
Int J App Pharm, Vol 18, Issue 1, 2026, 421-431Original Article
GC-MS (GAS CHROMATOGRAPHY-MASS SPECTROMETRY) ANALYSIS SEED KEBIUL (CAESALPINIA BONDUC (L) ROXB)
DENSI SELPIA SOPIANTI1,6*, MUCHSIN DOEWES1,2, TATAR SUMANDJAR1,3 , ETI PONCORINI PAMUNGKASARI1,4, PARAMASARI DIRGAHAYU1,5, RATIH PUSPITA FEBRINASARI1,2
, ETI PONCORINI PAMUNGKASARI1,4, PARAMASARI DIRGAHAYU1,5, RATIH PUSPITA FEBRINASARI1,2
1*Doctoral Program of Medical Sciences, Faculty of Medicine, Universitas Sebelas Maret, Surakarta, Indonesia. Jalan Ir. Sutami 36A Ir. Sutami 36A Surakarta Jawa Tengah Indonesia 57126,, Indonesia. 3Department of Internal Medicine, Faculty of Medicine, Universitas Sebelas Maret, Surakarta, Indonesia. 4Department of Public Health, Faculty of Medicine, Universitas Sebelas Maret, Surakarta, Indonesia. 5Department of Parasitology and Mycology, Faculty of Medicine, Universitas Sebelas Maret, Surakarta, Indonesia. 6Department of Pharmacy, Sekolah Tinggi Kesehatan Al-Fatah, Bengkulu, Indonesia
*Corresponding author: Densi Selpia Sopianti; *Email: dselpias@gmail.com
Received: 16 Jun 2025, Revised and Accepted: 15 Oct 2025
ABSTRACT
Objective: Kebiul (Caesalpinia bonduc (L) Roxb) is one of the medicinal plants that plays an important role in treating various diseases, but this plant is vulnerable to extinction. This study aims to explore the possibility of various biological compounds in the ethanol extract of the seed kernel and seed coat of kebiul using gas chromatography-mass spectrometry (GC-MS) analysis.
Methods: The seed coat was removed from the kernel of the kebiul seed. After being ground into a powder, the seed kernel and seed coat were dried in an oven set to 40 °C for 1 h, and grinding them into a fine powder. The powder was soaked in 70% ethanol at a 1:10 (w/v) ratio for 3 days at room temperature, with occasional stirring. The chemical composition of secondary metabolite in the ethanol extracts of both the seed kernel and seed coat of kebiul was analyzed using gas chromatography–mass spectrometry (GC-MS) instrumentation (ID ISQD1702517_1) at a temperature of 25.30 °C and humidity of 35%.
Results: In this GC-MS analysis, 87 bioactive phytochemical compounds were identified in the ethanol extract of seed kernel and 50 bioactive phytochemical compounds were identified in the ethanol extract of seed coat. Seven chemicals in the seed kernel ethanol extract obtained the highest relative peak area (>2%) from each chromatogram, with the highest compound at a retention time of 19.93 min, namely 6-Octadecenoic acid, and three compounds in the seed coat ethanol extract obtained the highest relative peak area (>2%) in this investigation, with the highest compound at a retention time of 19.65 min, namely 9,12-Octadecadienoic acid (Z,Z)-.
Conclusion: GC-MS analysis identified 87 phytochemical compounds were identified in the ethanol extract of kebiul seed kernel and 50 phytochemical compounds were identified in the ethanol extract of kebiul seed coat.
Keywords: Active compound, Caesalpinia bonduc (L) Roxb, GC-MS analysis
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.55764 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
In traditional medicine, various parts of the plant are consumed alone and formulated as polyherbs, along with other medicinal plants, which are further used to treat various diseases [1]. Kebiul seeds are bulging oblong pods measuring 5 to 7.5 cm, covered with spines, and containing one or two seeds per pod. The pods have short stems, and the seeds are 1-2 cm in size, round, hard, and bluish-gray in color with a smooth shiny surface [2]. All parts of the plant (roots, leaves, seeds, bark, and stem) have been used as herbal medicine for various diseases such as antipyretic, antiperiodic, anthelmintic, antibacterial, hydrocele, antioxidant, antitumor, and antidiabetic and also used for the treatment of skin diseases such as leprosy, as well as used for the treatment of paralysis and nervous complaints [2-4].
Medicinal plants are more prone to extinction [5]. However, the ability to regenerate entire plants from epicotyl explants using callus induction and shoot regeneration methods has been demonstrated. This provides an efficient and reproducible protocol for the regeneration of ethnomedicinal plants, which is important for in vitro propagation and large-scale species conservation [6]. Therefore, identifying the active ingredients in traditional medicinal plants used as therapeutic drugs is necessary for scientific validation purposes [7]. Gas Chromatography-Mass Spectrometry (GC-MS) has become a popular technique to identify volatile and non-volatile compounds in plant species. Many previous studies found phytochemical compounds in plant components using the GC-MS method [8]. There is currently relatively little scientific data on the characterization of chemicals found in seed kernels and seed coat kebiul. This study is an early attempt to use the GC-MS method to determine the secondary metabolite profile, which can reveal additional details about the distribution of bioactive chemicals in both seed sections. The main objective of this study is to identify the biological compounds present in the seed kernel and seed coat of kebiul, which are extracted using 70% ethanol and analyzed with the GC-MS method, allowing for their potential to be assessed through in vitro and in vivo tests aimed at discovering new compounds that may have therapeutic applications. Using different methods and solvents is expected to produce different compounds and concentrations.
MATERIALS AND METHODS
Sample preparation
Kebiul seeds from woods on Sumatra Island, Indonesia's Bengkulu province, served as the samples. At the General Hospital's Tawangmangu Traditional Health Service Functional Service Unit, tests for plant determination were conducted. Test report number TL.02.04/D. XI.6/22566.1061/2024, with synonym Caesalpinia bonduc (L) Roxb, was submitted by Dr. Sarjito of Karanganyar Regency, Central Java. After being separated into seed kernels and seed coats, the sample was ground into a powder. The seed coat was removed from the kernel of the kebiul seed. After being ground into a powder, the seed kernel and seed coat were dried in an oven set to 40 °C for 1 h, and grinding them into a fine powder. The powder was soaked in 70% ethanol at a 1:10 (w/v) ratio for 3 days at room temperature, with occasional stirring. After maceration, the mixture was filtered using whatman No. 1 filter paper. The residue was re-macerated twice under the same conditions. All filtrates were pooled and evaporated under reduced pressure at 40 °C using a rotary evaporator. The resulting extract was then stored in a sealed container at 4 °C for subsequent analysis.
Instrument GC-MS
This research is an exploratory study using ethanol extract from the seed kernel and seed coat of kebiul. The chemical composition of secondary metabolite in the ethanol extracts of both the seed kernel and seed coat of kebiul was analyzed using gas chromatography–mass spectrometry (GC-MS) instrumentation (ID ISQD1702517_1) at a temperature of 25.30 °C and humidity of 35%. The analysis was performed using a capillary column HP-5MS (30 m length × 0.25 mm internal diameter × 0.25 µm film thickness). Helium was used as the carrier gas at a constant flow rate of 1.0 ml/min. The injection volume was 1 µl** with a split ratio of 10:1. The GC oven temperature was programmed as follows: initial temperature 50 °C (held for 2 min), ramped to 280 °C at 10 °C/min, and held at 280 °C for 10 min. GC-MS analysis was conducted using electron ionization at 70 eV, scanning m/z values from 40 to 600. Detector and injector were maintained at 280 and 250 °C, respectively. Then, the compound identification was performed by comparing retention indices and mass spectra with entries in the National Institute of Standards and Technology (NIST) library database, as described [9].
RESULTS
By employing the maceration method to produce 70% ethanol extract of the seed kernel and seed coat of kebiul, 29.99 g of ethanol extract of the seed kernel with a yield of 2.14% and 26.81 g of ethanol extract of the seed coat of kebiul with a yield of 2.6% were obtained. The chromatogram of the kebiul seed kernel, along with its corresponding retention periods and peaks, was obtained by GC-MS analysis and is shown in fig. 2 and table 1.
a |
b |
c |
Fig. 1: Preparation of seed kernel and seed coat of kebiul. a. seeds, b. seed coat, c. seed kernel of kebiul
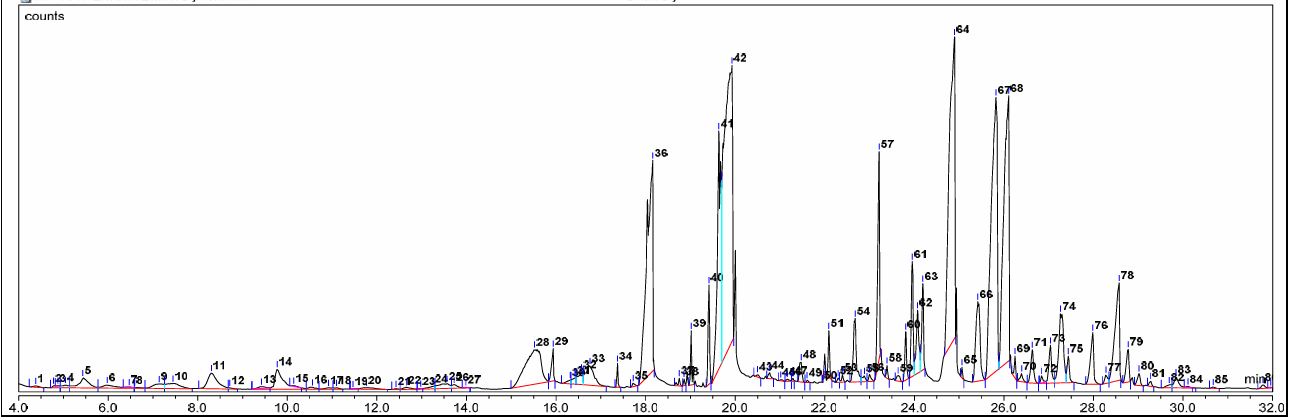
Fig. 2: Chromatogram of ethanol extract of kebiul seed kernel by GC-MS
Fig. 2 shows the chromatogram identified in the ethanol extract of the seed kernel. Individual compounds were identified by comparing their mass spectra database with the main library alliance, ensuring the listed spectra had a Similarity Index (SI) of more than 80% (NIST), and by comparing them with values published in the literature. The GC-MS analysis indicated that 87 phytochemical compounds were identified in the ethanol extract of seed kernels (tables 1 and 2).
Table 1: Compound of ethanol extract of kebiul seed kernel by GC-MS
| No. | Ret. time min |
Hit# 1 | Chemical formula | Mol. weight | SI Hit#1 | Rel. area % | Area counts*min |
| 1 | 4,38 | Sec-Butyl nitrite | C4H9NO2 | 103 | 652 | 0,05 | 12356481,038 |
| 2 | 4,78 | 2-Nitro-1-buten-3-ol | C4H7NO3 | 117 | 686 | 0,09 | 21175346,480 |
| 3 | 4,87 | Isoxazolidine-3,5-dicarboxylic acid, dimethyl ester | C7H11NO5 | 189 | 678 | 0,01 | 1435204,059 |
| 4 | 5,05 | N-Isopentyl-N-nitroso-pentylamine | C10H22N2O | 186 | 664 | 0,13 | 30663445,304 |
| 5 | 5,43 | Propanoic acid, 3-ethoxy-, ethyl ester | C7H14O3 | 146 | 736 | 0,69 | 166938220,038 |
| 6 | 5,98 | Alpha-l-rhamnopyranose | C6H12O5 | 164 | 730 | 0,32 | 77240815,627 |
| 7 | 6,47 | d-Glycero-d-ido-heptose | C7H14O7 | 210 | 663 | 0,02 | 4290477,110 |
| 8 | 6,59 | 2-Deoxy-D-galactose | C6H12O5 | 164 | 694 | 0,04 | 8648154,764 |
| 9 | 7,14 | Isosorbide Dinitrate | C6H8N2O8 | 236 | 706 | 0,33 | 80890210,831 |
| 10 | 7,45 | Glycerin | C3H8O3 | 92 | 739 | 0,49 | 118153069,053 |
| 11 | 8,30 | 4H-Pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl- | C6H8O4 | 144 | 842 | 1,05 | 254430558,682 |
| 12 | 8,71 | a-D-Galactopyranose, 2-(acetylamino)-2-deoxy- | C8H15NO6 | 221 | 694 | 0,03 | 7774744,820 |
| 13 | 9,42 | Isosorbide Dinitrate | C6H8N2O8 | 236 | 747 | 0,09 | 22094672,577 |
| 14 | 9,77 | 5-Hydroxymethylfurfural | C6H6O3 | 126 | 853 | 1,29 | 314249191,294 |
| 15 | 10,15 | 6-Acetyl-ß-d-mannose | C8H14O7 | 222 | 710 | 0,03 | 8442761,233 |
| 16 | 10,56 | Ascaridole epoxide | C10H16O3 | 184 | 733 | 0,09 | 21494313,997 |
| 17 | 10,92 | l-Gala-l-ido-octonic lactone | C8H14O8 | 238 | 712 | 0,07 | 16405387,642 |
| 18 | 11,10 | Dithiocarbamate, S-methyl-,N-(2-methyl-3-oxobutyl)- | C7H13NOS2 | 191 | 732 | 0,05 | 11323892,739 |
| 19 | 11,47 | Melezitose | C18H32O16 | 504 | 787 | 0,00 | 846197,660 |
| 20 | 11,77 | Melezitose | C18H32O16 | 504 | 787 | 0,15 | 36047631,307 |
| 21 | 12,43 | 1-Hexadecanol, 2-methyl- | C17H36O | 256 | 746 | 0,02 | 5128618,693 |
| 22 | 12,66 | Melezitose | C18H32O16 | 504 | 753 | 0,09 | 22299600,463 |
| 23 | 12,98 | Melezitose | C18H32O16 | 504 | 740 | 0,00 | 1121755,970 |
| 24 | 13,25 | Desulphosinigrin | C10H17NO6S | 279 | 745 | 0,11 | 26612156,948 |
| 25 | 13,56 | Dodecanoic acid, 3-hydroxy- | C12H24O3 | 216 | 740 | 0,38 | 91361996,381 |
| 26 | 13,72 | Melezitose | C18H32O16 | 504 | 741 | 0,14 | 33739231,949 |
| 27 | 13,99 | 2-Myristynoyl pantetheine | C25H44N2O5S | 484 | 775 | 0,01 | 2514248,128 |
| 28 | 15,52 | Ethyl a-d-glucopyranoside | C8H16O6 | 208 | 850 | 3,79 | 919183671,051 |
| 29 | 15,94 | Tetradecanoic acid | C14H28O2 | 228 | 874 | 0,42 | 102582161,109 |
| 30 | 16,32 | 3-O-Methyl-d-glucose | C7H14O6 | 194 | 761 | 0,10 | 23927031,993 |
| 31 | 16,42 | 3-O-Methyl-d-glucose | C7H14O6 | 194 | 771 | 0,16 | 39138063,552 |
| 32 | 16,57 | 3-O-Methyl-d-glucose | C7H14O6 | 194 | 766 | 0,41 | 100239213,835 |
| 33 | 16,76 | 3-O-Methyl-d-glucose | C7H14O6 | 194 | 677 | 1,14 | 276170844,873 |
| 34 | 17,37 | Hexadecanoic acid, methyl ester | C17H34O2 | 270 | 900 | 0,17 | 41806131,566 |
| 35 | 17,72 | 9-Hexadecenoic acid | C16H30O2 | 254 | 841 | 0,06 | 14857838,310 |
| 36 | 18,16 | l-(+)-Ascorbic acid 2,6-dihexadecanoate | C38H68O8 | 652 | 856 | 8,97 | 2177601507,849 |
| 37 | 18,75 | Ethanol, 2-(9-octadecenyloxy)-, (Z)- | C20H40O2 | 312 | 850 | 0,10 | 25223422,168 |
| 38 | 18,86 | n-Hexadecanoic acid | C16H32O2 | 256 | 817 | 0,05 | 13348651,915 |
| 39 | 19,02 | 9,12-Octadecadienoic acid (Z,Z)-, methyl ester | C19H34O2 | 294 | 923 | 0,47 | 113340275,316 |
| 40 | 19,41 | Androsta-4,16-dien-3-one | C19H26O | 270 | 714 | 0,99 | 240011900,417 |
| 41 | 19,64 | 9,12-Octadecadienoic acid, ethyl ester | C20H36O2 | 308 | 886 | 5,86 | 1422586259,826 |
| 42 | 19,93 | 6-Octadecenoic acid | C18H34O2 | 282 | 877 | 16,28 | 3951600262,929 |
| 43 | 20,50 | 1-Heptatriacotanol | C37H76O | 536 | 817 | 0,04 | 9685050,657 |
| 44 | 20,77 | 1-Heptatriacotanol | C37H76O | 536 | 825 | 0,09 | 22874119,034 |
| 45 | 21,02 | Cyclopropanebutanoic acid, 2-[[2-[[2-[(2-pentylcyclopropyl)methyl]cyclopropyl]methyl]cyclopropyl]methyl]-, methyl ester | C25H42O2 | 374 | 813 | 0,03 | 6862248,149 |
| 46 | 21,16 | 1-Heptatriacotanol | C37H76O | 536 | 811 | 0,05 | 11870633,118 |
| 47 | 21,29 | 1-Heptatriacotanol | C37H76O | 536 | 784 | 0,03 | 6601142,798 |
| 48 | 21,47 | 4,8,13-Cyclotetradecatriene-1,3-diol, 1,5,9-trimethyl-12-(1-methylethyl)- | C20H34O2 | 306 | 780 | 0,37 | 89915392,029 |
| 49 | 21,60 | 1-Heptatriacotanol | C37H76O | 536 | 815 | 0,03 | 6871881,498 |
| 50 | 21,94 | 1-Heptatriacotanol | C37H76O | 536 | 811 | 0,03 | 7184095,749 |
| 51 | 22,09 | 2-Phenyl-4-tret-butyl-7-methylindene | C20H22 | 262 | 746 | 0,36 | 88592175,622 |
| 52 | 22,30 | 1-Heptatriacotanol | C37H76O | 536 | 830 | 0,06 | 13499608,111 |
| 53 | 22,40 | Curan, 16,17,19,20-tetradehydro- | C19H22N2 | 278 | 664 | 0,09 | 21530222,532 |
| 54 | 22,68 | 2,4,6-Tri-t-butylbenzenethiol | C18H30S | 278 | 679 | 1,35 | 327352977,751 |
| 55 | 22,89 | 1-Heptatriacotanol | C37H76O | 536 | 756 | 0,14 | 33836126,080 |
| 56 | 23,00 | 1-Heptatriacotanol | C37H76O | 536 | 807 | 0,11 | 27317859,989 |
| 57 | 23,21 | Pregna-5,17(20)-dien-3-ol, (3ß,17E)- | C21H32O | 300 | 728 | 2,52 | 611086882,463 |
| 58 | 23,39 | Hexadecanoic acid, 1-(hydroxymethyl)-1,2-ethanediyl ester | C35H68O5 | 568 | 765 | 0,10 | 23951481,918 |
| 59 | 23,64 | 1-Heptatriacotanol | C37H76O | 536 | 794 | 0,13 | 31028509,408 |
| 60 | 23,81 | Gibbane-1,10-dicarboxylic acid, 4a,7-dihydroxy-1-methyl-8-methylene-, 1,4a-lactone, 10-methyl ester, (1a,4aa,4bß,10ß)- | C20H26O5 | 346 | 678 | 0,58 | 139667652,116 |
| 61 | 23,95 | 1,4,9(11)-Pregnatriene-3,20-dione, 21-acetoxy-17-hydroxy- | C23H28O5 | 384 | 668 | 1,51 | 367737480,175 |
| 62 | 24,07 | 1-Heptatriacotanol | C37H76O | 536 | 701 | 1,14 | 275818489,090 |
| 63 | 24,19 | 17Alpha-ethynyl-17beta-hydroxy-6beta-methoxy-3alpha,5-cyclo-5alpha-androstan-19-oic acid | C22H30O4 | 358 | 701 | 1,03 | 251033153,944 |
| 64 | 24,90 | Pregnenolone | C21H32O2 | 316 | 720 | 11,77 | 2858353832,214 |
| 65 | 25,06 | 6ß-Hydroxymethandienone | C20H28O3 | 316 | 707 | 0,07 | 18100793,977 |
| 66 | 25,42 | Norethindrone Acetate | C22H28O3 | 340 | 730 | 2,36 | 572240692,503 |
| 67 | 25,82 | 1H-Cyclopropa[3,4]benz[1,2-e]azulene-5,7b,9,9a-tetrol, 3-[(acetyloxy)methyl]-1a,1b,4,4a,5,7a,8,9-octahydro-1,1,6,8-tetramethyl-, 5,9,9a-triacetate, [1aR-(1aa,1bß,4aß,5ß,7aa,7ba,8a,9ß,9aa)]- | C28H38O9 | 518 | 662 | 11,21 | 2720558837,592 |
| 68 | 26,11 | 1H-Cyclopropa[3,4]benz[1,2-e]azulene-5,7b,9,9a-tetrol, 3-[(acetyloxy)methyl]-1a,1b,4,4a,5,7a,8,9-octahydro-1,1,6,8-tetramethyl-, 5,9,9a-triacetate, [1aR-(1aa,1bß,4aß,5ß,7aa,7ba,8a,9ß,9aa)]- | C28H38O9 | 518 | 722 | 9,58 | 2324559456,196 |
| 69 | 26,25 | 1H-Cyclopropa[3,4]benz[1,2-e]azulene-5,7b,9,9a-tetrol, 3-[(acetyloxy)methyl]-1a,1b,4,4a,5,7a,8,9-octahydro-1,1,6,8-tetramethyl-, 5,9,9a-triacetate, [1aR-(1aa,1bß,4aß,5ß,7aa,7ba,8a,9ß,9aa)]- | C28H38O9 | 518 | 681 | 0,21 | 50305606,222 |
| 70 | 26,40 | 5,16,20-Pregnatriene-3beta,20-diol diacetate | C25H34O4 | 398 | 756 | 0,16 | 37891353,402 |
| 71 | 26,63 | 1H-Cyclopropa[3,4]benz[1,2-e]azulene-5,7b,9,9a-tetrol, 1a,1b,4,4a,5,7a,8,9-octahydro-3-(hydroxymethyl)-1,1,6,8-tetramethyl-, 5,9,9a-triacetate, [1aR-(1aa,1bß,4aß,5ß,7aa,7ba,8a,9ß,9aa)]- | C26H36O8 | 476 | 758 | 0,65 | 157287190,038 |
| 72 | 26,84 | 1H-Cyclopropa[3,4]benz[1,2-e]azulene-5,7b,9,9a-tetrol, 3-[(acetyloxy)methyl]-1a,1b,4,4a,5,7a,8,9-octahydro-1,1,6,8-tetramethyl-, 5,9,9a-triacetate, [1aR-(1aa,1bß,4aß,5ß,7aa,7ba,8a,9ß,9aa)]- | C28H38O9 | 518 | 711 | 0,10 | 25002936,711 |
| 73 | 27,04 | 1H-Cyclopropa[3,4]benz[1,2-e]azulene-5,7b,9,9a-tetrol, 3-[(acetyloxy)methyl]-1a,1b,4,4a,5,7a,8,9-octahydro-1,1,6,8-tetramethyl-, 5,9,9a-triacetate, [1aR-(1aa,1bß,4aß,5ß,7aa,7ba,8a,9ß,9aa)]- | C28H38O9 | 518 | 746 | 0,74 | 180109102,256 |
| 74 | 27,27 | 1H-Cyclopropa[3,4]benz[1,2-e]azulene-5,7b,9,9a-tetrol, 1a,1b,4,4a,5,7a,8,9-octahydro-3-(hydroxymethyl)-1,1,6,8-tetramethyl-, 5,9,9a-triacetate, [1aR-(1aa,1bß,4aß,5ß,7aa,7ba,8a,9ß,9aa)]- | C26H36O8 | 476 | 784 | 2,32 | 563669675,482 |
| 75 | 27,44 | 4,13,20-Tri-O-methylphorbol 12-acetate | C25H36O7 | 448 | 762 | 0,42 | 101842917,656 |
| 76 | 27,99 | Phorbol 12,13,20-triacetate | C26H34O9 | 490 | 747 | 1,15 | 278563075,943 |
| 77 | 28,28 | Acetic acid, 17-(1-acetoxy-ethyl)-10,13-dimethyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-11-yl (ester) | C25H34O5 | 414 | 721 | 0,18 | 42548366,987 |
| 78 | 28,57 | 2-Cyclopenten-1-one, 3,4-dihydroxy-5-(3-methyl-2-butenyl)-2-(3-methyl-1-oxobutyl)-4-(4-methyl-1-oxo-3-pentenyl)- | C21H30O5 | 362 | 650 | 3,00 | 729450089,319 |
| 79 | 28,77 | 3-Pyridinecarboxylic acid, 2,7,10-tris(acetyloxy)-1,1a,2,3,4,6,7,10,11,11a-decahydro-1,1,3,6,9-pentamethyl-4-oxo-4a,7a-epoxy-5H-cyclopenta[a]cyclopropa[f]cycloundecen-11-yl ester, [1aR-(1aR*,2R*,3S*,4aR*,6S*,7S*,7aS*,8E,10R*,11R*,11aS*)]- | C32H39NO10 | 597 | 698 | 0,61 | 149225718,612 |
| 80 | 29,02 | 1H-Cyclopropa[3,4]benz[1,2-e]azulene-5,7b,9,9a-tetrol, 1a,1b,4,4a,5,7a,8,9-octahydro-3-(hydroxymethyl)-1,1,6,8-tetramethyl-, 9,9a-diacetate, [1aR-(1aa,1bß,4aß,5ß,7aa,7ba,8a,9ß,9aa)]- | C24H34O7 | 434 | 696 | 0,23 | 56766218,653 |
| 81 | 29,27 | Acetic acid, 17-(1-acetoxy-ethyl)-10,13-dimethyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-11-yl (ester) | C25H34O5 | 414 | 796 | 0,08 | 19652808,230 |
| 82 | 29,69 | Ethyl iso-allocholate | C26H44O5 | 436 | 770 | 0,11 | 26385262,922 |
| 83 | 29,84 | Pregn-5-ene-3,11,12,14,20-pentol, 11-acetate 12-(3-methylbutanoate), (3ß,11a,12ß,14ß)- | C28H44O7 | 492 | 717 | 0,38 | 91647832,814 |
| 84 | 30,11 | Butanoic acid, 1a,2,5,5a,6,9,10,10a-octahydro-5,5a-dihydroxy-4-(hydroxymethyl)-1,1,7,9-tetramethyl-11-oxo-1H-2,8a-methanocyclopenta[a]cyclopropa[e]cyclodecen-6-yl ester, [1aR-(1aa,2a,5ß,5aß,6ß,8aa,9a,10aa)]- | C24H34O6 | 418 | 782 | 0,02 | 6034424,424 |
| 85 | 30,67 | Stigmasterol | C29H48O | 412 | 766 | 0,03 | 8062119,845 |
| 86 | 31,78 | ?-Sitosterol | C29H50O | 414 | 802 | 0,10 | 23362737,090 |
| 87 | 31,96 | 9,12,15-Octadecatrienoic acid, 2-phenyl-1,3-dioxan-5-yl ester | C28H40O4 | 440 | 717 | 0,01 | 1880362,172 |
Table 2 summarizes the seven compounds in the seed kernel ethanol extract with the highest relative peak areas in this investigation, whereas fig. 4-10 summarize the apex pecan compound and real-time.
| Ret. time | 19,635 | 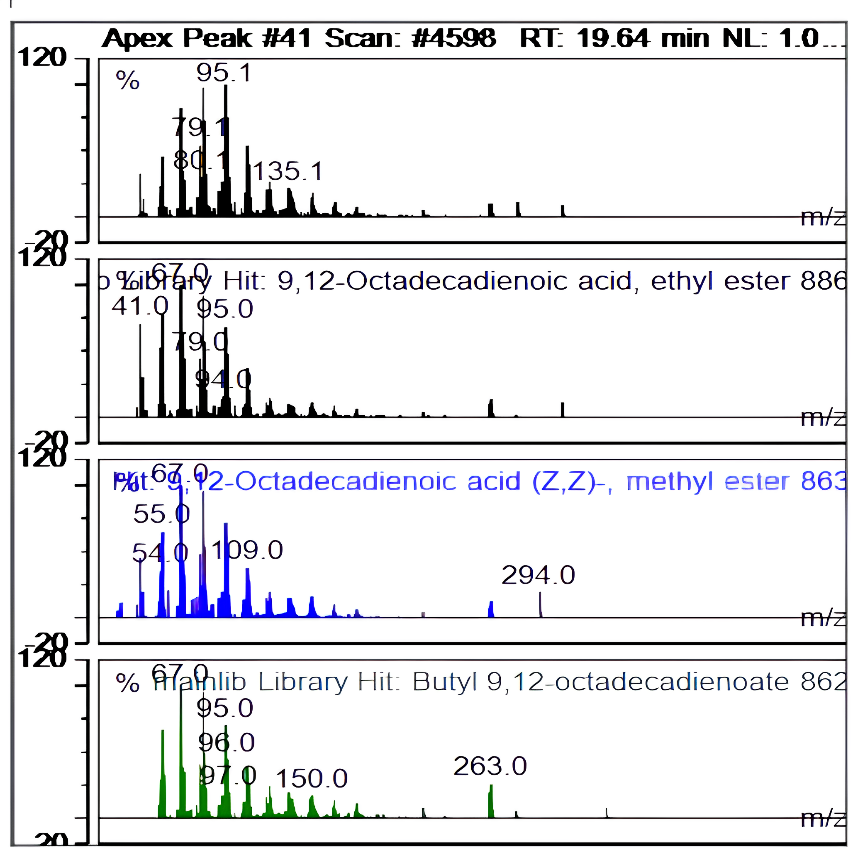 |
| Hit# 1 | ||
| Component: | 9,12-Octadecadienoic acid, ethyl ester | |
| SI: | 886 | |
| RSI: | 928 | |
| Library: | mainlib | |
| Hit# 2 | ||
| Component: | 9,12-Octadecadienoic acid (Z,Z)-, methyl ester | |
| SI: | 863 | |
| RSI: | 886 | |
| Library: | mainlib | |
| Hit# 3 | ||
| Component: | Butyl 9,12-octadecadienoate | |
| SI: | 862 | |
| RSI: | 882 |
Fig. 4: Apex peak 9,12-octadecadienoic acid, ethyl ester
Table 2: Compounds with various retention times, peak area percentages, molecular formulas, and molecular weights appeared after GC-MS analysis of the ethanol extract of kebiul seed kernel
| No | Name | RT* (Min) |
Peak area (%) | Molekuler formula | Molecular weight | Structure (2D PubChem) |
SI (NIST) |
| 1 | l-(+)-Ascorbic acid 2,6-dihexadecanoate | 18,16 | 8,97 | C38H68O8 | 652 |  |
856 |
| 2 | 9,12-Octadecadienoic acid, ethyl ester | 19,64 | 5,86 | C20H36O2 | 308 |  |
886 |
| 3 | 6-Octadecenoic acid | 19,93 | 16,28 | C18H34O2 | 282 | 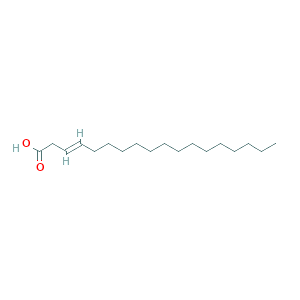 |
877 |
| 4 | Pregna-5,17(20)-dien-3-ol, (3ß,17E)- | 23,21 | 2,52 | C21H32O | 300 | 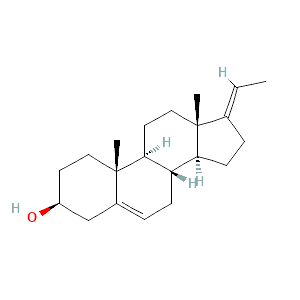 |
728 |
| 5 | Pregnenolone | 24,90 | 11,77 | C21H32O2 | 316 | 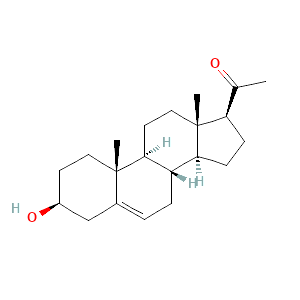 |
720 |
| 6 | 1H-Cyclopropa[3,4]benz[1,2-e]azulene-5,7b,9,9a-tetrol, 3-[(acetyloxy)methyl]- 1a,1b,4,4a,5,7a,8,9-octahydro-1,1,6,8-tetramethyl-, 5,9,9a-triacetate, [1aR-(1aa,1bß,4aß,5ß,7aa,7ba,8a,9ß,9aa)] |
25,82 | 11,21 | C28H38O9 | 518 | 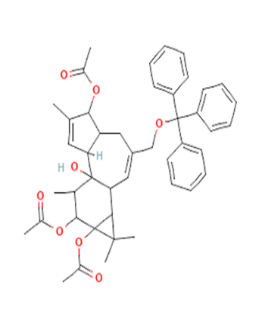 |
662 |
| 7 | 1H-Cyclopropa[3,4]benz[1,2-e]azulene-5,7b,9,9a-tetrol, 3- [(acetyloxy)methyl]-1a,1b,4,4a,5,7a,8,9-octahydro-1,1,6,8-tetramethyl-, 5,9,9a-triacetate, [1aR-(1aa,1bß,4aß,5ß,7aa,7ba,8a,9ß,9aa)]- |
26,11 | 9,58 | C28H38O9 | 518 | 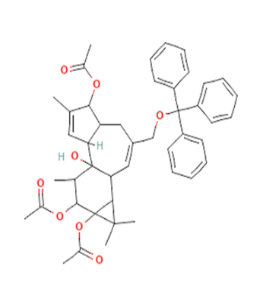 |
722 |
| Ret. Time: | 19,928 | 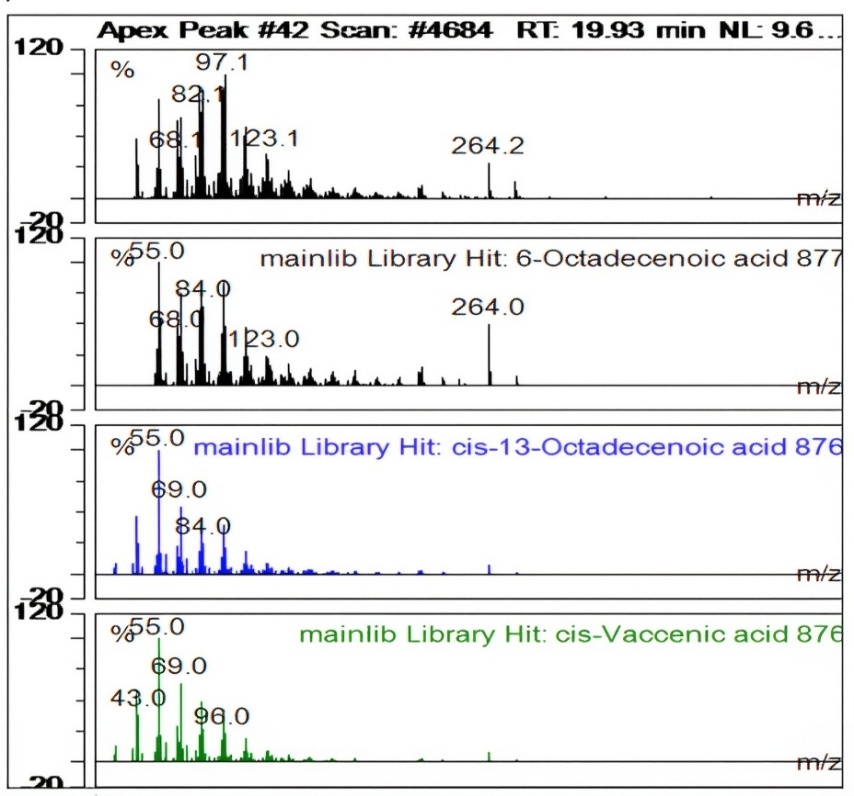 |
| Hit# 1 | ||
| Component: | 6-Octadecenoic acid | |
| SI: | 877 | |
| RSI: | 894 | |
| Library: | mainlib | |
| Hit# 2 | ||
| Component: | cis-13-Octadecenoic acid | |
| SI: | 876 | |
| RSI: | 880 | |
| Library: | mainlib | |
| Hit# 3 | ||
| Component: | cis-Vaccenic acid | |
| SI: | 876 | |
| RSI: | 879 |
Fig. 5: Apex peak 6-octadecenoic acid
| Ret. Time: | 18,159 | 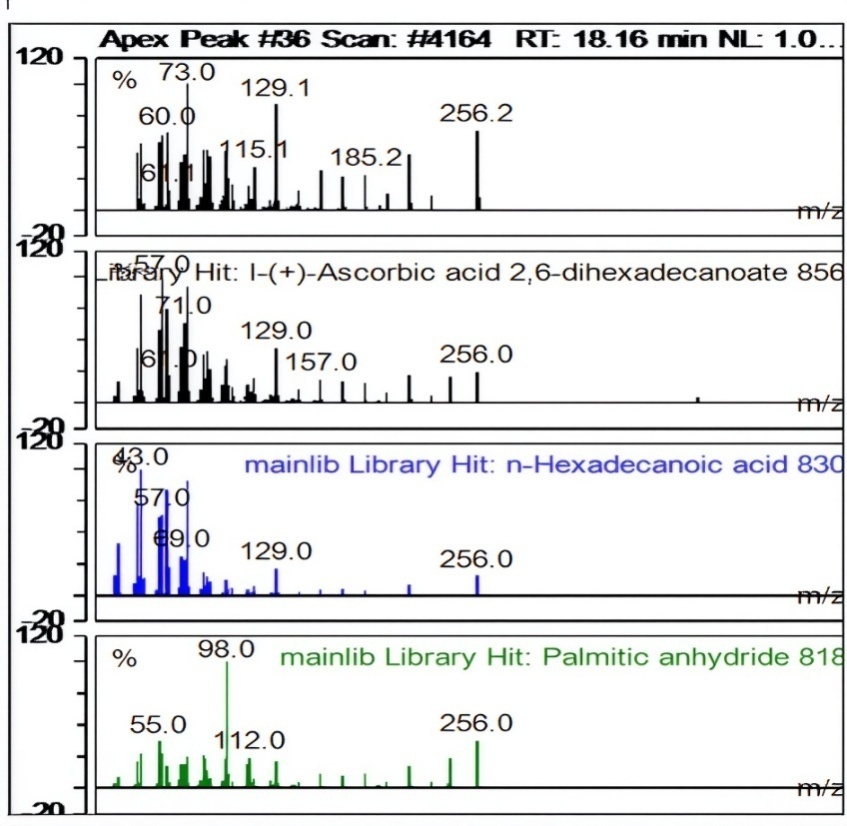 |
| Hit# 1 | ||
| Component: | l-(+)-Ascorbic acid 2,6-dihexadecanoate | |
| SI: | 856 | |
| RSI: | 856 | |
| Library: | mainlib | |
| Hit# 2 | ||
| Component: | n-Hexadecanoic acid | |
| SI: | 830 | |
| RSI: | 835 | |
| Library: | mainlib | |
| Hit# 3 | ||
| Component: | Palmitic anhydride | |
| SI: | 818 | |
| RSI: | 819 |
Fig. 6: Apex peak-(+)-ascorbic acid 2,6-dihexadecanoate
| Ret. Time: | 23,213 |  |
| Hit# 1 | ||
| Component: | Pregna-5,17(20)-dien-3-ol, (3ß,17E)- | |
| SI: | 728 | |
| RSI: | 758 | |
| Library: | mainlib | |
| Hit# 2 | ||
| Component: | Retinoic acid | |
| SI: | 715 | |
| RSI: | 735 | |
| Library: | mainlib | |
| Hit# 3 | ||
| Component: | Androst-4-en-11-ol-3,17-dione, 9-thiocyanato- | |
| SI: | 709 | |
| RSI: | 714 | |
| Library: | mainlib |
Fig. 7: Apex peak pregna-5,17(20)-dien-3-ol, (3ß,17E)-
| Ret. Time: | 26,108 | 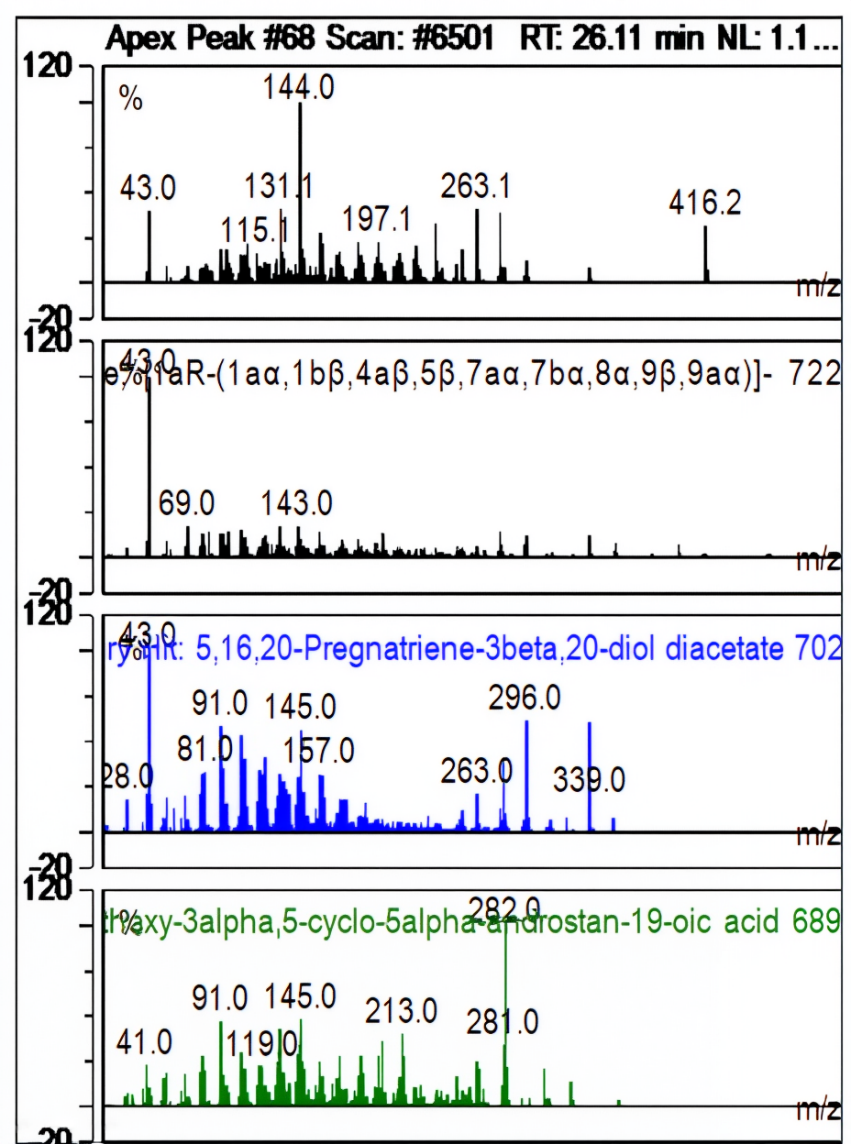 |
| Hit# 1 | ||
| Component: | 1H-Cyclopropa[3,4]benz[1,2-e]azulene-5,7b,9,9a-tetrol, 3-[(acetyloxy)methyl]-1a,1b,4,4a, 5,7a,8,9-octahydro-1,1,6,8-tetramethyl-, 5,9,9a-triacetate, [1aR-(1aa,1bß,4aß,5ß, 7aa,7ba,8a,9ß,9aa)]- | |
| SI: | 722 | |
| RSI: | 722 | |
| Library: | mainlib | |
| Hit# 2 | ||
| Component: | 5,16,20-Pregnatriene-3beta,20-diol diacetate | |
| SI: | 702 | |
| RSI: | 725 | |
| Library: | mainlib | |
| Hit# 3 | ||
| Component: | 17Alpha-ethynyl-17beta-hydroxy-6beta-methoxy-3alpha,5-cyclo-5alpha-androstan-19-oic acid | |
| SI: | 689 | |
| RSI: | 739 |
Fig. 8: Apek peak 1H-cyclopropa[3,4]benz[1,2-e]azulene-5,7b,9,9a-tetrol,3-[(acetyloxy)methyl]-1a,1b,4,4a,5, 7a,8,9-octahydro-1,1,6,8-tetramethyl-, 5,9,9a-triacetate, [1aR-(1aa,1bß,4aß,5ß,7aa,7ba,8a, 9ß,9aa)]-
| Ret. Time: | 24,897 | 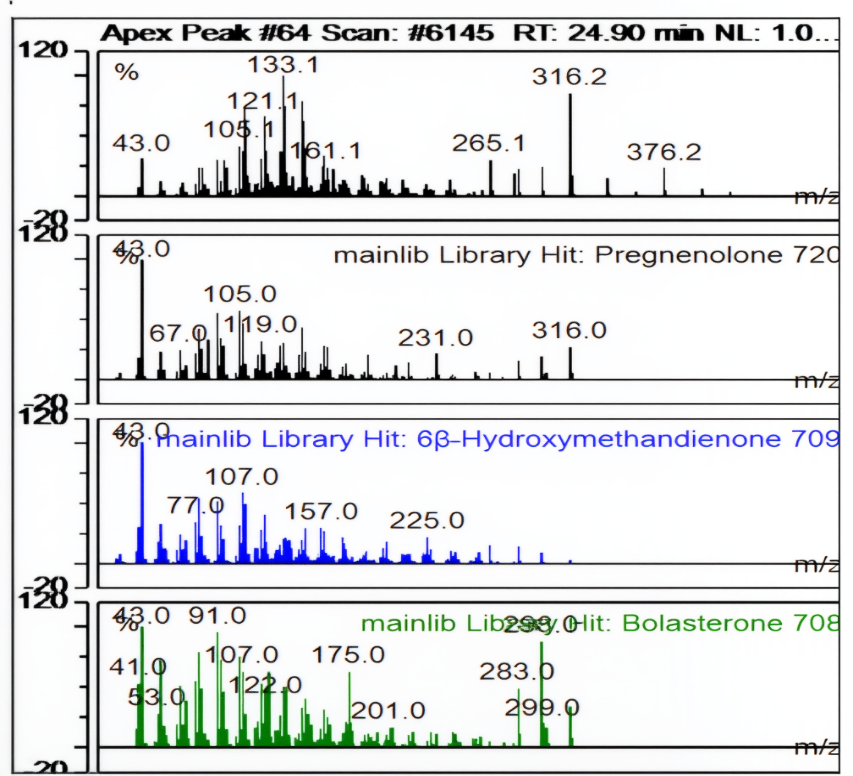 |
| Hit# 1 | ||
| Component: | Pregnenolone | |
| SI: | 720 | |
| RSI: | 750 | |
| Library: | mainlib | |
| Hit# 2 | ||
| Component: | 6ß-Hydroxymethandienone | |
| SI: | 709 | |
| RSI: | 747 | |
| Library: | mainlib | |
| Hit# 3 | ||
| Component: | Bolasterone | |
| SI: | 708 | |
| RSI: | 738 |
Fig. 9: Apek Peak pregnenolone
| Ret. Time: | 25,822 | 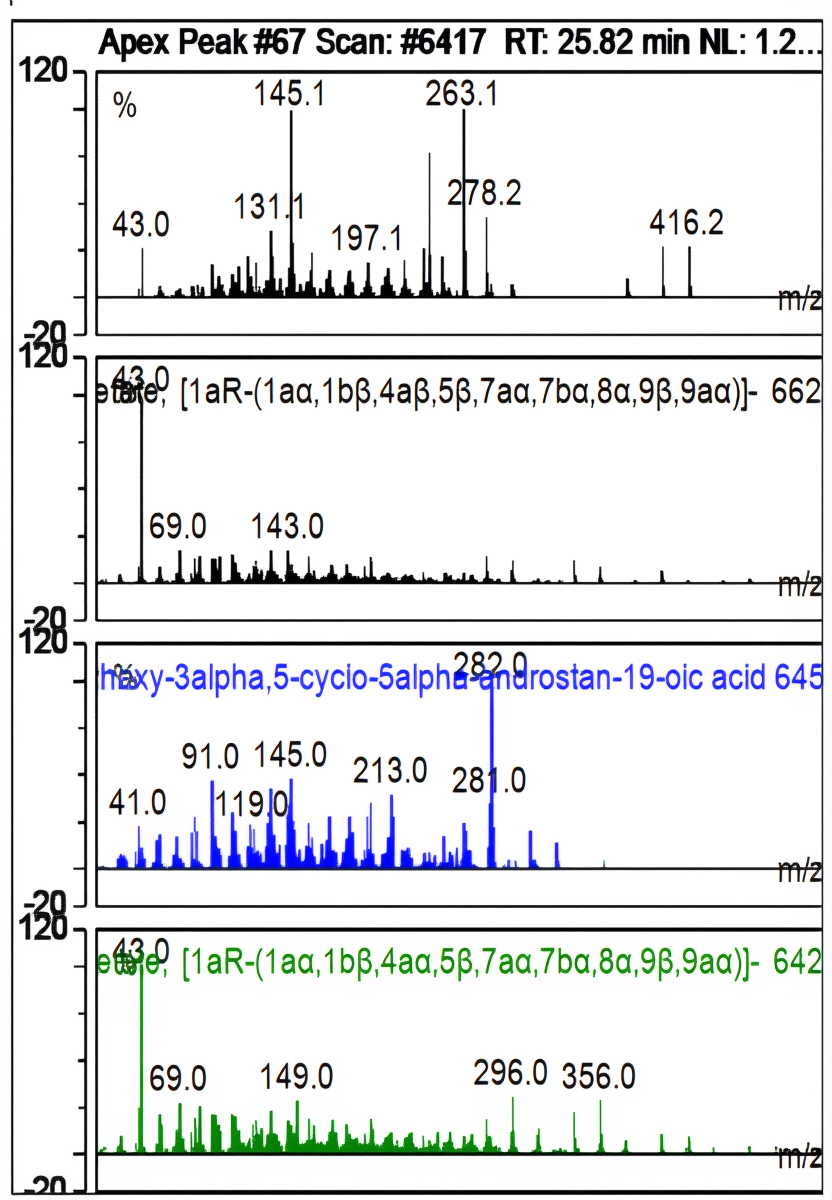 |
| Hit# 1 | ||
| Component: | 1H-Cyclopropa[3,4]benz[1,2-e]azulene-5,7b,9,9a-tetrol, 3-[(acetyloxy)methyl]-1a,1b,4,4a,5, 7a,8,9-octahydro-1,1,6,8-tetramethyl-, 5,9, 9a-triacetate, [1aR-(1aa,1bß, 4aß,5ß,7aa,7ba,8a,9ß,9aa)]- | |
| SI: | 662 | |
| RSI: | 663 | |
| Library: | mainlib | |
| Hit# 2 | ||
| Component: | 17Alpha-ethynyl-17beta-hydroxy-6beta-methoxy-3alpha,5-cyclo-5alpha-androstan-19-oic acid | |
| SI: | 645 | |
| RSI: | 704 | |
| Library: | mainlib | |
| Hit# 3 | ||
| Component: | 1H-Cyclopropa[3,4]benz[1,2-e]azulene-5,7b,9,9a-tetrol, 3-[(acetyloxy)methyl]-1a,1b,4,4a,5,7a,8,9-octahydro-1,1,6,8-tetramethyl-, 9,9a-diacetate, [1aR-(1aa,1bß, 4aa,5ß,7aa,7ba,8a,9ß,9aa)]- | |
| SI: | 642 | |
| RSI: | 642 | |
| Library: | mainlib |
Fig. 10: Apek Peak1H-Cyclopropa[3,4]benz[1,2-e]azulene-5,7b,9,9a-tetrol,3-[(acetyloxy)methyl] 1a,1b,4,4a,5,7a,8,9-octahydro-1,1,6,8-tetramethyl-,5,9,9a-triacetate, [1aR(1aa,1bß,4aß,5ß,7aa,7ba,8a,9ß,9aa)]-
Table 3: Chromatogram of ethanol extract of kebiul seed coat by GC-MS.
| No. | Ret. time min |
Hit# 1 | Chemical formula | Mol. weight | SI Hit#1 | Rel. area % |
Area counts*min |
| 1 | 4,40 | Butanoic acid, 4-butoxy- | C8H16O3 | 160 | 690 | 0,65 | 17149251,603 |
| 2 | 4,90 | 1-(3,3,3-Trifluoro-2-hydroxypropyl)piperidine | C8H14F3NO | 197 | 678 | 1,98 | 52600361,831 |
| 3 | 5,46 | Propanoic acid, 3-ethoxy-, ethyl ester | C7H14O3 | 146 | 809 | 2,92 | 77439842,012 |
| 4 | 6,15 | e-N-Formyl-L-lysine | C7H14N2O3 | 174 | 719 | 0,24 | 6348137,883 |
| 5 | 7,05 | DL-Arabinose | C5H10O5 | 150 | 665 | 2,72 | 72235304,066 |
| 6 | 7,52 | Glycerin | C3H8O3 | 92 | 834 | 5,19 | 137676830,908 |
| 7 | 7,97 | Isosorbide Dinitrate | C6H8N2O8 | 236 | 695 | 0,28 | 7424173,969 |
| 8 | 8,38 | Acetamide, N-(2-acetyl-3-oxo-4-isoxazolidinyl)- | C7H10N2O4 | 186 | 687 | 1,75 | 46451880,017 |
| 9 | 9,37 | 2-Myristynoyl pantetheine | C25H44N2O5S | 484 | 718 | 0,26 | 6865816,496 |
| 10 | 9,89 | 6-Acetyl-ß-d-mannose | C8H14O7 | 222 | 702 | 0,37 | 9807341,141 |
| 11 | 10,57 | Ascaridole epoxide | C10H16O3 | 184 | 720 | 0,20 | 5377155,199 |
| 12 | 11,10 | Z-(13,14-Epoxy)tetradec-11-en-1-ol acetate | C16H28O3 | 268 | 689 | 0,26 | 6789871,482 |
| 13 | 11,41 | d-Mannose | C6H12O6 | 180 | 765 | 0,31 | 8239081,820 |
| 14 | 12,56 | 1,2,3-Benzenetriol | C6H6O3 | 126 | 795 | 3,86 | 102304036,550 |
| 15 | 12,99 | Melezitose | C18H32O16 | 504 | 665 | 0,01 | 266193,201 |
| 16 | 13,40 | Melezitose | C18H32O16 | 504 | 747 | 1,07 | 28348426,477 |
| 17 | 13,58 | d-Gala-l-ido-octonic amide | C8H17NO8 | 255 | 719 | 0,04 | 933308,843 |
| 18 | 13,75 | Melezitose | C18H32O16 | 504 | 724 | 0,10 | 2628173,803 |
| 19 | 14,81 | Desulphosinigrin | C10H17NO6S | 279 | 718 | 0,46 | 12147900,801 |
| 20 | 15,02 | Melezitose | C18H32O16 | 504 | 751 | 1,94 | 51576313,489 |
| 21 | 15,28 | [1,1'-Bicyclopropyl]-2-octanoic acid, 2'-hexyl-, methyl ester | C21H38O2 | 322 | 695 | 2,44 | 64609697,162 |
| 22 | 15,89 | Desulphosinigrin | C10H17NO6S | 279 | 733 | 2,64 | 70056482,665 |
| 23 | 16,35 | 3-O-Methyl-d-glucose | C7H14O6 | 194 | 725 | 8,01 | 212444222,592 |
| 24 | 16,41 | 3-O-Methyl-d-glucose | C7H14O6 | 194 | 731 | 0,81 | 21420479,779 |
| 25 | 16,56 | 3-O-Methyl-d-glucose | C7H14O6 | 194 | 714 | 4,69 | 124442347,240 |
| 26 | 16,75 | 7-Methyl-Z-tetradecen-1-ol acetate | C17H32O2 | 268 | 707 | 1,02 | 27033533,752 |
| 27 | 16,90 | Dodecanoic acid, 2,3-bis(acetyloxy)propyl ester | C19H34O6 | 358 | 737 | 0,08 | 2119033,345 |
| 28 | 17,05 | Dodecanoic acid, 2,3-bis(acetyloxy)propyl ester | C19H34O6 | 358 | 726 | 0,38 | 10019404,547 |
| 29 | 17,37 | Hexadecanoic acid, methyl ester | C17H34O2 | 270 | 824 | 0,51 | 13473523,905 |
| 30 | 17,96 | n-Hexadecanoic acid | C16H32O2 | 256 | 875 | 6,45 | 171053124,069 |
| 31 | 18,82 | Heptadecanoic acid | C17H34O2 | 270 | 822 | 0,36 | 9504109,889 |
| 32 | 19,00 | 9,12-Octadecadienoic acid (Z,Z)-, methyl ester | C19H34O2 | 294 | 909 | 0,99 | 26351755,956 |
| 33 | 19,65 | 9,12-Octadecadienoic acid (Z,Z)- | C18H32O2 | 280 | 886 | 33,68 | 893553961,822 |
| 34 | 19,81 | Octadecanoic acid | C18H36O2 | 284 | 895 | 3,51 | 93133812,412 |
| 35 | 21,13 | 9,12,15-Octadecatrienoic acid, 2,3-dihydroxypropyl ester, (Z,Z,Z)- | C21H36O4 | 352 | 800 | 0,31 | 8280821,105 |
| 36 | 21,32 | 7-Methyl-Z-tetradecen-1-ol acetate | C17H32O2 | 268 | 792 | 0,27 | 7034298,457 |
| 37 | 21,43 | Ethyl iso-allocholate | C26H44O5 | 436 | 778 | 0,53 | 14171794,774 |
| 38 | 22,09 | 1,4-Naphthoquinone, 2-acetyl-5,8-dihydroxy-3-methoxy- | C13H10O6 | 262 | 719 | 0,37 | 9832281,596 |
| 39 | 22,62 | Hexadecanoic acid, 1-(hydroxymethyl)-1,2-ethanediyl ester | C35H68O5 | 568 | 791 | 0,64 | 17030916,494 |
| 40 | 23,15 | Ethyl iso-allocholate | C26H44O5 | 436 | 760 | 0,50 | 13224711,648 |
| 41 | 23,99 | 9,12-Octadecadienoic acid (Z,Z)-, 2,3-dihydroxypropyl ester | C21H38O4 | 354 | 826 | 0,89 | 23739730,221 |
| 42 | 24,68 | 6ß-Hydroxymethandienone | C20H28O3 | 316 | 737 | 1,64 | 43534942,669 |
| 43 | 25,32 | 7aH-Cyclopenta[a]cyclopropa[f]cycloundecene-2,4,7,7a,10,11-hexol, 1,1a,2,3,4,4a,5,6,7,10,11,11a-dodecahydro-1,1,3,6,9-pentamethyl-, 2,4,7,10,11-pentaacetate | C30H44O11 | 580 | 745 | 0,28 | 7490606,831 |
| 44 | 25,56 | 1H-Cyclopropa[3,4]benz[1,2-e]azulene-3-carboxaldehyde, 9a-(acetyloxy)-1a,1b,4,4a,5,7a,7b,8,9,9a-decahydro-4a,7b,9-trihydroxy-1,1,6,8-tetramethyl-5-oxo-, [1aR-(1aa,1bß,4aß,7aa,7ba,8a,9ß,9aa)]- | C22H28O7 | 404 | 613 | 1,64 | 43411487,584 |
| 45 | 27,82 | Prednisolone Acetate | C23H30O6 | 402 | 713 | 0,28 | 7344743,487 |
| 46 | 28,32 | Ethyl iso-allocholate | C26H44O5 | 436 | 711 | 0,73 | 19360722,136 |
| 47 | 28,51 | (+)--Tocopherol, O-methyl- | C29H50O2 | 430 | 778 | 0,44 | 11561126,095 |
| 48 | 29,74 | 9,12,15-Octadecatrienoic acid, 2-phenyl-1,3-dioxan-5-yl ester | C28H40O4 | 440 | 702 | 0,05 | 1402279,047 |
| 49 | 30,07 | Ethyl iso-allocholate | C26H44O5 | 436 | 800 | 0,36 | 9572451,194 |
| 50 | 31,75 | -Sitosterol | C29H50O | 414 | 865 | 0,91 | 24167148,635 |
Table 4 summarizes the three compounds in the ethanol extract of the seed coat that have the highest relative peak areas (%) in this investigation, whereas fig. 12-14 summarizes the apex peak compound and real-time.
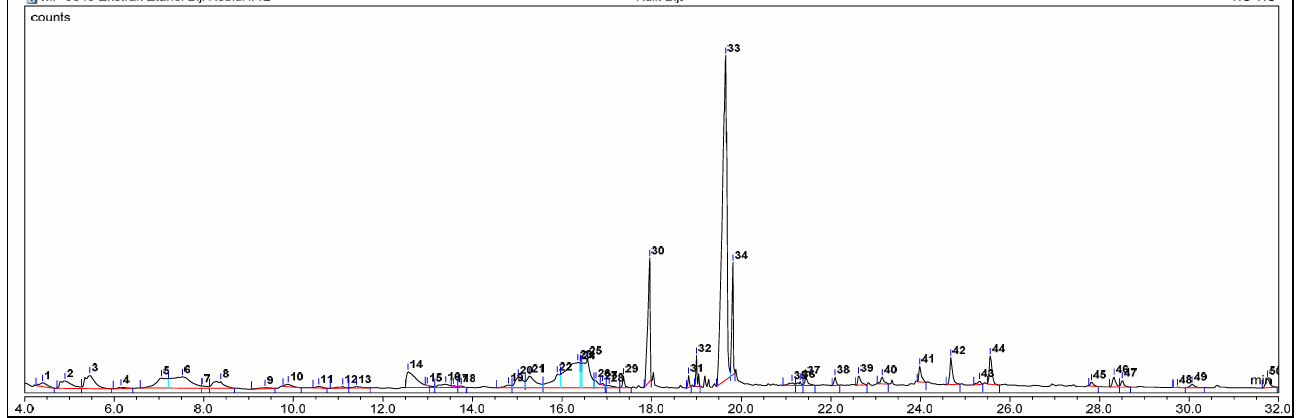
Fig. 11: Chromatogram of ethanol extract of kebiul seed coat by GC-MS
The chromatogram of the kebiul seed coat, as illustrated in fig. 11 and detailed in table 3, showcases the retention times and peaks identified through GC-MS analysis.
Fig. 11 presents the chromatogram obtained from the ethanol extract of the kebiul seed coat. Compounds were identified by comparing their mass spectra with a primary database, ensuring a Similarity Index (SI) exceeding 80% (NIST), and by cross-referencing with published literature values. GC-MS analysis identified 50 phytochemical compounds in the seed coat extract (table 3).
Table 4: Compounds with various retention times, percentage of peak area, molecular formula, and molecular weight appeared after GC-MS analysis of ethanol extract of kebiul seed coat
| No | Name | RT* (Min) |
Peak area (%) | Molekuler formula | Molecular weight | Structure | SI |
| 1 | n-Hexadecanoic acid | 17,96 | 6,45 | C16H32O2 | 256 | 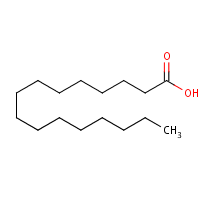 |
875 |
| 2 | 9,12-Octadecadienoic acid (Z,Z)- | 19,65 | 33,68 | C18H32O2 | 280 |  |
886 |
| 3 | Octadecanoic acid | 19,81 | 3,51 | C18H36O2 | 284 | 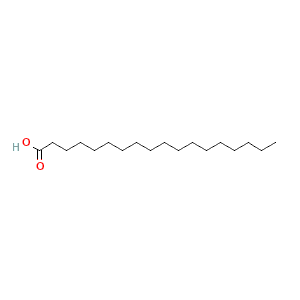 |
895 |
| Ret. Time: | 17,955 | 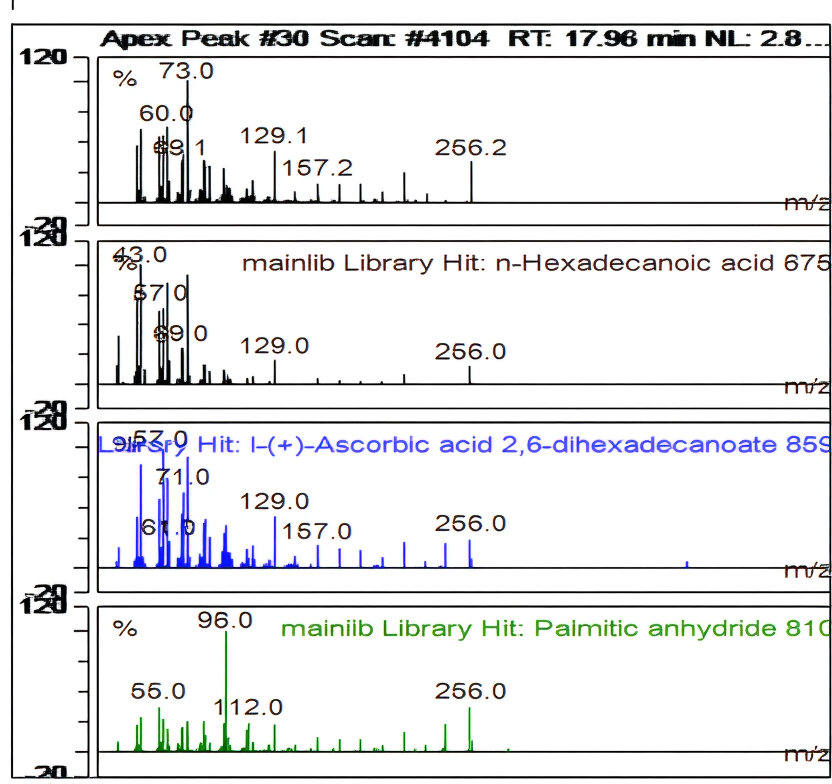 |
|||||
| Hit# 1 | |||||||
| Component: | n-Hexadecanoic acid | ||||||
| SI: | 875 | ||||||
| RSI: | 880 | ||||||
| Library: | mainlib | ||||||
| Hit# 2 | |||||||
| Component: | l-(+)-Ascorbic acid 2,6-dihexadecanoate | ||||||
| SI: | 859 | ||||||
| RSI: | 859 | ||||||
| Library: | mainlib | ||||||
| Hit# 3 | |||||||
| Component: | Palmitic anhydride | ||||||
| SI: | 810 | ||||||
| RSI: | 810 |
Fig. 12: Apex peak GC-MS hexadecanoic acid
| Ret. Time: | 19,652 | 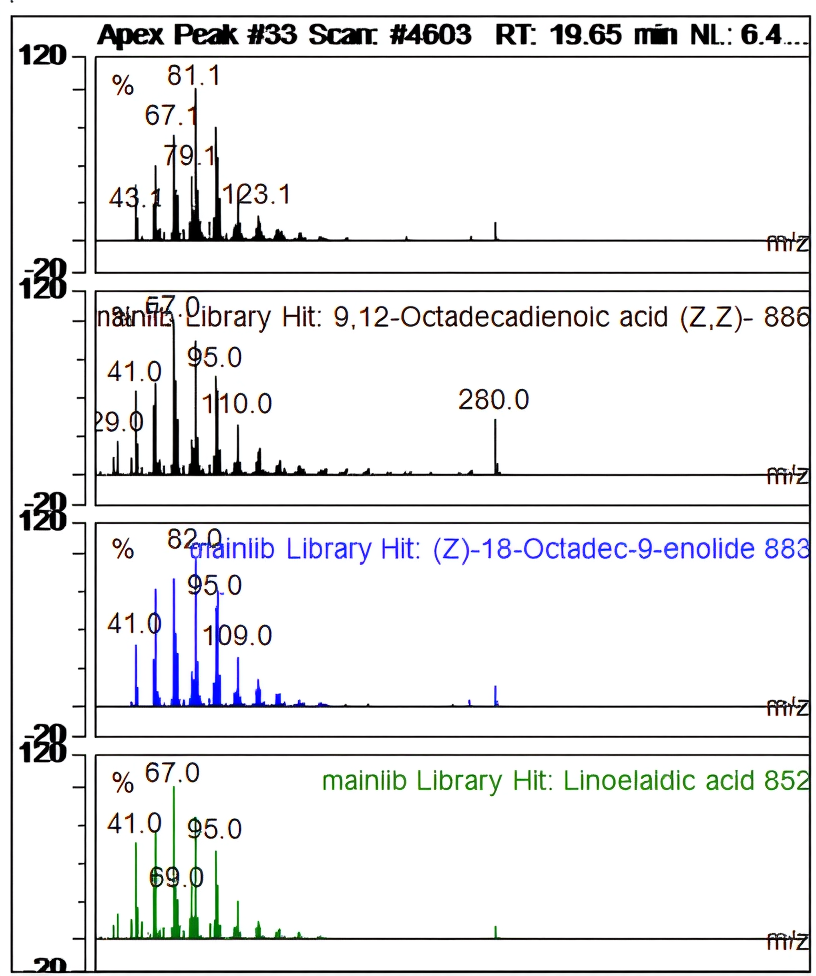 |
| Hit# 1 | ||
| Component: | 9,12-Octadecadienoic acid (Z,Z)- | |
| SI: | 886 | |
| RSI: | 886 | |
| Library: | mainlib | |
| Hit# 2 | ||
| Component: | (Z)-18-Octadec-9-enolide | |
| SI: | 883 | |
| RSI: | 896 | |
| Library: | mainlib | |
| Hit# 3 | ||
| Component: | Linoelaidic acid | |
| SI: | 852 | |
| RSI: | 922 |
Fig. 13: Apek peak 9,12-octadecadienoic acid (Z,Z)-
| Ret. time: | 19,812 | 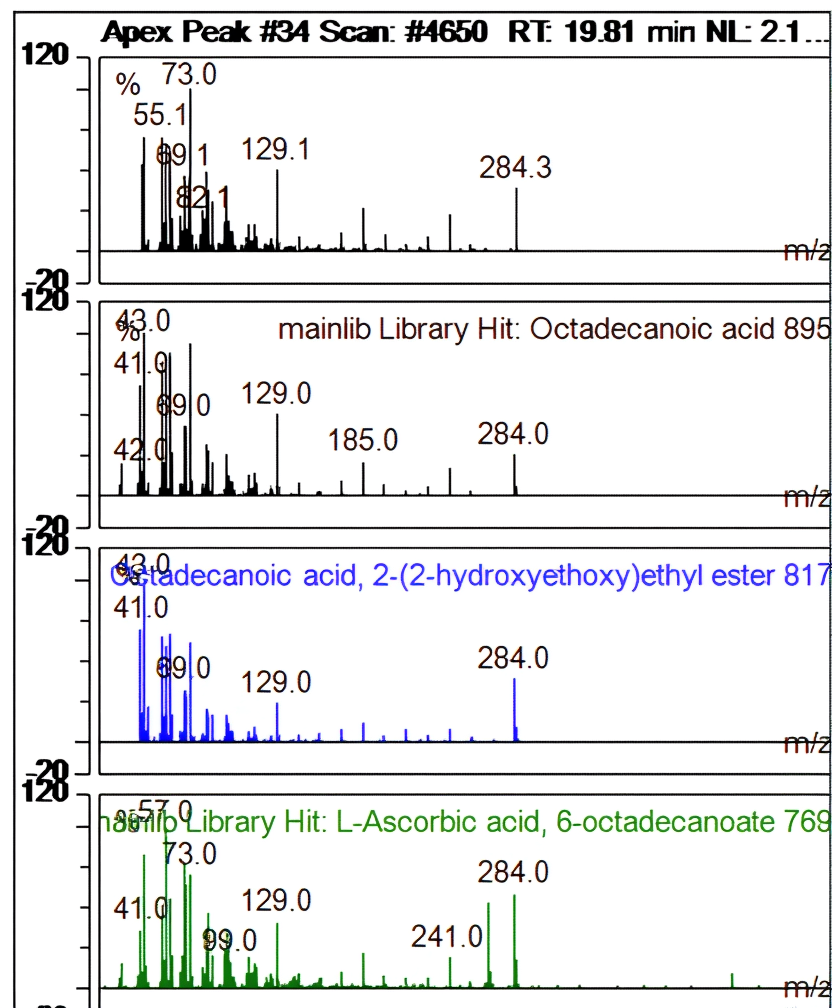 |
| Hit# 1 | ||
| Component: | Octadecanoic acid | |
| SI: | 895 | |
| RSI: | 899 | |
| Library: | mainlib | |
| Hit# 2 | ||
| Component: | Octadecanoic acid, 2-(2-hydroxyethoxy)ethyl ester | |
| SI: | 817 | |
| RSI: | 830 | |
| Library: | Mainlib | |
| Hit# 3 | ||
| Component: | L-Ascorbic acid, 6-octadecanoate | |
| SI: | 769 | |
| RSI: | 769 |
Fig. 14: Apex peak octadecanoic acid
In contrast to the main library, fig. 12-14 shows the compound's apex peak at a certain moment as well as its resemblance to other compounds.
DISCUSSION
The kingdom of plants contains an endless supply of active chemicals for medications. In the quest for resources and raw materials for the pharmaceutical sector, phytochemical approaches are crucial. Finding new drugs is a drawn-out process that includes several steps that must be taken one after the other. Fresh and dried plant materials can be extracted using various of methods, either with or without the use of solvents (expression, sublimation, distillation, etc). These methods may include the use of water, ether, acetone, methanol, ethanol, chloroform, etc. A key step in the analysis of medicinal plants is isolation, which involves several basic procedures, including prewashing, drying or freeze-drying plant materials, grinding to produce a homogenous sample, and frequently enhancing the kinetics of analytic extraction and increasing sample surface contact with the solvent system. Qualitative assays for screening phytochemical compounds are the main way to start phytochemical characterization. The last stages in the photochemical study of plants are the characterization and identification of the isolated and separated constituents [10].
The selection of plant material, phytochemical analysis, characterization, and pharmacological investigation are all steps in the drug development process. A thorough preclinical inquiry is then conducted before clinical trials begin. Until 1996, naturally occurring chemicals either inspired or directly derived almost 80% of pharmaceuticals. Between 1981 and 2019, 1881 new medications were approved; of these, about 23.5% were natural products or semisynthetic derivatives of natural products, and about 25% were either pharmacophores or natural product mimics. The assessment of natural products in novel drug discovery has been revolutionized by the emergence of new skills [11].
By utilizing both mass spectra and retention periods, GC-MS is an effective method for compound identification. The GC-MS chromatogram examination of the ethanol extract revealed that the ethanol extract of seeds contained 87 bioactive phytochemical compounds (fig. 2), whereas the ethanol extract of the seed coat contained 50 bioactive phytochemical compounds (fig. 11). Tables 1-4 highlight the three compounds in the ethanol extract of the seed coat and the seven compounds in the ethanol extract of the seed kernel with the largest relative peak areas, as well as the apex peak compound and real time. The amount of time it takes for a chemical to move from the GC column to the detector is known as the retention time. It is an essential criterion for compound identification. When compared to the mainlib collection, the mass spectrum's apex peak similarity and retention duration are matched to different chemicals (fig. 4-13).
The bioactive chemicals from the coral Junceella delicata were separated and then processed for structure elucidation using GC-MS and FTIR. These substances include ethyl aminomethyl formimidate, Gly-Gly, and 2-(2-Pyridyl)-4-methylthiazole-5-carboxylic acid, 7-Methoxy-2-methylquinolin-4-ol, fraxidin, 2-methyl-3-trans-propenylpyrazine, 3-tert-butylpyridine, acetaldehyde benzyl ethyl acetal, α-methylcinnamicacid, 4-ethoxycoumarin, 3-hydroxycoumarin, 2,4,7,9-tetramethyl-5-decyne-4,7-diol, 2,2-bis(3-allyl-4-hydroxyphenyl) propane, phenyltriethylammonium cation, and dodecanedioic acid [12]. Using spectroscopic data from the literature, five lignans (1–5), two phloroglucinols (6–7), five flavonoids (8–12), and four phenolics (13–16) were discovered from the leaves of Caesalpinia bonduc. The genus Caesalpinia yielded compounds 1–4, 6–7, 11, and 13–14, which are useful chemotaxonomic markers for Caesalpinia bonduc [13]. Determine secondary metabolites and analyze the methanol extract of Caesalpinia bonduc seeds using Gas Chromatography-Mass Spectrometry (GC-MS) and Fourier Transform Infrared (FTIR) as well as antibiotic activity. Alkaloids, flavonoids, saponins, tannins, and phenolics are detected by phytochemical screening of the methanol extract from Caesalpinia bonduc seeds. Three major chemicals were identified by the GC-MS analysis: 4-Methyl-1,3-Dioxolan-2-One, 2-Trimethylsilyl-1,3-dithiane, and Cholesta-5,7,9(11)-trien-3-ol acetate. The FTIR study revealed that the extract contains the functional groups O-H, N-H, C-N, C-H, C-O, C=C, C=O, C-S, and C-Si. Escherichia coli and Staphylococcus aureus growth was inhibited by the antibacterial activity methanol extract from Caesalpinia bonduc seeds at different doses [14].
This work used gas chromatography and mass spectroscopy to identify the bioactive chemicals found in the ethanol extracts of kebiul seed kernel and kebiul seed coat. Retention time (RT), molecular formula, molecular weight (BM), and concentration (peak area%) are all active principles. An intriguing technique for determining the concentration of certain active ingredients in herbs used in the food, pharmaceutical, cosmetics, environmental, and forensic industries is the GC-MS method, which is employed to analyze the extracted materials [15]. This method analyzes a combination of chemical components by combining two analytical techniques into one. The components of the mixture are separated by gas chromatography, and each component is examined independently by mass spectroscopy.
Utilize HPLC and GC-MS methodologies to examine the bioactive constituents of Matricaria chamomilla (chamomile) flowers, encompassing flavonoids, terpenoids, and polyphenols [16]. 20 chemicals were identified as a result of the n-hexane extract's gas chromatography-mass spectrometry (GC-MS) investigation. These comprised siloxanes, fatty acid esters, sterols (e. g., stigmasterol and ergost-5-en-3-ol), terpenoids (e. g., phytol and caryophyllene oxide), alkanes, and other hydrocarbons. Numerous substances, such as erucamide, phytol, and squalene, have been investigated for their known antibacterial and anticancer properties.
Caesalpinia bonduc is utilized in ointments including castor oil and seed powder, which can be applied topically to address hydrocele and orchitis [17]. Seed-derived oil can be applied topically to address convulsions and paralysis. The consumption of equal quantities of pepper and Caesalpinia bonduc seed powder has demonstrated antimalarial properties [18]. They utilized such grains as necklaces [19]. Seed oil moisturizes the skin and alleviates acne. Caesalpinia bonduc seeds, as a polyherbal formulation, are utilized in the treatment of diphtheria (7 g of mixed seeds) and pneumonia (5 g of mixed seeds), as indicated by an ethnobotanical study [20].
The seeds are utilized for several medicinal conditions, including hemostatic, laxative, anthelmintic, inflammation, colic, malaria, skin disorders, and leprosy, according to Singh and Raghav (2012). The seeds' extracts are also utilized as anthelmintic and anti-blennorrhagic medicines, and they are also used as tonics [22]. Traditionally, the seed extract is used with castor oil or honey to make this anthelmintic medication [23]. Traditional folk pre-treat intermittent fever with Caesalpinia bonduc juice for two weeks. To treat hydrocele and orchitis topically, seed powder has also been combined with castor oil to create an ointment [24]. According to a recent assessment by Sasidharan et al. (2021), the best method of treating health issues is to take roasted and powdered seeds orally. Similarly, a review examined the potential of Caesalpinia bonduc in treating polycystic ovarian syndrome (PCOS) [2]. To energize the body and ease pain, the seeds are roasted and ground into a powder. The seeds, which are made as a poultice with asafoetida, ghee, and salt to taste, are also used to treat postpartum stomach pain.
Colitis, diarrhea, and dysentery have all been treated using the seed coat. Amra, Haridra, and Palasa can be taken with roasted seed powder or leaf juice to treat worm infestation-induced irritation in the anal area. In India, the roasted seeds are marketed as Latakaranja after being mixed 1:1 with pippali. An approved herbal treatment for malaria, it can be administered at intervals of 0.5 g per day for 3–4 d [26]. The extract of this plant is an excellent treatment for an enlarged spleen caused by malaria [27]. Additionally, they reported that the plant's seeds are used to treat menstrual pain in the abdomen, boost menstrual flow during oligomenorrhea, and enhance uterine function. Because the seed coat is so effective at treating fluoride, it is utilized to absorb the fluoride present in drinking water. Therefore, the first step in identifying the active chemicals in this plant is to analyze the ethanol extract of the seed kernel and seed coat of kebiul using GC-MS.
CONCLUSION
GC-MS analysis revealed the ethanol extract of the kebiul seed coat included 50 phytochemical compounds, with the highest compound at a retention time of 19.65 min, namely 9,12-Octadecadienoic acid (Z,Z)-. Subsequently, n-Hexadecanoic acid exhibits a retention time of 17.96 min, while Octadecanoic acid has a retention time of 19.81 min. While the ethanol extract of the kebiul seed kernel contained 87 phytochemical compounds. Since these results are preliminary and only based on chemical identification, more investigation is required, with the highest compound at a retention time of 19.93 min, namely 6-Octadecenoic acid. Subsequently Pregnenoloneexhibits a retention time of 24.90 min, 1H-Cyclopropa[3,4]benz[1,2-e]azulene-5,7b,9,9a-tetrol,3-[(acetyloxy)methyl]-1a,1b,4,4a,5,7a,8,9-octahydro-1,1,6,8-tetramethyl-, 5,9,9a-triacetate, [1aR-(1aa,1bß,4aß,5ß,7aa,7ba,8a,9ß,9aa)]-exhibits a retention time of 25.82 min,1H-Cyclopropa[3,4]benz[1,2-e]azulene-5,7b,9,9a-tetrol, 3-[(acetyloxy)methyl]-1a,1b,4,4a,5,7a,8,9-octahydro-1,1,6,8-tetramethyl-, 5,9,9a-triacetate, [1aR-(1aa,1bß,4aß,5ß,7aa,7ba,8a,9ß,9aa)]-has a retention time of 23.21 min, l-(+)-Ascorbic acid 2,6-dihexadecanoate has a retention time of 18.16 min, and 9,12-Octadecadienoic acid, ethyl ester has a retention time of 19.64 min, and Pregna-5,17(20)-dien-3-ol, (3ß,17E)-has a retention time of 23.21 min.
ACKNOWLEDGMENT
The authors would like to express their gratitude to the Center for Higher Education Funding and Assessment, Ministry of Higher Education, Science, and Technology of the Republic of Indonesia, which has provided financial support for this study. The authors also extend their gratitude to thank Al Fathah Foundation Bengkulu and all those who helped with this research process.
AUTHORS CONTRIBUTIONS
Densi Selpia Sopianti: Conceptualization, Writing–Original Draft; Muchsin Doewes: Writing the Original Draft and conducting the Review and Editing; Tatar Sumandjar: Writing–Original Draft, Writing–Review and Editing; Eti Poncorini Pamungkasari: Methodology, Writing–Original Draft; Paramasari Dirgahayu: Methodology, Writing–Original Draft; Ratih Puspita Febrinasari: Methodology, Writing–Original Draft.
CONFLICT OF INTERESTS
The authors declare no conflict of interest
REFERENCES
Srinivasan P, Karunanithi K, Muniappan A, Singamoorthy A, Kadaikunnan S, Narayanan SP. Botany traditional usages phytochemistry pharmacology and toxicology of Guilandina bonduc L.: a systematic review. Naunyn Schmiedebergs Arch Pharmacol. 2024;397(5):2747-75. doi: 10.1007/s00210-023-02822-w, PMID 37987793.
Kandasamy V, Balasundaram U. Caesalpinia bonduc (L.) Roxb. as a promising source of pharmacological compounds to treat poly cystic ovary syndrome (PCOS): a review. J Ethnopharmacol. 2021 Oct 28;279:114375. doi: 10.1016/j.jep.2021.114375, PMID 34192600.
Billah MM, Islam R, Khatun H, Parvin S, Islam E, Islam SA. Antibacterial antidiarrhoeal and cytotoxic activities of methanol extract and its fractions of Caesalpinia bonducella (L.) Roxb leaves. BMC Complement Altern Med. 2013;13:101. doi: 10.1186/1472-6882-13-101, PMID 23663985.
Shukla S, Mehta A, Mehta P, Vyas SP, Shukla S, Bajpai VK. Studies on anti-inflammatory, antipyretic and analgesic properties of Caesalpinia bonducella F. seed oil in experimental animal models. Food Chem Toxicol. 2010 Jan;48(1):61-4. doi: 10.1016/j.fct.2009.09.015, PMID 19766160.
Vuorelaa P, Leinonenb M, Saikkuc P, Tammelaa P, Rauhad JP, Wennberge T. Natural products in the process of finding new drug candidates. Curr Med Chem. 2004 Jun;11(11):1375-89. doi: 10.2174/0929867043365116, PMID 15180572.
Hutton I. Rare plant surveys: Lord Howe Island. Report to NSW Scientific Committee. Sydney: NSW Department of Planning and Environment; 2016.
Cheruvathur MK, Britto J, Thomas TD. Callus induction and shoot regeneration from epicotyl explants of ethnomedicinally important Caesalpinia bonduc (L.) Roxb. Iran J Biotechnol. 2010;8(4):263-9.
Syarpin S, Permatasari S, Pujianto DA. Analysis of phytochemical constituents and antioxidant activity from the fractions of Luvunga sarmentosa root extract using LCMS/MS. Biodiversitas. 2023;24(2):733-40. doi: 10.13057/biodiv/d240208.
Tran TK, Ha PT, Henry RJ, Nguyen DN, Tuyen PT, Liem NT. Polyphenol contents gas chromatography mass spectrometry (GC–MS) and antibacterial activity of methanol extract and fractions of Sonneratia caseolaris fruits from Ben Tre Province in Vietnam. J Microbiol Biotechnol. 2024;34(1):94-102. doi: 10.4014/jmb.2304.04019, PMID 38282409.
Alamgir AN. Methods of qualitative and quantitative analysis of plant constituents. Prog Drug Res. 2018;74:721-804. doi: 10.1007/978-3-319-92387-1_9.
Goel B, Sahu B, Jain SK. Plant-derived drug discovery: introduction to recent approaches. In: Singh B, editor. Botanical leads for drug discovery. Singapore: Springer; 2020. p. 1-27. doi: 10.1007/978-981-15-5917-4_1.
Borate M, Zodape G. Extraction and isolation of bioactive compounds from coral Junceella delicata (Grasshoff, 1999) from west coast of Mumbai. Int J Pharm Pharm Sci. 2025 Jul;17(9):36-46. doi: 10.22159/ijpps.2025v17i9.54605.
Zhang TL, Yi QQ, Han YX, Zhao L, Gui SQ, Liu WX. Chemical constituents from the leaves of Callicarpa arborea Roxb. and their chemotaxonomic significance. Biochem Syst Ecol. 2023;110:104683. doi: 10.1016/j.bse.2023.104683.
Samriani S, Natsir H, Dali S, Arif AR. Phytochemical screening GC–MS, FTIR, and antibacterial activity of methanol extract of Caesalpinia bonduc (L.) Roxb. seeds. AIP Conf Proc. 2024 Aug 30;2774(1):030015. doi: 10.1063/5.0165110.
Uma B, Prabhakar K, Rajendran S, Sarayu LY. Studies on GC/MS spectroscopic analysis of some bioactive antimicrobial compounds from Cinnamomum zeylanicum. J Med Plants. 2009;8(31):125-31.
Parabathina RK, Dubey NN, Girgaonkar SS, Lolge VN, Narwade KK. Qualitative and quantitative analysis of flavonoids terpenoids and polyphenols in chamomile flowers by HPLC and GC–MS methods. Int J Pharm Pharm Sci. 2025 Jul;17(9):28-35. doi: 10.22159/ijpps.2025v17i9.54977.
Handa SS, Kaul MK. Supplement to cultivation and utilization of medicinal plants. Jammu-Tawi: RRL; 1996. p. 727-37.
Kokate CK. Practical pharmacognosy. 4th ed. Delhi: Vallabh Prakashan; 1997. p. 107-11.
D Souza M. Tribal medicine. 1st ed. Ahmednagar India: Social Center; 1998. p. 300.
Acharyya, BK, Sharma, HK. Folklore medicinal plants of Mahmora area Sivasagar district Assam. Indian Journal of Traditional Knowledge. 2004;3(4):365-72.
Singh V, Raghav PK. Review of pharmacological properties of Caesalpinia bonduc L. Int J Med Arom Plants. 2012;3(2):514-30.
Nazeerullah K, Sunil K, Pal SR, Neelam D. A pharmacognostic and pharmacological overview on Caesalpinia bonducella. Res J Pharm Biol Chem Sci. 2012;3:440-96.
Chopra RN, Nayar SL. Glossary of Indian medicinal plants. New Delhi: Council of Scientific and Industrial Research; 1956. p. 256.
Subramani V, Kamaraj M, Ramachandran B, Jerome Jeyakumar J. Phytochemical investigation and antimicrobial activity of Caesalpinia bonduc (Linn.) Roxb. Seeds. Int J Phytopharmacol. 2014;4(3):92-5. doi: 10.7439/ijpp.v4i3.102.
Sasidharan S, Kumar S, Das K, Nair H. Caesalpinia bonduc: a ubiquitous yet remarkable tropical plant owing various promising pharmacological and medicinal properties with special reference to the seed. Med Aromat Plants (Los Angeles). 2021;10(7):394. doi: 10.35248/2167-0412.21.10.394.
Singh PK, Dwivedi M, Prakash S, Dubey RK, Tiwari S, Kumar A. Pharmacognostical and phytochemical study on Caesalpinia bonduc L. (Latakaranja): an overview. World J Pharm Res. 2016;5(2):1512-20.
Nadkarni AK, Nadkarni KM. Indian materia medica. Rev. ed Bombay: Popular Book Depot; 1954.


