
Int J App Pharm, Vol 18, Issue 1, 2026, 542-550Original Article
MESALAZINE PREFORMULATION AND SOLUBILITY STUDY
RUSSUL HAMEED1*, MOWAFAQ M. GHAREEB2
1The Ministry of Health of the Republic of Baghdad, Iraq. 2Department of Pharmaceutics College of Pharmacy, University of Baghdad, Baghdad, Iraq
*Corresponding author: Russul Hameed; *Email: rusul.atia2200@copharm.uobaghdad.edu.iq
Received: 22 Jun 2025, Revised and Accepted: 29 Oct 2025
ABSTRACT
Objective: Aim of preformulation study is to develop the elegant, stable, effective and safe dosage form by assay solubility and stability study of MSL.
Methods: Preformulation study was done to check the purity and stability of drug by different steps of preformulation study, first of this step involve identification of melting point by DSC, and by using UV spectrophotometry can be detection of lambda max and calibration curve in ethanol and phosphate buffer pH7.4. Drug was dissolved in different media to evaluate its solubility and stability in The study includes solubility, stability, and pH of drug in different oil (Almond oil, Grape oil, Sesame oil, Clove oil, Olive oil, Jojoba oil, and Labrafil oil), different surfactants (Cremophor, Tween 40, Tween 80, PEG 400, PEG600, PEG200, Labrasol), different co surfactants (Oleic acid, Imm 948, Vitamin E, frankincense oil, Labrafac oil, and Imwitor30 with and without different antioxidants (sodium thiosulfate, sodium metabisulfite, vitamin C, and Vitamin E. FTIR done to check purity and identity of drug and to evaluate MSL-solubilizing media interaction.
Results: The results from the Preformulation studies show melting point and lambda max of mesalamine were found to be 285.5 °C and 330 nm respectively. MSL best condition of preparing in a solution in oil and acidic media at room temperature. The FTIR study of pure MSL confirms the purity of drug and FTIR show the interaction of drug with tween 40; in which FTIR of MSL-tween40 show chemical interaction.
Conclusion: from solubility study of MSL in deferent media show different concentration of drug dissolve in mg/ml and stability of drug was affected by type of media mainly pH of media in which oil media show better stability.
Keywords: Preformulation, Meslazine, Ulcerative colitis, Drug-excipients compatibility studies
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.55689 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Mesalazine (5-aminosalicylic acid) is the active component of sulfasalazine, which has been used for a long time in the treatment of ulcerative colitis and Crohn’s disease, either orally in different controlled-release forms or rectally in the form of suppositories, enemas and foams. Mesalazine (MSL), chemically known as 5-amino-2 hydroxybenzoic acid its chemical structure shown in fig. 1, is an anti-inflammatory drug used to treat and also maintain the remission of mild to moderate ulcerative colitis or Crohn's disease. MSL has been shown to be a potent scavenger of reactive oxygen species that play a significant role in the pathogenesis of inflammatory bowel disease, inhibition of natural killer cell activity, inhibition of antibody synthesis, inhibition of cyclooxygenase and lipoxygenase pathways and impairment of neutrophil function [1]. The purpose of preformulation testing is to determine how the physicochemical qualities of the drug ingredients and excipients will affect the final product's formulation, manufacturing technique, and pharmacokinetic and biopharmaceutical properties. To find out whether an API (new medicinal entity) is compatible with typical excipients, preformulation studies are useful. In order to increase bioavailability, it directs the formulator to select the appropriate drug substance form. It helps to learn everything there is to know about a new drug entity. It is also useful for figuring out how to keep formulations fresh. The development of a stable, effective, and safe dosage form can be aided by preformulation studies [2, 3]. Before starting the preformulation studies, we should know the properties of the drug, potency relative to the competitive products and the dosage form, literature search providing stability and decay data, the proposed route of drug administration, literature search regarding the formulation approaches, bioavailability and pharmacokinetics of chemically related drugs [4]. It is white to pinkish crystals, slightly soluble in water, 20 to 30 % absorbed following oral administrations [5]. The determination of the physicochemical properties and intrinsic solubility rate of mesalamine will provide a basis for meaningful characterization of the drug. Mesalazine is unstable in gastric acid, N-acetylated MSL is mainly excreted into the urine. After rectal administration, 10–35% of the drug is absorbed, whereas after oral administration, depending on the dose and the type of formulation used, 15–67% of the drug is absorbed and excreted into the urine, MSL is metabolized locally by the intestinal N-acetyltransferase 1 (NAT1), 5-ASA acts locally on the colonic epithelium and has multiple anti-inflammatory effects. Although the exact mechanism of action remains to be elucidated, the main mechanism includes inhibition of cyclooxygenase and lipoxygenase, subsequently leading to reduced production of prostaglandins and leukotrienes, respectively. After oral and rectal administration, systemic concentrations of 5-ASA are low (\ 0.5 lg/ml) compared with intraluminal colonic concentrations, and have been shown to increase with higher oral 5-ASA doses. To evaluate the concentration–effect relationship of 5-ASA, local tissue measurement of 5-ASA is preferred because of the topical mechanism of action [6, 7].
The objective of this article is to study of MES physicochemical properties; solubility and stability in different media, include oil, polymers and surfactant.
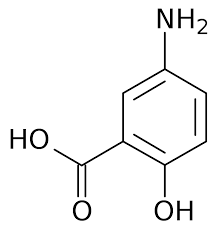
Fig. 1: Chemical structure of mesalazine [8]
MATERIALS AND METHODS
Material
Mesalazine was purchased from Hyperchem China and the oils (Labrosol, Transculol, Cremophare, Oleic acid, Almond oil, Grape oil, Sesame oil, Imm 948, Clove oil Olive oil, Peppermint oil, Vitamin E, Jojoba oil, Frankincenseoil, Labrafil oil Labrafac oil and Imwitor308) and other surfactant from Kathy chinaz.
Characterization of mesalazine
Before developing any dosage form, it is crucial to determine the physicochemical properties of the medicine through preformulation research. This information aids the formulator in developing a refined, stable, efficacious, and safe dosage form. Moreover, preformulation studies enable the validation of the drug's legitimacy.
Methods
UV-wavelength (lambda max)
The lambda max (λmax) of MSL was identified using two media: ethanol and phosphate buffer at pH 7.4. Precisely measured mesalamine (10 mg) was dissolved in a 10 ml volumetric flask containing ethanol for measurement of the drug's λmax in ethanol, and in 10 ml of phosphate buffer at pH 7.4 for detection of λmax in the phosphate buffer. The volume was increased to a level containing a sufficient concentration of ethanol and phosphate buffer at pH 7.4. This yielded a concentration of 100 µg/ml. Furthermore, several aliquots were generated from the stock solution. The aliquots underwent analysis using spectrophotometry (Shimadzu, UV-1800, Japan) [9].
Preparation of standard curve
Mesalazine (MSL) (10 mg) was accurately weighed and dissolved in a 100 ml volumetric flask containing phosphate buffer (pH 7.4) and ethanol separately. To get a 100 μg/ml concentration. This stock solution was used to prepare further standard solutions for the drug with appropriate dilution. From the standard stock solution, several aliquots were measured using a UV spectrophotometer (Shimadzu, UV-1800, Japan).
Solubility and stability Studies of MSL
The solubility experiments utilise the conventional shake flask approach. The solubility values of MSL in various media are determined as presented in table 1. Excess quantities of MSL are introduced into a plain tube containing 2 ml of different oils, surfactants, or co-surfactants until it reaches supersaturation. The tube is then securely closed and put on a water bath shaker. The medication solution was agitated for 48 h at 25 °C, the sample was centrifuged at 3000 rpm for 30 min, and then 0.5 ml of the supernatant was pulled out and diluted to 10 ml with ethanol. After that, the concentration was quantified by using an ethanol calibration curve in lambda max 331 nm using UV spectrophotometry [10].
Effect type of media on solubility
To understand the effect of the type of media on UV spectrophotometry absorbance of MSL, these different media affected the stability of the MSL preparation. Different oils, surfactants and co-surfactants are used, as shown in table 1.
Effect the type and concentration of antioxidant on solubility
To evaluate the effect of the type of antioxidant on UV spectrophotometry absorbance of MSL. When we use a different type of antioxidant, we evaluate how its concentration and type affect the discolouration of MSL. n. The amount of antioxidant mixed with MSL in a 50% ratio is represented by the formula code F*, as shown in table 1.
Table 1: The effect of type of media and present of antioxidant
| Experiments no. | Media | Experiments no. after add of antioxidant | Type of antioxidant |
| F1 | Labrosol | F1* | Sodium metabisulfite |
| F2 | Transcutol | F2* | Sodium metabisulfite |
| F3 | Cremophor | F3* | Sodium metabisulfite |
| F4 | Tween 40 | F4* | Sodium metabisulfite |
| F5 | Propylene glycol | F5* | Sodium metabisulfite |
| F6 | Tween 80 | F6* | Sodium metabisulfite |
| F7 | PEG 400 | F7* | Sodium metabisulfite |
| F8 | PGE600 | F8* | Sodium metabisulfite |
| F9 | PEG200 | F9* | Sodium metabisulfite |
| F10 | Oleic acid | F10* | Sodium metabisulfite |
| F11 | Almond oil | - | - |
| F12 | Grape oil | - | - |
| F13 | Sesame oil | - | - |
| F14 | Imm 948 | - | - |
| F15 | Clove oil | - | - |
| F16 | Olive oil | - | - |
| F17 | Peppermint oil | - | - |
| F18 | Vitamin E | - | - |
| F19 | Jojoba oil | - | - |
| F20 | Frankincense oil | - | - |
| F21 | Labrafil oil | F21* | Sodium metabisulfite |
| F22 | Labrafac oil | F22* | Sodium metabisulfite |
| F23 Imwitor308 | F23* | Sodium metabisulfite |
Fourier transform infrared spectroscopy (FTIR)
FTIR scanning of KBr pellets containing powder samples of pure drug, and MSL-Solubilizing media the wave number range 400-4000 cm-1 at a resolution of 4 cm-1, with speed of 2 mm/sec [11] grinding the sample with potassium bromide (KBr) and subsequently compacting it into a thin film disc using a precise technique.
Differential scanning calorimetric (DSC)
The thermodynamic characteristics of MSL were evaluated using a DSC-60 Plus instrument (Shimadzu, Japan). A sample weighing approximately 2 mg was placed in aluminum pans and heated from 30 to 400 °C at a rate of 10 °C/min, with nitrogen flowing at a rate of 50 ml/min [12].
RESULTS AND DISCUSSION
Melting point determination
DSC provides the information on the physical characteristics of the sample. According to the thermograms fig. 2, MSL presented a sharp endothermic peak at 285.5 °C, which is agree with references, Gupta K, Roy SB and Newton AMJ, Prabakaran [13, 14].
UV-wavelength (λmax) and standard curve
The lambda max of MSL in ethanol was 331 nm, which closed to value obtain by Rakesh Kumar Singh [15] and phosphate buffer pH 7.4 was 330.5 nm that closed to value in these references [9, 16], as shown in UV spectrophotometry fig. 3 and 4, and the calibration curve shown in fig. 5 and 6 for MSL in ethanol and phosphate buffer pH 7.4, and stander curve obtain from data in table 2. That have linear regression with R2 value equal to 0.9994 for ethanol stander curve and linear regression with R2 value equal 0.997 in phosphate buffer pH 7.4.
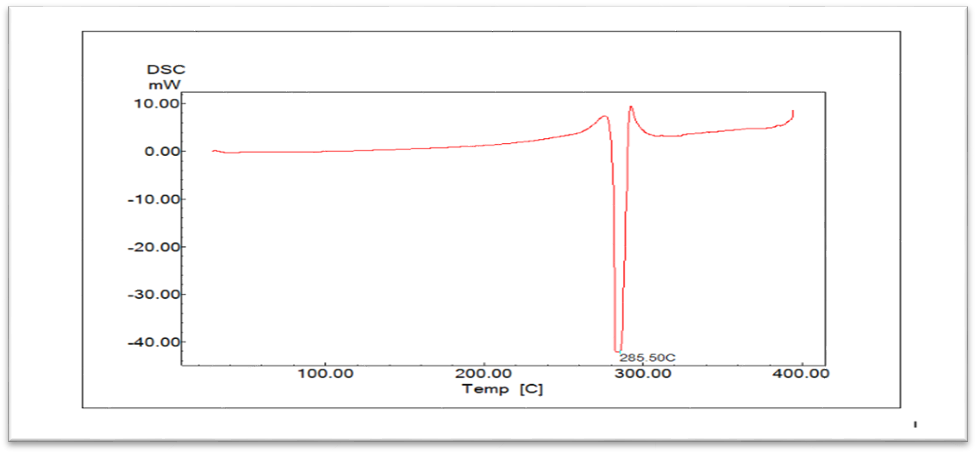
Fig. 2: Thermograms of mesalazine
Table 2: Linearity table of MSL in working standard (A) in ethanol (B) in phosphate buffer pH 7.4
| A | Concentration μg/ml | Absorbance | B | Concentration μg/ml | Absorbance |
| 0 | 0 | 0 | 0 | ||
| 20 | 0.219 | 5 | 0.094 | ||
| 30 | 0.306 | 10 | 0.213 | ||
| 40 | 0.4 | 15 | 0.316 | ||
| 50 | 0.501 | 20 | 0.463 | ||
| 60 | 0.609 | 25 | 0.562 | ||
| 70 | 0.71 | 30 | 0.712 | ||
| 80 | 0.824 | 35 | 0.827 | ||
| 90 | 0.92 | 40 | 0.981 |
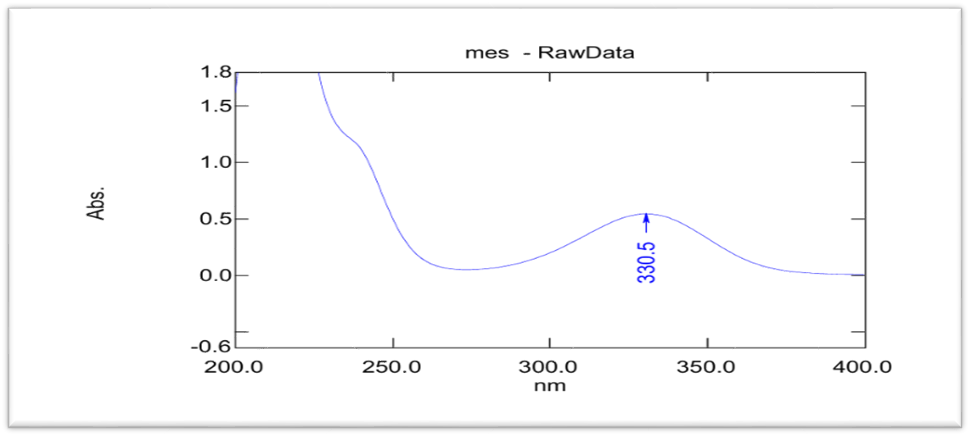
Fig. 3: Lambda max of mesalazine in phosphate buffer pH 7.4
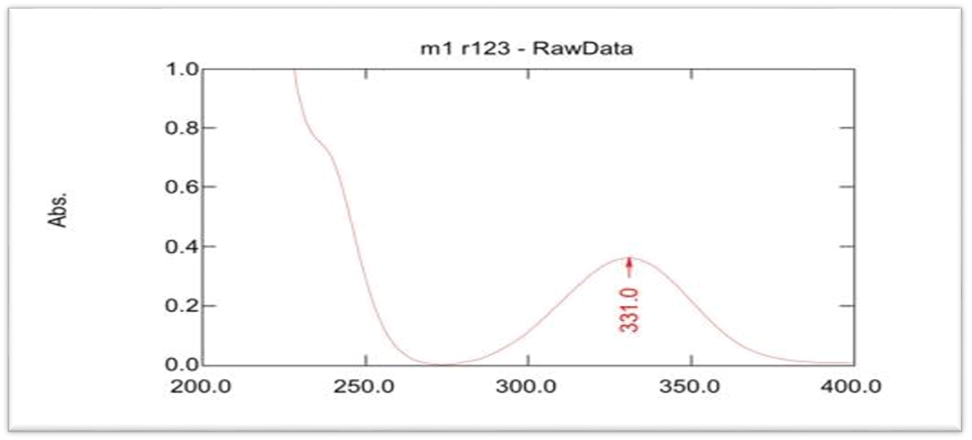
Fig. 4: Lambda max of mesalazine in ethanol
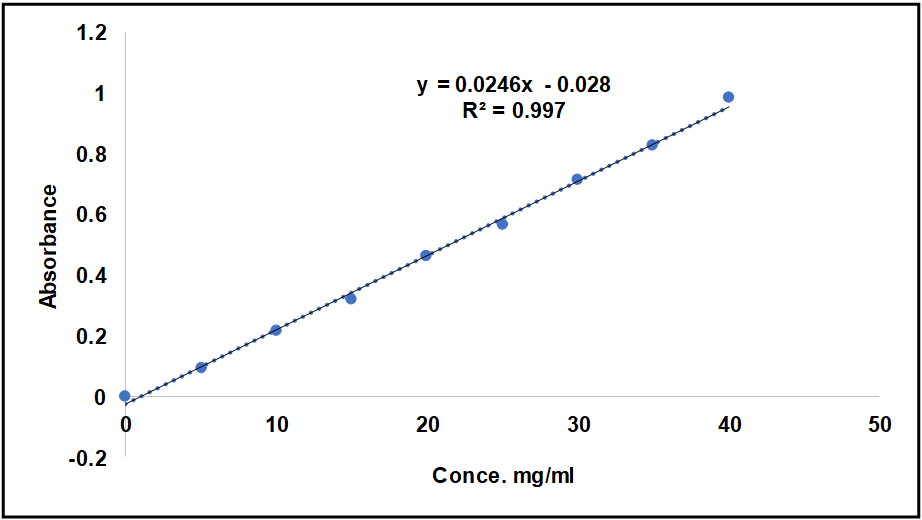
Fig. 5: Standard curve of MSL in phosphate buffer pH 7.4
Fig. 6: Standardc curve of MSL in ethanol
Solubility study
The solubility of MSL in different media, show result that MSL well suffer from changed in color in some of these media, as shown in table 3 and fig. 10.
The effect type of media on solubility study
When we used oil as media, as shown in table 3, the absorbance was detected and the concentration of dissolved MSL. The solubility of MSL in surfactant and co-surfactant was measured and the color change to deep brown was more significant in the case of using surfactant and co-surfactants than oils, as shown in table 3. The oxidation potentials facilitated the ranking of the compounds according to their susceptibility to oxidation. The color alteration of MSL upon dissolving in surfactant and co-surfactant media occurred within less than two hours, indicating a susceptibility to oxidation. Theoretically, MSL upon oxidation well loses its aromaticity, transforming into a quinone form as shown in fig. 7 [17]. As in E. S. Salih1; M. S. Al-Enizzi The investigation in their paper revealed that the blue dye product was generated exclusively in an alkaline medium; hence, the effects of several alkaline solutions (pH 9-12) and sodium hydroxide (pH>12) were examined [18].
Mesalazin is an acidic compound with pKa values of 2.30 and 5.69. It exhibits a pH-dependent solubility profile, with a pKa for the phenolic hydroxyl group (–OH) of MSL. This group possesses a pKa of around 5.8, indicating it donates a proton at somewhat elevated pH levels relative to the carboxylic acid group [18].
When we dissolve in basic media (surfactant and co-surfactant), the pH of these media is shown in table 3; the discoloration process develops well within 2 h., and the suggested mechanism of MSL oxidation developing in basic media forms intense dye [19].
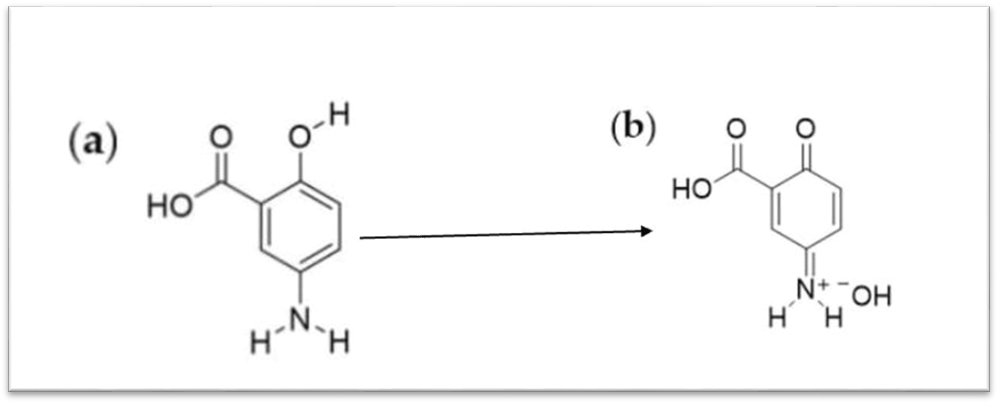
Fig. 7: Theoretical oxidation of mesalazine [16]
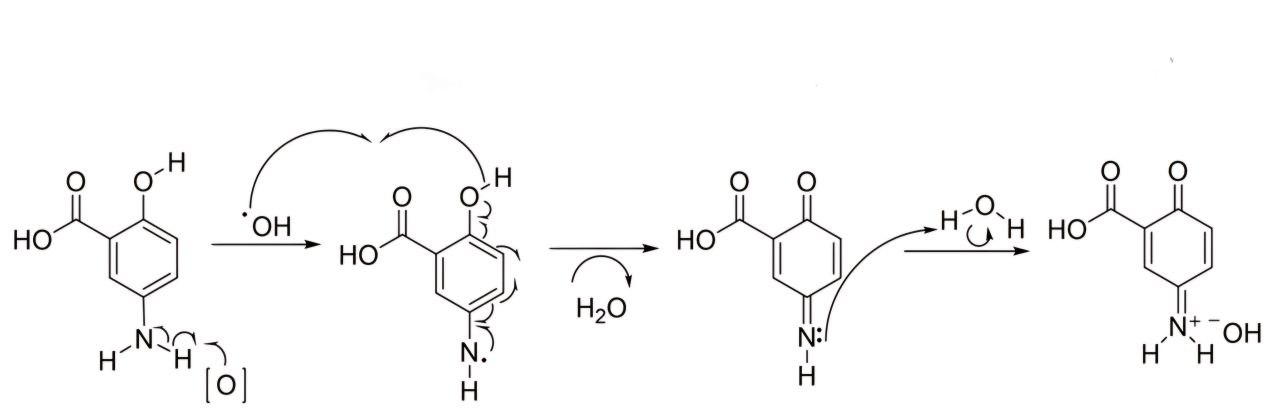
Fig. 8: Theoretical MSL mechanism of oxidation in basic media [19]
Table 3: Solubility value of MSL in different media
| Formula symbol | Media | Concentration mg/ml mean N±SD | Color changed |
| F1 | Labrosol | 52.04±0.002 | yes |
| F2 | Transcutol | 73.8± 0.00132 | yes |
| F3 | Cremophor | 101.24±0.023 | yes |
| F4 | Tween 40 | 118.02±0.02 | yes |
| F5 | Propylene glycol | 59.4±0.004 | yes |
| F6 | Tween 80 | 100.5±0.031 | yes |
| F7 | PEG 400 | 121.2±0.0021 | yes |
| F8 | PGE600 | 139.68±0.0019 | yes |
| F9 | PEG200 | 177.33±0.022 | yes |
| F10 | Oleic acid | 26.25±0.0012 | yes |
| F11 | Almond oil | 35.8±0.00133 | No |
| F12 | Grape oil | 9.78±0.0023 | No |
| F13 | Sesame oil | 1.84±0.011 | No |
| F14 | Imm 948 | 14.29±0.0011 | No |
| F15 | Clove oil | 5.12±0.005 | No |
| F16 | Olive oil | 5.47±0.017 | No |
| F17 | Peppermint oil | 181.8±0.007 | No |
| F18 | Vitamin E | 85.5±0.0041 | No |
| F19 | Jojoba oil | 2.92±0.009 | No |
| F20 | Frankincense oil | 4.59±0.018 | No |
| F21 | Labrafil oil | 27.2±0.026 | yes |
| F22 | Labrafac oil | 4.5±0.0056 | yes |
| F23 | Imwitor308 | 10.25±0.0044 | yes |
N=number of sampling which equal to 3 (mean of N±SD)
That mean there is relationship between intensity of color and pH of media. The effect of pH was studied by forming the colored product in the presence of various pH the absorbance of the proton transfer product was measured fig. 7 and 8 [20] In oil media, free fatty acids are recognized by the acidity index, which results in an acidic pH value for the oil. The acidity index of oil is defined by British Standard specifications as either the milligrams of potassium hydroxide necessary to neutralize the total acidity in 100 g of oil or as a percentage of oleic acid [21]. This indicates that there is no oxidation in the oil. In the solubility study of MSL in co-surfactants and surfactants with a pH value exceeding 5, as shown in fig. 10 [22] In fig. 9 show the chemical structure of some of media that show chemical interaction with MSL.
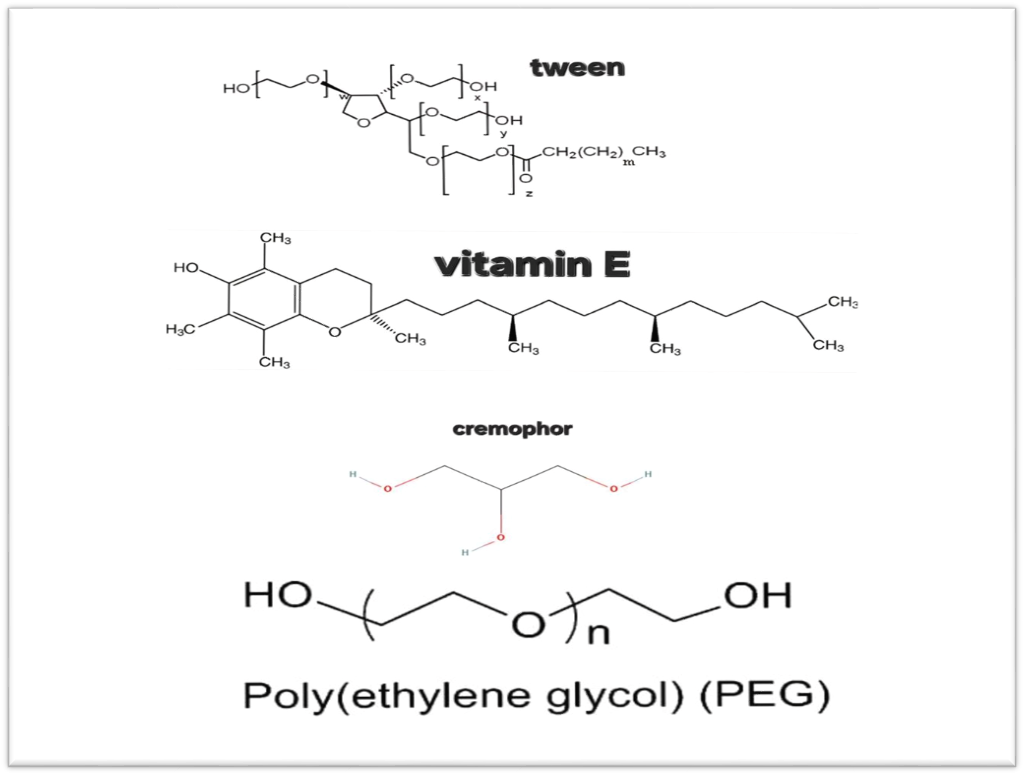
Fig. 9: Chemical structure of media used [21, 24]
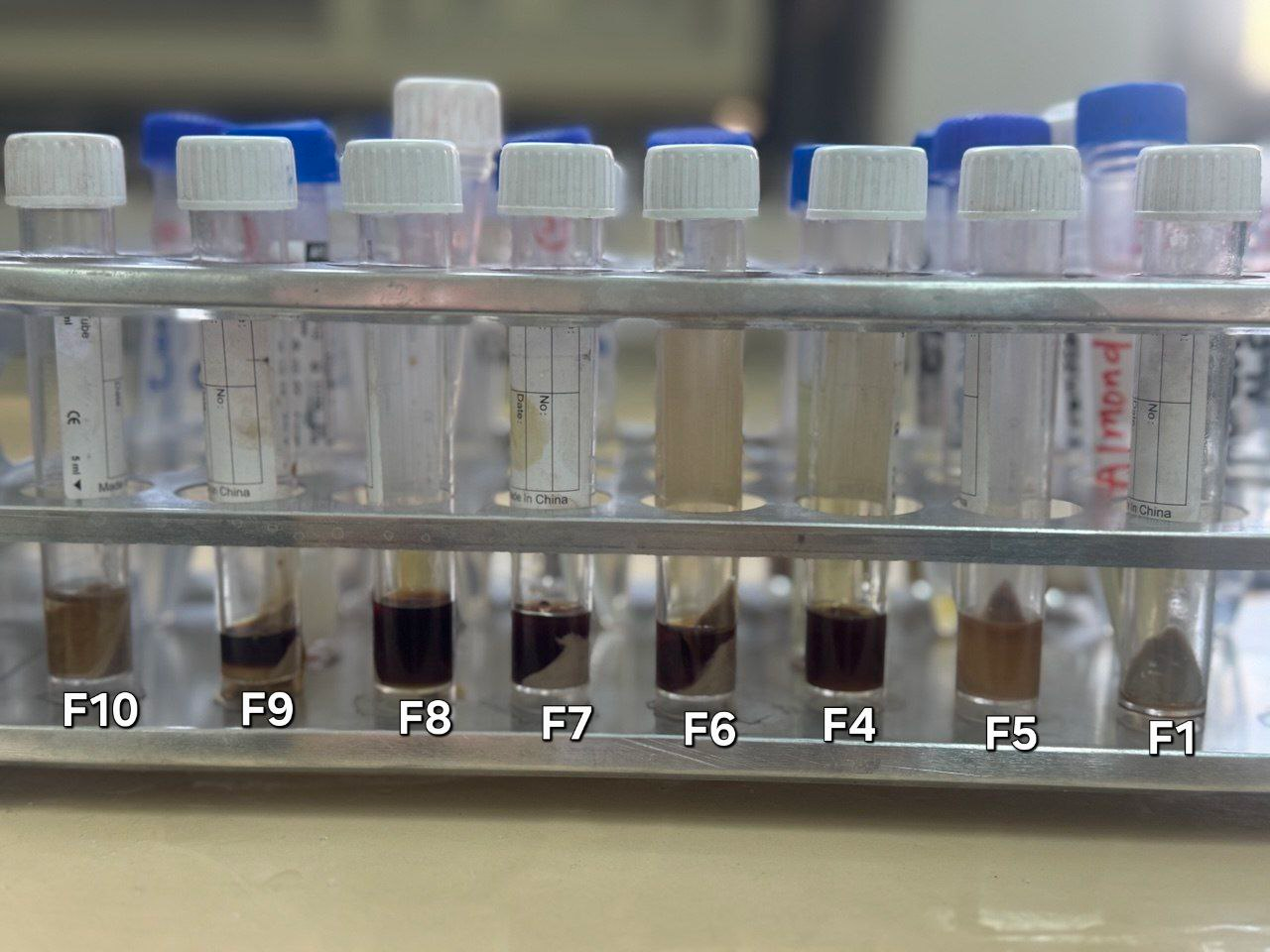
Fig. 10: MSL solution change in color that visually obtain during our analytical experiment in different oil and surfactant
Effect the type and concentration of antioxidant on solubility and stability study
As mention previously, the oxidation more becomes easy at basic media as in surfactant and co-surfactants, to resolve these problem we add antioxidant, at first we add sodium thiosulfate as antioxidant,the result we found there is no resolve in oxidation of MSL as shown in fig. 11, when we use vitamin C (ascorbic acid) as antioxidant, the oxidation did not resolve and the discoloration of solution still intense dark brown color and there is no reduction in oxidation rate [25] after we are use sodium metabisulfate the intensity of color decrease as shown in fig. 12.
As concentration of sodium metabisulfate increase, the solution well be more clear. to study the effect concentration of sodium metabisulfate on tanscutol MSL liquid shown in fig. 13 [16, 25].

Fig. 11: Effect of sodium thiosulfate on discoloration that visually obtain during our analytical experiment

Fig. 12: Effect use sodium metabisulfate on discoloration that visually obtain during our analytical experiment

Fig. 13: Effect of concentration of sodium metabisulfate as antioxidant on discoloration that visually obtain during our analytical experiment
Fourier transform infra-red spectroscopy (FTIR)
FT-IR spectrum of MSL appears, with peaks at 1354 for (OH bending) Carboxylic acids and 1620 cm-1 for (C=O) Carboxylic acids, the peak at 2553 cm-1 for O-H stretching (phenolic and carboxylic that Intramolecular hydrogen bonds), the 1190 cm-1 and 773.46 cm-1 for (C-N), Primary amine (N-H bend) at 1651 cm-1,the peak for C-H stretch of the aromatic group at 2978 cm, aliphatic stretching of C-H is allocated to 2783 cm-1. Aromatic C=C peaks occur in the range 1579 cm-1, that accepted with references [28-30] as show in fig. 14. Fig. 15 show the spectrum correspondent to Tween 40 showed that it possesses a methyl group C-H stretching at 2922 cm-1, an ester group at 1735 cm-1, an alkene group at 1643 cm-1, and an ether group at 1100 cm-1 [29] We chose F4 to detect MSL-solubilizing media interaction due to the most readily occurring discoloration and more intense color with Tween 40. the main peak of MSL was faded as shown in fig. 16, indicate there is chemical interaction, the peaks at 2978 cm-1 for C-H stretch of the aromatic group disappear, O-H streaching in 2553 cm-1 disappearand Aromatic C=C peaks occur in the range 1579 cm-1 also faded due to MSL loss its aromaticity.
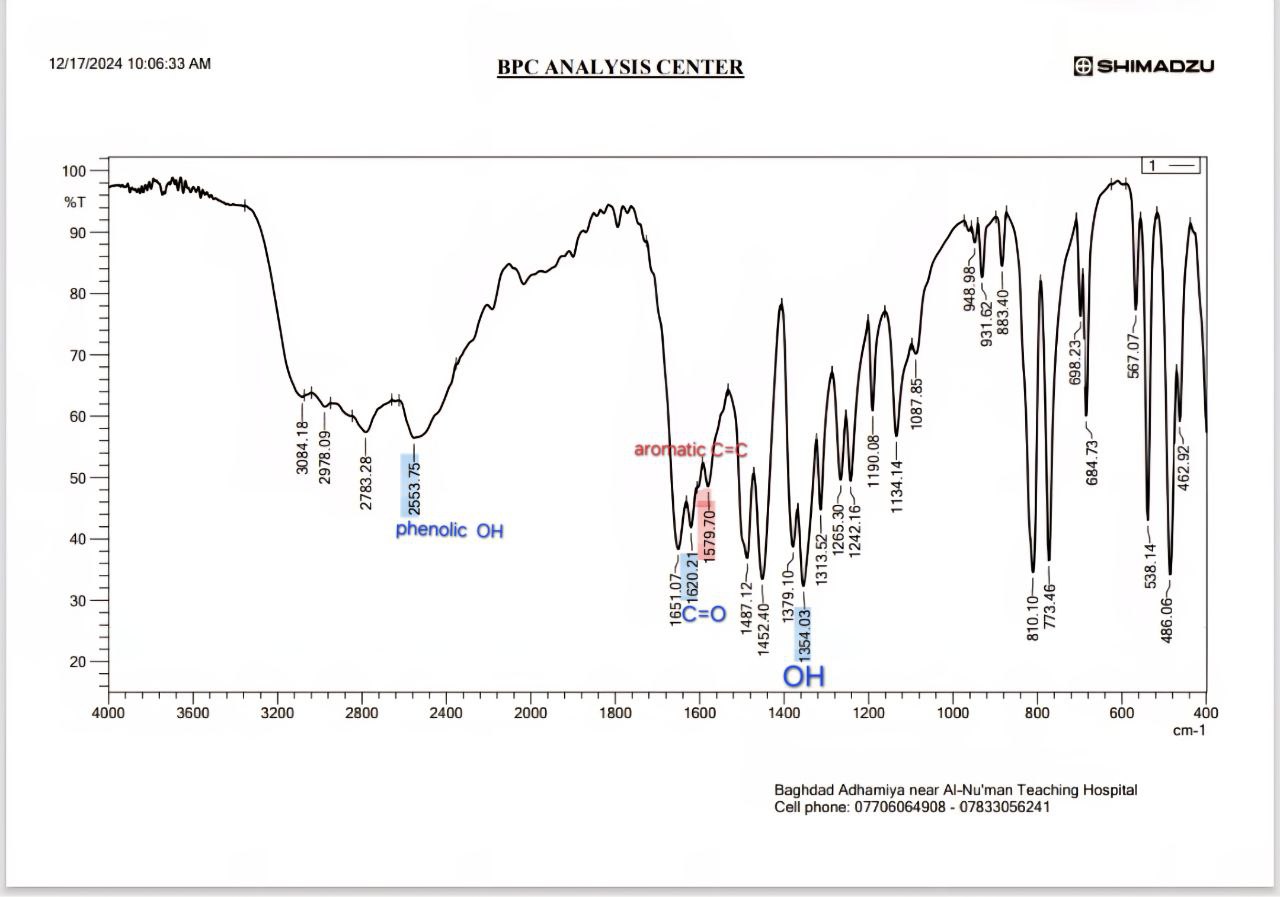
Fig. 14: Fourier transform infrared spectroscopy (FTIR) of pure MSL
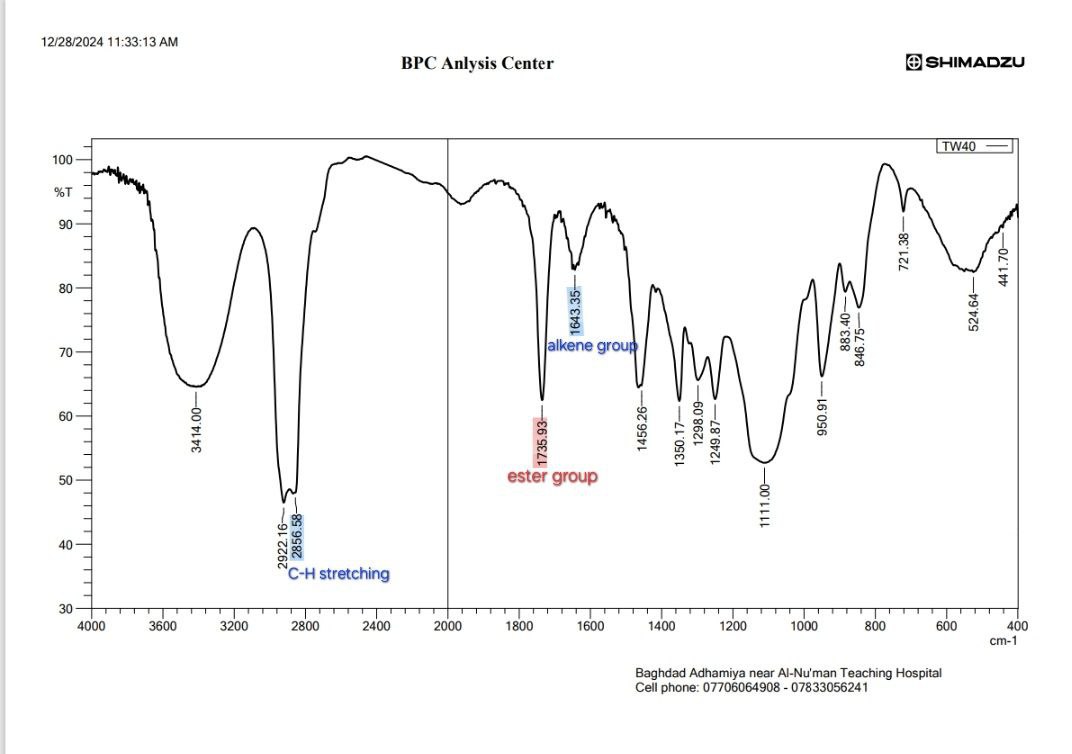
Fig. 15: Fourier transform infrared spectroscopy (FTIR) of tween 40
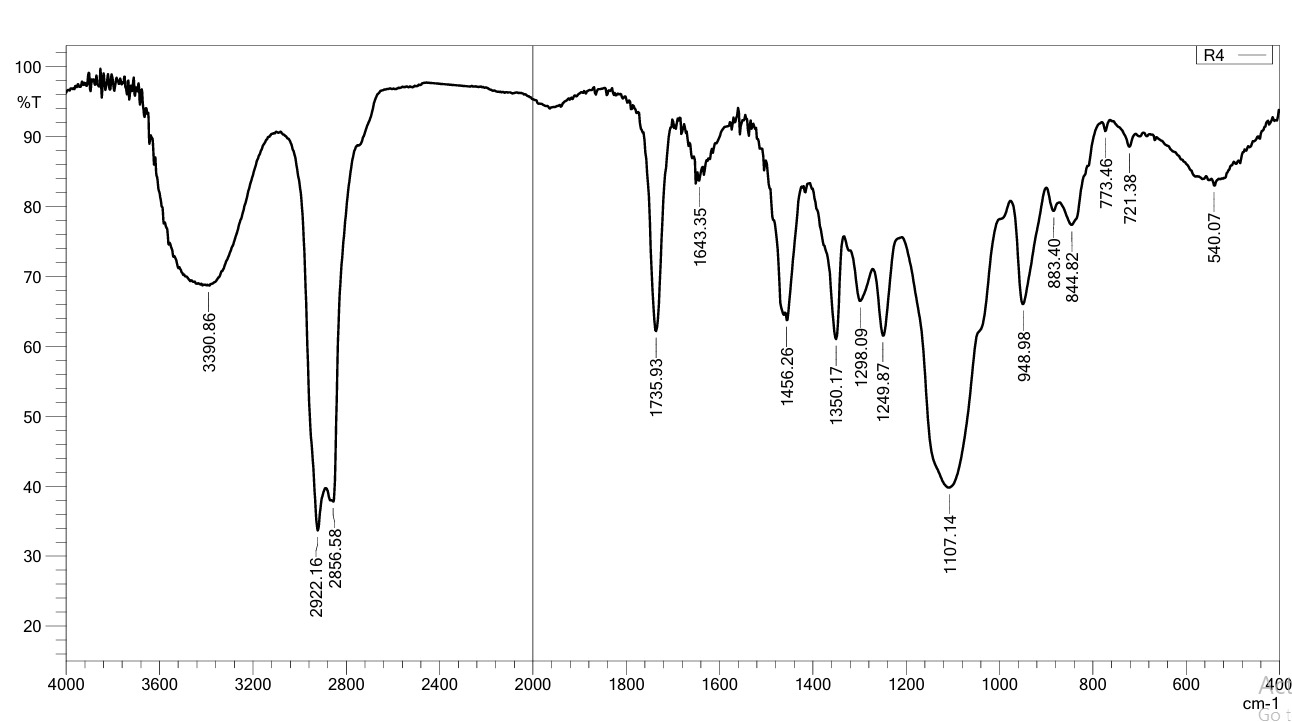
Fig. 16: Fourier transform infrared spectroscopy (FTIR) of MSL and tween 40 mixture
CONCLUSION
Mesalazine is the conventional treatment for inflammatory bowel disease. The pre-formulation studies facilitate the identification and confirmation of the drug's purity. In our research article, these studies include the characterization of the pure drug through UV spectrophotometry to determine lambda max and calibration curves in ethanol and phosphate buffer at pH 7.4, as well as the melting point obtained via DSC that accepted with previous study. The solubility analysis of MSL in various media which is show discoloration in surfactant and co-surfactant. An FTIR analysis showed on MSL in Tween 40 reveals a principal peak consistent with the reference. The FTIR spectrum of the MSL-Tween 40 combination demonstrates the absence of the primary peak of MSL, indicating a chemical interaction.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
Russul Hameed-Design and conceptualization of work, performed the work, analysed the data, DrMowafaq M. Ghareeb –Analysis of the data and manage of experimental work.
CONFLICT OF INTERESTS
Declared none
REFERENCES
Ahmed Elbashir A. Development and validation of spectrophotometric methods for the determination of mesalazine in pharmaceutical formulation. Med chem. 2014;4(3):361-6. doi: 10.4172/2161-0444.1000166.
Wani M, Rodge P, Baheti A, Polshettiwar S, Nandgude T, Tamboli F. Preformulation studies of glipizide: first step towards developing stable osmotic drug delivery system. Res J Pharm Technol. 2022;15(1):29-34. doi: 10.52711/0974-360X.2022.00006.
Jaitly A, Sharma R, Zafar R. Formulation and evaluation of cefuroxime axetil granules for oral suspension. Asian J Pharm Clin Res. 2023;16(7):180-6. doi: 10.22159/ajpcr.2023.v16i7.47397.
Chaurasia G. A review on pharmaceutical preformulation studies in formulation and development of new drug molecules. Int J Pharm Sci Res. 2016;7(6):2313–20. doi: 10.13040/IJPSR.0975-8232.7(6).2313-20.
Yadav AV, Yadav VB. Improvement of physicochemical properties of mesalamine with hydrophilic carriers by solid dispersion (kneading) method. Res J Pharm Technol. 2008;1(4):422-5.
Wadworth AN, Fitton A, Olsalazine. A review of its pharmacodynamic and pharmacokinetic properties and therapeutic potential in inflammatory bowel disease. Drugs. 1991;41(4):647-64. doi: 10.2165/00003495-199141040-00009, PMID 1711964.
Mazaher Haji Agha E, Barzegar Jalali M, Adibkia K, Hemmati S, Martinez F, Jouyban A. Solubility and thermodynamic properties of mesalazine in {2-propanol + water} mixtures at various temperatures. J Mol Liq. 2020;301:112474. doi: 10.1016/j.molliq.2020.112474.
Ye B, Van Langenberg DR. Mesalazine preparations for the treatment of ulcerative colitis: are all created equal? World J Gastrointest Pharmacol Ther. 2015;6(4):137-44. doi: 10.4292/wjgpt.v6.i4.137, PMID 26558148.
Al Obaidi MSM. Spectrophotometric determination of mesalazine by formation of ion pair complex. Eurasian Chem Commun. 2022 Feb;4(9):826-34. doi: 10.22034/ecc.2022.329317.1350.
Guler E, Poturcu K, Rahimpour E, Jouyban A. Determination of the mesalazine solubility at biorelevant temperature. IJAMSC. 2024;12(1):1-12. doi: 10.4236/ijamsc.2024.121001.
Thamer AK, Abood AN. Preparation and in vitro characterization of aceclofenac nanosuspension (ACNS) for enhancement of percutaneous absorption using hydrogel dosage form. Iraqi J Pharm Sci. 2021;30(2):86-98. doi: 10.31351/vol30iss2pp86-98.
Jassim ZE, Al Kinani KK, Alwan ZS. Preparation and evaluation of pharmaceutical cocrystals for solubility enhancement of dextromethorphan HBr. International Journal of Drug Delivery Technology. 2021; 11(4):1342–9. doi: 10.25258/ijddt.11.4.37.
Gupta K, Roy SB, Singhvi I. Preformulation study in the development of a tablet formulation for the treatment of ulcerative colitis. Int J Pharm Drug Anal. 2015;3(6):214-22.
Newton AM, Prabakaran L, Jayaveera KN. Formulation development, optimization and study on drug release kinetics of eudragit ® L100-HPMC E15 LV mixed film-coated colon-targeted Mesalamine tablets. Asian J Pharm. 2012;6(3):180-9. doi: 10.4103/0973-8398.104832.
Ghurghure S, Sawant K, Deokar SS. UV spectrophotometric method development and validation of mesalazine in bulk and solid dosage form (research article). Int J Pharm Biol Sci. 2021;11(1):131-5. doi: 10.21276/ijpbs.2021.11.1.17.
Kaur G, Chauhan V, Kumar K, Teotia D. Development and evaluation of mesalamine gastro-resistant tablets. J Adv Sci Res. 2019;10(4):106-16.
Aziz AT, Sultan SH. Spectrophotometric determination of mesalazine in pharmaceutical preparations by oxidative coupling reactions with m-aminophenol and 2,6-dihydroxybenzoic acid. Baghdad Sci J. 2019;16(4):1010-6. doi: 10.21123/bsj.2019.16.4(Suppl.).1010.
Salih E, Mohammed AE. Spectrophotometric assay of mesalazine in pharmaceutical preparations via oxidative coupling reaction with o-cresol and sodium metaperiodate. J Educ Sci. 2020;29(1):279-92. doi: 10.33899/edusj.2020.172815.
El Zein R, Ispas Szabo P, Jafari M, Siaj M, Mateescu MA. Oxidation of mesalamine under phenoloxidase- or peroxidase-like enzyme catalysis. Molecules. 2023;28(24):8105. doi: 10.3390/molecules28248105, PMID 38138595.
Dung NT, Dao DT, Hoat GD, Son NA. Development of spectrophotometric method for determination of ceftazidime with the Bratton–Marshall reagent in pharmaceutical preparation. JST. 2017;55(2):220. doi: 10.15625/0866-708X/55/2/8676.
Freeland JC. The determination of acidity in mineral oils. Johannesburg: Electricity Supply Commission; 1942. p. 49–52.
Gupta BS, Vallabhaneni MR. Electrochemical recognition and electrochemical oxidation of mesalazine drug at PMA modified glassy carbon electrode. Int J Pharm Investig. 2021;11(4):389-94. doi: 10.5530/ijpi.2021.4.70.
Jukic Spika M, Kraljic K, Skevin D. Tocopherols: chemical structure, bioactivity and variability in Croatian virgin olive oils. In: Boskou D, Clodoveo ML, editors. Products from Olive Tree. London (UK): InTech; 2016. p. 317–29. doi: 10.5772/64658.
Park NH, Manica M, Born J, Hedrick JL, Erdmann T, Zubarev DY. Artificial intelligence-driven design of catalysts and materials for ring-opening polymerization using a domain-specific language. Nat Commun. 2023;14(1):3686. doi: 10.1038/s41467-023-39396-3, PMID 37344485.
Kumar AA, Rani TS, Ganesh PS, Swamy BE. Electrochemical oxidation of masala zine drug at poly(glutamic acid) modified glassy carbon electrode. Anal Bioanal Electrochem. 2017;9(3):328-39.
Tan TC, Cheng LH, Bhat R, Rusul G, Easa AM. Effectiveness of ascorbic acid and sodium metabisulfite as anti-browning agent and antioxidant on green coconut water (Cocos nucifera) subjected to elevated thermal processing. Int Food Res J. 2015;22(2):631-7.
Lavoie JC, Lachance C, Chessex P. Antiperoxide activity of sodium metabisulfite a double-edged sword. Biochem Pharmacol. 1994;47(5):871-6. doi: 10.1016/0006-2952(94)90487-1, PMID 8135862.
Sharma M, Joshi B, Bansal M, Goswami M. Formulation and evaluation of colon-targeted tablets of mesalazine. J Drug Delivery Ther. 2012;2(5):24-36. doi: 10.22270/jddt.v2i5.290.
Alhadad KS, AL Salman HN. Chromatographic spectrophotometric determination using reverse-phase HPLC technique for mesalazine or mesalamine. Georgian Med News. 2023;342(342):58-65. PMID 37991958.
Alam MM, Tasneem F, Kabir AL, Rouf AS. Study of drug-drug and drug-food interactions of mesalazine through FTIR and DSC. Dhaka Univ J Pharm Sci. 2019;18(2):257-69. doi: 10.3329/dujps.v18i2.44466.
Simoes A, Ramos A, Domingues F, Luis A. Pullulan-tween 40 emulsified films containing geraniol: production and characterization as potential food packaging materials. Eur Food Res Technol. 2024;250(6):1721-32. doi: 10.1007/s00217-024-04514-y.