
Int J App Pharm, Vol 18, Issue 1, 2026, 130-140Reviewl Article
BASICS OF DEVELOPMENT OF NEW ANTIFUNGAL AGENTS IN OPHTHALMOLOGY: A REVIEW
ILYA KURGUZOV1*, IRINA LOGUNKOVA2, ELENA BAKHRUSHINA3, MADINA DURZHINSKAYA4, ANNA PLYUKHOVA5, ALEXANDRA KURGUZOVA6, MARIA NARBUT7
1Center for Master's Programs, I. M. Sechenov First Moscow State Medical University (Sechenov University), Moscow, Russia. 2,3A. P. Nelyubin Institute of Pharmacy, I. M. Sechenov First Moscow State Medical University (Sechenov University), Moscow, Russia. 4,5,6Department of Retinal and Optic Nerve Pathology, Federal State Budgetary Institution of Science "M. M. Krasnov Research Institute of Eye Diseases" (Krasnov Research Institute of Eye Diseases), Moscow, Russia. 7Department of Pathology of Optical Media of the Eye, Federal State Budgetary Institution of Science "M. M. Krasnov Research Institute of Eye Diseases" (Krasnov Research Institute of Eye Diseases), Moscow, Russia
*Corresponding author: Ilya Kurguzov; *Email: ilya.kurguzov@internet.ru
Received: 17 May 2025, Revised and Accepted: 30 Oct 2025
ABSTRACT
The aim of this work was to study the status of treatment of fungal eye infections. Recent data indicate a steady rise in diagnosed cases of fungal ocular diseases, particularly fungal keratitis-especially in developing countries. In some regions, the absence of locally available antifungal medications, despite increasing demand, contributes to this trend. Despite prolonged observation and efforts to combat fungal eye diseases, the global availability of antifungal drugs remains limited, especially for local use. These drugs are an integral part of comprehensive treatment, along with systemic drugs. The objective of this review work is to conduct a comparative analysis of molecules that have been evaluated in terms of antifungal activity, bioavailability, efficacy, and safety, considering the long duration of treatment and, consequently, increased risks of side effects.
For this purpose, a comprehensive review was conducted of materials published between 2015 and 2025 in international scientific databases, including PubMed, Scopus, Google Scholar, and ResearchGate. The search strategy was designed to capture relevant studies on antifungal ophthalmic therapies, dosage forms, and active pharmaceutical ingredients used in the treatment of fungal keratitis. Based on the reviewed literature, a SWOT analysis was performed, for the first time in this context, to systematically evaluate the strengths, weaknesses, opportunities, and threats associated with the principal dosage forms of ophthalmic antifungal drugs. This novel analytical approach enabled the identification of key directions for enhancing therapeutic efficacy, improving formulation design, and addressing clinical and regulatory gaps in the management of fungal ocular infections. The presented data can serve as a foundation for the development of improved drug formulations and delivery systems targeted directly to the affected ocular tissues-without preservatives, with controlled drug release-thereby enhancing treatment effectiveness, minimizing drug loss, and reducing patient risks associated with side effects and antifungal resistance. Furthermore, these advancements have the potential to significantly improve patient quality of life and increase the likelihood of full recovery, without irreversible visual impairment.
Keywords: Antifungal agents, Fungal keratitis, Treatment, Bioavailability, Side effects
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.55104 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
According to the World Health Organization (WHO), over 2.2 billion people worldwide suffer from various visual impairments caused by a variety of factors and diseases [1]. However, more than 90% of these problems can be prevented or successfully treated using modern medical technologies, including surgery, vision correction, and other innovative methods. Despite this, eye problems continue to significantly impact people's quality of life, especially in developing countries. Residents of low-income countries and rural areas constitute the largest group of those affected. This is attributable to the fact that people in such regions are less likely to seek qualified medical care, often ignoring symptoms or attempting to treat diseases on their own, which can lead to serious complications [1, 2].
Without proper treatment, vision problems can result in irreversible consequences, such as vision loss and total blindness. In high-income countries, the prevalence of blindness is approximately 1.94 cases per 1,000 people, whereas in low-income countries it is much higher-about 8.75 cases per 1000 people [1, 2]. These data highlight a significant disparity in access to medical care and the effectiveness of eye disease prevention across different regions of the world. Climatic and geographical factors also influence the frequency of eye diseases, leading to complete vision loss. In countries with tropical and subtropical climates (such as India, Thailand, Vietnam, and Bangladesh), high humidity, elevated temperatures, and abundant vegetation contribute to the spread of infectious eye conditions. Consequently, these countries experience a high incidence of bilateral blindness, caused both by infectious factors and traumatic injuries to the visual organs [3].
Blindness not only diminishes quality of life but also predisposes individuals to other serious health issues. People with vision loss are more likely to experience depression, cognitive impairment, and difficulties adapting to daily life. These problems are especially pronounced among older adults, where blindness can lead to social isolation, an increased risk of falls, and injuries. Additionally, such people often end up in nursing homes, where their psychoemotional well-being often deteriorates further [4-6].
One of the causes of blindness is corneal infections, collectively known as infectious keratitis, which can be caused by bacteria, fungi, viruses, or protozoa [7]. The etiology and prevalence of infectious keratitis causes vary according to the relative influence of climatic, socioeconomic, and predisposing factors. It is important to note that most published studies report on the incidence of non-viral infectious keratitis [8]. For instance, bacterial keratitis represents the most common type of infectious keratitis in most regions, including the UK (91–93%), North America (86–92%), South America (79–88%), Middle East (91.8%), and Australasia (93–100%). Several studies have demonstrated that fungi are the primary cause of infectious keratitis in Asia and Africa [9]. A population-based study conducted in several regions of China found that viral keratitis was more prevalent than bacterial and fungal keratitis. Compared to other causes of infectious keratitis, protozoal keratitis is less common, but in the last decade, an increasing number of protozoal keratitis cases have been reported, contact lens wearing the leading risk factor [8, 10]. However, this review specifically focuses on fungal keratitis. This infection can develop in various parts of the visual organ and depending on which part of the eye is affected, several types of fungal diseases are distinguished. The most common include fungal keratitis (affecting the cornea), fungal endophthalmitis (infection of internal ocular structures), fungal conjunctivitis (inflammation of the conjunctiva), and fungal blepharitis (involving the eyelids). If not diagnosed and treated early, these diseases can severely impair vision or even lead to its complete loss.
Therefore, prevention, early diagnosis, and effective treatment of eye diseases remain highly relevant issues. It is important to recognize that modern medicine possesses all necessary tools for preventing and treating most conditions associated with vision loss. However, the availability of such services and the level of public awareness of possible risks remain key factors for improving the health status of people on a global scale.
The purpose of this review is to identify major fungal infections affecting the eye and their treatment options, analyze the global market for antifungal ophthalmic drugs, and determine the optimal molecule and dosage form for the development of a new antifungal agent characterized by high efficacy, safety, and patient compliance.
MATERIALS AND METHODS
Materials for this review were collected from international scientific databases, including PubMed, Scopus, Google Scholar, and ResearchGate, covering publications from 2015 to 2025. The main dataset was obtained from medical publication databases. The material search was conducted using the keywords: antifungal agents and ophthalmology (1236 results), fungal keratitis and treatment (yielding approximately 2,400 results), antifungal drugs and ophthalmology (1277 results).
Inclusion criteria
The following parameters were used to determine study eligibility: Clinical trials, meta-analyses, and reviews focusing on antifungal agents specifically formulated for ophthalmic use. Studies investigating the epidemiology of ocular fungal infections. Research involving human or animal models. In vitro studies, only if they evaluated the efficacy of the drug in corneal/ocular tissue models.
Exclusion criteria
Studies were excluded if they met any of the following conditions: Focused on ocular diseases of non-fungal origin. Addressed only the differentiation between fungal and bacterial infections. Investigated non-ophthalmic delivery systems, including systemic antifungal therapies (e. g., intravenous or oral administration).
Considering the predefined inclusion and exclusion criteria, a total of 124 publications were analyzed during this review, of which 80 were ultimately cited as references.
Fungal infections of the eye
The two primary forms of fungal ocular infections are keratitis, which affects the corneal surface, and endophthalmitis, involving intraocular structures. Both conditions may initially mimic bacterial infections, complicating early diagnosis. Consequently, they pose a serious health threat and present significant clinical challenges due to their potential to cause severe visual impairment and the complexity of their diagnosis and treatment [11].
Fungal keratitis
Fungal keratitis is one of the most common fungal eye infections and constitutes a true ophthalmic emergency. The main causative agents are fungi from the genera Fusarium, Aspergillus, and Candida [1, 12]. These pathogens can penetrate ocular tissues through corneal damage, making the disease particularly dangerous. It typically manifests symptoms such as eye pain, photophobia, conjunctival hyperemia, tearing, and a foreign body sensation. If not diagnosed and treated promptly, fungal keratitis can rapidly progress to corneal ulceration, visual impairment, blindness, or even loss of the eye. Unlike bacterial keratitis, fungal keratitis often follows a more indolent course, leading to delayed presentation and diagnosis. As a result, patients frequently seek medical attention at an advanced stage of the disease, when significant corneal damage has already occurred [12-17].
According to the World Health Organization (WHO), keratitis is one of the leading causes of vision loss worldwide. The most comprehensive epidemiological review on fungal keratitis was published in 2021, based on literature spanning from 1946 to 2019. The authors estimated a minimum annual global incidence of approximately 1,051,787 cases, with the highest burden observed in Asia and Africa. When all culture-negative cases are presumed to be of fungal origin, this estimate rises to about 1,480,916 cases annually [1, 13, 18]. Recent studies from regions such as the United States, West and South Asia and Southeast Europe confirm that fungal keratitis remains prevalent and that its incidence is trending upward globally [19-21]. For example, based on study performed by Bongomin et al. (2024) on the epidemiology of fungal keratitis in Uganda, the country is estimated to have approximately 6,084 annual cases of fungal keratitis, with a rate of 13.3 cases per 100,000 people [22]. But in Ghana, fungal keratitis affects approximately 810 people out of 100,000 [23].
Fungal keratitis is more common in tropical climates, where high temperatures and humidity create favorable conditions for pathogenic fungi. In these regions, fungal keratitis accounts for 20–60% of all corneal lesions. Factors contributing to this high incidence include extensive involvement in agriculture, frequent exposure to dust and pollutants, and trauma related to outdoor work [24, 25].
One of the main risk factors for developing fungal keratitis is recent penetrating corneal trauma. This explains its higher prevalence among men and young adults, who are more likely to experience ocular injuries due to active lifestyles or occupational hazards [25, 26]. Additional risk factors include wearing soft Multifocal Contact Lenses (MCLs), which can harbor pathogens on their surfaces or promote biofilm formation-thus increasing infection risk [24]. Contact lens wear also reduces corneal regenerative capacity, further facilitating fungal proliferation [27].
Corneal transplantation can also predispose individuals to fungal infections. Studies indicate that prolonged storage of donor corneas increases contamination risk. The incidence of fungal keratitis following corneal transplantation is approximately 1.4 cases per 10,000 patients-accounting for about 63% of post-surgical infections in this context [26, 28]. Fungal keratitis can lead to serious complications, such as corneal opacity and complete vision loss. The prognosis worsens with delayed medical intervention; often, the disease is masked under other types of keratitis—leading to misdiagnosis or inappropriate treatment. Untimely or incorrect therapy significantly increases the risk of chronic corneal damage and irreversible blindness [24].
Therefore, timely diagnosis and appropriate treatment are crucial in managing fungal keratitis. This condition requires specialized attention, because its management differs from bacterial keratitis and often necessitates specific antifungal agents [29].
Fungal endophthalmitis
Fungal endophthalmitis is a severe infectious disease characterized by the invasion of fungi into the interior of the eye. It is one of the most serious ocular conditions, as its consequences can include complete loss of vision. Fungal endophthalmitis is most often caused by Candida albicans, although many other pathogenic fungi may also be responsible for the development of the infection. Depending on the route of infection, two types of endophthalmitis are distinguished: endogenous and exogenous [30].
Exogenous endophthalmitis
Exogenous endophthalmitis develops when an infection enters the eye from external sources. It can occur due to various factors, such as intraocular surgery, foreign bodies in the eye, or penetrating trauma. Fungi are responsible for exogenous endophthalmitis in 16-27% of cases, while the remaining cases are caused by bacterial or mixed infections, including 3.2% involving both bacteria and fungi [31, 32]. In these cases, the source of infection may be microorganisms from the normal eyelid flora or contaminated objects that caused trauma.
Previous trauma to the eye is the primary cause of exogenous endophthalmitis, accounting for 40-80% of all cases. The main fungal agents involved are species of Candida and filamentous fungi such as Fusarium solani and Aspergillus flavus. It is important to note that approximately 25% of cases of exogenous endophthalmitis develop after ocular surgical procedures. Additionally, fungal keratitis can lead to endophthalmitis, especially if fungal infections are not properly treated. In such cases, the risk of disease progression increases significantly. A major risk factor is the use of corticosteroids without concomitant antibiotics, as these drugs suppress immune defenses. Other risk factors include corneal perforation, immunosuppressive conditions, and dry eye syndrome [33].
Endogenous endophthalmitis
Endogenous endophthalmitis is less common, accounting for only 5-15% of cases. It occurs when infection spreads to the eye via the bloodstream from other infectious foci within the body. Patients with endogenous endophthalmitis often exhibit signs of fungemia (fungal presence in blood), indicating systemic fungal infection. Candida albicans is the most common causative agent of endogenous fungal endophthalmitis; fungi of this genus are believed to have a particular affinity (tropism) for ocular tissues. This form of endophthalmitis is more frequently observed in immunocompromised patients, such as those with human immunodeficiency virus (HIV) infection, kidney and liver disease, or cancer. Patients who use intravenous drugs, have diabetes mellitus, or are undergoing immunosuppressive therapy are also at higher risk. In some cases, multiple risk factors may be present simultaneously [25, 28, 34, 35]. Fungi account for 11.1–18% of endogenous endophthalmitis cases. The prognosis in endogenous fungal endophthalmitis tends to be very poor; delayed diagnosis and treatment often result in complete vision loss. The right eye is more commonly affected due to its more direct blood flow from the carotid artery; however, both eyes may be involved in about 25% of cases. Notably, infections caused by Aspergillus species tend to have a worse prognosis for visual recovery because they cause more aggressive forms of endophthalmitis that can lead to significant visual deterioration or even enucleation (removal of the eye) [36, 37]. The earlier treatment begins for endophthalmitis-particularly fungal-the better the chances for preserving vision. However, managing fungal endophthalmitis presents several challenges.
Treatment of fungal lesions of the eye. The global market of antifungal drugs for ophthalmology
The treatment of ocular fungal infections is a prolonged process that can last from several weeks to several months. A significant challenge in the treatment of these infections is the low bioavailability of topical antifungal agents, which necessitates increasing the frequency of dosing. This can lead to undesirable side effects, such as irritation and discomfort in the eye area. Despite these issues, the combined use of topical antifungal agents with systemic broad-spectrum drugs has demonstrated the best outcomes in treating ocular fungal diseases.
To identify the most effective treatment methods for fungal eye lesions, an analysis of the global market for antifungal drugs was conducted. Data from drug registries were utilized for this purpose. Information on the drugs is presented in table 1.
Table 1: Comparative analysis of efficacy and safety of commonly used ophthalmic antifungal agents
| Name of licensed product (LP)\ Regulatory approval | Manufacturer | Active ingredient and dosage form | Clinical efficacy/safety of active ingredient | References |
| Natacin (The United States)/FDA approval | Harrow IP | Natamycin (eye suspension) | Natamycin is effective against various fungal species, including Fusarium, Aspergillus, Alternaria, Candida, Cephalosporium, Colletotrichum, Curvularia, Lasodiplodia, Scedosporium, Trichophyton, and Penicillium. Natamycin is generally well tolerated; however, it is recognized as an epitheliotoxic agent when administered over prolonged periods. Reported adverse effects include conjunctival hyperemia, foreign body sensation, stinging and burning, and excessive lacrimation. In addition, the suspension particles may adhere to areas of epithelial defect, as well as accumulate in the fornices and along the lid margins, potentially complicating the clinical course. | [38, 39] |
| Swissflu (India)/local regulatory approval | Swissvision | Fluconazole (eye drops) | Fluconazole is effective in the management of Candida keratitis, particularly in cases with deep stromal involvement, due to its favorable intraocular penetration and low incidence of adverse effects. However, its antifungal spectrum is limited, with poor efficacy against most filamentous fungi, such as Fusarium and Aspergillus species. Overall, fluconazole is generally well tolerated, making it a valuable therapeutic option for Candida keratitis, but its use should be restricted to susceptible organisms. | [40, 41] |
| Vozole (Kenya)/local regulatory approval | Dawa Limited | Voriconazole (powder for preparation eye drops 1%) | Voriconazole demonstrates potent activity against a wide spectrum of fungi, including Candida albicans, Candida parapsilosis, Candida tropicalis, Aspergillus fumigatus, Aspergillus flavus, Fusarium solani, as well as less common pathogens such as Paecilomyces, Histoplasma, Scedosporium, Curvularia, and Acremonium species. His activity is similar to natamycin, although its efficacy against Mucorales is minimal. It should be noted that topical administration of voriconazole alone may result in sub-therapeutic drug levels; therefore, the inclusion of oral voriconazole in the treatment regimen is often recommended to achieve optimal therapeutic concentrations. Voriconazole eye drops are generally well tolerated, with minimal adverse effects such as mild ocular irritation, transient conjunctival hyperemia, and periocular dermatitis. |
[42-46] |
In the global pharmaceutical landscape, fluconazole ophthalmic medicines are represented by several products with expected comparable efficacy and safety profiles. Notable examples include Fluzamed, produced by World Medicine Pharmaceutical Company, which is approved and marketed in Turkey; Fluconazole Eye/Ear Drop 0.3%, produced by Lexicare Pharma, which is approved and marketed in India; Fluconazole Eye Drops/Ophthalmic Solution 0.5%, produced by Inopha, which is approved and marketed in China [47-49].
In addition to original formulations, several generic versions of natamycin are available on the pharmaceutical market, particularly in India. These include Zonat Eye Drops 5 ml, manufactured by Sunways Pvt Ltd; Nata-Aid Eye Drops 5 ml, manufactured by NRI Vision Care India Ltd [50]. These generics are expected to demonstrate comparable efficacy and safety profiles to the original product, and they contribute to improving access to topical antifungal therapy in regions with high prevalence of fungal keratitis.
There is significant heterogeneity in the availability of treatment for fungal keratitis across different regions worldwide. Populations in North America have access to original antifungal eye drops, and to innovative therapies are supported by major pharmaceutical companies and specialized eye care centers. Europe demonstrates steady growth in fungal keratitis treatment, driven by strong healthcare infrastructure and public health initiatives for early detection. Countries such as Germany, the UK, and Italy offer broad access to specialized ophthalmology services and invest in antifungal research. Increased awareness and academic outreach further improve patient outcomes. The Asia-Pacific region is the fastest-growing market for fungal keratitis treatments. Accessibility of citizens is supported by government health programs, improved diagnostics, and international research collaborations.
The most vulnerable are countries in Africa. Given the high incidence of the disease, not all antifungal medicines needed are readily available in Sub-Saharan African countries. In Ghana, for example, the antifungals that are easily accessible are 5-flucytosine and fluconazole, while amphotericin B is available only in Benin, Zambia, South Africa, and Ethiopia [23, 52]. Currently, ophthalmic antifungal medication is not available in Tanzania outside a few centers, which have to specifically import this medication [12, 53]. The situation is similar in Russia. To date, no topical antifungal agents have received marketing authorization from the regulatory authorities of the Russian Federation for the treatment of fungal keratitis.
The lack of necessary ophthalmic antifungal agents in many local pharmaceutical markets often forces patients to obtain these medications from other countries, significantly increasing the cost of treatment. In the absence of approved medicines, clinicians are frequently compelled to use topical antifungal agents off-label for the management of ocular infections. However, such practices are associated with considerable risks for patients, including challenges in managing adverse effects and determining appropriate dosages [26]. It is important to note that high concentrations of many antifungal agents can exert toxic effects on ocular tissues, potentially leading to necrosis, whereas low concentrations may fail to achieve the desired therapeutic effect due to limited ocular bioavailability. These factors present serious challenges for clinicians and underscore the need for targeted ophthalmic formulations that can effectively treat fungal infections of the eye while minimizing the risk of adverse effects [26].
Thus, the pronounced heterogeneity in the availability of antifungal agents for the treatment of fungal keratitis worldwide raises several important ethical considerations, including:
Equity and Justice: the lack of access to essential medicines for vulnerable populations constitutes a violation of the principle of equity in healthcare. All patients, irrespective of their geographic location or socioeconomic status, should have the right to effective and safe treatment for vision-threatening diseases.
Patient Safety: the necessity to use antifungal agents not approved for ophthalmic indications, often in inappropriate dosage forms and without sufficient safety data, exposes patients to the risks of toxicity, suboptimal dosing, and unpredictable adverse effects. Such practices may compromise patient safety and clinical outcomes.
Global Responsibility: Pharmaceutical companies, regulatory authorities, and the global health community bear a collective responsibility to address these disparities. This includes supporting the development and regulatory approval of ophthalmic antifungal agents, ensuring their availability, and promoting their inclusion in national essential medicines lists.
Therefore, there is an urgent need to develop novel, effective antifungal agents specifically designed for ophthalmic use. The drug development process should begin with the rational selection of active molecules with proven antifungal efficacy and favorable safety profiles.
Therefore, there is an urgent need to develop a comprehensive strategy to address this issue. Such a strategy should include expanding the geographical availability of antifungal agents already accessible to ophthalmologists and reducing the cost of these medications or rising federal health programs to ensure equitable access to essential treatment for economically disadvantaged populations. A critical component of this approach is the development of novel, effective antifungal agents specifically designed for topical ophthalmic use. The drug development process should commence with the rational selection of active molecules that demonstrate proven antifungal efficacy and favorable safety profiles.
Candidate active molecules in the development of antifungal drugs in ophthalmology
Amphotericin B
Its mechanism of action involves binding to ergosterol, a key component of fungal cell membranes. The molecule consists of two domains: hydrophobic and hydrophilic. The hydrophobic region binds to ergosterol, forming pores in the cell membrane, with the hydrophilic parts facing inward. This pore formation causes potassium leakage, disrupting cellular metabolism. Subsequently, magnesium ions leak out, and protons enter the cell, leading to acidification and, ultimately fungal cell death [54].
Amphotericin B exhibits broad-spectrum activity against fungi such as Candida spp., Aspergillus and Mucorales. However, resistance can develop in some strains, like Aspergillus terreus and Trichosporonasahii. Various administration routes have been described, including subconjunctival, intrastromal, intracameral, and intravitreal injections [37, 55]. Topical application is ineffective due to poor penetration through the blood-brain barrier and hematocellular barrier, limiting its use in ocular infections. In the 1980s, studies were conducted to evaluate the pharmacokinetics of topically applied amphotericin B in animal models. However, despite its high antifungal efficacy, the drug demonstrates poor penetration into ocular tissues, particularly through the intact cornea, which significantly limits its effectiveness in treating ocular infections. Subconjunctival administration has been associated with adverse events such as necrosis, sclera thinning, and scleritis [56]. Currently, amphotericin B is not approved for topical ophthalmic use, although it is occasionally employed off-label in clinical practice.
Natamycin
Natamycin is one of the primary drugs used in treating ocular fungal infections and is the only antifungal agent approved by the Food and Drug Administration (FDA) for topical use.
It is available as a 5% suspension due to its low water solubility [26, 57]. Natamycin is active against fungi from genera such as Fusarium, Aspergillus, Acremonium, and Candida. It exhibits fungicidal effects at high concentrations; at lower doses, it acts as a fungistatic agent. Like amphotericin B, natamycin’s large molecular size limits tissue penetration into deeper eye structures. For severe infections like endophthalmitis, the drug may be used in combination with other medications. Compared to amphotericin B and azoles, natamycin has a favorable toxicity profile and higher efficacy-particularly in keratitis caused by Candida fungi, where it improves clinical outcomes and reduces corneal perforation risk [58].
However, despite its advantages, natamycin also presents certain limitations. One of the main drawbacks is its poor retention on the corneal surface, primarily due to rapid tear turnover. As a result, less than 5% of the administered doses reach the target site, which negatively impacts both the duration of treatment and the required frequency of administration. Typical dosing involves instillation of one drop every 1–2 h during the first 3–4 days, followed by 6–8 applications per day for 14–21 d. This regimen demands a high level of adherence from the patient, and frequent dosing may lead to poor compliance or necessitate inpatient care [59].
Fluconazole
Fluconazole belongs to the azole class of antifungals and inhibits lanosterol-14-alpha-demethylase, which converts lanosterol to ergosterol, a key component of fungal cell membranes. This leads to increased cell membrane permeability and subsequent lysis of fungal cells. Fluconazole is effective against Candida and Cryptococcus, though some strains like Candida glabrata may exhibit resistance. This drug is recommended by the Infectious Diseases Society of America (IDSA) because of its lower toxicity compared to other antifungal drugs [37]. Because of its long elimination half-life, fluconazole can be administered once daily. Its molecules readily penetrate ocular tissues due to high water solubility and low molecular weight, allowing therapeutic concentrations to be achieved in intraocular structures. These properties make it suitable for topical application at a concentration of 0.3%. In addition, fluconazole is frequently used in combination with amphotericin B for the treatment of fungal keratitis, particularly when administered via subconjunctival injection [55, 60].
To increase therapeutic activity, research is being conducted to create new forms using nanotechnology approaches. For example, several authors have conducted studies using fluconazole encapsulated in liposomes. A study conducted on rabbits with Candida albicans infection showed that topical application of liposomes containing fluconazole at a concentration of 2 mg/ml significantly improved the healing process. In the group that received the liposomal form of fluconazole, complete corneal healing was achieved in 86.4% of cases within three weeks of therapy. At the same time, in the control group, which received a standard solution of fluconazole at the same concentration, only 50% of the animals showed complete healing [61].
Voriconazole
It is also an azole antibiotic but has a broader spectrum of action than fluconazole. Voriconazole is active against fungi of the genera Candida, Aspergillus, Fusarium, Scedosporium, Paecilomyces, Curvularia and others. Despite its efficacy, its use is associated with significant side effects that must be considered when prescribing both ophthalmic and systemic forms [60].
Flucytosine
The mechanism of action of flucytosine is based on its ability to undergo deamination in fungal cells with the help of specific enzymes, resulting in the formation of a toxic compound for the pathogen-5-Fluorouracil (5FU). 5-FU is subsequently converted into 5-fluorouridine triphosphate, which becomes incorporated into fungal RNA in place of uridylic acid, thereby disrupting RNA function and inhibiting protein synthesis. This is exerts a fungistatic effect, slowing down the growth and division of fungal cells [62]. Topically, flucytosine can be prepared at concentrations of 1–1.5%, often used alongside other antifungals like amphotericin B or azoles—especially in severe infections such as fungal endophthalmitis. Flucytosine is particularly effective in the treatment of diseases caused by pathogens such as Candida albicans and Cryptococcus. However, when flucytosine is used as monotherapy, there is a risk of resistance development, which limits its use without additional drugs [55]. The positive and negative characteristics of active pharmaceutical substances used in ophthalmology are summarized in table 2.
Table 2: SWOT analysis of active antifungal pharmaceutical substances [d2]
Substance/ References |
Strengths | Weaknesses | Opportunities | Threats |
| Amphotericin B/ [37, 54-56, 63] | Broad spectrum; good to moderate activity against filamentous fungi. | Poor penetration through the ocular barrier. Not commercially available. | Encapsulation in liposomal nanoparticles improves solubility and bioavailability, enhances clinical response, and reduces the toxic effect of the drug. | Side effects: cataract, transient iritis and corneal oedema. |
Natamycin/ [26, 57-59, 63] |
Commercially available, first choice for Fusarium; good activity against Aspergillus. |
Large concentrations are needed, frequent dosing, low corneal penetration, and less effective against Candida species. | Complexes with cyclodextrin increase drug solubility and raise the concentration of the active component. Development of novel delivery systems (polymeric nanoparticles, transferosome, micelles, ocular inserts (e. g., films), and others)-non-immunogenic, biocompatible, biodegradable, and low-toxicity formulations fig. 1. | There are strains that are immune to natamycin, making the therapy ineffective. |
Fluconazole/ [11, 37, 55, 60, 63-66] |
Lower toxicity; long elimination half-life; small molecular size; superior water solubility; commercially available; effective against yeast. | Forced prolonged therapy; frequent administration; it is ineffective against filamentous fungi such as Aspergillus and Fusarium. | Encapsulation in liposomes or nanoparticles significantly increases the effectiveness and the penetration of the drug into the cornea. | There is a global increase in fluconazole-resistant Candida auris, Candida glabrata, and Candida parapsilosis. About 7% of all Candida blood samples tested at Centers for Disease Control and Prevention (CDC) are resistant to fluconazole. An increasing trend of resistance is also observed among Cryptococcus isolates. |
| Voriconazole/ [59, 62, 63] | Broad spectrum; commercially available; intraocular involvement effective for deep keratitis. | Less effective than natamycin. | Studies have shown that encapsulating voriconazole in lipidic nanoparticles improves its solubility and permeability, provides sustained drug release, and potentially enhancing efficacy and patient compliance. | Severe side effects when applied topically. |
Flucytosine / [55, 63, 67] |
Efficacy against Candida albicans, Cryptococcus. Synergistic effect with topical amphotericin B to yeasts. | Monotherapy carries a risk of resistance development; narrow spectrum; low penetration into ocular tissues. | Use as concomitant therapy increases antifungal effect. | Side effects with topical application. |
A SWOT analysis was conducted on the active pharmaceutical substances mostly used in the production of topical formulations for the treatment of fungal keratitis. The results of the analysis confirmed both a general and a particularly critical issue associated with these active ingredients, while simultaneously highlighting a key direction for future development-namely, the growing resistance of fungal pathogens and the limited efficacy of currently available market therapies when used as monotherapy. The primary opportunity identified lies in enhancing bioadhesive properties and leveraging advanced technologies to improve ocular bioavailability.
Medication forms of ophthalmic drugs
Modern medicine offers a variety of ophthalmic dosage forms for the treatment of eye diseases, each with its own advantages and limitations:
Liquid dosage forms-represented by eye drops, as well as solutions, suspensions, and emulsions for ocular irrigation or administration into the conjunctival sac, intraocular space, or parabulbar region. This is the most common and simple way of drug delivery, but the forms have low bioavailability.
Soft dosage forms-such as ointments, gels, and creams for application to the eye or eyelids-allow the drug to remain longer in the conjunctival cavity, which increases bioavailability, but the route of administration is often considered less convenient for patients.
Solid dosage forms: ocular films, implants, and tablets used for preparing ophthalmic solutions often provide the most prolonged therapeutic effect among all dosage forms. However, their use typically requires specialized knowledge and training for proper administration, which may limit their accessibility in routine clinical practice [47].
Table 3 presents a more detailed analysis of positive and negative characteristics for the most encountered forms of drugs in the therapy of ophthalmic diseases. There are also presented strategies for improving drug bioavailability and outlining potential risks associated with administration.
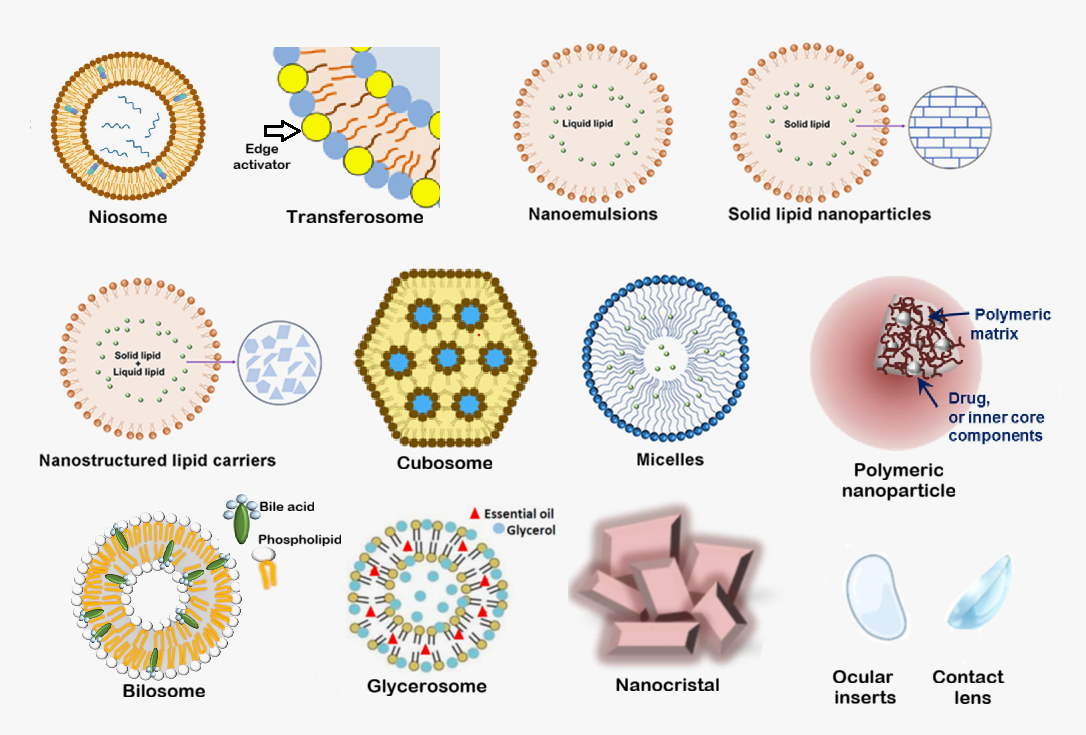
Fig. 1: Novel drug delivery systems have been used to enhance topical eye drug administration
Table 3: SWOT analysis of some dosage forms for ophthalmic use
| Dosage form | Strengths | Weaknesses | Opportunities | Threats | References |
| Eye drops | Easy to use; sterile administration; rapid absorption from the conjunctival cavity. | Low bioavailability; high dosing losses. | Use of microemulsions or liposomes to increase bioavailability; mucoadhesive polymers to increase corneal exposure fig. 2. | Possibility of microbial contamination of the contents. | [68-71] |
| Gels | Prolonged effect; long retention on the mucosa; cushioning and protective effect on the cornea. | Lack of dosing accuracy; temporary decrease in visual acuity after use. | Introduction of nanomolecules; the possibility of creating in situ systems based on gellan gum or carbopol; which will increase the release of the drug fig. 3. | Additional risks of contamination when administered. | [68-70] |
| Films | Exhibiting both aggregation stability and microbiological stability; can remain in the conjunctival cavity for a longer period, prolonged action. | Difficulty in insertion; special training required; sensation of foreign object in the eye during insertion. | Extemporaneous personalized manufacturing; the use of biodegradable polymers that can provide a prolonged effect and reduce the frequency of administration. | Potential release of drug substance into storage solution; drying of film during storage; traumatization during administration. | [72-75] |
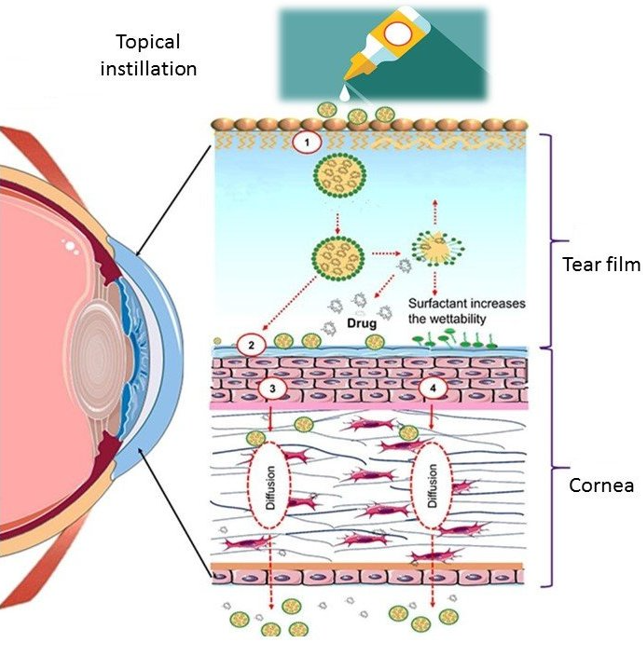
Fig. 2: Pathway of drug to a different part of the eye of topically administered microemulsions or liposomes. (1) Stabilization of the tear film through microemulsions or liposomes, which helps reduce nasolacrimal drainage and tear outflow, thereby prolonging drug residence time on the ocular surface, (2) Spreading of microemulsions or liposomes over the mucin layer, facilitating uniform distribution and interaction with the ocular surface, (3) Diffusion through the corneal epithelium and endothelium via the paracellular route, allowing drug molecules to pass between cells, (4) Transcellular diffusion, where microemulsions or liposomes penetrate directly through the corneal cells, enhancing drug delivery to deeper ocular tissues [76]
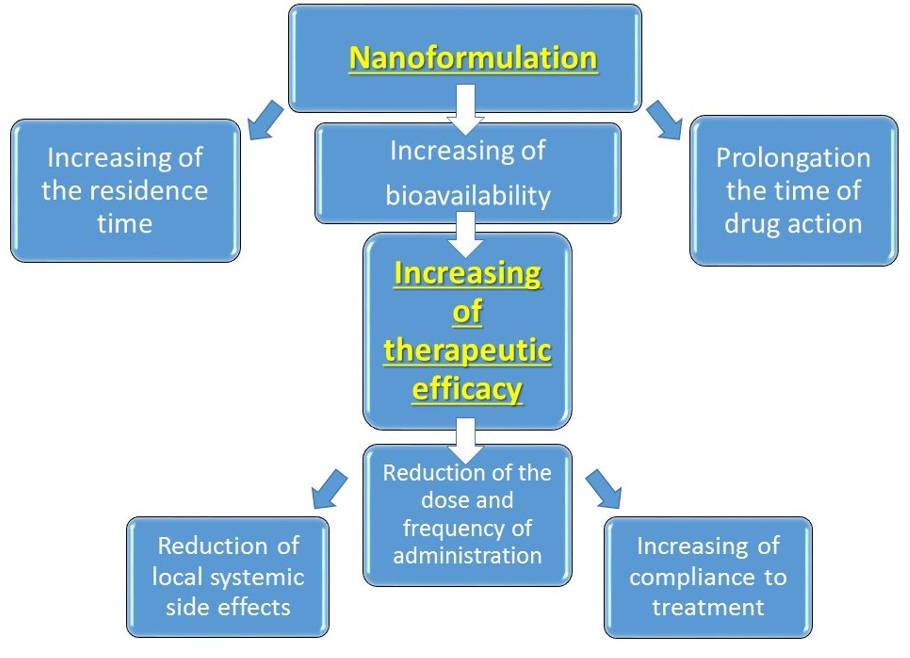
Fig. 3: Benefits of nanoformulated ophthalmic drug medicines
DISCUSSION
Analysis of pharmaceutical markets and scientific literature underscores the urgent need to develop effective antifungal ophthalmic drugs. The optimal dosage form for it to increase compliance may be eye drops with improved bioadhesion characteristics and the use of various technologies to increase bioavailability. In the context of increasing resistance to currently available topical eye drops used as monotherapy, combination therapies comprising complementary active ingredients are considered a particularly promising solution.
Modern types of packaging for ophthalmic drugs
Ocular films are usually packaged in blister or contour-mesh packaging, which ensures reliable protection and product integrity. In some cases, such packaging is equipped with a specialized applicator designed to minimize the risk of microbial contamination or mechanical damage to the eye during drug administration. This is especially important, as the mucous membrane of the eye is highly sensitive and vulnerable to external factors.
Gels for ophthalmic use are usually packaged in tubes with an elongated tip, which facilitate convenient administration into the conjunctival cavity. However, such packaging has significant drawbacks that can affect the quality and safety of treatment. Among the main problems are the lack of accurate dosing of the drug, which can lead to under-or overdosing, and the increased risk of microbial contamination. In clinical settings where gels are used, protection from external factors is essential to prevent bacterial ingress into the packaging and ensure product sterility.
Modern eyes drop packaging presents several challenges. Many currently available vials do not ensure accurate dosing, and the dropper tip may accidentally injure the eye or become a source of microbial contamination. Opened vials have a limited shelf life, and patients often fail to use the entire volume, resulting in wasted medication and unnecessary costs. A potential solution is the use of single-dose packaging, which in theory, eliminates the possibility of microbial contamination. However, in practice, the complete extraction of the drug from single-use containers is not always achievable. This forces manufacturers to refill vials, which in turn increases the cost of production. Additionally, the use of disposable packaging significantly increases the volume of production and the volume of materials, which also affects the final cost of the product.
In addition, most drops use preservatives, which can have adverse effects on ocular health, especially with long-term use. These excipients may irritate the corneal surface and contribute to the development or exacerbation of other eye conditions. Despite the proven toxicity of some preservatives, they remain widely used in commercial formulations, raising concerns among both clinicians and patients regarding their safety and tolerability.
All these challenges have driven the development of new, safer, and more effective ocular drug administration techniques and devices. One of these solutions is pump-assisted packaging, which controls the process of fluid reuptake and helps to avoid unnecessary loss of the drug. These devices are equipped with nozzles treated with silver ions that serve as an antibacterial agent. When the patient presses the vial, the drop passes through the antibacterial part of the nozzle, which helps to sanitize the medication and reduce the risk of infection. Such packaging solutions are manufactured by Aptarpharma (Switzerland) and Nemera (France), which testifies to the high quality and reliability of these technologies in the pharmaceutical industry.
Another innovative solution is the AcuStream® delivery system developed by Kedalion (USA). This device utilizes an ultrasonic stream of microdroplets to deliver medication to the eye. The droplets are reflected off a concave mirror integrated into the device, allowing for precise and rapid targeting of the ocular surface. The AcuStream® System can be either disposable or reusable, allowing the user to choose the most convenient option for treatment. However, the widespread adoption of such technologies in clinical practice may be hindered by several factors, including low patient compliance, high cost, and concerns regarding ease of use among both patients and healthcare professionals.
To further enhance dosing accuracy and minimize the risk of over-or underdose, advanced technologies have been introduced for liquid drug delivery. For example, the Novelia® system provides precise dosing based on the pressure required to deliver the correct dose and can be customized through a smartphone. This functionality allows both physicians and patients to monitor and manage the treatment process in real time, improving therapeutic outcomes and adherence.
In addition, the Novelia® dropper bottle is equipped with a secondary contamination protection mechanism, which includes a check valve. This design ensures that potentially contaminated droplets cannot flow back into the container, thereby significantly reducing the risk of microbial contamination. Furthermore, air intake occurs through a separate, filtered orifice, which also minimizes the likelihood of contamination and enhances the safety and convenience of use for the patient [69].
According to the data obtained, the use of the Novelia® system for storing voriconazole drops does not demonstrate significant advantages over traditional vials. Moreover, the implementation of this system may lead to an increase in production costs [73]. At the same time, switching to preservative-free forms of eye drops, while reducing the risks of toxic effects on the cornea, raises new questions. Currently, there are no large-scale studies confirming the long-term safety and stability of such preparations, especially under conditions of packaging integrity violations or improper storage. Thus, in a study [74], preservative-free eye drops for the treatment of dry eye syndrome were found to be contaminated with a drug-resistant strain of Pseudomonas aeruginosa. Even in single-use containers, the risk of contamination persists if the patient does not adhere to the application deadlines or opens the packaging incorrectly. Manufacturers mitigate this with sterile packaging technologies and barrier systems, but their effectiveness requires further study. Thus, the search for a balance between the safety of preservatives and the reliability of preservative-free solutions remains a relevant topic in modern pharmaceuticals.
Another disadvantage of innovative vials such as AcuStream® and Novelia® is their high cost, which is driven by a combination of technological, manufacturing, and regulatory factors. Although the initial cost of these devices is high, their use can lead to reduced overall treatment costs by increasing the bioavailability of drugs (reducing the loss of active ingredients), reducing the frequency of applications, preventing complications from improper dosing, and reducing the number of doctor visits due to improved treatment monitoring.
The problem of implementation is exacerbated by the conservatism of reimbursement systems: insurance companies are often unwilling to compensate for the higher initial cost, despite the potential long-term savings.
Thus, the high cost of these systems reflects not only the technological complexity, but also the investment in improving the safety, accuracy, and effectiveness of treatment. The key to widespread adoption will be to demonstrate not only the clinical efficacy, but also the cost-effectiveness of these systems.
Fungal ophthalmologic infections present significant diagnostic challenges in clinical practice, primarily due to their nonspecific symptomatology, which closely resembles bacterial lesions, as well as the difficulties associated with routine laboratory identification of the causative agent. These factors lead to critical delays in the initiation of etiotropic therapy, which are further exacerbated by the frequent late presentation of patients seeking medical care. Collectively, these issues contribute to disease progression and the development of severe complications. In addition, the storage of most ophthalmic medications requires low temperatures, which may further limit their availability, particularly in regions with hot climates and unstable energy supplies.
To optimize the care provided to this category of patients, a comprehensive approach is required, including the following future directions:
Strengthening international cooperation with the WHO and relevant nongovernmental organizations is essential for the implementation of centralized procurement programs aimed at reducing the cost of antifungal agents. Concurrently, the development and deployment of targeted educational initiatives for healthcare professionals are critical to enhancing the accuracy of differential diagnosis between fungal and bacterial ocular infections.
The development of thermostable dosage forms is a critical advancement that can substantially broaden the capacity for drug storage in environments lacking refrigeration infrastructure. This innovation is particularly relevant for improving therapeutic accessibility in tropical and subtropical regions, where the incidence of fungal keratitis is disproportionately high.
The inclusion of key antifungal agents, particularly commercially available medications —into national essential medicines lists represents a critical step toward ensuring equitable access to effective antifungal therapy for the general population.
Notably, there is a lack of pediatric-specific formulations of antifungal agents, which limits safe and effective treatment options for children with fungal keratitis. Although fungal keratitis is less frequently reported in pediatric populations, the current understanding of its clinical presentation, progression, and therapeutic response in children remains limited due to its low incidence. This knowledge gap may contribute to delayed, incorrect, or suboptimal treatment, potentially resulting in irreversible visual impairment. Childhood blindness is a lifelong condition that imposes substantial emotional, educational, and socioeconomic burdens on families and society. Therefore, prioritizing research into pediatric fungal keratitis and the development of targeted topical antifungal therapies is essential to ensure timely diagnosis and appropriate therapeutic interventions. Zhang et al. (2024) conducted a comprehensive retrospective analysis of pediatric fungal keratitis cases over a 25-year period (1996–2021), highlighting the clinical characteristics and treatment outcomes in children [77]. Among the antifungal agents evaluated, voriconazole demonstrated the highest drug sensitivity rate (up to 80%) and was identified as the most effective agent, with the most favorable minimum inhibitory concentration values. Importantly, voriconazole exhibited excellent corneal penetration, even through an intact corneal epithelium, making it particularly suitable for deep stromal infections. These findings underscore the potential of voriconazole as a preferred therapeutic option in pediatric fungal keratitis, especially in cases caused by Fusarium and Aspergillus species, which were the predominant pathogens in the study cohort.
Additionally, the potential of combination antifungal therapies remains insufficiently explored in clinical settings, despite growing evidence of their utility in managing resistant fungal infections. The severity of fungal keratitis is increasingly aggravated by the emergence of resistant fungal species, for which monotherapy may be inadequate. Combination therapy has demonstrated superior efficacy in such cases, offering broader coverage and reducing the risk of resistance development. In this context, one of the critical future directions is the development of novel ophthalmic formulations that combine antifungal agents, aiming to provide effective, noninvasive treatment options with minimal resistance potential. Jiang et al. (2020) demonstrated that the combination of voriconazole and chlorhexidine exhibits synergistic activity against Fusarium spp., suggesting enhanced therapeutic potential [78]. Similarly, Al-Hatmi et al. (2016) confirmed the synergistic efficacy of natamycin combined with voriconazole against most Fusarium strains, outperforming other combinations such as natamycin/itraconazole and natamycin/micafungin [79].
The hypothesis is currently being tested that topical natamycin 5% in combination with chlorhexidine 0.2% is superior to topical natamycin 5% alone in a two-group, single-blind, randomized controlled trial (ISRCTN, ISRCTN87195453, registered 08/27/2020) [80].
Despite these promising findings, significant gaps remain in clinical trial data. Most available studies are limited to in vitro analyses or small-scale observational reports, with few randomized controlled trials evaluating the long-term safety, pharmacokinetics, and comparative efficacy of combination therapies in diverse patient populations. There is also a lack of standardized protocols for evaluating antifungal combinations in ophthalmology. Addressing these gaps through well-designed clinical trials is essential to validate the therapeutic potential of combination regimens and guide evidence-based practice in the management of fungal keratitis.
CONCLUSION
Fungal eye infections remain a serious challenge due to diagnostic complexity, limited ophthalmic antifungal options, and rising resistance. Developing new broad-spectrum agents, improving existing formulations, and advancing drug delivery systems—such as eye drops, gels, and ocular films—are essential to enhance treatment efficacy and patient safety. While these approaches demonstrate theoretical potential, their clinical applicability must be validated through rigorous in vitro and in vivo studies.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
Ilya Kurguzov and Irina Logunkova: Conceptualization, Writing-Original draft preparation. Elena Bakhrushina: Conceptualization, Methodology, Writing-Reviewing and Editing. Madina Durzhinskaya, Anna Plyukhova, Alexandra Kurguzova and Maria Narbut: Writing-Reviewing and Editing.
CONFLICTS OF INTEREST
All authors have none to declare
REFERENCES
Burton MJ, Ramke J, Marques AP, Bourne RR, Congdon N, Jones I. The lancet global health commission on global eye health: vision beyond 2020. Lancet Glob Health. 2021 Apr;9(4):e489-551. doi: 10.1016/S2214-109X(20)30488-5, PMID 33607016, PMCID PMC7966694.
GBD 2019 Blindness and Vision Impairment Collaborators; Vision Loss Expert Group of the Global Burden of Disease Study. Causes of blindness and vision impairment in 2020 and trends over 30 y and prevalence of avoidable blindness in relation to vision 2020: the right to sight: an analysis for the global burden of disease study. Lancet Glob Health. 2021 Dec 1;9(2):e144-60. doi: 10.1016/S2214-109X(20)30489-7, PMCID PMC7820391.
Whitcher JP, Srinivasan M. Corneal ulceration in the developing world a silent epidemic. Br J Ophthalmol. 1997 Aug;81(8):622-3. doi: 10.1136/bjo.81.8.622, PMID 9349145, PMCID PMC1722289.
Taipale J, Mikhailova A, Ojamo M, Nattinen J, Vaatainen S, Gissler M. Low vision status and declining vision decrease health related quality of life: results from a nationwide 11 y follow-up study. Qual Life Res. 2019 Dec;28(12):3225-36. doi: 10.1007/s11136-019-02260-3, PMID 31401749, PMCID PMC6863947.
Heesterbeek TJ, Van Der Aa HP, Van Rens GH, Twisk JW, Van Nispen RM. The incidence and predictors of depressive and anxiety symptoms in older adults with vision impairment: a longitudinal prospective cohort study. Ophthalmic Physiol Opt. 2017 Jul;37(4):385-98. doi: 10.1111/opo.12388, PMID 28516509.
Whitson HE, Cousins SW, Burchett BM, Hybels CF, Pieper CF, Cohen HJ. The combined effect of visual impairment and cognitive impairment on disability in older people. J Am Geriatr Soc. 2007 Jun;55(6):885-91. doi: 10.1111/j.1532-5415.2007.01093.x, PMID 17537089.
Yan C, Zhao H, Pan Q, Xie X, Chen K, Zhang L. Novel treatments for bacterial keratitis: a review. Int J Pharm. 2025 Aug 20;681:125793. doi: 10.1016/j.ijpharm.2025.125793, PMID 40446871.
Stapleton F. The epidemiology of infectious keratitis. Ocul Surf. 2023 Apr;28:351-63. doi: 10.1016/j.jtos.2021.08.007, PMID 34419639.
Ting DS, Ho CS, Deshmukh R, Said DG, Dua HS. Infectious keratitis: an update on epidemiology causative microorganisms risk factors and antimicrobial resistance. Eye (Lond). 2021 Apr;35(4):1084-101. doi: 10.1038/s41433-020-01339-3.
Ilyas M, Stapleton F, Willcox MD, Henriquez F, Peguda HK, Rayamajhee B. Epidemiology of and genetic factors associated with Acanthamoeba keratitis. Pathogens. 2024 Feb 4;13(2):142. doi: 10.3390/pathogens13020142, PMID 38392880, PMCID PMC10892102.
Centers for Disease Control and Prevention. Fungal eye infections: about fungal eye infections. Atlanta (GA): CDC; 2024 Apr 24. Available from: https://www.cdc.gov/fungal-eye-infections/about/index.html. [Last accessed on 23 Jul 2025].
Burton MJ, Pithuwa J, Okello E, Afwamba I, Onyango JJ, Oates F. Microbial keratitis in East Africa: why are the outcomes so poor? Ophthal Epidemiol. 2011 Aug;18(4):158-63. doi: 10.3109/09286586.2011.595041, PMID 21780874, PMCID PMC3670402.
Brown L, Kamwiziku G, Oladele RO, Burton MJ, Prajna NV, Leitman TM. The case for fungal keratitis to be accepted as a neglected tropical disease. J Fungi (Basel). 2022 Oct 5;8(10):1047. doi: 10.3390/jof8101047, PMID 36294612, PMCID PMC9605065.
Hoffman JJ, Burton MJ, Leck A. Mycotic keratitis a global threat from the filamentous fungi. J Fungi (Basel). 2021 Apr 3;7(4):273. doi: 10.3390/jof7040273, PMID 33916767, PMCID PMC8066744.
Ali Shah SI, Shah SA, Rai P, Katpar NA, Abbasi SA, Soomro AA. Visual outcome in patients of keratomycosis at a Tertiary Care Centre in Larkana, Pakistan. J Pak Med Assoc. 2017 Jul;67(7):1035-8. PMID 28770882.
Roth M, Holtmann C, Daas L, Kakkassery V, Kurzai O, Geerling G. Results from the German fungal keratitis registry: significant differences between cases with and without a history of contact lens use. Cornea. 2021 Nov 1;40(11):1453-61. doi: 10.1097/ICO.0000000000002705, PMID 34633358.
Tananuvat N, Upaphong P, Tangmonkongvoragul C, Niparugs M, Chaidaroon W, Pongpom M. Fungal keratitis at a tertiary eye care in Northern Thailand: etiology and prognostic factors for treatment outcomes. J Infect. 2021 Jul;83(1):112-8. doi: 10.1016/j.jinf.2021.05.016, PMID 34038772.
Brown L, Leck AK, Gichangi M, Burton MJ, Denning DW. The global incidence and diagnosis of fungal keratitis. Lancet Infect Dis. 2021 Mar;21(3):e49-57. doi: 10.1016/S1473-3099(20)30448-5, PMID 33645500.
Yu B, Wang Q, Zhang L, Lin J, Feng Z, Wang Z. Ebselen improves fungal keratitis through exerting anti-inflammation anti-oxidative stress and antifungal effects. Redox Biol. 2024 Jul;73:103206. doi: 10.1016/j.redox.2024.103206, PMID 38796864, PMCID PMC11152752.
Benedict K, Gold JA, Smith DJ. Prevalence and features of fungal keratitis among US patients with commercial health insurance. JAMA Ophthalmol. 2024 Apr 1;142(4):386-8. doi: 10.1001/jamaophthalmol.2023.6825, PMID 38358752, PMCID PMC10870225.
Bala RK, Dange KJ, Patil GA. Practise and use of over-the-counter topical and traditional eye medicines and their effects in fungal keratitis cases in Tertiary Teaching Hospital in Northern India. Asian J Pharm Clin Res. 2024 Mar;17(3):103-6. doi: 10.22159/ajpcr.2024.v17i3.49534.
Bongomin F, Kwizera R, Namusobya M, Van Rhijn N, Andia Biraro I, Kirenga BJ. Re-estimation of the burden of serious fungal diseases in Uganda. Ther Adv Infect Dis. 2024 Feb 6;11:20499361241228345. doi: 10.1177/20499361241228345, PMID 38328511, PMCID PMC10848809.
Boakye Yiadom E, Odoom A, Osman AH, Ntim OK, Kotey FC, Ocansey BK. Fungal infections treatment and antifungal resistance: the sub-Saharan African context. Ther Adv Infect Dis. 2024 Nov 13;11:20499361241297525. doi: 10.1177/20499361241297525, PMID 39544852, PMCID PMC11562003.
Bongomin F, Gago S, Oladele RO, Denning DW. Global and multi-national prevalence of fungal diseases estimate precision. J Fungi (Basel). 2017 Oct 18;3(4):57. doi: 10.3390/jof3040057, PMID 29371573, PMCID PMC5753159.
Gopinathan U, Garg P, Fernandes M, Sharma S, Athmanathan S, Rao GN. The epidemiological features and laboratory results of fungal keratitis: a 10 y review at a referral eye care center in South India. Cornea. 2002 Aug;21(6):555-9. doi: 10.1097/00003226-200208000-00004, PMID 12131029.
Donovan C, Arenas E, Ayyala RS, Margo CE, Espana EM. Fungal keratitis: mechanisms of infection and management strategies. Surv Ophthalmol. 2022 May-Jun;67(3):758-69. doi: 10.1016/j.survophthal.2021.08.002, PMID 34425126, PMCID PMC9206537.
Maier P, Betancor PK, Reinhard T. Contact lens associated keratitis an often underestimated risk. Dtsch Arztebl Int. 2022 Oct 7;119(40):669-74. doi: 10.3238/arztebl.m2022.0281, PMID 35912449, PMCID PMC9830382.
Aldave AJ, DeMatteo J, Glasser DB, Tu EY, Iliakis B, Nordlund ML. Report of the eye bank association of America medical advisory board subcommittee on fungal infection after corneal transplantation. Cornea. 2013 Feb;32(2):149-54. doi: 10.1097/ICO.0b013e31825e83bf, PMID 23051906.
Deka DS, Khanikar D. A study of the prescribing pattern and outcome of antimicrobials in infectious eye diseases in a Tertiary Care Hospital. Asian J Pharm Clin Res. 2024 Aug;17(8):67-70. doi: 10.22159/ajpcr.2024v17i8.51416.
Haseeb AA, Elhusseiny AM, Siddiqui MZ, Ahmad KT, Sallam AB. Fungal endophthalmitis: a comprehensive review. J Fungi (Basel). 2021 Nov 22;7(11):996. doi: 10.3390/jof7110996, PMID 34829283, PMCID PMC8623405.
Long C, Liu B, Xu C, Jing Y, Yuan Z, Lin X. Causative organisms of post-traumatic endophthalmitis: a 20 y retrospective study. BMC Ophthalmol. 2014 Mar 25;14:34. doi: 10.1186/1471-2415-14-34, PMID 24661397, PMCID PMC3987925.
Sadiq MA, Hassan M, Agarwal A, Sarwar S, Toufeeq S, Soliman MK. Endogenous endophthalmitis: diagnosis management and prognosis. J Ophthal Inflamm Infect. 2015 Dec;5(1):32. doi: 10.1186/s12348-015-0063-y, PMID 26525563, PMCID PMC4630262.
Henry CR, Flynn HW JR, Miller D, Forster RK, Alfonso EC. Infectious keratitis progressing to endophthalmitis: a 15 y study of microbiology associated factors and clinical outcomes. Ophthalmology. 2012 Dec;119(12):2443-9. doi: 10.1016/j.ophtha.2012.06.030, PMID 22858123, PMCID PMC3490005.
Reginatto P, Agostinetto GJ, Fuentefria RD, Marinho DR, Pizzol MD, Fuentefria AM. Eye fungal infections: a mini review. Arch Microbiol. 2023 May 15;205(6):236. doi: 10.1007/s00203-023-03536-6, PMID 37183227, PMCID PMC10183313.
Klotz SA, Penn CC, Negvesky GJ, Butrus SI. Fungal and parasitic infections of the eye. Clin Microbiol Rev. 2000 Oct;13(4):662-85. doi: 10.1128/CMR.13.4.662, PMID 11023963, PMCID PMC88956.
Sridhar J, Flynn HW JR, Kuriyan AE, Miller D, Albini T. Endogenous fungal endophthalmitis: risk factors clinical features and treatment outcomes in mold and yeast infections. J Ophthal Inflamm Infect. 2013 Sep 20;3(1):60. doi: 10.1186/1869-5760-3-60, PMID 24053550, PMCID PMC3848556.
LY V, Sallam A. Fungal endophthalmitis. In: Treasure Island, FL: StatPearls Publishing; 2023 Jun 26. Available from: https://www.natacyn.com/ [Last accessed on 23 Jul 2025].
Natacyn. Nashville: Harrow IP, LLC; 2025.
Sharma N, Bagga B, Singhal D, Nagpal R, Kate A, Saluja G. Fungal keratitis: a review of clinical presentations treatment strategies and outcomes. Ocul Surf. 2022 Apr;24:22-30. doi: 10.1016/j.jtos.2021.12.001, PMID 34915188.
Swissvision. Fluconazole eye drops. Panchkula: Swissvision; 2025. Available from: https://www.swissvision.in/product/fluconazole-ee-drops. [Last accessed on 23 Jul 2025].
Al Badriyeh D, Neoh CF, Stewart K, Kong DC. Clinical utility of voriconazole eye drops in ophthalmic fungal keratitis. Clin Ophthalmol. 2010 May 6;4:391-405. doi: 10.2147/opth.s6374, PMID 20463910, PMCID PMC2866570.
Dawa Life Sciences. Vozole eye drops. Nairobi: Dawa Limited; 2025. Available from: https://dawalifesciences.com/product/vozole-eye-drops. [Last accessed on 23 Jul 2025].
Malhotra S, Khare A, Grover K, Singh I, Pawar P. Design and evaluation of voriconazole eye drops for the treatment of fungal keratitis. J Pharm (Cairo). 2014;2014:490595. doi: 10.1155/2014/490595, PMID 26556196, PMCID PMC4590801.
Jain K, Garhwal J, Swami T. Clinical profile and evaluation of topical voriconazole for management of fungal corneal ulcer in North Western Rajasthan. Delhi J Ophthalmol. 2018;29(1):35-8. doi: 10.7869/djo.374.
Sharma N, Bagga B, Singhal D, Nagpal R, Kate A, Saluja G. Fungal keratitis: a review of clinical presentations treatment strategies and outcomes. Ocul Surf. 2022 Apr;24:22-30. doi: 10.1016/j.jtos.2021.12.001, PMID 34915188.
Sahay P, Asif MI, Maharana PK, Titiyal JS. Periocular contact dermatitis with use of topical voriconazole 1% in mycotic keratitis. BMJ Case Rep. 2018 Sep 15;2018:bcr2018226498. doi: 10.1136/bcr-2018-226498, PMID 30219785, PMCID PMC6144350.
World Medicine Pharmaceutical Company. Fluzamed 0.3% Eye Drop. Istanbul: World Medicine Ilac San. Ve Tic A. S.; 2025. Available from: https://www.worldmedicine.com/translator/en/products/medicines?searchField=name&search=fluzamed. [Last accessed on 23 Jul 2025].
Lexicare Pharma Pvt. Ltd. Fluconazole eye/ear drop. Ankleshwar: Lexicare Pharma Pvt. Ltd; 2025. Available from: https://www.lexicarepharma.com/fluconazole-eye-ear-drop-5422867.html. [Last accessed on 02 Sep 2025].
International I. Fluconazole eye drops/ophthalmic solution 0.5%. In: Hangzhou: Inopha International Co, Limited; 2025. Available from: https://www.inopha.net/fluconazole-eye-drops-ophthalmic-solution-0-5. [Last accessed on 02 Sep 2025].
Sunways (India) Private Limited. ZONAT natamycin 5% eye drops. Mumbai: Sunways (India) Private Limited; 2025. Available from: https://zonatophthalmicsuspension|leadingeyecarespecialistsinindia|sunwaysindiapvtlt. [Last accessed on 02 Sep 2025].
Care NRIV. Nata-aid eye drop. New Delhi: NRI Vision Care India. LTD; 2025. Available from: https://nriglobalhealthcare/detail.com.jsp?cc=105andsrsltid=afmboortfi1xf8mkmueipliikbkebkyxfh1anghwhao0eanl8zhiup9c.
Makangara Cigolo JC, Oladele RO, Kennedy SB, Bignoumba M, Hassane HM, Bucumi V. Diagnostic capacity for fungal keratitis in Africa survey in 50 African countries. Ocul Surf. 2023 Oct;30:139-41. doi: 10.1016/j.jtos.2023.08.010, PMID 37660991.
Khor WB, Prajna VN, Garg P, Mehta JS, Xie L, Liu Z. The Asia cornea society infectious keratitis study: a prospective multicenter study of infectious keratitis in Asia. Am J Ophthalmol. 2018 Nov;195:161-70. doi: 10.1016/j.ajo.2018.07.040, PMID 30098351.
Hamill RJ. Amphotericin B formulations: a comparative review of efficacy and toxicity. Drugs. 2013 Jun;73(9):919-34. doi: 10.1007/s40265-013-0069-4, PMID 23729001.
Manzouri B, Vafidis GC, Wyse RK. Pharmacotherapy of fungal eye infections. Expert Opin Pharmacother. 2001 Nov;2(11):1849-57. doi: 10.1517/14656566.2.11.1849, PMID 11825321.
O Day DM, Head WS, Robinson RD, Clanton JA. Corneal penetration of topical amphotericin B and natamycin. Curr Eye Res. 1986 Nov;5(11):877-82. doi: 10.3109/02713688609029240, PMID 3490954.
Betbeze CM, Wu CC, Krohne SG, Stiles J. In vitro fungistatic and fungicidal activities of silver sulfadiazine and natamycin on pathogenic fungi isolated from horses with keratomycosis. Am J Vet Res. 2006 Oct;67(10):1788-93. doi: 10.2460/ajvr.67.10.1788, PMID 17014335.
Mascarenhas M, Chaudhari P, Lewis SA. Natamycin ocular delivery: challenges and advancements in ocular therapeutics. Adv Ther. 2023 Jun;40(8):3332-59. doi: 10.1007/s12325-023-02541-x, PMID 37289410.
Prajna NV, Krishnan T, Mascarenhas J, Rajaraman R, Prajna L, Srinivasan M. The mycotic ulcer treatment trial: a randomized trial comparing natamycin vs voriconazole. JAMA Ophthalmol. 2013 Apr;131(4):422-9. doi: 10.1001/jamaophthalmol.2013.1497, PMID 23710492, PMCID PMC3769211.
Alakkad A, Stapleton P, Schlosser C, Murdan S, Odunze U, Schatzlein A. Amphotericin B polymer nanoparticles show efficacy against candida species biofilms. Pathogens. 2022 Jan 7;11(1):73. doi: 10.3390/pathogens11010073, PMID 35056021, PMCID PMC8781556.
Bongomin F, Oladele RO, Gago S, Moore CB, Richardson MD. A systematic review of fluconazole resistance in clinical isolates of cryptococcus species. Mycoses. 2018 May;61(5):290-7. doi: 10.1111/myc.12747, PMID 29377368.
Schulz J, Kluwe F, Mikus G, Michelet R, Kloft C. Novel insights into the complex pharmacokinetics of voriconazole: a review of its metabolism. Drug Metab Rev. 2019 Aug;51(3):247-65. doi: 10.1080/03602532.2019.1632888, PMID 31215810.
Suman S, Kumar A, Saxena I, Kumar M. Fungal keratitis: recent advances in diagnosis and treatment. In: Rodriguez Garcia A, C Hernandez Camarena J, editors. Infectious eye diseases recent advances in diagnosis and treatment. Intech Open; 2021. doi: 10.5772/intechopen.98411.
Habib FS, Fouad EA, Abdel Rhaman MS, Fathalla D. Liposomes as an ocular delivery system of fluconazole: in vitro studies. Acta Ophthalmol. 2010 Dec;88(8):901-4. doi: 10.1111/j.1755-3768.2009.01584.x, PMID 19681761.
Aldosari BN, Ibrahim MA, Alqahtani Y, Abou El Ela AE. Formulation and evaluation of fluconazole nanosuspensions: in vitro characterization and transcorneal permeability studies. Saudi Pharm J. 2024 Jul;32(7):102104. doi: 10.1016/j.jsps.2024.102104, PMID 38841107, PMCID PMC11152688.
Daneshnia F, De Almeida Junior JN, Ilkit M, Lombardi L, Perry AM, Gao M. Worldwide emergence of fluconazole resistant Candida parapsilosis: current framework and future research roadmap. Lancet Microbe. 2023 Jun;4(6):e470-80. doi: 10.1016/S2666-5247(23)00067-8.
Sigera LS, Denning DW. Flucytosine and its clinical usage. Ther Adv Infect Dis. 2023 Apr 5;10:20499361231161387. doi: 10.1177/20499361231161387, PMID 37051439, PMCID PMC10084540.
Bakhrushina EO, Anurova MN, Demina NB, Lapik IV, Turaeva AR, Krasnuk II. Ophthalmic drug delivery systems. Razrabotka i Registracia Lekarstvennyh Sredstv. 2021;10(1):57-66. doi: 10.33380/2305-2066-2021-10-1-57-66.
Ivanov IS, Bakhrushina EO, Turaeva AR, Shatalov DO, Aydakova AV, Akhmedova DA. Approaches to the search of the optimum packaging of eye drops. Int J App Pharm. 2022;14(5):1-7. doi: 10.22159/ijap.2022v14i5.45402.
Wu Y, Liu Y, Li X, Kebebe D, Zhang B, Ren J. Research progress of in situ gelling ophthalmic drug delivery system. Asian J Pharm Sci. 2019 Jan;14(1):1-15. doi: 10.1016/j.ajps.2018.04.008, PMID 32104434, PMCID PMC7032175.
Kolawole OM, Cook MT. In situ gelling drug delivery systems for topical drug delivery. Eur J Pharm Biopharm. 2023 Mar;184:36-49. doi: 10.1016/j.ejpb.2023.01.007, PMID 36642283.
Choursiya K, Kandalkar S, Shaikh S, Gangurde P, Jadhav K, Nikam S. Advancements in ocular drug delivery system: a comprehensive review. Int J Pharm Sci. 2024 May 16;2(5):758-75. doi: 10.5281/zenodo.11204180.
Roche M, Lannoy D, Bourdon F, Danel C, Labalette P, Berneron C. Stability of frozen 1% voriconazole eye drops in both glass and innovative containers. Eur J Pharm Sci. 2020 Jan 1;141:105102. doi: 10.1016/j.ejps.2019.105102, PMID 31655210.
Mughal S, Sakina SK. Artificial tears: promising treatment or silent threat to public health. Health Sci Rep. 2023 Aug 24;6(8):e1508. doi: 10.1002/hsr2.1508, PMID 37636283, PMCID PMC10448608.
Lopez Cano JJ, Gonzalez Cela Casamayor MA, Andres Guerrero V, Herrero Vanrell R, Molina Martinez IT. Liposomes as vehicles for topical ophthalmic drug delivery and ocular surface protection. Expert Opin Drug Deliv. 2021 Jul; 18(7):819-47. doi: 10.1080/17425247.2021.1872542.
Singh M, Bharadwaj S, Lee KE, Kang SG. Therapeutic nanoemulsions in ophthalmic drug administration: concept in formulations and characterization techniques for ocular drug delivery. J Control Release. 2020 Oct 13;328:895-916. doi: 10.1016/j.jconrel.2020.10.025, PMID 33069743.
Zhang J, Chen H, Jhanji V, Zhang B, Xie L, Dong Y. Clinical characteristics and treatment outcomes of pediatric fungal keratitis. Infect Drug Resist. 2025 Mar 4;18:1271-8. doi: 10.2147/IDR.S507178, PMID 40059941, PMCID PMC11890013.
Jiang T, Tang J, Wu Z, Sun Y, Tan J, Yang L. The combined utilization of chlorhexidine and voriconazole or natamycin to combat fusarium infections. BMC Microbiol. 2020 Sep 5;20(1):275. doi: 10.1186/s12866-020-01960-y, PMID 32891143, PMCID PMC7487579.
Al Hatmi AM, Meletiadis J, Curfs Breuker I, Bonifaz A, Meis JF, De Hoog GS. In vitro combinations of natamycin with voriconazole itraconazole and Micafungin against clinical Fusarium strains causing keratitis. J Antimicrob Chemother. 2016 Apr;71(4):953-5. doi: 10.1093/jac/dkv421, PMID 26702918.
Hoffman JJ, Arunga S, Matayan E, Ebong A, Orishaba F, Makupa W. Randomised controlled trial of topical combination therapy chlorhexidine 0.2% and natamycin 5% versus topical natamycin 5% alone for fungal keratitis in East Africa: study protocol. Wellcome Open Res. 2025 Mar 28;10:165. doi: 10.12688/wellcomeopenres.21390.1, PMID 40495927, PMCID PMC12149813.