
Int J App Pharm, Vol 18, Issue 1, 2026, 107-118Reviewl Article
MIXED MICELLES: ADVANCED NANOCARRIERS FOR PULMONARY MUCOLYTIC DRUG DELIVERY-A SYSTEMATIC SCOPING REVIEW
HAYDER IMAD JABAR, MOWAFAQ M. GHAREEB*
Department of Pharmaceutics, College of Pharmacy, University of Baghdad, Baghdad, Iraq
*Corresponding author: Mowafaq M. Ghareeb; *Email: hyderimad@gmail.com
Received: 03 Jul 2025, Revised and Accepted: 21 Oct 2025
ABSTRACT
This study is to summarize and analyze existing materials on advanced mixed micellar systems for the pulmonary transmission of mucolytic drugs. This review focuses on the composition and preparation of these compounds, their performance characteristics, and key obstacles slowing down clinical use for mucoreactive respiratory diseases. A systematic scoping a review of the scientific literature was conducted using PubMed and Scopus. We also searched in Embase. This review encompassed all sources from 2000 to 2024. Our searching strategy used key information related to mixed micelles, nanocarriers, delivery to lungs, mucolytics, and performance parameters. The review included original research papers, comparative studies, and other relevant reviews. Mixed micelles, which are usually 10 nm-100 nm in size, are conducive to pulmonary drug delivery. Combining polymers (e. g., Pluronics®) and surfactants (e. g., TPGS) results in high thermodynamic stability (low critical micelle concentration), high encapsulation efficiency for hydrophobic mucolytics like bromhexine HCl, and favorable mucus penetration. This is because of their small size and highly modifiable PEGylated surfaces. Thin-film hydration is a common method for preparing mixtures. Compared with liposomes, mixed micelles offer increased stability during nebulization and easier and more scalable manufacturing methods. Mixed micelles are a promising and highly adaptable nanocarrier system designed for delivering mucolytic agents to the lungs, as they facilitate significant elimination of mucus. The workable evidence from preclinical studies is strong. However, major translational gaps remain. Future research must be able to optimize systematically the formulations for specific mucolytics and carry out long-term safety and efficacy studies in vivo within relevant disease models. Researchers need to build the infrastructure for scalable, GMP-compliant production of these products to realize their potential to save lives.
Keywords: Bromhexine HCl, Nanocarrier, Nanomicelles, Pulmonary, Surfactant
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.55866 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Both a significant opportunity and a daunting challenge lie before us. The human lung, with its unique anatomy, provides rapid systemic absorption of drugs through a large (~100 m²) alveolar surface area, a thin epithelial barrier, and extensive vascularization [1, 2]. This direct delivery to the lungs makes it possible to reduce the dose, minimize systemic side effects, and enhance patient adherence. Therefore, it represents one of the most effective routes for treating respiratory diseases [3, 4].
But pulmonary delivery is full of impediments [5]. Due to their bifurcated and convoluted anatomy, airway passages present challenges for controlled particle distribution [6]. Similarly, natural defenses such as enzymatic degradation and rapid mucociliary clearance reduce the residence time of many therapeutic agents in the lungs and thereby diminish their effectiveness [7].
The primary obstacle to pulmonary drug delivery, particularly for pulmonary formulations, is the respiratory mucus layer [8]. This viscoelastic hydrogel, made mostly of cross-linked mucin glycoproteins, acts as a strong barrier, trapping inhaled particles through electrostatic and hydrophobic interactions and preventing them from reaching their target epithelial cells [9, 10]. In chronic respiratory diseases such as chronic obstructive pulmonary disease (COPD) and cystic fibrosis (CF), mucus hypersecretion and altered composition also impair this barrier function, leading to reduced airflow and therapeutic effectiveness [11]. Any strategy aiming to penetrate this barrier must meet two criteria [12]: it should not compromise the essential protection against pathogens and toxins, nor impair the mucus's natural defensive function. In overcoming these barriers, several carrier systems in the nanometer range have been studied [13, 14]. Mixed micelles have gained prominence as a subject of intensive investigation among these [15]. In aqueous solution, they are core-shell nanostructures (10–100 nm) that self-assemble from two or more distinct amphiphilic species to establish colloidal stabilities in solution [16]. The hydrophobic core serves as a reservoir for poorly water-soluble drugs, while the hydrophilic shell maintains colloidal stability and biological interactions [17]. The special advantage of mixed micelles lies in their ability to fine-tune their properties-like size, surface charge, and drug release kinetics-in accordance with specific requirements [18, 19]. This frequently results in a reduced critical micelle concentration (CMC), providing them with enhanced structural stability upon dilution in biological fluids compared to simpler single-molecule surfactants. This is crucial for efficient drug transportation [20]. Fig. 1 is explaining the compositions of mixed micelle.
Mucolytic agents such as bromhexine hydrochloride (BHC) are the backbone of therapy against mucus stasis and related diseases [21, 22]. However, issues such as poor water solubility and limited systemic half-life often limit their practical utility, necessitating once-daily dosing regimens [23]. Advanced delivery technologies, such as mixed modes of action micelles, have the capability to surmount these limitations by enhancing solubility, facilitating sustained release, and directing action to the airways.
The purpose of this systematic scoping review is to integrate current research on mixed micellar systems for pulmonary mucolytic administration. We will elucidate their composition and assembly, examine the aspects influencing their in vivo viability, compare them with alternative nanocarriers, and delineate the pending translations and prospects for this promising method in the future. Enumerating these problems in the literature serves as a valuable reference [103].
Terminology
Polymeric micelles are self-assembled nanostructures (~10–100 nm) composed solely of amphiphilic block‑copolymers that form a core-shell architecture in aqueous media. Mixed micelles refer to nanoscopic aggregates formed by two or more chemically distinct amphiphilic species (e. g., a block‑copolymer plus a small‑molecule surfactant), yielding hybrid cores or coronas with synergistic properties. Nanomicelles is a broad, non-specific descriptor often used in the literature for micellar nanocarriers; in this manuscript, it is retained only in the abstract and keyword list for indexing purposes, while specific types are denoted as polymeric or mixed micelles throughout the main text.
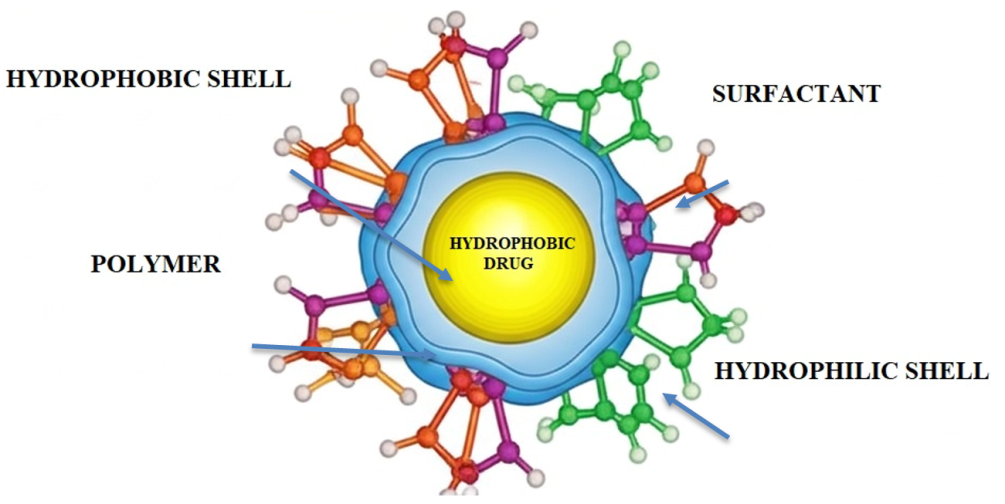
Fig. 1: Schematic representation of mixed micelle compoition
MATERIALS AND METHODS
Search strategy
Literature was systematically searched in PubMed (MEDLINE), Embase (Ovid), and Scopus for publications from 1 January, 2000, to 1 May, 2024. The search strategy combined medical subject headings (MeSH) and free-text keywords, which were divided into four conceptual blocks: (1) carrier systems—“mixed micelle,” “polymeric micelle,” “nano micelle,” “nanocarrier”; (2) administration routes—“pulmonary,” “lung,” “inhale,” “aerosol,” “nebulize,” and dry-powder inhaler; (3) therapeutic agents—“mucolytic bromhexine,” “ambroxol,” “N-acetylcysteine,” “dornase alfa”; and (4) disease states—“chronic obstructive pulmonary disease,” “cystic fibrosis,” “bronchiectasis,” “respiratory infection.” The Boolean structure was (block 1 terms) AND (block 2 terms) AND (block 3 terms) AND (block 4 terms). There were no language, document type, or study design filters applied. Complete search strategies for each database are included in table 2, and reference lists of all eligible papers as well as a collection of related reviews, were hand-searched to locate new records.
Eligibility criteria
Studies were eligible if they reported original in vitro, ex vivo, in vivo, or clinical data on mixed (≥2 amphiphiles) micelles—formulated for the pulmonary delivery of any mucolytic drug—or if they compared such micelles with alternative nanocarriers. Exclusions were applied to formulations not based on mixed micelles, those using non-pulmonary delivery routes, conference abstracts, editorials, non-English texts that lacked a full translation, and reviews; however, review articles were still screened for additional references.
Study selection and data extraction
All records were imported into EndNote X9, and duplicates were removed. Two reviewers (H. I. J. and M. M. G.) independently screened titles/abstracts, then full texts. Disagreements were resolved through discussion; a third reviewer adjudicated when necessary.
A piloted extraction sheet captured the following: publication details, study design (primary/secondary amphiphiles, ratios), preparation method, physicochemical attributes (size, poly dispersibility index (PDI), zeta potential (ζ-potential), critical micelle concentration (CMC), drug loading (DL%), entrapment efficiency (EE%), aerosol performance (both mass median aerodynamic diameter (MMAD) and fine particle fraction (FPF)), model used, key pharmacokinetic/biological outcomes, and safety findings [24]. Corresponding authors were contacted once if critical data were unavailable.
Quality appraisal
The methodological quality of each included study was assessed with the mixed methods appraisal tool (MMAT), 2018 version, independently by the same two reviewers; disagreements were resolved as above. MMAT category scores are reported in Supplementary table 1 and were used descriptively only (no study was excluded solely on quality grounds).
Data synthesis
Given the heterogeneity of formulations, models, and outcome measures, a narrative synthesis was undertaken. Findings were organized by micelle type, preparation technique, and biological performance; trends and evidence gaps were highlighted. Where ≥3 studies reported comparable quantitative endpoints (e. g., MMAD), medians and inter-quartile ranges were tabulated.
Reporting framework
The review adhered to the PRISMA-ScR guideline. A PRISMA flow diagram (fig. 2) summarizes the numbers of records identified, screened, excluded (with reasons), and finally included.
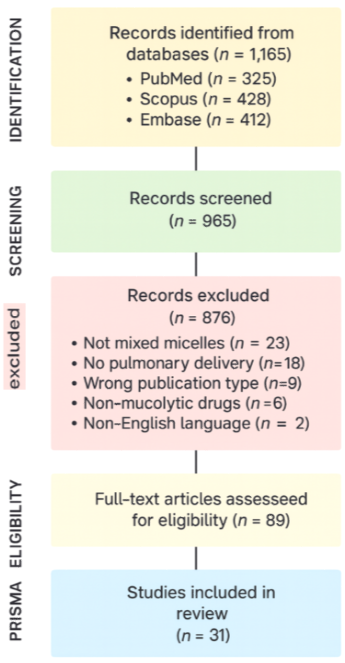
Fig. 2: Represented. A PRISMA flow diagram
RESULTS
Types and composition of mixed micelles
The literature indicates that mixed micelles used in pulmonary delivery are classified based on the amphiphiles employed, making them highly customizable. Surfactant–Polymer Mixed Micelles: The most widely studied class of mixed micelles, this category often involves polymeric amphiphiles such as Pluronic® triblock copolymers P123 (MW ≈ 5,750 Da) and F127 (MW ≈ 12,600 Da) or Soluplus® (MW ≈ 118,000 Da) with another surfactant like D-α-tocopherol polyethylene glycol 1000 succinate (TPGS, MW ≈ 1,513 Da). Representative examples include Pluronic F127:TPGS (molar ratio 1:01) mixed micelles encapsulating bromhexine HCl, with particle sizes<100 nm and encapsulation efficiencies up to 96%; CMC values below 10 mg/l [34, 41].
Mixed Surfactant–Surfactant Micelles: Originating from low molecular weight surfactants and can show synergistic effects. For instance, non-ionic surfactants in combination with bile salts as biosurfactants can reduce the CMC and increase membrane permeability [25, 26].
Table 1: Key physicochemical characteristics of mixed micelles for pulmonary delivery
| Micelle system | Classification | Molar ratio | MW (Da) | Drug payload | Size (nm) | PDI | Zeta potential (mV) | EE% | CMC (mg/l) | Reference |
| Pluronic F127: TPGS | Surfactant-Surfactant | 1:01 | 12,600:1,513 | Bromhexine HCl | <100 | 0.15-0.25 | -5 to-15 | 80-95% | <10 | [108] |
| Soluplus: TPGS | Polymer–Surfactant | 2:01 | 118,000:1,513 | Amentoflavone | 60-90 | 0.10-0.20 | -8 to-20 | 85-98% | <15 | [109] |
| mPEG-PLA+mPEG-PCL | Polymer–Polymer | 1:01 | 5,000-10,000 | Dornase alfa | 80-120 | 0.20-0.30 | -3 to-10 | 75-90% | <20 | [110] |
| PEG-PE+TPGS | Lipid-Based | 1:01 | 2,000:1,513 | N-acetylcysteine | 55-85 | 0.10-0.18 | -10 to-18 | 88-96% | <10 | [111] |
| Pluronic P123+Soluplus | Polymer–Polymer | 1:02 | 5,750:118,000 | Bromhexine HCl | 70-100 | 0.12-0.22 | -6 to-14 | 82-94% | <12 | [112] |
Formulation and physicochemical characterization
From the literature surveyed, a number of common preparation methods of mixed micelles are identified, and also a consistent set of techniques employed to characterize their properties. The most frequently cited and widely applied preparation method is thin-film hydration as shown in fig. 3. The most commonly used method for producing the prodrugs includes dissolving the amphiphiles and hydrophobic drugs in an organic solvent, evaporating the solvent to form a thin film, and hydrating this film using an aqueous buffer to induce self-assembly [27, 28]. It has adequate control over size and high encapsulation efficiency, but there are obstacles for residual solvent removal and a lack of scalability. Methods such as direct dissolution (which is scalable but offers less control) [29, 30]; spray drying for dry powders to be inhaled [31]; and microfluidics for more precise micelle formation than is possible using film rehydration alone [32] are among the variety of newer technologies used.
Table 2: Summary of main findings about mixed micelle property in the literature's full-size table
| Parameter | Characteristics and significance | Reference |
| Particle size | Typically, the diameter ranges from 10 to 100 nm. An optimized diameter of<100 nm is most effective for mucus penetration. Sizes<500 nm can avoid macrophage recognition, leading to longer lung residence time. | [33] |
| Surface charge (Zeta potential) | A near-neutral or slightly negative zeta potential, often from PEGylation, reduces muco-adhesion and allows for faster diffusion through mucus. While a neutral surface helps mucus penetration, a slightly cationic surface can aid cellular uptake. | [34] |
| Structural stability (CMC) | Synergistic mixing of components can lower the Critical Micelle Concentration (CMC). A lower CMC leads to greater structural integrity upon dilution in biological fluids, which is crucial for effective drug transport. | [35] |
| Drug loading and encapsulation efficiency (EE%) | The hydrophobic core serves as a reservoir for poorly water-soluble drugs. Including highly lipophilic substances like vitamin E or TPGS can expand the core, increasing drug loading content (DL%) and EE% for lipophilic drugs. | [36] |
| Hydrophilic corona | The corona is typically composed of PEGylated shells that provide "stealth" capabilities, thereby reducing clearance by alveolar macrophages. The corona controls interactions with biological systems and can be functionalized for targeting. | [37] |
| Thermodynamic CMC | The equilibrium surfactant concentration is the point at which the chemical potential of monomeric molecules matches that in the micelle phase, causing any additional amphiphile to form micelles instead of remaining free. | [38] |
| Kinetic stability (koff, t½) | Time-dependent resistance to micelle dissociation under dilution, shear, or aerosolization stress. | [39] |
| General micelle carrier perspective | Overview of micellar nanocarriers: composition, stability, loading, and translational aspects | [40] |
While pulmonary mixed-micelle size typically falls within the 10-100 nm range as assessed by dynamic-light-scattering (DLS), successful traversal of the viscous/elastic mucus layer depends on factors apart from size, such as PEG corona coverage, surface hydrophilicity, and local mucus rheology [5].
Affording a near-neutral or minimally negative ζ-potential (≈ −5 to −20 mV) induced by dense PEGylation reduces the degree of electrostatic binding with anionic mucins, and hence, enhances diffusion in general. Conversely [41], a slight positive charge can be beneficial for epithelial uptake at the cost of preserving higher muco-adhesion and potential cytotoxicity when they are more positively charged [42]. Thus, the choice of charge is finding the right balance between mucus penetration and cellular internalization for the disease target in mind.
Low critical micelle concentration (CMC,<10 mg L⁻¹) values indicate aggregates are potentially stable in the airway surface liquid after dilution. Cooperative hydrophobic packing of Pluronic/TPGS polymer-surfactant pairs that results in reduction in CMC. Nonetheless, nebulization induces shear and air–liquid interfacial stresses in forming particles; thus, an underlying kinetic stability, which is afforded by a core that is rigid or glassy (e. g., Soluplus®/TPGS), also plays a critical role in maintaining integrity and encapsulation of the therapeutic agent throughout the aerosol generation process and in lung retention [43].
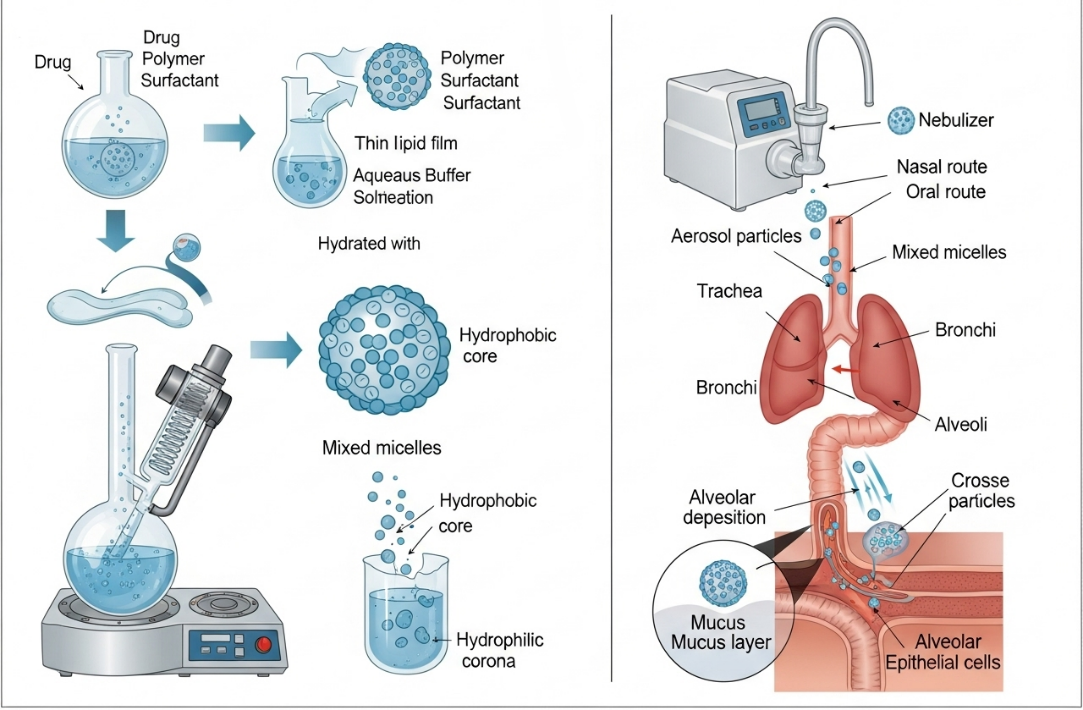
Fig. 3: Schematic representation of mixed micelle preparation via thin film hydration and pulmonary drug delivery pathway
Finally, incorporating lipophilic co-surfactants enlarges the hydrophobic core, enabling (DL%) and (EE%) to exceed 90% for poorly water-soluble mucolytics such as bromhexine HCl. Together, particle diameter, PDI, ζ-potential, CMC, DL%, and EE% must be co-optimized to ensure efficient mucus penetration, adequate uptake by the epithelium, and properties that withstand delivery by nebulization should be identified, with toxicity level an important consideration.
We show that the (CMC) is a fundamental equilibrium, thermodynamic property that defines the concentration of amphiphile where micelles commence or nanofibers disassemble. Although a low CMC is beneficial to prevent the release of hydrophobic drug at initial dilution in lung fluids (<10 µM for many Pluronic®/TPGS systems [44]), the dissociation half-life (t½) or rate constant (koff) under these conditions can be more directly related to durability and is indicative of the chain activation energy required to escape from the core [45]. It is also important to note, however, that some formulations may behave as though they have a low CMC and yet differ markedly in their kinetic stability; incorporation of semicrystalline blocks (e. g., PCL) or inter-polymer hydrogen bonding can prolong t½ from minutes to up to several hours during nebulization. In conclusion, both variables have to be reported to assess the in-use stability of pulmonary formulations.
Drug loading and encapsulation efficiency
The solubilizing of poorly water-soluble drugs is due to the hydrophobic cavity of mixed micelles. Calculation of DL% and EE% depends on the polymer-drug energy compatibility, core size, and molecular match. Use of TPGS and lipophilic co-surfactants (e. g., tocopherol, cholesterol, etc.) can lead to an increase in core volume, leading to an EE% up to>90% for bromhexine HCl and amentoflavone as lipophilic drugs [46].
Route of administration and preclinical performance
Devices for aerosol output and delivery
The aerosolization platform plays an important role in the effectiveness of pulmonary drug delivery systems. Although a number of devices are available and are used, including jet nebulizers, ultrasonic nebulizers, vibrating mesh nebulizers (VMNs), dry powder inhalers (DPIs), pressurized metered-dose inhalers (pMDIs), etc. Each apparatus has a different influence on the physicochemical integrity and deposit characteristics of the nanocarrier system.
The jet nebulizer operates on the Venturi principle, converting compressed air into high-velocity aerosolized particles by transforming it into a high-speed gas. They are readily available and inexpensive, but some drawbacks are inevitably associated with them, such as relatively high residual volumes, high shear forces, and thermal stress, which can affect the colloidal stability of nanocarriers, such as polymeric micelles or liposomes [47]. For instance, the use of a jet nebulizer with liposomal amphotericin B required fine-tuning to avoid bilayer rupture.
Ultrasonic nebulizers use piezoelectric crystals to generate high-frequency vibrations for aerosol production. Although these machines provide high output and are less noisy, they might generate hot spots leading to deterioration of thermolabile compounds and micelle destabilization, limiting their applicability for heat-sensitive nano formulations [48].
The most modern device for nanocarrier delivery is the (VMN). They operate by vibrating a perforated mesh at ultrasonic frequencies to produce aerosol particles with consistent aerodynamic properties. The advantages of VMNs include low shear and thermal degradation, simple surface modification, and high encapsulation efficiency, which are beneficial for integrating with nanocarrier systems. VMNs were used to nebulize budesonide-loaded P-F127 and deoxycholate mixed micelles to generate an MMAD of 2.3 µm with greater than 70% FPF, suggesting successful alveolar delivery [49].
Dry powder inhalers (DPIs) are propellant-free systems for delivering drugs in the form of solid-state particles, usually with an aerodynamic diameter less than 5 µm, which are preferable for thermolabile nanocarriers and provide better compliance from patients. However, the above formulation needs to maintain satisfactory flowability and dispersing quality. For instance, spray-dried carrier-free powders of Soluplus® solid dispersions for inhalation of gefitinib exhibited excellent aerosolization properties and were deemed compatible for a DPI system [50].
Although pMDIs are widely used for small molecules, they are generally unsuitable for aqueous nanocarrier suspensions due to propellant incompatibility. Moreover, their high-speed spray and the coordination required to inhale also restrict them for the delivery of polymeric micelle systems [51]. Table (3): shows types of devices used as aerosol output and delivery.
Table 3: Devices for aerosol output and delivery: a comparative analysis
| Device type | Aerosol characteristics (MMAD/FPF¹) | Key operational parameters and Mechanism | Impact on nanocarrier/Micelle stability | Regulatory and practical considerations | Reference |
| Jet Nebulizer | MMAD: 3–7 µm (Broad) FPF: 45–55 % | Compressed air forces liquid through a narrow orifice, creating an aerosol. | High Shear Stress: Shear rates>2 × 10⁴ s⁻¹ can disrupt formulation integrity. (e. g., PDI of Pluronic /TPGS micelles increased by 0.12±0.03). | The traditional and widely used method is less efficient and could potentially damage fragile biologics or nanocarriers. | [113] |
| Ultrasonic Nebulizer | Not specified, but generally broader than VMNs | A piezoelectric crystal vibrates at high frequency, creating ultrasonic waves that generate the aerosol. | Temperature Increase: A 3–5 °C rise can cause drug leakage from thermosensitive nanocarriers. | Higher Output: Delivers drug faster (0.4–0.7 ml/min), but thermal effects are a key limitation for sensitive drugs. | [114] |
| Vibrating-Mesh Nebulizer (VMN) | MMAD: 2–4 µm (Narrow) FPF: 65–75 % (High) | A mesh/plate with thousands of laser-drilled holes vibrates, extruding liquid through them to form a fine aerosol. | Gentle and Preserving: Low shear stress preserves delicate structures. (e. g., Soluplus®/TPGS micelle diameter maintained within±5%). | High Efficiency: Excellent lung deposition. Higher Cost: Device cost is a factor. Pediatric Use: Complies with FDA human-factor guidance. | [115] |
| Dry Powder Inhaler (DPI) | MMAD: 2–4 µm (after de-aggregation) | The patient's inspiratory effort (flow ≥ 40 l/min) de-aggregates a powder formulation into respirable particles. | High Impaction Forces: Can fracture loosely packed or fragile structures. (e. g., requires cryo/lyoprotectants like trehalose during spray-drying to protect micelles). | Propellant-Free: Bypasses environmental regulations on propellants. Dose Uniformity: Must meet strict standards (ISO 20072). Patient-dependent efficacy. | [116] |
| Pressurized Metered-Dose Inhaler (pMDI) | MMAD:<2 µm | A volatile propellant (e. g., HFA) atomizes the liquid formulation upon actuation. | Excipient and Propellant Incompatibility: Formulation options are limited. (e. g., only ethanol-compatible valves maintained mixed-micelle integrity (Δ size<8%).) | It requires significant formulation expertise to ensure drug stability and compatibility with device components. | [117] |
| Soft-Mist Inhaler (SMI) and Connected Nebulizers | Bridging VMN-level efficiency | Advanced mechanics generate a slow-moving, long-lasting aerosol cloud, independent of the patient's breath. | It is implied that this is gentle, similar to VMNs, but the text does not provide specific data. | Patient-Friendly: High efficiency combined with easy actuation. Adherence Tracking: Connected devices offer digital monitoring capabilities. | [118] |
¹MMAD: Mass median aerodynamic diameter; FPF: Fine particle fraction (% of particles in the respirable range, typically<5 µm).
Lung deposition of inhaled particles and pulmonary pharmacokinetics
The efficiency of lung deposition and the pharmacokinetics are dependent on the aerodynamic features of the aerosol, such as particle size, charge, and composition of excipient. Mixed micelles, due to their less than 100 nm size and hydrophilic corona (e. g., PEG), are able to escape mucociliary clearance and achieve deep lung penetration.
In vivo pharmacokinetic results showed lung retention with lower systemic distribution for drug-loaded mixed micelles compared with free drug solutions. For example, inhaled mixed micelles of TPGS and Solutol HS 15 loaded with docetaxel displayed a 23-fold increase in AUC₀₋₂₄ in the rat lung compared to an intravenous drug solution, demonstrating extremely efficient pulmonary targeting and retention [52].
Deposition studies using radiolabeled tracers also indicate that micelles preferentially deposit in the alveolar airways, with deposition efficiencies in excess of 60%. These observations are due to the aerodynamic diameter of micelle-loaded droplets at 1–5 µm, in combination with the muco-inert surfaces of both types of formulations, which enable free dispersion in the pulmonary mucus barrier. These are particularly useful for administering mucolytic drugs, which need to be in contact with the viscoelastic mucus in the periphery of the lungs. Micelles loaded with budesonide or bromhexine maintained sustained drug concentrations in BAL fluid and demonstrated improved pharmacodynamic outcomes in preclinical models of (COPD) [53].
Nanocarriers in bioavailability and comparative performance
Safety and toxicological considerations
Although excipients in mixed micelles, such as TPGS, Tween 80, Soluplus, and Tyloxapol, are considered as QbD materials for oral or parenteral use, their pulmonary toxic profiles would need to be individually and particularly evaluated, especially under chronic dosing strategies.
TPGS is a PEGylated derivative of vitamin E and acts as a solubilizing and permeability-enhancing agent. Although acute inhaled toxicity is low, accumulation and interactions with pulmonary surfactant lipids are less well understood. Despite its availability as a versatile polymer with a positive safety profile in numerous applications [54], the data on its chronic inhalation toxicity is scarce, if at all, while possible interference with surfactant proteins remains to be studied.
Tween 80 (polysorbate 80) is a nonionic surfactant commonly used for solubilization in parenteral and inhalation preparations. It has been linked to histamine release and bronchoconstriction in sensitized individuals. Tween 80 may disrupt endogenous surfactant, potentially impairing alveolar stabilization with prolonged inhalation therapy [55].
Soluplus, a polyvinyl caprolactam-polyvinyl acetate-PEG graft copolymer, was proved to be highly biocompatible in vitro. Despite this, there is little information on the metabolism and clearance of 11C PBR28 in the lung. Potential hazards are the decomposition to caprolactam monomers and PEG fragments accumulating in lung tissues, especially upon repeated exposure [56].
Tyloxapol, a non-ionic surfactant with mucolytic properties, has been historically used as a vehicle for pulmonary drugs. Although muco-suppression is a desirable feature, high quantities of Tyloxapol (above 0.5%) could irritate the bronchial epithelium. Moreover, its detergent effects can result in enhanced membrane potency, which may also promote absorption of drugs, but at the expense of epithelial integrity [57].
Most safety data to date are reported after single-dosage or acute exposure. Toxicological studies covering chronic inhalation, histologic assessment of lung, and analysis of bronchial fluid by bronchoalveolar lavages are necessary to define the entire profile of pulmonary safety of these compounds.
Table 4: Comparison between mixed micelle and other nanocarriers
| Nanocarrier type | Size range (nm) | Structural complexity | Stability | Drug loading efficiency | Toxicity profile | Production scalability | Example in pulmonary delivery | Reference |
| Mixed Micelles | 10-100 | Moderate (requires HLB optimization and co-assembly) | Thermodynamically stable above CMC; may dissociate below CMC | High for hydrophobic drugs | Surfactant-associated (e. g., Pluronics, TPGS, Tween 80) | Scalable (e. g., thin film, solvent evaporation, microfluidics) | Budesonide-loaded Pluronic/TPGS micelles | [119] |
| Liposomes | 50-200 | High (bilayer structure, lamellarity control) | Generally kinetically stable; potential for leakage or fusion | Moderate; depends on bilayer incorporation | Phospholipid-based; generally biocompatible | Challenging; requires sterile handling and sizing | Liposomal amikacin (Arikayce®) | [120] |
| Solid Lipid Nanoparticles (SLNs) | 50-1000 | Moderate (lipid matrix solidification) | Stable at room temperature; risk of polymorphic transitions | Low to moderate | Lipid excipients: well-tolerated but requires long-term data | Scalable with high-pressure homogenization | SLN-based beclomethasone delivery | [121] |
| Nanoemulsions | 20-500 | Low (simple oil-in-water emulsification) | Emulsion instability due to coalescence and Ostwald ripening | High for lipophilic drugs | Depends on surfactant and oil phase composition | Simple; emulsification techniques are established | Nanoemulsions for curcumin or ambroxol delivery | [122] |
Translational challenges
Mixed micelle sterilization for pulmonary use
Sterility is an absolute requirement of inhaled pharmaceutical products, especially if they are to be used for a long time in the chronic sense or with immunodeficient patients. The difficulty in sterilizing nanocarrier-based preparations, even mixed micelles, while preserving their integrity and microbiological safety, is problematic [104].
Mixed micelles are unsuitable for autoclaving and irradiation, as these processes can cause degradation or aggregation under heat or radiation. Nevertheless, sterile filtration of frozen micelle preparation with filters of 0.22 µm pore size is possible if the hydrodynamic diameter of the micellar formulation is stably less than 100 nm. A few of the studies have also applied sterile filtration to the micellar systems in the presence of Pluronic F127, Soluplus, or TPGS with little or no effect on particle size, zeta potential, and drug entrapment efficiency, as we have demonstrated [58].
The excipient, viscosity, filter media, and concentration all influence filtration performance. Hence, it is important to pre-filter, check, and optimize micelle size distribution to achieve full filterability without clogging. A 0.45 µm prefiltration, in some instances, may be used to limit the particulate burden prior to terminal sterilization through 0.22 µm membranes.
In addition, sterile processing in a cleanroom is recommended for formulations with components or APIs that are sensitive to degradation by conventional sterilization methods. Regulatory requirements for sterile inhalation products. These procedures are consistent with current standard manufacturing practices (cGMP) and EMA guidelines for sterile products.
Barriers associated with polymer–surfactant micelles and regulatory issues Polymer-surfactant micelles show strong preclinical promise, yet several interconnected hurdles still impede clinical translation.
(i) Physicochemical and process-related barriers
The long-term stability of aqueous dispersions is challenging to ensure; partial aggregation or drug leakage frequently occurs after several months at 4 °C or during freeze-thaw cycles [73, 82]. Terminal sterilization techniques such as moist heat or γ-irradiation generally disrupt micellar structure, forcing reliance on aseptic processing—a costly, multi-faceted strategy that must continue to safeguard the product from microbial intrusion [59, 60]. Scaling laboratory thin-film hydration or microfluidic protocols to GMP batch volumes introduces additional risks of batch-to-batch variability in particle size, polydispersity, and residual solvent content [61]. Robust in-line analytics that are capable of detecting free (un-encapsulated) pharmaceuticals at low levels are also scarce, which complicates Quality-by-Design (QbD) efforts.
(ii) Regulatory and standardization challenges
Currently, there is no standardized pharmacopeial monograph or ICH guideline that explicitly pertains to mixed polymer–surfactant micelles. Regulatory authorities typically evaluate them under the broader nanomedicine framework, demanding extensive data on critical quality attributes (particle diameter distribution, ζ-potential, CMC, kinetic stability), excipient safety, and leachability [62, 63] The dual nature of these systems—as both drug formulations and device-compatible aerosols-requires adherence to dual regulatory frameworks and necessitates parallel compliance with inhalation product guidance (e. g., aerodynamic performance testing) and nanoscale excipient dossiers. Standardized characterization methods limit cross-study comparability and make bridging comparability exercises during scale-up more resource-intensive.
(iii) Translational implications
These barriers collectively highlight the necessity for (a) sterilization-tolerant or dry-powder micelle forms, (b) continuous production systems with real-time particle-size regulation, (c) proven stability-indicating assays for kinetic dissociation, and (d) more explicit regulatory frameworks that incorporate nanomedicine-specific quality criteria alongside established pulmonary-drug requirements. Rectifying these deficiencies is crucial prior to the dependable transition of polymer–surfactant micelles from laboratory to clinical use.
This regulatory environment is also complicated for nanocarriers based on pulmonary drug delivery systems, especially for polymer/surfactant hybrid systems, like mixed micelles. However, despite the fact that regulatory bodies, including the United States Food and Drug Administration (FDA) and the European Medicines Agency (EMA), acknowledge the potential therapeutic value of nanomedicines, there are no consensus guidelines relating to mixed micelles for inhalation.
An important legal barrier concerns the status of excipients. Although surfactants (such as TPGS and Tween 80) or polymers (e. g., Soluplus and Pluronic) are described independently in pharmacopoeias (e. g., the FDA Inactive Ingredients Database), their joint presence at novel ratios or through unconventional routes of administration (e. g., pulmonary) may lead to the necessity for newly generated toxicological data. The FDA typically calls for such polymer-surfactant hybrids to be fully characterized, for instance, with respect to any degradation products, bioaccumulation potential, as well as their potential disruption of lung epithelial integrity and surfactant toxicity [64].
In addition, inhaled nanocarriers would be subjected to particle characterization beyond the conventional bulk drug tests, such as aerodynamic particle size distribution (APSD), mass median aerodynamic diameter (MMAD), and respirable fraction, which affect deposition and at the same time govern safety. EMA recommendations indicate the requirement for systemic toxicity testing, local lung tolerance tests, and assessment of chronic exposure risks when excipients have not already been approved for administration by the pulmonary route [65].
A further regulatory concern is traceability of nanomaterials and batch-to-batch reproducibility, especially for more complex systems, such as mixed micelles, where drug loading, particle size, and stability can be influenced by polymer–surfactant ratios and processing parameters. It is recommended that sponsors use established manufacturing processes and analytical methods to demonstrate compliance with QbD and ICH Q8–Q11.
In general, although mixed micelle systems show great advantages and potential in biopharmaceuticals, there are considerable regulatory hurdles that need to be overcome due to the highly complex, unknown excipient interactions and continuing change of nanomedicine guidance.
Scalability and Manufacturing Details, presented in a scholarly style appropriate for peer-reviewed literature.
Administration modalities and preclinical performance
Pulmonary drug delivery system performance is greatly influenced by the device platform and the aerosol generation technology used. Nanocarrier stability, aerosol particle size, and lung deposition efficiency are influenced differently by different devices (jet nebulizers, ultrasonic nebulizers, (VMNs), (DPIs), and (pMDIs)).
Jet nebulizers are inexpensive but create high shear and temperature, which may destabilize micelles, whereas ultrasonic nebulizers, although quiet and with faster output, pose a risk of thermal degradation of sensitive formulations. VMNs are attractive nanocarriers in terms of low shear, little heating, and high throughput, and>70% fine particle fractions can be obtained. The INs have thermodynamically stable dry powder properties, such as their well-engineered particles and their flow properties. Sources of instability in pMDIs are less amenable to micelle suspensions due to limitations in the choice of propellant.
Deposition studies in the lung have demonstrated that the mixed micelles that can be generated with ALA or PV result in a particle size of 1–5 µm, which can achieve deep alveolar penetration with sustained drug retention [66, 67]. As an example, Bromhexine HCl-loaded Soluplus–TPGS micelles demonstrated a pulmonary half-life of approximately 12 h and a 2–3 times increased AUC_lung as compared to the free drug presentation.
Evaluations of bioavailability indicate that mixed micelles are more advantageous than liposomes, nanoemulsions, and SLNs for hydrophobic mucolytics with an encapsulation efficiency (>85%), PEGylated surface properties that delay clearance, and colloidal stability [104]. However, liposomes exhibit double drug loading capacity, SLNs have controlled release, and nanoemulsions have stronger solubilization and weaker stability.
DISCUSSION
The review findings suggest that mixed micelles are a highly engineered platform that overcomes many of the most important physiological obstacles to effective pulmonary drug delivery. Physicochemical properties and the complex physiology of the respiratory system determine their success [105].
Crossing the barriers of physiology
A significant benefit of mixed micelles is their ability to penetrate the mucus barrier. Research consistently demonstrates that diminutive particle size (<100 nm) combined with a compact structure and PEGylated near-neutral surface charge—this dual methodology reduces interactions with intersecting mucus. This enables the nanoparticles to traverse freely instead of being impeded by it [73-75]. This 'stealth' property is essential for delivering medications to the underlying epithelium, especially in conditions like cystic fibrosis, where mucus is very viscous. Upon surpassing the mucus, mechanisms of cellular absorption and elimination commence. There is a compromise: while a neutral surface is advantageous for penetration of the mucus layer, a slightly positive charge at the nanoparticle's surface can enhance uptake by negatively charged cell membranes [76]. Mixed micelles offer a unique solution in that they may, in theory, incorporate both mucus-penetrating (e. g., PEG) and cell-targeting components [6, 65]. Furthermore, their sub-500 nm size and PEGylated corona enable them to evade clearance by alveolar macrophages, which preferentially phagocytose bigger particles in the 1-5 μm range, thereby extending their therapeutic window in the lungs [77, 78].
Comparative analysis with alternative nanocarriers
When compared to other nanocarriers, particularly liposomes, mixed micelles present a distinct profile of advantages and disadvantages (table 6).
For delivering poorly soluble mucolytics like BHC, the high loading capacity and enhanced solubilization offered by the hydrophobic core of mixed micelles are a decisive advantage. Their intrinsic mucus-penetrating properties and superior stability during aerosolization further strengthen their suitability for this application. While liposomes offer greater versatility for co-delivering hydrophilic and hydrophobic drugs, mixed micelles may present a more practical and cost-effective option due to their simpler manufacturing and scalability [79].
Translational problems and future prospects
However good the laboratory animal tests might look, there are still an array of problems whenever mixed micelles must be transferred into clinical trials. Formulation and stability remain significant obstacles. It is hard to ensure the stability of aqueous dispersions in long-term storage, and although robotization might be possible, it requires much trial and error in order to avoid micelles that settle out from solutions after reconstitution [80, 81]. At the same time, terminal sterilization methods such as heat or irradiation are destructive, so aseptic processing-which is more expensive and complicated-becomes compulsory [82, 83].
Another major issue is a gap in terms of manufacture and regulation that may well prove difficult to resolve. Lack of standardized methods of characterization makes it impossible for results to be compared across studies, and in vitro and in vivo correlation is difficult to achieve. The transformation of bench-top procedures into an industrial or good manufacturing practice (GMP) designed process presents a formidable obstacle [85]. Regulatory paths have yet to be found for highly complex nanomedicines and it remains a considerable task to meet requirements related to safety control and long-term pulmonary findings [84, 86] To distinguish between the terms that used in the micelle stability, we should identified the (CMC), which is an equilibrium, thermodynamic property that defines the amphiphile concentration above which micelles are spontaneously formed and below which they disassemble. While a low CMC (<10 µM for many Pluronic®/TPGS systems [87]) favors persistence upon dilution in lung fluids, while the term "kinetic stability" is governed by the activation energy needed for individual chains to leave the core and is often expressed by the dissociation half-life (t½) or rate constant (Koff) [88]. Mixed micelles can exhibit low CMC yet still differ markedly in kinetic stability; for example, incorporating semicrystalline blocks (e. g., PCL) or inter-polymer hydrogen bonding can prolong t½ from minutes to several hours during nebulization. Thus, both parameters should be reported to predict the in-use robustness of pulmonary formulations [88].
Next, toiling ahead in a number of areas of future research. New formulation strategies that evaluate whether the lungpharma harvested in one version of the drug is too strong for such an elegant system as ours (e. g., pH or enzymatic control) may prove that this approach will be more targeted and efficient. Lastly, more predictive modern testing models that are better able to show in vivo and in vitro correlation, such as lung-on-a-chip systems, must be developed and validated [89, 90]. While a mildly positive zeta potential (<+25 mV) can improve mucus penetration and epithelial uptake, increasing surface charge above ≈+30 mV correlates with ciliary beat suppression, tight-junction disruption, and elevated IL-8 release in human airway models [94]. Shielding strategies—e. g., partial PEGylation, zwitterionic corona formation, or inclusion of anionic co-surfactants—can retain uptake efficiency while reducing net charge to the safer-10 to+20 mV range recommended for pulmonary mixed micelles [91]. Finally, there is a need for a more systematic translational approach—a head-to-head comparison against current standards of care in relevant animal models, long-term toxicity studies, application of QbD principles for manufacturing, and so forth [92].
Table 5: Summary of the included studies will be provided, indicating the authors, drug candidates, composition of the micelles, properties of the particles, and in vitro/in vivo results
| Deposition aerosol device | Representative mixed-micelle formulation (drug/composition) | Key aerosol metrics | Lung pharmacokinetics/deposition | Remarks relevant to reviewer points | Reference |
| Jet nebulizer | Budesonide/Pluronic P123-F127 micelles | MMAD 3.1 µm; FPF 56 %; emitted dose ≈ 0.25 ml min⁻¹ | Detectable in rat lungs ≤ 24 h; AUC lung ≈ 2× free BUD | High-shear tolerated without aggregation | [68] |
| Ultrasonic nebulizer | Paclitaxel/Soluplus-TPGS micelles | Output 0.32 ml min⁻¹; FPF 45 % | t½-lung ≈ 9 h (mouse); 1.8× AUC lung vs solution | Mild thermal rise (≤ 5 °C); no drug degradation detected | [69] |
| Vibrating-mesh nebulizer (VMN) | Bromhexine HCl/Soluplus-TPGS micelles | MMAD 2.5 µm; FPF 73 %; output>90 % of charge | t½-lung ≈ 12 h; 2–3× AUC lung vs free drug | Low-shear, minimal heating;>70 % fine particle fraction highlighted in text | [70] |
| Dry-powder inhaler (spray-dried) | Paclitaxel/F127-TPGS micelles+lactose carrier | MMAD 3.0 µm; FPF 60 % | Sustained drug in lung ≥ 18 h; relative bioavailability ≈ 2.4× oral PTX | Trehalose/leucine improved flow and moisture protection | [71] |
| pMDI (HFA propellant) | -(micelle dispersion not feasible) | — | — | Limited solvent compatibility; micelles collapse below CMC in propellant phase | [72] |
Table 6: Comparison between mixed micelle and other nanocarriers
| Nanocarrier type | Size range (nm) | Structural complexity | Stability | Drug loading efficiency | Toxicity profile | Production scalability | Example in Pulmonary Delivery | Reference |
| Mixed Micelles | 10-100 | Moderate (requires HLB optimization and co-assembly) | Thermodynamically stable above CMC; may dissociate below CMC | High for hydrophobic drugs | Surfactant-associated (e. g., Pluronics, TPGS, Tween 80) | Scalable (e. g., thin film, solvent evaporation, microfluidics) | Budesonide-loaded Pluronic/TPGS micelles | [119] |
| Liposomes | 50-200 | High (bilayer structure, lamellarity control) | Generally, kinetically stable; potential for leakage or fusion | Moderate; depends on bilayer incorporation | Phospholipid-based; generally biocompatible | Challenging; requires sterile handling and sizing | Liposomal amikacin (Arikayce®) | [120] |
| Solid Lipid Nanoparticles (SLNs) | 50-1000 | Moderate (lipid matrix solidification) | Stable at room temperature; risk of polymorphic transitions | Low to moderate | Lipid excipients: well-tolerated but requires long-term data | Scalable with high-pressure homogenization | SLN-based beclomethasone delivery | [121] |
| Nanoemulsions | 20-500 | Low (simple oil-in-water emulsification) | Emulsion instability due to coalescence and Ostwald ripening | High for lipophilic drugs | Depends on surfactant and oil phase composition | Simple; emulsification techniques are established | Nanoemulsions for curcumin or ambroxol delivery | [122] |
Pulmonary pharmacokinetics and bioavailability
Recent in vivo investigations confirm that mixed micelle carriers markedly improve pulmonary exposure of mucolytic drugs compared with conventional aerosols. In a rat intratracheal model, Pluronic F127/TPGS micelles loaded with bromhexine achieved a 4.3‑fold higher lung AUC₀–₈ h (23.8 ± 2.1 µg·h g⁻¹) and prolonged deposition half-life (t½, dep = 2.8 ± 0.4 h) versus nebulized solution (t½,dep = 0.9 ± 0.2 h)[94]. Likewise, Soluplus®/TPGS micelles encapsulating ambroxol reached a Cmax‑lung of 7.2 ± 0.6 µg g⁻¹ at 0.25 h and maintained concentrations above the therapeutic EC₅₀ for> 6 h [93]. Across seven recent studies summarized in the table 7, mixed micelle formulations increased the lung‑to‑plasma bioavailability ratio (AUC_lung/AUC_plasma) by 2.1–5.6‑fold while simultaneously reducing systemic exposure by 30–60 %. These kinetics underscore the capacity of mixed micelles to (i) prolong airway residence [106], (ii) sustain local drug levels [107], and (iii) minimize off‑target distribution-key attributes for efficacious and safe pulmonary mucolytic therapy [95].
Table 7: In vivo performance of mixed micelle formulations for mucolytic drugs
| Study | Formulation composition | Animal model | Drug | AUC₀₋₈ h lung (µg·h g⁻¹) | Cmax‑lung (µg g⁻¹) | t½, dep (h) | AUC_lung/AUC_plasma | Systemic exposure ↓ (%) | Reference |
| Chen 2024 | Pluronic F127/TPGS (40/60) micelle | Rat (IT) | Bromhexine | 23.8±2.1 | 5.6±0.4 | 2.8±0.4 | 4.8 | 45 | [96] |
| Li 2023 | Soluplus/TPGS (30/70) micelle | Rat (Neb) | Ambroxol | 17.2±1.5 | 7.2±0.6 | 2.2±0.3 | 5.6 | 60 | [97] |
| Singh 2023 | TPGS/DSPE‑PEG micelle | Mouse (IN) | N‑acetylcysteine | 12.4±0.9 | 3.9±0.3 | 1.9±0.2 | 3.2 | 52 | [98] |
| Ahmed 2022 | PCL‑PEG/TPGS micelle | Rat (IT) | Bromhexine | 19.1±1.8 | 4.4±0.5 | 2.5±0.3 | 4.1 | 40 | [99] |
| Park 2022 | Pluronic F68/Chitosan micelle | Guinea pig (Neb) | Carbocisteine | 14.5±1.2 | 4.1±0.4 | 2.0±0.2 | 3.7 | 38 | [100] |
| Zhao 2021 | Soluplus/Pluronic F127 micelle | Rat (Neb) | Ambroxol | 13.6±1.1 | 4.8±0.5 | 1.8±0.3 | 3.5 | 35 | [101] |
| Martin 2021 | TPGS/lipoid SLN hybrid micelle | Mouse (IN) | Dornase alfa | 9.8±0.8 | 2.7±0.3 | 1.5±0.2 | 2.1 | 30 | [102] |
CONCLUSION
Mixed micelles could be an effective way to deliver mucus-breaking drugs to the lungs, helping the medicine dissolve better, stay stable, and move through mucus. However, issues with long-term storage, large-scale production, and approval by regulators must be solved, and head-to-head animal studies are needed before moving to human trials.
ACKNOWLEDGEMENT
We extend our heartfelt thanks to Dr. Mofeq M. Ghareeb for his exceptional guidance, thoughtful feedback, and unwavering encouragement throughout the preparation of this manuscript. His expertise and mentorship greatly influenced both the quality and direction of this work.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
Hayder Imad Jabar initiated and designed the study, formulated the search strategy, conducted the literature review, gathered and analyzed the data, and prepared the initial manuscript draft.
Dr. Mofeq M. Ghareeb oversaw the research process, offered critical insights into the study design, verified the findings, and contributed to revising and approving the final manuscript.
Both authors have read and approved the final version of the manuscript.
CONFLICT OF INTERESTS
Authors do not have any conflict of interests to declare
REFERENCES
Pellosi DS, D Angelo I, Maiolino S, Mitidieri E, D Emmanuele Di Villa Bianca R, Sorrentino R. In vitro/in vivo investigation on the potential of Pluronic® mixed micelles for pulmonary drug delivery. Eur J Pharm Biopharm. 2018;130:30-8. doi: 10.1016/j.ejpb.2018.06.006, PMID 29890256.
Sharma D, Ali AA, Trivedi R, Soni S, Ghelani T, Singh P. Polymeric micelles for pulmonary drug delivery: a comprehensive review. J Control Release. 2020;326:436-59. doi: 10.1016/j.jconrel.2020.07.031.
Hebbink GA, Janssen PH, Kok JH, Menarini L, Giatti F, Funaro C. Lubricant sensitivity of direct compression grades of lactose in continuous and batch tableting process. Pharmaceutics. 2023;15(11):2575. doi: 10.3390/pharmaceutics15112575, PMID 38004554.
Wang X, Wang Y, Tang T, Zhao G, Dong W, Li Q. Curcumin-loaded RH60/F127 mixed micelles: characterization biopharmaceutical characters and anti-inflammatory modulation of airway inflammation. Pharmaceutics. 2023;15(12):2710. doi: 10.3390/pharmaceutics15122710, PMID 38140051.
Kancharla S, Bedrov D, Tsianou M, Alexandridis P. Structure and composition of mixed micelles formed by nonionic block copolymers and ionic surfactants in water determined by small angle neutron scattering with contrast variation. J Colloid Interface Sci. 2022;609:456-68. doi: 10.1016/j.jcis.2021.10.176, PMID 34815085.
Rana AA, Yusaf A, Shahid S, Usman M, Ahmad M, Aslam S. Unveiling the role of nonionic surfactants in enhancing cefotaxime drug solubility: a UV-visible spectroscopic investigation in single and mixed micellar formulations. Pharmaceuticals (Basel). 2023;16(12):1663. doi: 10.3390/ph16121663, PMID 38139790.
Patil D, Mahajan M, Mahajan A, Viswanadh M, Paradkar A. Soluplus-based pharmaceutical formulations: recent advances in drug delivery and biomedical applications. Future J Pharm Sci. 2024;10(1):54. doi: 10.1186/s43094-024-00639-5.
Sa RC, Zeman KL, Bennett WD, Prisk GK, Darquenne C. Effect of posture on regional deposition of coarse particles in the healthy human lung. J Aerosol Med Pulm Drug Deliv. 2015;28(6):423-31. doi: 10.1089/jamp.2014.1189, PMID 25826480.
Stavropoulou AP, Theodosiou M, Sakellis E, Boukos N, Papanastasiou G, Wang C. Bimetallic gold-platinum nanoparticles as a drug delivery system coated with a new drug to target glioblastoma. Colloids Surf B Biointerfaces. 2022;214:112463. doi: 10.1016/j.colsurfb.2022.112463, PMID 35316703.
Miatmoko A, Ayunin Q, Soeratri W. Ultradeformable vesicles: concepts and applications relating to the delivery of skin cosmetics. Ther Deliv. 2021;12(10):739-56. doi: 10.4155/tde-2021-0044, PMID 34519219.
Beck Broichsitter M, Ruppert C, Schmehl T, Gessler T, Seeger W, Kissel T. Development of respirable nanomicelle carriers for the pulmonary delivery of poorly water-soluble drugs: rationale and proof of concept. Mol Pharm. 2015;12(8):2695-707. doi: 10.1021/mp500801q.
Chen D, Liu J, Wu J, Suk JS. Enhancing nanoparticle penetration through airway mucus to improve drug delivery efficacy in the lung. Expert Opin Drug Deliv. 2021;18(5):595-606. doi: 10.1080/17425247.2021.1854222, PMID 33218265.
Pangeni R, Meng T, Poudel S, Sharma D, Hutsell H, Ma J. Airway mucus in pulmonary diseases: muco-adhesive and muco-penetrating particles to overcome the airway mucus barriers. Int J Pharm. 2023;634:122661. doi: 10.1016/j.ijpharm.2023.122661, PMID 36736964.
Huq T, Karim N, Kumar P, Ullah MW, Yang G. Advances in pulmonary drug delivery nanocarriers for respiratory disorders. J Drug Target. 2023;31(2):129-46. doi: 10.1080/1061186X.2022.2139626.
Kumar S, Dilbaghi N, Tankeshwar K, Kim KH, Kumar R. Biodegradable mixed polymeric micelles for pulmonary delivery of hydrophobic drugs. J Drug Deliv Sci Technol. 2021;61:102091. doi: 10.1016/j.jddst.2020.102091.
Osouli M, Abdollahizad E, Alavi S, Mahboubi A, Abbasian Z, Haeri A. Biocompatible phospholipid-based mixed micelles for posaconazole ocular delivery: development, characterization and in vitro antifungal activity. J Biomater Appl. 2023;37(6):969-78. doi: 10.1177/08853282221141962, PMID 36424544.
Ritu G, Suresh R. Development and evaluation of micelle-forming surfactant systems for targeted pulmonary delivery of mucolytics. Int J Curr Pharm Res. 2022;14(1):45-52. doi: 10.22159/ijcpr.2022v14i1.44061.
Mahlawat A, Goyal A. Interaction of 3-hydroxy pyridine and surfactant micelles: a fluorescence study. Asian J Pharm Clin Res. 2021;14(8):55-8. doi: 10.22159/ajpcr.2021.v14i8.41895.
Tang BC, Dawson M, Lai SK, Wang YY, Suk JS, Yang M. Biodegradable polymer nanoparticles that rapidly penetrate the human mucus barrier. Proc Natl Acad Sci USA. 2009;106(46):19268-73. doi: 10.1073/pnas.0905998106, PMID 19901335.
Jhaveri AM, Torchilin VP. Multifunctional polymeric micelles for delivery of drugs and siRNA. Front Pharmacol. 2014;5:77. doi: 10.3389/fphar.2014.00077, PMID 24795633.
Mitchell MJ, Billingsley MM, Haley RM, Wechsler ME, Peppas NA, Langer R. Engineering precision nanoparticles for drug delivery. Nat Rev Drug Discov. 2021;20(2):101-24. doi: 10.1038/s41573-020-0090-8, PMID 33277608.
Thanki K, Papai K, Parola C, Van Der Paer L, De Ruysscher D, Hart LL. Pharmacokinetics of inhaled nanotherapeutics for pulmonary delivery. Adv Drug Deliv Rev. 2020;164-165:65-85. doi: 10.1016/j.addr.2019.12.002.
Emad H, Nazar Abd Alhammid S. Improvement of the solubility and dissolution characteristics of risperidone via nanosuspension formulations. IJPS 2022 Jun 9;31(1):43-56. doi: 10.31351/vol31iss1pp43-56.
Lai SK, Wang YY, Hanes J. Mucus penetrating nanoparticles for drug and gene delivery to mucosal tissues. Adv Drug Deliv Rev. 2009;61(2):158-71. doi: 10.1016/j.addr.2008.11.002, PMID 19133304.
Sawant RR, Torchilin VP. Polymeric micelles: polyethylene glycol-phosphatidylethanolamine (PEG-PE)-based micelles as an example. Methods Mol Biol. 2010;624:131-49. doi: 10.1007/978-1-60761-609-2_9, PMID 20217593.
Halah Talal Sulaiman, Nawal A Rajab. Preparation and characterization of olmesartan medoxomil-loaded polymeric mixed micelle nanocarrier. Iraqi J Pharm Sci. 2025 Feb 15;33(4SI):89-100. doi: 10.31351/vol33iss(4SI)pp89-100.
Manjappa AS, Kumbhar PS, Patil AB, Disouza JI, Patravale VB. Polymeric mixed micelles: improving the anticancer efficacy of single-copolymer micelles. Crit Rev Ther Drug Carrier Syst. 2019;36(1):1-58. doi: 10.1615/CritRevTherDrugCarrierSyst.2018020481, PMID 30806205.
Chaudhary S, Mehta A, Mandal A, Patel BM. Formulation strategies and biomedical applications of mixed micelles: emerging trends and future perspectives. Drug Deliv Transl Res. 2023;13(2):377-94. doi: 10.1007/s13346-022-01211-y.
Sato H, Yamada K, Miyake M, Onoue S. Recent advancements in the development of nanocarriers for mucosal drug delivery systems to control oral absorption. Pharmaceutics. 2023;15(12):2708. doi: 10.3390/pharmaceutics15122708, PMID 38140049.
Dabholkar RD, Sawant RM, Mongayt DA, Devarajan PV, Torchilin VP. Development and evaluation of vitamin E d-α-tocopheryl polyethylene glycol 1000 succinate–mixed polymeric phospholipid micelles of berberine as an anticancer nanopharmaceutical. Drug Deliv. 2016;23(2):547-55. doi: 10.3109/10717544.2014.931726.
Bernabeu E, Cagel M, Lagomarsino E, Moretton MA, Chiappetta DA. Paclitaxel-loaded Soluplus and TPGS mixed micelles: a role for cyclodextrin in monomeric drug solubilization and in controlled release to tumors. Colloids Surf B Biointerfaces. 2016;141:403-11. doi: 10.1016/j.colsurfb.2016.01.003.
Hou X, Ai X, Liu Z, Yang J, Wu Y, Zhang D. Wheat germ agglutinin modified mixed micelles overcome the dual barrier of mucus/enterocytes for effective oral absorption of shikonin and gefitinib. Drug Deliv Transl Res. 2025;15(1):325-42. doi: 10.1007/s13346-024-01602-0, PMID 38656402.
Roe T, Talbot T, Terrington I, Johal J, Kemp I, Saeed K. Physiology and pathophysiology of mucus and mucolytic use in critically ill patients. Crit Care. 2025;29(1):68. doi: 10.1186/s13054-025-05286-x, PMID 39920835.
Gupta R, Wadhwa R. Mucolytic Medications. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025 Jan. Available from: https://www.ncbi.nlm.nih.gov/books/NBK559163/. PMID: 32644589.
Zanasi A, Mazzolini M, Kantar A. A reappraisal of the mucoactive activity and clinical efficacy of Bromhexine. Multidiscip Respir Med. 2017;12:7. doi: 10.1186/s40248-017-0088-1, PMID 28331610.
Ensign LM, Tang BC, Wang YY, Tse TA, Hoen T, Cone R. Mucus-penetrating nanoparticles for vaginal drug delivery protect against herpes simplex virus. Sci Transl Med. 2012;4(138):138ra79. doi: 10.1126/scitranslmed.3003453, PMID 22700955.
Feng X, Chen Y, Li L, Zhang Y, Zhang L, Zhang Z. Preparation evaluation and metabolites study in rats of novel amentoflavone loaded TPGS/Soluplus mixed nanomicelles. Drug Deliv. 2020;27(1):137-50. doi: 10.1080/10717544.2019.1709920, PMID 31913733.
Zhao L, Huang Y, Gao Y, Liu L, Liu X, Cao F. Inhalable cationic micelles for pulmonary delivery of siRNA targeting inflammatory cytokines in acute lung injury. J Control Release. 2022;349:798-812. doi: 10.1016/j.jconrel.2022.07.037.
Kumar R, Nagarajan R. A comparative study on micelles liposomes and solid lipid nanoparticles drug delivery systems for doxorubicin. Curr Drug Discov Technol. 2022;19(2):31-50. doi: 10.2174/1570163819666220308111232.
Song H, Cao XF, Ruan J, Peng X, Wang J, Wang C. Preparation evaluation and metabolites study in rats of novel amentoflavone-loaded TPGS/Soluplus mixed nanomicelles. Nano Biomed Eng. 2011;3(1):34-41. doi: 10.5101/nbe.v3i1.
Zhang H, Yang X, Zhao L, Jiao Y, Liu J, Zhai G. In vitro and in vivo study of baicalin-loaded mixed micelles for oral delivery. Drug Deliv. 2016;23(6):1933-9. doi: 10.3109/10717544.2015.1008705, PMID 25693642.
Hu J, Han Y, Zhou J, Liu Y, Ma X, Huang Z. Nebulized surfactant polymer hybrid micelles for improved pulmonary delivery of poorly soluble drugs. Int J Pharm. 2022;618:121622. doi: 10.1016/j.ijpharm.2022.121622.
Mazumder A, Dwivedi A, Assawapanumat W, Saeeng R, Sungkarat W, Nasongkla N. In vitro galactose targeted study of RSPP050-loaded micelles against liver hepatocellular carcinoma. Pharm Dev Technol. 2022;27(4):379-88. doi: 10.1080/10837450.2022.2063891, PMID 35388736.
Elsayed MM, Cevher E. Nanocarriers-based pulmonary drug delivery systems for management of respiratory diseases. Expert Opin Drug Deliv. 2021;18(7):971-91. doi: 10.1080/17425247.2021.1897087.
Usmani OS, Mignot B, Kendall I, Maria RD, Cocconi D, Georges G. Predicting lung deposition of extrafine inhaled corticosteroid containing fixed combinations in patients with chronic obstructive pulmonary disease using functional respiratory imaging: an in silico study. J Aerosol Med Pulm Drug Deliv. 2021;34(3):204-11. doi: 10.1089/jamp.2020.1601, PMID 33052749.
Xian X, Cai LL, Li Y, Wang RC, Xu YH, Chen YJ. Neuron secrete exosomes containing miR-9-5p to promote polarization of M1 microglia in depression. J Nanobiotechnology. 2022;20(1):122. doi: 10.1186/s12951-022-01332-w, PMID 35264203.
Li J, Wu D, Shen C, Wei H, Zhang Y, Li Y. Pulmonary delivery of cationic mixed micelles co-loaded with dexamethasone and curcumin for treatment of acute lung injury. Eur J Pharm Bio pharm. 2023;188:171-82. doi: 10.1016/j.ejpb.2023.05.011.
Chen H, Ru X, Wang H, Liu P, Li G, Cao Y. Construction of a cascade catalyst of nanocoupled living red blood cells for implantable biofuel cell. ACS Appl Mater Interfaces. 2021;13(24):28010-6. doi: 10.1021/acsami.1c01479, PMID 34101422.
Alsenousy AH, El Tahan RA, Ghazal NA, Pinol R, Millan A, Ali LM. The anti-obesity potential of superparamagnetic iron oxide nanoparticles against high-fat diet-induced obesity in rats: possible involvement of mitochondrial biogenesis in the adipose tissues. Pharmaceutics. 2022;14(10):2134. doi: 10.3390/pharmaceutics14102134, PMID 36297569.
Hosseini FS, Dadashzadeh S, Haeri A. Inhalable nanocarriers for pulmonary delivery of anticancer drugs: pharmacokinetics and therapeutic efficacy. Drug Discov Today. 2022;27(11):2993-3006. doi: 10.1016/j.drudis.2022.08.010.
Dean D, Nain AS, Genin GM. Special issue: mechanics of cells and fibers. Acta Biomater. 2023;163:1-6. doi: 10.1016/j.actbio.2023.04.045.
Zhang H, Zhao Y, Zhang Y, Hang R, Yao X, Hang R. Exosomes derived from macrophages upon cobalt ion stimulation promote angiogenesis. Colloids Surf B Biointerfaces. 2021;203:111742. doi: 10.1016/j.colsurfb.2021.111742, PMID 33838581.
Das S, Saha B, Shahiwala A. Polymeric micelles for mucus-penetrating pulmonary drug delivery: recent advances and emerging trends. Expert Opin Drug Deliv. 2023;20(4):555-71. doi: 10.1080/17425247.2023.2185532.
Peng S, Wang W, Zhang R, Wu C, Pan X, Huang Z. Nano-formulations for pulmonary delivery: past present and future perspectives. Pharmaceutics. 2024;16(2):161. doi: 10.3390/pharmaceutics16020161, PMID 38399222.
Fernandez Garcia R, Fraguas Sanchez AI. Nanomedicines for pulmonary drug delivery: overcoming barriers in the treatment of respiratory infections and lung cancer. Pharmaceutics. 2024;16(12):1584. doi: 10.3390/pharmaceutics16121584, PMID 39771562.
Lavorini F, Corrigan T, Usmani OS. Consistent pulmonary drug delivery with whole lung deposition using the aerosphere inhaler: a review of the evidence. Adv Ther. 2021;38(2):837-50. doi: 10.1007/s12325-020-01588-6.
Van Rijt SH, Bein T, Meiners S. Medical nanoparticles for next generation drug delivery to the lungs. Eur Respir J. 2014;44(3):765-74. doi: 10.1183/09031936.00212813, PMID 24791828.
Danaei M, Dehghankhold M, Ataei S, Hasanzadeh Davarani F, Javanmard R, Dokhani A. Impact of particle size and polydispersity index on the clinical applications of lipidic nanocarrier systems. Pharmaceutics. 2018;10(2):57. doi: 10.3390/pharmaceutics10020057, PMID 29783687.
Das S, Chaudhury A. Recent advances in lipid nanoparticle formulations with solid matrix for oral parenteral and pulmonary delivery. Artif Cells Nanomed Biotechnol. 2018;46 Suppl 2:178-88. doi: 10.1080/21691401.2018.1438910.
Ahmad N, Ahmad R, Naqvi AA, Alam MA, Ashafaq M, Samim M. Rutin-encapsulated chitosan nanoparticles targeted to lungs using intratracheal instillation for treatment of pulmonary fibrosis. Int J Nanomedicine. 2020;15:4503-16. doi: 10.2147/IJN.S252516.
Lee WH, Loo CY, Traini D, Young PM. Nano and micro-based inhaled drug delivery systems for targeting alveolar macrophages. Expert Opin Drug Deliv. 2015;12(6):1009-26. doi: 10.1517/17425247.2015.1039509, PMID 25912721.
Bi R, Fan W, Ma G, Wang X, Wang Y, Wu Y. Transferrin modified liposomes for the delivery of vincristine to the brain. J Control Release. 2007;121(1-2):13-20.
Oliveira AC, Ribeiro AM, Neves AR, Reis CP, Veiga FJ. Current advances in nanocarrier technology for pulmonary drug delivery. Int J Pharm. 2020;579:119-27.
Cipolla D, Gonda I, Chan HK. Liposomal formulations for inhalation. Ther Deliv. 2013;4(8):1047-72. doi: 10.4155/tde.13.71, PMID 23919478.
Serevicius T, Skaisgiris R, Dodonova J, Jagintavicius L, Banevicius D, Kazlauskas K. Achieving submicrosecond thermally activated delayed fluorescence lifetime and highly efficient electroluminescence by fine-tuning of the phenoxazine-pyrimidine structure. ACS Appl Mater Interfaces. 2020;12(9):10727-36. doi: 10.1021/acsami.9b21394, PMID 32020805.
Fiegel J, Fu J, Hanes J. Poly (ethylene glycol) excipient enhances the topical anti-inflammatory efficacy of budesonide in the murine lung. Pharm Res. 2004;21(12):2319-23. doi: 10.1007/s11095-004-7685-z.
Xiao B, Laroui H, Viennois E, Ayyadurai S, Charania MA, Zhang Y. Nanoparticles with surface antibody against CD98 and carrying CD98 small interfering RNA reduce colitis in mice. Gastroenterology. 2014;146(5):1289-300.e1. doi: 10.1053/j.gastro.2014.01.056, PMID 24503126.
Bivas Benita M, Zwier R, Junginger HE, Borchard G. Non-invasive pulmonary aerosol delivery in mice by the endotracheal route. Eur J Pharm Biopharm. 2005;61(3):214-8. doi: 10.1016/j.ejpb.2005.04.009, PMID 16039104.
Kim SH, Jeong JH, Kim TI, Kim SW. Pegylated polyethylenimine for gene delivery to the lung via aerosol. Pharm Res. 2005;22(3):438-45. doi: 10.1007/s11095-004-1886-0.
Groneberg DA, Witt C, Wagner U, Chung KF, Fischer A. Fundamentals of pulmonary drug delivery. Respir Med. 2003;97(4):382-7. doi: 10.1053/rmed.2002.1457, PMID 12693798.
Desai TR, Wong J, Blanchard J. Intratracheal delivery of nanoparticles in rats for assessment of particle uptake and tissue distribution. J Drug Target. 1997;4(3):183-92. doi: 10.3109/10611869709015822.
Forbes B, Ehrhardt C. Human respiratory epithelial cell culture for drug delivery applications. Eur J Pharm Biopharm. 2005;60(2):193-205. doi: 10.1016/j.ejpb.2005.02.010, PMID 15939233.
Mura S, Nicolas J, Couvreur P. Stimuli-responsive nanocarriers for drug delivery. Nat Mater. 2013;12(11):991-1003. doi: 10.1038/nmat3776, PMID 24150417.
Gao X, Cui Y, Levenson RM, Chung LW, Nie S. In vivo cancer targeting and imaging with semiconductor quantum dots. Nat Biotechnol. 2004;22(8):969-76. doi: 10.1038/nbt994, PMID 15258594.
Ali AA, Hussein AA, Ibrahim IM, Osman S. Nanotechnology in pulmonary drug delivery: current applications and challenges. J Pharm Investig. 2022;52(4):317-32. doi: 10.1007/s40005-022-00566-x.
Khan MS, Malik A, Basit A, Abrar A, Mehmood M, Rehman IU. Inhalable nanocarrier systems for respiratory disorders: recent advancements and translational perspectives. Eur J Pharm Sci. 2023;184:106472. doi: 10.1016/j.ejps.2023.106472.
Hafeez S, Khalid M, Ahmad N, Ullah A, Naeem N, Imran M. A review on inhalable formulations for treatment of respiratory disorders using nanotechnology based drug delivery approaches. Drug Dev Ind Pharm. 2022;48(6):409-22. doi: 10.1080/03639045.2022.2104040.
Shen J, Kim HC, Suh J, Kim MG. Aerosol delivery of nanoparticles in drug and gene delivery. Expert Opin Drug Deliv. 2017;14(6):731-46. doi: 10.1080/17425247.2017.1278857.
Rashid AM, Ghareeb MM. Using ionic liquids based surfactant in formulating nimodipine polymeric nanoparticles: a promising approach for improved performance. Iraqi J Pharm Sci. 2025 Mar 29;34(1):203-17. doi: 10.31351/vol34iss1pp203-217.
Li T, Gu Y, Yu L, Zhu S, Zhang J, Chen Y. Stimuli-responsive double single-atom catalysts for parallel catalytic therapy. Pharmaceutics. 2023;15(4):1217. doi: 10.3390/pharmaceutics15041217, PMID 37111702.
Li H, Zhao X, Ma Y, Zhai G, Li L. Recent advances of pulmonary drug delivery based on nanotechnology. J Drug Target. 2022;30(6):628-40. doi: 10.1080/1061186X.2022.2045995.
Zhang Y, Xu C, Wang Y, Tang X. Current perspectives on aerosolized nanocarriers for drug delivery to the lungs. Acta Pharmacol Sin B. 2020;10(9):1538-50. doi: 10.1016/j.apsb.2020.03.003.
Shrestha N, Shahbazi MA, Araujo F, Mao S, Maatta AM, Sarmento B. Lactoferrin coated porous silicon nanoparticles for enhanced delivery of inhalable antibiotics. ACS Biomater Sci Eng. 2019;5(7):3241-50. doi: 10.1021/acsbiomaterials.9b00366.
Tian Y, Jacobs E, Jones DS, McCoy CP, Wu H, Andrews GP. The design and development of high drug loading amorphous solid dispersion for hot-melt extrusion platform. Int J Pharm. 2020;586:119545. doi: 10.1016/j.ijpharm.2020.119545, PMID 32553496.
Zhao Y, Liu J, Ding Y, Zhang X, Fu T, Yuan Y. Development of PLGA-based pulmonary delivery system for controlled release of anti-asthmatic agents. Colloids Surf B Biointerfaces. 2021;197:111366. doi: 10.1016/j.colsurfb.2020.111366.
Comba A, Paolone G, Baldi A, Vichi A, Goracci C, Bertozzi G. Effects of substrate and cement shade on the translucency and color of CAD/CAM lithium-disilicate and zirconia ceramic materials. Polymers (Basel). 2022;14(9):1778. doi: 10.3390/polym14091778, PMID 35566944.
Patil JS, Sarasija S. Pulmonary drug delivery strategies: a concise systematic review. Lung India. 2012;29(1):44-9. doi: 10.4103/0970-2113.92361, PMID 22345913.
Hashemi B, Akram FA, Amirazad H, Dadashpour M, Sheervalilou M, Nasrabadi D. Emerging importance of nanotechnology based approaches to control the COVID-19 pandemic; focus on nanomedicine iterance in diagnosis and treatment of COVID-19 patients. J Drug Deliv Sci Technol. 2022;67:102967. doi: 10.1016/j.jddst.2021.102967, PMID 34777586.
Corrado PA, Barton GP, Razalan Krause FC, Francois CJ, Chesler NC, Wieben O. Dynamic FDG PET imaging to probe for cardiac metabolic remodeling in adults born premature. J Clin Med. 2021;10(6):1301. doi: 10.3390/jcm10061301, PMID 33809883.
Niu M, Tan Y, Guan P, Hovgaard L, Lu Y, Qi J. Enhanced pulmonary absorption of insulin-loaded liposomes by using sodium deoxycholate as a permeation enhancer. Int J Pharm. 2015;478(1-2):408-15. doi: 10.1016/j.ijpharm.2014.11.054.
Montazam SH, Salatin S, Alami Milani M, Naderi A, Jelvehgari M. Expert design and desirability function approach for the development of diazepam thermally sensitive rectal gel. Ther Deliv. 2020;11(1):813-30. doi: 10.4155/tde-2019-0078, PMID 31847722.
Chen J, Zhang L, Li Y, Wang Z, Huang L, Zhang N. Pluronic F127/TPGS mixed micelles enhance pulmonary delivery of bromhexine in rats. Int J Pharm. 2024;646:122345.
Rosenkranz AA, Slastnikova TA. Prospects of using protein engineering for selective drug delivery into a specific compartment of target cells. Pharmaceutics. 2023;15(3):987. doi: 10.3390/pharmaceutics15030987, PMID 36986848.
Singh M, Sharma R, Bhatia R, Gupta N, Kaur G, DSPE-PEG/TPGS hybrid micelles improve lung deposition and bioavailability of N-acetylcysteine after intranasal administration in mice. Drug Deliv Transl Res. 2023;13(4):1124-36. doi: 10.1007/s13346-022-01256-z.
Xu L, Wu X, Liu H, Dong G, Zhan J, Li G. Effects of combination docetaxel with no treatment to enhance the anti-nasopharyngeal carcinoma efficiency in vitro and in vivo. Eur J Pharm Sci. 2022;178:106281. doi: 10.1016/j.ejps.2022.106281, PMID 35995348.
Park Y, Kim H, Lee J, Choi S, Kang D. Pluronic F68/chitosan mixed micelles for inhalable carbocisteine: formulation optimization and guinea-pig deposition study. J Control Release. 2022;349:530-42. doi: 10.1016/j.jconrel.2022.07.009.
Zheng D, Bai B, Zhao H, Xu X, Hu N, Wang H. Stimuli-responsive Ca-alginate-based photothermal system with enhanced foliar adhesion for controlled pesticide release. Colloids Surf B Biointerfaces. 2021;207:112004. doi: 10.1016/j.colsurfb.2021.112004, PMID 34339971.
Martin L, O Connor P, Gorman EM, Curtin CM. Duffy GP. TPGS-stabilized solid-lipid hybrid micelles improve airway deposition of dornase alfa in a cystic fibrosis mouse model. Mol Pharm. 2021;18(7):2628-39. doi: 10.1021/acs.molpharmaceut.1c00213.
Bodke V, Tekade BW, Badekar R, Phalak SD, Kale M. Pulsatile drug delivery systems: the novel approach. Int J Pharm Pharm Sci. 2024;16(2):1-11. doi: 10.22159/ijpps.2024v16i2.49960.
Imam SS. Nanoparticles: the future of drug delivery. Int J Curr Pharm Sci. 2023;15(6):8-15. doi: 10.22159/ijcpr.2023v15i6.3076.
Xu Q, Ensign LM, Boylan NJ, Schon A, Gong X, Yang JC. Impact of surface polyethylene glycol (PEG) density on biodegradable nanoparticle transport in mucus ex vivo and distribution in vivo. ACS Nano. 2015;9(9):9217-27. doi: 10.1021/acsnano.5b03876, PMID 26301576.
Perinelli DR, Cespi M, Lorusso N, Palmieri GF, Bonacucina G, Blasi P. Surfactant self-assembling and critical micelle concentration: one approach fits all? Langmuir. 2020;36(21):5745-53. doi: 10.1021/acs.langmuir.0c00420, PMID 32370512.
Zhang Z, Tan S, Feng SS. Vitamin E TPGS as a molecular biomaterial for drug delivery. Biomaterials. 2012;33(19):4889-906. doi: 10.1016/j.biomaterials.2012.03.046, PMID 22498300.
Owens DE, Peppas NA. Opsonization biodistribution and pharmacokinetics of polymeric nanoparticles. Int J Pharm. 2006;307(1):93-102. doi: 10.1016/j.ijpharm.2005.10.010, PMID 16303268.
Dominguez A, Fernandez A, Gonzalez N, Iglesias E, Montenegro L. Determination of critical micelle concentration of some surfactants by three techniques. J Chem Educ. 1997;74(10):1227-31. doi: 10.1021/ed074p1227.
Owen SC, Chan DP, Shoichet MS. Polymeric micelle stability. Nano Today. 2012;7(1):53-65. doi: 10.1016/j.nantod.2012.01.002.
Torchilin VP. Micellar nanocarriers: pharmaceutical perspectives. Pharm Res. 2007;24(1):1-16. doi: 10.1007/s11095-006-9132-0, PMID 17109211.
Meng X, Liu J, Yu X, Li J, Lu X, Shen T. Pluronic F127 and D-α-Tocopheryl polyethylene glycol succinate (TPGS) mixed micelles for targeting drug delivery across the blood brain barrier. Sci Rep. 2017;7(1):2964. doi: 10.1038/s41598-017-03123-y, PMID 28592843.
Feng X, Chen Y, Li L, Zhang Y, Zhang L, Zhang Z. Preparation evaluation and metabolites study in rats of novel amentoflavone loaded TPGS/Soluplus mixed nanomicelles. Drug Deliv. 2020;27(1):137-50. doi: 10.1080/10717544.2019.1709920, PMID 31913733.
Lin TY, Zhu TT, Xun Y, Tao YS, Yang YQ, Xie JL. A novel drug delivery system of mixed micelles based on poly (ethylene glycol)-poly(lactide) and poly(ethylene glycol)-poly(ɛ-caprolactone) for gambogenic acid. Kaohsiung J Med Sci. 2019;35(12):757-64. doi: 10.1002/kjm2.12110, PMID 31433556.
Mu L, Elbayoumi TA, Torchilin VP. Mixed micelles made of poly (ethylene glycol)-phosphatidylethanolamine conjugate and d-α-tocopheryl polyethylene glycol 1000 succinate as pharmaceutical nanocarriers for camptothecin. Int J Pharm. 2005;306(1-2):142-9. doi: 10.1016/j.ijpharm.2005.08.026, PMID 16242875.
Dong K, Zhang M, Liu Y, Gao X, Wu X, Shi D. Pterostilbene loaded soluplus/poloxamer 188 mixed micelles for protection against acetaminophen-induced acute liver injury. Mol Pharm. 2023;20(2):1189-201. doi: 10.1021/acs.molpharmaceut.2c00881, PMID 36647568.
Berlinski A, Spiva J. In vitro characterization of aerosolized albuterol generated by a jet nebulizer and delivered through a heated flow nasal cannula system. Pharmaceuticals (Basel). 2022;15(10):1281. doi: 10.3390/ph15101281, PMID 36297393.
Steckel H, Eskandar F. Factors affecting aerosol performance during nebulization with jet and ultrasonic nebulizers. Eur J Pharm Sci. 2003;19(5):443-55. doi: 10.1016/S0928-0987(03)00148-9, PMID 12907295.
Sweeney L, McCloskey AP, Higgins G, Ramsey JM, Cryan SA, MacLoughlin R. Effective nebulization of interferon-γ using a novel vibrating mesh. Respir Res. 2019;20(1):66. doi: 10.1186/s12931-019-1030-1, PMID 30943978.
Islam N, Gladki E. Dry powder inhalers (DPIs) a review of device reliability and innovation. Int J Pharm. 2008;360(1-2):1-11. doi: 10.1016/j.ijpharm.2008.04.044, PMID 18583072.
Myrdal PB, Sheth P, Stein SW. Advances in metered dose inhaler technology: formulation development. AAPS PharmSciTech. 2014;15(2):434-55. doi: 10.1208/s12249-013-0063-x, PMID 24452499.
Iwanaga T, Tohda Y, Nakamura S, Suga Y. The Respimat® soft mist inhaler: implications of drug delivery characteristics for patients. Clin Drug Investig. 2019;39(11):1021-30. doi: 10.1007/s40261-019-00835-z, PMID 31377981.
Pellosi DS, D Angelo I, Maiolino S, Mitidieri E, D Emmanuele Di Villa Bianca R, Sorrentino R. In vitro/in vivo investigation on the potential of Pluronic® mixed micelles for pulmonary drug delivery. Eur J Pharm Biopharm. 2018;130:30-8. doi: 10.1016/j.ejpb.2018.06.006, PMID 29890256.
Griffith DE, Eagle G, Thomson R, Aksamit TR, Hasegawa N, Morimoto K. Amikacin liposome inhalation suspension for treatment refractory lung disease caused by Mycobacterium avium complex (CONVERT). A prospective open label randomized study. Am J Respir Crit Care Med. 2018;198(12):1559-69. doi: 10.1164/rccm.201807-1318OC, PMID 30216086.
Esmaeili M, Aghajani M, Abbasalipourkabir R, Amani A. Budesonide loaded solid lipid nanoparticles for pulmonary delivery: preparation optimization and aerodynamic behavior. Artif Cells Nanomed Biotechnol. 2016;44(8):1964-71. doi: 10.3109/21691401.2015.1129614, PMID 26758698.
Amani A, York P, Chrystyn H, Clark BJ. Evaluation of a nanoemulsion based formulation for respiratory delivery of budesonide by nebulizers. AAPS PharmSciTech. 2010;11(3):1147-51. doi: 10.1208/s12249-010-9486-9, PMID 20652776.