Int J App Pharm, Vol 18, Issue 2, 2026, 345-353Original Article
QUALITY-BY-DESIGN ASSISTED DEVELOPMENT AND EVALUATION OF A MICROBALLOON-BASED GASTRORETENTIVE DRUG DELIVERY SYSTEM
MANIVASAKAM PRAKASH1,2, VENKATESWARAMURTHY NALLASAMY1,2*, SENTHIL VENKATACHALAM2, NEELAMEGARAJAN RAMAN1,2
1J. K. K. Nattaraja College of Pharmacy, Namakkal-638183, Tamil Nadu, India. 2The Tamil Nadu Dr. M. G. R Medical University, Chennai-600032, Tamil Nadu, India
*Corresponding author: Venkateswaramurthy Nallasamy; *Email: nvmurthi@gmail.com
Received: 26 Aug 2025, Revised and Accepted: 08 Jan 2026
ABSTRACT
Objective: To develop and optimize levofloxacin-loaded floating microballoons for gastroretentive delivery to improve local antibiotic concentrations against Helicobacter pylori (H. pylori), prolong gastric residence, and achieve extended release using a Quality by Design (QbD) approach.
Methods: Microballoons were prepared by a modified emulsion solvent diffusion technique using levofloxacin hemihydrate, eudragit RS100 (Eu RS100) and ethyl cellulose (EC) in ethanol: dichloromethane (1:1 v/v) and stabilized with 0.75% PVA. A Plackett–Burman design screened formulation and process variables; significant factors were optimized by a central composite design (CCD). Characterization included entrapment efficiency (EE), percentage yield, particle size and zeta potential, in vitro buoyancy, dissolution testing, fourier transform infrared spectroscopy, powder x-ray diffraction, differential scanning calorimetry, scanning electron microscopy and ICH Q1A (R2) stability testing at 25 °C/60% RH and 40 °C/75% RH. Data were modelled in Design-Expert® and kinetics evaluated.
Results: Plackett–Burman screening identified EC, Eu RS100 and stirring speed as critical. CCD optimization selected EC 400 mg and Eu RS100 392 mg (desirability 0.789) at fixed stirring (300 rpm). The validated optimized formulation showed a percentage yield of ≈85%, EE of 74.7%±1.2%, a mean particle size of 266.3±4.2 µm, a zeta potential between −15 and −25 mV, and a buoyancy of 79.1%±2.4% at 12 h. Twelve-hour cumulative release was 64.7 %±1.9 % with sustained, controlled release. Fourier transform infrared spectroscopy spectroscopy showed small changes in the spectrum that were consistent with hydrogen-bonding interactions (the drug did not break down). Powder x-ray diffraction and differential scanning calorimetry showed that levofloxacin lost its crystalline structure (from partial to complete amorphization) in the polymer matrix. Scanning electron microscopy verified the presence of spherical, hollow, porous microballoons characterised by distinct internal cavities. In terms of appearance, drug content, buoyancy, or release profile, stability testing revealed no significant changes.
Conclusion: Levofloxacin-loaded floating microballoons, developed using a QbD framework, provided high entrapment, prolonged gastric buoyancy and controlled release with robust stability. The formulation is a promising gastroretentive platform to enhance local gastric drug concentration and potentially improve H. pylori eradication.
Keywords: Levofloxacin, Floating microballoons, Gastroretentive drug delivery system, Eudragit RS100, Ethyl cellulose, Quality by design, Plackett-burman, Central composite design
© 2026 The Authors.Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i2.56652 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Helicobacter pylori (H. pylori) infection represents a persistent global health challenge, affecting approximately 44% of the adult population worldwide [1]. This pathogen is the main cause of gastric adenocarcinoma and a major cause of peptic ulcer disease and gastric MALT lymphoma. International consensus standards support its eradication. An analysis of 185 countries using GLOBOCAN 2022 and UN demographic predictions estimated 15.6 million lifetime stomach cancer cases among individuals born between 2008 and 2017, with 76% (approximately 11.9 million) linked to H. pylori. This underscores the potential effectiveness of population-level screening and treatment strategies [2]. Such a heavy disease burden accentuates the pressing demand for effective control strategies. The therapeutic scheme for H. pylori is being increasingly threatened. Once the cornerstone of therapy, clarithromycin-containing triple regimens are now shown to be less effective, owing to increasing antibiotic resistance. Clarithromycin resistance has surpassed 20-22% in numerous regions, while metronidazole resistance approaches 70% in certain populations, resulting in eradication rates that frequently fall below the 80% threshold required for first-line therapeutic acceptability [3, 4]. There are other regimens, like bismuth quadruple therapy and fluoroquinolone-based protocols, but they are hard to use in practice because of complicated dosing requirements, patients not following the rules, and the basic pharmacokinetic problem of rapid gastric emptying (less than 2 h). Levofloxacin exhibits enhanced bactericidal efficacy against clarithromycin-and metronidazole-resistant H. pylori strains, establishing it as a significant option for second-line and rescue therapies [5]. Conventional levofloxacin formulations do not achieve sufficient therapeutic efficacy due to rapid gastric transit, suboptimal local drug concentrations at the infection site, and extensive systemic distribution, which heightens adverse effects while diminishing mucosal bioavailability [6, 7]. Drug absorption through the gastric mucus layer from the lumen is more effective at killing H. pylori than basolateral absorption from systemic circulation. However, standard formulations don't keep the drug in the stomach long enough to take advantage of this pathway [8].
To overcome these limitations, gastroretentive drug delivery systems (GRDDS) have emerged as a revolutionary solution. GRDDS also maintain antibiotic levels that exceed the minimum inhibition concentrations in the stomach, the area of H. pylori colonization, for at least 12 h through the extension of the gastric retention period [9]. Single-unit GRDDS can empty the stomach "all-or-nothing", but multiparticulate systems distribute the dose over many subunits, which makes early emptying or device failure less of a problem. Floating microballoons hollow, low bulk density (<1 g/cm³) microspheres-offer a smart approach to combine the characteristics of multiparticulate formulations with prolonged drug-release kinetics. The drug is thus made to become more bioavailable at its intended site of action, while reducing its systemic exposure. Choice of appropriate polymer matrix components is crucial in achieving best results. In this work EC and Eu RS100 were chosen in order to enhance the release of the drug and the floating behavior. The EC has an intrinsic buoyancy (bulk density 0.4 g/cm³) and higher drug-entrapment property [10]. The introduction of Eu RS100 ensures capturing of drug at a release rate which is pH-insensitive, while controlling the membrane permeability [11]. This combination tackles the pair of challenges, i. e., retention of drug in the stomach and prolongation of its gastric acidic environment residence.
The researchers apply a Quality by Design (QbD) methodological approach, starting from the Plackett-Burman screening design [12] and then performing the CCD [13, 14] for the systematic production of FBAs containing levofloxacin salts suffering a floating behaviour. This methodological approach allows for a comprehensive examination of the formulation variables as well as each structure relationships which direct to the optimum settings that increases the therapeutic performance with the formulation stability.
MATERIALS AND METHODS
Materials
Levofloxacin hemihydrate was sourced from BLD Pharmatech Pvt. Ltd. (Hyderabad, India). Eu RS100 was purchased from Otto Chemie Pvt. Ltd. (Mumbai, India), and EC (viscosity grade 10 cP [9–11 mPa·s at 5% w/v in toluene/ethanol 80:20, 25 °C]; TCI, Tokyo, Japan). Polyvinyl Alcohol (PVA), dichloromethane, and methanol of analytical grade were used. All other solvents and chemicals employed in the study were of analytical or pharmacopoeial grade and used. The Design Expert program version 13 (Stat-Ease Inc., Minneapolis, MN, USA) was utilised for generating experimental designs, conducting statistical analyses, and implementing response surface methods.
Methods
Preparation, screening, and optimization of levofloxacin microballoons
Levofloxacin-loaded floating microballoons were formulated by a modified emulsion solvent diffusion technique [15, 16] following the parameters indicated by the experimental designs (Utilisation of Plackett-Burman for the screening phase and central composite for the optimisation phase). Briefly, accurately weighed levofloxacin hemihydrate, Eu RS100, and EC (viscosity grade 10 cP) were dissolved in 20 ml of ethanol: dichloromethane (1:1 v/v) to prepare the organic phase, which was added dropwise to 250 ml of 0.75% (w/v) PVA aqueous solution under continuous stirring at 300 rpm using a mechanical overhead stirrer (Remi Motors, Mumbai, India) fitted with a three-bladed propeller at controlled temperature. Stirring was continued for 2 h to promote solvent diffusion and microballoon formation, after which the microballoons were collected by filtration, washed with water, and dried, with all batches prepared in triplicate. For the screening strategy, a Plackett–Burman 12-run (PB12) design with 11 factors was established; five real factors (A-E) and six dummy variables (D1–D6), all at ±1 coded levels. The dummy columns were meaningless markers for enabling the estimation of background (pure) error and to reduce false positives. In Design-Expert® v13, ANOVA followed with treating D1–D6 as noise and pooling the sum of squares for the estimate mean square error for significance testing. The physical parameters were surfactant concentration (0.50%–0.75% w/v), stirring speed (300–600 rpm), temperature (40–45 °C), Eu RS100 (200, 400 and 800 mg) and EC (400, 600 and 800 mg). The responses (yield, EE, buoyancy and 12 h release) were fitted to first order model (Y = B₀+Σ Bᵢ Xᵢ) [17, 18]. EC and Eu RS100 concentrations and stirring speed were the important (p<0.05); for further optimization, the polymer levels was elevated as critical material attributes while stirring speed was held constant. While 600 rpm enhanced yield and EE in PB12, we maintained stirring at 300 rpm during optimization to prevent excessive shear-driven droplet break-up, which can decrease particle size, narrow the internal cavity, and increase apparent density-factors that negatively impact buoyancy and prolonged release. A constant speed of 300 rpm facilitated effective hollowing (gas entrapment) and resulted in particle sizes exceeding 200 µm, which are advantageous for float strength. A CCD was then applied to optimize EC (Factor A) and Eu RS100 (Factor B) concentrations at five levels across thirteen runs, evaluating responses (EE, particle size, buoyancy at 12 h, and drug release at 8-12 h) using a quadratic polynomial model [19] (Y = b₀+b₁A+b₂B+b₃A²+b₄B²+b₅AB), with data analyzed using Design-Expert® software (Stat-Ease Inc., Minneapolis, MN, USA) to define the design space aligned with the quality target product profile (high EE ≥70%, buoyancy ≥8 h, controlled release 60-70% in 8-12 h, optimized particle size and yield) and critical quality attributes [20].
Fourier transform infrared (FTIR) spectroscopy
FT-IR spectroscopy assessed drug-polymer interactions. Samples (2 mg each of levofloxacin, polymers, physical mixture, and optimized microballoons) were mixed with 200 mg potassium bromide (KBr, spectroscopic grade), ground, and compressed into pellets using a hydraulic press (Specac, Orpington, UK; 10 tons pressure). Spectra were recorded on a Shimadzu IRSpirit X Series FT-IR spectrometer (Shimadzu Corporation, Kyoto, Japan) from 4000 to 400 cm⁻¹ at 4 cm⁻¹ resolution, averaging 32 scans [21]. Peak shifts were analyzed for compatibility.
Powder X-ray diffraction (PXRD)
Crystallinity was assessed using PXRD. Diffraction patterns of levofloxacin, polymers, physical mixture, and optimized microballoons were obtained using a Rigaku SmartLab SE diffractometer (Rigaku Corporation, Tokyo, Japan) with Cu-Kα radiation (λ = 1.5406 Å) at 40 kV and 40 mA, scanning from 5° to 50° 2θ at 0.02° step size and 2°/min speed [22, 23]. Amorphous transformation was inferred from peak intensity reductions.
Differential scanning calorimetry (DSC)
Thermal properties were studied using a Shimadzu DSC-60 (Shimadzu Corporation, Kyoto, Japan). We used samples (5–10 mg) of levofloxacin, polymers, a physical mixture, and optimised microballoons in aluminium pans and heated them from 25 °C to 300 °C at a rate of 10 °C/min under a nitrogen purge (50 ml/min) [24]. Endothermic/exothermic peaks were compared to assess drug-polymer interactions.
Percentage yield
The practical yield was determined to assess process efficiency. After drying microballoons at ambient temperature (25 °C±2 °C) for 24 h until constant weight, they were weighed using an analytical balance (Sartorius Entris II, Göttingen, Germany; precision ±0.0001 g). We was calculatedthe percentage yield [24] as follows:
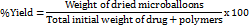
This method measures the amount of material is lost during formulation and is the standard way to determine if manufacturing is possible.
Entrapment efficiency
Dried microballoons (~10 mg) were placed in a 15-mL tube and extracted with 10 ml of methanol using sonication (40 kHz, 10 min), and cleared through centrifugation (3000 g, 5 min). The supernatant was diluted, and its absorbance was read at 294 nm (methanol as blank) on a Shimadzu UV-1800 spectrophotometer. Calibration curve amounted from 2 to 20 µg/ml (R²>0.999). Where EE (%) [25, 26] was calculated as:
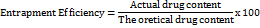
This approach was selected because it is a non-destructive technique and is well suited for microparticles.
Particle size and zeta potential analysis
To assess the physical stability and gastric retention potential, the particle size and zeta potential were measured. We mixed 10 mg of dried microballoons with 10 ml of water in a vortex for 2 min. A Zetasizer Nano ZSP (Malvern Panalytical, Worcestershire, UK) was used to measure the hydrodynamic diameter (Z-average), polydispersity index (PDI), and zeta potential at 25 °C. The angle of scattering was 173° and the refractive index for water was 1.33 [27]. We performed three measurements and reported the mean±SD. We chose this method because it is non-invasive and works well with particles that are only a few microns in size.
In vitro buoyancy study
In vitro buoyancy was evaluated to replicate gastric floating dynamics. In a glass beaker at 37 °C±0.5 °C, 100 ml of simulated gastric fluid (0.1 N HCl, pH 1.2, made with analytical grade HCl) was mixed with 100 microballoons. The mixture was stirred at 100 rpm using a Remi RQ-121/D magnetic stirrer (Remi Elektrotechnik Ltd., Mumbai, India). We counted the floating microballoons at 1, 4, 8, and 12 h after separating the particles that had sunk. We recognize that 100 rpm magnetic stirring can impose hydrodynamic forces that bias particles downward; for low-density microparticles, buoyancy evaluation in a USP paddle apparatus (50 rpm) provides gentler, more reproducible conditions. This is noted as a study limitation. The buoyancy percentage was computed [25, 26, 28] as:
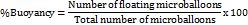
This method simulates gastric conditions and conforms to USP standards for floating systems.
In vitro drug release study
Drug release was studied to assess controlled release under gastric conditions using a LabIndia Disso 8000 dissolution tester (Labindia Instruments Pvt. Ltd., Mumbai, India; USP Apparatus 2, paddle method). Microballoons equivalent to 100 mg levofloxacin (based on EE) were placed in 900 ml of 0.1 N HCl (pH 1.2) at 37 °C±0.5 °C, with paddle speed at 50 rpm. At intervals (0.5, 1, 2, 4, 6, 8, 10, 12 h), 5 ml aliquots were withdrawn (with replacement), filtered through a 0.45 µm cellulose acetate filter, and analyzed using the Shimadzu UV-1800 UV-Vis spectrophotometer at 294 nm against a calibration curve (linearity: 1–50 µg/ml, R²>0.999). Cumulative drug release was plotted versus time [20]. The paddle method was chosen for its relevance to oral dosage forms, maintaining sink conditions.
Release kinetics studies
The cumulative drug release profile of the optimized formulation was fitted to zero-order, first-order, Higuchi, and Korsmeyer–Peppas models to elucidate the drug release mechanism. We used Microsoft Excel (Solver add-in) to do curve fitting and regression analysis. We found the best-fit model by looking for the highest correlation coefficient (r²). The diffusional exponent (n) from the Korsmeyer–Peppas equation was then used to sort the release mechanism into Fickian, non-Fickian, or Case II transport.
Surface morphology and elemental analysis
Surface morphology and internal structure were examined to confirm microballoon formation. Dried microballoons were mounted on aluminum stubs with double-sided carbon adhesive tape (Agar Scientific, Stansted, UK), coated with a 10 nm gold-palladium layer using a Cressington 108auto sputter coater, and imaged at 5–15 kV and magnifications (50× to 5000×) using a Carl Zeiss Sigma 300 Scanning Electron Microscope (SEM) (Carl Zeiss AG, Oberkochen, Germany). We selected this method because it can take high-resolution pictures. We performed energy dispersive x-ray (EDX) Spectroscopy analysis in situ under the same beam conditions, taking spectra at several surface points with a Zeiss SmartEDX detector. We used ZAF correction to process the spectra for quantitative C and O mapping, which confirmed the elemental composition and even distribution of the polymer across the microballoon shell [28].
Stability studies
We evaluated for stability according to International Council for Harmonisation (ICH) Q1A(R2) [29]. Optimised microballoons were kept in airtight glass vials at 25 °C±2 °C/60%±5% RH and 40 °C±2 °C/75%±5% RH in stability chambers (KL Lab, Chennai, India). Samples were analyzed at 0, 1, 2, and 3 mo for physical appearance, drug content (via UV-Vis), drug release, and buoyancy, as described [20, 21]. This ensured formulation robustness.
Statistical analysis
Experiments were performed in triplicate (n=3), with data as mean±SD [30]. Design-Expert version 13 (Stat-Ease Inc., Minneapolis, MN, USA) was used for experimental design, model fitting (first-order for Plackett-Burman, quadratic for CCD), and optimization. ANOVA evaluated model and factor significance (P<0.05 significant, P<0.01 highly significant), with R²>0.9 and a non-significant lack-of-fit validating model adequacy [31].
RESULTS AND DISCUSSION
Fourier transform infrared spectroscopy studies
The characteristic peaks in the FT-IR spectra of pure levofloxacin were approximately 1725 cm⁻¹ (COOH C=O) and 1622 cm⁻¹ (C=O stretching), and a band at about 1541 nm inflicted the signature of N–H bending as reported in the literature [21]. The bands were still in the physical mixture with intense reduction in the intensity revealing no noticeable incompatibility. The optimized microballoons showed suppression of the C=O band around 1622 cm⁻¹, emergence of a weak characteristic at ∼1452 cm⁻¹ and presence of a new broad feature about 3493 cm⁻¹. The latter is related to O–H stretching caused by hydrogen bonding, such as those between levofloxacin carboxyl/amine groups and polymer moieties or bound water. The minor changes and enhancement in intensity where observed for the non-covalent interaction positioned the drug with the polymer than a new chemical species. This also corresponds to improved dispersion, stability and overall controlled release.
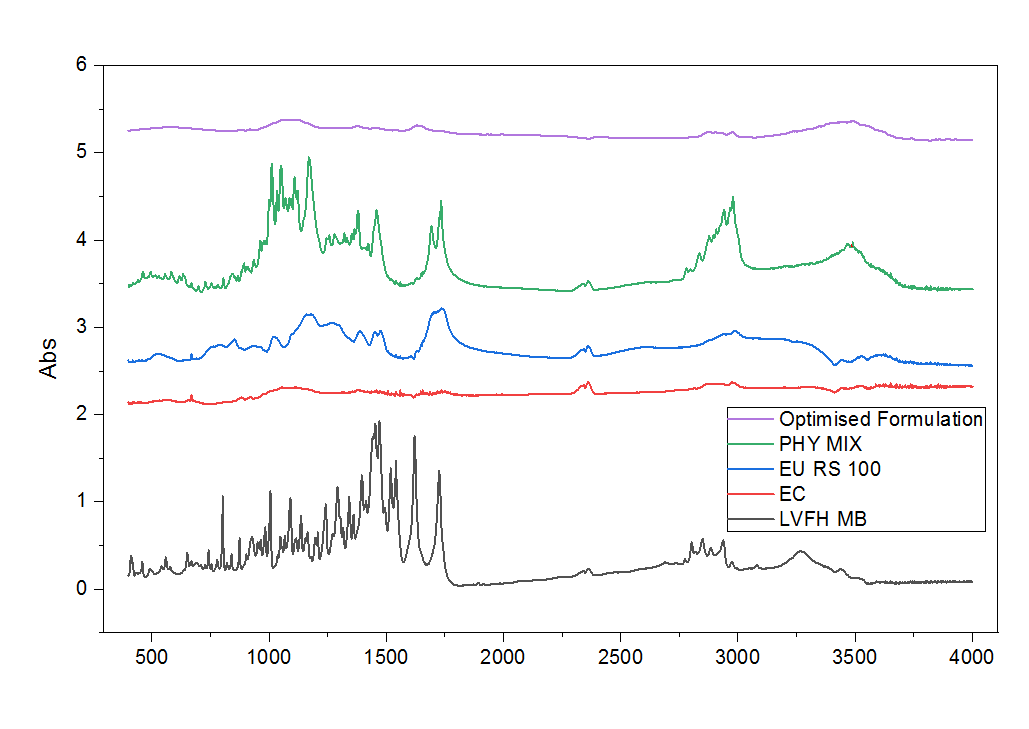
Fig. 1: FT-IR spectra (4000–500 cm⁻¹) of levofloxacin, EC, Eu RS100, physical mixture, and optimized microballoons. A new broad band at ~3493 cm⁻¹ with minor shifts near 1622 and 1452 cm⁻¹ supports hydrogen bonding (no new species)
Powder X-ray diffraction
We investigated the crystal state of levofloxacin in formulations by PXRD (fig. 2). XRD analysis Pure levofloxacin exhibited intense diffraction peaks at 19.5°, 23.1°, and 26.5° suggesting its crystalline nature. The polymers exhibited broad halos, in good agreement with the amorphous nature. The levofloxacin peaks were present even in the physical mixture, indicating no interaction at this stage. On the other hand, optimised microballoons exhibited a marked reduction, broadening, or disappearance of the crystalline peaks. This fact shows that there is transformation of levofloxacin from crystallization state to partially or completely amorphous form. This amorphization indicates that the drug molecules are distributed inside the polymeric matrix. That can increase the solubility of the drug and make it dissolve more quickly, aiding bioavailability. This is in accord by previously reported levofloxacin polymeric delivery systems in which a lower crystallinity contribute to better performance [33, 34].
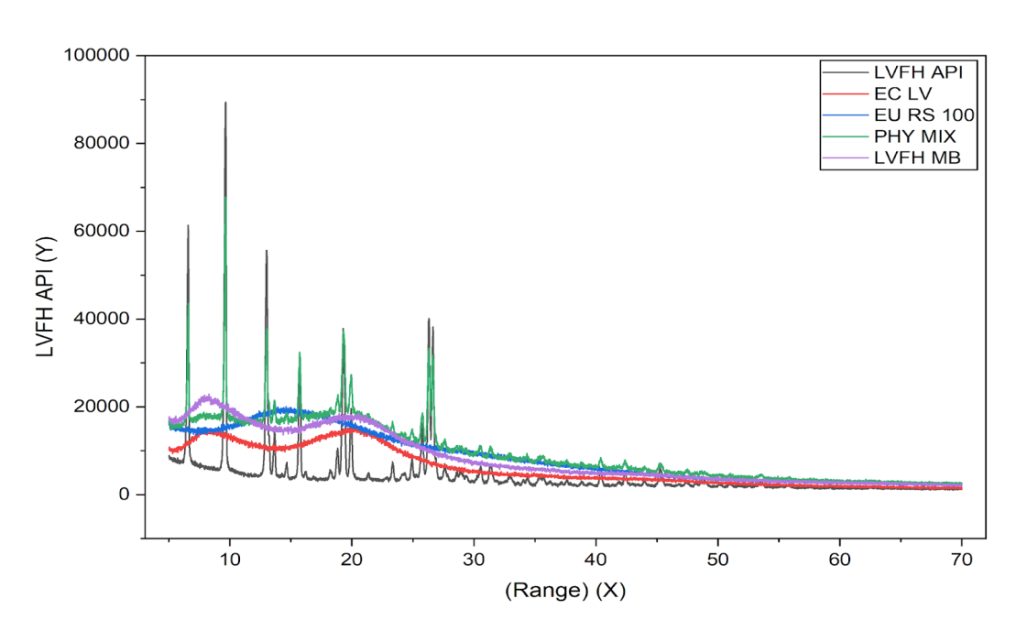
Fig. 2: PXRD patterns of pure drug, polymers, physical mixture, and optimized microballoons
Differential scanning calorimetry
Additional information about the solid-state behaviour of levofloxacin in the formulations was obtained from DSC thermograms (fig. 3). The melting point and crystalline nature of pure levofloxacin were indicated by a sharp endothermic peak at 232 °C. This distinctive melting endotherm was not present in the optimised microballoon formulation. Rather than a sharp drug melting peak, several broadened thermal events were seen at lower temperatures (88.6 °C, 117.8 °C, and 187.9 °C) as well as a weak transition at 216.8 °C. Levofloxacin may have changed into an amorphous or molecularly dispersed state within the polymeric matrix, as indicated by the disappearance of the characteristic crystalline melting signal. The developed gastroretentive microballoons sustained-release qualities are supported by this amorphization, which also improves drug solubility and helps ensure stable encapsulation [35].
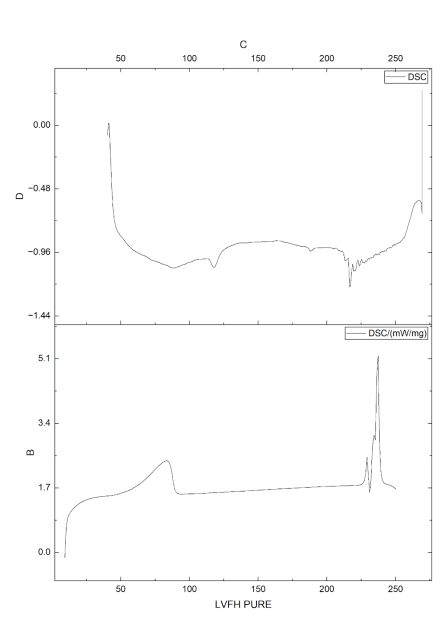
Fig. 3: DSC thermograms of pure drug, polymers, physical mixture, and optimized microballoons
Plackett-burman design (PBD) screening
In the PBD phase, twelve experimental runs screened the influence of five real factors (A: surfactant concentration 0.50–0.75%; B: stirring speed 300–600 rpm; C: stirring temperature 40–45 °C; D: Eu RS100 200–600 mg; E: EC 400–800 mg) and six dummy factors on percentage yield (72.6–88.4%), entrapment efficiency (EE, 76.9–91.9%), buoyancy (7.0–9.2 h), and drug release at 12 h (48.5–63.1%). Experimental data (mean±SD, triplicates, SD<5%) are presented in table 1. EC, Eu RS100 and stirring speed made (p<0.05) difference on Critical Quality Attributes (CQAs) through ANOVA, As expected, all dummy factor effects (D1 – D6) were nonsignificant (p>0.10), ensuring that the observed main effects of EC, Eu RS100, and level of stirring are indeed true process events and not due to random variations. Due to the hydrophobic nature of EC as well as their ability to form rigid matrix, EC and Eu RS100 showed an increase in yield, EE, and float-ability with decrease in release vice versa, as reported for polymeric floating microspheres [15, 19, 21, 31, 33]. Denser particle formation explained by high shear, decreased buoyancy and release and increased yield and EE as the stirring speed increased, respectively [24, 29]. Modest levels of PVA were sufficient to stabilise the emulsion considering insignificant effect of the surfactant [18, 30]. Run 12 (desirability = 0.978) was ranked first, with the yields of 88.4±2.1%, EE91.9±1.8%, buoyancy 9.1±0.3 h, and release 48.7±1.5%. According to QbD risk-based reduction principles, PMD successfully recognised a critical material attribute (CMA) (EC and Eu RS100) and a critical quality attribute (CQA) (stirring speed), which allowed screening eleven variables down to three for optimisation [22, 25, 35].
Table 1: Plackett–burman screening design (12 runs) for 11 variables: five real factors and six dummy factors (coded −1/+1). Real factors and actual levels: A (Surfactant, %), B (Stirring speed, rpm), C (Temperature, °C), D (Eudragit RS100, mg), E (Ethyl cellulose, mg). Dummy factors D1–D6 had no physical meaning and were included to estimate experimental error in effect screening ANOVA
| Run | A: Surfactant (%) | B: Stirring speed (rpm) | C: Temp. (°C) | D: Eu RS100 (mg) | E: EC (mg) | F-K: D1-D6 | Percentage Yield (%) | EE (%) | Buoyancy (h) | Drug release @ 12 h (%) | Overall desirability | Rank |
| 1 | 0.75 | 300 | 45 | 200 | 800 | - | 82.5 | 87.4 | 8.2 | 50.9 | 0.896 | 5 |
| 2 | 0.50 | 600 | 45 | 200 | 400 | - | 74.3 | 78.6 | 7.3 | 62.4 | 0.794 | 11 |
| 3 | 0.75 | 300 | 40 | 600 | 400 | - | 80.2 | 84.1 | 8.9 | 55.7 | 0.872 | 7 |
| 4 | 0.50 | 600 | 40 | 600 | 800 | - | 87.9 | 91.5 | 9.2 | 48.5 | 0.966 | 2 |
| 5 | 0.75 | 600 | 45 | 200 | 400 | - | 75.1 | 79.8 | 7.4 | 61.7 | 0.802 | 10 |
| 6 | 0.50 | 300 | 45 | 600 | 400 | - | 81.4 | 84.7 | 8.6 | 54.8 | 0.864 | 9 |
| 7 | 0.75 | 300 | 40 | 200 | 800 | - | 84.6 | 88.5 | 8.1 | 51.2 | 0.905 | 4 |
| 8 | 0.50 | 600 | 40 | 200 | 400 | - | 72.6 | 76.9 | 7.0 | 63.1 | 0.781 | 12 |
| 9 | 0.75 | 600 | 40 | 600 | 400 | - | 82.8 | 85.8 | 8.7 | 54.2 | 0.884 | 6 |
| 10 | 0.50 | 300 | 45 | 200 | 800 | - | 85.5 | 89.2 | 8.3 | 50.5 | 0.912 | 3 |
| 11 | 0.75 | 300 | 45 | 600 | 400 | - | 81.9 | 85.0 | 8.8 | 54.9 | 0.869 | 8 |
| 12 | 0.50 | 600 | 45 | 600 | 800 | - | 88.4 | 91.9 | 9.1 | 48.7 | 0.978 | 1 |
Responses are presented as means of three replicates (n = 3). No dummy factor was significant (p>0.05).
Central composite design (CCD) optimization
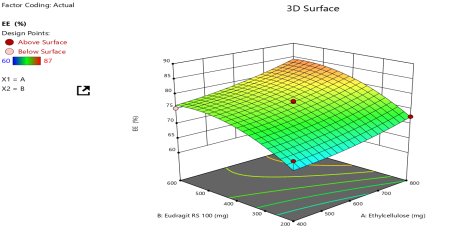
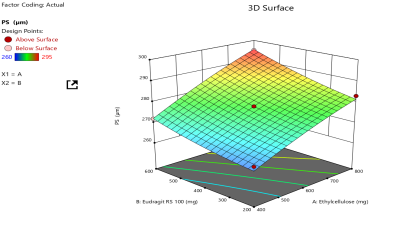
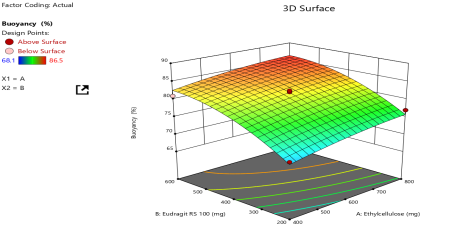
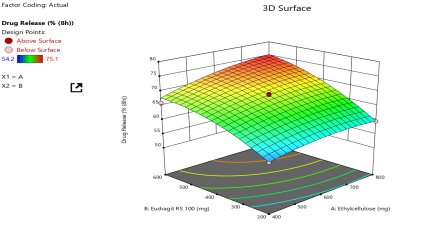
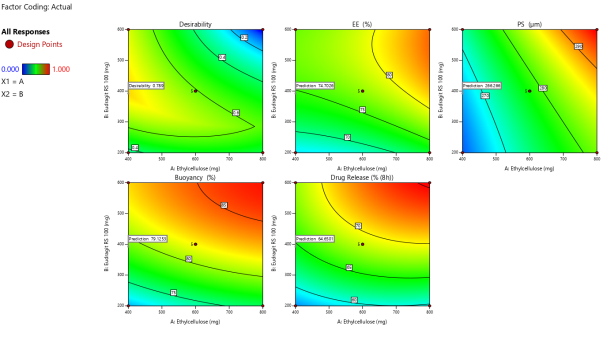
CCD explored EC (400–800 mg) and Eu RS100 (200–600 mg) at fixed stirring speed (300 rpm) over 13 runs (table 2). Responses included EE (60–87%), particle size (260–295 µm), buoyancy (68.1–86.5%), and 12 h release (54.2–75.1%). Quadratic models were significant (p<0.05, R²>0.93, nonsignificant lack-of-fit for most), validating predictive accuracy. EE was strongly influenced by both polymers (Model F = 12.86, p = 0.0020), where increased polymer levels enhanced drug retention by reducing diffusion from the organic to aqueous phase, aided by ionic interactions between Eu RS100 quaternary ammonium groups and levofloxacin carboxyl [28, 32, 38]. Particle size increased with polymer levels due to elevated viscosity during emulsification (Model F = 129.9, p<0.0001), consistent with droplet growth under viscous resistance [26, 34]. Buoyancy improved with polymers but declined at excessive Eu RS100 due to increased density, reflecting the need for balanced ratios to maintain porosity [20, 23, 37]. Drug release decreased with higher polymer levels due to thicker diffusion barriers (Model F = 48.99, p<0.0001), consistent with sustained systems based on EC/Eu RS100 blends [16, 21, 27].
Table 2: CCD run matrix and responses. “Space Type”: Factorial = cube corner points at coded levels −1 and+1; Axial = star points at ±α (α = 1.414 for a rotatable CCD); Center = coded level 0. Factors: A = Ethyl cellulose (mg, actual); B = Eudragit RS100 (mg, actual)
| Run | Space type | A: Ethyl cellulose (mg) | B: Eudragit RS100 (mg) | EE (%) | PS (µm) | Buoyancy (%) | Drug release at 12 h (%) |
| 1 | Axial | 600 | 682.843 | 81.1 | 289 | 86.5 | 75.1 |
| 2 | Factorial | 800 | 200 | 72.5 | 283 | 77.0 | 59.5 |
| 3 | Center | 600 | 400 | 77.2 | 278 | 81.0 | 67.9 |
| 4 | Center | 600 | 400 | 77.5 | 278 | 82.1 | 69.1 |
| 5 | Center | 600 | 400 | 77.8 | 278 | 82.5 | 68.7 |
| 6 | Axial | 600 | 117.157 | 60.0 | 270 | 68.1 | 54.2 |
| 7 | Axial | 317.157 | 400 | 73.0 | 260 | 78.5 | 63.4 |
| 8 | Axial | 882.843 | 400 | 87.0 | 290 | 83.4 | 70.6 |
| 9 | Center | 600 | 400 | 77.5 | 278 | 82.1 | 68.9 |
| 10 | Center | 600 | 400 | 76.9 | 278 | 82.1 | 68.9 |
| 11 | Factorial | 400 | 600 | 75.3 | 272 | 81.0 | 66.0 |
| 12 | Factorial | 800 | 600 | 80.4 | 295 | 85.2 | 73.5 |
| 13 | Factorial | 400 | 200 | 69.1 | 265 | 72.0 | 57.0 |
Responses are presented as means of three replicates (n = 3). EE = entrapment efficiency; PS = particle size; Buoyancy and Drug release reported at 12 h and in %.
Effects on individual CQAs
Percentage yield
Batch yields spanned 72.6–88.4% in PBD and =85% for the CCD-optimized batch. The positive main effects of EC and Eu RS100 on yield are consistent with polymer-driven stabilization of the primary emulsion and formation of a cohesive shell that limits material loss during solvent diffusion/evaporation (microballoon literature frequently reports>80% when the organic phase viscosity and polymer content are adequate). Increased stirring speed also improved yield in screening-likely by promoting more efficient solvent exchange and narrower droplet size distributions that resist coalescence-an effect noted in floating microsphere systems prepared by emulsion–solvent diffusion [15, 16, 19, 28]. The overall yield performance falls within the typical range for EC/Eu RS100 floating systems [9, 10, 23].
Entrapment efficiency
EE ranged 76.9–91.9% in PBD and 60–87% in CCD; the optimized batch achieved 74.7±1.2%. Both EC and ERS showed significant positive linear terms for EE, with a curvature (B²) at higher Eu RS100, indicating diminishing returns as the matrix saturates. Mechanistically, higher polymer levels increase the organic-phase viscosity and shorten the drug diffusion path length to the interface, curbing leaching into the aqueous phase during emulsification [16, 19, 26]. In Eu RS100-containing matrices, quaternary ammonium groups can engage in ionic/hydrogen-bonding interactions with levofloxacin functionalities, which has been invoked to rationalize improved retention in levofloxacin polymeric systems [22, 23, 34]. Achieving EE>70% is typical for GRDDS microspheres and aligns with reported values for amoxicillin/famotidine/riboflavin floating microballoons and levofloxacin gastric systems [15, 19, 23, 27, 28, 33].
Buoyancy study
Buoyancy was quantified in a beaker at 100 rpm; this setup may underestimate buoyancy compared with USP Apparatus II at 50 rpm. Future work using USP Apparatus II at 50 rpm is needed to confirm buoyancy under standardized hydrodynamic conditions. Buoyancy was 7.0–9.2 h in PBD and 68.1–86.5% at 12 h in CCD, with the optimal at 79.1±2.4% at 12 h exceeding the quality target product profile (QTPP) (>8 h). EC and Eu RS100 increased buoyancy by generating porous, low-density shells (from solvent diffusion and internal gas entrapment), a hallmark of hollow microspheres [15, 16, 19, 27, 28]. The quadratic penalty at high ERS levels likely reflects over-densification (thicker, less porous walls) that can trap less gas and increase apparent density, a trend noted in polymer-ratio studies of floating microspheres [19, 26, 28]. The performance is in line with gastroretentive design principles-adequate porosity/rigidity balance is required to both entrap gas (Eu RS100 swelling/voids) and resist collapse (EC rigidity) under gastric hydrodynamics [9, 10, 15, 27].
Drug release
12 h release ranged 48.5–63.1% (PBD) and 54.2–75.1% (CCD); the optimal batch was 64.7±1.9%. The negative influence of polymer on the release (origins of polymer effects on release) is due to a thicker diffusion barrier and lower matrix permeability, as the EC/Eu RS100 content increases (diffusion-controlled release) [9–11, 23]. Eu RS100 addition offers pH-independent permeability but at higher EC loadings, (compounded with EC) slows transport due to tortuosity and lower effective diffusivity–which explains the observed quadratic terms [10, 11, 16, 23]. The 12-h release ~65% falls in the 50–70% range commonly demonstrated for sustained gastric antibiotic release to balance retention of local concentration with limited burst [15, 23, 27, 33, 34].
(a) Entrapment efficacy |
(b) Particle size |
(c) Buoyancy |
(d) Drug release |
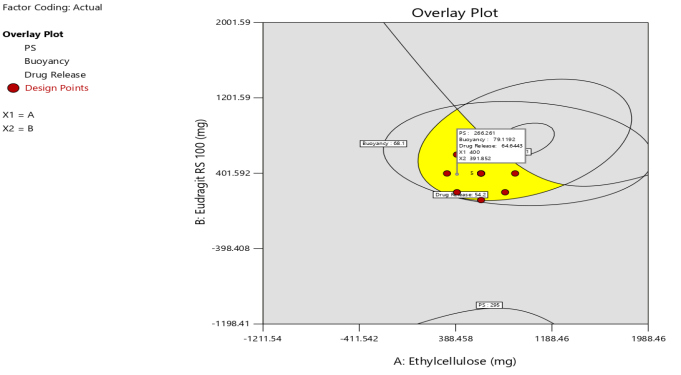
(e) Numerical Contour – all responses – design point |
(f) Graphical Contour – overlay plot – design point |
Fig. 4: Response-surface analysis for CCD: (a) EE, (b) Particle size, (c) Buoyancy, (d) Drug release, (e) Numerical desirability contour with design point, and (f) Overlay plot with design space and optimum
Particle size and zeta potential analysis
CCD producedparticle size 260–295 µm and the optimized one at 266.3±4.2 µm, while both EC and Eu RS100 gave larger particle size due to the higher viscosities of the organic phase to encourage greater initial droplets and thicker wall thickness prior to solidification [16, 19, 26]. The optimized batch measured 266±4 µm and retained buoyancy at 12 h, despite a decrease in EE compared with high-shear PB12 runs. Particle size of>100 µm has been suggested as the optimum size for floating microballoons, and a large particle size may be beneficial in terms of float strength and ease of gastric residence because premature passage through the gastrointestinal tract is minimized [15, 19, 27, 28]. At fixed stirring (300 rpm) and particle-size control, polymer levels controlled droplet rheology, agreeing with the CCD model [31]. The zeta potential (−15 to −25 mV) indicates moderate electrostatic stabilization. For rigid microparticles of 200–300 µm, physical stability during testing is governed primarily by gravitational settling rather than colloidal aggregation; therefore, a very high |ζ| is not required as it would be for nanoparticles. The observed ζ is adequate for routine handling and measurement without agglomeration [16, 19, 26].
Synthesis of effects and optimization outcome
Collectively, EC and Eu RS100 showed synergistic, yet non-linear control of microstructure: increasing EE, buoyancy, and particle size while reducing release (fig. 4), with quadratic terms guarding against over-densification. This is consistent with prior EC/Eu RS100 floating systems and levofloxacin GRDDS reports [15, 19, 23, 34]. The chosen compromise (EC 400 mg, Eu RS100 392 mg) maximized overall desirability (0.789) and validated well (EE 74.7%, particle size 266 µm, buoyancy 79.1%, 12-h release 64.7%)-supporting the QbD/DoE approach to define a robust design space [30, 31, 38-40].
Release kinetics studies
The optimized microballoons released 64.7±1.9% levofloxacin at 12 h. Kinetic analysis (Excel Solver) showed the best fit with the Korsmeyer–Peppas model (n = 0.727; R² = 0.9972), indicating anomalous (diffusion+polymer relaxation/erosion) transport. Goodness-of-fit ranked Korsmeyer–Peppas (R² = 0.9972)>First-order (R² = 0.9959)>Zero-order (R² = 0.9881)>Higuchi (R² = 0.9878). Although Higuchi also fit well—supporting a substantial diffusion component-the highest R² for K–P together with n ≈ 0.73 justifies concluding anomalous transport. This dual-mechanism release supports the design goal of an initial bactericidal phase followed by prolonged gastric exposure needed for H. pylori eradication [11, 23] and aligns with the observed hollow, porous morphology and partial drug amorphization.
Surface and internal structural characterization
SEM confirmed spherical, hollow, porous microballoons with 200–300 µm diameters (fig. 5A–B). EDX spectra (fig. 5C) corroborated the expected C/O composition of the EC/Eu RS100 shell; elemental analysis was used only to verify polymer presence and is not interpreted for release performance. These structural characteristics are typical of systems based on solvent evaporation, in which the formation of hollow cores due to the diffusion of volatile solvents improves gastroretentive performance. These findings are consistent with earlier studies on floating microballoons based on levofloxacin and eudragit, which showed comparable morphology–function relationships [7, 41].
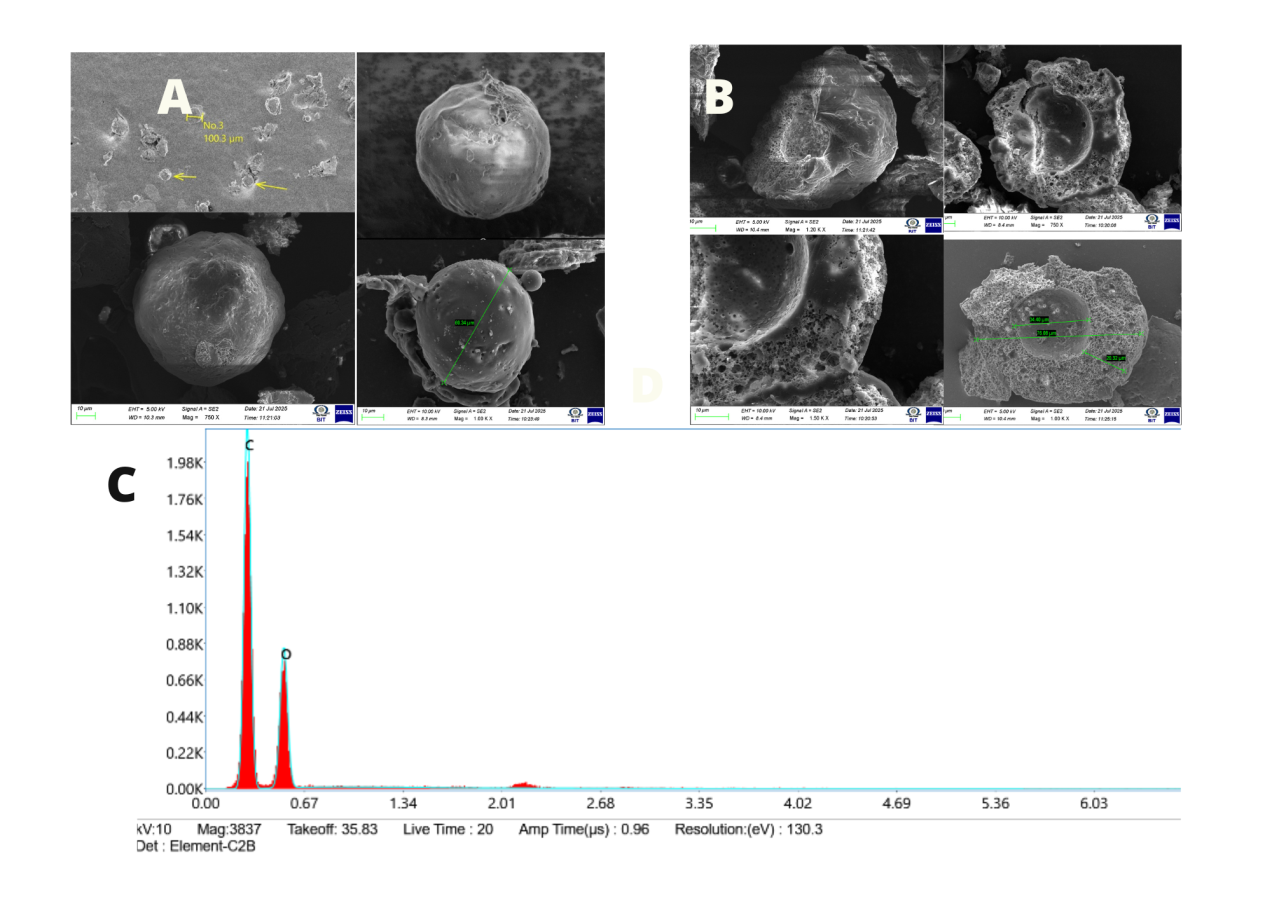
Fig. 5: SEM and EDX analysis of optimized levofloxacin-loaded microballoons. (A) Spherical shape, (B) Hollow cavity structure in cross-section, and (C) Elemental composition as shown by the EDX spectrum
Stability studies
Under ICH Q1A(R2) accelerated settings (40 °C/75 % RH), no notable alterations were detected over a period of 3 mo in appearance, assay, buoyancy, or release. Although promising, a minimum of six months of accelerated data is often necessary before suggesting a provisional shelf-life; thus, we provide these as preliminary stability results. The drug content (98.5–99.2%), buoyancy (78.0–79.5%), and drug release (63.8–65.2%) of optimised formulation has not showed significant difference with those of initial ones (p>0.05, one-way ANOVA), retaining the physical appearance. These results indicate the good stability for the polymeric matrix due to the resistance offered against degradation by moisture. The studied stability patterns are in line with literature data on polymeric GR systems, highlighting the protective role of pH-insensitive and hydrophobic polymers towards the formulation integrity [29, 42].
Overall, the investigation corroboratively confirms the achievement of the aim to fabricate levofloxacin-loaded floating microballoons for better gastro-retention, as evidenced from the excellent performance properties of the formulation that directly address the major constraints of conventional H. pylori eradication therapy. The high EE of the optimised formulation (74.7±1.2%) guarantees precise formulation and economizes on costs, while its sustained buoyancy (79.1±2.4% at 12 h) prolongs the gastric residence time from the usual 2-hour transit time in conventional formulations. This in turn permits more prolonged action of the drug on the site of H. pylori colonisation, in the gastric mucosa. The controlled delivery profile (which released 64.7±1.9% of the drug over 12 h via non-Fickian transport; n = 0.73) offers the combined advantage of a high initial killing effect, as well as maintaining the concentration of the drug within the therapeutic range. This property may be important to combat both planktonic and biofilm-associated H. pylori [43]. The QbD-driven development strategy was instrumental in achieving these outcomes. The methodical optimisation resulted in microballoons having the optimum particle size (266.3±4.2 µm) while neither facilitating early stomach evacuation nor collapsing during an accelerated mode. Importantly, the gastroretentive profile is expected to sustain therapeutically relevant levofloxacin levels in the gastric lumen and at the mucosal surface for extended periods, which may improve local antibacterial activity at the site of H. pylori colonization. Accordingly, this gastroretentive system represents an attractive approach to increase H. pylori eradication rates, and is anticipated that such dosing strategies could reduce the systemic drug exposure responsible for adverse effects and resistance development, addressing both the patient’s need for effective eradication therapy and the public health problem of preventing H. pylori-induced gastric malignancies.
CONCLUSION
This study developed and optimised levofloxacin-loaded floating microballoons using the QbD approach. The optimised formulation (EC 400 mg; Eu RS100 392 mg) attained a high encapsulation efficiency (74.7±1.2 %), sustained buoyancy (79.1±2.4 % at 12 h), regulated release (64.7±1.9 % at 12 h), and a particle size of approximately 266 µm. Screening and CCD modelling indicated that polymer concentrations are critical factors influencing EE, buoyancy, and release, with model adequacy validating the established design space. Solid-state and spectroscopic results corroborated non-covalent interactions between the drug and polymer, as well as diminished crystallinity within the matrix. No substantial alterations were seen under accelerated settings for three months; longer-term stability is recommended according to ICH recommendations. The platform signifies a viable gastro-retentive approach to augment local stomach exposure of levofloxacin and warrants in vivo assessment.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
Manivasakam Prakash-Conceptualization, Methodology, Investigation, Formal analysis, Writing-original draft; Venkateswaramurthy Nallasamy-Conceptualization, Methodology, Supervision, Writing-review and editing; Senthil Venkatachalam-Institutional support, Supervision, Writing-review and editing; Neelamegarajan Raman-Resources, Writing-review and editing.
CONFLICT OF INTERESTS
Declared none
REFERENCES
Hooi JK, Lai WY, Ng WK, Suen MM, Underwood FE, Tanyingoh D. Global prevalence of Helicobacter pylori infection: systematic review and meta-analysis. Gastroenterology. 2017 Aug;153(2):420-9. doi: 10.1053/j.gastro.2017.04.022, PMID 28456631.
Malfertheiner P, Megraud F, Rokkas T, Gisbert JP, Liou JM, Schulz C. Management of Helicobacter pylori infection: the Maastricht VI/Florence consensus report. Gut. 2022 Sep;71(9):1724-62. doi: 10.1136/gutjnl-2022-327745, PMID 35944925.
Savoldi A, Carrara E, Graham DY, Conti M, Tacconelli E. Prevalence of antibiotic resistance in Helicobacter pylori: a systematic review and meta-analysis in world health organization regions. Gastroenterology. 2018 Nov;155(5):1372-82.e17. doi: 10.1053/j.gastro.2018.07.007, PMID 29990487.
Chey WD, Howden CW, Moss SF, Morgan DR, Greer KB, Grover S. ACG clinical guideline: treatment of Helicobacter pylori infection. Am J Gastroenterol. 2024 Sep;119(9):1730-53. doi: 10.14309/ajg.0000000000002968, PMID 39626064.
Hu Y, Zhu Y, Lu NH. Recent progress in Helicobacter pylori treatment. Chin Med J (Engl). 2020 Feb 5;133(3):335-43. doi: 10.1097/CM9.0000000000000618, PMID 31929363.
Umamaheshwari RB, Ramteke S, Jain NK. Anti-Helicobacter pylori effect of mucoadhesive nanoparticles bearing amoxicillin in experimental gerbils model. AAPS PharmSciTech. 2004 Jun;5(2):e32. doi: 10.1208/pt050232, PMID 15760090.
Kim DH, Lee SW, Lee JH, Park JW, Park SM, Maeng HJ. Development of gastroretentive floating combination tablets containing amoxicillin trihydrate 500 mg and levofloxacin 125 mg for eradicating resistant helicobacter pylori. Pharmaceutics. 2024 Sep 24;16(10):1242. doi: 10.3390/pharmaceutics16101242, PMID 39458574.
Azab ET, Thabit AK, McKee S, Al Qiraiqiri A. Levofloxacin versus clarithromycin for Helicobacter pylori eradication: are 14 d regimens better than 10 d regimens? Gut Pathog. 2022 Dec 6;14(1):24. doi: 10.1186/s13099-022-00502-3, PMID 35668511.
Streubel A, Siepmann J, Bodmeier R. Gastroretentive drug delivery systems. Expert Opin Drug Deliv. 2006 Mar 28;3(2):217-33. doi: 10.1517/17425247.3.2.217, PMID 16506949.
Arora S, Ali J, Ahuja A, Khar RK, Baboota S. Floating drug delivery systems: a review. AAPS PharmSciTech. 2005 Oct 19;6(3):E372-90. doi: 10.1208/pt060347, PMID 16353995.
Korsmeyer RW, Gurny R, Doelker E, Buri P, Peppas NA. Mechanisms of solute release from porous hydrophilic polymers. Int J Pharm. 1983 May;15(1):25-35. doi: 10.1016/0378-5173(83)90064-9.
Bolton S, Bon C. Pharmaceutical statistics. 5th ed Bolton S, Bon C, editors. Boca Raton: CRC Press; 2009. p. 670. doi: 10.3109/9781420074239.
Gohel MC, Parikh RK, Nagori SA, Jena DG. Fabrication of modified release tablet formulation of metoprolol succinate using hydroxypropyl methylcellulose and xanthan gum. AAPS PharmSciTech. 2009 Mar 16;10(1):62-8. doi: 10.1208/s12249-008-9174-1, PMID 19148762.
Gannu R, Palem CR, Yamsani SK, Yamsani VV, Yamsani MR. Enhanced bioavailability of buspirone from reservoir-based transdermal therapeutic system optimization of formulation employing box–behnken statistical design. AAPS PharmSciTech. 2010 Jun 2;11(2):976-85. doi: 10.1208/s12249-010-9451-7, PMID 20517714.
Kawashima Y, Niwa T, Takeuchi H, Hino T, Itoh Y. Hollow microspheres for use as a floating controlled drug delivery system in the stomach. J Pharm Sci. 1992 Feb;81(2):135-40. doi: 10.1002/jps.2600810207, PMID 1372046.
Yadav A, Jain DK. In vitro characterization of gastroretentive microballoons prepared by the emulsion solvent diffusion method. J Adv Pharm Technol Res. 2010;1(1):56-67. doi: 10.4103/2231-4040.70524, PMID 22247832.
Vanaja K, Shobha Rani RH. Design of experiments: concept and applications of plackett burman design. Clin Res Regul Aff. 2007 Jan 10;24(1):1-23. doi: 10.1080/10601330701220520.
Kharb M, Tanwar YS. Development and statistical optimization of Gastroretantive floating microspheres of pregabalin prepared by W/O/O multiple emulsion method. Int J Appl Pharm. 2021 May 7;13(3):199-206. doi: 10.22159/ijap.2021v13i3.41001.
Awasthi R, Kulkarni GT. Development and characterization of amoxicillin loaded floating microballoons for the treatment of Helicobacter pylori induced gastric ulcer. Asian J Pharm Sci. 2013 Jun;8(3):174-80. doi: 10.1016/j.ajps.2013.07.023.
Awasthi R, Kulkarni GT, Pawar VK, Garg G. Optimization studies on gastroretentive floating system using response surface methodology. AAPS PharmSciTech. 2012 Mar 2;13(1):85-93. doi: 10.1208/s12249-011-9730-y, PMID 22135108.
Alnaqbi AY, Bhongade BA, Azzawi AA. New validated diffuse reflectance infrared fourier transform spectroscopic method for the quantification of levofloxacin in pharmaceutical dosage form. Indian J Pharm Sci. 2021;83(3):430-6. doi: 10.36468/pharmaceutical-sciences.791.
Kadri L, Carta M, Lampronti G, Delogu F, Tajber L. Mechanochemically induced solid-state transformations of levofloxacin. Mol Pharm. 2024 Jun 3;21(6):2838-53. doi: 10.1021/acs.molpharmaceut.4c00008, PMID 38662637.
Kumar V, Sodavat RK, Rathore GS. Formulation and evaluation of gastroretentive floating tablets of lovastatin using natural polymers. J Drug Deliv Ther. 2025 Jul 15;15(7):71-9. doi: 10.22270/jddt.v15i7.7280.
Varshosaz J, Tabbakhian M, Zahrooni M. Development and characterization of floating microballoons for oral delivery of cinnarizine by a factorial design. J Microencapsul. 2007 May;24(3):253-62. doi: 10.1080/02652040601162723, PMID 17454436.
Choudhary S, Jain A, Amin MC, Mishra V, Agrawal GP, Kesharwani P. Stomach specific polymeric low density microballoons as a vector for extended delivery of rabeprazole and amoxicillin for treatment of peptic ulcer. Colloids Surf B Biointerfaces. 2016 May;141:268-77. doi: 10.1016/j.colsurfb.2016.01.048, PMID 26859118.
Jain A, Pandey V, Ganeshpurkar A, Dubey N, Bansal D. Formulation and characterization of floating microballoons of nizatidine for effective treatment of gastric ulcers in murine model. Drug Deliv. 2015 Apr 3;22(3):306-11. doi: 10.3109/10717544.2014.891273, PMID 24601855.
Sato Y, Kawashima Y, Takeuchi H, Yamamoto H. In vitro and in vivo evaluation of riboflavin-containing microballoons for a floating controlled drug delivery system in healthy humans. Int J Pharm. 2004 May;275(1-2):97-107. doi: 10.1016/j.ijpharm.2004.01.036, PMID 15081141.
Ramachandran S, Shaheedha SM, Thirumurugan G, Dhanaraju MD. Floating controlled drug delivery system of famotidine loaded hollow microspheres (microballoons) in the stomach. Curr Drug Deliv. 2010 Jan 1;7(1):93-7. doi: 10.2174/156720110790396436, PMID 20044907.
International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH). Stability testing of new drug substances and products. ICH Q1A (R2). Geneva: ICH; 2003 Feb 6.
Boorugu R, Gadela RV. Eplerenone floating microspheres: radiographic and pharmacokinetic studies in rabbits. Int J Appl Pharm. 2024;16(2):231–6. doi: 10.22159/ijap.2024v16i2.49798.
Krishna SR, Ramu A, Vidyadhara S, Rani AP. Bioavailability enhancement by floating microballoons of dipyridamole and clopidogrel: in vivo pharmacokinetic study. Int J Appl Pharm. 2021;13(6):216–20. doi: 10.22159/ijap.2021v13i6.42316.
Mc Hugh ML. Multiple comparison analysis testing in ANOVA. Biochem Med (Zagreb). 2011;21(3):203-9. doi: 10.11613/BM.2011.029, PMID 22420233.
Maurya S, Malik JK, Kumar GP, Kumar S, Singh G, Singh SP. Preparation and evaluation of chitosan microspheres containing levofloxacin. Saudi J Med Pharm Sci. 2024 Feb 1;10(2):99-104. doi: 10.36348/sjmps.2024.v10i02.006.
Spizzirri UG, Hampel S, Cirillo G, Mauro MV, Vittorio O, Cavalcanti P. Functional gelatin-carbon nanotubes nanohybrids with enhanced antibacterial activity. Int J Polym Mater Polym Biomater. 2015 Sep 22;64(9):439-47. doi: 10.1080/00914037.2014.958833.
Perissutti B, Rubessa F, Moneghini M, Voinovich D. Formulation design of carbamazepine fast-release tablets prepared by melt granulation technique. Int J Pharm. 2003 Apr;256(1-2):53-63. doi: 10.1016/S0378-5173(03)00062-0, PMID 12695011.
Mozayad AN, Fouad MA, Elkady EF. Utilizing experimental design and desirability function in optimizing RP-HPLC method for simultaneous determination of some skeletal muscle relaxants and analgesics. Sci Rep. 2024 May 6;14(1):10360. doi: 10.1038/s41598-024-58381-4, PMID 38710733.
Krishna KA, Srivastava R. In vitro-in vivo studies on floating microspheres for gastroretentive drug delivery system: a review. Asian J Pharm Clin Res. 2021 Jan 5;14(1):13-26. doi: 10.22159/ajpcr.2021.v14i1.39183.
Beg S, Kaur R, Khurana RK, Rana V, Sharma T, Singh B. QbD-based development of cationic self-nanoemulsifying drug delivery systems of paclitaxel with improved biopharmaceutical attributes. AAPS PharmSciTech. 2019 Apr 21;20(3):118. doi: 10.1208/s12249-019-1319-x, PMID 30790136.
Parmar A, Kaur G, Kapil S, Sharma V, Sharma S. Central composite design-based optimization and fabrication of benzylisothiocynate-loaded PLGA nanoparticles for enhanced antimicrobial attributes. Appl Nanosci. 2020 Feb 23;10(2):379-89. doi: 10.1007/s13204-019-01185-0.
Senthilnathan B, Maheswaran A, Gopalasatheeskumar K, Masilamani K, Edros RZ. Formulation and evaluation of pregabalin loaded Eudragit S100 nanoparticles. Int J Eng Technol Sci. 2016 Dec 30;3(2):64-70. doi: 10.15282/ijets.6.2016.1.9.1059.
Hasan AA, Madkor H, Wageh S. Formulation and evaluation of metformin hydrochloride-loaded niosomes as controlled release drug delivery system. Drug Deliv. 2013 Apr 8;20(3-4):120-6. doi: 10.3109/10717544.2013.779332, PMID 23651102.
Reddy LH, Murthy RS. Floating dosage systems in drug delivery. Crit Rev Ther Drug Carrier Syst. 2002;19(6):553-85. doi: 10.1615/CritRevTherDrugCarrierSyst.v19.i6.20, PMID 12822735.
Saini S, Bhardwaj BY, Chhabra J, Kumar M, Pahwa R. In vivo monitoring strategies for evaluation of floating drug delivery systems. Int J Appl Pharm. 2022 Nov 7;14(6):28-33. doi: 10.22159/ijap.2022v14i6.45906.