
Int J App Pharm, Vol 18, Issue 2, 2026, 43-48Original Article
PREPARATION AND EVALUATION OF A CHITOSAN HYDROGEL-BASED SLOW-RELEASE SYSTEM LOADED WITH DOCETAXEL AND EUPHORBIA MICROSCIADIA FOR BREAST CANCER THERAPY IN A MURINE MODEL
MAJID ASADI-SAMANI1, DHIYA ALTEMEMY2, ARMIN AMINI3, MOHSEN SAFAEI4, ELHAM BIJAD5, AKRAM ALIZADEH6, PEGAH KHOSRAVIAN5*
1Cellular and Molecular Research Center, Basic Health Sciences Institute, Shahrekord University of Medical Sciences, Shahrekord, Iran. 2Department of Pharmaceutics, College of Pharmacy, Al-Zahraa University for Women, Karbala, Iraq. 3Student Research Committee, Isfahan University of Medical Sciences, Isfahan, Iran. 4Department of Tissue Engineering, School of Advanced Technologies in Medicine, Fasa University of Medical Sciences, Fasa, Iran. 5Medical Plants Research Center, Basic Health Sciences Institute, Shahrekord University of Medical Sciences, Shahrekord, Iran. 6Nervous System Stem Cells Research Center, Semnan University of Medical Sciences, Semnan, Iran
*Corresponding author: Pegah Khosravian; *Email: pegah.khosraviyan@gmail.com
Received: 11 Jun 2025, Revised and Accepted: 09 Jan 2026
ABSTRACT
Objective: To develop and evaluate a thermosensitive chitosan hydrogel co-loaded with docetaxel and a standardized E. microsciadia extract for breast cancer treatment in a murine 4T1 model.
Methods: The aerial parts of E. microsciadia were collected and identified. A chitosan hydrogel was prepared and loaded with docetaxel and Euphorbia extract. The morphological characteristics of the hydrogel were evaluated using scanning electron microscopy (SEM). Subsequently, the in vitro release profiles of docetaxel and Euphorbia extract from the hydrogel were determined. The antitumor efficacy of the prepared hydrogels was assessed in tumor-bearing mice by monitoring tumor size reduction, followed by histopathological examination of excised tumor tissues.
Results: SEM confirmed hydrogel crosslinking with 5–10 µm pore sizes versus 10–20 µm in controls. Initial release of 60% of both agents occurred within 10 h, reaching 80% by 72 h. Docetaxel and E. microsciadia significantly reduced tumor volumes compared to control (p<0.001). The combination treatment showed no significant difference from docetaxel alone (p>0.05), suggesting no synergistic effect. Histology revealed that combination therapy (docetaxel+E. microsciadia) exhibited superior tumor suppression, with significant cell degeneration and necrosis compared to other treatments.
Conclusion: The co-loaded thermosensitive chitosan hydrogel provides controlled release and effective in vivo antitumor activity. Lack of synergy warrants further optimization to enhance clinical relevance versus current docetaxel systems.
Keywords: Cancer, Euphorbia, Solubility, Controlled release, Animal model
© 2026 The Authors.Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i2.55517 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Breast cancer continues to be one of the most common cancers affecting women globally, and the development of therapeutic resistance remains a significant challenge, especially with taxane-based drugs like paclitaxel and docetaxel [1]. These agents exert their cytotoxic effects by disrupting microtubule dynamics, which not only halts cell division but also triggers apoptosis. However, resistance to taxanes develops through various mechanisms, such as structural changes in microtubules, increased drug efflux mediated by transporters like P-glycoprotein, and the deregulation of intracellular signaling pathways. To address this issue, new strategies are emerging, including the combination of antiangiogenic therapies with vascular normalization agents, inhibitors targeting resistance-related proteins, and anti-inflammatory agents [2, 3]. Combination therapy has become a cornerstone in cancer treatment, where multiple therapeutic agents are administered simultaneously. This strategy offers several benefits compared to monotherapy, notably delaying the emergence of resistant mutations in cancer cells by targeting multiple molecular pathways. Additionally, combining drugs can enhance therapeutic efficacy through a synergistic effect on shared cellular mechanisms [4, 5].
In parallel, medicinal plants have long been recognized for their therapeutic potential in cancer treatment. Specific plant species have garnered attention due to their potent anticancer properties. Phytochemicals derived from these plants can influence several vital biological processes, such as inducing apoptosis, reducing oxidative stress, inhibiting metastasis, and suppressing tumor angiogenesis. The Euphorbia genus, which is part of the Euphorbiaceae family, consists of over 2,000 species, many of which are known for their unique phytochemical compositions and historical medicinal uses. Particularly, Euphorbia species, including members of this genus, have demonstrated strong anticancer and antioxidant activities, primarily due to their flavonoid and triterpenoid compounds. Various species from this genus have been traditionally utilized to treat a variety of ailments, including cancer, with promising results seen in preclinical studies [6]. One such species, Euphorbia microsciadia, is rich in polyphenols and triterpenes, exhibiting cytotoxic effects against cancer cell lines, including those of breast and prostate cancer. These effects are mediated through mechanisms like the inhibition of cell proliferation and the induction of oxidative stress [7-9].
By leveraging both traditional plant-based remedies and modern combination therapies, new avenues for overcoming resistance in breast cancer treatment are emerging, providing hope for more effective therapeutic strategies in the future. However, a critical challenge in pharmacotherapy along this pathway is regulating the rate and extent of drug release. Conventional delivery methods often lead to rapid drug release, causing drug concentrations to spike and potentially result in toxicity and adverse effects [10]. Accordingly, sustained-release drug delivery systems have garnered significant research interest. These systems enable controlled release, maintaining stable plasma drug levels and prolonging drug-target tissue interactions, which collectively enhance therapeutic outcomes and reduce side effects. Hydrogels, as biodegradable and biocompatible materials, offer excellent platforms for such controlled delivery due to their porous, hydrophilic structures that respond to environmental stimuli like pH and temperature [11, 12]. Chitosan, a naturally derived, non-toxic polysaccharide, is particularly suited for drug delivery applications because of its bioadhesive properties and high permeability. Its amine functional groups facilitate interactions with cellular membranes and improve drug penetration [13, 14].
Building on the established limitations of docetaxel, such as dose-dependent toxicity and the potential for resistance, and the documented anticancer and antioxidant properties of Euphorbia microsciadia, particularly due to bioactive compounds like flavonoids, with rutin being a key active constituent, the present study aims to develop and evaluate a chitosan hydrogel-based combined drug delivery system. This system incorporates both docetaxel and E. microsciadia extract, targeting enhanced therapeutic efficacy in breast cancer treatment.
MATERIALS AND METHODS
Chemicals and reagents
The chemicals and reagents used in this study included chitosan (medium molecular weight, degree of deacetylation 75–85%) purchased from Sigma-Aldrich (Germany), docetaxel (99% pure) obtained from Sigma-Aldrich (Germany), and absolute ethanol from Merck (Germany). Acetic acid (99%) was sourced from Merck (Germany), while β-glycerophosphate (β-GP) was procured from Sigma-Aldrich (Germany). Phosphate-buffered saline (PBS, pH 7.4) and Tween 80 (0.5% v/v) were also purchased from Sigma-Aldrich (Germany). For flavonoid quantification, aluminum chloride and rutin standards were obtained from Sigma-Aldrich (Germany). The formalin (10%) and hematoxylin and eosin (H and E) staining kits used for histopathological analysis were sourced from Thermo Fisher Scientific (USA). All reagents were of analytical grade or higher, suitable for the experimental procedures conducted in this study.
Plant material collection and extract preparation
Aerial parts of E. microsciadia were collected during the flowering season from the Chaharmahal and Bakhtiari region of Iran. The plant species was authenticated by a qualified botanist, and a voucher specimen (SKMP-203) was deposited at the herbarium of Shahrekord University of Medical Sciences. The hydroalcoholic extract was obtained via maceration: dried plant material was soaked in a 70:30 ethanol–water solution for 72 h with intermittent stirring. The extract was filtered, concentrated under reduced pressure at 40 °C using a rotary evaporator, and stored at 4 °C until further use. The E. microsciadia extract used in this study was previously characterized phytochemically in our earlier work [7, 9]. This included quantification of total flavonoids and flavonols, antioxidant assessment, and HPLC analysis, which identified rutin as the major flavonoid.
Hydrogel preparation
Chitosan (medium molecular weight, degree of deacetylation 75–85%) was dissolved in 0.1 M acetic acid at a concentration of 4% (w/v) and stirred for 3 h to obtain a homogeneous solution. Docetaxel was first solubilized in a minimal volume of absolute ethanol to address its hydrophobicity. The pre-dissolved docetaxel and the E. microsciadia extract were then dispersed into the chitosan solution. Separately, β-glycerophosphate (β-GP, 20% w/v) was prepared in deionized water and cooled to 4 °C. The β-GP solution was slowly added dropwise to the chitosan mixture while maintaining the system on ice to prevent premature gelation. The final hydrogel formulation was stored at 4 °C until further analysis.
Morphological analysis
The hydrogel was frozen at –20 °C and lyophilized for 48 h. Samples were then gold-coated and examined by scanning electron microscopy (SEM) to assess pore structure and morphology.
In vitro drug release study
One g of hydrogel was placed in 500 ml of phosphate-buffered saline (PBS, pH 7.4) containing 0.5% (v/v) Tween 80 to ensure sufficient solubility of docetaxel. The system was maintained at 37 °C and agitated at 100 rpm. At predetermined intervals (0.5, 1, 2, 4, 6, 12, 24, 48, and 72 h), 1 ml of the release medium was withdrawn and replaced with an equal volume of fresh buffer. Docetaxel content was quantified using UV spectrophotometry at 210 nm, and total flavonoid release (expressed as rutin equivalents) was assessed at 385 nm using the aluminum chloride colorimetric method. The limitations of UV-based quantification, including potential overlap with polymer absorption, are acknowledged in the Discussion section.
Cell line and animal model
Murine 4T1 breast cancer cells were cultured in DMEM supplemented with 10% fetal bovine serum (FBS), 1% penicillin-streptomycin, and maintained at 37 °C in a 5% CO₂ atmosphere. Cells were used at passages 5–10. Female BALB/c mice (6–8 w old, 20–25 g) were obtained from an accredited animal facility. All animal procedures were conducted under approval of the Institutional Animal Care and Use Committee (Ethical Code: IR. SKUMS. REC.1397.166). Mice were housed under controlled conditions (22±2 °C, 12-hour light/dark cycle) with ad libitum access to food and water and acclimatized for one week prior to experimentation.
In vivo study design
Each mouse received a subcutaneous injection of 1×105 4T1 cells into the right flank. Once tumors became palpable (approximately 10 days' post-injection), animals were randomly divided into four groups (n=8 per group): (1) blank hydrogel, (2) hydrogel containing docetaxel, (3) hydrogel containing Euphorbia extract, and (4) hydrogel containing both agents. Treatments were administered via subcutaneous injection (2 ml per mouse). Tumor volume was measured daily using calipers and calculated as (length × width²)/2. On Day 10 post-treatment, animals were euthanized and tumors excised for histological evaluation [15].
Histopathological analysis
Tumors were sectioned, fixed in formalin, and stained with hematoxylin and eosin (H and E). Microscopic evaluation focused on mitotic activity, nuclear pleomorphism, inflammatory cell infiltration, and tubular architecture. While qualitative comparisons were performed in this study, future work will include semi-quantitative scoring systems to strengthen pathological interpretation.
Statistical analysis
Data were analyzed using SPSS software. Normal distribution was confirmed via Shapiro-Wilk test. Differences between groups were assessed by one-way ANOVA followed by Tukey’s post hoc test. A significance level of p<0.05 was considered statistically meaningful. Tumor volume data are presented as mean±standard deviation (SD).
RESULTS
Morphological evaluation of the hydrogel
Scanning electron microscopy (SEM) was used to assess the microstructure of both the plain and crosslinked chitosan hydrogels. The plain hydrogel exhibited a loosely connected porous architecture with pore diameters ranging from 10 to 20 µm. In contrast, the β-glycerophosphate crosslinked hydrogel displayed a denser and more compact structure, with a noticeable reduction in pore size to approximately 5–10 µm. This morphological change is indicative of successful crosslinking and improved network stability (fig. 1).
In vitro drug release profile
The release behavior of docetaxel and E. microsciadia extract from the crosslinked hydrogel was evaluated in PBS (pH 7.4) containing 0.5% Tween 80 over a 72 h period. Both agents showed a biphasic release profile. An initial burst release occurred within the first 10 h, during which approximately 60% of the encapsulated docetaxel and flavonoid content were released. This was followed by a slower and more controlled release phase, reaching nearly 80% cumulative release by 72 h. The burst effect is attributed to surface-associated drug diffusion, while the sustained phase reflects gradual matrix degradation and diffusion through the hydrogel network (fig. 2 and 3).
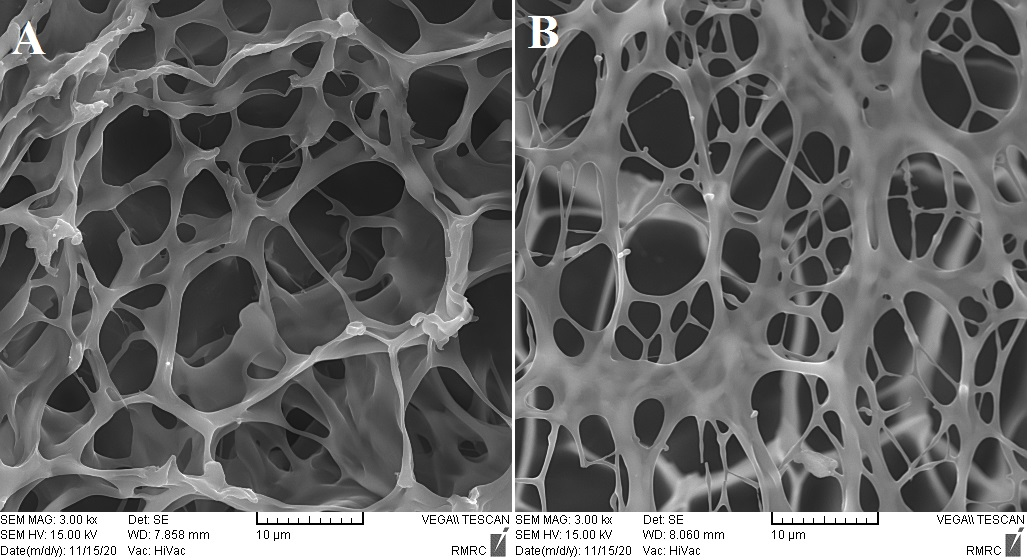
Fig. 1: Chitosan hydrogel (A) and cross-linked chitosan hydrogel (B)
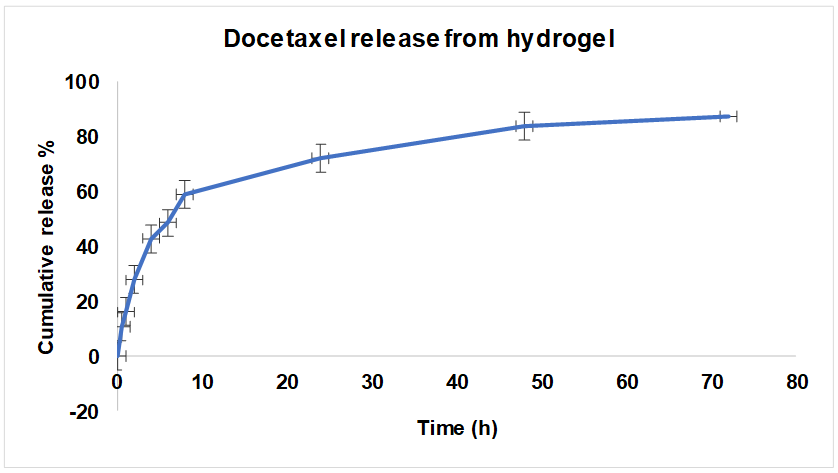
Fig. 2: Docetaxel release curve from cross-linked chitosan hydrogel at pH 7.4. n=3
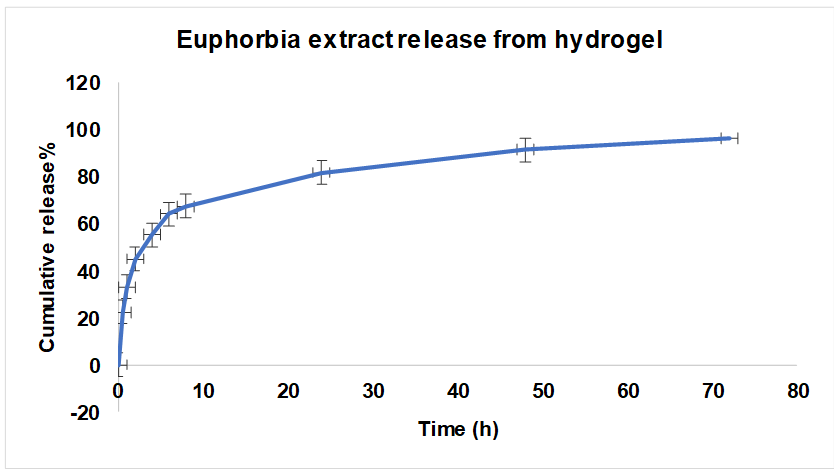
Fig. 3: Release curve of Euphorbia extract from cross-linked chitosan hydrogel at pH 7.4. n=3
In vivo antitumor efficacy
Following hydrogel administration, tumor volume was monitored daily for ten consecutive days in four treatment groups: blank hydrogel, docetaxel-loaded hydrogel, Euphorbia-loaded hydrogel, and a combination-loaded hydrogel. Statistical analysis using one-way ANOVA confirmed a significant difference in tumor volume among the groups (p<0.001).
Tukey’s test revealed significantly lower tumor volumes in both docetaxel and Euphorbia microsciadia groups compared to the hydrogel control group (p<0.01). No significant difference was observed between docetaxel and Euphorbia groups (p>0.05). The combination group (docetaxel+Euphorbia) showed a higher tumor volume than the docetaxel-only group (p<0.001), suggesting the lack of a synergistic effect (fig. 4).
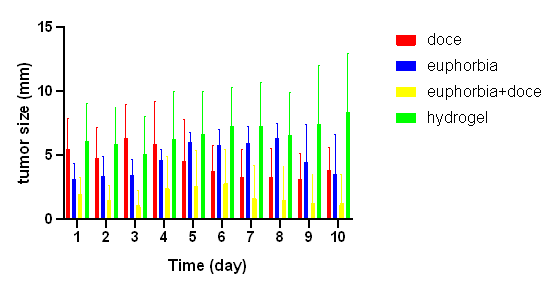
Fig. 4: Comparison of tumor size between study groups from days 1 to 10 after hydrogel injection. Doce: docetaxel; Euphorbia: Euphorbia microsciadia extract; hydrogel: chitosan hydrogel; Tukey’s test showed significantly smaller tumors in docetaxel and Euphorbia microsciadia groups compared to the hydrogel control (p<0.01). No significant difference was observed between docetaxel and Euphorbia (p>0.05). The combination therapy showed no synergistic effect.
Histopathological evaluation
Hematoxylin and eosin (H and E) staining of tumor tissues revealed clear differences in cellular architecture across the treatment groups. Tumors in the hydrogel group exhibited prominent mitotic figures, dense cellularity, nuclear pleomorphism, and moderate to severe inflammatory infiltration, indicating uncontrolled tumor growth. In contrast, the docetaxel group displayed significant reductions in mitotic activity, nuclear atypia, and inflammatory response, with clear evidence of necrotic regions. The E. microsciadia group showed moderate decreases in tumor cell density and structural disorganization, but to a lesser extent than the docetaxel group. The most pronounced cellular regression was observed in the combination therapy group (docetaxel and E. microsciadia), which demonstrated extensive tumor cell degeneration, increased necrosis, and a substantial reduction in viable tumor cells. These findings support the superior efficacy of the combination therapy in reducing tumor growth compared to the other treatments (fig. 5). The findings were consistent with the tumor volume data and are discussed accordingly.
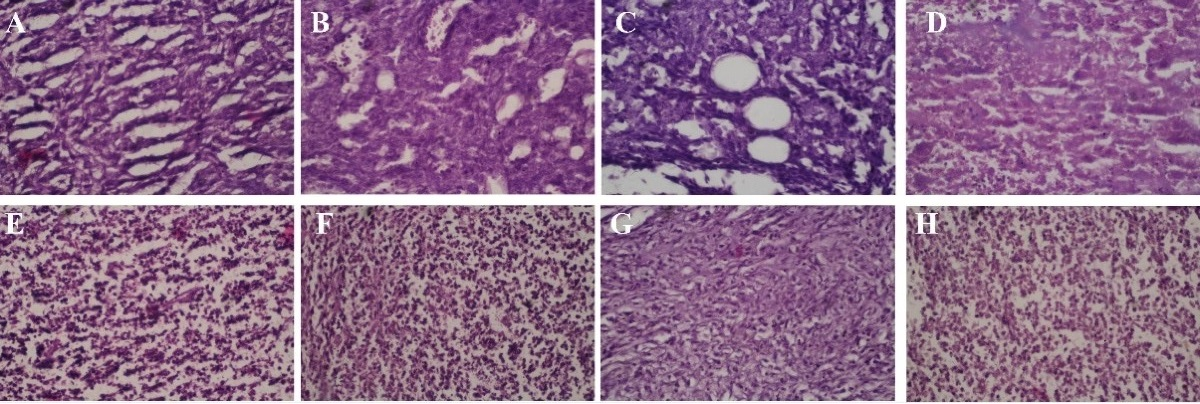
Fig. 5: Histopathological analysis of breast tumor tissues from mice treated with hydrogel (A, E), docetaxel (B, F), Euphorbia microsciadia extract (C, G), and the combination of docetaxel with E. microsciadia (D, H); H and E staining revealed significant differences in tumor morphology across treatment groups. The hydrogel group showed dense cellularity, mitotic figures, and inflammation, indicating uncontrolled growth. Docetaxel treatment reduced mitotic activity, nuclear atypia, and inflammation, with evidence of necrosis. The Euphorbia microsciadia group showed moderate tumor regression. The combination therapy (docetaxel+Euphorbia) resulted in the most pronounced tumor cell degeneration, necrosis, and reduced viable tumor cells, highlighting its superior efficacy.
DISCUSSION
The present study explored a sustained-release hydrogel system composed of chitosan crosslinked with β-glycerophosphate, designed to deliver both docetaxel and a hydroalcoholic extract of E. microsciadia in a murine model of breast cancer. The formulation strategy aimed to enhance local drug retention, reduce systemic toxicity, and examine whether the combination of a cytotoxic agent with a plant-derived extract could yield additive or synergistic antitumor effects. The in vitro drug release profile demonstrated a biphasic pattern characterized by an initial burst followed by a slower, sustained release phase. The burst likely resulted from the release of surface-associated drug, whereas the subsequent phase corresponded to matrix diffusion and gradual degradation. Although this release behavior may allow for early therapeutic concentrations, the relatively high burst magnitude (~60% in 10 h) suggests a need for further formulation optimization, potentially through adjustments in crosslinker ratio or incorporation of nanoparticulate carriers [13, 16]. Morphological assessment confirmed successful hydrogel formation, with SEM revealing reduced pore sizes in the crosslinked matrix, indicative of improved structural integrity and potential for controlled release [17].
In this study, we observed that all treatments with active agents significantly reduced tumor growth compared to the control group receiving hydrogel alone. However, the docetaxel-only group showed the most notable decrease in tumor volume, outperforming both the E. microsciadia extract and the combination treatment. Surprisingly, the combination of docetaxel and E. microsciadia resulted in even higher tumor volumes than the docetaxel group alone, indicating that no synergistic effect was present. These results suggest that, under the conditions tested, combining docetaxel with E. microsciadia extract did not enhance the treatment's effectiveness. The increase in tumor size seen in the combination group calls for further investigation into possible pharmacokinetic or physicochemical factors that might have interfered with the expected therapeutic outcomes of docetaxel. This observation underscores the importance of thoroughly evaluating drug–extract compatibility and interaction profiles in combination delivery systems [18].
Histological analysis revealed a decrease in mitotic activity, nuclear atypia, and inflammatory infiltration in the treated groups compared to the control, indicating an effective reduction in tumor aggressiveness. Among the treatments, the combination therapy (docetaxel with E. microsciadia) showed the most substantial improvement, with the greatest reductions in these parameters. This suggests that the combination therapy not only more effectively suppressed tumor cell proliferation but also resulted in a more significant reduction in inflammatory responses and nuclear polymorphism compared to the other treatments. These results underscore the superior anti-tumor potential of the combination therapy, positioning it as a promising strategy for achieving more robust and sustained tumor growth inhibition.
It is also important to acknowledge the limitations of the current work. Drug loading capacity and encapsulation efficiency were not quantified, nor were rheological properties, degradation kinetics, or molecular interaction data (e. g., FTIR or DSC) obtained. These parameters are essential for comprehensive formulation characterization and should be incorporated in subsequent research. Furthermore, the use of UV-based quantification methods, though practical, lacks the specificity of chromatographic techniques. Future studies should employ HPLC or LC-MS to ensure accurate and selective drug analysis.
From a translational standpoint, the subcutaneous route used in this model may not fully replicate clinically relevant delivery methods such as intravenous or intratumoral administration. While the current design offers valuable preclinical insight into formulation behavior and tumor response, further work is required to assess systemic pharmacokinetics, biodistribution, and toxicity, particularly with regard to organ-level biocompatibility. Additionally, no overt toxicity or behavioral abnormalities were observed in the animals during the study period, but detailed safety profiling—including histopathology of major organs—was beyond the scope of this investigation and should be prioritized in future work.
CONCLUSION
This study demonstrated the successful formulation of a thermosensitive chitosan hydrogel crosslinked with β-glycerophosphate, exhibiting reduced pore size (5–10 µm) compared to uncross linked hydrogel (10–20 µm) and confirming effective network formation. Both docetaxel and E. microsciadia extract displayed an initial burst release within the first 24 h, followed by a sustained release over 72 h. In the 4T1 murine breast cancer model, hydrogels loaded individually with docetaxel or Euphorbia extract significantly reduced tumor volume and improved histopathological parameters compared to controls. However, the combined hydrogel treatment did not demonstrate additive or synergistic antitumor effects over docetaxel alone. Furthermore, it is important to note that the subcutaneous hydrogel administration used in this study may not fully replicate clinically relevant drug delivery routes such as intravenous or intratumoral injections typically employed in metastatic breast cancer treatment. This limitation underscores the need for further investigation into optimized delivery strategies and formulation refinements to enhance the translational potential of this system. Overall, the chitosan hydrogel platform provides a promising controlled-release vehicle for docetaxel and natural extracts, warranting additional studies to better understand and improve combination efficacy.
ACKNOWLEDGMENT
We gratefully acknowledge the Vice Chancellor for Research and Technology at Shahrekord University of Medical Sciences for funding this project. We also extend our sincere thanks to the staff of the Phytochemical Laboratory at the Research Center for Medicinal Plants, Shahrekord University of Medical Sciences, for their invaluable support throughout the study.
FUNDING
This work was supported by a grant from the Vice Chancellor for Research and Technology of Shahrekord University of Medical Sciences, Iran (Grant No. 3677).
AUTHORS CONTRIBUTIONS
Majid Asadi-Samani: Conceptualization, Investigation, Data Curation, Writing-Review and Editing; Dhiya Altememy: Writing-Review and Editing, Data Curation; Pegah Khosravian: Conceptualization, Supervision, Data Curation, Writing-Review and Editing; Armin Amini, Mohsen Safaei, and Elham Bijad: Investigation, original draft writing; Akram Alizadeh: Conceptualization, Data Curation, original draft writing; All the authors have read and agreed to publish the manuscript.
CONFLICTS OF INTERESTS
The authors declare no conflicts of interest related to this study
REFERENCES
Khan MM, Yalamarty SS, Rajmalani BA, Filipczak N, Torchilin VP. Recent strategies to overcome breast cancer resistance. Crit Rev Oncol Hematol. 2024;197:104351. doi: 10.1016/j.critrevonc.2024.104351, PMID 38615873.
Abouzeid HA, Kassem L, Liu X, Abuelhana A. Paclitaxel resistance in breast cancer: current challenges and recent advanced therapeutic strategies. Cancer Treat Res Commun. 2025;43:100918. doi: 10.1016/j.ctarc.2025.100918, PMID 40215760.
Alalawy AI. Key genes and molecular mechanisms related to paclitaxel resistance. Cancer Cell Int. 2024;24(1):244. doi: 10.1186/s12935-024-03415-0, PMID 39003454.
Bayat Mokhtari RB, Homayouni TS, Baluch N, Morgatskaya E, Kumar S, Das B. Combination therapy in combating cancer. Oncotarget. 2017;8(23):38022-43. doi: 10.18632/oncotarget.16723, PMID 28410237.
Lee JH, Nan A. Combination drug delivery approaches in metastatic breast cancer. J Drug Deliv. 2012;2012(1):915375. doi: 10.1155/2012/915375, PMID 22619725.
Aleksandrov M, Maksimova V, Koleva Gudeva L. Review of the anticancer and cytotoxic activity of some species from genus Euphorbia. Agric Conspect Sci. 2019 Mar 25;84(1):1-5.
Asadi Samani M, Rafieian Kopaei M, Lorigooini Z, Shirzad H. A screening to determine total phenol and flavonoid content of some Iran’s medicinal plants grown in Chaharmahal va Bakhtyari province. Indian J Nat Prod Resour. 2019;9(4):296-302. doi: 10.56042/ijnpr.v9i4.22122.
Asadi Samani M, Rafieian Kopaei M, Lorigooini Z, Shirzad H. A screening of growth inhibitory activity of Iranian medicinal plants on prostate cancer cell lines. Biomedicine. 2018;8(2):8. doi: 10.1051/bmdcn/2018080208, PMID 29806586.
Asadi Samani M, Rafieian Kopaei M, Lorigooini Z, Shirzad H. A screening of anti-breast cancer effects and antioxidant activity of twenty medicinal plants gathered from Chaharmahal va Bakhtyari province Iran. J Pharm Pharmacogn Res. 2019;7(1):213-22. doi: 10.56499/jppres19.611_7.3.213.
Phalak SD, Bodke VI, Yadav RE, Pandav SA, Ranaware MA. A systematic review on nano drug delivery system: solid lipid nanoparticles (SLN). Int J Curr Pharm Sci. 2024;16(1):10-20. doi: 10.22159/ijcpr.2024v16i1.4020.
Khoee S, Kardani M. Hydrogels as controlled drug delivery carriers. Polymerization. 2013;2(4):16-27. doi: 10.22063/basparesh.2013.955.
Qiu Y, Park K. Environment-sensitive hydrogels for drug delivery. Adv Drug Deliv Rev. 2001;53(3):321-39. doi: 10.1016/s0169-409x(01)00203-4, PMID 11744175.
Popat A, Liu J, Lu GQ, Qiao SZ. A pH-responsive drug delivery system based on chitosan-coated mesoporous silica nanoparticles. J Mater Chem. 2012;22(22):11173. doi: 10.1039/c2jm30501a.
Thanou M, Verhoef JC, Junginger HE. Chitosan and its derivatives as intestinal absorption enhancers. Adv Drug Deliv Rev. 2001;50(Suppl 1):S91-101. doi: 10.1016/S0169-409X(01)00180-6, PMID 11576697.
Kalnina Z, Lieknina I, Kotelovica S, Petrovska R, Zvinys G, Petrosiute A. Development of 4T1 breast cancer mouse model system for preclinical carbonic anhydrase IX studies. FEBS Open Bio. 2025 May 15;15(8):1285-302. doi: 10.1002/2211-5463.70052, PMID 40371725.
Andrgie AT, Tsai HC. Hydrogels as local depots for on-demand therapeutic delivery: potential therapeutic approaches for tumor metastasis. Mater Adv. 2024;5(9):3629-43. doi: 10.1039/d4ma00122b.
Javdani M, Barzegar A, Khosravian P, Hashemnia M. Evaluation of inflammatory response due to use of controlled release drug delivery system of chitosan hydrogel loaded with buprenorphine and ketorolac in rat with experimental proximal tibial epiphysis defect. J Invest Surg. 2022;35(5):996-1011. doi: 10.1080/08941939.2021.1989728, PMID 34666588.
Riet K, Adegoke A, Mashele S, Sekhoacha M. Effective use of Euphorbia milii DCM root extract encapsulated by thermosensitive immunoliposomes for targeted drug delivery in prostate cancer cells. Curr Issues Mol Biol. 2024;46(11):12037-60. doi: 10.3390/cimb46110714, PMID 39590308.
Abo-Dola MA, Lutfi MF. Anti-inflammatory activity of Euphorbia aegyptiaca extract in rats. Int J Health Sci (Qassim). 2016;10(1):69-75. doi: 10.12816/0033255, PMCID PMC4791159, PMID 27004059.
Gras J. Ingenol mebutate: a new option for actinic keratosis treatment. Drugs Today (Barc). 2013;49(1):15-22. doi: 10.1358/dot.2013.49.1.1910723, PMID 23362492.