
Int J App Pharm, Vol 18, Issue 1, 2026, 20-32Reviewl Article
NANOPARTICLE-EMBEDDED MICRONEEDLES FOR ENHANCED TRANSDERMAL DELIVERY: ADVANCES AND APPLICATIONS
SMRITI MANIBHUSHANAM*, UMASHANKAR MARAKANAM SRINIVASAN, DAMODHARAN NARAYANASAMY
Department of Pharmaceutics, SRM College of Pharmacy, Faculty of Medicine and Health Science, SRM Institute of Science and Technology, Kattankulathur-603203, India
*Corresponding author: Smriti Manibhushanam; *Email: msmriti1009@gmail.com
Received: 03 Jul 2025, Revised and Accepted: 17 Oct 2025
ABSTRACT
Transdermal drug delivery (TDD) provides a non-invasive alternative to oral and injectable administration, avoiding gastrointestinal degradation and first-pass metabolism. Traditional TDD systems are limited to small, lipophilic drugs and are prone to skin irritation and low bioavailability. Although microneedles (MNs) have several advantages, they also suffer from limited drug-loading capacity, fast drug diffusion, and mechanical fragility. Recent developments combine nanoparticles with microneedles (NEMNs), improving drug stability, bioavailability, and controlled release. The types of MNs (solid, coated, hollow, dissolving, and hydrogel) and considerations regarding materials (silicon, metals, polymers) are presented alongside nanoparticles to enhance therapeutic outcomes. Advancements in manufacturing methods, such as 3D printing and micro molding, have facilitated the controlled deposition of nanoparticles and enabled scalable fabrication. Stimuli-induced NEMNs triggered by pH, glucose, light, and magnetic fields have applications in site-specific drug release for diabetes management, cancer treatment, wound healing, and the treatment of inflammatory ailments. Combining nanomaterials with biodegradable polymers enhances their strength and compatibility with living organisms. Although improvements have been made, nanoparticle stability, scalability, regulatory compliance, and long-term safety remain significant concerns. To encourage clinical translation, future directions will focus on AI-based closed-loop systems, biomaterials that can perform multiple functions, and eco-friendly manufacturing processes. Overcoming these barriers, NEMNs have the potential to serve as patient-centric treatments for both chronic and acute conditions, thereby closing the gap between nanotechnology and minimally invasive therapeutics.
Keywords: Transdermal drug delivery, Nanoparticle-embedded microneedles, Mechanical strength, Nanoparticles, Controlled release, Stimuli-responsive, Fabrication techniques
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.55869 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Transdermal drug delivery (TDD) provides a non-invasive route that bypasses first-pass metabolism and gastrointestinal degradation, thereby enhancing patient adherence through self-administration and reducing systemic side effects commonly associated with oral and injectable therapies [1]. Conventional transdermal formulations, such as gels, creams, and adhesive patches, are often limited to low molecular weight, moderately lipophilic drugs and frequently suffer from poor permeation, limited bioavailability, skin irritation, and adhesion-related pain. These constraints have spurred the development of microneedle (MN) technologies as a next-generation transdermal platform [2, 3].
Microneedles (MNs) are 25-2,000 µm projections that perforate the stratum corneum, creating temporary drug transport channels while minimizing discomfort by bypassing deeper nerves and vasculature [3, 4]. In contrast to conventional transdermal techniques, MNs enable the safe and low-risk administration of hydrophilic and macromolecular therapies, including proteins, peptides, insulin, and vaccinations [5]. Their many uses include wound healing, tissue regeneration (cardiac, bone, tendon, ophthalmic, oral, hair, and uterine) [6], dermatological therapies, contraception, vaccination delivery, and health monitoring [7]. Self-administration capabilities further enhance adherence and reduce the financial burden of healthcare. The choice of material and shape determines MNs mechanical performance. Silicon, metals, ceramics, glasses, and biocompatible and biodegradable polymers are among the materials used in fabrication [8].
Metals and ceramics exhibit strong fracture forces but are prone to brittle failure, whereas polymers are biocompatible and biodegradable but relatively weak. The fracture force, which is the maximum axial load before structural failure, must be greater than 0.1 to 0.5 N per needle, depending on the material and form, for reliable skin implantation [9]. Polymers, with optimized tip shapes (rectangular, pyramidal, cylindrical, conical, and quadrangular), reduce swelling, inflammation, and granuloma formation, unlike metals and ceramics, which are brittle and prone to bending or fracture [10, 11]. These advantages are, nonetheless, accompanied by several issues associated with MN systems, including the inability to easily load drugs in the micro-cavity space and a rapid diffusion of low-molecular-weight drugs. The small shipping capacity and various distribution channels complicate the distribution of high doses of vaccination and biologics. Proper experimental and clinical safety testing for skin irritation and immune response should also be conducted. Regulatory approval requires standardized tests of mechanical integrity, biocompatibility, and long-term tolerability [8, 12].
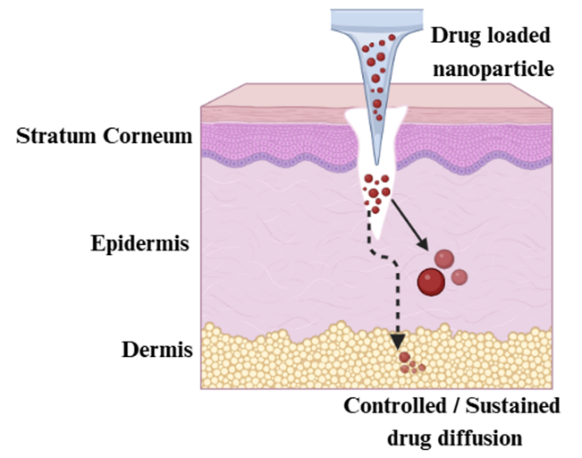
Fig. 1: Schematic of microneedle (MN)-mediated delivery of drug-loaded NPs into skin, enabling controlled and sustained drug release. Source: Created with BioRender
The limitations can be overcome by integrating nanoparticles (NPs) into MNs to create nanoparticle-embedded micro needles (NEMNs), which have a higher loading capacity, stability of a payload, and control over penetration. Researchers have also added NPs to MNs to increase their delivery. Nanoscale carriers (1–1000 nm) enhance drug solubility, prolong systemic circulation, enable site-specific targeting, and reduce systemic side effects. These carriers enable continuous, regulated release to enhance therapeutic efficacy by overcoming biological barriers, such as the blood-brain barrier [13, 14]. NPs, such as polymeric, liposomal, solid lipid, and metallic NPs, increase skin permeability, regulate drug release, and stabilize unstable drugs. Fig. 1 shows NEMNs depositing drug-loaded NPs into the skin for controlled, sustained diffusion.
In another study, dissolvable MNs containing chitosan NPs were constructed for the delivery of pramipexole dihydrochloride to Parkinson's disease, being able to attain a loading efficiency of ~0.12 mg/cm2 and sustained release with low skin irritation. This hybrid strategy not only takes advantage of the mechanical conduit of MNs and carrier properties of NPs to control the diffusion kinetics and bioavailability [15]. Comparison of transdermal drug delivery (TDD) systems, hypodermic needles, MNs, and NEMNs is shown in table 1, which shows the differences between the systems in invasiveness, drug loading, and bioavailability.
These advanced NEMN systems facilitate applications across vaccine delivery, cancer therapy, and delivery of biologics (e. g., insulin, gene therapies) [16].
Faraji et al. (2021) thoroughly reviewed microneedle technology, including the wide range of designs, materials, and manufacturing approaches based on lithography to more sophisticated additive fabrication techniques such as two-photon polymerization [11]. Upon this, Gowda et al. (2022) examined stimuli-responsive MNs whose activation is driven by endogenous (e. g., glucose) and external (e. g., light) stimuli to deliver drugs precisely and on demand with consideration of practical translation challenges [17]. This review also builds on these understandings, utilising the more advanced NEMNs as a superior method to enhance stability, drug loading capacity, and controlled release. It explains fabrication methods, scalability, regulatory issues, and biocompatibility of different NPs and provides a general picture of nanotechnology-based next-generation transdermal therapies.
Table 1: Comparative characteristics of transdermal delivery systems, hypodermic needles, MNs, and NEMNs
| Parameter | Conventional TDD | Hypodermic needle | MNs | NEMNs |
| Description | Semi-solid (emulsion/gel/ cream/ointment) or adhesive patch on skin | Hollow tube with sharp tip/opening | Micron-sized needles on a patch | Micron-sized needles embedded with NPs for improved delivery |
| Invasiveness | Noninvasive | Highly invasive | Minimally invasive | Minimally invasive |
| Pain | Painless | Painful | Painless | Painless |
| Drug loading | Limited to small molecules | High | Moderate | High |
| Bioavailability | Poor | Sufficient | Sufficient | Sufficient/Enhanced |
| Patient compliance | High | Low | High | High |
| Self-administration | Possible | Not possible | Possible | Possible |
| Mechanism of drug delivery | Passive diffusion across the skin | Direct injection into blood/tissue | Microchannels enable drug diffusion | Microchannels with nanoparticle-mediated enhanced penetration |
| References | [18] | [19] | [20] | [21] |
Search methodology
A thorough literature search was conducted in PubMed, Scopus, Web of Science, and the database of the Innovare Academics Journals. Keywords were nanoparticle-embedded microneedles, transdermal drug delivery, fabrication, controlled release, biocompatibility, and clinical translation. The search was restricted to peer-reviewed, English-language primary research articles published between June 2016 and July 2025. Any investigations that are not related to pharmaceutical use or do not provide experimental results were excluded. Further sources were identified by screening bibliographies of major articles, which covered a broad range of recent and classic developments in the discipline.
Microneedle classification and materials consideration
Continuing on the classification and material aspects briefly described above, the present section is devoted to microneedle types and fabrication materials, which are of particular importance in terms of nanoparticle integration within the framework of transdermal drug delivery systems.
Microneedle classification
As outlined in the Introduction, MNs vary by design and delivery method, each suited to distinct therapeutic applications. This section summarizes key structural types with emphasis on their roles in nanoparticle integration for enhanced transdermal delivery [22].
Solid microneedles (SMNs)
SMNs penetrate the stratum corneum to generate immediate microchannels, thereby boosting the skin’s permeability. These microchannels can then be used to apply drugs in pharmaceutical form, such as gels or patches. As described earlier in the Introduction, they are pre-treatment devices, which do not create residues when applied to the skin barrier (fig. 2) [23]. For example, tetanus toxoid-loaded chitosan NPs administered through SMNs resulted in an enhanced antigen uptake and immune response, which was considerably better than conventional administrations [24]. The typical materials used to construct SMNs include metals, such as silicon, nickel, cobalt alloy, palladium, and stainless steel.
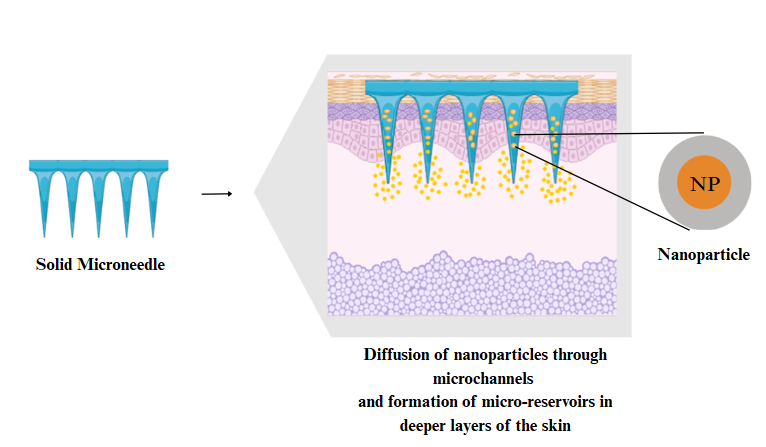
Fig. 2: Schematic representation of the mechanism involved in the release of NPs from SMNs. Source: Created with bio render
Coated microneedles (CMNs)
CMNs, as previously mentioned, are active agent carriers wherein active agents are deposited on the ends of the MNs in the form of a dissolvable surface film. They have a lower drug-loading capacity (usually less than 1 mg), but they are still useful in delivering small molecules, macromolecules, and vaccines quickly [25]. In nanoparticle-integrated systems, CMNs stabilize therapeutic payloads, allowing for controlled release upon skin insertion. A notable example is lipid bilayer-coated mesoporous silica NPs, which enhance antigen stability and significantly improve uptake by dendritic cells following microneedle application (fig. 3) [26].
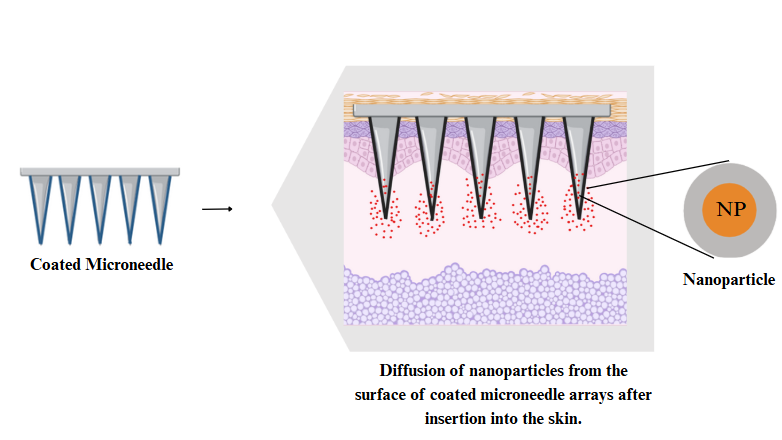
Fig. 3: Schematic representation of the mechanism involved in the release of NPs from surface CMNs. Source: created with bio render
Hollow microneedles (HMNs)
HMNs are characterized by a central opening that permits the direct delivery of therapeutics into the deep layers of the skin using pressure-based flow or diffusion [12]. They can support real-time liquid dosing and increased volume and are therefore the best suited for nanoparticle-laden formulations. Another method used in fabrication is microelectromechanical systems (MEMS)-based photolithography and deep reactive ion etching (Bosch process), and glass micropipette approaches [27]. A recent study has shown that HMNs loaded with polymeric, lipid-based, and dendrimeric NPs improved the delivery of plasmid DNA vaccines, resulting in a higher delivery rate, better antigen uptake, and enhanced immunity compared to the traditional subcutaneous injection method (fig. 4). Although these advantages exist, HMNs cannot be applied to nanoparticle suspensions. The lumen is small and may clog due to the aggregation of particles or high viscosity. Backflow during administration can cause poor delivery accuracy unless the MNs are placed deep or firmly. To address these problems and enhance clinical reliability, design innovations in HMN, such as anti-clogging coatings and the optimal channel geometry design, are under research [28].
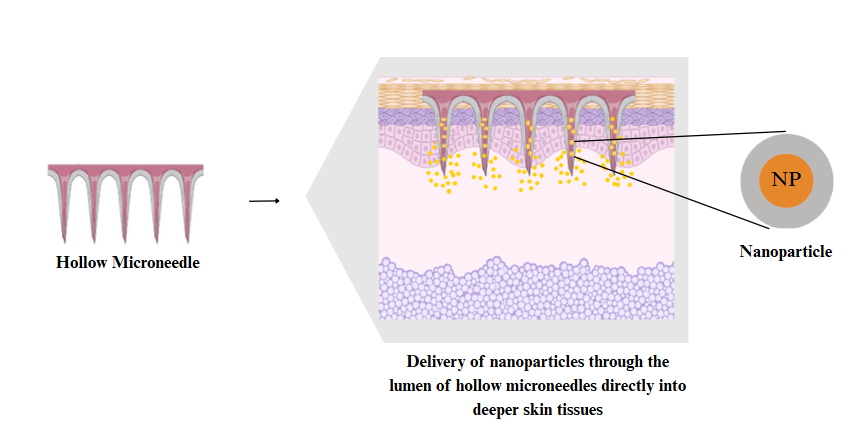
Fig. 4: Schematic representation of the mechanism involved in the release of NPs from HMNs. Source: created with bio render
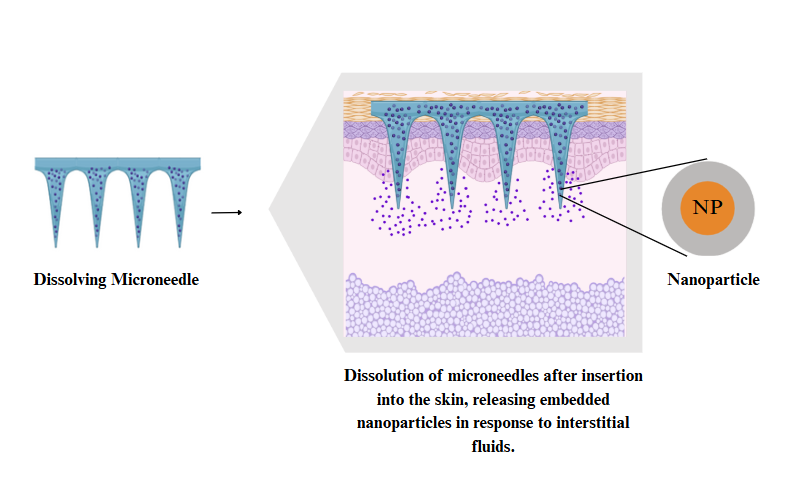
Fig. 5: Schematic representation of the mechanism involved in the release of NPs from DMNs. Source: created with bio render
Dissolving microneedles (DMNs)
DMNs are biodegradable polymers of polylactic acid (PLA), hyaluronic acid (HA), and poly (lactic-co-glycolic acid) (PLGA), which dissolve in the dermis of the skin to release drug payloads [29]. Most of the dissolved MNs are produced in the production facilities through solvent casting techniques using dissolved provisional materials [30]. They can be localized, sustained, and minimally invasive because they can be used as nanoparticle-loaded systems. A recent study reported that DMNs incorporating silver NPs in poly(ε-caprolactone) NPs, which interacted with bacterial enzymes, destroyed biofilms of Staphylococcus aureus and Pseudomonas aeruginosa over 60 h in the presence of bacteria. The effect of this approach was enhanced skin penetration, reduced toxicity, and extended antimicrobial efficacy and adherence in patients (fig. 5) [31].
Hydrogel microneedles (HG-MNs)
HG-MNs are made of cross-linked polymer matrices that swell when inserted into the skin and absorb interstitial fluid to cause drug diffusion [5, 32]. Although such MNs are generally used as conduits, not drug carriers, active agents may be added to an active drug film, tablet, or lyophilized reservoir under the patch. The design enables the delivery of a high dose, although it may take a considerable amount of time to be applied due to slow diffusion [33]. A recent study by Ding et al. (2024) showed that HG-MNs based on hyaluronic acid and loaded with vascular endothelial growth factor (VEGF) and Ritlecitinib/polyhydroxyalkanoates (PHA) NPs exhibited a dual-release profile: both VEGF and Ritlecitinib quickly diffused out of the swollen hydrogel, and the nanoparticle degraded, releasing Ritlecitinib [34]. According to Martins et al. (2024), the parameters of the HG-MN (e. g., its crosslinking density, porosity, and swelling rate) can be adjusted to move the prevalent mode, either diffusion or erosion. This interaction is fundamental to ensure that predictable and long-term delivery of drugs is achievable on hydrogel microneedle platforms. Developments in hydrogel chemistry are thus creating opportunities to develop tailored microneedle systems that can deliver contents in a controlled and targeted manner in therapeutic fields [35] (fig. 6).
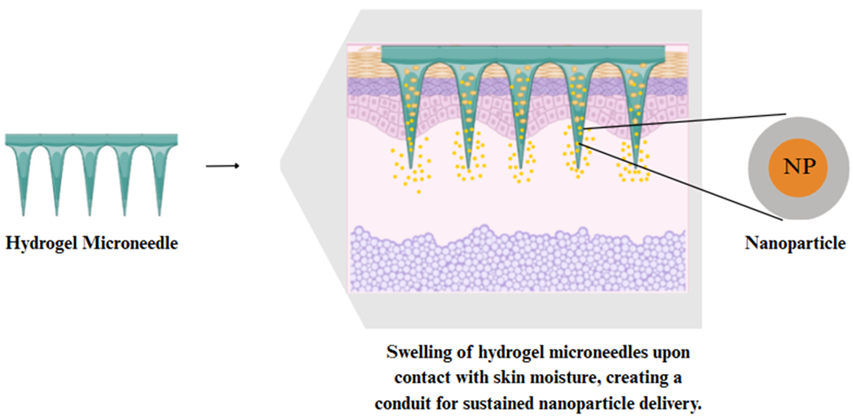
Fig. 6: Schematic representation of the mechanism involved in the release of NPs from HGMNs. Source: created with bio render
Materials for microneedle
MNs enter the stratum corneum without structural damage. Material choice affects NEMN mechanical strength, biocompatibility, and delivery. Metals provide durability, silicon enables precision microfabrication, polymers such as HA and PLGA facilitate controlled release, and hydrogels enable swelling-mediated distribution. The encapsulation, stability, and skin penetration of NPs depend on each platform, as listed in table 2, based on loading capacity, mechanical performance, degradation, and restrictions.
Table 2: Comparative properties of MN materials
| Material | Nanoparticle loading efficiency | Mechanical strength | Degradation timeline | Key drawback |
| Silicon | Low-Moderate | High (sharp, brittle) | Non-degradable | Brittle, expensive |
| Metals | Low-Moderate | Very High (skin penetration) | Non-degradable | Requires removal |
| Ceramics | Moderate | Very High (stiff, brittle) | Non-degradable | Brittle, heat-sensitive |
| Polymers | High | Moderate (tunable, degradable) | 2–6 w (PLGA) | Moisture sensitive, softer |
Silicon
Silicon also enables precise fabrication of MNs using photolithography and MEMS technologies, resulting in sharp, high-aspect-ratio arrays with controlled dimensions [36]. Monocrystalline silicon is better in terms of mechanical stability and precision penetration; therefore, it can be used in high-load transdermal delivery systems [37]. Although amorphous silicon is easier and more flexible to fabricate, it has a lower stiffness and reduces [38]. The silicon-based MNs can be applied most productively to MEMS-integrated nanoparticle systems where shape accuracy and uniform skin penetration are required.
Metals
Stainless steel and titanium are metals that provide remarkable mechanical properties and are highly utilized as MNs, requiring deep penetration beneath the skin. Stainless steel is both cost-effective and resistant to corrosion and is therefore popular in drug delivery and biosensing [39, 40]. Titanium is used to provide sensitive biologics and vaccines that have increased biocompatibility and resist rust [7]. However, metallic MNs, being non-biodegradable, will need to be removed later and may also typically require additional coatings to facilitate drug release.
Ceramic
Ceramic MNs commonly composed of alumina, zirconia, or calcium phosphate, have high mechanical and thermal stability and good biocompatibility [41]. Alumina, for example, serves as a durable carrier for NPs and biologics such as insulin and antibodies [42]. Another disadvantage of alumina is that it is typically sintered at high temperatures, making it challenging to incorporate thermolabile medications into the ceramic material [41]. This, however, is limited by its brittleness and high-temperature processing, which makes it unsuitable for thermosensitive drugs. Despite these challenges, ceramics provide a viable platform for MNs with NP-loaded structures that are stable.
Polymer
Polymers such as PLGA, PVA, and chitosan are also extensively used in MN fabrication, primarily due to their ability to load drugs, tunable mechanical properties, and low toxicity. PLGA is a biocompatible and controlled-release polymer that is efficient in this capacity [43]. However, with natural polymers, such as chitosan, it is essential to consider that the source, deacetylation procedure, and molecular weight can lead to variations from batch to batch. This may make it more difficult to reproduce in manufacturing and drug release [44]. On the contrary, PVA is highly hydrophilic, easy to manufacture, and dissolves in transdermal systems [45]. PVA, on the other hand, is very hydrophilic, simple to produce, and promotes dissolution in transdermal systems [46]. MNs based on polymers offer to deliver NPs in dissolved, hydrogel, and coated forms.
Types of nanoparticle used in microneedle systems
MN-enhanced drug delivery uses NPs, which improve solubility, stability, and release. The size, composition, and surface features of NPs influence their penetration depth, retention, and kinetics, with common systems including lipid-based, polymeric, inorganic, hybrid, and nanocrystal NPs [47]. To avoid aggregation and retain functionality, NPs must be carefully formulated when 3D printed onto microneedle matrices. Ziesmer et al. (2024) found that poorly distributed Silver/silicon dioxide (Ag/SiO₂) NPs in photothermal MNs form large clusters (>100 µm), affecting mechanical strength and thermal characteristics. In addition to this, UV curing is less efficient with a much higher loading. Even distribution and uniform manufacturing performance then require bead homogenization and concentration optimization [48].
Lipid-based nanoparticles
The most frequently employed nanoparticle in microneedle systems is lipid-based nanoparticles (LNPs), such as liposomes, niosomes, ethosomes, and solid lipid nanoparticles (SLNs), which can carry both hydrophilic and lipophilic drugs. Their lipid bilayers are compatible with the skin, increasing drug permeation and reducing degradation [49]. The sustained release profile, improvement of therapeutic activity, and reduction of the dosing interval can be ensured with the use of SLNs and nanostructured lipid carriers (NLCs). LNPs enhance transdermal absorption and avoid hepatic first-pass metabolism, which could potentially be used to deliver vaccines, peptides, or drugs with poor solubility when incorporated into MNs [50].
Polymeric nanoparticles
Polymeric nanoparticles (PNPs), derived from biocompatible and biodegradable polymers such as PLGA, PEG, PLA, and chitosan, provide a fine-tuned control of drug release kinetics when combined with microneedle systems. These NPs help encapsulated drugs avoid being degraded by enzymes and develop controlled or targeted delivery [51]. Their small scale dimension allows them to penetrate deeper into the skin, which increases drug retention and effectiveness. Additional tissue targeting can be enhanced by functionalization of the PNPs through ligand modification, which helps to minimize systemic side effects [52].
Inorganic nanoparticles
Inorganic NPs (gold, silver, iron oxide, and metal-organic frameworks) improve transdermal drug delivery efficiency by providing antibacterial, photothermal, and magnetic characteristics, as well as acting as Magnetic Resonance Imaging (MRI) contrast agents [47]. Their incorporation into NEMNs enhances treatment precision and diagnostic capabilities. Smaller metal oxides, such as zinc oxide (ZnO) and titanium dioxide (TiO₂), below 50 nm, produce reactive oxygen species (ROS), which can lead to oxidative stress and skin irritation. Their low biodegradability raises concerns regarding long-term tissue buildup and toxicity. Xia et al. (2024) have conducted recent studies that demonstrate the effective mitigation of these risks through the use of appropriate nanoparticle design, which includes surface functionalization, size optimization, and biodegradable microneedle matrices. Histopathological analyses have shown minimal toxicity after repeated administration. Thus, while inorganic NPs provide improved targeting and multifunctionality, including safety-focused design techniques, they are critical for effective clinical translation [53].
Nanocrystals
Nanocrystals are pure drug crystals with a high surface area and an almost 100% loading capacity, offering a carrier-free alternative for microneedle-mediated delivery. Their submicron size enhances the solubility and bioavailability of the poorly water-soluble drug [54]. Nanocrystals, when incorporated into MNs, can be used to successfully enter the stratum corneum and enable sustained and localized drug release. This would be especially valuable in cases of high-dose treatment for chronic conditions such as cancer and psoriasis, where non-invasive self-administered delivery is also highly desirable [55].
Hybrid carriers
Hybrid carriers also integrate synthetic and natural materials, including polymers, lipids, and inorganic NPs, to make them multifunctional in drug delivery with MNs. The systems provide fine control over the release kinetics of the drugs, increase payload stability, and offer better biocompatibility [56]. A notable example of hybrid carriers is lipid-polymer hybrid nanoparticles (LPNs) in microneedle systems. Zhao et al. have created DMN patches of LPNs that contain microRNA-218 (miR-218) in the form of encapsulations. The LPNs also exhibited a combination of liposomal biomimetic and polymer stability, which offers a higher payload protection and a targeted miRNA release. These patches successfully penetrated the skin, delivering NPs to the dermis with four times the bioavailability of topical preparations, exhibiting greater therapeutic effectiveness. The system demonstrated excellent mechanical strength, stability, and low cytotoxicity, demonstrating how hybrid NPs enable accurate, controlled, and biocompatible transdermal distribution [57].
Furthermore, Different nanoparticle platforms have varied release kinetics that influence their therapeutic value. For example, nanocrystals often exhibit a rapid burst release for immediate therapeutic action, while LNPs provide a more sustained and controlled administration that is ideal for long-term therapy. Table 3 compares the release patterns, burst characteristics, and therapeutic significance of these systems.
Table 3: Nanoparticle release behaviors and therapeutic suitability
| Nanoparticle type | Carrier class | Initial burst | Release kinetics | Duration | Therapeutic suitability | Reference |
| SLNPs | Lipid-based | High | Biphasic | ~5 d | Peptides, vaccines with a fast onset and prolonged effect | [58] |
| Ondansetron NPs | Polymeric | Moderate | Biphasic | ~12 h | Acute nausea treatment with enhanced permeation | [59] |
| Iron(II) NPs | Inorganic | 10-30% | Near-zero-order | Up to 12 d | Chronic dosing (e. g., anemia) with minimal frequency | [60] |
| Conventional Nanocrystals | Nanocrystal | Absent | Zero-order | ~24 h | Constant dosing for cardiovascular therapies | [61] |
| LPNs | Hybrid | Low-moderate | Biphasic | 1-7 d | Combination therapies, precise control, multifunctional delivery | [57] |
Adding NPs to MN devices has several advantages, including improved stability, more precise delivery, and a greater capacity for drug loading. Table 4 illustrates how NPs enhance MN performance and their associated limitations.
Table 4: Roles of NPs in MN drug delivery systems and key challenges for different nanoparticle types
| Role | Description | Nanoparticle type | Challenges | References |
| Enhanced drug loading capacity | Increases the concentration of therapeutic agents in MNs, optimizing drug delivery with smaller microneedle sizes. | Nanocrystals | Aggregation; poor wettability in polymer matrix; drug expulsion during drying | [61] |
| Lipid-based | Limited encapsulation of hydrophilic drugs; phase separation during storage | [57] | ||
| Controlled and sustained drug release | Enables the release of drugs over a specified period based on environmental conditions like pH or temperature | Polymer | Variability in degradation rate, unpredictable burst release | [59] |
| Hybrid | Interface instability and complex manufacturing increase batch variability | [57] | ||
| Targeted drug delivery | Functionalized NPs allow the precise delivery of drugs to specific cells or tissues. | Inorganic | Loss of ligand activity under fabrication heat; possible cytotoxicity | [60] |
| Hybrid | Multi-step functionalization increases complexity and batch variability | [57] | ||
| Improved biocompatibility and Stability | NPs increase the stability of the drug within MNs and enhance biocompatibility for safer use. | Lipid-based | Lipid crystallization under heat; drug leakage during storage | [58] |
| Polymer | Aggregation during storage, UV/thermal crosslinking, may degrade the matrix | [59] | ||
| Enhanced skin penetration | NPs improve the ability of MNs to penetrate the skin, facilitating the delivery of larger or more complex drug molecules. | Inorganic | Size-dependent limitation; risk of local irritation, tissue accumulation | [60] |
| Nanocrystals | Excessive particle hardness can damage the matrix or reduce insertion reliability | [61] | ||
| Diagnostics and biosensing | NEMNs can be used for real-time disease monitoring through nanoparticle-based sensors integrated into microneedle tips. | Inorganic | Photobleaching, heavy-metal leaching, and limited sensor lifespan | [60] |
| Stimuli-responsive drug release | NPs respond to environmental factors (eg, pH and temperature) to release drugs only when needed. | Hybrid | Premature release during storage, achieving a precise response is complex | [57] |
| Polymer | Variable trigger thresholds; inconsistent release under physiological fluctuations | [59] | ||
| Antimicrobial and Anti-inflammatory properties | Some NPs possess intrinsic antimicrobial or anti-inflammatory properties, offering benefits in wound care or infection prevention. | Inorganic | Dose-dependent cytotoxicity; risk of resistance; tissue accumulation | [60] |
| Hybrid | Heterogeneous particle size; complex synthesis affects safety and efficacy | [57] |
Fabrication techniques for NEMNs
The effectiveness of NEMNs is further enhanced by strategic nanoparticle loading techniques that regulate drug release kinetics and bioavailability. The sections that follow provide a detailed study of the NPs used in MNs and the key strategies employed to integrate them into these systems (fig. 7).
3D printing techniques
3D printing has transformed MN manufacture by providing exact control over shape, size, and internal architecture, as well as the ability to include biosensors, drug reservoirs, and diagnostics directly into the MN design [62]. The most common of these techniques is stereolithography (SLA) and digital light processing (DLP) due to their high resolution capabilities and ability to use nanoparticle-filled resins. SLA has lateral resolutions of about 20-50 μm, and DLP has around 50 μm, which is approximately a thousand times greater than the size range of NPs incorporated, ranging from 1-1000 nm. As a result, microneedle geometry is governed by printer resolution, whereas nanoparticle dispersion and uniformity depend exclusively on resin formulation and mixing protocols. The two techniques were compared in terms of speed, precision, and nanoparticle incorporation as shown in table 5.
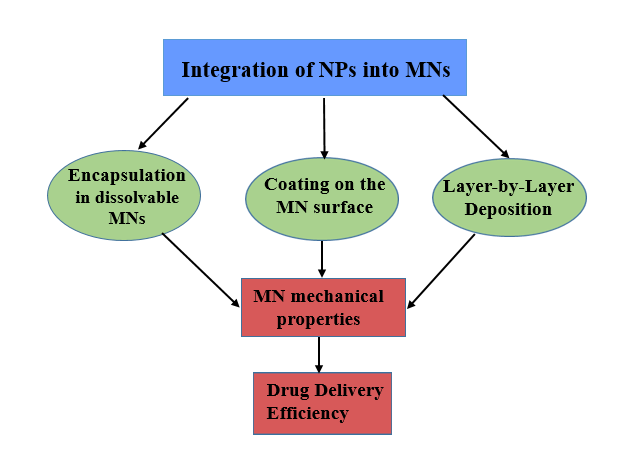
Fig. 7: Strategies for nanoparticle integration in MN systems and their impact on mechanical strength, drug loading, and release control. Source: created with bio render
Table 5: Comparison of 3D printing techniques for MN fabrication
| Technique | Resolution | Material compatibility | Speed | Nanoparticle integration | Limitations |
| SLA | ~20–50 µm | Uv-curable resins | Moderate | Precise nanoparticle control | Expensive materials, limited biocompatibility |
| DLP | ~50 µm | Uv-curable polymers | Fast | Homogeneous nanoparticle dispersion | Lower resolution compared to SLA |
Stereolithography
SLA is a common 3D printing technology relying on vat photopolymerization that creates complex geometry and produces very accurate MNs. SLA reveals a sheet of a particular liquid resin using a UV laser or light source, and allows complex microstructures to form with a smooth surface. Given that SLA reinforces resins at the nanoparticle level, it may be of significant help in printing MNs with NPs [63]. In photothermal MNs, Ziesmer et al. (2024) dispersed plasmonic Ag/SiO₂ nanoaggregates (primary particles tens of nm, aggregated order 100 nm) in UV-curable resin. To minimize agglomeration and improve optical performance, vortex mixing, ultrasonication, and bead homogenization were compared. The latter produced the smallest and most uniform agglomerates, thereby reducing scattering losses during curing. The geometric structure of the MNs consisted of two materials: a support base made of a defined transparent material and a structure of NPs as a functional material. This structure maintained print resolution and enabled spatial control of nanoparticle positioning, so that they could be activated by photothermal stimuli [48].
Digital light processing
DLP is a high-resolution, high-speed vat photopolymerization 3D printing technology suitable for the fabricating of NEMNs. Unlike point-by-point laser systems, DLP cures the entire resin layer with visible or UV light and, as such, can manufacture complex structures quickly. Petrova et al. (2025) showed that a hydrophilic hydrogel matrix could be used to produce dissolvable MNs with the desired geometry, uniform dispersion, and size of the nanocrystals (imiquimod) of about 74 nm and pH-dependent drug release. The ratios of the monomer to the photoinitiator played a significant role in the achievement of an appropriate curing and mechanical strength because the high hydrophilic content or the nanoparticle loading was shown to harm the polymerization and the integrity of the device. Overall, SLA offers greater resolution for selective nanoparticle placement, whereas DLP excels at speed and homogeneous inclusion. The shape of MN is all printer-dependent, whereas nanoparticle dispersion and mechanical stability are all determined by the formulation and resin composition [64].
Micromolding
Micromolding is another fabrication process that is scalable and precise and is extensively used to create NEMNs. Being a soft lithography method, it can be used to reproducibly fabricate biocompatible microstructures for TDD [65]. Multilayer and hybrid micromolding techniques, which enhance nanoparticle encapsulation, mechanical viability, and controlled drug release, are advanced strategies that allow for the spatial regulation of polymer-nanoparticle layering. The innovations increase the functional capacity of NEMNs to meet various therapeutic needs. Table 6 modifies and compares micromolding practices based on performance properties in NEMN manufacturing.
Table 6: Comparative assessment of MN manufacturing methods for nanoparticle integration and scalability
| Technique | Drug encapsulation efficiency | Mechanical strength | Scalability | Limitations | Heat-sensitive NPs compatibility | Aggregation-prone NPs compatibility |
| Solvent casting | High | Moderate | High | Requires solvent removal, risk of nanoparticle aggregation without stabilizers | High (room-temp drying) | Moderate needs stabilizers/ultrasonication needed for uniform nanoparticle dispersion |
| Hybrid micromolding | Very high | High | Moderate | Requires hydrogel synthesis and crosslinking; UV may affect photolabile NPs | High (no thermal curing) | High if NP remains stable during crosslinking chemistry and UV exposure; avoid for ROS or photo-sensitive formulations. |
| Multilayer micromolding | Moderate-high | Very high | Moderate | Time‑consuming layer assembly | High (ambient spray deposition) | Moderate–High in aqueous layers; Low–Moderate in organic solvent layers |
Solvent casting
Solvent casting micromolding is a typical and cost-effective method for producing NEMNs with a uniform distribution of NPs and a high rate of drug encapsulation. Zarei Chamgordani et al. (2024) made PLA MNs containing 1–5% silver NPs (~20 nm) by dissolving PLA in dichloromethane (DCM), dispersing AgNPs ultrasonically, and casting them into polydimethylsiloxane (PDMS) molds via centrifugation. The MNs (~1.36 mm height, 24–27 µm tip) released silver for 34 d with sustained antibacterial activity. Although residual DCM was not directly measured, drying and vacuum must have reduced the solvent concentration to below 1–2% w/w, which is within the ICH Q3C safety limits. Controlling the residual solvent is crucial because traces of DCM can affect PLA crystallinity, promote nanoparticle aggregation, and modulate release profiles. Nanoparticle stability and reproducible mechanical and biological performance are ensured by appropriate solvent selection, drying, and post-process steps [66].
Hybrid micromolding
Hybrid micromolding is the use of solvent-assisted nanoparticle incorporation in combination with polymer casting to improve drug loading and mechanical stability of NEMNs. The method will enable accurate control over the composition and geometry of materials and enhance mechanical strength. Tang et al. (2021) reported a hybrid micromolding in which poly (chondroitin sulfates-acrylamido-2-methyl propane sulfonic acid) hydrogels were hybridized with in situ synthesized gold nanoparticles (GNPs). The mixture of the prepolymer/GNP precursor was poured into PDMS molds, centrifuged to fill the cavities, and then UV-cured, which simultaneously crosslinked the hydrogel and synthesized GNPs. This combined process resulted in MNs (~800 μm long, ~300 μm bottom) with>0.4 N/tip strength, high swelling capacity, and the ability to selectively enrich positively charged metabolites from rat skin interstitial fluid [67].
Multilayer micromolding
Multilayer micromolding is a more sophisticated technique for preparing NEMNs, which are more stable mechanically and release drugs in a controlled manner over time. In this approach, multiple layers of various polymers are employed, including PLGA to enable slow release and PVP to facilitate fast breakdown. Each layer is filled with NPs of varied natures, such as AgNPs or polymeric carriers. Recently, micro-milled aluminum master molds were used to create the obelisk-shaped PLGA PVP MNs with stratified layers and acutely ended features. The polymer composition could change the release profiles of these NEMNs, which had a homogeneous distribution of NPs and a high compression strength. Adding AgNPs made the material stiffer, whereas using poly (methyl methacrylate) (PMMA) matrices made it easier to load drugs and less likely to break when inserted into the skin [68].
Electrohydrodynamic atomization
Electrohydrodynamic atomization (EHDA) is a novel method for coating NEMNs with high accuracy and consistency. Using an electric field, it breaks up polymer-nanoparticle suspensions into tiny droplets. This enables control of deposition onto MN surfaces without damaging their structure. A recent study utilized electrospray deposition (ESD) to coat HG-MNs with ovalbumin (OVA)-loaded poly(lactic-co-glycolic acid) NPs, achieving a deposition efficiency of approximately 30% while maintaining mechanical strength and insertion force (around 17 N) comparable to uncoated devices. Compared to traditional dip-coating or inkjet printing, EHDA produces thicker and more uniform nanoparticle layers, which are essential for achieving optimal drug delivery efficiency. Scanning electron microscopy confirmed the well-preserved morphology of MN, and EHDA was confirmed as a scalable and precise functionalization method for NEMNs [69].
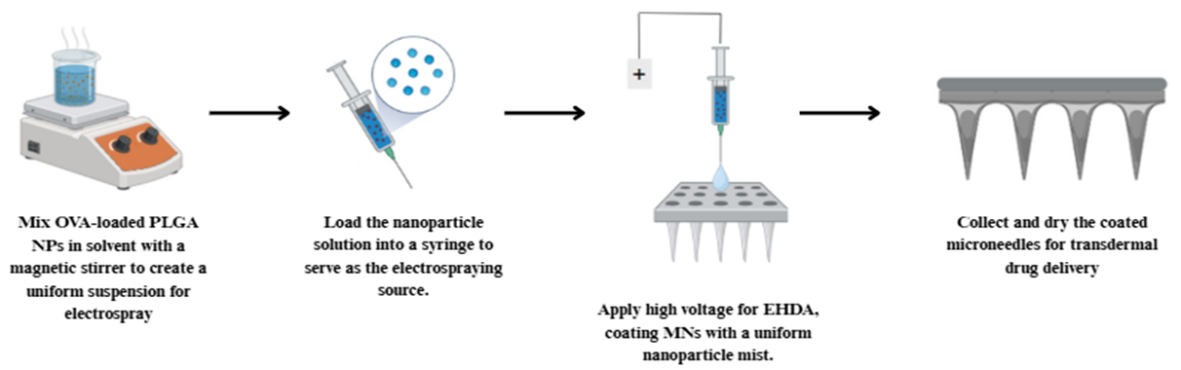
Fig. 8: Schematic representation of nanoparticle coating onto hydrogel-forming MNs using the EDHA technique. Source: created with bio render
Recent advances in NEMNs
Recent developments in NEMNs have made it much easier to deliver drugs in a controlled, efficient, and predictable manner in cancer treatment, diabetes management, and vaccine delivery.
Stimuli-responsive drug release mechanism
These MNs can effectively encapsulate bioactive agents and nanoparticles, thereby enhancing drug-loading capacity and attaining targeted therapeutic effects [70]. Compared to regular MNs, NEMNs have the benefit of being more precise when it comes to drug release, which lets them have both immediate and long-lasting delivery profiles. These MNs can respond to a wide range of stimuli, including physiological (pH, glucose concentration), physical (light, temperature), and mechanical (stretching, pressure) stimuli. The next section is devoted to investigating the underlying mechanism of NEMNs and their application in smart drug delivery. Table 7 summarizes the main stimuli-responsive NEMN designs, their biological triggers, nanoparticle compositions, biomedical applications, response times, scalability issues, and, more importantly, therapeutic results.
Biomedical applications of NEMENs
MNs with small particles can cure several diseases caused by diverse pharmaceutical delivery methods and disorders. Before reaching the circulation, MN drug delivery passes through the skin's barrier to reach the epidermis or higher dermis. It's painless, patient-friendly, and simple. Miniature MNs offer lesser dosages and minimize deep tissue harm. Controlling simple MN release rates limits dosage size, depth, and time. Precision dosage, penetration depth, and release patterns are possible using NPs.
Diabetic treatment
Diabetes is also known to cause high blood glucose due to a lack of insulin or failure to work properly, leading to fatigue, vision problems, and a high risk of being affected by vascular diseases. Normal injections used in diabetes are painful and can also cause tissue damage. Type 1 diabetes is treated using insulin injections, and type 2 diabetes is treated by lifestyle change and pharmaceutical therapy. To overcome these challenges, microneedle patches are developed to deliver encapsulated drugs in NPs in a targeted, painless, and physiologically responsive manner and offer a minimally invasive alternative.
Yang et al. (2020) developed a microneedle patch containing ZIF-8 NPs encapsulating glucose oxidase and insulin, which could realize self-controlled insulin release under the stimulation of high glucose. This minimally invasive system was able to sustain stable glucose levels, decrease the risk of hypoglycemia, and increase compliance in diabetic models, suggesting that it might have the potential to replace standard insulin injections [76]. Liu et al. (2018) developed an NIR-responsible dissolving microneedle system to precisely deliver transdermal metformin through bismuth nanodots (BiNDs) as photothermal agents and a thermoresponsive coating made of lauric acid. BiNDs were heated by NIR radiation, and the coating was melted to release metformin on demand, which significantly reduced blood glucose (~69.4%) without causing tissue damage or hypoglycemia, providing an effective and minimally invasive diabetes treatment [77]. Table 8 compares the pharmacokinetic parameters and response times of glucose-responsive platforms with those of the current standard care, which is continuous subcutaneous insulin infusion (CSII) pumps.
Cancer therapy
Malignant tumors pose a serious health risk, with surgical resection as the primary treatment. However, surgery may leave cancer cells behind, causing recurrence and lowering immunity. Chemotherapy and radiotherapy often have adverse side effects. To enhance therapy and prevent metastasis, new delivery methods are needed. Transdermal microneedle systems offer effective, minimally invasive, painless, and safe administration. Multifunctional combined microneedle systems hold promising clinical potential for improved tumor treatment. Although NEMNs have shown promising results in several cancer therapy applications, penetration of solid tumors is a major challenge due to the dense extracellular matrix (ECM) and tumor microenvironment barriers, such as high interstitial fluid pressure, dense collagen, and uneven blood flow. Conventional nanoparticle delivery typically penetrates 2 mm or less from the tumor periphery, with less than 1% of the particles reaching the target cancer cells, and exhibits preferential accumulation at the tumor periphery. Additionally, there is a decrease in penetration efficiency across and within tumors due to tumor heterogeneity. To overcome these challenges, NEMNs use new designs and localized delivery to improve nanoparticle distribution in tumors [78].
Ma et al. (2024) constructed dissolving MNs with hybrid lipid polymer NPs encapsulated with chlorin e6 (Ce6) for photodynamic therapy and tirapazamine (TPZ), a hypoxia-activated prodrug. Additionally, Ce6 generated reactive oxygen species and activated TPZ upon laser irradiation under hypoxic conditions, thereby enhancing the anticancer effects. The hyaluronic acid-based MNs penetrated deep into the dermis, controlled drug release, and effectively suppressed tumor growth in melanoma and squamous cell carcinoma models. They also suppressed tumor survival markers (NQO1, HIF-1α) and demonstrated high biocompatibility, which demonstrates that stimulus-responsive NEMNs could help with cancer hypoxia and enhance the effectiveness of local treatment [79]. A gelatin methacryloyl (GelMA) MN platform was also created to treat melanoma. The goal was to make curcumin more soluble and send it through crosslinked MN arrays in a controlled way. A 3D model of skin cancer spheroids showed much better tumor suppression and cytotoxicity than free curcumin. This biocompatible system offers localized therapy, minimizing systemic toxicity and increasing drug retention at tumor sites [80]. Another PVP-based dissolving microneedle system was developed to deliver liposomes loaded with indocyanine green (ICG) and reactive oxygen species (ROS)-responsive prodrug of doxorubicin (Pb-DOX), which was used for the treatment of melanoma. Upon microneedle injection, the liposomes were released into tumor tissues, and ROS production induced by near-infrared (NIR) light enabled photodynamic therapy and activation of Pb-DOX for chemotherapy. This formulation decreased systemic toxicity and markedly enhanced anti-melanoma activity. Furthermore, the system was also compatible with fluorescence and photoacoustic imaging for real-time monitoring of drug delivery and therapeutic response [81].
Table 7: Comparison of various stimuli-responsive NEMNs, including the mechanisms, applications, advantages, activation times, and key scalability challenges
| Stimulus type | Biological trigger and NPs | Biomedical application | Activation time | Scalability challenges | Key functional outcome | References |
| pH-responsive | The metal-organic framework Zeolitic Imidazolate Framework-8 (ZIF-8) degrades in acidic conditions (pH~5.0), releasing encapsulated curcumin. | Tumor therapy, inflammation | Significant release onset within 15 min;>80% cumulative release by 6 h | Multi‐step MBG–ZnO QD synthesis; precise QD size/charge; enzyme stability; reproducible pH‐dissolution kinetics | High drug loading (~40.5%), selective curcumin release in acidic tissue, prevents systemic leakage. | [70] |
| The pyridine-modified MNs trigger the release of lipid bilayer-coated mesoporous silica nanoparticles (LB-MSNs) as pH shifts from ~5.8 to neutral skin pH (~7.4 | Intradermal vaccine delivery | ~70% nanoparticle release within 30 min of skin insertion | Silicon MN fabrication; uniform LB coating; cold-chain for lipid NPs | pH-triggered release, enhanced antigen presentation, non-invasive delivery, and high dendritic cell uptake | [26] | |
| Glucose-responsive | Phenylboronic acid groups on gold nanoclusters (PBA-GNCs) release insulin as glucose disrupts covalent bonds | Type 1 Diabetes | Rapid response within 0.5 h; sustained control up to 2.8 d | Gold cost; GNC synthesis; cold-chain for insulin | Glucose-sensitive, self-regulated insulin release for 2.8 d; avoids hypo/hyperglycemia. | [71] |
| Zinc Oxide Quantum Dots (ZnO QDs) in Mesoporous Bioactive Glass (MBG) dissolve with local acidity triggered by high glucose | Diabetes mellitus | pH drop and QD dissolution within minutes; sustained release over hours | Multi-step synthesis; enzyme stability; reproducible kinetics | Prevents premature insulin release; pH-triggered gating system avoids side effects. | [72] | |
| Light-responsive | Polydopamine (PDA) nanoenzymes absorb Near-Infrared (NIR) light, heat releases drug+Reactive Oxygen Species (ROS) neutralization. | Atopic dermatitis, skin disease | Therapeutic temperature in ~3 min; drug release sustained 24–72 h | PDA loading; NIR accessibility; Hyaluronic Acid Methacrylate (HAMA) crosslinking | Controlled drug release, ROS neutralization, improved microcirculation | [73] |
| Magnetic-responsive | Magnetic Graphene Quantum Dots (MGQDs) in chitosan respond to a field, enabling on-demand drug release | Inflammatory diseases, remote therapy | Release starts within 1–2 min; tunable by field strength/duration | MGQD dispersion; MN reproducibility; field penetration | Tunable and remote drug release, improved conductivity, and biodegradability | [74] |
| Multi-responsive | Cerium/Manganese Oxide Nanoparticles (Ce/MnO₂ NPs) respond to Reactive Oxygen Species (ROS)+hypoxia, modulate the immune system | Rheumatoid arthritis | ROS clearance and immune shift within hours; sustained effect for days | Mixed-oxide NP synthesis; catalytic stability; MN loading reproducibility | Anti-inflammatory effect, M1 to M2 shift, targeted immune modulation | [53] |
| ZIF-8 with Glucose Oxidase (GOx) and Horseradish Peroxidase (HRP) enzymes—cascade for wound healing under high glucose | Diabetic wound healing | Microcurrent/ROS detectable within minutes; antibacterial effect within hours; complete wound closure by 7 d in vivo | Multi-stage fabrication; enzyme stability; MOF loading; sterility | Enzyme-triggered cascade promotes antimicrobial activity, tissue regeneration, and local glucose control. | [75] |
Table 8: Comparative pharmacokinetics and insulin response times of glucose-responsive microneedle platforms and CSII pumps
| Metric | CSII insulin pump | Glucose-responsive microneedle patch | Advantage |
| Lag time (onset of insulin action) | 30–60 min | 5–15 min | 4–6× faster initiation |
| Time to peak concentration (Tmax) | 60–90 min | 20–30 min | ~3× more rapid peak delivery |
| Postprandial glucose excursion control | Reduces excursions by ~30–50% | Reduces excursions by ~50–70% | Superior attenuation of early glucose peaks |
| Inter-and intra-patient variability | Coefficient of variation (CV) 20–30% | CV 10–15% | More consistent, physiologic insulin profiles |
Gout treatment
Gout, a chronic inflammatory arthritis, is caused by the accumulation of monosodium urate (MSU) crystals in the joints, which leads to severe pain, swelling, and inflammation. Oral colchicine and urinase-reducing drugs are traditional therapies with systemic side effects, poor patient compliance, and slow effects. Nonsteroidal anti-inflammatory drugs (NSAIDs) and corticosteroids have gastrointestinal side effects and cardiovascular risk, and colchicine has a narrow therapeutic index and a higher risk of toxicity at higher doses. MN-based drug delivery systems have emerged as a new alternative in the treatment of gout for solving these problems. They provide localized, sustained-release, minimally invasive methods aiming to enhance drug effectiveness, minimize systemic side effects, and increase patient compliance.
A recent study developed chitosan-based DMNs co-loaded with uricase-liposomes (UAO-LPO) and colchicine for the treatment of long-term gout. The MN system features a fast-dissolving release layer that delivers the drug-loaded tips to the skin, enabling sustained release for one week. Phospholipid micelle-encapsulated uricase made the enzyme work better and lowered the level of uric acid. Colchicine delivered inside cells lowered the toxicity of the whole body. Studies on people with acute and chronic gout have shown that the drugs lower inflammatory cytokines, reduce joint swelling, and have an anti-inflammatory effect that lasts longer [82]. In addition, a MN system was developed utilizing a UV-crosslinked polymer hydrogel matrix and demonstrated high mechanical strength (11.53 N per needle) and high swelling capacity. This design enables localized colchicine release with no burst toxicity and maintains anti-inflammatory activity for extended periods of time [83].
Chronic and infected wounds
Chronic wound healing is difficult, particularly with the presence of biofilm and bacterial infection. Biofilms exist as a compact extracellular matrix that imparts drug resistance, evades immune detection, and results in chronic inflammation, recurrent infections and poor tissue regeneration. MN patches are a minimally invasive approach for the local delivery of drugs to the infected wound sites.
To improve diabetic wound healing, Xuan et al. (2025) developed an amyloid-templated ceria nanoenzyme-reinforced MN system, which aims to reduce chronic inflammation, oxidative stress, and bacterial infections. The system lowers reactive oxygen species (ROS) and alleviates hypoxia in the wound environment. Glucose oxidase (GOx) breaks down glucose, further enhancing tissue recovery. In diabetic wound models, it resulted in improved wound closure (~95.7%), less inflammation, and fewer bacterial infections. Conclusion: MN-mediated, non-invasive, non-surgical, self-administered enzyme delivery through nanotechnology is a promising therapeutic option for diabetic wound healing and avoidance of complications [84]. To enhance mitochondrial content, Yao et al. developed hydrogel MN patches of adipose-derived stem cells that produced mitochondrial-rich extracellular vesicles (EVs) containing mitochondrial electron donors: metformin. EVs reduce oxidative stress, increase ATP production, and enhance the functioning of mitochondria, which are essential to the formation of chronic wounds. The MNs enhanced EV delivery into the skin and prolonged its duration, resulting in mitochondrial transfer, M2 macrophage polarization, angiogenesis, and collagen deposition. This new technique enhanced the radiation-induced kinetics of chronic wound healing, making the use of MN-nanoparticle systems as a regenerative wound treatment feasible. [85]. However, biofilm invasion is one of the main problems of chronic wounds because of a polymicrobial EPS matrix of more than 600 μm, which increases antimicrobial resistance. This thickness might decrease the efficiency of mechanical disruption by 600 μm-long MNs and restrict the diffusion of nanoenzyme payloads. A gradient-length array of needles and matrix-degrading enzymes, or ultrasonics, can be used to dissolve biofilm and transfer the payload to researchers [86].
Challenges in NEMNs
Multiple problems exist in the development of NEMNs despite their great potential in smart drug delivery systems. Table 9 summarizes major challenges and practical solutions for nanoparticle-loaded microneedle drug delivery technologies.
Table 9: Summary of key challenges and corresponding mitigation strategies for NEMNs systems
| Challenges | Description | Mitigation strategies | References |
| Nanoparticle Stability | NPs may degrade, aggregate, or lose activity during storage and fabrication. | Use freeze-drying (lyophilization) or very cold grinding (cryogenic milling) to keep NPs from clumping or breaking down, preserving their size and strength | [58] |
| Low drug loading efficiency | Low nanoparticle encapsulation affects drug concentration and delivery efficiency, and capacity. | Design MNs with tiny pores or multiple layers to hold more NPs, allowing higher drug payload and steady release | [47] |
| Uncontrolled drug release | Difficult to achieve precise, sustained, or stimuli-responsive drug release. | Use materials that respond to body changes like pH or temperature to release drugs exactly when and where needed. | [70] |
| Microneedle strength and penetration | Some MNs are too brittle or too soft, affecting skin penetration and drug absorption. | Adjust materials and their internal bonds to balance toughness and flexibility, ensuring proper skin penetration without breaking. | [66] |
| Biocompatibility and Toxicity | Some NPs may trigger immune responses or accumulate in tissues, raising safety concerns. | Coat NPs with safe substances (like PEG) to reduce immune reactions and prevent buildup in the body | [8] |
| GMP-compliant manufacturing and Scalability | Challenges in scaling up production under GMP, include cost reproducibility and quality control. | Use scalable methods like roller embossing and continuous micromolding for consistent quality and cost-effective production aligned with regulations. | [53] |
| Regulatory hurdles | Lack of standardized regulatory frameworks for NEMNs and extensive preclinical/clinical validation. | Promote collaboration among scientists, industry, and regulators to develop clear guidelines and thorough testing to ensure safety and effectiveness. | [57] |
Conclusion and future perspective
Nanoparticle-embedded microneedles (NEMNs) are a novel transdermal delivery platform that improves drug stability, targeting, and patient compliance by overcoming the limitations of conventional methods. Advances in material and fabrication, such as biocompatible hybrid composites and scalable 3D/4D printing, have realized increased mechanical robustness and cost-effectiveness. Translation to the clinic still prioritizes safety, efficacy, and manufacturing consistency, along with regulatory matters. NEMNs are regulated as combination products and, when there are analogous predicate devices, can be reviewed as an accelerated 510(k) approval; otherwise, new NEMNs may be subjected to a more vigorous de novo classification. Early regulatory engagement: Engaging with the FDA early in the development process can help clarify regulatory expectations and expedite market access.
The future development of NEMN will focuses on incorporating actual smart technologies, such as closed-loop control of insulin delivery based on real-time continuous glucose monitoring (CGM) and machine learning, for personalized treatment. Biodegradable materials, such as cellulose nanocrystals and LNPs, will replace metallic or persistent carriers, ensuring the sustainable maintenance of this technology. New MN shapes, such as gradient-length arrays, matrix-degrading enzymes, or enhanced ultrasound, will enable them to penetrate deeper into biofilms and better deliver drugs more effectively for both infectious and chronic diseases. Before transformative therapies can be widely used in clinics, safety and effectiveness must be proven. This will occur through clear rules, high-integrity validation, and partnerships among various sectors and academia.
Declaration of generative AI and AI-assisted writing tools
For manuscript preparation, the authors used Grammarly and Quillbot Premium to assist with copy-editing for gmar, style, and clarity. Fig. were created using BioRender, a professional illustration tool. The final version of the manuscript has been properly checked, verified, and approved by all the authors.
FUNDING
This review received no external funding.
AUTHORS CONTRIBUTIONS
Smriti Manibhushanam-Prepared the initial manuscript draft, conducted the literature review, structured the manuscript, and prepared the fig. and tables.
Umashankar Marakanam Srinivasan conceptualized the study and supervised the overall project.
Damodharan Narayanasamy-Primarily contributed through academic guidance and institutional support.
CONFLICT OF INTERESTS
The authors declare no conflict of interest
REFERENCES
Panda P, Mohanty T, Mohapatra R. Advancements in transdermal drug delivery systems: harnessing the potential of macromolecular-assisted permeation enhancement and novel techniques. AAPS PharmSciTech. 2025;26(1):29. doi: 10.1208/s12249-024-03029-9, PMID 39789371.
Bird D, Ravindra NM. Transdermal drug delivery and patches an overview. Med Devices Sens. 2020 Dec;3(6):e10069. doi: 10.1002/mds3.10069.
Tucak A, Sirbubalo M, Hindija L, Rahic O, Hadziabdic J, Muhamedagic K. Microneedles: characteristics materials production methods and commercial development. Micromachines. 2020;11(11):961. doi: 10.3390/mi11110961, PMID 33121041.
Ingrole RS, Azizoglu E, Dul M, Birchall JC, Gill HS, Prausnitz MR. Trends of microneedle technology in the scientific literature patents clinical trials and internet activity. Biomaterials. 2021;267:120491. doi: 10.1016/j.biomaterials.2020.120491, PMID 33217629.
Yang J, Liu X, Fu Y, Song Y. Recent advances of microneedles for biomedical applications: drug delivery and beyond. Acta Pharm Sin B. 2019;9(3):469-83. doi: 10.1016/j.apsb.2019.03.007, PMID 31193810.
Oliveira C, Teixeira JA, Oliveira N, Ferreira S, Botelho CM. Microneedles device: design fabrication and applications. Macromol. 2024;4(2):320-55. doi: 10.3390/macromol4020019.
Aldawood FK, Andar A, Desai S. A comprehensive review of microneedles: types materials processes characterizations and applications. Polymers (Basel). 2021;13(16):2815. doi: 10.3390/polym13162815, PMID 34451353.
Wu C, Yu Q, Huang C, Li F, Zhang L, Zhu D. Microneedles as transdermal drug delivery system for enhancing skin disease treatment. Acta Pharm Sin B. 2024;14(12):5161-80. doi: 10.1016/j.apsb.2024.08.013, PMID 39807331.
Ando D, Miyatsuji M, Sakoda H, Yamamoto E, Miyazaki T, Koide T. Mechanical characterization of dissolving microneedles: factors affecting physical strength of needles. Pharmaceutics. 2024;16(2):200. doi: 10.3390/pharmaceutics16020200, PMID 38399254.
Ahmed Saeed AL Japairai K, Mahmood S, Hamed Almurisi S, Reddy Venugopal J, Rebhi Hilles A, Azmana M. Current trends in polymer microneedle for transdermal drug delivery. Int J Pharm. 2020;587:119673. doi: 10.1016/j.ijpharm.2020.119673, PMID 32739388.
Faraji Rad Z, Prewett PD, Davies GJ. An overview of microneedle applications materials and fabrication methods. Beilstein J Nanotechnol. 2021;12:1034-46. doi: 10.3762/bjnano.12.77, PMID 34621614.
Avcil M, Celik A. Microneedles in drug delivery: progress and challenges. Micromachines (Basel). 2021;12(11):1321. doi: 10.3390/mi12111321, PMID 34832733.
Mitchell MJ, Billingsley MM, Haley RM, Wechsler ME, Peppas NA, Langer R. Engineering precision nanoparticles for drug delivery. Nat Rev Drug Discov. 2021;20(2):101-24. doi: 10.1038/s41573-020-0090-8, PMID 33277608.
Malik S, Muhammad K, Waheed Y. Emerging applications of nanotechnology in healthcare and medicine. Molecules. 2023;28(18):6624. doi: 10.3390/molecules28186624, PMID 37764400.
Phetporkha K, Saisawang C, Rattanavirotkul N, Thamphiwatana SD, Chantasart D. Dissolvable microneedles embedded with chitosan nanoparticles for transdermal delivery of pramipexole dihydrochloride in Parkinson’s disease. ACS Omega. 2025;10(35):39923-32. doi: 10.1021/acsomega.5c04299, PMID 40949249.
Hsu CY, Rheima AM, Kadhim MM, Ahmed NN, Mohammed SH, Abbas FH. An overview of nanoparticles in drug delivery: properties and applications. S Afr J Chem Eng. 2023;46:233-70. doi: 10.1016/j.sajce.2023.08.009.
Gowda BH, Ahmed MG, Sahebkar A, Riadi Y, Shukla R, Kesharwani P. Stimuli-responsive microneedles as a transdermal drug delivery system: a demand-supply strategy. Biomacromolecules. 2022;23(4):1519-44. doi: 10.1021/acs.biomac.1c01691, PMID 35274937.
Saini P, Mandal RK, Dubey A, Rahul Pal. Recent advances in transdermal drug delivery systems. J Pharm Insights Res. 2024;2(6):87-97. doi: 10.69613/p3cspw10.
Cheung K, Das DB. Microneedles for drug delivery: trends and progress. Drug Deliv. 2016;23(7):2338-54. doi: 10.3109/10717544.2014.986309, PMID 25533874.
Chen J, Ren H, Zhou P, Zheng S, Du B, Liu X. Microneedle mediated drug delivery for cutaneous diseases. Front Bioeng Biotechnol. 2022;10:1032041. doi: 10.3389/fbioe.2022.1032041, PMID 36324904.
Lu M, Zhang X, Luo Z, Zhao Y. Developing hierarchical microneedles for biomedical applications. Engineered Regen. 2023;4(3):316-27. doi: 10.1016/j.engreg.2023.04.004.
Jung JH, Jin SG. Microneedle for transdermal drug delivery: current trends and fabrication. J Pharm Investig. 2021;51(5):503-17. doi: 10.1007/s40005-021-00512-4, PMID 33686358.
Kim YC, Park JH, Prausnitz MR. Microneedles for drug and vaccine delivery. Adv Drug Deliv Rev. 2012;64(14):1547-68. doi: 10.1016/j.addr.2012.04.005, PMID 22575858.
Siddhapura K, Harde H, Jain S. Immunostimulatory effect of tetanus toxoid-loaded chitosan nanoparticles following microneedles assisted immunization. Nanomedicine. 2016;12(1):213-22. doi: 10.1016/j.nano.2015.10.009, PMID 26554391.
Nagarkar R, Singh M, Nguyen HX, Jonnalagadda S. A review of recent advances in microneedle technology for transdermal drug delivery. J Drug Deliv Sci Technol. 2020;59:101923. doi: 10.1016/j.jddst.2020.101923.
Tu J, Du G, Reza Nejadnik M, Monkare J, Van Der Maaden K, Bomans PH. Mesoporous silica nanoparticle-coated microneedle arrays for intradermal antigen delivery. Pharm Res. 2017;34(8):1693-706. doi: 10.1007/s11095-017-2177-4, PMID 28536970.
Nayak S, Suryawanshi S, Bhaskar V. Microneedle technology for transdermal drug delivery: applications and combination with other enhancing techniques. J Drug Delivery Ther. 2016;6(5):65-83. doi: 10.22270/jddt.v6i5.1285.
Pamornpathomkul B, Wongkajornsilp A, Laiwattanapaisal W, Rojanarata T, Opanasopit P, Ngawhirunpat T. A combined approach of hollow microneedles and nanocarriers for skin immunization with plasmid DNA encoding ovalbumin. Int J Nanomedicine. 2017;12:885-98. doi: 10.2147/IJN.S125945, PMID 28184159.
Ita K. Dissolving microneedles for transdermal drug delivery: advances and challenges. Biomed Pharmacother. 2017;93:1116-27. doi: 10.1016/j.biopha.2017.07.019, PMID 28738520.
Dugam S, Tade R, Dhole R, Nangare S. Emerging era of microneedle array for pharmaceutical and biomedical applications: recent advances and toxicological perspectives. Futur J Pharm Sci. 2021;7(1):19. doi: 10.1186/s43094-020-00176-1.
Permana AD, Anjani QK, Sartini, Utomo E, Volpe Zanutto F, Paredes AJ. Selective delivery of silver nanoparticles for improved treatment of biofilm skin infection using bacteria-responsive microparticles loaded into dissolving microneedles. Mater Sci Eng C Mater Biol Appl. 2021;120:111786. doi: 10.1016/j.msec.2020.111786, PMID 33545912.
Filho D, Guerrero M, Pariguana M, Marican A, Duran Lara EF. Hydrogel based microneedle as a drug delivery system. Pharmaceutics. 2023;15(10):2444. doi: 10.3390/pharmaceutics15102444, PMID 37896204.
Mohite P, Puri A, Munde S, Ade N, Kumar A, Jantrawut P. Hydrogel-forming microneedles in the management of dermal disorders through a non-invasive process: a review. Gels. 2024;10(11):719. doi: 10.3390/gels10110719, PMID 39590075.
Ding YW, Li Y, Zhang ZW, Dao JW, Wei DX. Hydrogel forming microneedles loaded with VEGF and ritlecitinib/polyhydroxyalkanoates nanoparticles for mini-invasive androgenetic alopecia treatment. Bioact Mater. 2024;38:95-108. doi: 10.1016/j.bioactmat.2024.04.020, PMID 38699241.
Martins CF, Garcia Astrain C, Conde J, Liz Marzan LM. Nanocomposite hydrogel microneedles: a theranostic toolbox for personalized medicine. Drug Deliv Transl Res. 2024;14(8):2262-75. doi: 10.1007/s13346-024-01533-w, PMID 38376619.
Hamdan I. Microneedle and drug delivery across the skin: an overview. Pharmacia. 2024;71:1-12. doi: 10.3897/pharmacia.71.e112503.
Bariya SH, Gohel MC, Mehta TA, Sharma OP. Microneedles: an emerging transdermal drug delivery system. J Pharm Pharmacol. 2012;64(1):11-29. doi: 10.1111/j.2042-7158.2011.01369.x, PMID 22150668.
Bolton CJ, Howells O, Blayney GJ, Eng PF, Birchall JC, Gualeni B. Hollow silicon microneedle fabrication using advanced plasma etch technologies for applications in transdermal drug delivery. Lab Chip. 2020;20(15):2788-95. doi: 10.1039/D0LC00567C, PMID 32632424.
Bhatnagar S, Gadeela PR, Thathireddy P, Venuganti VV. Microneedle-based drug delivery: materials of construction. J Chem Sci. 2019;131(9):1-128. doi: 10.1007/s12039-019-1666-x.
Cahill EM, Keaveney S, Stuettgen V, Eberts P, Ramos Luna P, Zhang N. Metallic microneedles with interconnected porosity: a scalable platform for biosensing and drug delivery. Acta Biomater. 2018;80:401-11. doi: 10.1016/j.actbio.2018.09.007, PMID 30201432.
Ita K. Ceramic microneedles and hollow microneedles for transdermal drug delivery: two decades of research. J Drug Deliv Sci Technol. 2018;44:314-22. doi: 10.1016/j.jddst.2018.01.004.
Bao L, Park J, Bonfante G, Kim B. Recent advances in porous microneedles: materials fabrication and transdermal applications. Drug Deliv Transl Res. 2022;12(2):395-414. doi: 10.1007/s13346-021-01045-x, PMID 34415566.
Kulkarni D, Gadade D, Chapaitkar N, Shelke S, Pekamwar S, Aher R. Polymeric microneedles: an emerging paradigm for advanced biomedical applications. Sci Pharm. 2023;91(2):27. doi: 10.3390/scipharm91020027.
Li H, Cui J, Zhang T, Lin F, Zhang G, Feng Z. Research progress on chitosan microneedle arrays in transdermal drug delivery. Int J Nanomedicine. 2024;19:12957-73. doi: 10.2147/IJN.S487313, PMID 39651356.
Liu C, Liu M, Li X, Hu Y, Zhang L, You FM. Unique advantages and applications of polysaccharide microneedles as drug delivery materials and in treatment of skin diseases. Nanoscale Adv. 2025;7(12):3631-54. doi: 10.1039/D4NA01083C, PMID 40417161.
Nguyen HX, Bozorg BD, Kim Y, Wieber A, Birk G, Lubda D. Poly (vinyl alcohol) microneedles: fabrication, characterization and application for transdermal drug delivery of doxorubicin. Eur J Pharm Biopharm. 2018;129:88-103. doi: 10.1016/j.ejpb.2018.05.017, PMID 29800617.
Zuo Y, Sun R, Del Piccolo N, Stevens MM. Microneedle mediated nanomedicine to enhance therapeutic and diagnostic efficacy. Nano Converg. 2024;11(1):15. doi: 10.1186/s40580-024-00421-w, PMID 38634994.
Ziesmer J, Sonden I, Venckute Larsson J, Merkl P, Sotiriou GA. Customizable fabrication of photothermal microneedles with plasmonic nanoparticles using low-cost stereolithography three-dimensional printing. ACS Appl Bio Mater. 2024;7(7):4533-41. doi: 10.1021/acsabm.4c00411, PMID 38877987.
Alimardani V, Abolmaali SS, Yousefi G, Rahiminezhad Z, Abedi M, Tamaddon A. Microneedle arrays combined with nanomedicine approaches for transdermal delivery of therapeutics. J Clin Med. 2021;10(2):181. doi: 10.3390/jcm10020181, PMID 33419118.
Santonocito D, Puglia C. Lipid nanoparticles and skin: discoveries and advances. Cosmetics. 2025;12(1):22. doi: 10.3390/cosmetics12010022.
Larraneta E, McCrudden MT, Courtenay AJ, Donnelly RF. Microneedles: a new frontier in nanomedicine delivery. Pharm Res. 2016;33(5):1055-73. doi: 10.1007/s11095-016-1885-5, PMID 26908048.
Chen M, Quan G, Sun Y, Yang D, Pan X, Wu C. Nanoparticles-encapsulated polymeric microneedles for transdermal drug delivery. J Control Release. 2020;325:163-75. doi: 10.1016/j.jconrel.2020.06.039, PMID 32629134.
Xia T, Zhu Y, Li K, Hao K, Chai Y, Jiang H. Microneedles loaded with cerium-manganese oxide nanoparticles for targeting macrophages in the treatment of rheumatoid arthritis. J Nanobiotechnology. 2024;22(1):103. doi: 10.1186/s12951-024-02374-y, PMID 38468261.
Jiang X, Zhao H, Li W. Microneedle mediated transdermal delivery of drug-carrying nanoparticles. Front Bioeng Biotechnol. 2022;10:840395. doi: 10.3389/fbioe.2022.840395, PMID 35223799.
Permana AD, Paredes AJ, Volpe Zanutto F, Anjani QK, Utomo E, Donnelly RF. Dissolving microneedle mediated dermal delivery of itraconazole nanocrystals for improved treatment of cutaneous candidiasis. Eur J Pharm Biopharm. 2020;154:50-61. doi: 10.1016/j.ejpb.2020.06.025, PMID 32649991.
Shan Y, Tan B, Zhang M, Xie X, Liao J. Restorative biodegradable two layered hybrid microneedles for melanoma photothermal/chemo co-therapy and wound healing. J Nanobiotechnology. 2022;20(1):238. doi: 10.1186/s12951-022-01426-5, PMID 35590414.
Zhao Y, Tian Y, Ye W, Wang X, Huai Y, Huang Q. A lipid polymer hybrid nanoparticle (LPN)-loaded dissolving microneedle patch for promoting hair regrowth by transdermal miR-218 delivery. Biomater Sci. 2022;11(1):140-52. doi: 10.1039/D2BM01454H, PMID 36385302.
Foram Bhatt, Dipti Patel. Fabrication and screening of solid lipid nanoparticles loaded microneedle patch for polycystic ovary syndrome treatment. Asian J Pharm Clin Res. 2025;18(7):109-14. doi: 10.22159/ajpcr.2025v18i7.54740.
Noor AH, Ghareeb MM. Release ondansetron hydrogen chloride nanoparticles. Int J Drug Deliv Technol. 2021;11:858-63.
Surekha B, Misra P, Thippaiah AC, Shamanna BR, Madathil A, Rajadurai M. A microneedle transdermal patch loaded with iron(ii) nanoparticles for non-invasive sustained delivery to combat anemia. Mater Adv. 2024;5(8):3247-56. doi: 10.1039/D3MA01166F.
Deshpande A, Mer V, Patel D, Thakkar H. Microneedle assisted transdermal delivery of carvedilol nanosuspension for the treatment of hypertension. RSC Pharm. 2024;1(3):472-83. doi: 10.1039/D4PM00038B.
Prabhu A, Baliga V, Shenoy R, Dessai AD, Nayak UY. 3D printed microneedles: revamping transdermal drug delivery systems. Drug Deliv Transl Res. 2025;15(2):436-54. doi: 10.1007/s13346-024-01679-7, PMID 39103595.
Olowe M, Parupelli SK, Desai S. A review of 3D-printing of microneedles. Pharmaceutics. 2022;14(12):2693. doi: 10.3390/pharmaceutics14122693, PMID 36559187.
Petrova E, Chvila S, Stepanek F, Zbytovska J, Lamprou DA. Imiquimod nanocrystal-loaded dissolving microneedles prepared by DLP printing. Drug Deliv Transl Res. 2025;15(1):158-70. doi: 10.1007/s13346-024-01567-0, PMID 38472727.
Luo X, Yang L, Cui Y. Microneedles: materials fabrication and biomedical applications. Biomed Microdevices. 2023;25(3):20. doi: 10.1007/s10544-023-00658-y, PMID 37278852.
Zarei Chamgordani N, Asiaei S, Ghorbani Bidkorpeh F, Babaee Foroutan M, Mahboubi A, Moghimi HR. Fabrication of controlled release silver nanoparticle polylactic acid microneedles with long-lasting antibacterial activity using a micro-molding solvent casting technique. Drug Deliv Transl Res. 2024;14(2):386-99. doi: 10.1007/s13346-023-01406-8, PMID 37578649.
Tang Y, Li S, Hu L, Sun X, Zhang B, Ji W. Hybrid poly(AMPS-CS)-Au microneedle arrays to enrich metabolites from skin for early disease diagnosis. Adv Healthc Mater. 2021;10(19):e2100764. doi: 10.1002/adhm.202100764, PMID 34028989.
Kim MJ, Park SC, Rizal B, Guanes G, Baek SK, Park JH. Fabrication of circular obelisk-type multilayer microneedles using micro-milling and spray deposition. Front Bioeng Biotechnol. 2018;6:54. doi: 10.3389/fbioe.2018.00054, PMID 29868571.
Angkawinitwong U, Courtenay AJ, Rodgers AM, Larraneta E, McCarthy HO, Brocchini S. A novel transdermal protein delivery strategy via electrohydrodynamic coating of PLGA microparticles onto microneedles. ACS Appl Mater Interfaces. 2020;12(11):12478-88. doi: 10.1021/acsami.9b22425, PMID 32066234.
Jung S, Chang S, Kim NE, Choi SO, Song YJ, Yuan Y. Curcumin/zeolitic imidazolate framework-8 nanoparticle-integrated microneedles for pH-responsive treatment of skin disorders. ACS Appl Nano Mater. 2022;5(9):13671-9. doi: 10.1021/acsanm.2c03884.
Zhang Y, Li J, Wu M, Guo Z, Tan D, Zhou X. Glucose responsive gold nanocluster loaded microneedle patch for type 1 diabetes therapy. ACS Appl Bio Mater. 2020;3(12):8640-9. doi: 10.1021/acsabm.0c01042, PMID 35019634.
Xu B, Cao Q, Zhang Y, Yu W, Zhu J, Liu D. Microneedles integrated with ZnO quantum-dot-capped mesoporous bioactive glasses for glucose-mediated insulin delivery. ACS Biomater Sci Eng. 2018;4(7):2473-83. doi: 10.1021/acsbiomaterials.8b00626, PMID 33435111.
Zhang Y, Zhang X, Wu X, Zhao Y. Photo-responsive polydopamine nanoenzyme microneedles with oxidative stress regulation ability for atopic dermatitis treatment. Nano Today. 2024;56:102241. doi: 10.1016/j.nantod.2024.102241.
Xie R, Li W, Shi K, Yang L, Chen H, Jiang S. Versatile platforms of microneedle patches loaded with responsive nanoparticles: synthesis and promising biomedical applications. Adv NanoBiomed Res. 2024;4(4):2300142. doi: 10.1002/anbr.202300142.
Zhang X, Wang Z, Jiang H, Zeng H, An N, Liu B. Self-powered enzyme-linked microneedle patch for scar-prevention healing of diabetic wounds. Sci Adv. 2023;9(28):eadh1415. doi: 10.1126/sciadv.adh1415, PMID 37450590.
Yang XX, Feng P, Cao J, Liu W, Tang Y. Composition-engineered metal–organic framework-based microneedles for glucose-mediated transdermal insulin delivery. ACS Appl Mater Interfaces. 2020;12(12):13613-21. doi: 10.1021/acsami.9b20774, PMID 32138507.
Liu D, Zhang Y, Jiang G, Yu W, Xu B, Zhu J. Fabrication of dissolving microneedles with thermal-responsive coating for NIR-triggered transdermal delivery of metformin on diabetic rats. ACS Biomater Sci Eng. 2018;4(5):1687-95. doi: 10.1021/acsbiomaterials.8b00159, PMID 33445325.
Seetharam AA, Choudhry H, Bakhrebah MA, Abdulaal WH, Gupta MS, Rizvi SM. Microneedles drug delivery systems for treatment of cancer: a recent update. Pharmaceutics. 2020;12(11):1101. doi: 10.3390/pharmaceutics12111101, PMID 33212921.
Ma J, Tai Z, Li Y, Li Y, Wang J, Zhou T. Dissolving microneedle-based cascade-activation nanoplatform for enhanced photodynamic therapy of skin cancer. Int J Nanomedicine. 2024;19:2057-70. doi: 10.2147/IJN.S443835, PMID 38482522.
Zhou X, Luo Z, Baidya A, Kim HJ, Wang C, Jiang X. Biodegradable β-cyclodextrin conjugated gelatin methacryloyl microneedle for delivery of water-insoluble drug. Adv Healthc Mater. 2020;9(11):e2000527. doi: 10.1002/adhm.202000527, PMID 32364331.
Liu F, Cheng Z, Yi H. NIR light activatable dissolving microneedle system for melanoma ablation enabled by a combination of ROS-responsive chemotherapy and phototherapy. J Nanobiotechnology. 2023;21(1):61. doi: 10.1186/s12951-023-01815-4, PMID 36814244.
Yang Y, Li Z, Huang P, Lin J, Li J, Shi K. Rapidly separating dissolving microneedles with sustained release colchicine and stabilized uricase for simplified long-term gout management. Acta Pharm Sin B. 2023;13(8):3454-70. doi: 10.1016/j.apsb.2023.02.011, PMID 37655319.
Bao C, Li Z, Liang S, Hu Y, Wang X, Fang B. Microneedle patch delivery of capsaicin-containing α-lactalbumin nanomicelles to adipocytes achieves potent anti-obesity effects. Adv Funct Materials. 2021;31(20):2011130. doi: 10.1002/adfm.202011130.
Xuan Q, Cai J, Gao Y, Qiao X, Jin T, Peydayesh M. Amyloid-templated ceria nanozyme reinforced microneedle for diabetic wound treatments. Adv Mater. 2025;37(15):e2417774. doi: 10.1002/adma.202417774, PMID 39995378.
Yao WD, Zhou JN, Tang C, Zhang JL, Chen ZY, Li Y. Hydrogel microneedle patches loaded with stem cell mitochondria-enriched microvesicles boost the chronic wound healing. ACS Nano. 2024;18(39):26733-50. doi: 10.1021/acsnano.4c06921, PMID 39238258.
Xu J, Danehy R, Cai H, Ao Z, Pu M, Nusawardhana A. Microneedle patch-mediated treatment of bacterial biofilms. ACS Appl Mater Interfaces. 2019;11(16):14640-6. doi: 10.1021/acsami.9b02578, PMID 30933463.