
Int J App Pharm, Vol 18, Issue 2, 2026, 72-83Original Article
IN SITU GELLING OCULAR FILMS OF LORATADINE: A MUCOADHESIVE PLATFORM FOR ENHANCED ANTIHISTAMINIC OCULAR DELIVERY
RASHA HAMEED AL-KAABY, LENA MURAD THOMAS*
Department of Pharmaceutics, College of Pharmacy, University of Baghdad, Baghdad-10071, Iraq
*Corresponding author: Lena Murad Thomas; *Email: linatomas@copharm.uobaghdad.edu.iq
Received: 27 Aug 2025, Revised and Accepted: 01 Jan 2026
ABSTRACT
Objective: The objective of this study was to prepare an in situ gelling, mucoadhesive ocular film of loratadine, with extended precorneal retention and sustained release.
Methods: The films were formulated using the solvent casting method, using hydroxypropyl methylcellulose K4M (HPMC K4M) as a film-forming polymer, sodium hyaluronate (SH), sodium alginate (SA), polyvinylpyrrolidone K30 (PVP K-30) as mucoadhesive polymers, and propylene glycol (PG) as a plasticizer.
Results: Among the fourteen formulations prepared (F1-F14), formula (F5), containing 1% HPMC K4M and 0.75% SA, exhibited high drug content (99.11±3.57 %), suitable pH (7.03±0.09), thickness (0.080±0.008 mm) and tensile strength (3.44±0.11 N/mm2). The film quickly hydrated to form a gel-like structure with proper mucoadhesion (4.436±0.09 N), and in vitro dissolution studies showed sustained drug release (99.43% over 12 h) that best fitted Korsmeyer–Peppas model, suggesting Fickian diffusion. The developed film showed no cytotoxicity or irritation to eyes in studies conducted on rabbits.
Conclusion: The optimized in situ gelling loratadine ocular film has desirable properties, and is promising for long-term topical ocular delivery of loratadine.
Keywords: Loratadine, Ocular drug delivery, In situ gelling film, Solvent casting method, Sustained release, Mucoadhesive film
© 2026 The Authors.Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.56656 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Ocular allergy is a focal eye condition affecting diverse populations with a markedly heightened incidence in recent years. Various factors, including dust, pollen, and petal lergens can trigger it. It has many manifestations that may be related to allergic rhinitis, such as itchy, irritated, red, and tearful eyes, which can significantly affect a patient's quality of life [1].
Topical ocular drug delivery is a commonly used approach for treating various ocular conditions and diseases, including allergies. Using traditional ocular dosage forms, such as eye drops and ointments, represents the easiest method of ocular drug delivery [2]. However, due to ocular physiological barriers, rapid drug dilution and removal from the site of application by lacrimal fluid and blinking, a short precorneal contact time is a major limitation that reduces the absorption of drugs and their duration, thereby necessitating a frequent dosing regimen to achieve effective therapeutic delivery to the eye [3].
Ocular films represent one alternative approach to overcoming the aforementioned limitations. These are sterile, solid, or semi-solid polymeric dosage forms, intended for application in the conjunctival sac. They permit accurate dosing, a more rapid onset of action, reduced systemic side effects, increased residence time in the eye, providing the required concentration at the site of action, and improving therapeutic effectiveness. This enables the achievement of sustained drug release and a decrease in dosing frequency, thereby increasing patient compliance. Nevertheless, some films may cause initial foreign body sensation, and those made using non-biodegradable polymers require removal once the drug has been depleted. Such inconvenience is overcome by using soluble, biocompatible polymers, which enable ocular film conversion to into a gel-like material with no need for their removal [4, 5].
Loratadine, a second-generation antihistamine, is commonly used for the treatment of urticaria, rhinitis, and conjunctivitis. According to the Biopharmaceutical Classification System (BCS), it is classified as a class II drug, having limited water solubility and high permeability. Moreover, it undergoes first-pass metabolism and has a pH-dependent solubility due to its mild basicity (pKa of 5.2) [6]. Topical ocular delivery of loratadine provide targeted, rapid and localized relief for symptoms of allergic conjunctivitis, while minimizing systemic side effects. Despite the favorable permeability of loratadine due to its inherent lipophilic nature, its poor solubility remains a major challenge for achieving therapeutically effective ocular concentrations upon topical use. Several formulation strategies have been investigated to increase its solubility. Among these are pluronic/carbomer-based thermo-responsive hydrogels, which undergo a sharp sol-to-gel transition when warmed by body temperature [7]. Loratadine-β-cyclodextrin complex have also been utilized in the development of loratadine gel and ocusert [8, 9]. Nanostructured lipid carriers embedded in a gel matrix (Ocugel) were also formulated, with significantly enhanced solubility and trans-corneal penetrability [10]. Although these systems improved solubility, limitations such as inaccurate dosing, blurred vision, and patient inconvenience still persist.
Polymeric blends are commonly used in various drug delivery systems, including ophthalmic films, to customize or improve film mechanical properties, ocular adhesion, and retention time, as well as to modify the drug release rate for controlled release applications [11]. Hydroxypropyl methylcellulose (HPMC) is a safe, biocompatible, linear, non-ionic cellulose polymer well-known for its high transparency, effective matrix-forming, and gelling abilities. It has versatile uses as a lubricating agent and a viscosity enhancer, in addition to its use in ocular controlled-release delivery systems, such as gels, inserts, or films [12-14]. However, it has a slow rate of hydration and moderate mucoadhesive properties, which can be enhanced by blending with other hydrophilic polymers to achieve the desired goal of the formulation or delivery system [15, 16]. Polyvinylpyrrolidone (PVP), a non-ionic, synthetic tertiary amide, is known for its high-water solubility, biocompatibility, and mucoadhesive properties. It has been used in ocular drug delivery systems as a film-forming agent and a component in contact lenses, where it acts as a lubricant or wetting agent, or in controlled drug delivery systems to modulate drug release kinetics [17, 18].
Sodium hyaluronate (SH) and sodium alginate (SA) represent two typical examples of natural, biodegradable polymers, well-recognized in literature for their hydrophilic and mucoadhesive properties, with wide application in a variety of ocular preparations [19, 20]. These polymers have the potential to provide a synergistic effect with HPMC, particularly in terms of flexibility, bioadhesion, and provide appropriate drug release kinetics [21, 22].
In this study, we propose the development of in situ gelling films of loratadine that offer drug solubilization using ethanol, with ease of production of a solid, accurate unit-dose drug delivery system using mucoadhesive, biocompatible soluble polymers that have the potential to achieve extended contact with ocular surfaces and provide prolonged drug release, thereby reducing the frequency of administration To the best of our knowledge, this is the first description of an ocular film that combines loratadine with the capacity to gel in situ, which adds the benefit of continuous release with the lack of need for removal by the patient as compared to solid ocular inserts. This makes it a promising ocular delivery substitute for lipid nanocarriers, hydrogels, ocuserts, and gels that have been previously studied.
MATERIALS AND METHODS
Materials
Loratadine was received as a gift from Sama Al Fayhaa Pharmaceutical Industries (Basrah, Iraq). HPMC K4M was purchased from Baoji Guokang Bio-Technology Co. Ltd (Shaanxi, China), hyaluronic acid sodium salt (M. WT 8000-15000 DA) was purchased from Shanghai BiDe Pharmaceutical Technology Co., Ltd. (Shanghi, China), sodium alginate was purchased from Avonchem Diagnostics Limited (UK), polyvinyl pyrrolidine K30 was purchased from Himedia Laboratories (Mumbai, India), porcine mucin was purchased from Beijing Jin Ming Biotechnology Co., Ltd. (Beijing, China), ethanol was purchased from Alpha Chemika (Mumbai, India), Tween 20® was purchased from Loba Chemie Pvt. Ltd. (Mumbai, India). Propylene glycol and the remaining chemicals used in the study were obtained from Thomas Baker Chemicals Pvt. Ltd (Mumbai, India).
Preparation of ocular films
Several ocular films of loratadine were prepared by the solvent casting method [23]. HPMC K 4M was used as a film-forming polymer, while SH, SA, or PVP K-30 were used as bioadhesive polymers as per composition given in table 1.
Each polymer was formulated as a 4% w/v stock solution and left overnight to allow complete hydration and swelling of the polymer. A stock solution (1% w/v) of the drug was also prepared by dissolving loratadine in ethanol. Films were prepared by mixing calculated volumes of the polymer solutions to obtain required concentration and 10% v/v PG used as a plasticizer. The mixture was stirred at 500 rpm using a magnetic stirrer (MHS-6, Witeg Labortechnik GmbH, Korea) for 30 min to allow homogeneity. Then, one milliliter of 1% loratadine ethanolic solution was added so that the final ethanol concentration in the film was 10% (v/v), and the total volume was made up to 10 ml with distilled water. Magnetic stirring was performed for 30 min to ensure homogenization, followed by sonication of the formulation mixtures for an additional 30 min in an ultrasonic water bath (Copley Scientific, England).
The prepared viscous dispersion was poured into a glass Petri dish (8 cm diameter) and left to dry at room temperature for 48 h. Dried films were carefully peeled off the Petri dish and cut into circular discs (7 mm diameter), each containing 76.5 µg of loratadine. The dry films were stored in aluminum foil and kept in a dry place until further evaluation.
Table 1: Composition of different formulations of loratadine ocular films*
| Code | HPMC K4M (%) | SH (%) | SA (%) | PVP K30 (%) | Loratadine (%) |
| F1 | 1 | 0.75 | 0.1 | ||
| F2 | 1.5 | 0.25 | 0.1 | ||
| F3 | 1.5 | 0.5 | 0.1 | ||
| F4 | 1.5 | 0.75 | 0.1 | ||
| F5 | 1 | 0.75 | 0.1 | ||
| F6 | 1.5 | 0.75 | 0.1 | ||
| F7 | 1.5 | 1 | 0.1 | ||
| F8 | 1.5 | 1.5 | 0.1 | ||
| F9 | 1.5 | 2 | 0.1 | ||
| F10 | 1 | 0.75 | 0.1 | ||
| F11 | 1.5 | 0.75 | 0.1 | ||
| F12 | 1.5 | 1 | 0.1 | ||
| F13 | 1.5 | 1.5 | 0.1 | ||
| F14 | 1.5 | 2 | 0.1 |
*All formulations contain 10% (v/v) PG as plasticizer.
Evaluation of ocular films
Organoleptic properties, weight, thickness, surface pH, and drug content
The prepared formulations were evaluated for film-forming ability and ease of peeling. Formed films were visually inspected for clarity (rated as opaque, semi-transparent, or transparent) by examining them through a numbered ruler and taking digital images [24, 25].
Film thickness was measured at different locations, including the center, using a digital micrometer [26]. The folding endurance was assessed by placing ocular film between the fingers and thumb, with successive folding and unfolding in the same position at the center, until it broke or up to 300 times, whichever came first. Films that can be folded more than 300 times without cracking or breaking are considered successful [27].
The pH of the formulations was measured with a digital pH meter (Hanna Instrument, Italy) at room temperature. Prepared film formulations were kept in contact with 1 ml distilled water for 15 min. Then, the pH meter electrode was placed close to the film surface for 1 min without disturbance, and pH was recorded [28].
To determine uniformity of weight, for each formulation batch, three films cut from different areas were weighed individually using an analytical balance. The average weight and standard deviation were recorded [29]. For drug content determination, three films were randomly selected from each formulation, cut into small pieces, and soaked in 10 ml ethanol overnight to extract the drug. The dispersion was filtered using a 0.45 µm filter syringe, and the absorbance of loratadine was recorded using a UV spectrophotometer (Shimadzu UV mini-1240, Japan) at λmax of 247 nm, with ethanol as the blank. The results were expressed as the average of three measurements with standard deviation [30].
Mechanical properties
The mechanical properties of films, including tensile strength and elongation at break, were determined using a tensile strength testing machine (HZ-1007A). A rectangular film strip (10 x 20 mm) was held between the two grips of the machine and pulled at a rate of 50 mm/min until it was torn. Film thickness was measured using a digital micrometer before each measurement. Tests were triplicated, and average values with standard deviation were reported [31]. The weight causing films to break was recorded as force at break and was used in the measurement of tensile strength using Equation (1):
Tensile strength (N/mm2) = (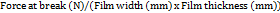
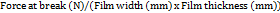 …. (1)
…. (1)
Percentage elongation at break was calculated according to Equation (2):
% Elongation at break = 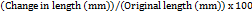
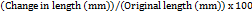 …. (2)
…. (2)
Film-swelling behavior
To determine the swelling index, each film was weighed, placed into a basket, and inserted into a 20 ml phosphate buffer (pH 7.4, 34±0.5 °C) in a Petri dish. The basket was removed at specified time intervals, up to 30 min, and placed on filter paper to remove excess buffer medium. It was then reweighed and returned to the same media [32]. The swelling index was calculated according to Equation (3):
Swelling index (%) 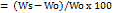
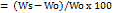 …. (3)
…. (3)
where Ws represents the weight of the hydrated film at each time interval, and Wο is the original weight of the dry film.
In vitro muco-adhesion test
Mucoadhesion of ocular films was measured as the force required to detach the films from a mucin disk using a texture analyzer (TA-XT plus, Stable Micro Systems, Godalming, UK) set in adhesive mode with a 5 Kg load cell using a cylindrical TA-probe. Mucin disks were prepared by compressing 500 mg mucin powder into a 15 mm diameter disk using a tablet machine (Erweka, Germany) and used as the adhesive surface. Each film was cut into a 20 mm circular disk and attached to the base of the probe using double-sided adhesive tape, then positioned at the mobile arm of the texture analyzer. Phosphate buffer (pH 7.4, 10 µl) was added to the mucin disk using a micro pipette. The probe was activated and the following parameters were set: applied force (0.01 N), pre-test speed (0.1 mm/s), test speed (1.0 mm/s), and contact time (60 sec). The used contact time is necessary to permit the creation of hydrogen bonds and other non-covalent interactions. The test was conducted until the film detached from the mucin disk, and the force required to detach the film was recorded and expressed as the force of adhesion (in Newtons, N). All measurements were performed in triplicate [33].
In vitro drug release and kinetic study
The in vitro drug release profile of ocular films was determined using a vertical Franz diffusion cell with a 7 ml receptor compartment. A dialysis membrane (molecular weight cutoff of 12,000-14,000 Dalton) was hydrated overnight with phosphate buffer (pH 7.4) before being cut into pieces and mounted between the receptor and donor compartments. Previously weighed ocular film was placed on the membrane. The receptor compartment was filled with phosphate buffer (pH 7.4) containing 1% Tween 20 in order to overcome the poor solubility of loratadine and maintain sink conditions, magnetically stirred at 50 rpm, with a temperature of 35±1 °C. A sample of 1 ml was withdrawn through the sampling port from the receptor compartment at specified time intervals up to 12 h, and replaced with an equal volume of prewarmed media after each sampling time to maintain a constant volume. The sink condition was maintained, and the collected samples were filtered through a 0.45 µm filter syringe. The samples were then analyzed spectrophotometrically at λmax 250 nm using phosphate buffer (pH 7.4) containing 1% Tween 20 as a blank. The cumulative amounts of loratadine released were calculated and plotted against time to produce the release profiles [34, 35].
To understand the drug release mechanism, dissolution data from the prepared ocular films were fitted to various kinetic equations, including zero-order, first-order, Higuchi, and Korsmeyer–Peppas equations. The goodness of fit of drug release was evaluated by the determination coefficient (R2) value [36]. The DDSolver software (DDSolver: An Excel Add-In Program for Modeling and Comparison of Drug Dissolution Profiles) was employed for this analysis [37].
Sterilization and sterility test
The selected ocular film was transferred into an 8 mm diameter Petri dish and placed in an ultraviolet sterilizer cabinet (Model SM-208, China). The film samples were irradiated with a short-wavelength UV beam (254 nm) on both sides for 15 min and subsequently were packaged in pre-sterilized aluminum foil. To confirm that UV exposure was effective in overcoming viable forms of bacteria and fungi, the formulated ocular film was subjected to sterility testing using the direct inoculation method with two culture media: fluid thioglycolate medium (FTGM) and soya bean-casein digest (SBCD) medium. Formulation was incubated for 5 d in FTGM at 30-35 °C and 3 d in SBCD at 20-25 °C. The tests were conducted on sterilized films designated as "Test", positive control (growth promotion), and negative control (sterility), and observations were recorded [38, 39].
Cytotoxicity study
In vitro cytotoxicity evaluation of selected ocular film was carried out using Normal Human Fibroblast (HNF) cell line, and percentage cell viability of both blank and medicated films was determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide compound (Elabscience, China) (MTT assay). According to ISO10993-5 recommendations, HNF cells were cultured in Minimum Essential Medium (MEM) (US Biological, USA) at a density of 3 × 105 cells/ml. The cultured cells were implanted in 96-well microplates (NEST Biotech, China) (100 μl/well) and incubated at 37 °C for 72 h until a monolayer confluence was achieved. Sterilized loratadine and blank films were placed in the wells containing culture media to be in contact with the cells and incubated for 24 h. The control (cells without ocular films) was a row of 96-well plates. After the incubation, MTT solution (28 µl of 2 mg/ml stock solution) was added to the media in each well, and the cells were incubated for an additional three hours. Subsequently, 100 μl of dimethyl sulfoxide (DMSO) was added to each well with gentle agitation to dissolve the purple formazan crystals, and the mixture was incubated for 15 min. Absorption was measured at 570 nm using a microplate reader (Gennex Lab, USA). All experiments were carried out in triplicate (mean±SD, n = 3). The percentage of cell viability was calculated using Equation (4) [40]:
Cell viability (%) =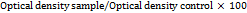
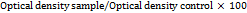 …. (4)
…. (4)
Ocular irritancy study and histological examination
A selected and sterilized ocular film was used for conducting the irritation study. The preclinical study protocol was approved by the Institutional Animal Ethical Committee (number: REC032624102R). Four male albino rabbits, weighing between 1.5 and 2 kg, were fed with green vegetables throughout the study. Selected sterilized ocular film was placed in the cul-de-sac of the rabbit's left eye, and the right eye served as a control. The eye was visually observed for 3 d for the presence of redness, tearing, and swelling. At the end of the test, the rabbits were sacrificed, and their eyes were excised, washed with phosphate buffer solution, fixed in a 10% (w/v) formalin solution for 72 h, dehydrated with alcohol, and embedded in melted paraffin. Tissues were sectioned into 5-μm-thick slices, placed on glass slides, and stained with hematoxylin and eosin for examination using a light microscope [41, 42].
Statistical analysis
The results are expressed as the mean±standard deviation. Statistical analysis was performed to assess the % swelling, adhesion force, % tensile strength, and in vitro drug release. Data were compared using GraphPad Prism software (version 8, GraphPad, USA) using a one-way analysis of variance (ANOVA) test followed by Tukey's multiple comparisons tests. Statistical significance is indicated by (*p<0.05, **p<0.01, ***p<0.001, and ****p<0.0001).
RESULTS AND DISCUSSION
Organoleptic properties, weight, thickness, surface pH, and drug content
Formulation of loratadine as in situ gelling ocular films offers a unique advantage over gels by eliminating dosing variability and allowing for accurate unit-dose administration. Additionally, compared to traditional solid ocular inserts, the film utilizes soluble, biocompatible polymers that enable it to convert into a gel-like material in-situ. This overcomes a major drawback of inserts, which reside in the need for removal, and thereby enhances patient compliance.
All 14 formulations peeled easily and had a smooth surface with no visible cracks, indicating a good film-forming ability with homogeneous distribution of all components. This demonstrates the adequacy of the solvent casting method for film preparation, which is considered a simple method compared to previously reported nanotechnology methods employed to prepare topically applied loratadine [10].
Visual inspection of the films (fig. 1) indicates that all films were clear and transparent, apart from those containing SA (F5-F9), which had a slightly yellowish color, mainly due to the faint color of SA. In our study, transparency is not considered a deciding factor for selection because the site of application is at the cornea or cul-de-sac, rather than the pupil, and thus vision will not be disturbed.

Fig. 1: Digital photographs of loratadine films containing (A) sodium hyaluronate, (B) sodium alginate, (C) PVP, and (D) appearance of circular discs cut from films
All films had a thickness ranging from 0.0525±0.01 to 0.128±0.009 mm, with low standard deviation values indicating uniformity. The average weight ranged from 7.6±0.282 to 10.7±0.163 mg, as shown in table 2.
Table 2: Evaluation parameters of loratadine ocular films*
| Code | Thickness (mm) | Weight (mg) | Drug content (%) | Surface pH |
| F1 | 0.106±0.008 | 8.66±0.205 | 99.22±3.86 | 7.13±0.04 |
| F2 | 0.072±0.016 | 7.60±0.282 | 97.05±2.65 | 7.06±0.04 |
| F3 | 0.102±0.009 | 7.73±0.047 | 94.34±2.83 | 7.06±0.04 |
| F4 | 0.112±0.007 | 9.50±0.163 | 95.31±1.80 | 7.06±0.04 |
| F5 | 0.080±0.008 | 9.20±0.163 | 99.11±3.57 | 7.03±0.09 |
| F6 | 0.088±0.017 | 9.63±0.235 | 94.55±2.31 | 7.03±0.09 |
| F7 | 0.116±0.013 | 10.30±0.216 | 101.38±1.21 | 7.36±0.04 |
| F8 | 0.118±0.009 | 10.46±0.124 | 100.30±3.97 | 7.43±0.04 |
| F9 | 0.128±0.009 | 10.70±0.163 | 98.24±1.80 | 7.43±0.04 |
| F10 | 0.052±0.01 | 8.30±0.163 | 99.65±1.33 | 7.16±0.04 |
| F11 | 0.084±0.004 | 8.33±0.262 | 97.26±2.17 | 7.16±0.04 |
| F12 | 0.086±0.004 | 8.36±0.169 | 97.81±1.47 | 6.90±0.08 |
| F13 | 0.090±0.006 | 9.06±0.169 | 102.14±2.12 | 6.90±0.08 |
| F14 | 0.094±0.01 | 9.30±0.081 | 96.07±1.08 | 6.90±0.08 |
Results are given as mean±SD, n=3.
The difference in weight and thickness for each formula had low standard deviation values, indicating film uniformity. Variations among different formulations are due to differences in the type and concentrations of polymers used. Both thickness and weight were found to increase with polymer concentration. However, differences were found to be non-significant (p>0.05) and, as per the literature, the obtained values were suitable for application to the eye without discomfort [43, 44].
Prepared films exhibited acceptable drug content, ranging from 94.34±2.83% to 102.14±2.12%, and were within pharmacopeial limits (85–115%), indicating uniform and homogeneous drug distribution within the polymeric matrix attained by solubilizing the drug, and implying a reliable method of preparation [45].
Surface pH measurements yielded values ranging from 6.9±0.08 to 7.43±0.04, which were compatible with the physiological pH of the eye surface and suggested that the prepared films would not cause ocular irritation [46].
Mechanical properties and folding endurance
Prepared films are intended to be applied to the eyes with hands, and therefore, they should be flexible and have proper strength for ease of handling and application. All films exhibited values of folding endurance exceeding 300, indicating film flexibility. For a more precise quantitative evaluation of mechanical properties, tensile strength (N/mm²) as a measure of breakability and elongation at break (%) as an indicator of film flexibility and elasticity were recorded. The obtained values are summarized in table 3.
Table 3: Mechanical parameters of prepared ocular films*
| Code | Folding endurance | Tensile strength (N/Mm2) | % Elongation |
| F1 | >300 | 3.32±0.12 | 96.90±3.34 |
| F2 | >300 | 7.83±0.17 | 143.36±3.93 |
| F3 | >300 | 5.16±0.11 | 111.90±3.92 |
| F4 | >300 | 4.80±0.14 | 122.56±5.92 |
| F5 | >300 | 3.44±0.11 | 111.40±3.93 |
| F6 | >300 | 6.25±0.08 | 121.75±5.48 |
| F7 | >300 | 5.14±0.07 | 138.19±2.58 |
| F8 | >300 | 4.62±0.07 | 123.65±3.63 |
| F9 | >300 | 3.35±0.07 | 153.87±4.11 |
| F10 | >300 | 4.42±0.09 | 124.85±2.92 |
| F11 | >300 | 4.95±0.10 | 84.84± 5.20 |
| F12 | >300 | 5.43±0.06 | 162.99±3.84 |
| F13 | >300 | 7.03±0.09 | 130.52±5.04 |
| F14 | >300 | 10.42±0.08 | 125.40±2.97 |
Results are given as mean±SD, n=3.
The tensile strength of formulations F1-F14 ranged from 3.32±0.12 N/mm2 to 10.42±0.08 N/mm2, and elongation at break (%) ranged from 84.842±5.20% to 162.99±3.84%. There is no specific range for optimum tensile strength for ocular films. Formulated films were sufficiently durable and flexible for handling, exhibiting better mechanical properties than those reported in the literature [47, 48]. Many reports indicate that the mechanical properties of the polymer blends in films are strongly affected by their composition and concentration of polymers used, and are improved with intermolecular interactions [49, 50]. Our obtained data indicate that increasing the concentration of film-forming polymer (HPMC K4M) in the polymer blends increases the tensile strength, as evident when comparing formulations F4, F6, and F11, which contain 1.5% HPMC, with formulations F1, F5, and F10, which contain 1% HPMC. This could be due to denser packing and higher polymer chain intertwining, which provides additional resistance to deformation, contributing to the enhanced tensile strength [51].
Amongst the three types of polymeric blends produced using different mucoadhesive polymers, the tensile strength of HMPC/PVP films (F11-F14) was highest and significantly increased when using higher concentrations of PVP; this could be due stronger intermolecular linkages between the similar PVP polymers as HPMC amount is reduced, and hence reduction in the space for sliding and stretching of polymeric chains, producing stronger films with lower percentage of elongation at break (%EB) [52]. For films produced with HPMC/SH (F2-F4) or HPMC/SA (F6-F9) polymeric blends, lower values of tensile strength were observed, primarily due to the lower molecular weight of these biopolymers compared to PVP, and the fact that biopolymers generally have low mechanical strength [53]. Moreover, a reduction in tensile strength was observed upon increasing the concentrations of SH or SA in the blend. This effect can be attributed to the ability of these polymers to promote structural discontinuity between HPMC chains. A similar observation was reported previously by Ghosal et al. [54].
The use of a relatively high concentration of PG, which served as a plasticizer in all formulations, contributed to the enhanced flexibility of the prepared films, as indicated by high values of % elongation at break. This can be attributed to the role of the plasticizer in intervening between polymeric chains and reducing their interaction and cohesion, making them less brittle. It has been reported that incorporating different plasticizers affected film blends of HPMC and caused significant changes in mechanical properties [55].
Although the relationship between tensile strength and elongation at break in general is inverse, it is not always the case for blends of different polymers, as the interaction among polymers affects mechanical performance [56].
Film-swelling behavior
The swelling behavior of films varies according to their composition, as it depends on the ability of the polymers to hydrate and retain fluids. This is considered important since excessive swelling can lead to discomfort, reduce mucoadhesion, and influence the drug dissolution profile [57]. The swelling behavior was investigated for all formulations, and different degrees of swelling were obtained as shown in fig. 2.
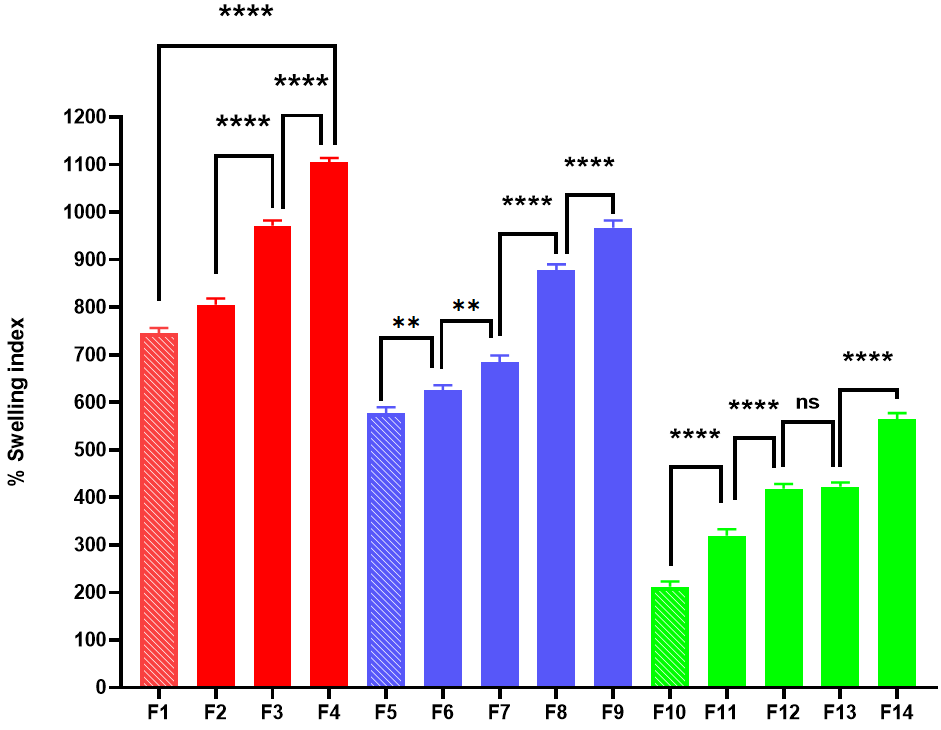
Fig. 2: Swelling index of loratadine ocular films (mean±SD, n = 3, *p<0.05, **p<0.01, ***p<0.001, and ****p<0.0001)
The % swelling was found to be minimum in formulation F10 (212.33±9.03 %) containing HPMC/PVP blend and maximum in formulation F4 (1104.66±7.58 %) containing HPMC/SH blend. Rapid swelling was observed in all formulations due to the hydrophilic nature of the polymers used and the high content of PG used as a plasticizer, which enhanced water absorption. As illustrated in fig. 2,% swelling index of HPMC/SH films increases from F1 to F4 with a statistically highly significant difference (p<0.0001) with each step-up (F1 to F2, F2 to F3, F3 to F4). Similarly, for HPMC/SA films, comparing consecutive formulations F5 to F9 showed a highly statistically significant difference (p<0.01) when comparing F5 to F6 or F6 to F7 and (p<0.0001) when comparing formulations F7 to F8 or F8 to F9. Finally, the % swelling index in HPMC/PVP formulations is lower overall compared to the others, and swelling increased in formulations F10 to F14 when compared to the preceding formulation with a statistically highly significant difference (p<0.0001) except for formulation F12 and F13, where the observed increase in % swelling was non-significant (p>0.05).
HPMC K4M is an amorphous hydrophilic polymer having hydroxyl groups in its structure, and the presence of hydrophilic bioadhesive polymers had a synergistic effect with HPMC K4M in enhancing swelling mainly due to high water penetration into the polymer matrix; this effect was more evident at higher concentrations due to more abundant sites for interaction with water [58] as evident when comparing formulations F4, F6, F11 containing 1% HPMC to formulations F1, F5, F10, containing 1.5 % HPMC K4, combined with SH, SA, and PVP as mucoadhesive polymers, respectively.
Both SH and SA are highly ionized at pH 7.4, having a high number of negatively charged carboxylate ions along their polymer backbone. This causes repulsion and polymer chain expansion, which enables more water penetration into the polymer matrix. However, the higher swelling ability of SH is related to its linear and less-structured chain, which allows for more free chain expansion and provides a higher swelling capacity compared to the rigid "egg-box" model of alginate, known for its ability to cross-link with cations [59-61]. As for HPMC/PVP-containing films, the lowest values of swelling observed can be attributed to the relatively limited space available for their chains to expand freely in the presence of other polymers. This finding was consistent with observations reported by El Gamal et al. [62].
In vitro muco-adhesion test
The mucoadhesive property of films has a significant impact on their therapeutic efficacy as it prolongs attachment duration. In vivo adhesion of ocular films to mucosal surfaces involves contact, hydration of films with mucin covering the cornea and the cul-de-sac area of the eye to promote interactions with mucin. Such a process is influenced by the type of polymer, concentration, molecular weight, chain flexibility, surface charge density, and ability to form non-covalent bonds with mucin, in addition to the influence of the surrounding physiological environment [63]. Using biodegradable, mucoadhesive, and hydrophilic polymers in the formulation of ocular films is advantageous, since applied films will not require removal after use [64].
All prepared films were evaluated for their bioadhesive ability by measuring their force of adhesion. Obtained values ranged from 1.179±0.052 N to 6.684±0.06 N, as shown in fig. 3.
The adhesive force is highest in the HPMC/SA (F5-F9) formulations, peaking at F9. The lowest force of adhesion is observed in the HPMC/PVP (F11 and F12) formulations, which are not significantly different (p>0.05).
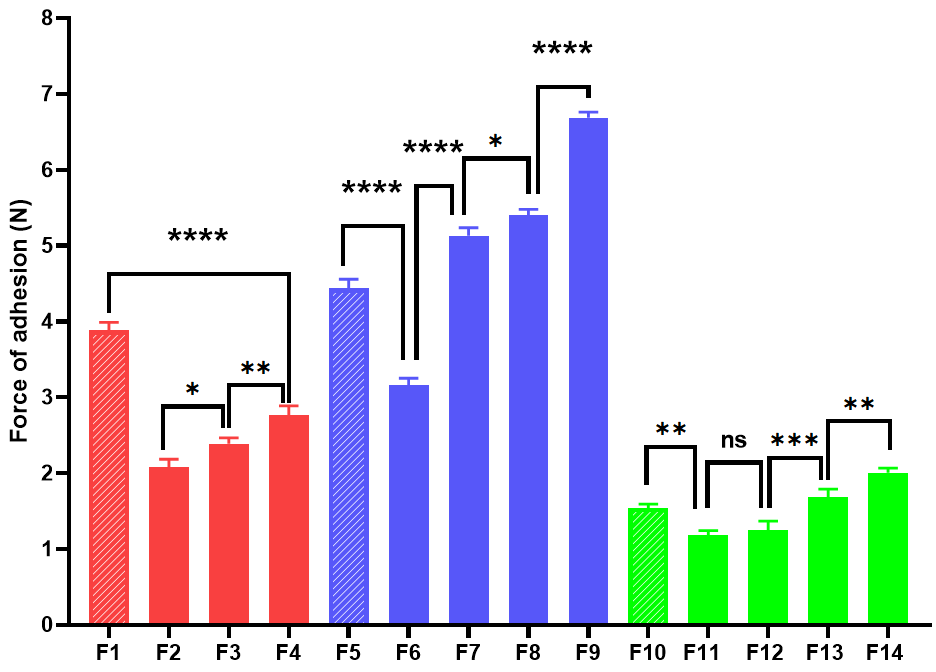
Fig. 3: Bio-adhesion force of loratadine ocular films (mean±SD, n = 3, *p<0.05, **p<0.01, ***p<0.001, and ****p<0.0001)
Results indicate that combining HPMC K4M with different mucoadhesive polymers provides adequate mucoadhesion to the films, making them suitable for ocular use, as reported in the literature [65].
Using one-way ANOVA and Tukey's multiple comparisons tests, it was revealed that most comparisons for adhesion show highly significant differences (p<0.0001), emphasizing that changes in formulation components (presumably polymer concentration or type) had a strong effect on bioadhesive properties. The Force of adhesion was reduced significantly (p<0.0001 for HPMC/SH or HPMC/SA and p<0.0001 for HPMC/PVP blends) with increasing the concentration of film-forming polymer (HPMC K4M), which is evident when comparing formulations F1, F5, F10 to F4, F6, and F11, respectively. This can be attributed to the moderate adhesive properties of HPMC and a reduction in the ratio of mucoadhesive polymer to total polymer content, resulting in a decrease in mucoadhesive characteristics.
Regarding the influence of using different types of mucoadhesive polymers in HPMC blended films, a statistically significant difference (p<0.05) in mucoadhesive strength was observed, which ranked as SA>SH>PVP in their adhesion strength.
Statistical analysis for the influence of increasing mucoadhesive polymers concentration reveals that the differences between all sequential HPMC/SH formulations (F1 to F2, F2 to F3, and F3 to F4) are all statistically significant (p<0.05or better). For formulations based on HPMC/SA, the substantial increase in adhesion from F6 to F7 is highly significant (p<0.0001), and the subsequent sequential increases from F8 to F9 or F9 to F10 are all statistically significant (p<0.05 or better).
As for HPMC/PVP-based films, the adhesive forces for F11 and F12 are not statistically different (p>0.05), representing the lowest bioadhesion observed overall. The subsequent increases from F12 to F13 are extremely significant (p<0.001), and for F13 to F14 are very significant (p<0.01), indicating a substantial improvement in bioadhesion towards F14.
The synergistic mucoadhesive effect provided to HPMC by the inclusion of highly charged, flexible polymers like SH or SA is mainly attributed to the presence of a higher number of free ionizable groups within the chains of these polymers. This causes extensive swelling, which becomes more apparent at higher concentrations, leading to increased interpenetration and interaction with the mucin layer through H-bonds or electrostatic forces, resulting in higher adhesion forces [65]. The superiority of SA over SH in mucoadhesion ability can be attributed to its higher molecular weight, which promotes better entanglement and more robust physical interactions compared to inadequate interpenetration of the short polymeric chain of low molecular weight SH. Moreover, the mucoadhesive behavior of these biopolymers aligns well with observations from their swelling study, where HPMC/SH polymeric blends exhibited higher swelling ability and lower bioadhesion strength, as overhydration reduces polymer entanglement and interaction with the mucin layer. This was in agreement with previously published data [66]. Regarding HPMC/PVP blends, the discussion based on the influence of charge on mucoadhesive strength does not apply, as PVP is neutral; therefore, among the three types of mucoadhesive polymers used, it had the lowest mucoadhesive property. The improvement of adhesion force with use of higher concentrations of mucoadhesive polymers is in accordance with that reported by previous studies [68, 69].
In vitro drug release and release kinetics
Drug release profiles of loratadine from prepared ocular films of HPMC blends with SH, SA, and PVP at various concentrations over 12 h are shown in fig. 4 (A-C). These formulations demonstrated a sustained release profile, representing a marked improvement compared to literature studies, which achieved only 4 h release [8, 9].
A biphasic release profile was observed, with an initial burst release of 26.99±1.96 % to 57.76±1.62 % within the first hour, primarily due to the release of the drug present at the film's surface. This was followed by a gradual, slower release, in the range of 59.75±4.30 % to 99.93±0.86 % over 12 h, occurring due to the synergistic influence of HPMC and mucoadhesive polymers in forming a gel layer, which increased the diffusion length and reduced the drug release rate. These results are consistent with previous reports [70-72].
Different mucoadhesive polymers exhibited varying release profiles when combined with HPMC, and the release profiles ranked as PVP>SA>SH. It was found that, after 12 h of dissolution study, the highest amount of drug released (99.93±0.86 %) was from formulation F10, containing 1% HPMC and 0.75% PVP, while the lowest (59.75±4.30 %) drug release was from formulation F9, containing 1.5% HPMC and 2% SA.
As expected, slower drug release rates were obtained using higher polymeric concentrations of HPMC or mucoadhesive polymers. However, statistical difference was only evident in the dissolution profiles of HPMC/SH or HPMC/SA blends at their highest polymeric concentrations (F4 or F9, respectively), and no significant reduction in drug release was observed in HPMC/PVP blends when compared even at their highest polymeric concentrations, indicating no significant influence of polymer concentrations on drug release rate.
The highest drug release rate observed when using PVP can be attributed to its highly hygroscopic nature and the rapid dissolution of the polymer, which forms pores in its structure. Additionally, the low swelling ability of the polymer caused no apparent gel-like effect to retard medium penetration and reduce drug release.
It is noteworthy that the degree of hydration and swelling can influence in vivo drug release from swellable matrices. Films containing HPMC/SH or HPMC/SA blends characterized by their high swelling nature had a lower drug release rate, mainly because the gel-like layer produced by the hydrated polymers, in addition to the increased viscosity of the microenvironment, which retards the penetration of the dissolution medium and reduces drug diffusion.
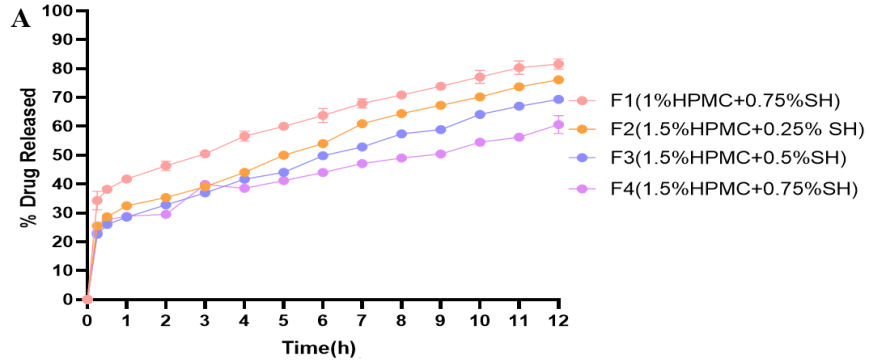
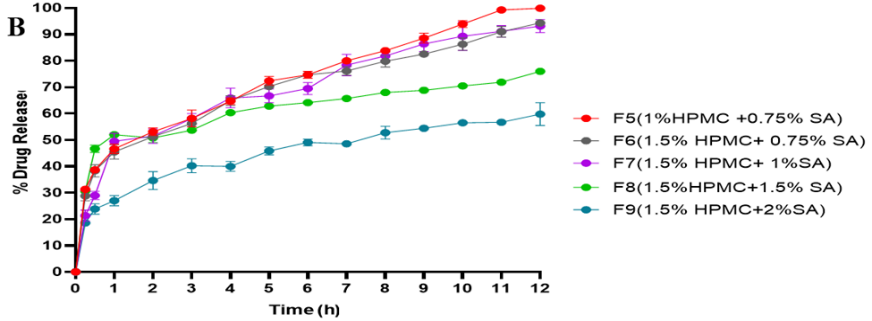
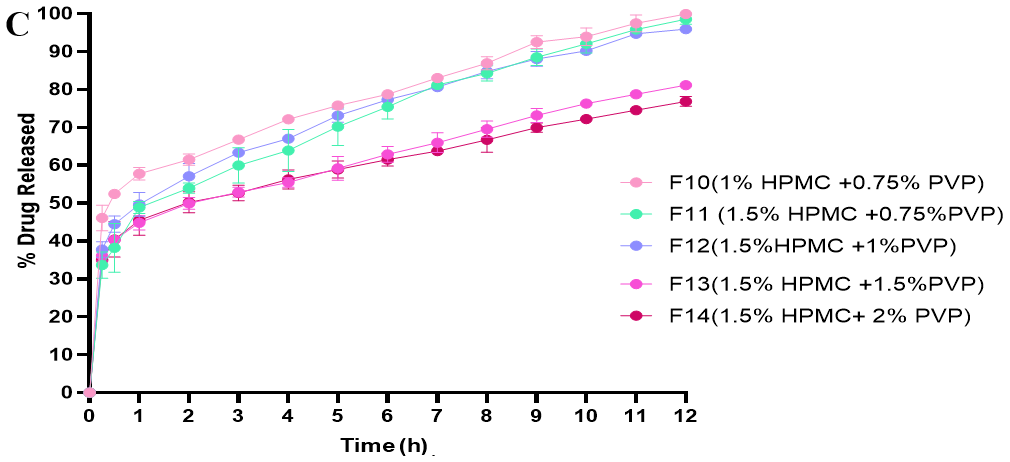
Fig. 4: In vitro drug release profiles of loratadine from (A) HPMC/SH-based films, (B) HPMC/SA-based films, (C) HPMC/PVP-based films
Table 4: Kinetic modeling of drug release data from loratadine ocular films
| Code | Zero order | First order | Higuchi | Korsmeyer-peppas | Release mechanism | |||||
| R2 | K0 | R2 | K1 | R2 | KH | R2 | K | n | ||
| F1 | 0.0300 | 8.559 | 0.5588 | 0.190 | 0.7913 | 25.879 | 1.000 | 1.000 | 1.000 | Case II |
| F2 | 0.4889 | 7.638 | 0.7517 | 0.139 | 0.9250 | 22.752 | 0.9767 | 32.562 | 0.245 | Diffusion |
| F3 | 0.4780 | 6.890 | 0.7177 | 0.115 | 0.9262 | 20.537 | 0.9776 | 28.783 | 0.306 | Diffusion |
| F4 | 0.0979 | 6.028 | 0.4045 | 0.094 | 0.8016 | 18.185 | 0.9733 | 29.034 | 0.253 | Diffusion |
| F5 | 0.3549 | 10.271 | 0.8226 | 0.307 | 0.9078 | 30.805 | 0.9982 | 45.177 | 0.238 | Diffusion |
| F6 | 0.2456 | 9.705 | 0.7843 | 0.271 | 0.8809 | 29.241 | 0.9944 | 43.615 | 0.244 | Diffusion |
| F7 | 0.3928 | 9.770 | 0.8543 | 0.269 | 0.9207 | 29.335 | 0.9585 | 40.739 | 0.359 | Diffusion |
| F8 | -0.7152 | 8.173 | 0.0923 | 0.186 | 0.4800 | 25.141 | 0.9524 | 46.709 | 0.164 | Diffusion |
| F9 | 0.2223 | 6.265 | 0.5538 | 0.101 | 0.8777 | 18.909 | 0.9959 | 28.026 | 0.300 | Diffusion |
| F10 | -0.3080 | 10.578 | 0.6048 | 0.469 | 0.6550 | 32.213 | 0.9998 | 58.013 | 0.161 | Diffusion |
| F11 | 0.2626 | 10.180 | 0.7925 | 0.309 | 0.8807 | 30.623 | 0.9966 | 46.571 | 0.233 | Diffusion |
| F12 | 0.0040 | 10.170 | 0.7145 | 0.342 | 0.7943 | 30.813 | 0.9996 | 49.976 | 0.194 | Diffusion |
| F13 | -0.1859 | 8.468 | 0.4091 | 0.189 | 0.7013 | 25.717 | 0.9990 | 44.886 | 0.160 | Diffusion |
| F14 | -0.4084 | 8.144 | 0.2583 | 0.176 | 0.6188 | 24.864 | 0.9991 | 44.806 | 0.165 | Diffusion |
To understand the drug release mechanism, the dissolution data were kinetically analyzed, and the kinetic parameters obtained for all formulations are summarized in table 4.
The results showed that release profiles from film formulations were best fitted with the Korsmeyer-Peppas model, as indicated by the highest regression coefficient (R2) values for this model. For formulation F1, the value of the release exponent, n, is equal to 1, indicating case II transport, where drug release is primarily controlled by polymer relaxation or swelling. Formulations F2-F14 had values of n less than 0.5, indicating a Fickian diffusion-controlled drug release mechanism. Such a mechanism is in agreement with swellable systems, where liquid penetration, rather than the relaxation rate of the polymeric chains, substantially affects the release mechanism. Such an effect was explained in previous work [73, 74].
Based on several desirable formulation properties, such as film adhesion force, flexibility, tensile strength, and a suitable profile of drug release, which provided initial fast and complete drug release over 12 h, formula F5 containing 1% HPMC and 0.75% SA was selected for in vivo irritation studies.
Sterilization and sterility test
The selected film formulation (F5) was sterilized using UV radiation, and the efficiency of this method was checked using fluid thioglycolate medium (FTGM) and soya bean-casein digest (SBCD) medium to assess the presence of bacteria and fungi, respectively, as evident in fig. 5.
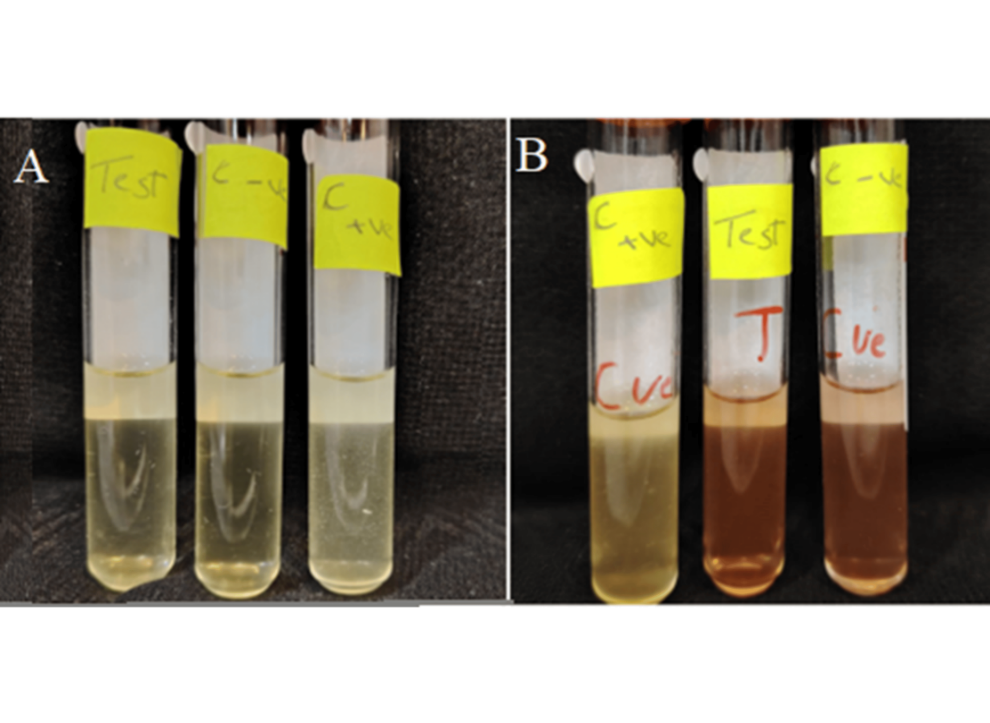
Fig. 5: Sterility test in (A) SBCD media, and (B) in FTGM media, showing (+ve control), sterilized film (Test), and (-ve control)
Visual inspection of tubes containing sterilized films (Test) shows no signs of turbidity indicative of microbial proliferation, similar to those observed in (-ve control) tubes, whereas signs of turbidity were evident in (+ve control) tubes, indicating microbial growth. Such observations confirm that the film was successfully sterilized using UV radiation and is suitable for in vivo studies. Although UV sterilization was appropriate for the specific experimental setup, other techniques like gamma irradiation or aseptic processing would be better alternatives for commercial scale-up.
Cytotoxicity study
The cytotoxic effects of loratadine films from formulation (F5) were tested for cytotoxicity towards normal human fibroblast (HNF) cells using an MTT assay, as shown in fig. 6A.
Both blank film and loratadine containing film (F5) showed cell viability of more than 80% following administration, in particular, compared to the untreated control (100%), cell viability was 91.25±2.51% for the medicated HPMC/SA film (F5) and 91.93±3.34%for the blank film, which is considered non-toxic and can therefore be used safely for ocular application [75].
Ocular irritancy study and histological examination
The cytotoxicity study does not guarantee the absence of in vivo irritation; therefore, ocular irritancy and histological examinations were performed. Ocular films are well tolerated upon application when no signs of tearing, swelling, or burning are evident.
An important observation in this study is that the films (F5) lost their shape forming a gel-like appearance which occurred after about 10 min of inclusion in the conjunctival sac, this signifies the formulations in situ gelling capability, which is essential for prolonging drug release and improving eye retention time, with the ability of the formed gel to disappear within 12 h after application. Observing the cornea and conjunctiva of rabbits' eyes after application of the formulation shows no signs of inflammation or tearing in the treated eye after 1 h and up to 72 h after insertion (fig. 6B). A histopathological study of the rabbit eye was conducted to support the results, and as shown in fig. 7, it revealed normal histology of the retina, sclera, ciliary body, and lens tissues when compared to the normal control eye, indicating that loratadine ocular film produced no ocular irritation in the rabbits' eyes.
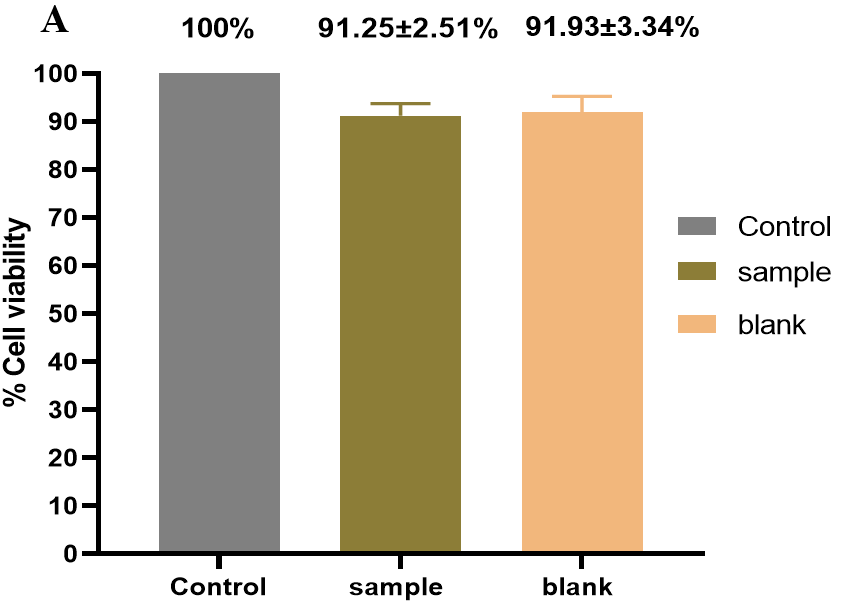
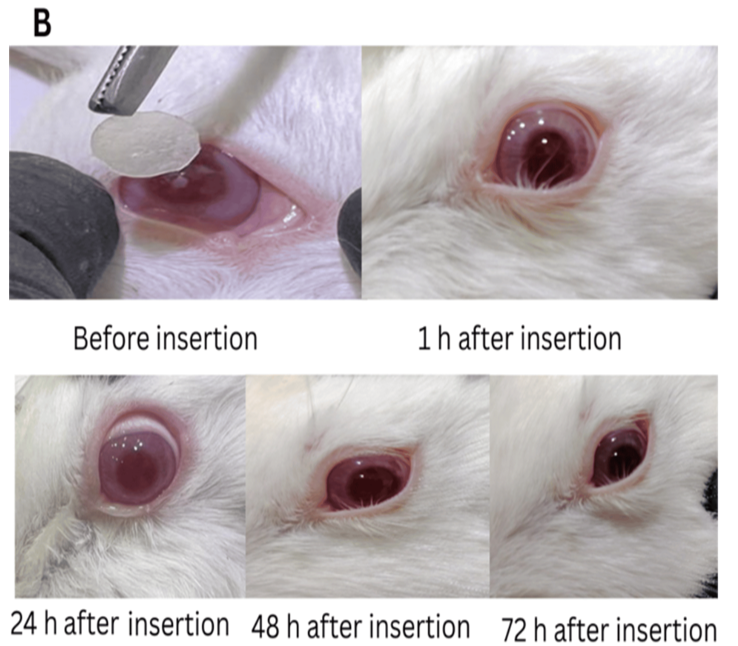
Fig. 6: (A) Cell viability (%) of selected loratadine ocular film (F5) and blank film measured using NHF and MTT method. (B) Eye irritancy study on the rabbit eye treated with selected loratadine ocular film (F5) over 3 d
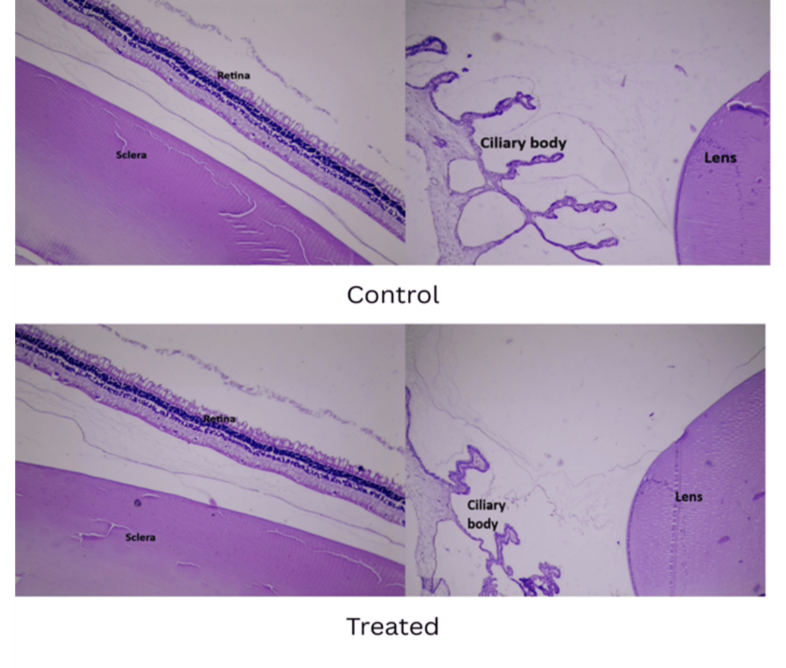
Fig. 7: Histological sections of retina, sclera, ciliary body, and lens. (HandE stain, 10× magnification)
CONCLUSION
This study suggests the formulation of an in-situ gel forming film for loratadine, having desirable pharmaceutical properties, using the solvent casting method. The obtained results indicate that combining HPMC K4M with different mucoadhesive polymers provides adequate mucoadhesive strength, having proper tensile strength, with an intermediate and sustained drug release behavior over a 12-hour period.
Prepared films offer the benefits of decreased application frequency and increased therapeutic efficacy. Moreover, the soluble nature of film components cancels the need for film removal after application, enhancing patient acceptability. Therefore, formulated films can be a suitable alternative to conventional eye drops for treating ocular allergy. However, in vivo/ex vivo corneal permeation was not evaluated, which is a limitation for the current study.
ACKNOWLEDGMENT
The authors would like to express the acknowledgement to the College of Pharmacy, University of Baghdad, Department of Pharmaceutics for providing equipment to facilitate this study.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
Rasha Hameed AL-Kaaby: Conceptualization, methodology, investigation, data curation, resources, software, formal analysis, visualization, original draft writing and editing.
Lena Murad Thomas: Conceptualization, methodology, supervision, writing—review and editing. All authors have read and agreed to the published version of the manuscript.
CONFLICTS OF INTERESTS
The authors declare no conflicts of interest
REFERENCES
Rodrigues J, Kuruvilla ME, Vanijcharoenkarn K, Patel N, Hom MM, Wallace DV. The spectrum of allergic ocular diseases-2020. Ann Allergy Asthma Immunol. 2021;126(3):240-54. doi: 10.1016/j.anai.2020.11.016, PMID 33276116.
Rozi MF, Mohmad Sabere AS. Review on conventional and novel topical ocular drug delivery system. Journal of Pharmacy. 2021;1(1):19-26. doi: 10.31436/jop.v1i1.32.
Jumelle C, Gholizadeh S, Annabi N, Dana R. Advances and limitations of drug delivery systems formulated as eye drops. J Control Release. 2020;321:1-22. doi: 10.1016/j.jconrel.2020.01.057, PMID 32027938.
Dosmar E, Walsh J, Doyel M, Bussett K, Oladipupo A, Amer S. Targeting ocular drug delivery: an examination of local anatomy and current approaches. Bioengineering (Basel). 2022;9(1):41. doi: 10.3390/bioengineering9010041, PMID 35049750.
Pelusi L, Mandatori D, Mastropasqua L, Agnifili L, Allegretti M, Nubile M. Innovation in the development of synthetic and natural ocular drug delivery systems for eye diseases treatment: focusing on drug-loaded ocular inserts contacts and intraocular lenses. Pharmaceutics. 2023;15(2):625. doi: 10.3390/pharmaceutics15020625, PMID 36839947.
Van Nguyen K, Dang TK, Vu LT, Ha NT, Truong HD, Tran TH. Orodispersible film incorporating nanoparticulate loratadine for an enhanced oral bioavailability. J Pharm Investig. 2023;53(3):417-26. doi: 10.1007/s40005-023-00613-2.
Sharif Makhmalzadeh BS, Salimi A, Niroomand A. Loratadine-loaded thermoresponsive hydrogel: characterization and ex-vivo rabbit cornea permeability studies. Iran J Pharm Res. 2018;17(2):460-9. PMID 29881404.
Abd El AE GOA, Shams ME, Maria DN. Formulation and in vitro evaluation of loratadine ocuserts. RGUHS J Pharm Sci. 2013;3(4):62-8. doi: 10.13140/RG.2.1.2297.4887.
Abd El-Gawad AE, Soliman OA, Shams ME, Maria DN. Formulation and in vitro evaluation of loratadine gels for ophthalmic use. RGUHS J Pharm Sci. 2014;4(2):62-9. doi: 10.5530/rjps.2014.2.5.
Abdelmonem R, Al-Samadi IE, El Nashar RM, Jasti BR, El-Nabarawi MA. Fabrication of nanostructured lipid carriers ocugel for enhancing Loratadine used in treatment of COVID-19 related symptoms: statistical optimization in vitro ex-vivo, and in vivo studies evaluation. Drug Deliv. 2022;29(1):2868-82. doi: 10.1080/10717544.2022.2115164, PMID 36065090.
Nyamweya NN. Applications of polymer blends in drug delivery. Futur J Pharm Sci. 2021;7(1):18. doi: 10.1186/s43094-020-00167-2.
Tundisi LL, Mostaco GB, Carricondo PC, Petri DF. Hydroxypropyl methylcellulose: physicochemical properties and ocular drug delivery formulations. Eur J Pharm Sci. 2021;159:105736. doi: 10.1016/j.ejps.2021.105736, PMID 33516807.
Ibrahim AG, Thomas LM. Formulation and evaluation of bilastine thermosensitive mucoadhesive ophthalmic in situ gel. Al-Rafidain J Med Sci. 2024;7(1):1-7. doi: 10.54133/ajms.v7i1.1014.
Al-Saedi ZH, Alzhrani RM, Boddu SH. Formulation and in vitro evaluation of cyclosporine-a inserts prepared using hydroxypropyl methylcellulose for treating dry eye disease. J Ocul Pharmacol Ther. 2016;32(7):451-62. doi: 10.1089/jop.2016.0013, PMID 27294697.
Ghadermazi R, Hamdipour S, Sadeghi K, Ghadermazi R, Khosrowshahi Asl A. Effect of various additives on the properties of the films and coatings derived from hydroxypropyl methylcellulose-a review. Food Sci Nutr. 2019;7(11):3363-77. doi: 10.1002/fsn3.1206, PMID 31762990.
Shukr M. Formulation in vitro and in vivo evaluation of lidocaine HCl ocular inserts for topical ocular Anesthesia. Arch Pharm Res. 2014;37(7):882-9. doi: 10.1007/s12272-013-0317-x, PMID 24395530.
Franco P, De Marco I. The use of poly(N-vinyl pyrrolidone) in the delivery of drugs: a review. Polymers. 2020;12(5):1114. doi: 10.3390/polym12051114, PMID 32414187.
Naga Priya KR, Bhattacharyya S, Ramesh Babu P. Formulation and evaluation of erodible ocular films of valacyclovir hydrochloride. Dhaka Univ J Pharm Sci. 2014;13(1):75-81. doi: 10.3329/dujps.v13i1.21866.
Huynh A, Priefer R. Hyaluronic acid applications in ophthalmology rheumatology and dermatology. Carbohydr Res. 2020;489:107950. doi: 10.1016/j.carres.2020.107950, PMID 32070808.
Hurtado A, Aljabali AA, Mishra V, Tambuwala MM, Serrano Aroca A. Alginate: enhancement strategies for advanced applications. Int J Mol Sci. 2022;23(9):4486. doi: 10.3390/ijms23094486, PMID 35562876.
Trastullo R, Abruzzo A, Saladini B, Gallucci MC, Cerchiara T, Luppi B. Design and evaluation of buccal films as paediatric dosage form for transmucosal delivery of ondansetron. Eur J Pharm Biopharm. 2016;105:115-21. doi: 10.1016/j.ejpb.2016.05.026, PMID 27267732.
Okeke OC, Boateng JS. Composite HPMC and sodium alginate-based buccal formulations for nicotine replacement therapy. Int J Biol Macromol. 2016;91:31-44. doi: 10.1016/j.ijbiomac.2016.05.079, PMID 27222284.
Khurana G, Arora S, Pawar PK. Ocular insert for sustained delivery of gatifloxacin sesquihydrate: preparation and evaluations. Int J Pharm Investig. 2012;2(2):70-7. doi: 10.4103/2230-973X.100040, PMID 23119235.
Jethava JK, Jethava GK. Design formulation and evaluation of novel sustained-release bioadhesive in-situ gelling ocular inserts of ketorolac tromethamine. Int J Pharm Investig. 2014;4(4):226-32. doi: 10.4103/2230-973X.143131, PMID 25426444.
Alwan ZS, Rajab NA. Preparation and characterization of febuxostat nanosuspension as a fast-dissolving oral film. Al-Rafidain J Med Sci. 2024;6(2):171-7. doi: 10.54133/ajms.v6i2.873.
Ghadhban HY, Ahmed KK. Nanosuspension-based repaglinide fast-dissolving buccal film for dissolution enhancement. AAPS PharmSciTech. 2024;25(6):161. doi: 10.1208/s12249-024-02868-w, PMID 38992175.
Wafa HG, Essa EA, El-Sisi AE, El Maghraby GM. Ocular films versus film-forming liquid systems for enhanced ocular drug delivery. Drug Deliv Transl Res. 2021;11(3):1084-95. doi: 10.1007/s13346-020-00825-1, PMID 32728811.
Radhi ZA, Ghareeb MM. Preparation and evaluation of rebamipide film using casting technique for local action. Iraqi J Pharm Sci. 2019;28(1):24-36. doi: 10.31351/vol28iss1pp24-36.
Kareem H, Rajab N. Formulation and characterization of eplerenone nanocrystal as sublingual fast-dissolving film. Iraqi J Pharm Sci. 2024;33(4):151-65. doi: 10.31351/vol33iss4pp151-165.
Alhamhoom Y, Said AK, Kumar A, Nanjappa SH, Wali D, Rahamathulla M. Sublingual fast-dissolving thin films of loratadine: characterization in vitro and ex vivo evaluation. Polymers. 2024;16(20):2919. doi: 10.3390/polym16202919, PMID 39458747.
Hermans K, Van Den Plas D, Kerimova S, Carleer R, Adriaensens P, Weyenberg W. Development and characterization of mucoadhesive chitosan films for ophthalmic delivery of cyclosporine a. Int J Pharm. 2014;472(1-2):10-9. doi: 10.1016/j.ijpharm.2014.06.017, PMID 24929014.
Aburahma MH, Mahmoud AA. Biodegradable ocular inserts for sustained delivery of brimonidine tartarate: preparation and in vitro/in vivo evaluation. AAPS PharmSciTech. 2011;12(4):1335-47. doi: 10.1208/s12249-011-9701-3, PMID 21979886.
Jovanovic M, Tomic N, Cvijic S, Stojanovic D, Ibric S, Uskokovic P. Mucoadhesive gelatin buccal films with propranolol hydrochloride: evaluation of mechanical mucoadhesive and biopharmaceutical properties. Pharmaceutics. 2021;13(2):273. doi: 10.3390/pharmaceutics13020273, PMID 33670448.
Abdelkader H, Pierscionek B, Alany RG. Novel in situ gelling ocular films for the opioid growth factor-receptor antagonist-naltrexone hydrochloride: fabrication mechanical properties mucoadhesion, tolerability and stability studies. Int J Pharm. 2014;477(1-2):631-42. doi: 10.1016/j.ijpharm.2014.10.069, PMID 25445974.
Sivadasan D, Venkatesan K, Mohamed JM, Alqahtani S, Asiri YI, Faisal MM. Application of 32 factorial design for loratadine-loaded nanosponge in topical gel formulation: comprehensive in vitro and ex vivo evaluations. Sci Rep. 2024;14(1):6361. doi: 10.1038/s41598-024-55953-2, PMID 38493177.
Costa P, Sousa Lobo JM. Modeling and comparison of dissolution profiles. Eur J Pharm Sci. 2001;13(2):123-33. doi: 10.1016/S0928-0987(01)00095-1, PMID 11297896.
Zhang Y, Huo M, Zhou J, Zou A, Li W, Yao C. DDSolver: an add-in program for modeling and comparison of drug dissolution profiles. AAPS J. 2010;12(3):263-71. doi: 10.1208/s12248-010-9185-1, PMID 20373062.
Shah P, Thakkar V, Anjana V, Christian J, Trivedi R, Patel K. Exploring of taguchi design in the optimization of brinzolamide and timolol maleate ophthalmic in-situ gel used in treatment of glaucoma. Curr Drug Ther. 2020;15(5):524-42. doi: 10.2174/1574885514666190916151506.
Hardia A. Preparation and evaluation of biodegradable ocular inserts of timolol maleate. IJPSM. 2021;6(3):21-38. doi: 10.47760/ijpsm.2021.v06i03.003.
Teba HE, Khalil IA, Gebreel RM, Fahmy LI, Sorogy HM. Development of antifungal fibrous ocular insert using the freeze-drying technique. Drug Deliv Transl Res. 2024;14(9):2520-38. doi: 10.1007/s13346-024-01527-8, PMID 38366116.
Jafer RS, J Kassab HJ. Development and characterization of lornoxicam-infused ocular gel for effective treatment of ocular inflammation in domestic cats. Iraqi J Vet Med. 2025;49(1):8-15. doi: 10.30539/pw6vsy73.
Fulgencio GdO, Viana FA, Ribeiro RR, Yoshida MI, Faraco AG, Cunha-Junior AdS. New mucoadhesive chitosan film for ophthalmic drug delivery of timolol maleate: in vivo evaluation. J Ocul Pharmacol Ther. 2012;28(4):350-8. doi: 10.1089/jop.2011.0174, PMID 22320419.
Mishra D, Gilhotra R. Design and characterization of bioadhesive in-situ gelling ocular inserts of gatifloxacin sesquihydrate. DARU J Pharm Sci. 2008;16(1):1-8.
Chughtai FR, Zaman M, Khan AH, Amjad MW, Aman W, Khan SM. Formulation and evaluation of sustained release ocular inserts of betaxolol hydrochloride using arabinoxylan from Plantago ovata. Pak J Pharm Sci. 2021;34(Suppl 3):S1069-74. doi: 10.36721/PJPS.2021.34.3.
British Pharmacopoeia Commission. London: the stationery office. Appendix XII C. Consistency of Formulated Preparations; 2023.
Nanda A, Das S, Sahoo RN, Nandi S, Swain R, Pattanaik S. Aspirin–hydrogel ocular film for topical delivery and ophthalmic anti-inflammation. J Serb Chem Soc. 2022;87(7-8):829-43. doi: 10.2298/JSC210504019N.
Dave V. Design and evaluation of aceclofenac ocular inserts with special reference to cataracts and conjunctivitis. J Chin Pharm Sci. 2013;22(5):449. doi: 10.5246/jcps.2013.05.066.
Noori MM, Al-Shohani AD, Yousif NZ. Fabrication and characterization of new combination ocular insert for the combined delivery of tinidazole and levofloxacin. Mater Today Proc. 2023;80:2652-9. doi: 10.1016/j.matpr.2021.07.008.
Ding C, Zhang M, Li G. Preparation and characterization of collagen/hydroxypropyl methylcellulose (HPMC) blend film. Carbohydr Polym. 2015;119:194-201. doi: 10.1016/j.carbpol.2014.11.057, PMID 25563960.
Wang Y, Jiang S, Chen Y, Qiu D, Weng Y. Synthesis and characterization of a novel composite edible film based on hydroxypropyl methyl cellulose grafted with gelatin. Gels. 2023;9(4):332. doi: 10.3390/gels9040332, PMID 37102944.
Kraisit P, Limmatvapirat S, Nunthanid J, Sriamornsak P, Luangtana Anan M. Preparation and characterization of hydroxypropyl methylcellulose/polycarbophil mucoadhesive blend films using a mixture design approach. Chem Pharm Bull (Tokyo). 2017;65(3):284-94. doi: 10.1248/cpb.c16-00849, PMID 27980251.
Salih SI, Jabur AR, Mohammed T, editors. The effect of PVP addition on the mechanical properties of ternary polymer blends. IOP Conf S Mater Sci Eng. 2018;433:012071. doi: 10.1088/1757-899X/433/1/012071.
Salih SI, Jabur AR, Mohammed T. The effect of PVP addition on the mechanical properties of ternary polymer blends. IOP Conf Ser: Mater Sci Eng. 2018;433:012071. doi: 10.1088/1757-899X/433/1/012071.
Jain D, Carvalho E, Banerjee R. Biodegradable hybrid polymeric membranes for ocular drug delivery. Acta Biomater. 2010;6(4):1370-9. doi: 10.1016/j.actbio.2009.11.001, PMID 19900594.
Ghosal K, Ranjan A, Bhowmik BB. A novel vaginal drug delivery system: anti-HIV bioadhesive film containing abacavir. J Mater Sci Mater Med. 2014;25(7):1679-89. doi: 10.1007/s10856-014-5204-6, PMID 24699799.
Wang Y, Zhang L, Liu H, Yu L, Simon GP, Zhang N. Relationship between morphologies and mechanical properties of hydroxypropyl methylcellulose/hydroxypropyl starch blends. Carbohydr Polym. 2016;153:329-35. doi: 10.1016/j.carbpol.2016.07.029, PMID 27561503.
Zhang L, Wang XF, Liu H, Yu L, Wang Y, Simon GP. Effect of plasticizers on microstructure compatibility and mechanical property of hydroxypropyl methylcellulose/hydroxypropyl starch blends. Int J Biol Macromol. 2018;119:141-8. doi: 10.1016/j.ijbiomac.2018.07.064, PMID 30016660.
Ravindran VK, Repala S, Subadhra S, Appapurapu AK. Chick chorioallantoic membrane model for in ovo evaluation of timolol maleate-brimonidine tartrate ocular inserts. Drug Deliv. 2014;21(4):307-14. doi: 10.3109/10717544.2013.845272, PMID 24134746.
Hernandez Gonzalez ME, Rodriguez Gonzalez CA, Valencia Gomez LE, Hernandez Paz JF, Jimenez Vega F, Salcedo M. Characterization of HPMC and PEG 400 mucoadhesive film loaded with retinyl palmitate and ketorolac for intravaginal administration. Int J Mol Sci. 2024;25(23):12692. doi: 10.3390/ijms252312692, PMID 39684402.
Tighsazzadeh M, Mitchell JC, Boateng JS. Development and evaluation of performance characteristics of timolol-loaded composite ocular films as potential delivery platforms for treatment of glaucoma. Int J Pharm. 2019;566:111-25. doi: 10.1016/j.ijpharm.2019.05.059, PMID 31129346.
Al-Mamari A, Shahitha F, Al-Sibani M, Al Saadi A, Al Harrasi A, Ahmad A. Novel antibacterial wound healing hydrogels based on HEC/SA/HA using green chemistry approach. Lett Appl Nanobiosci. 2023;12:69. doi: 10.1016/j.eurpolymj.2018.09.003.
Abou-Okeil A, Fahmy HM, El-Bisi MK, Ahmed-Farid OA. Hyaluronic acid/Na-alginate films as topical bioactive wound dressings. Eur Polym J. 2018;109:101-9. doi: 10.1016/j.eurpolymj.2018.09.003.
El Gamal SS, Naggar VF, Allam AN. Formulation and evaluation of acyclovir ophthalmic inserts. Asian J Pharm Sci. 2008;3(2):58-67.
Bayer IS. Recent advances in mucoadhesive interface materials, mucoadhesion characterization and technologies. Adv Materials Inter. 2022;9(18):2200211. doi: 10.1002/admi.202200211.
Nanda A, Sahoo RN, Pramanik A, Mohapatra R, Pradhan SK, Thirumurugan A. Drug-in-mucoadhesive type film for ocular anti-inflammatory potential of amlodipine: effect of sulphobutyl-ether-beta-cyclodextrin on permeation and molecular docking characterization. Colloids Surf B Biointerfaces. 2018;172:555-64. doi: 10.1016/j.colsurfb.2018.09.011, PMID 30218981.
Alzahrani A, Adel Ali Youssef AA, Senapati S, Tripathi S, Bandari S, Majumdar S. Formulation development and in vitro–ex vivo characterization of hot-melt extruded ciprofloxacin hydrochloride inserts for ocular applications: part I. Int J Pharm. 2023;630:122423. doi: 10.1016/j.ijpharm.2022.122423, PMID 36427695.
Pamlenyi K, Kristo K, Jojart Laczkovich O, Regdon G. Formulation and optimization of sodium alginate polymer film as a buccal mucoadhesive drug delivery system containing cetirizine dihydrochloride. Pharmaceutics. 2021;13(5):619. doi: 10.3390/pharmaceutics13050619, PMID 33925927.
Gilhotra RM, Gilhotra N, Mishra DN. Piroxicam bioadhesive ocular inserts: physicochemical characterization and evaluation in prostaglandin-induced inflammation. Curr Eye Res. 2009;34(12):1065-73. doi: 10.3109/02713680903340738, PMID 19958126.
Pan P, Svirskis D, Waterhouse GIN, Wu Z. Hydroxypropyl methylcellulose bioadhesive hydrogels for topical application and sustained drug release: the effect of polyvinylpyrrolidone on the physicomechanical properties of hydrogel. Pharmaceutics. 2023;15(9):2360. doi: 10.3390/pharmaceutics15092360, PMID 37765328.
Pınar Yagcılar A, Guven GK, Sefik Caglar E, Okur NU, Siafaka PI. Development and ex vivo / in vitro evaluation of sodium alginate/ hydroxypropyl methylcellulose films for dermal and/or transdermal delivery of p-hydroxycinnamic acid. J Holist Integr Pharm. 2025;6(2):175-83. doi: 10.1016/j.jhip.2025.06.002.
Everaert A, Wouters Y, Melsbach E, Zakaria N, Ludwig A, Kiekens F. Optimisation of HPMC ophthalmic inserts with sustained release properties as a carrier for thermolabile therapeutics. Int J Pharm. 2017;528(1-2):395-405. doi: 10.1016/j.ijpharm.2017.06.047, PMID 28624658.
Patil AV, Mahajan HS. Modified pea starch-based ocular films of azelastine hydrochloride: development and characterization. Carbohydr Polym Technol Appl. 2021;2:100078. doi: 10.1016/j.carpta.2021.100078.
Patel DP, Setty CM, Mistry GN, Patel SL, Patel TJ, Mistry PC. Development and evaluation of ethyl cellulose-based transdermal films of furosemide for improved in vitro skin permeation. AAPS PharmSciTech. 2009;10(2):437-42. doi: 10.1208/s12249-009-9224-3, PMID 19381831.
Perioli L, Ambrogi V, Angelici F, Ricci M, Giovagnoli S, Capuccella M. Development of mucoadhesive patches for buccal administration of ibuprofen. J Control Release. 2004;99(1):73-82. doi: 10.1016/j.jconrel.2004.06.005, PMID 15342182.
Sulaiman HT, Jabir SA, Kadhem Al-Kinani K. Investigating the effect of different grades and concentrations of PH-sensitive polymer on preparation and characterization of lidocaine hydrochloride as in situ gel buccal spray. Asian J Pharm Clin Res. 2018;11(11):401. doi: 10.22159/ajpcr.2018.v11i11.28492.
Taghe S, Mirzaeei S, Ahmadi A. Preparation and evaluation of nanofibrous and film-structured ciprofloxacin hydrochloride inserts for sustained ocular delivery: pharmacokinetic study in rabbit’s eye. Life (Basel). 2023;13(4):913. doi: 10.3390/life13040913, PMID 37109442.