
Int J App Pharm, Vol 18, Issue 1, 2026, 332-339Original Article
FABRICATION AND ASSESSMENT OF DIFFERENT COMPOSITE BIOMATERIAL SCAFFOLDS FOR SKIN REPAIR AND REGENERATION
MORUSU KEERTHANA1, KAMMARI RADHA1, MARUVAJALA VIDYAVATHI1*, RAYADURGAM VENKATASURESH KUMAR2
1Institute of Pharmaceutical Technology, Sri Padmavati Mahila Visvavidyalayam, Tirupati, India. 2Department of Surgery and Radiology, S. V. Veterinary University, Tirupati, India
*Corresponding author: Maruvajala Vidyavathi; *Email: vidyasur@rediffmail.com
Received: 14 Aug 2025, Revised and Accepted: 16 Jan 2026
ABSTRACT
Objective: The healing of a wound in the shortest possible time, with minimal pain and discomfort, is the prime objective of wound management. This study was focused to prepare and compare Chitosan and Gelatin polymers with sodium alginate (CS and GS) separately in the form of Composite scaffolds.
Methods: Solvent casting was chosen to prepare six different composite scaffolds in three stages, containing different proportions of chitosan: sodium alginate and gelatin: sodium alginate. The best among 6 scaffolds prepared in stage-1 was selected to study the effect of plasticizer (ethylene glycol) in 3 different concentrations in stage 2. The best of the different scaffolds of stage 2 was used to study the effect of cross-linking agent (CaCl2) in 2 different concentrations in stage 3. The best of different scaffolds of stage 3 was used for the evaluation of in vitro antibacterial activity and in vivo wound healing activity.
Results: Results of the study revealed that the increase in sodium alginate concentration shown the best physico-mechanical properties in both CS and GS, which further acquired the best properties with selected highest concentrations of crosslinker and plasticizer. Folding endurance of CGS11 and CCS11 scaffolds were found to be 363±0.57 and 353±18.05, respectively. CCS and CGS scaffolds showed better zone of inhibition against both Gram-positive and negative bacteria. The hydroxyproline and hexosamine contents released from group treated with CGS11 on day 11 were 4.369±0.59, 2.045±0.001, respectively which were found higher than contents released from groups treated with CCS 11 (2.484±0.05 and 1.478±0.001). Percentage wound contraction on day 14 of CGS11 and CCS 11 was found to be 99.95±4.97% and 95.75±3.64%, respectively. Photography of wounds and histopathological studies revealed that the experimental group IV treated with CGS showed better and faster healing than the control group and group treated with CCS 11.
Conclusion: Present study concluded that, among all formulations, the CGS11 scaffold exhibited superior wound contraction and histological regeneration compared to CCS11. Composite bio scaffolds can be efficiently used for wound healing to replace synthetic antibiotics. The Composition of bioscaffolds with chitosan and gelatin in combination with sodium alginate highly impacts and improves the physico-mechanical and therapeutic properties of the scaffolds, skin repair and regeneration.
Keywords: Tissue repair, Wound healing, Chitosan, Gelatin, Sodium alginate, Biomaterial scaffolds
© 2026 The Authors.Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i2.56532 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Scaffolds are temporary dressings of wounds, one of the best healing approaches used in wound healing. These scaffolds can carry drugs also for local action on a tissue near the site of application and promote healing at an injured area of the body. The demand for new wound healing materials with inherent antimicrobial properties is on the rise [1].
In that way of searching, Polysaccharides of low antigenic nature are a good choice in preparation of wound healing scaffolds. However, polysaccharides alone cannot provide the desirable physico mechanical properties suitable for application on the skin. Hence, the proposed work concentrated on combining the effects of more than one natural polysaccharide to construct new material surfaces to facilitate tissue regeneration during wound management. The composite scaffolds contain two or more polymers; and can be indicated for wound treatment due to their improved mechanical strength, transparency and excellent haemostatic properties [2-4].
The novelty of this study lies in the systematic comparative evaluation of chitosan–sodium alginate (CCS) and gelatin–sodium alginate (CGS) composite scaffolds for wound healing, optimized through sequential modification with plasticizer and crosslinker concentrations. It uniquely demonstrates the best polymer among gelatin and chitosan when combined with sodium alginate by evaluating physico-mechanical properties, anti-bacterial activity, and in vivo wound healing potential. The study highlights an innovative, biopolymer-based scaffold design that can effectively replace synthetic wound dressings while promoting faster and more complete skin regeneration.
FT-IR Compatibility studies and SEM analysis were helpful to ascertain the compatibility between polymers and surface morphology of the prepared scaffolds
MATERIALS AND METHODS
Materials
Chitosan, Gelatin and Sodium alginate were obtained from Himedia Laboratory Pvt. Ltd, Nashik, India Agar-agar, peptone, beef extract and other media components were obtained from Qualigens Fine Chemicals, Mumbai, India. Solvents used in the work were procured from Molychem Pvt. Ltd. Mumbai, India.
Methods
Stage 1: Preparation of chitosan-sodium alginate composite scaffolds (CS)
Chitosan (1% w/v) and sodium alginate (1.5%w/v) solutions were mixed at high speed with a composition shown in table 1. The prepared solutions were vigorously stirred by mechanical stirrer and vacuum-filtered for the removal of any entrapped air bubbles. Then the solution was cast into the scaffolds by pouring them in petri plates and dried. All the Chitosan-sodium alginate (CS) composite scaffolds (6 inches in length and 4.5 inches in width) were prepared by solvent casting technique [5].
Preparation of gelatin-sodium alginate composite scaffolds (GS)
Gelatin (1%w/v) and sodium alginate (1.5%w/v) solutions were mixed at high speed with a composition shown in table 1. These scaffolds were also prepared by following similar procedure mentioned in the preparation of chitosan-sodium alginate scaffolds [6, 7].
Stage-2: Study of the effect of plasticizer (Ethylene glycol)
The best among six scaffolds of both combinations, chitosan: sodium alginate (CS) and gelatin: sodium alginate (GS) composite scaffolds were selected and further, the effect of plasticizer concentration on the characteristics of scaffolds was studied by preparing PCS (Plasticizer: Chitosan: Sodium alginate) and PGS (Plasticizer: Gelatin: Sodium alginate) scaffolds with different concentrations of ethylene glycol by following a similar procedure and compositions shown in table 1.
Stage-3: Study of the effect of cross-linking agent
The best concentration of ethylene glycol was selected from PCS and PGS composite scaffolds and further, the effect of concentration of cross-linker on scaffolds was studied with the help of different concentrations of CaCl2 (cross-linker) as shown in table 1 with the following procedure.
Polymer solutions were prepared separately by dissolving chitosan, gelatin, and sodium alginate of a selected ratio in distilled water containing selected concentration of ethylene glycol at 30 °C. The solutions were mixed and stirred mechanically at 800 rpm for 1 hour to achieve complete dissolution. Later the mixture temperature rose to 70 °C and a solution of 1% w/v calcium chloride (CaCl2) was added slowly to the mixture to work as the primary cross-linking agent. This film-forming solution was then transferred into Petri plates. The plates were brought to an oven at 40 °C. Then, the scaffolds were removed from the petri plate and maintained on a support for 20 hr, at an ambient temperature, for drying [8]. The scaffolds were stored in a desiccator for a period of three days to reach the equilibrium moisture content before its characterization [9].
Characterization of scaffolds
The prepared scaffolds were characterized for Physico-mechanical properties like Thickness, Folding endurance, Swelling behaviour, tensile parameters and Texture parameters. Those were also evaluated for the anti-bacterial activity. All the tests were conducted in triplicate by random selection of scaffolds from three different places of the prepared scaffolds.
Table 1: Composition of different scaffolds
| S. No. | Code of scaffold | Number of parts | Amount of plasticizer (Ethylene glycol) (ml) | Crosslinking agent CaCl2 (1% w/v)(ml) |
||
Chitosan (1%w/v) (ml) |
Gelatin (1%w/v) (ml) | Sodium alginate (1.5%w/v) (ml) |
||||
| Stage 1: Chitosan-sodium alginate | ||||||
| 1 | CS1 | 1 | - | 10 | 0.2 | - |
| 2 | CS2 | 1 | - | 20 | 0.2 | - |
| 3 | CS3 | 1 | - | 30 | 0.2 | - |
| 4 | CS4 | 1 | - | 40 | 0.2 | - |
| 5 | CS5 | 1 | - | 50 | 0.2 | - |
| 6 | CS6 | 1 | - | 60 | 0.2 | - |
| Stage-2: Study of effect of plasticizer | ||||||
| 7 | PCS7 | 1 | - | 60 | 0.4 | - |
| 8 | PCS8 | 1 | - | 60 | 0.6 | - |
| 9 | PCS9 | 1 | - | 60 | 0.8 | - |
| Stage-3: Study of effect of cross-linking agent | ||||||
| 10 | CCS10 | 1 | - | 60 | 0.8 | 0.5 |
| 11 | CCS11 | 1 | - | 60 | 0.8 | 1 |
| Stage-1: Gelatin-sodium alginate | ||||||
| 12 | GS1 | - | 1 | 10 | 0.2 | - |
| 13 | GS2 | - | 1 | 20 | 0.2 | - |
| 14 | GS3 | - | 1 | 30 | 0.2 | - |
| 15 | GS4 | - | 1 | 40 | 0.2 | - |
| 16 | GS5 | - | 1 | 50 | 0.2 | - |
| 17 | GS6 | - | 1 | 60 | 0.2 | - |
| Stage-2: Study of effect of plasticizer | ||||||
| 18 | PGS7 | - | 1 | 60 | 0.4 | - |
| 19 | PGS8 | - | 1 | 60 | 0.6 | - |
| 20 | PGS9 | - | 1 | 60 | 0.8 | - |
| Stage-3: Study of effect of cross-linking agent | ||||||
| 21 | CGS10 | - | 1 | 60 | 0.8 | 0.5 |
| 22 | CGS11 | - | 1 | 60 | 0.8 | 1 |
Physico-mechanical properties
Thickness
Thickness was determined to find the uniformity in the scaffolds. The thickness of the scaffolds was measured using a screw gauge in triplicate and the average value was determined [10].
Folding endurance
The folding endurance test was used to find the flexibility of scaffolds. It was determined by repeatedly folding one scaffold at the same place till it broke. The number of times the scaffolds could be folded at the same place without breaking gives the value of folding endurance [10].
Swelling behaviour
The swelling behaviour of scaffolds was investigated at room temperature by exposing them to PBS solution. A known weight of scaffold (Wd) material was placed in the PBS solution for 30 min. The wet weight of the scaffold (Ww) was determined after blotting the scaffold surface with filter paper to remove excess surface water. The percentage of water absorption (W SW) known as degree of swelling of the scaffold was calculated using below expression:
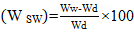
Tensile parameters
The Tensile parameters measure the ability of scaffolds to withstand the mechanical pressure or the force required to break the scaffolds. Tensile strength of the scaffolds was determined by using a texture analyzer (TA-XT PLUS stable system analyzer) then maximum force (N), maximum elongation at break (sec) and tensile strength (mpa) were calculated [10].
Texture parameters
The scaffolds may be exposed to force/pressure and causing deformation or rupture during manufacturing, packaging and transportation. This strength of a scaffold was quantified using the (TA-XT PLUS) Texture Analyzer by a penetration test using a 5 mm cylinder probe and the burst strength (rupture point) and burst time (time at which burst) were determined [11].
In vitro antibacterial activity
In vitro antibacterial activity of the best-selected scaffold was estimated by agar disc diffusion method using two g positive (Bacillus subtilis, Staphylococcus aureus) and two g negative bacteria (Escherichia coli, Pseudomonas aeruginosa) obtained from National chemical Laboratory, Pune. Nutrient Agar medium was poured into boiling tubes and sterilized by an autoclave. After sterilization, two g positive and g negative organisms were inoculated and the composite scaffolds of 8 mm2 area and thickness of 105 µm were placed on the solidified agar medium. The petri plates were incubated for 24 h at 37±1 C. The antibacterial activity was evaluated by measuring the zone of growth inhibition of bacteria. This was done in triplicate for the best composite scaffold with each bacteria and an average diameter was noted [12].
Polymer compatibility studies
Fourier transform infrared (FT-IR) analysis
This study was carried out to find out the compatibility between polymers used in scaffolds. The scaffold was kept in a sample holder and scanned from 4000 cm-1 to 400 cm-1 in FT-IR spectrophotometer. The possible interaction between chitosan, gelatin and sodium alginate was accessed by comparing FT-IR spectra of selected best composite scaffolds [13, 14].
Scanning electron microscopy (SEM) analysis
The surface morphology of the best composite scaffold was determined by scanning electron microscopic method (SEM, Zeiss Ultra-60). The scaffold was taken and mounted directly on the SEM stub, using double-sided, sticking tape and scanned in a high vacuum chamber with a focused electron beam emitted from the sample. Then, the SEM microphotographs of selected composite scaffolds were obtained [15].
In vivo studies
The best-selected scaffold based on the performance in in vitro tests was used to study the in vivo wound healing activity on albino rats by excision wound model after taking approval from IAEC (CPCSEA/1677/PO/Re/S/2012/IAEC/13).
Pathogen-free adult female albino rats obtained from Sri Venkateswara Enterprises, Bangalore, India, weighing 150 – 200 gm were selected. The rats were housed in polypropylene cages under standard laboratory conditions with a 12-hour light and dark cycle. The rats were fed with standard laboratory chow (Hindustan Lever Limited, Mumbai) and water. The rats were treated as per the designed protocol.
Procedure
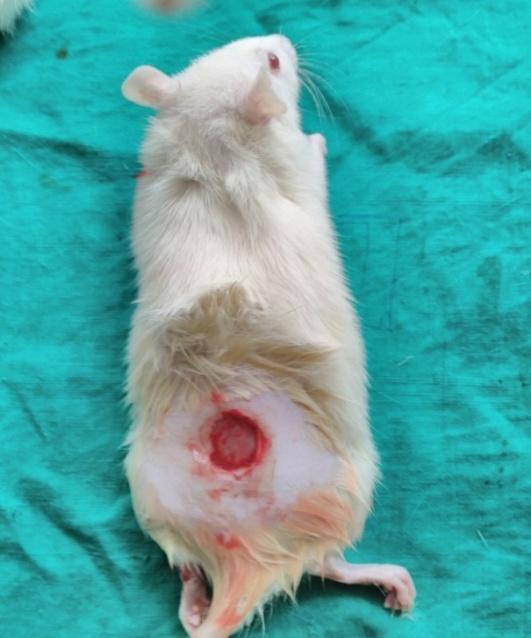
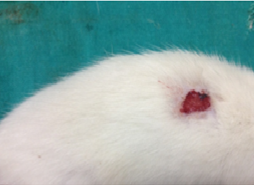
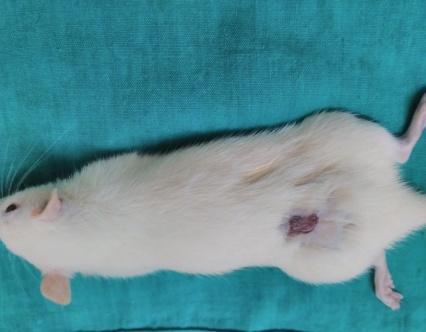
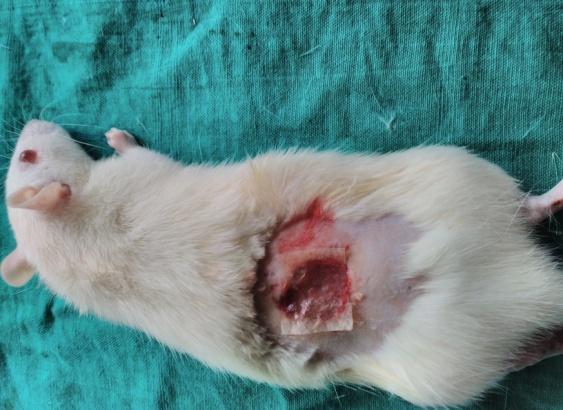
The anesthetized animal was placed on the operation table in a normal position. The dorsal fur of the animals was shaved with an electric clipper and the anticipated area (2 cm. length and 2 cm. width) of the wound to be created was outlined on the back of the animals on an inter-scapular region i. e., 5 mm. away from ears. Skin with full thickness from the demarcated area was excised to get a wound area of 2 sq. cm. After achieving homeostasis, the wound was blotted with sterile gauze in the control group (group-I) and the respective scaffolds were placed on the wound of animals in the treatment groups (groups II and III) and covered with sterile gauze. Then, the following parameters were determined at specific time intervals [16].
Percentage of wound contraction
This was measured to determine the reduction in wound area at different periods of treatment. It was determined by a graphical method. Wound area was measured on the 7th, 14th, 21st, and 28th post-wounding day by counting the number of squares of retraced wound area on graph paper. The degree of wound healing was calculated as % closure of the wound area from the original wound using a formula.
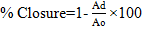
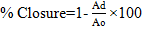
Ao – Wound area on day zero.
Ad-Wound area on corresponding days.
Biochemical studies
Granulation tissue was collected from two groups on 0 and 11th day and washed with cold saline (0.9% NaCl) to remove blood clots and stored in normal saline solution at-20 °C for estimation of the following biochemicals [17].
Estimation of hydroxyproline (mg/gm)
Granulation tissue weighing around 300 mg was homogenized in 10 ml of 6N HCl and heated for hydrolysis. Then these were washed and combined with the hydrolysate, methyl red was added and shaken thoroughly. This was followed by the adjustment of pH to 6 – 7. The sample was diluted to 50 ml with distilled water. To 2 ml of the above sample, 1 ml of chloramine –T solution and 1 ml perchloric acid were added. To all the test tubes 1 ml of PDAB solution was added. The absorbance of the purple-coloured solution was measured at 557 nm by using a calorimeter. A standard curve was prepared and the amount of hydroxyproline was determined and expressed as mg/gm wt. of tissue.
Estimation of hexosamine (µg/gm)
Hexosamine is an amino sugar, measured using a spectrophotometer at 530 nm. Granulation tissue weighing around 300 mg was homogenized in 10 ml of 6N HCl and subjected to hydrolysis. The pH of the hydrolysate was adjusted to 7 using 4 N NaOH. The sample was diluted to 50 ml with distilled water. To the 2 ml of above sample, 1 ml of 2% acetylacetone was added and heated to 96 °C for 40 min. The mixture was cooled and 5 ml of 96% ethanol was added followed by the addition of 1 ml of Ehrlich's reagent. After 1hour, the absorbance of this solution was measured at 530 nm. The amount of hexosamine was determined by comparing with a standard curve. Hexosamine content has been expressed as μg/gm wt. of the tissue.
Histopathological studies
Biopsy specimens for histopathological examination were collected on 7th, 14th, day of post-treatment from all the groups and preserved in 10% buffered formalin. They were processed by routine paraffin embedding technique. i. e., 5 to 6 microns thick sections were cut and stained with hematoxylin and eosin [18, 19]. The specimens were collected by trephining that involved skin tissue of both healing and normal skin using corneal trephiner.
Photography
The photographs of wounds from different groups were taken at specific intervals for visual comparison.
Statistical analysis
The results are expressed as mean±SD. Statistical analysis of in vitro parameters was tested with one-way ANOVA. The results of in vivo animal experiments were analysed by two tailed, unpaired student’s t-test. Statistical significance was set accordingly at p = 0.05 level.
RESULTS
Physico-mechanical properties
The results of physico-mechanical properties of all CS and GS scaffolds and results are shown in table 2 and 3 respectively
Zone of inhibition in in vitro antibacterial studies
Four different bacteria incubated with selected best scaffolds (CCS11, CGS11) along with one control and its diameter of zone of inhibitions (in cm) is noted. The CCS11 and CGS11 Scaffolds have shown different Zone of inhibition values for different bacteria. CCS11 and CGS11 showed zones of inhibition of 2.1±0.2 cm and 2.33±0.152 cm against Bacillus subtilis; 3.1±0.2 cm and 2.56±0.305 cm against Staphylococcus aureus; 3.0±0.05 cm and 2.7±0.264 cm against Escherichia coli; and 2.6±0.60 cm and 2.96±0.416 cm against Pseudomonas aeruginosa, respectively.
FT-IR spectroscopy
FT-IR spectra of selected the best composite scaffolds CCS-11and CGS-11 were shown in fig. 1 and 2.
SEM
Surface morphology of selected the best composite scaffolds was determined by scanning electron microscopy and its scanning electron microphotographs are shown in fig. 3.
In vivo studies
Percentage of wound contraction
The percentage of wound contraction in untreated and treated groups was measured on the 7th, 14th, 21st and 28th post-wounding day and the results are shown in table 4.
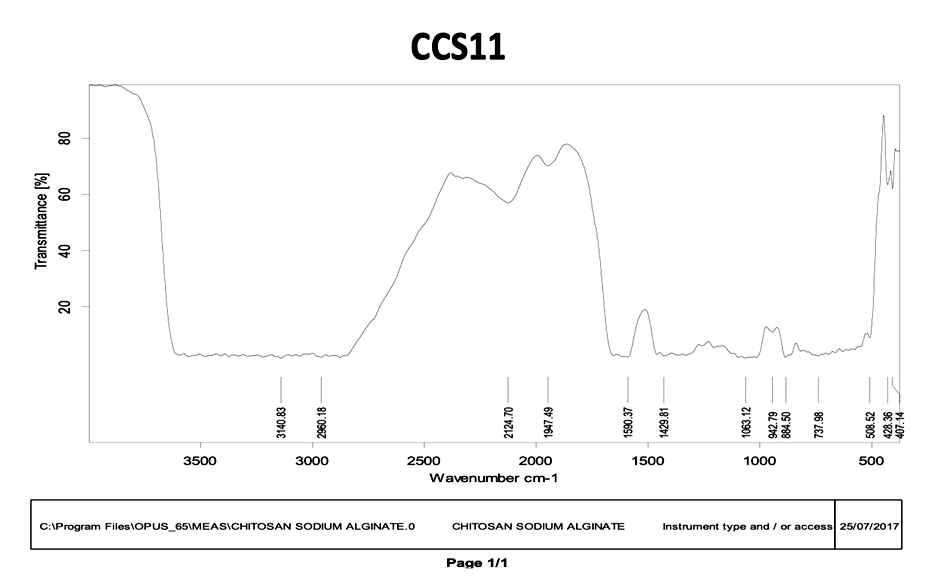
Fig. 1: FT-IR spectra of best selected chitosan composite scaffold (CCS-11) and gelatin composite scaffold (CGS-11)
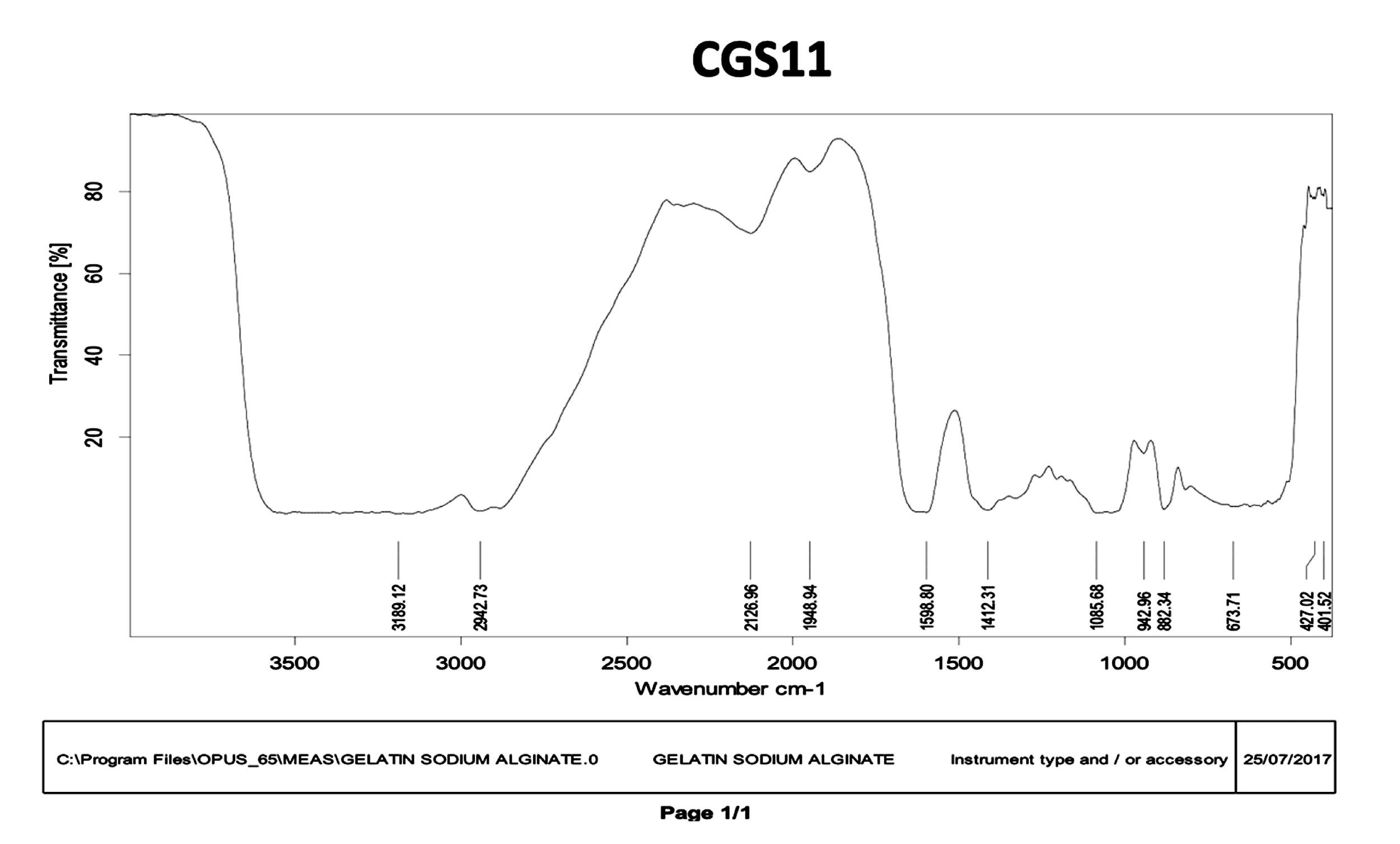
Fig. 2: FT-IR spectra of best selected gelatin composite scaffold (CGS-11)
 |
 |
| (a) | (b) |
Chitosan – Sodium Alginate (CCS11) |
|
 |
 |
| (a) | (b) |
Gelatin-Sodium alginate (CGS11) |
Fig. 3: SEM images of selected best scaffolds (CCS-11and CGS 11)
Table: 2 Results of different physico-mechanical parameters of CS scaffolds
| S. No. | Code of scaffold | Thickness (µm)(mean ±SD) |
Folding endurance (mean±SD) | Swelling behavior (%) (mean±SD) |
Tensile parameters | Texture parameters | ||
| Maximum force (N) | Max. elongation at break (sec) | Tensile strength (Mpa) | Burst strength (kg) | |||||
| 1 | CS1 | 54±1.57 | 179±3.78 | 5.05±0.28 | 0.97 | 0.7 | 0.98 | 0.14 |
| 2 | CS2 | 55.5±0.76 | 182±7.37 | 5.05±0.27 | 0.98 | 0.8 | 0.989 | 0.18 |
| 3 | CS3 | 59±3.51 | 205±3.13 | 8.5±0.47 | 0.98 | 0.82 | 0.98 | 0.18 |
| 4 | CS4 | 63±1.52 | 279±2.64 | 10.05±0.67 | 1.029 | 0.41 | 1.078 | 0.16 |
| 5 | CS5 | 69±5.29 | 291±1.52 | 12.49±0.72 | 0.98 | 1.2 | 0.999 | 0.23 |
| 6 | CS6 | 69.8±3.95 | 299±5.85 | 15.5±0.78 | 1.568 | 1.5 | 1.66 | 0.50 |
| 7 | PCS7 | 69.1±2.91 | 310±0.57 | 16.43±0.87 | 1.078 | 0.65 | 1.079 | 0.62 |
| 8 | PCS8 | 73±0.57 | 314±1.52 | 17.5±0.91 | 1.869 | 2.0 | 1.96 | 0.26 |
| 9 | PCS9 | 92±2.08 | 319±3.05 | 19.8±0.97 | 1.076 | 1.0 | 1.078 | 1.2 |
| 10 | CCS10 | 95.5±0.5 | 343±2.51 | 18.9±1.02 | 1.068 | 0.5 | 1.079 | 0.652 |
| 11 | CCS11 | 106±0.57 | 353±18.05 | 21.6±1.17 | 1.029 | 0.9 | 1.078 | 0.50 |
Value are expressed as mean±SD and sample size (n) =3
Biochemical studies
Estimation of Hydroxyproline and hexosamine contents: Hydroxyproline and hexosamine were estimated in granulated tissues of wounds of different groups of animals and results are shown in table 5. Hydroxyproline and hexosamine were found to have maximum content in group-I animals on the 11th d whereas group –II animals showed minimum hydroxyproline on the same day.
Histopathological studies
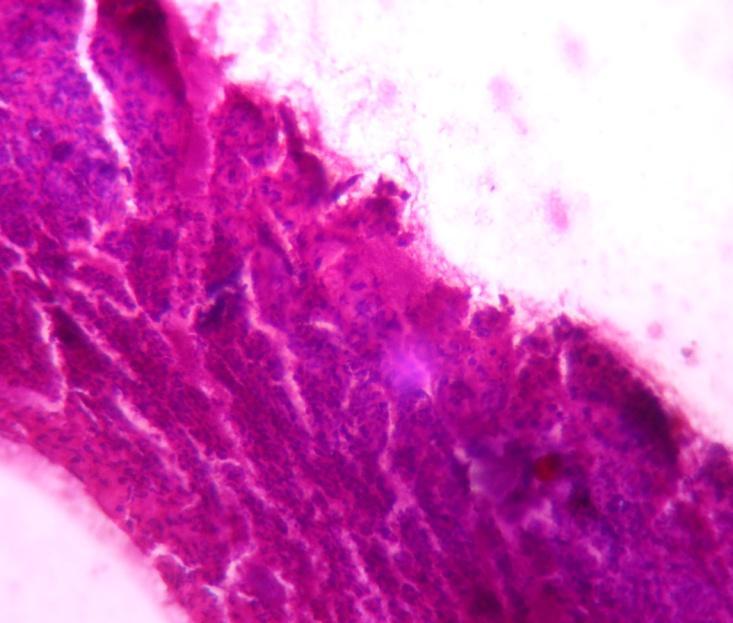
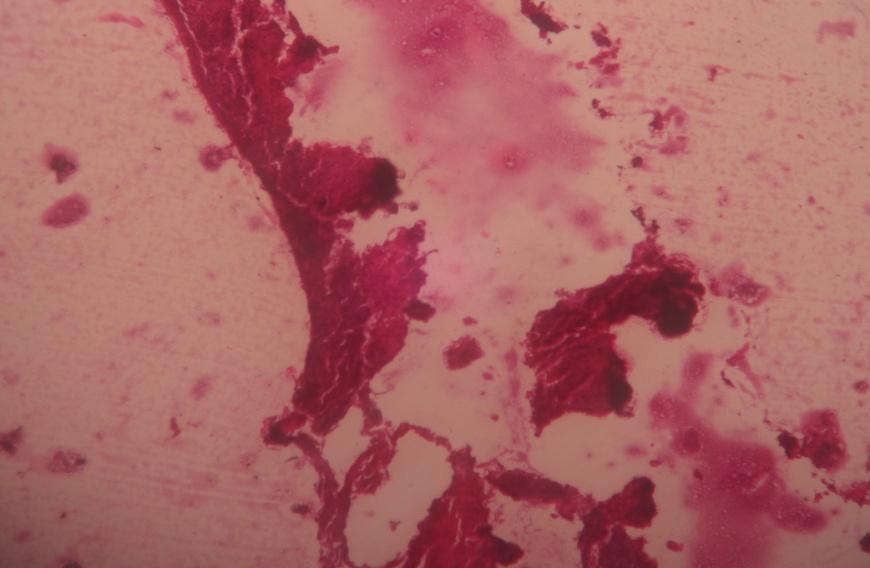
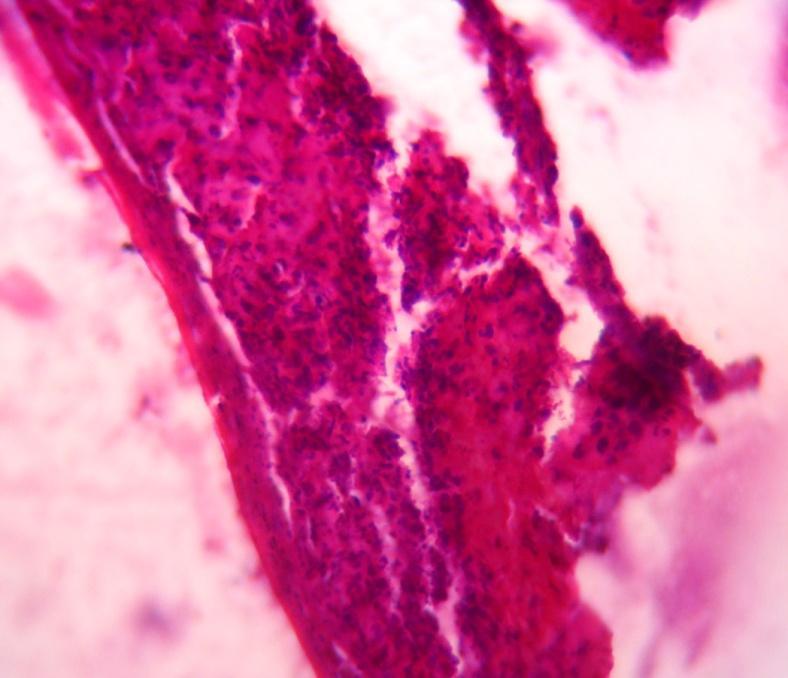
Photomicrographs representing histopathological characters were taken from samples collected from all three groups at 7th d and 14th d of treatment are shown in fig. 4.
Photography
The photographs showing wounds and healing of wounds at different time periods in various groups are shown in fig. 5.
Table 3: Results of different physico-mechanical parameters of GS scaffolds
| S. No. | Code of scaffold |
Thickness (µm) (mean ±SD) | Folding endurance (Mean±SD) | Swelling behaviour (%) (Mean±SD) |
Tensile parameters | Texture parameters | ||
| Maximum force (N) | Max. elongation at break (sec) | Tensile strength (Mpa) | Burst strength (kg) | |||||
| 1 | GS1 | 40±0.57 | 101±1.15 | 3.23±0.15 | 0.637 | 1.32 | 0.641 | 0.055 |
| 2 | GS2 | 44±1.73 | 159±0.57 | 3.69±0.19 | 1.176 | 2.5 | 1.225 | 0.60 |
| 3 | GS3 | 45±0.57 | 189±3.60 | 4.11±0.22 | 0.98 | 0.7 | 1.078 | 0.59 |
| 4 | GS4 | 49±1.52 | 236±1.52 | 4.00±0.61 | 1.225 | 2.5 | 1.264 | 0.30 |
| 5 | GS5 | 54±1.52 | 259±2.08 | 9.6±0.78 | 1.274 | 2.51 | 1.293 | 0.22 |
| 6 | GS6 | 61±1.52 | 264±2.64 | 11.9±0.79 | 1.078 | 1.1 | 1.079 | 0.50 |
| 7 | PGS7 | 65±2.08 | 266±4.16 | 13.0±0.81 | 0.98 | 1.1 | 0.99 | 0.65 |
| 8 | PGS8 | 86±2.08 | 290±1.52 | 18.10±0.92 | 0.98 | 1.4 | 0.99 | 1.1 |
| 9 | PGS9 | 98±3.60 | 314±3.05 | 19.93±0.96 | 1.22 | 1.5 | 1.263 | 1.9 |
| 10 | CGS10 | 101±1.54 | 329±5.05 | 20.94±1.08 | 0.98 | 1.5 | 1.078 | 0.72 |
| 11 | CGS11 | 104±3.21 | 363±0.57 | 22.09±1.12 | 0.989 | 1.1 | 1.079 | 0.8 |
Value are expressed as mean±SD and sample size (n) =3
Table 4: Percentage of wound contraction and biochemical contents at different time intervals in different groups
| Groups | (%) Wound contraction (mean±SD) | Hydroxyproline mg/gm (mean±SD) | Hexosamine mg/gm mean±SD) | |||||
| Day 7 | Day 14 | Day 21 | Day 28 | Day 0 | Day 11 | Day 0 | Day 11 | |
| Group I (untreated) | 15.6±2.35 | 37.5±6.29 | 79.5±7.36 | 94.5±5.48 | 0.950±0.03 | 2.484±0.05 | 1.479±0.004 | 1.478±0.001 |
| Group II (treated with CCS11) | 84.5±8.40* | 95.75±3.64* | 100* | 100* | 1.678±0.007* | 3.676±0.002* | 1.064±0.002 | 1.948±0.20 |
| Group III (treated with CGS11) | 70.35±5.88* | 99.95±4.97* | 100* | 100* | 1.921±0.016* | 4.369±0.59* | 0.973±0.03 | 2.045±0.001 |
Value are expressed as mean±SD and sample size (n)=3. *Significantly different from Group I at p<0.05.
| Group-I | |
| Day-7th | Day-14th |
 |
 |
| Group-II | |
| Day-7th | Day-14th |
 |
 |
| Group-III | |
| Day-7th | Day-14th |
 |
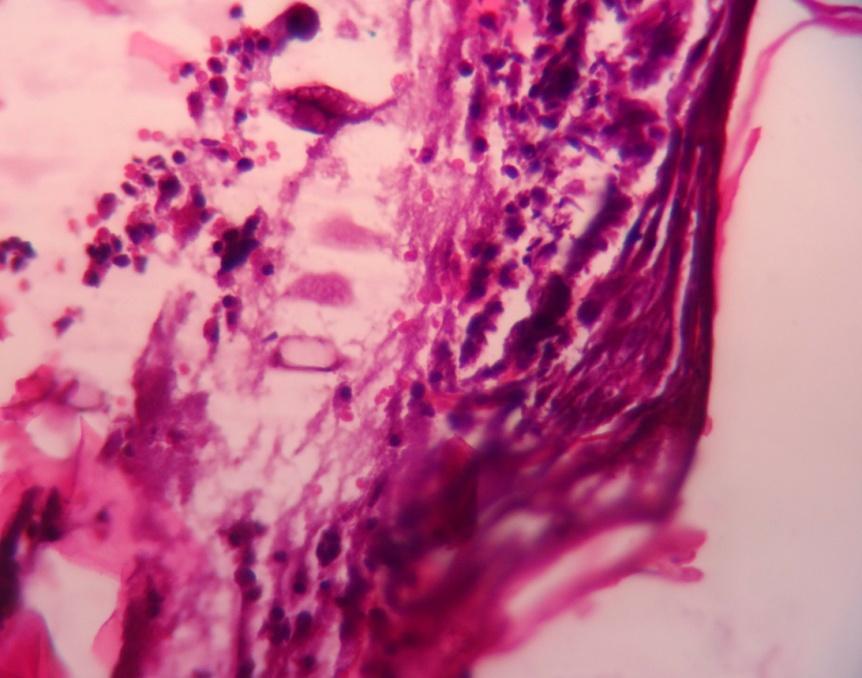 |
Fig. 4: Photomicrographs of wounds in group I, group II and group III
| Group-1 | |
| Day-0 | Day – 7 |
 |
 |
| Day – 14 | Day – 21 |
 |
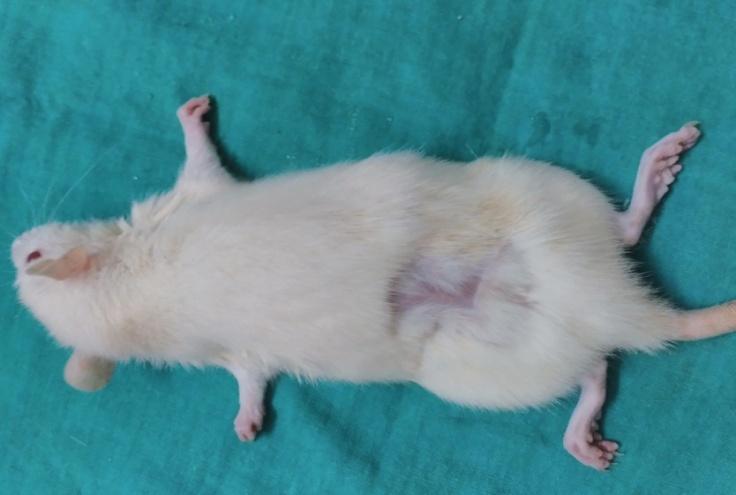 |
| GROUP-II | |
| Day-0 | Day – 7 |
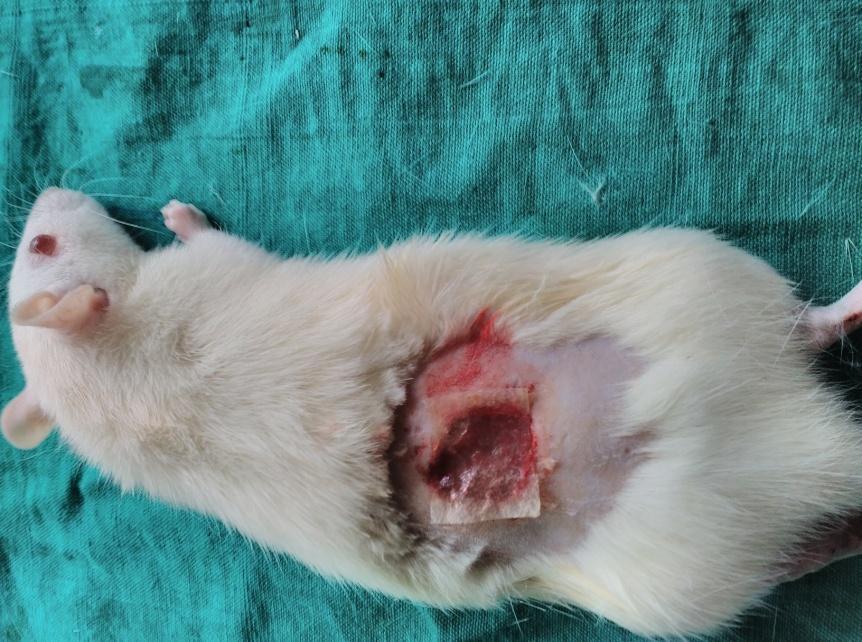 |
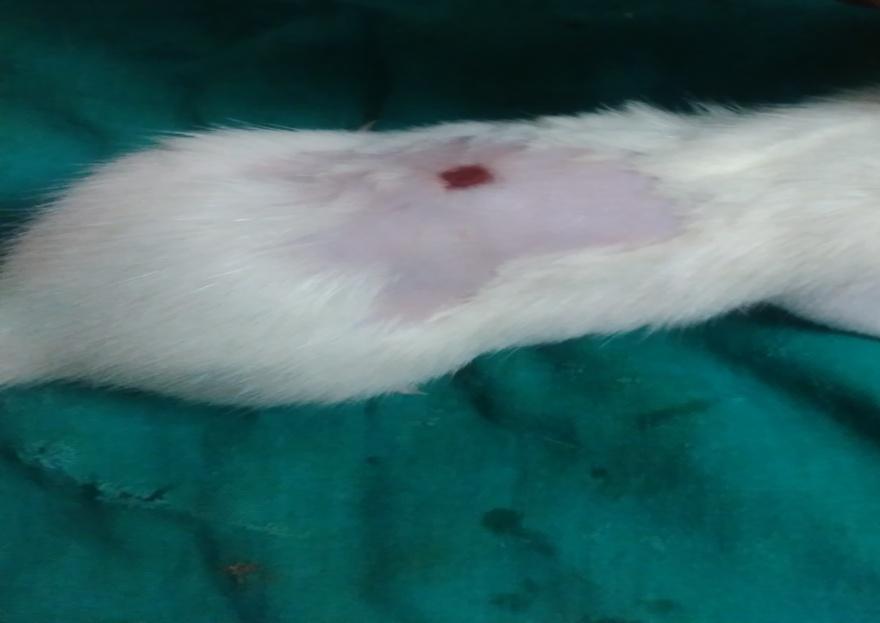 |
| Day – 14 | Day – 21 |
 |
 |
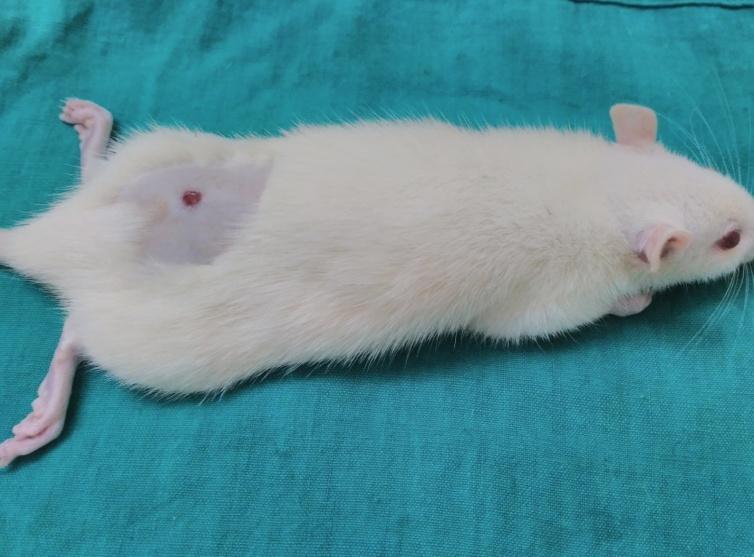
| GROUP-III | |
| Day-0 | Day – 7 |
 |
 |
| 14th Day | |
 |
Fig. 5: Photographs of wounds in Group I, Group II and Group III
Table 6: ANOVA results of chitosan composite scaffolds
| Parameter | F value | p-value |
| CS1-CS6 | ||
| Thickness | 13.26 | 0.000154 |
| Folding endurance | 469.81 | 2.51×10⁻¹³ |
| Swelling behavior | 158.62 | 1.58×10⁻¹⁰ |
| Tensile parameters | 40.97 | 3.92×10⁻⁷ |
| Texture parameters | 3.46 | 0.0363 |
| PCS7-PCS-9 | ||
| Thickness | 102.97 | 2.27×10⁻⁵ |
| Folding endurance | 15.33 | 0.00438 |
| Swelling behavior | 10.57 | 0.0108 |
| Tensile parameters | 45.71 | 0.000234 |
| Texture parameters | 28.67 | 0.00085 |
Table 7: ANOVA results of gelatine composite scaffolds
| Parameter | F value | p-value |
| GS1-GS6 | ||
| Thickness | 104.26 | 1.2×10⁻¹⁰ |
| Folding endurance | 2611.32 | 1.11×10⁻¹⁶ |
| Swelling behavior | 143.66 | 2.82×10⁻¹⁰ |
| Tensile parameters | 13,108.92 | 1.11×10⁻¹⁶ |
| Texture parameters | 143.66 | 2.82×10⁻¹⁰ |
| PGS7-PGS-9 | ||
| Thickness | 114.15 | 1.68×10⁻⁵ |
| Folding endurance | 110.37 | 1.85×10⁻⁵ |
| Swelling behavior | 47.88 | 0.000205 |
| Tensile parameters | 1.03 | 0.414 |
| Texture parameters | 29.14 | 0.000813 |
Statistical analysis
The statistical significance of the different parameters of both gelatin and chitosan composite scaffolds was tested using one way ANOVA and obtained F values and p values were noted in table 6 and 7
DISCUSSION
Chitosan, Gelatin and sodium alginate have proven wound healing properties when used alone. FTIR and X-ray analysis showed good compatibility between these polymers and hence the combinations were selected to study wound healing activity in the form of composite scaffolds [20, 21].
The effect of the concentration of plasticizer and cross-linking agent on the properties of these composite scaffolds was also studied. The thickness, folding endurance and swelling behaviour of different composite scaffolds were increased with increase in concentration of sodium alginate, indicating that the sodium alginate is increasing the flexibility and swelling property of scaffolds due to the hydrophilicity of sodium alginate [21].
Among six CS and GS scaffolds, 1:60 of chitosan-sodium alginate (CS6) and gelatin-sodium alginate (GS6) composite scaffolds were selected as the best to study the role of plasticizer in composite scaffolds, as these have the maximum tensile strength, swelling behavior and folding endurance as shown in table 2 and 3.
The thickness, folding endurance, and % swelling behaviour were significantly increased with an increase in plasticizer concentration in all composite scaffolds (table 6 and 7). There was no significant difference found in tensile parameters with increases in the concentration of plasticizer in all composite scaffolds (PCS, PGS), which may indicate the absence of the role of plasticizer in building tensile strength of scaffolds. (table 6 and 7) In contrast, the burst strength was significantly increased (p=0.05) with an increase in the concentration of the plasticizer which might be due to the contribution of elasticity by the plasticizer to scaffolds [22]. Then PCS9 and PGS9 were selected as the best scaffolds among the scaffolds that studied the effect of plasticizer due to their highest burst strength and burst time for further studies.
The effect of two concentrations of cross-linking agent on PCS9 (CCS10 and CCS11) and PGS9 (CGS10 and CGS11), was studied. The addition and increase in the concentration of cross-linking agent in scaffolds showed significantly (p=0.05 for CCS and p=0.1for CGS) increased thickness, and folding endurance in composite scaffolds and However, no significant difference (p=0.05) in swelling behaviour was found in all composite scaffolds, which indicated that the cross linking agent increased the flexibility of the film but not water absorption capacity. There was no discernible difference between the CS and GS scaffolds in terms of thickness and burst strength (p=0.05) [23, 24].
Then, based on all the above results CCS11 and CGS11 were selected as the best scaffold among all prepared composite scaffolds due to their highest folding endurance and swelling behavior, which are very important for application on skin and wound fluid absorption during wound treatment. Hence these were used for further studies of in vitro antibacterial studies, compatibility studies, surface morphology and in vivo studies.
The zone of inhibition of best-selected composite scaffolds against four different bacterial species found in anti-bacterial studies was different. The zone of inhibition of Bacillus subtilis and Pseudomonas aeruginosa was the highest with CGS11 and the zone of inhibition of Staphylococcus aureus and Escherichia coli was the highest with CCS11 scaffolds. However, from this, it can be suggested CCS11 exhibited higher inhibition against Staphylococcus aureus and E. coli, whereas CGS11 showed superior activity against P. aeruginosa and B. subtilis.
The variation in antibacterial activity of these scaffolds against four organisms may be due to differences in the nature of the cell membranes of organisms. It indicated that their antibacterial activity promotes wound healing and decreases the time required for healing.
The effect of CCS11 scaffolds may be due to the interaction of the positively charged amino group at the C-2 position of the glucosamine residue, which interacts with negatively charged microbial cell membranes, leading to the leakage of proteinnaceous and other intracellular constituents of the microorganisms [25].
Gelatin acts by surfactant-mediated hemostasis and offers a framework on wound for deposition of the cellular matrix to assist in healing [26]. It also proved that gelatin supports during the repetitive process when the gealtin sponge or foam is used as a hemostatic agent in experimental liver resections and injuries to large veins [27].
FT-IR spectrum of chitosan demonstrated the characteristic absorption peaks at 3363.81 cm-1 for O-H stretching, 1652.91 cm-1 for amide, 1678.10 cm-1 for N-H stretching,1072.74 cm-1for C=O stretching. FT-IR spectrum of gelatin demonstrated the characteristic absorption peaks at 1279.11 cm-1 for C=O stretching, 3260.08 cm-1 for N-H stretching and 2929.19 cm-1 for C-H stretching. FT-IR spectrum of sodium alginate demonstrated the characteristic absorption peaks at 1621.51 cm-1 for C=O stretching. The absorption peaks with selected best scaffolds. FT-IR spectrum of CCS11 demonstrated the characteristic absorption peaks at 3140.83 cm-1for O-H stretching, 1590.37 cm-1for C=O stretching (fig. 1). FT-IR spectrum of CGS11 demonstrated the characteristic absorption peaks at 3189.12 cm-1 for O-H stretching, 1598.80 cm-1for C=O stretching, almost similar to those obtained with the polymers (fig. 1 and 2). This confirmed the compatibility between polymers used in scaffolds.
The SEM images of CCS11 scaffolds (fig. 3) showed smooth, flimogenic continuous, compact and homogeneous, more uniform surfaces indicating complete miscibility of polymers. The SEM images of CGS11 scaffolds (fig. 3) showed more porosity and an interconnected porous structure with a rough surface which enhances compatible biological conditions for cell attachment, proliferation and differentiation [28]. This was proved by the fast-healing effect of CGS11 in in vivo studies. This porous structure is the reason for the increased swelling behaviour of scaffolds.
In vivo studies were conducted with the two selected best composite scaffolds (CCS11, CGS11), as per protocol (table 4). The percentage of wound contraction in all groups was measured on 7th, 14th, 21st and 28th post-wounding day to estimate the reduction in the wound area. There was a significant (p<0.05) difference in the percentage of wound contraction between the untreated group and treated groups (Group-II, III). 100% of wound contraction was observed in group III treated with a CGS11 within 14 days. It indicates that gelatin-sodium alginate scaffolds have shown more wound contraction than chitosan-sodium alginate scaffolds. Among the two composite scaffolds CGS11 i. e. gelatin-sodium alginate composite scaffold has shown the highest and faster contraction than chitosan-sodium alginate scaffolds.
Hydroxyproline is an amino acid exclusively found in connective tissue scleroproteins, collagen and elastin. Because of this restricted distribution, hydroxyproline has been widely used as an indicator of both the presence and metabolism of collagen. Hexosamine is a matrix molecule, which acts as a ground substance for the synthesis of a new extracellular matrix. It stimulates the synthesis of hyaluronic acid, which promotes extracellular matrix remodeling leading to better wound healing. As per table 4, the hydroxyproline and hexosamine values in the present study increased significantly from the day-1 and day-11 in all groups, which indicated an increased amount of collagen deposition and extracellular matrix with extension of time. Increased collagen content at the wound site is directly correlated with a number of fibroblasts being laid down and suggests early healing process. Both biochemical ingredients estimated were the highest in group III treated with CGS i. e., gelatin-sodium alginate composite scaffolds and were the lowest in group II treated with CCS i. e., chitosan-sodium alginate composite scaffolds among treated groups indicating that CGS promoted the generation of new extracellular matrix [29, 30].
In group II, wounds treated with chitosan-sodium alginate scaffolds the necrosis of dermis with more neutrophils on 7th day and the presence of cellular debris on the 14th day was observed. This suggested that gelatin-sodium alginate scaffolds may have more capacity for fast recovery and rapid epithelialization of skin than in the untreated and wounds treated with CS scaffolds. It might be due to its antibacterial action, which prevents further infections on the wound supporting for fast epithelialization by stimulation of wound healing environment. Chitosan-sodium alginate combination has not shown any difference in histopathological properties of wound when compared to untreated wounds and wounds treated with gelatin-sodium alginate composite scaffolds.
Based on the photography, the wound size was decreased in all groups on the 7th day, 14th and 21st day when compared to day zero. On 7th and 14th day, the size of wound was decreased in group treated with gelatin-sodium alginate composite scaffolds (CGS11) when compared to other groups on respective days which indicated that CGS11 composite scaffolds augmented the healing of wounds than wounds treated with chitosan-sodium alginate (CCS11) composite scaffolds.
CONCLUSION
The present study successfully fabricated and compared different composite scaffolds prepared using chitosan-sodium alginate and gelatin-sodium alginate for wound healing. Unlike previous CS/SA or GS/SA formulations, this study directly compares chitosan-and gelatin-based scaffolds under identical fabrication conditions to identify the optimal biopolymer combination for skin regeneration. From all the observations, it was found that gelatin-sodium alginate scaffolds showed more potential in wound healing than the chitosan-sodium alginate and this might be due to improved physico-mechanical properties of scaffolds. So, it can be concluded that altered physic mechanical properties of scaffolds can greatly influence the wound healing activity of scaffolds.
ACKNOWLEDGEMENT
Authors are grateful to, DBT-BUIDER, DST-CURIE funds to Sri Padmavati Mahila Visvavidyalayam, Tirupati and the authors gratefully acknowledge that the funding for this publication was provided by the Pradhan Mantri Uchchatar Shiksha Abhiyan (PM-USHA), under the Multi-Disciplinary Education and Research Universities (MERU) Grant sanctioned to Sri Padmavati Mahila Visvavidyalayam, Tirupati.
FUNDING
The authors received no financial support for the research but received funds for publication of work.
ETHICS STATEMENT
This study received authorisation from Institutional Animal ethics committee (IAEC (CPCSEA/1677/PO/Re/S/2012/IAEC/13)).
AUTHORS CONTRIBUTIONS
All authors contributed to conceptualization, experimentation, data analysis, and manuscript preparation. M. K. assisted in performed formulation work and drafting manuscript; R. K. performed formulation work and conducted in vivo studies; V. M. supervised and finalized the manuscript; S. K. R. V. assisted with histopathology interpretation.”
CONFLICTING OF INTERESTS
The authors declared no potential conflicts of interest with respect to the research.
REFERENCES
Stephane FF, Jules BK, Batiha GE, Ali I, Bruno LN. Extraction of bioactive compounds from medicinal plants and herbs. Nat Med Plants. 2022;147. doi: 10.5772/intechopen.98602.
Habanjar O, Diab Assaf M, Caldefie Chezet F, Delort L. 3D cell culture systems: tumor application advantages and disadvantages. Int J Mol Sci. 2021;22(22):12200. doi: 10.3390/ijms222212200, PMID 34830082.
Serrano Aroca A, Cano Vicent A, Sabater I Serra RS, El-Tanani M, Aljabali A, Tambuwala MM. Scaffolds in the microbial resistant era: fabrication materials properties and tissue engineering applications. Mater Today Bio. 2022;16:100412. doi: 10.1016/j.mtbio.2022.100412, PMID 36097597.
Li Z, Lin Z. Recent advances in polysaccharide-based hydrogels for synthesis and applications. Aggregate. 2021;2(2):e21. doi: 10.1002/agt2.21.
Abbasi AR, Sohail M, Minhas MU, Khaliq T, Kousar M, Khan S. Bioinspired sodium alginate-based thermosensitive hydrogel membranes for accelerated wound healing. Int J Biol Macromol. 2020;155:751-65. doi: 10.1016/j.ijbiomac.2020.03.248, PMID 32246960.
Varaprasad K, Jayaramudu T, Kanikireddy V, Toro C, Sadiku ER. Alginate-based composite materials for wound dressing application: a mini review. Carbohydr Polym. 2020;236:116025. doi: 10.1016/j.carbpol.2020.116025, PMID 32172843.
Badekar R, Bodke V, Tekade BW, Phalak SD. An overview on oral thin films: methodology characterization and current approach. Int J Pharm Pharm Sci. 2024;16(4):1-10. doi: 10.22159/ijpps.2024v16i4.50386.
Singh BN, Veeresh V, Mallick SP, Jain Y, Sinha S, Rastogi A. Design and evaluation of chitosan/chondroitin sulfate/nano-bioglass based composite scaffold for bone tissue engineering. Int J Biol Macromol. 2019;133:817-30. doi: 10.1016/j.ijbiomac.2019.04.107, PMID 31002908.
Eslami Z, Elkoun S, Robert M, Adjalle K. A review of the effect of plasticizers on the physical and mechanical properties of alginate-based films. Molecules. 2023;28(18):6637. doi: 10.3390/molecules28186637, PMID 37764413.
Wang L, Wang C, Wu S, Fan Y, Li X. Influence of the mechanical properties of biomaterials on degradability cell behaviors and signaling pathways: current progress and challenges. Biomater Sci. 2020;8(10):2714-33. doi: 10.1039/D0BM00269K, PMID 32307482.
Hamedi H, Moradi S, Hudson SM, Tonelli AE, King MW. Chitosan based bioadhesives for biomedical applications: a review. Carbohydr Polym. 2022;282:119100. doi: 10.1016/j.carbpol.2022.119100, PMID 35123739.
Li J, Zhuang S. Antibacterial activity of chitosan and its derivatives and their interaction mechanism with bacteria: current state and perspectives. Eur Polym J. 2020;138:109984. doi: 10.1016/j.eurpolymj.2020.109984.
Wathoni N, Shan CY, Shan WY, Rostinawati T, Indradi RB, Pratiwi R. Characterization and antioxidant activity of pectin from mangosteen rind. Heliyon. 2019;5(8):e02101. doi: 10.1016/j.heliyon.2019.e02299.
Derkach SR, Voron’ko NG, Sokolan NI, Kolotova DS, Kuchina YA. Interactions between gelatin and sodium alginate: UV and FTIR studies. J Dispers Sci Technol. 2020;41(5):690-8. doi: 10.1080/01932691.2019.1611437.
Lapomarda A, De Acutis A, De Maria C, Vozzi G. Pectin-based scaffolds for tissue engineering applications. In: Masuelli MA, editor. Pectins–the new-old polysaccharides. 2nd ed. London: IntechOpen; 2021. p. 146-216.
Nordin NN, Aziz NK, Naharudin I, Anuar NK. Effects of drug-free pectin hydrogel films on thermal burn wounds in streptozotocin-induced diabetic rats. Polymers (Basel). 2022;14(14):2873. doi: 10.3390/polym14142873, PMID 35890648.
Ibrahim N, Mohamed IN, Mohamed N, Mohd Ramli ES, Shuid AN. The effects of aqueous extract of Labisia Pumila (Blume) Fern Vill.-Var. Alata on wound contraction, hydroxyproline content and histological assessments in superficial partial thickness of second-degree burn model. Front Pharmacol. 2022;13:968664. doi: 10.3389/fphar.2022.968664, PMID 36313379.
Mojally M, Sharmin E, Alhindi Y, Obaid NA, Almaimani R, Althubiti M. Hydrogel films of methanolic Mentha piperita extract and silver nanoparticles enhance wound healing in rats with diabetes type I. J Taibah Univ Sci. 2022;16(1):308-16. doi: 10.1080/16583655.2022.2054607.
Keerthana M, Madeena D, Vidyavathi M, Kumar RS, Devasena Y. Development and characterization of curcumin-loaded chitosan–pectin composite bio-scaffolds. BPAS-Z. 2024;43B(2S):449–65.
Nagahama H, Maeda H, Kashiki T, Jayakumar R, Furuike T, Tamura H. Preparation and characterization of novel chitosan/gelatin membranes using chitosan hydrogel. Carbohydr Polym. 2009;76(2):255-60. doi: 10.1016/j.carbpol.2008.10.015.
Jimenez Gomez CP, Cecilia JA. Chitosan: a natural biopolymer with a wide and varied range of applications. Molecules. 2020;25(17):3981. doi: 10.3390/molecules25173981, PMID 32882899.
Diaz Montes E, Castro Munoz R. Trends in chitosan as a primary biopolymer for functional films and coatings manufacture for food and natural products. Polymers (Basel). 2021;13(5):767. doi: 10.3390/polym13050767, PMID 33804445.
Anal AK, Stevens WF, Remunan Lopez C. Ionotropic cross-linked chitosan microspheres for controlled release of ampicillin. Int J Pharm. 2006;312(1-2):166-73. doi: 10.1016/j.ijpharm.2006.01.043, PMID 16497452.
Archana D, Upadhyay L, Tewari RP, Dutta J, Huang YB, Dutta PK. Chitosan–pectin–alginate scaffold for tissue engineering. Int J Biotechnol. 2013;12(4):475-82.
Sharma UK. Disposable contact lens-based ocular delivery of moxifloxacin. Asian J Pharm Clin Res. 2021;14(11):105-11. doi: 10.22159/ajpcr.2021.v14i11.43090.
Foda NH, El-Laithy HM, Tadros MI. Implantable biodegradable sponges: effect of interpolymer complex formation of chitosan with gelatin on the release behavior of tramadol hydrochloride. Drug Dev Ind Pharm. 2007;33(1):7-17. doi: 10.1080/03639040600975188, PMID 17192246.
Jimenez Gomez CP, Cecilia JA. Chitosan: a natural biopolymer with a wide and varied range of applications. Molecules. 2020;25(17):3981. doi: 10.3390/molecules25173981, PMID 32882899.
Rotta J, Minatti E, Barreto PL. Determination of structural and mechanical properties, diffractometry and thermal analysis of chitosan and hydroxypropylmethylcellulose (HPMC) films plasticized with sorbitol. Cienc Tecnol Aliment. 2011;31(2):450-5. doi: 10.1590/S0101-20612011000200026.
Shahram E, Sadraie SH, Kaka G, Khoshmohabat H, Hosseinalipour M, Panahi F. Evaluation of chitosan-gelatin films for use as postoperative adhesion barrier in rat cecum model. Int J Surg. 2013;11(10):1097-102. doi: 10.1016/j.ijsu.2013.09.012, PMID 24090691.
Jahit IS, Nazmi I. Preparation and physical properties of gelatin/CMC/chitosan films. Int Food Res J. 2016;23(3):1068-74.