
Int J App Pharm, Vol 18, Issue 1, 2026, 119-129Reviewl Article
ORAL TRANSMUCOSAL DELIVERY: AN IN-DEPTH REVIEW OF BUCCAL PATCH FABRICATION, INNOVATIONS AND APPLICATIONS
KAMPANART HUANBUTTA1, TANUJ KUMAR2, SANDEEP KUMAR2, VANDANA KHARB3, TANIKAN SANGNIM4, KITTIPAT SUWANPITAK4, INDERBIR SINGH5*
1Department of Manufacturing Pharmacy, College of Pharmacy, Rangsit University, Pathum Thani, Thailand. 2Department of Pharmaceutics, Amar Shaheed Baba Ajit Singh Jujhar Singh Memorial College of Pharmacy, Bela, Rupnagar, India. 3Department of Pharmaceutics, Sachdeva College of Pharmacy, Gharuan, Mohali, India. 4Faculty of Pharmaceutical Sciences, Burapha University, 169, Seansook, Muang, Chonburi, Thailand. 5Chitkara College of Pharmacy, Chitkara University, Punjab, India
*Corresponding author: Inderbir Singh; *Email: inderbir.singh@chitkara.edu.in
Received: 23 Jul 2025, Revised and Accepted: 30 Oct 2025
ABSTRACT
Mucoadhesive buccal patches have attracted considerable interest over the last decade because of their potential as a novel drug delivery system (NDDS) for several therapeutic uses. Mucoadhesive buccal patches stick to the buccal mucosa, ensuring controlled and prolonged release of drugs, thereby largely avoiding first-pass metabolism and enhancing drug bioavailability. The present paper summarizes a thorough collection of mucoadhesive buccal patches with special emphasis on design, formulation, processing, and application in drug delivery. In addition, this article emphasizes new patent research dealing with mucoadhesive buccal patches. It provides an overview of the existing patents and discusses their novelty, usefulness, and potentiality for commercialization. The all-encompassing analysis contained within this article serves to provide as an informative tool for researchers, pharmaceutical scientists, and entrepreneurs interested in the development of mucoadhesive buccal patches as a potential drug delivery system. It also stimulates future research and innovation in this field, with potential for future advancement.
Keywords: Mucoadhesive, Buccal mucosa, Drug delivery, Penetration enhancers, Patents
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.56224 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
The oral route is the most commonly preferred method of drug administration among both patients and healthcare professionals. However, it is unsuitable for certain pharmaceutical categories, particularly peptides and proteins, due to enzymatic degradation in the gastrointestinal tract and extensive first-pass metabolism in the liver [1]. Recently, there has been a notable increase in research focused on targeted drug delivery to specific anatomical sites, aiming to optimize therapeutic availability and reduce dose-dependent side effects [2]. Consequently, various absorptive mucosal surfaces have emerged as promising sites for drug administration. For systemic drug delivery, the transmucosal route, which includes the mucosal linings of the oral, nasal, rectal, vaginal, and ocular cavities, offers several advantages over the peroral route. These include bypassing presystemic clearance (first-pass effect), and, depending on the drug, providing an enzymatic environment more favorable for absorption [3]. Mucoadhesion has gained attention for its ability to maintain the formulation at the absorption site, such as the buccal cavity, or to localize drug release at specific targets within the gastrointestinal tract [4, 5].
Among various mucosal routes, the buccal mucosa stands out due to its excellent accessibility, relatively low mobility, and favorable anatomical characteristics, making it an ideal site for controlled drug release from various dosage forms. Oral mucosal drug delivery enables both rapid and sustained drug release, while circumventing limitations such as enzymatic degradation and hepatic first-pass metabolism. This route supports both local and systemic drug action and offers greater convenience compared to non-oral alternatives. The ease of self-administration and removal of buccal patches further improve patient compliance. Moreover, patients can control treatment duration and discontinue use in the event of discomfort or emergency [6].
Among various mucosal routes, the buccal mucosa is particularly advantageous due to its accessibility, low mobility, and favorable anatomical features, making it suitable for controlled drug release. Oral mucosal delivery allows both rapid and sustained release while minimizing enzymatic degradation and partially reducing first-pass metabolism. However, the claim that buccal delivery completely “bypasses” first-pass metabolism is an oversimplification, as absorption is highly drug-dependent and influenced by physicochemical properties, residence time, and the extent of swallowing, which can still subject part of the dose to hepatic clearance [7]. This route supports both local and systemic drug action, while offering convenience, ease of self-administration, and improved patient compliance [8], as evidenced by patient-reported outcomes. Moreover, buccal patches allow patients to control treatment duration and discontinue use if discomfort or emergencies arise.
The richly vascularized oral mucosa facilitates efficient drug absorption into the systemic circulation via venous pathways such as the internal jugular, brachiocephalic, deep lingual, and facial veins. Buccal patches are favored for their ease of use and non-invasive application. This approach is particularly advantageous for peptide-and protein-based therapeutics, which are susceptible to enzymatic degradation. A buccal patch is a thin, modified-release dosage form composed of one or more polymeric layers containing the active drug and suitable excipients. It is not designed to dissolve during administration but instead incorporates a mucoadhesive polymer that adheres to the oral mucosa, enabling controlled drug release. After the prescribed duration, the patch is removed and appropriately discarded [4, 9].
This review was compiled by conducting a comprehensive literature search of scientific databases, including PubMed, Scopus, and Google Scholar, for the period of 2000–2024. The search utilized keywords such as “buccal patch,” “mucoadhesive film,” “oral transmucosal delivery,” “fabrication techniques,” “3D printing,” and “commercialization.” Articles published in English were included, while non-peer-reviewed articles and conference abstracts were excluded to ensure the quality and reliability of the cited literature.
Design and composition of buccal patches
The formulation and design of buccal patches are predicated on the precise combination of several constituent excipients and structural elements. The fundamental components include the active pharmaceutical ingredient (API), mucoadhesive polymers for mucosal adhesion, a protective backing membrane, penetration enhancers to improve bioavailability, and plasticizers to ensure mechanical flexibility. In terms of design, buccal patches are predominantly engineered as either matrix-type or reservoir-type systems. A comprehensive elucidation of each component and structural design is presented in the subsequent sections.
Structural design of buccal patches
Matrix type (Bi-directional)
Matrix-type Buccal Patches are characterized by a simple design where the active pharmaceutical ingredient (API), mucoadhesive polymers, and other additives are uniformly mixed into a single, homogeneous layer. This construction leads to bi-directional release, where the drug diffuses simultaneously into the buccal mucosa for absorption and into the oral cavity, as shown in fig. 1a. The primary advantages of this system are its simplicity and low cost of manufacturing. However, its main drawbacks are significant drug loss into the saliva, which reduces overall efficiency and bioavailability, and a lack of precise control over the drug release rate, which tends to decrease over time [10, 11].
Reservoir type (Uni-directional)
Reservoir type buccal patches feature a more complex, multi-layered structure designed for targeted delivery. This system contains the drug in a distinct reservoir, which is separated from the mucosa by a rate-controlling membrane and protected by an impermeable backing layer. This design facilitates uni-directional release, forcing the drug exclusively toward the cheek tissue as shown in fig. 1b. The main advantages include highly efficient and precisely controlled drug delivery, leading to more predictable therapeutic effects. The primary disadvantages are the increased complexity and cost of manufacturing, as well as the potential risk of "dose dumping," a rapid, unintended release of the entire drug content if the rate-controlling membrane is damaged [11].
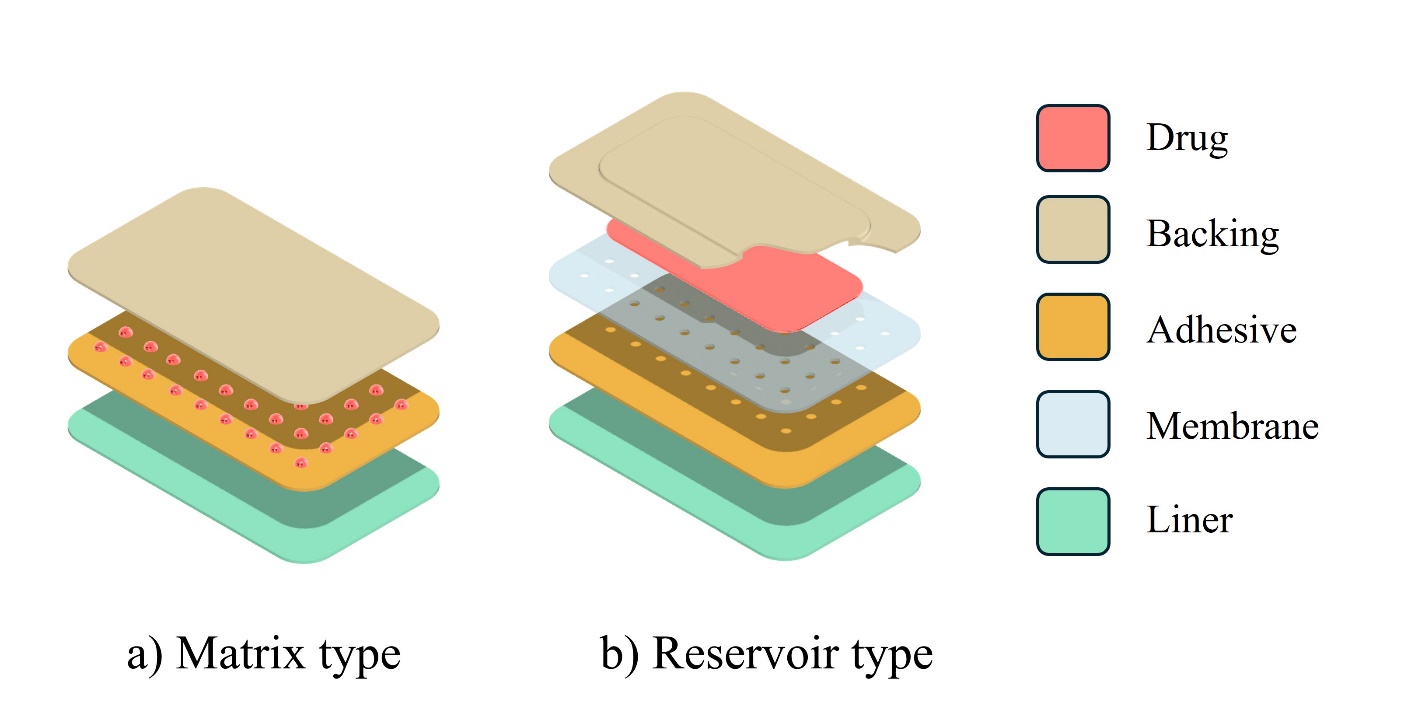
Fig. 1: Types of buccal patches (a) matrix type and (b) reservoir type
Composition of a buccal patch
Active pharmaceutical ingredient
In buccoadhesive drug delivery systems, achieving the desired therapeutic effect hinges on the drug maintaining prolonged contact with the oral mucosa. A drug’s ability to diffuse through the buccal mucosa and the patch matrix is significantly influenced by its physicochemical properties, including molecular weight, chemical stability, and melting point [12]. For a drug to be considered suitable for this delivery method, it must meet several essential criteria. The drug should be effective at a low dose and be absorbed passively through the oral mucosa, without the need for active transport mechanisms. This delivery route is often chosen for medications that experience significant first-pass metabolism or presystemic clearance when taken orally. Crucially, the active ingredient must not cause irritation or disrupt the natural microbial flora of the oral cavity [13].
Mucoadhesive polymers
Recent research has increasingly focused on the use of mucoadhesive materials in oral drug delivery, as the ability of a formulation to adhere to the buccal mucosa is critical for ensuring therapeutic efficacy. Mucoadhesive polymers enhance drug delivery by prolonging the residence time of the formulation at the site of absorption, thereby improving bioavailability and therapeutic outcomes [14]. An ideal mucoadhesive polymer must possess several key characteristics. The polymer and its degradation byproducts must be non-toxic and should not be absorbed through the gastrointestinal tract, and it must not cause irritation or damage to the mucosal membrane. Furthermore, the polymer should form strong non-covalent interactions with mucin on the epithelial cell surfaces and remain stable without breaking down during storage or throughout the product’s shelf life [15, 16]. Various types of mucoadhesive polymers, which are classified by source, solubility, and charge, are summarized in table 1.
Table 1: List of mucoadhesive polymers
| Criteria | Category | Examples |
| Source | Semi-Natural/Natural | Agarose, Gelatin, Hyaluronic Acid, Guar Gum [15, 17] |
| Synthetic | Cellulose Derivatives: Carboxymethyl cellulose, Sodium carboxymethyl cellulose, Hydroxyethyl cellulose, Hydroxypropyl Methyl Cellulose [13, 17] | |
| Poly (acrylic acid)-Based polymers: Polydactylies, Poly (2-hydroxyethylmethacrylate), Poly (acrylicacid-coethylhexylacrylate), Poly (methacrylate), CP, PC, PAA [17, 18] | ||
| Aqueous Solubility | Water Soluble | Cellulose Propionate, Hydroxyethyl cellulose, HPC (water<38 °C), HPMC (cold water), Sodium CMC [17, 18] |
| Water Insoluble | Sodium alginate, Chitosan, Ethyl Cellulose, Polycarbonate [13] | |
| Charge | Cationic | Sodium alginate, Chitosan, Ethyl Cellulose, Polycarbonate [13, 18] |
| Anionic | CMC, Pectin, PAA, PC, sodium alginate, Sodium CMC [17] | |
| Non-ionic | Hydroxypropyl Cellulose, Polyvinyl Alcohol, Polyvinylpyrrolidone [17, 18] |
*HPMC: hydroxypropyl methylcellulose; CP: cellulose phosphate; PC: poly(acrylic acid)-g-chitosan; PAA: polyacrylic acid; CMC: carboxymethyl cellulose.
Backing membrane
The backing membrane in buccal patches serves a critical function by directing drug release unidirectionally toward the buccal mucosa. It acts as a barrier to prevent drug diffusion into the oral cavity, thereby minimizing contact with saliva and reducing the risk of drug ingestion. An ideal backing membrane should be chemically inert, mechanically robust, and impermeable to both the drug and penetration enhancers. To maintain flexibility and provide structural support, the membrane is typically very thin, ranging from 75 to 100 µm. Common materials include polyethylene and polyester-based laminated paper, as well as polyglassine paper, multiport sheets, and cellophane-325. These materials must exhibit high flexibility, sufficient mechanical strength, and low water permeability to ensure optimal performance during application [16].
Penetration enhancers
Penetration enhancers facilitate drug transport across the mucosal barrier by interacting with structural components such as intercellular desmosomes, intracellular keratin, and epithelial lipids. These interactions transiently reduce the resistance of the stratified epithelium, thereby enhancing drug permeation. Mechanistically, they may directly interact with keratin within epithelial cells or disrupt intercellular lipids and proteins to improve drug diffusion [19]. For example, the transport of insulin nanoparticles across the buccal epithelium is enhanced by lipid rearrangement induced through electrostatic interactions between positively charged polymethacrylate and HPMC derivatives, enabling nanoparticle passage through intercellular spaces [20]. Recently, Zonula Occludens Toxin (ZOT) peptides, synthetic derivatives based on the active domain of the ZOT secreted by Vibrio cholerae, have been explored as penetration enhancers. Their efficacy stems from their ability to act as tight junction regulators, facilitating absorption by transiently and reversibly modulating and disassembling tight junctions [21]. Ionic liquids (ILs), defined as "liquid salts" formed by organic cations and anions and characterized by a melting point less than 100 °C, are utilized as novel chemical permeation enhancers to proactively augment drug permeation across human skin and mucosal systems, including dermal, nasal, corneal, buccal, and intestinal biobarriers. Their mechanism of action involves modifying the therapeutic payload by forming highly soluble API-ILs with balanced hydrophilicity/hydrophobicity, while simultaneously conditioning the biological barrier by affecting the lipid layers, perturbing their lamellar arrangement, and inducing an isotropic lipid phase [22]. Penetration enhancers are particularly valuable for macromolecules such as proteins, peptides, and hydrophilic low-molecular-weight drugs [23]. While combining penetration enhancers often enhances absorption, it’s important to note that prolonged use of these agents can potentially lead to local irritation or tissue damage [24]. Therefore, selection should consider the physicochemical properties of the active ingredient. Ideal enhancers should be non-toxic, biocompatible, non-irritating, pharmacologically inert, and organoleptically neutral (i. e., without affecting taste or odor) [25]. Table 2 summarizes the various types of penetration enhancers, their transport mechanisms, and key considerations.
Table 2: List of penetration enhancers
| Criteria | Examples | Transport mechanism |
| Surfactants [26-29] | Cationic: cetylpyridinium chloride | To make drugs dissolve better in water |
| Anionic: sodium lauryl sulfate, sodium dodecanoate | Intercellular lipid disruption and protein integrity | |
| Non-ionic: polyoxyethylene-9-lauryl ether, nonylphenoxypolyoxyethylene, polysorbates | Epithelial swelling is brought on by the hydrophobic interaction of surfactant and keratin fibrils | |
| Cyclodextrins [30, 31] | α, β, γ cyclodextrins, methylated cyclodextrins | Disruption of intracellular lipids and protein integrity |
| Polymers [32] | Trimethyl chitosan, poly-L-arginine, L-lysine, chitosan | Mucin interacts ionically with carboxyl and sulphate groups that are negatively charged |
| Fatty acids and their esters [33] |
Caprylic acid, lauric acid, linoleic acid, linolenic acid, oleic acid | Interact with the phospholipid domain to increase the fluidity of the membrane |
| Chelating agents [34, 35] | EDTA, polyacrylate, citric acid, salicylates | Complexes are created when the chelators react with Ca2+ions |
| Derived peptides [36] | Zonula occludens toxin peptides (Zot peptides) | Signaling activation, tight junction disassembly, and cytoskeletal change |
| Liquid salts [22] | Ionic liquids | Convert solid active pharmaceutical ingredients (APIs) into a fluid and barrier (skin) conditioning. |
*EDTA: Ethylenediaminetetraacetic acid
Plasticizers
Plasticizers are incorporated into polymeric films to enhance their flexibility, smoothness, and mechanical strength. Commonly used plasticizers include glycerol, propylene glycol, polyethylene glycol (PEG 200 and PEG 400), and castor oil. These agents improve the pliability of thin films, facilitating processing and handling. Additionally, plasticizers can act as penetration enhancers and promote drug release from the polymeric matrix. The choice of plasticizer depends on its ability to solvate the polymer and modify polymer–polymer interactions. When used at an optimal ratio with the polymer, plasticizers reduce molecular rigidity by increasing polymer chain mobility, resulting in improved elasticity and reduced brittleness [37].
Methods of preparation of buccal patches
Solvent casting method
The solvent casting method is a widely adopted technique for the preparation of buccal patches. In this process, water-soluble polymers and excipients are first dissolved in an aqueous medium, while the active drug and other hydrophobic components are dissolved separately in a suitable organic solvent. These two solutions are then combined and thoroughly mixed to create a homogeneous blend. This mixture is poured into a mold or casting plate and left to dry under controlled environmental conditions, resulting in the formation of a uniform film. Once the film is dried, a backing membrane is applied and the patch is further dried to complete the process. The final buccal patch is typically wrapped in aluminum foil or other protective packaging materials and stored appropriately for future use. Among its advantages, the solvent casting method allows for better uniformity in patch thickness and clarity when compared to extrusion techniques, and it produces flexible patches with enhanced physical properties, including smooth and glossy surfaces free of surface defects or die lines. However, the method does have notable drawbacks. It requires precision in forming a uniform patch and ensuring it can be removed intact from the casting surface. Additionally, it demands the use of polymers that are soluble in rapidly evaporating or water-based solvents. The reliance on organic solvents introduces further concerns, particularly regarding environmental safety and handling hazards.
Direct milling method
The direct milling method is a solvent-free approach for buccal patch fabrication, eliminating the need for liquids or organic solvents. In this technique, the drug and excipients are mechanically mixed using methods such as direct milling or kneading to achieve a homogeneous blend. The resulting mixture is then rolled onto a release liner to achieve the desired thickness. Subsequently, a backing membrane is applied to complete the patch. This solvent-free process offers advantages in terms of safety, reduced environmental hazards, and minimized health risks. Moreover, patches produced by this method demonstrate comparable performance to those prepared by solvent-based techniques, with only minor variations in certain characteristics. A notable advantage of this technique is that any polymer can be used, regardless of its solubility in water or organic solvents, and the absence of stirring during mixing simplifies the process. However, the method also presents challenges, such as difficulty in achieving uniform distribution of the active pharmaceutical ingredient (API) and the possibility of producing patches with inferior surface uniformity and mechanical properties compared to those obtained through solvent-based methods [38, 39].
Hot-melt extrusion method
Hot-melt extrusion (HME) is a solvent-free manufacturing technique that utilizes heat and pressure to mix materials and form them into thin films. The process begins by feeding a pre-blended mixture of the active pharmaceutical ingredient (API), thermoplastic polymer carriers, and plasticizers into the barrel of a hot-melt extruder. Within the heated barrel, the polymer melts while rotating screws homogenize the components into a uniform molten mass. This melt is then forced through a flat die to produce a film of the desired thickness, which is subsequently cooled and cut into individual patches. The method offers several advantages: it is an anhydrous and solvent-free process, making it particularly suitable for moisture-sensitive and solvent-sensitive drugs; it is a continuous manufacturing technique that provides high process efficiency with fewer operational steps; the intensive mixing within the extruder ensures excellent content uniformity in the final product; and it facilitates the formation of solid dispersions, thereby enhancing the bioavailability of poorly water-soluble drugs by dispersing them at the molecular level within the polymer matrix. However, HME also has limitations, as both the drug and polymer must exhibit thermal stability to withstand the high processing temperatures, which may otherwise cause degradation, and the process requires specialized, high-cost equipment [38].
Semisolid casting
The semisolid casting method is a modification of the solvent casting technique, developed to enable the incorporation of polymers with differing solubility profiles. In this process, a solution of a water-soluble film-forming polymer is first prepared, while an acid-insoluble polymer is separately dissolved in an alkaline medium. The two solutions are then blended together, and a plasticizer is added to form a homogeneous, viscous gel mass. This semisolid gel is subsequently cast onto a release liner and dried using temperature-controlled drums to yield the final film or patch. The method offers important advantages, such as the ability to combine polymers with distinct solubility and physicochemical properties, thereby allowing precise control over patch adhesion, swelling behavior, and drug release rate. It also facilitates the development of patches with unique structural characteristics, including biphasic designs. However, the technique has notable disadvantages: the formulation process is complex and highly sensitive to pH adjustments, and achieving consistent batch-to-batch reproducibility requires precise control over multiple processing variables.
3D printing
Three-dimensional (3D) printing, also known as additive manufacturing, is an emerging technology that provides innovative solutions to the formulation challenges of buccal film production. By constructing patches layer by layer from digital designs, 3D printing overcomes the drug-loading limitations of conventional methods [40]. Its multi-layered architecture enables a higher active pharmaceutical ingredient (API) content per unit area, making it particularly suitable for drugs requiring larger doses and addressing the constraint of limited mucosal surface area for absorption [38, 39, 41]. This approach also offers distinct advantages such as compartmentalization, where incompatible ingredients can be incorporated into separate layers within a single patch; controlled release, allowing complex drug release profiles to be precisely tailored; and personalization, enabling customization of dose, size, and shape to meet the needs of individual patients, thereby advancing personalized medicine. However, despite its potential, 3D printing faces limitations such as high equipment costs, longer production times compared to conventional methods, and the need for stringent process optimization to ensure reproducibility and regulatory compliance.
Technologies such as fused deposition modeling (FDM) and extrusion-based printing have already been applied successfully in buccal film fabrication [42]. For example, dexamethasone acetate buccal patches have been prepared using FDM, while adipic acid-containing buccal films have been developed by combining hot-melt extrusion with FDM-based 3D printing [43]. More recent advances from 2023–2024 have further expanded its potential, showcasing innovations such as in situ bioprinting for highly personalized therapies and the creation of QR-coded buccal patches that enable digital traceability and anti-counterfeiting measures [44]. Collectively, these developments underscore the transformative potential of 3D printing in designing sophisticated, patient-specific buccal drug delivery systems.
Inkjet printing
Inkjet printing is a non-contact, additive manufacturing technique that has been adapted for pharmaceutical applications. In the context of buccal patches, it is typically used as a secondary processing step to precisely deposit the active pharmaceutical ingredient (API) onto a pre-fabricated film (substrate), which may have been created using methods like solvent casting or hot-melt extrusion. The process involves formulating an "ink" by dissolving or dispersing the API in a suitable solvent system. This ink is then loaded into an inkjet printer, which uses either thermal or piezoelectric drop-on-demand technology to eject microscopic droplets onto the blank buccal film with high precision. By controlling the number and placement of these droplets, a specific dose and distribution pattern can be achieved [41]. A novel application of this technology is to print the API onto a substrate in the form of a readable QR code. In this concept, the drug itself constitutes the dark pixels of the code. This creates a dual-function patch that not only delivers the medication but also serves as an anti-counterfeiting measure. A patient or healthcare provider could scan the code with a smartphone to verify the drug's authenticity, access dosage information, or track the product's origin [45, 46].
Inkjet printing offers several notable advantages for buccal patch fabrication. It enables precision and personalization, allowing for highly accurate dosing down to the microgram level, which supports the development of personalized medicine by tailoring doses to individual patient needs and even enabling complex drug gradients or multi-drug patterns on a single patch. The technique is particularly suitable for sensitive drugs, as printing is typically performed at ambient temperatures, making it ideal for heat-sensitive molecules such as proteins, peptides, and other biologics that could degrade under high-temperature methods like hot-melt extrusion. Additionally, since the active pharmaceutical ingredient (API) is applied only to the surface, the bulk mechanical properties of the underlying film, such as flexibility and tensile strength, remain uncompromised, ensuring a robust final product. Inkjet printing can also stabilize APIs in an amorphous solid state, which improves solubility and enhances long-term stability. However, this method has limitations: drug loading is restricted by the solubility of the API in the ink vehicle and the absorption capacity of the substrate, and the process can be slower compared to high-throughput manufacturing techniques.
Electrospinning and electrospraying
Electrospinning and electrospraying are advanced electro-hydrodynamic atomization techniques employed to fabricate buccal patches with highly controlled micro-and nano-scale architectures [47-49]. Electrospinning generates ultrafine fibers from a polymer solution by applying a high electrical voltage, which causes a charged jet of polymer-drug solution to be ejected from a spinneret. As the jet travels toward a grounded collector, the solvent rapidly evaporates, resulting in a web of solid nanofibers that form a non-woven mat. This nanofibrous matrix is characterized by an exceptionally high surface-area-to-volume ratio and porosity [50], features that are highly advantageous for buccal drug delivery. These include enhanced drug release, as the large surface area increases the dissolution rate of poorly soluble drugs with tunable release kinetics; effective drug loading, since the nanofibrous matrix can serve as a highly efficient carrier for APIs; and improved mucoadhesion, as the flexible, porous structure conforms closely to the buccal mucosa, ensuring prolonged contact time.
Electrospraying, which operates on similar principles, utilizes solutions with lower polymer concentrations and viscosities as well as different operating parameters. Instead of producing continuous fibers, it atomizes the polymer-drug solution into fine charged droplets that, upon solvent evaporation, deposit micro-or nano-particles onto a collector surface. This allows for the creation of particle-based films or the coating of other substrates, such as electrospun mats, with drug-loaded particles. For instance, organogel films derived from poly(L-lactic-co-ε-caprolactone) have been fabricated using this technique, providing biodegradable and biocompatible options for buccal delivery. Together, electrospinning and electrospraying offer unparalleled control over the physical properties and nano-architecture of buccal patches, enabling the design of highly efficient and sophisticated drug delivery systems. However, their practical application is hindered by major limitations, including low production throughput that challenges scalability and the frequent use of toxic organic solvents, which raises significant safety and regulatory concerns [51].
Table 3: Comparative analysis of buccal patch fabrication techniques
| Technique | Equipment cost | Scalability/Throughput | Drug loading capacity | API stability suitability | Key advantage | Key limitation |
| Solvent Casting | Low | High | Moderate | Good for most APIs | Simple, low cost, high flexibility | Use of organic solvents, residual solvent risk |
| Direct milling | Low | High | Moderate-High | Excellent (solvent-free) | Solvent-free, simple process | Poor API uniformity, inferior mechanical properties |
| Hot-Melt Extrusion (HME) | High | Very High | High | Thermostable APIs only | Solvent-free, continuous process, enhances solubility | High temperature degrades thermolabile drugs |
| 3D Printing | Moderate-High | Low | Very High | Thermolabile (inkjet) or Thermostable (FDM) | Personalized dose/geometry, complex release profiles | Slow production speed, regulatory novelty |
| Inkjet Printing | Moderate | Low | Low | Thermolabile biologics | High precision dosing, ideal for sensitive drugs | Low drug loading, secondary processing step |
| Electro- spinning |
Moderate | Very Low | High | Good for most APIs | High surface area, enhanced dissolution | Very low throughput, use of toxic solvents |
Evaluations of buccal patches
A series of rigorous quality control tests are performed to evaluate the physical, mechanical, and performance characteristics of buccal patches. These evaluations are essential to ensure the safety, efficacy, batch-to-batch consistency, and overall quality of the final drug delivery system.
Weight variation
Weight variation is a fundamental quality control test employed to ensure dosage consistency within a batch of buccal patches. The procedure involves individually weighing a representative number of patches using a precision analytical balance to determine the mean weight and standard deviation. Each individual weight is then compared against the calculated average to assess compliance with the limits established by official pharmacopoeias. Uniformity in weight serves as a key indicator of manufacturing reproducibility and is directly correlated with drug content uniformity, which is essential for maintaining therapeutic safety, efficacy, and overall product quality [52].
Thickness
The thickness of buccal patches is a critical parameter for ensuring dimensional consistency, which directly influences drug content uniformity, controlled release rates, and patient comfort. Thickness measurements are performed using calibrated instruments such as a digital micrometer or screw gauge at multiple points (typically the center and four corners) across several patches. The mean thickness and standard deviation are then calculated to assess uniformity. This verification not only ensures dosage accuracy but also confirms that the patch dimensions are optimized for effective bioadhesion and performance [53, 54].
Folding endurance
Folding endurance evaluates the mechanical flexibility and structural integrity of a buccal patch, reflecting its ability to withstand physical stress during handling and application without breaking. The test involves repeatedly folding a single patch at the same crease until a crack forms or it breaks completely. The total number of folds endured before failure is recorded as the folding endurance value. A higher value indicates superior mechanical strength and flexibility, suggesting that the patch is less prone to brittleness and is more likely to maintain integrity within the dynamic environment of the buccal cavity [38].
Surface pH measurement
Surface pH measurement is an essential safety assessment to ensure that buccal patches are non-irritating and biocompatible with the oral mucosa. Deviations toward acidic or alkaline pH can lead to patient discomfort or mucosal irritation. For this test, a patch is hydrated on an agar plate or with simulated saliva, and a calibrated micro-pH electrode is placed in contact with the swollen surface to record the pH. An optimal surface pH, ideally in the range of 6.5–7.0, closely mimics natural saliva and supports patient comfort and mucosal compatibility [39].
Swelling study
The swelling study evaluates a buccal patch’s ability to absorb fluid from the oral cavity, a property critical for initiating mucoadhesion and controlling drug release. A pre-weighed dry patch (W₀) is immersed in simulated salivary fluid at 37 °C. The SSF was prepared using a phosphate buffer and supplemented with electrolytes such as NaCl and KCl, as well as divalent ions including CaCl₂ and MgCl₂ to reproduce physiological ionic strength. To better mimic human saliva, optional organic components such as urea, mucin, and α-amylase were incorporated, and the pH was adjusted to fall within the physiological range of 6.5–7.4 [55-58]. Since the pH and electrolyte content are critical for reproducibility, the preparation of SSF should ideally be standardized in accordance with protocols such as ISO 10271:2020 [59]. The swollen weight (Wₜ) is measured at predetermined intervals after carefully blotting excess surface moisture. The swelling index (%S) is calculated using the equation:
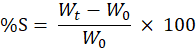
Where Wₜ is the weight at time t and W₀ is the initial weight.
This parameter provides valuable insight into the polymer matrix behavior and helps predict adhesion performance and drug release profiles in vivo [42], and advanced techniques such as magnetic resonance imaging (MRI), X-ray microtomography (XµT), atomic force microscopy (AFM), attenuated total reflectance Fourier transform infrared (ATR-FTIR) spectroscopy, fluorescence spectroscopy, texture analysis, and ultrasound imaging have been employed to monitor swelling and structural changes in real time [60].
Tensile strength
Tensile strength is defined as the maximum stress a buccal patch can withstand before rupture, serving as a critical indicator of its mechanical robustness and integrity. This parameter is typically measured using a tensiometer or a custom-designed (in-house) apparatus. In the in-house setup, a patch specimen is clamped between two clips—one fixed and the other connected to a movable pan [61]. The original length of the specimen is recorded, and incremental weights are gradually added to the pan until the patch ruptures. The force at break (N) is divided by the cross-sectional area (m²) of the film to calculate tensile strength (MPa) using the equation:
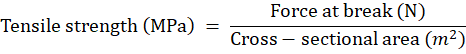
Additional mechanical parameters, such as strain and Young’s modulus, provide further insights into material behavior. Strain reflects the degree of elongation under stress, while Young’s modulus quantifies stiffness and resistance to deformation [62]. These metrics are essential to ensure that the patch maintains its structural integrity during handling, storage, and application—particularly when subjected to the dynamic environment of the buccal cavity.
Content uniformity
The content uniformity of the buccal patches was determined to ensure that every patch contains a consistent and uniform amount of the active pharmaceutical ingredient, in accordance with pharmacopeial standards. This evaluation adheres to the principles outlined in the United States Pharmacopeia (USP) General Chapter<905>Uniformity of Dosage Units.
For the assay, ten patches were randomly selected from a batch, and each was individually immersed in a vessel containing no more than 100 ml of isotonic phosphate buffer (pH 6.8) to simulate the physiological pH of saliva. The vessels were agitated for 1 to 8 h to facilitate complete dissolution of the API. After dissolution, aliquots of the solution were filtered through a 0.45 µm filter to remove undissolved excipients and subsequently diluted to fall within the calibrated range of the analytical method. Drug quantification was performed using a validated technique, such as UV-Vis spectrophotometry or high-performance liquid chromatography (HPLC).
The drug amount of each patch was calculated, and results were evaluated against USP acceptance criteria, which stipulate that individual dosage units must fall within a specified range around the label claim. This verification ensures dose-to-dose consistency, which is essential for patient safety and therapeutic efficacy [63].
Mucoadhesive strength
Mucoadhesive strength measures the force required to detach a buccal patch from a mucosal surface, providing a direct assessment of its adhesive performance and predicted residence time in the oral cavity. In a standard in vitro setup using a texture analyzer, the patch is affixed to a probe and pressed against a section of excised animal buccal mucosa or a standardized synthetic mucosal membrane (e. g., porcine, rabbit, or synthetic) with a defined force (in N) for a specified contact time (in seconds), following established concepts like those outlined in USP<1151>for general principles for evaluating dosage forms [64]. The probe is then retracted at a controlled speed, and the peak force needed to separate the patch from the tissue is recorded as the mucoadhesive strength. This parameter is critical for evaluating the ability of buccal patches to maintain prolonged contact with the mucosal surface under dynamic oral conditions [65].
In vitro drug release and permeation studies
In vitro permeation studies are conducted to evaluate a buccal patch’s capacity to deliver drugs across the mucosal barrier and predict its therapeutic performance. A Franz diffusion cell (fig. 2) is commonly employed, where the patch is placed on an excised buccal mucosa mounted between the donor and receptor compartments. The receptor compartment is filled with simulated salivary fluid or phosphate buffer (pH 6.8) maintained at 37 ± 0.5 °C and continually stirred to maintain sink conditions.
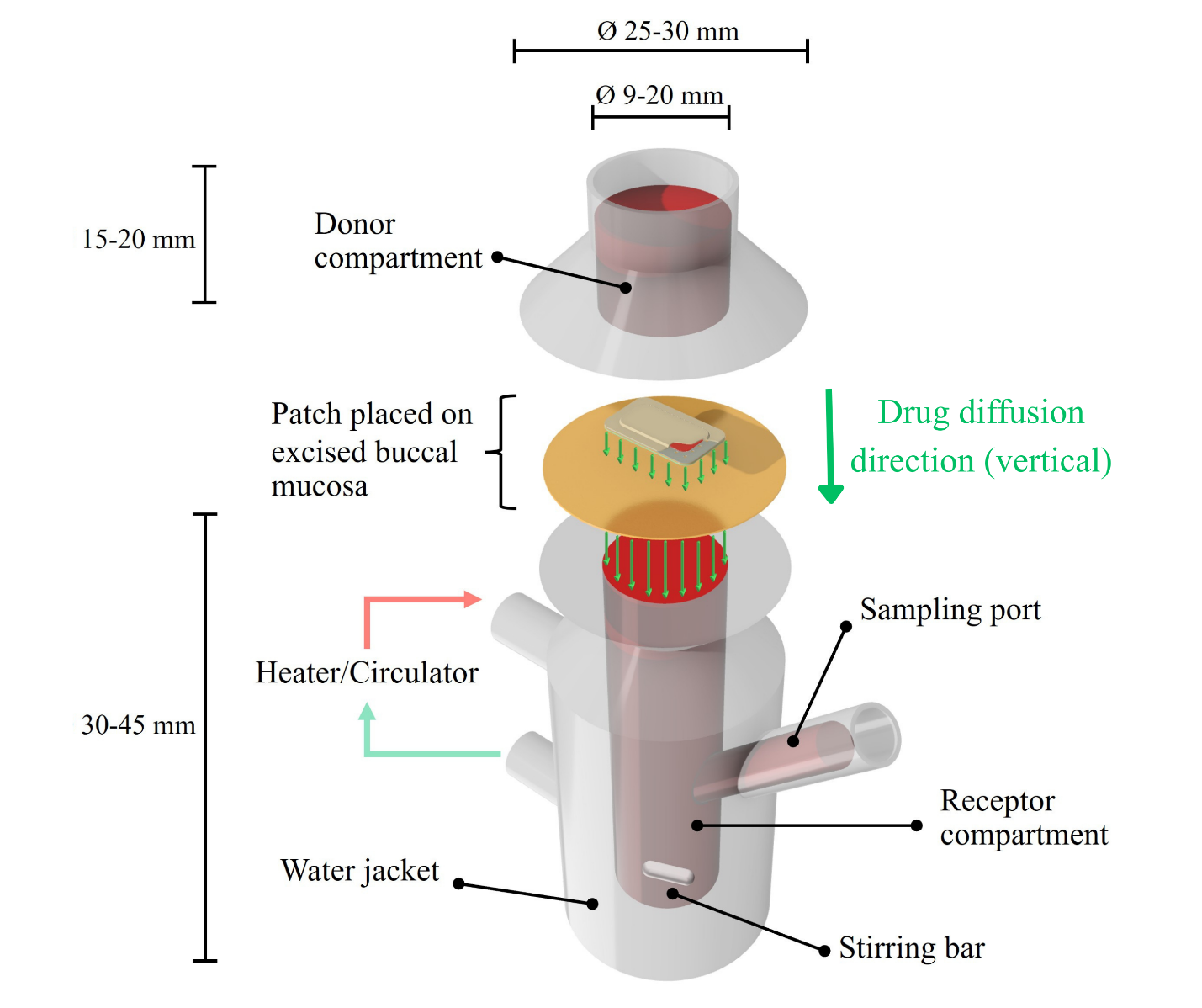
Fig. 2: Schematic of a franz diffusion cell for a permeation study for buccal patches
Aliquots are periodically withdrawn from the receptor compartment over a defined period and analyzed using validated methods. The permeation profile generated from these measurements provides insights into the rate and extent of drug absorption through the mucosa, which is critical for assessing formulation performance and optimizing therapeutic efficacy [66].
Morphological characterization (scanning electron microscopy)
Scanning electron microscopy (SEM) is employed to examine the surface morphology of buccal patches. Prior to imaging, a thin layer of gold is sputter-coated onto the surface of the patch under vacuum conditions to enhance conductivity and improve image resolution. The prepared samples are then examined under the SEM at an accelerating voltage of 80 kV to observe surface features such as texture, porosity, and homogeneity [67].
Stability study in human saliva
This biorelevant study evaluates the physical and chemical stability of buccal patches in conditions simulating the human oral cavity. Patches are incubated in pooled human saliva or a validated simulated salivary fluid at 37 °C for a period representative of their intended application time. Throughout the study, the patches are monitored for any changes in physical attributes such as color, shape, and structural integrity. At the conclusion, the residual drug content may also be analyzed to assess potential chemical degradation, ensuring the patch remains stable and effective during its use [68].
Challenges in buccal drug delivery
The buccal cavity presents a limited absorption area, which is further constrained by the size of the buccal patch. Continuous salivary flow can dilute the drug concentration at the absorption site and lead to inadvertent swallowing, removing a substantial amount of drug from its intended location. Additionally, saliva may degrade moisture-sensitive drugs, and there is a risk of accidental swallowing of the patch itself. Patients using buccal patches are often restricted from speaking, eating, or drinking, which can negatively affect compliance.
To overcome these limitations, an ideal buccal patch must adhere rapidly to the mucosa and maintain sufficient adhesive strength to remain in place throughout the intended duration. It should enable controlled and sustained drug release without impeding routine oral activities such as speaking, eating, or drinking, thereby ensuring patient comfort and compliance [52].
Applications of buccal patches
Buccal patches have demonstrated versatility in delivering both systemic and local therapies. Systemically, they are employed for managing hypertension (e. g., atenolol), hormone replacement therapy, treatment of angina pectoris (nitroglycerin), and pain management in cancer using opioid analgesics. They are also used in smoking cessation programs (nicotine) and for treating microbial infections in conditions such as periodontitis. Locally, buccal patches provide therapeutic benefits for oral disorders, including infections, mouth ulcers, dental caries, gingivitis, and stomatitis [53, 54]. This wide range of applications highlights the potential of buccal patches as targeted and effective drug delivery systems across diverse medical contexts.
Patents and marketed formulations
Table 4 provides an overview of patents related to buccal patches, highlighting innovations and technological advancements in this domain. Table 5 summarizes marketed buccal patch formulations, detailing their brand names, manufacturers, and therapeutic applications. Together, these tables offer valuable insights into both ongoing research and the practical implementation of buccal patch technology in healthcare.
An analysis of the patent landscape (table 4) reveals a significant trend towards enhancing drug delivery efficiency, improving patient compliance through more flexible and less irritating patch materials, and engineering sophisticated controlled-release mechanisms. On the commercial front, the variety of marketed formulations showcases the successful application of this technology in treating conditions that benefit from rapid absorption or avoidance of first-pass metabolism, such as acute pain, opioid dependence, and hormonal imbalances. This dynamic interplay between innovation and market availability not only highlights the clinical viability of buccal patches but also signals a promising future for further expansion into other therapeutic areas. For commercially related buccal patches (table 5), a diverse range of formulations are available to address various therapeutic needs. These patches are widely used for pain management, particularly in cancer patients, utilizing active drugs such as fentanyl and buprenorphine. They are also crucial for treating opioid dependence through combination formulations of buprenorphine and naloxone. Other significant applications include smoking cessation (nicotine), managing hypertension (carvedilol), providing topical anesthesia (lidocaine), and hormone therapy (androgens).
Table 4: Patents on buccal patches/films
| Title | Patent number | Inventors | Year |
| Buccal local anaesthetic [69] | WO1989010740A1 | Robert Nichol Boyes, et al. | 1989 |
| Desmopressin Buccal Patch Composition [70] | US5298256A | Ian R. Flockhart and Joseph H. Cort | 1994 |
| Transmucosal formulations of levosimendan [71] | US6399610B1 | KaukoKurkela, et al. | 2002 |
| Bioadhesivehot-melt extruded film for topical and mucosal adhesion applications and drug delivery and process for preparation and thereof [72] | US6375963B1 | Michael A. Repka, et al. | 2002 |
| Soft, adherent, soluble oral patch [73] | US20030124178A1 | Jeffrey Haley | 2003 |
| Water soluble film for oral administration with instant wettability [74] | US6709671B2 | Horst Georg Zerbe, et al. | 2004 |
| Transmucosal form of administration with reduced mucosal irritation [75] | WO2005000263A1 | Tina Rademacher and Christian Von Falkenhausen |
2005 |
| Edible film for transmucosal delivery of nutritional supplements [76] | WO006119286A1 | Maurice E. Durschlag and Gary S. Kehoe | 2006 |
| Film bandage for mucosal administration of actives [77] | WO2007084617A2 | Richard C. Fuisz | 2007 |
| Orally administrable films and preparation thereof [78] | WO2007125533A2 | Meir Haber, et al. | 2007 |
| Two Phase mucoadhesive Composition [79] | WO2008002353A1 | James E. Biegajski | 2008 |
| Canker sore patch [80] | WO2010006612A3 | Leonardo Malcovati | 2010 |
| Water soluble pharmaceutical film with enhanced stability [81] | WO2015083181A2 | Anwar Siraj Daud, et al. | 2015 |
| Bioadhesive films for local and/or systemic delivery [82] | US2016128947A1 | Jason Thomas McConville,et al. | 2016 |
| Buccal bioadhesive strip and method of treating snoring and sleep apnea [83] | US9549842B2 | Joseph E. Kovarik | 2017 |
| Sublingual and buccal film compositions [84] | US9687454B2 | Garry L. Myers, et al. | 2017 |
| Sublingual or buccal administration of dim for treatment of skin diseases [85] | WO2018051183 | Michael C. Scaife | 2018 |
| Mucoadhesive preparations, methods and applications thereof [86] | WO2018083643A1 | Leo Mavely, et al. | 2018 |
| Pharmaceutical dosage form for application to mucous membranes [87] | US20180036251A1 | Malte Bogdahn. et al. | 2018 |
| Pharmaceutical compositions for the transmucosal delivery of therapeutic peptides and proteins [88] | WO2019193204A1 | Martin Werle and Florian FÖGER | 2019 |
| Thin film with non-self-aggregating uniform heterogeneity and drug delivery systems made therefrom [89] | US20200163901A1 | Robert K. Yang, et al. | 2020 |
| Controlled release mucoadhesive systems [90] | US10758619B2 | Susan R. Mallery, et al. | 2020 |
| Multilayered buccal patch of antihypertensive drugs [91] | ZA202213524B | Vivek Subhash Tarate. et al. | 2022 |
| Detection and treatment of obstructive sleep apnea [92] | US20230083643A1 | Hoo-min D. Toong and William C. Altmann | 2023 |
| Dry eye treatment systems [93] | US20250228702A1 | Paul Badawi | 2025 |
| Controller for dry eye treatment systems [94] | US12257181B2 | David Badawi, et al. | 2025 |
Table 5: Marketed formulation of buccal patches
| Brand name | Active drug | Manufacturer | Uses |
| Nicoderm CQ | Nicotine | Pfizer | Help people to quit smoking |
| Anadrol-50 | Androgen | Thomson Healthcare | Used as Hormonal agent |
| Breaky-400mcg | Fentanyl | MEDA | Alleviates pain experienced by individuals with cancer |
| Fentanyl MTX Patch | Fentanyl | Sandoz | Decreases pain in cancer patients |
| Coreg | Carvediol | GlaxoSmithKline plc | Hypertension |
| DentiPatch | Lidocaine | Noven Pharmaceuticals | Topical anesthesia of the accessible mucous membranes |
| Suboxone | Buprenorphine, Naloxone | Reckitt Benckiser | Treat opioid dependence |
| Belbuca | Buprenorphine HCl | Endo Pharmaceuticals Inc | Opioid pain Medicine |
| Onsolis | Fentanyl | Meda Pharmaceuticals | Decreases pain in cancer patients |
| Bunavail | Buprenorphine, Naloxone | Biodelivery Sciences International Inc | Treat opioid dependence |
Barriers to commercialization of buccal patches
Despite the growing academic and industrial interest in buccal patch technologies, their successful clinical translation and commercialization remain limited due to a number of critical barriers. One of the foremost challenges is regulatory uncertainty, particularly for systems incorporating novel excipients, 3D printing, or nanofabrication techniques such as electrospinning. Regulatory agencies like the U. S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) require rigorous documentation for quality, safety, and efficacy, but there is currently no standardized regulatory pathway tailored specifically for buccal patch systems. This is especially problematic for personalized or hybrid devices, which may be classified as drug-device combinations and thus face additional scrutiny under complex approval frameworks such as the FDA’s Combination Product Rule (21 CFR Part 3) [95].
Safety concerns, particularly relating to the long-term use of permeation enhancers, also pose a significant barrier. Enhancers such as sodium lauryl sulfate (SLS), bile salts, and fatty acids have demonstrated efficacy in increasing drug permeability across the buccal mucosa. However, repeated or prolonged exposure to such agents can lead to mucosal irritation, inflammation, or epithelial damage, raising questions about their biocompatibility in chronic or high-dose therapies [96-98]. Regulatory authorities, including the FDA, have issued warnings about mucosal toxicity associated with certain surfactants, underscoring the need for thorough in vitro and in vivo biocompatibility testing before approval. This creates a bottleneck for otherwise promising formulations that fail to meet safety thresholds for long-term mucosal contact [99].
On the manufacturing and scalability front, translating laboratory-scale fabrication methods into commercial production remains a formidable challenge. While techniques like solvent casting and hot-melt extrusion are more amenable to industrial-scale manufacturing [100], advanced methods such as electrospinning, 3D printing, and inkjet deposition face issues related to low throughput, equipment cost, reproducibility, and GMP compliance [101, 102]. These methods often require specialized environmental controls and fine-tuned parameters to maintain consistency in patch architecture, drug loading, and mechanical properties. In particular, electrospinning suffers from low scalability and solvent toxicity concerns, whereas 3D printing, though highly customizable, requires extensive validation for process repeatability and dose uniformity [103].
Finally, commercialization hurdles extend beyond technical and regulatory aspects to include intellectual property (IP) competition, cost-effectiveness, and market adoption challenges. Many patents listed in recent years (table 4) demonstrate innovation in materials, multilayer structures, and drug release mechanisms, but relatively few have progressed to approved products (table 5). The long development timelines, uncertain return on investment, and reimbursement challenges make pharmaceutical companies hesitant to adopt buccal patches, especially for non-life-threatening conditions. Furthermore, lack of patient awareness and limited clinical trial data comparing buccal patches to conventional dosage forms contribute to slow market penetration. Thus, addressing these barriers will require coordinated efforts involving academia, industry, and regulatory agencies to create standardized evaluation protocols, incentivize GMP-compliant innovation, and streamline approval pathways.
Future perspectives
Buccal patches are evolving as a highly viable drug delivery system, offering precise dosing, controlled release, and enhanced stability. Future advancements, particularly in nanotechnology, are anticipated to enable targeted drug administration within the oral cavity, facilitating personalized therapies tailored to individual patient needs. Innovative strategies, such as incorporating antigens like ovalbumin into microneedles for effective mucosal vaccination, are under investigation. Moreover, the development of advanced bioadhesive materials aims to improve safety, biocompatibility, and patient comfort. With expanding applications beyond pain management and hormone therapy, buccal patches also hold promise for managing chronic conditions such as diabetes, cardiovascular diseases, and neurological disorders, thereby broadening their therapeutic potential.
CONCLUSION
Buccal patches represent a promising drug delivery platform with significant advantages, including precise dosing, controlled drug release, improved patient compliance, and rapid onset of action. While challenges such as limited absorption surface, salivary dilution, and patient-related restrictions persist, ongoing innovations in formulation design, manufacturing technologies, and evaluation methods are steadily overcoming these barriers. Continued research and technological progress are expected to establish buccal patches as a versatile, patient-centric drug delivery platform that contributes meaningfully to the advancement of modern healthcare.
ACKNOWLEDGEMENT
The authors express sincere gratitude to Amar Shaheed Baba Ajit Singh Jujhar Singh Memorial College of Pharmacy, Bela for the invaluable support and assistance in facilitating the literature search.
FUNDING
No funding was received
AUTHORS CONTRIBUTIONS
Kampanart Huanbutta: Writing-Reviewing and Editing, Visualization, Project administration; Tanuj Kumar: Writing-Original Draft; Sandeep Kumar: Writing-Original Draft; Vandana Kharb: Writing-Original Draft; Tanikan Sangnim: Writing-Original Draft; Kittipat Suwanpitak: Writing-Original Draft, Visualization; Inderbir Singh: Conceptualization, Writing-Reviewing and Editing, Supervision. All authors have read and agreed to the published version of the manuscript.
CONFLICT OF INTERESTS
There are no interests to declare
REFERENCES
Hearnden V, Sankar V, Hull K, Juras DV, Greenberg M, Kerr AR. New developments and opportunities in oral mucosal drug delivery for local and systemic disease. Adv Drug Deliv Rev. 2012;64(1):16-28. doi: 10.1016/j.addr.2011.02.008, PMID 21371513.
Adhikari SN, Nayak BS, Nayak AK, Mohanty B. Formulation and evaluation of buccal patches for delivery of atenolol. AAPS PharmSciTech. 2010;11(3):1038-44. doi: 10.1208/s12249-010-9459-z, PMID 20533098.
Heemstra LB, Finnin BC, Nicolazzo JA. The buccal mucosa as an alternative route for the systemic delivery of risperidone. J Pharm Sci. 2010;99(11):4584-92. doi: 10.1002/jps.22175, PMID 20845457.
Parmar HG, Jain JJ, Patel TK, Patel VM. Buccal patch: a technical note. Int J Pharm Sci Rev Res. 2010;4(3):178-82.
Rohani Shirvan A, Hemmatinejad N, Bahrami SH, Bashari A. Fabrication of multifunctional mucoadhesive buccal patch for drug delivery applications. J Biomed Mater Res A. 2021;109(12):2640-56. doi: 10.1002/jbm.a.37257, PMID 34190400.
Jacob S, Nair AB, Boddu SH, Gorain B, Sreeharsha N, Shah J. An updated overview of the emerging role of patch and film-based buccal delivery systems. Pharmaceutics. 2021;13(8):1206. doi: 10.3390/pharmaceutics13081206, PMID 34452167.
Sabra R, Kirby D, Chouk V, Malgorzata K, Mohammed AR. Buccal absorption of biopharmaceutics classification system III drugs: formulation approaches and mechanistic insights. Pharmaceutics. 2024;16(12):1563. doi: 10.3390/pharmaceutics16121563, PMID 39771541.
Korbonits M, Slawik M, Cullen D, Ross RJ, Stalla G, Schneider H. A comparison of a novel testosterone bioadhesive buccal system striant with a testosterone adhesive patch in hypogonadal males. J Clin Endocrinol Metab. 2004;89(5):2039-43. doi: 10.1210/jc.2003-030319, PMID 15126518.
Merkle HP, Anders R, Wermerskirchen A. Mucoadhesive buccal patches for peptide delivery. In: Bioadhesive drug delivery systems. Boca Raton, FL: CRC Press; 2024. p. 105-36. doi: 10.1201/9781003574484-6.
Venkatalakshmi R, Sudhakar Y, Chetty MC, Sasikala C, Varma MM. Buccal drug delivery using adhesive polymeric patches. Int J Pharm Sci Res. 2012;3(1):35-41. doi: 10.13040/IJPSR.0975-8232.3(1).35-41.
Morales JO, McConville JT. Manufacture and characterization of mucoadhesive buccal films. Eur J Pharm Biopharm. 2011;77(2):187-99. doi: 10.1016/j.ejpb.2010.11.023, PMID 21130875.
Shojaei AH, Berner B, Xiaoling L. Transbuccal delivery of acyclovir: I In vitro determination of routes of buccal transport. Pharm Res. 1998;15(8):1182-8. doi: 10.1023/A:1011927521627, PMID 9706047.
Shaikh R, Raj Singh TR, Garland MJ, Woolfson AD, Donnelly RF. Mucoadhesive drug delivery systems. J Pharm Bioallied Sci. 2011;3(1):89-100. doi: 10.4103/0975-7406.76478, PMID 21430958.
Gandhi PA, Patel M, Patel K, Patel N. A review article on mucoadhesive buccal drug delivery system. Int J Pharm Res Dev. 2011;3(5):159-73.
Mujoriya R, Dhamande K, Wankhede U, Angure S. A review on study of buccal drug delivery system. Innov Syst Eng. 2011;2(3):200-4.
Prajapati V, Bansal M, Sharma PK. Mucoadhesive buccal patches and use of natural polymer in its preparation a review. Int J PharmTech Res. 2012;4(2):582-9.
Salamat Miller N, Chittchang M, Johnston TP. The use of mucoadhesive polymers in buccal drug delivery. Adv Drug Deliv Rev. 2005;57(11):1666-91. doi: 10.1016/j.addr.2005.07.003, PMID 16183164.
Khutoryanskiy VV. Advances in mucoadhesion and mucoadhesive polymers. Macromol Biosci. 2011;11(6):748-64. doi: 10.1002/mabi.201000388, PMID 21188688.
Williams AC, Barry BW. Penetration enhancers. Adv Drug Deliv Rev. 2012;64:128-37. doi: 10.1016/j.addr.2012.09.032.
Morales JO, Huang S, Williams III RO, McConville JT. Films loaded with insulin-coated nanoparticles (ICNP) as potential platforms for peptide buccal delivery. Colloids Surf B Biointerfaces. 2014;122:38-45. doi: 10.1016/j.colsurfb.2014.05.025, PMID 25016543.
Gopalakrishnan S, Pandey N, Tamiz AP, Vere J, Carrasco R, Somerville R. Mechanism of action of ZOT-derived peptide AT-1002, a tight junction regulator and absorption enhancer. Int J Pharm. 2009;365(1-2):121-30. doi: 10.1016/j.ijpharm.2008.08.047, PMID 18832018.
Lu Y, Ma Y, Wu W. Ionic liquids: emerging chemical permeation enhancers. MedComm Biomaterials and Applications. 2024;3(2):e81. doi: 10.1002/mba2.81.
Aungst BJ. Absorption enhancers: applications and advances. AAPS J. 2012;14(1):10-8. doi: 10.1208/s12248-011-9307-4, PMID 22105442.
Iyer H, Khedkar A, Verma M. Oral insulin a review of current status. Diabetes Obes Metab. 2010;12(3):179-85. doi: 10.1111/j.1463-1326.2009.01150.x, PMID 20151994.
Ramadon D, McCrudden MT, Courtenay AJ, Donnelly RF. Enhancement strategies for transdermal drug delivery systems: current trends and applications. Drug Deliv Transl Res. 2022;12(4):758-91. doi: 10.1007/s13346-021-00909-6, PMID 33474709.
Park K, Kwon IC, Park K. Oral protein delivery: current status and future prospect. React Funct Polym. 2011;71(3):280-7. doi: 10.1016/j.reactfunctpolym.2010.10.002.
Nicolazzo JA, Reed BL, Finnin BC. Buccal penetration enhancers how do they really work? J Control Release. 2005;105(1-2):1-15. doi: 10.1016/j.jconrel.2005.01.024, PMID 15894393.
Som I, Bhatia K, Yasir M. Status of surfactants as penetration enhancers in transdermal drug delivery. J Pharm Bioallied Sci. 2012;4(1):2-9. doi: 10.4103/0975-7406.92724, PMID 22368393.
Hassan N, Ahad A, Ali M, Ali J. Chemical permeation enhancers for transbuccal drug delivery. Expert Opin Drug Deliv. 2010;7(1):97-112. doi: 10.1517/17425240903338758, PMID 20017661.
Palem CR, Kumar Battu S, Gannu R, Yamsani VV, Repka MA, Yamsani MR. Role of cyclodextrin complexation in felodipine-sustained release matrix tablets intended for oral transmucosal delivery: in vitro and ex vivo characterization. Pharm Dev Technol. 2012;17(3):321-32. doi: 10.3109/10837450.2010.535829, PMID 21265709.
Senel S, Kremer MJ, Kas S, Wertz PW, Hıncal AA, Squier CA. Enhancing effect of chitosan on peptide drug delivery across buccal mucosa. Biomaterials. 2000;21(20):2067-71. doi: 10.1016/S0142-9612(00)00134-4, PMID 10966016.
Padula C, Pescina S, Nicoli S, Santi P. New insights on the mechanism of fatty acids as buccal permeation enhancers. Pharmaceutics. 2018;10(4):201. doi: 10.3390/pharmaceutics10040201, PMID 30355980.
Jampilek J, Brychtova K. Azone analogues: classification, design and transdermal penetration principles. Med Res Rev. 2012;32(5):907-47. doi: 10.1002/med.20227, PMID 22886628.
Sood A, Panchagnula R. Peroral route: an opportunity for protein and peptide drug delivery. Chem Rev. 2001;101(11):3275-303. doi: 10.1021/cr000700m, PMID 11840987.
Hou SY, Flynn GL. Influences of 1-dodecylazacycloheptan-2-one on permeation of membranes by weak electrolytes. 1. Theoretical analysis of weak electrolyte diffusion through membranes and studies involving silicone rubber membranes. J Pharm Sci. 1997;86(1):85-91. doi: 10.1021/js9601905, PMID 9002465.
Cox DS, Gao H, Raje S, Scott KR, Eddington ND. Enhancing the permeation of marker compounds and enaminone anticonvulsants across Caco-2 monolayers by modulating tight junctions using zonula occludens toxin. Eur J Pharm Biopharm. 2001;52(2):145-50. doi: 10.1016/S0939-6411(01)00172-2, PMID 11522479.
Khanna R, Agarwal SP, Ahuja A. Preparation and evaluation of muco-adhesive buccal films of clotrimazole for oral Candida infections. Indian J Pharm Sci. 1997;59(6):299-305.
Lodhi M, Dubey A, Narayan R, Prabhu P, Priya S. Formulation and evaluation of buccal film of ivabradine hydrochloride for the treatment of stable angina pectoris. Int J Pharm Investig. 2013;3(1):47-53. doi: 10.4103/2230-973X.108963, PMID 23799205.
Perumal VA, Lutchman D, Mackraj I, Govender T. Formulation of monolayered films with drug and polymers of opposing solubilities. Int J Pharm. 2008;358(1-2):184-91. doi: 10.1016/j.ijpharm.2008.03.005, PMID 18430529.
Huanbutta K, Burapapadh K, Sriamornsak P, Sangnim T. Practical application of 3D printing for pharmaceuticals in hospitals and pharmacies. Pharmaceutics. 2023;15(7):1877. doi: 10.3390/pharmaceutics15071877, PMID 37514063.
Huanbutta K, Sriamornsak P, Singh I, Sangnim T. Manufacture of 2D-printed precision drug-loaded orodispersible film prepared from tamarind seed gum substrate. Appl Sci. 2021;11(13):5852. doi: 10.3390/app11135852.
Nafee NA, Ismail FA, Boraie NA, Mortada LM. Mucoadhesive buccal patches of miconazole nitrate: in vitro/in vivo performance and effect of ageing. Int J Pharm. 2003;264(1-2):1-14. doi: 10.1016/S0378-5173(03)00371-5, PMID 12972331.
Elkanayati RM, Chambliss WG, Omari S, Almutairi M, Repka MA, Ashour EA. Mucoadhesive buccal films for treatment of xerostomia prepared by coupling HME and 3D printing technologies. J Drug Deliv Sci Technol. 2022;75:103660. doi: 10.1016/j.jddst.2022.103660.
Oh BC, Jin G, Park C, Park JB, Lee BJ. Preparation and evaluation of identifiable quick response (QR)-coded orodispersible films using 3D printer with directly feeding nozzle. Int J Pharm. 2020;584:119405. doi: 10.1016/j.ijpharm.2020.119405, PMID 32387310.
Montenegro Nicolini M, Reyes PE, Jara MO, Vuddanda PR, Neira Carrillo A, Butto N. The effect of inkjet printing over polymeric films as potential buccal biologics delivery systems. AAPS PharmSciTech. 2018;19(8):3376-87. doi: 10.1208/s12249-018-1105-1, PMID 29934803.
Carou Senra P, Awad A, Basit AW, Alvarez Lorenzo C, Goyanes A. Harnessing handheld inkjet printing technology for rapid and decentralised fabrication of drug-loaded hydroxypropyl cellulose buccal films. Carbohydr Polym Technol Appl. 2025;9:100724. doi: 10.1016/j.carpta.2025.100724.
Huanbutta K, Sittikijyothin W, Sangnim T. Development and characterization of bilayer wound healing patch nanofiber fabricated by electrospinning. Journal of Nano Research. 2019;59:46-56. doi: 10.4028/www.scientific.net/JNanoR.59.46.
Sangnim T, Limmatvapirat S, Nunthanid J, Sriamornsak P, Sittikijyothin W, Wannachaiyasit S. Design and characterization of clindamycin-loaded nanofiber patches composed of polyvinyl alcohol and tamarind seed gum and fabricated by electrohydrodynamic atomization. Asian J Pharm Sci. 2018;13(5):450-8. doi: 10.1016/j.ajps.2018.01.002, PMID 32104419.
Huanbutta K, Sangnim T, Limmatvapirat S, Nunthanid J, Sriamornsak P. Design and characterization of prednisolone-loaded nanoparticles fabricated by electrohydrodynamic atomization technique. Chem Eng Res Des. 2016;109:816-23. doi: 10.1016/j.cherd.2016.03.004.
Teo WE, Ramakrishna S. A review on electrospinning design and nanofibre assemblies. Nanotechnology. 2006;17(14):R89-106. doi: 10.1088/0957-4484/17/14/R01, PMID 19661572.
Nguyen DN, Clasen C, Van Den Mooter G. Pharmaceutical applications of electrospraying. J Pharm Sci. 2016;105(9):2601-20. doi: 10.1016/j.xphs.2016.04.024, PMID 27287515.
Sudhakar Y, Kuotsu K, Bandyopadhyay AK. Buccal bioadhesive drug delivery a promising option for orally less efficient drugs. J Control Release. 2006;114(1):15-40. doi: 10.1016/j.jconrel.2006.04.012, PMID 16828915.
Seema KK, Teotia D, Deepak Teotia. A comprehensive review on buccal patches. GSC Biol and Pharm Sci. 2020;13(1):130-5. doi: 10.30574/gscbps.2020.13.1.0308.
Hanif M, Zaman M, Chaurasiya V. Polymers used in buccal film: a review. Des Monomers Polym. 2015;18(2):105-11. doi: 10.1080/15685551.2014.971389.
Gittings S, Turnbull N, Henry B, Roberts CJ, Gershkovich P. Characterisation of human saliva as a platform for oral dissolution medium development. Eur J Pharm Biopharm. 2015;91:16-24. doi: 10.1016/j.ejpb.2015.01.007, PMID 25603197.
Khan J, Williams R, Frith WJ, Menzies AR, Ang S, Chen S. Probing the interactions of chitosan based nanoparticles with mucin and their implications for oral drug delivery. Int J Pharm. 2017;532(1):521-30.
Klimek J, Hellwig E, Ahrens G. Artificial saliva formulations for in vitro studies on dental hard tissue demineralization: influence of composition and stability. Caries Res. 2020;54(2):123-31.
Marques MR, Loebenberg R, Almukainzi M. Simulated biological fluids with possible application in dissolution testing. Diss Technol. 2011;18(3):15-28. doi: 10.14227/DT180311P15.
International Organization for Standardization. Istry corrosion test methods for metallic materials. Geneva ISO; 2020.
Huanbutta K, Terada K, Sriamornsak P, Nunthanid J. Advanced technologies for assessment of polymer swelling and erosion behaviors in pharmaceutical aspect. Eur J Pharm Biopharm. 2013;83(3):315-21. doi: 10.1016/j.ejpb.2012.10.002, PMID 23178201.
Siddiqui M, Garg G, Sharma PK. A short review on a novel approach in oral fast-dissolving drug delivery system and their patents. Adv Biol Res. 2011;5(6):291-303.
Sangnim T, Sriamornsak P, Theerawatcharothai S, Fugnoot K, Wattanathammanon P, Vongsak B. Development of fast dissolving orodispersible films loaded with cannabis extract. Sci Eng Health Stud. 2022;16:e22050023. doi: 10.14456/sehs.2022.60.
Hiremath J, Sarfaraz M, Hiremath D, Sarudkar S. Preparation and physicochemical characterization of simvastatin-loaded mucoadhesive bilayered tablet. Indian J Nov Drug Deliv. 2009;1(1):18-24.
United States Pharmacopeia and National Formulary (USP 47-NF 42). Rockville (MD): United States Pharmacopeial Convention. In: United States Pharmacopeial Convention. Pharmaceutical dosage forms; 2024.
Anil A, Sudheer P. Design and evaluation of mucoadhesive buccal patch of ramipril. Int J Pharm Sci Rev Res. 2018;50(2):121-9.
Deshmane SV, Channawar MA, Chandewar AV, Joshi UM, Biyani KR. Chitosan based sustained release mucoadhesive buccal patches containing verapamil HCl. Int J Pharm Pharm Sci. 2009;1(1):216-29.
El Kamel AH, Ashri LY, Alsarra IA. Micromatricial metronidazole benzoate film as a local mucoadhesive delivery system for treatment of periodontal diseases. AAPS PharmSciTech. 2007;8(3):E75. doi: 10.1208/pt0803075, PMID 17915825.
Kumar A, Phatarpekar V, Pathak N, Padhee K, Garg M, Sharma N. Formulation development and evaluation of carvedilol bioerodable buccal mucoadhesive patches. Int J Compr Pharm. 2011;3(7):1-5.
Boyes RN, Perkins B, Strobel JN, Dunn RL. Noven pharmaceuticals inc assignee. Buccal local anaesthetic. World Patent WO 1989010740; 1989 Nov 16.
Flockhart IR, Cort JH. Cort Jitka East Riding Laboratories Ltd, Assignee. Desmopressin Buccal Patch Composition. United States Patent US5298256A; 1994 Apr 29.
Kurkela K, Marvola M, Larma I, Virtanen R, Karlsson M. Orion corporation assignee. Transmucosal formulations of levosimendan. United States Patent US6399610B1; 2002 Apr 4.
Repka MA, Repka SL, McGinity JW. Board of regents the University of Texas System Assignee. Bioadhesive hot-melt extruded film for topical and mucosal adhesion applications and drug delivery and process for preparation thereof. United States Patent US6375963B1; 2002 Apr 23.
Haley J. Soft adherent soluble oral patch. United States patent application US20030124178A1; 2003 Jul 3.
Zerbe HG, Guo JH, Serino A. LTS lohmann therapie-systeme AG, assignee. Water soluble film for oral administration with instant wettability. United States Patent US6709671B2; 2004 Mar 23.
Rademacher T, Falkenhausen CV. LTS Lohmann Therapie Systeme AG, Assignee. Transmucosal form of administration with reduced mucosal irritation. World Patent WO2005000263A1; 2005 Jan 6.
Durschlag ME, Kehoe GS. Edible film for transmucosal delivery of nutritional supplements. World patent WO 006119286A1; 2006 Nov 9.
Fuisz RC. Aquestive therapeutics inc assignee. Film bandage for mucosal administration of actives. World patent WO2007084617A2; 2007 Jul 27.
Haber M, Kristmundsdottir T, Skulason S, inventors. Biota Ltd, assignee. Orally administrable films and preparation thereof. World Patent WO2007125533A2; 2007 Nov 8.
Biegajski JE. Dow pharmaceutical sciences inc assignee. Two-phase mucoadhesive composition. World patent WO2008002353A1; 2008 Jan 3.
Malcovati L. Canker sore patch. World Patent WO2010006612A3; 2010 Jan 21.
Daud AS, Sapkal NP, Bonde MN. Water soluble pharmaceutical film with enhanced stability. World patent WO2015083181A2; 2015 Jun 11.
McConville JT, Morales JO, Ross A. UNM Rainforest innovations assignee. Bioadhesive films for local and/or systemic delivery. United States Patent Application US20160128947A1; 2016 May 12.
Kovarik JE. Seed health inc assignee. Buccal bioadhesive strip and method of treating snoring and sleep apnea. United States patent application US9549842B2; 2017 Jan 24.
Myers GL, Hilbert SD, Boone BJ, Bogue BA, Sanghvi P, Hariharan M. Indivior UK Ltd, assignee. Sublingual and buccal film compositions. United States Patent Application US9687454B2; 2017 Jun 27.
Scaife MC. Sublingual or buccal administration of dim for treatment of skin diseases. World patent WO2018051183; 2018 Mar 22.
Mavely L, Sonaje K, Indu AG, inventors. Axio biosolutions Pvt Ltd, assignee. Mucoadhesive preparations methods and applications thereof. World Patent WO2018083643A1; 2018 May 11.
Bogdahn M. Esocap AG, assignee. Pharmaceutical dosage form for application to mucous membranes. United States Patent Application US20180036251A1; 2018 Feb 8.
Werle M, Foger F. Cyprumed GmbH, assignee. Pharmaceutical compositions for the transmucosal delivery of therapeutic peptides and proteins. World Patent WO2019193204A1; 2019 Oct 10.
Yang RK, Fuisz RC, Myers GL, Fuisz JM. Aquestive therapeutics inc, assignee. Thin film with non-self-aggregating uniform heterogeneity and drug delivery systems made therefrom. United States Patent Application US20200163901A1; 2020 May 28.
Mallery SR, Larsen PE, Stoner GD, Schwendeman SP, Desai KG. Ohio State University, assignee. Controlled release mucoadhesive systems. United States Patent Application US10758619B2; 2020 Sep 1.
Tarate VS, Chougule PN, Chougule NB, Shinde VR, Shelke SS, Kulkarni AS. Multilayered buccal patch of antihypertensive drugs. South Africa Patent ZA 202213524B; 2023 May 31.
Toong HD, Altmann WC. Neurostim technologies LLC, assignee. Detection and treatment of obstructive sleep apnea. United States Patent Application US20230083643A1; 2023 Mar 16.
Badawi P. Sight sciences inc assignee. Dry eye treatment systems. United States Patent Application US20250228702A1; 2025 Jul 17.
Badawi D, Badawi P, Harshman S, O Keeffe D, inventors. Sight sciences inc assignee. Controller for dry eye treatment systems. United States Patent Application US12257181B2; 2025 Mar 25.
Food and Drug Administration (U. S.). Combination products. 21 C.F.R. Part 3. Washington, DC: U.S. Government Publishing Office; 2020.
Leoty Okombi S, Gillaizeau F, Leuillet S, Douillard B, Le Fresne Languille S, Carton T. Effect of sodium lauryl sulfate (SLS) applied as a patch on human skin physiology and its microbiota. Cosmetics. 2021;8(1):6. doi: 10.3390/cosmetics8010006.
Moghimipour E, Ameri A, Handali S. Absorption enhancing effects of bile salts. Molecules. 2015;20(8):14451-73. doi: 10.3390/molecules200814451, PMID 26266402.
Pat Y, Ogulur I, Yazici D, Mitamura Y, Cevhertas L, Kucukkase OC. Effect of altered human exposome on the skin and mucosal epithelial barrier integrity. Tissue Barriers. 2023;11(4):2133877. doi: 10.1080/21688370.2022.2133877, PMID 36262078.
Maher S, Geoghegan C, Brayden DJ. Safety of surfactant excipients in oral drug formulations. Adv Drug Deliv Rev. 2023;202:115086. doi: 10.1016/j.addr.2023.115086, PMID 37739041.
Reuther M, Rollet N, Debeaufort F, Chambin O. Orodispersible films prepared by hot-melt extrusion versus solvent casting. Int J Pharm. 2025;675:125536. doi: 10.1016/j.ijpharm.2025.125536, PMID 40164416.
Juarez Navarro KJ, Guarino V, Alvarez Perez MA. Converging electrospinning and 3D-printing technologies: from innovative design for tissue engineering to global patent trends and technology transfer. Fibers. 2025;13(6):83. doi: 10.3390/fib13060083.
Carou Senra P, Rodriguez Pombo L, Awad A, Basit AW, Alvarez Lorenzo C, Goyanes A. Inkjet printing of pharmaceuticals. Adv Mater. 2024;36(11):e2309164. doi: 10.1002/adma.202309164, PMID 37946604.
Mersanne A, Foresti R, Martini C, Caffarra Malvezzi C, Rossi G, Fornasari A. In-house fabrication and validation of 3D-printed custom-made medical devices for planning and simulation of peripheral endovascular therapies. Diagnostics (Basel). 2024;15(1):8. doi: 10.3390/diagnostics15010008, PMID 39795536.