
Int J App Pharm, Vol 18, Issue 2, 2026, 455-463Original Article
SIMULTANEOUS METHOD DEVELOPMENT AND VALIDATION OF GABAPENTIN AND CARBAMAZEPINE IN RAT PLASMA USING LC-MS/MS AND ITS APPLICATION TO PHARMACOKINETIC STUDIES
P. SRIVIDYA1, P. BHARATH1, SK. MUBEENA1, B. SREE RAMUDU2, D. RAMACHANDRAN1*
1Department of chemistry, University College of Sciences, Acharya Nagarjuna University, Nagarjuna Nagar-522510, Guntur (Dt), A. P., India. 2Department of Chemistry, Adikavi Nannaya University Campus, Tadepalligudem-534101, A. P., India
*Corresponding author: D. Ramachandran; *Email: ramchandran.anu22@gmail.com
Received: 30 Aug 2025, Revised and Accepted: 30 Dec 2025
ABSTRACT
Objective: For the bioanalytical method of gabapentin and carbamazepine using tiagabine as the internal standard (IS), a simple, rapid, accurate, specific, and repeatable Liquid Chromatography Tandem Mass Spectrometry (LC-MS/MS) approach was devised.
Methods: A Symmetry C18 column (150x4.6 mm, 3.5µ) and mobile phase of 0.1% formic acid in water and acetonitrile (70:30, v/v) are used in this article to summarise the latest advancements in bioanalytical LC-MS/MS procedures.
Results: Analysis was carried out within 7 min over a good linear concentration range from 6.0ng/ml to 240ng/ml (r2= 0.9999±0.004) for Gabapentin and 2ng/ml to 80ng/ml (r2=0.9998± 0.003) for Carbamazepine. Accuracy, precision, recovery, matrix effect and stability results were found to be within the suitable limits.
Conclusion: When used successfully for the examination of rat pharmacokinetic studies, the application indicates that all system appropriateness, specificity, linearity, and accuracy metrics are in excellent compliance with USFDA requirements.
Keywords: Gabapentin, Carbamazepine, LC-MS/MS, USFDA guidelines, Rat plasma
© 2026 The Authors.Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i2.56696 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
The anticonvulsant [1, 2] medicine carbamazepine is used to treat epilepsy and neuropathic pain [3, 4]; it goes by many brand names, including Tegretol. Used in conjunction with other drugs, it is an adjuvant therapy for schizophrenia [5, 6] and a second-line agent in bipolar disorder [7, 8]. It seems that carbamazepine, along with phenytoin and valproate, is just as effective for both localised and widespread seizures [9, 10]. Myoclonic or absence seizures will not respond to it [11]. In an effort to lessen carbamazepine's negative systemic effects, researchers have created photoswitchable analogues of the medication to allow for local and on-demand light-based modulation of its pharmacological activity (photopharmacology). Analgesia with noninvasive [12] illumination in vivo was shown in a rat model of neuropathic pain by one of these light-regulated drugs, carbadiazocine, which is based on a bridging azobenzene or diazocine. Common conditions treated with carbamazepine include neuropathic pain and seizure disorders. When traditional antipsychotic therapy for schizophrenia has not been successful, it is sometimes used off-label as a second-line treatment for bipolar disorder or in conjunction with an antipsychotic [13, 14]. Nevertheless, there is evidence that it is not effective in treating schizophrenia. Myoclonic and absence seizures do not respond to it. Carbamazepine may be just as effective (as measured by people continuing to take the medication) and efficacious (as measured by the medicine reducing seizure recurrence and improving remission) as phenytoin and valproate, but each person's medication choice should be carefully considered because more research is needed to find out which medication is best for newly-onset seizures.
The anticonvulsant drug gabapentin is used to treat neuropathic pain and partial seizures of epilepsy; it is marketed under many trade names, including Neurontin. This medicine is often used to alleviate neuropathic pain, which may be caused by diabetic neuropathy [15, 16], postherpetic neuralgia [17, 18], or central pain,. About 30–40% of patients treated with gabapentin for diabetic neuropathy or postherpetic neuralgia report a significant improvement in their symptoms, indicating a moderate level of effectiveness. Similar to other gabapentinoid medications [19], gabapentin reduces the activity of the α2δ-1 protein, which is encoded by the CACNA2D1 gene. This protein was first recognised as an auxiliary subunit of voltage-gated calcium channels. See Pharmacodynamics, however, for more on it. Gabapentin decreases the release of excitatory neurotransmitters [20], mostly glutamate, by attaching to α2δ-1. Consequently, it lessens the overexcitation of neural networks in the brain and spinal cord. The two most prevalent adverse effects are drowsiness and vertigo [21, 22]. Some serious adverse effects include respiratory depression [23] and allergic responses [24, 25]. There is a warning about the increased risk of suicide with gabapentin and all other FDA-approved antiepileptic medications. People who have renal problems should take lower dosages.
Some literatures were available for gabapentin [26-28] and carbamazepine [29-31] individually. The lack of a simultaneous method for these two specific drugs in rat plasma, highlighting the clinical or research need for such a method. The study's overarching goal was to establish a novel, sensitive, and fast LC-MS/MS technique for the concurrent determination of gabapentin and carbamazepine in rat plasma, with tiagabine serving as an internal reference (In the simultaneous procedure two deuterated standards are used for analysis, It was critical to separate. So that I selected same category drug of Tiagabine as an internal standard).
MATERIALS AND METHODS
Chemicals and reagents
Merck (India) Ltd. of Worli, Mumbai, India, supplied the acetonitrile and formic acid, water (HPLC grade). We obtained all of the active pharmaceutical ingredients (APIs) for gabapentin, tigabine, and carbamazepine from Zydus Cadila Healtha Care Ltd in Ahmedabad to use as reference standards.
Equipment
An HPLC system, namely a Waters Alliance e2695 type, was used in conjunction with a QTRAP 5500 triple quadrupole mass spectrometer. An operation was carried out using the ABSCIEX software [32].
LCMS
The examination was performed on a mass spectrometer QTRAP 5500 triple quadrupole instrument with a positive ion electrospray ionization interface. Working parameters of mass spectrometry after optimization as follows: Ion spray voltage 5500 V; temperature source 550 °C; Drying gas temperature 120-250 °C; Collision gas Nitrogen; Pressure 55 psi; Drying gas flow stream-5 ml/min; Declustering potential-40 V; Entrance potential-45 V; Exit potential-15 V; Capillary voltage-5500 V and Dwell time 1 sec respectively. MRM mode was employed to monitor ion pairs of mass: m/z 58.6342 → 172.2378, m/z 61.2047 → 237.2695 for Gabapentin and Carbamazepine, m/z 83.6591 → 376.5487 Tiagabine (Internal standard).
Chromatographic conditions
Symmetry C18 columns (150 x 4.6 mm, 3.5 micron) were used for isocratic mode at room temperature chromatographic separation. For the mobile phase, we used a 70:30 v/v combination of acetonitrile and 0.1% formic acid, with a flow rate of 1 ml/min. There was a 7 min runtime with an injection rate of 10µl.
Preparation of standard and internal control samples
Preparation of Gabapentin Parent Stock Solution (4800 ng/ml)
The standard solution of Gabapentin was prepared by accurately transferring 6 mg into 100 ml volumetric flask, dissolved the contents with diluent, and adjusted the final volume to the mark. The parent stock solution of the Gabapentin was prepared by transferring 0.8 ml to 10 ml volumetric flasks and adjusting the final volume with diluent.
Preparation of carbamazepine parent stock solution (1600 ng/ml)
The standard solution of Carbamazepine was prepared by accurately transferring 8 mg into 100 ml volumetric flask, dissolved the contents with diluent, and adjusted the final volume to the mark. The parent stock solution of the Carbamazepine was prepared by transferring 0.2 ml to 10 ml volumetric flasks and adjusting the final volume with diluent.
Preparation of standard stock solution (Gabapentin-480 ng/ml and carbamazepine-160 ng/ml)
Gabapentin and Carbamazepine combined working stock solution was prepared by transferring 1 ml of each drug working parent stock solution to a 10 ml volumetric flask, then diluting the mixture to the final volume with diluent.
Preparation of tiagabine (Internal standard) stock solution
The standard solution of Tiagabine was prepared by accurately transferring 6 mg into 100 ml volumetric flask, dissolved the contents with diluent, and adjusted the final volume to the mark. The parent stock solution of the Tiagabine was prepared by transferring 0.8 ml to 10 ml volumetric flasks and adjusting the final volume with diluent.
Preparation of standard solution
Here we used liquid-liquid extraction method for the extraction of drugs from plasma. We prepared the drug sample by transferring 0.5 ml of the combined standard stock solution into 2 ml centrifuged vials. To this 200 μl plasma, 500 μl internal standard stock, 300 μl acetonitrile, and 500 μl diluent were added, and the mixture was centrifuged at 4000 RPM for about 20 min. Later, the supernatant was collected and loaded into an HPLC vial.
Bioanalytical technique validation
Selectivity, sensitivity, linearity, accuracy, precision, matrix condition, recovery research, re-injection repeatability, and stability were all areas where the approach was shown to be valid [33, 34].
Selectivity
To ensure retention time selectivity and rule out interference, six separate rat plasma samples were analyzed.
Matrix effect
The matrix effect was determined by comparing the height-area ratio of six different drug-free plasma samples of gabapentin and carbamazepine. Six separate plasma lots were used in triplicate experiments conducted at MQC levels with an acceptable CV of ≤ 15%.
Precision and accuracy
Replicated analyses of internal control samples at four different quality control levels (LLOQC, LQC, MQC, and HQC) were used to get this result. The acceptable limit of CV for LQC, MQC, HQC is 15% and for LLOQ is 20%, the accuracy should be within 85%-115% for LQC, MQC, HQC and for LLQC is 80%-120%.
Recovery
The procedure involves extracting gabapentin and carbamazepine from six replicate samples at each internal control concentration. Standard height regions, both extracted and unextracted, are compared to one another to determine recovery [35].
Carry over
When a sample is diluted with blank matrix, any analyte that remains after the chromatographic system processes it with analyte concentrations over the Upper Limit of Quality Control (ULOQC) is considered a carryover [36].
Dilution integrity
Diluting a sample with blank matrix after spiked with an analyte concentration higher than the ULOQC should explain the dilution integrity [37].
Stability
Through contrasting the stability sample taken from a freshly prepared stock sample with the stability sample taken during the act of stock solution stability [38]. Plasma sample stability experiments were conducted with six replicates at both the LQC and HQC concentration levels. If the change is less than 15%, the analyte was deemed stable according to US FDA criteria [39]. We tested the integrity of spiking rat plasma that had been kept at room temperature for twenty-four hours. After being kept at room temperature in an auto sampler for twenty-four hours, the stability of spiked rat plasma was assessed. By comparing the extract plasma samples that were injected immediately with those that were re-injected after storage with wet extract stability at room temperature after 12 h and 18 h at 2-8 °C, the auto sampler stability (LQC, MQC, and HQC) was assessed. To test the repeatability of reinjection, we compared plasma samples that were extracted and injected right away with those that were reinjected after being stored in the dry extract stable at room temperature for 12 h and 18 h at-20±3 °C. The stability of the samples was tested by comparing them to newly spiked internal control samples and steadiness samples that had been frozen at-31 °C and thawed three times. The stability test was run for 7 d at 7 °C. The concentrations obtained after 24 h were compared to the starting concentration in order to evaluate the stability over the long term.
Pharmacokinetic study
Biological E Limited in Hyderabad, India, supplied six white New Zealand rats (about 250 g) for in vivo pharmacokinetic investigations. The Animal Ethics Committee of the Institute gave their stamp of approval to the study's protocol (Reg. No: 1250/PO/RcBi/S/09/CPCSEA). The animals were housed in similar laboratory conditions with access to endive, carrots, fresh corn (few amounts only). The animals were kept at a temperature of 21-24 °C and humidity was 50-55%. Before experimentation, all animals were made to fast overnight and had water ad libitum. Each rat was given the samples when it was fasting. Blood was collected into centrifugal radioimmunoassay vials containing EDTA from each animal at specified intervals through retro-orbital plexus, centrifuged at 4000 rpm for about 15–20 min to collect the supernatant plasma. Blood samples were collected from rats at 1, 2, 3, 4, 8, 12, 16, and 20 h after oral administration of Gabapentin and Carbamazepine. Following a temperature range of 2-8 °C, the blood was spun at 4000 rpm for 20 min. The transparent plasma supernatant was collected and kept at-30 °C until it could be analyzed. We used a newly developed analytical approach to determine the drug concentration in the plasma samples after subjecting them to liquid-liquid phase extraction. The animals were taken back to the animal shelter for rehabilitation after the research.
Plasma concentration data were used to assess the pharmacokinetic characteristics for the oral administration of carbamazepine and gabapentin. Standard pharmacokinetic parameters include area under the curve (AUC), maximum concentration (Cmax), time to peak concentration (Tmax), and the time at which Cmax occurred. From zero to the infinity point of the concentration-time curve, the data was measured using the trapezoidal rule approach. From the graph, we were able to get Cmax and Tmax. The mean±SD is used to represent all values. (SD) stands for standard deviation.
RESULTS AND DISCUSSION
Using electrospray ionisation yields the best results when using this method's air pressure chemical ionisation mode. Gabapentin and carbamazepine, when administered in the positive ion mode with a mobile phase flow rate of 10 µl/min, provide excellent sensitivity and signal stability when used continuously with electro spray ions.
Specificity and selectivity
Using optimized parameters, the selectivity and specificity of the method were determined by injecting six different animal-extracted blank plasma samples and plasma spiked with internal standards into the LC-MS/MS. The results (fig. 1-3) showed that plasma components don’t affect the analyte’s retention time value ranges or internal standards. This means that the new method is selective.
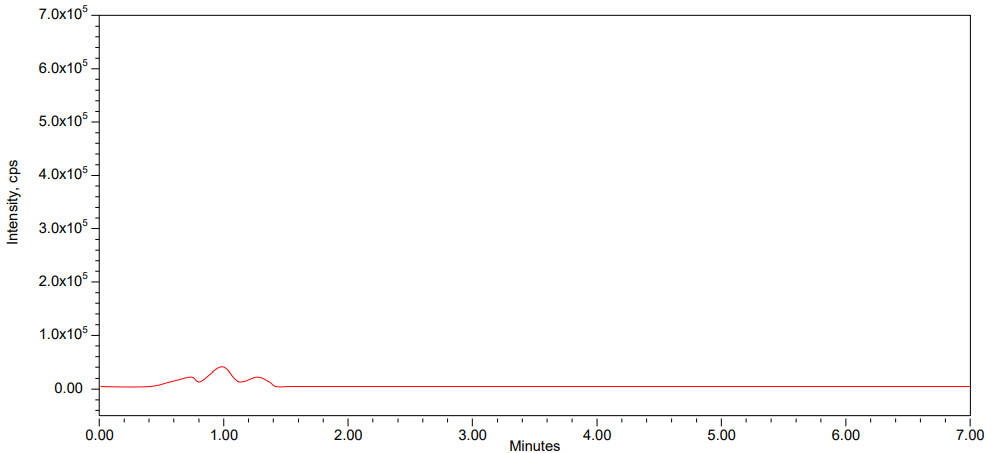
Fig. 1: Blank chromatogram
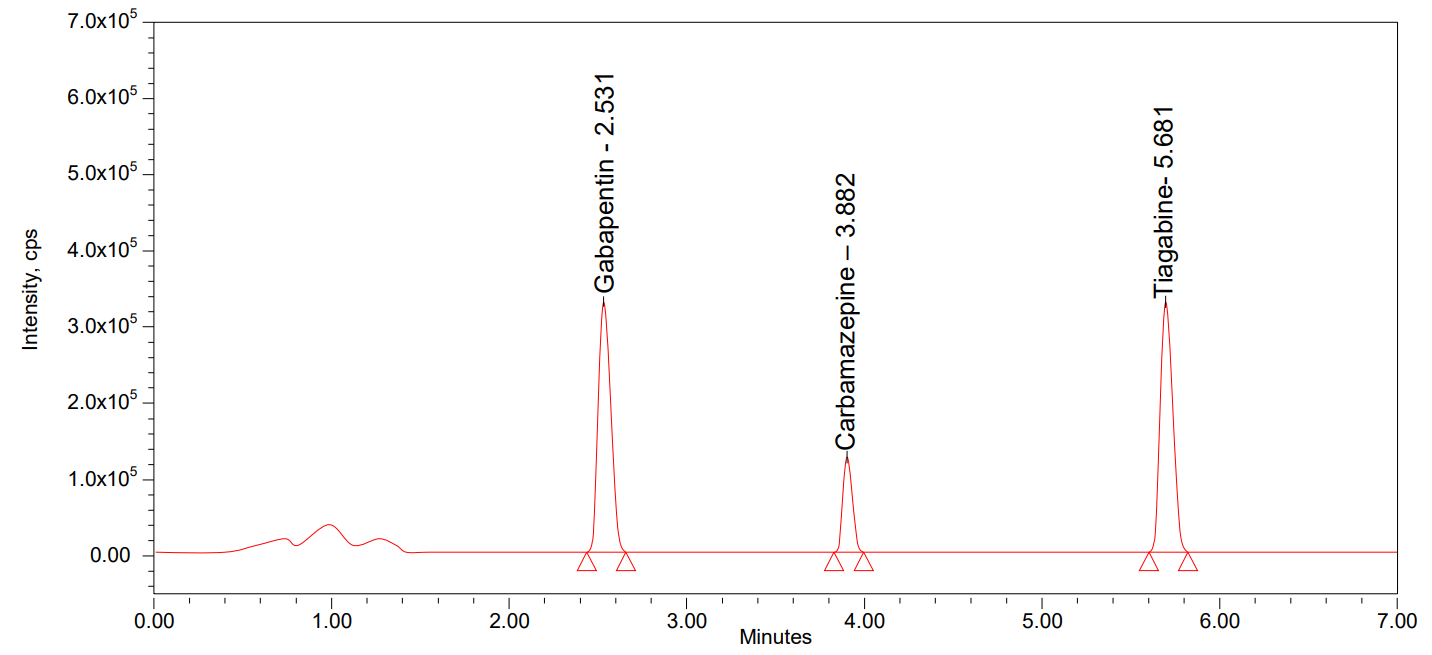
Fig. 2: Standard chromatogram
Matrix effect
Under these conditions, the matrix impact [40] on analyte ionisation is within an acceptable range, as the percent RSD (Relative Standard Deviation) for within-signal ion suppression/enhancement for gabapentin and carbamazepine in LC-MS/MS was one percent. Gabapentin had a LQC of 96.58 and an HQC of 98.11 in the matrix effect, whereas carbamazepine had 96.63 and 97.94%. The relative CVs for the two medications at the LQC and HQC levels were 0.89 and 0.10 and 1.74 and 0.27, respectively. This result shows that the analyte's ionization is affected by the matrix effect within the acceptable range.
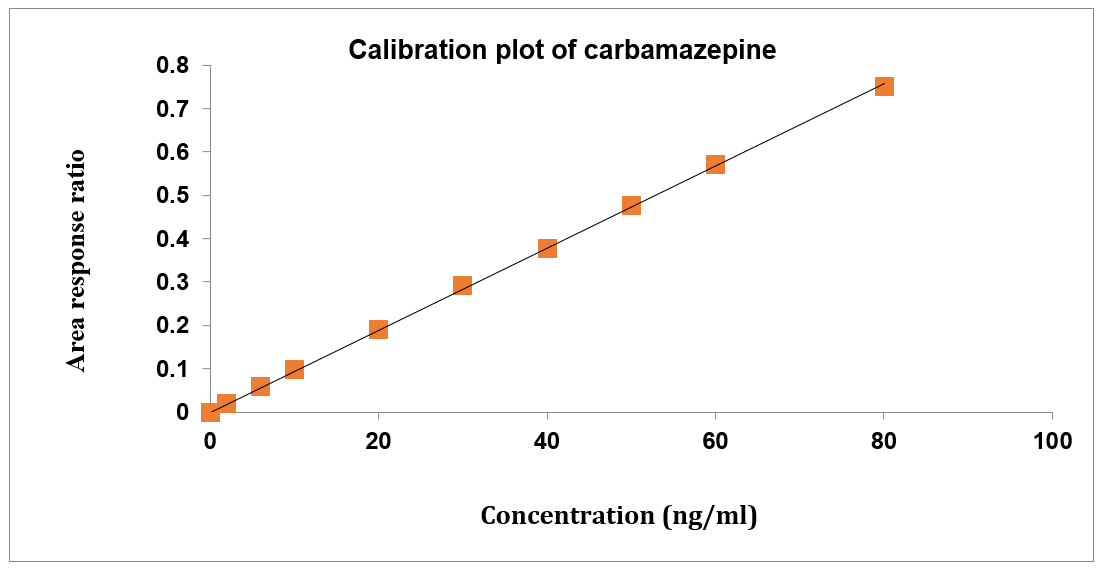
Linearity
Concentration had a direct correlation with the peak area ratio of the standards used for calibration. Both gabapentin and carbamazepine have concentration ranges of 6–240 ng/ml and 2-80 ng/ml, respectively. Table 1 shows the results of the linearity test for gabapentin and carbamazepine, and fig. 4 shows the calibration plots for both drugs [41]. The correlation coefficient for gabapentin and carbamazepine was determined to be 0.999, and the calibration curves seemed to be linear.
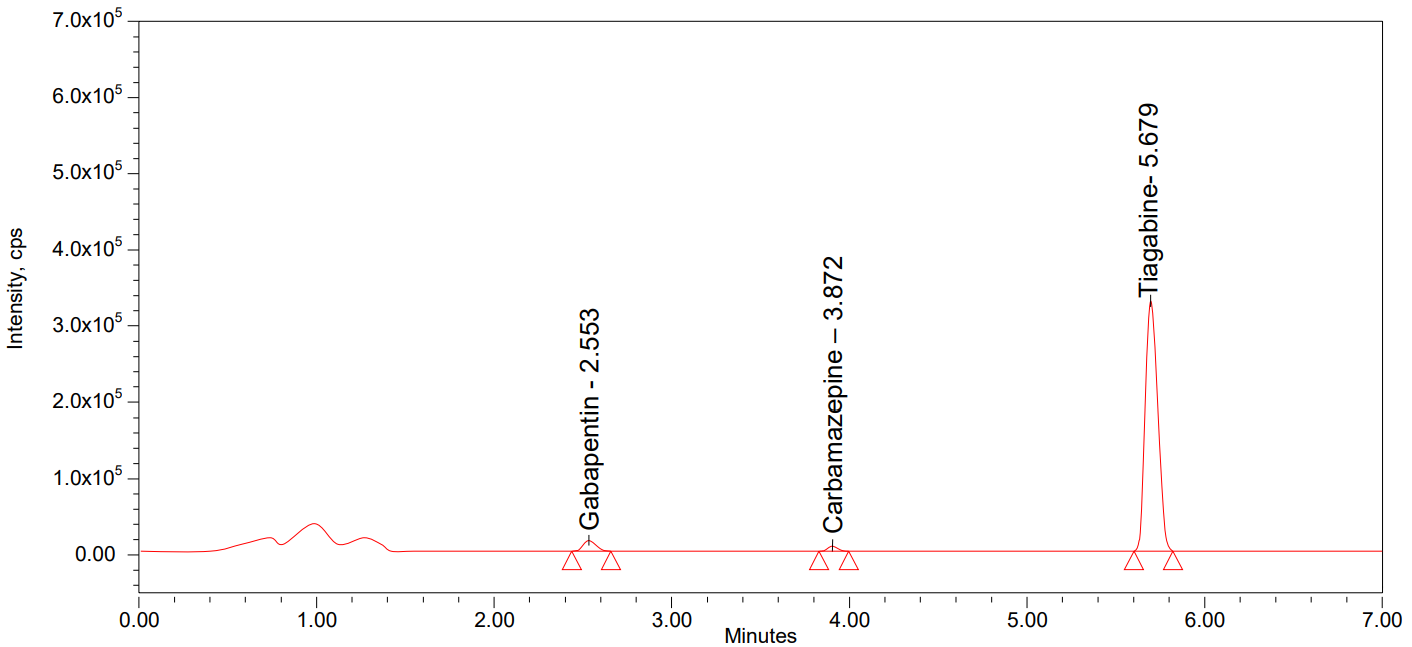
Fig. 3: LLOQ chromatogram
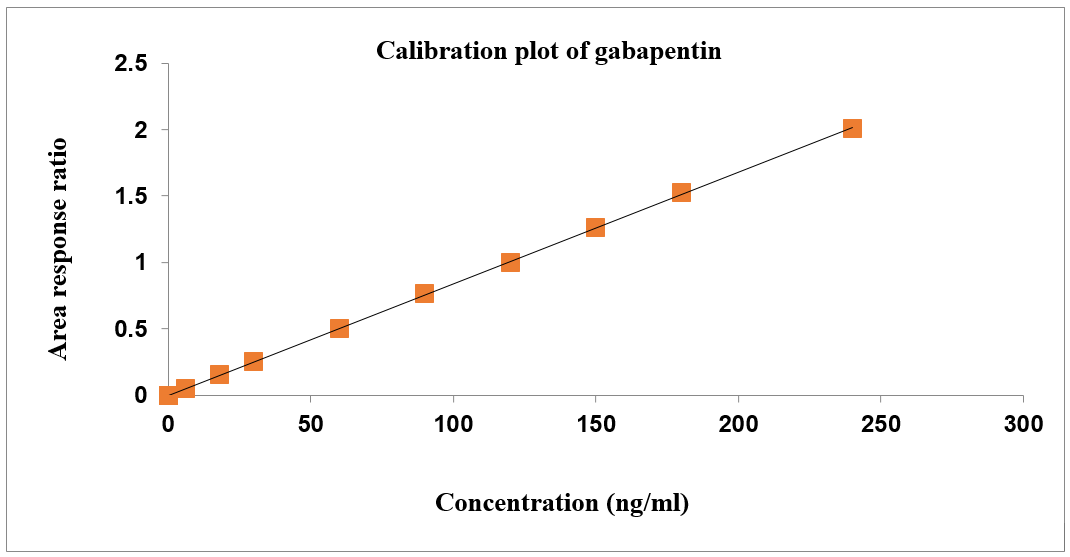
A
B
Fig. 4: Calibration plots of (A) Gabapentin and (B) Carbamazepine
Table 1: Results of linearity
| Linearity | Gabapentin | Carbamazepine | ||||||
| Conc. (ng/ml) | Analyte Area | IS Area | Area response ratio (Analyte/IS) | Conc. (ng/ml) | Analyte area | IS Area | Area response ratio (Analyte/IS) | |
| 1 | 6 | 0.174 | 3.254 | 0.053 | 2 | 0.065 | 3.254 | 0.020 |
| 2 | 18 | 0.507 | 3.235 | 0.157 | 6 | 0.193 | 3.235 | 0.060 |
| 3 | 30 | 0.823 | 3.247 | 0.253 | 10 | 0.318 | 3.247 | 0.098 |
| 4 | 60 | 1.639 | 3.265 | 0.502 | 20 | 0.621 | 3.265 | 0.190 |
| 5 | 90 | 2.466 | 3.229 | 0.764 | 30 | 0.942 | 3.229 | 0.292 |
| 6 | 120 | 3.268 | 3.258 | 1.003 | 40 | 1.229 | 3.258 | 0.377 |
| 7 | 150 | 4.095 | 3.242 | 1.263 | 50 | 1.543 | 3.242 | 0.476 |
| 8 | 180 | 4.921 | 3.231 | 1.523 | 60 | 1.848 | 3.231 | 0.572 |
| 9 | 240 | 6.576 | 3.274 | 2.009 | 80 | 2.461 | 3.274 | 0.752 |
| Slope | 0.0084 | Slope | 0.0095 | |||||
| Intercept | 0.00330 | Intercept | 0.00109 | |||||
| CC | 0.99993 | CC | 0.99982 | |||||
Precision and accuracy
The accuracy and precision [42] were determined by combining the individual test results from several internal control samples. The statistics presented made it very clear that the approach was spot on and productive. Tables 2 and 3 provide the accuracy findings for gabapentin and carbamazepine, respectively. Results for gabapentin range from 94.54 to 98.53 on the quality control samples, whereas those for carbamazepine are 94.46 to 98.86. The percentage of gabapentin and carbamazepine in the total internal control samples is less than 5%. The acceptable limit of recovery was 85-115% for LQC, MQC, HQC and for LLQC the limit is 80-120%. [We used Microsoft Office Excel's function of STDEV to calculate standard deviation and we get following results and %Accuracy also the same value as we use the formula (% measured/nominal *100)].
Recovery
The recoveries for Gabapentin and Carbamazepine at LQC, MQC and HQC levels the results demonstrated that the bioanalytical method had good extraction efficiency. This also showed that the recovery wasn’t hooked into concentration. The recoveries for Gabapentin (95.76%-98.10%) and Carbamazepine (95.55%-98.64%) at LQC, MQC and HQC levels and % CV ranged from 0.05-0.83 for Gabapentin and 0.70-1.34 for Carbamazepine. The results demonstrated that the bioanalytical method had good extraction efficiency [44].
Table 2: Precision and accuracy of gabapentin
| S. No. | HQC | MQC | LQC | LLQC |
| Nominal concentration (ng/ml) | ||||
| 180 | 120 | 18 | 6 | |
| Measured concentration (ng/ml) | ||||
| 1 | 177.8 | 117.7 | 17.2 | 5.6 |
| 2 | 175.7 | 118.4 | 17.6 | 5.7 |
| 3 | 178.8 | 118.1 | 17.2 | 5.8 |
| 4 | 178.1 | 118.9 | 17.6 | 5.7 |
| 5 | 176.4 | 117.3 | 17.4 | 5.6 |
| 6 | 177.2 | 117.2 | 17.3 | 5.5 |
| n | 6 | 6 | 6 | 6 |
| mean | 177.3 | 117.9 | 17.4 | 5.7 |
| SD | 1.12463 | 0.65253 | 0.19994 | 0.10087 |
| % CV | 0.63 | 0.55 | 1.15 | 1.78 |
| % Accuracy | 98.50% | 98.25% | 96.67% | 95.00% |
Table 3: Precision and accuracy of carbamazepine
| S. No. | HQC | MQC | LQC | LLQC |
| Nominal concentration (ng/ml) | ||||
| 60 | 40 | 6 | 2 | |
| Measured concentration (ng/ml) | ||||
| 1 | 59.2 | 39.4 | 5.8 | 1.8 |
| 2 | 59.0 | 39.7 | 5.7 | 1.9 |
| 3 | 59.2 | 39.5 | 5.8 | 1.9 |
| 4 | 59.1 | 39.6 | 5.7 | 1.9 |
| 5 | 59.1 | 39.4 | 5.7 | 1.9 |
| 6 | 59.1 | 39.5 | 5.6 | 1.9 |
| n | 6 | 6 | 6 | 6 |
| Mean | 59.1 | 39.5 | 5.7 | 1.9 |
| SD | 0.07037 | 0.10655 | 0.07037 | 0.03364 |
| % CV | 0.12 | 0.27 | 1.23 | 1.79 |
| % Accuracy | 98.50% | 98.75% | 95.00% | 95.01% |
Ruggedness
In all four quality control (HQC, LQC, MQC, and LLQC) samples, the percentage recoveries and percent CV of gabapentin and carbamazepine showed up within acceptable ranges when measured with two separate analysers using two separate columns. The method's findings demonstrated its robustness. For gabapentin, the percent recoveries varied between 96.79%-98.13%, while for carbamazepine, they ranged from 95.01% to 97.96%. For gabapentin, the %CV values were 0.42-0.49, while for carbamazepine, they were 1.11-1.27. The method's findings demonstrated its robustness.
Auto sampler carryover
Injecting blank rat plasma samples with LLQC and ULQC sequentially at the retention durations of gabapentin and carbamazepine did not result in an observable peak area response. Auto sampler carryover is not shown by this approach.
Stability
A solution stability study was conducted on gabapentin and carbamazepine, which were produced using diluent and then stored in a refrigerator at 2-8 °C. Stock solutions that were produced 24 h before to use were paired with fresh stock solutions. Both the bench top and auto sampler plasma stability remained constant during twenty-four hours, with the auto sampler maintaining stability for twenty-four hours at 20 °C. Future stability testing confirmed that both gabapentin and carbamazepine were stable for up to 24 h when stored at-30 °C. In long-term stability, the samples were stable up to 21 d, but on going to 28 d Gabapentin was not stable in LQC condition and Carbamazepine also not stable in both LQC and MQC conditions. In tables 4, 5, we can see the overall stability findings of carbamazepine and gabapentin.
In vivo pharmacokinetic evaluation
Fig. 5 shows the temporal patterns of plasma concentrations of gabapentin and carbamazepine in rats. The experimental formulations in both instances showed a bell-shaped curve on the graph. The fact that gabapentin and carbamazepine were detectable in the blood for 8 and 16 h after oral and intravenous dosing, respectively, suggests that the formulation was successful in releasing the drugs [44].
The results of the calculations of the pharmacokinetic parameters Cmax, Tmax, T1/2, AUC0-t, and AUC0-∞ are shown in table 6. Gabapentin had a Cmax of 116.360 ng/ml and carbamazepine a Cmax of 39.463 ng/ml. It was determined that the Tmax for carbamazepine was 8 h and that for gabapentin it was 3 h. Gabapentin and carbamazepine had t½ values of 8 h and 16 h, respectively. Gabapentin had an AUC0-t of 439 ng-h/ml and carbamazepine of 315 ng-h/ml. Table 6 displayed the pharmacokinetic parameters.
Table 4: Stability results of gabapentin
| Stability experiment spiked plasma | Mean area±SD | % CV | %Recovery |
| Bench top stability | LQC | 17.3±0.18708 | 1.08 |
| MQC | 117.9±0.42622 | 0.36 | |
| HQC | 177.4±0.51153 | 0.29 | |
| Auto sampler stability | LQC | 17.4±0.25626 | 1.46 |
| MQC | 118.1±0.30332 | 0.26 | |
| HQC | 177.6±0.26077 | 0.15 | |
| Long term (Day28) stability | LQC | 15.0±0.26394 | 1.76 |
| MQC | 102.1±0.25820 | 0.17 | |
| HQC | 153.6±0.34641 | 0.23 | |
| Wet extract 18 H stability | LQC | 17.2±0.22527 | 1.99 |
| MQC | 115.8±0.35777 | 0.31 | |
| HQC | 173.2±0.28810 | 0.17 | |
| Dry extract 18 H stability | LQC | 17.2±0.27869 | 1.60 |
| MQC | 115.7±0.18708 | 0.16 | |
| HQC | 172.9±0.31411 | 0.18 | |
| Freeze thaw stability | LQC | 17.4±0.27325 | 1.56 |
| MQC | 118.1±0.19408 | 0.16 | |
| HQC | 177.6±0.20976 | 0.12 | |
| Short term stability | LQC | 16.9±0.28284 | 1.66 |
| MQC | 114.4±0.21602 | 0.19 | |
| HQC | 171.3±0.20412 | 0.12 |
Mean±SD (n=6)
Table 5: Stability results of carbamazepine
| Stability experiment spiked plasma | Mean area±SD | % CV | %Recovery |
| Bench top stability | LQC | 5.8±0.10328 | 1.77 |
| MQC | 39.4±0.18619 | 0.47 | |
| HQC | 59.3±0.18708 | 0.32 | |
| Auto sampler stability | LQC | 5.7±0.08615 | 1.43 |
| MQC | 39.5±0.27869 | 0.71 | |
| HQC | 59.1±0.17889 | 0.30 | |
Long term (Day 28) stability |
LQC | 5.0±0.08944 | 1.79 |
| MQC | 33.7±0.26077 | 0.78 | |
| HQC | 52.1±0.28810 | 0.25 | |
| Wet extract 18 h stability | LQC | 5.7±0.08944 | 1.54 |
| MQC | 38.5±0.22804 | 0.59 | |
| HQC | 57.8±0.20012 | 0.35 | |
| Dry extract 18 h stability | LQC | 5.6±0.07528 | 1.34 |
| MQC | 38.2±0.21370 | 0.56 | |
| HQC | 57.9±0.22804 | 0.40 | |
| Freeze thaw stability | LQC | 5.8±0.07528 | 1.30 |
| MQC | 39.0±0.14142 | 0.36 | |
| HQC | 59.1±0.13784 | 0.23 | |
| Short term stability | LQC | 5.6±0.08165 | 1.47 |
| MQC | 37.9±0.11690 | 0.31 | |
| HQC | 57.1±0.14720 | 0.26 |
n=6
Table 6: Pharmacokinetic parameters of gabapentin and carbamazepine
| Pharmacokinetic parameters | Gabapentin | Carbamazepine |
| AUC0-t | 439 ng-h/ml | 315 ng-h/ml |
| Cmax | 116.360 ng/ml | 39.463 ng/ml |
| AUC0-∞ | 566 ng-h/ml | 361 ng-h/ml |
| tmax | 3 h | 8 h |
| T1/2 | 4.98 h | 3.5 h |
AUC0−∞: Area under the curve extrapolated to infinity, AUC0−𝑡: Area under the curve up to the last sampling time, Cmax: The maximum plasma concentration, Tmax: The time to reach peak concentration, T1/2: Time the drug concentration
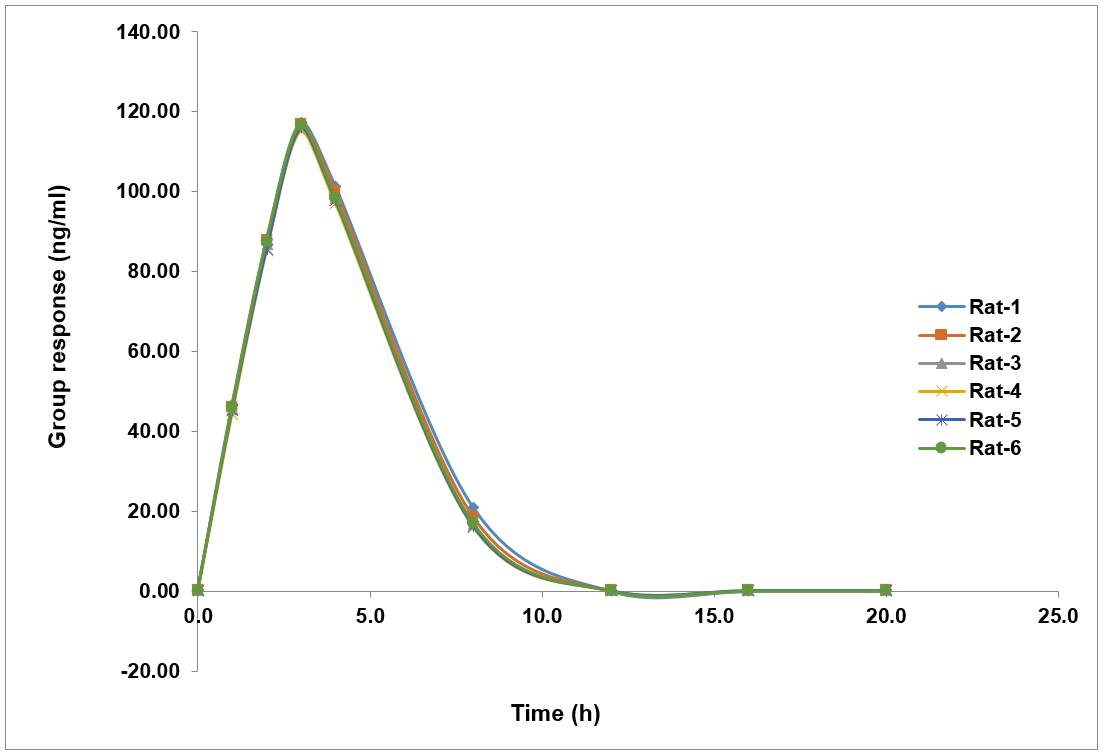
A
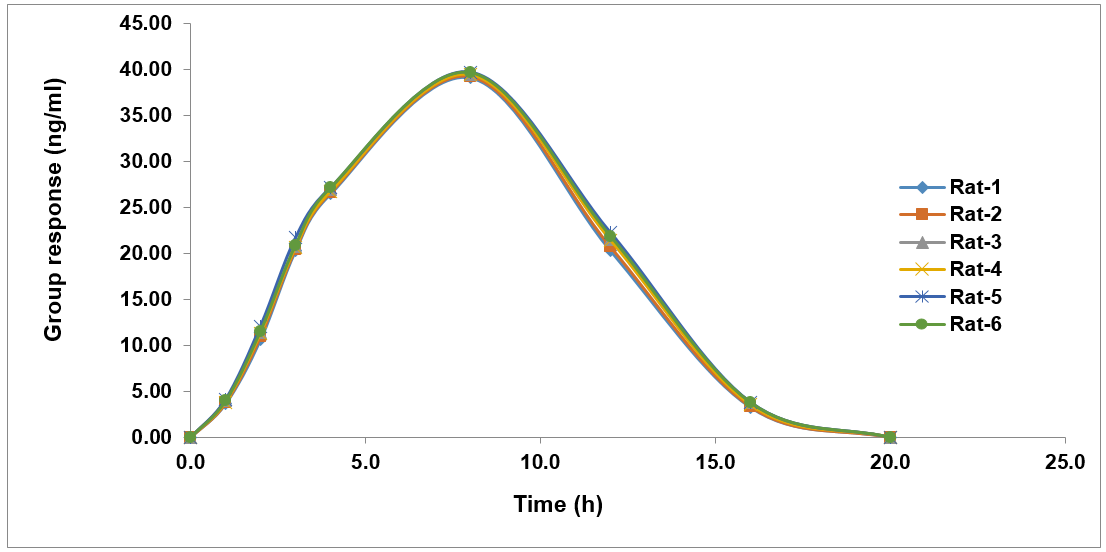
B
Fig. 5: Mean plasma concentration-time profile of (A) Gabapentin and (B) Carbamazepine
CONCLUSION
To determine carbamazepine and gabapentin in rat plasma, the first ever validated technique was a highly sensitive HPLC-ESI-LCMS/MS. In this case, we have a bioanalytical approach that is robust, rapid, and repeatable. Following USFDA requirements, this approach was verified. In order to see the studied analyte in bodily fluids and conduct pharmacokinetic research, a simple and effective technique was devised.
ACKNOWLEDGEMENT
I thankful to my guide for encouragement and supporting to finish this research work.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
All of the drug-related material has been compiled by Mubeena. The research samples and manuscript preparation were done by Bharath, Sree Ramudu, and Srividya. The data was checked by Ramachandran, who also read the manuscript.
CONFLICTS OF INTERESTS
Author declares that there have been no conflicts of interest
REFERENCES
Al-Otaibi F. An overview of structurally diversified anticonvulsant agents. Acta Pharm. 2019;69(3):321-44. doi: 10.2478/acph-2019-0023, PMID 31259739.
Pennell PB, Karanam A, Meador KJ, Gerard E, Kalayjian L, Penovich P. Antiseizure medication concentrations during pregnancy: results from the maternal outcomes and neurodevelopmental effects of antiepileptic drugs (MONEAD) study. JAMA Neurol. 2022;79(4):370-9. doi: 10.1001/jamaneurol.2021.5487, PMID 35157004.
Tadokoro T, Bravo Hernandez M, Agashkov K, Kobayashi Y, Platoshyn O, Navarro M. Precision spinal gene delivery-induced functional switch in nociceptive neurons reverses neuropathic pain. Mol Ther. 2022;30(8):2722-45. doi: 10.1016/j.ymthe.2022.04.023, PMID 35524407.
Kaur J, Ghosh S, Sahani AK, Sinha JK. Mental imagery as a rehabilitative therapy for neuropathic pain in people with spinal cord injury: a randomized controlled trial. Neurorehabil Neural Repair. 2020;34(11):1038-49. doi: 10.1177/1545968320962498, PMID 33040678.
Murrie B, Lappin J, Large M, Sara G. Transition of substance-induced brief and atypical psychoses to schizophrenia: a systematic review and meta-analysis. Schizophr Bull. 2020;46(3):505-16. doi: 10.1093/schbul/sbz102, PMID 31618428.
Rouy M, Saliou P, Nalborczyk L, Pereira M, Roux P, Faivre N. Systematic review and meta-analysis of metacognitive abilities in individuals with schizophrenia spectrum disorders. Neurosci Biobehav Rev. 2021;126:329-37. doi: 10.1016/j.neubiorev.2021.03.017, PMID 33757817.
Vieira IS, Barreto NT, Pedrotti Moreira F, Mondin TC, Simjanoski M, Cardoso TA. Sleep alterations in individuals recently diagnosed with bipolar disorder across different mood stages. Psychiatry Res. 2021;299:113824. doi: 10.1016/j.psychres.2021.113824, PMID 33756207.
Zhong R, Wang Z, Zhu Y, Wu X, Wang X, Wu H. Prevalence and correlates of non-suicidal self-injury among patients with bipolar disorder: a multicenter study across China. J Affect Disord. 2024;367:333-41. doi: 10.1016/j.jad.2024.08.231, PMID 39233245.
Chu H, Zhang X, Shi J, Zhou Z, Yang X. Antiseizure medications for idiopathic generalized epilepsies: a systematic review and network meta-analysis. J Neurol. 2023;270(10):4713-28. doi: 10.1007/s00415-023-11834-8, PMID 37378757.
Lindquist BE, Timbie C, Voskobiynyk Y, Paz JT. Thalamocortical circuits in generalized epilepsy: pathophysiologic mechanisms and therapeutic targets. Neurobiol Dis. 2023;181:106094. doi: 10.1016/j.nbd.2023.106094, PMID 36990364.
Andrade DM, Hamani C, Minassian BA. Treatment options for epileptic myoclonus and epilepsy syndromes associated with myoclonus. Expert Opin Pharmacother. 2009;10(10):1549-60. doi: 10.1517/14656560903025189, PMID 19527185.
Belzberg M, Mahapatra S, Perdomo Pantoja A, Chavez F, Morrison K, Xiong KT. Minimally invasive therapeutic ultrasound: ultrasound-guided ultrasound ablation in neuro-oncology. Ultrasonics. 2020;108:106210. doi: 10.1016/j.ultras.2020.106210, PMID 32619834.
Mueller C, John C, Perera G, Aarsland D, Ballard C, Stewart R. Antipsychotic use in dementia: the relationship between neuropsychiatric symptom profiles and adverse outcomes. Eur J Epidemiol. 2021;36(1):89-101. doi: 10.1007/s10654-020-00643-2, PMID 32415541.
Durrani US, Vasireddy S, Arshad MZ, Paracha A, Paracha MA, Waheed F. The effect of antipsychotics on prolactinoma growth: a radiological and serological analysis. Cureus. 2023;15(11):e49342. doi: 10.7759/cureus.49342, PMID 38143631.
Jahantigh Akbari N, Hosseinifar M, Naimi SS, Mikaili S, Rahbar S. The efficacy of physiotherapy interventions in mitigating the symptoms and complications of diabetic peripheral neuropathy: a systematic review. J Diabetes Metab Disord. 2020;19(2):1995-2004. doi: 10.1007/s40200-020-00652-8, PMID 33553048.
Xu L, Sun Z, Casserly E, Nasr C, Cheng J, Xu J. Advances in interventional therapies for painful diabetic neuropathy: a systematic review. Anesth Analg. 2022;134(6):1215-28. doi: 10.1213/ANE.0000000000005860, PMID 35051958.
Theresa MS, Brett S, Brant Jeannine M. Postherpetic neuralgia: epidemiology pathophysiology and pain management pharmacology. J Multidiscip Healthc. 2021;9:447-54. doi: 10.2147/JMDH.S106340.
Huerta MA, Garcia MM, Garcia Parra B, Serrano Afonso A, Paniagua N. Investigational drugs for the treatment of postherpetic neuralgia: systematic review of randomized controlled trials. Int J Mol Sci. 2023;24(16):12987. doi: 10.3390/ijms241612987, PMID 37629168.
Varadi G. Mechanism of analgesia by gabapentinoid drugs: involvement of modulation of synaptogenesis and trafficking of glutamate-gated ion channels. J Pharmacol Exp Ther. 2024;388(1):121-33. doi: 10.1124/jpet.123.001669, PMID 37918854.
Teleanu RI, Niculescu AG, Roza E, Vladacenco O, Grumezescu AM, Teleanu DM. Neurotransmitters-key factors in neurological and neurodegenerative disorders of the central nervous system. Int J Mol Sci. 2022;23(11):5954. doi: 10.3390/ijms23115954, PMID 35682631.
Chu EC, Zoubi FA, Yang J. Cervicogenic dizziness associated with craniocervical instability: a case report. J Med Cases. 2021;12(11):451-4. doi: 10.14740/jmc3792, PMID 34804305.
Chu EC, Chin WL, Bhaumik A. Cervicogenic dizziness. Oxf Med Case Rep. 2019;2019(11):476-8. doi: 10.1093/omcr/omz115, PMID 31844531.
N Weingarten T, Deljou A, Sprung J. Research and clinical implications of emerging evidence regarding patterns of postoperative opioid-induced respiratory depression. Biomol Biomed. 2025;25(2):327-37. doi: 10.17305/bb.2024.11123, PMID 39319856.
Bartha I, Almulhem N, Santos AF. Feast for thought: a comprehensive review of food allergy -2023. J2021 Allergy Clin Immunol. 2024;153(3):576-94. doi: 10.1016/j.jaci.2023.11.918, PMID 38101757.
Fukutomi Y, Kawakami Y. Respiratory sensitization to insect allergens: species components and clinical symptoms. Allergol Int. 2021;70(3):303-12. doi: 10.1016/j.alit.2021.04.001, PMID 33903033.
Tony RM, El Hamd MA, Gamal M, Saleh SF, Maslamani N, Alsaggaf WT. Green bio-analytical study of gabapentin in human plasma coupled with pharmacokinetic and bioequivalence assessment using UPLC-MS/MS. Separations. 2023;10(4):234. doi: 10.3390/separations10040234.
Datla S, Imam Pasha S, Koneru A. Various quantitative and qualitative methods for estimation of anti-epileptic drugs in bulk drug formulations and their biological samples a review. World J Pharm Res. 2019;8:579-608. doi: 10.20959/wjpr20192-14155.
Matar KM, Abdel Hamid ME. Rapid tandem mass spectrometric method for determination of gabapentin in human plasma. Chromatographia. 2005;61(9-10):499-504. doi: 10.1365/s10337-005-0542-7.
Sharwaree H, Vaishali U, Rupesh S, Mayuresh G. Development and validation of RP-HPLC method for determination of carbamazepine in rat plasma. Int J Biol Pharm Res. 2013;4(1):30-4.
Tonic Ribarska J, Sterjev Z, Cvetkovska E, Kuzmanovski I, Kiteva G, Suturkova L, Trajkovic Jolevska S. Optimization and validation of bioanalytical SPE‑HPLC method for the simultaneous determination of carbamazepine and its main metabolite carbamazepine‑10,11‑epoxide in plasma. Macedonian Pharm Bull. 2011;57:53–61. doi: 10.33320/maced.pharm.bull.2011.57.006.
Louati K, Mahfoudhi S, Safta F. Development of an analytical method for the plasma determination of five anti epileptics and their metabolites by HPLC: comparison with immunoanalytical methods. RRJCHEM. 2018;7:7-19.
Ramadevi P, Rambabu K. Bio analytical method development and validation for ezetimibe and pitavastain and its applications to pharmacokinetic studies in rat plasma by using LCMS/MS. Int J Res Pharm Sci. 2020;11(4):7854-62. doi: 10.26452/ijrps.v11i4.4670.
Eluru A, Surendra Babu K. Bio analytical method development and validation for aplidine in rat plasma and their pharmacokinetic studies by LCMS. WJPPS. 2019;8(11):1201-9. doi: 10.20959/wjpps201911‑15023.
Ramchandran D, Kethipalli A, Krishnamurthy M. Bioanalytical method development and validation of daunorubicin and cytrarabine in rat plasma by LC-MS/MS and its application in pharmacokinetic studies. J Pharm Sci Res. 2020;12:381-6.
Harsha K, Ravisankar P, Kumar Konidala S, Srinivasa Babu P. Application of newly developed and validated LC-MS/MS method for pharmacokinetic study of adagrasib and pembrolizumab simultaneously in rat plasma. J Chromatogr B. 2024;1241:124171. doi: 10.1016/j.jchromb.2024.124171.
Harsha K, Panchumarthy Ravisankar, Sathish Kumar Konidala, Srinivasa Babu Puttagunta. Quantification of belzutifan in biological samples: LC-MS/MS method validation and pharmacokinetic study in rats. Biomed Chromatogr. 2025;39:e70168.
Raju GE, Pottendla S, Yaparthi S. Bio analytical approach to Ensitrelvir estimation using liquid chromatography tandem mass spectrometry and its application to pharmaceutical research. Asian J Pharm Clin Res. 2025;18(3):25-9. doi: 10.22159/ajpcr.2025v18i3.53760.
Rathore MK, Mohan Reddy TR. Tandem mass spectrometric method for the trace level determination of 2-aminopyridine: a potential genotoxic impurity in tenoxicam API. Int J Pharm Pharm Sci. 2024;16(4):50-6. doi: 10.22159/ijpps.2024v16i4.49902.
Thomas A, Varkey J. Development and validation of a new RP-HPLC analytical method for the determination of etodolac succinic acid co crystals in spiked rabbit plasma. Int J Curr Pharm Sci. 2023;15(2):59-63. doi: 10.22159/ijcpr.2023v15i2.2098.
Raju GE, Pottendla S, Yaparthi S. Establishment and validation of LC-MS/MS technique for Lenacapavir quantification in rat plasma with application to pharmacokinetic assessment. J Appl Pharm Sci. 2025;15(5):112-20. doi: 10.7324/JAPS.2025.229006.
Raju GE, Pottendla S, Yaparthi S. Novel ultraperformance liquid chromatography method for concurrent estimation and pharmacokinetic analysis of favipiravir and molnupiravir in rat plasma. Asian J Pharm Clin Res. 2025;18(2):90-5. doi: 10.22159/ajpcr.2025v18i2.53662.
Talari S, Vejendla A, Shetty RK. Development and validation of a UPLC-MS/MS method for the simultaneous determination of verapamil and trandolapril in rat plasma: application to a pharmacokinetic study. Curr Pharm Anal. 2022;18(3):291-304. doi: 10.2174/1573412917666210302145711.
Sentat T, Lucida H, Widyati W, Nasif H. Development and validation of a bio analytical method for the therapeutic drug monitoring of amikacin in human plasma using ultraperformance liquid chromatography tandem mass spectrometry. Int J Appl Pharm. 2024;16(1):140-4. doi: 10.22159/ijap.2024.v16s1.30.
Prabhakara Rao K, Namburi L, A Amara Babu, Kalyani Koganti, Babji Palakeeti, Koduri SV, Srinivas. Related substances method development and validation of an LC-MS/MS method for the quantification of selexipag and its related impurities in rat plasma and its application to pharmacokinetic studies. SN Appl Sci. 2021;3:321. doi: 10.1007/s42452‑021‑04219‑x.