
Int J App Pharm, Vol 18, Issue 1, 2026, 187-192Original Article
NANOPARTICLE PREPARATION OF SNAKEHEAD FISH EXTRACT (CHANNA STRIATA) BY IONIC GELATION METHOD USING SODIUM ALGINATE AS POLYMER
WINTARI TAURINA, MOHAMAD ANDRIE*
Departement of Pharmacy, Faculty of Medicine, Tanjungpura University, Pontianak, West Kalimantan, Indonesia
*Corresponding author: Mohamad Andrie; *Email: andrie@pharm.untan.ac.id
Received: 24 Aug 2025, Revised and Accepted: 24 Nov 2025
ABSTRACT
Objective: The extract from the snakehead fish (Channa striata) is a rich source of albumin, a protein recognized for its wound healing properties. In this study, we developed albumin-loaded nanoparticles using sodium alginate as the polymer matrix. We also examined how varying sonication durations affected the characteristics of the nanoparticles.
Methods: Nanoparticles were prepared using ionic gelation using 0.1% sodium alginate and 0.01% calcium chloride (CaCl₂) as a cross-linker, stirred for 2 h at 1500 rpm, and sonicated for 1 h (Formula 1) or 2 h (Formula 2) using 45Hz frequency. After freeze-drying, characterization included visual appearance, %transmittance, particle size, and polydispersity index. Scanning electron microscope (SEM) and entrapment efficiency tests further evaluated the optimal formula.
Results: There were notable differences in the characteristics of the nanoparticles between F1 and F2. F1 exhibited an average transmittance of 93.22%, a particle size of 606.43 nm, and a polydispersity index of 0.563. In contrast, F2 demonstrated a higher transmittance of 99.41%, a smaller particle size of 483.26 nm, and a slightly lower polydispersity index of 0.558. Statistical analysis revealed a significant difference (p<0.05) between the two formulations. F2 was identified as the optimal formulation, achieving an entrapment efficiency of 96.63%, a zeta potential of-24.6 mV, and exhibiting a wrinkled particle morphology with aggregates visible at 10.000x magnification. The optimal ultrasonication time depends on how effectively the process dissolves and distributes the ingredients evenly, as longer mixing reduces the particle size.
Conclusion: Snakehead fish extract nanoparticles prepared with 0.1% sodium alginate and 2 h of sonication (F2) showed the best characteristics. After 7 days, the solution remained slightly cloudy with a few floating particles and a distinctive fish odor. The average %transmittance was 99.406%, particle size 483.266 nm, polydispersity index 0.558, zeta potential-24.6±0.294 mV, and entrapment efficiency 96.63±0.11%. SEM at 5000x and 10.000x magnification revealed mostly spherical, dense particles with rough surfaces.
Keywords: Snakehead fish extract, Nanoparticles, Ionic gelation, Sodium alginate
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.56622 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Fish is a major marine resource widely used as a source of animal protein. Snakehead fish contains higher protein levels than many other species, with 25.5% protein, along with albumin, omega-3, and omega-6 fatty acids that support wound healing [1, 2]. Its albumin aids tissue repair and offers a cost-effective alternative protein source [2]. However, the 30 μm particle size and sensitivity of albumin to temperature and pH can lead to denaturation and reduce its effectiveness [3]. The use of nanoparticle technology continues to increase due to its advantages, including enhanced bioavailability, improved solubility, larger particle surface area, and greater stability of active compounds. The large particle size of snakehead fish extract makes it difficult to penetrate the stratum corneum, thereby limiting its ability to pass through cell membranes. Converting the extract into nanoparticle form facilitates drug penetration through the stratum corneum. The encapsulation of albumin within nanoparticles protects the protein by embedding it in the polymer matrix, thereby preventing rapid degradation. To overcome these limitations, albumin nanoparticles from snakehead fish extract can be prepared using the ionic gelation method with sodium alginate as the polymer.
Nanoparticles are colloidal systems ranging in size from 10 to 1000 nm, offering benefits such as enhanced bioavailability, solubility, surface area, diffusion, penetration, and stability of active compounds [4, 5]. Ionic gelation, which involves cross-linking between polyelectrolytes and multivalent ions, strengthens the particle structure mechanically [6]. Due to their unique properties, nanoparticles can overcome the limitations of conventional therapies and are efficiently taken up by cells, making them promising delivery systems [7, 8]. The duration of sonication is a critical parameter in the formulation of nanoparticles. By using ultrasonic waves, sonication enhances the contact between the sample and solvent at room temperature, leading to the formation of particles with nanometer-scale diameters. Sodium alginate was selected for its ability to prolong contact time, enhance penetration, and increase efficacy. Snakehead fish extract nanoparticles were formulated via ionic gelation using positively charged CaCl2 and subsequently freeze-dried to produce a powder, aiming to improve stability, prolong shelf life, and protect heat-sensitive components.
This research aimed to formulate albumin nanoparticles from snakehead fish extract using the ionic gelation method with sodium alginate as a polymer. The study examined the physical and chemical characteristics of powder nanoparticles from snakehead fish extract, including visual appearance, percentage transmittance, particle size, zeta potential, particle morphology, and percentage entrapment efficiency.
MATERIALS AND METHODS
Materials
Snakehead fish (Channa striata) was sourced from a fisherman in Sungai Itik village, Kuburaya Regency, West Kalimantan, Indonesia. Sodium alginate (Eurofins), and calcium chloride (Eurofins).
Instruments
Scanning electron microscopy (SEM) (SU3500, America), particle size analyzer (PSA) and zeta potential analyzer (ZPA) (SZ-HORIBA 100, Japan), Spectrophotometer UV-Vis (Shimadzu UV 2600i, Japan), Photometer (Microlab 300, Netherland)
Collection and extraction of water phase snakehead fish extract
Snakehead fish (Channa striata) was determined at the Biology Laboratory, Department of Biology, Faculty of Mathematics and Natural Sciences, Tanjungpura University, Pontianak, West Kalimantan, with number 0164/A/lB/FMIPA/UNTAN/2024. The Snakehead fish used have weights ranging from 500 to 1000 s, obtained from fishermen in Itik River Village, Kuburaya Regency, West Kalimantan, Indonesia. The selection of snakehead fish was carried out by the Indonesian National Standard (SNI) No. 2729-2021. Snakehead fish includes a whole body with intact scales, free from parasites, without defects, and with a bright and shiny appearance. Afterward, we conduct a wet sorting process. The scales, fins, tail, and intestines are cleaned. Following that, the snakehead fish was washed using running water. The snakehead fish is chopped into several pieces to facilitate the extraction of the water phase. The snakehead fish meat is steamed at 70 °C for 30 min. The meat is wrapped in cloth and pressed with a hydraulic press to extract the essence of snakehead fish. Afterward, the extract was placed into a test tube and centrifuged at 1000 rpm for 60 minute, applying a relative centrifugal force (RCF) of approximately 11,180 × g for 30 min. The water phase of snakehead fish extract was taken, and preservatives DMDM hydantoin and parabens (methyl paraben and propyl paraben) were added.
Preparation Nanoparticle of water phase snakehead fish extract
Nanoparticles were initially prepared on a medium scale (60 ml) to determine the most optimal preparation method. Two formulations of alginate nanoparticles underwent sonication for different durations (1 h and 2 h) during preparation using 45Hz frequency. The sonication was performed at a power output of 200 W and 50% amplitude in pulse mode (5 sec on/5 sec off) for a total duration of 10 min. The sample volume was 20 ml, placed in a 50 ml glass beaker immersed in an ice bath to maintain the temperature below 30 °C. The total energy delivered was estimated at 120 kJ, equivalent to 6 kJ/ml. The volume ratio of the aqueous phases-snakehead fish extract, sodium alginate, and CaCl₂-was 2:1:1, respectively. The snakehead fish extract did not require dissolution, as it was already in liquid form. CaCl₂ was dissolved in distilled water, producing a transparent, clear solution. Similarly, the sodium alginate solution was prepared by dissolving sodium alginate in distilled water, yielding a transparent and colorless solution. Specifically, 15 g of fish extract was weighed, and 30 g of a 0.1% sodium alginate solution was placed into a 250 ml beaker. The extract was gradually added to the alginate solution, which was continuously stirred at 1500 rpm for 30 min. Subsequently, 15 g of a 0.01% CaCl₂ solution was added dropwise, and stirring was maintained for an additional 1.5 h at the same speed. The mixture was then subjected to sonication for either 1 or 2 h, depending on the formulation [9]. Additionally, the aqueous-phase nanoparticles containing snakehead fish extract were dried into powder form for further characterization. Observations were made on several physical parameters-including the presence of floating particles, sedimentation, solution color, sediment characteristics, particle shape, and turbidity-to determine the optimal formulation for subsequent evaluation.
Evaluation of physical characteristic nanoparticles
The nanoparticles from both formulations were subjected to physical characterization, including visual observation, % transmittance, particle size, and polydispersity index. The size, distribution, and zeta potential of the nanoparticles were determined using a particle size analyzer (psa), while transmittance was measured using UV/Vis spectrophotometry [13]. The optimum nanoparticle formulation was further analyzed using a SEM and tested for its zeta potential. Nanoparticle morphology was also assessed using SEM. This study presents a preliminary investigation into the ultrasonic-assisted preparation of Channa striata protein nanoparticles. Although the initial results are promising, the small sample size (n = 3) limits the statistical power and generalizability of the results. Future research involving larger sample sizes is essential to confirm the trends observed and support broader application of the method.
Evaluation of chemical characteristic nanoparticles
Entrapment efficiency of optimal formulation
The entrapment efficiency was determined by measuring the concentration of free albumin in the optimum formulation of the snakehead fish extract nanoparticles. Photometry using the bromocresol blue reagent was employed for this analysis. The nanoparticle suspension was centrifuged at 10.000 rpm for 30 min, and the supernatant was collected. A 10 µl** sample of the supernatant was transferred into a 1.5 ml tube, followed by the addition of 1000 µl** of bromocresol blue reagent. The mixture was then vortexed until homogeneous.
A blank solution was prepared by mixing 10 µl** of distilled water with 1000 µl** of the reagent. A standard solution was prepared using an albumin standard at a concentration of 200 mg/dl; 10 µl** of this standard was mixed with 1000 µl** of reagent. Each mixture was vortexed until homogeneous and incubated at room temperature (24–30 °C) for 10 min. Measurements were performed using a Microlab 300 photometer at a wavelength of 546 nm using the endpoint method. The drug entrapment efficiency (EE) was calculated as follows [11, 12]:
% Drug entrapment = 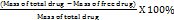
RESULTS
Collection and extraction of snakehead fish
Snakehead fish weighing 707 g were sourced from the Toampe ditch in Sungai Kakap District, West Kalimantan. The fish were selected based on the optimal albumin content criteria of 600 to 900 g per fish [10]. Processing began with sorting to select live fish in good condition, free of parasites, with fresh gills and a clean aroma [11]. The fish were then cleaned by removing scales, fins, and tails, cut into pieces, and steamed at 70 °C for 30 min to break down muscle cells and efficiently release nutrients [12, 13]. After steaming, the fish was pressed and centrifuged at 6000 rpm for 1 h to separate the water and oil phases. The water phase was carefully collected using a syringe. Finally, preservatives-1% DMDM hydantoin and 1.8% methyl and propyl parabens-were added, resulting in a 25.36% yield of the water-phase snakehead fish extract. This result is useful for determining the albumin content or total protein content of the extract in order to comprehensively characterize the raw material.
Preparation of nanoparticle
Nanoparticles were prepared using the ionic gelation method. Alginate, an anionic polymer, reacts with divalent cations such as CaCl₂ [14]. The interaction between CaCl₂ and sodium alginate results in the formation of bonds between Ca²⁺ ions from calcium chloride and the carboxylic groups of guluronic acid in sodium alginate, leading to a polyelectrolyte complex through opposing electrostatic forces and "egg-box" formation [15]. The resulting cross-links enhance the mechanical strength of the formed particles [14].
Table 1: Design of snakehead fish extract nanoparticle formula, Sodium Alginat, CaCl2
| Formula | Sodium alginate concentration (%) | CaCl2 concentration (%) | Water phase of snakehead fish extract concentration (ml) | Sonication time (h) |
| 1 | 0.1 | 0.01 | 15 ml | 1 |
| 2 | 0.1 | 0.01 | 15 ml | 2 |
Visual observation of nanoparticle
The nanoparticles formed with sodium alginate polymer appeared as a clear solution with a white haze in both formulas. This phenomenon is due to the formation of colloids resulting from the interaction between the negative charges of the crosslinking agent and the positive charges of the polymer, known as opalescence [16]. The electrostatic interaction causes the colloidal particles to have like charges. The movement of these particles leads to random collisions in all directions, a phenomenon known as Brownian motion.
The stirring and sonication duration influence the nanoparticles with Na-alginate polymer, as shown in table 2. The reduction of sediment and suspended particles occurs with increasing stirring time of the nanoparticles because the longer the stirring, the more particles are broken down into nano-sized particles. The intensity of solvent molecules contacting the polymer increases, resulting in smaller particle sizes [17]. Sonication affects the suspended particles produced due to sound waves and vibrations that separate particle agglomerates and break them into smaller sizes. The longer the process duration, the more complete the bond breakage and aggregate dispersion, while maintaining small and homogeneous particle sizes. Besides being a simple process, the sonication method also offers advantages as it is faster and more efficient in producing nanoparticles [18].
Table 2: Visual observation of nanoparticle
| Formula | Visual observation |
| Clarity | |
| 1 | Clear |
| 2 | Clear |
Characterization of nanoparticle
Physicochemical characterization of nanoparticles is essential for affect the absorption release of active substances, influence their interactions with biological components, including proteins and tissue membranes. Characterization of water phase nanoparticles of snakehead fish extract was carried out in powder form with the freeze-dry method. Freeze-dry is one of the drying methods that has the advantage of maintaining the quality of extracts from snakehead fish that can leave a sample air content of 1% which meets the requirements of raw materials from nature in pharmaceutical preparations. The powder form to extend the shelf life, the suspension of snakehead fish extract nanoparticles was freeze-dried. Physicochemical characterization of nanoparticles, as the main drug carrier, includes assessment of their shape, size, surface properties, and entrapment efficiency [18].
Table 3: Characterization results of alginate-snakehead fish extract nanoparticle
| Formulation | Sonication time (H) | Sodium alginate Concentration (%) |
CaCl2 concentration (%) |
Sodium alginate: CaCl2: snakehead fish extract | Nanoparticle characterization results |
| Particle size (nm) | |||||
| 1 | 1 | 0,1 | 0.01 | 2:1:1 | 593.6 |
| 613.6 | |||||
| 609.1 | |||||
| Average | 605.433±8.56 | 0.563± | 93.221 | ||
| 2 | 2 | 0,1 | 0.01 | 2:1:1 | 486.9 |
| 482.3 | |||||
| 480.6 | |||||
| Average | 483.26 | 0.558 | 99.406 |
Description: CaCl2= Calcium Chloride; PI = Polydisperse Index. (Results are expressed as a mean±SD, n=3)
% Transmittance
% Transmittance is used as a quantitative measure of the clarity of a solution or dispersed system. Based on table 3, the transmittance of nanoparticles sonicated for 1 h was 93.221%, while the transmittance of nanoparticles sonicated for 2 h increased to 99.406%. A transmittance value close to 100% suggests that the resulting nanoparticles form clear and transparent dispersions, indicating particle sizes within the nanometer scale [19]. The increase in sonication time results in higher % transmittance, likely due to more effective breakdown of particles into uniform nanometer-sized structures. This uniform dispersion allows light to pass through the solution more freely during transmittance measurement [20, 21].
Particle size
The particle size of the sample was determined using a particle size analyzer (PSA) based on the dynamic light scattering (DLS) method [1]. A reduction in particle size increases the total surface area, thereby accelerating drug release. However, smaller particles tend to aggregate during storage, which can negatively affect the stability of the formulation. In this study, sodium alginate and calcium chloride (CaCl₂) were utilized at concentrations of 0.10% and 0.01%, respectively. Excessive CaCl₂ concentrations may influence particle formation due to the generation of extensive cross-linking between Ca²⁺ ions and alginate anions. This interaction may also lead to the formation of insoluble precipitates, resulting in an increase in particle size. The results showed that the smallest particle size was obtained in Formula 2 (2 h sonication), measuring 483.26 nm, as presented in table 2. A decrease in sedimentation and suspended particles was also observed with increasing sonication duration during the nanoparticle preparation process. This can be attributed to the extended sonication time, which facilitates the breakdown of larger particles into nanoscale sizes and enhances solvent-polymer interactions, thereby contributing to further particle size reduction [2]. The particle size decreases with the increase of applied force due to collisions between particles, which enhance the intensity of interaction between the polymer and the extract, resulting in smaller particles, increase in sonication time results in smaller and more homogeneous particle sizes, leading to stable nanoparticles with a reduced tendency for aggregation. This phenomenon occurs because the shock waves generated by cavitation during ultrasonication are capable of breaking down particle agglomerates into smaller units and preventing re-agglomeration, thereby producing a more uniform and stable dispersion system.
Polydispersity index
Nanoparticles with a polydispersity index value of 1 have a very wide size distribution and contain large particles or aggregates that can undergo sedimentation. Table 2 shows the polydispersity index value of nanoparticles with sodium alginate 0,1% (sonication for 1 h) is 0.563±0.030 and sonication for 2 h is 0,558±0,0344. This polydispersity index value is below 0.7 which means it is the upper range where the distribution algorithm operates best and is quite homogeneous with a polydispersity index value close to 0 [24]. Sonication nanoparticle for 2 h produces nanoparticles with a smaller polydispersity index, this is because the optimum ultrasonication time is judged by the ability to dissolve and distribute a material evenly in the system [25]. The cavitation and vibrations generated by ultrasonication reduce the particle size by breaking down particle agglomerates into smaller units and preventing re-agglomeration; thus, the longer the sonication time, the more uniform the fragmented particles become, as bond disruption and aggregate dispersion occur more effectively, resulting in a more homogeneous system [3].
Table 4: Normality test
| Tests of normality | Sonication | Shapiro-wilk |
| Statistic | ||
| Particle Size | 2 H | 0.934 |
| 1 H | 0.908 | |
| Polidispersity Index | 2 H | 0.878 |
| 1 H | 0.807 | |
| %Transmittance | 2 H | 1.000 |
| 1 H | 0.998 | |
| a. Lilliefors significance correction |
Normal distributed test data Sig Shapiro-Wilk above 0.05. The normality test is shown the significant results of Shapiro Wilk in all test groups show sig values >0.05 so it can be concluded that the data characteristics water phase of snakehead fish extract nanoparticle is normally distributed.
Table 5: Independent sample t-test
| Independent samples test | Levene's test for equality of variances | t-test for equality of means |
| F | Sig. | |
| Particle size | Equal variances assumed | 4.832 |
| Equal variances not assumed | ||
| Polidispersity index | Equal variances assumed | .020 |
| Equal variances not assumed | ||
| % transmittance | Equal variances assumed | 1.937 |
| Equal variances not assumed |
The results of the significance test are presented in table 5. The analysis shows that, for the transmittance and particle size parameters under the assumption of equal variances, the sig. value (2-tailed) was<0.05. This indicates that, under the conditions of this study, there is a significant difference between the transmittance and particle size of 0.1% alginate subjected to 2 h of sonication compared to 1 h of sonication. In contrast, the analysis of the polydispersity index under the assumption of equal variances yielded a sig. value (2-tailed)>0.05, indicating no significant difference between the polydispersity index values of alginate nanoparticle formulations subjected to 2 h and 1 h of sonication in this study conditions. Based on these statistical results, it can be concluded that significant differences were observed in the transmittance and particle size between snakehead fish extract alginate nanoparticle formulations sonicated for 2 h and those sonicated for 1 h. The optimal formulation, which produced alginate nanoparticles from snakehead fish extract with good physical characteristics, was F2 (sonicated for 2 h).
Based on these statistical results, it can be concluded that there is a significant difference in transmittance and particle size between the two formulations, but not in polydispersity index. The optimal formulation for producing alginate nanoparticles from snakehead fish extract was Formula 2 (sonication for 2 h), as it demonstrated superior physical characteristics.
This can be attributed to the ultrasonication process, which uses sound waves and vibrations to break down particle agglomerates into smaller sizes. A longer sonication duration enhances the disruption of bonds and promotes better dispersion of aggregates, resulting in smaller and more uniform nanoparticles. In addition to being a simple method, ultrasonication is also a rapid and efficient technique for producing nanoparticles within the nanometer range [18].
Zeta potential
After conducting preliminary tests such as transmittance, particle size test and polydispersity index, the optimal formula for alginate (F2) was obtained so that further tests were carried out, namely zeta potential. Zeta potential describes the condition of the surface charge of nanoparticles, which is sufficient to cause repulsive forces between the particles. A good zeta potential value of>±30 indicates that the repulsion force is greater than the attractive force, thus increasing the stability of the dispersion system. The result of zeta potential of water phase snakehead fish extract nanoparticle with polymer sodium alginate shows-24.6 mV (>±30 mV). A zeta potential value of 24.6 mV indicates that the nanoparticle suspensions in all formulations can be categorized as colloidal systems with moderate physical stability. This suggests that the surface charge of the droplets is predominantly anionic, contributing to a shift toward negative zeta potential values. An optimal sonication time allows the particles to be evenly distributed due to bond disruption and more effective dispersion of aggregates, resulting in a more homogeneous system. This promotes particle interactions that reduce aggregation; however, the addition of stabilizers is still required to maintain the stability of the nanoparticle system [4].
Nanoparticles morphology
SEM characterization is used to observe the morphology of a particle [24]. SEM analysis was carried out on the optimized nanoparticle powder of snakehead fish extract using alginate polymer (F2). At 5000x and 10.000× magnification, the nanoparticles appeared coated but showed an irregular, non-spherical shape with rough surfaces and visible aggregates (fig. 1). Ideally, nanoparticles should be spherical to improve their ability to enter target cells. The presence of aggregates may be caused by van der Waals forces, which are stronger at the nanoscale due to the large surface area of the particles. As a result, particles tend to stick together and form clumps [25]. The uneven surface might be due to incomplete polymer cross-linking during the formulation process and the adhesive properties of albumin are influenced by charged amino acids, such as lysine, which undergo electrostatic interactions that enhance its adhesion [26]. This phenomenon may also occur because the freeze-drying process imposes significant stress, affecting the stability of colloidal nanoparticle suspensions, thereby making them susceptible to aggregation or irreversible fusion between particles. Such issues can be mitigated by the addition of protective agents, namely cryoprotectants such as trehalose, sucrose, glucose, and mannitol [7-9]. Aggregation of nanoparticles during lyophilization is often driven by freezing-and drying‑induced stresses such as ice‑crystal formation and solute‑concentration at ice–liquid interfaces, which promote particle–particle contact and fusion. Cryoprotectants like sucrose or trehalose act by forming a vitrified matrix or by shielding particles, thereby reducing mechanical stress and isolation of individual nanoparticles to mitigate aggregation [26].
(a) |
(b) |
Fig. 1: Nanoparticles morphology using SEM magnification of 5000x (a) and magnification of 10000x (b)
%Entrapment efficiency
A good nanoparticle delivery system has high absorption efficiency, which is influenced by the drug's affinity with the nanoparticle-forming polymer, polymer composition, molecular weight, and the interaction between the drug and the polymer [9]. Absorption efficiency (entrapment efficiency/EE) is used to evaluate the ability of sodium alginate to protect active substances in nanoparticle form [27]. For example, snakehead fish extract nanoparticles with 0.1% sodium alginate sonicated for 2 h showed an absorption efficiency of 96.63%, which is considered good as it exceeds 80% [28]. A higher EE value indicates a stronger bond between the carrier system and the active substance, helping to protect it from environmental degradation. Sodium alginate contains negatively charged carboxyl groups that electrostatically interact with positively charged calcium ions (Ca²⁺) from CaCl₂, forming an "egg-box" structure that effectively entraps albumin molecules. The high entrapment efficiency (>96%) demonstrates that sodium alginate is an optimal polymer for albumin encapsulation. Converting albumin into nanoparticle form enhances its therapeutic effectiveness due to the smaller particle size, which allows faster drug penetration through the stratum corneum, while encapsulation within sodium alginate also extends the shelf life by protecting the protein from degradation.
CONCLUSION
Nanoparticles formulated from Channa striata (snakehead fish) extract using 0.1% sodium alginate and processed via ultrasonication for 2 h demonstrated optimal physicochemical properties. The formulation remained relatively stable over 7 days, with minimal floating particles, slight turbidity, and a characteristic odor of the extract of snakehead fish. The average % transmittance was 99.406%, with a particle size of 483.266 nm, a polydispersity index of 0.528, and a zeta potential of −24.6±0.294 mV, indicating moderate stability. The entrapment efficiency was notably high at 96.63±0.11%. Scanning electron microscopy at 5000× and 10,000× magnification revealed nanoparticles with a predominantly spherical shape, dense structure, and rough surface.
ACKNOWLEDGMENT
These findings support the potential of snakehead fish albumin-based nanoparticles as a promising platform for biomedical applications. Given the established wound-healing, anti-inflammatory, and regenerative properties of Channa striata albumin, the nanoparticle formulation may serve as a suitable candidate for incorporation into topical delivery systems, particularly for wound care. Further in vivo studies and formulation development are warranted to explore its clinical utility in dermal and tissue regeneration therapies.
AUTHORS CONTRIBUTIONS
Mohamad Andrie contributed to the conceptualization of the study, preparation of materials, execution of experiments, data collection, and drafting of the manuscript. Wintari Taurina contributed to study design, data analysis, interpretation of results, critical revision of the manuscript, and overall supervision of the research.
CONFLICT OF INTERESTS
The authors declare no conflict of interest.
REFERENCES
1. Windy YM, Dilla KN, Claudia J, Noval N, Hakim AR. Karakterisasi dan formulasi nanopartikel ekstrak tanaman bundung (Actinoscirpus grossus) dengan variasi konsentrasi basis kitosan dan Na-TPP menggunakan metode gelasi ionik. J Surya Medika. 2022;8(3):25-9. doi: 10.33084/jsm.v8i3.4495.
2. Taurina W, Sari R, Hafinur UC, Wahdaningsih S, Isnindar I. Optimization of stirring speed and stirring time toward nanoparticle size of chitosan-siam citrus peel (Citrus nobilis L. var Microcarpa) 70% ethanol extract. Majalah Obat Tradisional. 2017 Apr 16;22(1):16. doi: 10.22146/tradmedj.24302.
3. Prihantini M, Zulfa E, Prastiwi LD, Yulianti ID. Pengaruh waktu ultrasonikasi terhadap karakteristik fisika nanopartikel kitosan ekstrak etanol daun SUJI (Pleomele angustifolia) dan uji stabilitas fisika menggunakan metode cycling test. J Ilmu Farmasi Farmasi Klin. 2020;16(2):125. doi: 10.31942/jiffk.v16i02.3237.
4. Nemeth Z, Csoka I, Semnani Jazani R, Sipos B, Haspel H, Kozma G. Quality by design-driven zeta potential optimisation study of liposomes with charge imparting membrane additives. Pharmaceutics. 2022;14(9):1798. doi: 10.3390/pharmaceutics14091798, PMID 36145546.
5. Guge SR, Lukum A, Kunusa WR. Pembuatan nano kitosan dengan menggunakan metode gelasi ionik. Jamb J Chem. 2024;6(1):1-8. doi: 10.37905/jambchem.v6i1.21843.
6. Juliantoni Y, Hajrin W, Subaidah WA. Nanoparticle formula optimization of Juwet seeds extract (Syzygium cumini) using simplex lattice design method. JBT. 2020;20(3):416-22. doi: 10.29303/jbt.v20i3.2124.
7. Daskar A, Utami PI, Astuti IY, Antoni F. Formulasi dan karakterisasi nanopartikel ekstrak daun senggani (Melastoma malabathricum L.) pada berbagai variasi komposisi kitosan dengan metode gelasi ionik. Jurnal Farmasi Aisyah. 2022;2(1):46-56. Available from: https://journal.aisyahuniversity.ac.id/index.php/JFA/article/download/najimis/357. [Last accessed on 16 Dec 2025].
8. Heinritz C, NG XJ, Scheibel T. Bio-inspired protein-based and activatable adhesion systems. Adv Funct Materials. 2023;34(35):2303609. doi: 10.1002/adfm.202303609.
9. Ambarwati R. Pembuatan nanopartikel albumin menggunakan metode desolvasi sebagai alternatif sistem pembawa. Fitofarmaka: Jurnal Ilmiah Farmasi. 2019;9(1):35-9. doi: 10.33751/jf.v9i1.1258.
10. Arfiyanti, Ariyanti D. Makanan tambahan ibu hamil untuk mencegah stunting dalam upaya meningkatkan kualitas SDM bagi pertahanan negara. Jurnal Kimia Saintek dan Pendidikan. 2022;6(2):56-65. doi: 10.51544/kimia.v6i2.3494.
11. Andrie M, Sihombing D. Efektivitas sediaan salep yang mengandung ekstrak ikan gabus (Channa striata) pada proses penyembuhan luka akut stadium II terbuka pada tikus jantan galur wistar rats. Pharm Sci Res. 2017;4(2):88-101. doi: 10.7454/psr.v4i2.3602.
12. Waicang R, Maria R, Herawati T. Pengaruh suplemen ekstrak ikan gabus pada pasien nephrotic syndrome. Jurnal Penelitian Kesehatan Suara Forikes. 2022;13(3):600-3.
13. Tungadi R, Wicita P. Formulation optimization and characterization of snakehead fish (Ophiocephalus striatus) powder nanoemulgel. Braz J Pharm Sci. 2020;56(17337):1-8. doi: 10.1590/s2175-97902019000417337.
14. Zdzisaw ES. Food quality and standards pertaining to fish. In: food quality and standards. In: encyclopedia of life support systems (EOLSS) developed under the auspices of UNESCO. Paris, France: EOLSS Publishers. 2009;2(10):134.
15. Indonesian National Standard. SNI 2729:2021 Fresh Fish. Jakarta: National Standardization Agency; 2021.
16. Dina Rahmawanty, Risa Ahdyani, Malikhatun N, Prima Happy Ratnapuri. Nanoparticle preparation and characterization of Haruan fish (Channa striata) extract containing albumin from South Kalimantan with ionic gelation method. Int J Drug Deliv. 2017;9(2):47. doi: 10.5138/09750215.2070.
17. Salmatia S, Isamu KT, Sartinah A. Pengaruh proses perebusan dan pengukusan terhadap kandungan albumin dan proksimat ikan GABUS (Channa striata). Fish Protech. 2020;3(1):67. doi: 10.33772/jfp.v3i1.11606.
17. Andrie M, Taurina W. Nanoencapsulation of ethanol extract of papaya leaf (Carica papaya linn.) using chitosan and testing its effectiveness as an anti-inflammatory. Int J App Pharm. 2024;16(2):264-71. doi: 10.22159/ijap.2024v16i2.49992.
18. Ariani L, Wand Purwanto URE. Nanoparticle formulation of hibiscus leaf extract (Hibiscus rosa-sinensis L.). Semarang: STIFAR; 2021.
19. Supraba W, Juliantoni Y, Ananto AD. The effect of stirring speeds on the entrapment efficiency in a nanoparticles formulation of Java plum’s seed ethanol extract (Syzygium cumini). Acta Chim Asiana. 2021;4(1):197-03. doi: 10.29303/aca.v4i1.50.
20. Betala S, Mohan Varma M, Abbulu K. Formulation and evaluation of polymeric nanoparticles of an antihypetensive drug for gastroretention. J Drug Delivery Ther. 2018;8(6):82-6. doi: 10.22270/jddt.v8i6.2018.
21. Qonitannisa S, Fadli A, Sunarno. Synthesis of nanochitosan by ionic gelation method using acetic acid solvent with variation of chitosan concentration. J Online Mhs BiD Tek Dan Sains. 2020;7(2):1-4.
22. Guge SR, Lukum A, Kunusa WR. Nano chitosan production using ionic gelation method. Jamb J Chem. 2024;6(1):1-8. doi: 10.37905/jambchem.v6i1.21843.
23. Ngafif A. Optimization of sodium alginate and calcium chloride (CaCl2) as cross-linking agents of ethanol extract nanoparticles of katuk leaves (Sauropus androgynus (l.) merr). Berkala Ilmiah Mahasiswa Farmasi. 2020;7(2):13-23. doi: 10.48177/bimfi.v7i2.33.
24. Rahmatullah S, Permadi YW, Agmarina SN. Testing of nanoparticle character ionic gelation method of extract and tablet of african leaf (Vernonia amygdalina del.). J Wiyata. 2021;8(2):147-51.
25. Maharani P, Ikasari E, Purwanto U, Bagiana I. Optimization of na-alginate and ca-chloride in nanoparticles of purified fucoidan extract from brown seaweed (Sargassum polycystum). Pharm Med J. 2022;5(2):38-45. doi: 10.35799/Pmj.V5i2.45100.
26. Picco AS, Mondo GB, Ferreira LF, De Souza EE, Peroni LA, Cardoso MB. Protein corona meets freeze-drying: overcoming the challenges of colloidal stability, toxicity and opsonin adsorption. Nanoscale. 2021;13(2):753-62. doi: 10.1039/D0NR06040B, PMID 33232428.

