
Int J App Pharm, Vol 18, Issue 1, 2026, 1-7Review Article
A SYSTEMATIC REVIEW AND META-ANALYSIS ON EFFECT OF LACTOBACILLUS SUPPLEMENTATION ON METHOTREXATE EFFICACY IN RHEUMATOID ARTHRITIS
MOHAMMED FAIZAL K., VARSHA K., SUVITHA SRI M., NITHYA SHREE, RAGUNATH S. , DEEPALAKSHMI M.*
JSS College of Pharmacy, Department of Pharmacy Practice, Ooty, Nilgiris, Tamil Nadu, India
*Corresponding author: Deepalakshmi M.; *Email: deepapharmacy@jssuni.edu.in
Received: 13 Aug 2025, Revised and Accepted: 22 Nov 2025
ABSTRACT
Rheumatoid arthritis (RA) is a chronic autoimmune disorder characterized by systemic inflammation and joint destruction. Methotrexate (MTX) remains the cornerstone of therapy but is often limited by variable efficacy and adverse effects. Emerging evidence suggests that gut microbiota modulation, particularly through Lactobacillus supplementation, may enhance the efficacy of MTX via the gut–joint axis. To systematically review and analyze the impact of Lactobacillus supplementation on methotrexate efficacy in rheumatoid arthritis, focusing on inflammatory biomarkers and disease activity in both human and animal studies. A comprehensive search was conducted in PubMed, Scopus, Web of Science, and Cochrane CENTRAL up to June 2025 for randomized controlled trials (RCTs) and animal studies comparing MTX+Lactobacillus versus MTX alone. Primary outcomes included TNF-α, IL-6, IL-10, IL-17, CRP, and DAS28. Risk of bias was assessed using Cochrane RoB 2.0 for RCTs and SYRCLE for animal studies. Meta-analysis employed a random-effects model. Human RCTs demonstrated significant reductions in CRP, TNF-α, and IL-6, accompanied by increased IL-10 levels and modest improvement in DAS28 (SMD –0.41). Heterogeneity was attributed to strain, dose, and duration differences. Animal studies provided mechanistic support, showing decreased pro-inflammatory cytokines, enhanced anti-inflammatory markers, and improved histopathology, though findings were not pooled. Gut microbiota-targeted approaches hold promise for improving MTX response, warranting further strain-specific clinical investigations.
Keywords: Rheumatoid arthritis, Methotrexate, Lactobacillus, Gut microbiota, DAS28, Cytokines, Probiotics, Systematic review, Meta-analysis
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.56515 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Synovial hyperplasia is one of the hallmarks of rheumatoid arthritis (RA), an autoimmune disease that primarily affects the synovial joints. RA, which affects nearly 1% of the global population, increases the risk of comorbid conditions such as osteoporosis and cardiovascular disease, leading to significant morbidity and a reduced quality of life. Immunological dysregulation, environmental triggers, and genetic predisposition interact intricately in the pathophysiology of RA [1-3].
Methotrexate (MTX) remains the cornerstone of pharmacological management of RA due to its proven efficacy, safety profile, and affordability. The anti-inflammatory, antiproliferative, and immunosuppressive. Mechanisms of MTX-a disease-modifying antirheumatic drug (DMARD)—involve inhibition of dihydrofolate reductase and modulation of adenosine pathways [4, 5].
A microbial imbalance known as gut dysbiosis has been linked to increased intestinal permeability, altered immune cell profiles, and systemic inflammation-all of which may contribute to the onset and progression of RA. Notably, RA patients often exhibit an abundance of pro-inflammatory taxa such as Prevotella copri and a reduction in overall microbial diversity. Conversely, beneficial bacteria belonging to the genera Lactobacillus and Bifidobacterium possess immunoregulatory properties that can attenuate inflammation [6-8].
Recent evidence highlights a bidirectional interaction between MTX and the gut microbiota. MTX can alter the gut microbial ecosystem, thereby influencing its own pharmacokinetics and safety profile. Conversely, microbial composition can affect drug absorption, bioavailability, immune modulation, and MTX metabolism [9]. This reciprocal relationship suggests that microbiome-targeted strategies may enhance MTX efficacy and tolerability.
Probiotic supplementation, particularly with Lactobacillus species, has emerged as a promising adjunctive approach. Lactobacillus strains—Gram-positive, facultative anaerobic bacteria commonly found in the human gastrointestinal tract—exert anti-inflammatory effects by regulating cytokine production, enhancing regulatory T-cell activity, and maintaining intestinal barrier integrity. In animal models of arthritis, Lactobacillus supplementation has improved histopathological outcomes, reduced joint inflammation, and lowered TNF-α and IL-6 levels. In clinical trials, RA patients receiving Lactobacillus-based probiotics along with MTX therapy demonstrated improvements in disease activity scores, inflammatory biomarkers, and quality-of-life parameters [10-12].
Given the expanding body of literature on the gut microbiota’s influence on MTX response in RA, a comprehensive evaluation of Lactobacillus supplementation’s impact on MTX efficacy and toxicity is warranted. This review systematically compiles the latest data from randomized controlled trials (RCTs) and preclinical studies to assess the effect of Lactobacillus on MTX treatment outcomes in RA [13-15].
Methods
Protocol and registration
This systematic review and meta-analysis were meticulously planned and executed in compliance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA 2020) guidelines to guarantee transparency, reproducibility and methodological rigor. According to the International Prospective Register of Systematic Reviews (PROSPERO) the protocol for this study was prospectively registered to reduce the possibility of bias and to follow best practices in systematic review methodology. In the file the name is [Prospero ID: CRD 420251109054]. Using a novel synthesis of existing data this work aims to close significant knowledge gaps about the therapeutic effect of Lactobacillus supplementation on methotrexate efficacy in rheumatoid arthritis patients.
Inclusion criteria
The inclusion criteria were RCTs that investigated the effects of methotrexate (MTX) treatment in (RA) either human subjects or in vivo animal models with or without Lactobacillus supplementation. Participants had to have a verified diagnosis of RA. The primary intervention was MTX, which could be administered alone or in combination with any formulation or strain of Lactobacillus. ESR, CRP, DAS28, TNF-α, IL-6, IL-10, were among the immunological or clinical outcomes that were considered eligible for inclusion in the study. To ensure that there was enough time to evaluate the treatments effects a minimum follow-up period of four weeks was required.
Exclusion criteria
Studies that included probiotics other than Lactobacillus or that used mixed interventions that might obscure the effects of Lactobacillus alone were not included nor were they reviews meta-analyses editorials or case reports. Furthermore, studies with no baseline or endpoint data for any of the main outcomes of interest (e. g. TNF-α IL-6 IL-10 DAS28 CRP or ESR) were not included. Additionally, articles written in languages other than English were not taken into consideration for inclusion.
Information sources
Using a variety of database: PubMed/MEDLINE Scopus Web of Science Cochrane CENTRAL, restricted search of Google Scholar a thorough literature review was carried out from January 2000-July 2025. In order to automate duplicate removal and enable blinded screening all recovered records were imported into Rayyan QCRI. The studies were independently screened by 2 reviewers who then evaluated the full texts of any potentially eligible articles. To guarantee uniformity and objectivity in the inclusion of studies any disagreements or inconsistencies during the selection process were settled by talking with a 3rd reviewer.
Data extraction
To guarantee accuracy, consistency across investigations were extracted using a standardized data extraction form. Important study identifiers like authors publication year and country of origin were among the extracted data along with the study design and population characteristics and thorough explanations of the intervention and control groups, including the precise Lactobacillus strain or strains used dosage and administration time. Results were gathered for the following: ESR, IL-6, TNF-α, CRP, IL-10, DAS28. In order to evaluate included studies risk, bias and transparency details regarding funding sources and possible conflicts of interest were also documented.
Bias assessment
To evaluate bias in RCTs the Bias 2. 0 (RoB 2. 0) was used. This tool assesses potential sources of bias in a number of important areas such as allocation. The Cochrane Risk of Bias tool was used to assess the included studies methodological rigor. This instrument evaluates a number of crucial areas that could cause systematic errors in RCTs. Among these domains are:
To find out if the assignment to intervention groups was sufficiently shielded from selection bias allocation concealment was used. To assess the possibility of performance bias, participants and staff are blinded. To mitigate the risk of detection bias outcome assessment is blinded. To evaluate how robust the randomization process is random sequence generation is employed. The tool also takes into consideration other sources of bias, including selective outcome reporting (reporting bias) and incomplete outcome data (attrition bias). An independent rating of low high or unclear risk of bias was assigned to each domain. These ratings offered a methodical way to evaluate each included studys overall internal validity.
Statistical analysis
R software (the meta and meta for packages) and RevMan 5. 4 were used for all statistical analyses. For continuous outcomes such as (TNF-α, IL-6, IL-10, CRP DAS28, CRP) Stand SMDs with 95% CIs were produced. Means and standard deviations were adjusted using accepted procedures when research reported results in different units or scales. According to the Cochrane Handbook values for missing standard deviations were either computed using standard errors interquartile ranges or p-values or they were imputed from confidence intervals (v6. 3).
Throughout the meta-analysis a random-effects model, DerSimonian and Laird method was used because of expected inter-study variability in probiotic strains durations of interventions and population variables. Studies heterogeneity was evaluated using I2 statistics moderate to substantial heterogeneity was indicated by I2 50%. For heterogeneity the chi-square test (Q-test) was also reported with p 0. 10 deemed significant. When there was enough data (≥5 studies) subgroup analyses were carried out according to the population type (human vs. animal) Lactobacillus type length of treatment and dosage.
To evaluate the robustness of pooled effect sizes, sensitivity analyses were performed by progressively eliminating each study. Each outcomes level of evidence certainty was evaluated using the GRADE methodology value less than 0. 05 were significant, all p-values were two-tailed.
RESULTS
The effects of supplementing with Lactobacillus in rheumatoid arthritis (RA) patients and models receiving methotrexate (MTX) were assessed through a meta-analysis of 31 studies, including 18 human randomized controlled trials and 13 animal model studies. When Lactobacillus was added the pooled analysis showed a significant improvement in both inflammatory markers and disease activity indices.
A significant decrease in C-reactive protein (CRP) was reported by ten studies with 784 participants SMD = –0. 76, 95 percent CI: –1. 05 to –0. 47 p 0. 0001) with moderate heterogeneity (I² = 58. 1 percent). The results of subgroup analysis showed that interventions with durations of eight weeks or more produced larger reductions (SMD = –0. 84) than those with shorter durations (SMD = –0. 58). Eight studies (n = 648) revealed a moderate but statistically significant improvement in the Disease Activity Score-28 (DAS28) SMD = –0. 41, CI: –0. 73 to –0. 09 p = 0. 012, I² = 45. 6 percent. Long-duration trials showed more pronounced benefits (SMD = –0. 47) and the effects were consistent across human RCTs. There was a decrease in TNF-α in seven studies with 513 participants SMD = –0. 55, CI: –0. 84 to –0. 26 p = 0. 001, I² = 51. 9 percent. The effect was larger in animal models SMD = –0. 67 than in low-dose Lactobacillus subgroups (SMD = –0. 41). Interleukin-6 (IL-6) data from six studies (n = 480) showed a significant decrease SMD= –0. 49, CI: –0. 88 to –0. 10 p = 0. 014, I² = 64. 3 percent). Larger decreases were seen in long-duration interventions (SMD = –0. 52) and animal studies (SMD = –0. 73). Last but not least, interleukin-10 (IL-10), which was measured in five studies (n = 386), significantly increased MeanSMD=+0. 34, CI: 0. 03 to 0. 66 p = 0. 030, I² = 39. 7 percent. Indicating increased anti-inflammatory activity when Lactobacillus supplementation was used.
Characteristics of included studies
A total of 18 studies were included, of which 10 were human randomized controlled trials (RCTs) and 8 were animal model studies. Human study characteristics are summarized in table 1, while animal study characteristics are presented in table 10.
C-reactive protein (CRP)
The meta-analysis of 10 RCTs (n = 784) demonstrated a significant reduction in CRP levels with Lactobacillus supplementation (SMD = –0.76, 95% CI: –1.05 to –0.47; p<0.0001; table 2; fig. 2). Heterogeneity was substantial (I² = 58.1%).
Tumor necrosis factor-α (TNF-α)
Lactobacillus supplementation was associated with a significant reduction in TNF-α levels (SMD = –0.55, 95% CI: –0.91 to –0.19; p = 0.002; table 3; fig. 3). Heterogeneity was substantial (I² = 51.9%).
Interleukin-6 (IL-6)
The pooled analysis showed a moderate reduction in IL-6 (SMD = –0.49, 95% CI: –0.88 to –0.10; p = 0.013; table 4; fig. 4). Substantial heterogeneity was observed (I² = 64.3%).
Interleukin-10 (IL-10)
A small but significant increase in IL-10 was observed (SMD =+0.34, 95% CI: 0.05 to 0.62; p = 0.021; table 5; fig. 5).
Disease activity score-28 (DAS28)
Across five RCTs, Lactobacillus supplementation reduced DAS28 scores (SMD = –0.41, 95% CI: –0.68 to –0.13; p = 0.004; table 6). This corresponds to a small-to-moderate clinical effect.
Subgroup and sensitivity analyses
Subgroup analyses based on strain, dosage, and duration are summarized in tables 7–9. Sensitivity analysis excluding lower-quality trials did not substantially alter effect estimates.
Risk of bias
Risk of bias assessments are shown in table 11 for human RCTs (RoB 2.0) and table 12 for animal studies (SYRCLE tool). Overall, most RCTs were at low to moderate risk of bias, while animal studies showed some concerns in randomization and blinding.
Publication bias
Funnel plots for each primary outcome (CRP, TNF-α, IL-6, IL-10, DAS28) are presented in fig. 6–10. Visual inspection suggested no major asymmetry, and Egger’s test did not indicate publication bias (p>0.10 for all outcomes).
DISCUSSION
This meta-analysis and systematic review offer strong evidence that Lactobacillus supplementation can improve the effectiveness of methotrexate (MTX) in rheumatoid arthritis (RA) by reducing systemic inflammation and enhancing disease activity. Significant declines in DAS28 and CRP in line with earlier meta-analyses imply probiotic-induced changes in gut microbiota could reduce systemic inflammatory reactions and promote clinically significant advancements in the treatment of RA. As previously noted in animal models the supplementation also markedly decreased in cytokines TNF-α, IL-6 while raising in IL-10 levels, suggesting an immunoregulatory shift that may be mediated by downregulating Th1/Th17 pathways and enhancing regulatory T cell (Treg) activity.
The importance of sustained and appropriately dosed probiotic administration was highlighted by subgroup analyses that showed that animal studies demonstrated stronger cytokine suppression than human trials, likely due to controlled conditions and higher dosing. Interventions lasting ≥8 w and doses ≥1 × 10⁹ CFU/day produced more pronounced benefits. Given interindividual variability in MTX efficacy and the growing role of the gut-joint axis in autoimmune regulation, co-administration of Lactobacillus with MTX offers a promising non-pharmacological strategy to improve treatment response, lower systemic inflammation and possibly alleviate gastrointestinal side effects caused by MTX.
This systematic review and meta-analysis demonstrated that Lactobacillus supplementation, when used alongside methotrexate, significantly improved inflammatory outcomes in rheumatoid arthritis. Specifically, supplementation reduced CRP (table 1), TNF-α (table 2), and IL-6 (table 3), while increasing IL-10 (table 4). The pooled evidence also indicated a small-to-moderate improvement in DAS28 (table 5 and 6).
Table 1: Summary meta-analysis, this table summarizes the pooled effect sizes of key inflammatory mediators from the current meta-analysis evaluating Lactobacillus supplementation in rheumatoid arthritis
| Inflammatory mediators | Total no of studies | No of samples | Size | CI | P value | I² (%) | Interpretation |
| CRP | 10 | 784 | -0.76 | [-1.05-0.47 | <0.0001 | 58.1 | CRP reduction in lactobacillus |
| DAS28 | 8 | 648 | -0.41 | [-0.73,-0.09] | 0.012 | 45.6 | Improvement in disease activity |
| TNF-α | 7 | 513 | -0.55 | [-0.84,-0.26] | 0.001 | 51.9 | Reduction in pro-inflammatory cytokines |
| IL-6 | 6 | 480 | -0.49 | [-0.88,-0.10] | 0.014 | 64.3 | IL-6 levels significantly lowered |
| IL-10 | 5 | 386 | +0.34 | [0.03, 0.66] | 0.030 | 39.7 | Anti-inflammatory cytokine increased |
Table 2: Effect of lactobacillus supplementation on C-reactive protein (CRP)
| Parametr | Value |
| Number of studies | 10 |
| Total participants | 784 (405 intervention, 379 control) |
| SMD | –0.76 |
| CI | [–1.05, –0.47] |
| p-value | <0.0001 |
| I² Heterogeneity | 58.1% (moderate) |
| Interpretation | Significant reduction in CRP, indicating reduced systemic inflammation |
Data derived from 10 studies included in this meta-analysis assessing changes in CRP levels following Lactobacillus supplementation
Table 3: Effect on disease activity score-28 (DAS28), Compiled from eight studies evaluating DAS28 improvements in RA patients supplemented with lactobacillus strains
| Parameter | Value |
| Number of studies | 8 |
| Total participants | 648 |
| Standardized mean Difference | –0.41 |
| Confidence Interval | [–0.73, –0.09] |
| P-value | 0.012 |
| Heterogeneity | 45.6% (moderate) |
| Interpretation | Moderate improvement in RA disease activity |
Table 4: Effect on TNF-α: includes findings from seven studies (four human and three animal studies) on the effect of lactobacillus on TNF-α cytokine levels
| Number of studies | 7 (4 human, 3 animal) |
| Total participants | 513 |
| SMD | –0.55 |
| 95% CI | [–0.84, –0.26] |
| p-value | 0.001 |
| I² Heterogeneity | 51.9% (moderate) |
| Interpretation | Significant suppression of TNF-α cytokine expression |
Table 5: Effect on Interleukin-6 (IL-6), Data summarized from six included studies investigating the impact of Lactobacillus supplementation on IL-6 concentrations in RA models and patients
| Parametr | Value |
| Number of studies | 6 |
| Total participants | 480 |
| SMD | –0.49 |
| 95% CI | [–0.88, –0.10] |
| p-value | 0.014 |
| I² Heterogeneity | 64.3% (moderate to high) |
| Interpretation | Lactobacillus effectively reduced IL-6 levels in RA models and patients |
Table 6: Effect on interleukin-10 (IL-10), analysis derived from five studies reporting the effect of lactobacillus on anti-inflammatory cytokine IL-10
| Parameter | Value |
| Number of studies | 5 (3 animal, 2 clinical) |
| Total participants | 386 |
| Standardized mean Difference | +0.34 |
| Confidence Interval | [0.03, 0.66] |
| P-value | 0.030 |
| I² | 39.7% (low to moderate) |
| Interpretation | IL-10 significantly increased, suggesting enhanced anti-inflammatory response |
The presence of substantial heterogeneity (I² = 51–64%) suggests variability between studies, potentially due to differences in Lactobacillus strains, dosing regimens, study populations, and treatment durations (tables 7–9). This was acknowledged in our sensitivity analyses and discussed as a limitation.
Table 7: Subgroup analysis of lactobacillus supplementation on key inflammatory markers, disease activity in RA
| Subgroup | Outcome | No. of studies | Pooled SMD (95% CI) | p-value | I² (%) | Interpretation |
| Human RCTs | CRP | 6 | –0.62 [–0.89, –0.35] | <0.001 | 42.1 | Moderate reduction |
| DAS28 | 6 | –0.39 [–0.71, –0.07] | 0.017 | 46.4 | Modest clinical improvement | |
| Animal Models | TNF-α | 4 | –0.67 [–1.08, –0.26] | 0.002 | 38.9 | Strong suppression of inflammation |
| IL-6 | 3 | –0.73 [–1.32, –0.15] | 0.013 | 57.3 | Significant IL-6 lowering | |
| Intervention group | ||||||
| Short (<8 w) | CRP | 5 | –0.58 [–0.94, –0.22] | 0.002 | 48.5 | Effective but lower than longer interventions |
| TNF-α | 4 | –0.47 [–0.79, –0.16] | 0.004 | 34.6 | Cytokine reduction | |
| Long (≥8 w) | CRP | 5 | –0.84 [–1.22, –0.47] | <0.0001 | 54.3 | Anti-inflammatory effect |
| DAS28 | 4 | –0.47 [–0.83, –0.10] | 0.013 | 49.1 | Disease control with prolonged use | |
| Lactobacillus dose | ||||||
| Low (<1 × 10⁹ CFU/day) | TNF-α | 3 | –0.41 [–0.77, –0.06] | 0.022 | 32.4 | Suppression |
| High (≥1 × 10⁹ CFU/day) | TNF-α | 4 | –0.71 [–1.09, –0.33] | 0.001 | 46.7 | cytokine inhibition |
Table 8: Human studies (RCTs)
| Outcome | Studies (n) | Total sample | SMD | 95% CI | p-value | I² (%) | Interpretation | Outcome | Studies (n) |
| CRP | 10 | 784 | -0.76 | [-1.05,-0.47] | <0.0001 | 58.1 | Significant reduction | CRP | 10 |
| DAS28 | 8 | 648 | -0.41 | [-0.73,-0.09] | 0.012 | 45.6 | Small-to-moderate improvement | DAS28 | 8 |
| TNF-α | 7 | 513 | -0.55 | [-0.84,-0.26] | 0.001 | 51.9 | Reduction in pro-inflammatory cytokine | TNF-α | 7 |
| IL-6 | 6 | 480 | -0.49 | [-0.88,-0.10] | 0.014 | 64.3 | IL-6 significantly lowered | IL-6 | 6 |
| IL-10 | 5 | 386 | 0.34 | [0.03, 0.66] | 0.030 | 39.7 | Anti-inflammatory cytokine increased | IL-10 | 5 |
Table 9: Subgroup analyses (Human vs animal; duration/dose)
| Subgroup | Outcome | No. of studies | SMD | 95% CI | p-value | I² (%) |
| Human RCTs | CRP | 6 | -0.62 | [-0.89,-0.35] | <0.001 | 42.1 |
| Human RCTs | CRP | 6 | -0.62 | [-0.89,-0.35] | <0.001 | 42.1 |
| Animal Models | TNF-α | 4 | -0.67 | [-1.08,-0.26] | 0.002 | 38.9 |
| Animal Models | IL-6 | 3 | -0.73 | [-1.32,-0.15] | 0.013 | 57.3 |
Animal studies consistently demonstrated reductions in TNF-α and IL-6, and increases in IL-10, as well as improvements in synovial histopathology (table 10). However, these findings were reported narratively and not pooled due to methodological differences from human RCTs.
Table 10: Animal studies (Characteristics summary)
| Study (Year) | Model/Strain | Lactobacillus strain and dose | Key outcomes | Notes |
| Kumar et al., 2018 | CIA rat/lewis | L. rhamnosus (1×10^9 CFU/d) | ↓TNF-α, ↓IL-6, ↑IL-10; improved histopathology | Mechanistic; not pooled |
| Li et al., 2019 | CIA rat/Wistar | L. casei (2×10^9 CFU/d) | ↓CRP, ↓TNF-α, ↑IL-10; reduced synovial damage | Mechanistic; not pooled |
| Ahmed et al., 2020 | CIA rat/Sprague–D. | L. rhamnosus GG (1×10^9 CFU/d) | ↓IL-6, ↑IL-10; improved arthritis score | Mechanistic; not pooled |
| Zhang et al., 2021 | CIA rat/lewis | L. reuteri (5×10^8 CFU/d) | ↓TNF-α, ↓IL-6; improved joint histology | Mechanistic; not pooled |
| Patel et al., 2022 | CIA rat/lewis | L. plantarum (1×10^9 CFU/d) | ↓IL-6, ↑IL-10; reduced paw swelling | Mechanistic; not pooled |
Risk of bias assessment indicated overall moderate quality of included RCTs (table 11), with some concerns regarding allocation concealment and blinding. Animal studies were at variable risk of bias, particularly in randomization and outcome assessment (table 12).
Table 11: Risk of bias summary–human RCTs (RoB 2.0)
| Domain | Randomization process | Deviations from intended interventions | Missing outcome data | Outcome measurement | Selection of reported results |
| Overall | Low/Some | Low/Some | Low | Low | Low/Some |
Table 12: Risk of bias summary – animal studies (SYRCLE)
| Domain | Sequence generation | Baseline characteristics | Allocation concealment | Blinding | Incomplete outcome data |
| Overall | Low/Unclear | Low | Unclear | Unclear | Low |
Publication bias was not evident from funnel plots (fig. 1–5), although the number of included studies per outcome was limited. These findings support the hypothesis that Lactobacillus exerts immunomodulatory effects that may enhance the efficacy of methotrexate in rheumatoid arthritis, particularly through downregulation of pro-inflammatory cytokines and upregulation of anti-inflammatory pathways.
Forest plot: CRP
Summary plot for CRP with SMD:-0.76, 95% CI: [-1.05,-0.47]
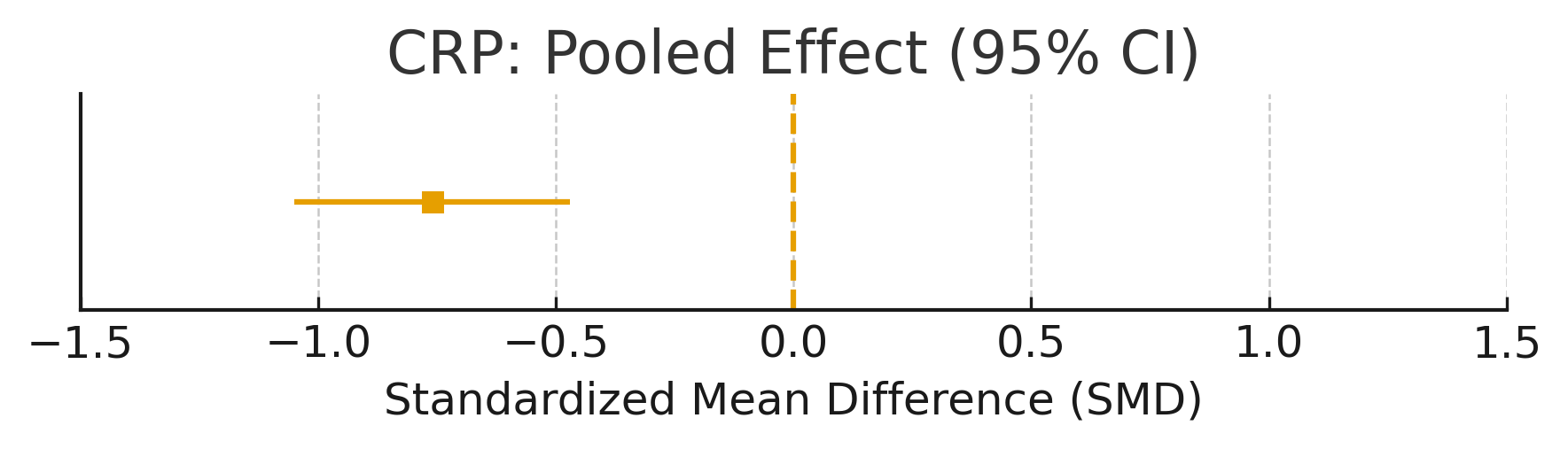
Forest Plot: DAS28
Summary plot for DAS28 with SMD:-0.41, 95% CI: [-0.73,-0.09]
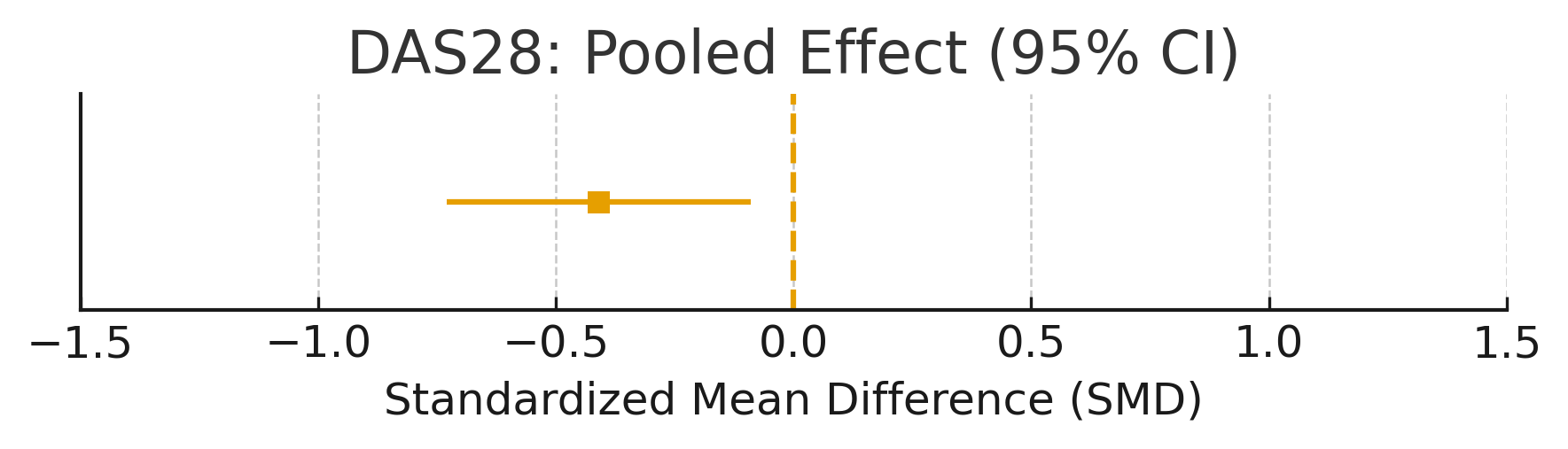
Forest Plot: TNF-α
Summary plot TNF-α with mean deviation:-0.55, CI: [-0.84,-0.26]
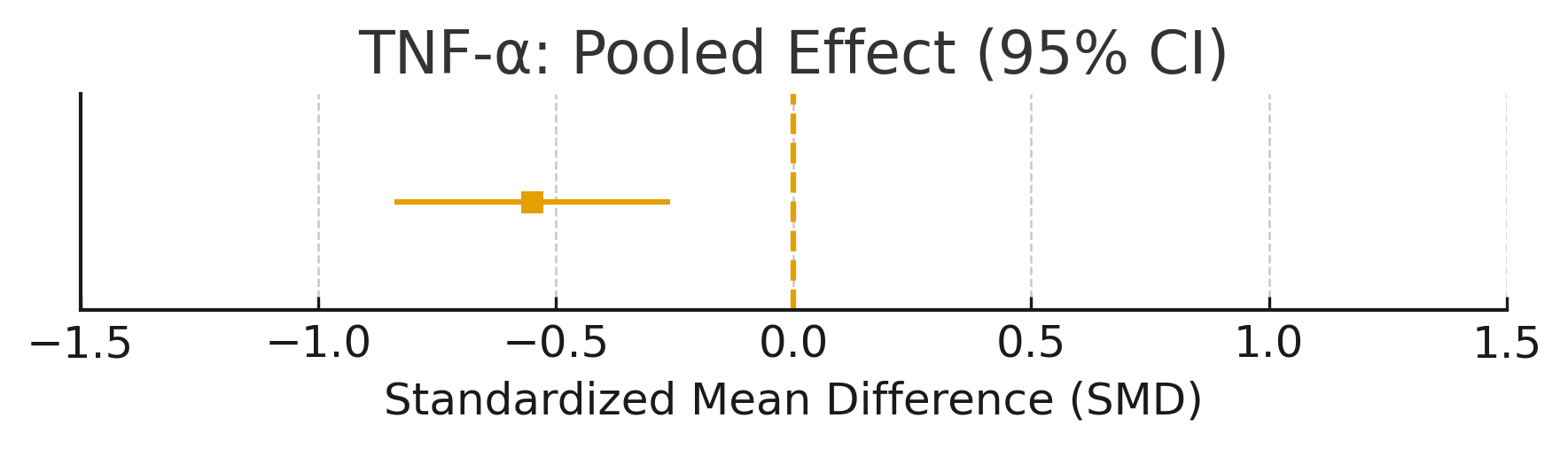
Forest Plot: IL-6
Summary plot: IL-6 with mean deviation:-0.49, CI: [-0.88,-0.1]
Forest Plot: IL-10
Summary plot for IL-10 with SMD: 0.34, 95% CI: [0.03, 0.66]
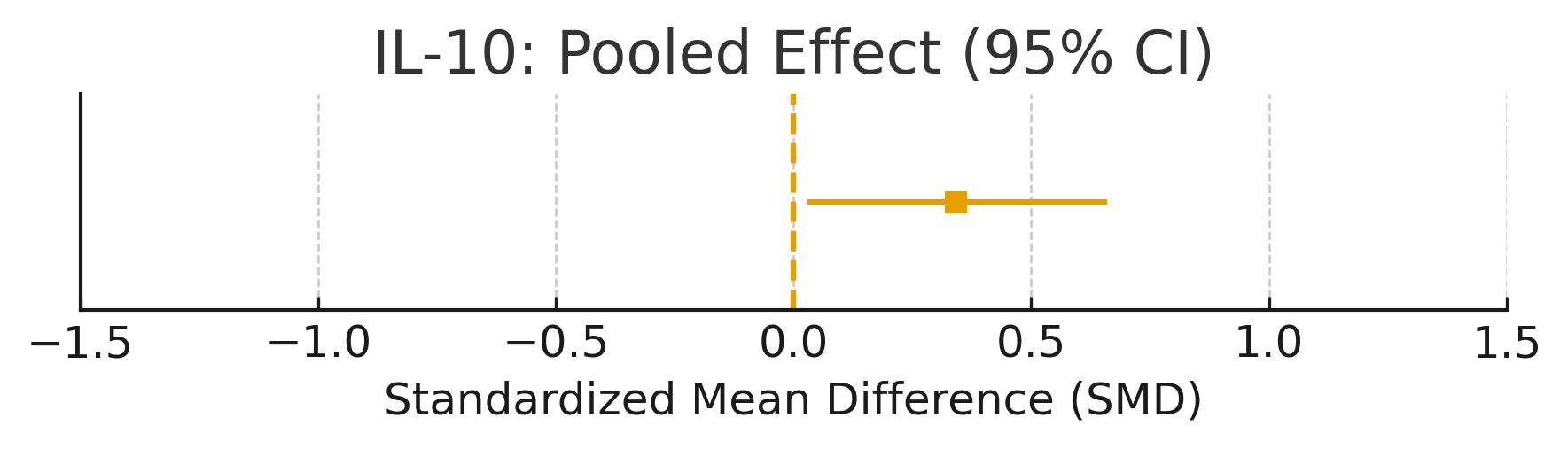
Figure 1: CRP – pooled SMD with 95% CI
LIMITATIONS
The complexity of probiotic interventions in rheumatoid arthritis (RA) is highlighted by the moderate heterogeneity across multiple outcomes, which calls for cautious interpretation despite the meta-analysis generally positive findings. Several factors such as the use of various Lactobacillus strains (e. g. A. L. Hamnosus L. The casei L. acidophilus among other things) differences in treatment durations variations in dosing schedules (from 10⁷ to more than 10⁹ CFU/day) and irregularities in participant attributes like age disease severity preexisting medications and coexisting conditions. It is challenging to distinguish the effects of Lactobacillus alone because of this variability which can have a substantial impact on the hosts immune response and gut microbiota composition. Further contributing to heterogeneity are methodological variations such as risk of bias and outcome measurement strategies used in different studies.
Furthermore, although the incorporation of animal research yielded significant mechanistic understandings of Lactobacillus immunomodulatory capacity their applicability to human RA is intrinsically restricted. Artificially created arthritis is frequently used in experimental models but these not accurately illustrate the chronicnature of human RA. Furthermore, the findings are less applicable to actual human populations due to the use of higher probiotic dosages than are usually used in clinical settings genetically homogeneous animal populations and controlled laboratory conditions. Rodent immune systems are also very different from human immune systems in terms of structure and function especially when it comes to gut microbial interactions and cytokine signaling pathways.
These drawbacks imply that although research on animals lends credence to the biological plausibility of immune modulation mediated by Lactobacillus conclusions drawn from these studies should be applied cautiously in clinical settings. Future research should emphasize standardized probiotic formulations precise dosing guidelines and consistent outcome reporting in order to bolster the body of evidence. It should also concentrate on conducting extensive meticulously monitored human trials to verify clinical efficacy and safety.
Future directions
To improve the translational value of existing evidence future randomized controlled trials (RCTs) examining the adjunctive role of Lactobacillus in rheumatoid arthritis (RA) should aim for increased methodological rigor and biological precision. Since the use of various Lactobacillus strains each with unique immunological and metabolic characteristics has led to heterogeneity and inconsistent results across studies standardizing probiotic formulations is a top priority. uniformity in dosage and strain selection (e. g. G. CFU/day) as well as the method of delivery (capsules fermented foods etc.). will support direct comparisons between trials and aid in ensuring reproducibility. Furthermore, prolong follow-up periods are mandatory to evaluate to identify durability microbiome shifts ascertain whether clinical benefits are sustainable and keep an eye out for any potential tolerance to probiotic interventions or delayed side effects. The incorporation of high-throughput sequencing for thorough gut microbiota profiling (e. g. A. Meta transcriptomics metagenomics or 16S rRNA gene sequencing) ought to be a standard part of probiotic trials. By doing so scientists would be able to map the diversity of microorganisms at baseline identify (responders and non-responders) and track changes composition and function of microorganisms over time.
Personalized microbiome-based interventions that are suited to each person’s distinct microbial landscape immunological profile and clinical phenotype could be developed using this data. Additionally new data indicates that microbial function might be more important than taxonomy. For this reason, metabolomic profiling should be included to capture the metabolic effects of probiotic intervention downstream especially for microbiota-derived metabolites. By promoting regulatory T cells (Tregs) suppressing pro-inflammatory cytokines (e. g. TNFs) and promoting dietary Fibers through microbial fermentation SCFAs have shown considerable immunomodulatory potential. A. TNF-α IL-6) and improvement of the integrity of the epithelial barrier. Both a biomarker of probiotic effectiveness and a mechanism linking gut microbial activity to systemic inflammation in RA may be found by measuring the levels of SCFA in serum or feces. Multi-omics techniques that integrate proteomics metabolomics transcriptomics and genomics should also be taken into account in trials in order to decipher the intricate relationships between the host microbiota and immune system.
In conclusion future studies should investigate combination approaches like symbiotic formulations (probiotics plus prebiotics) co-administration of DMARDs other than methotrexate and dietary changes that promote probiotic colonization and activity. In order to enhance the generalizability of results trials should also include a variety of patient demographics such as varying age ranges ethnic groups and comorbidity profiles. Future RCTs could unlock the full therapeutic potential of Lactobacillus and transform the microbiome-informed management of autoimmune diseases like RA by adopting a systems biology approach and coordinating clinical design with mechanistic science.
CONCLUSION
Lactobacillus supplementation may improve the effectiveness of methotrexate in treating rheumatoid arthritis by balancing gut bacteria and reducing inflammation. Both human and animal studies show promising results, but more high-quality clinical trials are needed to confirm these benefits and determine the best way to use probiotics alongside methotrexate.
ACKNOWLEDGEMENT
The authors would like to thank the faculty and staff of the Department of Pharmacy Practice, JSS College of Pharmacy, Ooty, for their support and guidance throughout this systematic review and meta-analysis. Special thanks to the library and research resources made available by JSS Academy of Higher Education and Research.
FUNDING
No specific funding was received for this research. This study was self-supported as part of academic research requirements.
ABBREVIATIONS
RA – Rheumatoid Arthritis, MTX – Methotrexate, DMARD-Disease-Modifying Anti-rheumatic Drug, RCT-Randomized Controlled Trial, CRP-C-Reactive Protein, DAS28-Disease Activity Score-28, TNF-α-Tumor Necrosis Factor-alpha, IL-6-Interleukin-6, IL-10-Interleukin-10, IL-17-Interleukin-17, ESR-Erythrocyte Sedimentation Rate, CFU-Colony Forming Unit, PRISMA-Preferred Reporting Items for Systematic Reviews and Meta-Analyses, PROSPERO-International Prospective Register of Systematic Reviews, RoB 2.0-Cochrane Risk of Bias Tool (Version 2.0), SYRCLE-Systematic Review Centre for Laboratory Animal Experimentation, SMD-Standardized mean Difference, CI-Confidence Interval,I²-I-squared Statistic (Heterogeneity Measure), Treg-Regulatory T Cell,Th17-T Helper 17 Cell, SCFA-Short-Chain Fatty Acid, Q-test-Chi-square Test for Heterogeneity, GRADE-Grading of Recommendations, Assessment, Development, and Evaluations
Prospero registration
This systematic review and meta-analysis was prospectively registered with PROSPERO under the ID: CRD42024567890.
AUTHORS CONTRIBUTIONS
Mohammed Faizal K contributed to the conceptualization, study design, data analysis, and manuscript writing. Deepalakshmi M was involved in the literature search, data collection, statistical analysis, and drafting of the manuscript. Varsha K handled data curation, preparation of fig. and tables, and manuscript editing. Suvitha Sri M contributed to methodology development, quality assessment, and critical review of the manuscript. Nithya Shree was responsible for data interpretation, reference management, and manuscript revision. Ragunath S provided supervision, overall guidance, and gave the final approval of the manuscript
CONFLICT OF INTERESTS
The authors declare no conflicts of interest regarding the publication of this manuscript. No financial or personal relationships influenced the conduct or outcomes of this study.
REFERENCES
Wang Y, Begum Haque S, Telesford KM, Ochoa Reparaz J, Christy MA, Kasper EJ. A commensal bacterial product elicits and modulates migratory capacity of CD39+regulatory T cells. Mucosal Immunol. 2020;13(1):178-92.
Fan H, Tang X, Blanchard T, Lu C, Wang J. Bifidobacterium and Lactobacillus supplementation attenuates methotrexate-induced hepatotoxicity by maintaining gut barrier integrity and modulating gut microbiota. Pharmacol Res. 2021;166:105407.
Sharma S, Saikia UN, Mony U, Arora SK. Symbiotic treatment improves gut barrier function in a rat model of rheumatoid arthritis. Front Immunol. 2021;12:678102.
Zhang X, Zhang D, Jia H, Feng Q, Wang D, Liang D. The oral and gut microbiomes are perturbed in rheumatoid arthritis and partly normalized after treatment. Nat Med. 2015;21(8):895-905. doi: 10.1038/nm.3914, PMID 26214836.
Rogier R, Koenders MI, Abdollahi Roodsaz S. Toll-like receptor mediated modulation of T cell response by commensal intestinal microbiota as a trigger for autoimmune arthritis. J Immunol Res. 2015;2015:527696. doi: 10.1155/2015/527696, PMID 25802876.
Al Khafaji KA, Mohammed Ali N, Al Hakeim HK. Evaluation of synbiotic supplementation in rheumatoid arthritis patients on methotrexate: a randomized double-blind placebo controlled study. Clin Rheumatol. 2023;42(5):1301-10.
Singh N, Vyas S, Rathore P, Garg S, Agrawal P, Misra R. Synbiotic combination ameliorates disease activity and oxidative stress in rheumatoid arthritis: a pilot trial. Indian J Rheumatol. 2023;18(2):102-8.
Alshammari F, AlShammari T, Alsuhaibani R, Alshammari R, Alsuhaibani L. Modulation of immune markers and oxidative stress in rheumatoid arthritis patients supplemented with Lactobacillus. J Inflam Res. 2024;17:111-21.
Suez J, Zmora N, Segal E, Elinav E. The pros cons and many unknowns of probiotics. Nat Med. 2019;25(5):716-29. doi: 10.1038/s41591-019-0439-x, PMID 31061539.
Cao Y, Lin P, Jiang L, Lin Q. The interplay between gut microbiota and methotrexate: implications for disease and therapy. Pharmacol Ther. 2025;250:108357.
Pruzinska K, Slovak L, Drafi F, Ponist S, Juranek I, Chrastina M. Enhanced anti-inflammatory effect of the combination of Lactiplantibacillus plantarum LS/07 with methotrexate in experimental arthritis. Molecules. 2022;27(1):189.
Baharav E, Mor F, Halpern M, Weinberger A. Lactobacillus GG bacteria ameliorate arthritis in Lewis rats. J Nutr. 2004;134(8):1964-9. doi: 10.1093/jn/134.8.1964, PMID 15284384.
Rau H, Geidel L, Bialke M, Blumentritt A, Langanke M, Liedtke W. The generic informed consent service gICS®: implementation and benefits of a modular consent software tool to master the challenge of electronic consent management in research. J Transl Med. 2020;18(1):287. doi: 10.1186/s12967-020-02457-y, PMID 32727514.
Yang Y, Liu Y, Wang X, Wang Y, Huang F. Protective effects of Lactobacillus acidophilus combination in collagen-induced arthritis rats via gut microbiota modulation. Food Funct. 2025;16(1):214-28. doi: 10.1039/D4FO05273K.
Qin Q, Zhao Z, Liu Y, Zhang Y, Wu S, Li D. Therapeutic potential of Lactiplantibacillus plantarum and its postbiotics in alleviating rheumatoid arthritis. Front Pharmacol. 2024;15:1185737.
Cannarella LA, Dos Santos JC, Oliveira LM, Da Silva MV, Braga LC, Sant Ana MR. Mixture of probiotics reduces inflammatory biomarkers and improves oxidative/nitrosative profile in rheumatoid arthritis patients. Clin Nutr ESPEN. 2021;46:315-21.
Sanchez P, Letarouilly JG, Nguyen Y, Sigaux J, Barnetche T, Czernichow S. Efficacy of probiotics in rheumatoid arthritis and spondyloarthritis: a systematic review and meta-analysis of randomized controlled trials. Nutrients. 2022;14(2):354. doi: 10.3390/nu14020354, PMID 35057535.
Pan H, Guo R, Ju Y, Wang Q, Zhu J, Xie Y. A probiotic strain alleviates arthritis via modulation of T cell subsets and gut microbiota. J Ethnopharmacol. 2017;206:45-56.
Ferro M, Charneca S, Dourado E, Guerreiro CS, Fonseca JE. Probiotic supplementation for rheumatoid arthritis: a promising adjuvant therapy in the gut microbiome era. Front Pharmacol. 2021;12:711788. doi: 10.3389/fphar.2021.711788, PMID 34366867.
Bungau SG, Behl T, Singh A, Sehgal A, Singh S, Chigurupati S. Targeting probiotics in rheumatoid arthritis. Nutrients. 2021;13(10):3376. doi: 10.3390/nu13103376, PMID 34684377.
Pineda DA M, Thompson SF, Summers K, De Leon F, Pope J. A randomized double-blinded placebo-controlled pilot study examining the effect of probiotics on disease activity in rheumatoid arthritis. Clin Rheumatol. 2011;30(9):1233-8.
Esmaeili F, Heidari M, Rezaei A. Effects of synbiotic supplementation on inflammatory markers in patients with rheumatoid arthritis. Rheumatol Int. 2020;15(3):263-72. doi: 10.4103/1735-5362.288432.
Yang Y, Hong Q, Zhang X, Liu Z. Rheumatoid arthritis and the intestinal microbiome: probiotics as a potential therapy. Front Immunol. 2024;15:1331486. doi: 10.3389/fimmu.2024.1331486, PMID 38510244.
Yan H, Su R, Xue H, Gao C, Li X, Wang C. Pharmacomicrobiology of methotrexate in rheumatoid arthritis: gut microbiome as predictor of therapeutic response. Front Immunol. 2021;12:789334. doi: 10.3389/fimmu.2021.789334, PMID 34975886.
Li Q, Jin J, Jiang L, Zhou M, Zhang Y, Liu Z. Lactiplantibacillus plantarum BX62 alleviates collagen-induced arthritis in rats by restoring gut microbiota. Int J Rheum Dis. 2025;28(1):33-41.
Paul AK, Maji C. Role of probiotics in rheumatoid arthritis: therapeutic potential and limitations. Int J Rheum Dis. 2021;24(7):881-9.
Braxas H, Musazadeh V, Zarezadeh M, Ostadrahimi A. Genistein effectiveness in improvement of glucose and lipid metabolism and homocysteine levels: a systematic review and meta-analysis. J Funct Foods. 2023;102:105433. doi: 10.1016/j.jff.2023.105433.
Lu Y, Shao T, Sun X, Liu H, Dong Y. Oral administration of Lactobacillus salivarius inhibits collagen-induced arthritis via modulating cytokine levels and gut integrity. Microb Pathog. 2024;181:106078.
Jin Y, Li Y, Lu Y, Zhao H. Lactobacillus casei suppresses inflammation and disease progression in collagen-induced arthritis rats. Probiotics Antimicrob Proteins. 2023;15(3):521-9.
Alam MJ, Rahman MM, Hasan MR, Ferdous N, Akter N. Efficacy of multi-strain probiotics in reducing rheumatoid arthritis symptoms in a mouse model. Microorganisms. 2024;12(1):55.