
Int J App Pharm, Vol 18, Issue 2, 2026, 60-64Original Article
CHITOSAN-ROSE ESSENTIAL OIL FILMS: EVALUATION OF PHYSICAL INTEGRITY AND BIOPROTECTIVE ACTIVITIES
RULA M. DARWISH1*, ALI H. SALAMA2
1Department of Pharmaceutics and Pharmaceutical Technology, School of Pharmacy, the University of Jordan, Amman-11942, Jordan. 2Department of Pharmaceutical Chemistry, Faculty of Pharmaceutical Sciences, The Hashemite University, Zarqa-13133, Jordan
*Corresponding author: Rula M. Darwish; *Email: rulamdarwish1@gmail.com
Received: 16 Nov 2025, Revised and Accepted: 29 Dec 2025
ABSTRACT
Objective: To develop and characterize chitosan-based films enriched with rose essential oil as a natural, bioactive wound dressing.
Methods: Films were prepared using chitosan as the matrix polymer with rose essential oil incorporated at 0.5%, 1%, and 2% (w/w) concentrations. Physicochemical properties, antioxidant capacity, and antimicrobial activity were evaluated to assess their potential for wound healing applications.
Results: The films exhibited uniform thickness (0.15–0.22 mm) and weight (0.28–0.38 g) with skin-compatible pH (5.8–6.2). Swelling capacity was decreased within creasing roseoil concentration (0.5%: ~250%, 1%: ~188%, 2%: ~120%), indicating controlled fluid uptake. Antioxidant activity rose in a concentration-dependent manner (0.5%: 35% ±2, 1%: 48% ±3, 2%: 62% ±4 DPPH inhibition). Antimicrobial tests showed increased inhibition zones versus control for S. aureus (12–18 mm vs 8 mm), E. coli (10–16 mm vs 7 mm), and C. albicans (8–14 mm vs 6 mm). These findings support enhanced bioactivity with higher rose oil loading.
Conclusion: Chitosan films enriched with rose essential oil demonstrated favorable physicochemical, antioxidant, and antimicrobial properties, supporting their potential as effective natural wound dressings and bioactive delivery systems.
Keywords: Chitosan, Rose essential oil, Wound dressing, Antioxidant activity, Antimicrobial
© 2026 The Authors.Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i2.56521 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Wound healing is a complex, multistage biological process involving hemostasis, inflammation, proliferation, and remodeling, all of which work together to restore tissue integrity after injury. This biological process can be disrupted by several factors, including microbial infection, systemic conditions such as diabetes and vascular disorders and excessive oxidative stress [1, 2]. Among these factors, infection remains one of the most problematic complications because it intensifies local inflammation that contributes to extensive tissue damage, prolongs recovery time, and increases treatment costs [3]. As a result, the development of advanced wound dressings has focused on preventing both primary and secondary infections while simultaneously supporting optimal healing.
An ideal wound dressing should, therefore, provide effective antimicrobial protection, maintain moist environment that promotes natural tissue repair, and avoid damaging surrounding healthy tissue. Natural bioactive compounds, in particular essential oils, have gained significant attention due to their therapeutic potential, anti-inflammatory and antioxidant properties, and lower toxicity compared with many synthetic agents [4–6].
Rose essential oil, derived from Rosa damascena Mill. (Damask rose), is one such essential oil, extensively investigated for its medicinal applications. It is obtained through steam distillation of rose petals, yielding an aromatic oil rich in monoterpenes, sesquiterpenes, and phenolic constituents [7]. These bioactive molecules contribute to a range of wound-relevant activities. Rose oil has demonstrated potent anti-inflammatory effects through modulation of cytokines such as IL-6 and TNF-α, along with broad-spectrum antimicrobial activity against bacteria, fungi, and viruses [8].
Despite these promising properties, the direct clinical use of rose oil is limited by its volatility, poor water solubility, and instability, all of which reduce the effective bioavailability of its active components. Advanced delivery systems including films, gels, and nanoemulsions, have been developed to overcome these challenges by enhancing stability and enabling controlled release. Films are particularly advantageous because they maintain a moist wound environment, support cell migration, reduce scar formation, and act as a protective barrier against contaminants, while ensuring gentle contact with the wound surface [9].
Based on these considerations, this study aims to evaluate roseoil-incorporated chitosan films as a novel wound care approach by examining their physicochemical characteristics, antimicrobial activity against common wound pathogens (Staphylococcus aureus and Escherichia coli), and antioxidant capacity to counteract oxidative stress, a major factor contributing to delayed wound healing.
MATERIALS AND METHODS
Chemicalas and reagents
Chitosan (medium molecular weight, degree of deacetylation ≥75%) was obtained from Sigma-Aldrich (St. Louis, MO, USA). Glacial acetic acid (analytical grade) and glycerol were purchased from Merck (Darmstadt, Germany). Rose essential oil was obtained from a certified commercial supplier (Biosef) and stored in airtight amber vials at 4 °C until use.
2,2-Diphenyl-1-picrylhydrazyl (DPPH) radical and methanol (HPLC/analytical grade) used for antioxidant analysis were purchased from Sigma-Aldrich (USA). Phosphate-buffered saline (PBS, pH 7.4) was prepared using PBS tablets supplied by Oxoid (UK) and distilled water.
Mueller–Hinton agar and Sabouraud dextrose agar were obtained from Oxoid (UK). All reagents were of analytical grade and used without further purification.
Preparation of rose oil films
Chitosan-based films were produced using the solvent casting method. Chitosan (1.5% w/v) was dissolved in 1% (v/v) acetic acid under continuous stirring at room temperature for 24 h, followed by filtration to remove any undissolved residues. Glycerol (1% w/v) was then added as a plasticizer, and the mixture was stirred for an additional 30 min to ensure uniform distribution.
Rose essential oil was incorporated into the chitosan–glycerol solution at three concentrations (0.5%, 1.0%, and 2.0% w/w relative to the chitosan mass). The oil was emulsified using probeul trasonication (200 W nominal power, 40% amplitude, pulsed 5 s on/5 s off for a total of 10 min). The beaker containing the 50 ml mixture (in a 100 ml vessel) was kept in an ice bath to maintain the temperature below 30 °C and prevent thermal degradation of the oil’s bioactive compounds [10].
The resulting emulsions were poured into leveled petri dishes and left to dry at room temperature for 48 h. After drying, the films were carefully peeled off and stored in a desiccator at room temperature until further testing. A control film consisting of chitosan and glycerol without rose oil was prepared under identical conditions for comparative analysis.
Physical characterization of films thickness measurement
Film thickness was determined using a digital micrometer (±0.01 mm). Measurements were taken at five random points per film, and the mean value was reported to ensure uniformity of film formation [11].
PH measurement
The pH of the films was measured to evaluate compatibility with skin. A 1 cm² sample was immersed in 10 ml of distilled water for 1 h, and the pH of the solution was measured using a calibrated pH meter. Maintaining a skin-compatible pH is crucial to avoid irritation and support optimal wound healing [12].
Weight uniformity
Five circular film specimens (2 cm in diameter) were individually weighed using an analytical balance, and the mean weight was calculated. This procedure verified the uniformity of film fabrication and ensured reproducibility of the films’ physical characteristics [13].
Swelling test
The swelling behavior of the wound films was evaluated by assessing their capacity to absorb wound exudates and maintain a moist environment, a key factor in wound healing. Film samples (2 × 2 cm) were weighed before and after immersion in phosphate-buffered saline (PBS, pH 7.4) at 37 °C to simulate physiological conditions. The procedure was repeated at predetermined time intervals over one hour. After each interval, the films were removed, gently blotted with filter paper to remove excess surface fluid, and reweighed. The swelling ratio (DS) was calculated using the following equation:
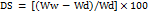
where Ww represents the wet weight of the film and Wd denotes its dry weight [14].
Antioxidant activity (DPPH ASSAY)
The antioxidant activity of the films was evaluated using the 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging assay. Ten milligrams of each film were dissolved in 10 ml of methanol to obtain a stock solution (1 mg/ml). One milliliter of this solution was mixed with 1 ml of 0.1 mmol DPPH in methanol and incubated in the dark for 30 min at room temperature. The absorbance was measured at 517 nm using a UV–Vis spectrophotometer.
The percentage of radical scavenging activity was calculated according to the following equation:
The percentage scavenging activity was calculated using the following formula:
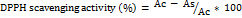
where Abs control is the absorbance of the DPPH solution without sample and Abs sample is the absorbance of the solution with the film extract.
Antimicrobial activity
The antimicrobial activity of the films was assessed against Staphylococcus aureus (ATCC 6538), Escherichia coli (ATCC 25922), and Candida albicans (ATCC 10231) using the agar diffusion method. Film discs (6 mm diameter) were aseptically placed on Mueller–Hinton agar (for bacteria) or Saburou dextrose agar (for fungi) previously inoculated with standardized microbial suspensions.
Bacterial inoculate were adjusted to a 0.5 McFarland standard (~1 × 10⁸ CFU/ml), while fungal inoculum density was standardized to 1–5×10⁶ CFU/ml following CLSI M44 guidelines. Plates were incubated at 37 °C for 24 h, and inhibition zone diameters were measured using a digital caliper. Each assay was performed in triplicate to ensure reproducibility [15].
Statistical analysis
All experiments were conducted in triplicate (n = 3). Data were expressed as mean±standard deviation (SD). Statistical comparisons among groups were performed using one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test. Differences were considered statistically significant at p<0.05 [16].
RESULTS AND DISCUSSION
Film preparation and appearance
Chitosan films incorporated with rose essential oil at concentrations of 0.5%, 1.0%, and 2.0% (w/w) were successfully prepared using the solvent casting technique. The resulting films were transparent, flexible, and smooth, exhibiting a faint yellow coloration that became more intense with increasing oil concentration, indicating successful incorporation of rose oil into the chitosan matrix (fig. 1). These visual characteristics are consistent with previous reports on essential oil–loaded biopolymer films [17].



A B C
Fig. 1: Film morphology (A: 0.5 % rose oil, B: 1% Rose oil, C: 2% rose oil)
Table 1: Physical properties of chitosan films incorporated with rose oil
| Property | 0.5% Rose oil | 1.0% Rose oil | 2.0% Rose oil |
| Thickness (mm) | 0.15±0.02 | 0.18±0.02 | 0.22±0.03 |
| pH | 5.8±0.2 | 6.0±0.2 | 6.2±0.2 |
| Weight (g) | 0.28±0.05 | 0.32±0.06 | 0.38±0.07 |
Value are mean±SD (n = 3).
Physical propertiesss
The physicochemical parameters of the prepared films are summarized in table 1.
A gradual increase in both thickness and weight was observed with higher concentrations of rose oil, likely due to the increased incorporation of oil droplets within the polymer matrix. This trend agrees with earlier findings where essential oil inclusion led to proportional increases in film thickness and mass [18].
All films demonstrated skin-compatible pH values (5.8–6.2), which closely align with the natural pH of human skin. Maintaining this slightly acidic pH is advantageous for wound management, as it minimizes irritation, supports the skin barrier, and inhibits microbial colonization. The observed uniformity in physical parameters reflects the reproducibility and consistency of the film fabrication process [18].
Swelling behavior
The swelling behavior of chitosan–rose oil films is shown in fig. 2. Swelling ratios after one hour were 250% for 0.5% rose oil, 188% for 1.0%, and 120% for 2.0%. The reduction in swelling with increasing oil concentration is attributed to the hydrophobic nature of rose oil, which limits water absorption within the polymeric network.
Controlled swelling is critical for wound dressings, as it helps regulate exudate absorption and maintains a moist environment without oversaturation. The moderate swelling of the 1% and 2% formulations suggests suitability for wounds with low to moderate exudate levels. These results are consistent with other studies on hydrophobic essential oil–incorporated chitosan films.
Antioxidant activity (DPPH assay)
The antioxidant activity of the films increased proportionally with rose oil concentration (fig. 3). DPPH radical inhibition was 35±2% for 0.5%, 48±3% for 1.0%, and 62±4% for 2.0% rose oil.
This concentration-dependent enhancement is attributed to the phenolic and terpenoid constituents of rose essential oil, known for their free radical scavenging capacity [20]. By reducing oxidative stress, these compounds may promote tissue regeneration and accelerate wound healing. The findings corroborate previous research demonstrating that essential oil incorporation enhances the antioxidant potential of chitosan-based biomaterials.
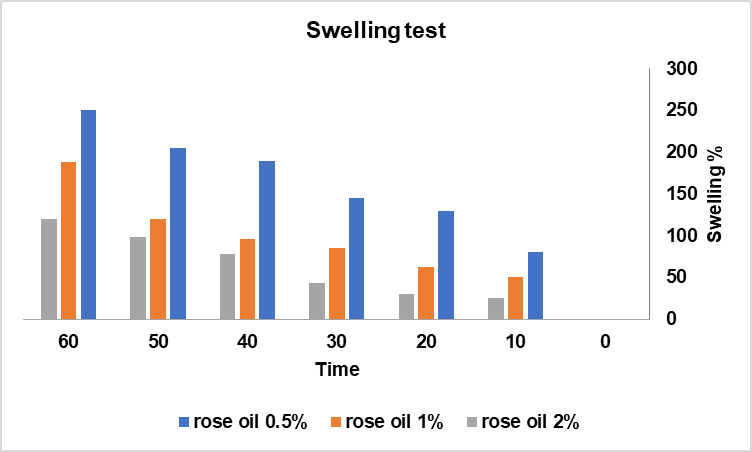
Fig. 2: Swelling test for different concentration of rose oil, values are mean (n = 3) [19]
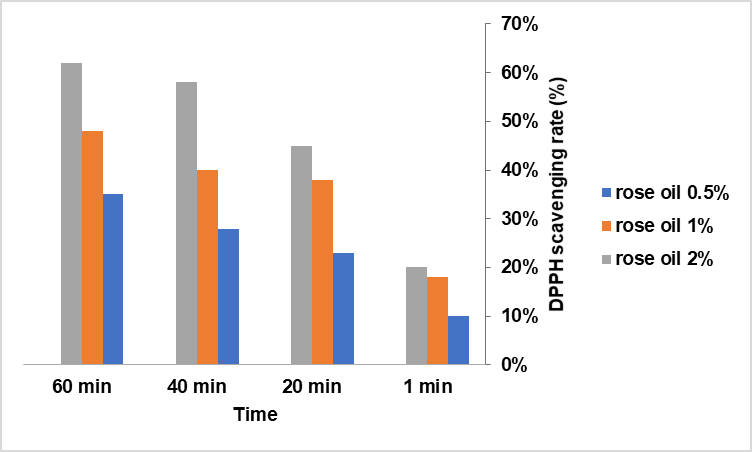
Fig. 3: DPPH assay for the rose oil, values are mean (n = 3)
Table 2: Antimicrobial activity of chitosan films with varying rose oil concentrations
| Microorganism | 0.5% rose oil (mm) | 1.0% rose oil (mm) | 2.0% rose oil (mm) | Control (Chitosan+glycerol) (mm) |
| S. aureus | 12.12±0.05 | 15.23±0.046 | 18.45±0.003 | 8.15±0.005 |
| E. coli | 10.23 ±0.23 | 13.43±0.038 | 16.40±0.031 | 7.37±0.006 |
| C. albicans | 8.00±0.005 | 11.25±0.09 | 14.36±0.002 | 6.45±0.002 |
Value are mean±SD (n = 3).
Antimicrobial activity
The antimicrobial efficacy of the chitosan–rose oil films against Staphylococcus aureus, Escherichia coli, and Candida albicans is presented in table 2.
The films exhibited significant antimicrobial activity that increased with rose oil concentration (p<0.05, ANOVA). The strongest inhibition zones were observed for S. aureus, suggesting greater susceptibility of gram-positive bacteria compared to gram-negative strains, which possess an outer membrane that restricts permeability.
Chitosan itself exerts intrinsic antimicrobial action due to its cationic amino groups, which interact with negatively charged microbial membranes, leading to cell leakage and death. The enhanced efficacy observed in the rose oil–incorporated films reflect a synergistic effect between chitosan and the bioactive terpenes of rose oil [21].
Furthermore, the antifungal activity against C. albicans highlights the films’ broad-spectrum potential. This property is particularly relevant for chronic wounds, where fungal colonization can delay healing and complicate infection control [22].
Collectively, these findings suggest that rose oil incorporation enhances both the antioxidant and antimicrobial performance of chitosan films, rendering them promising candidates for multifunctional wound dressings.
All in all, the developed films exhibited desirable physicochemical stability, controlled swelling, skin-compatible pH, and potent bioactivity. The concentration-dependent enhancement of antioxidant and antimicrobial properties underscores the formulation’s potential as a natural, biocompatible alternative to synthetic dressings. The integration of rose essential oil within the chitosan matrix represents a synergistic approach that combines mechanical integrity with sustained therapeutic efficacy.
CONCLUSION
This study successfully developed and characterized chitosan-based films incorporated with rose essential oil for potential wound-healing applications. The films exhibited desirable physicochemical properties, including uniform thickness, controlled swelling behavior, and skin-compatible pH values. A concentration-dependent increase in antioxidant and antimicrobial activity was observed, highlighting the contribution of rose oil’s phenolic and terpenoid constituents.
These findings demonstrate that rose oil–enriched chitosan films combine mechanical stability with bioactive functionality, making them promising candidates for natural, biocompatible wound dressings. Their dual antioxidant and antimicrobial properties may contribute to faster healing and infection prevention, addressing key challenges in chronic wound management.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
R. M. D. was responsible for the conceptualization of the study, supervision of the research process, and critical revision of the manuscript. R. M. D. and A. H. S. jointly conducted the experimental design, data acquisition, data analysis, and initial drafting of the manuscript. All authors have read and approved the final version of the manuscript.
CONFLICT OF INTERESTS
Declared none
REFERENCES
Yeung YF. Complications in vascular and endovascular surgery: how to avoid them and how to get out of trouble. Surgical Practice. 2012 Aug;16(3):124. doi: 10.1111/j.1744-1633.2012.00608.x.
Harrison DG. Basic science: pathophysiology: oxidative stress. J Am Soc Hypertens. 2014 Aug 1;8(8):601-3. doi: 10.1016/j.jash.2014.07.002, PMID 25151322.
Leaper D, Assadian O, Edmiston CE. Approach to chronic wound infections. Br J Dermatol. 2015 Aug 1;173(2):351-8. doi: 10.1111/bjd.13677, PMID 25772951.
Hamedi A, Zomorodian K, Safari F. Antimicrobial activity of four medicinal plants widely used in Persian folk medicine. Res J Pharmacognosy. 2015 Jan;2(1):25-33.
Wang H, Chen Y, Wang L, Liu Q, Yang S, Wang C. Advancing herbal medicine: enhancing product quality and safety through robust quality control practices. Front Pharmacol. 2023 Sep 25;14:1265178. doi: 10.3389/fphar.2023.1265178, PMID 37818188.
Criollo Mendoza MS, Contreras Angulo LA, Leyva Lopez N, Gutierrez Grijalva EP, Jimenez Ortega LA, Heredia JB. Wound healing properties of natural products: mechanisms of action. Molecules. 2023 Jan 6;28(2):598. doi: 10.3390/molecules28020598, PMID 36677659.
Baumann LS. Rosa Damascena. Skin Allergy News. 2011 Aug;42(8):18. doi: 10.1016/s0037-6337(11)70421-1.
Masyita A, Mustika Sari R, Dwi Astuti A, Yasir B, Rahma Rumata N, Emran TB. Terpenes and terpenoids as main bioactive compounds of essential oils their roles in human health and potential application as natural food preservatives. Food Chem X. 2022;13:100217. doi: 10.1016/j.fochx.2022.100217, PMID 35498985.
Chrubasik C, Duke RK, Chrubasik S. The evidence for clinical efficacy of rose hip and seed: a systematic review. Phytother Res. 2006 Jan;20(1):1-3. doi: 10.1002/ptr.1729, PMID 16395741.
Borbolla Jimenez FV, Pena Corona SI, Farah SJ, Jimenez Valdes MT, Pineda Perez E, Romero Montero A. Films for wound healing fabricated using a solvent casting technique. Pharmaceutics. 2023 Jul 9;15(7):1914. doi: 10.3390/pharmaceutics15071914, PMID 37514100.
Walton J, Alexander MR, Fairley N, Roach P, Shard AG. Film thickness measurement and contamination layer correction for quantitative XPS. Surf Interface Anal. 2016 Jan 22;48(3):164-72. doi: 10.1002/sia.5934.
Chopra H, Bibi S, Kumar S, Khan MS, Kumar P, Singh I. Preparation and evaluation of chitosan/PVA-based hydrogel films loaded with honey for wound healing application. Gels. 2022 Feb 11;8(2):111. doi: 10.3390/gels8020111, PMID 35200493.
Mariani F, Serafini M, Gualandi I, Arcangeli D, Decataldo F, Possanzini L. Advanced wound dressing for real-time pH monitoring. ACS Sens. 2021 Jun 2;6(6):2366-77. doi: 10.1021/acssensors.1c00552, PMID 34076430.
Gasti T, Hiremani VD, Sataraddi SP, Vanjeri VN, Goudar N, Masti SP. UV screening, swelling and in vitro cytotoxicity study of novel chitosan/poly (1-vinylpyrrolidone-co-vinyl acetate) blend films. Chem Data Collect. 2021 Jun;33:100684. doi: 10.1016/j.cdc.2021.100684.
Ait Ouahioune L, Wrona M, Becerril R, Salafranca J, Nerin C, Djenane D. Ceratonia siliqua L. kibbles seeds and leaves as a source of volatile bioactive compounds for antioxidant food biopackaging applications. Food Packaging Shelf Life. 2022;31:100764. doi: 10.1016/j.fpsl.2021.100764.
Abdollahzadeh E, Nematollahi A, Hosseini H. Composition of antimicrobial edible films and methods for assessing their antimicrobial activity: a review. Trends Food Sci Technol. 2021;110:291-303. doi: 10.1016/j.tifs.2021.01.084.
Nguyen TA. Characterization of chitosan-based active film incorporating with gallic acid. IJMRA. 2023 Aug 21;6(8):3693-8. doi: 10.47191/ijmra/v6-i8-40.
Elshamy S, Khadizatul K, Uemura K, Nakajima M, Neves MA. Chitosan-based film incorporated with essential oil nanoemulsion foreseeing enhanced antimicrobial effect. J Food Sci Technol. 2021 Jan 22;58(9):3314-27. doi: 10.1007/s13197-020-04888-3, PMID 34366449.
Molina Torres MA, Gimenez PA, Mercadal PA, Alvarez Igarzabal CI, Gonzalez A. Enhancing the properties of soy protein films via riboflavin photo-crosslinking and their application in preventing photo-oxidation of chia oil. Food Hydrocoll. 2024 May;155:110185. doi: 10.1016/j.foodhyd.2024.110185.
Gunay Z, Yıldırım HK, Rahim SC. Evaluation of bioactive components and antioxidant activities of Rose canina L. Viburnum opulus L. Berberis vulgaris L. and Berberis integerrima L. subjected to different extraction methods. Croat J Food Sci Technol. 2025 Jun 27;17(1):20-36. doi: 10.17508/cjfst.2025.17.1.02.
Yan D, Li Y, Liu Y, Li N, Zhang X, Yan C. Antimicrobial properties of chitosan and chitosan derivatives in the treatment of enteric infections. Molecules. 2021 Nov 25;26(23):7136. doi: 10.3390/molecules26237136, PMID 34885715.
Park KJ, Lee JS, Jo HJ, Kim ES, Lee HG. Antimicrobial and indicator properties of edible film containing clove bud oil-loaded chitosan capsules and red cabbage for fish preservation. Int J Biol Macromol. 2022 Jan;196:163-71. doi: 10.1016/j.ijbiomac.2021.12.027, PMID 34920069.