
Int J App Pharm, Vol 18, Issue 1, 2026, 617-624Original Article
ERTUGLIFLOZIN MITIGATES LIVER DAMAGE DURING SEPSIS: EVIDENCE FROM A MURINE MODEL
SALSABEEL H. ALRUFAIE1, ALI M. JANABI2*
1AL-Najaf AL-Ashraf Teaching Hospital, Al Najaf Al Ashraf Health Directorate, Najaf, Iraq. 2Department of Pharmacology and Toxicology, Faculty of Pharmacy, University of Kufa, Najaf, Iraq
*Corresponding author: Ali M. Janabi; *Email: alim.hashim@uokufa.edu.iq
Received: 19 Aug 2025, Revised and Accepted: 03 Nov 2025
ABSTRACT
Objective: This study sought to examine the liver-protective effects of ertugliflozin in male mice subjected to CLP-induced sepsis.
Methods: Overall, 24 male albino Swiss mice, aged eight to twelve weeks and weighing 25–35 g, were segregated into four separate groups (n=6 each): the Sham, CLP, CLP+DMSO, and CLP+ertugliflozin groups. In the sham group, the mice were anaesthetized and subjected to laparotomy without cecal ligation and puncture (CLP), whereas in the sepsis group, the mice underwent CLP. The CLP+DMSO group received DMSO (the vehicle for ertugliflozin) through intraperitoneal injection one hour before CLP. In the CLP+ertugliflozin group, 20 mg/kg was administered via the intraperitoneal method one hour before CLP. Biochemical indicators (GPT, GOT, IL-6, TNFα, MDA, GSH, Bax, pERK1/2, and caspase-3) and histological changes were assessed.
Results: ELISA was used to investigate the levels of the study parameters in hepatic tissues. Compared with those in the sham group, the levels of TNFα, IL6, Bax, caspase3, MDA and phospho-ERK1/2 were significantly elevated in the mice subjected to sepsis via caecal ligation and puncture (CLP), whereas the GSH level was significantly decreased. In contrast, treatment of septic mice with ertugliflozin significantly reduced these parameters in comparison with those of the sepsis group. Furthermore, histopathological examination revealed that the occurrence of sepsis via CLP resulted in significant liver damage compared with that in the sham group. Conversely, the treatment of mice subjected to CLP with ertugliflozin significantly mitigated liver injury.
Conclusion: Ertugliflozin, which inhibits SGLT2, has significant hepatoprotective effects on CLP-induced sepsis, mainly via its antioxidant, anti-inflammatory, and antiapoptotic effects.
Keywords: Ertugliflozin, Extracellular regulated kinases 1/2, Cecal ligation and puncture, Liver damage, Sepsis
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.56581 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Sepsis constitutes a worldwide health concern and is a primary cause of death, particularly in those with weakened immunity, such as the pediatric population, geriatric individuals, and immunocompromised patients, and those in intensive care units [1]. Sepsis is a life-threatening illness characterized by organ failure, mostly resulting from the host's immunological response to infection [2]. Among the affected organs, the liver is especially vulnerable and is often one of the first organs to sustain damage during sepsis [3]. Hepatic dysfunction in sepsis occurs in 34% to 46% of cases and is a significant predictor of death associated with sepsis. The mortality risk linked to sepsis-induced hepatic failure varies between 54% and 68% [4], which exceeds the mortality rate attributable to sepsis-induced pulmonary dysfunction [5].
The pathogenesis of sepsis involves the recognition of pathogen-associated proteins (PAMPs) and damage-associated molecular patterns (DAMPs), which bind to toll-like receptors (TLRs) on immune cells. Thus, sepsis is initiated by gene transcription, which results in inflammatory conditions, metabolism of cells, and adaptive immune responses [6]. Inflammation caused by increased levels of inflammatory mediators results in tissue destruction and ultimately multiple organ dysfunction. The liver is an essential component of the primary defensive system against infection. In the initial stages of sepsis, Kupffer cells are crucial for the elimination of germs and endotoxins because they release the proinflammatory meditators IL-6 and TNFα [7]. Although these cytokines facilitate the eradication of infections, they concurrently cause liver damage and provoke the secretion of more inflammatory cytokines in a phenomenon known as a storm of cytokines. This ultimately leads to a broad inflammatory reaction and damage to organs [4]. Additional mechanisms cause sepsis-induced liver injury, including bacterial toxins, tissue hypoperfusion [8], microvascular dysfunction [9], and reactive oxygen species (ROS) and proinflammatory generation, further exacerbating liver injury [10]. ROS can directly harm cells or tissues and can indirectly initiate a cascade of damage-associated signalling pathways [11]. It is hypothesized that ertugliflozin exerts hepatoprotective effects against sepsis-induced liver injury by modulating the ERK1/2 signalling pathway, thereby reducing inflammation, oxidative stress, and apoptosis and ultimately preserving hepatic structure and function.
The current study aimed to evaluate the hepatoprotective effects of ertugliflozin in a standardized CLP-induced sepsis model. This objective was addressed by assessing biochemical and histological markers of inflammation, oxidative stress, and apoptosis and by examining the modulation of ERK1/2 activation and translocation.
MATERIALS AND METHODS
Animal preparation
Overall, 24 adult albino Swiss mice, aged eight to twelve weeks and weighing 25–35 g, were obtained from the animal husbandry facility at the Faculty of Science at the University of Kufa. The animals were housed in the animal facility of the Faculty of Science at the University of Kufa. The animals were housed in a distinct room with a 12 h daylight and 12 h night cycle at a temperature of 24±2 °C and a relative humidity of 60–65% [12]. The mice were provided with a conventional diet of food and water. All experimental protocols received approval from the Institutional Animal Care and Use Committee (IACUC) at the University of Kufa following the submission of the required applications (2122,23\1\2025).
Study design
Randomized selection was employed to allocate 24 adult male Swiss albino mice into four groups (n = 6 per group): Sham, CLP, CLP+DMSO, and CLP+ertugliflozin. In the sham group, the mice were anaesthetized and subjected to a laparotomy but not to surgery. CLP: This group was used as a negative control. In contrast, animals in the CLP group underwent CLP; the cecum was accessed through a 1.5 cm midline abdominal incision and subsequently connected just below an ileocecal valve via a 5.0 suture, resulting in almost 75% of the cecal length being ligated, which led to high-grade sepsis [13]. A bilateral pair of caecal punctures was performed via a 22-determine needle [14]. The needle was subsequently delicately extracted, and the cecum was compressed. A minimal quantity of faces was expelled through the two apertures. The cecum was repositioned anatomically, the incision was sutured using 6.0 silk surgical thread, and 1cc of prewarmed (37 °C) normal saline was administered intraperitoneally immediately post-surgery to compensate for heat and fluid loss during the treatment [15]. The CLP+DMSO group received DMSO for ertugliflozin by i. p. injection for 1 h before CLP. In the CLP+ertugliflozin group, 20 mg/kg was injected intraperitoneally for 1 h prior to the CLP [16]. During the procedure, the mice received an injection of 100 mg/kg ketamine and 10 mg/kg xylazine into their peritoneum [17, 18].
Preparation of ertugliflozin
Ertugliflozin has a solubility of 100 mg/2 ml in DMSO and must be prepared immediately prior to usage. Dosage was delivered intraperitoneally on the basis of body weight.
Sampling techniques
Collection of blood samples
The mice were euthanized 24 h after CLP or sham surgery. A volume of blood (approximately 1 ml) was extracted from the heart via the intracardiac technique, and the samples were permitted to clot for between ten and twenty minutes at ambient temperature. The serum was subsequently extracted from the whole blood via centrifugation at 3000 rotations per minute for a duration of ten minutes [19, 20]. The sera were subsequently stored at-80 °C for the determination of serum GPT "glutamate pyruvate transaminase" and GOT "glutamate oxaloacetate transaminase" activities.
Biochemical analysis by tissue sampling
The harvested liver tissues were dissected, rinsed, and partitioned into two segments: the right portion was homogenized and preserved at-80 degrees Celsius until homogenization, utilizing a high-speed ultrasonic fluid processor in a one--to-ten W/V phosphate-buffered sodium chloride solution with 1% Triton X-100 and a protease inhibitor cocktail [21]. The homogenate was centrifuged for 10 min at 5000 rpm at 4 °C, and the resulting supernatants were subjected to various biochemical assays: MDA (malondialdehyde): Cat. No. SL0370Mo and Bax (Bcl2 associated × protein): Cat. No. SL0766Mo; a mouse ELISA kit from Sunlong Biotech Co., Hangzhou, China; and other methods [GSH (reduced glutathione): Cat. No. EA0123Mo; TNFα (tumor necrosis factor alpha): Cat. No. E0117Mo; IL6 (interleukin 6): Cat. No. E0049Mo; and Caspase3: Cat. No. E1513Mo]: a mouse ELISA kit from Bioassay Technology Laboratory, Hangzhou, China.
Tissue samples for immunohistochemistry were cut into 3-micrometer-thick sections on positively charged slides, deparaffinized and heated for 20 min. Then, the blocking agent peroxidase was added for 5 min, followed by washing buffer, and the primary antibody phosphoERK1/2 [Elabscience Bionovation Inc., USA, Cat. No. E-AB-20869] was added for thirty minutes, followed by washing buffer. Afterwards, the secondary antibody "mouse/rabbit poly detector plus DAB HRP brown" was added [BIO SB, USA, Cat. No. BSB 0269] for thirty minutes, followed by washing buffer. Subsequently, HRP (horse radish peroxidase) was added for 30 min, followed by washing buffer, followed by counterstaining with hematoxylin. Five samples were subsequently randomly selected from each group, and five points of view were randomly chosen from every selected sample for imaging via a light microscope (100× and 400×). The results represent the quick H score, which is produced by multiplying the positive cell proportion (P) by the cell intensity (I).
While tissue sampling for histopathology
After 24 h of tissue fixation in 10% formalin, tissue processing started with the use of an automated tissue processor for 20 h, which included the following steps: (A) dehydration in different alcohol concentrations from 50% to 100%. (B) Clearing in xylene. (C) Paraffin impregnation. (D) Paraffin embedding, followed by tissue sectioning via microtome and subsequent deparaffinization, tissue staining, and hematoxylin and eosin staining, including the following steps: (A) rehydration in different alcohol concentrations from 100% to 70% each for 2 min. (B) Distilled water was added for 1 min. (C) Hematoxylin for 1:30 min. (D) Rowing in tap water for 10 min. (E) Eosin staining for 1 mo. (F) Different alcohol concentrations from 70% to 100% each for 2 min. (H) Clearing in xylene followed by mounting.
The pathologists scrutinized the slides for liver damage via light microscopy at magnifications of X100 or X400. Histopathological alterations of the liver were classified according to the Batts Ludwig scoring system as necroinflammatory activity grade: Grade 0: no portal, periportal or lobular activity. Grade 1: minimal patchy or infrequent. Grade 2: mild (some or all portal tracts) with lobular activity (mildly little hepatocyte damage). Grade 3: moderate (all portal tracts), moderate lobular activity. Grade 4: severe (may have bridging fibrosis), steatosis activity grade and fibrosis grade [22].
Statistical analysis
Analyses were performed via "GraphPad Prism 8", California, USA. The data are displayed as the means±standard errors of the means. One-way analysis of variance (ANOVA) was employed, followed by the Benforonni test. Changes in histopathology were compared between groups via a combination of Fisher’s exact test and the Kruskal‒Wallis one-way analysis of variance with Dunn’s test for multiple comparisons. The statistically significant level was set at P<0.05 in all tests.
RESULTS
Sepsis was sustained for 24 h. One hour prior to CLP, the mice were administered DMSO (as a vehicle) or ertugliflozin (20 mg per kg) intraperitoneally [32] or left untreated (sham and CLP groups). A range of biochemical and histological indicators were examined to assess the degree of injury.
Effects on hepatic enzymes
Significant increases in the serum GOT and GPT were observed in the CLP group compared with those in the sham group. Compared with the CLP group, the CLP group treated with ertugliflozin presented significantly lower serum liver function marker (GOT, GPT) levels, whereas the CLP group treated with vehicle, DMSO, presented no significant changes in the serum liver enzyme levels (fig. 1).
Effect on tissue inflammation
Significant elevations in hepatic tissue IL-6 and TNFα levels were noted in the CLP group compared with those in the sham group. The CLP+ertugliflozin group presented significantly reduced levels of tissue inflammatory markers (IL6and TNFα). Compared with those in the CLP group, the liver IL6 and TNFα levels in the CLP+DMSO group did not significantly differ from those in the CLP group (fig. 2).
Effects on oxidative stress markers
Compared with those in the sham group, a significant increase in liver MDA and a significant reduction in liver GSH were observed in septic mice. Compared with those in septic mice, hepatic tissue levels of GSH were significantly increased, whereas MDA levels were significantly decreased in the CLP+ertugliflozin group. Compared with those in the CLP group, the levels of both MDA and GSH in the CLP group treated with vehicle or DMSO were not significantly different (fig. 3).
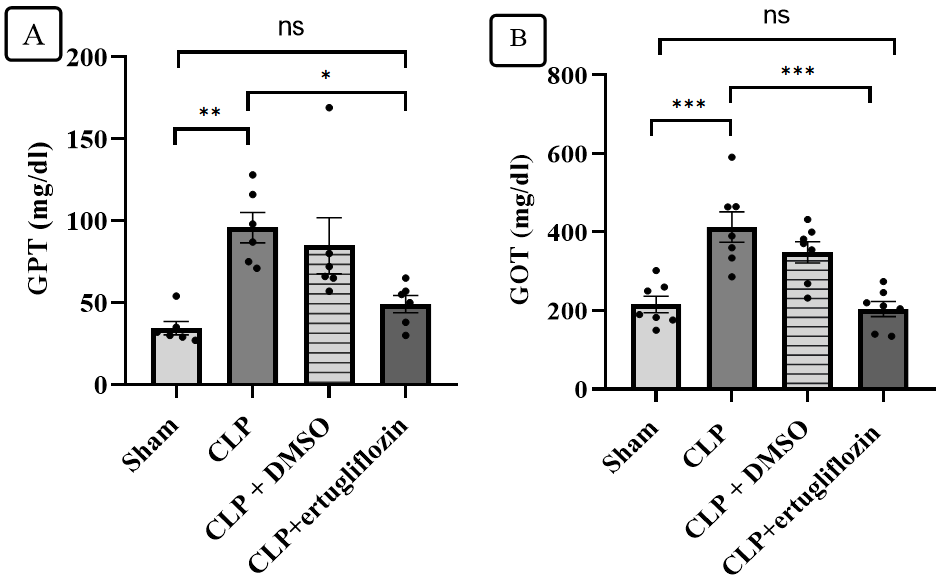
Fig. 1: Analysis of liver function indicators. A. GPT tier of study groups, B. GOT tier of study groups (6 animals in each group). Sepsis was induced in the mice via caecal ligation and puncture for 24 h. The mice were pretreated with vehicle (DMSO) or ertugliflozin (20 mg/kg) or left untreated (sham and CLP groups). Serum concentrations of GPT and GOT were assessed via a spectrophotometric technique. The results were examined via one-way ANOVA and Bonferroni's multiple comparison test for thorough interpretation. The results are shown as the mean±SEM, *P<0.05, **P<0.01, ***P<0.001
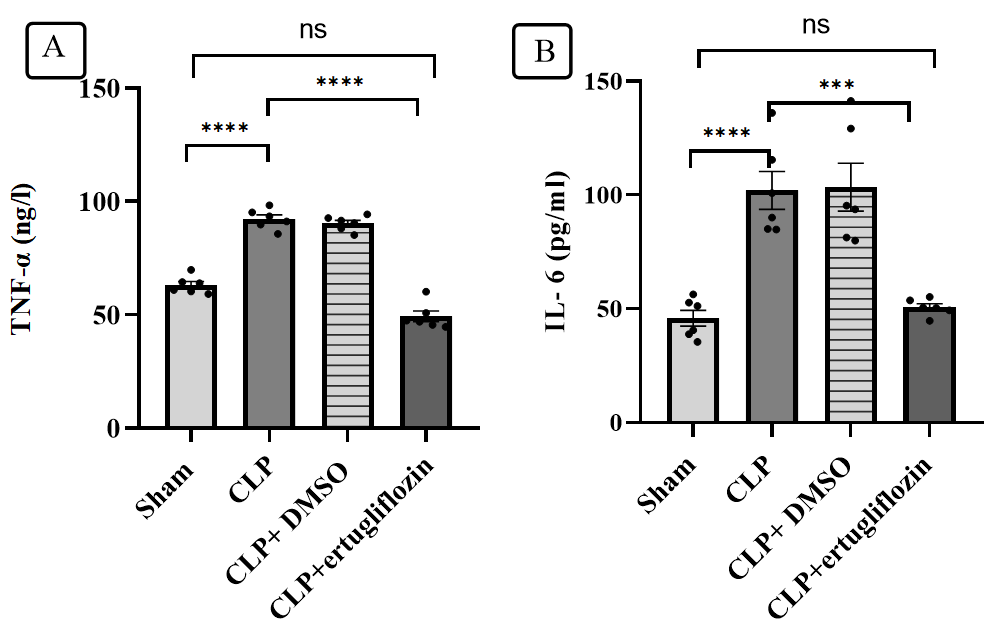
Fig. 2: Hepatic proinflammatory marker analysis. A. TNFα levels in the study groups. B. IL-6 levels in the study groups (6 animals in each group). The mice were subjected to sepsis by CLP for 24 hr. The mice were pretreated with vehicle (DMSO) or ertugliflozin (20 mg/kg) or left untreated (sham and CLP groups). TNF alpha and IL6 levels were determined via ELISA kits. The results were analysed via one-way ANOVA and Bonferroni's different comparisons test for multiple interpretation. The data are shown as the means±SEMs, ***P<0.001, ****P<0.0001
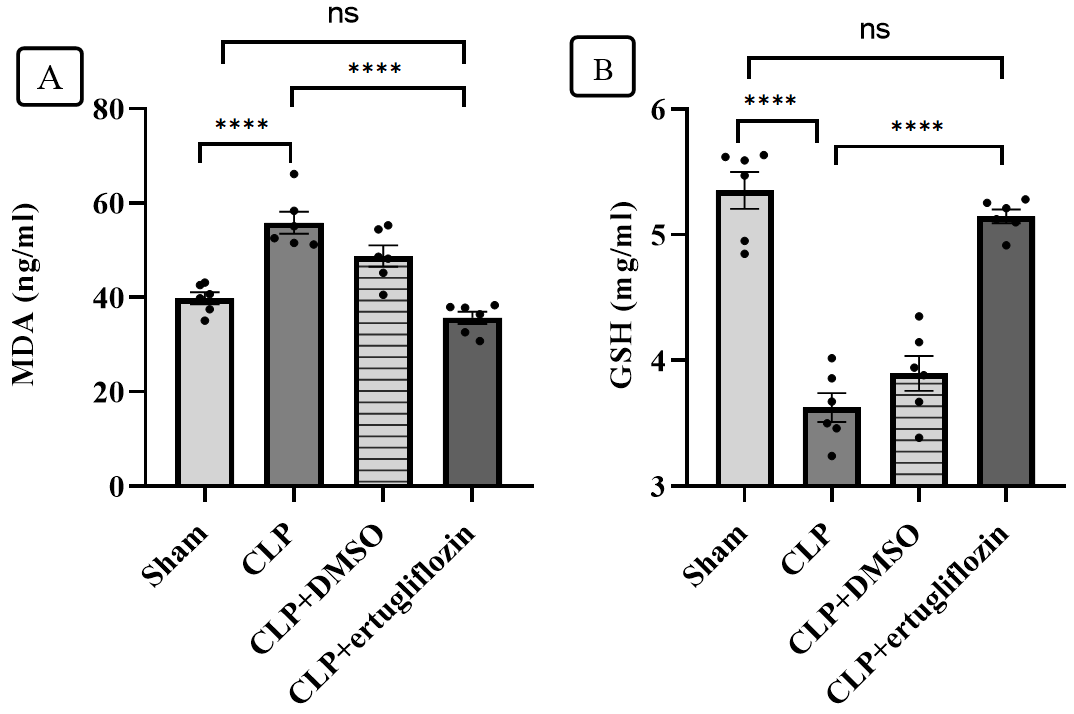
Fig. 3: Hepatic oxidative stress analysis. A. MDA levels in the study groups. B. GSH levels in the study groups (6 animals in each group). The mice were subjected to sepsis by CLP for 24 hr. The mice were pretreated with vehicle (DMSO) or ertugliflozin (20 mg/kg) for 1 hr. before CLP or left untreated (sham and CLP groups). The concentrations of the oxidative marker (MDA) and the antioxidant (GSH) were determined via ELISA kits. The data were analysed via one-way ANOVA and Bonferroni's different comparisons test for multiple interpretations. The data are shown as the means±SEMs, ****P<0.0001
Effects on the expression of apoptosis markers
Compared with those in the sham group, significant increases in caspase 3 and Bax levels were observed in the liver tissue of the CLP group. Compared with those in the CLP+ertugliflozin group, the levels of the apoptotic markers caspase-3 and Bax in the liver decreased significantly. Compared with those in the CLP group, the levels of both caspase 3 and Bax in the CLP group treated with vehicle or DMSO were not significantly different (fig. 4).
Effect on hepatic tissue damage
Histopathological examination was performed to evaluate the degree of hepatic injury 24 h after CLP. Compared with those in the sham group, the liver tissues in the CLP and CLP+DMSO groups were significantly damaged. Conversely, histopathological analysis of the liver in the CLP+ertugliflozin group revealed a slight degree of necroinflammation and steatosis (fig. 5, fig. 6). Fibrosis was not detected in any of the study groups.
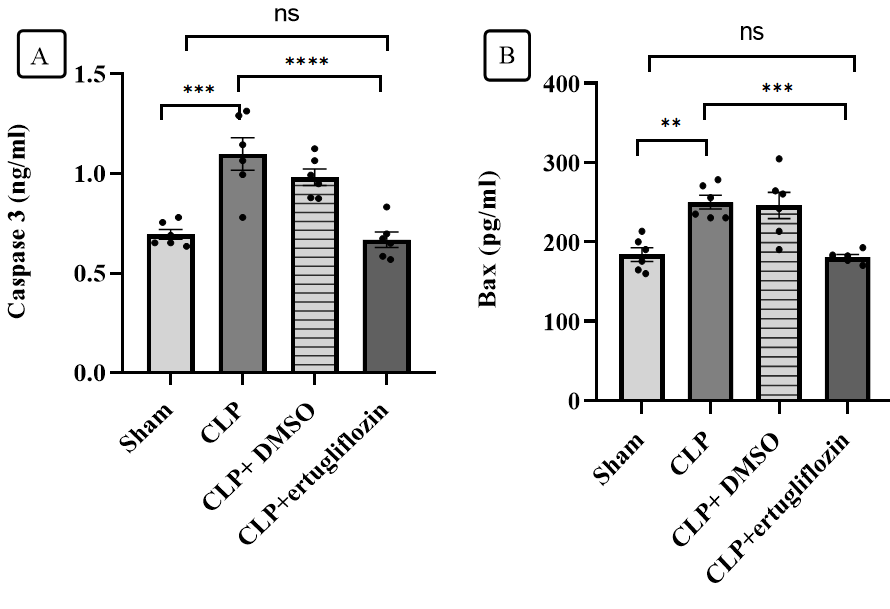
Fig. 4: Hepatic apoptosis marker analysis. A. Caspase 3 levels in the study groups. B. Bax levels in the study groups (6 animals in each group). The mice were subjected to sepsis by CLP for 24 h. The mice were pretreated with vehicle (DMSO) or ertugliflozin (20 mg/kg) for 1 hr. before CLP or left untreated (sham and CLP groups). The concentrations of proapoptotic markers (caspase 3) and Bax were determined via ELISA kits. The data were analysed via one-way ANOVA and Bonferroni's multiple comparisons test for multiple interpretation. The data are shown as the means±SEMs, **P<0.01, ***P<0.001, ****P<0.0001
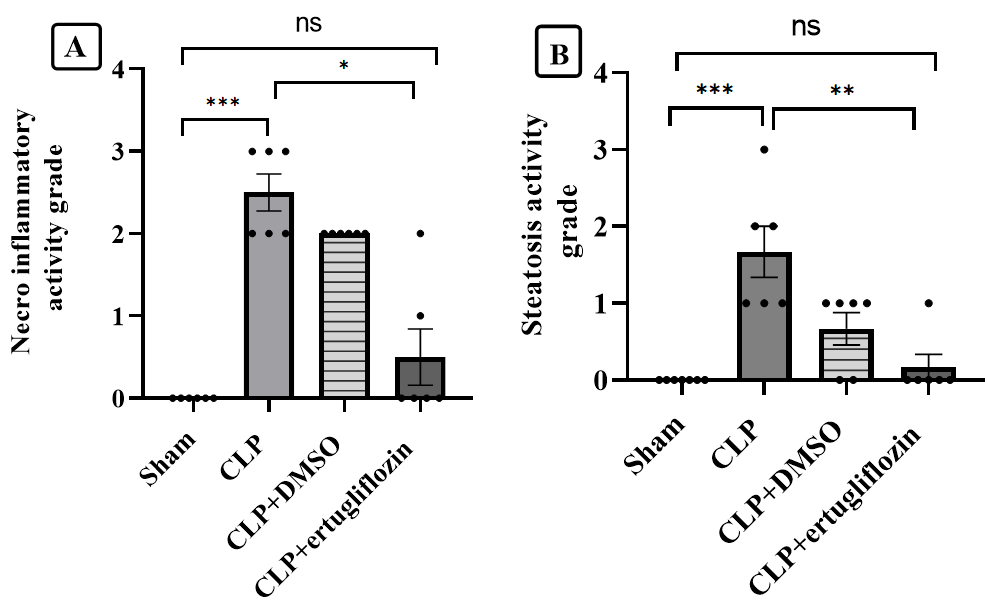
Fig. 5: Hepatic tissue damage analysis. A. Necroinflammatory activity grades of the study groups. B. Steatosis activity grades of the study groups (6 animals in each group). The mice were subjected to sepsis by CLP for 24 hr. The mice were pretreated with vehicle (DMSO) or ertugliflozin (20 mg/kg) for 1 hr. before CLP or left untreated (sham and CLP groups). The histopathologist assessed the grade of liver damage. The data were analysed via nonparametric methods, namely, the Kruskal‒Wallis test and Dunn’s test for multiple comparisons. The results are shown as the means±SEMs, *P<0.05, **P<0.01, ***P<0.001
Ertugliflozin mitigated sepsis-induced activation of ERK1/2 in hepatic tissue
Immunohistochemical analysis revealed a significant increase in phospho-ERK 1/2 in the hepatic tissue of the CLP and CLP+DMSO groups compared with that of the sham group. Conversely, ertugliflozin therapy significantly diminished the sepsis-induced increase in nuclear expression and activation of ERK1/2 (fig. 7 and fig. 8).
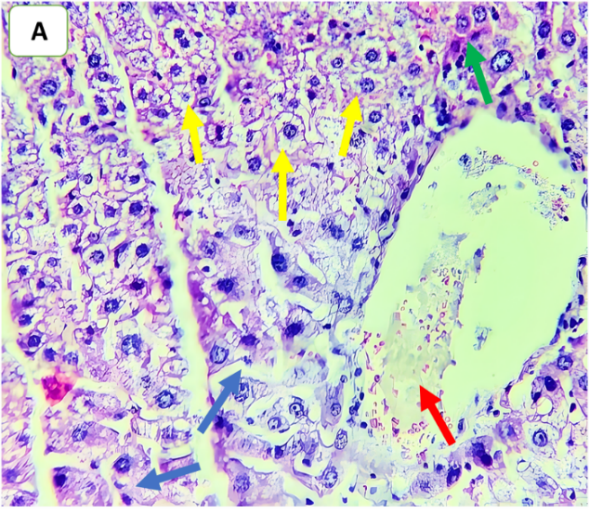
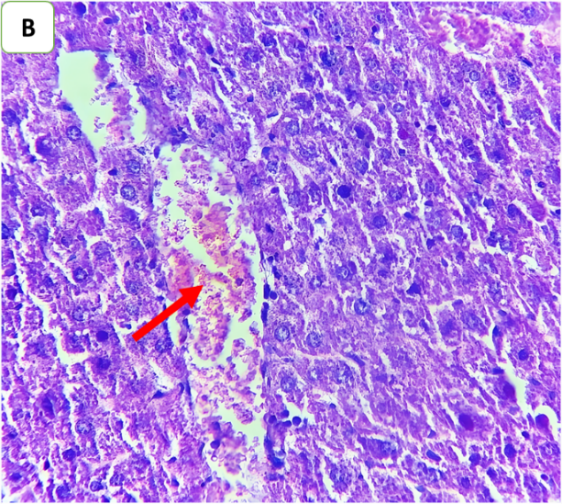
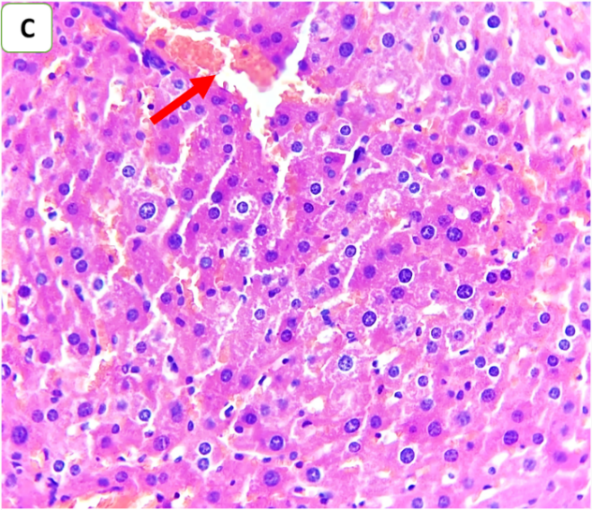
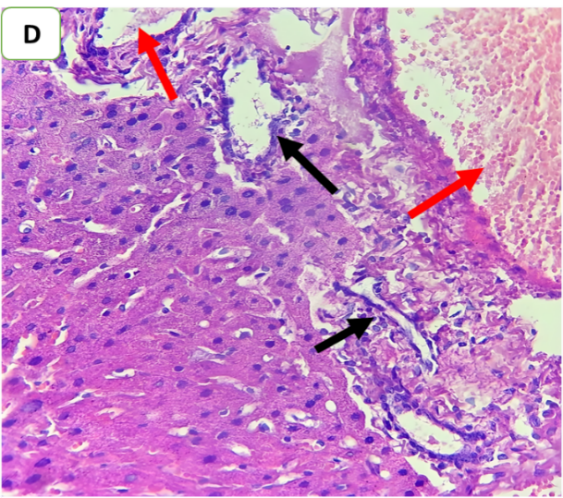
Fig. 6: Effects of ertugliflozin on histopathological findings (6 animals in each group): A. CLP group: animals underwent CLP-induced sepsis, with a necroinflammatory grade of 3, liver tissue with vascular congestion (red arrow), hepatocytes ballooning (blue arrows), apoptotic cells (green arrow) and steatosis (yellow arrows). B. Sham group: animals in this group were anaesthetized only and then laparotomized without the induction of sepsis by CLP, and the necroinflammatory grade was zero liver tissue with vascular congestion (red arrows). C. CLP+ertugliflozin group: animals were intraperitoneally injected with ertugliflozin (20 mg/kg) 1 h before CLP, the necroinflammatory grade was zero, and vascular congestion occurred (red arrow). D. CLP+DMSO group: animals were intraperitoneally injected with DMSO (vehicle for ertugliflozin) 1 h before CLP, the necroinflammatory grade was 2, and the liver tissue exhibited mild inflammation (black arrow). Vascular congestion (red arrows). A and BandCandD X 400, HandE staining
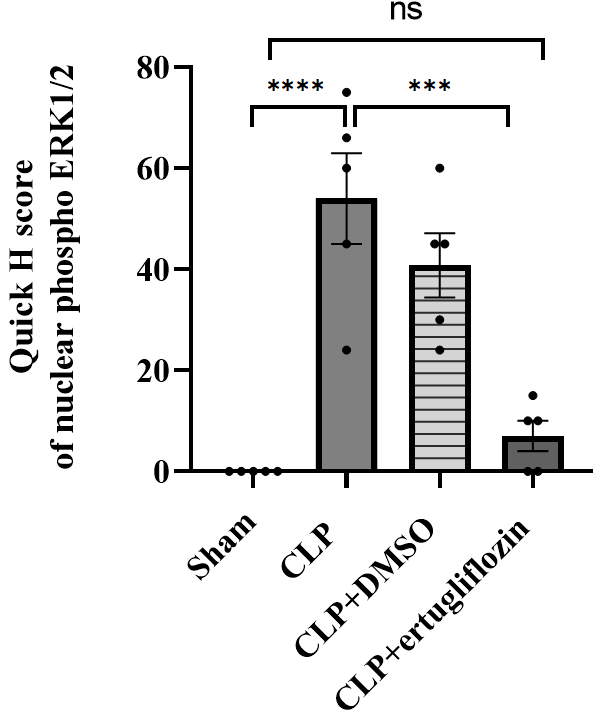
Fig. 7: Hepatic phospho-ERK1/2 analysis. The mice were subjected to sepsis by CLP for 24 hr (5 animals in each group). The mice were pretreated with vehicle (DMSO) or ertugliflozin (20 mg/kg) for 1 h before CLP or left untreated (sham and CLP groups). Liver pERK1/2 levels were determined by IHC. The data were analysed via one-way ANOVA and Bonferroni's different comparisons test. The results are shown as the mean±SEM, ***P<0.001, ****P<0.0001
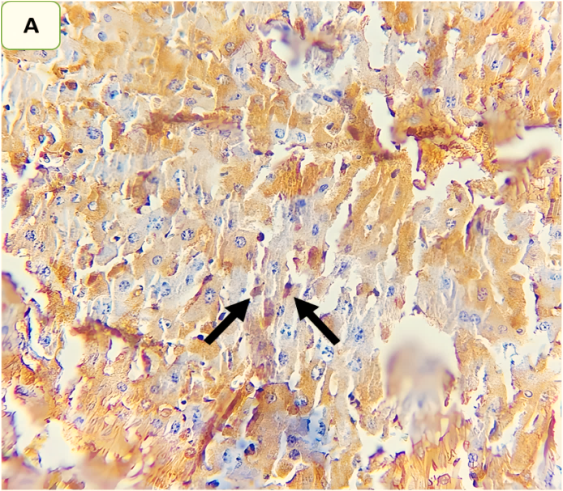
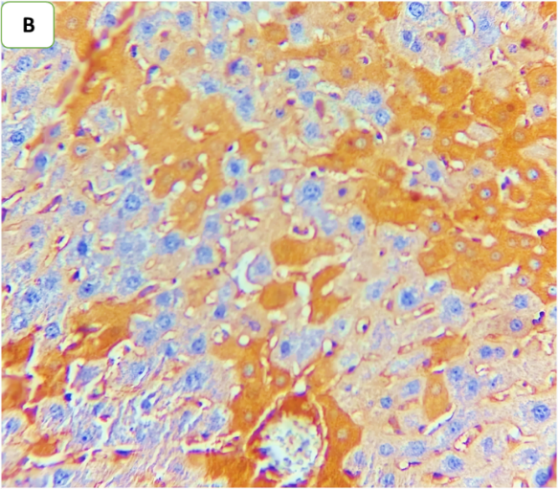
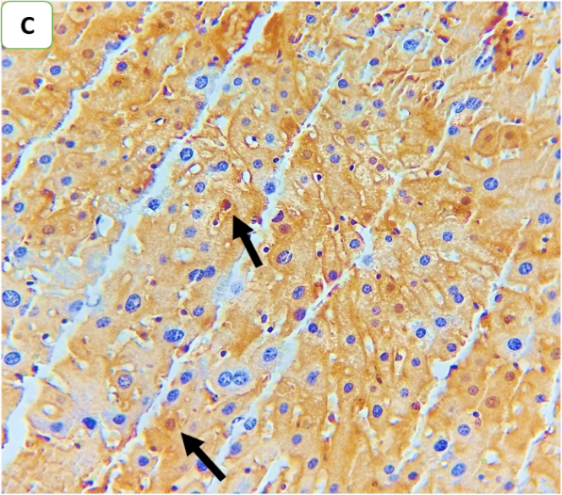
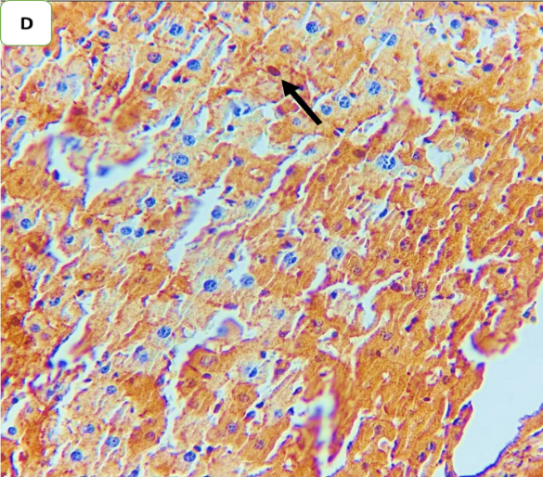
Fig. 8: Effect of ertugliflozin on phospho-ERK1/2 by immunohistochemistry (five animals in each group): A. CLP group: animals underwent CLP-induced sepsis; the mean quick H score of nuclear expression for this group was 54, with moderate-intensity nuclear expression in 20% of the examined tissue (black arrows). B. Sham group: animals in this group were anaesthetized only and then laparotomized without the induction of sepsis by CLP, and the mean quick H score of nuclear expression was zero. C. CLP+ertugliflozin group: animals were intraperitoneally injected with ertugliflozin (20 mg/kg) for 1 hr before CLP, the mean quick H score of nuclear expression for this group was 7, with liver tissue with 5% intense nuclear expression (black arrow). D. CLP+DMSO group: animals were intraperitoneally injected with DMSO (vehicle for ertugliflozin) for 1 hr before CLP, the mean quick H score of nuclear expression was 40.8, indicating that strong nuclear staining (black arrows) was present in the liver tissue. A, B, C, D X 400, IHC
DISCUSSION
Sepsis is a multifaceted inflammatory disease syndrome induced by a pathogenic bacterium and its byproducts. Prolonged treatment of sepsis may lead to severe sepsis and septic shock, potentially resulting in multiple organ dysfunction syndrome. Bacterial sepsis infiltrates the bloodstream and leads to the dissemination of different microbial byproducts. Host cells activate innate immune responses [23], which induce detrimental metabolic changes resulting in systemic inflammatory reactions, tissue dysfunction, and ultimately multiple organ failure [24]. The prevalence of sepsis-related conditions varies between 34% and 46% for individuals with liver dysfunction and between 1.3% and 22% for those with liver failure [5]. Factors leading to liver impairment during sepsis include uncontrolled systemic inflammation, hepatic ischemia, and dysregulated cell death [25]. Apoptotic cell death is crucial in the progression of organ damage during sepsis. Jeschke et al. revealed that insulin enhances hepatic integrity by reducing hepatic cell mortality and suppressing hepatic inflammatory responses in endotoxemic rats [26]. Furthermore, inhibiting apoptosis enhances the prognosis of animals suffering from severe sepsis [27]. On the basis of these results, we propose that ertugliflozin may improve liver dysfunction during sepsis by reducing hepatic apoptosis, inflammation, and oxidative stress.
In this study, there was a significant increase in the inflammatory markers TNFα and IL-6 in the sepsis group and vehicle group. These results are supported by other studies that reported that TNFα and IL-6 are associated with organ damage and mortality, suggesting that they are potentially valuable prognostic indicators of sepsis-induced organ impairment [28]. However, in the group treated with ertugliflozin, the levels of proinflammatory markers were significantly lower than those in the other groups, suggesting that ertugliflozin has an anti-inflammatory effect [29].
The inflammatory markers assessed in our study were notably decreased in the livers of the mice that were pretreated with ertugliflozin. These findings indicate that ertugliflozin possesses significant anti-inflammatory properties related to liver injury. To our knowledge, no published research has examined the impact of ertugliflozin on liver injury induced by sepsis, suggesting that our work is novel. Abd Uljaleel and colleagues demonstrated that "mice treated with ertugliflozin exhibited significantly decreased levels of inflammatory cytokines in the lungs" [16]. A separate study demonstrated that terzepatide (a dual agonist of GLP-1 and GIP) significantly reduced IL-6 levels during ischemia‒reperfusion injury in rat kidneys [18].
Compared with those in the endotoxemic group, significant decreases in the MDA levels were detected in the liver samples of the mice in the ertugliflozin pretreatment group, whereas the GSH level was significantly increased. Another study revealed that GSH levels are reduced in animals with renal ischaemic reperfusion injury [30, 31]. Inhibitors of SGLT2 have shown distinct cardiovascular advantages in diabetic kidney disease, perhaps due to their role in establishing a favourable balance among oxidant-generating systems and those offering antioxidant defences [32].
Sepsis causes hepatic apoptosis, resulting in organ damage and dysfunction [33]. Consequently, the inhibition of apoptosis mitigates the hepatocellular damage caused by sepsis [34]. The activation of caspase-3 is a critical step in executing programmed cell death. The balance between proapoptotic (Bax) and antiapoptotic (Bcl-2) proteins is essential for cell survival. Sepsis frequently disturbs this balance by increasing Bax expression and caspase3 levels, hence facilitating apoptosis. This study revealed a "notable elevation in caspase3 and Bax activity in liver tissue homogenates in the sepsis and DMSO groups compared with the sham group". This study revealed that pretreatment with ertugliflozin significantly decreased caspase3 activity and Bax expression in injured liver tissues. This study revealed significant histological changes in the liver 24 h after CLP-induced sepsis. The detected changes included portal inflammation, centrilobular inflammation, hepatocyte ballooning, apoptosis, and steatosis. The diminished pathological score noted in the CLP+ertugliflozin group was attributed to increased steatosis, reduced ballooning and apoptosis within hepatic tissue, and decreased infiltration of inflammatory cells, suggesting that this medication may have the capacity to help protect the liver from endotoxemia-induced liver injury. These data substantiate the notion that ertugliflozin can inhibit apoptosis during sepsis-induced liver damage. The anti-inflammatory, antiapoptotic, and antioxidant properties of ertugliflozin contribute to the demonstrated protection against hepatic damage generated by sepsis. This result is supported by other studies that revealed that ipragliflozin (another analogue of an SGLT2 inhibitor) can diminish the brain damage score in mice subjected to CLP-induced polymicrobial infection [35]. The proteins of the MAPK (mitogen-activated protein kinase) family, encompassing the p38 MAPK, ERK, and JNK subgroups, can be activated via phosphorylation in response to apoptotic and external stimuli. In our investigation, the phosphorylation levels of ERK1/2 were examined. The results indicated that the level of phosphorylated ERK1/2 in the sepsis group significantly increased and that ERK1/2 translocated to the nucleus following CLP, indicating a tendency toward elevated levels of phosphorylated ERK, which was correlated with apoptosis and inflammation. Following treatment with ertugliflozin, a reduction in p-ERK1/2 expression and translocation in hepatocytes was noted. These results suggested that ertugliflozin suppressed the phosphorylation of ERK1/2, which is involved in reducing apoptosis and inflammation through the inhibition of NF-KB and other transcription factors responsible for apoptosis and the production of proinflammatory cytokines. Numerous studies have shown that "p38 MAPK can be phosphorylated by the release of ROS-regulated Bax translocation" [36]. Moreover, hypoxia and subsequent reoxygenation of the liver may increase the levels of caspases and proapoptotic cofactors via ERK upregulation [37]. Suppression of the MAPK pathway is typically considered a protective measure against HIRI. Astaxanthin pretreatment safeguards against hepatocyte apoptosis and hepatic ischaemia‒reperfusion injury by blocking the ERK1/2 signalling pathway, suggesting a viable therapeutic approach [38]. In HIRI, necrosis induces liver damage, subsequently activating autophagy via ERK1/2 activation. Necrostatin-1 alleviates this damage by reducing the activity of ERK1/2 [39]. Finally, in the present study, ertugliflozin demonstrated hepatoprotective effects in sepsis, which were associated with the modulation of ERK1/2 phosphorylation. The role of ERK1/2 signalling in sepsis appears to be context dependent, as previous studies have reported both protective and detrimental outcomes depending on the extent and duration of activation. Our findings suggest that these agents may promote balanced regulation of ERK1/2 activity, restoring physiological signalling rather than inducing excessive activation. This interpretation may help reconcile conflicting reports regarding ERK1/2 function in sepsis. Moreover, accumulating evidence indicates that the protective actions of ertugliflozin extend beyond SGLT2 inhibition, involving additional mechanisms such as the suppression of oxidative stress and the inhibition of inflammation. These SGLT2-independent pathways mitigate sepsis-induced liver injury.
CONCLUSION
This is the first effort to utilize ertugliflozin for the management of sepsis-induced liver injury. Ertugliflozin mitigated injury by reducing ROS generation and inflammatory cytokine expression, as well as the activation and translocation of ERK1/2, while enhancing the expression of antioxidant indicators such as GSH and decreasing the expression of caspase3 and Bax. Pretreatment with ertugliflozin has a protective effect against liver injury caused by sepsis.
Ethical considerations
The research and protocol of this study were in accordance with the guidelines of animal studies and were approved by the Institutional Animal Care and Use Committee (IACUC) at the University of Kufafollowing the submission of the required applications (2122,23\1\2025).
Study highlights
Hepatic dysfunction in sepsis occurs in 34% to 46% of cases and is a significant predictor of death associated with sepsis. Pretreatment with ertugliflozin has a protective effect against liver injury caused by sepsis.
FUNDING
The authors received no financial support for the research, authorship, and/or publication of this article.
AUTHORS CONTRIBUTIONS
All the authors contributed equally to this article. Alrufaie, SH contributed to data collection, statistical analysis, and draft writing. Janabi, AM contributed to the main idea, design of the study and critical revision.
CONFLICT OF INTERESTS
The authors declare that they have no conflicts of interest.
REFERENCES
Fleischmann C, Scherag A, Adhikari NK, Hartog CS, Tsaganos T, Schlattmann P. Assessment of global incidence and mortality of hospital-treated sepsis: current estimates and limitations. Am J Respir Crit Care Med. 2016;193(3):259-72. doi: 10.1164/rccm.201504-0781OC, PMID 26414292.
Seymour CW, Liu VX, Iwashyna TJ, Brunkhorst FM, Rea TD, Scherag A. Assessment of clinical criteria for sepsis: for the third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA. 2016;315(8):762-74. doi: 10.1001/jama.2016.0288, PMID 26903335.
Saini K, Bolia R, Bhat NK. Incidence predictors and outcome of sepsis-associated liver injury in children: a prospective observational study. Eur J Pediatr. 2022;181(4):1699-707. doi: 10.1007/s00431-022-04374-2, PMID 35020050.
Woznica EA, Inglot M, Woznica RK, Lysenko L. Liver dysfunction in sepsis. Adv Clin Exp Med. 2018;27(4):547-51. doi: 10.17219/acem/68363, PMID 29558045.
Yan J, Li S, Li S. The role of the liver in sepsis. Int Rev Immunol. 2014;33(6):498-510. doi: 10.3109/08830185.2014.889129, PMID 24611785.
Rubio I, Osuchowski MF, Shankar Hari M, Skirecki T, Winkler MS, Lachmann G. Current gaps in sepsis immunology: new opportunities for translational research. Lancet Infect Dis. 2019;19(12):e422-36. doi: 10.1016/S1473-3099(19)30567-5, PMID 31630991.
Traeger T, Mikulcak M, Eipel C, Abshagen K, Diedrich S, Heidecke CD. Kupffer cell depletion reduces hepatic inflammation and apoptosis but decreases survival in abdominal sepsis. Eur J Gastroenterol Hepatol. 2010;22(9):1039-49. doi: 10.1097/MEG.0b013e32833847db, PMID 20300005.
Waseem N, Chen PH. Hypoxic hepatitis: a review and clinical update. J Clin Transl Hepatol. 2016;4(3):263-8. doi: 10.14218/JCTH.2016.00022, PMID 27777895.
La Mura V, Pasarin M, Meireles CZ, Miquel R, Rodriguez Vilarrupla A, Hide D. Effects of simvastatin administration on rodents with lipopolysaccharide-induced liver microvascular dysfunction. Hepatology. 2013;57(3):1172-81. doi: 10.1002/hep.26127, PMID 23184571.
Strnad P, Tacke F, Koch A, Trautwein C. Liver guardian modifier and target of sepsis. Nat Rev Gastroenterol Hepatol. 2017;14(1):55-66. doi: 10.1038/nrgastro.2016.168, PMID 27924081.
Jia Y, Li Z, Feng Y, Cui R, Dong Y, Zhang X. Methane-rich saline ameliorates sepsis induced acute kidney injury through anti-inflammation, antioxidative and antiapoptosis effects by regulating endoplasmic reticulum stress. Oxid Med Cell Longev. 2018;2018:4756846. doi: 10.1155/2018/4756846, PMID 30581532.
Qassam H, Janabi AM, Gaen KK, Hadi NR. Dimethyl fumarate attenuates liver injury in a mouse model of cecal ligation and puncture by modulating inflammatory angiogenic and pyroptotic pathways. BMC Pharmacol Toxicol. 2025;26(1):134. doi: 10.1186/s40360-025-00968-2, PMID 40676712.
Jeong SJ, Han SH, Kim CO, Choi JY, Kim JM. Anti-vascular endothelial growth factor antibody attenuates inflammation and decreases mortality in an experimental model of severe sepsis. Crit Care. 2013;17(3):R97. doi: 10.1186/cc12742, PMID 23710641.
Ibrahim YF, Fadl RR, Ibrahim S, Gayyed MF, Bayoumi A, Refaie M. Protective effect of febuxostat in sepsis-induced liver and kidney injuries after cecal ligation and puncture with the impact of xanthine oxidase interleukin 1β, and c-Jun N-terminal kinases. Hum Exp Toxicol. 2020;39(7):906-19. doi: 10.1177/0960327120905957, PMID 32054342.
Siempos II, Lam HC, Ding Y, Choi ME, Choi AM, Ryter SW. Cecal ligation and puncture-induced sepsis as a model to study autophagy in mice. J Vis Exp. 2014;(84):e51066. doi: 10.3791/51066, PMID 24561344.
Abd Uljaleel A, Hassan E. Protective effect of ertugliflozin against acute lung injury caused by endotoxemia model in mice. Iran J War Public Health. 2023;15(1):67-75. doi: 10.58209/ijwph.15.1.67.
Alaasam ER, Janabi AM, Al Buthabhak KM, Almudhafar RH, Hadi NR, Alexiou A. Nephroprotective role of resveratrol in renal ischemia reperfusion injury: a preclinical study in sprague-dawley rats. BMC Pharmacol Toxicol. 2024;25(1):82. doi: 10.1186/s40360-024-00809-8, PMID 39468702.
Alkhafaji GA, Janabi AM. GIP/GLP-1 dual agonist tirzepatide ameliorates renal ischemia/reperfusion damage in rats. Int J App Pharm. 2025;17(2):165-73. doi: 10.22159/ijap.2025v17i2.53156.
Q Jallawee H, Janabi AM. Trandolapril improves renal ischemia reperfusion injury in adult male rats via activation of the autophagy pathway and inhibition of inflammation oxidative stress and apoptosis. J Biosci Appl Res. 2024;10(6):114-27. doi: 10.21608/jbaar.2024.315239.1077.
Alkhafaji GA, Janabi AM. Protective effects of bexagliflozin on renal function in a rat model of ischemia-reperfusion injury; an experimental animal study. J Nephropharmacol. 2025;14(2):e12760. doi: 10.34172/npj.2025.12760.
Tiba AT, Qassam H, Hadi NR. Semaglutide in renal ischemia-reperfusion injury in mice. J Med Life. 2023;16(2):317-24. doi: 10.25122/jml-2022-0291, PMID 36937464.
Chowdhury AB, Mehta KJ. Liver biopsy for assessment of chronic liver diseases: a synopsis. Clin Exp Med. 2023;23(2):273-85. doi: 10.1007/s10238-022-00799-z, PMID 35192111.
Esmon CT, Fukudome K, Mather T, Bode W, Regan LM, Stearns Kurosawa DJ. Inflammation sepsis and coagulation. Haematologica. 1999;84(3):254-9. PMID 10189392.
Jacobi J. Pathophysiology of sepsis. Am J Health Syst Pharm. 2002;59(Suppl 1):S3-8. doi: 10.1093/ajhp/59.suppl_1.S3, PMID 11885412.
Kobashi H, Toshimori J, Yamamoto K. Sepsis-associated liver injury: incidence, classification and the clinical significance. Hepatol Res. 2013;43(3):255-66. doi: 10.1111/j.1872-034X.2012.01069.x, PMID 22971102.
Jeschke MG, Rensing H, Klein D, Schubert T, Mautes AE, Bolder U. Insulin prevents liver damage and preserves liver function in lipopolysaccharide-induced endotoxemic rats. J Hepatol. 2005;42(6):870-9. doi: 10.1016/j.jhep.2004.12.036, PMID 15885358.
Hotchkiss RS, Tinsley KW, Swanson PE, Chang KC, Cobb JP, Buchman TG. Prevention of lymphocyte cell death in sepsis improves survival in mice. Proc Natl Acad Sci USA. 1999;96(25):14541-6. doi: 10.1073/pnas.96.25.14541, PMID 10588741.
Pettila V, Hynninen M, Takkunen O, Kuusela P, Valtonen M. Predictive value of procalcitonin and interleukin 6 in critically ill patients with suspected sepsis. Intensive Care Med. 2002;28(9):1220-5. doi: 10.1007/s00134-002-1416-1, PMID 12209268.
Kareem RT, Abass MK. A potential anti-inflammatory effect of ertugliflozin in animal model. South Asian Res J Pharm Sci. 2024;6(3):84-8. doi: 10.36346/sarjps.2024.v06i03.006.
Alaasam ER, Janabi AM. Erythropoietin protects against renal ischemia/reperfusion injury in rats via inhibition of oxidative stress inflammation and apoptosis. J Contemp Med Sci. 2023;9(4):233-8. doi: 10.22317/jcms.v9i4.1405.
Alsaaty EH, Janabi AM. Moexipril improves renal ischemia/reperfusion injury in adult male rats. J Contemp Med Sci. 2024;10(1):25-30. doi: 10.22317/jcms.v10i1.1477.
Llorens Cebria C, Molina Van Den Bosch M, Vergara A, Jacobs Cacha C, Soler MJ. Antioxidant roles of SGLT2 inhibitors in the kidney. Biomolecules. 2022;12(1):143. doi: 10.3390/biom12010143, PMID 35053290.
Gao X, Yan X, Yin Y, Lin X, Zhang Q, Xia Y. Therapeutic targeting of apoptosis inhibitor of macrophage/CD5L in sepsis. Am J Respir Cell Mol Biol. 2019;60(3):323-34. doi: 10.1165/rcmb.2018-0272OC, PMID 30326743.
Yoon JH, Gores GJ. Death receptor-mediated apoptosis and the liver. J Hepatol. 2002;37(3):400-10. doi: 10.1016/S0168-8278(02)00209-X, PMID 12175638.
Mohammad AR, Shnaien AA, Alabsawy SK, Hassan ES. Protective effect of ipragliflozin on acute brain injury induced by endotoxemia in mice. Iran J War Public Health. 2023;15(3):225-31. doi: 10.58209/ijwph.15.3.225.
Park GB, Choi Y, Kim YS, Lee HK, Kim D, Hur DY. ROS-mediated JNK/p38-MAPK activation regulates Bax translocation in sorafenib-induced apoptosis of EBV-transformed B cells. Int J Oncol. 2014;44(3):977-85. doi: 10.3892/ijo.2014.2252, PMID 24402682.
Haddad JJ. The role of Bax/Bcl-2 and pro-caspase peptides in hypoxia/reperfusion-dependent regulation of MAPK(ERK): discordant proteomic effect of MAPK(p38). Protein Pept Lett. 2007;14(4):361-71. doi: 10.2174/092986607780363925, PMID 17504094.
Li S, Takahara T, Fujino M, Fukuhara Y, Sugiyama T, Li XK. Astaxanthin prevents ischemia-reperfusion injury of the steatotic liver in mice. PLOS One. 2017;12(11):e0187810. doi: 10.1371/journal.pone.0187810, PMID 29121675.
Hong JM, Kim SJ, Lee SM. Role of necroptosis in autophagy signaling during hepatic ischemia and reperfusion. Toxicol Appl Pharmacol. 2016;308:1-10. doi: 10.1016/j.taap.2016.08.010, PMID 27521978.