
Int J App Pharm, Vol 18, Issue 1, 2026, 551-557Original Article
INCREASED SUSCEPTIBILITY TO GASTRIC ULCERATION IN DIABETIC RATS: SYNERGISTIC EFFECT OF QUERCETIN AND PIOGLITAZONE
GHASSAN ADEL ALKHALIFAH1, PREM KUMAR NANJUNDAN2*, AFZAL HAQ ASIF1*, SREEHARSHA NAGARAJA3
1Department of Pharmacy Practice, College of Clinical Pharmacy, King Faisal University, Al-Ahsa-31982, Saudi Arabia. 2*Department of Pharmacology, Krupanidhi College of Pharmacy, Bangalore-560035, India. 3Department of Pharmaceutical Sciences, College of Clinical Pharmacy, King Faisal University, Al-Ahsa-31982, Saudi Arabia
*Corresponding author: Prem Kumar Nanjundan; *Email: premkrupanidhi@gmail.com
Received: 20 Apr 2025, Revised and Accepted: 27 Oct 2025
ABSTRACT
Objective: This research delves into the potential therapeutic benefits of quercetin, a prominent flavonoid, in addressing diabetes and associated ulcers, emphasizing its synergistic effects with oral hypoglycemic agent, pioglitazone.
Methods: Sprague-Dawley rats received a high-fat diet and 35 mg/kg streptozotocin to mimic human diabetic condition. These rats were exposed to drug (indomethacin) and alcohol induced gastric ulceration, to study the flavonoid’s effect alone and in combination with pioglitazone (low doses) in type 2 diabetes enhanced gastric mucosa sensitivity towards ulcerogenic insults. Quercetin and pioglitazone administered alone and in combination over eight-week period. OGTT, antioxidant status was estimated at the end of the study. Ulcer score and index were also determined by using respective standards.
Results: Monotherapy of pioglitazone (low dose) and quercetin exhibited limited effectiveness in increasing insulin sensitivity and ulcer prevention. Blood sample analyses, unveiled elevated antioxidant levels in the combination group compared to monotherapy. Notably, low dose combinations of quercetin and pioglitazone effectively displayed better healing-promoting properties. The combination of low doses of quercetin and pioglitazone demonstrated superior efficacy in ameliorating resistance towards insulin, physiological restoration, and prevention of stomach ulcers in diabetic rats.
Conclusion: The synergistic effects of quercetin and pioglitazone at low doses offers promising avenues for slowing the progression of diabetic gastric ulcers by enhancing antioxidant levels and maintaining physiological balance. Our findings open new possibilities for innovative treatment strategies in the realm of diabetes and related complications.
Keywords: Diabetic ulcers, Insulin resistance, Pioglitazone, Quercetin, Antioxidant levels
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.54668 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Ulcer formation stems from a discrepancy in the acidic peptic secretions and weakened mucosal resilience, the diabetic condition negatively impacts various gastrointestinal functions. In insulin-dependent diabetic rats, both starvation and stress contribute to cancer genic responses of gastric mucosa [1]. Diabetes amplifies vulnerability of mucosa layer to ulcer-causing factors, predisposing individuals to gastric ulcers. This highlights the complex interplay between diabetes and gastrointestinal well-being, emphasizing the increased likelihood of ulcer development in diabetic states due to compromised mucosal integrity and heightened responsiveness to cancer genic triggers [2]. In diabetic animals, multiple ulcerogens, like ethanol and non-steroidal anti-inflammatory agents caused increase vulnerability of gastric mucosa, i. e increase in susceptibility of gastric mucosa in causing ulceration [3, 4].
Lipid peroxidation is suggested as a crucial factor inulcerogenesis, contributing to acute hemorrhagic ulceration through increased back diffusion of acid [5]. Non-insulin-dependent diabetic (NIDDM) rats display an elevated predisposition to ulceration across studied gastric ulcer models. Despite varied causative factors, the root cause of ulcer genesis is perceived as an imbalance between offensive and defensive factors. Optimal glycemic control and achieving nonglycemic goal, like the antioxidant status as well as lipid levels are imperative in preventing long-lasting diabetes induced comorbid conditions [6]. Diabetic ulcer genesis is mitigated by exogenous supplements of antioxidants that could regularize the gastric motility, impaired duodenal bicarbonate secretion [7, 8].
In streptozotocin-induced diabetic rats, flavonoids like quercetin foster pancreatic islet regeneration, likely enhancing insulin release. Notably, quercetin's antioxidant-scavenging activity delays lipid peroxidation and hinders CuZ-induced LDL oxidation by chelating copper ions [9-11]. The restoration of antioxidant enzymes in diabetic ulcer was due to protective nature of quercetin, a powerful antioxidant [12]. Pioglitazone exhibited gastric ulcer healing in rats by activating PPAR-ɣ receptors and hence stimulating angiogenesis near to the margin of ulcer [13]. Proton pump inhibitors (PPI) are widely meant for treating gastric ulcers but on long term use, they are associated with renal disease, calcium and vitamin B12 deficiencies [14]. Proton pump inhibitors are bound to increase the levels of gastrin via negative feedback [15] and gastrin has been proved to secrete insulin in animal models [16, 17]. So simultaneously usage of PPI with oral hypoglycemics could lead into hypoglycemic episodes when therapy is for a longer duration of time. This study aims to investigate the impact of quercetin and pioglitazone and their low dose combination on gastric mucosa of diabetic rats that were susceptible to ulcerogenic stimuli as well as prevent hypoglycemic episodes
MATERIALS AND METHODS
Drugs and chemicals
Streptozotocin, omeprazole and and absolute ethanol were aquired from Sigma Aldrich Ltd, USA; Quercetin and pioglitazone from Sisco Research Laboratories, India. Analytical grade chemicals were used for the rest of the protocols.
Sprague Dawley rats (150-180g),received from our institutional animal house, wereshelteredinawell-ventilated animal facility, at a regulated temperature of 25°±5 °C and a 12:12 day-night cycle. Experimental methodology along with the protocol was approved by the InstitutionalAnimal Ethics Committee (KCP/IAEC-34/2011-12), in accordance with the standard conditions and guidelines established by the Committee for control and supervision of experiments onanimals (CCSEA). Handling of animals was as per the guidelines of the ethical committee.
Experimental models
Induction of type 2 diabetes in rats through high-fat diet and low-dose streptozotocin treatment
Rats was fed with modified diet for few weeks, then given i. p injections of subdiabetagenic dose of streptozotocin i. e 35 mg/kg following an overnight fast. To prevent hypoglycemic mortality, those injected with streptozotocin received a 5% w/v glucose for six hours post dosage of streptozotocin. Diabetic classification relied on non-fasted plasma glucose levels ≥300 mg/dl, assessed from tail vein blood samples using an AccuCheck glucose diagnostic kit [18].
Experimentdesign
Rats were assigned into eight groups (n=08). Group I: Normal Control (untreated, non-diabetic rats), received salineorally. The rats that developed diabetes were split into various groups such as Group II: Negative control (untreated, diabetic rats), Group III: Diabetic rats with low dose Pioglitazone treatment (07 mg/kg) [19], Group IV: Diabetic rats with Quercetin therapy (100 mg/kg) [20]. Diabetic rats with low dose of pioglitazone (07 mg/kg)+Quercetin treatment (50 mg/kg)was considered as Group V. Omeprazole (n=10) and sucralfate (n=10) were used as standard in respective animal model. The therapy commenced prior to diabetes induction and continued for 8 w.
Single and multiple dose studies in rats with diabetes
Single dose study
The study involved segregating animals into five groups, including Group I –Non diabetic rats (Control), Group II-diabetic control rats, group III received quercetin (50 mg/kg), while the fourth group was admin istered pioglitazone (7 mg/kg). Blood sugar levels measured at specified time.
Multiple dose study
In the prolonged multi dose study, oral treatment of the sample was performed daily for ten days. Serum glucose concentration was assessed from tail-tip blood sample son days 1, 3, 7, and 11 post-administrations. Additionally, body weights were recorded on the 10th day for all animals [21].
Oral glucose tolerance test (OGTT)
Post twelve hour fasting state, oraldoseof2.0g/kg glucose was given to rats and blood sugar levels estimated at specified intervals [22].
Recovery from drug-induced gastric ulcers: Investigation of free radical scavenging capabilities
Rats develop gastric ulcers through a five-day regimen of indomethacin administration (5 mg/kg,p. o). After ulcer formation, treatment will be administered for five days, with the control group receiving only the vehicle and omeprazole was used as standard drug. The rats were sacrificed using anaesthetic ether. The stomach was removed; ulcerscore and ulcerindex was determined [23, 24]. Theglandularportion ofthestomach used for the estimation of mucin content [25], total proteins [26] antioxidant factors like superoxide dismutase activity [27], and catalase activity [28].
The number of ulcers was noted and the severity was recorded with the following scores: 0 = no ulcer, 1 = superficial ulcer, 2 = deep ulcers, 3 = perforation.
Evaluation
An ulcer index UI is calculated by the formula: UI=UN+US+UP *10-1
UN – Average number of ulcers per animal, US – Average of severity score, UP – percentage of animals with ulcers [29]
Ethanolinducedulcer
Prior to the administration of 90% ethanol (1 ml/200 gm), albino rats underwent a 36-hour fast. The drug was given one hour before ethanol administration. Following ethanol exposure, the animals were sacrificed one hour later, and the stomach was isolated for the determination of the ulcer index. Sucralfate was used as standard drug [30].
Evaluation of antioxidant levels in tissue homogenate
Stomach homogenate preparation
Post conclusion of treatment period, stomachwas homogenizedwith cold 0.1 N perchloric acidand disodium EDTA. Isopropyl homocholine served as an internal standard. The homogenized mixture was then subjected to centrifugation (48 degree centigrade) for 20 min at 10,000 rpm [31].
Quantification of gastric content
The gastric glandularmucouswasmeasured. Excised stomach was soaked for a period of two hours in 0.1%alcian blue. Stomach was cleaned for two times using sucrose (0.25 M). Magnesium chloride (0.5 m) was used to remove the dye and the obtained solution was mixed with diethyl ether. The optical density of the aqueous phase was measured at 605 nm and the concentration (expressedinmg) of dye was calculated by using a standard curve. Finally, the dye’s weight was revealed over the weight of the stomach [32].
Quantification oftotalprotein
Protein concentration was measured through Lowry's method, using bovine serum albumin (BSA) as the benchmark standard [33].
Quantification of enzyme activity
A solution comprising of gastric tissue homogenate (100 µl), sodium carbonate (01 ml), NBT (0.4 ml), and EDTA (0.2 ml) was prepared. The resulting supernatant was utilized to assess superoxide dismutaseand catalase activity [34].
StatisticalInterpretation of the data
The information is laid out in the form of mean±standard error of the mean (SEM). Group distinctions were assessed utilizing one-way analysis of variance (ANOVA), succeeded by Tukey’s post hoc multiple comparison examination. Statistical significance was determined for P values less than 0.05 [35].
RESULTS
Single as well as repeated dose study of quercetin and pioglitazone in rats
Two weeks, post STZ administration, the rats exhibited elevated blood glucose concentration than control rats. In the single dose study, quercetin and pioglitazone exhibited decline in blood glucose concentration by 26.24 % as well as 08.6 % respectively. Repeated dosage study confirms that the quercetin as well as pioglitazone displaying a remarkable decline in blood concentration of glucose from 38.2 to 42.6 % and 14.23 to 17.86% respectively between 9th and 11th d of treatment. Quercetin being more effective than pioglitazone.
Effect of quercetin, pioglitazone and their combination on glucose tolerance in diabeticrats
In glucose tolearance test, we observed a decline of blood glucose concentration was exceptional with quercetin alone treated rats at 2 h post glucose load. The % increase in blood glucose level at 30th min and 60th min was more pronounced in rats treated with pioglitzone than the group treated with quercetin. i. e. quercetin exhibtedmore tolerance towards glucose loadthan pioglitazone. Percentage decrease of blood glucose shown by pioglitzone alone was more pronounced at 2nd h in contrast to quercetin’s response. Quercetin (Low dose) as well as pioglitazone (Low dose) when administered together, unveiled a remarkable decline in glucose concentration of blood in comparison with untreated diabetic rats and with group of diabetic rats treated with pioglitazone alone (graph 1).
Effect of quercetin, pioglitazone and quercetin plus pioglitazoneon antioxidant’s status
A remarkable short fall of content of super oxide dismutase as well as catalase was observed in post eight week treated diabetic rats and marked comparative response was noticed when matched with control rats. Post eight weeks treatment period, quercetin, pioglitazone and their combination restored the short fall of super oxide dismutase as well as catalase but the combined therapy of quercetin and pioglitazone resulted in a better response (table 1) than when treated alone.
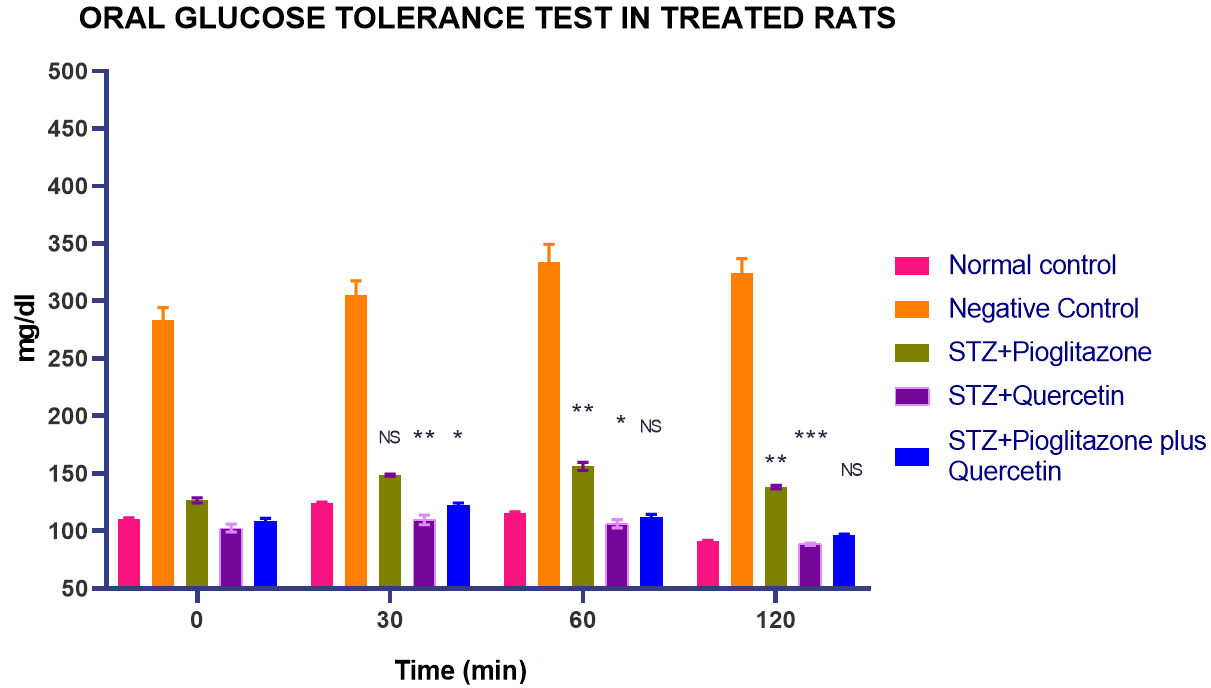
Graph 1: Quercetin alone and in combination with pioglitazone on OGTT in treated rats. All values are represented as mean±SEM, n= 10, ***p<0.001, **p<0.01, *p<0.05, NS = Not significant when all groups were compared with normal control group
Table 1: The influence of quercetin, pioglitazone and their combination on mucin content (μg/gm), total proteins (mg/ml), and SOD (U/mg of protein), CAT (U/mg of protein) observed in indomethacin-induced gastric ulcers model
| Parameter | Mucin content (μg/gm) | Total proteins | SOD (U/mg) | CAT (U/mg) |
| Normal Control | 0.55±0.14 | 18.5±1.7 | 21.3±1.58 | 17.3±1.11 |
| Negative Control | 0.097±0.022*** | 8.7±0.83*** | 9.08±0.74*** | 4.56±0.66*** |
| Omeprazole | 2.24±0.28a*** | 12.65±0.87a*** | 10.3±0.49*** | 7.11±0.91*** |
| Sucralfate | 1.42 ±0.49a*** | 11.75±0.25a*** | 9.45±0.22*** | 5.16±0.31*** |
| Pioglitazone (Pio) | 0.15±0.022*** | 14.06±0.80a*** | 18.9±0.66a** | 13.6±0.9a** |
| Quercetin (QE) | 1.24±0.16a*** | 14.6±0.61a*** | 16.75±0.92a** | 12.05±0.77a*** |
| Pio+QE | 1.74±0.13a*** | 15.9±0.85a* | 17.3±0.9a* | 14.7±0.84a* |
Results are mean±SEM of 6 rats in each group. One-way ANOVA followed by Tuckey’s test for multiple compar isons was applied for comparing the parameters with NC and DC groups. The difference was considered to be significant when ***P<0.001, **P<0.01, *P<0.05 when compared to normal control group and aP<0.001, bP<0.01, cP<0.05 when compared to diabetic control group. Pio – Pioglitazone, QE-Quercetin
Assessment of the anti-ulcer activity in ulcer models induced by indomethacin and ethanol
Ethanol-ulcerated rats displayeda remarkable (p<0.001) percentage elevation of ulcerated area in comparison with control group (table 2.1). In contrast, pre-treatment with sucralfate, low dose of pioglitazone (Pio) and Quercetin (QE) and their combination (Pio+QE) manifested a less mucosal injury as witnessed by a significant (p<0.01) decline in percentage of ulcerated area by 81.46%, 19.87%, 41.75 %, and 43.05 %, respectively, comparedF to untreated ethanolulcerated rats. Combination of pioglitazone and quercetin showed a better protective effect than when treated alone. (table 2.1)
Table 2: Effect of quercetin (QE), pioglitazone and their combination on ulcer ındex and ulcer score
Ethanol induced ulcer model
| Groups | Ulcer ındex | Ulcer score |
| Normal Control | 1.59±0.12 | 7.5±0.42 |
| Negative Control | 3.04±0.10*** | 15.1±0.56*** |
| Sucralfate | 0.11±0.02a*** | 2.8±0.70a*** |
| Pioglitazone (Pio) | 1.46±0.05a!!! | 12.1±1.1***!!! |
| Quercetin (QE) | 0.88±0.05a***!!! | 8.8±0.03a!!! |
| Pio+QE | 0.41±0.06a***!! | 8.6±0.40a!!! |
The Mean±SEM for 6 rats/group underwent one-way ANOVA with Tukey’s test. Significance levels: ***P<0.001, **P<0.01, *P<0.05 vs. NC; aP<0.001, bP<0.01, cP<0.05 vs. DC; !!!P<0.001, !!P<0.01, !P<0.05 vs. sucralfate-treated group. Pio – Pioglitazone, QE-Quercetin
Drug (Indomethacin)-ulcerated rats unveileda exceptional (p<0.001) percentage surge of ulcerated area in comparison with control group (table 2.2). In contrary, pre-treatment with a standard drug, omeprazole, low dose of pioglitazone (Pio) and Quercetin (QE) and their combination (Pio+QE) demonstrated a less mucosal injury as observed by a significant (p<0.01) decline in percentage of ulcerated area by 76 %, 18.54 %, 39.33 %, and 52.25 %, respectively, compared to untreated drug induced ulcerated rats. Low dose pioglitazone and quercetin elicited the best protective effect than when treated alone (table 2.2) (fig. 1).
Macroscopic features of gastric ulcers in different groups (Indomethacin and Ethanol Models)
Efect of quercetin (QE) and pioglitazone (Pio) and their combination (QE+Pio) on lesion score records obtained from the histological examination in Indomethacine-induced gastric ulcer in rats.
Table 3: Indomethacin induced ulcer model
| Groups | Ulcer ındex | Ulcer score |
| Normal Control | 1.62±0.18 | 10.8±0.70 |
| Negative Control | 2.32±0.49*** | 17.8±0.96*** |
| Omeprazole | 0.34±0.08a*** | 4.3±0.55a*** |
| Pioglitazone (Pio) | 1.91±0.05c!!! | 14.5±0.56c**!!! |
| Quercetin (QE) | 1.17±0.07a!!! | 10.8±0.60a!!! |
| Pio+QE | 0.97±0.04a!! | 8.5±0.61a!!! |
The Mean±SEM for 6 rats/group underwent one-way ANOVA with Tukey’s test. Significance levels: ***P<0.001, **P<0.01, *P<0.05 vs. NC; aP<0.001, bP<0.01, cP<0.05 vs. DC; !!!P<0.001, !!P<0.01, !P<0.05 vs. omeprazole-treated group. Pio – Pioglitazone, QE-Quercetin
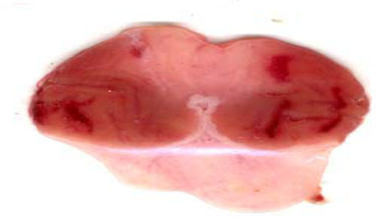
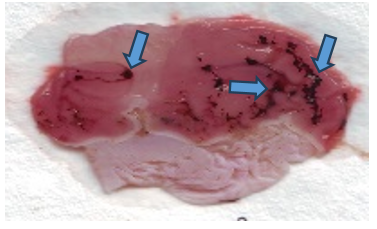
Normal control Negative control
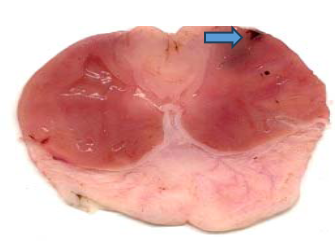
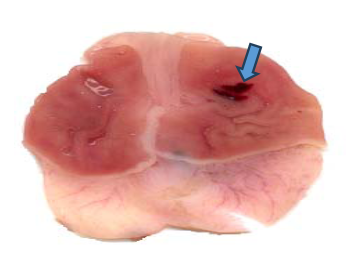
Sucralfate Omeprazole
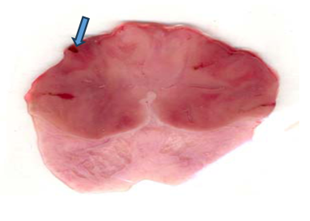
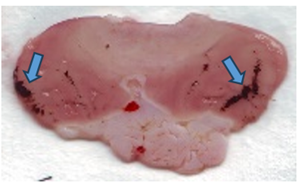
QE PIO
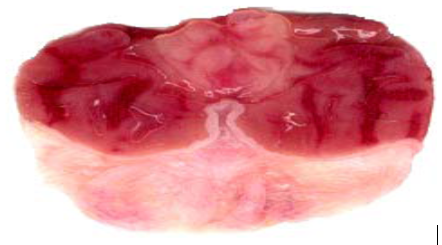
QE+PIO
Fig. 1: Efect (Photographic representation) of pioglitazone (Pio) and quercetin (Q) and their combination (Pio+Q) on gross gastric lesions in diabetic gastric ulcer in rats. Blue arrow: ulcerated areas. (Scale= 1 mm)
Table 4: Macroscopic features of gastric ulcers in different groups (Indomethacin and Ethanol Models)
| Group | Erosion/ulceration | Hemorrhagic patches | Infammatory cells infltrate | Blood vessels congestion | Oedema |
| Normal Control | - | - | - | - | - |
| Negative Control | +++ | +++ | ++ | +++ | +++ |
| Omeprazole | - | - | ++ | ++ | ++ |
| Pioglitazone (Pio) | ++ | ++ | + | ++ | ++ |
| Quercetin (QE) | + | - | + | + | ++ |
| Pio+QE | - | - | + | - | + |
Nil (-) = no lesion was evidenced,+= mild lesion records evidenced in less than 15% of examined tissue sections,++= moderate lesion records evidenced in 16–35% of examined tissue sections,+++= severe lesion records evidenced in more than 35% of examined tissue sections. Pio – Pioglitazone, QE-Quercetin
Table 5: Efect of quercetin (QE) and pioglitazone (Pio) and their combination (QE+Pio) on lesion score records obtained from the histological examination in Ethanol-induced gastric ulcer in rats
| Group | Erosion/ulceration | Hemorrhagic patches | Infammatory cells infltrate | Blood vessels congestion | Oedema |
| Normal Control | - | - | - | - | - |
| Negative Control | +++ | +++ | ++ | +++ | +++ |
| Sucralfate | - | - | ++ | + | ++ |
| Pioglitazone (Pio) | + | + | + | + | ++ |
| Quercetin (QE) | ++ | - | ++ | ++ | ++ |
| Pio+QE | - | - | + | + | + |
Nil (-) = no lesion was evidenced,+= mild lesion records evidenced in less than 15% of examined tissue sections,++= moderate lesion records evidenced in 16–35% of examined tissue sections,+++= severe lesion records evidenced in more than 35% of examined tissue sections. Pio – Pioglitazone, QE-Quercetin
DISCUSSION
Metabolic disorder is designated by congregate of unhealthy changes such as obesity, impaired glucose tolerance (IGT) and resistance to insulin activity. Modification of diet containing high amount of fructose was developed in our laboratory [18] because the incidences of developing diabetic complications is more vulnerable when fructose is consumed instead of glucose [36, 37]. The complicatedness and the vulnerability of complexity raises as the uncontrolled hyperglycemic state escalate [38]. Gastric ulceration in NIDDM is predisposed due to its defenselessness to ulcerogenic stimuli has been reported several times. Visceral autonomic neuropathy [39], caused due to increase in free radicals is generated by uncontrolled diabetes mellitus. The therapeutic agents possessing both anti-hyperglycemic and anti–ulcer activities are the choice of agents to treat diabetes induced ulcer. These agents could prove more cost-effective and less incidence of adverse effects.
Quercetin, among many flavonoids, causes enhanced hypoglycemic condition through promotion of beta cell proliferation and insulin secretion. We observed a marked decline in postprandial glucose strength and enhanced insulin sensitivity index from 30 min onwards during OGTT in diabetic rats and was identical with previous results that were proved in earlier studies [40, 41]. Choi et al. also demonstrated that the quercetin at a dose of 100 mg/kg, ameliorated resistance towards insulin. It also lowered the sustained raise of glucose post 30 min during OGTT [42]. Gohlke et al. observed that the plasma concentration of glucose declined, which might be due to elevated insulin release and insulin sensitivity when quercetin was administered to diabetic rats [43]. Quercetin was proven to heal gastric ulcer caused by indomethacin in diabetic rats [44].
The increase in insulin sensitivity produced by quercetin was much pronounced when compared with the low dose of pioglitazone. L. Past studies have proven that quercetin exhibited the mechanism as that of rosiglitazone in enhancing the utility of glucose through glucose transports [45, 46]. Querctin could be effective in diminishing fasting as well as postprandial blood glucose concentration in diabetic animal model [47]. High doses of quercetin and pioglitazone, when administered for a prolong time might lead to hypoglycaemic incidences. Previous studies have also show that pioglitazone has the tendency to cause severe fluid retention. Hence, adopted a combined reduced dose of both quercetin and pioglitazone to reduce the hypoglycemic when treated for a longer period. This combination had shown significant increase in insulin sensitivity and reduced the postprandial glucose level significantly, which is very much essential in preventing the increase in susceptibility of gastric mucosa to ulcerogenic stimuli. Our earlier study demonstrated the combined effect of an antidiabetic drug (acarbose) with Syzygium cumini seed extract in countering the susceptibility of diabetic gastric mucosa towards ulcerogenic stimuli. This combination was much superior to quercetin [20]. Syzygium cumini and acarbose have been proven to possess the α-glucosidase inhibition that is very much essential in curbing the postprandial glucose level. Similarly, Joshi M. C. et al. proved the α-glucosidase indering action of quercetin [48].
Pretreatment with pioglitazone has reduced the percentage of ulerative area when compared to untreated ethanol ulcerated rats [49]. Brzozowski et al. have demonstrated the gastric ulcer healing effect of pioglitazone via the stimulation PPAR-ɣ receptors and enhance the angiogenesis around ulcer margin [50]. Previous study has mentioned the anti-inflammatory effect of pioglitazone was through the suppression of proinflammatory cytokines and cyclooxygenase-2 [51]. AMPK signaling pathway corrects resistance towards insulin that is independent to the insulin-regulated mechanism for the generation of GLUT4. Quercetin as well as insulin-sensitizing agents, are proved for the management type 2 diabetes via AMPK pathway activation [52, 53].
In the present study, the pioglitazone (low dose) had shown less significant effect in decreasing postprandial glucose level and could be the reason for the less ulcer protective effect but the low dose combination of both the agents (quercetin and pioglitazone) had a noteworthy effect in decreasing the postprandial glucose level, thus ameliorating diabetes induced susceptibility to ulcerogenic stimuli.
Type 2 diabetes rats have exhibited an enhanced propensity towards gastric ulceration due to elevated offensive factors and simultaneous decline in antioxidant status there by increasing the ulcer index values [54, 55]. The elevated offensive and submissive defensive factors were ameliorated due to the effect of quercetin on antioxidant scavenging activity, delay in peroxidation of lipids as well as reduced copper induced LDL oxidation. Identical results were documented elsewhere also [56-58]. Segovia G. R et al. have proven that quercetin was effective in preventing ulcer in diabetic rats by restoring the levels of SOD and catalase [59]. Similarly, present study has also exhibited an increase in antioxidant enzyme status in hyperglycemic rats on quercetin therapy alone and as well as when combined with pioglitazone.
The combined effect of low dose of quercetin and pioglitazone have proven more effective in decreasing the vulnerability gastric lining towards ulcerogenic agents due to their balancing nature effect between offensive as well as defensive factors on gastric mucosa on elevated acid – pepsin secretion prompted by uncontrolled diabetic condition.
In view of the above evaluation, we agree that the simultaneous therapy of quercetin with pioglitazone at doses lower than that of conventional usage would be beneficial in preventing the susceptibility of diabetic gastric mucosa towards ulcerogenic agent by amelioarting oxidative stress as well as by effective postprandial glucose suppression leading to gastro protective effect.
CONCLUSİON
Approach of reduced dosage of both agents provides ulcer amelioarting effect in diabetic gastric mucosa susceptible to ulcerogenic agents as well preventing hypoglycemia. In constinuation of this study, further study is neccessary to prevent the adverse effect (hypoglycemia) of the conventional antidiabetic agents when therapy is for a prolong period. Before proposing for clinical trial, our study outcome has opened new possibilities for the delay in the progression or prevention the susceptibility of diabetic gastric mucosa towards ulcerogenic stimuli and long term study is essential before proposing for clinical trials inorder to validate efficacy in diabetic patients with gastric ulcers.
ACKNOWLEDGMENT
This work was supported by the Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research, King Faisal University, Saudi Arabia [Project No. KFU253279].
FUNDING
Nil
AUTHORS CONTRIBUTIONS
Prem Kumar Nanjundan: supervising, conceptualizing, literature review, writing original draft, and critical evaluation,;Ghassan Adel Alkhalifah: data curation, literaturereview, writing original draft; SreeharshaNagaraja: criticalevaluation, editing, and proof reading; Afzal Haq Asif: critical evaluation, editing, and proof reading.
CONFLICT OF INTERESTS
Declared none
REFERENCES
Tashima K, Korolkiewicz R, Kubomi M, Takeuchi K. Increased susceptibility of gastric mucosa to ulcerogenic stimulation in diabetic rats role of capsaicin-sensitive sensory neurons. Br J Pharmacol. 1998 Aug;124(7):1395-402. doi: 10.1038/sj.bjp.0701974, PMID 9723950, PMCID PMC1565532.
Vador N, Jagtap AG, Damle A. Vulnerability of gastric mucosa in diabetic rats its pathogenesis and amelioration by Cuminum cyminum. Indian J Pharm Sci. 2012;74(5):387-96. doi: 10.4103/0250-474X.108413, PMID 23716866.
Goldin E, Ardite E, Elizalde JI, Odriozola A, Panes J, Pique JM. Gastric mucosal damage in experimental diabetes in rats: role of endogenous glutathione. Gastroenterology. 1997;112(3):855-63. doi: 10.1053/gast.1997.v112.pm9041247, PMID 9041247.
Brzozowska I, Targosz A, Sliwowski Z, Kwiecien S, Drozdowicz D, Pajdo R. Healing of chronic gastric ulcers in diabetic rats treated with native aspirin nitric oxide (NO)-derivative of aspirin and cyclooxygenase (COX)-2 inhibitor. J Physiol Pharmacol. 2004;55(4):773-90. PMID 15613743.
Ommurugan B, Rao V. Pharmacotherapy of peptic ulcer disease and latest research. In: Maria Roesler BM, editor. Gastritis new approaches and treatments. Gastritis new approaches and treatments. IntechOpen; 2019. doi: 10.5772/intechopen.86386.
Chandra P, Kaleem M, Sachan N, Pathak R, Alanazi AS, Alsaif NA. Gastroprotective evaluation of Medicago sativa L. (Fabaceae) on diabetic rats. Saudi Pharm J. 2023;31(11):101815. doi: 10.1016/j.jsps.2023.101815, PMID 37860685.
Perdichizzi G, Bottari M, Pallio S, Fera MT, Carbone M, Barresi G. Gastric infection by Helicobacter pylori and antral gastritis in hyperglycemic obese and in diabetic subjects. New Microbiol. 1996;19(2):149-54. PMID 8722311.
Takehara K, Tashima K, Takeuchi K. Alterations in duodenal bicarbonate secretion and mucosal susceptibility to acid in diabetic rats. Gastroenterology. 1997 Feb;112(2):418-28. doi: 10.1053/gast.1997.v112.pm9024295, PMID 9024295.
Begum AN, Terao J. Protective effect of quercetin against cigarette tar extract-induced impairment of erythrocyte deformability. J Nutr Biochem. 2002;13(5):265-72. doi: 10.1016/S0955-2863(01)00219-4, PMID 12015156.
Janisch KM, Williamson G, Needs P, Plumb GW. Properties of quercetin conjugates: modulation of LDL oxidation and binding to human serum albumin. Free Radic Res. 2004;38(8):877-84. doi: 10.1080/10715760410001728415, PMID 15493462.
Benherlal PS, Arumughan C. Chemical composition and in vitro antioxidant studies on Syzygium cumini fruit. J Sci Food Agric. 2007 Nov;87(14):2560-9. doi: 10.1002/jsfa.2957, PMID 20836162.
Gonzalez Segovia R, Quintanar JL, Salinas E, Ceballos Salazar R, Aviles Jimenez F, Torres Lopez J. Effect of the flavonoid quercetin on inflammation and lipid peroxidation induced by Helicobacter pylori in gastric mucosa of guinea pig. J Gastroenterol. 2008;43(6):441-7. doi: 10.1007/s00535-008-2184-7, PMID 18600388.
Brzozowski T, Konturek PC, Pajdo R, Kwiecien SN, Konturek S, Targosz A. Agonist of peroxisome proliferator-activated receptor γ (PPAR-γ): a new compound with potent gastroprotective and ulcer healing properties. Inflammopharmacology. 2005;13(1-3):317-30. doi: 10.1163/156856005774423908, PMID 16259750.
Sivri B. Trends in peptic ulcer pharmacotherapy. Fundam Clin Pharmacol. 2004 Feb;18(1):23-31. doi: 10.1111/j.1472-8206.2004.00203.x, PMID 14748750.
Boj Carceller D. Proton pump inhibitors: impact on glucose metabolism. Endocrine. 2013;43(1):22-32. doi: 10.1007/s12020-012-9755-3, PMID 22886351.
Rooman I, Lardon J, Bouwens L. Gastrin stimulates beta-cell neogenesis and increases islet mass from transdifferentiated but not from normal exocrine pancreas tissue. Diabetes. 2002 Mar;51(3):686-90. doi: 10.2337/diabetes.51.3.686, PMID 11872667.
Bodvarsdottir TB, Hove KD, Gotfredsen CF, Pridal L, Vaag A, Karlsen AE. Treatment with a proton pump inhibitor improves glycaemic control in Psammomys obesus a model of type 2 diabetes. Diabetologia. 2010 Oct;53(10):2220-3. doi: 10.1007/s00125-010-1825-6, PMID 20585936.
Kumar NP, Annamalai AR, Thakur RS. Antinociceptive property of Emblica officinalis Gaertn. (Amla) in high fat diet-fed/low dose streptozotocin induced diabetic neuropathy in rats. Indian J Exp Biol. 2009 Sep;47(9):737-42. PMID 19957886.
Basavarajappa GM, Nanjundan PK, Alabdulsalam A, Asif AH, Shekharappa HT, Anwer MK. Improved renoprotection in diabetes with combination therapy of Coccinia indica leaf extract and low-dose pioglitazone. Separations. 2020;7(4):58. doi: 10.3390/separations7040058.
Jonnalagadda A, Karthik KM, Prem Kumar N. Combined effect of Syzygium cumini seed kernel extract with oral hypoglycemics in diabetes-induced increase in susceptibility to ulcerogenic stimuli. J Diabetes Metab. 2013;4(1):2-6. doi: 10.4172/2155-6156.1000236.
Sahoo PK, Padhy KM, Pradhan D, Tripathy G, Bhoi R, Pattanayak S. Antidiabetic and antioxidant activity of ethanolic extract of Sapindus trifoliatus Linn. Int J Pharm Bio Sci. 2010;1(2):1-8. doi: 10.5555/20113372253.
Kennard MR, Nandi M, Chapple S, King AJ. The glucose tolerance test in mice: sex drugs and protocol. Diabetes Obes Metab. 2022;24(11):2241-52. doi: 10.1111/dom.14811, PMID 35815375.
Vogel HG, editor. Drug discovery and evaluation. Berlin, Heidelberg: Springer Berlin Heidelberg; 2008. doi: 10.1007/978-3-540-70995-4.
Majumdar B, Ray Chaudhuri SG, Ray A, Bandyopadhyay SK. Effect of ethanol extract of Piper betle linn leaf on healing of NSAID-induced experimental ulcer a novel role of free radical scavenging action. Indian J Exp Biol. 2003;41(4):311-5. PMID 15255639.
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the folin phenol reagent. J Biol Chem. 1951;193(1):265-75. doi: 10.1016/S0021-9258(19)52451-6, PMID 14907713.
Elstner EF, Heupel A. Inhibition of nitrite formation from hydroxylammoniumchloride: a simple assay for superoxide dismutase. Anal Biochem. 1976 Feb;70(2):616-20. doi: 10.1016/0003-2697(76)90488-7, PMID 817618.
Eva ML. Mechanism of pH-dependent hydrogen peroxide cytotoxicity in vitro. Arch Biochem Biophys. 1988;365:362-72. doi: 10.1016/0003-9861(88)90139-7.
Vogel WH, Scholkens BA, Sandow J. Drug discovery and evaluation: pharmacological assays. 2nd ed. Berlin: Springer-Verlag; 2002.
Brzozowski T, Konturek PC, Konturek SJ, Kwiecien S, Pajdo R, Brzozowska I. Involvement of endogenous cholecystokinin and somatostatin in gastroprotection induced by intraduodenal fat. J Clin Gastroenterol. 1998;27 Suppl 1:S125-37. doi: 10.1097/00004836-199800001-00020, PMID 9872509.
Ajeigbe KO, Olaleye SB, Oladejo EO, Olayanju AO. Effect of folic acid supplementation on oxidative gastric mucosa damage and acid secretory response in the rat. Indian J Pharmacol. 2011 Sep;43(5):578-81. doi: 10.4103/0253-7613.84976, PMID 22022004, PMCID PMC3195131.
Yokotani K, Murakami Y, Okada S, Wang M, Nakamura K. Histamine H(3) receptor-mediated inhibition of endogenous acetylcholine release from the isolated vascularly perfused rat stomach. Eur J Pharmacol. 2000 Mar 24;392(1-2):23-9. doi: 10.1016/S0014-2999(00)00085-6, PMID 10748268.
Sarosiek J, Rourk RM, Piascik R, Namiot Z, Hetzel DP, McCallum RW. The effect of esophageal mechanical and chemical stimuli on salivary mucin secretion in healthy individuals. Am J Med Sci. 1994 Jul;308(1):23-31. doi: 10.1097/00000441-199407000-00006, PMID 8010333.
Mæhre HK, Dalheim L, Edvinsen GK, Elvevoll EO, Jensen IJ. Protein determination method matters. Foods. 2018;7(1):5. doi: 10.3390/foods7010005, PMID 29301260.
Engelbrecht I, Horn S, Giesy JP, Pieters R. Determining superoxide dismutase content and catalase activity in mammalian cell lines. MethodsX. 2023;11:102395. doi: 10.1016/j.mex.2023.102395, PMID 37791011.
Mishra P, Singh U, Pandey CM, Mishra P, Pandey G. Application of student’s t-test analysis of variance and covariance. Ann Card Anaesth. 2019 Oct-Dec;22(4):407-11. doi: 10.4103/aca.ACA_94_19, PMID 31621677, PMCID PMC6813708.
Sakai M, Oimomi M, Kasuga M. Experimental studies on the role of fructose in the development of diabetic complications. Kobe J Med Sci. 2002 Dec;48(5-6):125-36. PMID 12594356.
Reed MJ, Meszaros K, Entes LJ, Claypool MD, Pinkett JG, Gadbois TM. A new rat model of type 2 diabetes: the fat-fed streptozotocin-treated rat. Metabolism. ScienceDirect. 2000;49(11):1390-4. doi: 10.1053/meta.2000.17721, PMID 11092499.
Ahmad M, Qureshi R, Arshad M. Traditional herbal remedies used for the treatment of diabetes from district attock (Pakistan). Pak J Bot. 2009;41(6):2777-82.
Shinde J, Taldone T, Barletta M, Kunaparaju N, Hu B, Kumar S. α-glucosidase inhibitory activity of Syzygium cumini (Linn.) Skeels seed kernel in vitro and in Goto-Kakizaki (GK) rats. Carbohydr Res. 2008;343(7):1278-81. doi: 10.1016/j.carres.2008.03.003, PMID 18374320.
Adewole SO, Caxton Martins EA, Ojewole JA. Protective effect of quercetin on the morphology of pancreatic β-cells of streptozotocin-treated diabetic rats. Afr J Tradit Complement Altern Med. 2006 Aug;4(1):64-74. doi: 10.4314/ajtcam.v4i1.31196, PMID 20162074, PMCID PMC2816429.
Youl E, Bardy G, Magous R, Cros G, Sejalon F, Virsolvy A. Quercetin potentiates insulin secretion and protects INS-1 pancreatic β-cells against oxidative damage via the ERK1/2 pathway. Br J Pharmacol. 2010 Oct;161(4):799-814. doi: 10.1111/j.1476-5381.2010.00910.x, PMID 20860660, PMCID PMC2992896.
Choi HN, Jeong SM, Huh GH, Kim JI. Quercetin ameliorates insulin sensitivity and liver steatosis partly by increasing adiponectin expression in ob/ob mice. Food Sci Biotechnol. 2015;24(1):273-9. doi: 10.1007/s10068-015-0036-9.
Gohlke A, Ingelmann CJ, Nurnberg G, Weitzel JM, Hammon HM, Gors S. Influence of 4-week intraduodenal supplementation of quercetin on performance, glucose metabolism and mRNA abundance of genes related to glucose metabolism and antioxidative status in dairy cows. J Dairy Sci. 2013;96(11):6986-7000. doi: 10.3168/jds.2013-6852, PMID 24054306.
Khaleel EF, Mostafa DG, Abdel Aleem GA. Gastroprotective effect of flavonoid quercetin and coenzyme Q10 in indomethacin-induced gastric ulcers in normal and diabetic rats. J Dent Med Sci. 2015 Dec;14(12):58-71. doi: 10.9790/0853-141245871.
Moreira L, Araujo I, Costa T, Correia Branco A, Faria A, Martel F. Quercetin and epigallocatechin gallate inhibit glucose uptake and metabolism by breast cancer cells by an estrogen receptor independent mechanism. Exp Cell Res. 2013;319(12):1784-95. doi: 10.1016/j.yexcr.2013.05.001, PMID 23664836.
Dhanya R, Arya AD, Nisha P, Jayamurthy P. Quercetin a lead compound against type 2 diabetes, ameliorates glucose uptake via AMPK pathway in skeletal muscle cell line. Front Pharmacol. 2017;8:336. doi: 10.3389/fphar.2017.00336, PMID 28642704.
Kim JH, Kang MJ, Choi HN, Jeong SM, Lee YM, Kim JI. Quercetin attenuates fasting and postprandial hyperglycemia in animal models of diabetes mellitus. Nutr Res Pract. 2011 Apr;5(2):107-11. doi: 10.4162/nrp.2011.5.2.107, PMID 21556223, PMCID PMC3085798.
Joshi MC, Dorababu M, Prabha T. Effects of Pterocarpus marsupium on NIDDM-induced rat gastric ulceration and mucosal offensive and defensive factors. Indian J Pharmacol. 2004 Oct;36(5):296-302.
Mahmoud SA, Elkhoely A, El Sayed EK, Ahmed AA. Enhanced upregulation of SIRT1 via pioglitazone and ligustrazine confers protection against ethanol-induced gastric ulcer in rats. Naunyn Schmiedebergs Arch Pharmacol. 2024;397(8):6177-95. doi: 10.1007/s00210-024-03026-6, PMID 38441571.
Brzozowski T, Konturek PC, Pajdo R, Kwiecien SN, Konturek S, Targosz A. Agonist of peroxisome proliferator-activated receptor gamma (PPAR-γ): a new compound with potent gastroprotective and ulcer healing properties. Inflammopharmacology. 2005;13(1-3):317-30. doi: 10.1163/156856005774423908, PMID 16259750.
Konturek PC, Brzozowski T, Kania J, Konturek SJ, Kwiecien S, Pajdo R. Pioglitazone a specific ligand of peroxisome proliferator activated receptor-gamma accelerates gastric ulcer healing in rat. Eur J Pharmacol. 2003;472(3):213-20. doi: 10.1016/S0014-2999(03)01932-0, PMID 12871756.
Dhanya R, Arya AD, Nisha P, Jayamurthy P. Quercetin a lead compound against type 2 diabetes ameliorates glucose uptake via AMPK pathway in skeletal muscle cell line. Front Pharmacol. 2017;8:336. doi: 10.3389/fphar.2017.00336, PMID 28642704.
Fryer LG, Parbu Patel A, Carling D. The anti-diabetic drugs rosiglitazone and metformin stimulate AMP-activated protein kinase through distinct signaling pathways. J Biol Chem. 2002;277(28):25226-32. doi: 10.1074/jbc.M202489200, PMID 11994296.
Dorababu M, Joshi MC, Bhawani G, Kumar MM, Chaturvedi A, Goel RK. Effect of aqueous extract of neem (Azadirachta indica) leaves on offensive and diffensive gastric mucosal factors in rats. Indian J Physiol Pharmacol. 2006;50(3):241-9. PMID 17193895.
Yoshizumi M, Tsuchiya K, Suzaki Y, Kirima K, Kyaw M, Moon JH. Quercetin glucuronide prevents VSMC hypertrophy by angiotensin II via the inhibition of JNK and AP-1 signaling pathway. Biochem Biophys Res Commun. 2002;293(5):1458-65. doi: 10.1016/S0006-291X(02)00407-2, PMID 12054679.
Begum AN, Terao J. Protective effect of quercetin against cigarette tar extract-induced impairment of erythrocyte deformability. J Nutr Biochem. 2002;13(5):265-72. doi: 10.1016/S0955-2863(01)00219-4, PMID 12015156.
Janisch KM, Williamson G, Needs P, Plumb GW. Properties of quercetin conjugates: modulation of LDL oxidation and binding to human serum albumin. Free Radic Res. 2004;38(8):877-84. doi: 10.1080/10715760410001728415, PMID 15493462.
Benherlal PS, Arumughan C. Chemical composition and in vitro antioxidant studies on Syzygium cumini fruit. J Sci Food Agric. 2007 Nov;87(14):2560-9. doi: 10.1002/jsfa.2957, PMID 20836162.
Gonzalez Segovia R, Quintanar JL, Salinas E, Ceballos Salazar R, Aviles Jimenez F, Torres Lopez J. Effect of the flavonoid quercetin on inflammation and lipid peroxidation induced by Helicobacter pylori in gastric mucosa of guinea pig. J Gastroenterol. 2008;43(6):441-7. doi: 10.1007/s00535-008-2184-7, PMID 18600388.