
Int J App Pharm, Vol 18, Issue 2, 2026, 253-263Original Article
CHARACTERIZATION OF POULTRY EGG SHELL POWDER AS TABLET MATRIX BASE
SANJIBAN UTPALKUMAR SARKAR1, NITYANANDA MONDAL1, CHOWDHURY MOBASWAR HOSSAIN2*
1Department of Pharmaceutics, B. C. D. A. College of Pharmacy and Technology, 78/1 Jessore Road (S), Hridaypur, Kolkata-700127, West Bengal, India. 2Department of Pharmaceutical Technology, Maulana Abul Kalam Azad University of Technology, NH-12, Haringhata, Simhat, Nadia-741249, West Bengal, India
*Corresponding author: Chowdhury Mobaswar Hossain; *Email: drcmhossain@gmail.com
Received: 27 Oct 2025, Revised and Accepted: 22 Dec 2025
ABSTRACT
Objective: The present study focused on in-depth characterization of processed poultry egg shell powder (PESP) as a natural tablet diluent and a functional matrix base.
Methods: The eggshells were processed into a fine powder with a high-speed laboratory grinder, sieved through the #60 mesh. Processed PESP was characterized in terms of micromeritics, surface morphology by scanning electron microscopy (SEM), particle size distribution by bi-laser diffraction (BLD), crystallinity by x-ray diffraction (XRD) spectroscopy, functional groups identification by fourier transform infrared spectroscopy (FTIR), phase changing properties by differential scanning calorimetry (DSC), determination of microbial load and in vitro cytotoxic study by3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) using the human adult low calcium high temperature (HaCaT) keratinocyte cell line on PESP. Prepared tablets, both placebo and drug-loaded with PESP as matrix base were studied for tableting properties.
Results: Processed poultry eggshell powder (PESP) exhibited irregular, porous particles with a median size of 208 µm, indicating good flow ability and potential for uniform compaction. X-ray diffraction confirmed high crystallinity, while FTIR analysis showed no significant interactions with standard excipients or the model drug, suggesting chemical compatibility. Differential scanning calorimetry revealed phase transitions above 110 °C, indicating thermal stability without significant decomposition. Microbial evaluation demonstrated bacterial and fungal counts within pharmacopeial limits, and in vitro MTT assays on HaCaT keratinocytes showed no detectable cytotoxicity up to 1000 µg/ml. PESP also displayed acceptable compressibility and flow, supporting its suitability as a matrix excipient for tablet formulations. Overall, these findings highlight PESP as a safe, biocompatible, and pharmaceutically promising excipient.
Conclusion: Overall, as a proof-of-concept, preliminary findings demonstrated that PESP possessed functional properties and exhibited optimal tableting properties.
Keywords: Excipients, Processed, Placebo, Drug loaded, Compressible
© 2026 The Authors.Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i2.56682 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Tablet dosage forms are still the most widely used pharmaceutical dosage forms [1]. Tablets essentially include active pharmaceutical ingredients (API) and additives or excipients. An important part among these excipients is the diluent-also called bulking agent-which forms the bulk and aids in providing the desired mechanical and physical properties of tablets, including compressibility and uniformity of mass [2]. Lactose, microcrystalline cellulose (MCC), dicalcium phosphate (DCP), and modified starches are the most commonly used diluents, having been widely adopted due to their established functionality and compatibility [3, 4]. However, literature reports suggested that some diluents, although generally regarded as pharmacologically inactive and safe, may cause adverse effects in sensitive individuals or when administered in large amounts [5–7]. There is indeed a need for natural alternatives and sustainability. In recent years, the pharmaceutical industry has shown a rising interest in natural excipients derived from plant, animal, or mineral sources [8, 9]. These natural excipients are highly desirable due to their biodegradability, biocompatibility, and potential for reduced toxicity compared to certain synthetic counterparts [10]. Reports have indicated that some conventional diluents currently in use may be associated with adverse effects in sensitive individuals [11]. Lactose has been reported to cause bloating, diarrhoea, and abdominal cramps [12]. MCC, a widely used diluent, has been reported to cause allergic reactions, gastrointestinal discomfort, and flare-ups of irritable bowel syndrome (IBS) in predisposed individuals [13, 14]. Intestinal obstruction has been reported at very high intakes of MCC, especially from nutraceuticals [15]. MCC, as the bulk former in oro-dispersible tablets (ODT), may also impart a dry-mouth sensation or chalky taste [16]. Cellulose intolerance or hypersensitivity has been reported with cellulose derivative excipients as well [17]. Although still under-explored, excipients of natural origin-many of which are already recognized as nutraceuticals-represent a promising future option for tablet diluents in pharmaceutical formulations [18, 19]. During literature review, it was observed that there has been extensive research on the application of plant-derived excipients in pharmaceutical formulations, while comparatively limited literature is available on animal-derived excipients, particularly eggshell powder, as potential tablet diluents [20]. Only a few studies have addressed its micromeritic properties, compressibility, flow characteristics, and compatibility with various active pharmaceutical ingredients [21, 22]. These knowledge gaps highlighted the need for systematic investigations aimed at characterizing eggshell powder comprehensively. Such studies are essential to establish its functionality and safety as a sustainable and viable alternative to conventional diluents [23].
The present study focused on in-depth characterization of processed poultry eggshell powder (PESP) as a natural tablet diluent and a functional matrix base. In the current research, the investigation was focused on determining the applicability of processed poultry eggshell powder (PESP) as a natural and sustainable diluent for tablets. The primary objectives included transformation of eggshell waste into a standardized fine powder for pharmaceutical purposes. PESP was to be characterized in terms of micromeritics, particle size distribution studies in terms of bi-laser diffraction (BLD), scanning electron microscopy (SEM), X-ray diffraction (XRD) spectroscopy, differential scanning calorimetry (DSC), Fourier transform infrared (FTIR) spectroscopy, microbial studies, in vitro toxicity study (MTT), and accelerated stability studies. As proof-of-concept, preparation of placebo and drug-loaded trial batches of tablets by direct compression and wet granulation, with PESP as the major diluent, followed by evaluation of tableting properties and % cumulative drug release, were included in the study for assessing the functionality of PESP as a possible matrix base of natural origin [24].
MATERIALS AND METHODS
Collection and processing of poultry eggshell powder
Eggshells were collected from local sources, cleaned, boiled, oven-dried, and ground to fine powder using a high-speed laboratory grinder. Particles of 60# undersize, particles that were less than 250 µm in size, were included in the study and were thoroughly investigated for their diluent or bulk forming property [25].
Procurement of model drug and standard excipients
Metoprolol Succinate was used as the model drug and was received as a gift sample from Farma Hub Overseas, Haridwar. Standard tablet excipients were obtained as gift samples from Nitika Pharmaceuticals, Nagpur. All other chemicals used were of pharmacopoeial and research grade [26].
Characterization methodology
Processed poultry eggshell powder (PESP) was subjected to pre-formulation and micromeritic studies, including fundamental and derived powder properties [27]. BLD study was used to determine the particle size distribution. SEM was used to study surface morphology and texture [28]. XRD determined crystal arrangements [29], DSC assessed thermal behavior [30], FTIR analyzed functional groups [31, 32], Microbial studies were carried out for total aerobic count and fungal contamination [33].
In vitro cytotoxicity was evaluated using the MTT assay on mammalian cell lines [34]
The cytotoxicity of the provided PESP sample on HaCaT (procured from NCCS Pune) cell line was determined by MTT Assay. The cells (10,000 cells/well) were cultured in a 96-well plate for 24 h in DMEM (Dulbecco's Modified Eagle Medium-AT149-1L-HIMEDIA) supplemented with 10% FBS (foetal bovine serum-HIMEDIA-RM 10432) and 1% antibiotic solution (Penicillin-Streptomycin-Sigma-Aldrich P0781) at 37 °C with 5% CO₂. The next day, cells were treated with different concentrations of the samples from 1–1000 µg/ml based on the serial dilution method. Stock solution of samples was prepared in DMSO and further diluted to get different concentrations in incomplete cell culture medium (without FBS). After incubation for 24 h, MTT (5 mg/ml) was added to the cell culture and further incubated (Air-jacketed CO₂ incubator-Heal Force-HF90) for 2 h. Cells without treatment were considered as control and cells without MTT were considered blank. At the end of the experiment, culture supernatant was removed, and the cell layer matrix was dissolved in 100 µl dimethyl sulfoxide (DMSO – SRL-Cat no. 67685) and read in an ELISA plate reader (iMark, Bio-Rad, USA) at 540 nm. IC₅₀ was calculated by using GraphPad Prism-6. Images were captured under an inverted microscope (Olympus EK2) using a camera (AmScope digital camera 10 MP Aptima CMOS). Additionally, a positive control with a cytotoxic agent, paclitaxel, from 1–1000 ng/ml concentration based on the serial dilution method, was used to determine probable reduced viability in the presence of the cytotoxic agent for validation of the assay.
Where:
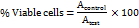
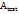 = Absorbance of the test sample
= Absorbance of the test sample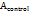 = Absorbance of the control
= Absorbance of the control
Accelerated Stability studies carried on PESP
Accelerated Stability Testing of PESP as per ICH guidelines, Q1A(R2), specifically under accelerated conditions: 40 °C±2 °C/75% RH±5% RH using stability chamber for 6 mo. PESP was stored in amber-colored high-density polyethylene bottles. Samples were withdrawn periodically and tested for set parameters.
Tablet formulation and evaluation
Two sets of preliminary tablet formulations were prepared to qualitatively assess the performance of processed PESP as a matrix-forming excipient. The placebo formulations (F1–F6) and drug-loaded formulations (F7–F8) incorporated different proportions of processed PESP, hydroxy propyl methyl cellulose (HPMC E 5), and cross-carboxymethylcellulose sodium (CCS) as depicted in Tables 9 and 12. These variations were not intended for direct comparative optimization but rather to explore the general tableting feasibility and processability of PESP in different compositional ranges. Formulations were designed as proof-of-concept formulations. Trial batches of uncoated tablets of processed PESP as diluent with other standard excipients were prepared. Three batches were prepared by direct compression and three by wet granulation. 8-station rotary tablet press was used for the compression. The compressed placebo batches were evaluated for tableting properties. The placebo batches were subjected to statistical comparison by one-way analysis of variance (ANOVA). Based on interpretation of the statistical data, selection of best performing batches in terms of tableting properties were done and placebo batches from both the category of direct compression and wet granulation were loaded with the model drug, Metoprolol Succinate. The drug-loaded batches were evaluated for tableting properties and % cumulative drug release. SEM studies of the tablet core were carried out to reveal the matrix structure. FTIR analysis study was carried out to determine the drug-excipient interactions.
RESULTS AND DISCUSSION
Characterization of PESP
Observations and findings in terms of particle morphology, shape, surface texture, particle length, agglomeration characteristics, fractures and cracks, color and odor are summarized in table 1. SEM studies (fig. 1) revealed surface texture and fracture patterns of PESP particles [28].
Table 1: Characterization of processed poultry egg shell powder
| Characterization | Observations, findings |
| Particle Morphology | The particles are irregular and rough-surfaced, indicating a non-uniform breakage process (e. g., mechanical grinding). |
| Shape | Particles are often angular, plate-like, or flaky. |
| Surface Texture | Rough, the surface texture is porous, which is typical of biogenic calcium carbonate structures |
| Agglomeration | Poly-disperse, aggregated, some particles appear angular, while others are more rounded, suggesting partial sintering or agglomeration |
| Fractures and Cracks | Low to moderate; some particles are loosely clustered |
| Color and Odor | White and odorless |
*All observations are qualitative in nature
Micromeritic properties
Fundamental and derived properties related to size range, bulk density, tapped density, angle of repose, compressibility index, Hausner’s ratio, polydispersity index. Observation and findings are summarized in table 2. Processed PESP exhibited acceptable bulk density, tapped density, compressibility index, and angle of repose, indicating fair flow and compressibility [27].

Fig. 1: Scanning electron microscopy image of processed poultry egg shell powder at 10000 x determined by Hitachi S4800
Table 2: Micromeritic properties of processed poultry egg shell powder
| Micromeritics | Observations, findings | Remarks |
Particle Size Distribution Span Index Uniformity Index Specific Surface Area (m²/kg) D [3,2] (Sauter mean, µm) D [4,3] (De Brouckere mean, µm) Dv (10) (percent undersize values, µm) Dv(50) (median particle size, µm) |
1.997 1.59 103.2 58.1 225 32.2 208 |
-moderate polydispersity -good flow, narrow size distribution -fines, surface-roughness contribute notably to the total surface area. -much smaller than the volume median. -more uniform, coarse, better flowability. -minor fraction of fines -Substantial coarse fraction, good flowability due to reduced inter-particle friction. Determined by Malvern Master Sizer 3000 |
| Bulk Density (g/ml) | 0.566 | Porous, void spaces |
| Tapped Density (g/ml) | 0.706 | Moderate packing density |
| Angle of Repose (˚) | 25.56 | Good Flow, Determined by classical funnel method |
| Carr’s Index (Compressibility Index %) | 19.83 | Fair flow, passable |
| Hausner’s Ratio | 1.24 | Fair flow, passable |
*All data are representative of mean of three independent determinations
The morphological and micromeritic properties of processed PESP demonstrated its suitability for tablet manufacturing. SEM revealed that the particles are irregular, rough-surfaced, and porous. This irregularity and surface roughness is advantageous, as it promotes inter-particulate bonding during compression, contributing to the structural integrity and hardness of the final tablet core, often comparable to the mechanical performance seen with dicalcium phosphate or microcrystalline cellulose. Micromeritic analysis showed an acceptable angle of repose of 25.56° and a Carr’s Index of 19.83%, classifying the powder as having good flow and acceptable compressibility. Furthermore, bi-laser diffraction (BLD) studies reported a median particle size (Dv50) of 208 µm. This substantial coarse fraction suggests a reduced inter-particle friction compared to very fine powders, confirming the fair flowability necessary for high-speed direct compression.
Instrumental analysis
Various instrumental analysis viz., XRD, FTIR, DLS and DSC performed on PESP for further in-depth characterization, with findings are summarized in table 3. XRD confirmed crystalline calcium carbonate content [29]. FTIR showed characteristic carbonate peaks, while DSC confirmed thermal stability [30-32].
Table 3: Instrumental characterization of processed poultry egg shell powder
| Tests | Observations, findings | Remarks |
x-ray diffraction (XRD study) |
d-spacing range: 11.57666 Å to 1.44452 Å Anode material: Copper (Cu) intended Wavelength type: K-Alpha |
This d-spacing range suggests that poultry eggshell powder exhibits a highly crystalline structure, typical of biogenic calcium carbonate (mainly calcite) and potentially some minor organic matrix traces; determined by BRUKER D8 Focus |
Fourier transform infrared spectroscopy (FTIR) |
Wave no. Possible functional group 3740.77 cm-1 O–H stretch 2359.12 cm-1 CO₂ overtone or C≡N 1690.51 cm-1C=O stretch (amide I or carboxyl) 1457.61 cm-1 C–O/CO₃²⁻ symmetric stretch 1418.20 cm-1 CO₃²⁻ asymmetric stretch 872.03 cm-1 CO₃²⁻ out-of-plane bending 771.09 cm-1 CO₃²⁻ in-plane bending 390.56 cm-1 Ca–O lattice vibrations 362.61 cm−1 Ca–O or lattice mode |
• Major component: Calcium carbonate (CaCO₃) in the calcite form shown by strong carbonate peaks at ~1418, 872 and 771 cm⁻¹). • Minor components: Organic residues like roteins or membrane fragments, C=O around 1690 cm⁻¹. • Hydroxyl peaks (3740 cm⁻¹) may be surface-adsorbed water or hydroxyl-containing ompounds. Determined by SHIMADZU FTIR IRSPIRIT-X series |
| differential scanning calorimetry (DSC) | Integral (Total Enthalpy): – 438.64 mJ Normalized enthalpy: – 199.38 J/g Onset temperature: 45.14 °C Peak temperature: 82.88 °C End set temperature: 135.13 °C |
The DSC profile of processed egg shell powder shows a prominent endothermic transition in the range of ~45–135 °C, indicating moisture release and minor organic matrix decomposition. The absence of sharp peaks above 200 °C implies no significant decomposition revealed by Mettler Toledo 822e |
† All data are representative of independent determinations
XRD verified the highly crystalline structure, primarily composed of calcite (calcium carbonate). This crystalline nature provides the essential mechanical strength and hardness-contributing properties required of a tablet matrix excipient. FTIR spectroscopy showed characteristic carbonate peaks at approximately 1418, 872, and 771 cm⁻¹, and confirmed that processed PESP is chemically compatible with the model drug (Metoprolol Succinate) and standard excipients, as no new or disappearing peaks were observed in the final tablet blend. DSC revealed thermal stability, with no significant decomposition peaks above 200 °C, confirming that processed PESP can withstand the heat generated during high-speed tableting without degradation.
Microbial studies
The results are summarized in table 4. Microbial studies revealed that both bacterial and fungal counts were found to be well within the permissible limits as specified by the United States Pharmacopeia, confirming microbiological safety [33].
Table 4: Microbial tests performed on processed poultry egg shell powder
| Tests | Observations, findings | Remarks |
| Total viable plate count | Aerobic bacterial count: 50cfu/g Total combined yeasts/molds count: Less than 10cfu/g E. coli Absent Salmonella spp. Absent Staphylococcus aureus Absent |
Count ≤1000 cfu/g, Acceptable Count ≤1000 cfu/g, Acceptable PESP met excellent microbiological quality standards |
*value are representative of mean of three independent determinations (n=3)
The total viable plate count was well within the permissible limits specified by the United States Pharmacopeia (Aerobic bacterial count: 50 cfu/g; Fungal count:<10 cfu/g). This confirms that the rigorous processing method successfully achieved the necessary microbiological quality standards for a pharmaceutical excipient.
In vitro toxicity
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay was performed using the human adult low calcium high temperature (HaCaT) keratinocyte cell line on processed PESP [34]. The observations in terms of mean cell viability % and subsequent interpretations revealed that PESP exhibited no significant cytotoxicity as IC₅₀ did not reached within tested dose range of 1-1000µg/ml, which suggested excellent biocompatibility and no detectable toxicity, even at the highest tested concentrations of 1000 µg/ml. Paclitaxel, a cytotoxic agent used as positive control, reached IC₅₀ close to 100ng/ml. The findings are summarized in table 5. Cell viability images of both PESP and paclitaxel are summarized in table 6 and table 7, respectively with possible interpretations. A critical finding was the excellent safety profile of the processed PESP.
Table 5: In vitro toxicity determination assay performed on human adult low calcium high temperature keratinocyte cell line for processed poultry egg shell powder and paclitaxel (mean cell viability %)
| PESP concentration (µg/ml) | Mean cell viability % | Paclitaxel concentration (ng/ml) | Mean cell viability % | Interpretation |
| 1 | 99.97±2.78 | 1 | 94.40±2.72 | Processed poultry egg shell powder: IC₅₀ didn’t reached at maximum concentration of 1000 µg/ml Paclitaxel: IC₅₀ reached close to 100 ng/ml |
| 10 | 87.88±3.81 | 10 | 83.36±2.30 | |
| 50 | 86.25±5.50 | 50 | 67.94±0.89 | |
| 100 | 85.45±7.69 | 100 | 45.70±1.34 | |
| 250 | 82.01±6.01 | 250 | 31.30±0.84 | |
| 500 | 65.48±6.78 | 500 | 21.06±0.56 | |
| 1000 | 64.29±3.18 | 1000 | 11.30±0.87 |
*value are expressed as mean±standard deviation (n = 4); IC₅₀ = Half-maximal inhibitory concentration
Accelerated stability studies carried on PESP
PESP sample was evaluated periodically as stated in table 8. Accelerated stability test results were satisfactory as per international council for harmonization (ICH) of technical Requirements for pharmaceuticals for human use guidelines, Q1A(R2) for the studied parameters. Microbial studies revealed the aerobic bacterial count was between 50 – 70 cfu and fungal count less than 10 cfu over the entire study duration, which indicated that if PESP was stored properly in airtight containers, chances of microbial spoilage was less. FTIR studies carried periodically revealed there were no new, additional and disappearing peaks, which suggested processed PESP was chemically stable.
Table 8: Record sheet for accelerated stability testing of processed poultry egg shell powder specifically, under accelerated conditions: 40 °C±2 °C/75% relative humidity±5%
| Parameters | Days | ||||||
| 0 | 30 | 60 | 90 | 120 | 150 | 180 | |
| Moisture Content (%) | 1.5-2.0 | 2.0-2.5 | 2.0-2.5 | 2.0-3.5 | 2.0-3.5 | 2.5-3.5 | 3.4-3.8 |
| Bulk Density (g/ml) | 0.566 | 0.543 | 0.548 | 0.533 | 0.476 | 0.481 | 0.484 |
| Tapped Density (g/ml) | 0.706 | 0.698 | 0.701 | 0.741 | 0.631 | 0.625 | 0.616 |
| Compressibility Index (%) | 19.83 | 22.20 | 21.87 | 28.07 | 24.57 | 23.04 | 21.43 |
| Hausner’s Ratio | 1.2473 | 1.29 | 1.28 | 1.39 | 1.33 | 1.30 | 1.27 |
*All data are representative of mean of three independent determinations (n=3)
Table 6: Image-based assessment of cell viability for processed poultry egg shell powder as test sample
| PESP concentration (µg/ml) | Cell viability images (100x) | Dominant cell morphology | Interpretation of cell viability |
| Control | 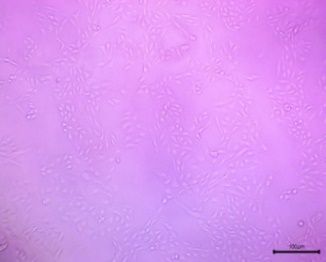 |
The cells in the image exhibit a spindle-shaped, elongated, or fibroblast-like morphology. | The culture appears healthy and viable. The cells are well-spread, uniform in appearance, and there is a high density of attached cells. |
| 1 | 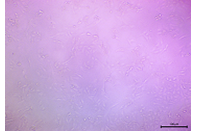 |
The cells appeared mostly confluent with a dense monolayer with normal fibroblastic/keratinocyte-like morphology. | High Viability: Minimal or no cytotoxic effect |
| 10 | 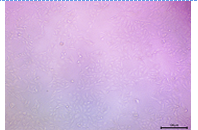 |
The cell density was still high, and the cells maintained a largely normal morphology. | High Viability: The sample remained largely non-cytotoxic. |
| 50 |  |
The cells were still numerous and well-spread, maintained their typical morphology. The monolayer was intact. | Appreciable Viability |
| 100 | 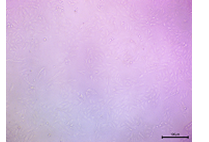 |
The cell monolayer appeared slightly less dense, but the cells that were present showed good attachment and morphology. | Moderate-High Viability |
| 250 | 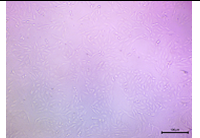 |
The overall cell density started to show a noticeable decrease. Many cells still were holding the morphology. | Moderate Viability |
| 500 | 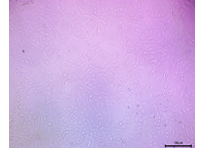 |
There was a clear reduction in cell density, and the cells started to appear more rounded or stressed. | Lower Viability still not cytotoxic |
| 1000 | 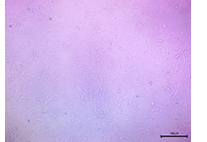 |
The cell density was significantly low, with fewer attached cells and more detached or morphologically compromised cells. | Lowest Viability, still not cytotoxic to an extent, at this dose IC₅₀ didn’t reached. |
*Images were captured under inverted microscope at 100x (Olympus ek2) using camera (AmScope digital camera 10 MP Aptima CMOS)
Table 7: Image-based assessment of cell viability for paclitaxel as positive control
| Paclitaxel concentration (ng/ml) | Cell viability images (100x) |
Dominant cell morphology | Interpretation of cell viability |
| Control | 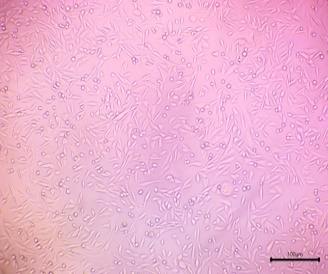 |
The cells in the image primarily display a spindle-shaped, elongated, or fusiform morphology. | Based on the visual evidence, the culture has high predicted viability, consistent with a healthy untreated control group. |
| 1 | 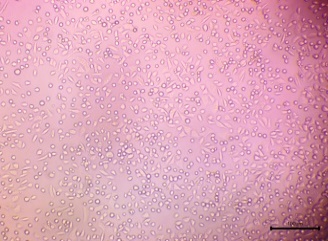 |
Mixture of spindle-shaped/polygonal (adherent) and round cells. | High Viability. Many cells were adherent and intact, indicating a low cytotoxic effect |
| 10 |  |
Predominantly spindle-shaped/polygonal (adherent) cells, with fewer round cells. | High Viability. Strong cell adherence, indicated minimal cytotoxic effect |
| 50 |  |
Mixture of spindle-shaped/polygonal (adherent) and round cells | Moderate Viability. Adherence was still notable, but the presence of more rounded cells suggested increasing toxicity |
| 100 |  |
Mixture of spindle-shaped/polygonal (adherent) and round cells. | Decreasing Viability. The cytotoxic effect was becoming more pronounced, leading to cell rounding/detachment. IC₅₀ reached closed to this concentration. |
| 250 |  |
Overwhelmingly round cells. Very few spindle-shaped/polygonal cells are visible. | Low Viability. High cytotoxic effect resulted in widespread cell rounding and detachment/death. |
| 500 |  |
Almost exclusively round cells. Very few, if any, adherent cells. | Very Low Viability. Severe cytotoxic effect |
| 1000 |  |
Almost exclusively rounded cells. Very few, if any, adherent cells. | Very Low Viability. Severe cytotoxic effect with least % of viable cells. |
*Images were captured under inverted microscope at 100x (Olympus ek2) using camera, (AmScope digital camera 10 MP Aptima CMOS)
Formulation and evaluation of compressed tablets
6-Placebo trial batches of tablets were prepared; 3 batches were prepared by direct compression (F1–F3) and 3 batches by wet granulation (F4–F6). An 8-station rotary tablet press was used for the compression. Formulations are summarized in table 9, and evaluation of all the placebo batches is summarized in table 10. Statistical analysis in terms of one-way ANOVA performed on the data obtained for tableting properties and subsequent interpretation of data revealed significant differences in hardness and disintegration time (p<0.05). The data analysis and interpretation are summarized in table 11. F1 (direct compression) showed the lowest disintegration time with acceptable hardness and friability, while F5 (wet granulation) demonstrated the lowest friability and optimal hardness. Thus, one placebo formulation from each category of direct compression (F1) and wet granulation (F5) was selected for incorporation of Metoprolol Succinate as a model drug, yielding batches F7 and F8 [36]. Formulation of drug-loaded trial batches is summarized in table 12, and evaluation of the same is summarized with interpretation in table 13. Tableting properties were found to be satisfactory for all the batches. SEM of tablet cores revealed porous internal structure [28]. Drug-loaded tablets were subjected to SEM studies for depiction of internal structure of the tablet core (fig. 4), which revealed porous nature with a matrix bed presence. Drug-loaded F7 and F8 exhibited>89% cumulative release as given in table 14. R² (coefficient of determination) values in terms of release kinetics indicated non-Fickian diffusion for both F7 and F8, with Korsmeyer–Peppas and Higuchi models fitting well [37], as depicted in table 15. The drug release profile for F7 and F8 in the form of graphical representation demonstrated that Higuchi and Korsmeyer–Peppas were the predominant fit (fig. 5). Furthermore, release exponent (n), which was between 0.6 to 0.7, indicated that drug release might have been governed by both diffusion and polymer relaxation/swelling. The combination of PESP as the matrix base and hydroxypropyl methylcellulose low viscosity grade (HPMC-E5) as binder resulted in approximately 90% drug release within 60 min, indicating a conventional release pattern. The hydrophilic and porous nature of PESP facilitated rapid matrix hydration and erosion, while the low viscosity grade HPMC improved tablet cohesiveness without forming a diffusion-limiting gel barrier, as it hydrated and dispersed rapidly, probably thereby promoting faster drug release. Overlay of IR spectrum showed no drug–excipient incompatibility (fig. 6); no new peaks or significant peak shifts were noted in the blend spectrum, suggesting no chemical interaction between Metoprolol Succinate, PESP, and the excipients. The FTIR spectra confirmed formulation compatibility and chemical stability of the blend; the findings are summarized in table 16. Overall, as a proof of concept, these preliminary findings demonstrated that PESP possessed functional properties suitable for both direct compression and granulation by the wet binding technique. The study successfully established PESP’s role as a compressible, stable, and pharmaceutically compatible excipient. Future studies employing statistical design of experiments (DoE) and controlled variable optimization are required to quantitatively delineate the influence of processed PESP concentration on mechanical and release behavior.
Table 9: Formulation of placebo trial batches of tablets with processed poultry egg shell powder (PESP) as matrix base
| Ingredients | F1 | F2 | F3 | F4 | F5 | F6 |
| All quantities in mg | ||||||
| Egg Shell Powder | 250 | 300 | 350 | 200 | 250 | 300 |
| Hydroxy Propyl Methyl Cellulose (Grade E5) | 40 | 50 | 60 | 40 | 50 | 60 |
| Cross Carmellose Sodium | 30 | 40 | 50 | 30 | 40 | 50 |
| Purified Water | --- | --- | --- | qs | qs | qs |
| Talc | 10 | 10 | 10 | 10 | 10 | 10 |
| Magnesium Stearate | 10 | 10 | 5 | 10 | 10 | 5 |
| Avg. Wt. (Per tablet) | 340 | 410 | 475 | 340 | 360 |
*F1, F2, F3 represents batches prepared by direct compression; F5, F6 represents batches prepared by wet granulation
Table 10: Evaluation of uncoated placebo tablets prepared with processed poultry egg shell powder (PESP) as matrix base
| Parameters | F1 | F2 | F3 | F4 | F5 | F6 |
| Shape | Bi-concave | Bi-concave | Bi-concave | Bi-concave | Bi-concave | Bi-concave |
| Thickness (mm) | 4.5-4.75 | 5.5 – 5.65 | 5.8-6.1 | 5.5 – 5.6 | 6.2 – 6.22 | 6.42-6.45 |
| Diameter (mm) | 11.0 | 11.0 | 11.0 | 11.0 | 11.0 | 11.0 |
Hardness (Kg/cm3) Mean |
5.0-5.1 5.03±0.058 |
5.5 – 5.6 5.56±0.058 |
6.0-6.5 6.26±0.252 |
5.5 – 6.0 5.60±0.252 |
5.9-6.0 5.95±0.050 |
6.0-6.5 6.23±0.252 |
Friability (%) Mean |
0.40-0.41 0.403±.006 |
0.42-0.44 0.430±0.010 |
0.42-0.43 0.427±0.006 |
0.44-0.46 0.453±0.012 |
0.38-0.39 0.387±0.20 |
0.50-0.53 0.517±0.015 |
Weight Variation (± 5 % Range) |
Within Limits |
Within Limits |
Within Limits |
Within Limits |
Within Limits |
Within Limits |
*All values are expressed as mean±standard deviation (n = 3)
Table 11: Statistical analysis in terms of one-way analysis of variance (ANOVA) applied to placebo tablets
| Parameter | F-value | p-value | Interpretation |
| Hardness (Kg/cm²) | 35.565 | 8.61 × 10⁻⁷ | Highly significant difference (p<0.01) among F1–F6; F5 has high hardness without exceeding limits, F1 has adequate hardness for quick release. |
| Friability (%) | 2.373 | 0.1022 | No statistically significant difference (p>0.05); all batches within acceptable friability limits (<1%), F5 has lowest friability, F1 also low. |
| Disintegration Time (min) | 176.953 | 8.27 × 10⁻¹¹ | Highly significant difference (p<0.01); F1 disintegrates fastest, F5 also quick but more balanced with strength. |
*Mean±standard deviation (n = 3); statistical significance determined using one-way analysis of variance (p<0.05); batches F1, F5selected for incorporation of Metoprolol Succinate as model drug
Table 12: Formulation of drug-loaded trial batches of tablets with processed poultry egg shell powder as matrix base
| Ingredients | F7 | F8 |
| All quantities in mg | ||
| Metoprolol succinate | 12.5 | 12.5 |
| Processed poultry egg shell powder | 250 | 250 |
| Hydroxy propyl methyl cellulose (Grade E5) | 40 | 50 |
| Cross carmellose sodium | 30 | 40 |
| Purified water | --- | qs |
| Talc | 10 | 10 |
| Magnesium stearate | 10 | 10 |
| Avg. Wt. (Per tablet) | 352.5 | 372.5 |
*F7-direct compression; F8-wet granulation. F1, F5-incorporated with drug after due statistical analysis, whereby additional 12.5 mg in average weight of tablets increased with respect to the placebo batches in consideration
Table 13: Evaluation of uncoated drug-loaded tablets prepared with processed poultry egg shell powder as matrix base
| Parameters | F7 | F8 | Remarks |
| Shape | Bi-concave | Bi-concave | Uniform |
Scanning electron microscopy (SEM) findings (Fragmented Tablets) Particle Arrangement Surface Texture Fusion/Compaction Porosity Fractures or Cracks Matrix Continuity |
Densely packed, irregular particles Rough with fractured crystal edges Particles appeared tightly bound, high composed structures were observed in F8 Low – very few visible pores Minor-micro fractures Consistent and dense |
Tablets prepared by direct compression and wet granulation methods revealed a compact, dense matrix core. The surface was rough and jagged with minimal porosity, indicating strong compressibility and tablet cohesion. | |
| Thickness (mm) | 4.6-4.7 | 5.9 – 6.1 | Acceptable |
| Diameter (mm) | 11.0 | 11.0 | Uniform |
Hardness Range observed (Kg/cm3) mean Hardness (Kg/cm3) |
5.0 – 5.1 5.033±0.058 |
5.75 – 5.8 5.783±0.029 |
Acceptable |
Friability Range (%) mean Friability (%) |
0.40-0.41 0.403±0.006 |
0.43-0.44 0.437±0.006 |
Passable, Acceptable |
| Weight Variation (± 5 % Range) | Within Limits | Within Limits | Passable, Acceptable |
Disintegration Time (Minutes) mean Disintegration Time (Minutes) |
1.20-1.21 1.203±0.006 |
1.41-1.42 1.417±0.006 |
Passable, Acceptable |
| % Drug Content | 96.22 | 98.35 | Passable, Acceptable |
| % cumulative drug release (CDR) | 89.92 | 93.55 | Passable, Acceptable |
| Fourier transform infrared spectroscopy (FTIR) | 3700 cm⁻¹ (O-H stretching), 1600 cm⁻¹ (C=O stretching), 1450–1350 cm⁻¹ (carbonate stretch), and 600–500 cm⁻¹ (Ca-O vibration) are still visible. 1700–1500 cm⁻¹: This range is consistent with amide and C=O stretches, which is likely due to the 1300 cm⁻¹–1100 cm⁻¹: Characteristic of C-N stretching or C-H bending vibrations from Metoprolol succinate. Significant Peaks: The peaks at 1300 cm⁻¹ and 1170 cm⁻¹ may be related to the aromatic nature of Metoprolol, as well as C-H and C-N stretches that are unique to this compound. |
There is no disappearance of any functional groups. Few functional groups related to the drug; Metoprolol succinate, were observed. Overall, No significant peak shifts or disappearance observed – indicates compatibility. |
*Scanning electron microscopy findings are qualitative, ‡All tablet evaluation data except % cumulative drug release is representative of mean of three independent determinations (n=3), †% cumulative drug release data are mean of six determinations (n=6)


a b
Fig. 4: a. Scanning electron microscopy image of F7 trial batch (drug loaded, prepared by direct compression) with processed poultry egg shell powder as matrix base at 5000 x determined by Hitachi S4800, b. Scanning electron microscopy image of F8 trial batch (drug loaded, wet granulation) with processed poultry egg shell powder as matrix base at 5000 x determined by Hitachi S4800
Table 14: % Cumulative drug release vs. time (minutes) for drug-loaded batches of tablets
| Time (Minutes) | % CDR, F7 | % CDR, F8 |
| 0.0 | 0.00 | 0.00 |
| 5.0 | 18.35 | 25.36 |
| 10.0 | 25.62 | 38.22 |
| 20.0 | 41.35 | 53.85 |
| 25.0 | 47.22 | 68.84 |
| 30.0 | 63.89 | 75.33 |
| 45.0 | 71.92 | 86.42 |
| 60.0 | 89.12 | 93.55 |
*value are mean of six determinations (n=6)
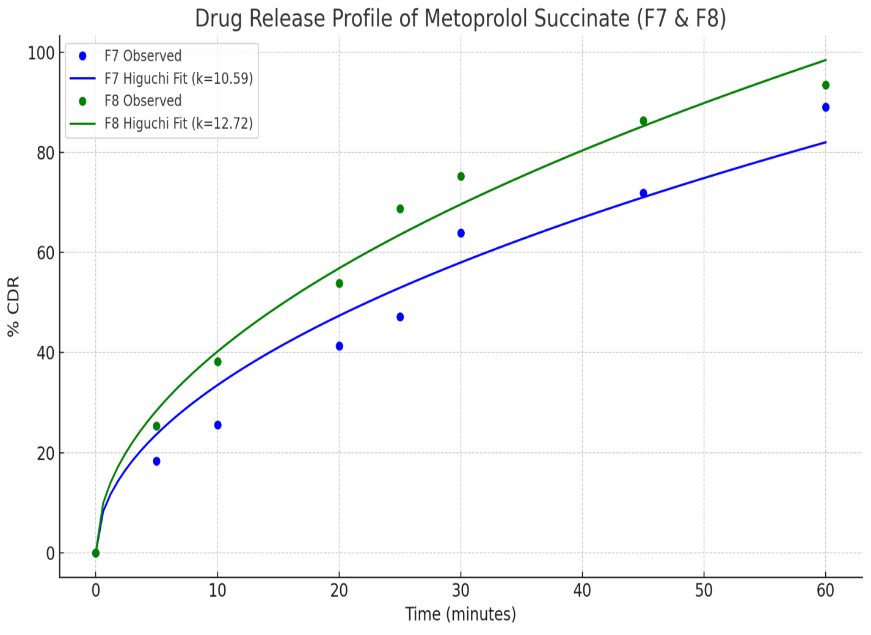
Fig. 5: Release profile in terms of %cumulative drug release vs. time (minutes) for drug-loaded batches of tablets prepared with processed poultry egg shell powder as matrix base
Table 15: Drug release kinetics summary for drug-loaded batches of tablets prepared with processed poultry egg shell powder as matrix base
| Formulation | Zero order R² | First order R² | Higuchi R² | Korsmeyer-peppas R² | Release exponent (n) | Interpretation |
| F7 | 0.9539 | 0.3707 | 0.9602 | 0.9879 | 0.699 | Korsmeyer-Peppas is the best fit with Non-Fickian release mechanism |
| F8 | 0.8734 | 0.4793 | 0.9851 | 0.9851 | 0.596 | Higuchi and Korsmeyer-Peppas both are the best fit with Non-Fickian release mechanism |
*DD Solver; Microsoft excel add-in specifically designed for dissolution/drug release data analysis was used for determination of coefficient of determination (R²) and release exponent (n)
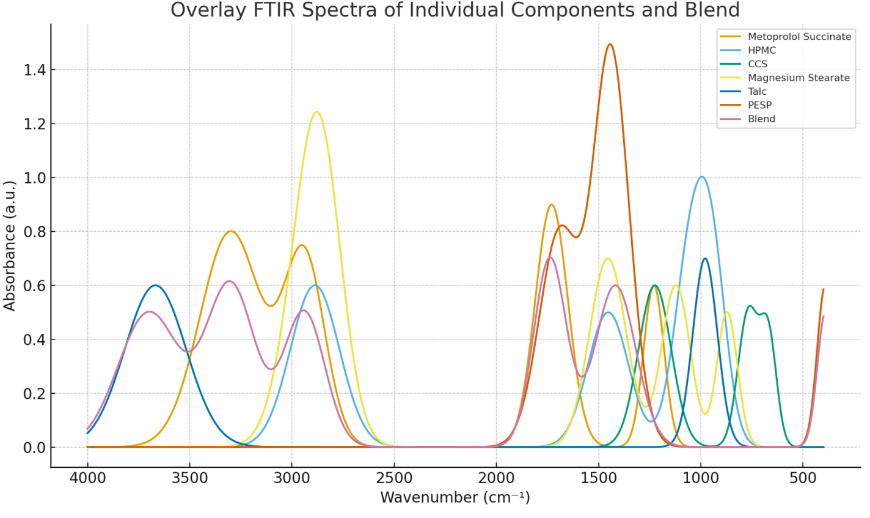
Fig. 6: Overlay IR spectrum of processed egg shell powder, Metoprolol Succinate, individual excipients and blend
Table 16: FTIR analysis summary for processed poultry egg shell powder, processed poultry egg shell powder-metoprolol succinate tablet blend with other standard excipients
| Sample | Key functional groups observed | Peaks (cm⁻¹) | Interpretation/Remark |
| Processed poultry egg shell powder | C–O Stretching, C=O Stretching, CO₃²⁻ asymmetric Stretching, Ca–O lattice vibrations | ~1457.61,1690.51, 1418.20,390.56 |
Major component: Calcium carbonate (CaCO₃) in the calcite form, strong carbonate peaks at ~1418, 872, characteristic carbonate peaks confirm CaCO₃ (calcite form) structure |
| Metoprolol Succinate | O–H, N–H, C–H, C=O, C–O Stretching | ~3300, 2940, 1730, 1230 | All expected peaks present for drug; confirms structural integrity |
| Hydroxy Propyl Methyl Cellulose (Grade E5) | C-H Stretching, C-H Bending, C–O Stretching, O-H Bending |
~2887.99,1453.31,943.08, 1051.29,1453.31 |
All characteristic, expected peaks present |
| Cross Carmellose Sodium | O-H Bending, C-H Rocking, C–O Stretching | ~1225.42,668.61,768.94, 1225.42 |
Observed peak wave numbers are consistent with Reference wave numbers |
| Magnesium Stearate | C-H Stretching, C-H Rocking, C–O Stretching, C-H Bending, | ~2850.72,2915.22,675.06, 871.41,1121.51,1455.46 |
All characteristic, expected peaks present, typical fatty acid salt peaks confirmed |
| Talc | O-H Stretching, C–O Stretching | ~3667.67,978.91 | All characteristic, expected peaks present, typical silicate peaks of talc confirmed |
Blend (PESP, Metoprolol Succinate, Hydroxy Propyl Methyl Cellulose, Cross-Carmellose Sodium, Magnesium Stearate) |
CO₃²⁻ asymmetric Stretching, Ca–O lattice vibrations, O-H Stretching, N–H Stretching, C=O Stretching, C-H Stretching, O-H Bending, C-H Rocking, C–O Stretching | ~1418, 390.56,3700,3300, 2940,1738, 2887,1225.42, 668.61, 675, |
Overlay IR spectrum revealed; no significant peak shifts or disappearance observed – indicates physico-chemical compatibility |
† All data are representative of independent determinations; Infrared spectra were obtained, analyzed and interpreted for each ingredient and the blend.
CONCLUSION
The present study systematically characterized processed poultry eggshell powder (PESP) and demonstrated its suitability as a natural, safe, and sustainable matrix base for tablet formulation. The micromeritic data (angle of repose = 25.56°, Carr’s Index = 19.83%) indicated good flow and compressibility, findings comparable to those reported for eggshell-derived calcium carbonate by few other studies, with similar flow indices suitable for direct compression. The median particle size (Dv₅₀ = 208 µm) was consistent, which suggested considerable powder uniformity and packing characteristics. Instrumental analyses, including SEM, XRD, FTIR, and DSC confirmed a porous morphology, high crystallinity (calcite form), and thermal stability of processed PESP. These findings closely aligned with previously reported observations, confirming that the structural integrity of biogenic calcium carbonate remained stable under formulation conditions. The microbial load remained well within pharmacopeial limits, while MTT assay results revealed>60% cell viability even at 1000 µg/ml, consistent with the biocompatibility reported in few studies carried out earlier. When formulated into placebo and drug-loaded tablets, PESP-based formulations exhibited acceptable hardness (≈ 5–6 kg/cm²), friability (<0.5%), rapid disintegration (≈ 1–1.4 min), and>89% cumulative drug release. These performance parameters were comparable to or better than those achieved with conventional diluents such as microcrystalline cellulose or dicalcium phosphate, as discussed in earlier studies. The non-Fickian release kinetics observed in both F7 and F8 formulations confirmed a diffusion–erosion-controlled release mechanism facilitated by the porous matrix structure of processed PESP. Overall, the present findings reinforce that processed poultry eggshell powder is a pharmaceutically acceptable, biocompatible, and environmentally sustainable excipient. Its comparable physicochemical and mechanical characteristics to conventional excipients, coupled with its non-toxic profile and renewable origin, highlight PESP’s potential as an eco-friendly matrix base and a value-added approach for repurposing eggshell waste in pharmaceutical solid dosage development.
ACKNOWLEDGEMENT
"The authors express their sincere gratitude to the Management of BCDA College of Pharmacy and Technology for providing the necessary facilities to carry out this research. The authors are also indebted to Nishka Labs, Hyderabad, for their high-end instrumental analysis and support. They also hereby acknowledge Aaakar Biotech, Lucknow, for conducting the in vitro cell line toxicity study. The authors are thankful to Sree Balaji Test Labs, New Delhi, for the microbiological studies, Choksi Labs. Ltd., Vapi, for carrying out the particle size distribution by bi-laser diffraction study, and Sura Pharma Labs, Hyderabad, for the additional tablet dissolution studies. Finally, the authors would like to express their sincere gratitude towards Farma Hub Overseas, Haridwar, for providing the candidate drug sample, and Nitika Pharmaceuticals, Nagpur, for providing the necessary industrial-grade excipients."
ABBREVIATIONS
SEM – scanning electron microscopy, BLD –bi-laser diffraction, XRD – x-ray diffraction, FTIR – Fourier transform infrared spectroscopy, DSC – differential scanning calorimetry, MTT – 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT assay for cell viability), API – active pharmaceutical ingredient, MCC – microcrystalline cellulose, IBS – irritable bowel syndrome, ODTs – orally disintegrating tablets, DLS – dynamic light scattering, ANOVA – analysis of variance, HaCaT – human adult low calcium high temperature keratinocytes (an immortalized, Human keratinocyte cell line), % CDR-percent cumulative drug release, cfu – colony forming unit
FUNDING
No funding received from any end
DATA AVAILABILITY
Data available from the Corresponding author upon reasonable request.
Statement of Usage of Artificial Intelligence
No AI platform was used in the manuscript preparation.
AUTHORS CONTRIBUTIONS
First author: Concept and design, Data acquisition; Second author: Data compilation, interpretation; Corresponding author: Critical revision of the manuscript, Critical evaluation.
CONFLICT OF INTERESTS
Declared none
REFERENCES
Aulton ME, Taylor K. Aulton’s pharmaceutics: the design and manufacture of medicines. 5th ed. London: Elsevier; 2018.
Rowe RC, Sheskey PJ, Quinn ME, editors. Handbook of pharmaceutical excipients. 6th ed. London: Pharmaceutical Press; 2009.
Podczeck F. Excipients in the pharmaceutical industry. Profiles Drug Subst Excipients Relat Methodol. 2007;33:3-34.
Alderborn G. Tablets and compaction. In: Aulton ME, Taylor K, editors. Aulton’s pharmaceutics. 5th ed. London: Elsevier; 2018. p. 505-40.
Kaushal N, Kaur H, Goyal AK. Pharmaceutical excipients and drug allergies: a review. J Pharm Investig. 2021;51:633-47.
Dooms M. Interactions and intolerance associated with cellulose derivatives used as excipients. Acta Clin Belg. 2018;73(5):365-9.
Patel A, Jacob S, Patil J, Bhargava H. Excipients limitations: challenges in drug formulation and delivery. J Pharm Drug Deliv Res. 2022;11(2):18459.
Shirwaikar A, Shirwaikar A, Prabu SL, Kumar GA. Herbal excipients in novel drug delivery systems. Indian J Pharm Sci. 2008;70(4):415-22. doi: 10.4103/0250-474X.44587, PMID 20046764.
Paradkar AR, Mahadik KR, Pawar AP. Natural polysaccharides as pharmaceutical excipients: a review. Drug Dev Ind Pharm. 2021;47(11):1697-710.
Shakouri A, Wollina U. Time to change theory; medical leech from a molecular medicine perspective leech salivary proteins playing a potential role in medicine. Adv Pharm Bull. 2021;11(2):261-6. doi: 10.34172/apb.2021.038, PMID 33880347.
Yu D, Hoag SW. The impact of diluents on the compaction dissolution and physical stability of amorphous solid dispersion tablets. Int J Pharm. 2024;654:123924. doi: 10.1016/j.ijpharm.2024.123924, PMID 38395318.
Figueiredo A, Auxtero MD, Santo M, Casimiro A, Costa IM. Risks of dairy derived excipients in medications for lactose intolerant and cow milk protein allergic patients. Sci Rep. 2024;14(1):15631. doi: 10.1038/s41598-024-66380-8, PMID 38972872.
Bhatia M, Ahuja G. Hypersensitivity reactions to pharmaceutical excipients: an overview. J Appl Pharm Sci. 2015;5(9):123-31.
Eswaran S, Tack J, Chey WD. Food: the forgotten factor in the irritable bowel syndrome. Gastroenterol Clin North Am. 2011;40(1):141-62. doi: 10.1016/j.gtc.2010.12.012, PMID 21333905.
Cherukuri R, Singh S, Gaddam S. Intestinal obstruction from excessive intake of microcrystalline cellulose in supplements: a case report. Clin Toxicol (Phila). 2018;56(4):345-8.
Agarwal S, Kumar SL, Garg R. Investigative study on impact of solid: liquid lipid ratio and stabilizer amount on some characteristics of nanostructure lipid carriers of quetiapine fumarate. Int J Pharm Investig. 2019;9(2):47-52. doi: 10.5530/ijpi.2019.2.10.
Schreiber R, Guillet C. Case report: severe IgE-mediated hypersensitivity to carboxymethylcellulose with tolerance to crosscarmellose and microcrystalline cellulose. Front Allergy. 2025;6:1663395. doi: 10.3389/falgy.2025.1663395, PMID 41210305.
Jani GK, Shah DP, Prajapati VD, Jain VC. Gums and mucilages: versatile excipients for pharmaceutical formulations. Asian J Pharm Sci. 2009;4(5):309-23.
Fan D, Fang Q. Siderophores for medical applications: imaging sensors and therapeutics. Int J Pharm. 2021;597:120306. doi: 10.1016/j.ijpharm.2021.120306, PMID 33540031.
Mohamed EM, Hassan MF. Eggshell as a potential pharmaceutical excipient: a review. Int J Pharm Sci Rev Res. 2019;54(2):12-8.
Ameh T, Sayyadi R, Ibrahim Y. Evaluation of eggshell powder as a pharmaceutical excipient in tablet formulations. J Pharm Res Int. 2020;32(21):1-11.
Boonyuen S, Thumanu K, Srichana T. Utilization of eggshell waste as pharmaceutical excipient: physicochemical and safety studies. Mater Sci Eng C. 2020;115:111094.
Olorunsola EO, Adikwu MU. The potential of animal-derived excipients in drug formulation: challenges and opportunities. Afr J Pharm Pharmacol. 2013;7(9):451-9.
Patel B, Patel K, Patel H. Evaluation of eggshell-derived calcium carbonate as a natural excipient for tablet formulation. Int J Pharm Pharm Sci. 2022;14(4):88-95.
United States Pharmacopeia (USP). United States Pharmacopeia and national formulary USP 46−NF. Rockville (MD): United States Pharmacopeial Convention; 2023. p. 41.
International council for harmonisation of technical requirements for pharmaceuticals for human use (ICH). Validation of analytical procedures: Q2(R2). Geneva: ICH; 2022.
Hickey AJ, Mansour HM. Pharmaceutical powder and particles: fundamentals and applications. Berlin: Springer; 2018.
Goldstein JI, Newbury DE, Michael JR, Ritchie NW, Scott JH, Joy DC. Scanning electron microscopy and X-ray microanalysis. 4th ed. Berlin: Springer; 2018. doi: 10.1007/978-1-4939-6676-9.
Klug HP, Alexander LE. X-ray diffraction procedures. 2nd ed. New York: John Wiley & Sons; 1974.
Hohne GW, Hemminger WF, Flammersheim HJ. Differential scanning calorimetry. 2nd ed. Berlin: Springer; 2003. doi: 10.1007/978-3-662-06710-9.
Stuart BH. Infrared spectroscopy: fundamentals and applications. Chichester: John Wiley & Sons; 2004. doi: 10.1002/0470011149.
Bhattacharjee S. DLS and zeta potential What they are and what they are not? J Control Release. 2016;235:337-51. doi: 10.1016/j.jconrel.2016.06.017, PMID 27297779.
United States Pharmacopeia (USP). Microbiological examination of nonsterile products: microbial enumeration tests. USP<61>. Rockville (MD): United States pharmacopeial convention; 2023.
Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983;65(1-2):55-63. doi: 10.1016/0022-1759(83)90303-4, PMID 6606682.
Banker GS, Rhodes CT. Modern pharmaceutics. 4th ed. Boca Raton, FL: CRC Press; 2002.
Bolton S, Bon C. Pharmaceutical statistics: practical and clinical applications. 5th ed. Boca Raton, FL: CRC Press; 2010.
Korsmeyer RW, Gurny R, Doelker E, Buri P, Peppas NA. Mechanisms of solute release from porous hydrophilic polymers. Int J Pharm. 1983;15(1):25-35. doi: 10.1016/0378-5173(83)90064-9.