
Int J App Pharm, Vol 18, Issue 1, 2026, 325-331Original Article
TOPICAL LEVODOPA-CARBIDOPA EYE DROPS FOR MYOPIA CONTROL: A PILOT STUDY IN A RABBIT MODEL OF FORM-DEPRIVATION MYOPIA
CHRISTINA ARITONANG1,2,4, IRWANTO IRWANTO3,4*, RENI PRASTYANI2,4
1Doctoral Program of Medical Science, Faculty of Medicine, Universitas Airlangga, Surabaya-60132, Indonesia. 2Department of Ophthalmology, Faculty of Medicine, Universitas Airlangga, Surabaya-60286, Indonesia. 3Department of Child Health, Faculty of Medicine, Universitas Airlangga, Surabaya-60286, Indonesia. 4Dr. Soetomo General Academic Hospital, Surabaya-60286, Indonesia
*Corresponding author: Irwanto Irwanto; *Email: irwanto@fk.unair.ac.id
Received: 14 Jul 2025, Revised and Accepted: 27 Oct 2025
ABSTRACT
Objective: Dopaminergic drugs may be repurposed to modulate ocular growth. Our study aims to evaluate the safety, stability, and efficacy of a novel levodopa-carbidopa topical ophthalmic formulation for controlling myopia progression in the form-deprivation myopia (FDM) rabbit model.
Methods: A 4:1 molar ratio of levodopa to carbidopa was formulated into an eye drop with a pH of 5.8 and stored at 4 °C in light-protected glass containers. We used 14 New Zealand white rabbits, which received form-deprivation myopia induction and were divided into two groups: seven into the control group and the other seven into the intervention group, the latter receiving treatment with the prepared levodopa-carbidopa formulation. Ocular safety was assessed using a slit-lamp exam, Schirmer’s test, and behavioral observations. Efficacy was represented by axial length and refraction, with retinal tissues being harvested for ELISA quantification of dopaminergic markers.
Results: Levodopa-carbidopa formulation remained clear and stable with no visual signs of microbial contamination or degradation during the study period. No signs of ocular irritation or systemic toxicity were observed. Schirmer’s test values showed no significant difference pre-and post-treatment, proving the absence of drug-induced ocular irritation (p>0.05). Treatment with levodopa–carbidopa significantly inhibited axial elongation (18.16 mm vs. 23.12 mm, p = 0.013) and reversed myopic refractive error (4.57±1.06 vs. 2.64±1.41, p = 0.013) compared to the control group. Retinal tyrosine hydroxylase (TH) and dopamine receptor D2 (D2R) expression were significantly elevated in the treated group (p = 0.006 and p = 0.036, respectively), supporting enhanced dopaminergic signaling.
Conclusion: Topical levodopa–carbidopa is a stable, well-tolerated formulation that suppresses progression of myopia in a rabbit model. The therapeutic effect is associated with enhanced retinal dopamine activity. These findings further support a pharmacologic strategy for myopia control.
Keywords: Dopaminergic signaling, Levodopa-carbidopa, Myopia, Ocular pharmacology, Health
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.56071 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Over the past ten years, myopia has emerged as one of the most important global public health concerns. Myopia is prevalent in Asia, where it affects over 80% of school-age children in some cities [1]. The etiology of myopia includes environmental factors such as excessive screen time and limited time outdoors, as well as genetic predisposition [2]. The underlying molecular processes that contribute to axial elongation are still being studied.
Studies have indicated that dopamine functions as a negative feedback mechanism that inhibits excessive ocular growth, making dopaminergic signaling a crucial modulator [3, 4]. Animal models of induced myopia have shown decreased dopamine levels, confirming the neurotransmitter’s function in ocular development [4]. Research suggests that enhancing dopaminergic signaling could slow down or even reverse myopic progression [4, 5]. Experimental studies in various species have investigated the potential of increasing dopamine levels or dopamine receptor activity to inhibit myopic growth in form deprivation myopia (FDM) models [5–7].
Administration of synthetic dopamine or its metabolic precursor, such as levodopa, has been shown to suppress myopia by increasing retinal dopamine synthesis and release [8, 9]. It is well known that the dopamine precursor levodopa can pass through physiological barriers, such as the blood-brain barrier. Systemic bioavailability and therapeutic efficacy are improved when combined with carbidopa, which prevents levodopa from being converted to dopamine peripherally [10]. Because of these characteristics, the combination is a desirable option for topical ophthalmic administration of localized retinal targeting. This approach not only enhances the delivery of dopamine directly to the retina but also minimizes systemic side effects [9].
Intravitreal dopamine in a lid-suture FDM model rabbits have been conducted, but FDM induction using this model may cause corneal flattening [11, 12]. Another recent research investigated the use of topical dopamine in an FDM-induced rabbit model with a diffuser. The result of the study demonstrated topical dopamine’s efficacy in reducing axial elongation with no histopathological toxicity in the eye [12]. Topical eye drop experiments of levodopa-carbidopa have been conducted in chicks, yet differences in avian anatomy, such as the presence of cartilaginous layers in the sclera, necessitate further investigation in mammals [8-10].
Previous studies have not conducted experiments on topical eye drops of levodopa-carbidopa in mammals with FDM. Thus, this study aims to formulate and assess a levodopa-carbidopa eye drop formulation's pharmacological stability, efficacy in inhibiting axial elongation, and ocular safety in animal models, particularly rabbits with FDM. This study also investigates the biochemical changes associated with the intervention of levodopa-carbidopa, especially related to the dopamine receptors and tyrosine hydroxylase (TH) expression in the retina. The findings from this research could offer helpful information regarding the potential application of levodopa-carbidopa as a pharmacological management for myopia and provide new insights into retinal dopamine signaling to help control myopia.
MATERIALS AND METHODS
Ethical approval
Authorization to conduct experiments using animals was approved by the Animal Care and Use Committee, Faculty of Veterinary Medicine, Universitas Airlangga (No. 2. KEH.160.10.2023). All of the protocols related to experimental animals were performed according to the Association for Research in Vision and Ophthalmology’s (ARVO) Statement for the Use of Animals in Ophthalmic and Vision Research. Ocular measurements were conducted after a local anesthetic of tetracaine 0.5% was administered and anesthetized with ketamine (30 mg/kg) and xylazine (5 mg/kg) before tissue collection.
Study period and location
This research was performed during January-March 2025 at the Institute of Tropical Diseases, Universitas Airlangga, Surabaya. This study focused on establishing the formulation for levodopa-carbidopa combination therapy as topical eye drops and assessing its formulation, its relevant stabilizers, handling requirements, and dose optimization strategies. In addition, an animal study model was performed to experiment with the ocular delivery route of topical levodopa-carbidopa and its efficacy in inhibiting myopia progression and safety profiles.
Formulation of levodopa-carbidopa
Based on previous studies, a 4:1 molar ratio of levodopa to carbidopa was selected to mimic systemic use while maintaining safety. This study used 3-(3,4-dihydroxyphenyl)-L-alanine (Tokyo Chemical Industry, Japan, D0600-25G) and carbidopa monohydrate (Tokyo Chemical Industry, Japan, C2450-1G). Ascorbic acid is commonly used as a stabilizer in pharmaceutical formulations due to its potent antioxidant properties, which help prevent the oxidative degradation of sensitive compounds such as catecholamines, including levodopa and carbidopa [16]. In this study, the formulation is supplemented with 0.1% ascorbic acid (pH 2-4) as a stabilizer to prevent oxidative degradation. Furthermore, it was dissolved in 1 liter of 1× phosphate-buffered saline with pH 7.4. The final pH of the solution was adjusted from an acidic pH to a pH closer to neutral (pH of 5.8), designed to enhance corneal permeability without compromising the integrity of the blood-ocular barrier [13]. The levodopa-carbidopa eye drops were formulated in a sterile lab at the Pharmacology Laboratory at the Faculty of Pharmacy, Universitas Airlangga.
Avoiding light exposure is one way to increase the stability of levodopa and carbidopa formulations [14]. Thus, the solution is kept in glass bottles sealed with rubber stoppers and covered with black duct tape to prevent light damage and minimize degradation. The temperature was also regulated to avoid degradation. All of the glass bottles were gathered into a single, large glass container, which was kept in a refrigerator at 4 °C. According to studies, the formulation degrades by less than 5% in 4 d at 4 °C [14]. To prepare the concentration of levodopa-carbidopa ophthalmic solution, the required amount was calculated based on the stock concentration of levodopa (15 mmol) and carbidopa (3.75 mmol), with each multiplied by its respective molecular weight (table 1). Each eye drop was standardized to a volume of 50 µl** (0.05 ml). Dosage calculations yielded a final concentration of 0.15 mg levodopa and 0.05 mg carbidopa. Levodopa-carbidopa eye drops were given twice daily for five days to treatment groups of fourteen rabbits each. This totaled 140 individual doses, requiring a total volume of 7,000 µl** (7 ml). Each bottle was filled with 10 ml of formulation to provide further backup for the liquid with the surface intact. For larger-scale preparation, a 50 ml levodopa solution would require approximately 76 mg of levodopa, while a 75 ml carbidopa solution would require about 150 mg of carbidopa, calculated based on the amount of drug per drop and total volume needed.
Table 1: Dosage calculation
| Properties | Levodopa | Carbidopa |
Molecular weight Concentration |
197.19 g/mol 15 mmol |
244.25g/mol 3,75 mmol |
| Conversion | = 15 × 10-3 M/l × 197.19 g/mol = 2.95785 g/l |
= 3.75 × 10-3 M/l × 244.25 g/mol = 0.9159375 g/l |
| Drug administration | 2×/day, 50 µl** | 2×/day, 50 µl** |
| Dose in 50 µl** | 50 µl** × 2.95785 g/l = 0.15 mg | 50 µl** × 0.9159375 g/l = 0.05 mg |
Experimental design
New Zealand white rabbits (n = 14), which were healthy young male rabbits, were obtained from a certified breeder specifically for research and were housed under standard laboratory conditions. Rabbits were kept in temperature-controlled rooms with laminar air flow. Rabbits were divided into a control group, which only received FDM and an intervention group, which received FDM followed by levodopa-carbidopa (LC). The required sample size per group was calculated using the Lemeshow formula for comparing two means. Substituting these values yielded an estimated minimum sample size of 5.16 animals per group. To account for potential attrition, non-measurable data, or experimental loss, we increased this by 20%, resulting in 7 animals per group. Given two experimental groups, the total sample size was therefore 14 animals (n = 7 per group). Similarly, our chosen group size is consistent with prior experimental myopia studies in rabbits and other animal models, where typical group sizes range from 8 animals to achieve adequate power for morphological and biochemical endpoints [14]. The statistical analysis was conducted by a qualified statistician based in Universitas Airlangga, Surabaya, Indonesia.
Before induction of FDM, rabbits underwent a 7 d acclimatization period in a controlled environment (temperature 22 °C; 12 h cycle of light/dark) and were provided with commercial feed, vegetables, and unlimited access to drinking water. Rabbits. Health and ocular status were confirmed before inclusion. After acclimatization, form-deprivation myopia was induced by monocular occlusion of the right eye for 30 consecutive days using a black plastic cap occluder. The occluder was then secured with Leukoplast (OneMed, Indonesia). The occlusion was maintained and replaced every three days under dim light illumination conditions. After the completion of the 30 d FDM induction, axial length and refractive status were measured in all groups to establish baseline pre-treatment parameters. These measurements served to confirm the successful induction of myopia and to ensure comparability between groups prior to intervention. Subsequently, the assigned interventions for each group were administered daily over a five-day treatment period. Previous studies in avian models have demonstrated that short-duration treatment with levodopa-carbidopa for four days can induce significant anti-myopic effects [8]. The dosage used in this study was one drop of 50 µl** given twice daily (09:30 and 13:30).
Evaluation
Formulation stability was monitored via visual inspection (color, clarity) and pH measurement. Schirmer tests using blotting strips of paper were performed before data collection on the efficacy of levodopa-carbidopa. Axial length and refractive status measurements were conducted after a local anesthetic of tetracaine 0.5% was administered topically (Cendo, Indonesia). Ocular safety was performed using slit lamp examination. Besides ocular safety, behavioral changes were noted by full-time professional veterinarians who are on duty and conduct regular ward rounds daily. Schirmer’s test was used as a quantitative indicator of tear film production to evaluate potential ocular surface irritation or lacrimal gland dysfunction before and after topical administration, providing a non-invasive measure of formulation safety. Schirmer’s test was conducted pre-and post-treatment.
Data collection
Axial length was measured using non-contact biometry using 10 MHz A-scan ultrasound (PacScan 300 Plus; Sonomed, USA) and an autorefractokeratometer (ARK) (Handyheld Ref/Keratometer HandyRef-K; NIDEK, Japan). Axial lengths were retrieved from the mean values of five measurements. Refractive status was measured using streak retinoscopy (RX 3A; Neitz, Japan). All data collection of axial length and refractive status were done by the principal investigator to minimize inter-observer reliability variation. After retrieving axial and refractive status data, rabbits were anesthetized with ketamine (30 mg/kg) and xylazine (5 mg/kg). Tissue collection from the retina was done using eye enucleation. Enucleated eyes were then harvested for retinal tissue and dissected under sterile conditions in a laminar flow cabinet with red light illumination. Retinal tissue (10–20 mg) was homogenized with 1500 μl PRO-PREP (Intron Biotechnology, South Korea) lysis buffer and protease inhibitor, incubated on ice, and centrifuged at 13,000 rpm at 4 °C for 5 min, after which the supernatant was collected for protein analysis. The retinal tissue was extracted from the FDM animal model using the PRO-PREP reagent. Concentrations of TH, dopamine receptor D1 (D1R) and dopamine receptors D2 (D2R) were quantified in retinal tissue using ELISA according to the manufacturer’s protocol using a specific kit for rabbits (D1R, D2R, and TH using Reed Biotech Ltd 96T, China) in Dr. Soetomo General Academic Hospital, Surabaya. Absorbance was measured at a designated wavelength using a microplate reader, and concentrations were determined by interpolating absorbance values onto a standard curve.
Statistical analysis
Statistical analysis was performed in SPSS v.26 (IBM, USA). Data normality was assessed using the Shapiro-Wilk test. Data are presented as mean±standard deviation (SD) for evenly distributed data, while unequally distributed data are presented as median (interquartile range [IQR]). Pre-and post-treatment results within groups were analyzed using the paired t-test or Wilcoxon signed-rank tests. Meanwhile, comparisons between the control and treatment groups were analyzed using an independent t-test or the Mann-Whitney U test. P values of less than 0.05 were considered statistically significant.
RESULTS
Formulation characteristics and its stability
The storage approach using glass bottles, rubber stoppers, and light-blocking tape was effective in preserving the formulation’s integrity. The levodopa-carbidopa ophthalmic solution produced a transparent, colorless liquid at room temperature, exhibiting no visible particles or precipitates throughout the study duration. The results demonstrated that ascorbic acid served as an effective antioxidant, reducing oxidative damage during the experiment. The final pH was consistently maintained at 5.8 to optimize drug permeability in the cornea and minimize ocular irritation. No discoloration, turbidity, or precipitate formation was observed in any of the samples visually assessed over the 10 d study period pH range, showing only a slight deviation from the baseline pH of 5.8. No leakage, evaporation, or external contamination was detected in any of the stored samples. Refrigeration at 4 °C further contributed to maintaining the solution’s chemical and microbiological stability. No fungal or bacterial growth was observed in any vials, as confirmed by visual and olfactory inspection. The prepared 10 ml volume was sufficient to cover all 140 administrations with minimal residual wastage, demonstrating precise volume planning and efficient dosing strategies. No reflux or overflow of the instilled drops was noted in the animals, indicating efficient administration without excessive drainage or loss of formulation.
Ocular safety and tolerability
Before the enucleation process for retinal harvesting, slit lamp examination showed no evidence of conjunctival hyperemia, corneal clouding, opacity, or edema, abnormal discharge or excessive tearing (lacrimation), or signs of anterior chamber reaction or flare. No signs of ocular irritation were observed following the administration of the levodopa-carbidopa formulation over the 5 d dosing period. Rabbits exhibited no scratching behavior or grooming of the periocular region, suggesting no discomfort or irritant sensation after instillation. Furthermore, no blepharospasm occurred, ocular reflexes remained intact, and blinking frequency was unaltered, further supporting the tolerability of the formulation.
Table 2: Schirmer’s test results (mm/5 min) in the control and treated groups
| Groups | Pre-treatment | Post-treatment | Pre vs post-treatment p-value | FDM vs LC (Post) p-value |
| FDM controls (n = 7) | 15.29±4.89 | 15.42±3.41 | 0.948 | 0.820 |
| FDM+LC (n = 7) | 16.71±4.54 | 15.86±3.48 | 0.457 |
Sample size (n) is 7 for all groups. Significance is set at p<0.05. LC: levodopa-carbidopa. Data are presented as mean±SD.
Schirmer’s test showed no significant difference between pre-treatment and post-treatment values in either group: In the FDM control group, tear production remained stable (15.29±4.89 mm/5 min pre-treatment vs 15.42±3.41 mm/5 min post-treatment, p = 0.948), indicating that form deprivation alone did not alter lacrimal function. Similarly, in the levodopa–carbidopa-treated group, there was no significant change (16.71±4.54 mm/5 min pre-treatment vs 15.86±3.48 mm/5 min post-treatment, p = 0.457), suggesting that the drug formulation did not impair tear secretion. Furthermore, the between-group comparison of post-treatment values also showed no statistically significant difference (p = 0.820), reinforcing that the levodopa-carbidopa formulation was comparable to the control, which did not receive any treatment of topical ocular drugs, in terms of ocular surface safety. Schirmer's test stayed at baseline values in all of the rabbits that were treated, indicating no epithelial surface damage due to the formulation.
Throughout the study, all rabbits maintained normal activity levels, with feeding and grooming behavior remaining consistent with pre-treatment baselines. Routine daily assessments did not reveal any abnormal behavior, lethargy, or reduced food intake. No adverse systemic signs were recorded. Animals tolerated the topical eye drop procedure well, with no requirement for anesthesia or sedation. These results indicated that the formulation did not produce systemic toxicity or stress when applied topically to the eye.
Efficacy of levodopa-carbidopa in inhibiting myopia progression
The noticeable shift from pre-treatment to post-treatment values in the analysis of axial length measurements (fig. 2) indicates a significant increase in axial elongation in the FDM control group. On the other hand, the group treated with levodopa–carbidopa showed a significantly lower increase in axial length, with post-treatment values remaining relatively close to baseline. These results imply that topical levodopa–carbidopa successfully inhibited the axial elongation caused by form deprivation. Analytical findings in axial length are also supported by corresponding changes in refractive status. Following treatment, the diopter values of the FDM control group decreased, which is consistent with a myopic shift. In contrast, levodopa-carbidopa-treated FDM showed an increase in refractive power following treatment, which may indicate a reduction in myopic shift or a refractive shift toward hyperopia. When combined, the functional (refraction) and anatomical (axial length) data provide compelling evidence for the role of topical levodopa–carbidopa-induced dopaminergic stimulation in regulating the progression of myopia in this experimental model.
Following treatment, the axial length analysis showed notable variations within and between groups. Before treatment, there were no statistically significant differences in the axial lengths of the levodopa–carbidopa and FDM control groups. In contrast, the FDM control group's post-treatment axial length increased significantly to 23.12±2.34 mm, which is consistent with progressive axial elongation caused by myopia. Levodopa–carbidopa-treated rabbits, on the other hand, showed a significantly reduced axial length after treatment, with a median value of 18.16 mm (IQR: 18.01–21.25 mm), indicating that the intervention successfully inhibited ocular growth.
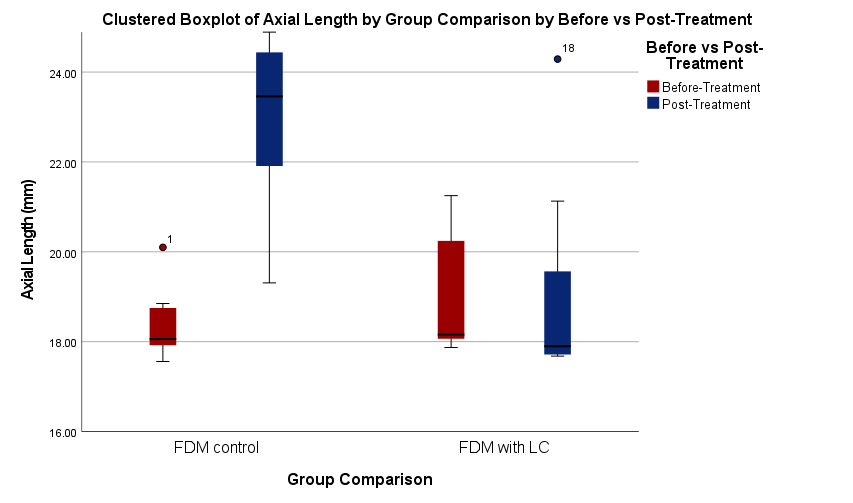
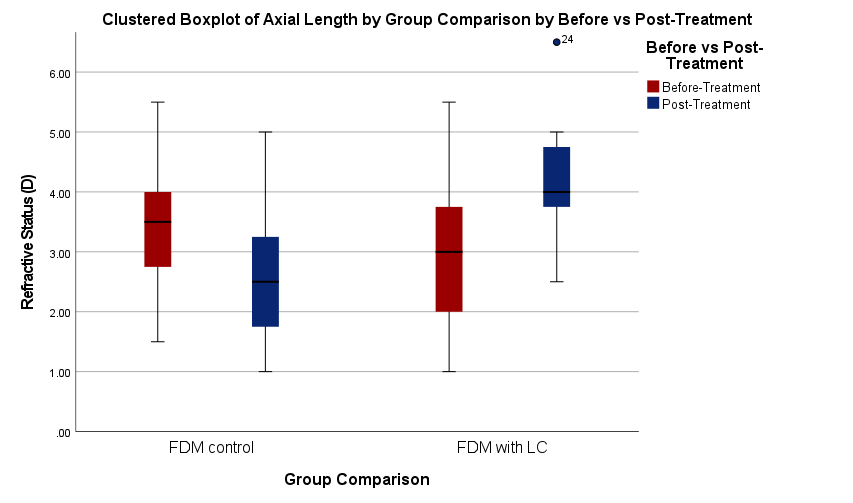
Fig. 2: Clustered boxplot of axial length and refractive status by group (n = 7 for both groups). FDM: Form deprivation myopia, LC: levodopa-carbidopa
Table 3: Changes in axial lengths (mm) in the control and treated groups
| Groups | Pre-treatment | Post-treatment | Pre vs post-treatment p-value | FDM vs LC (Post) p-value |
| FDM controls | 18.44±0.86 | 23.12±2.34 | 0.003* | 0.013* |
| FDM+LC | 18.06 (17.92-18.85) | 18.16 (18.01-21.25) | 0.018* |
Sample size (n) is 7 for all groups. Significance is set at p<0.05. LC: levodopa-carbidopa. Data are presented as mean±SD.
Statistical analysis using within-group comparisons showed significant axial elongation in the FDM control group (p = 0.003), validating the successful induction of myopia. Although the difference between the axial lengths before and after treatment was statistically significant (p = 0.018) in the levodopa–carbidopa group, it was significantly less than in the untreated group. Further evidence that levodopa–carbidopa eye drops prevent form-deprivation-induced axial elongation was provided by the significant intergroup difference (p = 0.013) found when comparing the post-treatment axial lengths between the FDM control and levodopa–carbidopa groups.
Table 4: Changes in refractive status (D) in the control and treated groups
| Groups | Pre-treatment | Post-treatment | Pre vs post-treatment p-value |
FDM vs LC (Post) p-value |
| FDM controls | 3.43±1.33 | 2.64±1.41 | 0.001* | 0.013* |
| FDM+LC | 3.00±1.53 | 4.57±1.06 | 0.001* |
Sample size (n) is 7 for all groups. Significance is set at p<0.05. LC: levodopa-carbidopa. Data are presented as mean±SD for evenly distributed data.
Changes in refractive status also paralleled the axial length findings. At this developmental stage, rabbits’ ocular parameters are still developing, and it is well-documented that young rabbits exhibit baseline hyperopia. Diopter values in the FDM control group significantly decreased from 3.43±1.33 D at baseline to 2.64±1.41 D following treatment (p = 0.001), suggesting a refractive status shift. However, refractive values in the levodopa–carbidopa group showed a significant improvement from 3.00±1.53 D before treatment to 4.57±1.06 D after treatment (p = 0.001), indicating a shift towards hyperopia and a partial recovery of refractive function. This reduction in myopic shift implies that levodopa–carbidopa therapy may both prevent additional myopic progression and return refractive status to normal emmetropia. The FDM control and treated groups’ post-treatment refractive values showed a statistically significant difference (p = 0.013) when compared intergroup, further confirming the therapeutic effectiveness of levodopa–carbidopa-induced dopaminergic stimulation.
The molecular effects of levodopa–carbidopa treatment were evaluated by biochemically analyzing retinal dopaminergic markers. TH, the enzyme that limits the rate of dopamine synthesis, was expressed at a significantly higher level in the treated group (12.74±1.23 ng/ml) than in the FDM control group (10.85±0.76 ng/ml), with a p of 0.006. This increase in TH supports the hypothesis that levodopa–carbidopa enhances retinal dopamine synthesis.
Table 5: Level of TH, D1R, and D2R expression post-treatment in the control and treated groups
| Group | TH (ng/ml) | D1R (pg/ml) | D2R (pg/ml) | |||
| Mean±SD | p-value | Mean±SD | p-value | Mean±SD | p-value | |
| FDM controls | 10.85±0.76 | 0.006* | 3520±861 | 0.081 | 1300±124 | 0.036* |
| FDM+LC | 12.74±1.23 | 4334±730 | 1468±140 | |||
Sample size (n) is 7 for all groups. Significance is set at p<0.05. LC: levodopa-carbidopa. Data are presented as mean±SD.
While the D1R levels were higher in the levodopa–carbidopa group (4334±730 pg/ml) compared to the FDM control group (3520±861 pg/ml), the difference did not reach statistical significance (p = 0.081). This suggests a possible trend that may require a larger sample size or longer treatment period to validate. In contrast, expression of the D2R was significantly higher in the treated group (1468±140 pg/ml) than in the FDM controls (1300±124 pg/ml), with a p of 0.036. This finding implies that levodopa–carbidopa may preferentially activate or upregulate D2R signaling, which is known to play a role in suppressing abnormal ocular growth in experimental myopia models. Collectively, these results indicate that levodopa–carbidopa therapy exerts both anatomical and molecular effects that are beneficial for inhibiting the progression of FDM. The observed improvements in axial length and refractive status, along with upregulation of TH and D2R expression, support the potential of dopaminergic pathways as a target for therapeutic intervention in myopia control.
DISCUSSION
Our study evaluated the safety, stability, and therapeutic efficacy of a novel levodopa–carbidopa topical ophthalmic formulation for the treatment of myopia using a rabbit model with FDM. Our findings demonstrate that this dopaminergic combination can be formulated as a stable eye drop preparation, is well tolerated in rabbit eyes, and significantly reduces axial elongation and myopic refractive shifts without inducing ocular toxicity.
The formulation was developed based on well-established pharmaceutical principles. Ascorbic acid was used to stabilize levodopa by preventing oxidative degradation, a common limitation in aqueous dopamine-based preparations [15]. Additionally, the pH was adjusted to balance corneal permeability and ocular comfort, per prior pharmacokinetic and tolerability studies [13]. These optimizations are essential when repurposing a drug class known for systemic use, such as levodopa-carbidopa for Parkinson’s disease, for localized ocular delivery. In systemic therapy, co-administration with carbidopa prevents the peripheral metabolism of levodopa [16, 17]. As a result, levodopa’s plasma half-life increases, its bioavailability to the central nervous system is enhanced, and therapeutic doses can be reduced [16, 17]. Translating this strategy to the retina allows for more efficient retinal dopamine delivery while minimizing systemic side effects, which is a key consideration for long-term use, particularly in pediatric patients.
The biological rationale for enhancing retinal dopaminergic signaling in myopia management is well supported by prior literature. Numerous studies across animal models—including chicks, guinea pigs, mice, and primates—have shown that dopamine and its precursors play a protective role against myopic eye growth, with low retinal dopamine levels consistently linked to myopia development [4, 5, 8, 9, 11, 18]. While each model offers specific insights, rabbits present a practical and relevant compromise: their eye size, anatomical structure, and pharmacokinetic properties offer more translational relevance than smaller animal models, while still being more ethically and economically feasible than non-human primates [19].
In this study, we observed no clinical signs of ocular irritation, such as conjunctival hyperemia or corneal erosion. Tear production, assessed via Schirmer tear testing, remained within the reported physiological range for rabbits (range: 0–15 mm/min) [20]. No statistically significant differences in Schirmer’s tear testing were observed before and after treatment. This finding is important given that Schirmer’s tear testing values in rabbits are known to be more sensitive to increases caused by ocular irritation than to decreases due to tear deficiency [20]. The absence of a change suggests that the levodopa–carbidopa formulation did not provoke a reflex tearing response or interfere with tear secretion mechanisms.
From a translational perspective, the use of eye drops offers substantial practical advantages over more invasive options such as intravitreal injections. This is especially relevant in the context of pediatric myopia management, where compliance and safety are critical. While intravitreal injections provide direct access to the retina, their invasiveness and associated risks limit clinical applicability. Previous studies, such as those by Thomson et al. emphasized the possible benefits of levodopa in chick models [8, 9]. Our study extends this line of investigation by demonstrating the effectiveness and safety of this therapeutic strategy in mammals.
The addition of carbidopa to levodopa significantly enhanced the formulation’s therapeutic effect. This is consistent with the pharmacological mechanism whereby carbidopa inhibits aromatic L-amino acid decarboxylase (AAAD), preventing premature conversion of levodopa and thereby enhancing local availability. This mechanism, well established in systemic applications, appears to extend to ocular tissues. Although the exact distribution of AAAD within the eye remains poorly defined, its expression in blood vessels and systemic circulation suggests that it may be present in ocular vasculature, including the choroid and ciliary body [21, 22]. Our findings lend indirect support to this hypothesis, though further investigation is warranted.
The refractive improvement observed in treated eyes went beyond stabilization, showing a shift toward emmetropia or mild hyperopia. This reduction in myopic shift is not typically seen with other monotherapies, such as dopamine, atropine, or levodopa alone in avian models [8, 9, 11, 23]. The enhanced efficacy observed with the levodopa-carbidopa combination may therefore be attributed to improved retinal dopamine availability. In addition to reducing the required dose of levodopa, this approach may also minimize the potential for systemic side effects such as nausea, dizziness, and fatigue, which have been reported in levodopa-based treatments for amblyopia [24].
At the molecular level, our data revealed upregulation of TH and D2R expression in the retina following treatment. This suggests a functional restoration of dopaminergic signaling, which is typically downregulated in the context of FDM. TH, as the rate-limiting enzyme in dopamine synthesis, is often upregulated in response to increased substrate availability (levodopa) and enhanced dopaminergic activity, especially when peripheral metabolism is reduced by carbidopa [17]. Dopamine receptor signaling, particularly via the D2 pathway, has been implicated in modulating axial elongation. We observed no significant change in D1R expression, supporting the notion that D2R-mediated signaling plays a more dominant role in the retina’s response to dopaminergic stimulation. While studies in chicks have shown D2R dominance, mouse models have sometimes indicated D1R involvement [25–27]. These discrepancies likely reflect interspecies differences in retinal architecture, such as photoreceptor distribution and receptor localization, reinforcing the need for comparative studies across species [24].
Emerging research underscores the pivotal role of dopamine receptor balance, particularly between D1-like and D2-like receptors, in regulating refractive development. D1R and D2R signaling often exhibit complementary yet opposing roles, where D1R activation generally promotes excitatory pathways and D2R activation modulates inhibitory feedback. A comprehensive review demonstrates that overactivation of D1-like receptors tends to promote hyperopia, whereas D2-like receptor overactivation is linked to myopia progression [4]. This nuanced interplay suggests that even a subtle increase in D1R expression (as indicated by a non-significant trend, p = 0.081) might signal a shift toward a molecular environment counteracting myopic progression. The non-significant but positive direction of change in D1R expression in our study may indicate an early molecular response that precedes robust morphological effects, particularly given the short treatment duration (5 d).
Currently, atropine eye drops, which works primarily through non-selective muscarinic receptor antagonism, which influences scleral remodeling and retinal signaling pathways, is the current standard for myopia control. Clinical trials have effectively shown slowed progression of myopia, but often causing side effects like photophobia, blurred near vision, and rebound myopia after stopping treatment [28]. Levodopa-carbidopa targets retinal dopamine pathways, offering a potentially more direct mechanism with fewer side effects and possible neuroprotective benefits. However, LC remains experimental, with limited clinical data for unclear issues regarding optimal dosing, formulation, and rebound effects. While promising as an alternative or adjunct, LC requires further clinical validation before routine use.
Despite these promising findings, several limitations must be acknowledged. First, the study’s duration was relatively short, thus limiting the ability to assess the long-term therapeutic effect or detect potential rebound after cessation. Second, pharmacokinetic profiling of intraocular levodopa and dopamine levels was not performed, which would help establish a dose–response relationship. Third, while the formulation was well tolerated over the study period, chronic safety under prolonged dosing remains and optimal topical delivery for ocular use to be established [29]. Future work should include imaging like optical coherence tomography (OCT) and functional testing such as electroretinography (ERG) and extended follow-up periods.
This stability of the LC formulation was assessed visually over a 7 d period (5 d of therapy), with no changes in color, odor, or turbidity and also digital pH meter was the sole parameter used for stability evaluation. While HPLC measurement is preferable, such measurement is very expensive in our country, thus, this is a certain limitation on our study. A significant change of suppressed FDM progression was noted following 5 d of LC eye drop administration. Further studies are needed to confirm the bioavailability of levodopa-carbidopa or dopamine levels in ocular tissues.
CONCLUSION
This study confirms that levodopa and carbidopa can be effectively used as a topical ophthalmic formulation to control myopia. The formulation was stable both physically and chemically, was well-tolerated in the rabbit model, and did not result in eye irritation or systemic toxicity. In a model of form-deprivation myopia, treatment with topical levodopa–carbidopa significantly inhibited axial elongation and reversed refractive error. Molecular analysis further demonstrated that tyrosine hydroxylase and dopamine D2R were both upregulated, supporting the dopaminergic signaling in the myopia. These findings support the potential of topical levodopa–carbidopa eye drops as a promising candidate for further preclinical and translational investigation for myopia management.
ABBREVIATIONS
FDM: Form-Deprivation Myopia, LC: levodopa-carbidopa, TH: tyrosine hydroxylase, D1R: dopamine receptor D1, D2R: dopamine receptor D2
ACKNOWLEDGEMENT
The authors thank the Doctoral Program at the Faculty of Medicine, Universitas Airlangga; the Institute of Tropical Diseases, Faculty of Pharmacy, Universitas Airlangga; and Dr. Soetomo General Academic Hospital for their support in this research.
AUTHORS CONTRIBUTIONS
Each author affirms that they are in charge of the article's writing and content, and the final manuscript was approved by all authors. Contributions from the authors: The original manuscript was written by CI, RP, and II, who also created the protocol and analyzed the experimental data. After conducting experiments, CI examined and assessed the results.
CONFLICTS OF INTERESTS
All authors declare no conflicts of interest.
REFERENCES
Morgan IG, French AN, Ashby RS, Guo X, Ding X, He M. The epidemics of myopia: aetiology and prevention. Prog Retin Eye Res. 2018;62:134-49. doi: 10.1016/j.preteyeres.2017.09.004, PMID 28951126.
Pan CW, Ramamurthy D, Saw SM. Worldwide prevalence and risk factors for myopia. Ophthalmic Physiol Opt. 2012;32(1):3-16. doi: 10.1111/j.1475-1313.2011.00884.x, PMID 22150586.
Feldkaemper M, Schaeffel F. An updated view on the role of dopamine in myopia. Exp Eye Res. 2013 Sep;114:106-19. doi: 10.1016/j.exer.2013.02.007, PMID 23434455.
Zhou X, Pardue MT, Iuvone PM, Qu J. Dopamine signaling and myopia development: what are the key challenges. Prog Retin Eye Res. 2017;61:60-71. doi: 10.1016/j.preteyeres.2017.06.003, PMID 28602573.
Dong F, Zhi Z, Pan M, Xie R, Qin X, Lu R. Inhibition of experimental myopia by a dopamine agonist: different effectiveness between form deprivation and hyperopic defocus in guinea pigs. Mol Vis. 2011;17:2824-34. PMID 22128230.
Chen S, Zhi Z, Ruan Q, Liu Q, Li F, Wan F. Bright light suppresses form-deprivation myopia development with activation of dopamine D1 receptor signaling in the ON pathway in retina. Invest Ophthalmol Vis Sci. 2017;58(4):2306-16. doi: 10.1167/iovs.16-20402, PMID 28431434.
Shu Z, Chen K, Wang Q, Wu H, Zhu Y, Tian R. The role of retinal dopamine D1 receptors in ocular growth and myopia development in mice. J Neurosci. 2023;43(48):8231-42. doi: 10.1523/JNEUROSCI.1196-23.2023, PMID 37751999.
Thomson K, Karouta C, Morgan I, Kelly T, Ashby R. Effectiveness and safety of topical levodopa in a chick model of myopia. Sci Rep. 2019;9(1):18345. doi: 10.1038/s41598-019-54789-5, PMID 31797988.
Thomson K, Morgan I, Kelly T, Karouta C, Ashby R. Coadministration with carbidopa enhances the antimyopic effects of levodopa in chickens. Invest Ophthalmol Vis Sci. 2021;62(4):25. doi: 10.1167/iovs.62.4.25, PMID 33877264.
Haddad F, Sawalha M, Khawaja Y, Najjar A, Karaman R. Dopamine and levodopa prodrugs for the treatment of Parkinson’s disease. Molecules. 2017;23(1):40. doi: 10.3390/molecules23010040, PMID 29295587.
Kim DH, Hwang JM, Yang HK. Topical dopamine application on form-deprivation myopia in rabbits. Life (Basel). 2025;15(3):461. doi: 10.3390/life15030461, PMID 40141806.
Barathi VA, Boopathi VG, Yap EP, Beuerman RW. Two models of experimental myopia in the mouse. Vision Res. 2008;48(7):904-16. doi: 10.1016/j.visres.2008.01.004, PMID 18289630.
Schuerer N, Stein E, Inic Kanada A, Pucher M, Hohenadl C, Bintner N. Implications for ophthalmic formulations: ocular buffers show varied cytotoxic impact on human corneal limbal and human conjunctival epithelial cells. Cornea. 2017;36(6):712-8. doi: 10.1097/ICO.0000000000001199, PMID 28399036.
Remenar J, Almarsson O, Meehan AJ, Zhong Z. Pharmaceutical compositions and method of using levodopa and carbidopa. US Patent US8815950B2. United States: United States Patent and Trademark Office; 2014.
Sanchez Rivera AE, Corona Avendano S, Alarcon Angeles G, Rojas Hernandez A, Ramirez Silva MT, Romero Romo MA. Spectrophotometric study on the stability of dopamine and the determination of its acidity constants. Spectrochim Acta A Mol Biomol Spectrosc. 2003;59(13):3193-203. doi: 10.1016/S1386-1425(03)00138-0, PMID 14583295.
Aminoff MJ. Pharmacologic management of Parkinsonism and other movement disorders. In: Vanderah TW, editor. Katzung’s basic and Clinical Pharmacology. 16th ed. McGraw-Hill Medical; 2023.
Daubner SC, Le T, Wang S. Tyrosine hydroxylase and regulation of dopamine synthesis. Arch Biochem Biophys. 2011;508(1):1-12. doi: 10.1016/j.abb.2010.12.017, PMID 21176768.
Yan T, Xiong W, Huang F, Zheng F, Ying H, Chen JF. Daily injection but not continuous infusion of apomorphine inhibits form deprivation myopia in mice. Invest Ophthalmol Vis Sci. 2015;56(4):2475-85. doi: 10.1167/iovs.13-12361, PMID 25744977.
Baumans V. Environmental enrichment for laboratory rodents and rabbits: requirements of rodents rabbits and research. ILAR J. 2005;46(2):162-70. doi: 10.1093/ilar.46.2.162, PMID 15775025.
Whittaker AL, Williams DL. Evaluation of lacrimation characteristics in clinically normal New Zealand white rabbits by using the schirmer tear test i. J Am Assoc Lab Anim Sci. 2015;54(6):783-7. PMID 26632789.
Sorriento D, Santulli G, Del Giudice C, Anastasio A, Trimarco B, Iaccarino G. Endothelial cells are able to synthesize and release catecholamines both in vitro and in vivo. Hypertension. 2012;60(1):129-36. doi: 10.1161/hypertensionaha.111.189605, PMID 22665130.
De Souza GA, Godoy LM, Mann M. Identification of 491 proteins in the tear fluid proteome reveals a large number of proteases and protease inhibitors. Genome Biol. 2006;7(8):R72. doi: 10.1186/gb-2006-7-8-r72, PMID 16901338.
Thomson K, Morgan I, Karouta C, Ashby R. Levodopa inhibits the development of lens-induced myopia in chicks. Sci Rep. 2020;10(1):13242. doi: 10.1038/s41598-020-70271-z, PMID 32764736.
Repka MX, Kraker RT, Beck RW, Atkinson CS, Bacal DA, Bremer DL. Pilot study of levodopa dose as treatment for residual amblyopia in children aged 8 y to younger than 18 y. Arch Ophthalmol. 2010;128(9):1215-7. doi: 10.1001/archophthalmol.2010.178, PMID 20837811.
Ward AH, Siegwart JT, Frost MR, Norton TT. Intravitreally administered dopamine D2-like (and D4), but not D1-like, receptor agonists reduce form-deprivation myopia in tree shrews. Vis Neurosci. 2017;34:E003. doi: 10.1017/S0952523816000195, PMID 28304244.
Ashby R, McCarthy CS, Maleszka R, Megaw P, Morgan IG. A muscarinic cholinergic antagonist and a dopamine agonist rapidly increase ZENK mRNA expression in the form-deprived chicken retina. Exp Eye Res. 2007;85(1):15-22. doi: 10.1016/j.exer.2007.02.019, PMID 17498696.
Ashby R, Kozulin P, Megaw PL, Morgan IG. Alterations in ZENK and glucagon RNA transcript expression during increased ocular growth in chickens. Mol Vis. 2010;16:639-49. PMID 20405027.
Zhao C, Cai C, Ding Q, Dai H. Efficacy and safety of atropine to control myopia progression: a systematic review and meta-analysis. BMC Ophthalmol. 2020;20(1):478. doi: 10.1186/s12886-020-01746-w, PMID 33287746.
Tinu TS, Thomas L, Kumar BA. Polymers used in ophthalmic in situ gelling system. Int J Pharm Sci Rev Res. 2013;20(1):176-83.