
Int J App Pharm, Vol 18, Issue 2, 2026, 418-423Original Article
OPTIMISATION AND CHARACTERIZATION OF TERBINAFINE-LOADED TOPICAL SOLID LIPID NANOPARTICLES
DIVYA KARUPPAIAH1, RAJKUMAR KARUNAKARAN2, AYYAPPAN THIYAGARAJAN1, JAMAL MOIDEEN MUTHU MOHAMED3, KRISHNA PRABHA NADUCHAMY4*
1Department of Pharmaceutics, Karpaga Vinayaga Institute for Pharmaceutical Sciences, Chengalpattu-603308, India. 2Department of Pharmaceutics, Nargund College of Pharmacy, Bangalore, India. 3Department of Pharmacology, Faculty of Medicine, Manipal University College Malaysia, Jalan Batu Hampar, Bukit Baru-75150 Melaka, Malaysia. 4Department of Pharmacology, SRM College of Pharmacy, SRMIST, Kattankulathur-603203, Tamil Nadu, India
*Corresponding author: Krishna Prabha Naduchamy; *Email: prabhasnc23@gmail.com
Received: 18 Jul 2025, Revised and Accepted: 16 Jan 2026
ABSTRACT
Objective: The present study aimed to prepare a nanoformulation using GMS and/or Precirol ATO5and Tween 80 (1% and 2%) to develop and optimize solid lipid nanoparticles with the potential to enhance oral bioavailability.
Methods: The factorial design was selected to study the impact of two solid lipid formulation parameters on the particle size (PS), zeta potential (PS), entrapment efficiency (EE), and in vitro drug release. The emulsification technique was used to develop solid lipid nanoparticles, and the prepared nanoparticles were evaluated using design of expert (DoE) software.
Results: The drug estimation was found linear between 5 and 40µg/ml, and the melting point was found to be 195 °C. Enhancing the lipid content (GMS or precirol ATO5) resulted in an increase in PS. However, increasing the concentration of the surfactant (Tween 80) reduced the PS. Among the two lipids used as a qualitative factor, the desirability function was more with precirol ATO5 formulations. The numerical optimization suggested an optimized formulation containing 2% Tween 80 and 5.5% precirol ATO5. Among the experimental batches, formulation F6 (2% Tween 80 and 3% precirol ATO5) exhibited a PS of 98.3±5.12 nm, an EE of 81.5±6.27%, and a drug release of 77.5±8.09% over 10 h.
Conclusion: The TB-loaded SLN were successfully formulated by the emulsification method. Among the two lipids used as a qualitative factor, the desirability function was more with precirol ATO5 formulations. This may potentially enhance oral bioavailability, as suggested by nanoscale PS and improved in vitro drug release.
Keywords: Solid lipid nanoparticles, Terbinafine, Glyceryl monostearate, Precirol ATO5, DoE, FTIR
© 2026 The Authors.Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i2.56153 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Nanoparticles are a type of innovative drug delivery system that can release medication at an optimal pace at the intended site of action. Nanoformulations provide the versatility to employ a wide range of polymers, encompassing synthetic, natural, biodegradable, and non-biodegradable types [1]. The nanoparticles have a size range of 1 to 1000 nm, while nanoparticles within the range of 50-500 nm are deemed appropriate, depending on the method of administration [2]. The particles in the nanosize range contribute to improved stability of the compounds, controlled drug release and better bioavailability are effective at overcoming permeability barriers.
Nanomaterials enhance the effectiveness of both new and existing medications, such as natural products, and enable targeted detection using disease-specific biomarkers. Common problems associated with drugs exhibiting low solubility encompass insufficient bioavailability and/or erratic absorption [3]. Solid lipid nanoparticles (SLNs) are a cutting-edge method for delivering medications directly to the brain and overcoming the challenges posed by the blood-brain barrier [4]. Small size, vast surface area, and high drug loading are some of the special qualities that SLNs offer, which serve to enhance the efficacy of medications and other materials [5, 6].
Terbinafine hydrochloride (TB), a synthetic allylamine compound, demonstrates strong and wide-ranging fungicidal effects by blocking the action of squalene epoxidase. However, its oral bioavailability is limited due to extensive first-pass metabolism and high plasma protein binding. Nanocarrier-based systems may offer a strategy to improve oral absorption [7]. Glycerol monostearate, a monoglyceride, is a white, odourless powder and hygroscopic in nature beside, it is utilized as an emulsifier. Precirol oil dissolves in chloroform and dichloro methane butnotin ethanol, mineral oil, and water [8, 9].
To situate our own work in relation to contemporary advances in topical SLN science, we highlight here some of the contemporary scientific literatures that indicate the potential of SLNs in improving dermal delivery of drugs in general [10]. Topical SLNs have also indicated their potential in improving retention and availability of drugs into and from skin in a maximized form and their occlusive and moisturizing action in several in vivo and in vitro human and clinical studies, including significant advancements in in vivo models where vitamin A and tretinoid SLNs proved to be effective and showed favourable skin tolerability and improved stability, and corticosteroid SLN gels showed significantly increased dermal deposition and permeability compared to conventional preparations [11].
In the present study, emulsification technique was employed to create SLNs of TB, with glyceryl monostearate or Precirol ATO5 serving as the lipid base. The prepared SLNs were analysed for their particle size (PS), zeta potential (ZP), polydispersibility index (PDI), entrapment efficiency (EE), and in vitro drug release.
MATERIALS AND METHODS
Terbinafine hydrochloride (TB) was provided by Dr. Reddy’s Laboratories, Hyderabad. Precirol ATO5 was obtained from Active Pvt. Ltd. Mumbai, and glycerol monostearate from Central Drug House Pvt Ltd, New Delhi. Ethanol was obtained from Karnataka Fine Chem. Pvt Ltd, Bangalore, and Tween 80 from Thomas Baker (Chemical) Pvt Ltd, Ambernath. The design of expert (DoE) software, version 11.1.2.0, was employed for statistical study. All the other chemicals utilized were of either reagent or pharmaceutical grade. The FTIR spectra were recorded using an FTIR-100 spectrophotometer, Shimadzu, Japan. The absorption maxima were detected using a UV spectrophotometer (UV-1800), Shimadzu, Japan. PS and size distribution were analysed by using Nano Partica SZ100, Horiba Scientific.
Evaluation of pre-formulation parameters
Pre-formulation evaluation is the preliminary phase in the development of dosage forms for a pharmaceutical ingredient. Pharmaceutical characterization involves examining the physical and chemical properties of a medicinal component both on its own and when mixed with other substances called excipients. The primary objective of the evaluation is to yield valuable information for the development of stable dosage forms appropriate for large-scale production.
Measurement of melting temperature
The melting temperature of the drug sample, TB, was measured using the capillary tube test. A minute quantity of pulverized substance was inserted into the slender capillary tube and sealed on one end through the process of liquefaction. The capillary was inserted into the melting point device [12]. A thermometer was inserted into the equipment that had previously been submerged in the liquid paraffin. Upon reaching a designated temperature, the drug underwent a process of melting, indicating its melting point.
Determination of λ max.
To determine the wavelength of maximum absorption (λ max), TB 1 mg/ml was prepared in ethanol, then 0.15 ml was made up to 10 ml using phosphate buffer pH7.4 and scanned in the UV wavelength range of 200-400 nm. The absorption maxima indicated in the graph was designated as λmax for the pure TB solution.
Preparation of TB calibration curve
The primary stocksolution1 was prepared by dissolving 10 mg of TB in 0.5 ml of ethanol, followed by volume adjustment with a pH 7.4 phosphate buffer solution containing 1000 μg/ml or 1 mg/ml. The secondary stock solution was prepared by 1 ml of above stock was diluted to 10 ml using pH 7.4 phosphate buffer (100 μg/ml). The volume of 0.5, 1.0, 1.5, 2.0, 2.5, 3.0, 3.5, and 4.0 ml were diluted to 10 ml using phosphate buffer to getaliquots of 5, 10, 15, 20, 25, 30, 35, and 40 µg/ml, respectively. The buffer solution was served as the blank, and three sets of standard solutions were prepared to repeat the standard graph. The absorbance was measured at 283 nm using a UV-Visible Spectrophotometer [13].
Drug-excipients compatibility study
FT-IR analysis was performed to determine the compatibility or interaction with the chemical makeup after adding the excipients. A 2 mg of samples, mixed with the excipients GMS, Precirol ATO5, and Tween 80, was combined with IR-grade KBr in a ratio of 1:10. The mixture was then subjected to a pressure of 10 tons using a hydraulic press, forming a clear pellet [14]. The pellet was analysed using an IR spectrophotometer from 4000 to 400 cm-1.
Formulation of solid lipid nanoparticles
The lipid phase, consisting of Precirol ATO5 or glyceryl monostearate was melted the above its melting point, approximately 60 °C. TB (1% w/w) was dissolved in 0.5 ml of ethanol and incorporated into the molten lipid phase to obtain a uniform lipid–drug mixture. The aqueous phase was prepared by dissolving Tween 80 (1% or 2% w/w) in 50 ml of deionized water and heated to the same temperature (≈60 °C). The hot lipid phase was then slowly added to the aqueous phase under continuous magnetic stirring (REMI2) at 2000 rpm. Stirring was continued for 4 h to allow solvent evaporation and formation of a hot nanoemulsion [15]. The resulting dispersion was allowed to cool naturally to room temperature under continuous stirring, leading to solidification of the lipid phase and formation of TB-loaded solid lipid nanoparticles.
Design of experiments (DoE)
DoE was utilized to optimize the independent variables to achieve the desired value of dependent variables. 2² full factorial design with one qualitative (categorical) factor was employed using Design-Expert® software. Two quantitative factors, between 80 concentration and lipid concentration-were studied along with one qualitative factor, namely lipid type (glyceryl monostearate or Precirol ATO5), as shown in table 1. Where the amounts of two excipients, Tween 80 and two different lipids such as GMS or Precirol ATO5, were selected as the independent variables. The levels for Tween 80 were considered as-1 and+1, corresponding to 1% and 2%, respectively [16]. For GMS or Precirol ATO5, the levels were considered as-1 and+1, corresponding to 3% and 6%, respectively. All other formulations and processing variables were constant throughout the trial.
Table 1: 2² full factorial design with one qualitative (categorical) factor of TB
| Factor | Name | Units | Minimum | Maximum |
| A | Tween 80 | Quantitative % | 1 | 2 |
| B | GMS and precirol ATO5 | Quantitative % | 3 | 6 |
Numerical method
Two quantitative independent variables (Tween 80 concentration and lipid concentration) and one qualitative independent variable (type of lipid: GMS or Precirol ATO5) were considered in the statistical design. They are (i) the amount of Tween 80 as factor A, (ii) the amount of lipid as factor B. These three factors are varied in their concentration. The amount of tween 80 was used in range of 1–2% and GMS or Precirol ATO5 was used within the range of 3-6%. The amount of drug used was a constant 1%, as shown in table 2.
Table 2: Composition of various TB-loaded SLNs
| S. No. | Batch code | Amount of drug (%) | Amount of GMS (%) | Amount of precirol (%) | Tween 80 (%) | Distilled water (ml) |
| 1 | F1 | 1 | 3 | 1 | 50 | |
| 2 | F2 | 3 | 2 | |||
| 3 | F3 | 6 | 1 | |||
| 4 | F4 | 6 | 2 | |||
| 5 | F5 | 3 | 1 | |||
| 6 | F6 | 3 | 2 | |||
| 7 | F7 | 6 | 1 | |||
| 8 | F8 | 6 | 2 |
Evaluation of nanoparticles
All measurements were performed in triplicate using the same sample dispersion, and the reported values represent instrumental repeatability rather than batch-to-batch variation.
PS and PDI analysis
The average PS and size distribution are critical characteristics that affect the physiochemical properties and biological behavior of nanoparticles. The PS must range from 10 to 100 nm. The polydispersity index is a metric that characterizes the PS distribution of synthesized nanoparticles. The dimensionless number, derived from the autocorrelation function, varies from 0.01 for monodisperse particles to 0.5-0.7 for broad PS distributions. The prepared nanoparticle sample was diluted with double-distilled water [17]. The PS and distribution were assessed using the analyser (Horiba Scientific Nano Partica SZ100). The PS of each formulation was measured thrice, and the mean as well as standard deviation were calculated (n=3).
Determination of ZP
The surface charge was assessed by evaluating the electrophoretic mobility of the nanoparticles, which is a key factor in determining the colloidal stability. 1 ml of the prepared SLNs sample was diluted with 10 ml of double-distilled water [18]. The samples were placed into the cuvette containing electrodes and the ZP was analysed by transmitting laser-scattered light through a cuvette (Nano Partica – SZ100, Horiba Scientific).
%EE
The EE was determined by separating the unentrapped drug from the SLN dispersion using centrifugation. Briefly, 2 ml of TB-loaded SLNs was mixed with 0.5 ml of ethanol to facilitate solubilization of the free drug present in the dispersion medium without disrupting the solid lipid matrix [19]. The mixture was centrifuged at 6000 rpm for 30 min at 25±1 °C. The supernatant was suitably diluted with phosphate buffer pH 7.4 and analysed spectrophotometrically at 283 nm. The % EE was calculated using the following equation.
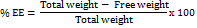
Drug content
1 ml of the dispersed nanoparticle sample was sonicated in 5 ml of acetone, and the volume was adjusted to 100 ml with phosphate buffer at pH 7.4. 1.5 ml of this solution was diluted with same buffer to 10 ml to get a concentration within the standard graph range, and the drug content was quantified by UV-Spectrophotometry at 283 nm.
In vitro drug release studies
The drug release of pure TB and TB-loaded SLNs were examined by using dialysis bag technique in a phosphate buffer with a pH of 7.4. A dialysis membrane possessing a specific molecular weight cutoff of 12000 to 14000 Da and a pore size of 2.4 nm was used to encapsulate the TB nanoparticle, corresponding to 10 mg of drug. The dialysis membrane was soaked in distilled water for 24 h [20]. The TB sample was introduced into the dialysis bag, and both ends of the bag were securely sealed. The bag was then immersed in 100 ml of dissolving medium, using a glass rod to prevent it from floating. The medium was constantly stirred at 100 rpm, using a magnetic stirrer, at 37±0.5 °C. Consistent concentration was maintained by replacing them with an equal amount of fresh dissolution medium with every 2 h. The samples were examined using spectrophotometry at a wavelength of 283 nm. Although improved in vitro drug release and reduced PS are indicative of enhanced dissolution and absorption potential, no in vivo pharmacokinetic evaluation was performed in the present study to directly confirm improved oral bioavailability.
Numerical optimization
Numerical optimisation explores the design space using models created during analysis. It can optimize any combination of several objectives. The various objectives encompass as maximization, minimization, targeting, maintain within a certain range, none (for responses exclusively), and establishing an exact value (for factors exclusively). Each parameter used in the optimization must have both a minimum and maximum level defined, as shown in table 3.
RESULTS AND DISCUSSION
Preliminary investigations were conducted to assess the purity of the drug prior to the formulation. The purity was assessed by determination of melting point and λmax. The melting temperature of TB was found to be 195 °C, thus indicating the purity of the obtained drug sample [21].
Calibration curve of TB
Determination of λ max
The standard solution of TB was analysed using a UV spectrometer within the wavelength range of 200-400 nm. The absorption maxima were determined to be at 283 nm. Sample were prepared from the stock solution for the calibration curve, and the absorbance was measured at 283 nm, as shown in table 4.
Table 3: Constraints for the chemicals to prepare a new nanoparticle formulation using Tween 80 as liquid phase and precirol ATO5 as lipid phase
| Name | Goal | Lower limit | Upper limit |
| A: Tween | In range | 1 | 2 |
| B: Concentration of lipid | In range | 3 | 6 |
| Type of lipid | In range | GMS | Precirol ATO5 |
| PS (nm) | minimize | 98.3 | 209.6 |
| ZP (mV) | In range | -30 | 30 |
| EE (%) | maximize | 70.5 | 83.1 |
| 10 h drug release | maximize | 66.2 | 83 |
Table 4: Absorbance of standard solution of TB at pH 7.4 phosphate buffer
| Concentration (µg/ml) | Absorbance (Mean±SD) |
| 0 | 0 |
| 5 | 0.120±0.0202 |
| 10 | 0.239±0.0145 |
| 15 | 0.351±0.0150 |
| 20 | 0.458±0.0467 |
| 25 | 0.553±0.0289 |
| 30 | 0.657±0.0139 |
| 35 | 0.749±0.0276 |
| 40 | 0.876±0.0198 |
Value are expressed as mean±standard deviation (SD, n = 3).
The calibration curve plotted against the concentration range of 5 to 40 µg/ml showed excellent linearity with a high correlation coefficient of R² = 0.9993, while its positive proportionality relationship of concentration to absorbance confirmed its reliability and suitability for analysis of the drug concentration by this method. This relationship of proportionality can be represented by fig. 1.
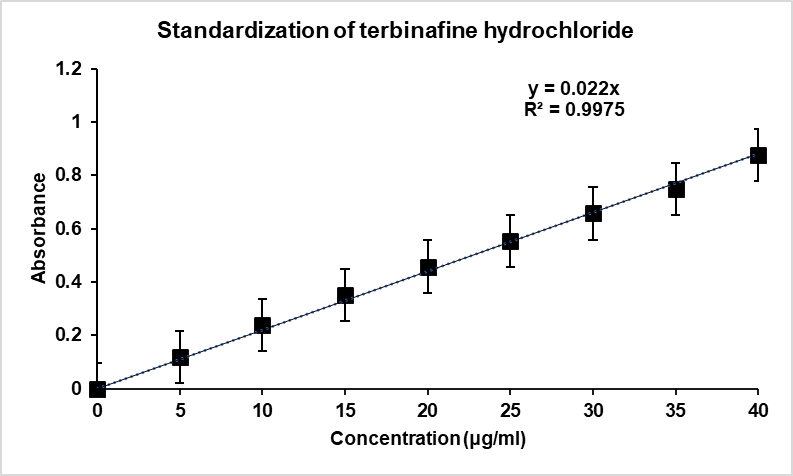
Fig. 1: Calibration curve for TB at pH 7.4 Phosphate buffer
Drug-excipient compatibility study
The FTIR spectrum of pure TB showed characteristic absorption bands corresponding to its functional groups (fig. 2a). Broad bands observed in the region of 3200–3000 cm⁻¹ were attributed to N–H stretching vibrations, while peaks in the range of 2950–2850 cm⁻¹ corresponded to aliphatic C–H stretching shown in table 5. The absorption band around 1600–1500 cm⁻¹ was associated with aromatic C=C stretching, and peaks in the region of 1300–1000 cm⁻¹ were assigned to C–N stretching vibrations [22].
Table 5: Major characteristic FTIR peaks of TB
| S. No. | Functional group | Wavenumber (cm-1) | Observed peak value |
| 1. | N-H Stretching | 3200-3000 cm-1 | 3041.84 cm-1 |
| 2. | Aliphatic C-H stretching | 2950-2850 cm-1 | 2565.41 cm-1 |
| 3. | Aromatic C=C stretching | 1600-1500 cm-1 | 1556.61 cm-1 |
| 4. | C-N stretching | 1300-1000 cm-1 | 1132.25 cm-1 |
The FTIR spectra of TB-loaded SLNs containing GMS, Precirol ATO5, and Tween 80 exhibited all the characteristic peaks of the drug without significant shifts, disappearance, or formation of new peaks shown in fig. 2b. This indicates the absence of chemical interaction between TB and the excipients, confirming compatibility within the formulation [23].
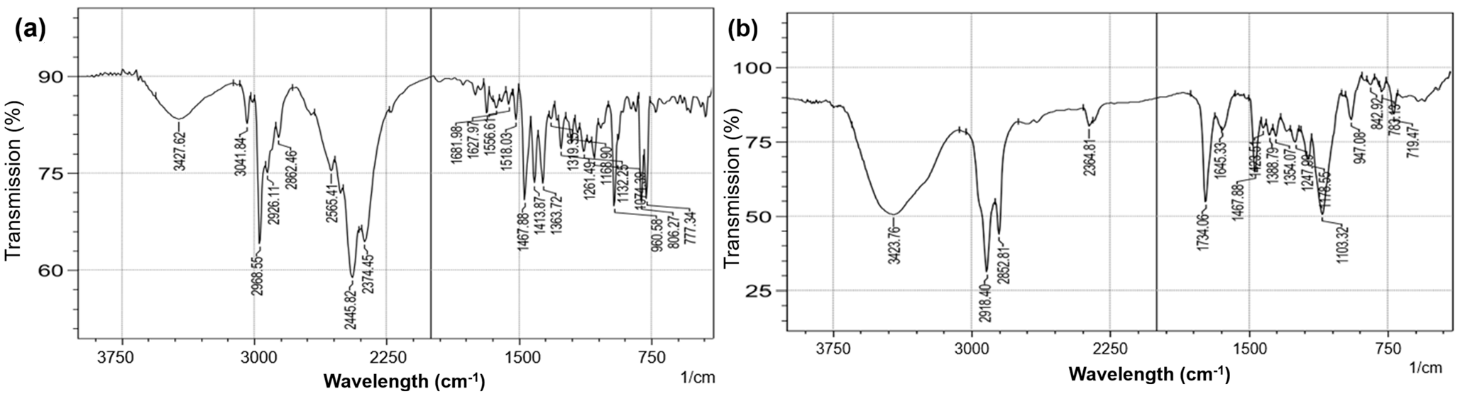
Fig. 2: FTIR spectrum of (a) pure TB and (b) TB-loaded SLNs showing retention of characteristic drug peaks
Evaluation of solid lipid nanoparticles
TB-loaded SLNs were successfully prepared, and different solid lipids were developed to obtain uniform nanosized particle. Utilizing a 2² full factorial design incorporating a qualitative lipid-type factor, eight batches of TB-loaded SLNs were produced by altering three determinants: drug to lipid ratio, surfactant concentration (% w/v), and volume of the organic phase, with results documented in table 6. The relatively low standard deviation values reflect the repeatability of instrumental measurements obtained from the same formulation batch and do not represent inter-batch variability. It was noted that formulation F6 refers to an experimental batch containing 3% Precirol ATO5 and 2% Tween 80 as per the factorial design [16].
The 5.5% Precirol ATO5 composition corresponds to the numerically optimized formulation predicted by Design-Expert software, which was not part of the initial factorial batches but was generated through numerical optimization. The PS and % EE of 8 batches showed a variation from 98.3±5.12 to 209.6±8.46 nm and 70.5±5.02 to 83.1±6.18%, respectively. The in vitro release data demonstrated a gradual and sustained release of TB, with cumulative drug release ranging from 66.2±4.98 to 83.0±8.43% over a period of 10 h (fig. 3). However, ethanol was used in a minimal volume solely to extract unentrapped TB from the aqueous phase. While this method is commonly employed for preliminary EE estimation, more refined techniques such as ultrafiltration or dialysis-based separation may provide greater accuracy and should be considered in future studies [24].
Table 6: The results of 2² full factorial design with one qualitative (categorical) factor of TB. All the parameters were measured thrice (n=3), and the mean as well as standard deviation (mean±SD) were representing instrumental repeatability
| Run | PS (nm) | PDI | ZP (mV) | EE (%) | Drug content (%) | 10-h drug release (%) |
| 1 | 137.2±6.21 | 0.43±0.02 | -29.3±1.01 | 70.5±5.02 | 94.2±7.13 | 66.2±4.98 |
| 2 | 107.5±6.08 | 0.28±0.03 | -22.2±1.18 | 80.2±6.09 | 96.8±7.32 | 74.2±4.13 |
| 3 | 209.6±8.46 | 0.21±0.02 | -35.4±1.21 | 75.3±5.10 | 94.1±8.43 | 71.4±5.02 |
| 4 | 143.4±7.07 | 0.31±0.03 | -25±1.32 | 82.5±7.01 | 98.8±8.18 | 82.3±6.18 |
| 5 | 125.5±6.30 | 0.48±0.08 | -15.2±0.81 | 71.5±5.17 | 95.1±8.30 | 68.3±7.62 |
| 6 | 98.3±5.12 | 0.51±0.05 | -21.2±1.05 | 81.5±6.27 | 97.1±7.92 | 77.5±8.09 |
| 7 | 206.9±8.92 | 0.38±0.06 | -12.6±0.81 | 76.1±5.14 | 94.8±8.68 | 70.5±6.38 |
| 8 | 148.5±7.36 | 0.27±0.07 | -20.2±1.67 | 83.1±6.18 | 99.2±9.28 | 83.0±8.43 |
PS = particle size (nm); PDI = polydispersity index; ZP = zeta potential (mV); EE = entrapment efficiency (%). Data are expressed as mean±standard deviation (SD), representing instrumental repeatability (n=3).
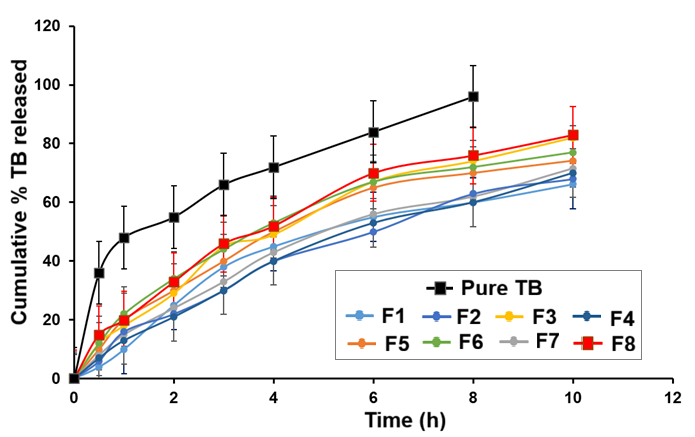
Fig. 3: Cumulative % of TB released from pure TB and TB-loaded SLNs (F1-F8)
Chen and co-researchers were reported that Terbinafine SLNs were developed as a method of delivering the drug topically, aiming to address the issues of lengthy treatment durations and frequent administration associated with the antifungal agent. The nanoparticles were prepared using a microemulsion technique with glyceryl behenate, glyceryl monostearate, and glyceryl palmitostearate serving as the solid lipid phases. The surfactant used was Tween 80 and the Cremophor series, while propylene glycol was used as the cosurfactant [25]. Topical administration of TB offers advantages such as precise delivery and targeting to the specific area of the skin that is afflicted, a low dosage required, and reduced risks of drug-related toxicities [26].
The ZP values of the TB-loaded SLNs ranged from-12.6±0.81 to-35.4±1. 21mV. Although absolute ZP values greater than ±30 mV are generally associated with electrostatically stabilized dispersions, the presence of the non-ionic surfactant Tween 80 contributes significantly to steric stabilization of the nanoparticles. The hydrophilic polyoxyethylene chains of Tween 80 form a stabilizing layer around the SLNs, reducing particle–particle interactions and aggregation even at relatively lower ZP values [27]. Therefore, the observed ZP values, in combination with steric effects, indicate acceptable colloidal stability of the formulated SLNs. Short-term observation of the SLN dispersions did not show any visible aggregation or phase separation during the study period, indicating acceptable initial physical stability of the formulations [28].
The FT-IR results of pure TB showed that the sample was free from impurities, and the characteristic peaks lie within the range of standard values. Analysis of the spectra of a TB mixture alongside the pure compound revealed that the excipient does not interact with the peaks of TB. The results of FTIR obtained from this study was like the results of Chauhan and his co-researcher [29]. The in vitro drug release profiles of formulations F1 to F8 demonstrated a sustained release pattern over a period of 10 h. However, detailed kinetic modelling was not performed in the present study, and therefore, the exact release mechanism could not be conclusively established [30].
LIMITATION
The present study focused primarily on formulation development and in vitro characterization of TB-loaded SLNs. Long-term physical and chemical stability studies under accelerated and real-time storage conditions were not performed, which represents a limitation of the work. Comprehensive stability evaluation will be undertaken in future studies to assess the changes in PS, ZP, PDI, EE (%), and drug release behaviour over time.
CONCLUSION
Terbinafine-loaded SLNs were successfully formulated and characterized, and they possessed good particle size, zeta potential, polydispersity index, entrapment efficiency, and sustained drug release, and there were no interactions between drug and excipient evidenced by FT-IR spectra. Factorial designs showed that increasing lipid concentration increased particle size and zeta potential, whereas Tween 80 concentration decreased particle size and surface charge. Precirol ATO5 showed a smaller particle size, better particle size distribution, higher entrapment efficiency, and drug release compared to GMS, and they followed zero-order and non-Fickian release kinetics. Numerical optimization showed that optimal formulation could be 5.5% Precirol ATO5 and 2% Tween 80, which is consistent with theoretical predictions. Although optimized SLNs possess good physicochemical properties and in vitro drug release profile, more studies are needed to assess their stability and efficacy in addition to their applications as therapeutic drug carriers in upcoming years.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
DIVYA KARUPPAIAH: Writing – Investigation, original draft, Validation, Software, Data curation. RAJKUMAR KARUNAKARAN: Formal analysis, writing – review and editing, Funding acquisition. AYYAPPAN THIYAGARAJAN: Formal analysis, Validation, Software, Data curation. KRISHNA PRABHA NADUCHAMY: Writing – review and editing, Software, Data curation. JAMAL MOIDEEN MUTHU MOHAMED: Conceptualization, Writing – review and editing, Visualization, Supervision, Project administration.
CONFLICT OF INTERESTS
The authors declare no conflict of interest
REFERENCES
Joseph TM, Kar Mahapatra D, Esmaeili A, Piszczyk L, Hasanin MS, Kattali M. Nanoparticles: taking a unique position in medicine. Nanomaterials (Basel). 2023;13(3):574. doi: 10.3390/nano13030574, PMID 36770535.
Lu Q, Yu H, Zhao T, Zhu G, Li X. Nanoparticles with transformable physicochemical properties for overcoming biological barriers. Nanoscale. 2023;15(32):13202-23. doi: 10.1039/D3NR01332D, PMID 37526946.
Lam PL, Wong WY, Bian Z, Chui CH, Gambari R. Recent advances in green nanoparticulate systems for drug delivery: efficient delivery and safety concern. Nanomedicine (Lond). 2017;12(4):357-85. doi: 10.2217/nnm-2016-0305, PMID 28078952.
Egwu CO, Aloke C, Onwe KT, Umoke CI, Nwafor J, Eyo RA. Nanomaterials in drug delivery: strengths and opportunities in medicine. Molecules. 2024;29(11):2584. doi: 10.3390/molecules29112584, PMID 38893460.
Yetisgin AA, Cetinel S, Zuvin M, Kosar A, Kutlu O. Therapeutic nanoparticles and their targeted delivery applications. Molecules. 2020;25(9):2193. doi: 10.3390/molecules25092193, PMID 32397080.
Dhiman N, Awasthi R, Sharma B, Kharkwal H, Kulkarni GT. Lipid nanoparticles as carriers for bioactive delivery. Front Chem. 2021;9:580118. doi: 10.3389/fchem.2021.580118, PMID 33981670.
Maxfield L, Preuss CV, Bermudez R. Terbinafine. In: Treasure Island, FL: StatPearls Publishing; 2025. Available from: https://www.ncbi.nlm.nih.gov/books/NBK545218.
Zhao M, Chen L, Liu F, Zhong F, Chen M, Jin H. The impact of glycerol monostearate’s similarity to fats and fatty acid composition of fats on fat crystallization, destabilization and texture properties of ice cream. J Sci Food Agric. 2023;103(14):6837-48. doi: 10.1002/jsfa.12768, PMID 37278491.
Chen J, Chen Z, Wang W, Wang L, Zheng J, Wu S. Effects of commonly used surfactants poloxamer 188 and tween 80, on the drug transport capacity of intestinal glucose transporters. AAPS PharmSciTech. 2024;25(6):163. doi: 10.1208/s12249-024-02881-z, PMID 38997614.
Ryan A, Patel P, Ratrey P, O Connor PM, O Sullivan J, Ross RP. The development of a solid lipid nanoparticle (SLN)-based lacticin 3147 hydrogel for the treatment of wound infections. Drug Deliv Transl Res. 2023;13(9):2407-23. doi: 10.1007/s13346-023-01332-9, PMID 36964439.
Safta DA, Bogdan C, Moldovan ML. SLNs and NLCs for skin applications: enhancing the bioavailability of natural bioactives. Pharmaceutics. 2024;16(10):1270. doi: 10.3390/pharmaceutics16101270, PMID 39458602.
OJ, Young J. True melting point determination. Methods Mol Biol. 2013;18:203-8. doi: 10.1333/s00897132500a.
Li CX, Elhassan GO, Abdoun SA, Khan RA, Goyal M, Bansal M. Ultra-fast synthesis of curcumin-loaded silver nanoparticles: improved physicochemical properties for drug delivery. Int J App Pharm. 2025;17(1):216-23. doi: 10.22159/ijap.2025v17i1.52647.
Mohamed JM, Alqahtani A, Menaa F, Kayarohanam S, Fatease AA, Alqahtani T. In vitro physical characterizations and docking studies on carvedilol nanocrystals. Crystals. 2022;12(7):988. doi: 10.3390/cryst12070988.
Moideen MM, Karuppaiyan K, Kandhasamy R, Seetharaman S. Skimmed milk powder and pectin decorated solid lipid nanoparticle containing soluble curcumin used for the treatment of colorectal cancer. J Food Process Engineering. 2020;43(3):e13246. doi: 10.1111/jfpe.13246.
Sivadasan D, Venkatesan K, Mohamed JM, Alqahtani S, Asiri YI, Faisal MM. Application of 32 factorial design for loratadine-loaded nanosponge in topical gel formulation: comprehensive in vitro and ex vivo evaluations. Sci Rep. 2024;14(1):6361. doi: 10.1038/s41598-024-55953-2, PMID 38493177.
Yadav K, Yadav D, Kumar S, Narra K, El-Sherbiny M, Al-Serwi RH. Natural biodegradable and polymeric nanoparticles for the delivery of noscapine for cancer treatment. Biomass Convers Biorefin. 2022;14(21):27609-21. doi: 10.1007/s13399-022-03334-y.
Alqahtani A, Raut B, Khan S, Mohamed JM, Fatease AA, Alqahtani T. The unique carboxymethyl fenugreek gum gel loaded itraconazole self-emulsifying nanovesicles for topical onychomycosis treatment. Polymers (Basel). 2022;14(2):325. doi: 10.3390/polym14020325, PMID 35054731.
Satheesh V, Mohamed JM, El-Sherbiny M, Othman G, Al-Serwi RH, Thilagar S. Sunlight-assisted green synthesis of silver nanoparticles using Zizania latifolia extract: toward antimicrobial applications. Biomass Conv Bioref. 2024;14(21):27633-43. doi: 10.1007/s13399-022-03363-7.
Mohamed JM, Alqahtani A, Ahmad F, Krishnaraju V, Kalpana K. Pectin co-functionalized dual layered solid lipid nanoparticle made by soluble curcumin for the targeted potential treatment of colorectal cancer. Carbohydr Polym. 2021;252:117180. doi: 10.1016/j.carbpol.2020.117180, PMID 33183627.
Kanakapura B, Penmatsa VK. Analytical methods for determination of terbinafine hydrochloride in pharmaceuticals and biological materials. J Pharm Anal. 2016;6(3):137-49. doi: 10.1016/j.jpha.2016.01.003, PMID 29403974.
Iizhar SA, Syed IA, Satar R, Ansari SA. In vitro assessment of pharmaceutical potential of ethosomes entrapped with terbinafine hydrochloride. J Adv Res. 2016;7(3):453-61. doi: 10.1016/j.jare.2016.03.003, PMID 27222750.
Ravishankar S. Textbook of Pharmaceutical Analysis. 5th ed. Tirunelveli: Rx Publications; 2019.
Cibotaru D, Celestin MN, Kane MP, Musteata FM. Comparison of liquid–liquid extraction, microextraction and ultrafiltration for measuring free concentrations of testosterone and phenytoin. Bioanalysis. 2022;14(4):195-204. doi: 10.4155/bio-2021-0249, PMID 35034505.
Chen YC, Liu DZ, Liu JJ, Chang TW, Ho HO, Sheu MT. Development of terbinafine solid lipid nanoparticles as a topical delivery system. Int J Nanomedicine. 2012;7:4409-18. doi: 10.2147/IJN.S33682, PMID 22923986.
Gaba B, Fazil M, Khan S, Ali A, Baboota S, Ali J. Nanostructured lipid carrier system for topical delivery of terbinafine hydrochloride. Bull Fac Pharm Cairo Univ. 2015;53(2):147-59. doi: 10.1016/j.bfopcu.2015.10.001.
Toopkanloo SP, Tan TB, Abas F, Alharthi FA, Nehdi IA, Tan CP. Impact of quercetin encapsulation with added phytosterols on bilayer membrane and photothermal-alteration of novel mixed soy lecithin-based liposome. Nanomaterials (Basel). 2020;10(12):2432. doi: 10.3390/nano10122432, PMID 33291386.
Pal P. Formulation development and evaluation of antifungal proniosomal gel for topical application. World J Pharm Res. 2017;6:656-74. doi: 10.20959/wjpr20175-8300.
Yadav RP, Chauhan MK. Terbinafine-loaded chitosan scaffold for fungal wound treatment. Int J Pharm Sci Res. 2020;11:3806-18. doi: 10.13040/IJPSR.0975-8232.11.3806-18.
Abobakr FE, Fayez SM, Elwazzan VS, Sakran W. Formulation and optimization of terbinafine HCL solid lipid nanoparticles for topical antifungal activity. Int J Pharm Pharm Sci. 2019;11(12):16-25. doi: 10.22159/ijpps.2019v11i12.35560.