
Int J App Pharm, Vol 18, Issue 2, 2026, 136-142Original Article
THERAPEUTIC POTENTIAL OF NIGELLA SATIVA OIL CREAM FOR ECZEMA TREATMENT: FORMULATION, STABILITY AND CLINICAL ASSESSMENT
BASSEL HUSSEIN1, ABDULLAH H. MAAD2,3*, WAEL ALAGHAWANI4, RAMA AZIZ5, NOUR ALHABBAL6
1Department of Pharmaceutics, Faculty of Pharmacy, Syrian Private University, Damascus, Syria. 2Department of Pharmaceutics, College of Pharmacy, University of Al-Ameed, Karbala, Iraq. 3Department of Pharmacy Practice, College of Clinical Pharmacy, Hodeidah University, Al-Hodeidah, Yemen. 4Department of Pharmacology, Faculty of Pharmacy, Al-Ittihad Private University, Daraa, Syria. 5Faculty of Agriculture, Damascus University, Damascus, Syria, 6Department of Pharmaceutics, Faculty of Pharmacy, Al-Sham Private University, Damascus, Syria
*Corresponding author: Abdullah H. Maad; *Email: dr.ph.abdullah.maad@gmail.com
Received: 04 Sep 2025, Revised and Accepted: 24 Dec 2025
ABSTRACT
Objective: This study was aimed at developing and evaluating a stable topical cream based on the Nigella sativa oil and to establish the therapeutic value of the product in the moderate to severe patients with the eczematous patients.
Methods: Nigella sativa oil was cold-pressed to obtain it and its composition was determined by gas chromatography-mass spectrometry (GC-MS). The prepared cream formulations were tested on physicochemical characteristics, microbial quality, and stability in accelerated conditions. The most stable formulation (Formula 3) was 30 d clinically tested on 18 patients with moderate to severe eczema.
Results: GC–MS analysis showed that thymoquinone was the major component, and it represented 64.17 percent of the 18 components identified. Formula 3 had the optimum viscosity (23,300±18.6 cps), spreadability (14.28±0.1 g•cm/s), and stability among the four formulations. The clinical assessment revealed that there was a dramatic improvement of the Eczema Area and Severity Index (EASI) score, at baseline: 18.9± 4.1 and 6.2±2.3, after 30 days (p<0.001). No adverse effects were observed.
Conclusion: Nigella sativa oil cream was stable, safe, and clinically effective in the reduction of the symptoms of eczematous lesions. Such results endorse its prospect as a natural remedy of eczema, though bigger controlled clinical studies are advisable to corroborate the status.
Keywords: Clinical assessment, Eczema, Herbal cream formulation, Nigella sativa, Stability, Thymoquinone
© 2026 The Authors.Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i2.54168 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Eczema or dermatitis is a collection of chronic inflammatory skin diseases, which are defined by peeling, redness, dryness, and itch of the skin. Despite having varied etiologic factors and clinical symptoms, all of the subtypes share frequent symptoms such as pruritus, erythema, and impaired skin barrier function [1]. Depending on the look and degree of local harm, eczema is generally classified into two basic patterns, acute and chronic, with an intermediate form sometimes being mentioned [2]. The interaction between endogenous and exogenous factors is the primary cause of the condition, which often leads to hypersensitivity responses [3-5]. Conventional medical therapies of eczema are aimed at symptom relief and prevention of exacerbations. They include emollients for skin moisturizing, topical corticosteroids for control of the inflammatory process, and antihistamines for itching. For more severe cases of eczema, systemic immune suppressants or biologic therapies can be offered as treatment options [5, 6]. However, long-term use of corticosteroids therapy has the potential to lead to adverse effects such as skin atrophy and systemic complications [7]. Moreover, the treatment outcomes frequently have substantial variability among patients encountered in the clinical setting, and therapeutic goals are often not achieved to the level desired. Many conventional medications have unwanted side effects as well [8]. As with many diseases, there has been growing interest in natural and traditional remedies for managing skin hypersensitivity conditions like eczema. The World Health Organization (WHO) has estimated that approximately 80% of the population in developing countries relies on traditional medicines for their primary healthcare needs [9, 10]. Plants, as a rule, contain different bioactive compounds from a wide range of chemical classes. Therefore, it is reasonable to incorporate plant extracts or their active principles in the formulation of topical preparations for the treatment of eczema, particularly from plants that have traditionally been used to treat skin conditions. An example from this group of plants is Nigella sativa. This particular plant has gained attention due to its anti-inflammatory, antioxidant, and immunomodulatory potential [11, 12]. Thymoquinone, a key active ingredient in Nigella sativa, has been established in the scientific literature to inhibit pro-inflammatory cytokines as well as oxidative stress pathways implicated in the pathogenesis of eczema [13-15]. The seeds and their oil, usually derived through cold pressing, are regularly used in the flavoring of bread, cheese, and other items. Despite its ancient application, scientific work on this plant and its components is still being done. The oil of Nigella sativa is consumed for its potential health benefits, but while the oil's properties have been largely investigated, the pharmacological effects are generally attributed to thymoquinone and the contributions of other components have been studied less [16, 17]. Numerous biochemical and biophysical techniques have been employed to gain better insights into the biological activities of its components, including volatile monoterpenes such as α-thujene, α-pinene, sabinene, and 3-carene. The integration of plant extracts and essential oils into nano-carrier systems in order to enhance their bioavailability, stability, and skin penetration is a rising trend in contemporary pharmaceutics [18, 19]. Recent studies have shown the activity of the black seed oil as an antioxidant, antitumor, antibacterial, and anti-inflammatory. One of the most widely known uses is its protective effect against oxidative stress due to free radical [20]. The potential of Nigella sativa oil as a topical treatment for eczema is still unexplored, despite its well-established anti-inflammatory, antimicrobial, and wound-healing qualities. The incorporation of Nigella sativa into scientifically designed topical formulations has received little attention in prior research, which has mostly concentrated on oral administration or crude extracts. Furthermore, there aren't many carefully made, stability-tested Nigella sativa oil creams that have been clinically tested for safety and effectiveness in eczema patients [21, 22]. Therefore, this study seeks to address this gap by developing a stable N. sativa oil cream and evaluating its physicochemical characteristics and therapeutic potential through clinical testing in patients with moderate-to-severe eczema.
MATERIALS AND METHODS
Materials
Nigella sativa seeds were procured from a local herbal market in Damascus, Syria. Stearic acid, paraffin wax, lanett O, sodium lauryl sulfate (SLS), tween 80, and tocopherol acetate were procured from TNN Development Limited (Shanghai, China). Methyl paraben, propyl paraben, and triethanolamine (TEA) were purchased from Salicylate and Chemicals Pvt Ltd (Mumbai, India). Glycerin was obtained from Luken (Barcelona, Spain). Carbomer 940 was sourced from TNJ Chemical (Anhui, China).
Extraction of oil
Nigella sativa oil was extracted using the cold pressing method. Seeds were mechanically pressed at 25 °C. The oil and crushed seed fibers were separated by soaking the mixture overnight at 25 °C, followed by filtration to obtain the fixed oil suitable for formulation [23].
Analysis of Nigella sativa oil
The chemical composition of the Nigella sativa oil was analyzed by gas chromatography-mass spectrometry (GC/MS) (Agilent MS-GC, model 5937, California USA). The oven temperature program started at 60 °C and was ramped to 240 °C at 10 °C/min, then further to 290 °C at 4 °C/min. The percentages of the components were determined based on the area of each one relative to the total area of all components. Compounds were identified by comparison of their mass spectra and retention indices with those in the NIST library and/or by comparison with authentic standards [24].
Formulation of cream
Four cream formulations containing 5% Nigella sativa oil were prepared with varying concentrations of stearic acid, carbomer 940, and emulsifiers (Tween 80 or SLS), as shown in table 1. The oily phase (stearic acid, paraffin wax, Lanett O, tocopherol acetate) and aqueous phase (SLS or Tween 80, glycerin, purified water) were heated separately to 75 °C. The oily phase was gradually added to the aqueous phase under continuous stirring while cooling. At 40 °C, the black seed oil and preservatives were incorporated, and the mixture was stirred until room temperature. The pH was adjusted to 6.5 using TEA where necessary. The four formulations were developed to examine how the type of emulsifier and the level of thickening polymer influence the physical and stability characteristics of the Nigella sativa oil cream. Carbomer 940 was added to Formula 3 and 4 at two levels, 0.5 and 1 %, respectively, to determine its influence on viscosity and spreadability. Sodium Lauryl Sulfate (SLS) and Tween 80 were chosen as emulsifying agents for comparative analysis of emulsification efficiency. The systematic variation enabled the discovery of the most stable, well-balanced formulation to be further clinically assessed.
Table 1: Composition of Nigella sativa oil cream
| Ingredient | Formula 1 | Formula 2 | Formula 3 | Formula 4 |
| Nigella sativa oil | 5% | 5% | 5% | 5% |
| Stearic acid | - | - | 2% | 2% |
| Carbomer 940 | - | - | 0.5% | 1% |
| Tween 80 | 1% | - | 1% | 1% |
| SLS | - | 1% | - | - |
| Paraffin wax | 3% | 3% | 3% | 3% |
| Lanett O | 2% | 2% | 2% | 2% |
| Tocopherol acetate | 0.1% | 0.1% | 0.1% | 0.1% |
| Glycerin | 4% | 4% | 4% | 4% |
| Methyl paraben | 0.2% | 0.2% | 0.2% | 0.2% |
| Propyl paraben | 0.5% | 0.5% | 0.5% | 0.5% |
| TEA | QS | QS | QS | QS |
Evaluation of cream
The prepared creams were evaluated thoroughly to determine their physicochemical properties, microbial content and general stability [25]. All tests were done three times unless indicated to the contrary.
Appearance tests
The visual examination was performed to assess how the cream looked like, its colour, smell and feel. The color was documented to enable consistency of the formulations, whereas the aroma was evaluated to tell whether it contained offensive smells.
Homogeneity and phase separation
To analyze homogeneity and phase separation, the cream was placed in a thin layer on a clean glass slide and observed under the visible light on whether coarse particles, air bubbles, or phase separation occurred.
pH measurement
The pH was measured using a calibrated digital pH meter, WTW Bensh pH meter 7110, Germany, at 25 °C. One g of cream was dissolved in 10 ml of distilled water and mixing thoroughly.
Viscosity measurement
The viscosity was determined with the help of Brookfield viscometer (Model DV2T, USA). About 50 g of cream were put in sample holder and viscosity values were measured at 25 °C.
Spreadability assessment
The parallel plate method was used to calculate spreadability. A given amount of the cream was put between two glass slides and compression was done to bring the thickness to the same level. The measurement to time the separation of the two slides was done and the spreadability was calculated using the formula:
Spreadability = 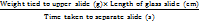
Microbial content testing
Testing of microbes was guided by USP<61>and<62>of non-sterile topical products. This involved the determination of total bacterial count (TBC), total fungal count (TFC) and identification of particular pathogens, including Escherichia coli, Staphylococcus aureus, Klebsiella pneumonia and Bacillus cereus.
Stability testing
The prepared creams were put through short-term stability tests, in accelerated conditions 40 °C and 75% Relative humidity (RH), over a period of 4 w in which parameters like color changes, pH changes, viscosity changes, spreadability and microbial content were observed.
Clinical study
A clinical study was performed to obtain preliminary clinical data on the optimized Nigella sativa oil cream, Formula 3. Among the four prototypes developed, Formula 3 was considered for clinical investigation due to its excellent physicochemical profile, balanced viscosity and spreadability, a microbial compliance profile, and accelerated stability study results. The total number of participants is18, aged between 18-40 years of age, wherein patients will be selected based on a confirmed diagnosis of eczema by a qualified dermatologist. A single-arm, open-label pilot study design was used to ensure the reliability of the results with minimal bias. The study was done in accordance with the Declaration of Helsinki and approved by the Scientific Research Ethics Committee, Faculty of Pharmacy, Al-Ittihad Private University, Damascus, Syria (Approval No. 12/24). Prior to enrollment, all participants provided written informed consent.
Study design and methodology
The participants were randomly assigned to the chosen formula (Formula 3) that was followed during the course of 30 d. All the participants were counseled not to use the cream more than twice a day, in the morning and also in the evening. The creams were labelled with different numbers to remain in blinding. The weekly assessments were preceded by the baseline assessments aimed at determining the changes in the severity of eczemas and the overall condition of the skin.
Assessment parameters
The burdensomeness of the disease is measured with the Eczema Area and Severity Index (EASI) that takes into account the area and extent of the manifestations of redness, thickening, scratching and lichenification. Patient Reports-Based Outcomes: The participants responded to a homogenous survey assessing their subjective experience on their symptoms, including itch severity and overall satisfaction with the treatment. The negative events or side effects were recorded during trial under the Safety Monitoring.
Statistical analysis
The data were expressed as mean±SD. A statistical comparison was made to compare the formulations according to their physicochemical characteristics and improvement in the severity of eczema. Analysis of Variance (ANOVA) was conducted to identify the significant differences between formulations. Tukey Really Significant Difference (HSD) test was used to conduct post-hoc comparisons. A p-value that fell below 0.05 was found to be statistically significant.
RESULTS
Nigella sativa oil analysis
Analysis of Nigella sativa oil using GC/MS revealed 18 peaks, indicating the presence of 18 compounds in the oil as shown in fig. 1. The components of the oil were analyzed and summarized in table 2.
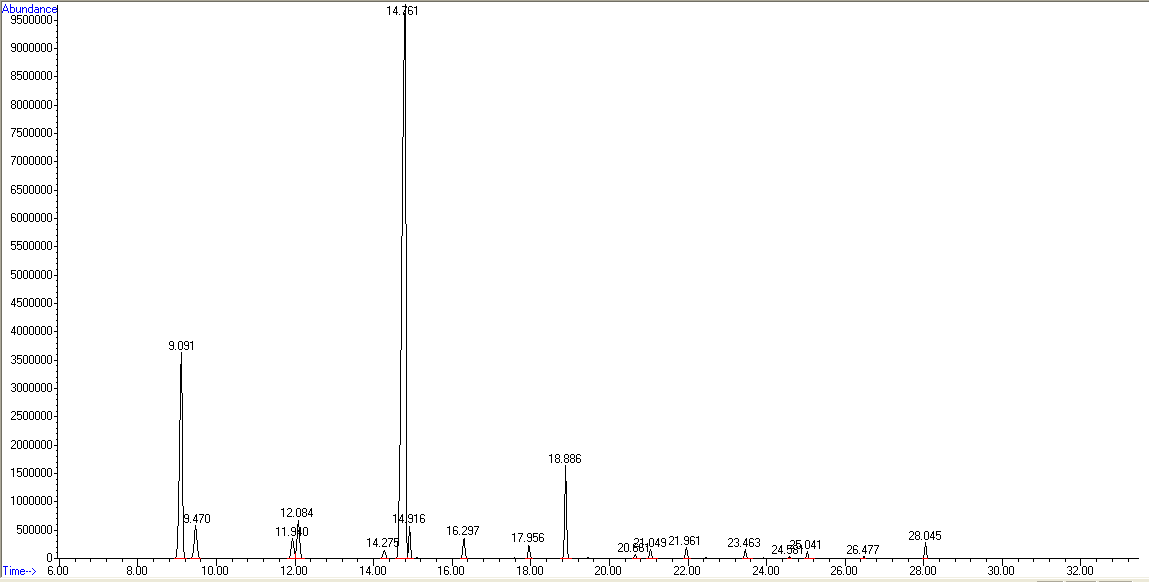
Fig. 1: Gas chromatography-mass spectrometry diagram illustrates the peaks of Nigella sativa oil components
Table 2: The main active constituents in Nigella sativa oil
| Peak number | Retention time | Percentage % | Compound |
| 1 | 9.0902 | 16.5968 | Alpha.-Thujene |
| 2 | 9.4704 | 2.6847 | Alpha.-Pinene |
| 3 | 11.9415 | 1.5594 | Beta.-Phellandrene |
| 4 | 12.084 | 2.8585 | Beta.-Pinene |
| 5 | 14.2767 | 0.3833 | Benzene, 1-methyl-2-(1-methylethyl)-(1-Methyl-2-isopropylbenzene |
| 6 | 14.7587 | 64.171 | Thymoquinone. |
| 7 | 14.9149 | 1.6621 | d-Limonene |
| 8 | 16.2998 | 1.1954 | Gamma.-Terpine |
| 9 | 17.9562 | 0.7151 | Gamma.-Terpinene |
| 10 | 18.8862 | 5.0077 | 1,4-Cyclohexadiene, 1-methyl-4-(1-methylethyl)- |
| 11 | 20.658 | 0.1972 | 1,4-Cyclohexadiene, 3-ethenyl-1,2-dimethyl- |
| 12 | 21.0518 | 0.4833 | 1,4-Cyclohexadiene, 1-methyl-4-(1-methylethyl)-(CAS) |
| 13 | 21.9615 | 0.5558 | Cis(2-((1E,3Z)-1,3-Butadienyl)cyclopentyl)methanol |
| 14 | 23.4618 | 0.4533 | 2,5-Cyclohexadiene-1,4-dione, 2-methyl-5-(1-methylethyl)-p-Cymene-2,5-dione |
| 15 | 24.5819 | 0.0707 | DELTA.3-Carene |
| 16 | 25.0435 | 0.328 | Carvacrol |
| 17 | 26.4759 | 0.0874 | Alpha.-Longipinene |
| 18 | 28.0441 | 0.7933 | (+)-Longifolene |
Results of evaluation test
The findings of the evaluation tests of the prepared Nigella sativa oil cream formulations are summarized in table 3. All 4 formulations had satisfactory physicochemical properties, including white color, characteristic odor, no phase separation, good homogeneity, and compliance with microbial content standards. The pH values were relatively close to the physiological skin range, suggesting that they would have no negative effects on the skin and suitable for topical application. Viscosity gradually increased among formulations, from 11,960 cps in Formula 1 to 31,405 cps in formula 4. The viscosity of Formula 3 and 4 was significantly higher than formula 1 and 2 (p<0.05). On the contrary, Formulas 3 and 4 had significantly lower spread ability than Formulas 1 and 2 (p<0.05). Of the creams tested, Formula 3 exhibited the most balanced profile of adequate viscosity (23,300 cps) to provide stability while also being acceptable spreadability (14.28 g·cm/s) for application. Based on these findings, Formula 3 was selected as the most optimal formulation for subsequent clinical evaluation in eczema patients.
Table 3: Physicochemical evaluation of Nigella sativa oil cream formulations
| Parameter | Formula 1 | Formula 2 | Formula 3 | Formula 4 |
| Color | White Cream | White Cream | White Cream | White Cream |
| Odor | Characteristic | Characteristic | Characteristic | Characteristic |
| Phase Separation | No phase separation | No phase separation | No phase separation | No phase separation |
| Homogeneity | Good | Good | Good | Good |
| pH Value* | 6.50±0.04a | 6.51±0.01a | 6.50±0.04a | 6.50±0.02a |
| Viscosity (cps)* | 11960±25.2a | 13513±35.2a | 23300±18.6b | 31405±20.4c |
| Spreadability (g‧cm/s)* | 20.87±0.11a | 19.02±0.09a | 14.28±0.13b | 11.35±0.07b |
| Microbial Content | Compliant | Compliant | Compliant | Compliant |
ᵃ,ᵇ,ᶜDifferent letters signify a significant difference. Means within each row that share the same letter are not significantly differing (p>0.05); means that share different letters are significantly differing (p<0.05) and were determined using one-way ANOVA with Tukey’s HSD test. Cps stands for Centipoise. *Mean±Standard deviation, n= 3.
Stability results
The findings of the stability tests of the creams prepared under accelerated conditions are detailed in table 4. All the formulations maintained acceptable pH values (6.39–6.51), and remained free from any microbial contamination throughout the storage period of 28 d. Formula 2 and Formula 4 underwent minimal changes in appearance (yellowing), while Formulas 1 and 3 retained their white color. Viscosity was shown to measurably decrease with storage time, combined with a slight increase in spread ability. Overall, formulation 3 was demonstrated the most stable physicochemical properties compared to the other formulations. Since formulation 3 shown the best results with stability under an accelerated condition, it was selected as the best formulation. Despite the formulations showed good short-term stability under accelerated conditions, but long-term stability studies are required to assign a definitive shelf life.
Table 4: Stability testing of Nigella sativa oil cream formulations under accelerated conditions
| Days | Temperature | Parameter | Formulations | |||
| F1 | F2 | F3 | F4 | |||
| 0 | Room temperature 25±1 °C |
Appearance | White Cream | White Cream | White Cream | White Cream |
| pH Value* | 6.50±0.04 | 6.51±0.01 | 6.50±0.04 | 6.50±0.02 | ||
| Viscosity (cps)* | 11960±25.2 | 13513±35.2 | 23300±18.6 | 31405±20.4 | ||
| Spreadability* (g•cm/s) | 20.87±0.11 | 19.02±0.09 | 14.28±0.13 | 11.35±0.07 | ||
| Microbial Content | Compliant | Compliant | Compliant | Compliant | ||
| 14 | T= 40±2 °C RH% = 75± 5% |
Appearance | No change | Yellowish white | No change | Yellowish white |
| pH Value | 6.44±0.03 | 6.42±0.05 | 6.48±0.02 | 6.46±0.05 | ||
| Viscosity (cps) | 8518±19.2 | 10211±24.0 | 17465±12.1 | 21340±14.8 | ||
| Spreadability (g•cm/s) | 30.58±0.14 | 23.89±0.07 | 16.97±0.14 | 14.31±0.11 | ||
| Microbial Content | Absent | Absent | Absent | Absent | ||
| 28 | T= 40±2 °C RH% = 75± 5% |
Appearance | No change | Yellowish white | No change | Yellowish white |
| pH Value* | 6.43±0.05 | 6.39±0.03 | 6.47±0.01 | 6.44±0.04 | ||
| Viscosity (cps)* | 8510±11.7 | 10090±16.0 | 17440±10.6 | 21215±16.9 | ||
| Spreadability * (g•cm/s) | 30.95±0.08 | 25.10±0.05 | 17.25±0.13 | 15.76±0.06 | ||
| Microbial Content | Absent | Absent | Absent | Absent | ||
*Mean±SD, n= 3, cps: Centipoise, F: Formulation, T: Temperature, RH: Relative humidity.
Table 5: Individual EASI scores and clinical outcomes at baseline and after 30 D of treatment with Nigella sativa oil cream (formula 3)
| Case | Sex | Age | Affected area | Baseline EASI | Day 30 EASI | % Change | Subjective assessment |
| 1 | M | 25 | Forehead | 21.0 | 1.8 | 91.4 | Complete recovery |
| 2 | M | 18 | Left elbow | 22.4 | 2.0 | 91.1 | Complete recovery |
| 3 | F | 26 | Right hand | 17.3 | 1.5 | 91.3 | Complete recovery |
| 4 | F | 22 | Both hands | 23.1 | 8.9 | 61.5 | Mild improvement |
| 5 | M | 19 | Both hands | 21.8 | 9.2 | 57.8 | Mild improvement |
| 6 | F | 24 | Both hands | 18.0 | 7.6 | 57.8 | Mild improvement |
| 7 | F | 22 | Both hands | 19.4 | 8.4 | 56.7 | Mild improvement |
| 8 | F | 23 | Feet fingers | 22.0 | 9.0 | 59.1 | Mild improvement |
| 9 | F | 24 | Cheeks | 16.3 | 7.2 | 55.8 | Mild improvement |
| 10 | F | 25 | Shoulders and face | 17.8 | 8.3 | 53.4 | Mild improvement |
| 11 | F | 23 | Face | 19.1 | 9.5 | 50.3 | Mild improvement |
| 12 | F | 39 | Left leg | 18.5 | 9.4 | 49.2 | Mild improvement |
| 13 | F | 24 | Forehead and nose | 22.2 | 10.6 | 52.3 | Mild improvement |
| 14 | F | 23 | Forehead | 17.4 | 8.6 | 50.6 | Mild improvement |
| 15 | F | 37 | Feet bottom | 20.5 | 12.5 | 39.0 | Light improvement |
| 16 | F | 40 | Face | 21.6 | 13.2 | 38.9 | Light improvement |
| 17 | F | 40 | Whole face | 18.7 | 12.1 | 35.3 | Light improvement |
| 18 | M | 21 | Back and shoulders | 23.4 | 14.8 | 36.8 | Light improvement |
Table 6: Summary of EASI score statistics for the study group
| Parameter | Baseline (Mean±SD) | Day 30 (Mean±SD) | % Reduction (Mean±SD) | p-value |
| EASI Score | 18.9±4.1 | 6.2±2.3 | 67.2±10.5 | <0.001 |
Clinical trial results
Eighteen patients with moderate-to-severe eczema were treated with 30 d of twice-daily applications of the optimized Nigella sativa oil cream (Formula 3). The outcomes are summarized in table 5. All patients had a mean EASI score of 18.9±4.1 at baseline, which indicates moderate-to-severe disease severity. The mean EASI score dropped to 6.2±2.3 after 30 d of treatment (p<0.001). A significant clinical improvement was indicated by the mean percentage decrease in the EASI score, which was 67.2±10.5%. Using percentage change in EASI as a measure, three patients (16.7%) achieved complete recovery (>90% reduction in EASI); eleven patients (61.1%) had mild improvement (50-90% reduction); and four patients (22.2%) had light improvement (<50% reduction). All participants completed the study, and no adverse effects or hypersensitivity reactions were observed. Representative clinical outcomes of patients are presented in fig. 2.
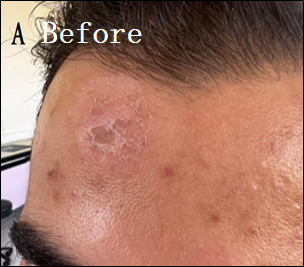
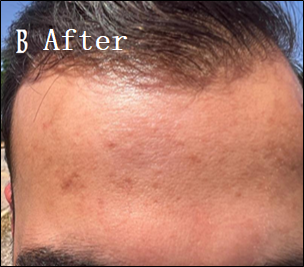
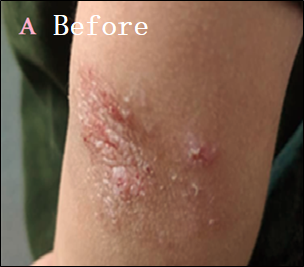
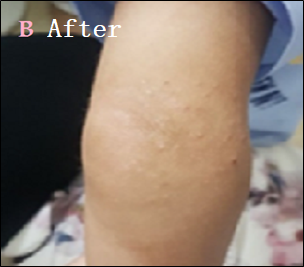
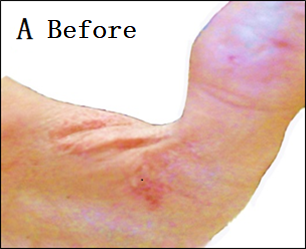
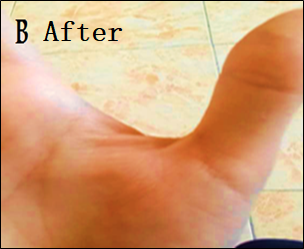
Fig. 2: Clinical outcomes of eczema patients before and after treatment using Nigella sativa oil cream for 30 d. (A) Before treatment. (B) After 30 d of treatment
DISCUSSION
This study confirms the practicality of creating a stable topical cream with Nigella sativa oil and provides early clinical evidence of its efficacy in eczema. Extraction by cold pressing preserved the compounds that were sensitive to heat, in particular thymoquinone, which can be destroyed by high temperature. This also justifies the relevance of this method to pharmaceutical uses [23]. The quality of the oil is important because the slightest changes in the chemical structure of the oil may influence its medicinal properties. We confirmed the presence of thymoquinone as the dominant constituent using our GC/MS analysis, which confirms previous studies that have associated this chemical to anti-inflammatory and antioxidant effects on skin diseases [24, 26]. We also found other terpenes that were present, such as α-thujene or α-pinene, which also supports the antimicrobial and protective activities of the oil [17]. Treating inflammation, as well as the microbial imbalance so common in eczema, the combination of these two has potential mechanistic explanations of the clinical benefit that was observed. The investigations carried out in the formulation point to the necessity to find a balance between the viscosity and spreadability in order to achieve the optimal topical administration. Formula 3 was ideal, considering its balanced viscosity and spreadability, ensuring both the stability of formulation and a patient-friendly application. Very high viscosity, as with Formula 4, may impede spreading and lower the rate of drug diffusion, while too low viscosity, such as in Formulas 1 and 2, compromises the stability. The intermediate viscosity of Formula 3 ensured good skin adhesion and homogenous application, enabling better delivery of active principles. Some of these patterns have been observed in other studies on herbal preparations, in which moderate levels of viscosity lead to a corresponding level of acceptability and skin adhesion [26-28]. Also, the cream formula (formula 3) was more able to retain its physicochemical characteristics in the course of stability analysis under the accelerated environment than the other prototypes. This is very important since herbal remedies are highly infamous due to instability due to oxidation and coloration. In this way, tocopherol acetate should have helped to stabilize the preparation by decreasing the oxidative stress [29]. It means that Nigella sativa oil can be successfully added to stable topical dosage forms when the excipients are used correctly. In cases of patients with moderate-to-severe eczema, the clinical examination is clear evidence of the effectiveness of the treatment. All patients tolerated the cream well, without any side effects, and all participants either fully recovered or showed partial improvement. These findings agree with an investigation conducted by Yousefi et al. [19], who found out that Nigella sativa is as effective as betamethasone in treating hand eczema and has fewer side effects. Our findings also validate the conclusions of Nasiri et al. [11], who carried out a systematic review and meta-analysis study to indicate the clinical efficacy of Nigella sativa in dermatologic disorders. Moreover, as pointed out by Hannan et al. [17], the absence of adverse effects in our study provides testimony to the fact that it is safe. These findings, therefore, suggest that Nigella sativa oil cream may be an effective and well-tolerated complementary natural treatment for eczema. Several limitations of the present study need to be acknowledged. The small sample size (n = 18) constrains the statistical power and generalizability of clinical outcomes. The study used a single-arm, non-controlled design; no placebo or vehicle comparator was included. The duration of the stability study and the clinical assessment periods was short (4 w and 30 d, respectively). Confirmation and extension of these preliminary results should be done in future studies, including larger randomized controlled trials with extended follow-up, comparative vehicle arms, and full long-term stability testing in accordance with ICH guidelines.
CONCLUSION
GC/MS analysis identified the existence of 18 bioactive compounds in Nigella sativa oil, with thymoquinone at 64.17% as the major constituent. All the prepared cream formulas of Nigella sativa oil demonstrated acceptable physicochemical properties, pH compatibility with skin, microbial safety, and homogeneity. Of these, Formula 3 showed an optimal balance between viscosity and spreadability, apart from excellent stability under accelerated storage conditions. Clinical evaluation of Formula 3 against moderate to severe eczema revealed a significant reduction in EASI scores, indicating a significant therapeutic improvement without any adverse side effects. These results suggest that the optimized Nigella sativa oil cream (Formula 3) has great potential as a safe and effective topical therapy for eczema. Further large-scale and controlled clinical studies are recommended to establish efficacy and elucidate mechanisms.
ACKNOWLEDGMENT
The authors would like to thank all the volunteers who participated in this research and all of the staff of the Faculty of Pharmacy, Al-Ittihad Private University, Syria, who assisted them in any way that they needed.
FUNDING
This research received no external funding.
AUTHORS CONTRIBUTIONS
This study was designed by Bassel Hussein, who was responsible for the entire methodology design, data collection, and extraction. Abdullah H Maad was responsible for writing the first draft of the manuscript, for data analysis, and for developing the recipe. Wael Alaghawani and Nour Alhabbal were in charge of patient enrollment, clinical assessment, and data curation. Rama Aziz conducted GC/MS analysis and interpreted the data. The final version of the text has been read and approved by all authors.
CONFLICT OF INTERESTS
The authors declare that there aren’t any competing interests with respect to this publication.
REFERENCES
Brown SJ. Atopic eczema. Clin Med (Lond). 2016;16(1):66-9. doi: 10.7861/clinmedicine.16-1-66, PMID 26833520.
Wollenberg A, Werfel T, Ring J, Ott H, Gieler U, Weidinger S. Atopic dermatitis in children and adults diagnosis and treatment. Dtsch Arztebl Int. 2023;120(13):224-34. doi: 10.3238/arztebl.m2023.0011, PMID 36747484.
Hanifin JM, Baghoomian W, Grinich E, Leshem YA, Jacobson M, Simpson EL. The eczema area and severity index-a practical guide. Dermatitis. 2022;33(3):187-92. doi: 10.1097/DER.0000000000000895, PMID 35594457.
Yang X, Pei X. Eczema compound patents herbal combination rules research based on complex system entropy clustering: a review. Medicine (Baltimore). 2022;101(49):e32005. doi: 10.1097/MD.0000000000032005, PMID 36626430.
Tramontana M, Hansel K, Bianchi L, Sensini C, Malatesta N, Stingeni L. Advancing the understanding of allergic contact dermatitis: from pathophysiology to novel therapeutic approaches. Front Med (Lausanne). 2023;10:1184289. doi: 10.3389/fmed.2023.1184289, PMID 37283623.
Eichenfield LF, Tom WL, Chamlin SL, Feldman SR, Hanifin JM, Simpson EL. Guidelines of care for the management of atopic dermatitis: section 1 diagnosis and assessment of atopic dermatitis. J Am Acad Dermatol. 2014;70(2):338-51. doi: 10.1016/j.jaad.2013.10.010, PMID 24290431.
Hengge UR, Ruzicka T, Schwartz RA, Cork MJ. Adverse effects of topical glucocorticosteroids. J Am Acad Dermatol. 2006;54(1):1-15. doi: 10.1016/j.jaad.2005.01.010, PMID 16384751.
Vieira BL, Lim NR, Lohman ME, Lio PA. Complementary and alternative therapies in dermatology. J Am Acad Dermatol. 2006;55(3):502-16. doi: 10.1016/j.jaad.2006.03.004.
World Health Organization. Integrating traditional medicine in health care. Geneva: World Health Organization; 2023.
Negi S, Verma S, RAJ P, Singh A. Novel approaches in ethosomal drug delivery: application in dermatology and beyond. Int J App Pharm. 2025;17(5):85-93. doi: 10.22159/ijap.2025v17i5.54400.
Nasiri N, Ilaghi Nezhad M, Sharififar F, Khazaneha M, Najafzadeh MJ, Mohamadi N. The therapeutic effects of Nigella sativa on skin disease: a systematic review and meta-analysis of randomized controlled trials. Evid Based Complement Alternat Med. 2022;2022:7993579. doi: 10.1155/2022/7993579, PMID 36518853.
De Oliveira YL, De Sa Resende A, Martins Filho PR, De Moura TR. Role of triggering receptor expressed on myeloid cells-1 (TREM-1) in COVID-19 and other viral pneumonias: a systematic review and meta-analysis of clinical studies. Inflammopharmacology. 2022;30(3):1037-45. doi: 10.1007/s10787-022-00972-6, PMID 35347523.
Chovatiya R, Paller AS. JAK inhibitors in the treatment of atopic dermatitis. J Allergy Clin Immunol. 2021;148(4):927-40. doi: 10.1016/j.jaci.2021.08.009, PMID 34437922.
Patil SB. Medicinal significance of Nigella sativa and its seeds: recent updates. Int J Pharm Sci Res. 2023;14(9):4277-84. doi: 10.13040/IJPSR.0975-8232.14(9).4277-84.
Al-Naqeep G, Ismail M. Nutritional quality of the black cumin seeds (Nigella sativa L.) and oil. J Am Oil Chem Soc. 2009;86:373-8. doi: 10.1007/s11746-008-1321-7.
Gholamnezhad Z, Havakhah S, Boskabady MH. Preclinical and clinical effects of Nigella sativa and its constituent thymoquinone: a review. J Ethnopharmacol. 2016;190:372-86. doi: 10.1016/j.jep.2016.06.061, PMID 27364039.
Hannan MA, Rahman MA, Sohag AA, Uddin MJ, Dash R, Sikder MH. Black cumin (Nigella sativa L.): a comprehensive review on phytochemistry, health benefits, molecular pharmacology and safety. Nutrients. 2021;13(6):1784. doi: 10.3390/nu13061784, PMID 34073784.
Khudhur ZO, Maad AH, Ghanimi H, Abdolmaleki A. Fullerene nanoparticle as new therapeutic agent for the nervous system disorders. Nanomed J. 2024;11(4):342-59. doi: 10.22038/NMJ.2024.78043.1903.
Yousefi M, Barikbin B, Kamalinejad M, Abolhasani E, Ebadi A, Younespour S. Comparison of therapeutic effect of topical Nigella with betamethasone and eucerin in hand eczema. J Eur Acad Dermatol Venereol. 2013;27(12):1498-504. doi: 10.1111/jdv.12033, PMID 23198836.
Rahmani AH, Aly SM. Nigella sativa and its active constituents, thymoquinone shows pivotal role in the diseases prevention and treatment. Asian J Pharm Clin Res. 2015;8(1):48-53.
Hwang JR, Cartron AM, Khachemoune A. A review of Nigella sativa plant-based therapy in dermatology. Int J Dermatol. 2021;60(12):e493-9. doi: 10.1111/ijd.15615, PMID 33899217.
Shivathaya N, Surve R, Sawant R, Khot S, Biradar K, Verma R, Gorav A. Formulation and in vitro evaluation of ethanolic extract of polyherbal face cream. Int J Curr PharmSci. 2022;14(2):41-7. doi: 10.22159/ijcpr.2022v14i2.1950.
Rahim MA, Shoukat A, Khalid W, Ejaz A, Itrat N, Majeed I. A narrative review on various oil extraction methods, encapsulation processes fatty acid profiles oxidative stability and medicinal properties of black seed (Nigella sativa). Foods. 2022;11(18):2826. doi: 10.3390/foods11182826, PMID 36140949.
Kmail A, Said O, Saad B. How thymoquinone from nigella sativa accelerates wound healing through multiple mechanisms and targets. Curr Issues Mol Biol. 2023;45(11):9039-59. doi: 10.3390/cimb45110567, PMID 37998744.
Tan PL, Rajagopal M, Chinnappan S, Selvaraja M, Leong MY, Tan LF. Formulation and physicochemical evaluation of green cosmeceutical herbal face cream containing standardized mangosteen peel extract. Cosmetics. 2022;9(3):46. doi: 10.3390/cosmetics9030046.
Kohandel Z, Farkhondeh T, Aschner M, Samarghandian S. Anti-inflammatory effects of thymoquinone and its protective effects against several diseases. Biomed Pharmacother. 2021;138:111492. doi: 10.1016/j.biopha.2021.111492, PMID 33743334.
Badwaik CB, Lade UB, Agarwal T, Barsagade P, Nandgave M, Gaddamwar N. Formulation and evaluation of herbal face cream. Int J Pharm Res Appl. 2022;17(1):955-60.
Ahmed Y, Maad AH, Hassan HA, Abdallah DB, Yousef M, Kadhum H. Evaluation of carboxymethyl millet starch and pregelatinized millet starch as pharmaceutical excipients using factorial experimental designs. Int J App Pharm. 2025;17(2):321-8. doi: 10.22159/ijap.2025v17i2.52161.
Goncalves GM, Srebernich SM, Souza JA. Stability and sensory assessment of emulsions containing propolis extract and/or tocopheryl acetate. Braz J Pharm Sci. 2011;47(3):585-92. doi: 10.1590/S1984-82502011000300016.