
Int J App Pharm, Vol 18, Issue 1, 2026, 493-500Original Article
FORMULATION AND EVALUATION OF ASCORBIC ACID TABLETS USING MICROCRYSTALLINE CELLULOSE (MCC) ISOLATED FROM OIL PALM EMPTY FRUIT BUNCHES (OPEFB)
ELISA PUTRI1*, FAZRINA ZAHARA2, MUTTAQIN3
1,2Department of Pharmacy, Faculty of Health Sciences, Universitas Sains Cut Nyak Dhien, Aceh-24415, Indonesia. 3Department of Computer Engineering, Faculty of Engineering, Universitas Sains Cut Nyak Dhien, Aceh-24415, Indonesia
*Corresponding author: Elisa Putri; *Email: elisa.putri@uscnd.ac.id
Received: 21 Dec 2024, Revised and Accepted: 24 Nov 2025
ABSTRACT
Objective: This study aimed to evaluate the effect of the composition of microcrystalline cellulose (MCC) isolated from oil palm empty fruit bunches (OPEFB) on the quality attributes of ascorbic acid tablets.
Methods: Oil palm empty fruit bunches were processed into powder, α-cellulose was isolated and hydrolyzed to produce MCC. The MCC was characterized for physicochemical properties. Four tablet formulations (F1–F4) containing varying proportions of OPEFB-derived MCC and commercial Avicel® MCC were prepared. Tablets were evaluated for weight uniformity, size uniformity, hardness, friability, and disintegration time. Experimental tablets were compared with a commercially available ascorbic acid tablet to assess the suitability of OPEFB-derived MCC as an alternative excipient.
Results: MCC isolated from OPEFB was suitable as an excipient for ascorbic acid tablets mean tablet weights for F1–F4 and the commercial tablet were 265.00 mg, 266.50 mg, 285.00 mg, 287.00 mg, and 312.00 mg, respectively. Hardness testing indicated that tablets containing OPEFB MCC were harder than those with Avicel® MCC and commercial tablets; however, all formulations exhibited hardness values below 4 kg. Friability values for F1–F4 and the commercial tablet were 0.46%, 0.76%, 0.15%, 0.30%, and 0.00%, respectively (all<1%). Disintegration time were 8.90, 6.51, 9.16, 5.39, and 11.68 min for F1–F4 and the commercial tablet, respectively (all ≤15 min).
Conclusion: OPEFB-derived MCC has potential as an excipient in ascorbic acid tablets and influences their physical quality attributes. While all formulations met pharmacopeial standards for friability and disintegration time, improvements are required for weight uniformity and hardness.
Keywords: Ascorbic acid, Filler, Microcrystalline cellulose, Tablet, OPEFB
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.53387 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
In Indonesia, oil palm plantations are currently expanding, as evidenced by the steady increase in plantation area and oil palm production since 2012. In 2012, the total plantation area was 1,076,081 hectares, which increased to 16.38 million hectares by 2022. Of this, smallholder oil palm plantations accounted for 6.94 million hectares. The rapid development of the palm oil industry requires serious attention, particularly regarding its impact on environmental preservation. Palm oil waste, especially oil palm empty fruit bunches (OPEFB), will become a serious problem if not managed properly. Appropriate handling is essential to maintain environmental cleanliness and order [1].
The negative impacts of palm oil mills on surrounding communities include significant environmental pollution. Some mills discharge hazardous and toxic waste into rivers without considering the composition of the substances present, which can deteriorate water quality. Industrial processes also contribute to environmental health problems by producing waste that damages facilities and ecosystems. Community consultations revealed that ecological pollution from processed palm oil waste can produce an unpleasant odor and create breeding grounds for flies. Additionally, this waste can generate leachate liquid formed from the decomposition of waste, particularly solid organic waste. Leachate may contain harmful substances, including chemicals, heavy metals, and microorganisms from the decomposing matter. If not properly managed, leachate can contaminate surrounding soil and water sources, posing threats to public health and ecosystems [2].
Environmental pollution from factory effluents is not only an ecological concern but also a pressing social issue. Neglecting the long-term effects of solid, liquid, and gaseous industrial waste can threaten public health and the sustainability of ecosystems. For instance, soil-polluting waste can seep into clean water sources, endangering the health of residents who rely on them. Therefore, there is an urgent need to enforce stricter regulations and promote the conversion of waste into valuable products, thereby increasing the economic value of OPEFB waste.
At present, OPEFB is primarily used commercially for compost and briquette production. The main components of OPEFB solid waste are cellulose (43%), hemicellulose (24%), and lignin (21%). These components can be converted into various chemicals, materials, and high-value products (2). The composition of OPEFB can be seen in table 1. One of the most valuable components is cellulose, which can be processed into microcrystalline cellulose (MCC). MCC is particularly relevant in biomaterial applications due to its biocompatibility and non-toxic nature [3].
Cellulose can be isolated using various methods, including mechanical techniques (e. g., high-pressure and ultrasonic methods), chemical techniques (e. g., organosolv, strong acid hydrolysis, alkaline solvents, ionic liquids, and oxidation processes), and biological approaches (e. g., enzyme-assisted extraction). In this study, a chemical cellulose isolation method was employed [4]. The process began with hydrolysis using strong acids such as sulfuric acid, hydrochloric acid, or perchloric acid, which effectively degrade the amorphous regions of plant fibers and hydrolyze hemicellulose into xylose and other sugars. The second step involved delignification using sodium hydroxide (NaOH) to remove lignin. The third stage was bleaching to further break down remaining lignin and impurities, typically using oxidizing agents such as hydrogen peroxide or hypochlorite [5].
Cellulose is a linear polymer of glucose. Based on its solubility in alkalis, cellulose can be classified into alpha, beta, and gamma cellulose. MCC is a purified, partially depolymerized cellulose with the formula(C6H10O5)n, produced by treating alpha-cellulose with mineral acids (type Ib). This polysaccharide consists of a linear chain of β-1,4-D-anhydroglucopyranosyl units, with degrees of polymerization ranging from several hundred to over ten thousand. The raw material for MCC production is typically plant-derived pulp [6].
Cellulose derivatives are widely used as excipients in pharmaceutical formulations, such as ethyl cellulose, methylcellulose, carboxymethyl cellulose, and hydroxypropyl methylcellulose, in oral, topical, and injectable dosage forms. The widespread use of cellulose is due to its inertness and biocompatibility in humans [7].
In the pharmaceutical field, MCC is one of the most common excipients used in directly compressed tablets, functioning as a filler, binder, and disintegrant. MCC is typically obtained by hydrolyzing alpha-cellulose with mineral acids [8]. The most widely used commercial MCC product is marketed under the trade name Avicel®.
Avicel® is used in directly compressed tablets as both a filler and a disintegrant, resulting in rapid tablet disintegration and drug release due to its ability to enhance penetration of the dissolution medium. However, Avicel® is relatively expensive, and because it typically constitutes 50–80% of the tablet formulation, its high cost contributes significantly to the overall price of the product [9]. Therefore, this study investigates the potential of MCC isolated from OPEFB as a cost-effective alternative to Avicel® in the production of ascorbic acid tablets.
MATERIALS AND METHODS
The materials used in this study were ascorbic acid, ethylenediamine, benzene, oil palm empty fruit bunches (OPEFB), ethanol, hydrochloric acid, hydrogen peroxide, iodine, spray-dried lactose (LSD), magnesium stearate, potassium chromate, potassium dihydrogen phosphate, potassium iodide, sodium sulfide p. a., talc, and zinc chloride.
Preparation of 3.5% HNO3 mixture
A total of 54.6 ml of 65% HNO3 was added to 10 mg NaNO2, then diluted with distilled water in a 1000 ml volumetric flask to the mark line and homogenized.
Preparation of 2% NaOH solution
A total of 10 g of NaOH was dissolved in 100 ml of distilled water in a beaker, allowed to cool, then transferred into a 500 ml volumetric flask, topped up with distilled water to the mark, and homogenized.
Preparation of 17.5% NaOH solution
A total of 87.5 g of NaOH was dissolved in 100 ml of distilled water in a beaker, allowed to cool, then transferred into a 500 ml volumetric flask, topped up with distilled water to the mark, and homogenized.
Preparation of 2% Na2S2O3 solution
A total of 10 g of Na2S2O3 was placed into a 500 ml volumetric flask, topped up with distilled water to the mark, and homogenized.
Preparation of 1.75% NaOCl solution
A total of 73 ml of 12% NaOCl was placed into a 500 ml volumetric flask, topped up with distilled water to the mark, and homogenized.
Preparation of 10% H2O2 solution
A total of 167 ml of 30% H2O2 was placed into a 500 ml volumetric flask, topped up with distilled water to the mark, and homogenized.
Preparation of 48.84% H2SO4 solution
A total of 245 ml of 98% H2SO4, was carefully added to a 500 ml volumetric flask containing 200 ml of distilled water, allowed to cool, then topped up with distilled water to the mark and homogenized.
Isolation of microcrystalline cellulose (MCC)
Preparation of OPEFB
OPEFB was washed with water and dried in the sun until completely dry. The material was cut into small pieces, ground into powder, and sieved through an 80-mesh sieve.
Isolation of α-cellulose from OPEFB
A total of 75 g of OPEFB powder was placed in a beaker, mixed with 1 l of 3.5% HNO₃ containing 10 mg NaNO₂, and heated at 90 °C for 2 h. The mixture was filtered, and the residue was washed until the filtrate was neutral. The residue was then treated with 750 ml of a solution containing 2% NaOH and 2% Na₂S₂O₃ at 50 °C for 1 hour, filtered, and washed until a neutral pH was reached. Bleaching was carried out using 250 ml of 1.75% NaOCl at 70 °C for 30 min, followed by filtration and washing to neutral pH. Purification of α-cellulose was conducted using 500 ml of 17.5% NaOH at 80 °C for 30 min, filtered, and washed until neutral. A final bleaching step was performed with 10% H₂O₂ at 60 °C, followed by filtration and washing. The α-cellulose obtained was dried in an oven at 60 °C [10, 11].
Hydrolysis of α-cellulose to obtain MCC
The oven-dried α-cellulose was hydrolyzed with either 2.5 N HCl or 48.84% H₂SO₄ under reflux for 15 min. The resulting product was washed with distilled water until neutral, dried at 40 °C, and milled to obtain OPEFB-derived MCC [12].
Characterization of OPEFB MCC
Scanning electron microscope (SEM)
The surface morphology of the OPEFB MCC was observed using a Scanning Electron Microscope (SEM, Hitachi 3000, Japan) at 15 kV and 32.5 mA. Samples were pre-coated with carbon and gold before being mounted on the SEM stub to reduce electron charging and improve image clarity [12].
Flow rate, bulk density, and tapped density
Bulk density was determined by pouring pre-sieved granules (180 mesh) into a graduated cylinder through a wide funnel and measuring both volume and weight. The average was calculated from three replicates. Tapped density was determined as the mass-to-volume ratio after mechanical tapping, expressed in g/cm³ [13].
Bulk Density (BD) = 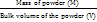
Tapped Density (BD) = 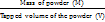
Carr index and hausner ratio
The Carr Index and Hausner Ratio are indicators used to evaluate the flowability and compressibility of granules. The Carr Index measures the compressibility of granules by comparing bulk density and tapped density, with values obtained from the average of three repetitions [14]. The Hausner Ratio assesses powder flowability and is calculated as the ratio of tapped density to bulk density [15].
Carr′ s index (%) = 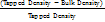 x 100
x 100
Hausner′ s Ratio = 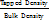
Flow rate and angle of repose
Granular flow characteristics were evaluated based on flow rate and angle of repose. A total of 100 g of granules was placed into a funnel, and the flow rate was determined using a stopwatch. Timing began when the funnel opening was unblocked and continued until all granules had exited. The maximum flow duration for 100 g of granules was recorded as 10 seconds. The frictional force between granules was measured using the angle of repose, defined as the maximum possible angle between the surface of the powder pile and the horizontal plane [12].
Tan θ = 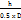
Where θ is the angle of repose, h is the height (cm), and D is the diameter (cm).
Tablet formulation
The ingredients used in the tablet formulation were ascorbic acid, amprotab, avicel, modified MCC, magnesium stearate, and talc. In this study, four ascorbic acid tablet formulations (n = 4) were developed, each differing in the type and quantity of microcrystalline cellulose (MCC) used as excipient. Formulation 1 (F1) contained 150 mg of ascorbic acid and 130 mg of MCC derived from oil palm empty fruit bunches, whereas Formulation 2 (F2) contained 150 mg of ascorbic acid with no Avicel added. Formulation 3 (F3) consisted of 150 mg of ascorbic acid and 136 mg of MCC, and Formulation 4 (F4) contained 150 mg of ascorbic acid and 136 mg of Avicel. The ingredients were blended using a Kris blender for 2 min, after which the mixtures underwent preformulation testing.
Table 1: Tablet formula of ascorbic acid using MCC isolated OPEFB
| Ingredients | Formula | |||
| F1 | F2 | F3 | F4 | |
| Ascorbic acid (mg) | 150 | 150 | 150 | 150 |
| Modified MCC (mg) | 130 | 0 | 136 | 0 |
| Avicel (mg) | 0 | 130 | 0 | 136 |
| Magnesium stearate (mg) | 6 | 6 | 6 | 6 |
| Talc (mg) | 3 | 3 | 3 | 3 |
| Amprotab (mg) | 10 | 10 | 10 | 10 |
Information: F1 (Formulation1) = Ascorbic acid 150 mg: MCC 130 mg; F2 (Formulation 2) = Ascorbic acid 150 mg: Avicel 0 mg; F3 (Formulation3) = Ascorbic acid 150 mg: MCC 136 mg; F4 (Formulation 4) = Ascorbic acid 150 mg: Avicel 136 mg
Tablet evaluation
Weight uniformity
Twenty tablets were weighed individually, and the average weight was calculated. The weight of each tablet was then compared with the average weight. According to the USP, no more than two tablets should deviate from the mean weight by more than the specified percentage, and no tablet should deviate by more than twice that percentage [16].
Size uniformity
Twenty tablets were randomly selected and measured individually for thickness and diameter using a micrometer. The average diameter and thickness were calculated. Tablets were considered acceptable in terms of size uniformity if the average diameter was not less than 1⅓ times the average thickness and not more than three times the average thickness.
Hardness
Tablet hardness, also referred to as crushing strength, can be influenced by factors such as poor flow properties and powder moisture content [17]. A YD-3 hardness tester was used to measure tablet hardness.
Friability
Twenty tablets from each batch were randomly selected and weighed. The friability test was performed using a Roche-type friabilator at a total of 100 revolutions (25 rpm for 4 min). The tablets were removed, cleaned, and reweighed. The mean of triplicate readings was recorded, and the standard deviation was calculated [18].

Disintegration time
Disintegration time is defined as the time required for a tablet to break down into small particles. The test was conducted using a disintegration apparatus, with one tablet placed in each of the six tubes and a disk placed on top of each tablet. The apparatus was maintained at 37±0.5 °C using distilled water and operated at 30 cycles per minute. The time for complete disintegration, with no palpable residual mass, was recorded. The mean disintegration time and standard deviation were calculated [19].
Statistical analysis
All experiments were conducted in triplicate, and results were expressed as mean±standard deviation. The physical properties of ascorbic acid tablets were analyzed using SPSS 26.0. Data for each variable were tested for normality and homogeneity before being analyzed using a one-way Analysis of Variance (ANOVA) for parametric data or a Kruskal–Wallis test for non-parametric data at a 95% confidence level (p<0.05). For significant results, a univariate test at a 95% confidence level (p<0.05) was performed, followed by Duncan’s post hoc test.
Fig. 1: Research flow chart
RESULTS AND DISCUSSION
MCC yield and characterization
The isolation of α-cellulose was carried out through a delignification process using 3.5% HNO₃ and NaNO₂ to remove lignin content, followed by a swelling treatment with 2% NaOH and 2% Na₂SO₃ to remove hemicellulose. Pulping was then performed, involving bleaching with 1.75% NaOCl solution and purification of α-cellulose using 17.5% NaOH to eliminate β-cellulose and γ-cellulose. A final bleaching step with 10% H₂O₂ was conducted to obtain pure, white α-cellulose, which was subsequently dried in an oven at 60 °C.
Table 1: MCC yield percentage
| Weight of OPEFB powder | Weight of α-cellulose | Weight of MCC | Percentage MCC (%) |
| 75 g | 21 g | 13.5 g | 64.3% |
From 75 g of OPEFB powder, 21 g of α-cellulose, and 13.5 g of MCC were obtained, corresponding to a yield of 64.3% from α-cellulose to MCC.
Table 2: Granule evaluation results±SDa
| Evaluation parameter | F1 | F2 | F3 | F4 | USP 41 requirements | p-value |
| Bulk density (g/cm3) | 0.528±0.001 | 0.527±0.001 | 0.532±0.001 | 0.541±0.002 | 0.910>0.05 | |
| Tapped density (g/cm3) | 0.561±0.009 | 0.557±0.012 | 0.562±0.010 | 0.580±0.011 | 0.423>0.05 | |
| Carr’s index (%) | 6.02 | 5.36 | 5.36 | 6.68 | <10% | 0.698>0.05 |
| Hausner’s ratio | 1.064±0.018 | 1.050±0.028 | 1.057±0.021 | 1.072±0.021 | <1,25 | 0.583>0.05 |
| Angle of repose () | 24.11±1.01 | 21.60±0.29 | 22.56±1.78 | 21.29±0.54 | <30 | 0.139>0.05 |
| Flow rate (g/s) | 1.26±0.14 | 1.29±0.20 | 1.50±0.05 | 1.39±0.17 | <10 s/100 g | 0.787>0.05 |
aResult are expressed as a mean ±SD, n=4, Condition: Specification limits or criteria used for evaluation, p-value: Probability value indicating statistical significance (threshold set at<0.05)
Bulk density is a parameter used to describe the particle size characteristics of a material. The bulk density of granules mainly depends on particle size distribution, particle shape, and the tendency of particles to form intra-or inter-particle voids, or to create a non-porous mass. Bulk densities ranged from 0.527 to 0.541 g/cm³, and tapped densities ranged from 0.557 to 0.580 g/cm³ for F1–F4. F4 had the highest bulk density, while F2 had the lowest. These results indicate that F4 exhibited faster flow properties in the unconsolidated granule state. Bulk density influences tapped density, which is defined as the weight of the powder divided by its volume in a graduated cylinder after tapping [20].
Evaluation of bulk density and tapped density is important for determining the compressibility of granule masses. Tapped density is highly dependent on particle shape, as smaller particles tend to produce a more compact mass compared to larger granules. From table 2, it can be observed that the tapped density values in all formulas are higher than their respective bulk density values. This indicates good packing behavior, a higher tapped density with minimal variation from bulk density suggests that the granules are well compressed and have a strong potential to form dense, stable tablets under compression [21].
The Hausner ratio assesses the flowability and compressibility of powders by comparing tapped density to bulk density [21]. A Hausner ratio below 1.25 is considered good. Table 2 shows that all formulations met this requirement, indicating that both MCC and Avicel powders possess good flow properties. One reason microcrystalline cellulose is widely preferred as a diluent is its low bulk density. Excipients with low density and broad particle size distribution exhibit high dispersion potential by weight, optimal packing density, and effective mixing with the active drug [22].
The Carr index is used to evaluate granule compressibility. Carr’s index values were below 10% and Hausner ratios below 1.25 for all formulations, indicating acceptable flow properties. F1 and F3 had the lowest Carr index values, followed by higher values for F2 and F4. This suggests that F2 and F3 have lower compressibility, and therefore slightly better flowability, compared to F1 and F4. The inclusion of OPEFB-derived MCC in F2 and F3 contributed to this outcome, as its morphological characteristics may reduce particle cohesion [23].
The angle of repose is another parameter used to assess powder flow. It is the angle formed between the base plane and the slope of a conical pile of powder after pouring [23]. Based on table 2, the angle of repose for all formulas ranged from 20° to 25°. According to USP 41 standards, an ideal angle of repose is below 30°, indicating that all formulas displayed good flow properties suitable for the tableting process [24].
Flow rate refers to the ability of granules to flow, which is a critical parameter in tablet compression, especially during die filling. Flow rate affects tablet weight uniformity, which directly impacts dosage precision. Factors influencing flow rate include particle size, particle size distribution, particle shape, and particle density [25].
The flow rate data show that all formulas had similar results and met the set requirements. Granules with a flow rate>10 g/s are categorized as very good, 4–10 g/s as good, 1.6–4 g/s as poor, and<1.6 g/s as very poor [25]. Based on these criteria, all five samples fell into the very poor category, with flow rates below 1.6 g/s. F1 had the fastest flow rate at 1.26 s, while F3 had the slowest at 1.50 s. This variation is attributed to differences in excipient type and proportion, particularly between Avicel and OPEFB MCC. Avicel’s porous structure improves granule fluidity by reducing interparticle cohesion, giving F1 the fastest flow rate. Conversely, the high OPEFB MCC content in F3 likely increased interparticle cohesion due to its physicochemical characteristics, such as particle size and crystal form, resulting in the slowest flow rate [26]. Generally, smaller granule particle sizes increase cohesion, causing clumping and reducing flow speed.
Statistical analysis (ANOVA or Kruskal–Wallis as appropriate) showed no significant differences among formulations (p>0.05) for the granule parameters measured.
Tablet evaluation
Tablet morphology using Scanning Electron Microscopy (SEM).
The morphological analysis results obtained using SEM are shown in fig. 1. In fig. 1(A), MCC powder exhibits a fibrous and elongated structure with uneven particle size distribution, a rough surface, and a rigid surface texture. This indicates that the wood-derived powder is still coated with lignin. Such a fibrous structure can influence the flow properties and compressibility of granules. To obtain more uniform and higher-quality MCC powder, ultrasonic isolation can be employed with variations in extraction temperature. The cavitation effect in the sonication process can break and remove the lignin layer and part of the hemicellulose, enabling their separation and dissolution in NaOH solution as black liquor [27]. Fig. 1(B) shows that Avicel MCC powder has a rod-shaped, shorter, and hollow structure.

Fig. 2: SEM images of MCC obtained from (A) OPEFB; (B) Avicel pH 102 at magnification x3.000
Increasing acid concentration during extraction converts holocellulose into alpha-cellulose, forming uniform fibrils with smooth surfaces. However, higher acid concentrations can also produce shorter fibrils with rougher surfaces, as shown in fig. 2(A), 2(B), and 2(C)[28]. Structural variations in cellulose are caused by the depolymerization of cellulose polymers into shorter-chain MCC [29]. Acid treatment significantly reduces fiber width, with the average width of MCC decreasing from 10 μm to 3 μm as the H₂SO₄ concentration increases from 5% to 25%. The shape and diameter of OPEFB MCC are highly dependent on acid concentration and the acid-to-fiber ratio. OPEFB MCC generally has a smooth outer surface and irregular shape, likely due to the removal of silica, hemicellulose, and lignin [30].

Fig. 3: SEM images of MCC obtained from OPEFB with varying sulfuric acid concentrations: (A) MCC 5%, (B) MCC 15%, (C) MCC 25%
Yugatama et al. (2012) reported that the particle size of Avicel pH 102 ranges from 13.33 to 200 μm, with irregular shapes, uneven surface textures, and obtuse angles [31]. The following is a comparison of morphological images from several literatures:

Fig. 4: Comparison of SEM images of MCC obtained from Avicel pH 102 [31, 14]
The results of the tablet morphology evaluation for each formulation are presented as follows: fig. 5(A)-F1 Tablets containing 130 mg OPEFB MCC powder exhibit rough surfaces with many sharp edges and voids, resulting in a less compact structure. This may contribute to better granule flow but slower compression during tablet formation. Fig. 5(B)-F3 Tablets containing 136 mg OPEFB MCC filler display a smoother surface compared to F1. The elongated, fibrous structure indicates better compaction and more uniform particle distribution, which may enhance compression by improving interparticle bonding.

Fig. 5: SEM images of tablets with OPEFB MCC filler: (A) F1, (B) F3 at magnification x3.000
Fig. 6(A) – F2: Tablets with 130 mg Avicel filler exhibit a rough, coarse surface similar to F1, but with more uniform particle distribution. However, the tablets appear less dense, possibly due to the smaller amount of filler used. Fig. 6(B)-F4 with 136 mg Avicel filler shows a more elongated surface. The structure is more compact and dense, which is due to the agglomeration of components and the greater amount of advice, thus providing a better binding effect and producing a more homogeneous particle distribution [32]. Fig. 6(C)-Commercial ascorbic acid tablets show the smoothest, most uniform, and compact surface among all samples.
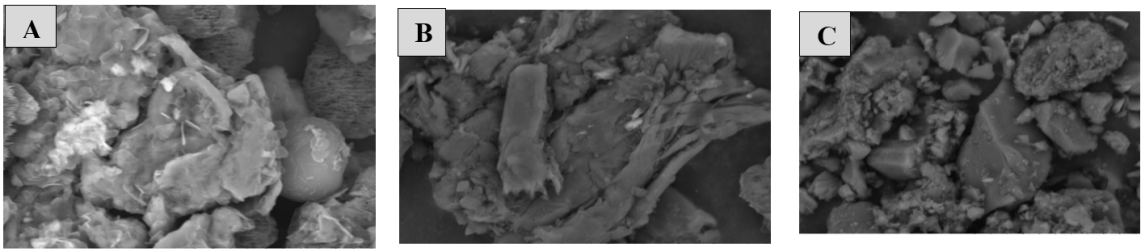
Fig. 6: SEM images of tablets with Avicel filler: (A) F2, (B) F4, and (C) commercial ascorbic acid tablet at magnification x3.000
MCC contains hydrogen bonds between adjacent cellulose molecules, enabling the formation of multiple hydrogen bonds that contribute to its high compaction capacity even under low compression forces [33]. Therefore, to improve tablet compaction when using OPEFB MCC, the amount of MCC in the formulation can be increased.
Table 3: Summarizes tablet characteristics compared with USP 41 requirements±SDb
| Evaluation parameter | F1 | F2 | F3 | F4 | Commercial ascorbic acid | USP 41 requirements |
p-Value |
| Weight uniformity (mg) | 265.00±8.272 | 266.50±13.089 | 285.00±15.044 | 287.00±12.607 | 312.00±6.156 | <5% | 0.937 |
| Size uniformity (mm) | 0.9±0.00 | 0.9±0.00 | 0.95±0.00 | 0.95±0.00 | 1±0.00 | 11/3t<d<3t | 1.000 |
| Thickness (T) (mm) | 0.52±0.00 | 0.45±0.00 | 0.85±0.00 | 0.40±0.00 | 0.32±0.00 | 0.260 | |
| 4/3T ≤ D ≤ 3T | 0.69<0,9<1.56 | 0.6<0.9<1.35 | 1.13>0.95<2.55 | 0.53<0.95<1.2 | 0.43<1>0.96 | ||
| Hardness (kg) | 3.29±0.40 | 0.98±0.23 | 3.82±2.60 | 1.12±0.87 | 2.92±0.06 | 4-8 kg | 0.012 |
| Friability (%) | 0.46 | 0.76 | 0.15 | 0.3 | 0.00 | <1% | 0.042 |
| Disintegration time (min) | 8.90±1.40 | 6.51±0.74 | 9.16±2.36 | 5.39±0.73 | 11.68±1.03 | <15 menit | 0.033 |
bResult are expressed as an mean±SD, n = 4, and positive group withcommercial ascorbic acid. Condition: Specification limits or criteria used for evaluation, p-value: Probability value indicating statistical significance (threshold set at<0.05).
Weight uniformity
Weight uniformity assesses the consistency of material filled into molds and compressed into tablets, which directly influences dosing accuracy and therapeutic safety. According to USP 41 (2020), for tablets with an average weight>250 mg, no more than two tablets may deviate by more than 5% from the mean. mean tablet weights were 265.00±8.27 mg (F1), 266.50±13.09 mg (F2), 285.00±15.04 mg (F3), 287.00±12.61 mg (F4), and 312.00±6.16 mg (commercial). According to USP, for tablets with a mean weight>250 mg, no more than two tablets may deviate by more than 5% from the mean. The formulations did not meet this requirement uniformly, suggesting variability in fill weight or die filling. ANOVA results showed no significant differences (p>0.05). The deviations may be attributed to the less uniform particle size distribution of OPEFB MCC compared to Avicel MCC, as well as differences in excipient density [26].
Size uniformity
Tablet size uniformity ensures consistent dimensions within a batch. USP 41 specifies that the diameter should be between 4/3 and 3 times the thickness. ANOVA results showed no significant differences (p>0.05), but F3 did not meet the requirement, likely due to the non-uniform particle size distribution of OPEFB MCC. Such variability can affect compression behavior, leading to inconsistent density and dimensions. Tablet thickness is influenced by fill weight, compression force, and bulk density [26]. Factors that affect tablet thickness include compression during tablet molding, the amount of granule mass filled in the tablet molding chamber, and the density of the printed tablet mass [34].
Hardness
Measured hardness values were below the USP recommended range (4–8 kg) for all formulations. OPEFB MCC-containing formulations (F1 and F3) exhibited higher hardness than Avicel®-containing formulations (F2 and F4), indicating better binding capacity; however, absolute values remained below the ideal range (e. g., F1: 3.29±0.40 kg; F3: 3.82±2.60 kg). ANOVA indicated significant differences (p<0.05). The higher hardness in OPEFB MCC tablets suggests stronger binding capacity, but the overall values did not meet the specified criteria. This shows that OPEFB MCC contributes to better binding properties during the compression process. Variations in hardness values highlight the significant impact of the combination of fillers and physical properties on the ability of granules to form strong and homogeneous tablets [26].
Friability
Friability measures resistance to mechanical stress, with acceptable limits being<1% weight loss (USP 41). All formulations met the USP criterion (<1%): F1 = 0.46%, F2 = 0.76%, F3 = 0.15%, F4 = 0.30%, commercial = 0.00%. Friability decreased with higher OPEFB MCC content, likely due to denser, more compact granules with lower porosity, which improved mechanical strength. ANOVA revealed significant differences (p = 0.042). Based on the results in table 3, all formulations showed tablet friability values below 1%. As the amount of OPEFB MCC increased, the friability value decreased. This was due to denser and more compact granules with lower porosity, making the tablets stronger and less fragile. It can be concluded that all four formulations met the requirements for tablet friability.
Disintegration time
Disintegration time measures how quickly tablets break down in liquid, ensuring timely drug release. The Indonesian Pharmacopeia (3rd ed.) specifies<15 min for uncoated tablets [8]. According to the 3rd Edition of the Indonesian Pharmacopeia, the acceptable disintegration time for uncoated tablets should not exceed 15 min. All formulations disintegrated within 15 min (F1: 8.90±1.40 min; F2: 6.51±0.74 min; F3: 9.16±2.36 min; F4: 5.39±0.73 min; commercial: 11.68±1.03 min). All formulations met this requirement, with ANOVA showing significant differences (p<0.05). Variations were influenced by binder/disintegrant composition, compression force, and hardness. OPEFB MCC tablets tended to have slightly longer disintegration times than Avicel MCC tablets, consistent with their higher hardness. The difference in disintegration time was influenced by the presence of binders and disintegrants in the tablet. In addition, excipients, compression pressure during tablet making, and tablet hardness also affected the disintegration time [26].
Rum (2019) reported that MCC from nang de pina (60% w/w) produced tablets with a hardness of 5.06 kg and a disintegration time of 5.91 min [35]. Tarigan (2019) found corn cob MCC tablets had a hardness of 4.18 kg, friability of 0.018%, and a disintegration time of 8.5 min. These results highlight that MCC source and properties strongly influence tablet mechanical strength and disintegration [36].
Microcrystalline cellulose is widely used as a diluent with excellent binding properties, especially in direct compression. While it is self-disintegrating and requires minimal lubricant, it does not eliminate the need for dedicated disintegrants and lubricants. When used at 10–50% w/w, MCC can complement disintegrants to achieve optimal disintegration performance [33, 37].
Microcrystalline Cellulose (MCC) remains a pivotal excipient in the realm of pharmaceutical formulation, owing to its proven utility, functional diversity, and regulatory endorsement. Its physicochemical robustness, marked by exceptional compressibility, binding efficacy, chemical inertness, and wide-ranging drug compatibility, renders it highly suitable for numerous dosage forms, including conventional tablets, capsules, oral suspensions, and emerging drug delivery platforms. Structurally, MCC is derived via partial acid hydrolysis of α-cellulose, a process that eliminates amorphous regions and yields a crystalline, fibrous material. This microstructure confers high mechanical integrity and crystallinity, facilitating its dual function as a filler and binder in both direct compression and wet granulation methodologies. Various commercial grades, such as MCC 101, 102, 200, and silicified MCC (SMCC), offer differentiated particle sizes and flow characteristics, enabling tailored application in diverse formulation scenarios, particularly in high-speed tablet manufacturing [38].
CONCLUSION
This study demonstrated the successful isolation of microcrystalline cellulose (MCC) from oil palm empty fruit bunch (OPEFB) waste and its application as an excipient in ascorbic acid tablet formulations. The resulting tablets met the friability and disintegration time requirements; however, weight uniformity, size uniformity, and hardness require further optimization. These findings highlight the potential of OPEFB-derived MCC as a sustainable excipient source. Future work should focus on enhancing particle size uniformity, optimizing MCC content, and exploring modifications with functional excipients, such as hydroxypropyl methylcellulose, to improve overall tablet performance and expand its applicability in pharmaceutical formulations.
FUNDING
This research was supported by the Ministry of Education, Culture, Research, and Technology. The authors also acknowledge the contributions of all team members involved in the completion of this work.
AUTHORS CONTRIBUTIONS
All authors contributed equally. Conceptualization: E. Putri. Design: E. Putri, F. Zahara, M. Muttaqin. Data collection and processing: F. Zahara. Analysis and interpretation: E. Putri, F. Zahara. Literature search and writing: E. Putri. Final approval: E. Putri.
CONFLICT OF INTERESTS
The authors declare no conflict of interest.
REFERENCES
Badan Pusat Statistik. Statistics Indonesia. Palm Oil Statistics in Indonesia; 2012.
Alfiani DL, Rofita HD, Efendi I, Fatmawati. The impact of palm oil industry waste disposal on community livelihood sustainability in Indragiri Hulu Regency. Sci J Ilm Sain Teknol. 2024;3(2):773-80.
Dewanti DP. Potensi selulosa dari limbah tandan kosong kelapa sawit untuk bahan baku bioplastik ramah lingkungan. Jurtekling. 2018;19(1):81. doi: 10.29122/jtl.v19i1.2644.
Effendi DB, Rosyid NH, Nandiyanto AB, Mudzakir A. Review: sintesis nanoselulosa. J Integr Proses. 2015;5(2):61-74. doi: 10.36055/jip.v5i2.19.
Azami YI, Musfiroh I, Muchtaridi PR, Pratiw R, Putra ON. Isolation of cellulose from Siwalan fiber (Borassus flabellifer L.) using response surface methodology. Int J App Pharm. 2023;15(2):48-51. doi: 10.22159/ijap.2023.v15s2.09.
Yohana Chaerunisaa A, Sriwidodo S, Abdassah M. Microcrystalline cellulose as pharmaceutical excipient. In: Ahmad U, Akhtar J, editors. Pharmaceutical formulation design: recent practices. IntechOpen; 2020. doi: 10.5772/intechopen.88092.
Jackson JK, Letchford K, Wasserman BZ, Ye L, Hamad WY, Burt HM. The use of nanocrystalline cellulose for the binding and controlled release of drugs. Int J Nanomedicine. 2011;6:321-30. doi: 10.2147/IJN.S16749, PMID 21383857.
Rustiani E, Agustina R, Andini S. Formulation and pharmaceutical quality evaluation of tablets containing extract of Cinnamomum burmannii BARK and Colocasia esculenta (L.) Schott leaves. JMPI. 2023;9(1):27-34. doi: 10.35311/jmpi.v9i1.292.
Laili N, Komala AM, Maulida H, Suprapto S. Optimization of sago starch (Metroxylon rumphii) as a Co-processed excipient in theophylline tablets. Pharmacon J Farm Indones. 2019;14(2):72-81. doi: 10.23917/pharmacon.v14i2.6422.
Ohwoavworhua FO, Adelakun TA. Phosphoric acid-mediated depolymerization and decrystallization of α-cellulose obtained from corn cob: preparation of low crystallinity cellulose and some physicochemical properties. Trop J Pharm Res. 2007;4(2):509-16. doi: 10.4314/tjpr.v4i2.14627.
Ohwoavworhua FO, Adelakun TA. Non-wood fibre production of microcrystalline cellulose from Sorghum caudatum: characterisation and tableting properties. Indian J Pharm Sci. 2010;72(3):295-301. doi: 10.4103/0250-474X.70473, PMID 21188036.
Rao MR, Sonavane V, Kulkarni S, Magar M, Zope A, Karanjkar P. Design of transdermal patch of ketoprofen by full factorial design for treatment of rheumatoid arthritis. J Drug Delivery Ther. 2019;9(2):197-205. doi: 10.22270/jddt.v9i2.2549.
Randis R, Darmadi DB, Gapsari F, Sonief AA. Isolation and characterization of microcrystalline cellulose from oil palm fronds biomass using consecutive chemical treatments. Case Studies in Chemical and Environmental Engineering. 2024;9:100616. doi: 10.1016/j.cscee.2024.100616.
Agustin N, Abdassah M. Isolasi dan karakterisasi selulosa mikrokristal dari nanas (Ananas comosus (L.) Merr.) [Isolation and characterization of microcrystalline cellulose derived from pineapple (Ananas comosus (L.) Merr.)]. Pharm: J farm Indones. 2021;18(1):111-21. doi: 10.30595/pharmacy.v18i1.10277.
Deshpande KB, Ganesh NS. Orodispersible tablets: an overview of formulation and technology. Int J Pharm Bio Sci. 2011;2(1):726-34.
Aruna M, Samreen SHR, Shaik Harun Rasheed. Formulation and evaluation of fast-disintegrating tablets of metoprolol succinate using various superdisintegrants. Int J Res Pharm Sci & Tech. 2019;1(2):79-83. doi: 10.33974/ijrpst.v1i2.150.
Bhosale NR, Kolte NS. Formulation development and evaluation of orally disintegrating tablet of chlorpheneramine maleate by sublimation technique. Int J Pharm Pharm Sci. 2019;11(9):28-36. doi: 10.22159/ijpps.2019v11i9.34387.
Gupta A, Mishra AK, Gupta V, Bansal P, Singh R, Singh AK. Recent trends of fast dissolving tablet-an overview of formulation technology. Int J Pharm Biol Arch. 2010;1(1):1-10.
Gaur K, Tyagi LK, Kori ML, CS Sharma, Nema RK. Formulation and characterization of fast-disintegrating tablet of aceclofenac by using sublimation method. Int J Pharm Sci Drug Res. 2011;3(1):19-22. doi: 10.25004/IJPSDR.2011.030104.
Patel SS, Patel NM. Development of directly compressible co-processed excipient for dispersible tablets using 32 full factorial design. Int J Pharm Pharm Sci. 2009;1(1):25-48.
Akseli I, Hilden J, Katz JM, Kelly RC, Kramer TT, Mao C. Reproducibility of the measurement of bulk/tapped density of pharmaceutical powders between pharmaceutical laboratories. J Pharm Sci. 2019;108(3):1081-4. doi: 10.1016/j.xphs.2018.10.009, PMID 30326209.
Silalahi K, Husni P. Review: application of microcrystalline cellulose in pharmaceuticals. Farmaka. 2018;16(1):380–8.
Al Hakim NA, Nur Fauziyyah SA, Purnamasari N, Faramayuda F. Formulation and physical evaluation of Orthosiphon aristatus leaf extract granule preparation as herbal beverage candidate. Majalah Obat Tradisional. 2024;29(3):286. doi: 10.22146/mot.91009.
USP-NF. The United States pharmacopeia and the national formulary. NF 36. 2018;7011:41.
Aminingsih T, Rahayu SY, Yulianita Y. Formulation of instant granule containing nano calcium from the shell of freshwater mussels (Anodonta woodiana) for autism children. IJPST. 2018;1(1):49-56. doi: 10.24198/ijpst.v1i1.16125.
Belali NG, Chaerunisaa AY, Rusdiana T. Isolation and characterization of microcrystalline cellulose derived from plants as excipient in tablet: a review. Indo J Pharm. 2019;1(2):55-61. doi: 10.24198/idjp.v1i2.21515.
Subhedar PB, Gogate PR. Alkaline and ultrasound-assisted alkaline pretreatment for intensification of delignification process from sustainable raw materials. India: Chemical Engineering Department Institute of Chemical Technology. 2014;21(1):216–25. doi: 10.1016/j.ultsonch.2013.08.001.
Ismail F, Ali Othman NE, Abdul Wahab N, Abdul Hamid F, Abdul Aziz A. Preparation of microcrystalline cellulose from oil palm empty fruit bunch fibre using steam-assisted acid hydrolysis. ARFMTS. 2021;81(1):88-98. doi: 10.37934/arfmts.81.1.8898.
Mat Soom R, Abd Aziz A, Wan Hassan WH, Top M AG. Solid-state characteristics of microcrystalline cellulose from oil palm empty fruit bunch fibre. J Oil Palm Res. 2009;21:613-20.
Fahma F, Iwamoto S, Hori N, Iwata T, Takemura A. Isolation preparation and characterization of nanofibers from oil palm empty-fruit-bunch (OPEFB). Cellulose. 2010;17(5):977-85. doi: 10.1007/s10570-010-9436-4.
Yugatama A, Maharani L, Pratiwi H, Ikaditya L. Characteristics testing of microcrystalline cellulose from Nata de Coco compared to Avicel pH 101 and Avicel pH 102. In: Current Breaktrough in Pharmacy Materials and Analyses Pharmaceutical Technology; 2012.
Cahyani IM, Lukitaningsih E, Adhyatmika A, Sulaiman TN. Preparation and characterization of microcrystalline cellulose for pharmaceutical exipient: a review. Trop J Nat Prod Res. 2022;6(10):1570-5. doi: 10.26538/tjnpr/v6i10.3.
Ali J, Saigal N, Baboota S, Ahuja A. Microcrystalline cellulose as a versatile excipient in drug research. J Young Pharmacists. 2009;1(1):6. doi: 10.4103/0975-1483.51868.
Maulana ML, Alfian M, Faizah N. Effervescent tablet formulation of turmeric extract (curcuma Domestica) with Artocarpus heterophyllus seed strach as a binder and siraitia grosvenorii as a sweetener. Int J App Pharm. 2025;17(1):94-100. doi: 10.22159/ijap.2025.v17s1.14.
Rum IA, Lestari H, Santoso R. Preparation and characterization of microcrystalline cellulose derived from Nata de Pina for use as an excipient in tablet formulations. J Pharmacopolium. 2019;1(3):149-61.
Tarigan AM, Panjaitan RM, Tampubolon A. Isolasi selulosa dari Tongkol jagung sebagai bahan pengisi pembuatan tablet klorfeniramin maleat cetak langsung. Jurnal Ilmiah PANNMED. 2019;10(1):1-10. doi: 10.36911/pannmed.v10i1.190.
Bala R, Khanna S, Pawar PK. Formulation and optimization of a fast-dissolving intraoral drug delivery system for clobazam using response surface methodology. J Adv Pharm Technol Res. 2013;4(3):151-9. doi: 10.4103/2231-4040.116785, PMID 24083203.
Janvalkar M, Kaloji P, Shettigar R. Microcrystalline cellulose in pharmaceutical formulations: a comprehensive review on applications concentrations and functional attributes. Int J Pharm Sci. 2025;3(4):3098-112. doi: 10.5281/zenodo.15283152.