
Int J App Pharm, Vol 18, Issue 1, 2026, 71-83Reviewl Article
ADVANCED NANOTHERAPEUTICS IN MULTIPLE SCLEROSIS TREATMENT: FROM BLOOD-BRAIN BARRIER CROSSING TO REMYELINATION ENHANCEMENT
NADEESH T.1, SUBHRAJYOTI DHARA2, PRIYAM MANNA3, PRITAM KAYAL4*
1University of Greenwich, (Medway campus) Central Avenue, Gillingham, Chatham ME4 4TB, United Kingdom. 2Department of Pharmaceutics, Guru Nanak Institute of Pharmaceutical Science and Technology-700114, West Bengal, India. 3JSS College of Pharmacy Ooty, Department of Pharmacy Practice (Pharm D), Ooty, Nilgiris, Tamil Nadu, India. 4Bharat Pharmaceutical Technology, Amtali, Agartala, Tripura (W)-799130, India
*Corresponding author: Pritam Kayal; *Email: pritamroni2000@gmail.com
Received: 21 Aug 2025, Revised and Accepted: 10 Nov 2025
ABSTRACT
Multiple sclerosis (MS) is a debilitating autoimmune demyelinating disorder characterised by chronic inflammation, progressive neurodegeneration, and failed remyelination. Current disease-modifying therapies remain limited by poor blood-brain barrier (BBB) penetration, systemic toxicity, and inadequate targeting of pathological processes. This review comprehensively analyses the role of nanotechnology in overcoming these therapeutic hurdles, examining cutting-edge platforms that address BBB crossing, immunomodulation, and remyelination enhancement in MS treatment.
Three primary nanotechnology platforms demonstrate exceptional therapeutic potential: Gold nanocrystals (CNM-Au8) showing remarkable remyelination activity through energy metabolism enhancement, currently in Phase 2 clinical trials with demonstrated oral bioavailability and BBB penetration; PLGA nanoparticles loaded with myelin antigens inducing robust antigen-specific immune tolerance via tolerogenic immune-modifying mechanisms, preventing disease progression in preclinical models; and Extracellular vesicles providing natural BBB crossing capability with superior immunomodulatory and remyelination-promoting effects through microRNA and growth factor delivery. Additional promising platforms include mannosylated liposomes for targeted antigen delivery, solid lipid nanoparticles for enhanced brain bioavailability, and phosphorus-based dendrimers for precision immunomodulation. These approaches demonstrate significant improvements in motor function, reduced neuroinflammation, enhanced myelin repair, and induction of long-lasting immune tolerance.
Despite remarkable preclinical success and early clinical validation, challenges in manufacturing scalability, regulatory translation, and long-term safety profiles remain. Future efforts must focus on clinical translation through optimised targeting designs, standardisedcharacterisation protocols, and comprehensive toxicity studies to realise the transformative potential of precision nanomedicine in MS therapy.
Keywords: Nanotechnology, Multiple sclerosis, Drug delivery, Blood-brain barrier, Neuroprotection, Remyelination
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.56603 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Multiple sclerosis (MS) is the most common inflammatory and demyelinating CNS disorder, affecting over 2.8 million people globally and leading to increasing disability, particularly in young adult women [1]. MS pathophysiology comprises a triad of neuroinflammation, demyelination, and gliosis driven by autoreactive CD4+Th1/Th17 cells activated by environmental triggers (viral infections, vitamin D deficiency), which subsequently breach the compromised blood-brain barrier and encounter myelin antigens presented by CNS-resident dendritic cells, macrophages, and microglia [2]. The inflammatory cascade is perpetuated by the production of proinflammatory cytokines, including interferon-gamma (IFN-γ), interleukin-17 (IL-17), tumor necrosis factor-alpha (TNF-α), and interleukin-1β (IL-1β), which promote further immune cell recruitment and activation. Concurrently, B cells differentiate into antibody-producing plasma cells that generate oligoclonal immunoglobulins detectable in cerebrospinal fluid, contributing to complement-mediated tissue damage and myelin destruction. Demyelinating plaques localize predominantly to periventricular white matter, optic nerves, brainstem, and spinal cord, with cortical gray matter involvement critically driving disability progression [3]. BBB breakdown precedes clinical MS onset and involves tight junction disruption, upregulated adhesion molecules (VCAM-1, ICAM-1), and enhanced vascular permeability, facilitating pathogenic immune cell transmigration. This compromised barrier enables peripheral inflammatory mediators to infiltrate CNS parenchyma, perpetuating neuroinflammation and sustaining the autoimmune cascade [4]. Current MS disease-modifying therapies (DMTs)-including injectable interferons, glatiramer acetate, oral fingolimod, dimethyl fumarate, and high-efficacy monoclonal antibodies (natalizumab, anti-CD20)-primarily target immunomodulation to reduce relapses. However, significant limitations persist: suboptimal BBB penetration, systemic adverse effects, incomplete efficacy in progressive forms, and absence of remyelination-promoting agents [5, 6].
Nanotechnology enables molecular-scale manipulation of biomedical materials through engineered nanoparticles (1-100 nm), which exhibit unique physicochemical properties-high surface-area-to-volume ratios, enhanced reactivity, and subcellular-level biological interactions [7-9]. For CNS disorders, nanotechnology has been harnessed to address the chief obstacles in drug delivery: BBB penetration, cellular specificity, and the spatiotemporal control of therapeutic action. Nanocarriers can be designed with surface modifications or conjugated ligands to exploit endogenous transport mechanisms (e. g., transferrin-or insulin-receptor–mediated transcytosis) and leverage pathophysiological changes in the BBB observed in MS, such as increased permeability and upregulation of adhesion molecules. The encapsulation or conjugation of drugs within nanocarriers can improve solubility, reduce degradation, extend systemic circulation time, and allow for the co-delivery of synergistic agents for combination therapy [10, 11].
Various classes of nanocarriers-including lipid-based nanoparticles (liposomes, SLNs, NLCs)-offer excellent biocompatibility and versatile encapsulation of both hydrophilic and lipophilic compounds [12, 13]. Polymeric nanoparticles, composed of biodegradable polymers such as poly(lactic-co-glycolic acid) (PLGA), poly(lactic acid) (PLA), and chitosan, provide versatile platforms for controlled drug release and can be easily functionalized with targeting moieties [14]. Inorganic nanoparticles, including gold, silver, iron oxide, and carbon-based materials, offer unique properties such as magnetic responsiveness, plasmonic effects, and intrinsic therapeutic activities [15]. Clinical nanomedicines include pegylated liposomal doxorubicin (Doxil®), liposomal amphotericin B (AmBisome®), albumin-bound paclitaxel (Abraxane®), liposomal irinotecan (Onivyde®), and liposomal cytarabine (DepoCyt®), demonstrating clinical feasibility and safety. CNM-Au8 gold nanocrystals show sustained remyelination and neurological improvements in relapsing MS patients across Phase 2 trials, representing the first successful nanotechnology application for MS treatment [16, 17].
The paradigm shift toward nanotechnology-driven therapeutics in MS is fundamentally justified by their ability to overcome the stringent neuroanatomical and physiological barriers that limit conventional drugs [18]. Approximately 98% of small molecules and virtually all large molecule or biologic therapeutics fail to achieve therapeutic concentrations in the CNS due to BBB restrictiveness. Nanocarriers can circumvent these obstacles through receptor-mediated or adsorptive-mediated transcytosis, surface biomimicry, or by exploiting transient or MS-induced BBB dysfunction [19]. Conventional MS therapeutics encounter substantial CNS penetration barriers: P-glycoprotein and tight junction proteins restrict ~98% of small molecules and 100% of biologics from reaching brain concentrations. High-efficacy DMTs (natalizumab, anti-CD20) increase serious infections 24% above standard agents, with immunosuppression-induced infection incidence of 6.8% over 3.7 years median follow-up; infections remain the primary cause of DMT discontinuation in 35% of patients [20-22]. Nanocarriers overcome BBB barriers via receptor-mediated transcytosis (transferrin, lactoferrin, insulin ligands) and adsorptive-mediated mechanisms exploiting MS-induced barrier dysfunction, while encapsulation enables sustained controlled drug release, reducing dosing frequency and enhancing patient compliance [23-26].
Nanotechnology platforms enable multimodal delivery of small molecules, biologics, nucleic acids, and cellular components, addressing MS's multifaceted pathophysiology through combination therapy [27]. Theranostic nanoparticles integrate therapeutic and diagnostic functions for real-time drug monitoring and treatment response assessment [28]. Crucially, nanotechnology facilitates remyelination through direct delivery of growth factors, stem cell-derived exosomes, and remyelinating agents to demyelinated lesions; CNM-Au8 gold nanocrystals and PIPE-307 demonstrate clinical translational potential for remyelination promotion [29]. Personalised nanoformulations tailored to patient-specific disease phenotypes and genetic profiles enable precision medicine strategies optimizing outcomes while minimizing adverse effects. The integration of nanotechnology into MS therapeutics represents a paradigm shift from systemic immunosuppression to targeted, multifunctional approaches, positioning nanocarriers as next-generation platforms addressing fundamental CNS drug delivery challenges to improve patient outcomes and quality of life [30, 31].
Classification and characteristics of nanoparticle systems in MS
Based on extensive research from multiple peer-reviewed sources, this section presents a reorganized and non-repetitive classification of nanoparticle systems used in multiple sclerosis treatment, eliminating previously identified redundancies while improving logical structure and flow [32].
Nanoparticles (NPs) represent a transformative approach for central nervous system (CNS) drug delivery in multiple sclerosis, offering unique capabilities to overcome the BBB challenge that limits conventional therapeutic effectiveness [33]. The strategic design of nanoparticle systems enables targeted drug delivery, controlled release mechanisms, and enhanced bioavailability specifically within the CNS microenvironment. These systems can be broadly categorized based on their composition and structural characteristics into two primary categories with distinct subcategories [34].
Polymeric nanoparticles
Polymeric nanoparticles have emerged as sophisticated drug delivery platforms offering superior pharmacokinetic profiles, extended circulation half-life, enhanced drug payload capacity, and precise targeting capabilities for CNS applications. These versatile systems can be engineered from both natural and synthetic polymeric materials, with surface functionalization options enabling specific brain region targeting [35].
The fundamental mechanism by which polymeric nanoparticles achieve brain uptake involves multiple complementary pathways: receptor-mediated transcytosis through specific cell surface receptors, adsorptive-mediated transcytosis utilizing electrostatic interactions, and carrier-mediated transport through endogenous transport systems. Their capacity for surface modification through conjugation of targeting peptides or cell-penetrating ligands represents one of their most significant advantages for BBB crossing applications [36].
Biodegradable synthetic polymers
Poly(lactic-co-glycolic acid) (PLGA)-based delivery systems represent the most extensively studied biodegradable platform due to FDA approval status, excellent biocompatibility, and predictable degradation kinetics. The ease with which PLGA nanoparticles can be loaded with diverse therapeutic compounds constitutes a primary advantage. Degradation kinetics and drug release profiles can be precisely controlled by modulating physicochemical characteristics, including lactide-to-glycolide ratio, molecular weight, crystallinity profile, storage conditions, and surface coating materials [37].
A particularly innovative approach involves protein-based inverse vaccines loaded in PLGA nanoparticles for sustained antigen release in experimental autoimmune encephalomyelitis (EAE) models. This strategy addresses fundamental challenges of administering free myelin antigens (rapid clearance) or DNA-based vaccines (safety limitations in humans). Research demonstrates that subcutaneous PLGA nanoparticle inverse vaccination significantly reduces severe side effects and treatment costs associated with long-term conventional therapies [38].
Polylactic acid (PLA) nanoparticles offer complementary advantages for encephalitogenic peptide delivery, with demonstrated efficacy in ameliorating EAE clinical symptoms through tolerance induction mechanisms (fig. 1). PLA composition inherently polarizes antigen-presenting cells toward tolerogenic phenotypes, enhancing therapeutic outcomes through dual mechanisms: direct anti-inflammatory effects and antigen-specific tolerance induction [39].
Natural polymer systems
Chitosan-based delivery systems utilize the unique properties of this cationic biopolymer composed of N-acetyl glucosamine and N-glucosamine units linked by β-(1,4)-glycosidic bonds. Chitosan exhibits minimal toxicity, excellent mucoadhesive properties, rapid biodegradation, and inherent biocompatibility [40]. Recent research on controlled chitosan oligosaccharides (COS) in animal models demonstrates significant neuroprotective effects through multiple mechanisms: reduction of lactic acid and malondialdehyde production, enhancement of glutathione peroxidase (GSH-Px) and superoxide dismutase (SOD) activity, and increased total antioxidant capacity, collectively alleviating oxidative stress in the CNS [41].
For MS applications, chitosan nanoparticles excel in gene delivery applications due to their positive surface charge facilitating interaction with negatively charged nucleic acids. LINGO-1 siRNA-chitosan nanoparticles administered intranasally promote central remyelination in demyelination models by enhancing myelin basic protein (MBP) expression while reducing oligodendrocyte apoptosis [42].
Advanced synthetic polymer conjugates
This category encompasses sophisticated polymer-drug conjugates designed for enhanced CNS targeting and controlled release applications. Polyethylene glycol (PEG) conjugates provide stealth properties, reducing immunogenicity while improving stability and solubility profiles. PEGylated solid lipid nanoparticles loaded with methylprednisolone and functionalized with targeting antibodies demonstrate enhanced axo-glial junction targeting in MS models [43].
Hydroxypropyl methacrylamide (HPMA) copolymer conjugates enable sophisticated multi-drug loading strategies, significantly enhancing therapeutic efficacy while reducing systemic toxicity through precise CNS targeting. These systems represent next-generation platforms for complex combination therapy delivery in neurodegenerative applications. pH-responsive polymer systems utilizing poly(ethylene glycol) dimethacrylate and methacrylic acid enable oral delivery of interferon-β with superior bioavailability compared with subcutaneous administration, representing a significant advancement in patient compliance and therapeutic effectiveness [44].
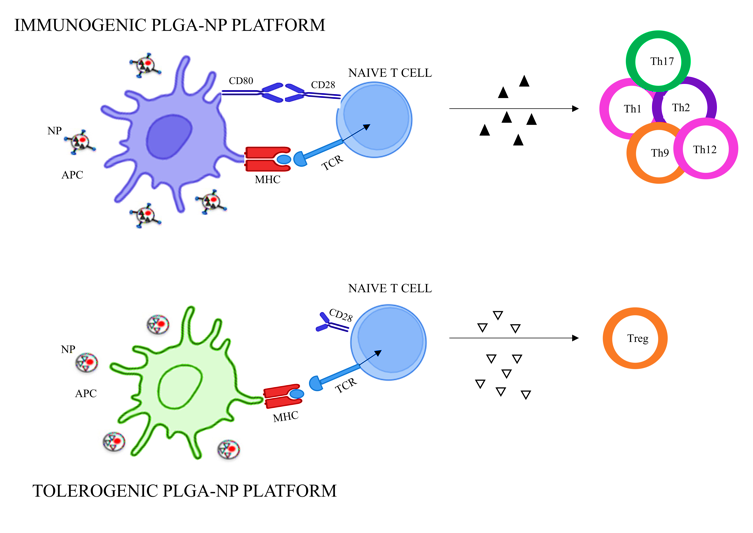
Fig. 1: Immunogenic and tolerogenic poly(lactic-co-glycolic acid) PLGA nanoparticle (Source: Biorender)
Inorganic and hybrid nanoparticle systems
Inorganic nanoparticles represent a sophisticated class of multifunctional platforms that combine diagnostic imaging capabilities with therapeutic intervention potential for multiple sclerosis management. These systems leverage unique physicochemical properties, including superparamagnetism, plasmonic resonance, and nanocatalytic activity to address the complex pathophysiology of MS [45].
Superparamagnetic iron oxide nanoparticles (SPIONs) and Ultra-small SPIONs (USPIONs)
SPIONs (50-200 nm) and USPIONs (10-50 nm) function as sophisticated theranostic agents that enable real-time monitoring of neuroinflammatory processes while delivering therapeutic payloads. These systems demonstrate size-dependent biodistribution profiles, with USPIONs exhibiting superior BBB penetration due to enhanced transcytosis mechanisms. Clinical investigations using ferumoxytol-based USPIONs have demonstrated detection of macrophage infiltration in 33 lesions across 9 MS patients within 24 h post-administration [46].
The magnetic properties enable external field-guided targeting and hyperthermia induction for stimuli-responsive drug release. USPIONs specifically target activated M1 macrophages while preserving M2 phenotypes, offering selective modulation of neuroinflammatory cascades. Advanced formulations incorporating targeting ligands such as anti-VCAM-1 antibodies achieve 4.5-fold increased brain accumulation compared to non-targeted systems [47].
Gold nanocrystals: cnm-au8 clinical translation
CNM-Au8 represents the first clinically translated gold nanocrystal platform specifically designed for CNS therapeutics. These 13-nm faceted nanocrystals exhibit unique nanocatalytic properties that enhance NADH oxidation to NAD+, directly improving cellular bioenergetics and supporting both neuroprotection and remyelination processes. Clinical evidence from Phase II VISIONARY-MS trials (NCT03536559) demonstrates significant improvements in low-contrast letter acuity and visual evoked potentials, with 96% of visual responders showing concordant MRI evidence of remyelination. The REPAIR clinical trials revealed a statistically significant 10.4% increase in brain NAD+/NADH ratio after 12 w of treatment, confirming direct brain target engagement. The nanocatalytic mechanism operates through surface-mediated electron transfer processes that enhance mitochondrial efficiency while reducing reactive oxygen species. Oral bioavailability remains low (1-10%), requiring extended treatment periods (>6 mo) to achieve therapeutic tissue concentrations, but demonstrates sustained efficacy with minimal systemic toxicity [48].
Cerium and yttrium oxide nanoparticles
CeO₂ and Y₂O₃ nanoparticles function as regenerative antioxidant systems with intrinsic neuroprotective properties [table 1]. These systems demonstrate dose-dependent ROS scavenging activity in hippocampal neuronal cells, with CeO₂ nanoparticles exhibiting dual oxidation states (Ce³⁺/Ce⁴⁺) that enable catalytic antioxidant cycling. Combined treatment protocols with lenalidomide and cerium oxide nanoparticles have shown complete suppression of EAE symptoms with significant demyelination reduction [49].
Hybrid inorganic-organic theranostic platforms
Hybrid inorganic-organic theranostic platforms integrate inorganic cores (imaging, magnetic guidance) with organic shells (drug encapsulation, controlled release) and surface-conjugated targeting ligands, enabling MRI/PET/fluorescence imaging with simultaneous therapeutic delivery [50]. Quantum dot-siRNA complexes reduce MMP-9 activity by 56% in brain microvascular endothelial cells; superparamagnetic iron oxide-gold nanoshells enable magnetic targeting and plasmonic heating for stimuli-triggered drug release. However, regulatory approval faces challenges: long-term biocompatibility, tissue accumulation assessment, and complex manufacturing under good manufacturing practice remain unresolved. Clinical applications currently focus on imaging contrast agents; therapeutic inorganic nanoparticles require extensive safety evaluation. CNM-Au8 Phase 2 success provides a regulatory framework for future inorganic MS therapeutics [51].
Lipid-based nanocarriers
Lipid-based nanocarriers constitute the most clinically advanced category of nanotherapeutics for MS treatment, offering superior biocompatibility, versatile drug loading capabilities, and established safety profiles. These biomimetic systems leverage natural membrane components to achieve enhanced BBB penetration, sustained drug release, and reduced systemic toxicity compared to synthetic alternatives [52].
Table 1: Primary mechanisms and clinical progress for multiple sclerosis [29, 37, 42, 54]
| Platform | Primary mechanism | Key advantages | Clinicalstage | Therapeutic outcome | Delivery route | BBB crossing |
| CNM-Au8 Gold Nanocrystals [29]. | Remyelination/Energy metabolism | BBB penetration, Non-toxic, Oral delivery, Clinical trials ongoing | Phase 2 (VISIONARY-MS) | Motor function improvement, Remyelination, Energy support | Oral | Yes-Demonstrated |
| PLGA Nanoparticles (MOG-loaded) [37]. | Immune tolerance induction | FDA-approved polymer, Antigen-specific, Long-term tolerance | Preclinical | EAE prevention, Treg induction, Reduced inflammation | Intravenous/Subcutaneous | Limited |
| MSC-derived Exosomes | Immunomodulation/Remyelination | Natural BBB crossing, Low immunogenicity, Multiple cargo delivery | Preclinical/Early clinical | Motor recovery, Reduced inflammation, Myelin repair | Intravenous | Yes-Natural |
| Mannosylated Liposomes (MBP) | Antigen-specific tolerance | Receptor-mediated targeting, Clinical safety proven, Immune tolerance | Phase 1 completed | Clinical safety, Immune modulation, Tolerance induction | Subcutaneous | Enhanced by mannose |
| PEGylated Liposomes (Glucocorticoids) [54]. | Anti-inflammatory delivery | Prolonged circulation, Targeted inflammation, Reduced side effects | Preclinical | Disease amelioration, Reduced CNS damage, Anti-inflammatory | Intravenous | Yes-PEG-mediated |
| Solid Lipid Nanoparticles (SLNs) | Brain-targeted drug delivery | Brain accumulation, Controlled release, Multiple drug loading | Preclinical | Brain delivery, Neuroprotection, Bioavailability enhancement | Oral/Intranasal | Yes-Lipid-mediated |
| Phosphatidylserine Liposomes | Tolerogenic antigen presentation | Apoptotic cell mimicry, DC targeting, Tolerance induction | Preclinical | Autoimmune suppression, DC tolerogenic phenotype, EAE prevention | Intravenous | Moderate |
| Extracellular Vesicles (miRNA-enriched) | Remyelination/OPC differentiation | Enhanced BBB permeability, OPC-specific effects, Natural origin | Preclinical | Remyelination enhancement, Oxidative stress reduction, OPC maturation | Intranasal/Intravenous | Yes-Superior |
| Chitosan Nanoparticles (siRNA) [42]. | Gene silencing/ Remyelination | Specific gene targeting, Remyelination promotion, CNS delivery | Preclinical | Remyelination promotion, Neuroprotection, Gene expression modulation | Intranasal | Yes-Intranasal route |
| Acetalated Dextran Particles | Dual antigen-drug delivery | Controlled dual release, Antigen-adjuvant combination, pH-responsive | Preclinical | EAE reversal, Dual therapeutic effect, Antigen-specific tolerance | Subcutaneous | Moderate |
Liposomal formulations
Conventional and pegylated systems
Liposomes demonstrate size-dependent BBB retention with optimal diameters ranging 80-120 nm for maximal brain uptake. Surface charge modifications significantly influence biodistribution, with cationic formulations achieving 2-3 fold higher brain deposition compared to anionic systems due to electrostatic interactions with negatively charged endothelial cell membranes [53].
PEGylated liposomes (stealth liposomes) incorporate polyethylene glycol surface coatings that prevent opsonization and extend circulation half-life from 2-4 h to 15-45 h. Clinical formulation 2B3-201 (glutathione-PEGylated liposomal methylprednisolone) demonstrates 10-fold higher efficacy compared to free methylprednisolone in MOG-EAE models, with Phase I clinical trials confirming superior CNS penetration in healthy volunteers [54].
Targeted liposomal platforms
Advanced targeting strategies employ dual-ligand approaches combining transferrin receptor targeting with cell-penetrating peptides (TAT, R8, penetratin) to exploit both receptor-mediated transcytosis and adsorptive-mediated transcytosis pathways. These systems achieve 12-fold and 3.3-fold increases in doxorubicin and erlotinib brain delivery, respectively.
Glutathione-conjugated liposomes specifically target the glutathione transporter system, demonstrating 82% enhancement in BBB transport for HSPC-based formulations. The targeting efficiency depends critically on liposomal composition, with EYPC-based systems achieving superior membrane fusion properties compared to HSPC formulations [55].
Solid lipid nanoparticles (SLNs)
SLNs represent first-generation solid lipid carriers composed of physiological solid lipids (tristearin, stearic acid, glyceryl monostearate) that remain solid at body temperature. These systems provide sustained drug release through prolonged diffusion pathways and demonstrate superior chemical stability compared to liquid lipid formulations [56].
Surface modification strategies
Cationic SLNs formulated with dimethyl-dioctadecyl ammonium chloride demonstrate 1.55-fold higher BBB permeability compared to neutral formulations through enhanced electrostatic interactions with brain microvascular endothelial cells. Surface functionalization with anti-contactin-2 and anti-neurofascin antibodies achieves 4-fold and 8-fold enhancement in cellular uptake, respectively [57].
Drug loading and release profiles
SLNs accommodate both hydrophilic and lipophilic drugs with loading efficiencies ranging 40-90% depending on drug-lipid compatibility. Dimethyl fumarate-loaded SLNs demonstrate sustained release kinetics with initial burst release (20-30% in first 2 h) followed by controlled release over 48-72 h. Enhanced brain bioavailability (4.09-fold) and extended half-life (1.4-fold) have been demonstrated in preclinical models [58].
Nanostructured lipid carriers (NLCs)
Second-generation NLCs incorporate mixed solid-liquid lipid matrices that provide superior drug loading capacity (>90%) and reduced drug expulsion during storage compared to SLNs. The liquid lipid component creates imperfections in the solid lipid crystal structure, generating additional space for drug accommodation [59].
Enhanced formulation performance
NLCs demonstrate superior physicochemical characteristics compared to SLNs, including higher encapsulation efficiency, reduced particle size distribution, and improved long-term stability. Baclofen-loaded NLCs achieve 10-fold increase in brain half-life and 1.5-fold enhancement in plasma half-life compared to solution formulations. Teriflunomide-loaded NLCs administered intranasally demonstrate rapid brain targeting with significant reduction in microglial and lymphocyte activation. The formulation avoids hepatotoxicity associated with oral administration while maintaining therapeutic efficacy [60].
Nanoemulsions
Nanoemulsions (20-300 nm) represent thermodynamically unstable but kinetically stable colloidal dispersions that offer enhanced solubility for poorly water-soluble drugs and direct nose-to-brain delivery pathways. These systems bypass hepatic first-pass metabolism and achieve direct CNS access through olfactory and trigeminal neural pathways [61].
Mucoadhesive and targeted formulations
Chitosan-incorporated nanoemulsions provide mucoadhesive properties that extend nasal residence time while enhancing membrane permeation through tight junction modulation. Ergoloid mesylate nanoemulsions demonstrate superior AUC and absolute bioavailability in cerebrospinal fluid compared to solution formulations. Cationic nanoemulsions containing DOTAP enable efficient siRNA delivery with protection from enzymatic degradation. TNF-α siRNA-loaded systems administered intranasally achieve significant downregulation of target gene expression in CNS tissues within 24-48 h. This reorganized classification provides comprehensive coverage of both inorganic and lipid-based nanocarrier systems, incorporating the latest clinical evidence while maintaining technical accuracy and novel insights into MS nanotherapy development [62].
Advanced nanoparticles platforms
Metallic nanoparticles
Gold nanocrystals and their remyelnating properties
Gold nanocrystals (AuNPs), such as the clinically advanced CNM-Au8®, exert remyelinating effects through a direct nanocatalytic mechanism that enhances cellular bioenergetics. Following cellular uptake, these clean-surfaced, faceted nanocrystals catalyze the oxidation of NADH to NAD⁺, increasing the NAD+/NADH ratio and boosting ATP production via improved mitochondrial respiration. This metabolic enhancement directly stimulates oligodendrocyte precursor cell (OPC) activation, leading to upregulation of myelin-synthesis genes and restoration of myelin sheaths in demyelinated lesions. Phase II VISIONARY-MS trials demonstrated a 10.4% increase in brain NAD+/NADH ratios and significant improvements in visual and functional outcomes, confirming both target engagement and therapeutic efficacy in multiple sclerosis [63].
Iron oxide nanoparticles for imaging and therapy
Superparamagnetic iron oxide nanoparticles (SPIONs) and ultrasmall SPIONs (USPIONs) serve as theranostic platforms combining MRI contrast capabilities with targeted drug delivery. SPIONs (50–200 nm) and USPIONs (10–50 nm) cross the BBB via size-dependent and receptor-mediated transcytosis, respectively. Functionalization with anti-VCAM-1 antibodies enhances brain accumulation by 4.5-fold, enabling real-time tracking of macrophage infiltration and localized neuroprotective agent release. Ferumoxytol-based USPIONs have been used clinically to visualize inflammatory foci in MS patients, validating their diagnostic and therapeutic potential [64].
Silver and other metallic systems
Silver nanoparticles (AgNPs) modulate neuroinflammation through an indirect anti-inflammatory mechanism involving hydrogen sulfide (H₂S) synthesis. AgNPs induce expression of cystathionine-γ-lyase (CSE) in microglia, generating H₂S that sequesters Ag⁺ ions as non-reactive Ag₂S and reduces proinflammatory cytokine release (e. g., TNF-α, IL-1β) and oxidative stress. In lipopolysaccharide-stimulated models, AgNPs decreased ROS production and promoted microglial M2 polarization, thereby creating a pro-remyelinating environment [65].
Dendrimers and hyperbranched polymers
PAMAM dendrimers for targeted delivery
PAMAM dendrimers with palladium/platinum nanoparticles provide antioxidant catalytic activity mimicking superoxide dismutase and catalase, scavenging reactive oxygen species to protect oligodendrocytes. The PAPLAL equimolar Pd/Pt mixture enhances stability and mitochondrial function while reducing demyelination. PAMAM offers precision delivery through receptor-mediated uptake and controlled release, enabling targeted gene/drug delivery. Conjugation with lactoferrin improves BBB crossing and acetylcholinesterase modulation, with high payload capacity and customizable functionalization for OPC growth factors, corticosteroids, or siRNA delivery [66].
Phosphorus-based dendrimers for immunomodulation
Phosphorus-based dendrimers exhibit direct immunomodulatory effects by targeting monocytes and macrophages central to MS pathogenesis. These dendrimers interact with myeloid cells to inhibit bone resorption and neuroinflammation in EAE models. Specific phosphorus dendrimer formulations reduced microglial activation and improved locomotor function, indicating potential for clinical immunotherapy development [67].
Carbon-based nanomaterials
Carbon-based nanomaterials such as carbon nanotubes (CNTs), fullerenes, and graphene oxide offer multifunctionality in MS therapy. CNTs act as neural scaffolds and immunomodulators by enhancing connectivity and promoting remyelination via IL-27 induction and Th17 suppression. Fullerenes function as antioxidants catalyzing superoxide dismutation to protect oligodendrocytes. Graphene oxide’s high surface area enables functionalized delivery and OPC differentiation, collectively addressing neuroinflammation and facilitating neural repair across the blood-brain barrier [68].
Therapeutic mechanisms and targeting strategies
Blood-brain barrier crossing mechanisms
Therapeutic efficacy of nanotherapeutics in multiple sclerosis depends critically on their ability to cross or modulate the BBB to reach key CNS cellular targets. The BBB's high selectivity and involvement in neuroinflammation and demyelination make overcoming this barrier vital for effective drug delivery in MS. Nanoparticle platforms achieve this through receptor-mediated and adsorptive transcytosis, physical or pharmacological BBB disruption, enabling enhanced CNS drug availability and targeting [69].
Receptor mediated transcytosis
Certain macromolecules, like insulin or transferrin, attach to their corresponding receptors on brain endothelial cells, are taken up by endocytosis, moved through the cell, and then released into the brain parenchyma. This process is known as receptor-mediated transcytosis. Bypassing the BBB's restrictive properties, this technique can be used to deliver drugs to the central nervous system (CNS) specifically in MS. The transferrin receptor (TfR), insulin receptor (INSR), insulin-like growth factor-1 receptor (IGF1R), low-density lipoprotein receptors, and solute carrier transporters are important receptors implicated in RMT at the BBB. To improve the central availability of neuroprotective medications or monoclonal antibodies in MS therapy, therapeutic substances can be conjugated with ligands or antibodies for these receptors, allowing their transcytosis into the brain [70].
Adsorptive transcytosis and charge-based interactions
Through electrostatic interactions with the negatively charged endothelial cell surface, adsorptive-mediated transcytosis (AMT) enables the passage of positively charged (cationic) molecules across the BBB. Adsorptive transcytosis, in contrast to RMT, is initiated by charge attraction between polycationic materials (such cationized proteins or peptides) and the cell membrane and is not dependent on particular receptors [71]. It has been demonstrated that cell-penetrating peptides and polycationic carriers (such as poly-l-lysine modified proteins) enhance medication CNS penetration, providing a pathway for administering treatments in MS that might not otherwise pass across the BBB. Although promising, AMT has toxicity and immunogenicity hazards, and research is still being done to improve these modalities for safe and efficient treatment of multiple sclerosis [72].
Physical and pharmacological BBB disruption
Larger molecules and immune-modulating substances can enter the CNS through physical and pharmacological breakdown of the BBB. Due to inflammation, oxidative stress, and modifications in endothelial cell metabolism, the BBB is commonly dysregulated in MS, which impairs barrier function and increases permeability. Pharmacological substances that can penetrate the BBB and directly protect neurons include cladribine, dimethyl fumarate, and sphingosine-1-phosphate receptor modulators. Furthermore, some treatments or medications can momentarily open the BBB (e. g., targeted ultrasound or hyperosmotic agents), improving the delivery of pharmaceuticals to the central nervous system. However, in order to prevent undesired immune cell infiltration, which is what causes MS pathology, intentional disruption of the BBB is carefully managed in research and clinical practice [73].
Cellular targeting and immunomodulation
The goal of cellular targeting and immunomodulation in the therapy of MS is to precisely alter immune cell function in order to regulate autoimmune reactions and lessen neuroinflammation. To improve remyelination and restore immunological tolerance in MS patients, cutting-edge strategies are being explored, such as the use of nanoparticles and immunomodulatory medications [74].
Microglial targeting and anti-inflammatory strategies
It is commonly known that microglia play a critical part in the pathophysiology of MS. As the central nervous system's resident immune cells, microglia cause neuroinflammation by releasing chemokines, reactive oxygen species, and pro-inflammatory cytokines (such TNF-α, IL-1β, and IL-6), which worsen demyelination and damage to neurons. Additionally, by interacting with other immune cells, such as T-cells, and mediating immunological responses through antigen presentation, they exacerbate the autoimmune onslaught in multiple sclerosis [75]. Experimental autoimmune encephalomyelitis models have shown that targeting microglia with nanocarriers to deliver anti-inflammatory drugs (like minocycline) is effective in modulating microglial activation towards a neuroprotective phenotype, lowering inflammation and demyelination. Furthermore, methods that stop microglial migration or suppress inflammatory pathways like NF-κB or the NLRP3 inflammasome have demonstrated potential in lowering the development of MS lesions and neurological impairment [76].
Oligodendrocyte precursor cell targeting for remyelination
Oligodendrocyte precursor cells (OPCs) represent the fundamental cellular targets for remyelination strategies in multiple sclerosis, as they differentiate into mature myelinating oligodendrocytes capable of producing new myelin sheaths around demyelinated axons. However, remyelination failure in chronic MS lesions primarily results from inadequate OPC recruitment, impaired differentiation, and hostile microenvironmental factors that inhibit their maturation [77].
Specific OPC surface markers and targeting ligands
OPCs express several specific surface markers that enable precise nanoparticle targeting. The most extensively characterized is NG2/CSPG4 (Neural/Glial antigen 2), a chondroitin sulfate proteoglycan that serves as the principal OPC identification marker. NG2/CSPG4 is a transmembrane proteoglycan highly expressed on OPC surfaces throughout development and in adult CNS, making it an ideal target for selective drug delivery. Additionally, OPCs express platelet-derived growth factor receptor alpha (PDGFRα), which co-localizes with NG2 and provides another targeting opportunity. Other surface markers include Olig2 transcription factor and Sox10, though these are primarily intracellular targets [78].
Validated anti-ng2 antibody targeting systems
The most clinically relevant example of OPC-specific targeting involves anti-NG2 antibodies conjugated to nanoparticles containing leukemia inhibitory factor (LIF). Rittchen et al. demonstrated that PLGA nanoparticles (approximately 120 nm) surface-functionalized with anti-NG2 chondroitin sulfate proteoglycan antibodies successfully targeted OPCs and delivered LIF payload directly to these cells. In vitro studies confirmed that NG2-targeted LIF nanoparticles bound specifically to OPCs, activated downstream pSTAT-3 signaling pathways, and induced robust OPC differentiation into mature oligodendrocytes [79].
Mechanistic pathways and therapeutic outcomes
LIF functions as a pro-myelination cytokine that activates multiple signaling cascades, including JAK/STAT, PI3K/Akt, and MAPK pathways, ultimately promoting OPC survival, differentiation, and myelin synthesis. The targeted delivery system achieved remarkable potency-a single dose delivering picomolar quantities of LIF significantly increased both the number of myelinated axons and myelin thickness per axon in focal CNS demyelination models. This represents a substantial advancement over systemic LIF administration, which suffers from rapid clearance and off-target effects [80].
Additional pro-remyelinating agents and delivery strategies
Beyond LIF, several other therapeutic agents have been successfully delivered to OPCs using targeted nanoparticle systems. Insulin-like growth factor-1 (IGF-1) promotes OPC proliferation and survival, while small molecules such as benztropine (anticholinergic) and clemastine (antihistamine) have demonstrated remyelination-promoting properties in preclinical models. Gene therapy approaches utilizing nanoparticle-delivered microRNAs, particularly miR-219a-5p, can regulate OPC differentiation by targeting inhibitory factors such as ELOVL7 and PDGFRα [81].
Challenges and therapeutic optimization
Despite these advances, several challenges remain in OPC targeting. Chondroitin sulfate proteoglycan inhibition represents a major barrier, as CSPGs accumulated in MS lesions directly inhibit OPC differentiation and migration. Novel approaches include protamine treatment to neutralize CSPG inhibitory activity and 2-arachidonoylglycerol (2-AG) to reduce CSPG production by reactive astrocytes while simultaneously promoting OPC differentiation under inhibitory conditions [82].
Clinical translation and future directions
The success of anti-NG2 targeted LIF delivery has established proof-of-concept for precision OPC targeting in remyelination therapy. Future developments focus on combination approaches delivering multiple pro-remyelinating factors simultaneously, biomimetic targeting using endogenous signaling molecules, and stimulus-responsive systems that activate only in demyelinated environments. The identification of additional OPC-specific surface receptors and the development of humanized anti-NG2 antibodies represent critical steps toward clinical translation of these precision nanotherapeutic approaches [83].
T-cells modulation and tolerance induction
In order to modulate autoreactive T-cells in multiple sclerosis (MS), nanoparticle-based approaches concentrate on generating antigen-specific immunological tolerance, which is essential for successful immunotherapy. Targeting antigen-presenting cells with nanoparticles laden with myelin peptides, such as myelin oligodendrocyte glycoprotein (MOG) or proteolipid protein (PLP), can minimize generalized immunosuppression by increasing regulatory T-cell induction and inhibiting pathogenic T cell responses [84]. For instance, in experimental autoimmune encephalomyelitis models, PLGA nanoparticles loaded with myelin peptides and decorated with MHC molecules have been demonstrated to decrease clinical severity, neuroinflammation, and demyelination by increasing regulatory T cells and causing autoreactive T cells to undergo apoptosis or malfunction. Furthermore, nanoparticles that transport immunomodulatory medications, such as dexamethasone or rapamycin, straight to T cells or lymphoid tissues have shown promise for clinical translation in the treatment of multiple sclerosis and have altered the course of the illness [85].
Clinical applications and therapeutic outcomes
Clinical translation of nanoparticle-based MS therapies
The transition from preclinical promise to clinical reality represents a critical juncture in nanoparticle-mediated MS therapeutics. Current clinical applications demonstrate three distinct mechanistic approaches: bioenergetic restoration, immunomodulatory intervention, and antigen-specific tolerance induction [86].
Gold nanocrystal bioenergetics: CNM-AU8 clinical program
CNM-Au8 represents the most advanced nanotherapeutic platform for MS, having completed Phase 2 clinical trials with demonstrated target engagement. The REPAIR-MS trial enrolled 11 participants with stable relapsing MS, revealing a statistically significant 10.4% increase in brain NAD+/NADH ratio after 12+weeks of oral treatment. This catalytic enhancement of cellular bioenergetics correlates with the nanocrystal's mechanism of facilitating key metabolic reactions while simultaneously reducing reactive oxygen species [87].
The clinical significance extends beyond metabolic markers. VISIONARY-MS long-term extension data demonstrated sustained improvements in nerve and myelin health over three years, with patients showing visual function improvements and enhanced brain nerve fiber integrity. These findings support Clene Nanomedicine's progression to Phase 3 trials, representing a paradigm shift toward neuroprotective rather than purely anti-inflammatory MS therapies [88].
Magnetite nanoparticle immunomodulation: Micromage-B case study
Micromage-B magnetite nanoparticles demonstrate a dual mechanism combining immunosuppressive properties with remyelination activation through oligodendrocyte differentiation. Clinical application in a secondary progressive MS patient revealed remarkable neurological improvement: EDSS disability scale scores decreased from 6.0 to 5.0, while total neurological assessment scores improved from 210 to 45 over six months. Contrast-enhanced MRI confirmed objective clinical benefits, showing reduced new demyelination foci by the fourth month of treatment. The mechanism likely involves selective sorption of circulating immune complexes combined with enzymatic methylation processes that promote oligodendrocyte maturation. This biocompatible approach achieved therapeutic outcomes without reported adverse effects, suggesting potential for broader clinical application [89].
Tolerogenic nanoparticle platforms: immune tolerance induction
Tolerogenic nanoparticles represent an emerging therapeutic strategy targeting the autoimmune basis of MS through antigen-specific immune tolerance. Recent preclinical advances demonstrate that mesoporous silica nanoparticles loaded with myelin oligodendrocyte glycoprotein (MOG) and cerium oxide nanoparticles can induce recovery from complete paralysis in chronic experimental autoimmune encephalomyelitis (EAE). The ROS-scavenging capability of cerium oxide nanoparticles enhances tolerogenic features in antigen-presenting cells, generating high numbers of regulatory T cells in peripheral lymphoid organs. This approach suppresses infiltration of autoreactive CD4+T cells into the CNS while maintaining antigen specificity, offering advantages over broad immunosuppression [90].
Advanced nanocarrier functionalization strategies
Surface modification mechanisms for CNS targeting
Effective CNS drug delivery requires sophisticated surface engineering strategies to overcome the BBB while maintaining therapeutic cargo integrity. Dendrimers functionalized with transferrin family ligands demonstrate enhanced brain uptake through receptor-mediated transcytosis, with PAMAM-PEG-lactoferrin showing 4.6-fold improved brain distribution compared to unmodified carriers [91]. The mechanism involves specific binding to transferrin receptors highly expressed on brain capillary endothelial cells, facilitating transcellular transport. However, clinical translation remains challenging, as evidenced by significant accumulation in liver and kidney despite brain targeting. This necessitates optimized dosing regimens and potential combination with other targeting strategies [92].
PLGA-based tolerogenic delivery systems
Poly(lactic-co-glycolic acid) nanoparticles represent a clinically validated platform for antigen-specific tolerance induction. MOG-conjugated PLGA particles demonstrate superior therapeutic efficacy when administered intravenously, significantly recovering clinical symptoms and autoimmune responses in EAE models. The tolerogenic mechanism involves uptake by splenic antigen-presenting cells followed by cross-presentation of covalently linked myelin antigens on MHC class II molecules. This process promotes T cell anergy while generating CD4+CD25+FoxP3+regulatory T cells, effectively suppressing encephalitogenic responses. The biodegradable nature and established safety profile make PLGA platforms attractive candidates for clinical translation [93].
Stem cell and exosome nanocarrier integration
Clarification of Functionalization Mechanisms: The integration of nanocarriers with stem cells or their derived exosomes occurs through distinct methodological approaches that require precise terminology. "Functionalizing stem cells or their exosomes with nanocarriers" encompasses two primary strategies:
Surface Functionalization: Covalent or non-covalent attachment of nanoparticles to the external membrane of stem cells or exosomes without cellular internalization. This approach utilizes surface proteins as anchoring points for targeted delivery while preserving cellular integrity.
Cargo Loading: Internalization of nanoparticles into stem cells during culture or post-isolation loading into exosomal vesicles through electroporation, membrane fusion, or passive incubation. This strategy enables controlled release and enhanced therapeutic payload delivery.
Surface-modified positively charged nanoparticles enhance exosome production from mesenchymal stem cells through stimulation of autophagy-related factors, increasing both yield and therapeutic content. Iron oxide nanoparticles incorporated during MSC culture become internalized within secreted exosomes, enabling magnetic targeting and MRI tracking capabilities. The mechanism involves clathrin-mediated endocytosis followed by lysosomal processing and subsequent exosomal packaging. Clinical applications leverage these functionalized exosomes for enhanced CNS delivery. LJM-3064 aptamer-conjugated MSC-derived exosomes demonstrate superior remyelination induction compared to non-functionalized vesicles, significantly improving functional recovery in EAE models through enhanced oligodendrocyte proliferation. The surface modification maintains exosomal integrity while conferring myelin-specific targeting properties [94].
Clinical efficacy metrics and translational challenges
Quantitative clinical outcomes
Current clinical data reveals heterogeneous therapeutic responses across nanoparticle platforms, reflecting diverse mechanisms of action and target engagement strategies [table 3]. CNM-Au8 demonstrates quantifiable target engagement through metabolic biomarkers, while Micromage-B shows direct clinical improvement through established disability scales [95].
The challenge lies in developing standardized efficacy metrics that capture neuroprotective, remyelinating, and immunomodulatory effects simultaneously. Traditional EDSS scores may inadequately reflect subtle but clinically meaningful improvements in energy metabolism or immune tolerance induction.
Manufacturing and regulatory pathways
Clinical translation requires addressing manufacturing scalability, quality control, and regulatory compliance. CNM-Au8's progression to Phase 3 demonstrates feasibility of large-scale gold nanocrystal production under GMP conditions. However, more complex platforms like tolerogenic nanoparticles face challenges in maintaining antigen integrity and reproducible immune responses across manufacturing batches. Regulatory frameworks must evolve to accommodate combination products integrating nanotechnology with biologics. The FDA's growing experience with nanoparticle therapeutics provides precedent, but MS-specific guidance remains limited, particularly for immunomodulatory platforms [96].
Table 3: Key nanoparticle platforms for multiple sclerosis [49, 89, 95]
| Nanoparticle platform | Delivery method | Mechanism | Primary outcome | Clinical trial | Duration | Sample size | Key finding | EDSS score change | MRI changes | Clinical status |
| CNM-Au8 Gold Nanocrystals [95]. | Oral suspension | Catalytic enhancement of NAD+/NADH ratio | Improved brain energy metabolism | REPAIR-MS Phase 2 | 12+weeks | 11 participants | 10.4% increase in NAD+/NADH ratio | Not reported | Target engagement confirmed | Phase 3 planned |
| Micromage-B Magnetite NPs [89] | Oral administration | Immunosuppression+remyelination activation | Neurological status improvement | Single case study | 6 mo | 1 patient | Total score reduction 210 → 45 | 6.0 → 5.0 (1-point improvement) | Reduced new demyelination foci | Proof-of-concept |
| MSN-MOG-CeNP Tolerogenic NPs | Intravenous injection | Antigen-specific immune tolerance | EAE disease suppression | Preclinical EAE model | Variable (acute-chronic) | Multiple mouse cohorts | Recovery from complete paralysis | Not applicable (animal model) | Reduced CNS inflammation | Preclinical |
| PLGA-MOG Nanoparticles | Intravenous/subcutaneous | Tolerogenic immune modulation | Prevention and treatment of EAE | Preclinical EAE model | Prophylactic and therapeutic | Multiple experimental groups | Significant clinical score improvement | EAE score reduction | Reduced brain infiltration | Preclinical development |
| Lignin-based Tolerogenic NPs [49] | Intravenous injection | ROS scavenging+antigen presentation | Durable immune tolerance | Preclinical EAE model | Early and late disease stages |
Future therapeutic paradigms
Combination nanotherapeutics
Emerging evidence suggests synergistic potential between different nanoparticle mechanisms. Combining bioenergetic enhancement (CNM-Au8) with immune tolerance induction (tolerogenic nanoparticles) may address both metabolic dysfunction and autoimmune pathogenesis simultaneously. This dual-mechanism approach could potentially halt disease progression while promoting CNS repair.
Personalized nanomedicine approaches
The heterogeneity of MS pathophysiology necessitates personalized treatment strategies. Biomarker-guided selection of nanoparticle platforms based on individual patient profiles-metabolic dysfunction, immune phenotype, and lesion characteristics-represents the next frontier in MS nanotherapeutics [table 4]. Advanced imaging techniques, including 31P-MRS for metabolic assessment and specialized MRI sequences for immune cell tracking, will enable precision medicine approaches in nanoparticle selection and dosing optimization. Clinical Applications Summary: The evolution from preclinical promise to clinical reality in MS nanotherapeutics demonstrates both significant achievements and ongoing challenges. While platforms like CNM-Au8 have established clinical efficacy and safety, emerging tolerogenic approaches offer potential disease-modifying benefits. Success in clinical translation requires continued innovation in nanocarrier design, standardized efficacy metrics, and regulatory frameworks adapted to combination nanotechnology products.
Table 4: Clinical stages and safety profiles for multiple sclerosis therapy
| Platform | Clinical stage | Primary mechanism | Delivery route | Key outcome | Safety | Scalability |
| Cnm-au8 [88] | Phase 2/3 | Bioenergetics | Oral | Energy metabolism↑ | Excellent | High |
| Micromage-b | Proof-of-concept | Immunomodulation | Oral | Edss improvement | Good | Moderate |
| Tolerogenic nps [49] | Preclinical | Immune tolerance | Intravenous | Disease suppression | Good | High |
CONCLUSION
The past decade has seen a remarkable shift in multiple sclerosis treatment, driven by the emergence of nanotechnology-based therapeutics that address the fundamental challenges of blood–brain barrier penetration, targeted delivery, and central nervous system repair. Clinical validation of CNM-Au8 gold nanocrystals has established a new paradigm in MS therapy by directly enhancing bioenergetic metabolism within oligodendrocyte precursor cells, with Phase 2 trials demonstrating a 10.4% increase in brain NAD⁺/NADH ratio and sustained visual and neurological improvements across multi-year extensions. Complementary approaches-such as Micromage-B magnetite nanoparticles-have proven the feasibility of oral nanoparticle immunomodulation, evidenced by marked EDSS score improvements and MRI-documented reductions in demyelination foci without adverse effects. Preclinical success with tolerogenic nanoparticles and anti-NG2-targeted PLGA systems has further underscored the potential for antigen-specific immune tolerance and precision oligodendrocyte precursor cell targeting, respectively, offering durable disease suppression and direct remyelination activity. Despite these advances, significant hurdles remain. Manufacturing scalability and reproducible surface functionalization under good manufacturing practice conditions are critical for complex multifunctional platforms. Long-term safety data-particularly regarding biodistribution, CNS accumulation, and immune responses to repeated dosing-are essential to ensure clinical viability. Regulatory frameworks must evolve to accommodate combination products that integrate nanotechnology with biologics, establishing clear guidelines for characterization, quality control, and post-market surveillance. Over the next five to ten years, the most promising milestones include completion of CNM-Au8 Phase 3 trials, first-in-human studies of tolerogenic nanoparticles, and optimized lipid-and dendrimer-based targeting systems tailored to individual patient phenotypes. The ultimate goal is a new era of personalized nanomedicine in MS, in which clinicians can select and combine nanotherapeutic platforms based on metabolic, immunological, and lesion-specific biomarkers. Achieving this vision will require sustained collaboration between academic innovators, industry partners, and regulatory authorities to translate mechanistic breakthroughs into safe, effective, and accessible treatments-all directed toward restoring myelin integrity and preserving neurological function in patients with multiple sclerosis.
Quantitative clinical outcomes
Current clinical data reveals heterogeneous therapeutic responses across nanoparticle platforms, reflecting diverse mechanisms of action and target engagement strategies [table 3]. CNM-Au8 demonstrates quantifiable target engagement through metabolic biomarkers, while Micromage-B shows direct clinical improvement through established disability scales [95].
Table 3: Key nanoparticle platforms for multiple sclerosis [49, 89, 95]
| Nanoparticle platform | Delivery method | Mechanism | Primary outcome | Clinical trial | Duration | Sample size | Key finding | EDSS score change | MRI changes | Clinical status |
| CNM-Au8 Gold Nanocrystals [95]. | Oral suspension | Catalytic enhancement of NAD+/NADH ratio | Improved brain energy metabolism | REPAIR-MS Phase 2 | 12+weeks | 11 participants | 10.4% increase in NAD+/NADH ratio | Not reported | Target engagement confirmed | Phase 3 planned |
| Micromage-B Magnetite NPs [89] | Oral administration | Immunosuppression+remyelination activation | Neurological status improvement | Single case study | 6 mo | 1 patient | Total score reduction 210 → 45 | 6.0 → 5.0 (1-point improvement) | Reduced new demyelination foci | Proof-of-concept |
| MSN-MOG-CeNP Tolerogenic NPs | Intravenous injection | Antigen-specific immune tolerance | EAE disease suppression | Preclinical EAE model | Variable (acute-chronic) | Multiple mouse cohorts | Recovery from complete paralysis | Not applicable (animal model) | Reduced CNS inflammation | Preclinical |
| PLGA-MOG Nanoparticles | Intravenous/subcutaneous | Tolerogenic immune modulation | Prevention and treatment of EAE | Preclinical EAE model | Prophylactic and therapeutic | Multiple experimental groups | Significant clinical score improvement | EAE score reduction | Reduced brain infiltration | Preclinical development |
| Lignin-based Tolerogenic NPs [49] | Intravenous injection | ROS scavenging+antigen presentation | Durable immune tolerance | Preclinical EAE model | Early and late disease stages |
The challenge lies in developing standardized efficacy metrics that capture neuroprotective, remyelinating, and immunomodulatory effects simultaneously. Traditional EDSS scores may inadequately reflect subtle but clinically meaningful improvements in energy metabolism or immune tolerance induction.
Manufacturing and regulatory pathways
Clinical translation requires addressing manufacturing scalability, quality control, and regulatory compliance. CNM-Au8's progression to Phase 3 demonstrates feasibility of large-scale gold nanocrystal production under GMP conditions. However, more complex platforms like tolerogenic nanoparticles face challenges in maintaining antigen integrity and reproducible immune responses across manufacturing batches. Regulatory frameworks must evolve to accommodate combination products integrating nanotechnology with biologics. The FDA's growing experience with nanoparticle therapeutics provides precedent, but MS-specific guidance remains limited, particularly for immunomodulatory platforms [96].
Future therapeutic paradigms
Combination nanotherapeutics
Emerging evidence suggests synergistic potential between different nanoparticle mechanisms. Combining bioenergetic enhancement (CNM-Au8) with immune tolerance induction (tolerogenic nanoparticles) may address both metabolic dysfunction and autoimmune pathogenesis simultaneously. This dual-mechanism approach could potentially halt disease progression while promoting CNS repair.
Personalized nanomedicine approaches
The heterogeneity of MS pathophysiology necessitates personalized treatment strategies. Biomarker-guided selection of nanoparticle platforms based on individual patient profiles-metabolic dysfunction, immune phenotype, and lesion characteristics-represents the next frontier in MS nanotherapeutics [table 4]. Advanced imaging techniques, including 31P-MRS for metabolic assessment and specialized MRI sequences for immune cell tracking, will enable precision medicine approaches in nanoparticle selection and dosing optimization. Clinical Applications Summary: The evolution from preclinical promise to clinical reality in MS nanotherapeutics demonstrates both significant achievements and ongoing challenges. While platforms like CNM-Au8 have established clinical efficacy and safety, emerging tolerogenic approaches offer potential disease-modifying benefits. Success in clinical translation requires continued innovation in nanocarrier design, standardized efficacy metrics, and regulatory frameworks adapted to combination nanotechnology products.
Table 4: Clinical stages and safety profiles for multiple sclerosis therapy
| Platform | Clinical stage | Primary mechanism | Delivery route | Key outcome | Safety | Scalability |
| Cnm-au8 [88] | Phase 2/3 | Bioenergetics | Oral | Energy metabolism↑ | Excellent | High |
| Micromage-b | Proof-of-concept | Immunomodulation | Oral | Edss improvement | Good | Moderate |
| Tolerogenic nps [49] | Preclinical | Immune tolerance | Intravenous | Disease suppression | Good | High |
CONCLUSION
Nanotechnology-based therapeutics have catalyzed a paradigm shift in MS treatment through direct BBB penetration and targeted CNS delivery. CNM-Au8 gold nanocrystals demonstrated clinical efficacy in phase 2 VISIONARY-MS trials with sustained visual and neurological improvements over multi-year extensions, supported by 10.4% increases in brain NAD⁺/NADH ratio. Micromage-B magnetite nanoparticles achieved marked EDSS score reductions (from 6.0 to 5.0 over 6 mo) and MRI-documented demyelination focus reduction (by month 4). Preclinical tolerogenic nanoparticles and anti-NG2-targeted PLGA systems show promise for antigen-specific immune tolerance and oligodendrocyte precursor cell targeting. Critical challenges remain: manufacturing scalability under good manufacturing practice, long-term biodistribution and CNS accumulation data, and regulatory frameworks accommodating combination products. Over five to ten years, completion of CNM-Au8 Phase 3 trials and first-in-human tolerogenic nanoparticle studies represent essential milestones toward personalized nanomedicine in MS, integrating metabolic, immunological, and lesion-specific biomarkers to optimize therapeutic outcomes and preserve neurological function.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
Nadeesh T: Literature review, Data curation, Writing-original draft, Evaluation; Subhrajyoti Dhara: Literature review, Data curation, and Writing-original draft; Priyam Manna: Writing-original draft, Conceptualization, Critical Evaluation; Pritam Kayal: Review and editing, Supervision, Evaluation, Visualization.
CONFLICTS OF INTERESTS
The authors declare no conflict of interest
REFERENCES
Mohammed EM. Understanding multiple sclerosis pathophysiology and current disease-modifying therapies: a review of unaddressed aspects. Front Biosci (Landmark Ed). 2024 Nov 19;29(11):386. doi: 10.31083/j.fbl2911386, PMID 39614433.
Oh J, Bar Or A. Emerging therapies to target CNS pathophysiology in multiple sclerosis. Nat Rev Neurol. 2022;18(8):466-75. doi: 10.1038/s41582-022-00675-0, PMID 35697862.
Lorenzut S, Negro ID, Pauletto G, Verriello L, Spadea L, Salati C. Exploring the pathophysiology, diagnosis and treatment options of multiple sclerosis. J Integr Neurosci. 2025;24(1):25081. doi: 10.31083/JIN25081, PMID 39862004.
Yong HY, Yong VW. Mechanism based criteria to improve therapeutic outcomes in progressive multiple sclerosis. Nat Rev Neurol. 2022;18(1):40-55. doi: 10.1038/s41582-021-00581-x, PMID 34732831.
Haki M, Al Biati HA, Al Tameemi ZS, Ali IS, Al Hussaniy HA. Review of multiple sclerosis: epidemiology etiology pathophysiology and treatment. Medicine. 2024;103(8):e37297. doi: 10.1097/MD.0000000000037297, PMID 38394496.
Balasa R, Barcutean L, Mosora O, Manu D. Reviewing the significance of blood–brain barrier disruption in multiple sclerosis pathology and treatment. Int J Mol Sci. 2021;22(16):8370. doi: 10.3390/ijms22168370, PMID 34445097.
Piehl F. Current and emerging disease modulatory therapies and treatment targets for multiple sclerosis. J Intern Med. 2021 Jun;289(6):771-91. doi: 10.1111/joim.13215, PMID 33258193.
Panghal A, Flora SJ. Nano-based approaches for the treatment of neuro-immunological disorders: a special emphasis on multiple sclerosis. Discov Nano. 2024 Oct 28;19(1):171. doi: 10.1186/s11671-024-04135-0, PMID 39466516.
Damavandi AR, Mirmosayyeb O, Ebrahimi N, Zalpoor H, Khalilian P, Yahiazadeh S. Advances in nanotechnology versus stem cell therapy for the theranostics of multiple sclerosis disease. Appl Nanosci. 2023 Jun;13(6):4043-73. doi: 10.1007/s13204-022-02698-x.
Jan Z, Mollazadeh S, Abnous K, Taghdisi SM, Danesh A, Ramezani M. Targeted delivery platforms for the treatment of multiple sclerosis. Mol Pharm. 2022 Jul 4;19(7):1952-76. doi: 10.1021/acs.molpharmaceut.1c00892, PMID 35501974.
Tapeinos C, Battaglini M, Ciofani G. Advances in the design of solid lipid nanoparticles and nanostructured lipid carriers for targeting brain diseases. J Control Release. 2017;264:306-32. doi: 10.1016/j.jconrel.2017.08.033, PMID 28844756.
Mohapatra R, Giri D, Vijayakumar D, Alagendran S, Saavedra DG, Jena JP. Nanomaterials in biomedical applications: a review. J Adv Biol Biotechnol. 2025 Apr 5;28(3):996-1009. doi: 10.9734/jabb/2025/v28i32157.
Thi TT, Suys EJ, Lee JS, Nguyen DH, Park KD, Truong NP. Lipid-based nanoparticles in the clinic and clinical trials: from cancer nanomedicine to COVID-19 vaccines. Vaccines. 2021 Apr 8;9(4):359. doi: 10.3390/vaccines9040359, PMID 33918072.
Wang Y, Li P, Kong L. Chitosan modified PLGA nanoparticles with versatile surface for improved drug delivery. AAPS PharmSciTech. 2013 Jun;14(2):585-92. doi: 10.1208/s12249-013-9943-3, PMID 23463262.
Burlec AF, Corciova A, Boev M, Batir Marin D, Mircea C, Cioanca O. Current overview of metal nanoparticles synthesis, characterization and biomedical applications with a focus on silver and gold nanoparticles. Pharmaceuticals (Basel). 2023 Oct 4;16(10):1410. doi: 10.3390/ph16101410, PMID 37895881.
Dhiman N, Awasthi R, Sharma B, Kharkwal H, Kulkarni GT. Lipid nanoparticles as carriers for bioactive delivery. Front Chem. 2021 Apr 23;9:580118. doi: 10.3389/fchem.2021.580118, PMID 33981670.
Belousov AN. Efficiency of nanoparticles (micromage-B) in the complex treatment of multiple sclerosis. Clin Res Clin Rep. 2023;2(2):1-8. doi: 10.31579/2835-8325/010.
Martinelli C, Pucci C, Battaglini M, Marino A, Ciofani G. Antioxidants and nanotechnology: promises and limits of potentially disruptive approaches in the treatment of central nervous system diseases. Adv Healthc Mater. 2020 Feb;9(3):e1901589. doi: 10.1002/adhm.201901589, PMID 31854132.
Jimenez A, Estudillo E, Guzman Ruiz MA, Herrera Mundo N, Victoria Acosta G, Cortes Malagon EM. Nanotechnology to overcome blood brain barrier permeability and damage in neurodegenerative diseases. Pharmaceutics. 2025 Feb 20;17(3):281. doi: 10.3390/pharmaceutics17030281, PMID 40142945.
Wu D, Chen Q, Chen X, Han F, Chen Z, Wang Y. The blood-brain barrier: structure, regulation and drug delivery. Signal Transduct Target Ther. 2023 May 25;8(1):217. doi: 10.1038/s41392-023-01481-w, PMID 37231000.
Gonzalez Lorenzo M, Ridley B, Minozzi S, Del Giovane C, Peryer G, Piggott T. Immunomodulators and immunosuppressants for relapsing remitting multiple sclerosis: a network meta-analysis. Cochrane Database Syst Rev. 2024;1(1):CD011381. doi: 10.1002/14651858.CD011381.pub3, PMID 38174776.
Luo Q, Yang J, Yang M, Wang Y, Liu Y, Liu J. Utilization of nanotechnology to surmount the blood brain barrier in disorders of the central nervous system. Mater Today Bio. 2025 Jan 4;31:101457. doi: 10.1016/j.mtbio.2025.101457, PMID 39896289.
Liu J, Wang T, Dong J, Lu Y. The blood brain barriers: novel nanocarriers for central nervous system diseases. J Nanobiotechnology. 2025 Feb 26;23(1):146. doi: 10.1186/s12951-025-03247-8, PMID 40011926.
Xie J, Shen Z, Anraku Y, Kataoka K, Chen X. Nanomaterial-based blood brain-barrier (BBB) crossing strategies. Biomaterials. 2019 Dec 1;224:119491. doi: 10.1016/j.biomaterials.2019.119491, PMID 31546096.
Han L. Modulation of the blood brain barrier for drug delivery to brain. Pharmaceutics. 2021;13(12):2024. doi: 10.3390/pharmaceutics13122024, PMID 34959306.
Bai X, Smith ZL, Wang Y, Butterworth S, Tirella A. Sustained drug release from smart nanoparticles in cancer therapy: a comprehensive review. Micromachines. 2022 Sep 28;13(10):1623. doi: 10.3390/mi13101623, PMID 36295976.
Sharma A, Sharma N, Singh S, Dua K. Review on theranostic and neuroprotective applications of nanotechnology in multiple sclerosis. J Drug Deliv Sci Technol. 2023 Mar 1;81:104220. doi: 10.1016/j.jddst.2023.104220.
Ojha S, Kumar B. A review on nanotechnology based innovations in diagnosis and treatment of multiple sclerosis. J Cell Immunother. 2018 Dec 1;4(2):56-64. doi: 10.1016/j.jocit.2017.12.001.
Gharagozloo M, Bannon R, Calabresi PA. Breaking the barriers to remyelination in multiple sclerosis. Curr Opin Pharmacol. 2022 Apr 1;63:102194. doi: 10.1016/j.coph.2022.102194, PMID 35255453.
Maheshwari S, Verma A, Singh A, Wasim R, Shariq M, Akhtar J. Emergence of nanomaterials in the management of multiple sclerosis. In: Ansari MM, Suresh AK, Akhtar N, editors. Emergence of sustainable biomaterials in tackling inflammatory diseases Smart. Singapore: Springer; 2025. p. 323-40. doi: 10.1007/978-981-96-2112-5_12.
Takei EK. The use of nanoparticles in the diagnosis and treatment of multiple sclerosis: a scoping review. STEM Fellowship J. 2024 May 23;10(1):59-73. doi: 10.17975/sfj-2024-005.
Nuzzo D, Picone P. Multiple sclerosis: focus on extracellular and artificial vesicles nanoparticles as potential therapeutic approaches. Int J Mol Sci. 2021;22(16):8866. doi: 10.3390/ijms22168866, PMID 34445572.
Bilorosiuk M, Steinman L, Koppisetti S, Hariri R, Leibovitch EC, Jacobson S. Nanomedicine in demyelinating disease application to diagnosis and therapy in multiple sclerosis. In: Kateb B, Heiss JD, Yu JS, Hsieh M, editors. The Textbook of Nanoneuroscience and Nanoneurosurgery. Berlin: Springer Nature; 2024 Nov 14. p. 477-96. doi: 10.1007/978-3-030-80662-0_29.
Tarannum S, Jain K. Drug delivery strategies in multiple sclerosis huntington’s disease and other neurodegenerative diseases. In: Mishra A, Kulhari H, editors. Drug delivery strategies in neurological disorders: challenges and opportunities. Singapore: Springer; 2023. p. 375-403. doi: 10.1007/978-981-99-6807-7_16.
El Say KM, El Sawy HS. Polymeric nanoparticles: promising platform for drug delivery. Int J Pharm. 2017 Aug 7;528(1-2):675-91. doi: 10.1016/j.ijpharm.2017.06.052, PMID 28629982.
Osorio Querejeta I, Carregal Romero S, Ayerdi Izquierdo A, Mager I, A NL, Wood M. MiR-219a-5p enriched extracellular vesicles induce OPC differentiation and EAE improvement more efficiently than liposomes and polymeric nanoparticles. Pharmaceutics. 2020 Feb 21;12(2):186. doi: 10.3390/pharmaceutics12020186, PMID 32098213.
Osorio Querejeta I, Carregal Romero S, Ayerdi Izquierdo A, Mager I, A NL, Wood M. MiR-219a-5p enriched extracellular vesicles induce OPC differentiation and EAE improvement more efficiently than liposomes and polymeric nanoparticles. Pharmaceutics. 2020;12(2):186. doi: 10.3390/pharmaceutics12020186, PMID 32098213.
Haider M, Abdin SM, Kamal L, Orive G. Nanostructured lipid carriers for delivery of chemotherapeutics: a review. Pharmaceutics. 2020;12(3):288. doi: 10.3390/pharmaceutics12030288, PMID 32210127.
Cappellano G, Comi C, Chiocchetti A, Dianzani U. Exploiting PLGA-based biocompatible nanoparticles for next-generation tolerogenic vaccines against autoimmune disease. Int J Mol Sci. 2019;20(1):204. doi: 10.3390/ijms20010204, PMID 30626016.
Manna S, Banerjee S, De A, Banerjee S, Das S, Rakshit P. Therapeutic and diagnostic implications of carbon dot: an advancement in the avenue towards cancer diabetes and neurodegenerative disorders. Pharm Nanotechnol. 2024 Sep 27. doi: 10.2174/0122117385314533240824090949, PMID 39350419.
Zierfuss B, Larochelle C, Prat A. Blood brain barrier dysfunction in multiple sclerosis: causes consequences and potential effects of therapies. Lancet Neurol. 2024 Jan 1;23(1):95-109. doi: 10.1016/S1474-4422(23)00377-0, PMID 38101906.
Amoriello R, Memo C, Ballerini L, Ballerini C. The brain cytokine orchestra in multiple sclerosis: from neuroinflammation to synaptopathology. Mol Brain. 2024;17(1):4. doi: 10.1186/s13041-024-01077-7, PMID 38263055.
Zhang S, Yang Y, LV X, Zhou X, Zhao W, Meng L. Exosome cargo in neurodegenerative diseases: leveraging their intercellular communication capabilities for biomarker discovery and therapeutic delivery. Brain Sci. 2024 Oct 23;14(11):1049. doi: 10.3390/brainsci14111049, PMID 39595812.
Kulkarni M, Patel K, Patel A, Patel S, Desai J, Patel M. Nanomaterials as drug delivery agents for overcoming the blood–brain barrier: a comprehensive review. ADMET DMPK. 2024 Feb 13;12(1):63-105. doi: 10.5599/admet.2043, PMID 38560713.
Zhang S, Yang Y, LV X, Zhou X, Zhao W, Meng L. Exosome cargo in neurodegenerative diseases: leveraging their intercellular communication capabilities for biomarker discovery and therapeutic delivery. Brain Sci. 2024 Oct 23;14(11):1049. doi: 10.3390/brainsci14111049, PMID 39595812.
Mahmoudi M, Sahraian MA, Shokrgozar MA, Laurent S. Superparamagnetic iron oxide nanoparticles: promises for diagnosis and treatment of multiple sclerosis. ACS Chem Neurosci. 2011 Mar 16;2(3):118-40. doi: 10.1021/cn100100e, PMID 22778862.
Rocca MA, Preziosa P, Barkhof F, Brownlee W, Calabrese M, De Stefano N. Current and future role of MRI in the diagnosis and prognosis of multiple sclerosis. Lancet Reg Health Eur. 2024 Sep 1;44:100978. doi: 10.1016/j.lanepe.2024.100978, PMID 39444702.
Zhang R, Kiessling F, Lammers T, Pallares RM. Clinical translation of gold nanoparticles. Drug Deliv Transl Res. 2023;13(2):378-85. doi: 10.1007/s13346-022-01232-4, PMID 36045273.
Phan NM, Nguyen TL, Shin H, Trinh TA, Kim J. ROS-scavenging lignin-based tolerogenic nanoparticle vaccine for treatment of multiple sclerosis. ACS Nano. 2023 Dec 5;17(24):24696-709. doi: 10.1021/acsnano.3c04497, PMID 38051295.
Collorone S, Coll L, Lorenzi M, Llado X, Sastre Garriga J, Tintore M. Artificial intelligence applied to MRI data to tackle key challenges in multiple sclerosis. Mult Scler. 2024 Jun;30(7):767-84. doi: 10.1177/13524585241249422, PMID 38738527.
Ghosh S, Bhatti GK, Sharma PK, Kandimalla R, Mastana SS, Bhatti JS. Potential of nano-engineered stem cells in the treatment of multiple sclerosis: a comprehensive review. Cell Mol Neurobiol. 2023;44(1):6. doi: 10.1007/s10571-023-01434-5, PMID 38104307.
Al Sayadi GM, Verma A, Choudhary Y, Sandal P, Patel P, Singh D. Solid lipid nanoparticles (SLNs): advancements in modification strategies toward drug delivery vehicle. Pharm Nanotechnol. 2023 Apr 1;11(2):138-54. doi: 10.2174/2211738511666221026163303, PMID 36305142.
Greco G, Sarpietro MG. Liposome assisted drug delivery in the treatment of multiple sclerosis. Molecules. 2024 Oct 3;29(19):4689. doi: 10.3390/molecules29194689, PMID 39407617.
Juhairiyah F, De Lange EC. Understanding drug delivery to the brain using liposome-based strategies: studies that provide mechanistic insights are essential. AAPS J. 2021 Oct 28;23(6):114. doi: 10.1208/s12248-021-00648-z, PMID 34713363.
Zhao Y, Zhang J, Cheng X, Huang W, Shen S, Wu S. Targeting L-selectin lymphocytes to deliver immunosuppressive drug in lymph nodes for durable multiple sclerosis treatment. Adv Sci (Weinh). 2023 Jul;10(20):e2300738. doi: 10.1002/advs.202300738, PMID 37170724, PMCID PMC10369270.
Satapathy MK, Yen TL, Jan JS, Tang RD, Wang JY, Taliyan R. Solid lipid nanoparticles (SLNs): an advanced drug delivery system targeting brain through BBB. Pharmaceutics. 2021 Jul 31;13(8):1183. doi: 10.3390/pharmaceutics13081183, PMID 34452143.
Bove RM, Green AJ. Remyelinating pharmacotherapies in multiple sclerosis. Neurotherapeutics. 2017 Oct 1;14(4):894-904. doi: 10.1007/s13311-017-0577-0, PMID 28948533.
Li PY, Bearoff F, Zhu P, Fan Z, Zhu Y, Fan M. PEGylation enables subcutaneously administered nanoparticles to induce antigen-specific immune tolerance. J Control Release. 2021 Mar 10;331:164-75. doi: 10.1016/j.jconrel.2021.01.013, PMID 33450320.
Agrawal M, Saraf S, Saraf S, Dubey SK, Puri A, Patel RJ. Recent strategies and advances in the fabrication of nano lipid carriers and their application towards brain targeting. J Control Release. 2020;321:372-415. doi: 10.1016/j.jconrel.2020.02.020, PMID 32061621.
Jiao Y, Yang L, Wang R, Song G, Fu J, Wang J. Drug delivery across the blood–brain barrier: a new strategy for the treatment of neurological diseases. Pharmaceutics. 2024 Dec 19;16(12):1611. doi: 10.3390/pharmaceutics16121611, PMID 39771589.
Niu X, Chen J, Gao J. Nanocarriers as a powerful vehicle to overcome blood–brain barrier in treating neurodegenerative diseases: focus on recent advances. Asian J Pharm Sci. 2019 Sep 1;14(5):480-96. doi: 10.1016/j.ajps.2018.09.005, PMID 32104476.
Bonferoni MC, Rossi S, Sandri G, Ferrari F, Gavini E, Rassu G. Nanoemulsions for “nose-to-brain” drug delivery. Pharmaceutics. 2019 Feb 17;11(2):84. doi: 10.3390/pharmaceutics11020084, PMID 30781585.
Kaye AD, Sala KR, Dethloff D, Norton M, Moss C, Plessala MJ. The evolving use of gold nanoparticles as a possible reversal agent for the symptoms of neurodegenerative diseases: a narrative review. Cureus. 2024 Jul 18;16(7):e64846. doi: 10.7759/cureus.64846, PMID 39156432.
Li Z, Bai R, Yi J, Zhou H, Xian J, Chen C. Designing smart iron oxide nanoparticles for MR imaging of tumors. Chem Biomed Imaging. 2023 Jul 24;1(4):315-39. doi: 10.1021/cbmi.3c00026, PMID 37501794.
Xu L, Wang YY, Huang J, Chen CY, Wang ZX, Xie H. Silver nanoparticles: synthesis medical applications and biosafety. Theranostics. 2020;10(20):8996-9031. doi: 10.7150/thno.45413, PMID 32802176.
Dehkordi AA, Mollazadeh S, Talaie A, Yazdimamaghani M. Engineering PAMAM dendrimers for optimized drug delivery. Nano Trends. 2025 Feb 16;9:100094. doi: 10.1016/j.nwnano.2025.100094.
Hayder M, Garzoni M, Bochicchio D, Caminade AM, Couderc F, Ong Meang V. Three dimensional directionality is a pivotal structural feature for the bioactivity of azabisphosphonate capped poly(PhosphorHydrazone) nanodrug dendrimers. Biomacromolecules. 2018 Feb 14;19(3):712-20. doi: 10.1021/acs.biomac.7b01398, PMID 29443507.
Garate Velez L, Quintana M. Carbon based nanomaterials: interactions with cells brain therapies and neural sensing. J Mater Sci: Mater Eng. 2025 Dec;20(1):1-23. doi: 10.1186/s40712-025-00236-5.
Haqqani AS, Belanger K, Stanimirovic DB. Receptor mediated transcytosis for brain delivery of therapeutics: receptor classes and criteria. Front Drug Deliv. 2024 Mar 12;4:1360302. doi: 10.3389/fddev.2024.1360302, PMID 40836978.
Zhang W, Liu QY, Haqqani AS, Leclerc S, Liu Z, Fauteux F. Differential expression of receptors mediating receptor-mediated transcytosis (RMT) in brain microvessels brain parenchyma and peripheral tissues of the mouse and the human. Fluids Barriers CNS. 2020 Jul 22;17(1):47. doi: 10.1186/s12987-020-00209-0, PMID 32698806.
Banks WA, Rhea EM, Reed MJ, Erickson MA. The penetration of therapeutics across the blood–brain barrier: classic case studies and clinical implications. Cell Rep Med. 2024 Nov 19;5(11):101760. doi: 10.1016/j.xcrm.2024.101760, PMID 39383873.
Pulgar VM. Transcytosis to cross the blood brain barrier new advancements and challenges. Front Neurosci. 2019 Jan 11;12:1019. doi: 10.3389/fnins.2018.01019, PMID 30686985.
Schreiner TG, Romanescu C, Popescu BO. The blood–brain barrier a key player in multiple sclerosis disease mechanisms. Biomolecules. 2022 Apr 2;12(4):538. doi: 10.3390/biom12040538, PMID 35454127.
Yang G, Van Kaer L. Therapeutic targeting of immune cell autophagy in multiple sclerosis: Russian roulette or silver bullet? Front Immunol. 2021 Aug 31;12:724108. doi: 10.3389/fimmu.2021.724108, PMID 34531871.
Mado H, Adamczyk Sowa M, Sowa P. Role of microglial cells in the pathophysiology of MS: synergistic or antagonistic? Int J Mol Sci. 2023 Jan 17;24(3):1861. doi: 10.3390/ijms24031861, PMID 36768183.
Vermersch P, Airas L, Berger T, Deisenhammer F, Grigoriadis N, Hartung HP. The role of microglia in multiple sclerosis: implications for treatment with Bruton’s tyrosine kinase inhibitors. Front Immunol. 2025;16:1495529. doi: 10.3389/fimmu.2025.1495529, PMID 40443664.
Tepavcevic V, Lubetzki C. Oligodendrocyte progenitor cell recruitment and remyelination in multiple sclerosis: the more the merrier? Brain. 2022;145(12):4178-92. doi: 10.1093/brain/awac307, PMID 36093726.
Derdelinckx J, Cras P, Berneman ZN, Cools N. Antigen-specific treatment modalities in MS: the past the present and the future. Front Immunol. 2021;12:624685. doi: 10.3389/fimmu.2021.624685, PMID 33679769.
Nally FK, De Santi C, McCoy CE. Nanomodulation of macrophages in multiple sclerosis. Cells. 2019 Jun 5;8(6):543. doi: 10.3390/cells8060543, PMID 31195710.
Rittchen S, Boyd A, Burns A, Park J, Fahmy TM, Metcalfe S. Myelin repair in vivo is increased by targeting oligodendrocyte precursor cells with nanoparticles encapsulating leukaemia inhibitory factor (LIF). Biomaterials. 2015 Jul 1;56:78-85. doi: 10.1016/j.biomaterials.2015.03.044, PMID 25934281.
Zveik O, Rechtman A, Ganz T, Vaknin Dembinsky A. The interplay of inflammation and remyelination: rethinking MS treatment with a focus on oligodendrocyte progenitor cells. Mol Neurodegener. 2024 Jul 12;19(1):53. doi: 10.1186/s13024-024-00742-8, PMID 38997755.
Pulgar VM. Transcytosis to cross the blood brain barrier new advancements and challenges. Front Neurosci. 2019;12:1019. doi: 10.3389/fnins.2018.01019, PMID 30686985.
Horwitz DA, Bickerton S, Koss M, Fahmy TM, La Cava A. Suppression of murine lupus by CD4+ and CD8+ Treg Cells induced by T cell-targeted nanoparticles loaded with interleukin-2 and transforming growth factor β. Arthritis Rheumatol. 2019 Apr;71(4):632-40. doi: 10.1002/art.40773, PMID 30407752.
Horwitz DA, Bickerton S, La Cava A. Strategies to use nanoparticles to generate CD4 and CD8 regulatory T cells for the treatment of SLE and other autoimmune diseases. Front Immunol. 2021;12:681062. doi: 10.3389/fimmu.2021.681062, PMID 34211471.
Horwitz DA, Bickerton S, Koss M, Fahmy TM, La Cava A. Suppression of murine lupus by CD4+ and CD8+ Treg cells induced by T cell-targeted nanoparticles loaded with interleukin-2 and transforming growth factor β. Arthritis Rheumatol. 2019 Apr;71(4):632-40. doi: 10.1002/art.40773, PMID 30407752.
Chountoulesi M, Demetzos C. Promising nanotechnology approaches in treatment of autoimmune diseases of central nervous system. Brain Sci. 2020 Jun 2;10(6):338. doi: 10.3390/brainsci10060338, PMID 32498357.
Robinson AP, Zhang JZ, Titus HE, Karl M, Merzliakov M, Dorfman AR. Nanocatalytic activity of clean-surfaced faceted nanocrystalline gold enhances remyelination in animal models of multiple sclerosis. Sci Rep. 2020;10(1):1936. doi: 10.1038/s41598-020-58709-w, PMID 32041968.
Vucic S, Menon P, Huynh W, Mahoney C, Ho KS, Hartford A. Efficacy and safety of CNM-Au8 in amyotrophic lateral sclerosis (Rescue-ALS study): a phase 2, randomised double-blind placebo-controlled trial and open label extension. E Clinical Medicine. 2023 Jun 1;60:102036. doi: 10.1016/j.eclinm.2023.102036, PMID 37396808.
Dong S. Targeted magnetic iron oxide nanoparticles for tumor imaging and therapy. Int J Nanomedicine. 2008;3(3):311-21. doi: 10.2147/IJN.S2824.
La Mothe RA, Kolte PN, Vo T, Ferrari JD, Gelsinger TC, Wong J. Tolerogenic nanoparticles induce antigen-specific regulatory T cells and provide therapeutic efficacy and transferrable tolerance against experimental autoimmune encephalomyelitis. Front Immunol. 2018 Mar 2;9:281. doi: 10.3389/fimmu.2018.00281, PMID 29552007.
Park DJ, Yun WS, Kim WC, Park JE, Lee SH, Ha S. Improvement of stem cell-derived exosome release efficiency by surface-modified nanoparticles. J Nanobiotechnology. 2020 Dec 7;18(1):178. doi: 10.1186/s12951-020-00739-7, PMID 33287848.
Kenison JE, Stevens NA, Quintana FJ. Therapeutic induction of antigen-specific immune tolerance. Nat Rev Immunol. 2024 May;24(5):338-57. doi: 10.1038/s41577-023-00970-x, PMID 38086932.
Gholamzad M, Baharlooi H, Shafiee Ardestani MS, Seyedkhan Z, Azimi M. Prophylactic and therapeutic effects of MOG-conjugated PLGA nanoparticles in C57BL/6 mouse model of multiple sclerosis. Adv Pharm Bull. 2020 Jul 26;11(3):505-13. doi: 10.34172/apb.2021.058, PMID 34513625.
Tan F, Li X, Wang Z, Li J, Shahzad K, Zheng J. Clinical applications of stem cell-derived exosomes. Signal Transduct Target Ther. 2024 Jan 12;9(1):17. doi: 10.1038/s41392-023-01704-0, PMID 38212307.
Belousov AN, Belousova EY, Mysyk AV. A clinical case of the successful application of magnetite nanoparticles in the complex treatment of multiple sclerosis. J Recent Adv Nanomed & Nanotech. 2025;1(3):555565. doi: 10.19080/JRANN.2024.01.555566.