from lab to industrial scale. Int J Pharm. 2020;573:118817. doi: 10.1016/j.ijpharm.2019.118817, PMID 31678520.
Duangjit S, Opanasopit P, Rojanarata T, Ngawhirunpat T. Characterization and in vitro skin permeation of meloxicam loaded liposomes versus transfersomes. J Drug Deliv. 2011;2011(1):418316. doi: 10.1155/2011/418316, PMID 21490750.
Mazhar D, Haq NU, Zeeshan M, Ain QU, Ali H, Khan S. Preparation characterization and pharmacokinetic assessment of metformin HCl loaded transfersomes co-equipped with permeation enhancer to improve drug bioavailability via transdermal route. J Drug Deliv Sci Technol. 2023;84:104448. doi: 10.1016/j.jddst.2023.104448.
Hussain M, Hafeez A, Kushwaha SP. Nanoformulation mediated transdermal delivery of anti-diabetic drugs: an updated review. Intell Pharm. 2023;1(4):192-200. doi: 10.1016/j.ipha.2023.08.009.
Abdallah MH, Abu Lila AS, Shawky SM, Almansour K, Alshammari F, Khafagy ES. Experimental design and optimization of nano transfersomal gel to enhance the hypoglycemic activity of silymarin. Polymers (Basel). 2022;14(3):508. doi: 10.3390/polym14030508, PMID 35160498.
Ramkanth S, Anitha P, Gayathri R, Mohan S, Babu D. Formulation and design optimization of nano transferosomes using pioglitazone and eprosartan mesylate for concomitant therapy against diabetes and hypertension. Eur J Pharm Sci. 2021;162:105811. doi: 10.1016/j.ejps.2021.105811, PMID 33757828.
Malakar J, Sen SO, Nayak AK, Sen KK. Formulation optimization and evaluation of transferosomal gel for transdermal insulin delivery. Saudi Pharm J. 2012;20(4):355-63. doi: 10.1016/j.jsps.2012.02.001, PMID 23960810.
Hussain M, Hafeez A, Kushwaha SP. Nanoformulation mediated transdermal delivery of anti-diabetic drugs: an updated review. Intell Pharm. 2023;1(4):192-200. doi: 10.1016/j.ipha.2023.08.009.
Aouta E, Patra CN. Design optimization and characterization of combined ethosomal transdermal patch of glimepiride and duloxetine drug regimen for diabetes and associated neuropathic pain management. Curr Drug Ther. 2022;17(5):359-68. doi: 10.2174/1574885517666220525122859.
Raghav SS, Kumar B, Sethiya NK, Singhal M, Alhowyan A, Kalam MA. Potential of hybrid lecithin chitosan kaempferol ethosomes for the treatment of diabetic foot ulcer: in vitro and in vivo investigation. J Drug Deliv Sci Technol. 2024 Sep;98:105927. doi: 10.1016/j.jddst.2024.105927.
Bodade SS, Shaikh KS, Kamble MS, Chaudhari PD. A study on ethosomes as mode for transdermal delivery of an antidiabetic drug. Drug Deliv. 2013;20(1):40-6. doi: 10.3109/10717544.2012.752420, PMID 23311652.
Fathima H, Srilatha KS, Geethalakshmi A, Sequeira C. Formulation and evaluation of vildagliptin ethosomal gel for diabetes mellitus. Arch Pharm Sci Res. 2023;13:150-6.
Maleki H, Khoshnevisan K, Sajjadi Jazi SM, Baharifar H, Doostan M, Khoshnevisan N. Nanofiber based systems intended for diabetes. J Nanobiotechnology. 2021;19(1):317. doi: 10.1186/s12951-021-01065-2, PMID 34641920.
Song J, Kim M, Lee H. Recent advances on nanofiber fabrications: unconventional state of the art spinning techniques. Polymers. 2020;12(6):1386. doi: 10.3390/polym12061386, PMID 32575746.
Lee H, Kim IS. Nanofibers: emerging progress on fabrication using mechanical force and recent applications. Polym Rev. 2018;58(4):688-716. doi: 10.1080/15583724.2018.1495650.
Panda BP, Wei MX, Kumar N, Shivashekaregowda H, Patnaik S. Design fabrication and characterization of PVA/PLGA electrospun nanofibers carriers for improvement of drug delivery of gliclazide in type-2 diabetes. Proceedings. 2020;78(1):14.
Alamer AA, Alsaleh NB, Aodah AH, Alshehri AA, Almughem FA, Alqahtani SH. Development of imeglimin electrospun nanofibers as a potential buccal antidiabetic therapeutic approach. Pharmaceutics. 2023;15(4):1208. doi: 10.3390/pharmaceutics15041208, PMID 37111693.
Shoukat R, Khan MI. Carbon nanotubes: a review on properties synthesis methods and applications in micro and nanotechnology. Microsyst Technol. 2021;27(12):4183-92. doi: 10.1007/s00542-021-05211-6.
Zaman S, Hussain S, Butt FK, Jianguo X, Zhu C. Functionalization of carbon nanotubes by a facile chemical method and its application in anti-diabetic activity. J Nanosci Nanotechnol. 2017;17(11):8557-61. doi: 10.1166/jnn.2017.15178.
Peters CJ, Hindmarsh PC, Thompson RJ. Insulin pump therapy. Paediatr Child Health. 2017 Apr 1;27(4):160-5. doi: 10.1016/j.paed.2017.02.002.
Peters CJ, Hindmarsh PC, Thompson RJ. Insulin pump therapy. Paediatr Child Health. 2017;27(4):160-5. doi: 10.1016/j.paed.2017.02.002.
Nimri R, Nir J, Phillip M. Insulin pump therapy. Am J Ther. 2020;27(1):e30-41. doi: 10.1097/MJT.0000000000001097, PMID 31833871.
Mishra V, Nayak P, Sharma M, Albutti A, Alwashmi AS, Aljasir MA. Emerging treatment strategies for diabetes mellitus and associated complications: an update. Pharmaceutics. 2021;13(10):1568. doi: 10.3390/pharmaceutics13101568, PMID 34683861.
Buchwald H, Rohde TD, Dorman FD, Skakoon JG, Wigness BD, Prosl FR. A totally implantable drug infusion defice: laboratory and clinical experience using a model with single flow rate and new design for modulated insulin infusion. Diabetes Care. 1980;3(2):351-8. doi: 10.2337/diacare.3.2.351, PMID 6993152.
Gin H, Renard E, Melki V, Boivin S, Schaepelynck Belicar P, Guerci B. Combined improvements in implantable pump technology and insulin stability allow safe and effective long term intraperitoneal insulin delivery in type 1 diabetic patients: the EVADIAC experience. Diabetes Metab. 2003;29(6):602-7. doi: 10.1016/s1262-3636(07)70075-7, PMID 14707889.
Duckworth WC, Saudek CD, Henry RR. Why intraperitoneal delivery of insulin with implantable pumps in NIDDM? Diabetes. 1992;41(6):657-61. doi: 10.2337/diab.41.6.657, PMID 1587393.
Brouwer E, Knol R, Vernooij AS, Van Den Akker T, Vlasman PE, Klumper FJ. Physiological based cord clamping in preterm infants using a new purpose built resuscitation table: a feasibility study. Arch Dis Child Fetal Neonatal Ed. 2019;104(4):F396-402. doi: 10.1136/archdischild-2018-315483, PMID 30282674.
Miller KM, Foster NC, Beck RW, Bergenstal RM, DuBose SN, DI meglio LA. Current state of type 1 diabetes treatment in the U.S: updated data from the T1D exchange clinic registry. Diabetes Care. 2015;38(6):971-8. doi: 10.2337/dc15-0078, PMID 25998289.
Blackman SM, Raghinaru D, Adi S, Simmons JH, Ebner Lyon L, Chase HP. Insulin pump use in young children in the T1D exchange clinic registry is associated with lower hemoglobin A1c levels than injection therapy. Pediatr Diabetes. 2014;15(8):564-72. doi: 10.1111/pedi.12121, PMID 24494980.
Willi SM, Miller KM, DI Meglio LA, Klingensmith GJ, Simmons JH, Tamborlane WV. Racial ethnic disparities in management and outcomes among children with type 1 diabetes. Pediatrics. 2015;135(3):424-34. doi: 10.1542/peds.2014-1774, PMID 25687140.
Maikawa CL, Chen PC, Vuong ET, Nguyen LT, Mann JL, D aquino AI. Ultra fast insulin pramlintide co-formulation for improved glucose management in diabetic rats. Adv Sci (Weinh). 2021;8(21):e2101575. doi: 10.1002/advs.202101575, PMID 34499434.
Nawaz MS, Shah KU, Khan TM, Rehman AU, Rashid HU, Mahmood S. Evaluation of current trends and recent development in insulin therapy for management of diabetes mellitus. Diabetes Metab Syndr. 2017;11 Suppl 2:S833-9. doi: 10.1016/j.dsx.2017.07.003, PMID 28709853.
Marin Garaundo E, Torre Beteta RL, Munive Degregori A, Alvitez J, Barja Ore J, Mayta Tovalino F. Use of artificial pancreas in the management of diabetes mellitus: a bibliometric study. Saudi J Med Med Sci. 2023;11(4):332-8. doi: 10.4103/sjmms.sjmms_12_23, PMID 37970460.
Breton MD, Kanapka LG, Beck RW, Ekhlaspour L, Forlenza GP, Cengiz E. A randomized trial of closed loop control in children with type 1 diabetes. N Engl J Med. 2020 Aug;383(9):836-45. doi: 10.1056/NEJMoa2004736, PMID 32846062.
DE Jongh D, Bunnik E, Ozcan B, Zietse R, Massey E, Vanguard Consortium. The bio-artificial pancreas to treat type 1 diabetes: perspectives from healthcare professionals in the netherlands. J Clin Transl Endocrinol. 2024;38:100372. doi: 10.1016/j.jcte.2024.100372, PMID 39502713.
Romer AI, Sussel L. Pancreatic islet cell development and regeneration. Curr Opin Endocrinol Diabetes Obes. 2015;22(4):255-64. doi: 10.1097/MED.0000000000000174, PMID 26087337.
Farhadieh RD, Bulstrode N, Cugno S. Plastic and reconstructive surgery: Approaches and techniques; 2015.
DE Bueno M, LGB. 3D modeling for comparison of surgically treated intracranial arachnoid cysts in children. Biomed J Sci Tech Res. 2018;10(3):1.
Hopt UT, Drognitz O. Pancreas organ transplantation. Short and long term results in terms of diabetes control. Langenbecks Arch Surg. 2000;385(6):379-89. doi: 10.1007/s004230000124, PMID 11127521.
Hasse JM. Nutrition assessment and support of organ transplant recipients. JPEN J Parenter Enteral Nutr. 2001;25(3):120-31. doi: 10.1177/0148607101025003120, PMID 11334061.
Snydman DR. Infection in solid organ transplantation. Transpl Infect Dis. 1999;1(1):21-8. doi: 10.1034/j.1399-3062.1999.10103.x, PMID 11428968.
Kotton CN, Kumar D, Caliendo AM, Asberg A, Chou S, Snydman DR. International consensus guidelines on the management of cytomegalovirus in solid organ transplantation. Transplantation. 2010;89(7):779-95. doi: 10.1097/TP.0b013e3181cee42f, PMID 20224515.
Paya CV. Fungal infections in solid organ transplantation. Clin Infect Dis. 1993;16(5):677-88. doi: 10.1093/clind/16.5.677, PMID 8507760.
Kelly C, Parke HG, McCluskey JT, Flatt PR, MC Clenaghan NH. The role of glucagon and somatostatin secreting cells in the regulation of insulin release and beta cell function in heterotypic pseudoislets. Diabetes Metab Res Rev. 2010;26(7):525-33. doi: 10.1002/dmrr.1111, PMID 20718077.
Bernard AB, Lin CC, Anseth KS. A microwell cell culture platform for the aggregation of pancreatic β-cells. Tissue Eng Part C Methods. 2012;18(8):583-92. doi: 10.1089/ten.TEC.2011.0504, PMID 22320435.
Dunbar CE, High KA, Joung JK, Kohn DB, Ozawa K, Sadelain M. Gene therapy comes of age. Science. 2018;359(6372):eaan4672. doi: 10.1126/science.aan4672, PMID 29326244.
XU R, LI H, Lai Yin T, Hsiang FU K, LU H, Lam KS. Diabetes gene therapy: potential and challenges. Curr Gene Ther. 2003;3(1):65-82. doi: 10.2174/1566523033347444.
Wong MS, Hawthorne WJ, Manolios N. Gene therapy in diabetes. Self Nonself. 2010;1(3):165-75. doi: 10.4161/self.1.3.12643, PMID 21487475.
Tuduri E, Bruin JE, Kieffer TJ. Restoring insulin production for type 1 diabetes. J Diabetes. 2012;4(4):319-31. doi: 10.1111/j.1753-0407.2012.00196.x, PMID 22429761.
Jaen ML, Vila L, Elias I, Jimenez V, Rodo J, Maggioni L. Long term efficacy and safety of insulin and glucokinase gene therapy for diabetes: 8 y follow up in dogs. Mol Ther Methods Clin Dev. 2017 Apr 5;6:1-7. doi: 10.1016/j.omtm.2017.03.008, PMID 28626777.
Torchilin VP. Recent approaches to intracellular delivery of drugs and DNA and organelle targeting. Annu Rev Biomed Eng. 2006;8:343-75. doi: 10.1146/annurev.bioeng.8.061505.095735, PMID 16834560.
Mali S. Delivery systems for gene therapy. Indian J Hum Genet. 2013 Jan;19(1):3-8. doi: 10.4103/0971-6866.112870, PMID 23901186.
Kaufmann KB, Buning H, Galy A, Schambach A, Grez M. Gene therapy on the move. EMBO Mol Med. 2013;5(11):1642-61. doi: 10.1002/emmm.201202287, PMID 24106209.
Yamaoka T. Gene therapy for diabetes mellitus. Curr Mol Med. 2001;1(3):325-37. doi: 10.2174/1566524013363717, PMID 11899081.
Bevacqua RJ, Dai X, Lam JY, GU X, Friedlander MS, Tellez K. CRISPR based genome editing in primary human pancreatic islet cells. Nat Commun. 2021;12(1):2397. doi: 10.1038/s41467-021-22651-w, PMID 33893274.
Wang CH, Lundh M, FU A, Kriszt R, Huang TL, Lynes MD. CRISPR engineered human brown like adipocytes prevent diet induced obesity and ameliorate metabolic syndrome in mice. Sci Transl Med. 2020;12(558):eaaz8664. doi: 10.1126/scitranslmed.aaz8664, PMID 32848096.
RGX-314 Gene therapy administered in the suprachoroidal space for participants with diabetic retinopathy without center involved diabetic macular edema (ci-dme). AbbVie. NCT04567550; 2023.
Extension of phase 3 gene therapy for painful diabetic neuropathy. Helixmith Co, Ltd. NCT04055090; 2023.
P53 gene therapy in treatment of diabetes concurrent with hepatocellular carcinoma. Shenzhen SI Biono GeneTech Co. Ltd. NCT02561546; 2015.
Gene therapy for painful diabetic neuropathy. Helixmith Co, Ltd. NCT01002235; 2019.
Phase. 3 Gene therapy for painful diabetic neuropathy. Helixmith Co, Ltd. NCT02427464; 2022.
Gene therapy to improve wound healing in patients with diabetes. Tissue repair company. NCT00065663; 2007.
VEGF Gene Transfer for Diabetic Neuropathy. Losordo Douglas M. D. NCT00056290; 2010.
ADVM-022 Intravitreal gene therapy for dme. Adverum Biotechnologies Inc. NCT04418427; 2023.
Mccall MD, Toso C, Baetge EE, Shapiro AM. Are stem cells a cure for diabetes? Clin Sci (Lond). 2009;118(2):87-97. doi: 10.1042/CS20090072, PMID 19807695.
Sheik Abdulazeez S. Diabetes treatment: a rapid review of the current and future scope of stem cell research. Saudi Pharm J. 2015;23(4):333-40. doi: 10.1016/j.jsps.2013.12.012, PMID 27134533.
Bonner Weir S, Baxter LA, Schuppin GT, Smith FE. A second pathway for regeneration of adult exocrine and endocrine pancreas a possible recapitulation of embryonic development. Diabetes. 1993;42(12):1715-20. doi: 10.2337/diab.42.12.1715, PMID 8243817.
Elbahwy IA, Ibrahim HM, Ismael HR, Kasem AA. Enhancing bioavailability and controlling the release of glibenclamide from optimized solid lipid nanoparticles. J Drug Deliv Sci Technol. 2017 Apr;38:78-89. doi: 10.1016/j.jddst.2017.02.001.
Mumuni MA, Kenechukwu FC, Ofokansi KC, Attama AA, Diaz DD. Insulin loaded mucoadhesive nanoparticles based on mucin chitosan complexes for oral delivery and diabetes treatment. Carbohydr Polym. 2020;229:115506. doi: 10.1016/j.carbpol.2019.115506, PMID 31826394.
Liu Y, Zeng S, JI W, Yao H, Lin L, Cui H. Emerging theranostic nanomaterials in diabetes and its complications. Adv Sci (Weinh). 2022;9(3):e2102466. doi: 10.1002/advs.202102466, PMID 34825525.
Yan Y, Cai H, Yang M. The application of nanotechnology for the diagnosis and treatment of endocrine disorders: a review of current trends toxicology and future perspective. Int J Nanomedicine. 2024 Sep 25;19:9921-42. doi: 10.2147/IJN.S477835, PMID 39345911.