
Int J App Pharm, Vol 18, Issue 1, 2026, 62-70Reviewl Article
LIMITATION AND ADVANCE IN THE DEVELOPMENT OF ORODISPERSIBLE FILMS FOR DRUG DELIVERY
B. MUKESH, M. DHIYANESHWARAN, C. GOKUL KUMAR, N. SHABARISHA, M. V. SARANYA, G. RAMYA, K. THAMARAISELVI, ARUN RADHAKRISHNAN*
Department of Pharmaceutics, JKKN College of Pharmacy, Kumarapalayam-638183, Namakkal, Tamil Nadu, India
*Corresponding author: Arun Radhakrishnan; *Email: arunpharma93@gmail.com
Received: 24 Jul 2025, Revised and Accepted: 20 Nov 2025
ABSTRACT
Orodispersible films (ODFs) are increasingly recognized as an important innovation in drug delivery, as they offer quick disintegration, better bioavailability, and greater comfort for patients. They are especially beneficial for children, the elderly, and those who have challenging in swallowing, since ODFs dissolve in the mouth and allow drugs to be absorbed through a sublingual or buccal route, avoiding metabolism by the liver. This review discusses the development of ODFs in detail, including formulation strategies, manufacturing methods, the current market landscape, and limitations that still affect these systems. The discussion incorporates significant details from an extensive review of recently published papers and patents on the subject, focusing on material screening, design considerations, and production processes. The review discusses physicochemical properties, stability issues, regulatory considerations, and challenges associated with industrial production, typically from pharmaceutical and engineering perspectives. While a number of advances have been made, challenges such as limited drug load and stability issues, and product scale-up still remain. This article attempts to identify these gaps to suggest areas for future research to improve performance, scale-up and commercialisation to enable ODFs for patients in need of products other than a conventional dosage form.
Keywords: Orodispersible films (ODFs), Personalized medicine, Bioavailability, Patient compliance, Polymer
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.56232 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
The oral route is currently considered the gold standard in the pharmaceutical industry due to its perceived superiority in patient compliance, cost-effectiveness, and convenience for drug delivery. Owing to its high patient adherence compared to alternative routes, the oral route remains the predominant form of administration for the majority of therapeutic agents intended to elicit systemic effects [1].

Fig. 1: Administration of an Orodispersible film, *This fig. was created using AI tools for academic and illustrative purposes
In recent years, orodispersible films have gained recognition as an alternative to conventional dosage forms. These thin oral films containing Active Pharmaceutical Ingredients (API) can rapidly release medication through oral or sublingual administration, providing both systemic and local effects [2].
There are numerous additional names for these thin films, such as oral soluble films, mucoadhesive films, buccal films, wafers, ocular films, transmucosal films, oral strips, orodispersible films [ODFs], and oral thin films. The Food and Drug Administration (FDA) simply refers to it as a soluble film, but the European Medicines Agency defines an orodispersible film as one that dissolves readily in the oral cavity [3-5].
Scope and Purpose
Orodispersible films (ODFs) are gaining importance as a potent drug delivery system because of their simplicity of use, rapid disintegration, and potential to increase drug absorption [6, 7]. ODFs are considered a precious alternative to conventional solid and liquid oral dosage forms.
ODFs are being proposed as an alternative to the conventional oral dosage forms, which can be solid or liquid. Orodispersiblefilms, also known as oral drug strips or dissolving films, release medications through oral absorption and have a mechanism that delivers the medication straight to the bloodstream. For first-pass metabolism drugs, it is an alternative platform [8].
For drug delivery in patients with dysphagia of conventional dosage forms like capsules or tablets, orodispersible films (ODFs) are being formulated as a quick, efficient, and patient-friendly mode of drug delivery [9].
Market data and statistics on ODF adoption trends
In recent years, the orodispersible film (ODF) market has demonstrated robust growth, driven by increasing demand for patient-friendly dosage forms and advancements in film technologies. As illustrated in fig. 2, industry analyzes project the global ODF market value to rise from USD 3.19 billion in 2024 to USD 3.52 billion in 2025, and further to USD 7.98 billion by 2033, reflecting a Compound Annual Growth Rate (CAGR) of approximately 9.45 percent. This upward trajectory underscores expanding applications in pharmaceuticals, nutraceuticals, and personalized medicine, particularly in developing economies where ease of administration and rapid onset of action are prioritized.
Market forecasts indicate that ODFs will sustain significant growth through the next decade, propelled by factors such as rising prevalence of chronic diseases, expansion of RandD activities, and consumer preference for fast-dissolving, water-free dosage forms. Fig. 2 presents the estimated global market value of ODFs from 2024 through 2033, highlighting key milestones in market expansion and projected revenue trends [13].
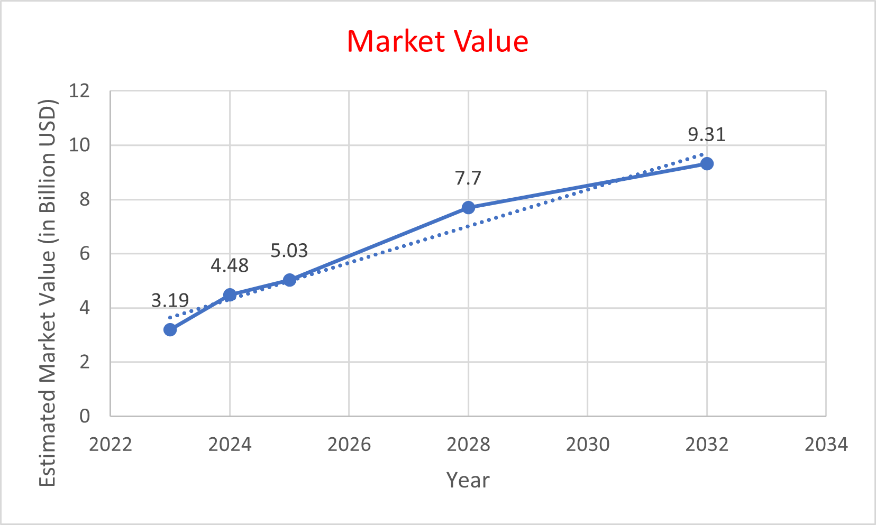
Fig. 2: Projected global market value of orodispersible films (ODFs) from 2023 to 2033 [10, 11]
Significance of ODFs in improving patient outcomes
Orodispersible films (ODFs) come with some great benefits that can really help patients. They're especially handy for folks who struggle with regular pills or liquids since they're easy to take and taste better. Because ODFs dissolve quickly in the mouth, the medicine gets absorbed faster, which means that it starts working sooner [12].
They also bypass the stomach, which can help ensure a more consistent dosage, particularly for conditions that require immediate relief, such as pain, allergies, or mental health issues [6]. Additionally, patients can more easily adhere to their treatment plans, as these films can be customized to deliver the appropriate dosage for each individual [14, 15].
This review aims to critically evaluate the development, formulation approaches, manufacturing techniques, and commercialization challenges of Orodispersible Films (ODFs), while identifying existing limitations and prospects in the field.
Fig. 3: Entry into systemic circulation, *This fig. was created using AI tools for academic and illustrative purposes
MATERIALS AND METHODS
A comprehensive literature search was conducted across PubMed, Scopus, Web of Science, and Google Scholar databases. The search strategy employed the following keywords: orodispersible films, oral thin films, mucoadhesive buccal films, 3D-printed films, nanoparticle-embedded films, hot-melt extrusion, and solvent casting. Articles published between January 2000 and June 2025 were considered to capture both foundational developments and the most recent advances.
RESULTS
Evolution of Orodispersible Films
Numerous orodispersible films (ODFs) have received FDA approval for both prescription and over-the-counter medications.
Although ODFs have existed since the 1970s, they began to gain popularity and recognition as a drug delivery system only after Pfizer introduced Listerine PocketPaks®-refreshing breath strips-in 2001 [16].
ODF milestones and innovations
The development of orodispersible films (ODFs) has experienced significant turning points in their evolution, progressing from a novel concept to an advanced drug delivery system. ODFs were initially proposed as a new oral dosage form; however, their prominence emerged in the early 2000s when the FDA approved the first thin film formulations for over-the-counter products. Recent innovations have expanded ODFs beyond being merely an alternative dosage form, enabling accurate dose customization through technologies such as 3D printing and personalized medicine. ODFs represent next-generation pharmaceutical technology, offering a wide range of potential clinical applications due to the growing market demand for easy-to-use and effective drug delivery systems [17-19].
The orodispersible film (ODF) market has evolved significantly through its patient-centric design capability, offering rapid dissolution and no need for water, which is convenient for patients in specific populations, including pediatric, geriatric, and dysphagic patients. ODFs are widely used in therapeutic areas including pain management, allergy, and neurological indications. Having traced the key milestones that have shaped ODF development and footpaths into personalized formulation, we now turn to the chemical and mechanical considerations that underlie their formulation.
Formulation strategies
The process of formulating orodispersible films (ODFs) involves selecting polymers, plasticizers, active pharmaceutical ingredients (APIs), and other excipients to achieve optimal properties. An ideal ODF should disintegrate quickly, possess sufficient mechanical strength, accommodate a high drug load, and promote patient compliance [20].
Table 1: ODF milestones and innovations
| Period | Breakthrough | Details |
| Pre-2010 | Early Development | 2001: Pfizer introduced Listerine PocketPaks, the first commercial ODF product, used as a breath freshener. 2010: FDA approved Zuplenz (ondansetron) for nausea and vomiting, and Suboxone (buprenorphine/naloxone) for opioid dependence, validating ODFs as a drug delivery system. |
| 2010–2020 | Technological Advancements | 2015: The European Pharmacopoeia and USP formally defined ODFs, providing regulatory clarity. 2018: Improved solvent casting and hot-melt extrusion enhanced film properties like flexibility, tensile strength, and disintegration. 2020: 3D printing of ODFs enabled personalized medicine, allowing custom dosages for pediatric and geriatric patients |
| 2020–2023 | Innovations in Formulation | 2021: Nanoparticle-embedded ODFs enhanced the bioavailability of poorly soluble drugs. 2022: Taste-masking technologies (e. g., Electronic tongue systems) improved acceptability, particularly for pediatric use. 2023: Integration of natural polymers (chitosan, pullulan) improved ODF stability, biocompatibility, and expanded applications in nutraceuticals. |
| Recent Innovations | Expansion into the Food Industry | 2024: ODFs gained traction as carriers for dietary supplements, vitamins, and bioactive compounds like curcumin and melatonin. 2025: ODFs are expanded into the food industry as carriers for dietary supplements, vitamins, and bioactive compounds. |
| Future Challenges | Future Prospects and Challenges | Challenges remain, including limited drug-loading capacity, sensitivity to humidity, and the need for standard quality control methods. Research continues on new applications in mental health, oncology, and personalized medicine. |
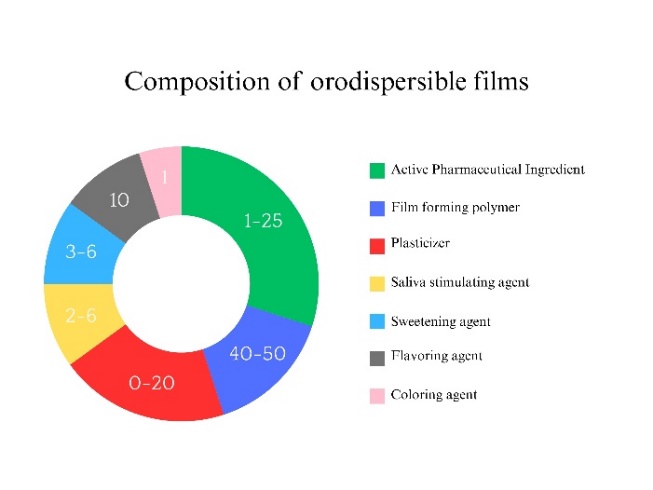
Fig. 4: Schematic representation of the typical composition of orodispersible films [20]
As illustrated in fig. 4, the typical composition of an ODF integrates these excipient classes in a balanced formulation to achieve rapid film disintegration (<30 sec), adequate tensile strength (≥10 MPa), and content uniformity (85–115%).
Active pharmaceutical ingredient (API)
Biopharmaceutics Classification System (BCS) Class I medications are the most commonly used in oral dissolving films (ODFs) due to their high solubility and permeability compared to other drug classes. The active pharmaceutical ingredient (API) can only be incorporated into medications considered potent, given the film’s low weight (less than 200 mg). Factors such as solubility, pharmacokinetic properties, and physical and chemical stability all influence the selection of APIs. It is possible to use 1% to 25% w/w of API, in the form of solid dispersions or nanocrystals, to develop an ODF with a total film weight under 200 mg. Various drug types-such as antibiotics, central nervous system agents (including antidepressants), and inhalable formulations like pressurized metered-dose inhalers (pMDIs)-have been explored in relation to ODFs [23].
Novel techniques aimed at enhancing solubility and bioavailability, such as cyclodextrin complexation, nanosuspensions, and wet milling to reduce particle size, continue to be evaluated. 3D printing technologies, such as Fused Deposition Modeling (FDM), improve the dissolution properties of the active pharmaceutical ingredient (API) by allowing the formation of porous films or by amorphizing the API. Multilayered ODFs for combination therapy are a very recent concept but present significant challenges due to limitations in drug loading [24].
In advanced ODF formulations, multilayer structures separate functions across distinct strata-mechanical support, drug release, and mucoadhesion-thereby overcoming limitations of single-layer films such as weak tensile strength or active substance incompatibility. A representative schematic of this architecture is shown in fig. 5.
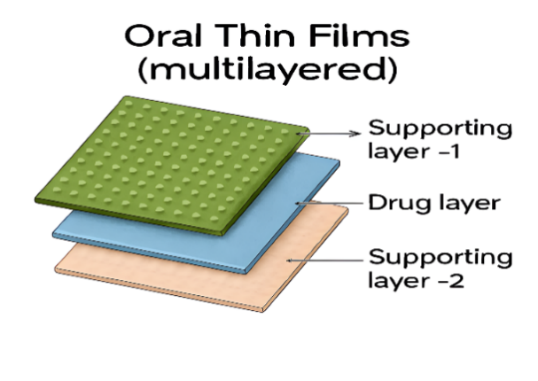
Fig. 5: Layers of orodispersible films, *This fig. was created using AI tools for academic and illustrative purposes
Orodispersible films have progressively evolved beyond single-layer formulations to multilayer architectures that enable separation of incompatible actives, enhanced mechanical integrity, and controlled release.
Excipients
Ideal orodispersible films (ODFs) for future applications must be carefully considered in terms of composition and formulation techniques to improve patient experience and medication delivery [2].
Table 2: Typical excipients used in orodispersible films (ODF) preparation
| Excipients | Examples of excipients | References |
| Polymer | Natural: Pullulan, gelatin, maltodextrin, hydroxypropyl methyl cellulose, chitosan, sodium alginate Synthetic: carboxymethyl cellulose, hydroxypropyl cellulose, polyvinyl pyrrolidone |
[25] |
| Plasticizers | Glycerin, sorbitol, mannitol, propylene glycol | [31] |
| Surfactants | Sorbitanoleate, glycerilmonolinoleate | [50] |
| Taste-masking agents | Essential oil natural or synthetic flavors, cyclodextrin | [22] |
| Sweeteners | Sucrose, fructose sucralose, saccharin, cyclamate, acesulfame K | [35] |
| Coloring Agents | Titanium dioxide | [39] |
| Stabilizing Agents | Hydroxypropyl cellulose (HPC), polyvinyl pyrrolidone (PVP), xanthan gum, sodium alginate | [41] |
| Saliva stimulators | Citric acid, malic acid, tartaric acid | [37] |
Polymer
The film-forming polymer is the most important component of an ODF, constituting the largest proportion of the film’s dry weight (40–50%). A film-forming polymer provides mechanical strength and, more importantly, the film-forming properties [25]. Common film-forming polymers include Hydroxypropyl Methylcellulose (HPMC) (E3, E5, E15), Sodium Carboxymethyl Cellulose (NaCMC), gelatin, Polyvinyl Alcohol (PVA), Polyvinylpyrrolidone (PVP), Polyethylene Oxide (PEO), pullulan, starch derivatives, and various gums [26–30]. The performance of film-forming polymers in orodispersible films varies significantly under different environmental and formulation conditions, necessitating comprehensive evaluation across multiple scenarios [25, 26].
Moisture sensitivity
HPMC exhibits superior moisture resistance among hydrophilic polymers, retaining structural integrity at 75% RH, while mannitol coating further reduces water uptake by ~40% for moisture-sensitive APIs [27]. PVA shows high moisture absorption and rapid disintegration (17–890 s), though tackiness above 65% RH necessitates moisture-barrier packaging. Chitosan demonstrates moderate moisture sensitivity with antimicrobial activity, beneficial for pediatric formulations. Gelatin absorbs>15% moisture but preserves flexibility more effectively than synthetic polymers under variable humidity [28].
High drug loading
HPMC maintains uniform drug content up to 30% loading (5 mg/1.5 cm²), but higher loadings reduce tensile strength by ~45% and prolong disintegration (>60 s). PVA films allow up to 50% loading; however, uniformity declines beyond 40% (CV>10%). HPMC/PVA (1:1) blends achieve 25–30% loading with tensile strength>2 MPa offering superior performance under high-loading conditions [29].
Pediatric formulations
PVA exhibits excellent biocompatibility for pediatric use with minimal oral irritation, supported by favorable toxicity profiles. HPMC E5 shows superior compatibility with taste-masking agents, maintaining integrity with cyclodextrins and flavors while enabling>85% drug release within 10 min. Chitosan–gelatin blends enhance flexibility (elongation>200%), reducing film breakage during pediatric administration [30].
Plasticizers
The selection and optimization of plasticizers in ODF formulations present a complex interplay between mechanical enhancement and formulation stability that demands critical evaluation beyond traditional empirical approaches. While plasticizers fundamentally reduce glass-transition temperatures and improve film flexibility, their integration creates multifaceted challenges that significantly impact commercial viability and therapeutic performance [31, 32].
The conventional recommendation of 0-20% w/w plasticizer concentration oversimplifies the optimization challenge, as this range fails to account for polymer-specific interactions and environmental stability requirements [32, 33]. Glycerol, despite its widespread use, exhibits a biphasic behavior that undermines long-term stability: at concentrations below 10%, it provides inadequate flexibility leading to film brittleness, while concentrations above 15% create excessive tackiness and moisture absorption that compromises packaging integrity and shelf-life [34]. This narrow therapeutic window highlights a fundamental limitation in current plasticizer selection paradigms [33, 34].
More critically, the industry's reliance on hygroscopic plasticizers like glycerol and propylene glycol creates an inherent contradiction. While these agents improve mechanical properties under controlled conditions, they simultaneously increase moisture sensitivity, the primary cause of ODF degradation in real-world storage conditions. Polyethylene glycol (PEG) variants demonstrate superior moisture resistance but at the cost of reduced compatibility with hydrophilic polymers like HPMC, leading to phase separation during film formation [35].
Plasticizer optimization in ODFs represents a fundamental constraint in aligning regulatory, formulation, and manufacturing requirements. Citrate esters, though safer, demand higher concentrations that reduce drug loading, while co-plasticizer systems lack mechanistic models to guide optimization, limiting predictability and scalability. Manufacturing processes impose divergent demands-solvent casting favors low-viscosity agents at the cost of uniformity, and hot-melt extrusion requires thermal stability often at the expense of flexibility, producing fragmented, process-dependent solutions. The absence of standardized performance metrics specific to ODFs further restricts rational selection, as disintegration, mechanical integrity, and patient acceptability must be optimized simultaneously. Closing this gap requires mechanistic frameworks linking plasticizer–polymer–environment interactions to long-term stability, shifting development from empirical screening toward predictive, scalable formulation strategies.
Sweetening and flavoring agent
Sweeteners are generally incorporated either individually or in combination, with concentrations typically ranging from 3% to 6% w/w. In the case of oral thin films (ODFs), various approaches are employed to mask the bitter or unpleasant taste of active drugs. Recently, researchers have shown considerable interest in the use of lipids because of their potential in flavor-masking applications [22].
Sweeteners used in ODFs are broadly classified into two categories: natural and artificial. Among the natural options, glycyrrhizinic acid-a triterpenoidsaponin obtained from the roots and rhizomes of licorice (Glycyrrhiza glabra)-is well recognized. It has been reported to possess a sweetness intensity at least 30 times greater than that of sucrose.
The natural sweetener rebaudioside A (steviol glycosides), isolated from stevia, has sweetness many times more than that of sucrose, being greater than 200-300 times [36]. The polyol alcohol mannitol is commonly employed as a sugar alcohol sweetener in ODFs due to the fact that it enhances the physical strength of the dosage form. In addition, since mannitol is not hygroscopic and possesses excellent stability and compatibility with active substances, it is useful for ensuring the long-term stability of the dosage form [38].
Saliva-stimulating agent
Saliva stimulant compounds, also known as sialagogues, are substances that may stimulate the mouth to secrete more saliva. Saliva stimulant compounds may benefit individuals who have disorders such as xerostomia, dry mouth, or who may be unable to produce sufficient saliva [38]. Salivation-inducing materials like citric, malic, lactic, ascorbic, and tartaric acids are also typical salivation-inducing materials used in dissolving strip formulations. Typically, separately or together, these chemicals appear with concentrations from 2 to 6% by weight with respect to the film [37].
Coloring agents
Pigments such as titanium dioxide, which impart a lighter or opaque appearance to the film, and iron oxides, which impart red, yellow, or brown colors to the film, are common Coloring agents in ODFs. The film is also colored blue with FD and C approved Coloring agents such as indigo carmine and brilliant blue dyes, green with fast green food Coloring (FCF), and yellow with tartrazine dye. The ODF preparation must have the agents at a maximum concentration of 1% by weight [39].
Stabilizing agents
Stabilizing ingredients are key constituents of ODFs to keep their structural integrity, prevent weakening, and ensure the film's desired properties [40]. Stabilizing chemicals help to enhance the film's flexibility, slipperiness, and quick break-up. Some of the widely used stabilizing ingredients include HPC, PVP, xanthan gum, sodium alginate, locust bean gum, carrageenan, gelatin, sorbitol, and surfactant [41].
With the physicochemical and mechanical properties defined, the focus shifts to how these formulations are realized at scale through various manufacturing techniques.
Manufacturing techniques
The production of orodispersible films (ODFs) is a very complex process that requires accuracy in formulation as well as manufacturing processes to make sure that the end product is efficient, stable, and able to release the Active Pharmaceutical Ingredient (API) at the required therapeutic level. This characteristic maximizes the drug's bioavailability, which enables faster therapeutic response than traditional oral dosage forms like tablets and capsules [42].
Solvent casting method
Solvent casting is the most widely used method for ODF production. The production of small batches of ODF and multi-layer films intended to deliver fixed combinations of medication can also be modified to solvent casting. Physically incompatible Active Pharmaceutical Ingredients (APIs) can be injected at different intensities onto multiple layers of film [41].
The solvent casting technique for preparing orodispersible films (ODFs) has a number of key downsides. First, it is extremely dependent on volatile organic solvents, which cause environmental, safety, and regulatory issues because of their toxicity, flammability, and possible residual presence in the final product. Secondly, obtaining good film thickness and homogeneity is difficult because uneven drying conditions can produce defects such as cracking, peeling, or drug unevenness. Thirdly, the process is inappropriate for heat-labile or moisture-labile Active Pharmaceutical Ingredients (APIs), since the drying step can break down these compounds. Moreover, the addition of high drug loads or low solubility drugs can adversely affect the mechanical strength and disintegration time of the film. Finally, the process is labor-intensive, energy-consuming, and requires special equipment, which makes it less scalable and economical for commercial-scale production [44].
Hot-melt extrusion
For ODFs, hot melt extrusion has also been contemplated as a solvent-free production process. Extrusion certainly has many advantages over solvent casting, such as the removal of solvents and drying. However, the process of melting may affect the stability of the polymer, flavor, or Active Pharmaceutical Ingredient (API) [45].
A major limitation of this process is the high processing temperature, which can degrade temperature-sensitive Active Pharmaceutical Ingredients (APIs) or excipients, thereby restricting their applicability for certain drugs. Moreover, the process demands strict control over parameters such as temperature, screw speed, and pressure, making it complex and requiring advanced equipment and expertise. The choice of suitable thermoplastic polymers and plasticizers is also limited, as they must withstand high temperatures while providing the desired film characteristics, which may reduce formulation flexibility. Additionally, achieving uniform drug distribution and consistent film thickness remains challenging, particularly for drugs with low solubility or high melting points [46].
Electrostatic spray deposition
Electrostatic spray deposition (ESD) for orodispersible films (ODFs) leverages an electrohydrodynamic atomization process in which a polymer–drug solution is pumped through a charged nozzle and breaks into fine, monodisperse droplets that are directed by an electric field onto a grounded substrate. Unlike powder‐based coatings, liquid‐phase ESD enables precise control over film thickness (typically 10–100 µm) and drug loading by adjusting flow rate, applied voltage (5–15 kV), and nozzle–substrate distance (5–20 cm). The process typically employs aqueous or benign organic solvents, reducing residual solvent concerns and accommodating heat‐sensitive active substances. Following deposition, films are dried under controlled humidity (≤ 30% RH) and temperature (25–40 °C) to preserve mechanical integrity and ensure uniform drug distribution. This technique has been used to fabricate ODFs with rapid disintegration (<30 s) and tensile strength ≥ 0.5 MPa, suitable for high‐potency Active Pharmaceutical Ingredients (APIs) in doses up to 20 mg per strip [48].
Limitations of liquid‐phase ESD include the need for precise parameter control to avoid “satellite” droplet formation, scalability constraints at high throughput (>1 m²/h), and sensitivity to solution conductivity and viscosity, which must be optimized (conductivity 0.1–1 mS/cm; viscosity ≤ 200 cP) to maintain droplet stability and deposition uniformity. Despite these challenges, ESD represents a solvent‐efficient, low‐temperature alternative to solvent casting and hot‐melt extrusion, particularly advantageous for personalized or small‐batch ODF manufacturing. Active Pharmaceutical Ingredients [49, 50].
Electrospinning method
The electrospinning process consists of the generation of nanoscale fiber filaments by transforming the spherical structure of polymer solution droplets into a conical structure. Because of their distinctive characteristics and potential applications, electrospun nanofibers have attracted significant interest. Such nanofibers possess excellent mechanical properties, a high surface area to volume ratio, and can be designed to exhibit specific functions such as tissue engineering, filtration, controlled release, and energy storage applications [51].
A multilayer film was successfully fabricated through sequential electrospinning, where ethyl cellulose nanofibers formed the outer layers and curcumin-loaded gelatin nanofibers formed the inner layer [52].
Electrospinning, though a highly promising method of producing orodispersible films (ODFs), has numerous limitations that keep it from extensive use in drug formulations. These include the lack of uniformity in drug distribution and film thickness, among others. Electrospinning is, in most cases, also subject to low product yields and process speeds, limiting its applicability in large-scale production. Maintaining uniform electric field strength and jet stability over large-area collectors is technically demanding; minor fluctuations in voltage or ambient conditions (humidity, temperature) lead to fiber diameter variability and bead formation, compromising film homogeneity and mechanical properties. Throughput is inherently low-single-needle setups yield just milligrams per hour-so multi-needle or needleless configurations introduce complex inter-jet interactions and require precise synchronization to prevent fiber fusion or deposition defects[53]. Electrospinning is perhaps not suited for heat-sensitive or unstable Active Pharmaceutical Ingredients (APIs) since the high voltage and solvent evaporation process might degrade these compounds [54].
Nanotechnology method
Recent advances have integrated various nanoparticulate systems into ODFs to overcome solubility and bioavailability limitations. Self-nanoemulsifying drug delivery systems (SNEDDS) loaded into ODFs form fine oil-in-water emulsions upon contact with saliva, enhancing dissolution of lipophilic drug substances such as curcumin and cannabidiol. Polymeric nanoparticles-commonly fabricated from PLGA, chitosan, or zein-provide controlled drug release and protect heat-or moisture-sensitive drug substances during film formation. Nanocrystals, produced by wet‐milling or precipitation, increase surface area and saturation solubility; for instance, indomethacin nanocrystals embedded in HPMC films achieved a 4-fold faster dissolution rate compared to microsuspensions [55].
However, nanoparticle incorporation poses formulation challenges. Particle aggregation during solvent casting can lead to dose non-uniformity unless stabilizers (e. g., poloxamers, surfactants) are carefully optimized. Polymer–nanoparticle compatibility must be assessed via techniques like DSC and XRD to prevent crystallization or phase separation that could compromise mechanical integrity. Additionally, residual organic solvents used in nanoparticle suspensions may plasticize the film matrix adversely, necessitating stringent solvent removal protocols [56].
3D printing technology
Three-dimensional printing has enabled precise, on-demand manufacture of ODFs with tailored dosage and geometry. Extrusion-based printing (Fused Deposition Modeling, FDM) extrudes drug–polymer filaments (e. g., PVA–paracetamol) at elevated temperatures (~150–180 °C), permitting layer-by-layer construction of films with thicknesses as low as 50 µm. Inkjet printing deposits picoliter droplets of Active Pharmaceutical Ingredient (API) solution onto preformed polymer sheets, offering exceptional resolution (<20 µm) and minimal thermal stress-ideal for thermolabile biologics. Stereolithography (SLA), though less common, uses photopolymerizable resins to achieve sub-10-µm feature sizes, but requires photoinitiators whose safety in oral administration remains under investigation [57].
Higher resolution (e. g., Inkjet) drastically slows production speed, hindering scale-up. Thermal degradation of active substances during FDM calls for thermogravimetric analysis (TGA) screening to select heat-stable drugs or incorporate plasticizers that lower extrusion temperature [58]. Regulatory pathways for 3D-printed ODFs are nascent; current FDA guidance addresses 3D-printed solid oral dosage forms without film-specific criteria. Demonstrating bioequivalence for bespoke geometries and ensuring device validation (e. g., Printer calibration, in-process controls) are critical for market approval [59].
Real-world examples include FDM-printed ondansetron films with variable dose gradients for pediatric use, and inkjet-printed miconazole nitrate ODFs achieving uniform 2 mg doses with rapid disintegration (<15 s). These studies underscore the potential of 3D printing to revolutionize personalized ODF manufacture while highlighting the need for robust quality-by-design frameworks and harmonized regulatory guidelines.
DISCUSSION
The evolution of orodispersible films (ODFs) has brought significant advantages, including enhanced patient compliance due to ease of administration and improved bioavailability through bypassing first-pass metabolism. Technological progress in nanoparticle incorporation and 3D printing allows tailored dosing and improved drug release profiles.
However, formulating ODFs faces inherent challenges such as limited drug loading capacity, especially for poorly soluble or unstable APIs, as discussed in the formulation strategies. Additionally, taste masking and moisture sensitivity impose formulation difficulties impacting palatability and stability.
Manufacturing techniques, while varied, also present limitations. Solvent casting is cumbersome and difficult to scale, whereas hot-melt extrusion is restricted by processing temperatures affecting heat-sensitive drugs. Emerging methods like electrospinning and electrostatic spray deposition offer promise but are hindered by scalability and process control issues.
Regulatory challenges further compound these issues, given the hybrid nature of ODFs as drug-device combinations, requiring extensive validation, which increases development time and costs. Challenges in demonstrating bioequivalence and ensuring consistent quality remain significant barriers to commercialization.
To overcome these limitations, ongoing research into novel polymers and plasticizers, advanced manufacturing technologies with in-line process monitoring, and regulatory frameworks tailored for ODFs is critical. Addressing these will be instrumental in realizing the full potential of ODFs within both pharmaceutical and nutraceutical applications.
Regulatory and commercialization challenges
Orodispersible films (ODFs) have yet to achieve widespread acceptance, primarily due to several regulatory and commercialization challenges. As hybrid drug–device systems, ODFs must undergo extensive and rigorous evaluation by regulatory agencies such as the FDA and the European Medicines Agency (EMA) to ensure safety, efficacy, and quality. The need for advanced manufacturing controls to achieve consistency in drug content, dissolution behavior, and mechanical strength further adds complexity and cost to production. Additional hurdles arise from their novel route of administration, which involves different absorption pathways. This often requires bioequivalence studies to be conducted with highly specific and carefully designed methodologies. From a commercialization standpoint, high manufacturing costs-driven by specialized equipment, the use of moisture-sensitive materials, and difficulties in scaling up production-present significant barriers. Moreover, patient and prescriber perceptions of ODFs as unconventional dosage forms can contribute to lower market acceptance. Beyond these factors, other obstacles include patent restrictions, the necessity of demonstrating cost-effectiveness to qualify for reimbursement, and meeting strict legislative requirements [60, 61].
Content uniformity for low-dose active pharmaceutical ingredients (APIs)
ODFs often incorporate potent active substances at low dose levels (<25 mg/film), making content uniformity critical. The United States Pharmacopeia (USP)<905>Uniformity of Dosage Units requires that no more than two individual units may deviate outside ±25% of the label claim, with none outside ±30%. For low-dose films, manufacturers must demonstrate robust process controls, such as in-line near-infrared (NIR) or Raman spectroscopy-to monitor API distribution during film formation. For example, Zuplenz® (ondansetron ODF) underwent extensive content uniformity studies, employing a twin-screw solvent-casting process and validated NIR end-point detection to ensure each strip met USP<905>criteria [62].
Bioequivalence definitions for local vs. systemic delivery
Unlike orally disintegrating tablets, ODFs can deliver drugs via buccal or sublingual absorption (local) or via gastrointestinal uptake post-swallowing (systemic). Regulatory guidance for ODF bioequivalence is often extrapolated from Orally Disintegrating Tablet (ODT) guidelines:
FDA: 21 CFR 320.25 permits a pharmacokinetic study when systemic absorption predominates, using Cmax and AUC comparisons against the reference product [63].
EMA: The Reflection Paper on the Use of in vitro–in vivo correlations for Orally Disintegrating Dosage Forms advises a tailored crossover study, with site-specific sampling (e. g., mucosal versus plasma) where local action is intended [64].
For Suboxone® (buprenorphine/naloxone ODF), a crossover bioequivalence study measured both plasma AUC and local mucoadhesive residence time, demonstrating equivalence in systemic exposure and adequate film retention for buccal delivery [65].
Stability testing for moisture-sensitive matrices
ODFs’ polymeric matrices (e. g., HPMC, pullulan) are highly hygroscopic, making moisture uptake a major stability risk. The International Council for Harmonisation (ICH) Q1A(R2) guideline on stability testing must be supplemented by:
Accelerated moisture stress at 25 °C/75% RH and 40 °C/75% RH, with periodic testing of disintegration time, tensile strength, and API assay.
Real-time packaging studies, evaluating both primary (blister foil with desiccant) and secondary (carton) systems for moisture barrier performance.
Franceschinis et al. demonstrated that pullulan-based ODFs maintained<5% weight gain after 6 mo at 25 °C/60% RH only when packaged in alu-alu blisters with desiccant sachets [66].
Harmonization and regulatory pathways in emerging markets
Emerging-market regulators (e. g., India’s CDSCO) often lack specific ODF guidelines but refer to ICH Q8–Q10. Harmonization efforts by the International Generic Drug Regulators Program (IGDRP) are promoting:
Common dossier templates require detailed film-formation methodology, in-process controls, and specific packaging validation.
Convergence workshops to align moisture barrier testing and bioequivalence approaches, particularly for low-and middle-income countries where ambient humidity varies widely [67].
CONCLUSION
Orodispersible films are a mature, patient-centric platform that pairs rapid disintegration with bioavailability gains for pediatric, geriatric, and dysphagic patients amid growing pharma and nutraceutical adoption. While the FDA uses soluble film and EMA uses orodispersible film, advances in 3D printing and nanoparticle embedding allow for on-demand microgram dosing, multilayers, and dose gradients. Remaining gaps include scale-up and uniformity limits in solvent casting, electrospinning, and electrostatic spray deposition, moisture sensitivity from hygroscopic matrices, and the need for ODF-specific bioequivalence and quality guidance. Priorities are continuous roll-to-roll hot‑melt extrusion with in-line NIR or Raman to meet USP 905, harmonized humidity‑cycling and mechanical‑stress stability models, robust taste masking with saliva stimulants and barrier packaging, and collaborative pathways with clear criteria for systemic or local action and for personalized 3D‑printed nanoparticle‑enabled products.
ABBREVIATIONS
ODFs: Orodispersible Films, API: Active Pharmaceutical Ingredient, FDA: Food and Drug Administration, EMA: European Medicines Agency, ICH: International Council for Harmonisation, HPMC: Hydroxypropyl Methyl Cellulose, PVA: Polyvinyl Alcohol, PVP: Polyvinyl Pyrrolidone, PEO: Polyethylene Oxide, NaCMC: Sodium Carboxymethyl Cellulose, ODTs: Orally Disintegrating Tablets, BCS: Biopharmaceutics Classification System, PEG: Polyethylene Glycol, GI: Gastrointestinal, 3D: Three-Dimensional, FDM: Fused Deposition Modeling, ESD: Electrostatic Spray Deposition, QbD: Quality by Design, GMP: Good Manufacturing Practice, CAGR: Compound Annual Growth Rate
FUNDING
Nil
AUTHORS CONTRIBUTIONS
B. Mukesh, A. M. Diyaneshwaran, N. Sabharisha, C. GokulKumar contributed to conceptualization, formal analysis, methodology, software, original draft, review, and editing. Arun Radhakrishnan, G. Ramya, K. Thamaraiselvi contributed to the investigation, visualization, review writing, and editing.
CONFLICTS OF INTERESTS
All authors have none to declare.
REFERENCES
Guo B, Sofias AM, Lammers T, Xu J. Image-guided drug delivery: nanoparticle and probe advances. Adv Drug Deliv Rev. 2024;206:115188. doi: 10.1016/j.addr.2024.115188, PMID 38272185.
Rathod A, Sonawane M, Pawar K, Nikam V, Tamboli A. Innovative drug delivery system oral fast dissolving film: a comprehensive review. RJPDFT. 2025;17(2):149-63. doi: 10.52711/0975-4377.2025.00022.
Zaki RM, Alfadhel M, Devanatha Desikan Seshadri VD, Albagami F, Alrobaian M, Tawati SM. Fabrication and characterization of orodispersible films loaded with solid dispersion to enhance rosuvastatin calcium bioavailability. Saudi Pharm J. 2022;31(1):135-46. doi: 10.1016/j.jsps.2022.11.012, PMID 36685296.
Garsuch V, Breitkreutz J. Novel analytical methods for the characterization of oral wafers. Eur J Pharm Biopharm. 2009;73(1):195-201. doi: 10.1016/j.ejpb.2009.05.010, PMID 19482082.
Hoffmann EM, Breitenbach A, Breitkreutz J. Advances in orodispersible films for drug delivery. Expert Opin Drug Deliv. 2011;8(3):299-316. doi: 10.1517/17425247.2011.553217, PMID 21284577.
Jacob S, Boddu SH, Bhandare R, Ahmad SS, Nair AB. Orodispersible films: current innovations and emerging trends. Pharmaceutics. 2023;15(12):2753. doi: 10.3390/pharmaceutics15122753, PMID 38140094.
Sharma R, Joshi D, Singh B, Semwal N. A review on orodispersible film a novel approach of drug delivery system. World J Bio Pharm Health Sci. 2022;12(1):1-11. doi: 10.30574/wjbphs.2022.12.1.0130.
Chen F, Liu H, Wang B, Yang L, Cai W, Jiao Z. Physiologically based pharmacokinetic modeling to understand the absorption of risperidone orodispersible film. Front Pharmacol. 2020;10:1692. doi: 10.3389/fphar.2019.01692, PMID 32116683.
He M, Zhu L, Yang N, Li H, Yang Q. Recent advances of oral film as platform for drug delivery. Int J Pharm. 2021;604:120759. doi: 10.1016/j.ijpharm.2021.120759, PMID 34098053.
Borges AF, Silva C, Coelho JF, Simoes S. Oral films: current status and future perspectives: I Galenical development and quality attributes. J Control Release. 2015;206:1-19. doi: 10.1016/j.jconrel.2015.03.006, PMID 25747406.
Straits Research. Oral thin films market size share and demand by 2033. Report Code SRHI3641DR. 2025 May. p. 110. Available from: https://straitsresearch.com/report/oral-thin-films-market. [Last accessed on 07 Oct 2025].
Badekar R, Bodke V, Tekade BW, Phalak SD. An overview on oral thin films-methodology characterization and current approach. Int J Pharm Pharm Sci. 2024;16(4):1-10. doi: 10.22159/ijpps.2024v16i4.50386.
Cupone IE, Sansone A, Marra F, Giori AM, Jannini EA. Orodispersible film (ODF) platform based on maltodextrin for therapeutical applications. Pharmaceutics. 2022;14(10):2011. doi: 10.3390/pharmaceutics14102011, PMID 36297447.
Ruchika KN, Khan N, Dogra SS, Saneja A. The dawning era of oral thin films for nutraceutical delivery: from laboratory to clinic. Biotechnol Adv. 2024;73:108362. doi: 10.1016/j.biotechadv.2024.108362, PMID 38615985.
Musazzi UM, Khalid GM, Selmin F, Minghetti P, Cilurzo F. Trends in the production methods of orodispersible films. Int J Pharm. 2020;576:118963. doi: 10.1016/j.ijpharm.2019.118963, PMID 31857185.
Ferlak J, Guzenda W, Osmalek T. Orodispersible films-current state of the art, limitations advances and future perspectives. Pharmaceutics. 2023;15(2):361. doi: 10.3390/pharmaceutics15020361, PMID 36839683.
Li Y, Zhao M, Zhao MY, Li B, Tian JL. Advances in oral dissolving film research in the food field. Food Prod Process and Nutr. 2025;7(1):1-15. doi: 10.1186/s43014-024-00285-X.
Palezi SC, Fernandes SS, Martins VG. Oral disintegration films: applications and production methods. J Food Sci Technol. 2022 Sep;60(10):2539-48. doi: 10.1007/s13197-022-05589-9, PMID 37599841.
Khalid GM, Selmin F, Musazzi UM, Gennari CG, Minghetti P, Cilurzo F. Trends in the characterization methods of orodispersible films. Curr Drug Deliv. 2021;18(7):935-46. doi: 10.2174/1567201818999201210212557, PMID 33305704.
Singh Rawat B, Badola A, Kumar Rajput S, Semalty A, Kumar R, Pandey S. A review on orodispersible film-based novel drug delivery system. Res J Pharm Technol. 2024;17(7):3480-8. doi: 10.52711/0974-360X.2024.00544.
Banerjee S, Joshi U, Singh A, Saharan VA. Lipids for taste masking and taste assessment in pharmaceutical formulations. Chem Phys Lipids. 2021;235:105031. doi: 10.1016/j.chemphyslip.2020.105031, PMID 33352198.
Sannala CK, Mac Lean C, Larsen F, Van OS S, Jadhav P, Shore N. A model-informed drug development approach to design a phase 3 trial of teverelix drug product in advanced prostate cancer patients with increased cardiovascular risk. Clin Pharmacol Drug Dev. 2024;13(8):915-29. doi: 10.1002/cpdd.1415, PMID 38757461.
Tian Y, Lin J, Jing H, Wang Q, Wu Z, Duan Y. Recent progress in orodispersible films-mediated therapeutic applications: a review. Med Comm. 2023;2(2):e1034. doi: 10.1002/mba2.34.
Sinha S, Sonali, Garg V, Thapa S, Singh S, Chauhan M. Empagliflozin containing chitosan-alginate nanoparticles in orodispersible film: preparation, characterization pharmacokinetic evaluation and its in vitro anticancer activity. Drug Dev Ind Pharm. 2022;48(7):279-91. doi: 10.1080/03639045.2022.2108829, PMID 35913103.
Pacheco MS, Barbieri D, Da Silva CF, De Moraes MA. A review on orally disintegrating films (ODFs) made from natural polymers such as pullulan, maltodextrin starch and others. Int J Biol Macromol. 2021 May 1;178:504-13. doi: 10.1016/j.ijbiomac.2021.02.180, PMID 33647337.
Mishra A, Pathak AK. Plasticizers: a vital excipient in novel pharmaceutical formulations. Curr Res Pharm Sci. 2017 May;7(1):1-10. doi: 10.24092/CRPS.2017.070101.
Abdelkader H, Abdel Aleem JA, Mousa HS, Elgendy MO, Al Fatease A, Abou-Taleb HA. Captopril polyvinyl alcohol/sodium alginate/gelatin-based oral dispersible films (ODFs) with modified release and advanced oral bioavailability for the treatment of pediatric hypertension. Pharmaceuticals (Basel). 2023;16(9):1323. doi: 10.3390/ph16091323, PMID 37765131.
Chen K, Sun Y. Development and optimization of oral dissolution films for enhanced delivery of ebastine-loaded solid lipid nanoparticles. Int J Nanomedicine. 2025;20:6963-81. doi: 10.2147/IJN.S521504, PMID 40462833.
Suksawat T, Brniak W, Lyszczarz E, Wesoly M, Ciosek Skibinska P, Mendyk A. Orodispersible dosage forms with Rhinacanthin rich extract as a convenient formulation dedicated to pediatric patients. Pharmaceuticals (Basel). 2024;17(8):994. doi: 10.3390/ph17080994, PMID 39204099.
Wang C, Li Z, Zhai H, Shen X, Li F, Zhang Q. Targeted blocking of EGFR and GLUT1 by compound H reveals a new strategy for treatment of triple-negative breast cancer and nasopharyngeal carcinoma. Eur J Pharm Sci. 2024;198:106789. doi: 10.1016/j.ejps.2024.106789, PMID 38710335.
Liew KB, Tan YT, Peh KK. Effect of polymer plasticizer and filler on orally disintegrating film. Drug Dev Ind Pharm. 2014;40(1):110-9. doi: 10.3109/03639045.2012.749889, PMID 23311593.
Olechno K, Basa A, Winnicka K. “Success depends on your backbone”-about the use of polymers as essential materials forming orodispersible films. Materials (Basel). 2021;14(17):4872. doi: 10.3390/ma14174872, PMID 34500962.
Kandil H, El Desouky FG. Plasticizer modulation of dynamic mechanical properties and dielectric performance in sodium alginate-based biopolymer films. J Inorg Organomet Polym. 2025;35(7):5790-804. doi: 10.1007/s10904-025-03623-9.
Mathur S, Bulchandan N, Parihar S, Shekhawat GS. Critical review on steviol glycosides: pharmacological toxicological and therapeutic aspects of high potency zero caloric sweetener. Int J Pharmacol. 2017;13(7):916-28. doi: 10.3923/ijp.2017.916.928.
Peteliuk V, Rybchuk L, Bayliak M, Storey KB, Lushchak O. Natural sweetener Stevia rebaudiana: functionalities, health benefits and potential risks. Excli J. 2021;20:1412-30. doi: 10.17179/excli2021-4211, PMID 34803554.
Maharjan S, Adhikari S, KC N, Dura S, Manandhar M. Formulation and evaluation of ondansetron oral dispersible tablet using different natural disintegrants. AJAST. 2021;5(4):82-92. doi: 10.38177/ajast.2021.5410.
Mitchell H. Sweeteners and sugar alternatives in food technology. 2nd ed. Oxford: Wiley-Blackwell; 2007. doi: 10.1002/9780470996003.
Pezik E, Gulsun T, Sahin S, Vural I. Development and characterization of pullulan-based orally disintegrating films containing amlodipine besylate. Eur J Pharm Sci. 2021;156:105597. doi: 10.1016/j.ejps.2020.105597, PMID 33065224.
El Bary AA, Al Sharabi I, Haza A BS. Effect of casting solvent film-forming agent and solubilizer on orodispersible films of a polymorphic poorly soluble drug: an in vitro/in silico study. Drug Dev Ind Pharm. 2019;45(11):1751-69. doi: 10.1080/03639045.2019.1656733, PMID 31416366.
Manda P, Popescu C, Juluri A, Janga K, Kakulamarri PR, Narishetty S. Micronized zaleplon delivery via orodispersible film and orodispersible tablets. AAPS PharmSciTech. 2018;19(3):1358-66. doi: 10.1208/s12249-017-0924-9, PMID 29352403.
Singh N, Sweta S, Jindal S. A concise overview on orodispersible film along with their formulation and characterization technique’s. RJPDFT. 2024;16(1):98-106. doi: 10.52711/0975-4377.2024.00016.
Jannini EA, Vignesh SO, Hassan T. Next-generation pharmaceuticals: the rise of sildenafil citrate ODF for the treatment of men with erectile dysfunction. Ther Deliv. 2025;16(4):365-78. doi: 10.1080/20415990.2024.2445501, PMID 39801170.
Speer I, Preis M, Breitkreutz J. Prolonged drug release properties for orodispersible films by combining hot-melt extrusion and solvent casting methods. Eur J Pharm Biopharm. 2018;129:66-73. doi: 10.1016/j.ejpb.2018.05.023, PMID 29792911.
Reuther M, Rollet N, Debeaufort F, Chambin O. Orodispersible films prepared by hot-melt extrusion versus solvent casting. Int J Pharm. 2025;675:125536. doi: 10.1016/j.ijpharm.2025.125536, PMID 40164416.
Trinh TH, Tran HT. Processing from natural products to effective pharmaceutical drugs: involvement of hot melt extrusion technique. Nat Prod Res. 2025;39(1):1-24. doi: 10.1080/14786419.2025.2538096, PMID 40729595.
Zhang J, Sun X, Shao R, Liang W, Gao J, Chen J. Polycation liposomes combined with calcium phosphate nanoparticles as a non-viral carrier for siRNA delivery. J Drug Deliv Sci Technol. 2015;30:1-6. doi: 10.1016/j.jddst.2015.09.005.
Kang D, Kim J, Kim I, Choi KH, Lee TM. Experimental qualification of the process of electrostatic spray deposition. Coatings. 2019;9(5):294. doi: 10.3390/coatings9050294, PMID 31721973.
Lai W, Di L, Zhao C, Tian Y, Duan Y, Pan Y. Electrospray deposition for electronic thin films on 3D freeform surfaces: from mechanisms to applications. Adv Materials Technologies. 2024;9:2400192. doi: 10.1002/admt.202400192.
Salawi A. An insight into preparatory methods and characterization of orodispersible film-a review. Pharmaceuticals (Basel). 2022;15(7):844. doi: 10.3390/ph15070844, PMID 35890143.
Madapally VD, Pandima Devi M. Fabrication of nanofibres by electrospinning using keratin from waste chicken feathers PVA and AgNPs. Int J Pharm Pharm Sci. 2019;11(8):78-84. doi: 10.22159/ijpps.2019v11i8.33637.
Wang P, Li Y, Zhang C, Feng F, Zhang H. Sequential electrospinning of multilayer ethylcellulose/gelatin/ethylcellulose nanofibrous film for sustained release of curcumin. Food Chem. 2020;308:125599. doi: 10.1016/j.foodchem.2019.125599, PMID 31648098.
Misra R, Fung G, Sharma S, Hu J, Kirkitadze M. Assessment of tunable resistive pulse sensing (TRPS) technology for particle size distribution in vaccine formulations a comparative study with dynamic light scattering. Pharm Res. 2024;41(5):1021-9. doi: 10.1007/s11095-024-03698-y, PMID 38649535.
Luraghi A, Peri F, Moroni L. Electrospinning for drug delivery applications: a review. J Control Release. 2021;334:463-84. doi: 10.1016/j.jconrel.2021.03.033, PMID 33781809.
Garcia Astrain C, Ahmed S, Pavon E, Smyth M, Larraneta E. 3D printed mucoadhesive orodispersible films loaded with nanocrystals for enhanced delivery of poorly soluble drugs. Addit Manuf. 2024;82:104038. doi: 10.1016/j.addma.2024.104038.
Steiner D, Tidau M, Finke JH. Embedding of poorly water-soluble drugs in orodispersible films-comparison of five formulation strategies. Pharmaceutics. 2022;15(1):17. doi: 10.3390/pharmaceutics15010017, PMID 36678646.
Muehlenfeld C, Duffy P, Yang F, Zermeno Perez D, El Saleh F, Durig T. Excipients in pharmaceutical additive manufacturing: a comprehensive exploration of polymeric material selection for enhanced 3D printing. Pharmaceutics. 2024;16(3):317. doi: 10.3390/pharmaceutics16030317, PMID 38543211.
Elbl J, Racaniello GF, Simon MC. Development of 3D printed multi-layered orodispersible films for personalized drug delivery. Drug Deliv Transl Res. 2023;13(4):1234-45. doi: 10.1007/s13346-023-01045-6, PMID 36912345.
Kuril A, Ambekar A, Nimase B, Giri P, Nikam P, Desai H. Exploring the potential of 3D printing in pharmaceutical development. Int J Curr Pharm Sci. 2023;15(6):31-42. doi: 10.22159/ijcpr.2023v15i6.3085.
Borges AF, Silva C, Coelho JF, Simões S. Oral films: current status and future perspectives II intellectual property technologies and market needs. J Control Release. 2015;206:108-21. doi: 10.1016/j.jconrel.2015.03.012, PMID 25776737.
Costa JS. A mini-review on drug delivery through wafer technology. Int J Pharm. 2019;558:178-90. doi: 10.1016/j.ijpharm.2019.01.031.
Ahmed SU, Gorukanti SR, Chowdhury TA; Barr Laboratories Inc. Assignee. Ondansetron orally disintegrating tablets. US Patent US7390503B2; 2008 Jun 24.
US Food and Drug Administration. Guidelines for the conduct of an in vivo bioavailability study. Washington, DC: U.S. Government Publishing Office; 2021. Available https://www.law.cornell.edu/cfr/text/21/320.25. [Last accessed on 07 Oct 2025].
European Medicines Agency. ICH M13A guideline on bioequivalence for immediate-release solid oral dosage forms. In: Amsterdam: European Medicines Agency; 2024. Available from: https://www.ema.europa.eu/en/documents/scientific-guideline/ich-m13a-guideline-bioequivalence-immediate-release-solid-oral-dosage-forms_en.pdf. [Last accessed on 07 Oct 2025].
Sullivan JG, Webster L. Novel buccal film formulation of buprenorphine naloxone for the maintenance treatment of opioid dependence: a 12 w conversion study. Clin Ther. 2015;37(5):1064-75. doi: 10.1016/j.clinthera.2015.02.027, PMID 25823919.
Pinal R. Enhancing the bioavailability of poorly soluble drugs. Pharmaceutics. 2024;16(6):758. doi: 10.3390/pharmaceutics16060758, PMID 38931880.
European Medicines Agency. ICH. In: Stability testing of new drug substances and drug products-scientific guideline. Vol. Q1A(R2). London, UK: European Medicines Agency; 2003. Available from: https://www.ema.europa.eu/en/ich-q1a-r2-stability-testing-new-drug-substances-drug-products-scientific-guideline. [Last accessed on 07 Oct 2025].