
Int J App Pharm, Vol 18, Issue 1, 2026, 41-61Reviewl Article
TIMELINE MANAGEMENT STRATEGIES: ENABLING DEVELOPING COUNTRIES TO DELIVER COMPLEX GENERICS TO GLOBAL MARKETS
GEDALA VENKATA MURALI MOHAN BABU1, NI LUH PUTU NURSHANTI1, HARRY MARTHA WIJAYA1, RAYMOND RUBIANTO TJANDRAWINATA1,2*
1Formulation Department, Dexa Development Center, Dexa Medica, Industri Selatan VII Blok PP No. 7, Jababeka Industrial Estate, Cikarang, West Java-17550, Indonesia. 2Center for Pharmaceutical and Nutraceutical Research and Policy, Atma Jaya Catholic University of Indonesia, Jakarta Selatan-12930, DKI Jakarta, Indonesia
*Corresponding author: Raymond Rubianto Tjandrawinata; *Email: raymond@dexa-medica.com
Received: 17 Dec 2024, Revised and Accepted: 22 Oct 2025
ABSTRACT
This review analyzes effective project timeline management strategies for complex generic drug development in developing countries, addressing challenges like regulatory inefficiencies, limited infrastructure, and workforce constraints. It provides actionable recommendations to align product development with financial and organizational goals in resource-constrained settings. The paper highlights the growing role of developing countries in the global generics market and examines challenges specific to complex generics. It traces the evolution of timeline management from traditional methods to advanced project management tools, which have reduced development cycle times by 20-30% in adopting organizations. Key factors such as organizational size, workforce psychology, and cultural contexts are analyzed, with strategies to mitigate their impact on timelines. The review emphasizes establishing meaningful milestones, coordinating cross-functional activities, and addressing emerging challenges. It underscores the need to synchronize milestones across organizational, financial, and product factors, adapting lessons from established countries to developing nations. A multifaceted approach integrating regulatory expertise, advanced project management tools, and financial planning is proposed to overcome technical and regulatory hurdles while ensuring financial sustainability and market responsiveness. By adopting these principles, enterprises in developing countries can enhance their ability to supply high-quality generic medicines, improving healthcare accessibility and outcomes. The review aims to equip policymakers and pharmaceutical companies with actionable insights for optimizing development timelines, improving regulatory compliance, and enhancing cross-functional coordination.
Keywords: Complex generics, Developing countries, Timeline management, Pharmaceutical industry, Generic drug development, Project management, Regulatory compliance, Bioequivalence, Organizational culture, Workforce psychology
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.53438 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
In the complex world of the quality-driven pharmaceutical industry, the development of generic products is fundamental to ensure everyone everywhere has access to healthcare. These generic versions of popular drugs are more reasonably priced and bioequivalent to branded drugs, enabling more patients to get lifesaving treatments on time. By helping to manufacture and distribute generic medications, developing nations have a great chance to meet local healthcare requirements while establishing themselves as major participants in the global pharmaceutical markets. This is particularly important given that the global generic drugs market size, which was calculated at $468.08 billion in 2025, is estimated to grow to $728.64 billion by 2034 [1].
On the other hand, complex generics, which encompass products with complex active ingredients, formulations, delivery systems, or drug-device combinations, represent a growing segment of the pharmaceutical industry [2, 3]. These products include inhalers, long-acting injectables, ophthalmic suspensions, transdermal patches, and are particularly useful to patients with chronic conditions such as diabetes, asthma, schizophrenia, and cancer [4]. The specialized segment of complex generics is projected to grow from USD 90.68 billion in 2025 to USD 197.59 billion by 2035, at a compound annual growth rate (CAGR) of 8.1% [5]. Within this sector, complex injectables hold a dominant 63% share, while oncology is the leading therapeutic area at 37%, driven by increasing demand for affordable, high-complexity treatments, which also presents opportunities for industry leaders.
However, the development of complex generics is fraught with significant challenges, particularly in the area of timeline management, which is critical for ensuring timely market entry and patient access to these essential medications. These challenges are exacerbated in developing countries, where limited infrastructure, regulatory inefficiencies, and lack of access to advanced project management tools further delay development [2, 6-9]. For example, the introduction of generic versions of atorvastatin, the active ingredient in Pfizer’s blockbuster cholesterol-lowering drug Lipitor, by companies such as Mylan and Ranbaxy, stands as a prime example of successful timeline management in the generic drug industry. This launch, which occurred in 2011 following the expiration of Lipitor’s primary patent, was facilitated by effective project scheduling and regulatory strategy, leading to a dramatic increase in patient access to this essential medication at a significantly reduced cost [10].
This review distinguishes itself from existing literature [11-19], by offering a deeply focused analysis of timeline management strategies tailored to the unique resource constraints and complex regulatory environments prevalent in developing countries. Unlike broader reviews, it critically examines the transition from traditional to more sophisticated project management tools, highlighting their practical limitations and applicability in resource-constrained settings for complex generics development. Furthermore, it proposes actionable insights and strategic recommendations to empower emerging nations to address domestic healthcare needs while establishing themselves as formidable players in the global pharmaceutical market by mastering regulatory environments and optimizing their product portfolios [20]. While this review provides a comprehensive analysis of timeline management, it does not delve into the detailed chemistry, pharmacology, or specific clinical trial methodologies of complex generics, focusing instead on the strategic and operational aspects crucial for market entry and sustained global competitiveness.
This review aims to identify and analyze the key timeline management challenges in the development of complex generics, with a focus on strategies to overcome these challenges in developing countries. Specifically, it will explore how regulatory hurdles, supply chain inefficiencies, and limited access to advanced project management tools contributed to a 20-30% reduction in development cycle times in organizations that adopted these strategies [20], contributing to delays in the development and approval of complex generics.
To ensure a comprehensive and reproducible analysis, a systematic literature review was performed. The search strategy involved the use of databases such as PubMed, Scopus, and Google Scholar. Keywords and search terms included: “complex generics timeline”, “timeline management in complex generics”, “regulatory delays in generic drug development,” “project scheduling in complex generics,” and “bioequivalence studies complex formulations”. The search was limited to publications in English up to 2025, encompassing original research articles, review articles, regulatory guidelines (Food and Drug Administration (FDA), European Medicines Agency (EMA), and World Health Organization (WHO)), websites, and relevant industry reports. Studies focusing solely on traditional small molecule generics or unrelated to project timeline considerations were excluded.
This review article examines the problem of timeline management in the development of complex generics, particularly for countries wishing to position themselves as reliable suppliers on the global market. It critically analyzes the transition from traditional project management methods to more sophisticated tools and techniques, highlighting their limitations in the context of complex generics development, particularly in resource-constrained settings. Additionally, it addresses how emerging nations may overcome challenges and become key competitors in the global pharmaceutical industry by using suitable policies and mastering regulatory environments.
Importance of time: a critical factor in complex generic drug development
“Time” in the context of generic drug development is much more than the duration of the process. It can be viewed as one of the most valuable components that define the process and encompasses various stages of research and development till market authorizations and distribution. As the case may be with this pharmaceutical industry, the manufacturing process is complex, and there are high standards of regulation amidst the operations, which call for proper planning and management. Any delay in one or the other stage of development poses a serious threat that increases the costs of the process, misses the beneficial market window, and, as a result, impairs patients’ access to necessary medicines. The stakes are even higher in the developing countries as the consequences of the crisis bear a greater impact on them. To enter the international market of generics, the portfolio should prove the ability to launch a product on the international market within the shortest time, correspond to high quality standards, and know how to navigate through the legal/regulatory requirements of the country. If this is not done, it has adverse effects on credibility and limits access to lucrative markets [21-24].
Global equity, cost-effectiveness, policy implications, and ethical considerations in timeline management
The increasing prevalence of advanced therapeutic medicinal products and immunotherapies, while offering transformative potential, introduces complex challenges related to equitable access, particularly in low-and middle-income countries [25]. This disparity is exacerbated by the high costs associated with these innovative treatments, which often render them inaccessible within healthcare systems operating on constrained budgets [25]. This global inequity necessitates a concerted effort to address the multifaceted barriers, including scientific complexities, regulatory hurdles, economic constraints, and policy-related challenges, that currently limit the widespread adoption and accessibility of advanced therapies [26]. Addressing these issues requires a multi-pronged approach involving novel financial models, regulatory adaptations, and collaborative partnerships between academic institutions, pharmaceutical companies, and governments to ensure treatments reach all populations [27].
Global equity
Timeline management strategies play a crucial role in promoting global equity in access to essential medicines. Efficient timelines can expedite the availability of affordable generic medicines in developing countries, addressing critical healthcare needs and reducing disparities.
Equitable access: Streamlined regulatory processes and efficient development timelines can ensure that essential medicines reach patients more quickly, improving health outcomes and reducing health inequalities [28].
International collaboration and local production: Collaborative initiatives between developed and developing countries are vital for facilitating technology transfer, knowledge sharing, and capacity building [27]. This also implicitly supports local manufacturing capabilities in developing countries.
Cost-effectiveness
The cost implications of different timeline management strategies are significant, particularly for developing countries with limited resources.
Optimized resource allocation: Efficient timeline management can minimize development costs by reducing delays, optimizing resource utilization, and streamlining regulatory processes [29]. Accelerating drug development can yield financial benefits and improved affordability.
Value for money and strategic partnerships: Generic medicines offer significant cost savings for patients and healthcare systems [30]. Collaborations with international organizations and private sector partners are crucial for providing access to funding, expertise, and technology, thereby reducing the financial burden on developing countries.
Policy implications
Policy changes can significantly support more efficient timeline management and increase access to generics in developing countries.
Regulatory harmonization: Harmonizing regulatory pathways across different countries is crucial for streamlining approval processes and reducing duplicative efforts, which facilitates faster market entry for generics [31].
TRIPS flexibilities: Utilizing Trade-Related Aspects of Intellectual Property Rights flexibilities, such as compulsory licensing, enables developing countries to produce or import generic medicines even when patents are in force [32].
Incentivizing innovation: Implementing policies that incentivize pharmaceutical companies to invest in the development of complex generics can promote innovation and increase the availability of affordable medicines [33].
Ethical considerations
Balancing speed to market with ensuring product quality and safety is an ethical imperative in timeline management.
Quality assurance and regulatory oversight: Robust quality control systems, comprehensive stability studies, strict adherence to international standards, and effective regulatory oversight are essential to ensure the safety and efficacy of generic medicines, prevent substandard or falsified drugs from entering the market, and maintain public trust [34].
Transparency and accountability: Transparency in regulatory processes and accountability for product quality are crucial to build confidence in generic medicines and promote their widespread adoption. The balance between accelerating access to breakthrough therapies and generating robust evidence for their safety and efficacy presents ethical challenges [35].
By addressing these considerations, policymakers and pharmaceutical companies can work together to enhance timeline management, promote global equity, and ensure that patients in developing countries have access to safe, effective, and affordable medicines.
Brief outlay of development process of complex generics
Complex generic drugs are pharmacological formulations based on complex active pharmaceutical substances, routes of administration, or complex compositions that are generic copies of original drugs. The flow diagram shows that creating and developing complex generics needs a whole strategy, as depicted in fig. 1. There are challenges that are different for each phase that need to be handled completely in order to get regulatory approval and a successful launch into the market. Here are the most important phases in making complex generics [7, 36-43].
Early-stage planning
Regulatory Framework: The process begins with a thorough understanding of specific regulatory requirements (e.g., FDA guidance) and preparing the abbreviated new drug application (ANDA). This first phase must be finished in order to set up the project timeline and make sure that everyone follows the rules.
Intellectual Property Strategy: A full patent landscape assessment is needed to uncover methods around current patents. This investigation finds possible impediments. This step may save money by preventing expensive problems later on in the development process.
Reference Product Selection: The appropriate Reference Listed Drug (RLD) should be identified, and its complexity must be assessed to find out its suitability for establishing bioequivalence and comparability.
Development and manufacturing
Analytical Method Development: Robust analytical methods for the Active Pharmaceutical Ingredients (API), excipients, and formulation are developed and validated. Reliable analytical methods are essential for ensuring consistent product quality and comparability.
Quality by Design (QbD) Principles: QbD strategies find and reduce risks by creating a design space for important material characteristics and process parameters. Using QbD principles from the start makes it less likely that there will be problems with production and makes the product stronger.
Formulation Considerations: After looking at the RLD, formulation to find its most critical quality attributes (CQAs), a new formulation should be made that exhibits similar drug release and stability.
Manufacturing Process Development: Scalable manufacturing processes should be established, considering the complexities of specialized equipment. Process controls must be implemented to maintain consistency.
Technology Transfer: The manufacturing process should be optimized, and pilot batches should be manufactured for evaluation. Efficient technology transfer is crucial for scaling up production while maintaining product quality.
Stability Studies: Comprehensive stability studies should be conducted under various conditions to determine shelf life and evaluate compatibility with packaging materials.
Testing and regulatory submission
Bioequivalence Studies: Studies are designed to demonstrate bioequivalence to the RLD, including pharmacokinetic and pharmacodynamics assessments.
Clinical Considerations: If applicable, clinical studies are planned to evaluate safety and efficacy, ensuring therapeutic equivalence.
Regulatory Submission and Approval: Pre-submission meetings should be organized to present data and gather insights from regulators. A comprehensive ANDA submission package should be prepared.
Commercialization
Market Analysis and Commercial Strategy: To make sure the launch goes well, one needs to look at the market, the competition, and pricing strategies. The marketing strategy purpose is to set the sophisticated generic product apart from its competitors.
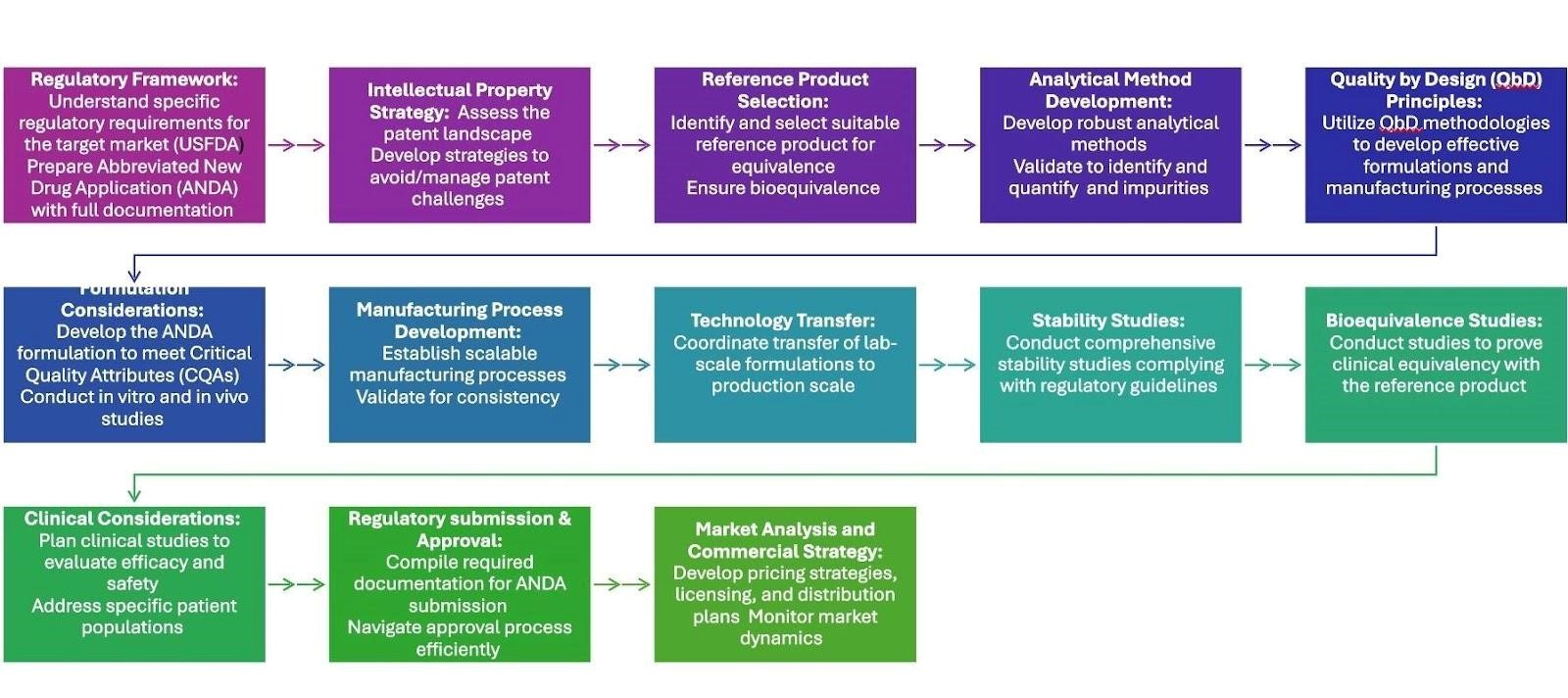
Fig. 1: Flowchart illustrating the development process for complex generic drugs, highlighting critical milestones such as formulation development, regulatory submissions, clinical trials, and market approval
The development of generic drugs can be divided into two main categories, as detailed in table 1: a) Simple Generics and b) Complex Generics, each with its own set of challenges and opportunities. Simple generics, which include straightforward products like tablets or capsules, are easier to develop because they often mimic the reference drug closely and use fewer ingredients. Their manufacturing processes are conventional, and they require less extensive clinical testing, focusing mainly on basic pharmacokinetic and physicochemical evaluations. However, the market for simple generics is highly competitive, leading to price pressures due to the sheer number of manufacturers. On the other hand, complex generics involve more sophisticated active ingredients, specialized delivery systems, or advanced manufacturing techniques like nanosizing or encapsulation. These products demand extensive research, more rigorous bioequivalence studies, and often face a complex patent landscape, especially around delivery systems or manufacturing processes. Regulatory hurdles are also higher, requiring more detailed data to prove equivalence to the reference drug. While the market for complex generics is less crowded due to the higher barriers to entry, their development is more resource-intensive, often leading to delays in production and market launch. In essence, simple generics offer a quicker, less complicated route but face fierce competition, whereas complex generics, though more challenging to develop, may provide a stronger market position once they overcome the initial hurdles. Understanding these differences is key for companies aiming to navigate the generic drug landscape effectively [44, 45].
Table 1: Differences between simple and complex generics and impact on timelines
| Aspects | Simple generics | Complex generics | Timeline implications [44, 45] |
| Definition | Typically involves drugs with straightforward compositions and delivery systems (e. g., tablets, capsules). | Involves products that are more intricate in API properties or their composition, delivery systems, or manufacturing processes or combination of drug-device. | More complex development leading to longer timelines. |
Patent Complexity |
Fewer patents; easier to manage. | While a large number of patents must be considered or navigated around device or delivery system-related patents, complex manufacturing process or composition. | Increased time for patent analysis, freedom-to-operate assessments, and potential litigation. |
| Resources | Resources such as raw materials, skilled manpower, manufacturing facilities are available and easier to manage. | Resources are very limited, and a definite commitment and effort are required to manage. | Resource scarcity may cause delays in procurement, training, and scaling manufacturing. |
Formulation Complexity |
Formulations have few excipients and are usually direct copies of the reference listed drug (RLD). | Formulations may include complex APIs, specialized delivery mechanisms, or complex excipient interactions. | Extensive formulation studies may prolong timelines. |
Manufacturing Processes |
Generally employs conventional manufacturing methods (e. g., tableting, capsule filling). | Often involves advanced manufacturing techniques (e. g., aseptic processing, emulsification, nanosizing, and encapsulation technology). | Scaling up production can be challenging and time-consuming. |
Clinical studies Requirements |
Less extensive bioequivalence (BE) studies are needed; often simpler pharmacokinetic assessments. | More extensive bioequivalence (BE) studies may be required, including consideration of in vitro, pharmacokinetic, and pharmacodynamic equivalence. | Proving bioequivalence (BE) can extend timelines significantly. |
Regulatory Requirements |
Less extensive studies are needed; often physicochemical assessments are sufficient. | More comprehensive data submissions may be required to demonstrate equivalence to the RLD and more studies. | Navigating regulatory pathways can take more time. |
Market Competition |
Competes in a more crowded market, often leading to price erosion due to many manufacturers. | Often faces fewer competitors due to higher barriers to entry related to complexity and development costs. | Slower competition may provide more time to enter market, but the complexity of products need more time. |
The increasing role of developing countries in global generics markets: opportunities and consequences
The dramatic rise of the global generics market over the past few years has exposed developing nations to exciting new opportunities to meet the growing demand for reasonably priced, high-quality pharmaceutical treatments. These countries have a one-of-a-kind chance to enhance their pharmaceutical manufacturing capabilities, consolidate their place in the global arena, and significantly contribute to improving healthcare accessibility on a global scale [8, 46]. Importantly, by supplying developing countries with affordable and high-quality generic medication goods, Indian companies have solidified their dominance in the global antiretroviral treatment sector. Increasing access to lifesaving Human Immunodeficiency Virus/Acquired Immunodeficiency Syndrome (HIV/AIDS) treatments in areas with low resources is largely attributable to their proficiency in developing affordable, FDA-approved antiretroviral medications [47].
It must be acknowledged, however, that emerging nations encounter substantial obstacles in the global generics market on a regular basis. Some of these obstacles include healthcare providers’ and patients’ generally negative views of generic medical products, complicated regulatory hurdles that make it difficult to enter the market, and murky pricing that makes people worry about whether or not they will be able to afford these lifesaving medications. In order to tackle these intricate problems, developing nations must quickly become the world’s go-to source for affordable, high-quality generics [48]. There are new and exciting opportunities for pharmaceutical companies in developing countries due to the emergence of biosimilars. These are pharmaceuticals that are biologically produced and closely resemble existing authorized biologics without major differences in their clinical properties. With their increasing knowledge of complex drug formulation and manufacturing, these nations can join the expanding biosimilars market and provide affordable, high-quality biosimilars that can increase global healthcare access to lifesaving biological treatments [46].
Pharmaceutical businesses in emerging nations are actively reshaping their strategies by cultivating a diversified talent pool in response to shifting patent laws. They are able to successfully manage global competitiveness and foster collaborative connections with stakeholders on a global scale because of their all-encompassing strategy. These companies may improve their knowledge in several areas, putting them in a good position to take advantage of the growth opportunities in the generics market [49].
In addition, the prevalence of substandard and fake medical supplies poses a serious threat to the healthcare system and the economy in many developing countries. Patients face serious health risks from these fake drugs, which may include inaccurate or hazardous ingredients, and the public’s faith in healthcare institutions might be eroded as a result. Consequently, developing nations must master the complexities of dealing with substandard and counterfeit drugs. Important pharmaceuticals cannot be tampered with if we want to keep improving public health and keeping the economy stable [50]. One possible response to these issues is to increase international trade in generics via market-driven initiatives [51].
The generics industry is now undergoing significant changes, which are expected to have far-reaching consequences for people and healthcare systems throughout the world. With the growing need for low-cost, high-quality generic medications, developing countries now have an unprecedented opportunity to shape the future trajectory of the generics industry. Such alterations are expected to significantly alter the accessibility and cost-effectiveness of basic pharmaceutical supplies, improving health-related results for varied people worldwide [52].
Challenges in developing complex generics in developing countries
The development of complex generic products poses significant challenges, particularly in less developed countries. Key issues include limited financial resources, not being able to get high-quality excipients and APIs, not having enough skilled workers, not having enough knowledge of complex formulation and analytical techniques, and not being able to make enough at the research and development (R&D) stage [53]. Also, proving bioequivalence through detailed analytical characterization and advanced bioequivalence testing, handling complicated manufacturing processes that need specialized technology and strict quality control measures, and dealing with complicated regulatory issues, such as a lot of documentation and following changing regulations, make things even harder [53]. Intellectual property laws and patent protections also make it harder for indigenous people to do research and share knowledge [54, 55].
In contrast, developed countries have implemented strategies to mitigate these challenges, offering valuable lessons for emerging markets. For instance, the United States Food and Drug Administration (USFDA) launched the Critical Path Initiative to modernize the scientific process of drug development, particularly for complex generics. This initiative emphasizes the use of advanced analytical tools, modeling, and simulation to streamline bioequivalence testing and reduce development timelines [40, 56]. Developing countries could adapt similar approaches by investing in advanced analytical technologies and fostering collaborations between regulatory bodies, academia, and industry to build local expertise [57].
Supply chain and raw material challenges
Developing countries often face difficulties in sourcing high-quality excipients and APIs, with many relying heavily on imports from a few dominant suppliers, such as China [58]. On the other hand, wealthy nations have made their supply chains more diverse and put strict quality control procedures in place to make sure that raw materials are reliable. For instance, the FDA’s Drug Supply Chain Security Act (DSCSA) sets up a system for keeping track of and tracing drugs to make sure there are no shortages and that they are of good quality [59]. Developing countries could adopt similar supply chain security measures and invest in local production of APIs and excipients to reduce dependency on imports [59].
Bioequivalence challenges
Proving bioequivalence is one of the most significant hurdles in complex generic development. Complex generics, such as extended-release injectable depot medications and topical dosage forms [60], require sophisticated testing to demonstrate equivalence in drug release mechanisms, stability, and pharmacokinetics [56]. The FDA has established product-specific guidances (PSGs) for specific complex generics in developed countries. These PSGs include thorough advice on how to perform bioequivalence studies for these products [39, 57]. These guides provide manufacturers clear, defined regulations to follow when doing bioequivalence testing, which may be quite complicated. Developing nations might use comparable guiding frameworks that are specific to their own situations. This would make bioequivalence studies clearer and more efficient [61].
The FDA has also pushed for the adoption of in vitro-in vivo correlation (IVIVC) models to cut down on the number of large clinical studies that are needed, especially for complex formulations like modified-release medicines [56, 61]. By leveraging IVIVC, manufacturers can predict in vivo performance based on in vitro data, significantly reducing costs and development time. Emerging markets might benefit from using these models, especially when there is not much clinical trial infrastructure [40, 43].
Regulatory hurdles
The lack of strong or uniform regulations in generic drug approvals is another major issue, particularly in developing countries [53, 62]. The Generic Drug User Fee Amendments (GDUFA) have simplified the evaluation of generic pharmaceuticals in the United States by increasing transparency and decreasing approval delays. Research in regulatory science to address challenging issues specific to generics, such as developing new methods to assess bioequivalence, is also funded by GDUFA [41, 63]. Developing countries should consider implementing comparable pricing structures to fund the development of their regulatory competence and the acceleration of the approval procedure for complex generics. Table 2 shows the details that the FDA employs to approve generics, which is the ANDA, in comparison with the European Union (EU) and India. The ANDA approval expected timeline is 12-30 mo based on complexity. For complex generics or those with modifications to approved drugs, the 505(b)(2) pathway is used, potentially extending the timeline to 12-60 mo depending on complexity [9, 64-66]. Two major issues are the complex 505(b)(2) procedure and the stringent BE criteria. While BE studies are necessary for all generics, additional clinical trials may be necessary for more complex generics [39]. Through pre-IND meetings and the issuance of product-specific guidance papers, the FDA provides firms with early feedback and provides them direction while developing complex generics meeting the requirements [67]. Developing countries should consider implementing similar procedures, which may help in developing complex generics faster.
Regulations are now more uniform throughout the European Union as a result of the Centralized Procedure, which allows a single marketing authorization to be valid in any EU member state [68]. For complex generics, such as transdermal patches or liposomal formulations, this approach streamlines the regulatory process by reducing superfluous processes [39, 61]. The Generic Medicinal Product Dossier is the procedure used for generics by the EMA, as shown in table 2. There will be a 210-day initial review period, but that may be extended to 12-18 mo if necessary. Time frames ranging from twelve to sixty months are used in the Hybrid Application for complex, modified generics. Similar to what is required in the US, BE studies are necessary, and further clinical trials could be necessary for complex generics [39]. The European Union also places a premium on data protection and authorizing products to sell only in their respective markets. Even if generic alternatives get the green light from regulators, this may delay their market entry. Data protection for eight years, market protection for two, and a possible extension of one year are all outlined in the “8+2+1” rule [70, 71]. Similarly, developing nations should study regional harmonization initiatives such as the African Medicines Agency (AMA) to establish standardized regulations that facilitate the approval and sale of complex generics in many countries simultaneously [72]. It is the hope of the AMA that standardizing regulations across all African nations would facilitate the approval of generic pharmaceuticals more quickly and easily, thus increasing access to essential medications.
In India, the Central Drugs Standard Control Organization (CDSCO) oversees the approval of complex generics through the Abbreviated New Drug Application pathway [73]. Referring to table 2, the CDSCO utilizes the Abbreviated New Drug Application for generics, with a general timeline of 6 mo or longer, depending on complexity. For complex generics with modifications, Form 44 is used, also potentially taking 12-60 mo. Bioequivalence studies are mandatory, and additional clinical trials may be required for complex formulations [74]. A key challenge is that CDSCO guidelines are less detailed compared to the US/EU, and pre-submission meetings are less formalized. This, coupled with regulatory delays and resource constraints, contributes to longer timelines. Post-marketing surveillance and Phase IV trials may also be required. Unlike the US and EU, India lacks specific guidance for complex generics, which can lead to uncertainties and delays. Moreover, the regulatory landscape in India is characterized by a complex interplay of central and state regulations, which can further complicate the approval process [44, 75]. Varying interpretations of regulatory requirements and inconsistent enforcement across different states can pose significant challenges for generic manufacturers [76]. By adopting more structured regulatory frameworks and harmonizing processes, developing countries can better manage the approval timelines for complex generics, ensuring faster access to affordable medicines.
Intellectual property and technology transfer
Intellectual property (IP) rights and patent protections make it hard for developing nations to make complex generics [54, 55]. However, developed countries have put in place ways to preserve intellectual property while also taking care of public health. For instance, the Hatch-Waxman Act in the US lets generic medication manufacturers fight patents and get generic pharmaceuticals on the market faster [77]. Developing nations might make similar changes to their laws to promote competitiveness and cut down on their need for imported pharmaceuticals.
In addition, voluntary license agreements and public-private partnerships have worked well in developed economies to let people share knowledge and technology [76]. For example, the Medicines Patent Pool (MPP) has made it possible for developing nations to make generic copies of protected pharmaceuticals by working out deals with patent holders. Emerging economies might use similar methods to get access to modern manufacturing technology and know-how for making complex generics.
Infrastructure and workforce development
It is much harder to make complex generics in developing nations since they do not have the right infrastructure or trained workers [62]. In developed markets, programs like the FDA’s Emerging Technology Program promote the use of new production technologies, such as continuous manufacturing, which may make things more efficient and better [78, 79]. Similar initiatives that concentrate on boosting local capacity via training, technological transfer, and infrastructure investment might help developing nations as well.
While the challenges of developing complex generics in less developed countries are significant, lessons from developed markets offer a roadmap for overcoming these obstacles. By adopting best practices such as the FDA’s Critical Path Initiative, product-specific guidances, and regulatory harmonization efforts, developing countries can streamline bioequivalence testing, reduce regulatory hurdles, and enhance local manufacturing capabilities [39, 40, 77]. Additionally, legislative reforms, voluntary licensing agreements, and supply chain security measures can facilitate technology transfer and ensure the availability of high-quality raw materials [57]. Ultimately, a comprehensive strategy that combines regulatory modernization, investment in infrastructure, and capacity-building is essential for emerging markets to fully harness the potential of complex generic drug development [61, 62, 80].
Table 2: Comparison of regulatory pathways and requirements for generic drugs across USA, EU, and India
| Aspect [81-87] | US (FDA) | EU (EMA) | India (CDSCO) |
Regulatory Pathway |
ANDA (Abbreviated New Drug Application) for generics. | Generic Medicinal Product Dossier for generics. | ANDA (Abbreviated New Drug Application) for generics. |
| 505(b)(2) for complex generics with changes to approved drugs. | Hybrid Application for complex generics with modifications. | Form 44 for complex generics with modifications. | |
Approval Timeline |
ANDA: 12-30 mo | Generic Dossier: 210 d however, it may increase to 12-18 mo as queries to be answered | AND: 6 mo and more based on complexity |
| 505(b)(2): 12-60 mo (depending on complexity). | Hybrid Application: 12-60 mo. | Form 44: 12-60 mo (due to additional requirements). | |
Bioequivalence Requirements |
Bioequivalence (BE) studies required for all generics. | BE studies required for all generics. | BE studies required for all generics. |
| Complex generics may require additional clinical trials. | Complex generics may require additional clinical trials. | Complex generics may require additional clinical trials. | |
| Clinical Trials | Phase IIII trials may be required for complex generics. | Phase IIII trials may be required for complex generics. | Phase IIII trials may be required for complex generics. |
Regulatory Flexibility |
FDA Guidance documents provide clarity for complex generics. | EMA Guidelines provide clarity for complex generics. | CDSCO Guidelines are less detailed compared to US/EU. |
| Pre-IND meetings available for early feedback. | Scientific Advice available for early feedback. | Pre-submission meetings are less formalized. | |
Post Approval Requirements |
Post-marketing surveillance and Phase IV trials may be required. | Post-marketing surveillance and Phase IV trials may be required. | Post-marketing surveillance and Phase IV trials may be required. |
| Key Challenges | Stringent BE requirements and complexity of 505(b) (2) pathway. | Complexity of hybrid applications and multicountry submissions. | Delays in approvals due to regulatory backlog and resource constraints. |
Impact on Timeline |
Longer timelines for complex generics due to additional requirements. | Longer timelines for complex generics due to multicountry submissions. | Longer timelines due to regulatory delays and lack of clarity. |
The evolution of timeline management: a historical perspective
Time management has been a fundamental part of human endeavor since the dawn of civilization, as individuals and companies battle with the challenges of appropriately organizing their scant resources. Time management theory is evolving as a four-stage process reflecting the changing points of view and methods used to maximize production and accomplish desired objectives [88].
Notes and memoranda help individuals record their tasks, appointments, and deadlines; therefore, early on, they were the most often used tools in time management [88]. As the concept evolved, the use of calendars and schedules became more evident, therefore enabling a more orderly approach to time management. The subsequent phase saw a move toward the emphasis on the notion of priority as individuals and businesses realized the importance of detecting and focusing on the most critical duties and activities. This approach aimed to simultaneously ensure that the most important objectives were fulfilled and balance capacity with productivity.
Particularly with data visualization and project execution, timeline management has a lengthy history. Constructing massive structures like the pyramids, ancient societies displayed project management skills [89, 90]. Originally seen in 1765, the first graphical timeline representation evolved into a standard tool for showing chronological events [91]. Timelines and basic tools for project management enable one to track progress and distribute resources [92]. Particularly in clinical research, they are very useful because they increase coordination and assist in reducing money waste [93]. In software development, timelines provide a means to address time-related issues like planning and late delivery [94]. Though they are crucial for historical information, historians have mainly neglected timelines [95]. Recent research indicates that evidence-based timelines improve project retrospectives by way of accurate memory and constructive discussions on past events [96].
The time management for the development of pharmaceutical products has evolved dramatically throughout the last several decades. Historically, long-term processes dominated drug development; this strategy is defined by the median of 36 years from research initiation to FDA approval. Recent studies, however, show that development schedules have not altered and that lengths of regulatory review have decreased [97]. Among other strategies to optimize timelines, the industry has adopted project portfolio management, critical chain project management, and systematic techniques to product design [98-100]. These methods aim to boost efficiency and concentrate on crucial housework. Factors influencing development timeframes include therapy category, route of administration, and company size [101]. The specific characteristics of pharmaceutical development, such as the need for extensive testing and vast alternative ways of generating data, might cause their complexity [102]. Sevoflurane illustrates how easily a global market might be reached and is considered as one good example [103].
Generic drugs provide unique challenges and need distinct strategies in development, unlike innovative drugs, which call for a sense of belongingness while working on generic product development. Aiming for products release around patent expiration dates [104, 105], generic manufacturers must manage challenging patent landscapes and exclusivity rights [104]. The generic drug development process, often shorter than that of innovators, has a median duration of 8.3 years from conception to submission [106]. Important steps include API selection, reference product selection, formulation development, and bioequivalent research [107]. Effective development requires careful planning, process standardization, and cooperation to simultaneously minimize time, cost, and quality [108]. Regulatory organizations strongly influence development strategies; initiatives like the FDA’s Critical Path focus on scientific concerns unique to generics [40]. Initial understanding of product life cycle management and patent tactics can help generic manufacturers negotiating the competitive environment [102, 109].
The current perspective of time management underlines even more the need for balancing, as psychologists and researchers highlight the need to address the individual’s information processing capacity and the intricate interplay among various aspects impacting productivity.
Effective time management has only become more important as managers and employees handle the ever-complex and fast-paced business environments. Direct influence on performance and career development makes the capacity to properly manage and utilize time a critical ability for achieving both personal and corporate goals. Studies on time management and other aspects of personal and organizational development have recently examined their linkages. Time management has been shown to enhance employee performance as it allows workers to prioritize their activities, make smarter decisions, and eventually assist the corporate objectives to be attained more effectively. Moreover, the concept of time management has been expanded from the personal level to the corporate one, as it is understood that effective time management at the organizational level may provide greater profitability, efficiency, and output.
In the end, the assessment of time management reveals the changing points of view and strategies used to maximize productivity and accomplish desired outcomes. As the concept shapes their performance, effective time management is still rather crucial for individuals as well as for companies.
Timeline management methodologies
Any complex development process depends critically on good time management as it ensures the effective completion of tasks, adherence to deadlines, and best use of resources. The efficient development of generics depends on the use of appropriate methods that solve the particular features of the project surroundings. Two well-known methods that provide different ways to effectively control deadlines are the Agile and Waterfall approaches.
Waterfall methodology
Because it gives the project manager total control over the full project lifecycle, the Waterfall Methodology [110] is often seen as the most efficient approach in project management. This rigorous approach ensures a comprehensive review and design stage, therefore enabling the team to address evolving system requirements. Requirements collecting, system design, implementation, verification, and maintenance comprise a sequential process differentiated by clearly defined phases in a Waterfall approach. It is important to create exact starting plans as every step needs to be completed before the following one begins.
Advantage of the waterfall methodology
Regarding structure and clarity, the Waterfall method provides a well-defined period that helps all stakeholders to understand their responsibilities. Well-stated deliverables highlight significant benchmarks, therefore encouraging responsibility. The Waterfall technique’s sequential approach guarantees adherence to regulatory criteria and seamless phase transitions by encouraging exact documentation at all levels. The project’s linear approach toward completion helps to increase its predictability and facilitates the projection of the completion dates. Still, the Waterfall technique could not be flexible enough to meet changing demands, which would result in large costs during the latter stages of the project [110].
Assessment of suitability for generic development
The Waterfall technique is especially good for the early stages of complex generic development, such as regulatory filings and the initial design phases, when precise documentation and following regulatory norms are very important. For instance, the Waterfall method worked well during the first step of regulatory filing for the development of a biosimilar product. This made sure that all the documentation fulfilled the FDA’s strict standards [111].
Agile methodology
Agile methods differ from the Waterfall approach in that their iterative character provides greater flexibility in meeting evolving needs [110, 112, 113]. Since the Agile development approach allows quick deployment of small, incremental software releases within short development cycles, it is particularly appropriate for complex and broad development projects. Teams try to provide operational components at the end of every sprint and actively engage in continuous feedback loops. Agile methodologies are particularly effective in environments characterized by uncertainty or shifting demands; thus, they are appropriate for developing broad ideas where fast iterations might provide important insights.
Advantages of agile methodologies
Agile teams’ agility and reactivity help them to quickly adapt to changes in project scope, increasing market demands, or legislative requirements. This degree of flexibility helps companies to make required changes at all phases of expansion. The iterative approach encourages constant communication and collaboration among stakeholders, hence improving transparency and inclusiveness in the decision-making process. Agile methods, by their focus on gradually releasing functional components, usually permit speedier market entry for generics than waiting for the completion of an entire project.
Assessment of suitability for generic development
Agile works very well in the final phases of generic development, such as clinical trials and post-market surveillance, where quick iterations and feedback are very important. For example, Agile methods were used during the clinical trial phase of making complex generics. This made it possible for the team to swiftly respond to new data and regulatory comments, which sped up the time it took to get the drug to market [114].
The agile approach presents challenges in the field of regulatory compliance, where tight adherence to predefined timelines and record-keeping are of great relevance, notwithstanding these advantages [115].
Combining agile with waterfall techniques
Organizations are seeing more and more the benefits of hybrid approaches, which combine Agile and Waterfall process elements. A hybrid approach lets project teams be flexible in iterative development and still provides a methodical foundation for critical procedures.
For the initial regulatory filings and general project structure, a project could use a Waterfall technique, for instance, while using agile practices all through the development phases, including rigorous examination and changes based on feedback from stakeholders. By allowing companies to adapt their approach to the particular needs of every project, this hybrid model helps them to increase operational efficiency and reduce time estimates [116]. Any complicated development process depends critically on it, as it guarantees successful completion of tasks, meeting of deadlines, and efficient use of resources [117].
Evaluation of suitability for generic development
The hybrid technique is especially useful for complex generic development projects that need to follow strict regulations but also be able to change as needed. One real-world example of this is the creation of a biosimilar generic product. The hybrid approach was used to make sure that the development process followed all the regulations during the early stages, while also allowing for improvements to be made during the clinical development phase. This made the process more efficient and compliant [118, 119].
Tools and approaches to be considered for the timeline management of complex generics
Complex generic drugs pose unique challenges for regulatory approval due to their intricate formulations, delivery systems, or active ingredients [39]. Regulatory frameworks for these products are often unclear, necessitating a combination of in vitro, pharmacokinetic, and pharmacodynamic studies to demonstrate bioequivalence [120]. The FDA and industry are exploring quantitative methods and modeling approaches to support complex generic development, including physiologically-based pharmacokinetic modeling and data analytics [42, 120]. Research priorities identified include complex injectables, drug-device combinations, and inhalation products [41]. Regulatory agencies are working to provide clearer guidelines and scientific advice to facilitate approvals [9]. Advanced project management tools may be beneficial for managing the complexity of these drug development projects [121]. Overall, there is a need for harmonization of regulatory frameworks to streamline the approval process for complex generics [36]. Fig. 2 presents various timeline management tools and associated processes as a mind map to illustrate the possible options.
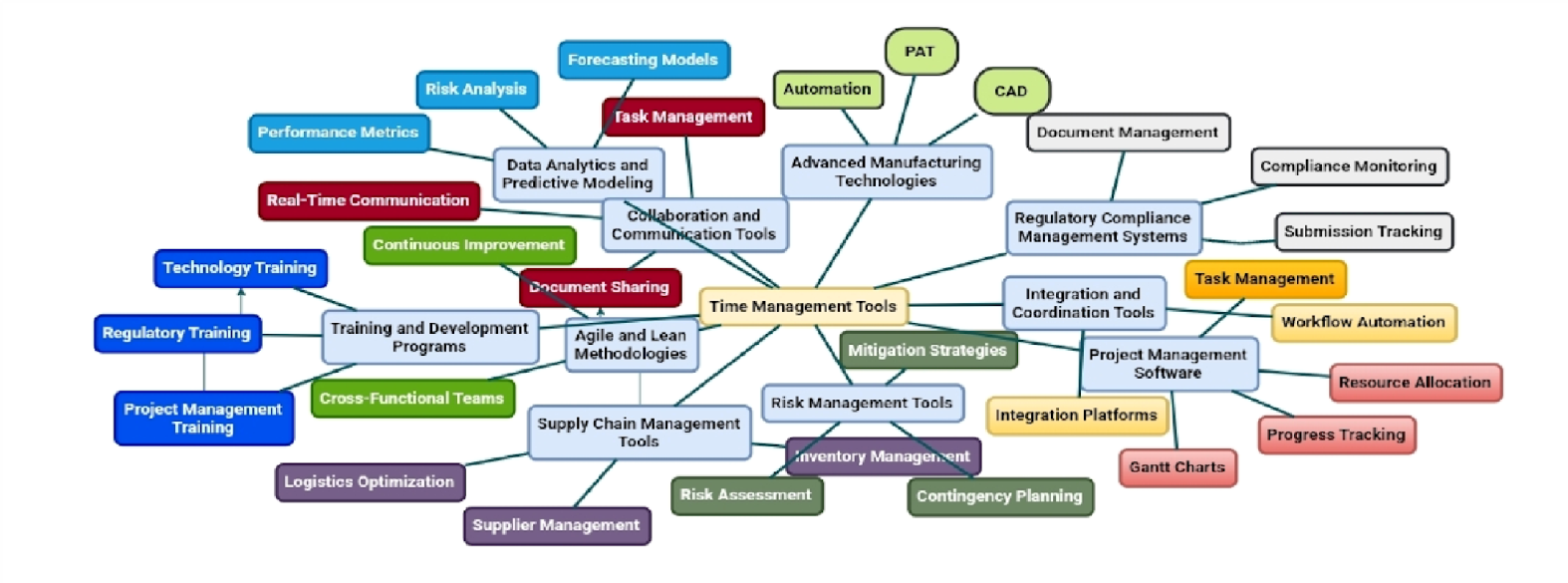
Fig. 2: Overview of timeline management tools and associated processes
The major timeline management tools for complex generics are: 1) Project management tools; 2) Advanced manufacturing technology; 3) Data analysis and predictive modeling; 4) Supply chain management tools; 5) Regulatory compliance handling mechanism; 6) Agile and lean approaches; 7) Risk management tools; 8) Collaboration and communication platforms; 9) Training programs; and 10) Integration and coordination technologies to help manage the timeline for complex generic drug development.
Project management software, such as Gantt charts, task management, resource allocation, and progress tracking platforms like Microsoft Project, Asana, and Trello, are all examples of tools and methodologies for managing complex timelines. Advanced manufacturing technologies, such as automation, process analytical technology (PAT), and computer-aided design (CAD), may help to simplify production and enhance quality consistency. Data analytics and predictive modeling, including forecasting models, risk analysis, and performance indicators, may improve timeline planning and forecasting utilizing tools such as Tableau, R, and Python. Inventory, logistics, and supplier management solutions, as well as regulatory compliance systems for document organization and submission monitoring, are all useful to improve timeliness. Agile and lean approaches, risk management tools, collaboration and communication platforms, training programs, and integration and coordination technologies may help to optimize the timelines for complex generic drug development to avoid delays.
Pharmaceutical businesses that strategically use these tools and techniques may negotiate the particular obstacles of generic drug development and launch the products effectively while guaranteeing quality and compliance.
Influence of size of organization, culture of organization and country and psychology of workforce in managing timelines effectively
Managing timelines effectively within organizations is vital for successful project completion and is shaped by various factors, including the organization’s size, its culture, the national culture, and the psychological aspects of its workforce. Effective project managers need to grasp not only the technical details of a project but also the economic, legal, institutional, and psychological elements that can affect the project’s schedule.
Previous research has shown that the scope of an organization has an influence on its culture, staff morale, and overall performance effectiveness [122, 123]. Larger corporations tend to possess more organized procedures and superior planning skills, although smaller enterprises may exhibit exceptional proficiency in information exchange [124]. Effective time management strategies are crucially dependent on the organizational culture [125]. Firm size and experience directly impact its capacity to handle project delays, with smaller and younger enterprises encountering specific difficulties in site operations and financial management [126]. To optimize effectiveness, organizations should balance the extensive resources of larger companies with the agility of smaller ones [127].
Both organizational and national cultures play significant roles in time management and project outcomes. Elements, such as management support and employee empowerment, enhance time management practices [128]. National cultural dimensions, like individualism versus collectivism, affect time perception and decision-making [129]. Different cultures approach management and planning differently: Anglo-American cultures tend to emphasize decentralization and quantitative methods, while East Asian cultures focus more on long-term planning and collective decision-making [130]. Multicultural initiatives include both obstacles and possibilities arising from diversity, necessitating meticulous management throughout the project’s lifespan [131]. Organizational problems usually cause project delays, which emphasize the importance of effective project management and education in developing countries [132]. Success in cross-cultural management and international business depends on a thorough awareness of these cultural aspects [133, 134].
To properly control schedules and improve output, one must first grasp the psychology of labor management. Crucial factors influencing organizational success include temporal perception, motivation, emotional state, and communication abilities [135, 136]. Effective workforce management techniques, such as regular performance reviews and helpful criticism, may greatly increase worker performance [137]. Modern HR management depends on psychological approaches being included in human resources practices [138]. Using techniques combining psychological understanding with efficient time management could greatly increase output [139]. Managers should create a good psychological environment if they want to improve team dynamics and general performance [140]. Effective time management and the maximization of staff productivity depend on an awareness of these psychological aspects [141].
In essence, effective time management in businesses calls for a careful mix of components, including the size of the company, its culture, the national culture, and the psychology of the employees. Dynamic project managers have to be adept in managing these elements to ensure the timely and effective completion of initiatives. Hence, a summary of thoughts on the above aspects is presented in the following sections.
Timeline management in large pharma companies
Major pharmaceutical companies depend on strategic time management as their sophisticated organizational structures and large resources provide new opportunities and challenges [142]. Usually, delays in decision-making result in delays in research and regulatory affairs; organizations of this nature sometimes confront complicated hierarchies and specialized divisions, which may also cause significant coordination problems [93, 143]. Complex bureaucratic systems might hinder the decision-making process as project changes must be approved by many tiers of management, causing probable delays [143].
Project plans are heavily influenced by organizational structure [144]. In large companies, multi-tiered management systems may lead to protracted approval processes as changes must pass through many tiers of hierarchy [143]. Specialized departments, such as regulatory affairs, might provide challenges that greatly affect the general progress of a project [93]. Minimizing these delays and ensuring that every department runs in harmony depend on open communication and cooperation.
Effective time management depends much on the distribution of resources. Large-scale pharmaceutical companies with significant resources, such as modern technologies and highly qualified staff, that ideally enable comprehensive planning and quick issue resolution, have great power. Still, the way these resources are coordinated across many departments may cause inefficiencies [142]. One example of this is the possible domino effect of delays in procuring necessary technology on project plans. Simplified communication and flexible resource management techniques are thus essential for the optimization of resource utilization and avoidance of major losses [21].
Allocating resources well across many departments has advantages as well as challenges [142]. Enough resources provide the ability to implement effective planning and risk management strategies, even if the complex character of creating specialized teams may lead to communication problems and different objectives [143, 144]. Along with careful planning and advanced forecasting models, effective resource management may improve project outcomes [144].
To effectively manage schedules, big pharmaceutical companies ultimately have to be proficient at negotiating complex organizational structures and large resources. Big corporations face challenges related to bureaucracy, coordination, and resource efficiency, even if their size and experience provide benefits [142, 143]. To increase resilience and sustained competitiveness, future plans should give integrated communication technologies [21], adaptive project management approaches [144], and proactive risk management top priority. In the dynamic pharmaceutical sector, the precise balance between controlled processes and flexibility guarantees rapid market reaction and helps to optimize timeline management.
Psychological elements influencing time management at work environments
Effective management of timelines depends on an awareness of the psychological aspects related to the workforce, both on the team level and throughout many functional divisions [145]. The psychological components directly affect the ability of team members to follow deadlines and achieve project goals by influencing the interactions, communication channels, and cooperative efforts among them. The upcoming discussion focuses on important psychological elements influencing timeline management in team configurations and within the many roles performed in a company.
Team dynamics and their connection to timeline management
Cohesion and trust
Observable cohesiveness among team members creates a trusting atmosphere and supports open lines of communication elements judged essential for effective teamwork and timely project completion. The inclination to share relevant information, voice concerns, and ask for help is much enhanced when team members have high levels of trust. Higher degrees of cohesiveness might match faster resolution of issues and a unified approach to meet deadlines. On the other hand, a lack of confidence might lead to reluctance to face clear problems, compromising accepted deadlines.
Motivation and engagement importance
One assumes that employee engagement and productivity levels are much influenced by the degree of motivation [146]. Teams marked by increased drive show resilience in facing obstacles and show a tendency toward responsibility for their given duties. Teams driven by inspiration generally set deadlines and adhere to them more effectively. On the other hand, team members who show disengagement might later fail to achieve deadlines because of less dedication or enthusiasm for the current project.
Conflict resolution
The relevance of conflict resolution is evident as team dynamics naturally lead to conflicts [147]. The way disagreements are handled may greatly affect the way timeframes are managed. Using successful conflict resolution strategies typified by open communication and negotiation can improve group projects. Quick resolution of problems enables teams to maintain concentration on their activities with respect to deadlines. When disagreements continue, however, diversion occurs, which reduces output and results in delays.
Understanding and appreciating diverse perspectives
Every functional division, including operations, marketing, and finance, adds special skills and knowledge to each project. Acknowledgment and respect of these many points of view is probably going to increase imagination and improve ability to solve problems. Collaborative efficacy rises and agreement is reached more quickly when team members acknowledge and respect contributions made by others, resulting from different roles, hence optimizing timeframes [148]. On the other hand, the existence of misinterpretation or a lack of respect among many participants might cause delays in project execution or decision-making.
Communication styles
Different communication styles and terminology are used in functional divisions sometimes; therefore, causing misunderstanding. Good teamwork depends on skillful navigation of these communication differences. Clear and regular communication helps teams to be in line and guarantees group understanding about deliverables and schedules. Inadequate communication may lead to differences of expectations and unsatisfied deadlines.
Shared goals and accountability
The development of common goals across functional divisions, along with the encouragement of an accountable-centric culture, might help to strengthen cooperation and increase dedication to a deadline [148]. Effective coordination of activities results from team members across functions realizing their various roles in the pursuit of a shared aim; therefore, supporting deadline compliance. On the other hand, a lack of responsibility might lead to uneven contributions that finally cause delays.
Role of emotional intelligence
Self-awareness and regulation
People with high degrees of emotional intelligence have a better capacity to control their own emotional reactions, which influences team dynamics as well as interrelation dynamics across operational departments [149]. Self-aware team members may identify events when their emotional states might interfere with their job responsibilities or social contacts. This understanding helps one to be more inclined toward more positive behavior and cooperative efforts kept in accordance with accepted project schedules.
Empathy and relationship management
Empathetic team members may more fully understand the points of view and needs of their colleagues, therefore creating an environment fit for support and cooperation [151]. A workplace marked by compassion creates conditions fit for honest communication and a desire to help each other. This then stimulates improved capacity for addressing problems and adherence to given deadlines.
Motivation theories in timeline management
Intrinsic vs. extrinsic motivation
Understanding the underlying causes of employee motivation, whether they come from extrinsic sources (rewards, recognition) or internal ones (personal fulfillment), will help greatly guide the strategies used in establishing and controlling deadlines [148]. Projects that provide opportunities for intrinsic motivation, such as challenge and autonomy, often help to maintain involvement and meet deadlines more successfully. On the other hand, those who rely exclusively on extrinsic motivators may see a decrease in excitement should recognition attempts lack timeliness or sufficient.
Goal-setting theory
Setting particular, quantifiable, reasonable, pertinent, and time-limited objectives might help to increase motivation and focus [148]. Members of a team who have unambiguous, precisely defined objectives that fit up with project schedules are likely to participate actively in trying to meet those deadlines. On the other hand, vague or too high objectives might cause irritation and result in noncompliance with deadlines.
In summary, effective management of deadlines depends much on the psychological aspects of the workforce, which include intra-team relationships and interdepartmental dynamics. The way teams work and fulfill their temporal responsibilities is much influenced by elements like trust, motivating factors, conflict resolution techniques, communication modalities, emotional intelligence, and goal articulation. By comprehensively understanding and addressing these psychological characteristics, companies may improve their project management strategies, thereby improving compliance with deadlines and supporting overall project success. Adoption of policies emphasizing psychological well-being and a collaborative culture would not only help to maximize schedules but also produce a staff more involved and productive.
Role of culture of country and organization in timeline management
Controlling deadlines goes beyond just noting events on a calendar. It is shaped by the cultural setting of the nation as well as the engaged company. Knowing these cultural subtleties will help one establish, present, and follow timelines more effectively [152]. The following sections will help to explain how the culture of an organization or country influences the performance of timeline management.
Timeline management and national culture
Timelines are regulated in great part by national culture; the common values, beliefs, and actions of individuals from a certain nation.
Time orientation
Different societies have different conceptions of time. In mono-chronic societies, such as Germany and the United States, for instance, time is seen as a straightforward flow. Essentials are timeliness and following a timetable. In poly-chronic societies, on the other hand, those seen in many Mediterranean and Latin American nations, relationships can take precedence over rigorous deadlines, resulting in more flexible project timelines.
Communication styles
Deadlines are spoken about differently in several civilizations. High-context cultures, including Japan and many Arab nations, rely on nonverbal signals and indirect communication. Sometimes this leads to misperceptions about deadlines. Conversely, low-context societies such as those of the Netherlands and Scandinavian nations value straightforward, unambiguous communication, which facilitates timeline management and understanding.
Risk of tolerance
Different societies also show different tolerance to uncertainty and danger. For nations like Greece and Japan, which have high uncertainty avoidance, for instance, there is a strong inclination for thorough preparation and set deadlines to prevent shocks. Conversely, nations with less uncertainty avoidance, such as the United States and New Zealand, are often more flexible in changing their schedules when conditions do.
Hofstede’s cultural dimensions and their impact on timeline management in complex generic pharma projects
Hofstede’s Cultural Dimensions Theory provides a robust framework for understanding how cultural differences influence project management, particularly in the pharmaceutical industry, where complex generic drug development involves rigorous regulatory compliance, scientific challenges, and cross-border collaborations [151]. Developing countries often face additional hurdles due to varying cultural norms affecting decision-making, risk tolerance, and team dynamics within project management contexts. We expand on each dimension with real-world examples and suggest mitigation strategies to enhance efficiency as follows.
Power distance – centralized decision-making delays
Power Distance measures societal acceptance of hierarchical authority. High power distance index (PDI) cultures (e.g., India, Mexico) typically exhibit more rigid organizational structures, which can slow down approval processes [152].
Impact on complex generics projects
In high PDI environments like India, regulatory submissions for complex generics may involve multiple layers of hierarchical clearances, potentially extending timelines. Similarly, project decision-making in high PDI settings might be centralized, requiring extensive approvals before local managers can act autonomously.
Mitigation strategies
Pre-approval engagement with regulators to foster direct interaction and potentially reduce bureaucratic delays.
Empower mid-level managers with clearly defined decision-making authority for specific aspects of projects.
Incorporate buffer timelines into project schedules to account for potential hierarchical approval delays.
Uncertainty avoidance – regulatory hesitation and risk aversion
Societies with a high uncertainty avoidance index (UAI) (e. g., Japan, Germany) prefer structured processes and resist ambiguity [153].
Impact on complex generics projects
Regulatory bodies in high UAI countries, such as Japan’s Pharmaceuticals and Medical Devices Agency, may mandate extensive bioequivalence studies and detailed data requirements, which can prolong drug approval timelines. This preference for thoroughness can also lead project teams to engage in over-planning, potentially delaying the initiation of manufacturing or other project phases.
Mitigation strategies
Submit precedent-based regulatory packages, referencing approvals from other stringent agencies to streamline reviews where acceptable.
Adopt adaptive clinical trial designs that allow for flexibility and adjustments based on accumulating data and regulatory feedback.
Utilize predictive analytics to inform risk assessments and potentially accelerate decision-making processes.
Individualism vs. collectivism – team dynamics and decision speed
Collectivist cultures (e. g., China, Brazil) prioritize group harmony and consensus, which can influence decision-making speed but enhance collective execution [154]. Individualist cultures (e. g., the US) typically favor individual initiative and faster decision-making, though this can sometimes lead to coordination gaps.
Impact on complex generics projects
In collectivist environments, achieving broad stakeholder alignment, such as required by Brazil’s ANVISA for certain generic approvals, might extend initial decision-making timelines. Cross-cultural teams, particularly those involving partners from collectivist and individualist backgrounds, can experience communication challenges or misunderstandings that impact project flow.
Mitigation strategies
Localize project ownership by appointing in-country liaisons who can bridge cultural gaps and facilitate communication.
Conduct pre-study alignment workshops for collectivist teams to establish shared objectives and foster consensus early on.
Implement integrated project management software to centralize communication and improve coordination across diverse teams.
Long-term vs. short-term orientation – Balancing speed vs. quality
Long-term cultures (e. g., South Korea) often emphasize future rewards and perseverance, which can translate into a willingness to invest in lengthy research and development cycles [98]. Short-term cultures (e. g., the US) tend to focus on immediate results and quick commercialization.
Impact on complex generics projects
In long-term-oriented pharmaceutical markets, companies may accept extended development cycles for complex generics to ensure comprehensive quality and long-term market success. Conversely, a strong short-term orientation might lead to pressure to accelerate regulatory filings, potentially resulting in incomplete submissions or quality issues.
Mitigation strategies
Implement milestone-driven contracts to ensure quality benchmarks are met at various stages of development.
Educate stakeholders on the long-term cost implications of rushed submissions, including potential rework and regulatory delays.
Align incentives to reward both timely progress and adherence to quality standards throughout the development process.
Masculinity vs. femininity – productivity vs. sustainability
Masculine cultures (e.g., Japan) generally value assertiveness, competition, and achievement, which can drive aggressive deadlines and a strong focus on productivity [155]. Feminine cultures (e. g., Sweden) prioritize cooperation, quality of life, and work-life balance, potentially leading to a more measured approach to project timelines.
Impact on complex generics projects
In highly masculine cultures, project teams might experience pressure to meet demanding deadlines, which could elevate the risk of burnout or errors if not managed effectively. Conversely, in feminine cultures, a stronger emphasis on employee well-being and sustainable practices could influence project scheduling, such as accommodating vacation periods or fostering work-life balance.
Mitigation strategies
Implement rotational staffing models to manage workload and prevent exhaustion in high-pressure environments.
Incorporate flexible deadlines where appropriate, particularly in regions that prioritize work-life balance.
Introduce wellness programs and support systems to maintain team health and productivity over the long term.
Hofstede’s model highlights how local norms affect timelines in pharmaceutical projects. By tailoring strategies, such as adaptive trials for high-UAI regulators or decentralized approvals in high-PDI countries, developing markets can accelerate complex generic drug development without compromising quality. This approach not only optimizes regulatory navigation but also enhances the global accessibility of essential medicines, fostering sustainable growth within the pharmaceutical sector. Furthermore, understanding these cultural nuances facilitates more effective cross-border collaborations and strategic market entry, ultimately fostering a competitive yet ethically grounded global pharmaceutical landscape.
Organizational culture and timelines management
Organizational culture means shared values, common beliefs, and practices that influence how an organization operates [156]. The culture of the organization plays a very crucial role in the handling of timelines, as explained below.
Leadership style
Approval of adjustments to timelines may be sluggish and difficult in hierarchical companies; therefore, possibly postpone development. On the other hand, in companies with a flat structure, decisions are sometimes taken faster, which allows more flexible schedule changes [157].
Cooperation against individualism
Companies that respect cooperation usually establish deadlines from many angles, which help to create more reasonable deadlines [158]. Conversely, companies that prioritize personal performance might feel under pressure to reach deadlines at the price of quality, as the emphasis is on individual successes [159].
Change readiness
Timeline management is significantly influenced by the degree of change an organization can accommodate. Like IT startups, creative and agile businesses are often better at changing their schedules depending on fresh ideas. Organizations that are opposed to change, on the other hand, might find it difficult to modify their timelines, therefore causing possible delays and dissatisfaction [160].
Intersection of national and organizational culture
Managing timeframes may become more complex when national and corporate cultures cross [161]. For example, a global firm running in a nation with a more flexible attitude to time may have to negotiate these variations to match local cultural norms with corporate objectives.
Cultural training
Giving cultural training may be very beneficial for businesses operating across several cultures. It helps staff members appreciate different ways of handling timeline management, therefore improving cooperation and reducing misunderstandings [162].
Tailored strategies
Better project results may result from customizing timeline management techniques to represent national as well as company cultures. When working with teams from cultures that stress relationships, for example, allowing flexibility in timeframes can improve teamwork and project success.
Control of project schedules calls for much more than just establishing deadlines. It calls for knowledge of the country and corporate cultures. Leaders and project managers who value these cultural aspects will be able to properly express objectives, create more reasonable timeframes, and foster improved cooperation in many environments. Respecting and adjusting to cultural diversity helps to balance schedule control and support more effective initiatives.
Creating milestones across product, finance, and organizational dimensions and prioritization
Successful new product development in the healthcare technology industry, including the generic pharmaceutical sector, requires a multidimensional approach that aligns the product, finance, and organizational axes of the value creation process [163]. This approach helps companies manage the interplay between these essential dimensions and achieve critical value milestones along the product development journey.
As outlined in the literature, the key principles of successful new product development include market advantage, product readiness, value-driven program execution, learning and confirming, and regulatory collaboration [163]. These principles must be carefully balanced and adapted to the specific needs of each generic product development program, guided by industry experience and informed decision-making.
Effectively coordinating the value milestones across these three dimensions: product, finance, and organization, is crucial for navigating the complexities of new generic product development. In today’s competitive business environment, organizations must prioritize the alignment of their product development initiatives with financial objectives and overarching organizational goals. Coordinating value milestones across product, finance, and organizational dimensions not only maximizes the potential for success but also enriches the decision-making process. Fig. 3 below depicts how valuable the milestones and subdivisions can influence timeline management.
Value milestones are critical performance indicators that monitor progress, facilitate communication, and encourage accountability throughout the product development lifecycle. These milestones, whether quantifiable (e. g., revenue targets) or qualitative (e. g., consumer feedback), are critical for measuring progress and engaging stakeholders. The optimal synchronization of value milestones includes three key aspects: product, finance, and organization [58, 164].
Fig. 3: The influence of milestones and subdivisions on timeline management
The Product dimension spans the whole product lifecycle, from concept creation to final delivery, with a focus on user feedback and agile development methods. In this context, tactics such as cross-functional team integration, modular design adoption, and continuous improvement promotion are applied. The Finance dimension focuses on economic feasibility, such as budget calculation, return on investment analysis, and risk reduction. Financial plans include the creation of detailed models, the implementation of scenario studies, and the ongoing engagement of stakeholders. The Organizational dimension ensures alignment with the organization’s strategic goals by emphasizing cultural aspects and active engagement of leadership. This dimension’s techniques include establishing clear governance structures, managing change efficiently, and aligning performance incentives to encourage cross-departmental collaboration [165, 166].
By synchronizing product, finance, and organizational value milestones, companies can enhance their ability to develop high-impact solutions, manage resources effectively, and foster a culture of innovation and accountability [167-173]. Following workflow diagram for milestone prioritization while developing complex generics depicted in fig. 4, which are broken down into five key phases: Initiation, Planning, Execution, Monitoring and Control, and Closure. Each phase is further organized into three dimensions: Product, Finance, and Organizational. Each dimension includes specific tasks and considerations relevant to that phase. The diagram provides a comprehensive overview of the activities and focus areas across the entire lifecycle, ensuring a holistic approach to milestone prioritization.
The harmonization of milestones across product, finance, and organizational dimensions calls for integrated planning. This cohesive approach facilitates bridging gaps between various functions and enables companies to maximize value creation. Table 3 highlights the benefits and implementation strategies to improve project performance.
Challenges in coordination
While the benefits of coordinating value milestones are clear, organizations may face several challenges in this endeavor.
Table 3: The benefits and implementation strategies to improve project performance
| Benefits [174-178] | Implementation |
| Improved coordination | Break down tasks into smaller activities, assign responsibilities, and use project management software. |
| Efficient resource allocation | Use resource management tools to assign resources based on project requirements and availability |
| Enhanced visibility and transparency | Implement a centralized system or dashboard to monitor project timelines and progress. |
| Potential for faster project delivery | Analyze dependencies, critical paths, and optimize task scheduling to reduce project completion time |
| Risk mitigation | Identify and manage risks proactively by considering potential delays and incorporating contingencies. |
Fig. 4: Workflow diagram for milestone prioritization of complex generics
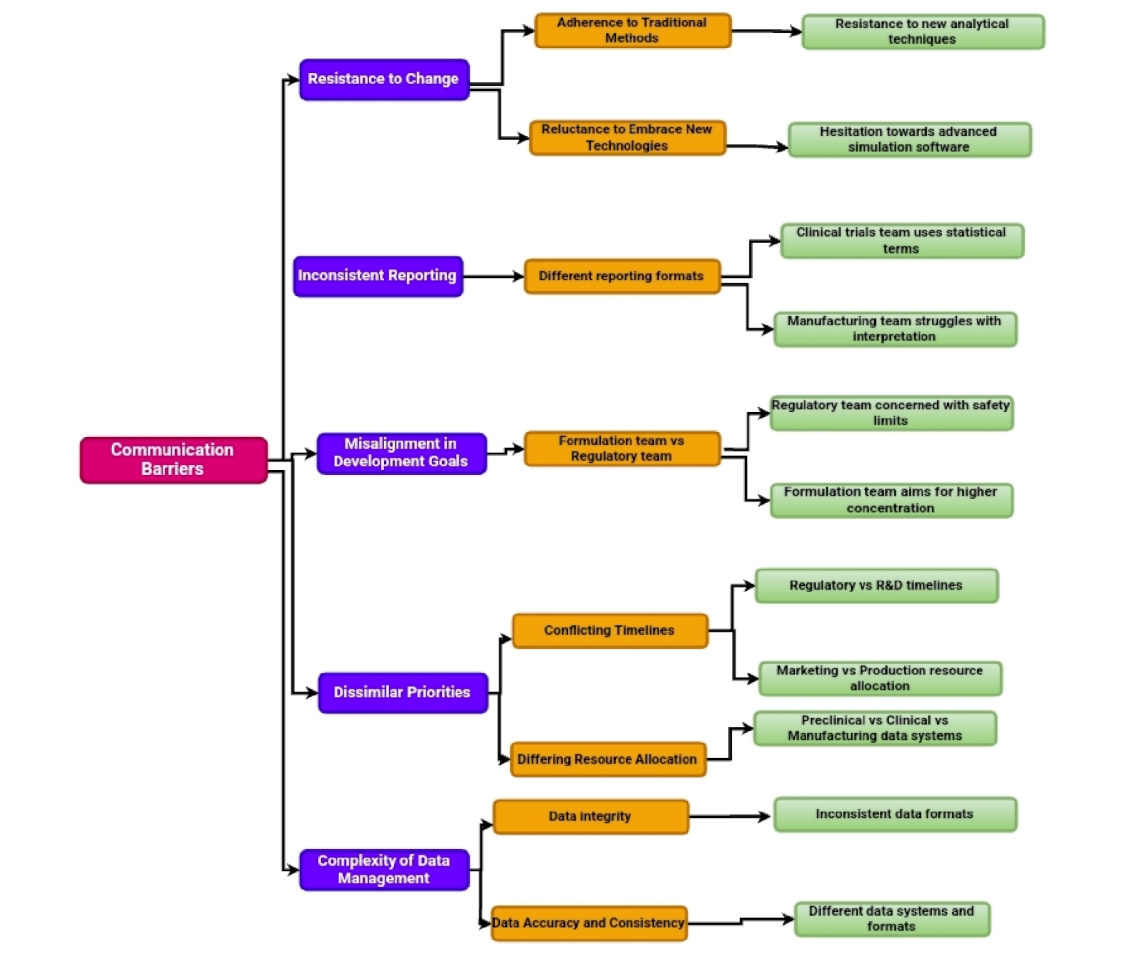
Fig. 5: Types of communication barriers in an organization
Fig. 5 shows some of the most common communication problems that come up throughout the development process. These problems come from a number of main causes, such as resistance to change, inconsistent reporting, development goals that do not match up, agendas that are at odds with one another, and the challenges of managing data. Resistance to change may lead to inefficiency if people refuse to alter their methods of doing things and do not want to use new technology. Different ways of reporting might make it tougher for people to understand each other and work together. Development teams often have different points of view, which may lead to conflicting aims. This is also frequent within formulation and regulatory groups. When schedules do not match up, priorities are different, and resources are not evenly distributed, it makes communication increasingly harder. Data management may be complicated, which can make it hard to communicate and make decisions quickly. This is because there can be problems with data integrity, accuracy, and consistency across numerous data systems and formats.
Best practices for effective coordination
To successfully coordinate value milestones across product, finance, and organizational dimensions, organizations can adopt the following best practices:
Regular training and development: invest in training programs that promote cross-functional understanding and develop skills necessary for collaboration.
Fostering a collaborative culture: promote a culture of respect and collaboration where contributions from all teams are valued. This can lead to a more engaged workforce and better decision-making.
Establishing metrics for success: create clear and quantifiable metrics that define success for each dimension, ensuring that all teams are aligned and accountable.
Emphasizing leadership support: ensure that leadership is visibly supportive of collaborative efforts and continually communicates the importance of value milestone coordination.
Utilizing technology: leverage software solutions that facilitate project management and enhance communication across teams.
Strategies for achievement: a framework for timeline management efficiently
In the quest to tackle numerous challenges and position themselves as dependable developmental contributors and manufacturers of intricate generics, nations of developing status are necessitated to give precedence to the enactment of substantial strategies for timeline management. Such endeavors entailed embodying a multifaceted approach, presiding over the ensuing pivotal elements.
The efficient execution of projects in many spheres depends on the best schedule management. Many companies have identified certain strategies as the most effective for boosting output and fulfilling deadlines. Successful schedule management is examined in this review article through the prism of technology, supply chain logistics, intellectual property (IP) demands, teamwork, quality control, talent development, financing, flexibility, sustainability, and stakeholder participation.
Technologically improving procedures
Project management supported by technology has transformed accepted practices. Advances in technology and software provide rapid, precise observation of team member correspondence, resource allocation, and activity level. Among other project management tools, Microsoft Project, Asana, and Trello are meant to let teams effectively meet benchmarks and project deadlines. Moreover, adopting collaboration tools like Microsoft Teams and Slack helps eradicate delays from poor communication, therefore fostering a more linked working atmosphere.
Optimizing supply chain logistics performance
Good supply chain management guarantees on-time completion of projects. Modern inventory control systems, such as SAP and Oracle, help companies maximize stock levels and prevent delays brought on by either too low or too high inventory levels. Strategic contacts with reliable logistics providers help to guarantee continuous delivery, therefore enhancing the general effectiveness of timeline control.
Navigating through intellectual property hurdles
Problems in maintaining a competitive edge and in preserving inventions depend on a strong awareness of intellectual property (IP) rights, as well as efficient management. Companies that want to deter competitors from violating copyrights must give patent and trademark filing first priority. Frequent intellectual property audits help to improve regulatory compliance and point out areas needing a change in intellectual property strategy, therefore safeguarding the project schedule from legal issues.
Fostering collaboration and knowledge sharing
Effective information flow within companies and encouragement of a cooperative culture may greatly increase creative capacity and problem-solving abilities. Establishing cross-functional teams of people from several backgrounds may hasten problem-solving and provide a wide spectrum of viewpoints. Moreover, the use of knowledge management systems such as centralized databases may enable team members to properly apply their particular skills and help information flow.
Creating robust quality assurance strategies
Successful fulfillment of high standards depends on a thorough quality assurance (QA) system that also lowers rework-related delays. Although regular quality assurance audits and feedback loops help to pinpoint areas for development, creating standard operating procedures (SOPs) for important jobs guarantees consistency. Giving quality top attention helps companies maximize their approaches to scheduling management.
Talent development and capacity building
Staff members must have the necessary skills to meet project needs, which call for efficient use of resources for talent development. Particularly in technical capabilities and project management, training courses and seminars let employees improve their skills. Moreover, creating mentorship initiatives could boost professional development and help to ensure the team’s ability to meet objectives by means of information flow.
Securing financing and investments
Maintaining project progress calls on both thorough awareness of financial needs and the ability to acquire such money. Companies should weigh grant and subsidy possibilities to support significant projects while preserving ownership integrity. Moreover, the process of creating convincing presentations for potential investors might help to properly acquire the necessary funds, thereby guaranteeing that financial assistance satisfies the expected project schedules and conforms to an environment of dynamic markets.
Adapt to changing market dynamics
The success of activities depends on our capacity to react fast to changes in the dynamic market of today. By means of market research instruments such as Nielsen or Statista, companies may remain current on consumer expectations and important trends. Agile project management techniques let teams react fast to comments and new priorities, therefore leaving them behind.
Incorporating measures of sustainability
Including sustainability in project management not only helps the surroundings but also has the possibility to increase brand recognition and provide long-term financial gains. Businesses should surely welcome greener goods and provide tools for energy-efficient technologies. These sustainable practices might appeal to customers who care about the environment and assist the company to improve its financial situation.
Stakeholder involvement and alignment
Including people in the planning and decision-making processes helps to match and inspire the drive required to finish projects on time. Regular stakeholder meetings and the use of feedback instruments like questionnaires or discussion sessions help to build trust by means of, including comments from stakeholders to help companies control challenges and improve the outcomes of projects. Eventually, good time management is difficult and calls for a thorough plan involving several ideal solutions.
Prioritization of tasks
Timeline management is much more effective when chores are correctly ranked with respect to their relevance, urgency, and interdependence. Depending on their degree of urgency, the Eisenhower Matrix, which may be used to classify tasks: 1) Urgent and Important, 2) Important but Not Urgent, 3) Urgent but Not Important, or 4) Neither Urgent nor Important, is a useful tool for arranging chores. In this sense, you may make forward plans to prevent issues and allocate duties based on priorities. Furthermore, the Critical Path Method (CPM) and other project management strategies assist in determining the longest interdependent sequence of activities that, taken together, provide the shortest possible project length. With the Critical Path Method (CPM), project managers may identify important tasks, track development, and react quickly. Integrated project management software based on modern critical path methods (CPM) offers timely and valuable information that facilitates wise decision-making and effective project completion.
To facilitate the practical application of these strategies, the following consolidated timeline management framework is essential. This framework integrates key activities, tools, and responsible parties across the project lifecycle, ensuring a holistic approach to managing complex generics development. The following example, given in fig. 6, provides a structured overview of an actionable framework for timeline management to guide policymakers and pharmaceutical companies in optimizing their processes.
Fig. 6: Actionable framework for timeline management
By adhering to this framework and its associated checklist items, organizations can systematically address the challenges of complex generic development, reduce time to market, and improve access to essential medicines in developing countries. This framework should be adapted with an altered timeline to specific organizational and project needs for maximum effectiveness. Regular monitoring, stakeholder coordination, and proactive risk management are key to navigating the complexities of developing country environments and delivering impactful products.
This review has highlighted the critical importance of effective timeline management in enabling developing countries to deliver complex generics to global markets. By outlining successful methodologies and addressing key challenges, the foundation is set for actionable strategies that can drive progress. However, to fully realize this potential, concrete steps are needed from both policymakers and pharmaceutical companies. The following recommendations provide a practical framework for advancing these efforts and fostering greater access to essential medicines.
Recommendations for policymakers and pharmaceutical companies
To enhance the practical relevance and impact of timeline management strategies in complex generic drug development, the following recommendations are directed to policymakers and pharmaceutical companies:
For policymakers
Harmonize regulatory pathways: work towards the harmonization of regulatory requirements across different countries for complex generics. This includes streamlining bioequivalence studies, documentation, and approval processes. Encourage international collaborations among regulatory agencies to share best practices and reduce redundancies.
Incentivize innovation: implement policies that incentivize pharmaceutical companies to invest in the development of complex generics, particularly in areas of unmet medical need. This can include tax incentives, expedited review processes, and market exclusivity periods.
Strengthen intellectual property enforcement: ensure robust enforcement of intellectual property rights while also promoting access to medicines. This can be achieved through balanced patent laws, voluntary licensing agreements, and compulsory licensing provisions in cases of public health emergencies.
Invest in infrastructure and training: provide funding and support for the development of infrastructure, technology, and skilled workforce in developing countries. This includes establishing quality control laboratories, training programs for pharmaceutical professionals, and technology transfer initiatives.
Promote transparency and information sharing: encourage transparency and information sharing among stakeholders, including regulatory agencies, pharmaceutical companies, healthcare providers, and patients. This can be achieved through the establishment of public databases, educational campaigns, and stakeholder engagement forums.
Address substandard and counterfeit drugs: implement measures to combat the proliferation of substandard and counterfeit drugs, including strengthening regulatory oversight, enhancing enforcement capabilities, and promoting public awareness.
For pharmaceutical companies
Adopt agile and lean methodologies: implement agile and lean methodologies in the development process to improve efficiency, reduce waste, and adapt to changing market conditions. This includes using cross-functional teams, iterative development cycles, and continuous improvement processes.
Invest in technology and automation: invest in advanced manufacturing technologies, such as automation, process analytical technology (PAT), and computer-aided design (CAD), to simplify production and enhance quality consistency.
Develop robust quality management systems: implement robust quality management systems to ensure the safety, efficacy, and quality of complex generic drugs. This includes using quality by design (QbD) principles, conducting comprehensive stability studies, and implementing strict quality control measures.
Establish strategic partnerships: establish strategic partnerships with suppliers, manufacturers, and distributors to ensure a reliable and cost-effective supply chain. This includes diversifying sources of active pharmaceutical ingredients (APIs), implementing inventory management systems, and negotiating favorable contracts.
Foster a culture of collaboration: foster a culture of collaboration within the organization and with external stakeholders. This includes promoting teamwork, communication, and knowledge sharing among employees, as well as engaging with regulatory agencies, healthcare providers, and patients.
Embrace sustainability: Incorporate sustainability principles into the development and manufacturing process to reduce environmental impact and improve long-term financial performance. This includes using green manufacturing practices, reducing waste, and promoting energy efficiency.
Continuous monitoring and adaptation: Keep monitoring the project and adapt to changes, and be open to use updated technologies.
International collaboration to address API supply chain challenges
The global supply chain for active pharmaceutical ingredients (APIs) is complex and vulnerable to disruptions, as demonstrated by recent events such as the COVID-19 pandemic. International collaboration is essential to address these challenges and ensure a stable and reliable supply of APIs. Specific strategies include:
Diversification of API sources: encourage pharmaceutical companies to diversify their sources of APIs to reduce reliance on a single country or region. This can be achieved through the establishment of strategic partnerships with API manufacturers in different countries and regions.
Investment in API manufacturing capacity: provide funding and support for the development of API manufacturing capacity in developing countries. This can help to reduce dependence on established API suppliers and promote local production.
Harmonization of API quality standards: work towards the harmonization of API quality standards across different countries to ensure the safety and efficacy of APIs used in complex generic drugs. This includes the establishment of common testing methods, inspection procedures, and certification programs.
Establishment of API stockpiles: consider the establishment of API stockpiles in strategic locations to ensure a buffer supply in case of disruptions. This can be achieved through international collaborations among governments, regulatory agencies, and pharmaceutical companies.
Promotion of transparency and information sharing: encourage transparency and information sharing among stakeholders about the API supply chain, including sources of APIs, manufacturing capacity, and inventory levels. This can help to identify potential vulnerabilities and prevent disruptions.
Strengthening trade agreements: negotiate trade agreements that promote the free flow of APIs across borders while also ensuring the protection of intellectual property rights and the enforcement of quality standards.
Joint research and development: foster joint research and development efforts among countries to develop innovative API manufacturing technologies and improve the efficiency and sustainability of the API supply chain.
By implementing these recommendations, policymakers and pharmaceutical companies can enhance the practical relevance and impact of timeline management strategies in complex generic drug development. This will help to ensure that patients have access to safe, effective, and affordable medicines, regardless of where they live.
CONCLUSION
In conclusion, effective timeline management is crucial for the successful development of complex generics in developing countries. The review underscores key challenges such as regulatory inefficiencies, limited infrastructure, and workforce constraints, which can hinder timely market entry. By adopting advanced project management tools, establishing clear milestones, and enhancing cross-functional coordination, developing nations can significantly reduce development cycle times. This multifaceted approach not only addresses regulatory and technical hurdles but also aligns product development with financial sustainability, ultimately improving access to high-quality generic medicines and enhancing healthcare outcomes. Policymakers and pharmaceutical companies are encouraged to implement these actionable insights to optimize their drug development processes in the complex generics sector.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
Gedala Venkata Murali Mohan Babu: Investigation, Conceptualization, Writing, Review and Visualization; Ni Luh Putu Nurshanti: Review, Editing, and Visualization; Harry Martha Wijaya: Review and Analysis; Raymond Rubianto Tjandrawinata: Conceptualization and Supervision.
CONFLICT OF INTERESTS
There is no conflict of interest from all the authors
REFERENCES
Globe Newswire. Generic drugs market size worth US $728.64 bn by 2034, driven by affordability, patent expirations and government support. 2025 Apr 16. Available from: https://www.globenewswire.com/news-release/2025/04/16/3062706/0/en/genericdrugs‑market‑size‑worth‑728‑64‑bn‑by‑2034‑driven‑by‑affordability‑patent‑expirations‑and‑government‑support.html.
Demina NB, Bakhrushina EO, Anurova MN, Merkushova AG, Pomytkina MV, Rastopchina OV. Complex generics as a trend of modern pharmaceutical development. Int J App Pharm. 2024;16(3):71-7. doi: 10.22159/ijap.2024v16i3.49937.
Stern S, Coghlan JP, Krishnan V, Raney SG, Babiskin A, Jiang W. Research and education needs for complex generics. Pharm Res. 2021 Dec;38(12):1991-2001. doi: 10.1007/s11095-021-03149-y, PMID 34950975.
Panchal K, Katke S, Dash SK, Gaur A, Shinde A, Saha N. An expanding horizon of complex injectable products: development and regulatory considerations. Drug Deliv Transl Res. 2022;13(2):433-72. doi: 10.1007/s13346-022-01223-5, PMID 35963928.
Market F. Insights complex generics market. Available from: https://www.futuremarketinsights.com/reports/complex-genericsmarket#:~:text=The%20complex%20generics%20market%20is,affordable%2C%20high-complexity%20treatments. [Last accessed on 18 Jun 2025].
Walenga RL, Babiskin AH, Zhao L. In silico methods for development of generic drug device combination orally inhaled drug products. CPT Pharmacometrics Syst Pharmacol. 2019;8(6):359-70. doi: 10.1002/psp4.12413, PMID 31044532, PMCID 6618094.
Renukuntla J, Palakurthi SS, Bolla PK, Clark BA, Boddu SH, Manda P. Advances in in vitro bioequivalence testing methods for complex ophthalmic generic products. Int J Pharm. 2022;627:122209. doi: 10.1016/j.ijpharm.2022.122209, PMID 36162609.
Haider R. The role and impact of generics in pharmaceutical markets. JDD. 2024 May 6;9(3):1-5. doi: 10.31579/2578-8949/155.
Klein K, Borchard G, Shah VP, Fluhmann B, McNeil SE, De Vlieger JS. A pragmatic regulatory approach for complex generics through the US FDA 505(j) or 505(b) (2) approval pathways. Ann N Y Acad Sci. 2021;1502(1):5-13. doi: 10.1111/nyas.14662, PMID 34296458.
Ranbaxy. Ranbaxy announces launch of atorvastatin generic Lipitor® in the US; 2011 Dec 01. Available from: https://www.daiichisankyo.com/media/press_release/detail/index_3727.html. [Last accessed on 18 Jun 2025].
Shaikh R, Yadav K, Thummar K, Maheriya B, Chauhan S. Development of complex generics: insights into trends challenges and market opportunities. J Generic Med. 2025;21(1):4-16. doi: 10.1177/17411343251313962.
Ranjan R. Development of complex generics and similar biological products: an industrial perspective of reverse engineering. AAPS PharmSciTech. 2025 Mar 26;26(4):95. doi: 10.1208/s12249-025-03087-7, PMID 40140232.
Rajput A, Pillai M, Ajabiya J, Sengupta P. Integrating quantitative methods and modeling and analytical techniques in reverse engineering; a cutting-edge strategy in complex generic development. AAPS PharmSciTech. 2025 Mar 26;26(4):92. doi: 10.1208/s12249-025-03067-x, PMID 40140161.
Lunawat S, Bhat K. Complex generic products: insight of current regulatory frameworks in US EU and Canada and the need of harmonisation. Ther Innov Regul Sci. 2020 Sep;54(5):991-1000. doi: 10.1007/s43441-020-00114-6, PMID 31960290.
Klein K, Stolk P, De Bruin ML, Leufkens HG, Crommelin DJ, De Vlieger JS. The EU regulatory landscape of non-biological complex drugs (NBCDs) follow-on products: observations and recommendations. Eur J Pharm Sci. 2019 May 15;133:228-35. doi: 10.1016/j.ejps.2019.03.029, PMID 30953753.
Rocco P, Musazzi UM, Franze S, Minghetti P. Copies of nonbiological complex drugs: generic hybrid or biosimilar. Drug Discov Today. 2019 Jan;24(1):250-5. doi: 10.1016/j.drudis.2018.08.003, PMID 30086403.
Kuribayashi R, Goto K, Ogawa T. Trend analysis of regulatory approvals for generics and biosimilars in Japan: 15 y of PMDA during fiscal years 2009-2023. AAPS J. 2024 Nov 12;26(6):113. doi: 10.1208/s12248-024-00989-5, PMID 39532822.
Zagalo DM, Simoes S, Sousa J. Regulatory science approach in pharmaceutical development of follow on versions of non-biological complex drug products. J Pharm Sci. 2022 Oct;111(10):2687-713. doi: 10.1016/j.xphs.2022.07.015, PMID 35901943.
Hussaarts L, Muhlebach S, Shah VP, McNeil S, Borchard G, Fluihmann B. Equivalence of complex drug products: advances in and challenges for current regulatory frameworks. Ann N Y Acad Sci. 2017 Nov;1407(1):39-49. doi: 10.1111/nyas.13347, PMID 28445611.
Zaidi FB, Ahmed SE, Makkad M. Role of effective project management in reducing drug development cost. Asian J Pharm Clin Res. 2017 Mar 1;10(3):53. doi: 10.22159/ajpcr.2017.v10i3.16186.
Kolluri S, Lin J, Liu R, Zhang Y, Zhang W. Machine learning and artificial intelligence in pharmaceutical research and development: a review. AAPS J. 2022 Jan 1;24(1):19. doi: 10.1208/s12248-021-00644-3, PMID 34984579.
Dave CV, Sinha MS, Beall RF, Kesselheim AS. Estimating the cost of delayed generic drug entry to medicaid. Health Aff (Millwood). 2020 Jun;39(6):1011-7. doi: 10.1377/hlthaff.2019.00673, PMID 32479219.
Chorniy A, Bailey J, Civan A, Maloney M. Regulatory review time and pharmaceutical research and development. Health Econ. 2021 Jan;30(1):113-28. doi: 10.1002/hec.4180, PMID 33078483.
Wang Y, Li J, Anupindi R. Manufacturing and regulatory barriers to generic drug competition: a structural model approach. SSRN Journal. 2018;69(3):1323-34. doi: 10.2139/ssrn.3145635.
Kamusheva M, Turcu Stiolica A, Gierczynski J, Subtirelu MS, Czech M, Petrova G. Do advanced therapies have a future in the low and middle income countries the case of Bulgaria Romania and Poland. Front Public Health. 2021 Aug 23;9:729847. doi: 10.3389/fpubh.2021.729847, PMID 34497796.
Olaghere J, Williams DA, Farrar J, Buning H, Calhoun C, Ho TW. Scientific advancements in gene therapies: opportunities for global regulatory convergence. Biomedicines. 2025;13(3):758. doi: 10.3390/biomedicines13030758, PMID 40149734.
Fu X, Buckley PJ, Sanchez Ancochea D, Hassan I. The world has a unique opportunity: accelerating technology transfer and vaccine production through partnerships. J Int Bus Policy. 2021 Nov 10;5(3):406-15. doi: 10.1057/s42214-021-00124-7.
Ahonkhai VI, Martins SF, Portet A, Lumpkin M, Hartman D. Speeding access to vaccines and medicines in low and middle-income countries: a case for change and a framework for optimized product market authorization. PLOS One. 2016 Nov 16;11(11):e0166515. doi: 10.1371/journal.pone.0166515, PMID 27851831, PMCID 5112794.
Beierle J, Algorri M, Cortes M, Cauchon NS, Lennard A, Kirwan JP. Structured content and data management enhancing acceleration in drug development through efficiency in data exchange. AAPS Open. 2023 May 8;9(1):11. doi: 10.1186/s41120-023-00077-6, PMID 37193559.
Albrecht M, Hoer A, Zimmermann A. Value of generic medicines: an health economics study. J Pharm Policy Pract. 2015 Oct 5;8(S1):p15. doi: 10.1186/2052-3211-8-s1-p15, PMCID 4602184.
Lakkis MM. Global and regional drug regulatory harmonization initiatives. Drug Inf J. 2010 May 1;44(3):289-97. doi: 10.1177/009286151004400311.
Wong H. The case for compulsory licensing during COVID-19. J Glob Health. 2020 Jun;10(1):010358. doi: 10.7189/jogh.10.010358, PMID 32509289, PMCID 7242884.
Pogge T. Just rules for innovative pharmaceuticals. Philosophies. 2022 Jul 12;7(4):79. doi: 10.3390/philosophies7040079.
Nwoke J. Regulatory compliance and risk management in pharmaceuticals and healthcare. Int J Health Sci. 2024 Sep 8;7(6):60-88. doi: 10.47941/ijhs.2223.
Djordjevic D, McFadyen A, Anderson JA. Ethical challenges and opportunities in the development and approval of novel therapeutics for rare diseases. J Med Access. 2023 Jun 6;7:27550834231177507. doi: 10.1177/27550834231177507, PMID 37323852.
Avhad PA, Chalikwar SS, Bhairav BA. A comprehensive review on complex generics. Bull of the Kar Univ. ‘Bio Med Geo’ Ser. BMG. 2022 Mar 30;105(1):129-35. doi: 10.31489/2022bmg1/129-135.
Sarikhani Y, Bastani P, Ahmadzadeh M, Dorosti H, Sara JA. Challenges of pharmaceutical supply chain in a developing country: a qualitative study. Thai J Pharm Sci. 2025;43(3):179-85. doi: 10.56808/3027-7922.3049.
Haq I, Muselemu OM. Blockchain technology in pharmaceutical industry to prevent counterfeit drugs. IJCA. 2018 Mar 19;180(25):8-12. doi: 10.5120/ijca2018916579.
Patil S, Kumar S, Rao DM, Rewatkar KG. Current regulatory framework and challenges for the approval of complex generics in the US and the EU CIS. 2024 Jan 30;2:e2210299X269535. doi: 10.2174/012210299x269535231203164108.
Lionberger RA. FDA Critical path initiatives: opportunities for generic drug development. AAPS J. 2008 Feb 20;10(1):103-9. doi: 10.1208/s12248-008-9010-2, PMID 18446510.
Van Der Pol KH Van Der, Aljofan M, Blin O, Cornel JH, Rongen GA, Woestelandt AG. Drug repurposing of generic drugs: challenges and the potential role for government. Appl Health Econ Health Policy. 2023 Jul 3;21(6):831-40. doi: 10.1007/s40258-023-00816-6, PMID 37398987.
Jayachandran P. Advances in quantitative methods and modeling for complex generic drugs and opportunities to hybridize learnings between innovator and generic drug developers. CPT Pharmacometrics Syst Pharmacol. 2023 Apr 27;12(5):549-51. doi: 10.1002/psp4.12968, PMID 37113050.
Lee CY, Chen X, Romanelli RJ, Segal JB. Forces influencing generic drug development in the United States: a narrative review. J Pharm Policy Pract. 2016 Sep 22;9(1):26. doi: 10.1186/s40545-016-0079-1, PMID 27688886.
Sreedevi A, Musmade PB, Bhat K, Dharmagadda S, Janodia MD, Bhat BB. A deep dive into the development of complex generics: a comprehensive review. J Appl Pharm Sci. 2024 Oct;14(10):1-14. doi: 10.7324/japs.2024.204337.
Raw AS, Wu L. Scientific considerations in the approval of complex generics. In: Sasisekharan R, Lee SL, Rosenberg A, Walker LA, editors. The science and regulations of naturally derived complex drugs. Cham: Springer International Publishing; 2019. p. 157-73. doi: 10.1007/978-3-030-11751-1_9.
Chhabra H, Mouslim MC, Kashiramka S, Rathore AS. Dynamics of biosimilar uptake in emerging markets. Expert Opin Biol Ther. 2022 Jun;22(6):679-88. doi: 10.1080/14712598.2022.2076557, PMID 35535988.
Ahmad PA, IA, Dehury RK, Behera MR. Access to essential medicines in India: the role of innovations, patents and intellectual property rights. Asia Pac J Health Manag. 2024;19(3):1-8. doi: 10.24083/apjhm.v19i3.1697.
Delgado Herrera L, Boone N, Kannenberg J, Van Gelderen M, Ogawa K, Beth Blauwet M. Enhanced globalization and speed to market for innovative medicines. ACCTRA. 2015;2(3):176-85. doi: 10.2174/2213476X03666160127001615.
Nakashima A, Kodama K, Sengoku S. Strategic trends of pharmaceutical companies: a growth model through regional focus and inter-regional alliances. Drug Discov Today. 2023 Jul;28(7):103611. doi: 10.1016/j.drudis.2023.103611, PMID 37164307.
Pathak R, Gaur V, Sankrityayan H, Gogtay J. Tackling counterfeit drugs: the challenges and possibilities. Pharmaceut Med. 2023 Jul;37(4):281-90. doi: 10.1007/s40290-023-00468-w, PMID 37188891.
Bakkert Hart IM, Ohana D, Venhuis BJ. Current challenges in the detection and analysis of falsified medicines. J Pharm Biomed Anal. 2021 Apr 15;197:113948. doi: 10.1016/j.jpba.2021.113948, PMID 33582458.
Vinothkumar V, Prabhu Kumar A. Speed of new product development enhancement through supply chain management in a generic pharmaceutical company. Int J Manag Stud. 2018 Apr 1;52(3):109. doi: 10.18843/ijms/v5i2(3)/14.
Mhando F, Berenge HT, Yue J, Gao L, Makundi EA, Malebo HM. Challenges and solutions for local pharmaceutical production in Tanzania. Voice Publ. 2025 Jan 1;11(2):318-31. doi: 10.4236/vp.2025.112023.
Pezzola A, Sweet CM. Global pharmaceutical regulation: the challenge of integration for developing states. Global Health. 2016 Dec 20;12(1):85. doi: 10.1186/s12992-016-0208-2, PMID 27998293.
Bansal S, Mahendiratta S, Kumar S, Sarma P, Prakash A, Medhi B. Collaborative research in modern era: need and challenges. Indian J Pharmacol. 2019 May-Jun;51(3):137-9. doi: 10.4103/ijp.IJP_394_19, PMID 31391680.
O Brien MN, Jiang W, Wang Y, Loffredo DM. Challenges and opportunities in the development of complex generic long-acting injectable drug products. J Control Release. 2021 Aug 1;336:144-58. doi: 10.1016/j.jconrel.2021.06.017, PMID 34126170.
Vieira M, Andia T, Karim O, Srishti SA, Pineda SA, Alonso Ruiz A. Rising pharmaceutical innovation in the Global South: a landscape study. J Pharm Policy Pract. 2023 Nov 27;16(1):155. doi: 10.1186/s40545-023-00669-3, PMID 38012700, PMCID PMC10680326.
Pava ML, Tucker EL. Effects of geopolitical strain on global pharmaceutical supply chain design and drug shortages. arXiv; 2023 Jan 1. doi: 10.48550/arxiv.2308.07434.
US Food and Drug Administration. Drug Supply Chain Security Act (DSCSA): law and policies. Silver Spring (MD): US Food and Drug Administration. Available from: https://www.fda.gov/drugs/drug-supply-chain-security-act-dscsa/drug-supply-chain-security-act-law-and-policies.
Sivaraman A, Banga A. Quality by design approaches for topical dermatological dosage forms. RRTD. 2015;4:9-21. doi: 10.2147/RRTD.S82739.
Gordon J, Potthast H, Stahl M, Rago L. World health organisation (WHO). In: Kanfer I, editor. Bioequivalence requirements in various global jurisdictions. Cham: Springer International Publishing. 2017. p. 307-31. doi: 10.1007/978-3-319-68078-1_11.
Merchant HA, Babar ZU, Hussain IM. A leap towards enforcing medicines prescribing by generic names in low and middle-income countries (LMICs): pitfalls limitations and recommendations for local drug regulatory agencies. J Pharm Policy Pract. 2022 Dec 22;15(1):104. doi: 10.1186/s40545-022-00501-4, PMID 36550588.
Hassen HK, Mekasha YT, Tegegne AA, Ozalp Y. A narrative review on problems in product quality regulatory system constraints and the concept of quality by design as a solution for quality assurance of African medicines. Front Med (Lausanne). 2024 Oct 3;11:1472495. doi: 10.3389/fmed.2024.1472495, PMID 39421861.
Franco P, Jain R, Rosenkrands Lange E, Hey C, Koban MU. Regulatory pathways supporting expedited drug development and approval in ICH member countries. Ther Innov Regul Sci. 2022;57(3):484-514. doi: 10.1007/s43441-022-00480-3, PMID 36463352.
K CP, MP, SA, RS. Concept of 505 (b) (2) application: benefits and challenges. Int J Drug Reg Affairs. 2018;2(2):16-20. doi: 10.22270/ijdra.v2i2.127.
Salminen WF, Wiles ME, Stevens RE. Streamlining nonclinical drug development using the FDA 505(b) (2) new drug application regulatory pathway. Drug Discov Today. 2018;24(1):46-56. doi: 10.1016/j.drudis.2018.07.005, PMID 30041014.
US Food and Drug Administration. Pre‑ANDA Program; 2022. Available from: https://www.fda.gov/drugs/generic‑drugs/pre‑anda‑program.
Nahler G. Dictionary of Pharmaceutical Medicine. Wien: Springer‑Verlag; 2009. p. 48. doi: 10.1007/978-3-211-89836-9_363.
Sullivan JO, Blake K, Berntgen M, Salmonson T, Welink J, Pharmacokinetics Working Party. Overview of the European medicines agencys development of product-specific bioequivalence guidelines. Clin Pharmacol Ther. 2018;104(3):539-45. doi: 10.1002/cpt.957, PMID 29319156.
Generic Applications in the EU, Patents and Exclusivity; 2011. Available from: https://gabionline.net/reports/generic-applications-in-the-eu-patents-and-exclusivity. [Last accessed on 10 Nov 2025].
Ellen FM, Pascale Boulet, Brook K Baker. Data exclusivity exceptions and compulsory licensing to promote generic medicines in the European Union: a proposal for greater coherence in European pharmaceutical legislation. J Pharm Policy Pract. 2017;10(1):19. doi: 10.1186/s40545-017-0107-9, PMID 28670455.
Dansie LS, Odoch WD, Ardal C. Industrial perceptions of medicines regulatory harmonization in the East African community. PLOS One. 2019;14(6):e0218617. doi: 10.1371/journal.pone.0218617, PMID 31216323.
Patel BD, Suthar SP, Mansuri AM, Joshi VD. A comparative analysis of generic drug assessment and regulatory approval in the USA, Europe and India. Zhongguo Ying Yong Sheng Li Xue Za Zhi. 2024;40:e20240029. doi: 10.62958/j.cjap.2024.029, PMID 39384543.
Chakraborty K, Yadav K. Drug approval process in US, Europe and India and its regulatory requirements: a review. Int J Drug Regul Aff. 2018;6(3):31. doi: 10.22270/ijdra.v6i3.266.
Ramteke A, Sufi MB, Deolekar R, Pathik C. A review on CDSCO: Central Drug Standard Control Organisation. IJARSCT. 2023;3(1):569-71. doi: 10.48175/ijarsct-14084.
RA, VG, Nelavala A. Analysis of regulatory strategies for generic product approval across Asian nations. Int JExp Biomed Res. 2024;3(1):16-22. doi: 10.26452/ijebr.v3i1.557.
Kannappan S, Darrow JJ, Kesselheim AS, Beall RF. The timing of 30 m stay expirations and generic entry: a cohort study of first generics, 2013-2020. Clin Transl Sci. 2021 Sep;14(5):1917-23. doi: 10.1111/cts.13046, PMID 33982425.
US Food and Drug Administration. GDUFA III user fee resource management program. Silver Spring (MD): FDA. Available from: https://www.fda.gov/industry/generic-drug-user-fee-amendments/gdufa-iii-user-fee-resource-management-program. [Last accessed on 11 Nov 2025].
Melocchi A, Briatico Vangosa F, Uboldi M, Parietti F, Turchi M, Von Zeppelin D. Quality considerations on the pharmaceutical applications of fused deposition modeling 3D printing. Int J Pharm. 2021 Jan 5;592:119901. doi: 10.1016/j.ijpharm.2020.119901, PMID 33010399.
Ngum N, Ndomondo Sigonda M, Walker S, Salek S. Regional regulatory harmonisation initiatives: their potential contribution to the newly established African medicines agency. Regul Toxicol Pharmacol. 2023;145:105497. doi: 10.1016/j.yrtph.2023.105497, PMID 37778434.
The FDA Group. Abbreviated New Drug Applications (ANDA) explained: a quick-guide. Boston (MA): The FDA Group; 2023 July 12. Available from: https://www.thefdagroup.com/blog/abbreviated-new-drug-applications-anda.
Drug patent watch-transform data into market domination; 2025. Available from: https://www.drugpatentwatch.com/blog/the-505b2-drug-patent-approval-process-uses-and-potential-advantages/?srsltid=AfmBOoqhq-3KJFTul2nYz9IoKxsKEn9INRXIaMX1IWx35Sy0bwmlCd.
Aggarwal S & Associates. Generic drug assessment and approval process in India; 2025 June 16. Available from: https://aggarwalsandassociates.com/generic-drug-assessment-and-approval-process-in-india/. [Last accessed on 11 Nov 2025].
Zoman SS, Khokale A. Regulatory frameworks adopted by the US and EU for bringing complex generics in the market. J Chem Health Risks. 2024;14(6):1187-96.
Bhatt M, Tank S, Shah J, Maheshwari D. Regulatory framework and disparities of complex generics in United States, European Union and Latin America. Journal of Generic Medicines. 2023;19(3):130-40. doi: 10.1177/17411343231194755.
Pribluda VS, Koduri C, Ibrahim S, Blake KV. Expanding global access to complex generics: the case for regulatory convergence. Regulatory Affairs Professionals Society; 2025. Available from: https://media.raps.org/m/55966497433bd03/original/25-2_pribluda-et-al.pdf. [Last accessed on 10 Nov 2025].
Raval P, Patel K, Patel K, Patel A, Gupta M, Bodhe S. An overview on comparative study of generic drug approval process in USA, India and Australia. Int J Pharmacol Res. 2025;31(1):1.
Xu W. A research on the influence of college student time management on their academic development. In: Proceedings of the 2020 5th International Conference on Modern Management and Education Technology (MMET 2020). Paris, France: Atlantis Press; 2020. p. 104-9. doi: 10.2991/assehr.k.201023.019.
Weaver P. The origins of modern management. South Melbourne (VIC): Mosaic Project Services Pty Ltd; 2007. Available from: https://mosaicprojects.com.au/pdf_papers/p050_origins_of_modern_management.pdf. [Last accessed on 10 Nov 2025].
Kwak YH. Brief history of project management; 2023. Available from: http://home.gwu.edu/~kwak/PM_History.pdf. [Last accessed on 10 Nov 2025].
Luthy C. The late origins of the timeline or: three paradoxes explained. Ann Sci. 2024;82(1):1-43. doi: 10.1080/00033790.2023.2289524, PMID 38308816.
Pamuji S, Baharuddin KM, Novianto AY, Kasmin K, Wibowo EP. Time management: the key to success in managing complex projects. J Markcount Finance. 2024;2(3):361-70. doi: 10.70177/jmf.v2i3.1470.
Gupta G, Mahor R, Pawar N, Gumashta R, Gumashta J. Program management challenges of clinical studies: a qualitative critical assessment. J Family Med Prim Care. 2023 Apr 1;12(4):625-31. doi: 10.4103/jfmpc.jfmpc_1151_22, PMID 37312779.
Khan UU, Ali Y, Garai Fodor M, Csiszarik Kocsir A. Application of project management techniques for timeline and budgeting estimates of startups. Sustainability. 2023 Nov 1;15(21):15526. doi: 10.3390/su152115526.
Poskett J. The history of science the ‘big picture’ and the politics of scale. BJHS Themes. 2024 Dec 1;9:1-15. doi: 10.1017/bjt.2024.24.
Lehtinen TO, Itkonen J, Lassenius C. Recurring opinions or productive improvements-what agile teams actually discuss in retrospectives. Empir Software Eng. 2016 Nov 3;22(5):2409-52. doi: 10.1007/s10664-016-9464-2.
Wong AK, Mooghali M, Ramachandran R, Ross JS, Wallach JD. Use of expedited regulatory programs and clinical development times for FDA-approved novel therapeutics. JAMA Netw Open. 2023 Aug 31;6(8):e2331753. doi: 10.1001/jamanetworkopen.2023.31753, PMID 37651145.
Bussgen M, Bussgen MA. Pipeline progress and portfolio management of the top 30 pharma companies over the past two decades. J Pharm Policy Pract. 2023 Sep 28;16(1):109. doi: 10.1186/s40545-023-00612-6, PMID 37770995.
Souza FB, Moraes AA. Analise da aplicacao da gestao de projetos por corrente critica no processo de desenvolvimento de produtos e na gestão de portfolio de um fabricante de aeronaves. Gest Prod. 2016 Aug 25;23(3):473-85. doi: 10.1590/0104-530x1953-15.
Yang S, Hu X, Zhu J, Zheng B, Bi W, Wang X. Aspects and implementation of pharmaceutical quality by design from conceptual frameworks to industrial applications. Pharmaceutics. 2025;17(5):623. doi: 10.3390/pharmaceutics17050623, PMID 40430914.
Stewart S, Giambalvo J, Vance J, Faludi J, Hoffenson S. A product development approach advisor for navigating common design methods, processes and environments. Designs. 2020 Feb 14;4(1):4. doi: 10.3390/designs4010004.
Kuwashima K. Exploring the characteristics of pharmaceutical product development. Annals Bus Admin Sci. 2015 Jun 14;14(3):161-70. doi: 10.7880/abas.14.161.
Yang C, Deng B, Wen Q, Guo P, Liu X, Wang C. Safety profiles of sevoflurane in pediatric patients: a real-world pharmacovigilance assessment based on the FAERS database. Front Pharmacol. 2025 Feb 10;16:1548376. doi: 10.3389/fphar.2025.1548376, PMID 39995419.
Reddy MV, Ganesh GN, Ahmed SS, Rajendra PK, Babu E. Research on pharmaceutical product life cycle management challenges faced by generic manufacturers for US approval. Indian J Pharm Educ Res. 2022 Apr 1;56(2 Suppl):s347-55. doi: 10.5530/ijper.56.2s.105.
Berger J, Dunn JD, Johnson MM, Karst KR, Shear WC. How drug life-cycle management patent strategies may impact formulary management. Am J Manag Care. 2016 Oct 1;22(16 Suppl):S487-95. PMID 28719222.
Lehmann S, Allard R, Boehler Y. The European Medicines Agency clinical data website enables insights into clinical development timelines and strategy. Open Access J Clin Trials. 2019 Oct 15;11:37-56:S205842. doi: 10.2147/OAJCT.
Hasan MI, Shimu SA, Akther A, Jahan I, Hamiduzzaman M, Hasan AH. Development of generic drug products by pharmaceutical industries considering regulatory aspects: a review. J Biosci Med. 2021 Jan 1;9(10):23-39. doi: 10.4236/jbm.2021.910003.
Pramod K, Tahir MA, Charoo NA, Ansari SH, Ali J. Pharmaceutical product development: a quality by design approach. Int J Pharm Investig. 2016;6(3):129-38. doi: 10.4103/2230-973x.187350, PMID 27606256, PMCID 4991121.
Reddy MV, Ganesh G, Babu B, Jagadeesan R, Mr PK. Risk assessment of failures in generic drug development and approval procedure under competitive generic drug therapy and patent challenge exclusivities provided by the United States food and Drug Administration. Acta Marisiensis Seria Medica. 2022 Mar 1;68(1):28-34. doi: 10.2478/amma-2022-0004.
Hao L, Zhang J, Ma X. Development trend of software process model. IOP Conf Ser: Mater Sci Eng. 2019;569(5):052077. doi: 10.1088/1757-899x/569/5/052077.
Amaral C, Rodrigues AR, Veiga F, Bell V. Biosimilar medicines: from development process to marketing authorization by the EMA and the FDA. Appl Sci. 2024;14(17):7529. doi: 10.3390/app14177529.
Micic L. Agile methodology selection criteria: IT start-up case study. IOP Conf Ser: Mater Sci Eng. 2017 May 1;200:012031. doi: 10.1088/1757-899x/200/1/012031.
Veiga AP. Project success in agile development projects. arXiv; 2017 Jan 1. doi: 10.48550/arxiv.1711.06851.
Lodha A. Agile: open innovation to revolutionize pharmaceutical strategy. Imperial J Interdiscip Res. 2016;2(12):39-41.
Alsaadi M, Lisitsa A, Khalaf M, Qasaimeh M. Investigating the capability of agile processes to support medical devices regulations: the case of XP, scrum, and FDD with EU MDR regulations. In: Huang DS, Huang ZK, Hussain A, editors. Intelligent computing methodologies: 15th International Conference, ICIC 2019, Nanchang, China, August 3-6, 2019, proceedings, part III. Cham: Springer International Publishing; 2019. p. 581-92. doi: 10.1007/978-3-030-26766-7_53.
Kaur K, Jajoo A, Manisha. Applying agile methodologies in industry projects: benefits and challenges. In: Proceedings of the 2015 International Conference on Computing Communication Control and Automation (ICCUBEA) Pune, India; 2015 Feb 1. p. 832‑6. doi: 10.1109/ICCUBEA.2015.166.
Heimicke J, Chen R, Albers A. Agile meets plan-driven hybrid approaches in product development: a systematic literature review. Proc Des Soc: Des Conf. 2020 May 1;1:577-86. doi: 10.1017/dsd.2020.259.
Minghetti P, Musazzi UM, Casiraghi A, Rocco P. Old active ingredients in new medicinal products: is the regulatory path coherent with patients expectations. Drug Discov Today. 2020 Aug;25(8):1337-47. doi: 10.1016/j.drudis.2020.05.013, PMID 32485225.
Li EC, Abbas R, Jacobs IA, Yin D. Considerations in the early development of biosimilar products. Drug Discov Today. 2015;20Suppl2:1-9. doi: 10.1016/j.drudis.2014.12.017, PMID 25572407.
Cristofoletti R, Schmidt S, Diniz A. Non-procrustean pathways for complex generic drugs development. Ther Deliv. 2018 Sep 1;9(9):605-7. doi: 10.4155/tde-2018-0047, PMID 30189807.
Remington K, Pollack J. Tools for complex projects. Vol. 1. London: Routledge; 2016. doi: 10.4324/9781315550831.
Dobre OI. Differences of organizational culture between small and large enterprises. Ovidius University Annals Economic Sciences Series. 2016;16(1):296-301.
Nazarian A, Atkinson P. Organisational size as a moderator of the culture-effectiveness relationship: the case of the private sector in Iran. Organ Cult Int J. 2015 Jul 9;14(2):1-16. doi: 10.18848/2327-8013/CGP/v14i02/50951.
Genedy M, Hellerstedt K, Naldi L, Wiklund J. Growing pains in scale-ups: how scaling affects new venture employee burnout and job satisfaction. J Bus Venturing. 2023 Nov 22;39(2):106367. doi: 10.1016/j.jbusvent.2023.106367.
Yasar H, Sagsan M. The mediating effect of organizational stress on organizational culture and time management: a comparative study with two universities. Sage Open. 2020 Apr 1;10(2):1-11. doi: 10.1177/2158244020919507.
Maqsoom A, Choudhry RM, Umer M, Mehmood T. Influencing factors indicating time delay in construction projects: impact of firm size and experience. Int J Constr Manag. 2019 May 9;21(12):1251-62. doi: 10.1080/15623599.2019.1613206.
Van Neerijnen P Van, Figge P, Tempelaar MP, Schippers MC. Ambidexterity: size matters, reflexive climate and organizational TMS’s influence and the contingent effect of size. J Small Bus Manag. 2021 Oct 21;61(6):3121-54. doi: 10.1080/00472778.2021.1981917.
Shayegan S, Yavari N, Bazrkar A. Human resource development practices and organizational performance: examining the mediating role of transformational leadership style. Work Pap Oper Manag. 2022 Jul 5;13(2):64-85. doi: 10.4995/wpom.17243.
Tang J, Yang J, Ye W, Khan SA. Now is the time: the effects of linguistic time reference and national time orientation on innovative new ventures. J Bus Venturing. 2021 Aug 9;36(5):106142. doi: 10.1016/j.jbusvent.2021.106142.
Malmi T, Bedford DS, Bruhl R, Dergard J, Hoozee S, Janschek O. The use of management controls in different cultural regions: an empirical study of anglo-saxon germanic and nordic practices. J Manag Control. 2022 Jul 3;33(3):273-334. doi: 10.1007/s00187-022-00342-x.
Huang J. The challenge of multicultural management in global projects. Procedia Soc Behav Sci. 2016 Jul 1;226:75-81. doi: 10.1016/j.sbspro.2016.06.164.
Islam MS, Trigunarsyah B. Construction delays in developing countries: a review. J Constr Eng Proj Manag. 2017;7(1):1-12. doi: 10.6106/jcepm.2017.3.30.001.
Khan MA, Panarina E. The role of national cultures in shaping the corporate management cultures: a four-country theoretical analysis. JEECAR. 2017 Apr 25;4(1):25. doi: 10.15549/jeecar.v4i1.152.
Huang H. Research on the importance of cross-cultural integration in international business management. Mod Econ Manag Forum. 2023 Dec 26;4(6):171. doi: 10.32629/memf.v4i6.1502.
Starkey K, Starkey K. Time and work: a psychological perspective. In: Blyton P, Hassard J, Hill S, Starkey K, editors. Time. Time, work and organization. London: Routledge; 2017. p. 35-56. doi: 10.4324/9781315267272-3.
Sayfudinova NZ, Nagumanova AI. Influence of psychological factors on the success of an organization and management activities. Ekonomika i Upravlenie: Problemy Resheniya. 2024 Jan 1;4/7(145):79-83. doi: 10.36871/ek.up.p.r.2024.04.07.012.
Kumari N. Increasing employees efficiency through workforce management. JBTP. 2023;11(3):77. doi: 10.22158/jbtp.v11n3p77.
Amritharaj SD. Role of psychology in human resource management. Int J Res Hum Resour Manage. 2019;1(1):17-20. doi: 10.33545/26633213.2019.v1.i1a.5.
Romero Perez C, Sanchez Lissen E. Scientific narratives in the study of student time management: a critical review. RIMCIS. 2022;11(2):60-86. doi: 10.17583/rimcis.10322.
Voskoboynikov F. In: Dalcher D, editor. Managing projects in a world of people, strategy and change. London: Routledge; 2018. p. 39-44. doi: 10.4324/9780429449741-5.
Jeyabharathy DP. Psychological dimensions of time management among employees. IJSREM. 2023 Jan 5;7(1):1-5. doi: 10.55041/ijsrem17408.
Schuhmacher A, Hinder M, Von Stegmann Und Stein A, Hartl D, Gassmann O. Analysis of pharma R&D productivity a new perspective needed. Drug Discov Today. 2023;28(10):103726. doi: 10.1016/j.drudis.2023.103726, PMID 37506762.
Parpiev K. Overemphasis on hierarchy and bureaucracy leading to slow decision-making and a lack of agility; 2023 Jan 1. doi: 10.2139/ssrn.4361544.
Murthy NN, Nagaraja HN, Rikhtehgar Berenji HR. Managing concurrency in cyclical projects under stochastic task environments: vaccine development projects during pandemics. Prod Oper Manag. 2022 Nov 9;32(3):951. doi: 10.1111/poms.13907, PMID 36718233.
Aeon B, Faber A, Panaccio A. Does time management work. A meta-analysis. PLOS One. 2021;16(1):e0245066. doi: 10.1371/journal.pone.0245066, PMID 33428644.
Rajesh E, Lal R, Mathuraswamy P, Ahamed SBI, Gaidhani S. Motivational theories and their applicability in the workplace. Journal of Informatics Education and Research. 2024 Jan 1;4(2):3111-7. doi: 10.52783/jier.v4i2.1225.
Yesudas R, Castelle K, Joiner KF, Bradley JM, Efatmaneshnik M. Addressing tensions of overlapping project management and systems engineering with the elegance of a complex systems governance approach. Int J Syst Syst Eng. 2020 Jan 1;10(2):164. doi: 10.1504/ijsse.2020.109143.
Ewim CP, Achumie GO, Adeleke AG, Okeke IC, Mokogwu C. Developing a cross-functional team coordination framework: a model for optimizing business operations. Int J Frontline Res Multidiscip Studies. 2024 Oct 9;4(1):15-34. doi: 10.56355/ijfrms.2024.4.1.0030.
Watanabe WC, Shafiq M, Nawaz MJ, Saleem I, Nazeer S. The impact of emotional intelligence on project success: mediating role of team cohesiveness and moderating role of organizational culture. Int J Eng Bus Manag. 2024 Mar 5;16:18479790241232508. doi: 10.1177/18479790241232508.
Luckmann P, Laumann M. How national culture impacts the success of project customer engagement: an empirical investigation. Procedia Comput Sci. 2016 Jan 1;100:95-102. doi: 10.1016/j.procs.2016.09.128.
Zhang Y, Marquis C, Filippov S, Haasnoot HJ, Van Der Steen M. The challenges and enhancing opportunities of global project management: evidence from Chinese and Dutch cross-cultural project management. SSRN Electron J. 2015 Jan 1. doi: 10.2139/ssrn.2807439.
Abu Rumman MA, Alzeyadat AA. The influence of delegation of authority and management control on the effectiveness of managerial decision-making in the Arab pharmaceutical manufacturing company limited in Al-Salt City in Jordan. Int J Bus Admin. 2019;10(4):13. doi: 10.5430/ijba.v10n4p13.
Malik TH. Culturally imprinted anxiety and the itinerary of clinical trial projects for its management. Cross-Cult Res J Comp Soc Sci. 2021;55(2-3):148-78. doi: 10.1177/1069397120967309.
Oyekunle D, Amodu O, Lateef O. Cultural dynamics in UK project environments: exploring the impact of individualistic and collectivist cultures on project outcomes. OALib. 2024;11(5):1-21. doi: 10.4236/oalib.1111573.
Boubakri N, Chkir I, Saadi S, Zhu H. Does national culture affect corporate innovation? International evidence. J Corp Fin. 2021;66:101847. doi: 10.1016/j.jcorpfin.2020.101847.
Piwowar Sulej K. Organizational culture and project management methodology: research in the financial industry. Int J Manag Projects Bus. 2021 Mar 30;14(6):1270-89. doi: 10.1108/ijmpb-08-2020-0252.
Reitzig M. How to get better at flatter designs: considerations for shaping and leading organizations with less hierarchy. J Organ Des. 2022 Jan 27;11(1):5-10. doi: 10.1007/s41469-022-00109-7.
Beer R, Qi A, Rios I. Team building and incentive schemes in collaborative projects; 2024 Jan 1. p. 47. doi: 10.2139/ssrn.4872792.
Basten D, Muller M, Ott M, Pankratz O, Rosenkranz C. Impact of time pressure on software quality: a laboratory experiment on a game-theoretical model. PLOS One. 2021 Jan 15;16(1):e0245599. doi: 10.1371/journal.pone.0245599, PMID 33449957.
Furxhi G. Employee’s resistance and organizational change factors. EJBMR. 2021 Mar 12;6(2):30-2. doi: 10.24018/ejbmr.2021.6.2.759.
De Waal AA De, De Boer FA De. Project management control within a multicultural setting. J Strategy Manag. 2017 Mar 27;10(2):148-67. doi: 10.1108/jsma-03-2016-0020.
Liu C. Employee performance in multinational corporations: the impact of cross-cultural training in the globalized era. EHSS. 2024 Apr 19;29:338-43. doi: 10.54097/z8s79850.
Iqbal M, Suzianti A. Improvement of new product development process by evaluating the existing development approach: lesson learned from pharmaceutical and ICT companies. AIP Conf Proc. 2020 Jan 1;2227:040009. doi: 10.1063/5.0001007.
Suvvari SK. Project portfolio management: best practices for strategic alignment. Int J Prof Bus Rev. 2023 Dec 29;8(12):e04987. doi: 10.26668/businessreview/2023.v8i12.4987.
Kummer TF, Schmiedel T. Reviewing the role of culture in strategic information systems research: a call for prescriptive theorizing on culture management. Commun Assoc Inf Syst. 2016;38:122-44. doi: 10.17705/1cais.03805.
Chang MH. Research on modern corporate cost management and cost control methods. Int J Glob Econ Manag. 2024 Apr 25;2(3):320-6. doi: 10.62051/IJGEM.v2n3.38.
Vinayavekhin S, Phaal R. Improving synergy in strategic planning: enablers and synchronization assessment framework (SAF). Int J Innov Technol Manag. 2020 Jan 31;17(2):2050009. doi: 10.1142/s0219877020500091.
Lopez D, Oliver M. Integrating innovation into business strategy: perspectives from innovation managers. Sustainability. 2023 Apr 11;15(8):6503. doi: 10.3390/su15086503.
Mwaikambo L, Brittingham S, Ohkubo S, Salem RM, Sama DJ, Sow F. Key factors to facilitate locally driven family planning programming: a qualitative analysis of urban stakeholder perspectives in Africa and Asia. Global Health. 2021 Jul 3;17(1):75. doi: 10.1186/s12992-021-00717-0, PMID 34217354, PMCID 8254949.
Akroyd C, Biswas SS, Chuang S. How management control practices enable strategic alignment during the product development process. Adv Manag Acc. 2016. p. 99-138. doi: 10.1108/s1474-787120150000026004.
Sunmola HO. Evaluation of motivating and requiring factors for milestones in IT projects. Procedia Manuf. 2020;51:1469-77. doi: 10.1016/j.promfg.2020.10.204.
Ojra J, Opute AP, Alsolmi MM. Strategic management accounting and performance implications: a literature review and research agenda. Future Bus J. 2021;7(1):64. doi: 10.1186/s43093-021-00109-1.
Zhang W, Zeng X, Liang H, Xue Y, Cao X. Understanding how organizational culture affects innovation performance: a management context perspective. Sustainability. 2023 Apr 14;15(8):6644. doi: 10.3390/su15086644.
Menon S. Best practices and implementation challenges in effective project management. Int Bus Res. 2024 Mar 27;17(2):66. doi: 10.5539/ibr.v17n2p66.
Wegrecka Chudzik K, Jankowski L, Duda J. Evolving role of PMO in contemporary project management. Commun Int Proc. 2024 Jan 1;2024(5). doi: 10.5171/2024.4343524.
Szekely B, Kesmarki Gally SE, Lakner Z. Hybrid Project Management: scoping review. Proj Leadersh Soc. 2025 Mar 1;6:100182. doi: 10.1016/j.plas.2025.100182.
Misbahuddin M, Maarif MS. The linkage between leadership style of project manager and project performance: evidence from the telecommunication industry. 2024 Jan 1;9(3):163-82. doi: 10.5267/j.jpm.2024.6.001.
Bolanowski M, Ganzha M, Maciaszek L, Paprzycki M, Slezak D, editors. Proceedings of the 19th Conference on Computer Science and Intelligence System, Warsaw, Poland. ACSIS; 2024 Oct 23-26;39:429-41.