
Int J App Pharm, Vol 18, Issue 1, 2026, 33-40Reviewl Article
REGULATORY AND FUNCTIONAL FRONTIERS IN PRECISION ONCOLOGY
VIVEK REDDY MURTHANNAGARI1*, KARUNAKARAN MOORTHY1, SYED SUHAIB AHMED2, INAMUL HASAN MADAR3
1,2Department of Regulatory Affairs, JSS College of Pharmacy, JSS Academy of Higher Education and Research, Ooty, Nilgiris, Tamil Nadu, India. 3Department of Pharmaceutics, Yenepoya Pharmacy College and Research Centre, Yenepoya (Deemed to be University), Mangalore, Karnataka-575018, India. 4Centre for Integrative Omics Data Science (CIODS), Yenepoya (Deemed to be University), Mangalore, Karnataka-575018, India
*Corresponding author: Vivek Reddy Murthannagari; *Email: mvivekreddy@jssuni.edu.in
Received: 21 Jul 2025, Revised and Accepted: 14 Nov 2025
ABSTRACT
Precision oncology has come a long way, largely thanks to our growing understanding of the molecular changes that drive cancer. By identifying these alterations, researchers and clinicians can now tailor treatments more effectively, offering what’s often called the right drug for the right patient at the right dose and at the right time. This approach has opened new possibilities in how we classify diseases, design clinical trials and use biomarkers and health technology to guide decisions. Modern tools like next-generation sequencing (NGS), RNA analysis and immune profiling have made it possible to analyse tumours and even detect genetic material like cell-free DNA from blood samples. These technologies help identify specific mutations or markers that could influence treatment. However, while the potential is enormous, there are still some challenges. For example, interpreting large volumes of genetic data can be tricky and there’s always the risk of false positives or unexpected findings. Plus, whole-genome sequencing and transcriptome profiling can still be expensive and time-consuming. To keep pace, regulatory agencies like the FDA in the U. S. and the EMA in Europe have put frameworks in place to ensure that precision therapies are developed responsibly. The FDA, for instance, encourages simultaneous development of diagnostics and treatments. Meanwhile, the UK’s MHRA has launched initiatives like the Precision Medicine Catapult to speed up innovation and translation from lab to clinic. Looking ahead, scientists are exploring even more refined strategies, such as functional precision oncology. Instead of relying solely on genetic sequencing, this approach incorporates real-time data about how tumours behave and respond to drugs, offering a more dynamic and personalised way to choose the most effective treatment.
Keywords: Precision treatment in oncology, Food and Drug Administration (FDA), European Medicines Agency (EMA), Molecular profiling, Targeted therapies
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.56182 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Precision medicine, also known as personalised medicine, stands out compared to traditional medicine by adapting treatment individually[1]. Traditional medicine is evidence-based medicine, which is considered a ‘one-size-fits-all’ (1-of-n) approach, which shows that the treatment is used for a large group of people with the same disease [2]. Alternatively, Precision medicine shows a ‘one-size-fits-one’ (n-of-1) approach where the treatment is provided individually for better outcomes, as shown in fig. 1 [3]. Precision medicine is commonly referred to as giving the right drug to the right patient at the right dose at the right time. In precision medicine, biomarkers are found and validated to determine how a patient will react to treatments. Cancer has a range of cell types and clinical design involves discovering and continuously testing predictive biomarkers for drug reactions to improve clinical trials and regulatory approval, as well as selecting therapies according to best practices [4]. The cause of cancer worldwide is projected to be 14 million in 2012 and 24 million in 2030 for which no country has a proper treatment to solve [5]. To prevent cancer, one must have a diverse perspective, ranging from sub-microscopy to micropolitics and identify the significance of molecular profiling and the biological background of the study, which leads to precision medicine [6]. To proceed with precision medicine also has major regulatory consequences; additionally, new clinical designs to recognise, confirm and targeted therapies need more knowledge of regulatory committees in institutions and the USFDA to establish that a useful approach reaches people beneficially without compromising their safety [7]. Regulatory body plays a major role in assessing the safety, efficacy and quality of personalised medicine and also in the timely approval of novel therapies for patients. For therapeutic decision-making on biomarker data, the FDA and EMA play a major role [8, 9].
Advances in precision medicine in oncology
The new cancer treatment is improving rapidly because of advances in DNA sequencing technology. The technical improvement has more knowledge on understanding cancer biology in terms of whole genome sequencing (WGS), targeted therapies, genotyping and bioinformatics. WGS is the core technology that supports the sequencing of cancer genomes. First-generation sequencing or Sanger sequencing (order of nucleotides in DNA sequencing) has been the chief method of DNA sequencing for 30 years. It is a successful technique, but the cost and improper output have led to the evaluation of Next-Generation Sequencing (NGS) as shown in fig. 2 [10]. NGS is a novel technique, primarily launched on the market by 454 life Sciences in 2005. The establishment of NGS has made it possible for screening a wide set of genes in one complete test, to recognise changes in the inadequate biopsy tissue, which is an inaccessible procedure [11]. Precision medicine in the treatment of cancer may be attained through analysis known as a liquid biopsy. The word liquid biopsy was introduced by Pantel and Alix-Panabieres to have knowledge of circulating tumor nucleic acid present in exosomes and particulars of platelets associated with cancer. An apparatus that permits the application of personalised therapy strategies is the liquid biopsy. Liquid biopsies are used as an instrument that is progressively being studied to record cancer development at once and lead to comprehensive work. The latest study has marked the use of liquid biopsies, concentrating mainly on inventions and their systematic and clinical strength. The section that consists of the liquid biopsies is circulating tumor cells, circulating tumor nucleic acids, free cells contained in exosomes and micro vesicles [12]. However, the clinical application of liquid biopsies faces technical and analytical limitations. The sensitivity is highly dependent on the analyte (e. g., cfDNA, CTCs), tumor stage and cancer type. Key challenges include variable detection thresholds, stringent pre-analytical sample handling requirements and the current inability to reliably detect certain structural variants using cfDNA [10]. A detailed overview of the performance characteristics and considerations for different liquid biopsy components is provided in table 6. In targeted therapies, notable examples include treatments for KRASG12C mutations, which were previously thought to be undruggable, as well as therapies targeting Exon 20 insertions and RET mutations and methods for delivering chemotherapies more precisely, such as the use of antibody-drug conjugates. These are specialized drugs that can deliver chemotherapy directly to cancer cells, particularly in lung cancer, enhancing treatment effectiveness while minimizing damage to healthy cells [13].
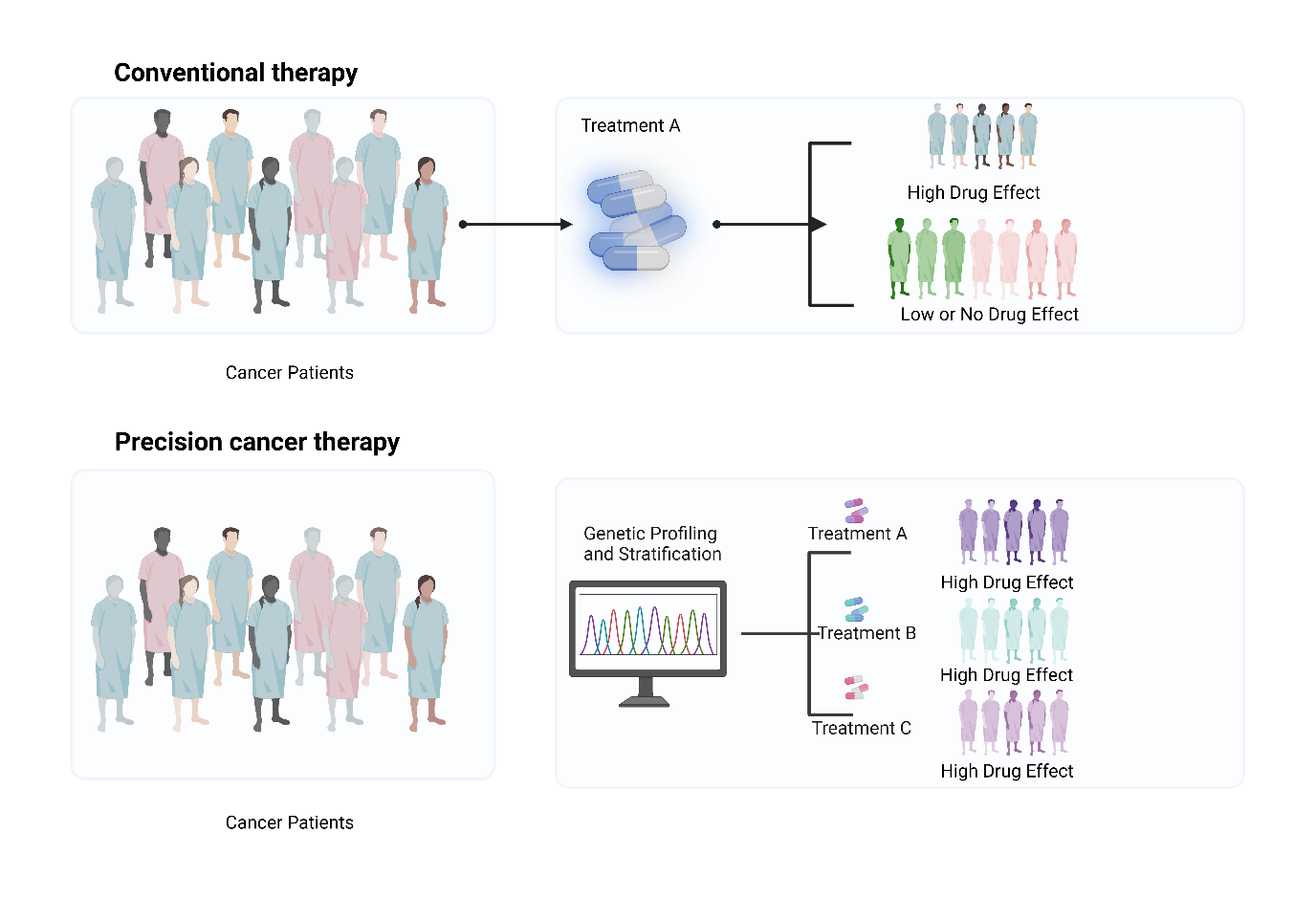
Fig. 1: Unlike traditional therapies that affect both healthy and cancerous cells, precision treatment specifically targets tumor-specific molecular alterations, minimising harm to normal tissue
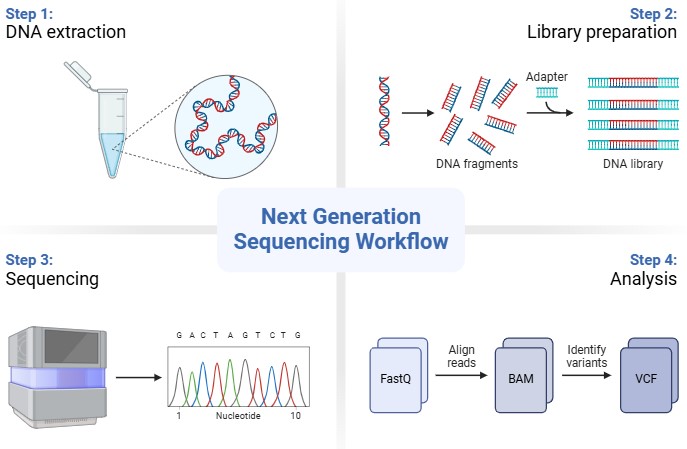
Fig. 2: Nucleic acid extraction, library preparation, sequencing, data analysis and interpretation
Table 1: Comparison of next-generation sequencing (NGS) platforms
| Platform | Read length and throughput | Detection sensitivity | Error rate | Cost per sample (approx.) | Input requirements | Notes | Reference |
| Illumina (NovaSeq, Next Seq) | Short read; up to 6 Tb/run | Detects SNVs down to 1% VAF | 0.1–1% | $500–1,200 | 10–50 ng DNA | Clinical gold standard | [11] |
| Ion Torrent (PGM, Proton) | Short read; semiconductor sequencing | 1–5% VAF | 1–2% | $400–800 | 10–20 ng DNA | Faster, indel error-prone | [10] |
| Oxford Nanopore | Long-read (10 kb–1 Mb) | Detects SVs>95% | 5–15% (improving) | $600–900 | 50–200 ng DNA | Real-time sequenci-ng | [14] |
| PacBio HiFi | Long-read, Q30+ | SNVs, SVs, phasing | <1% | $800–1,500 | ≥100 ng DNA | High accuracy; costly | [15] |
| Hybrid Approaches | Illumina+Nanopore/PacBio | Combines SNV+SV detection | <1% | >$1,000 | Multiple inputs | Used in comprehensive profiling | [16] |
The tumor elements consist of Circulating Tumor Cells (CTC), cell-free DNA (cfDNA), proteins, metabolites, extracellular vesicles and cell-free RNA (cfRNA) [14]. Tissue biopsy is commonly considered a main technique for the prognosis and prevention of many diseases. Tissue biopsy is used to study only mutations, whereas liquid biopsy shows on-time challenges, such as struggling to obtain them in the same location itself, the lack of noticeable tumor and the fundamental clinic-related method and deficiencies in routine testing. In 2013, the USFDA approved the first liquid biopsy test, the Cell search CTC enumeration platform (K073338-FDA), capable of detecting modern tumor cells in breast, colon and prostate cancer based on CTC count. All these novel techniques lead to a new place for research known as omics. The omics, which is known for the extensive study of the capacity, connection and activity of diverse molecules in cells of an organism [16]. Identification of actionable mutation and biomarker in clinical decision making such as (v-raf murine sarcoma viral oncogene homolog B1) BRAF V600 mutation for melanoma, (Epidermal Growth Factor Receptor) EGFR and (Anaplastic Lymphoma Kinase) ALK for lung cancer, (Human Epidermal Growth Factor Receptor 2) HER2 overexpression for breast cancer and (Kirsten Rat sarcoma oncogene homologue) KRAS mutation for colorectal cancer [17]. The main aim of personalised medicine is to acquire support from potential therapeutic targets for treatment selection. The biomarkers used in personalised medicine offer a way for companion diagnostics [18].
Table 2: Analytical sensitivity, pre-analytical considerations and detection thresholds of precision oncology tools
| Biomarker/Tool | Typical sensitivity | Detection threshold | Key pre-analytical considerations | Key limitations | References |
| cfDNA (NGS) | 70–85% (advanced); 40–50% (early-stage) | VAF 0.1–0.5% | Plasma separation within 2 h; EDTA tubes; –80 °C storage | Low tumor shedding in early stages; challenges detecting structural variants. | [19] |
| CTCs (Cell Search) | 40–70% (cancer-type dependent) | ≥1 CTC/7.5 ml blood | EpCAM-based enrichment | Misses epithelial-mesenchymal transition (EMT) CTCs; low abundance. | [20] |
| cfRNA | 60–75% (for fusion detection) | Requires intact RNA | Rapid stabilization critical (RIN>7) | High RNA fragility; lack of standardized protocols. | [21] |
| Exosomal DNA/RNA | Variable; lower in early-stage | Not standardized | Isolation method highly variable (e. g., ultracentrifugation, precipitation) | Technical heterogeneity; clinical utility still under investigation. | [22] |
| CAR-T Therapy | N/A | N/A | Patient-specific T-cell apheresis | Manufacturing Bottlenecks: Vein-to-vein time: 3-5 w. Cost: $375,000-$600,000 USD. Failure rate: ~5-10%. | [23] |
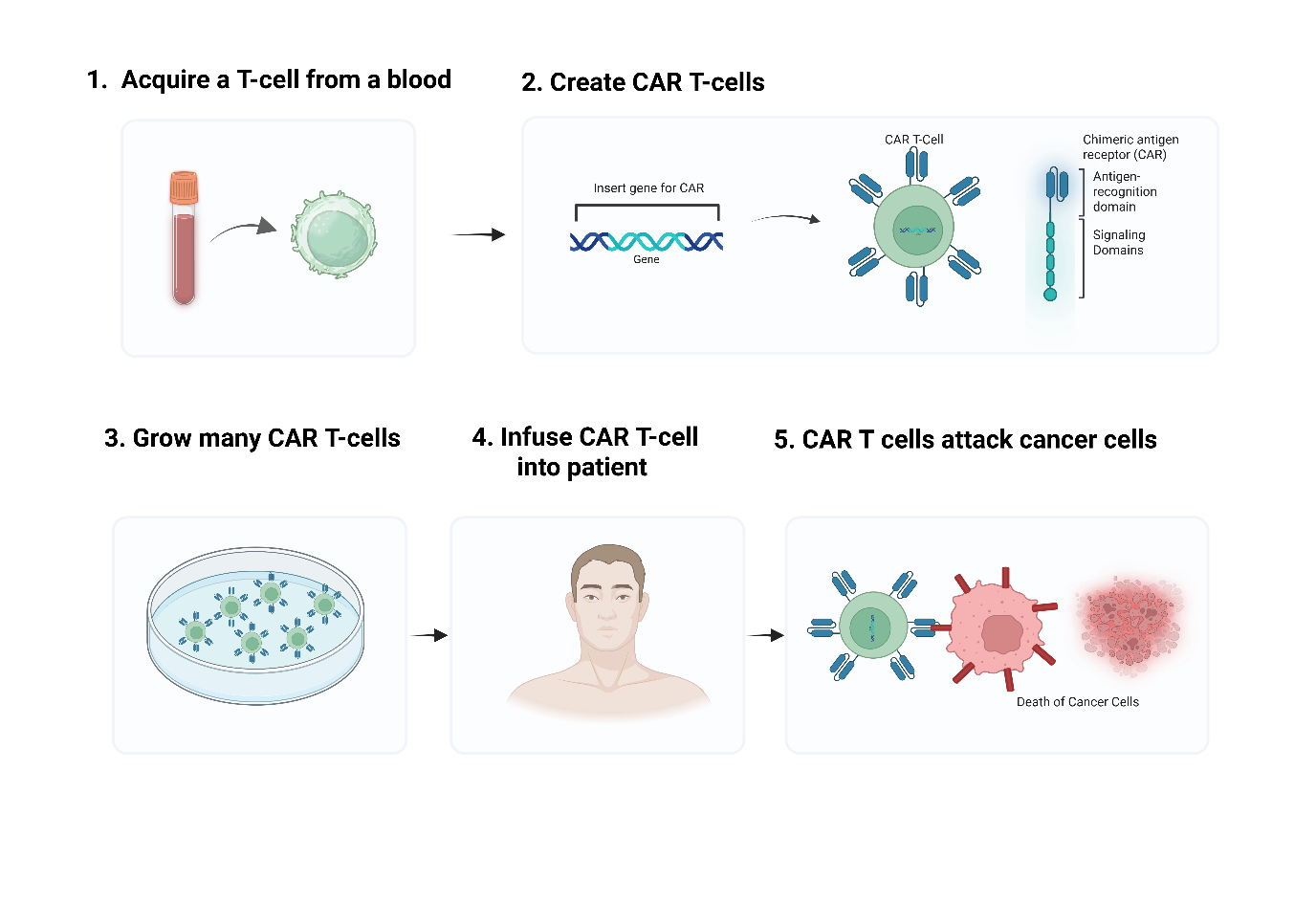
Fig. 3: An immunotherapy that uses genetically modifies T cells to fight against cancer
Targeted therapies and immunotherapies
Targeted therapy stops the pathway, which is important for cancer growth and maintenance, which increases the life span of the cancer patients by offering more precise interventions with potentially fewer side effects compared to traditional chemotherapy [24]. Where the immunotherapy seeks to stimulate a host immune response that results in sustained, long-term tumor destruction. The protein Imatinib, which is a tyrosine kinase inhibitor of Abelson Kinase (ABL), is present in aberrantly activated form in Chronic Myeloid Leukaemia (CML) due to a chromosomal translocation, which leads to the Breakpoint Cluster Region (BCR)-ABL fusion. The favourable outcome of imatinib in CML treatment has paved the way for the introduction of molecular targeted therapies in cancer [25]. The monoclonal antibody trastuzumab, which is used in targeted therapy for the treatment of breast cancer in which Human Epidermal Growth Factor Receptor 2 (HER2) is overexpressed, affects approximately 20-30% of breast cancer patients [26]. The checkpoint inhibitors, such as those targeting Programmed Cell Death Protein 1 (PD1), manage to modulate the immune response. Manifestation of PD1 on T-cells allows tumor cells to evade anti-tumor immunity by suppressing T-cell activity; thus, blocking of PD1 is the main immunotherapeutic tactic for cancer. Humanised monoclonal anti-PD1 antibody Pembrolizumab (Keytruda) was examined in various cancers across multiple cancers, which was approved by the USFDA for the treatment of advanced cancer [17] in 2014 initially for advanced melanoma, later expanding to non-small cell lung cancer (NSCLC) [27]. Based on end results, related T cells have developed to indicate a Chimeric Antigen Receptor (CAR) particular for Cluster of Differentiation 19 (CD19) B lymphocyte molecule, which has been approved by the USFDA for the prognosis of refractory pre-B cell acute lymphoblastic leukaemia and diffuse large B cell lymphatic leukaemia. CAR T cells are the earliest gene transfer therapy to obtain market approval by the USFDA in 2017, as shown in fig. 3. Despite their remarkable efficacy, the widespread adoption of CAR-T therapies is constrained by significant manufacturing bottlenecks. The process is complex and autologous (patient-specific), leading to long production timelines, high costs and potential manufacturing failures, which can limit patient access [32-34]. These quantitative challenges are summarized alongside liquid biopsy limitations in table 4.
Immunotherapy section only covers PD-1/PD-L1, CAR-T. No LAG-3/TIGIT or TME-driven resistance mechanisms
Immunotherapy has transformed cancer treatment by activating the immune system against tumors. Foundational approaches such as PD-1/PD-L1 and CTLA-4 inhibitors (e. g., Pembrolizumab, Ipilimumab) have achieved durable responses across multiple cancers. CAR-T cell therapies, like Tisagenlecleucel, further exemplify precision immunotherapy by engineering patient T cells to target tumor-specific antigens.
Beyond these established strategies, next-generation immune checkpoints such as LAG-3 and TIGIT are emerging to address resistance and non-responding populations. The FDA-approved combination of Relatlimab (anti-LAG-3) with Nivolumab validates LAG-3 as a therapeutic target, while anti-TIGIT agents like Tiragolumab show potential in PD-L1–high tumors. Additionally, the tumor microenvironment (TME), comprising immune and stromal components, plays a crucial role in modulating response and resistance to immunotherapy, emphasizing the need for integrated strategies targeting both immune checkpoints and TME-driven pathways.
Table 3: Expanding the immunotherapy landscape beyond PD-1 and CAR-T
| Target | Mechanism of action | Example agent(s) | Key indication/status | Significance |
| PD-1/PD-L1 | Blocks inhibitory signal on T-cells, restoring anti-tumor activity. | Pembrolizumab, Nivolumab, Atezolizumab | Approved for multiple cancers (e. g., melanoma, NSCLC). | Foundation of modern immunotherapy; provides durable responses in a subset of patients [28]. |
| CTLA-4 | Blocks inhibitory signal on T-cells, primarily in lymphoid tissue; promotes T-cell activation. | Ipilimumab | Approved for melanoma. | First checkpoint inhibitor to show survival benefit in melanoma; often used in combination with PD-1 inhibitors [29] |
| CAR-T Cells | Genetically engineered T-cells targeting specific tumor antigens. | Tisagenlecleucel (anti-CD19), Idecabtagenevicleucel (anti-BCMA) | Approved for B-cell ALL, DLBCL, Multiple Myeloma. | Pioneering personalized cell therapy for hematologic malignancies; demonstrates potential for long-term remission [25] |
| LAG-3 | Co-inhibitory receptor; dual blockade with PD-1 enhances T-cell reinvigoration. | Relatlimab+Nivolumab | Approved for advanced melanoma. | First approved LAG-3 inhibitor; provides a new combination strategy to overcome PD-1 resistance [30] |
| TIGIT | Inhibitory receptor on T/NK cells; blockade may enhance anti-tumor immunity. | Tiragolumab+Atezolizumab | Phase III trials in NSCLC (mixed outcomes). | A promising but complex target; highlights need for biomarker-driven selection and combination strategies [31] |
| TME-Targeting | Counteracts immunosuppressive factors in the tumor microenvironment. | Anti-CD73, Anti-TGF-β, CSF-1R inhibitors | Mostly in preclinical/early clinical trials. | Critical for overcoming resistance to existing immunotherapies; represents the next frontier in immuno-oncology [32] |
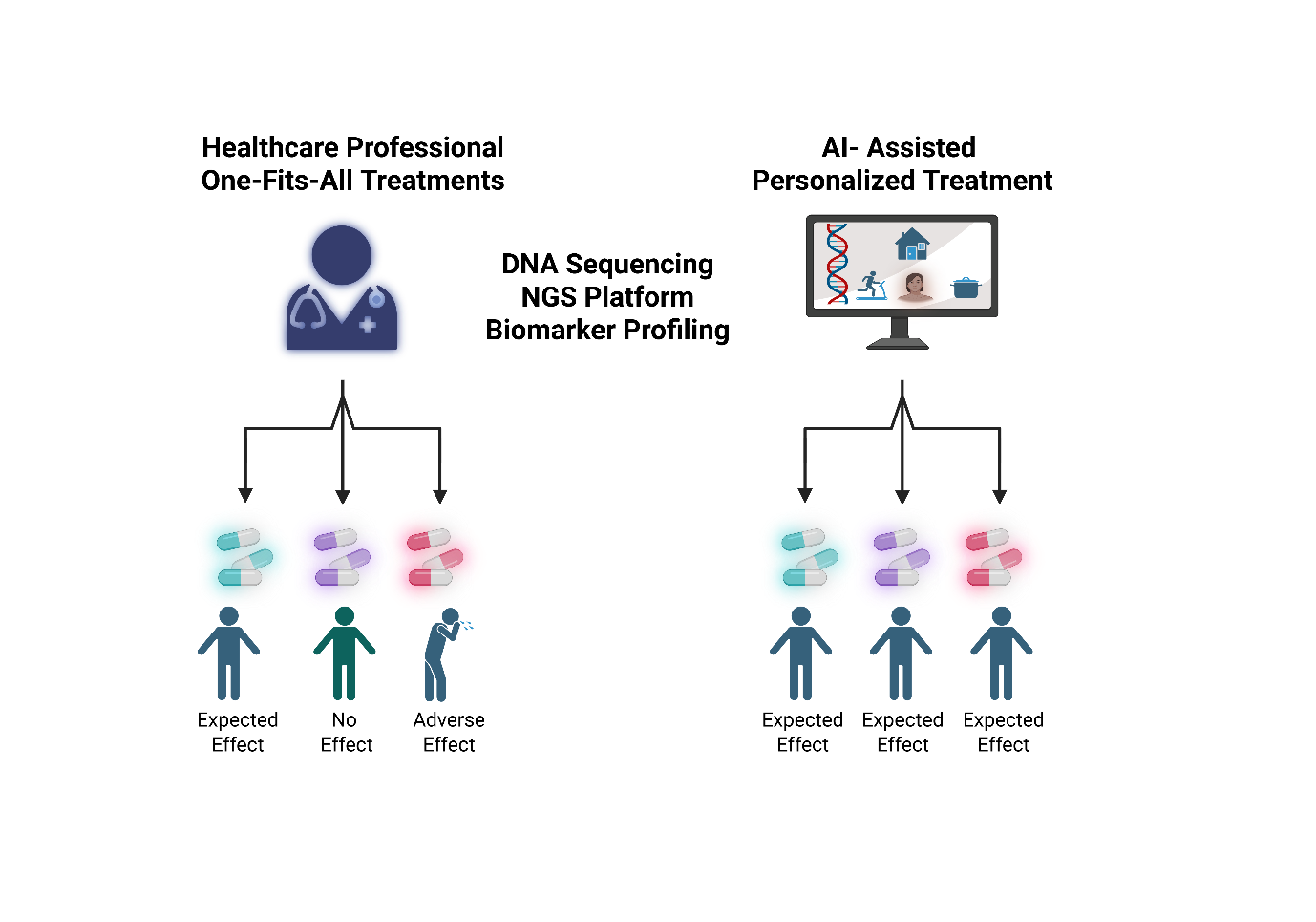
Fig. 4: The main difference between one-fits-all treatment vs AI-driven precision medicine for the benefit of human beings
AI in precision cancer
Novel medicine has stepped into precision, prevention and personalization rather than developing treatment for the whole population and making the same medical decisions based on a few physical traits. Artificial Intelligence (AI) is a pivotal technology that brought this technique to everyday practice and cultural transformation, as shown in fig. 4 [33]. By using AI, healthcare professionals have come to a result that can detect diseases like cancer or cardiovascular disorders by producing more accurate diagnosis methods. Analytics and insights, shared expertise, data and security are the three main elements for the implementation of artificial intelligence in healthcare [34]. Adoption of AI in the real world, its ability to function accurately in a reliable, safe and generalizable manner is critical as its accuracy [35]. Deep neural networks (DNNs) excel in classifying large medical images, histopathology images and achieving high AUC scores in controlled research settings. However, the real-world clinical application of these models faces challenges, including potential biases in training datasets and current regulatory hurdles that must be addressed for broader implementation. AI is also advancing in early cancer detection via liquid biopsies. Furthermore, AI is playing an increasingly pivotal role in drug discovery by identifying novel therapeutic targets and optimizing clinical trial designs for precision oncology therapies [36].
Table 4: Comparative analysis of regulatory frameworks and emerging challenges in precision medicine
| Aspect | United states (US) | European union (EU) | Japan |
| Expedited Programs | Fast Track, Breakthrough Therapy, Accelerated Approval, Regenerative Medicine Advanced Therapy (RMAT) | PRIME (Priority Medicines), Conditional Marketing Authorization | Sakigake Designation, Conditional Early Approval System |
| Approval Mechanism | Accelerated Approval based on surrogate endpoints; requires confirmatory trials. | Conditional Approval with post-marketing obligations; requires confirmatory data. | Conditional, time-limited approval with reassessment. |
| Key Challenge: Confirmatory Evidence | Risk of withdrawal if post-market trials fail (e. g., several oncology indications have been withdrawn). | Similar risk: mandatory post-authorization efficacy studies must confirm benefit to maintain conditional approval. | Re-assessment required; product may be withdrawn if efficacy not confirmed within the designated review period. |
| Key Challenge: Diagnostics and IVDR | Evolving oversight of Laboratory Developed Tests (LDTs) creates regulatory uncertainty. | IVDR (2017/746) significantly increases evidence requirements for CDx, threatening test availability and viability. | PMDA oversight is established, but the impact of IVDR equivalence on market strategy for global companies is a key consideration. |
| Key Challenge: Test Standardization | Discordance rates between different CDx assays (e. g., IHC vs. NGS) can complicate treatment decisions. | Lack of harmonization across member states and between notified bodies can lead to variable test performance. | Efforts towards Comprehensive Genomic Profiling (CGP) aim to standardize, but platform-specific differences persist. |
| Focus Area | Tumor-agnostic therapies, Real-World Data (RWD) utilization, evolving LDT regulation. | Biomarker-driven therapies, omics, cross-border genomic analysis, adapting to IVDR implementation. | Advanced genomic diagnostics, Real-World Evidence (RWE) and innovative biomarker integration. |
Global regulatory authorities in the US, EU and Japan have introduced accelerated pathways to expedite the approval of precision oncology therapies and companion diagnostics. Despite these efforts, practical challenges remain. A key issue is the withdrawal of approvals when post-marketing confirmatory trials fail to demonstrate clinical benefit, emphasizing the risks of expedited approvals and the need for stronger post-market surveillance [37]. The European Union’s in vitro Diagnostic Regulation (IVDR) further intensifies scrutiny and evidence requirements for diagnostics, aiming to improve safety and clinical validity. However, its stringent standards may delay market entry, limit access to essential biomarker tests and create bottlenecks for new targeted therapies [38].
Additional challenges include assay discordance and regulatory ambiguity around Laboratory Developed Tests (LDTs). Variability between testing platforms, such as next-generation sequencing (NGS) and immunohistochemistry (IHC)can lead to inconsistent biomarker results, complicating clinical decisions and underscoring the need for standardized testing guidelines [39]. Meanwhile, evolving FDA oversight of LDTs introduces uncertainty for laboratories and clinicians relying on these tests for emerging biomarkers lacking approved companion diagnostics, potentially slowing innovation and clinical translation [40].
Innovation and collaboration
The theory of basket trials is that a diagnosis based on tumor cell modification will be beneficial, independent of the organ where the disease develops. This increases the trial’s ability to register participants depending on cancer cell mutation and tumor kinds. Basket trials have gained significant regulatory attention, particularly from agencies like the FDA and EMA, adapting frameworks to evaluate these histology-agnostic designs through initiatives like master protocols, which streamline the assessment of multiple therapies or commitment to advancing precision cancer medicine. As opposed to basket trials, which are histology-agnostic designs, umbrella trials try to pair patients with the same tumor subtype (lung adenocarcinoma) with various targeted treatments; these patients are usually selected based on the biomarker that the treatments are targeting [41]. Real-World Data (RWD) serves as the foundation for Real World Evidence (RWE) and can be obtained from a variety of sources, including patient registries, Healthcare databases, claims databases, patient networks and wearable-generated data, as well as electronic health records (EHRs). Both RWD and RWE are major keys for regulatory agencies, while RWE obtained from observational studies is increasingly accepted for post-approval safety monitoring and provides a response to pharmacoeconomic questions. RWE provides sufficient proof for regulatory 22, particularly in precision oncology, where traditional randomised controlled trials (RCTs) may be infeasible due to a small patient population with aspecific genetic profile [42]. An approach for creating plans to quickly progress therapeutic advances in cancer research is the use of public-private partnership (PPP) which should be for the benefit of the public, especially if public funds are involved and should tackle an unmet need or scientific objective that neither party can handle on their own or that is better served by working together. PPP helps to enlarge the total investment in cancer research by promoting the merging of resources, reducing financial burdens and mitigating risk for patientsthrough shared expertise and infrastructure. Initiatives like the Cancer Moonshot and the Innovative Medicine initiative illustrate the power of collaboration in advancing precision oncology and improving cancer care [43].
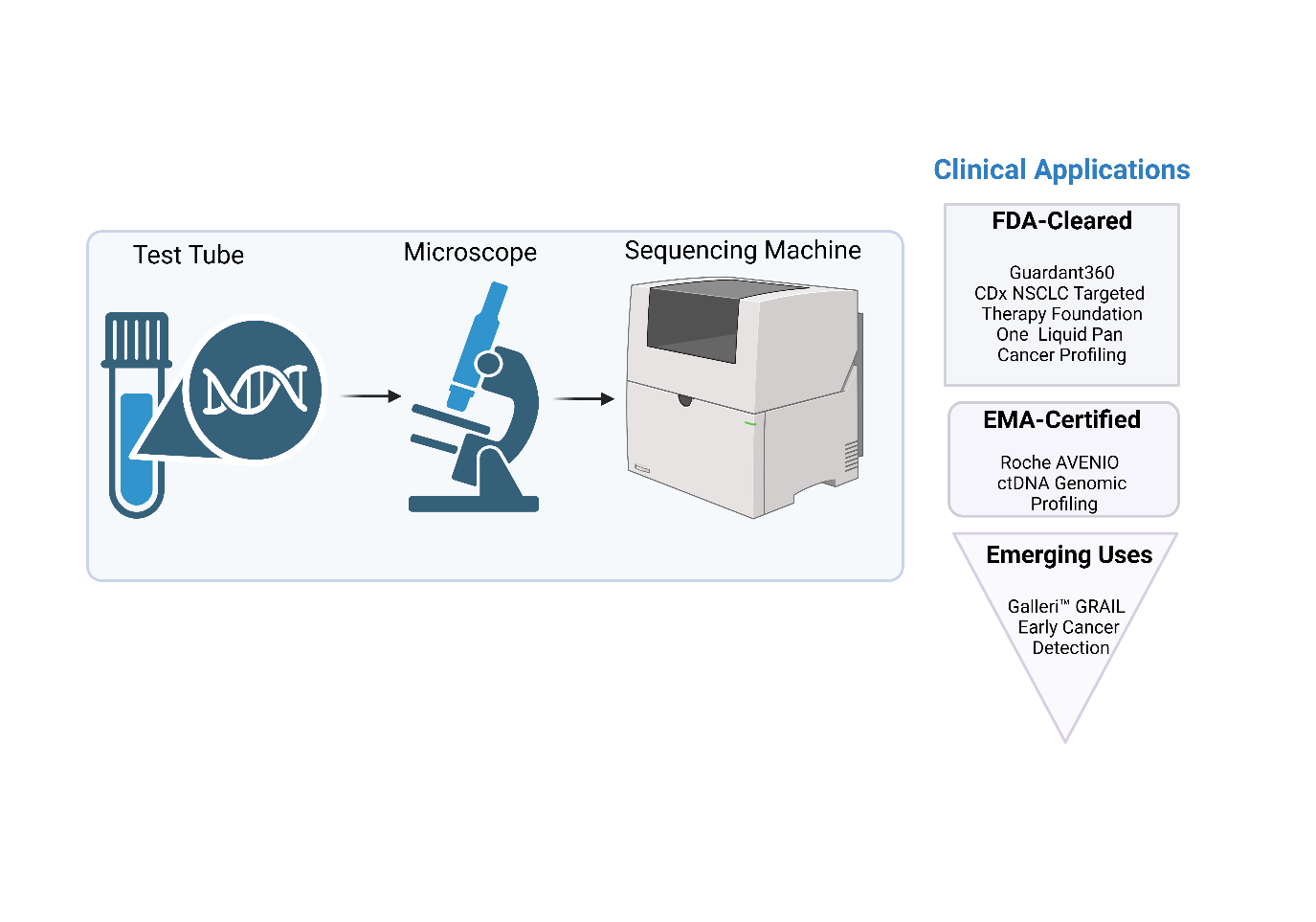
Fig. 5: Clinical applications of liquid biopsy with regulatory touch points
Table 5: Approved precision oncology therapies and companion diagnostics
| Drug name | Target/Biomarker | Companion diagnostic (CDx) | Regulatory body and approval year | Indication | Notes |
| Trastuzumab (Herceptin) | HER2 overexpression | HER2 IHC/FISH Test | FDA (1998); EMA (2000) | HER2-positive breast cancer | One of the first targeted therapies; CDx co-approval set a precedent for precision oncology. |
| Imatinib (Gleevec) | BCR-ABL fusion | Philadelphia chromosome test | FDA (2001); EMA (2001) | Chronic Myeloid Leukemia (CML) | Pioneered targeted therapy for specific genetic mutations. |
| Pembrolizumab (Keytruda) | PD-L1 expression/MSI-H/dMMR | PD-L1 IHC Test/MSI-H Test | FDA (2017-tissue-agnostic for MSI-H); EMA (2018) | Multiple cancers (e. g., NSCLC, colorectal) | First tissue-agnostic approval based on biomarker, not tumor location. |
| Larotrectinib (Vitrakvi) | NTRK gene fusion | NGS-based tests for NTRK fusion | FDA (2018); EMA (2019) | Solid tumors with NTRK fusion | Tissue-agnostic approval; highlights importance of NGS in precision diagnostics. |
| Olaparib (Lynparza) | BRCA1/2 mutations | BRCA mutation test (e. g., Myriad BRACAnalysisCDx) | FDA (2014 for ovarian); EMA (2014) | Ovarian, breast, prostate cancers | Emphasizes role of genetic testing in identifying eligible patients. |
Targeted therapy inhibits specific molecular pathways that are crucial for cancer growth and maintenance [15], thereby increasing the lifespan of cancer patients by offering more precise interventions. While these therapies often avoid the broad cytotoxic side effects associated with traditional chemotherapy (such as severe neutropenia and alopecia), they are associated with a distinct and often significant profile of mechanism-based toxicities. The promise of fewer side effects requires qualification; for instance, kinase inhibitors frequently cause high rates of dermatological (e. g., rash, hand-foot syndrome), gastrointestinal (e. g., diarrhea) metabolic (e. g., hyperglycemia, hyperlipidemia) adverse events. Severe (Grade ≥3) adverse events are not uncommon, as illustrated in table 6.
Furthermore, the application of targeted therapies is limited by the prevalence of actionable biomarkers. A minority of patients with a specific cancer type often harbor the required mutation and access is further constrained by the availability and reimbursement of companion diagnostics. For example, while EGFR mutations are a cornerstone of lung cancer treatment, they are present in only approximately 10-15% of Western patients with non-small cell lung cancer (NSCLC) [A2]. This highlights that targeted therapies, despite their efficacy in eligible subpopulations, are not a universally applicable solution.
Table 6: Spectrum of severe toxicities and patient eligibility for select targeted therapies
| Drug class/Example | Primary target | Common severe (Grade ≥3) adverse events (>10% unless noted) | Approximate biomarker eligibility in relevant cancer type |
| EGFR Inhibitors (e. g., Osimertinib) | EGFR | Diarrhea (5%), Rash (5%), Interstitial Lung Disease (3%) | ~15% of unselected NSCLC in North America/Europe (EGFR mutations) [44] |
| BRAF/MEK Inhibitors (e. g., Dabrafenib+Trametinib) | BRAF V600E | Pyrexia (6%), Fatigue (5%), Elevated Liver Enzymes (ALT/AST 11%) | ~40% of Cutaneous Melanoma (BRAF V600 mutations) [45] |
| KRAS G12C Inhibitors (e. g., Sotorasib) | KRAS G12C | Hepatotoxicity (ALT increase 15%), Diarrhea (10%), Nausea (2%) | ~13% of Lung Adenocarcinomas (KRAS G12C mutation) [46] |
| CDK4/6 Inhibitors (e. g., Palbociclib) | CDK4/6 | Neutropenia (66%), Leukopenia (31%) | ~70% of Metastatic Breast Cancer (HR+/HER2-phenotype) [47] |
| BTK Inhibitors (e. g., Ibrutinib) | BTK | Hypertension (14%), Atrial Fibrillation (6%), Bleeding (2%) | Indicated for specific B-cell malignancies (e. g., CLL, MCL) [48] |
Economic and access considerations
Despite the transformative potential of precision oncology, its widespread adoption faces significant economic and accessibility hurdles. The development of therapies for small, biomarker-defined populations often results in high per-patient costs, creating challenges for healthcare reimbursement systems. Health technology assessment bodies frequently evaluate these therapies using cost-per-QALY thresholds and many precision oncology drugs exceed accepted benchmarks, leading to restricted coverage and delayed patient access.
Furthermore, equitable access to these advances is not guaranteed. A genomic divide exists, with disparities observed across geographic, socioeconomic and racial lines. Patients in low-resource settings and from underrepresented minority groups are less likely to receive genomic testing or be enrolled in the clinical trials that are the gateway to novel therapies. The underrepresentation of diverse populations in genomic databases also risks perpetuating biases in biomarker discovery and AI algorithm development. Addressing these issues of cost-effectiveness and health equity through innovative funding models, policy interventions and inclusive research practices is essential to ensure that the promise of precision medicine reaches all patients in need.
CONCLUSION
Precision medicine is redefining the landscape of cancer care by enabling treatments that are tailored to the unique genetic and molecular profiles of individual patients. Innovations such as next-generation sequencing, liquid biopsies and targeted therapies are not only improving diagnostic accuracy but also enhancing therapeutic outcomes. While challenges such as high costs, data complexity and regulatory constraints persist, the collaborative efforts of global regulatory agencies, healthcare providers and researchers are accelerating the safe and effective integration of precision approaches into clinical practice. As artificial intelligence, real-world evidence and integrative omics technologies continue to advance, the future of oncology is poised to become increasingly personalized, leading to more effective, efficient and patient-centric care.
ACKNOWLEDGEMENT
The authors would like to thank the Department of Science and Technology-Fund for Improvement of Science and Technology Infrastructure (DST-FIST) and Promotion of University Research and Scientific Excellence (DST-PURSE) and Department of Bio Technology-Boost to University Interdisciplinary Life Science Departments for Education and Research program (DBT-BUILDER) for the facilities provided in our department. The authors also extend their gratitude to Yenepoya (Deemed to be University), Mangalore, for providing the necessary infrastructure and support through the Centre for Integrative Omics Data Science.
FUNDING
No external funding was received for this study.
AUTHORS CONTRIBUTIONS
Karunakaran designed, performed, analysed the data, and wrote the manuscript and revised it. vivek supervised the data collected and reviewed. Inamul helped in reviewing, data compilation, designing framework of article and manuscript components. Suhaib performed data analysis.
CONFLICT OF INTERESTS
All authors declare that there is no conflict of interest.
REFERENCES
Gameiro GR, Sinkunas V, Liguori GR, Auler Junior JO. Precision medicine: changing the way we think about healthcare. Clinics (Sao Paulo). 2018;73:e723. doi: 10.6061/clinics/2017/e723, PMID 30517307.
Pettitt D, Smith J, Meadows N, Arshad Z, Schuh A, Di Giusto D. Regulatory barriers to the advancement of precision medicine. Expert Rev Precis Med Drug Dev. 2016 May 3;1(3):319-29. doi: 10.1080/23808993.2016.1176526.
Chude Okonkwo UK, Paul BS, Vasilakos AV. Enabling precision medicine via contemporary and future communication technologies: a survey. IEEE Access. 2023;11:21210-40. doi: 10.1109/ACCESS.2022.3175573.
Tang X, Berger MF, Solit DB. Precision oncology: current and future platforms for treatment selection. Trends Cancer. 2024;10(9):781-91. doi: 10.1016/j.trecan.2024.06.009, PMID 39030146.
MG, SG, Rajagopal S, Angamuthu G, Dhandapani NV. Revolutionizing drug development: how ai and machine learning are shaping the future of medicine-a review. Int J Appl Pharm. 2025 May 7;17(3):148-56. doi: 10.22159/ijap.2025v17i3.53457.
Stewart BW, Bray F, Forman D, Ohgaki H, Straif K, Ullrich A. Cancer prevention as part of precision medicine: ‘plenty to be done’. Carcinogenesis. 2016;37(1):2-9. doi: 10.1093/carcin/bgv166, PMID 26590901.
Gonzalez Angulo AM, Hennessy BT, Mills GB. Future of personalized medicine in oncology: a systems biology approach. J Clin Oncol. 2010 Jun 1;28(16):2777-83. doi: 10.1200/JCO.2009.27.0777, PMID 20406928.
Mishra N, Dasari A. Artificial intelligence and 3D printing in pharmaceuticals: a new frontier in personalized drug manufacturing. Int J Pharm Pharm Sci. 2025 Jan 1;7(2):15-22. doi: 10.33545/26647222.2025.v7.i2a.188.
Sharma S, Chaubey A, Pathan MN, Tyagi S. AI-powered virtual screening for drug discovery: methods and challenges. Int J Pharm Pharm Sci. 2024;6(2):157-64. doi: 10.33545/26647222.2024.v6.i2b.136.
Tran B, Dancey JE, Kamel Reid S, Mc Pherson JD, Bedard PL, Brown AM. Cancer genomics: technology discovery and translation. J Clin Oncol. 2012 Feb 20;30(6):647-60. doi: 10.1200/JCO.2011.39.2316, PMID 22271477.
Wakai T, Prasoon P, Hirose Y, Shimada Y, Ichikawa H, Nagahashi M. Next-generation sequencing-based clinical sequencing: toward precision medicine in solid tumors. Int J Clin Oncol. 2019 Feb;24(2):115-22. doi: 10.1007/s10147-018-1375-3, PMID 30515675.
Fernandez Lazaro D, Garcia Hernandez JL, Garcia AC, Cordova Martinez A, Mielgo Ayuso J, Cruz Hernandez JJ. Liquid biopsy as novel tool in precision medicine: origins, properties identification and clinical perspective of cancer’s biomarkers. Diagnostics (Basel). 2020 Apr;10(4):215. doi: 10.3390/diagnostics10040215, PMID 32294884.
Desai A, Reddy NK, Subbiah V. Top advances of the year: precision oncology. Cancer. 2023;129(11):1634-42. doi: 10.1002/cncr.34743, PMID 36946766.
Chen M, Zhao H. Next-generation sequencing in liquid biopsy: cancer screening and early detection. Hum Genomics. 2019 Aug 1;13(1):34. doi: 10.1186/s40246-019-0220-8, PMID 31370908.
Frampton GM, Fichtenholtz A, Otto GA, Wang K, Downing SR, He J. Development and validation of a clinical cancer genomic profiling test based on massively parallel DNA sequencing. Nat Biotechnol. 2013 Nov;31(11):1023-31. doi: 10.1038/nbt.2696, PMID 24142049.
Olivier M, Asmis R, Hawkins GA, Howard TD, Cox LA. The need for multi-omics biomarker signatures in precision medicine. Int J Mol Sci. 2019;20(19):4781. doi: 10.3390/ijms20194781, PMID 31561483.
Vidwans SJ, Turski ML, Janku F, Garrido Laguna I, Munoz J, Schwab R. A framework for genomic biomarker actionability and its use in clinical decision making. Oncoscience. 2014 Oct 22;1(10):614-23. doi: 10.18632/oncoscience.90, PMID 25593991.
Mankoff DA, Edmonds CE, Farwell MD, Pryma DA. Development of companion diagnostics. Semin Nucl Med. 2016 Jan 1;46(1):47-56. doi: 10.1053/j.semnuclmed.2015.09.002, PMID 26687857.
Huang CC, Du M, Wang L. Bioinformatics analysis for circulating cell-free DNA in cancer. Cancers. 2019 Jun 11;11(6):805. doi: 10.3390/cancers11060805, PMID 31212602.
Rusnakova DS, Aziri R, Dubovan P, Jurik M, Mego M, Pin Ak D. Detection significance and potential utility of circulating tumor cells in clinical practice in breast cancer. Oncol Lett. 2024 Oct 17;29(1):10. doi: 10.3892/ol.2024.14756, PMID 39492933.
Hasegawa N, Kohsaka S, Kurokawa K, Shinno Y, Takeda Nakamura I, Ueno T. Highly sensitive fusion detection using plasma cell-free RNA in non‐small-cell lung cancers. Cancer Sci. 2021 Oct;112(10):4393-403. doi: 10.1111/cas.15084, PMID 34310819.
Coughlan C, Bruce KD, Burgy O, Boyd TD, Michel CR, Garcia Perez JE. Exosome isolation by ultracentrifugation and precipitation and techniques for downstream analyses. Curr Protoc Cell Biol. 2020 Sep;88(1):e110. doi: 10.1002/cpcb.110, PMID 32633898.
Lopes AG, Noel R, Sinclair A. Cost analysis of vein-to-vein CAR T-cell therapy: automated manufacturing and supply chain. Cell Gene Therapy Insights. 2020 Jun 2;6(3):487-510. doi: 10.18609/cgti.2020.058.
Stegmeier F, Warmuth M, Sellers WR, Dorsch M. Targeted cancer therapies in the twenty-first century: lessons from imatinib. Clin Pharmacol Ther. 2010;87(5):543-52. doi: 10.1038/clpt.2009.297, PMID 20237469.
June CH, O Connor RS, Kawalekar OU, Ghassemi S, Milone MC. CAR T cell immunotherapy for human cancer. Science. 2018 Mar 23;359(6382):1361-5. doi: 10.1126/science.aar6711, PMID 29567707.
Barbier L, Declerck P, Simoens S, Neven P, Vulto AG, Huys I. The arrival of biosimilar monoclonal antibodies in oncology: clinical studies for trastuzumab biosimilars. Br J Cancer. 2019 Jul;121(3):199-210. doi: 10.1038/s41416-019-0480-z, PMID 31257362.
Kwok G, Yau TC, Chiu JW, Tse E, Kwong YL. Pembrolizumab (Keytruda). Hum Vaccin Immunother. 2016 Nov 1;12(11):2777-89. doi: 10.1080/21645515.2016.1199310, PMID 27398650.
Tang Q, Chen Y, Li X, Long S, Shi Y, Yu Y. The role of PD-1/PD-L1 and application of immune-checkpoint inhibitors in human cancers. Front Immunol. 2022 Sep 13;13:964442. doi: 10.3389/fimmu.2022.964442, PMID 36177034.
Buchbinder EI, Desai A. CTLA-4 and PD-1 pathways: similarities, differences and implications of their inhibition. Am J Clin Oncol. 2016 Feb;39(1):98-106. doi: 10.1097/COC.0000000000000239, PMID 26558876.
Cillo AR, Cardello C, Shan F, Karapetyan L, Kunning S, Sander C. Blockade of LAG-3 and PD-1 leads to co-expression of cytotoxic and exhaustion gene modules in CD8+ T cells to promote antitumor immunity. Cell. 2024 Aug;187(16):4373-4388.e15. doi: 10.1016/j.cell.2024.06.036, PMID 39121849.
Ghasemi K. Tiragolumab and TIGIT: pioneering the next era of cancer immunotherapy. Front Pharmacol. 2025 Jun 11;16:1568664. doi: 10.3389/fphar.2025.1568664, PMID 40567374.
Fang H, De Clerck YA. Targeting the tumor microenvironment: from understanding pathways to effective clinical trials. Cancer Res. 2013 Aug 15;73(16):4965-77. doi: 10.1158/0008-5472.CAN-13-0661, PMID 23913938.
Mesko B. The role of artificial intelligence in precision medicine. Expert Rev Precis Med Drug Dev. 2017 Sep 3;2(5):239-41. doi: 10.1080/23808993.2017.1380516.
Alvarez Machancoses O, DeAndres Galiana EJ, Cernea A, Fernandez De La Vina J, Fernandez Martinez JL. On the role of artificial intelligence in genomics to enhance precision medicine. Pharmgenomics Pers Med. 2020 Mar 19;13:105-19. doi: 10.2147/PGPM.S205082, PMID 32256101.
Johnson KB, Wei WQ, Weeraratne D, Frisse ME, Misulis K, Rhee K. Precision medicine AI and the future of personalized health care. Clin Transl Sci. 2021 Jan;14(1):86-93. doi: 10.1111/cts.12884, PMID 32961010.
Bhinder B, Gilvary C, Madhukar NS, Elemento O. Artificial intelligence in cancer research and precision medicine. Cancer Discov. 2021;11(4):900-15. doi: 10.1158/2159-8290.CD-21-0090, PMID 33811123.
Beaver JA, Howie LJ, Pelosof L, Kim T, Liu J, Goldberg KB. A 25 y experience of US food and drug administration accelerated approval of malignant hematology and oncology drugs and biologics: a review. JAMA Oncol. 2018 Jun 1;4(6):849-56. doi: 10.1001/jamaoncol.2017.5618, PMID 29494733.
Lubbers BR, Schilhabel A, Cobbaert CM, Gonzalez D, Dombrink I, Bruggemann M. The new EU regulation on in vitro diagnostic medical devices: implications and preparatory actions for diagnostic laboratories. Hemasphere. 2021 May;5(5):e568. doi: 10.1097/HS9.0000000000000568, PMID 33898932.
Kalemkerian GP, Narula N, Kennedy EB, Biermann WA, Donington J, Leighl NB. Molecular testing guideline for the selection of patients with lung cancer for treatment with targeted tyrosine kinase inhibitors: American society of clinical oncology endorsement of the college of American pathologists/international association for the study of lung cancer/association for molecular pathology clinical practice guideline update. J Clin Oncol. 2018 Mar 20;36(9):911-9. doi: 10.1200/JCO.2017.76.7293, PMID 29401004.
Spitzenberger F, Patel J, Gebuhr I, Kruttwig K, Safi A, Meisel C. Laboratory-developed tests: design of a regulatory strategy in compliance with the international state-of-the-art and the regulation (EU) 2017/746 (EU IVDR in vitro diagnostic medical device regulation). Ther Innov Regul Sci. 2022 Jan;56(1):47-64. doi: 10.1007/s43441-021-00323-7, PMID 34291407.
Tsimberidou AM, Muller P, Ji Y. Innovative trial design in precision oncology. Semin Cancer Biol. 2022 Sep 1;84:284-92. doi: 10.1016/j.semcancer.2020.09.006, PMID 33022355.
Baumfeld Andre E, Reynolds R, Caubel P, Azoulay L, Dreyer NA. Trial designs using real-world data: the changing landscape of the regulatory approval process. Pharmacoepidemiol Drug Saf. 2020 Oct;29(10):1201-12. doi: 10.1002/pds.4932, PMID 31823482.
Herbst RS, Blumenthal G, Khleif SN, Lippman SM, Meropol NJ, Rosati K. Optimizing public-private partnerships to support clinical cancer research. J Natl Cancer Inst. 2025;117(7):1305-10. doi: 10.1093/jnci/djae279, PMID 39666955.
Soo RA, Reungwetwattana T, Perroud HA, Batra U, Kilickap S, Tejado Gallegos LF. Prevalence of EGFR mutations in patients with resected stages I to III NSCLC: results from the EARLY-EGFR study. J Thorac Oncol. 2024 Oct;19(10):1449-59. doi: 10.1016/j.jtho.2024.06.008, PMID 38880172.
Daud A, Tsai K. Management of treatment related adverse events with agents targeting the MAPK pathway in patients with metastatic melanoma. Oncologist. 2017 Jul 1;22(7):823-33. doi: 10.1634/theoncologist.2016-0456, PMID 28526719.
Arbour KC, Riely GJ. Systemic therapy for locally advanced and metastatic non–small cell lung cancer: a review. JAMA. 2019 Aug 27;322(8):764-74. doi: 10.1001/jama.2019.11058, PMID 31454018.
Yang L, Xue J, Yang Z, Wang M, Yang P, Dong Y. Side effects of CDK4/6 inhibitors in the treatment of HR+/HER2- advanced breast cancer: a systematic review and meta-analysis of randomized controlled trials. Ann Palliat Med. 2021 May;10(5):5590-9. doi: 10.21037/apm-21-1096, PMID 34107710.
Pal Singh S, Dammeijer F, Hendriks RW. Role of Bruton’s tyrosine kinase in B cells and malignancies. Mol Cancer. 2018 Dec;17(1):57. doi: 10.1186/s12943-018-0779-z, PMID 29455639.