
Int J App Pharm, Vol 18, Issue 1, 2026, 582-592Original Article
PREPARATION AND IN VITRO EVALUATION OF CANDESARTAN CILEXETIL –BASED NANOPARTICLES AS FAST DISSOLVING ORAL FILM
INAS F. ABDULRAZZAQ*, MALATH H. OUDAH, ABULFADHEL AL-SHAIBANI, YASMIEN A. SALAL
Department of Pharmaceutics, Faculty of Pharmacy, University of Kufa, Al-Najaf, Iraq
*Corresponding author: Inas F. Abdulrazzaq; *Email: enasf.abdulrazzaq@uokufa.edu.iq
Received: 02 Aug 2025, Revised and Accepted: 31 Oct 2025
ABSTRACT
Objective: The present work aims to formulate candesartan cilexetil (CC) as nanoparticles oral film to enhance solubility of CC, and promote its dissolution rate, and this could produce rapid therapeutic action of drug.
Methods: The nanoparticles prepared by solvent/antisolvent precipitated method then formulated as fast dissolving oral films by casting method. CC nanoparticles formulated by different polymer: drug ratio which was 1:1 and 2:1 hydroxypropyl methylcellulose (HPMC), and polyvinylpyrrolidone (PVP) with different molecular weight like, HPMCE5, HPMCE50, PVPK15 and PVPK30. The effect of polymer type, polymer ratio and injected volume on the size and specific surface area of prepared particle were tested.
Results: The bested formula FCC3 was found with a smallest size equal to 37 nm and specific surface area of 54.66m2/g. The FCC3 formula was dried by freeze drier and tested by field emission scanning electron microscope (FESEM), differential scanning calorimetry (DSC) and X-ray powder diffraction analysis (XRD) to determine surface morphology, crystallinity and compatibility, then formulated as fast dissolving oral film. The drug content in the 4 cm2 prepared film was distributed between 93.00%±1.75 to 98.25%±1.25 and disintegrate in 25 min. The in vitro release of candesartan nanoparticle from oral film was 68% in 2 min and complete to 100% at 4 min.
Conclusion: The results confirmed fast release and better dissolution rate of prepared candesartan nanoparticle than pure drug.
Keywords: Candesartan nanoparticle, Fast dissolving film, Solubility, In vitro release
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.56372 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Solubility of drug is important factor in design of oral dosage form. The poor water solubility leads to poor drug efficiency by effecting on the dissolution and bioavailability of drug [1, 2]. 40% of pharmaceutical ingredients estimated to have poor aqueous solubility [3]. Several methods were used to enhance the solubility of insoluble drug which may contain chemical and physical techniques. Nanonization of drug particles have an excellent chance to solve the solubility problems, improve the bioavailability and therapeutic activity of active pharmaceutical ingredients [4]. The oral thin film can be used buccal and sublingually to improve onset of action, enlarged effectiveness and removing side effects of the drug [5]. The polymeric oral film can be designed to controlled and regulate of many delivery routes, that have efficient role in marketing process. Delivery systems of fast dissolving drug have taking approval and acceptance as novel delivery system, for their simplest of administration and well patient obedience as well as not require water for swallowing thus preferable for elderly and pediatric patients. A small particle size with large surface area principals to fast disintegration and release of the active ingredient and therefore a fast onset of action could be accomplished [6]. Candesartan is widely used in management of hypertension, heart failure, myocardial infarction [7]. Candesartan cilexetil (CC) is white to off-white powder have a melting point equal to 170 °C. Chemically CC (C33H34N6O6) is 2-ethoxy-3-[21-cyclohexyl (1H-tetrazol-5-yl) biphenyl-4ylmethyl]-3H-benzoimiadazole-4-carboxylic acid 1-cyclohexyloxycarbonyloxy ethyl ester, has a molecular weight equal to 610.67. The pure drug practically insoluble in water and sparingly soluble in methanol with a pKa value of 6.0 [8, 9]. CC is prodrug hydrolyzed to candesartan through absorption process from the gastrointestinal tract, is an angiotensin II receptor antagonist [10]. The following study aimed to prepare and evaluate CC nanoparticles loaded fast dissolving film to enhance drug solubility and fasten the onset of action CC nanoparticles that fabricated by casting method.
MATERIALS AND METHODS
Materials
Candesartan cilexetil was purchase from WuxiHexia Chemical Company, Chin. hydroxypropyl methylcellulose (HPMCE5, HPMCE15, HPMCE50) Gromax Chemicals–USA, polyvinylpyrrolidone (PVPK15 and PVPK30) ALPHA Chemika-India. Acetone buying from Romil, UK, Disodium hydrogen orthophosphate, Hydrochloric acid 37% and sodium Chloride from BDH Laboratory England, potassium dihydrogen orthophosphate from Fine Chem-India.
Method
Preparation of CC nanoparticles
Nanoparticles formulas of CC were prepared by solvent/antisolvent precipitation method [11]. A particular amount of pure drug was dissolved in five milliter of acetone as a solvent then the solution injected in to antisolvent solution (50 ml of water at 1:10 ratio for FCC1-FCC8 and 25 ml of water at 1:5 ratio for FCC9-FCC12) containing specific amount of different grade of stabilizer (HPMCE5, HPMCE50, PVPK15 and PVPK30) at 1 ml/min upon injection, Candesartan nanoparticles were produced immediately, the nanoparticles solution sonicated for 5 min be for reading the particle size then dried by using freeze drying lyophilizer to get dry powder of CC nanoparticles. So that the choices formulation was dried using 2% mannitol as a cryoprotectant in a vacuum freeze drier at a controlled temperature of (-50) °C, the operating pump at a pressure lower than 0.055 mmHg for 48–72 h. The constituent and variable preparation conditions of formulas are recorded in table 1.
Evaluation of particle size, specific surface area (SSA) and poly dispersity Index
The average particle size, (SSA) and poly dispersity index measured by using dynamic light scattering ABT-9000 Nano laser that measure the series of size distribution for all formula as a liquid sample before dried.
Evaluate of formulation variables effect on the properties of prepared nanoparticles
Effect of type and concentration of polymer
FCC1-FCC8 formulas were prepared containing different polymers of (HPMCE5, HPMCE50, PVPK15 and PVPK30) with different concentration at polymer: drug percentage of 1:1and 2:1in the formulation of CC nanoparticles.
Table 1: Components of CC nanoparticles formulated with different type of polymers and different polymer drug ratio (1:1, 2:1) in FCC1-FCC8, and different solvent: antisolvent ratio (1:10) in (FCC1-FCC8) and (1:5) in (FCC9-FCC12)
| Formula No. | Candesartan (mg) | HPMCE5 (mg) | HPMCE50 (mg) | PVPK15 (mg) | PVPK30 (mg) | Water (ml) |
| FCC1 | 50 | 50 | 50 | |||
| FCC2 | 50 | 50 | 50 | |||
| FCC3 | 50 | 50 | 50 | |||
| FCC4 | 50 | 50 | 50 | |||
| FCC5 | 50 | 100 | 50 | |||
| FCC6 | 50 | 100 | 50 | |||
| FCC7 | 50 | 100 | 50 | |||
| FCC8 | 50 | 100 | 50 | |||
| FCC9 | 50 | 50 | 25 | |||
| FCC10 | 50 | 50 | 25 | |||
| FCC11 | 50 | 50 | 25 | |||
| FCC12 | 50 | 50 | 25 |
Effect of injected volume
FCC9-FCC12 was prepared to determine the effect of solvent: antisolvent ratio at 1:10 and 1:5 of acetone to water.
Characterization of CC nanoparticles dried powder
Drug content and loading efficiency
The tests was done by taking 16 mg of CC lyophilized nanoparticles and dissolved in dry volumetric flask containing 30 ml of simulated saliva with 0.7% tween 20 and sonicated for 15 min then complete the solution volume to 100 ml with same solvent. The solution filtrated and the amount of drug present in each formula was determined spectrophotometrically using UV-visible spectrophotometer. The loading efficiency of prepared nanoparticles was calculated from the actual and theoretical contents [12].
Field emission scanning electron microscope (FESEM)
CC nanoparticles of selected formula was tested by Field emission scanning electron microscope (MIRA3 TESCAN), it was established by dusting of dried powder directly on carbon tape, then taken image by altered magnification powers.
Differential scanning calorimetry (DSC)
DSC was used to evaluate the crystallinity of sample. The solid powder of selected formula engaged in constricted aluminum pans and raised temperature at the rate of 20 °C/min compared to an empty pan of aluminum as a reference.
X-ray powder diffraction analysis (XRD)
The X-ray diffraction was applied to evaluate the molecular and atomic structure; it can be used to prove the crystallinity of nanoparticles so this test applied to check whether formula is crystalline or amorphous. The study was recognized by powder X-ray diffraction at 2θ.
Fourier transforms infrared spectroscopy (FTIR)
FTIR determined by grounded solid sample with potassium bromide and compressed in special disc then measured under infrared spectroscopy. The samples that studied were pure CC, polymer, nanoparticles of selected formula. The spectrum obtained was in between the wave numberof 4000-400 cm-1 [13].
Preparation of CC nanoparticles as oral film
Solvent casting was used to synthesize CC nanoparticles into an oral film [14]. 16 mg of candesartan cilexetile nanoparticles from the chosen formula (FCC3), which is equivalent to 8 mg of pure candesartan, must be synthesized for a film covering an area of roughly 4 cm2. The film was made by slowly adding 448 mg of HPMC-E15 to 12.5 ml of purified water, stirring, and waiting 60 min to make sure the entire polymer was dissolved. After that, 0.45 ml of glycerin was added as a plasticizer, and stirring regularly for an additional 30 min. Subsequently, the powdered weighted CC nanoparticles were gradually added while stirring for 60 min. The gel was left for 120 min in order to extract any trapped air bubbles. The resulting gel was cast onto a 56 cm2 petri dish and allowed to dry for 48 h at 25 °C. The computation of the necessary candesartan nanoparticle weight for the preparation involved dividing the 56 cm2 petri dish area by the 4 cm2 dosage area. This resulted in 14 films, each comprising 16 mg of CC nanoparticles. Consequently, 224 mg of candesartan nanoparticle powder were needed for the entire petri dish.
Evaluation of oral film of CC nanoparticles
Visual examination
The physical characteristics of the films, such as their consistency, color, and homogeneity, were examined visually in the oral thin film research. The visual inspection of the films and tactile quality assessment confirmed these variables [15].
Weight variation
We take ten oral films of CC nanoparticles each of 4 cm2 in size and their individual weights were recorded, with the average weight being computed. The created oral film's homogeneity had been verified by the weight variation test [16].
Thickness
To ensure that the film thickness is uniform, a thickness test is necessary. The thickness of the film was measured using a digital Vernier caliper (Mitutoyo, Japan) at five separate locations: the center and four corners. For ten films, the average and standard deviation were determined [17].
Folding endurance
By repeatedly folding the oral film at a fixed place until it broke, the folding endurance of candesartan oral film was measured. The evaluation of folding endurance is indicated by the recorded folding number prior to the film breaking [18].
The surface pH of oral film
The pH of the solution was measured with a pH meter after the oral thin film of CC nanoparticles was added to 5 milliliters of pure water. For this test, three trials were conducted, and the average value was determined [19].
Drug content
The 4 cm2 candesartan cilexetile nanoparticles oral film was added to 100 ml of simulated saliva solution, and the nanoparticles were stirred for 30 min to fully dissolve them. A 1 ml sample was taken, diluted with the simulated saliva solution, and then UV analysis at the media's λ max was performed. For this experiment, three trials were completed, and the average value and standard deviation were computed [20].
Disintegration test
By placing a film inside the little container of cleaned water, the disintegration time was indicated, and the time was recorded till it disintegrated. Three duplicates of the experiment were run, and the average value was determined [21].
Swelling index of oral film
Place 4 cm2 films of CC nanoparticles that have been weighed (W0) into a stainless steel basket that has been previously weighed (the basket of tablet dissolving equipment). The oral film-containing basket was submerged in a 35 ml artificial saliva (pH 6.75) solution in a petri plate. Until a steady weight was found, the weight growth of the oral film was monitored several times. Equation number one was used to determine the degree of swelling (1) [22].
The degree of swelling = (Wt-W0)/W0…. Eq. (1).
The experiment was carried out in triplicate, and the mean value was computed, where Wt and W0 represent the weight of the film sample at time t and time zero, respectively.
In vitro dissolution test of CC nanoparticles loaded oral film
Utilizing a USP type II dissolution device (paddle type), the oral films of CC nanoparticles in vitro dissolution test were established. A modified paddle was used to rotate at 50 rpm and 37±0.5 °C while oral films containing 16 mg of candesartan nanoparticles of a chosen formula and 8 mg of raw candesartan were allowed to dissolve in 500 ml of simulated saliva pH 6.75 containing 0.7% of polysorbate 20 (using a customized sinker) [23]. Five milliliter samples were extracted using a syringe on multiple occasions at intervals of (0.5, 1, 2, 3, 4, 5, 10, 15, 20, 25, and 30 min). The samples were then filtered through a 0.22 µm membrane filter and diluted with dissolution media, subsequently examined at the reported λ max using UV spectrophotometry [24]. After every sample, the same amount of simulated saliva was added. Plotting the percentage of cumulative drug release versus time allowed for the construction of the drug's dissolution profile. The test was performed in triplicate, and the mean value was calculated.
Statistical analysis
The test results were statistically assessed using one-way analysis of variance (ANOVA) at p≤0.05. The results are shown as mean samples±standard deviation (SD).
RESULTS AND DISCUSSION
Evaluations of CC nanoparticles
All CC nanoparticles prepared formulas was tested and the size characterized by ABT-9000 Nano laser. The measured size for all formula was in the nanosize (560 nm-37 nm) as shown in table 2. All tested formulas were revealed monodisperse PDI. This study exposed a drop in particle size and consequently high surface area of CC nanoparticles compared with pure powder of drugs [25].
Table 2: CC nanoparticles particle size, specific surface area (SSA), and polydispersity index (PDI)
| Formula no. | Average particle size (nm)±SD | SSA (m2/g) | PDI |
| FCC1 | 236.5±0.800 | 9.34 | 0.012 |
| FCC2 | 334±10.000 | 6.45 | 0.008 |
| FCC3 | 37±1.000 | 54.66 | 0.019 |
| FCC4 | 150±1.000 | 14.96 | 0.015 |
| FCC5 | 270±0.800 | 10.04 | 0.017 |
| FCC6 | 560±10.000 | 4.42 | 0.013 |
| FCC7 | 150±8.000 | 14.43 | 0.011 |
| FCC8 | 177±7.000 | 13.44 | 0.014 |
| FCC9 | 315±7.000 | 6.45 | 0.021 |
| FCC10 | 555±9.000 | 4.04 | 0.008 |
| FCC11 | 220±1.000 | 9.97 | 0.010 |
| FCC12 | 280±6.000 | 8.12 | 0.007 |
Results are given as mean± SD (n=3)
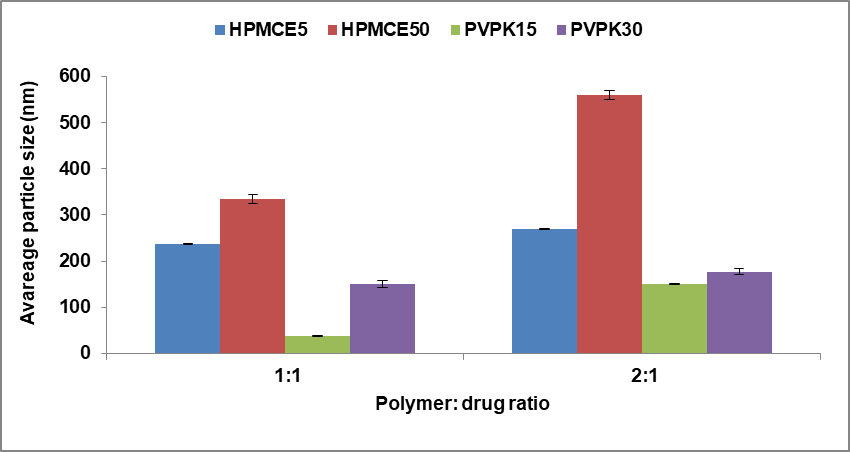
Fig. 1: Effect of kind and concentration of polymers on average CC nanoparticles size, mean±SD (n=3)
Effect of polymer type and its concentration
The influence of polymer type on mean particle size was studied on formulas FCC1-FCC4 at polymer: drug ratio (1:1). The results showed that are markable decrease in nanoparticle size obtained with PVP<HPMC, this effect might be due to increase in the affinity and decrease in viscosity. Different particle size range obtained from HPMC and PVP polymers, the different grade of polymer gave different size range as showed in fig. 1, and thus due differ in molecular weight, viscosity and in substitution group. The HPMC polymer have hydroxyl propyl group and different viscosity, the lowest viscous grade (E5<E50), also the differences in drug affinity and viscosity of PVP polymer (PVPK15<PVPK30). So, the smallest particle size in FCC3 was obtained with PVPK15 significantly (p≤0.05) decrease in size of particles (37 nm) than other polymer used due to high affinity of this polymer to candesartan nanoparticle [26]. Changing the polymer concentration was studied in formulas FCC1-FCC8 under all polymers: drug percentage of 1:1, 2:1. And it showed that the increase in polymer concentration above 1:1 ratio caused a notable increase in mean particle size which is agree with the finding of Matteucci et al., 2006 and this might be due to the higher thickness of polymer layer around each particle that led to aggregation of particles and/or the high number of polymer chains that associated in the diffusion process that lead to increasing in polymer-polymer interaction [27].
Effect of injected volume
The effects of changing the ratio of injected drug solvent solution to stabilizer antisolvent solution on the mean size of the nanoparticles formed for all polymers type was shown in fig. 2. It has been shown that the solvent: antisolvent ratio (1:10) led to a remarkable decrease in the size of the prepared CC nanoparticles and this may attributed to faster supersaturation and rapid neucleation that dominates over crystal growth [28].
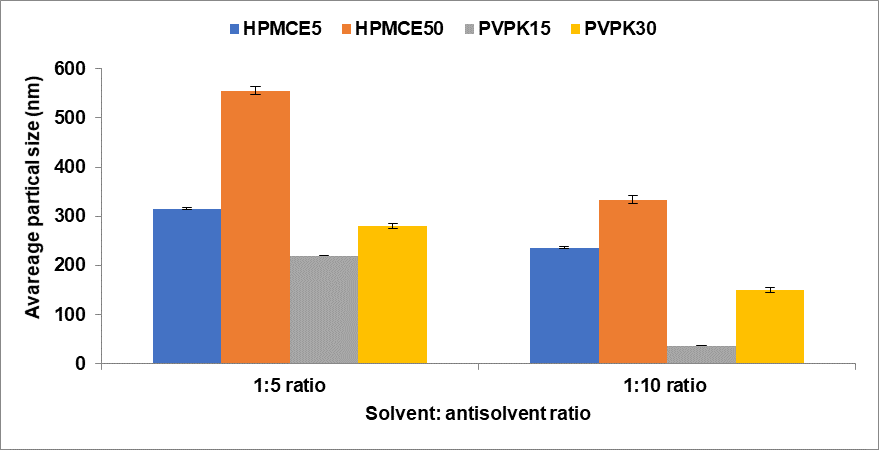
Fig. 2: Effect of injected volume on average CC nanoparticles size, mean± SD (n=3)
Drug content and loading efficiency
The demonstrated drug content result from the formula FCC3 was 7.8±0.06 mg with a loading efficiency of 96.3±0.04%. So the using of solvent/antisolvent method gives good drug content and small amount of drug loosed during preparation.
Field emission scanning electron microscope
The nanoparticles shape and topography were determined by using FESEM. It was established that good surface morphology and smallest particle size of FCC3 formula than raw drug powder, the FESEM image for pure powder with 10Kx while the FESEM image of FCC3 at 200Kx as showed in fig. 3 and 4. This effect might be due to adsorption of the polymer on the surface of drug.
Differential scanning calorimetry (DSC)
The differential scanning calorimetry of candesartan showed sharp endothermic peak at 171.5 °C which is near to its melting point that represent the crystalline state of drug as showed in fig. 5. [29]. While, the DSC of FCC3 formula indicated to a reduced intensity of peak and temperature in comparison to raw drug and polymer as in fig. 6 and 7. These results indicate the decrease of crystallinity percent of drug and/or translate to amorphous form that enhances solubility of drug.

Fig. 3: FESEM of pure drug
X-ray powder diffraction analysis (XRD)
The X-Ray powder diffraction profile of raw CC as in fig. 8, shows high intensity sharp peak for 2θ value at 9.8, 18.5 and 23.2⁰. While, the decline in intensity and larger diffraction peaks that presented in XRD of FCC3 formula as in fig. 9 that means, drug had converted to an amorphous form [30].

Fig. 4: FESEM of FCC3 formula
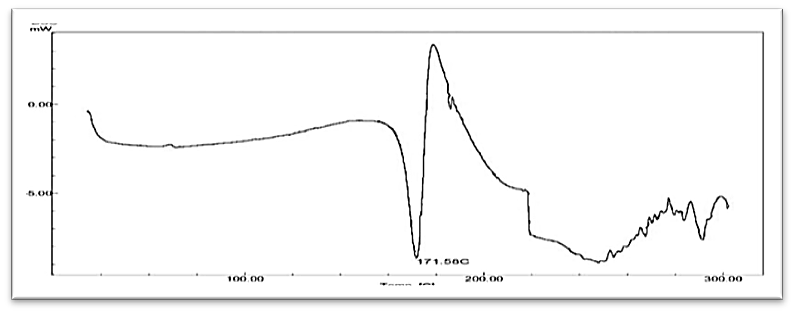
Fig. 5: DSC of raw candesartan
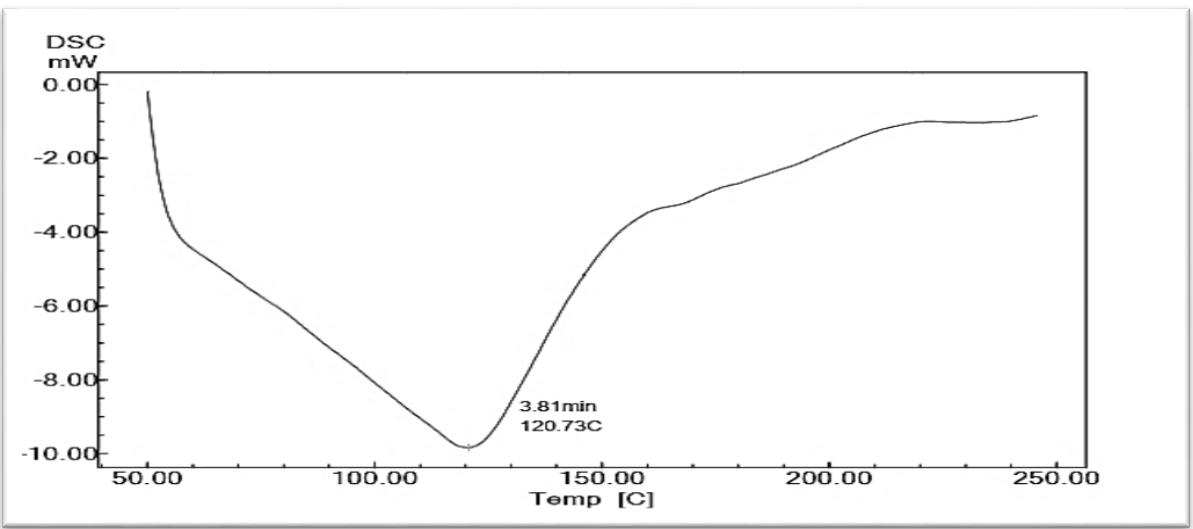
Fig. 6: DSC of PVP
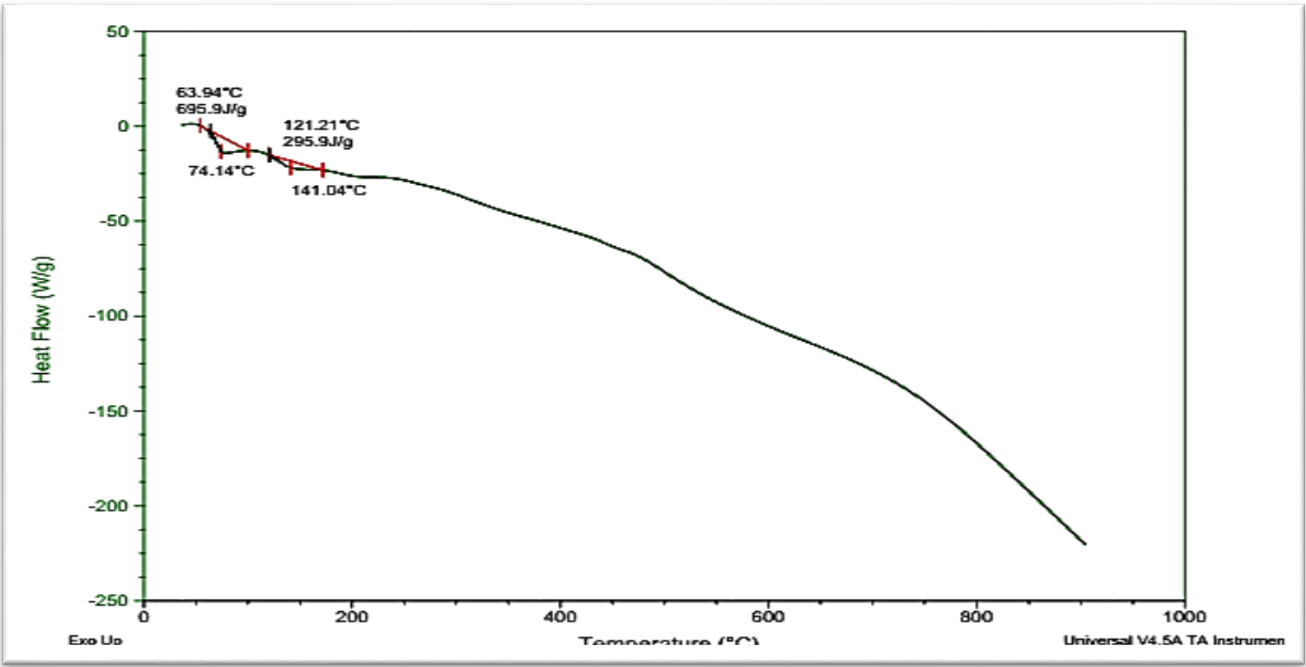
Fig. 7: DSC of FCC3
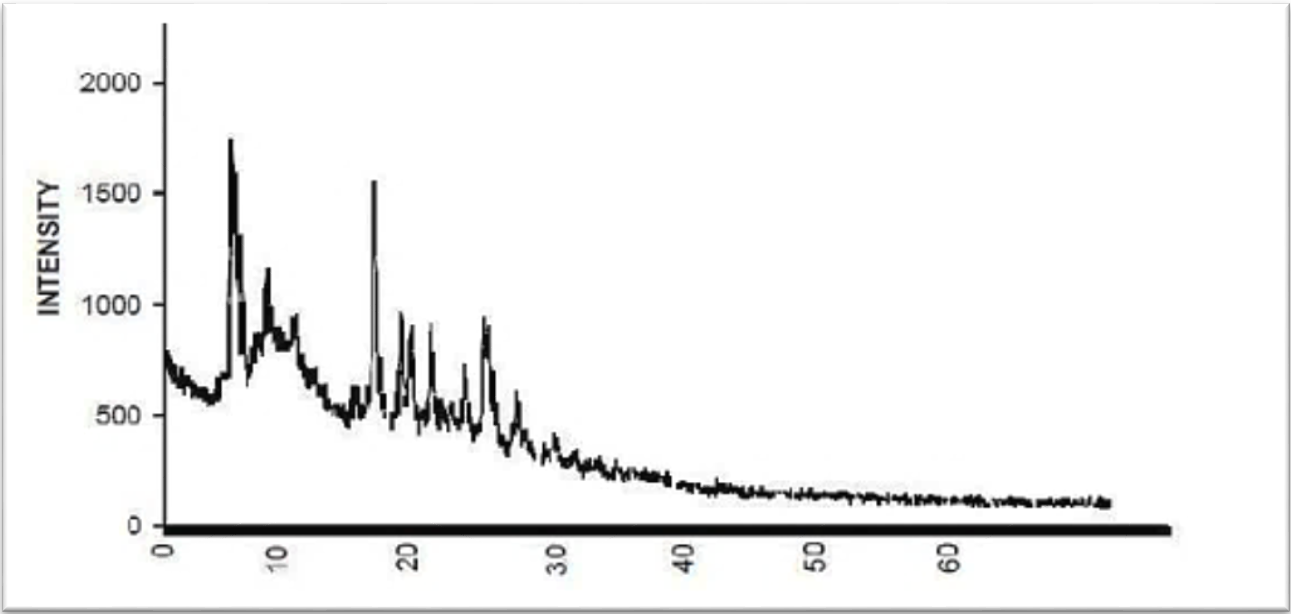
Fig. 8: XRD of raw CC
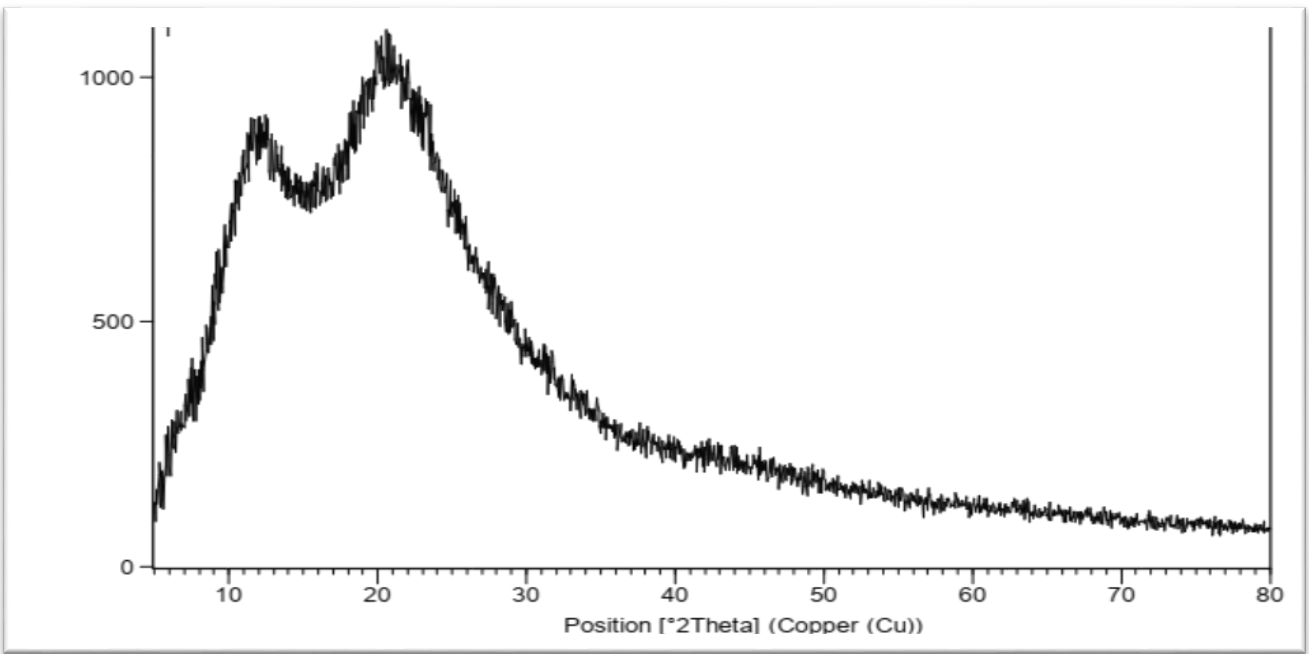
Fig. 9: XRD of FCC3
FT-IR study
The results of the peaks of FTIR tests absorbance bands of raw CC powder were shown in table 3 and fig. 10.
Visual Examination of CC oral film
The 2*2 cm2 film of CC nanoparticles made from HPMC-E15 was white, thin, and smooth, with a good candesartan nanoparticle distribution as in fig. 13.
Weight variation of oral film
The mean weight of ten films was 33.45 mg±0.2; this reduced standard deviation indicates that the weight of the oral films from CC was constant.
Thickness of oral film and folding endurance
The ten films that have been produced have a thickness explained in the table 4. The thickness uniformity of CC nanoparticles film was exposed to low standard deviation values, indicating approval of the dosage accuracy and, thus, the casting method's success for oral thin-film formulations. The folding endurance of CC nanoparticles oral film was found to be in range of (135–160 times) for film number one to film number ten respectively in table 4.
Surface pH study
The pH of CC nanoparticles films was measured and found to be between (6.4±0.05-6.9±0.08) this pH is close to neutral as possible to prevent irritation to mucosa.
Table 3: FTIR characteristic peaks of raw CC
| Functional group | Characteristic peaks of CC observed in IR region (cm-1) |
| Aromatic C-H Stretching | 2941.24 |
| – C=O Stretching | 1755.1 |
| C-N Stretching | 1614.31 |
| ‒C–O Stretching | 1244 |
| O-Subistitution | 750.26 |
The FTIR of polymer and FCC3 formula as represented in fig. 11 and 12 were studied by FTIR spectroscopy. The result of the prepared nanoparticles formula showed no alteration in shifting of location of characteristic peaks that demonstrates no main interaction between drug and PVP polymer [31].
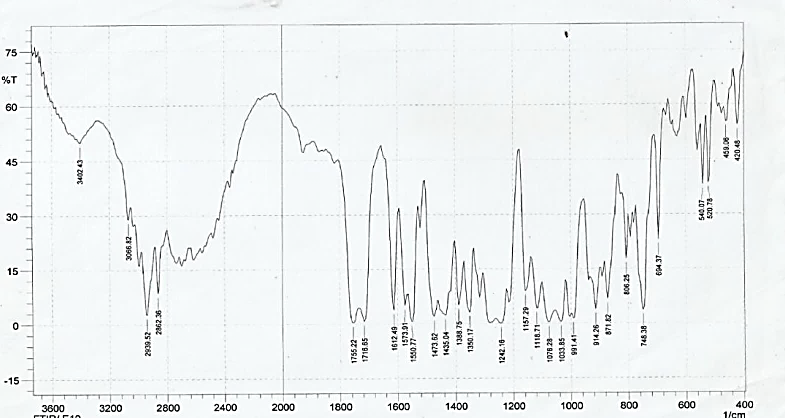
Fig. 10: FT-IR spectrum of pure candesartan cilexetil
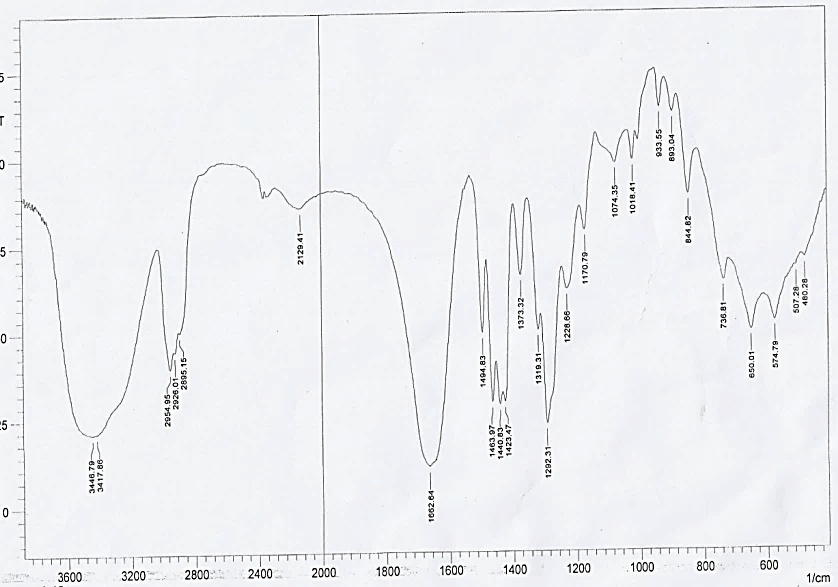
Fig. 11: FTIR spectrum for PVP polymer
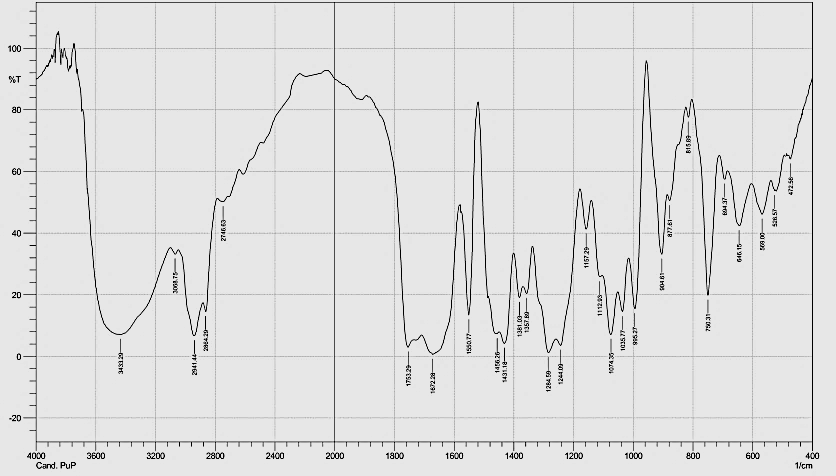
Fig. 12: FTIR spectrum for FCC3 nanoparticles

Fig. 13: Oral thin film of CC nanoparticles
Table 4: Thickness and folding endurance of CC nanoparticles oral film
| No. of film | Thickness (mm)±SD | Folding endurance±SD |
| 1 | 0.134± 0.015 | 160±1.2 |
| 2 | 0.132±0.017 | 156±2 |
| 3 | 0.133±0.017 | 155±2.1 |
| 4 | 0.134±0.017 | 149±1.4 |
| 5 | 0.135±0.017 | 143±1.5 |
| 6 | 0.136±0.015 | 136±2.3 |
| 7 | 0.131±0.016 | 159±2.5 |
| 8 | 0.133±0.015 | 140±1.7 |
| 9 | 0.132±0.016 | 138±1.2 |
| 10 | 0.134±0.013 | 135±1.1 |
Results are given as mean± SD (n=3)
Drug content uniformity
All CC films were found to contain uniform amount of the candesartan nanoparticles. The drug distributed in the range of 93.00±1.75 to 98.25±1.25. Thus, the oral film of CC nanoparticles met the standards of (BP 2011) content uniformity which is (85-115%) [32].
Disintegration time
The oral film made from CC nanoparticles disintegrated in a range of (25±0.013) sec, falling within the 30 sec window specified for the peri-oral disintegrating tablet. This recommendation is helpful for the development of oral thin film formulation [33].
Swelling index of oral film
Swelling index of the prepared oral film consist of CC nanoparticles showed in fig. 14, complete hydration of film was present at 2 min.
In vitro dissolution test of CC nanoparticles as oral film
The oral thin films of CC nanoparticles where tested for dissolution profile using 500 ml simulated saliva at 50rpm and 37 °C and compared with oral film of raw candesartan as shown in fig. 15. The percent of release in one minutes and time to reach 100% of release this comparison were done between film prepared from CC nanoparticles and other prepared from raw candesartan. The cumulative proportion of CC released from a film that contains nanoparticles of CC in one minute and three minute was 49% and 82 % respectively, and reach to 100% at time 4 min. Conversely, cumulative percent of release of pure CC film in one minute and three minute was 19% and 52 % respectively, and where failed to achieve full release (100%) because pure drug was not well dissolved in artificial saliva, so this rate limiting step reason for low bioavailability of candesartan, while oral film containing candesartan nanoparticles provided the best dissolution profile and significant quick release. From these result showed a very rapid disintegration of cc nanoparticles oral film (25s), indicating that the polymeric matrix is not limiting factor for drug release. However, the subsequent nanoparticles release and dissolution government the overall release profile, as reflected by 68% drug release after two minutes. Therefore the dissolution process of cc oral film is rate limiting step [34].
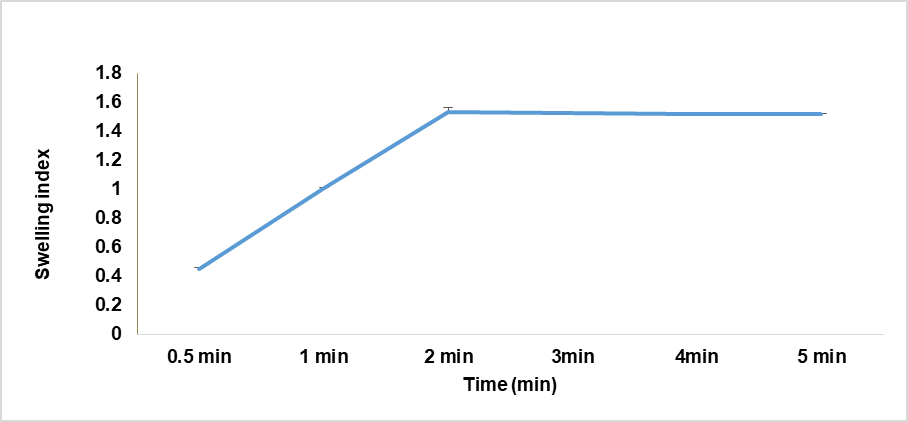
Fig. 14: Swelling index of oral film consist of CC nanoparticles with use HPMC-E15, mean± SEM (n=3)
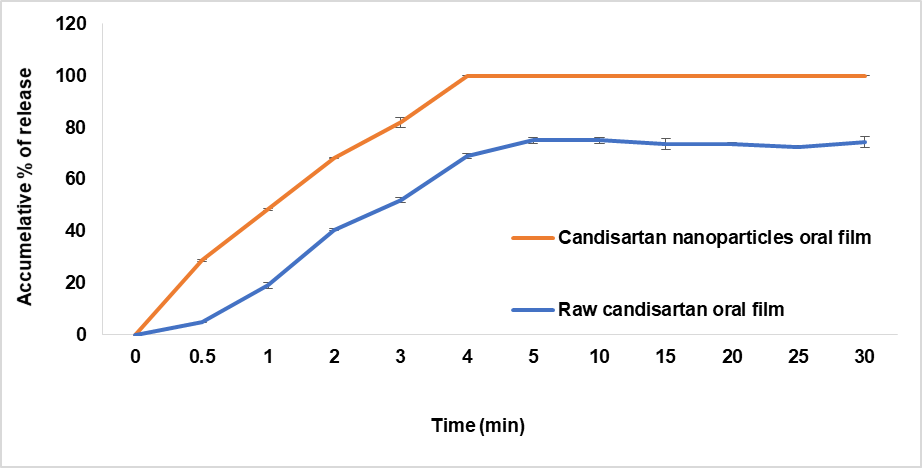
Fig. 15: In vitro release profile of oral film in simulated saliva with 0.7% tween 20 (mean± SEM, n=3)
CONCLUSION
In this study, the solvent: antisolvent precipitation process was used to create candesartan nanoparticles. Then, the choices formula was formulated as oral film by solvent casting method. The prepared film evaluated to confirm the uniformity of the content, thickness and appropriate dissolution profile. The oral candesartan nanoparticles film could be choice to accelerate the onset of drug action and improve the dissolution profile of pure drug.
ACKNOWLEDGEMENT
The authors give thanks and gratitude to the department of pharmaceutics, faculty of pharmacy, the University of Kufa for everything they have done for the completion of this research.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
All the authors have contributed equally.
CONFLICTS OF INTERESTS
Declared none
REFERENCES
Williams HD, Trevaskis NL, Charman SA, Shanker RM, Charman WN, Pouton CW. Strategies to address low drug solubility in discovery and development. Pharmacol Rev. 2013 Jan;65(1):315-499. doi: 10.1124/pr.112.005660, PMID 23383426.
Bhalani DV, Nutan B, Kumar A, Singh Chandel AK. Bioavailability enhancement techniques for poorly aqueous soluble drugs and therapeutics. Biomedicines. 2022 Aug 23;10(9):2055. doi: 10.3390/biomedicines10092055, PMID 36140156, PMCID PMC9495787.
Vijayaraj S, Omshanthi B, Anitha S, Sampath Kumar KP. Synthesis and characterization of novel sulphoxide prodrug of famotidine. Indian J Pharm Educ Res. 2014 Oct;48(4):35-44. doi: 10.5530/ijper.48.4.6.
Da Silva FL, Marques MB, Kato KC, Carneiro G. Nanonization techniques to overcome poor water-solubility with drugs. Expert Opin Drug Discov. 2020 Jul;15(7):853-64. doi: 10.1080/17460441.2020.1750591, PMID 32290727.
Ambrus R, Kocbek P, Kristl J, Sibanc R, Rajko R, Szabo Revesz P. Investigation of preparation parameters to improve the dissolution of poorly water-soluble meloxicam. Int J Pharm. 2009 Nov 3;381(2):153-9. doi: 10.1016/j.ijpharm.2009.07.009, PMID 19616609.
Kawale KA, Autade NB, Narhare HS, Mhetrea RL. A review on fast-dissolving oral film. Asian J Pharm Clin Res. 2023 Oct;16(10):7-17. doi: 10.22159/ajpcr.2023.v16i10.48099.
Asif H, Sabir A, Moloy M, Parminder S, Bhasin A. Review on candesartan: pharmacological and pharmaceutical profile. J Appl Pharm Sci. 2011 Dec;1(10):12-7.
Shaikh SM, Avachat AM. Enhancement of solubility and permeability of candesartan cilexetil by using different pharmaceutical interventions. Curr Drug Deliv. 2011 Jul;8(4):346-53. doi: 10.2174/156720111795767997, PMID 21453263.
Vairappan K, Ayarivan P, Lakshmanan R, Sundaravadivelu J. Development and validation of a dissolution test with reversed-phase high performance liquid chromatographic analysis for candesartan cilexetil in tablet dosage forms. Arab J Chem. 2011;9(1 Suppl);S867-73. doi: 10.1016/J.ARABJC.2011.09.011.
Sweetman S, editor. Martindale: the complete drug reference. 36th ed. London, UK: Pharmaceutical Press; 2009.
Wu CY, Wang W. Application of antisolvent precipitation method for formulating excipient-free nanoparticles of psychotropic drugs. Pharmaceutics. 2022 Apr 8;14(4):819. doi: 10.3390/pharmaceutics14040819, PMID 35456653, PMCID PMC9029518.
Paudel A, Ameeduzzafar ISS, Imam SS, Fazil M, Khan S, Hafeez A. Formulation and optimization of candesartan cilexetil nano lipid carrier: in vitro and in vivo evaluation. Curr Drug Deliv. 2017;14(7):1005-15. doi: 10.2174/1567201813666161230141717, PMID 28034361.
Reddy E. Enhancement of candesartan cilexetil by self emulsifying drug delivery system. Int J Pharm Sci Res. 2012 Jul;3(7):2098-104. doi: 10.13040/IJPSR.0975-8232.3(7).2098-04.
Keshari A, Sharma PK, Parvez N. Fast dissolving oral film: a novel and innovative drug delivery system. Int J Pharm Sci Res. 2014 Mar;5(3):92-5.
Bawane S, Teirandhe R, Pande SD. Formulation and evaluation of oral fast dissolving film of bisoprolol fumarate. Int J Pharm Drug Anal. 2018 Feb;6(2):105-15.
Jelvehgari M, Montazam SH, Soltani S, Mohammadi R, Azar K, Montazam SA. Fast dissolving oral thin film drug delivery systems consist of ergotamine tartrate and caffeine anhydrous. Pharm Sci. 2015 Sep;21(2):102-10. doi: 10.15171/PS.2015.24.
Saleem MA, Naheed MD, Murali YD, Jaydeep P, Dhaval M. Preparation and evaluation of mupirucin loaded polymer composite films. J Drug Deliv Ther. 2012 May;2(3):167-72. doi: 10.22270/jddt.v2i3.166.
Pallavi K, Pallavi T. Formulation and evaluation of fast dissolving films of eletriptan hydrobromide. Int J Curr Pharm Sci. 2017 Jan;9(2):59-63. doi: 10.22159/ijcpr.2017v9i2.17386.
Sultana F, Arafat M, Pathan SI. Preparation and evaluation of fast dissolving oral thin film of caffeine. Int J Pharm Biol Sci. 2013 Jan;3(1):153-61.
Choudhary DR, Patel V, Patel H, Kundawala A. Formulation and evaluation of quick dissolving film of ondansetron hydrochloride. Int J Pharmacol Res. 2011 Dec;3(4):31-50.
Mushtaque M, Muhammad IN, Fareed Hassan SM, Ali A, Masood R. Development and pharmaceutical evaluation of oral fast dissolving thin film of escitalopram: a patient friendly dosage form. Pak J Pharm Sci. 2020 Jan;33(1):183-9. PMID 32122847.
Irfan M, Rabel S, Bukhtar Q, Qadir MI, Jabeen F, Khan A. Orally disintegrating films: a modern expansion in drug delivery system. Saudi Pharm J. 2016 Sep;24(5):537-46. doi: 10.1016/j.jsps.2015.02.024, PMID 27752225, PMCID PMC5059831.
Hoppe K, Sznitowska M. The effect of polysorbate 20 on solubility and stability of candesartan cilexetil in dissolution media. AAPS PharmSciTech. 2014 Oct;15(5):1116-25. doi: 10.1208/s12249-014-0109-8, PMID 24871550, PMCID PMC4179655.
Aparna C, Anusha M, Manisha B. Enhancement of dissolution of candesartan cilexetil. Asian J Pharm Clin Res. 2023 Mar;16(3):148-51. doi: 10.22159/ajpcr.2023.v16i3.46626.
Dizaj SM, Vazifehasl Zh, Salatin S, Adibkia Kh, Javadzadeh Y. Nanosizing of drugs: effect on dissolution rate. Res Pharm Sci. 2015 Mar-Apr;10(2):95-108. PMID 26487886, PMCID PMC4584458.
Ghobad H, Cheraghali S. Effect of biocompatible polymers on the physicochemical properties of fenofibrate in nanoparticle system. J Chem Pharm Res. 2015;7(12):913-9.
Matteucci ME, Hotze MA, Johnston KP, Williams RO. Drug nanoparticles by antisolvent precipitation: mixing energy versus surfactant stabilization. Langmuir. 2006 Oct 10;22(21):8951-9. doi: 10.1021/la061122t, PMID 17014140.
Dong Y, Ng WK, Shen S, Kim S, Tan RB. Preparation and characterization of spironolactone nanoparticles by antisolvent precipitation. Int J Pharm. 2009 Jun 22;375(1-2):84-8. doi: 10.1016/j.ijpharm.2009.03.013, PMID 19481693.
Thadkala K, Nanam PK, Rambabu B, Sailu C, Aukunuru J. Preparation and characterization of amorphous ezetimibe nanosuspensions intended for enhancement of oral bioavailability. Int J Pharm Investig. 2014 Jul;4(3):131-7. doi: 10.4103/2230-973X.138344, PMID 25126526, PMCID PMC4131384.
Thirupathi A, Reddy A, Narayana A, Meshram S, Sampathi S. Solid state characterization of the polymorphic changes in candesartan cilexetil solid dispersion with polyethylene glycol 8000. Int J Pharm Sci Res. 2014 Jun;5(6):2362-8. doi: 10.13040/IJPSR.0975-8232.5(6).2362-68.
Ashok Kumar U, Suresh G. Preparation and in vivo evaluation of candesartan cilexetil solid dispersions. Asian J Pharm Clin Res. 2021;14(8):129-33. doi: 10.22159/ajpcr.2021.v14i8.42037.
British Pharmacopoeia. Uniformity of dosage units. In: Appendix XII C, Consistency of formulated preparations. London: The Stationery Office; 2011.
Saab M, Mehanna MM. Disintegration time of orally dissolving films: various methodologies and in vitro/in vivo correlation. Pharmazie. 2019 Apr 1;74(4):227-30. doi: 10.1691/ph.2019.8231, PMID 30940306.
Mahesh RD, Umang KG, Dhaval DM, Kalpesh AP, Ravi M, NR S. Formulation optimization and characterization of candesartan cilexetil nanosuspension for in vitro dissolution enhancement. Afr J Pharm Pharmacol. 2015 Feb;9(5):102-13. doi: 10.5897/AJPP2013.3887.