
Int J App Pharm, Vol 18, Issue 2, 2026, 143-149Original Article
DEVELOPMENT AND CHARACTERIZATION OF NON-PROPELLANT FOAM INCORPORATED ACYCLOVIR FOR TREATMENT OF HERPES INFECTION
PRAJITHA BIJU1*, VIVEK GHATE2, SHAILA LEWIS3, ASHWINI PRABHU4
1Department of Pharmaceutics, Yenepoya Pharmacy College and Research Centre, Yenepoya (Deemed to be University), Mangaluru-575018, Karnataka, India. 2Yenepoya Technology Incubator, Yenepoya (Deemed to be University), Mangalore-575018, India. 3Department of Pharmaceutics, Manipal College of Pharmaceutical Sciences, Manipal-576104, India. 4Division of Cancer Research and Therapeutics (CaRT), Yenepoya Research Centre, Yenepoya (Deemed to be University), Mangaluru-575018, Karnataka, India
*Corresponding author: Prajitha Biju; *Email: prajithabiju@yenepoya.edu.in
Received: 19 Jul 2025, Revised and Accepted: 23 Dec 2025
ABSTRACT
Objective: Herpes simplex virus (HSV) belongs to the herpes viridae family and primarily spreads through skin-to-skin contact. The study aimed to formulate topical anti-herpes non-propellant foam (NPF) incorporating acyclovir, offering a convenient alternative to conventional formulations. NPF is an optically transparent, thermodynamically stable, and light in consistency and evenly spreadable, thereby enhancing the permeation and poor permeability of acyclovir.
Methods: The acyclovir nanoemulsion was formulated by the emulsification method and dispensed through a container, which, upon actuation, produced the foam. The NPF was characterized by globule size analysis, pH, viscosity, and foaming performances. The NPF was evaluated for the drug permeation ex vivo excised skin, and deposition, while cytocompatibility was determined in vitro in human keratinocytes, respectively.
Results: The nanoemulsion had a globule size, pH, and polydispersity(PDI) of about 68.00 to 110.00 nm, pH of ~6.30 and a PDI of<0.200. The nanoemulsion viscosity was (⁓0.90 mPa. s, Spindle TSP-2, 20 rpm at 20 °C,), indicating the feasibility of expulsion from the container. Once actuated, the foam stability was 33.33% in 3 min, the expansion time was 100 sec, and density was 0.28g/ml, with a measured bubble size of 90.00 to 200.00 nm. The drug permeation of acyclovir was 411.147 per cm2 (µg/ml) with a flux rate of 0.89µg/cm2/h, and a permeability coefficient of 0.1789 in ex vivo studies with drug deposition in the skin up to ~78%. In addition, cytocompatibility studies proved acyclovir NPF to be safe.
Conclusion: The work suggests that NPF to be a new promising dosage form for topical application. The formulation when applied to the skin, instantly disappeared with no residue and was able to cover large body surfaces, presenting a no-touch application.
Keywords: Non-propellent foam, Acyclovir, Nanoemulsions, Skin irritation, Topical formulation
© 2026 The Authors.Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i2.56156 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
According to the World Health Organization (WHO), herpes is an infection caused by the herpes simplex virus (HSV), affecting approximately 493 million people worldwide [1]. When broken down by age, it was estimated that 3.7 billion people under 50 (67%) are infected with HSV-1, while 491 million individuals aged 15–49 y (13%) are affected by HSV-2 [2]. Among immune-compromised individuals, the mortality rate due to HSV can be alarmingly high, reaching up to 60%, while in immune-competent individuals, it remains significant at 30%. While HSV-1was most transmitted through oral contact, leading to oral herpes affecting the mouth and surrounding areas, it can also be spread through sexual contact, resulting in genital herpes by HSV-2.
Recent studies reveal that the majority of HSV infections are asymptomatic; however, when symptoms do occur, they often manifest as cold sores, painful blisters, or ulcers that may recur over time and last long in some cases. Eczema herpeticum was the secondary infection caused by HSV 1 or HSV 2 observed usually in impaired skin epidermis caused by Atopic dermatitis or Psoriasis [3]. As a pure drug, acyclovir is a synthetic purine (nucleoside analog), whose antiviral activity was dependent upon its metabolism within herpes-infected cells to sequentially form the mono-, di-and tri-phosphates, which eventually inhibit replication of the viral DNA polymerase enzyme [4]. Though acyclovir provides the desired outcome in Herpes infection, its poor penetration and growing evidence of microbial resistance have raised concerns about effective treatment. The study conducted by Nair et al., to quantify acyclovir in layers of skin observed a low skin uptake and rapid clearance of acyclovir from the target site [5]. However, the studies performed by John et al. to check the efficacy of different dosage forms confirmed formulations containing aqueous vehicles achieved cutaneous healing with reduced virus shedding [6]. The research findings align in parallel; the non-propellent foam (NPF) was an aqueous-based formulation having high drug permeation and retention with equal or more efficacy than the cream.
Acyclovir has a molecular weight of 225 daltons and a maximal solubility in water of 1.3 mg/ml at 25 °C and has a dissociation constant (pKa) of 2.27 and 9.25 [7].
Fig. 1: Structure of acyclovir
Foam-based therapies have become increasingly popular due to their cosmetic appeal and suitability for application on hair-bearing areas of the skin. Their non-sticky nature, ease of self-application without product contamination, and convenience for both geriatric and pediatric patients make foam formulations particularly patient-friendly [8]. The foam drug delivery system increases the effectiveness of therapy by its instant absorption and covers larger surface areas of the body, thus improving patient compliance [9]. NPF is a lotion formulated as o/w emulsions containing no propellant gases. They are the colloidal dispersions containing medicaments dispersed (hydrophobic or lipophilic) in a blend of lipids/oils and water, emulsified using a surfactant, which upon actuation can be dispensed as a quick-breaking porous system, from a non-propellant pump [10]. They are highly porous and break into a small amount of supersaturated emulsion at the interface of the skin, allowing an effective permeation of the drug. The present study investigated the NPF containing acyclovir for its effective permeation and thus can improve the bioavailability of the drug.
MATERIALS AND METHODS
Acyclovir (CAS NO#59277-89-3; Yarrow Chemicals), Tween 20 (CAS NO# 9005-64-5 ≥ 99% pure, Balaji Chemicals, Gujarat), Labrafac was a kind gift from Gattefosse, India Pvt. Ltd., Mumbai, Benzyl Alcohol (Balaji Chemicals, Gujarat), Ethanol (Balaji Chemicals Gujarat), Double-distilled deionized water was used for all the experiments. magnetic stirrer (Remi Laboratory Instruments, India), Nano ZetaSizer (Malvern Instruments), DV2T Viscometer (AMETEK Brookfield, USA), digital image processing method in ImageJ (U. S. National Institutes of Health, Bethesda, Maryland, USA). All the chemicals of analytical grade were used for the study.
Experimental methods
Solubility studies for the selection of oil phase, surfactant, and Smix ratio
Selection of oil phase
The solubility of the acyclovir in different oil was determined by taking a specified oil (Labrafac, palm oil and olive oil) in a beaker into which an excess drug was added in small quantity and stirred gently to the heated oil (50 °C) until a small quantity of drug deposit in the bottom of the beaker undissolved. The drug is pre-and post-weighed to check the amount of drug dissolved in the oil [11].
Selection of the surfactant
The fixed volume (10 ml) of surfactant was taken in a beaker into which excess drug was added and stirred until the acyclovir was completely dissolved in the surfactant. The study used Tween 20®, Tween 80®, and Span 20® as the surfactant [12].
Selection of Smix ratio (Surfactant: Co-surfactant) for non-propellent foam formulation
The varying volume (10 ml) of Tween 20® and ethanol (co-surfactant) was taken in different beakers, into which excess acyclovir was added and stirred. The beaker, having a transparent appearance with no settling of drug was considered as the Smix ratio for the preparation of acyclovir nanoemulsion [12].
Preparation of the foam
The NPF was nanoemulsions prepared by a simple method involving the addition of the oil phase dropwise into the aqueous phase. Accurately 5.0 g) of acyclovir was dissolved in 77.50 ml of water, placed on the magnetic stirrer (500 rpm, Remi 5MLH, Q-20A, Mumbai, India) maintained at 30 °C. The Smix was previously prepared and added to the water phase, stirred until completely dissolved. Labrafac (0.50 ml) was slowly added to the water phase and stirred until a uniform mixture was formed without any sign of phase separation. The formulation was bath sonicated for 5 min, at 30 °C, 220 V and 50 Hz, power density of 0.2W/cm2 (Patel Scientific Instruments Private Limited, Ahmedabad, India). The emulsion was transferred to the spray bottle and actuated to generate the foam. A set of blank NPF was prepared by varying the concentration of composition and finally composition was optimised based on the foam foaming ability. Blank NPF and test foam (nAcycNPF) were prepared and evaluated by comparing with Cetaphil moisturizing foaming face wash (Galderma Skin Health, India) as a reference standard serve as a vehicle reference for evaluating foamability [13].
Evaluation of NPF-based formulation
Physical appearance and quality, pH and globule size estimation
The NPF was visually examined either to be viscous or runny in nature and rated using mathematical symbols [14]. The pH of the foams was measured at 25 °C using a pH electrode containing a calomel glass electrode that was pre-calibrated using buffer pH 4.0, 7.0, and 9.0. Malvern NanoZS instrument was used to measure the size of the globules. The emulsion was diluted in the ratio of 1:10 using Milli-Q water. Samples were analyzed in triplicate, and the mean values of the three were taken and given in table 3. The transmission electron microscopy image of the optimized nanoemulsion is depicted as fig. 3.
Table 1: Formulation composition (5%) of foams and the uses of various formulation ingredients
| S. No. | Ingredients | Use | Formulation and composition (%w/v) | |
| nAcycNPFa | NPFb | |||
| 1 | Acylovir | Anti-Viral Agent | 5.0 | - |
| 2 | Tween 20 | Solubilizer, foaming agent | 18.0 | 18.0 |
| 3 | Ethanol | Co-surfactant, emulsifier | 3.0 | 3.0 |
| 4. | Labrafac™ Lipophile WL 1349 | Emollient and oily vehicle | 0.50 | 0.50 |
| 5. | Distilled water | Water phase | 77.50 | 77.50 |
| 6. | Benzyl Alcohol | Preservative | 1.0 | 1.0 |
anAcycNPF: acyclovir non-propellent foam; bNPF: blank non-propellent foam
Foamability and foam expansion
After the measurement of globule size, the emulsion was transferred into the propellant-free foam container. Briefly, the formulations (VOLactual, 10 ml) was then actuated and discharged into a glass measuring cylinder (25 ml). The volume occupied by the foam in the measuring cylinder was noted immediately after actuation (VO Lintial) and after three min (VOL final) [2].
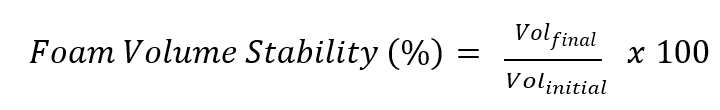 …… Eq. 1
…… Eq. 1
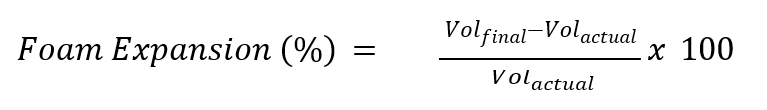 ……. Eq. 2
……. Eq. 2
Relative foam density
Relative foam density (RFD) was measured by placing water as the reference standard. The relative foam density was measured using the equation:
![]() …. Eq. 3
…. Eq. 3
Mf= Mass of the foam
Mw= Mass of the water
Determination of foam viscosity
The viscosity of the preparation and marketed formulation was determined using IKA Rotavisc hi-vi Heli, Spindle: TSP-3. The studies were conducted at 20 rpm, and the temperature was maintained at 25 °C. The studies were carried out in triplicate and viscosity was expressed in mPa.s [15].
Bubble size estimation
The test sample was actuated onto the watch glass without disturbing the photograph (Nikon Z 5 Mirrorless Digital Camera with 24-200 mm Lens), which was captured immediately. To estimate the bubble size the image was uploaded to ImageJ Software (National Institutes of Health, USA). Approximate diameters (scale 100 µm) of 100 bubbles was measured. The results were then loaded in Origin 2024b Software (graphing and analysing), and a histogram was plotted [15].
Drug content analysis
NPF was actuated one time to a volumetric flask of 10 ml measuring volume and diluted using 10 ml phosphate buffer pH 5.5 until a clear solution was formed. The solution was filtered and after subsequent dilution, the drug content was measured using an UV spectrophotometer (Shimadzu UV 1900, Model No 00101) at 257 nm. Drug content was determined using the following equation.
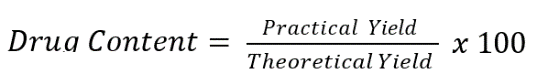 …. Eq. 4
…. Eq. 4
Ex vivo permeation studies
Vertical type of Franz diffusion cells using excised porcine skin was used to perform the ex vivo drug diffusion studies. The previously calibrated volume of the formulation was actuated into the donor compartment, and 5 ml buffer (pH 5.5) was placed in the receptor compartment; the porcine skin (pre-treated) of a thickness of 500µm, exposing a surface area of 3.799 cm2, was used. The sample from the receptor compartment was withdrawn for all 45 min, and replaced with fresh buffer, and the resultant sample, after suitable dilution, was analysed using a UV spectrophotometer at 257 nm. The permeation profile was obtained by plotting a graph of cumulative drug permeation versus time. The flux (μg/cm2/h), and permeability coefficient were calculated from the slope of the linear portion of the cumulative drug permeated (μg/cm2) versus the time (h) profile [15, 16]. Further, the surface of the skin was cleared with cotton swab and soaked in water. After 24 h, it was homogenised and analysed using UV spectrophotometer to determine the drug retained in layers of skin.
Evaluation of cytocompatibility of the test formulations
Human keratinocytes (HaCaT) were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% FBS and 1% antibiotic-antimycotic solution. They were subcultured upon attaining 70% confluence and used for the experiments after three consecutive passages. The cytocompatibility of the test formulations was assessed using Methyl Thiazolyl Tetrazolium (MTT) assay [17]. Briefly, HaCaT cells were seeded onto 96-well microtiter plates at a density of 5000 cells/well and incubated at 37C and 5% CO2. After adherence, they were treated with the test formulations at different concentrations viz., 5, 10, 25, 50, 100, 250, 500, 750, and 1000 µg/ml and incubated for 24 h. Spent media was removed, and MTT reagent was added to the wells and further incubated for 4 h. Formazan crystals were solubilized using DMSO. Absorbance was recorded at 570 nm using a multimode microplate reader (Fluo STAR Omega, BMG Labtech). Percentage cell viability was calculated concerning the untreated control.
RESULTS
NPF are oil-in-water emulsions that expand as foams upon actuation with the aid of a propellant-free pump. This formulation helps in the solubilization of both hydrophilic and lipophilic drugs. In the current study, the model drug used was acyclovir, and according to the literature, when applied topically, it effectively shortens the duration of viral shedding and accelerate the healing of herpes simplex virus infections. However, its poor permeability remains a significant limitation.
Thus, the permeability of acyclovir was enhanced by formulating it as an emulsion, which serves as a drug delivery carrier, thereby improving its stability. For the ease of application and targeting the larger surface area, the resulting formulation was subsequently incorporated into a foam container and actuated.
Table 2: Selection of surfactants and oil phase for the formulation of nanoemulsion
| Oil | Solubility* (mg drug/g) | Surfactant | Globule size*(nm) | Entrapment efficiency*(%) |
| Labrafac lipophile WL 1349 | 120.47 ±10.25 | Tween 20 | 92.2±0.57 | 84.37±0.17 |
| White palm kernel oil | 70.69±5.48 | Tween 80 | 101.8±0.84 | 85.51±0.04 |
| Castor oil | 67.44±9.17 | Span 60 | 110.8±0.35 | 87.87±0.84 |
*Data are presented as mean±SD (n = 3)
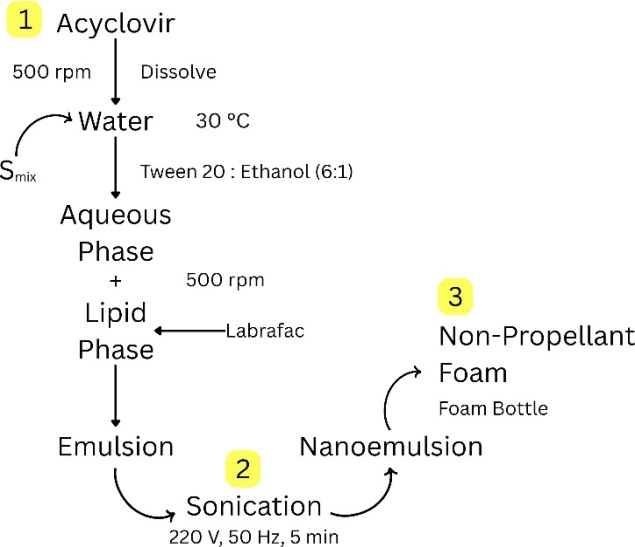
Fig. 2: Schematic representation of the formulation of NPF containing acyclovir. Steps (1) shows the scheme for the preparation of the emulsion, consisting of the aqueous phase (the drug Acyclovir, Smix and Water) while the lipid phase consisted of Labrafac; (2) sonication of the emulsion to form a nanoemulsion, and (3) filling the foam bottle with the nanoemulsion, which upon actuation will form the non-propellant foam
Preformulation studies
Preparation of NPF and nAcycNPF
The oil phase was optimized for the preparation of nanoemulsion, followed by the most critical step, observing the surfactant and co-surfactant (Smix) ratio required to stabilize the emulsion. After establishing the optimal Smix ratio and the oil phase, the compatibility between the oil phase and Smix concentration was fine-tuned by varying their concentrations by formulating the nanoemulsion. The formulation was sonicated for 5 min to prevent particles from settling before being transferred to a foam container for evaluation.
Evaluation of the NPF
Appearance, pH, and globule size distribution
All the formulations were clear and runny, allowing the formulation to pass through the actuated pump easily. The pH of the foam ranged from 5.0 to 6.8, closer to the skin pH. The pH in this range maintains the skin's natural environment, inhibiting the superinfection caused by harmful bacteria and enhancing enzyme activity for improved wound healing [18]. In the current study, nanoemulsion measured globule size from 68.17±1.06 to 111.25±0.04 nm (table 3), which is represented visually using TEM images (fig. 3). The nanoscale droplets facilitate deeper skin penetration, enabling more effective drug delivery to subdermal layers. Based on this observation, formulation (S5) was considered for further studies.
Table 3: Data on appearance, pH, PDI, viscosity, and globule size of different formulations
| Formulation code | Appearance | pH | PDI | Viscosity at 37 °C±2 °C (mPa. s) | Globule size (nm) |
| S1 | Clear | 6.3 | 0.203±0.091 | 0.99±0.45 | 111.25 ±0.04 |
| S2 | Clear | 6.8 | 0.199±0.01 | 0.93±0.32 | 98.47 ±0.04 |
| S3 | Clear | 6.0 | 0.197±0.021 | 0.90 ±0.64 | 99.95 ±0.75 |
| S4 | Clear | 6.3 | 0.185±0.01 | 0.87 ±0.81 | 96.17 ±2.25 |
| S5 | Clear | 6.2 | 0.181±0.089 | 0.80 ±0.02 | 68.17 ±1.06 |
Data are presented as mean±SD (n = 3), PDI=Polydispersity index
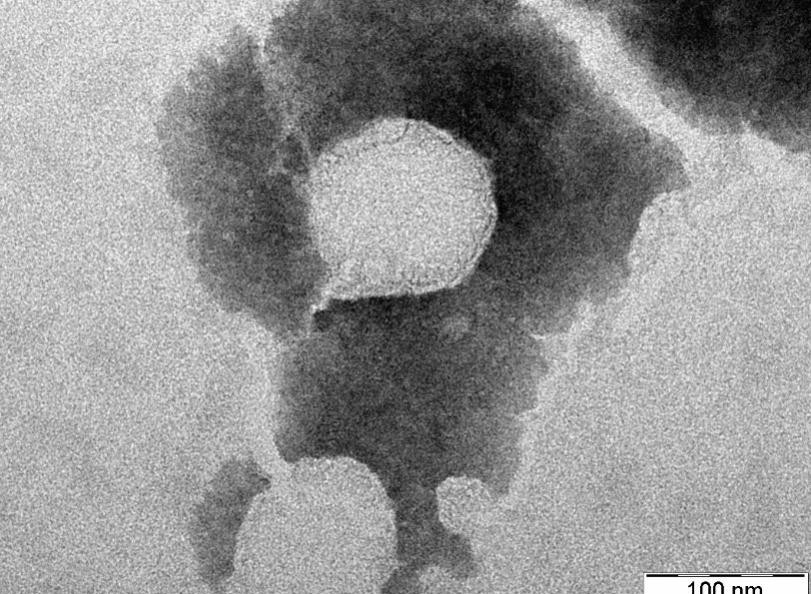
Fig. 3: TEM image of optimized nanoemulsion containing Smix 6:1 in the formulation of NPF
Foamability and foam expansion
The foam expansion and foamability stability were100% and 33% in 3 min for the optimized formulation, respectively (10).
Relative foam density (RFD)
The optimized foam was found to have a density 28% higher than that of water, which can be attributed to the addition of surfactant to the formulation.
Determination of foam viscosity
The reference product (Cetaphil) had a viscosity of 10.09 mPa.s and was readily flowable from the container. The NPF formulated also had comparable viscosities (0.87–0.90 mPa. s) as mentioned in table 3, indicating the feasibility of expulsion from the container.
Drug content analysis
The drug content was determined in triplicate, and the concentration of the drug in each actuation (equivalent to 0.5 ml) was found to be 22.20 mg/ml.
Bubble size estimation of the NPF
Upon comparing the bubble size in NPF with the droplet size in nanoemulsions, the foam bubbles were significantly larger, with diameters typically ranging from 90 to 200 µm. This large size is due to the air-filled nature of the spherical bubbles, which increases the foam volume while keeping it lightweight (i. e., Ostwald ripening). The larger bubble size aids in the spreading of the foam over the skin, making it ideal for surface-level application [13, 21]. In contrast, the smaller globule size in nanoemulsions achieved a more stable dispersion, enhancing the bioavailability.
Ex vivo permeation studies of NPF
Ex vivo skin permeation of acyclovir from nAcycNPF formulations was evaluated using excised porcine skin and quantified by UV spectrophotometry. Formulation S5 with Smix ratio 6:1 (surfactant: cosurfactant) showed a maximum drug release of 411.147 per cm2 (µg/ml) across the skin at the end of 495 min (fig. 5). The linear portion of the cumulative drug permeated (μg/cm2) versus time (h) profile at steady state conditions had a flux rate of 0.89µg/cm2/h and a permeability coefficient of 0.1789. Further, the skin deposition of acyclovir NPF showed enhanced skin deposition (78.00 %). These results were compared with reported permeation studies of plain acyclovir gel, which showed more permeation across the skin with less concentration of acyclovir deposited onto the skin. Contrary to it, acyclovir NPF exhibited higher permeability, since the globules with a size<400 nm can pass through the skin membrane easily, increasing the drug's permeability. The selection of ethanol demonstrated significantly higher skin permeation and retention, which is supported by a study conducted by steingrimsdottir et al. But the use of ethanol is associated with the drawback, which can drive drugs into deeper skin layers and systemic circulation. The lag time reflects the time taken for the drug to saturate and traverse the membrane before steady-state flux is reached. Surfactants increase hydration of the stratum corneum and alter lipid ordering, this can increase partitioning and solubility of the drug within the membrane, thereby enhancing drug flux. However, our present study used a minimal volume of ethanol solely to ensure complete oil miscibility and prevent phase separation.
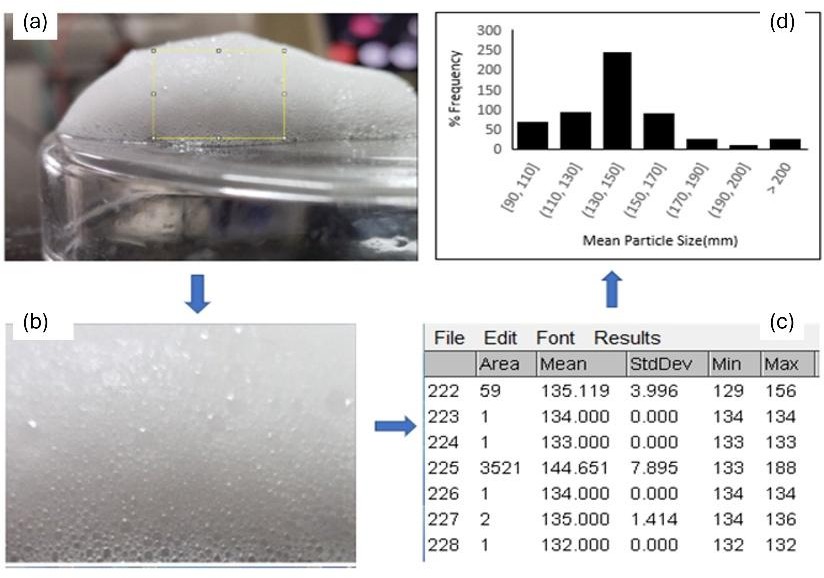
Fig. 4: Bubble size estimation of NPF; photographic images of the foam bubbles (a); digital image processing of the bubble size (b); analysed using ImageJ software (c); and bubble size distribution (d)
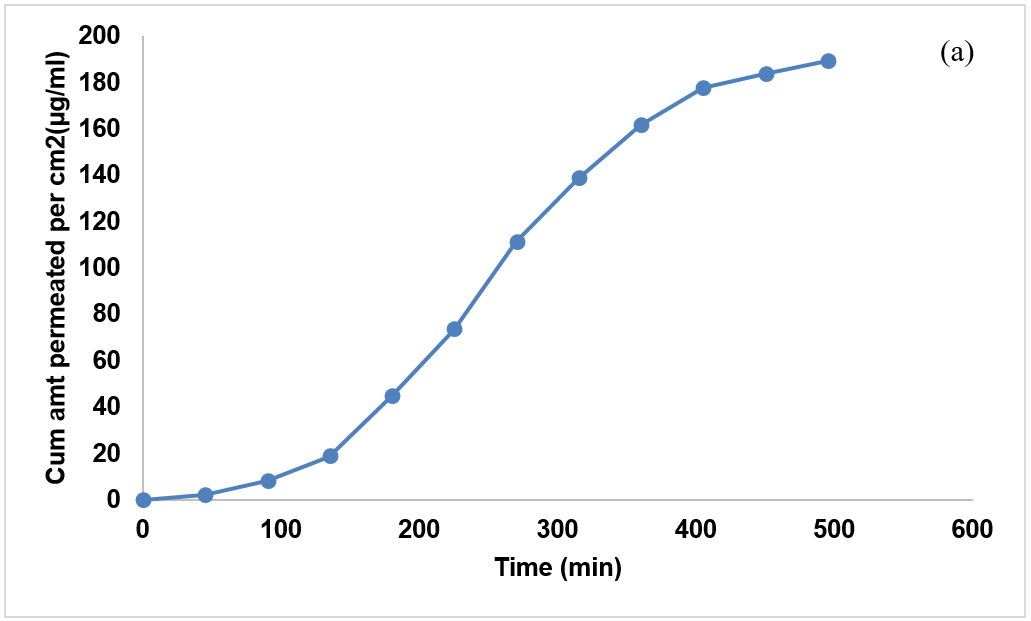
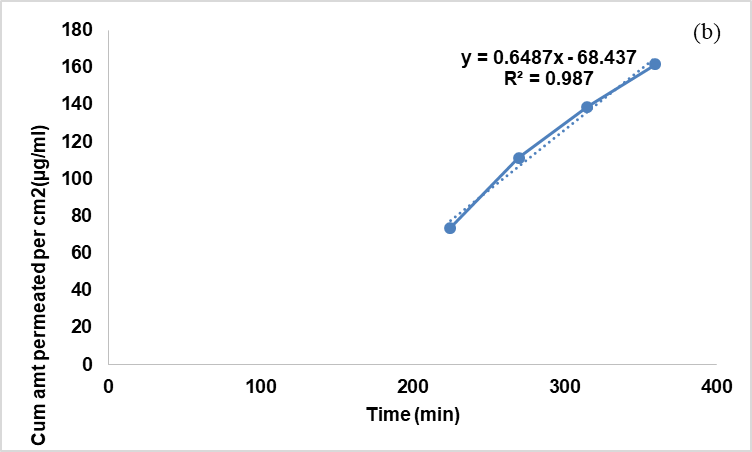
Fig. 5: Ex vivo permeation studies for NPF formulation (S5) using porcine skin for 495 min (a) and the plot of the linear portion of the permeation profile used in the calculation of the flux (b)
Evaluation of cytocompatibility of the test formulations
According to Wei et al., patients under acyclovir treatment must be observed for signs and symptoms of renal failure; hence, the safe concentration must be analysed. Keratinocytes, being the major cell type in mammalian skin, are considered the optimum in vitro model for assessing the safety of cosmetic formulations. Keratinocytes are also involved in regulating inflammatory responses mediated by exposure to cosmetic products [21, 22]. The results of the MTT assay on human keratinocytes depicted the cytocompatibility of acyclovir formulation at the tested concentrations ranging from 5-1000 µg/ml, which covers the actual therapeutic concentration (50μg/ml) delivered by the formulation. Thus, the concentrations used in the cytotoxicity assays do represent realistic exposure levels. Cell viability was maintained in the compatible range in the lower concentrations and slightly alleviated in the higher concentration, after 24 h of exposure to the test formulation. These results confirmed the safety of acyclovir formulation for skin use.
Hence, our study tested the cytocompatibility of acyclovir foam formulation on human keratinocytes, which did not exert any toxicity on the tested cells. It is explicitly acknowledged that the assay concentrations were limited to this range, and effects at concentrations outside this range were not evaluated, which should be considered in further studies.
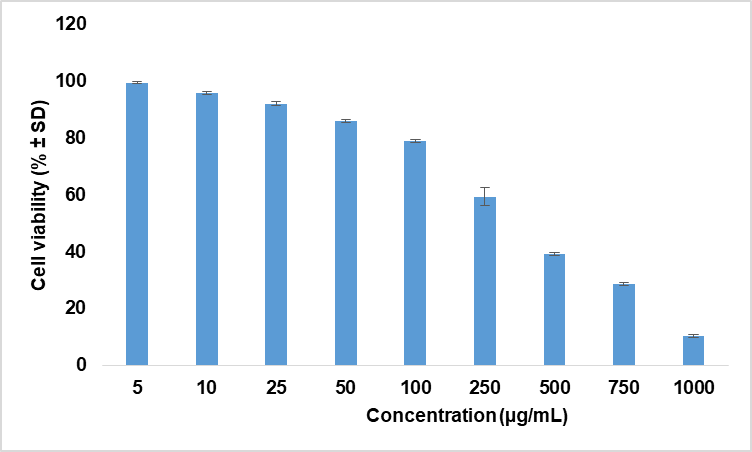
Fig. 6: Cytocompatibility of acyclovir NPF at the tested concentrations ranging from 5-1000 µg/ml, data are presented as mean±SD (n = 3)
DISCUSSION
Preformulation studies performed on acyclovir observed that Labrafac showed maximum solubility. Labrafac, a mixture of medium-chain triglycerides, mainly consists of caprylic (C8) and capric (C10) acid, then other short-chain fatty acids. It was observed that the surfactant Tween 20 solubilised acyclovir better compared to other surfactants. According to the report published by Schmidt RF et al. solubilization capacity of Tween 20 increased with an addition of suitable co-surfactants, by significantly reducing the interfacial tension and enhancing its capacity to solubilize oil [23]. The Smix ratio was fixed based on the homogeneity in the solution, with serial dilution, Tween 20 and ethanol in ratio 6:1 produced a clear solution with no sign of precipitation.
The current study formulated a nanoemulsion containing a Smix ratio (6:1v/v) along with other ingredients. Findings reported by Akram S et al. detail that the oil phase (Labrafac) alone has no influence on globule size and PDI but a change in the size distribution probably reflects variations in the affinities between the surfactants and oil [19]. S1measured the largest size due to the considerably high concentration of oil phase, resulting the surfactant concentration insufficient to reduce the interfacial tension. On increasing the concentration of surfactants by maintaining the oil concentration fixed, the globule diameter gradually decreased, with no significant changes appearance and PDI of the formulation. Overall, the size of the globule was observed to be in the nanometer range, spherical shape, with low polydispersity (<0.25); with no sign of creaming, cracking, and phase inversion on storage for several days. These properties make the emulsion thermodynamically stable.
Sugumar et al., studied the impact of Tween 80 on stabilizing the eugenol oil component on nanoemulsion and explained based on the HLB value of Tween 80, as the same polar head with smaller hydrophobic tails, causing the different molecular packing at the continuous phase-droplet interface, resulting in the droplets with a mean diameter of 55 nm. The formulation (S5) with Smix (6:1) and 0.5 ml of Labrafac measured the globule size of 68.00 ±1.06 nm and aligned with the requirement for dermal delivery.
The foamability of the NPF depends on the surfactants and oil phase, which are inversely proportional to the foam expansion. The breakdown and subsequent liquefaction of the foam are critical in providing intimate contact of the nanoemulsion globules with the skin tissues. The breakdown of the foam essentially allows the formulation to spread easily on the infected surface with minimal touch and disappear rapidly from the surface. Also, foam volume stability indicates the time the bubble remains viable on the skin upon application. Since the foam contains bubbles of different sizes, it takes different times to break the bubbles and allow easy penetration of the medication into the skin, but overall, all the bubbles collapse within 2 min. The current formulation contained a small volume of Labrafac, hence the surfactant concentration to coat the oil phase is less, which in turn helps in making the foam lighter. The light foam has a high air-to-liquid ratio, tends to spread more evenly, and may absorb faster, promoting rapid delivery of active ingredients like acyclovir to the skin [24].
Formulations with lower viscosity often produce larger bubbles, while higher viscosity can restrict foam expansion, resulting in a thicker, denser foam. In nanoemulsions, viscosity generally increases with a higher oil-to-surfactant ratio. An oil phase is typically added to nanoemulsions to improve the solubility of poorly soluble drugs. However, because acyclovir is highly water-soluble, the oil phase has no role in solubility enhancement but helps primarily to maintain the spherical structure of globules and enhances the foam's texture. Water plays a crucial role in the topical delivery of drugs. Increasing the water content up to 70% enhanced acyclovir's skin permeation and retention, as it is water-soluble and forms a concentration gradient that facilitates better diffusion through the skin's lipid bilayer. It has been suggested that increasing the concentration of surfactants enhances the drug’s affinity for the emulsion vehicle [22]. At the same time, it reduces the drug's thermodynamic activity, facilitating a slower release of the drug from the vehicle to the skin. This results in sustained drug release and improved drug deposition within the skin layers [25, 26].
CONCLUSION
Herpes infection is caused by Simplex Herpes Virus (HSV), resulting in painful blisters or ulcers that gradually spread throughout the body and can recur over time. Our research suggests that NPF could be a better alternative treatment for existing marketed formulations since foams can cover a larger surface area, non-sticky minimizing the dose and associated side effects. On topical application, first-episode primary herpes, the mean duration of viral shedding is 4 days. This application will effectively prevent the possibility of microbial infections and aid in the quicker proliferation of skin tissues. And the time to complete crusting of lesions present at the initiation of therapy takes about 7 days, which represents the shorter duration of viral shedding with no recurrent herpes. Investigations suggested that the microemulsion approach for topical delivery is feasible and improves the drug's solubility, permeability, and therapeutic effect by minimizing the dose and associated side effects. They are easy to formulate, transport, store, and use and have a long shelf-life.
ACKNOWLEDGEMENT
The authors gratefully acknowledge the funding of this work by the Yenepoya University Seed Grant Ref. No.: YU/REG/ACA/Seed Grant/2025. The authors thank Gattefosse India Private Limited, Mumbai, for generously providing Labrafac used in this study.
ABBREVIATIONS
Simplex Herpes Virus (HSV); Nonpropellent Foam (NPF); World Health Organization (WHO); Human keratinocytes (HaCaT); Dulbecco’s Modified Eagle’s Medium (DMEM).
AUTHORS CONTRIBUTIONS
All authors contributed to the study's conception and design. Material preparation, data collection, and analysis were performed by Prajitha Biju, Vivek Ghate, Shaila Lewis and Ashwini Prabhu. All authors read and approved the final manuscript.
CONFLICTS OF INTERESTS
Prajitha Biju, Vivek Ghate, Shaila Lewis and Ashwini Prabhu declare no conflict of interest.
REFERENCES
Looker KJ, Magaret AS, Turner KM, Vickerman P, Gottlieb SL, Newman LM. Global estimates of prevalent and incident herpes simplex virus type 2 infections in 2012. PLOS One. 2015;10(1):e114989. doi: 10.1371/journal.pone.0114989, PMID 25608026.
James C, Harfouche M, Welton NJ, Turner KM, Abu-Raddad LJ, Gottlieb SL. Herpes simplex virus: global infection prevalence and incidence estimates, 2016. Bull World Health Organ. 2020;98(5):315-29. doi: 10.2471/BLT.19.237149, PMID 32514197.
Esser M. Ueber eine kleine epidemie von pustulosis acuta. Ann Paediatr. 1941;157:156-61.
De Clercq E, Field HJ. Antiviral prodrugs the development of successful prodrug strategies for antiviral chemotherapy. Br J Pharmacol. 2006;147(1):1-11. doi: 10.1038/sj.bjp.0706446, PMID 16284630.
Nair AB. Quantification of uptake and clearance of acyclovir in skin layers. Antivir Ther. 2016;21(1):17-25. doi: 10.3851/IMP2970, PMID 26046929.
Zigrayova D, Mikusova V, Mikus P. Advances in antiviral delivery systems and chitosan‑based polymeric and nanoparticulate antivirals and antiviral carriers. Viruses. 2023;15(3):647. doi: 10.3390/v15030647.
Gnann JW, Barton NH, Whitley RJ. Acyclovir: mechanism of action pharmacokinetics safety and clinical applications. Pharmacotherapy. 1983;3(5):275-83. doi: 10.1002/j.1875-9114.1983.tb03274.x, PMID 6359082.
Eita AS, Makky AM, Anter A, Khalil IA. Atorvastatin-loaded emulsomes foam as a topical antifungal formulation. Int J Pharm X. 2022;4(4):100140. doi: 10.1016/j.ijpx.2022.100140, PMID 36465276.
Arzhavitina A, Steckel H. Foams for pharmaceutical and cosmetic application. Int J Pharm. 2010;394(1-2):1-17. doi: 10.1016/j.ijpharm.2010.04.028, PMID 20434532.
Kurowska A, Ghate V, Kodoth A, Shah A, Shah A, Vishalakshi B. Non-propellant foams of green nano-silver and sulfadiazine: development and in vivo evaluation for burn wounds. Pharm Res. 2019;36(8):122. doi: 10.1007/s11095-019-2658-8, PMID 31218556.
Baghel S, Nair VS, Pirani A, Sravani AB, Bhemisetty B, Ananthamurthy K. Luliconazole-loaded nanostructured lipid carriers for topical treatment of superficial Tinea infections. Dermatol Ther. 2020;33(6):e13959. doi: 10.1111/dth.13959, PMID 32618400.
Kumar S, Wadhwa K, Pahwa R, Ali J, Baboota S. Screening of surfactant mixture ratio for preparation of oil-in-water nanoemulsion: a technical note. J Appl Pharm Sci. 2024;14(10):121-7. doi: 10.7324/JAPS.2024.180648.
Mirtic J, Papathanasiou F, Temova Rakusa Z, Gosenca Matjaz M, Roskar R, Kristl J. Development of medicated foams that combine incompatible hydrophilic and lipophilic drugs for psoriasis treatment. Int J Pharm. 2017;524(1-2):65-76. doi: 10.1016/j.ijpharm.2017.03.061, PMID 28359820.
Bibi S, Kaur R, Henriksen Lacey M, McNeil SE, Wilkhu J, Lattmann E. Microscopy imaging of liposomes: From cover slips to environmental SEM. Int J Pharm. 2011;417(1):138-50. doi: 10.1016/j.ijpharm.2010.12.021.
Asghar Z, Jamshaid T, Sajid-ur-rehman M, Jamshaid U, Gad HA. Novel Transethosomal gel containing miconazole nitrate; development, characterization and enhanced antifungal activity. Pharmaceutics. 2023;15(11):2537. doi: 10.3390/pharmaceutics15112537, PMID 38004517.
Khattab A, Shalaby S. Optimized ciclopirox-based eudragit RLPO nail lacquer: effect of endopeptidase enzyme as permeation enhancer on transungual drug delivery and efficiency against onychomycosis. AAPS PharmSciTech. 2018;19(3):1048-60. doi: 10.1208/s12249-017-0917-8, PMID 29138987.
Kaur M, Singh K, Jain SK. Luliconazole vesicular-based gel formulations for its enhanced topical delivery. J Liposome Res. 2020;30(4):388-406. doi: 10.1080/08982104.2019.1682602, PMID 31631734.
Plumb JA. Cell sensitivity assays: clonogenic assay. Methods Mol Med. 2004;88:159-64. doi: 10.1385/1-59259-406-9:159, PMID 14634226.
Vedha Hari BN, Narayanan N, Dhevedaran K. Efavirenz-eudragit E-100 nanoparticle-loaded aerosol foam for sustained release: in vitro and ex-vivo evaluation. Chemical Papers. 2015;69(2):358-67. doi: 10.1515/chempap-2015-0005.
Schmidt RF, Prause A, Prevost S, Doutch J, Gradzielski M. Phase behavior and structure of a biocompatible microemulsion based on Tween 20, 2-ethylhexylglycerine and isopropyl palmitate in water. Colloid Polym Sci. 2023;301(7):753-62. doi: 10.1007/s00396-023-05119-9.
Akram S, Anton N, Omran Z, Vandamme T. Water-in-oil nano-emulsions prepared by spontaneous emulsification: new insights on the formulation process. Pharmaceutics. 2021;13(7):1030. doi: 10.3390/pharmaceutics13071030, PMID 34371723.
Farkas D, Kallai Szabo N, Saradi Kesztyus A, Lengyel M, Magramane S, Kiss E. Investigation of propellant-free aqueous foams as pharmaceutical carrier systems. Pharm Dev Technol. 2021;26(3):253-61. doi: 10.1080/10837450.2020.1863426, PMID 33307920.
Souto EB, Cano A, Martins Gomes C, Coutinho TE, Zielinska A, Silva AM. Microemulsions and nanoemulsions in skin drug delivery. Bioengineering (Basel). 2022;9(4):158. doi: 10.3390/bioengineering9040158, PMID 35447718.
Chen L, Annaji M, Kurapati S, Ravis WR, Jayachandra Babu R. Microemulsion and microporation effects on the genistein permeation across dermatomed human skin. AAPS PharmSciTech. 2018;19(8):3481-9. doi: 10.1208/s12249-018-1150-9, PMID 30140994.
Elshall AA, Ghoneim AM, Abdel Mageed HM, Osman R, Shaker DS. Ex vivo permeation parameters and skin deposition of melatonin-loaded microemulsion for treatment of alopecia. Futur J Pharm Sci. 2022;8(1):4-10. doi: 10.1186/s43094-022-00418-4.
Pa D, Phuvisaa B, Kiruthika S, Mohamed A, Abirami D, Gokul K. Design and optimization of acyclovir-loaded solid lipid nanoparticles: a sustained release approach. Int J Appl Pharm. 2025;17(1):355-64. doi: 10.22159/ijap.2025v17i1.51877.