
Int J App Pharm, Vol 18, Issue 1, 2026, 242-249Original Article
QBD-GUIDED DESIGN OF NLC-BASED BUCCAL FILMS OF A PHYTOCONSTITUENT FOR ENHANCED CHEMOPREVENTION
HIMAN PATEL1, MANISHA SUNIL LALAN2*
1Department of Pharmaceutics, Babaria Institute of Pharmacy, BITS Edu Campus, Vadodara, Gujarat (India). 2Department of Pharmaceutics, Parul Institute of Pharmacy and Research, Faculty of Pharmacy, Parul University, Limda, Waghodia-391760, Vadodara, Gujarat (India)
*Corresponding author: Manisha Sunil Lalan; *Email: manisha.lalan33664@paruluniversity.ac.in
Received: 04 Sep 2025, Revised and Accepted: 01 Dec 2025
ABSTRACT
Objective: This study aimed to develop and optimize a nanostructured lipid carrier (NLC)-based hybrid buccal film of quercetin (QCT) using a Quality by Design (QbD) approach for potential chemoprevention of oral cancer.
Methods: QCT-loaded NLCs were prepared via the solvent injection technique using pre-screened excipients. A Design of Experiments (DoE) framework using the Box-Behnken Design was employed to assess the influence of total lipid content, surfactant concentration, and liquid lipid proportion on critical quality attributes—namely, particle size and drug entrapment efficiency. Transmission electron microscopy (TEM) was used to analyze morphology. Bilayer hybrid buccal films were cast using ethyl cellulose as the backing layer and a blend of hydroxypropyl methylcellulose and Carbopol 934P for the drug-loaded matrix. These films were evaluated for mechanical strength, in vitro drug release, ex vivo buccal permeation, stability, and cytotoxicity using cell line studies.
Results: QCT-NLCs were successfully formulated with glyceryl monostearate (solid lipid), Transcutol HP and Capmul MCM (liquid lipids), and surfactants including soya lecithin and Poloxamer 188. DoE analysis indicated that higher surfactant levels reduced particle size, while increased lipid content enlarged particles but improved entrapment efficiency. The optimized formulation had particle size of 144 nm and an entrapment efficiency of 90.21%, as verified by TEM. The resultant hybrid film demonstrated sustained drug release (80% over 8 h), excellent tensile strength (3.93 MPa), and was stable under accelerated conditions. Ex vivo studies confirmed 72.8% buccal drug permeation in 6 h. Cytotoxicity assays validated the formulation’s safety and chemopreventive potential.
Conclusion: The QCT-loaded NLC hybrid buccal film demonstrated potential as a safe and effective strategy for oral cancer chemoprevention.
Keywords: Quercetin, Chemoprevention, Hybrid system, Buccal film, Nanocarrier
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.56754 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Oral squamous cell carcinoma (OSCC) is one significant cause of both cancer morbidity and mortality [1]. Oral cancer, which includes lip cancer, tongue cancer, mouth cancer, and pharyngeal cancer, is an important global health problem. The total number of new cases alone for 2020, according to the Global Cancer Observatory, is above 377,000 with total deaths exceeding 177,000 [1, 2]. The cancer is most common in India, covering approximately 30% of all cancers and mainly attributed to massive tobacco use, betel quid chewing, and alcohol intake [1]. All of these risk factors lead to chronic inflammation and oxidative stress, which induces carcinogenesis via DNA damage and unrestrained cell growth.
The localized chemopreventive strategies that can arrest malignant progression at the mucosal surface are gaining more attention. Phytochemicals, plant-derived natural products, are emerging as promising candidates in cancer chemoprevention, acting through multiple pathways involved in tumor initiation, promotion, and progression [3]. Quercetin (3,3',4',5,7-pentahydroxyflavone), a flavonoid found in onions, apples, tea, and red wine, possesses potent antioxidant, anti-inflammatory, and anticancer activities. Its anticancer actions are multitargeted, including pro-apoptosis, anti-oxidative, and cell-cycle modulation [3-6]. Quercetin exposes cancer cells to apoptosis via ROS-mediated pathways, alleviates oxidative DNA damage, modulates key proteins for cell cycle control (cyclin D1 and p53), and inhibits the metastasis of tumor cells by regulating matrix metalloproteinases. Recent findings indicate that quercetin exhibits selective toxicity to OSCC cells by inducing S and G2/M phase arrest along with inhibition of cells' proliferation through the PI3K-AKT-mTOR pathway. Quercetin produced dose-dependent chemopreventive effects against OSCC induced chemically in animal models by reducing tumor volume, invasion, and migration while enhancing apoptosis [7, 8].
For all of its advantages, quercetin faces a significant hindrance in the translation into clinical practice: it is poorly soluble in water (approximately 0.01 mg/ml), has a low bioavailability (less than 10% by the oral route), is rapidly metabolized, and does not remain stable in physiological environments [3-6]. These issues are caused by conventional oral administration, which leads to extensive first-pass metabolism, limiting the systemic exposure. Developing of advanced drug delivery systems is essential to overcome these barriers by enhancing solubility, stability, and targeted delivery [9].
Nanotechnology has revolutionized cancer therapeutics and targeting drugs to patients with a very controlled release of drugs. In the second generation, lipid nanoparticles made up of solid and liquid lipids have other advantages in comparison to solid lipid nanoparticles (SLNs), including up to 90% high drug loading capacity, physical stability, and reduced drug release during storage. NLCs offer the potential for increased encapsulation of lipophilic phytochemicals such as quercetin, releasing them over a longer duration, improving bioavailability, and reducing toxicity [9-13]. Waiving the condition for hepatic first-pass metabolism, mucoadhesive films would be ideal for localized delivery in oral cancer through buccal administration, as the buccal mucosa provides a vascularized, non-keratinized surface for rapid absorption. These films, using hybrid NLC-based buccal can integrate the benefits of nanotechnology and mucoadhesion to offer sustained release and prolonged contact at the site of action, which is important for chemoprevention in high-risk oral mucosa [14-17].
This study utilized a Quality by Design (QbD) approach, incorporating Box-Behnken design (BBD), to systematically optimize quercetin-loaded NLCs integrated into hybrid buccal films. The Box–Behnken design allowed quantitative mapping of formulation factors to CQAs (particle size and EE), while film construction aimed to maximize mucoadhesion and mechanical robustness for clinical handling [18]. The goal was to enhance quercetin's solubility, stability, and localized bioavailability for effective oral cancer chemoprevention, addressing gaps in current delivery strategies.
MATERIALS AND METHODS
Quercetin was procured from Sigma-Aldrich (India). Glyceryl monostearate (GMS), capmul MCM, transcutol HP, poloxamer 188, soya lecithin, ethyl cellulose, polyethylene glycol (PEG) 400, hydroxypropyl methylcellulose (HPMC) K15, and carbopol 934P were obtained from commercial suppliers (e. g., Gattefossé, Abitec Corporation). All other reagents and chemicals were of analytical grade and procured from SD finechem, Mumbai.
Preparation of NLCs
Preliminary trials screened excipients based on solubility studies: GMS showed high solubility for quercetin (>50 mg/g), Capmul MCM and Transcutol HP enhanced miscibility. Process parameters were optimized to minimize aggregation. NLCs were produced by a solvent‑injection method. The lipid phase was prepared by dissolving quercetin (1% w/v) and soya lecithin (1% w/v) in a mixture of solid lipid (GMS) and liquid lipids (Capmul MCM and Transcutol HP) at varying ratios, heated to 70 °C to ensure complete melting and dissolution. Ethanol (5 ml) was added as a co-solvent to the lipid phase to enhance solubility. The aqueous phase consisted of poloxamer 188 (surfactant, 1-3% w/v) dissolved in distilled water (q. s. to 100 ml), heated to the same temperature. The lipid phase was injected dropwise (1 ml/min) into the aqueous phase under magnetic stirring at 2500 rpm for 30 min, followed by ultrasonication (Probe sonicator, 20 kHz) for 4 min at 60% amplitude to reduce particle size. The dispersion was cooled to room temperature and stored at 4 °C for further use [19].
Experimental design (DoE)
A three-factor, three-level BBD was employed using Design-Expert software (Stat-Ease, USA). Independent variables: total lipid content (X1: 5-15%), surfactant concentration (X2: 1-3%), liquid lipid content (X3: 20-40%). Responses: particle size (Y1, minimize) and entrapment efficiency (Y2, maximize). Seventeen runs generated by the software (Design-Expert® version 13, Statease Inc., USA) were conducted (table 1,2). Quadratic models were fitted, and ANOVA assessed significance (p<0.05). Representative coded equations were used to guide optimization and were validated experimentally. Graphical optimization identified the design space for desired attributes (PS<200 nm, EE>80%) [20].
Film fabrication
Bilayer films were solvent cast wherein EC backing layer provided unidirectional release. Ethyl cellulose and PEG 400 were dissolved in dichloromethane under stirring for 2 h. The solution was cast onto a glass mold (10 cm²) and dried at 40 °C for 12 h. The mucoadhesive layer contained HPMC (2–3% w/v) and Carbopol 934P (0.25–0.5% w/v) with plasticizer (PEG‑400/glycerol) and incorporated the optimized NLC dispersion. HPMC K15 and Carbopol 934P were dispersed in NLC dispersion (equivalent to 1 mg quercetin/cm²) with glycerol and stirred overnight for hydration. Optimized quercetin-NLC dispersion was incorporated and homogenized. The mixture was cast over the backing layer and dried at 40 °C for 24 h. Films were peeled, cut into 2 cm² patches, and stored in desiccators. Films were characterized for thickness, mechanical properties, surface pH, drug content, folding endurance, and in‑vitro release and ex‑vivo permeation (porcine buccal mucosa) and cytotoxicity assessments [20, 21].
Characterization of NLC
Particle size, zeta potential, and polydispersity index (PDI)
Measured using dynamic light scattering (Malvern Zetasizer, Nano ZS) at 25 °C, diluting samples 1:10 with water [19]. The estimations were done at 25±1ºC, repeating thrice for each sample and considering the average value.
Entrapment efficiency (EE) and drug loading (DL)
EE was determined by ultracentrifugation (15,000 rpm, 30 min, 4 °C) using a cooling centrifuge (Remi India). Supernatant was removed and diluted suitably with methanol. Free drug in supernatant was quantified by UV spectrophotometry at 370 nm [20].
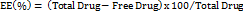
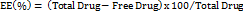 …. (1)
…. (1)
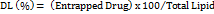
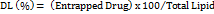 …. (2)
…. (2)
High-resolution transmission electron microscopy (HR-TEM)
Samples were negatively stained with 2% phosphotungstic acid and imaged. The instrument (JEOL JEM-2100, Japan) was operated at 120KV [19, 20].
Characterization of hybrid film
Thickness
The thickness of the film was assessed using a micrometer at four to six-different places across the surface of the film and the average thickness was recorded [22].
Tensile strength
A texture Analyzer (TexturePro CT, Brookefield Engg. Co.) equipped with a cell load of 10 kg was employed to measure the tensile strength of the film. A 2 × 4 cm2 film was anchored between the two clamps and pulled at a rate of 1 mm/s. The recording of the tensile strength was done at the point of film breakage [14]. The below equation was used to calculate the tensile strength [22]:
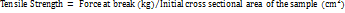 … (3)
… (3)
Surface pH
2% w/v agar in isotonic phosphate buffer pH 6.8 was dissolved and agar plate was gelled at room temperature. Buccal films were placed on the surface of the agar plate for 2 h to allow swelling. Surface pH was measured by placing a glass electrode (Electrolab, India) or pH indicator paper directly on the surface of the swollen film [21].
Folding endurance
The number of folds required to break the film or to develop visible cracks is considered as the folding endurance, which is indicative of brittleness of the film. A 2×2 cm2 film was taken and subjected to repetitively folding the film at the same place till visible cracks appeared [22].
Drug content
A 2 x 1 cm2 film was cut and dissolved in methanol followed by sonication for 3 min the methanolic solution was filtered through 0.22-micron filter paper and analyzed by UV (ultraviolet) spectroscopic method [22].
Moisture uptake
Films were placed in a desiccator maintained at ambient conditions for 24 h. The desiccatorwas loaded with saturated potassium chloride solution to maintain relative humidity of 84%. The films were taken out post 24h and weighed. Percentage of moisture uptake was calculated by the following formula [23].
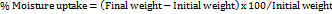 … (4)
… (4)
Swelling index
Buccal film was weighed (W1) and placed in the petri plate containing phosphate buffer pH 6.8. Buccal films were removed after 4h and placed on filter paper to drain out excess surface water and weighed again (W2). Swelling index (SI) was calculated using the formula [24, 25]:
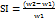 ……. (5)
……. (5)
Where; SI = Swelling index, W2 = Final weight, W1 = Initial weight
In vitro drug release
A preactivated dialysis membrane (Mol. wt. 12000–14000) was placed between the donor and receptor compartments of a Franz diffusion cell. The buccal film (1 cm x 1 cm) along with 2 ml of release medium (PBS with 20% ethanol) was placed in the donor compartment. The assembly was jacketed and thermostatically maintained at 37 °C by circulating water bath. Atspecific time intervals till 8 h, 2 ml of the sample was withdrawn from the receptor chamber and replenished with fresh buffer. The withdrawn aliquot was diluted appropriately and the drug release was spectroscopically analyzed [19].
Ex-vivo buccal permeation
Porcine buccal mucosa mounted on Franz diffusion cells (area 2.5 cm², receptor: PBS with 20% ethanol). Film was applied on the buccal mucosa (1 cm x 1 cm), drug permeation was monitored for 8 h. The porcine separated buccal epithelium was mounted between the two chambers and the entire assembly was placed on a magnetic stirrer by putting a magnetic bead coated with Teflon. The diffusion cell was maintained at 37 °C using circulating water jacket. 2 ml of the sample was withdrawn at regular intervals and replenished with receptor medium. The sample withdrawn was for analyzing it spectrophotometrically [26].
Cytotoxicity assay
The cytotoxicity of the nanostructured lipid carrier (NLC)-based buccal film was assessed using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay on the KB human oral epidermoid carcinoma cell line [27, 28]. KB cells were maintained in Dulbecco’s modified Eagle medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin at 37 °C in a humidified incubator with 5% CO₂. Cells were seeded at a density of 1 × 10⁴ cells per well in 96-well plates and allowed to adhere for 24 h prior to treatment. The NLC dispersion was serially diluted in medium to achieve concentrations ranging from 0.1 to 100 µg/ml based on QCT content. Cells were exposed to these dilutions, along with blank NLC and untreated controls, for 24 h. Subsequently, the medium was replaced with fresh medium containing 0.5 mg/ml MTT, and the plates were incubated for 4 h at 37 °C to allow formazan crystal formation. The supernatant was discarded, and 100-200 µl** of dimethyl sulfoxide (DMSO) was added to each well to solubilize the crystals, with gentle shaking for 10-30 min. Absorbance was measured at 570 nm using a microplate reader, and cell viability was calculated as a percentage relative to untreated controls using the formula: (absorbance of treated cells/absorbance of control cells) × 100 [27].
Stability study
The stability of lyophilized nanoparticles and the hybrid buccal film was conducted for 30 days at 4ºC and 25 °C±2 °C and 60%±5% RH in stability chambers. Stability studies are undertaken for analyzing any changes during the storage of formulation. The nanoparticles were analyzed for particle sizeand entrapment efficiency. The buccal films were assessed for changes in folding endurance, drug content, and in vitro drug release [28].
RESULTS AND DISCUSSION
The preliminary studies helped to identify the suitable solid and liquid lipids for the preparation of NLCs. The process variables like rpm of stirring, time for sonication, rate of addition of lipids were optimized initially and kept constant during the subsequent experiments. The optimized process parameters were stirring at 2500 rpm, 4 min sonication, rate of addition of lipid-1 ml/min, respectively. Preliminary trials identified lipid content, surfactant concentration, and liquid lipid content as key parameters for NLC optimization. The identified parameters are similar to the previously reported studies [19, 29].
A QbD guided optimization process was undertaken to formulate the most appropriate NLC formulation of QCT. A Box Behnken design was used to optimize the formulation, wherein the identified critical material attributes (CMA) or the independent variables were total lipid content (X1), surfactant concentration (X2), liquid lipid content (X3). The critical quality attributes (CQA) which were influenced by them were particle size (Y1) and entrapment efficiency (Y2). The formulation batches were prepared according to the composition outlined in the design matrix and were characterized for the response variables.
Table 1: The variables in the box-behnken design
| Independent variables | Variable levels | ||
| Low level (-1) | Central level (0) | High level (+1) | |
| % Total lipid (X1) | 5 | 10 | 15 |
| % Surfactant (X2) | 1 | 2 | 3 |
| % Liquid lipid (X3) | 20 | 30 | 40 |
Table 2: Design matrix of box-behnken design and the observed responses
| Batch No. | % Total lipid (X1) | % Surfactant (X2) | % Liquid lipid (X3) | Y1: PS (nm) | Y2: EE (%) |
| F1 | 5 | 1 | 30 | 147.8 | 74 |
| F2 | 15 | 1 | 30 | 128.4 | 89 |
| F3 | 5 | 3 | 30 | 1055.1 | 70 |
| F4 | 15 | 3 | 30 | 384.5 | 86 |
| F5 | 5 | 2 | 20 | 125.3 | 63 |
| F6 | 15 | 2 | 20 | 130.2 | 69 |
| F7 | 5 | 2 | 40 | 981.3 | 61 |
| F8 | 15 | 2 | 40 | 532.5 | 66 |
| F9 | 10 | 1 | 20 | 483.8 | 63 |
| F10 | 10 | 3 | 20 | 140.7 | 80 |
| F11 | 10 | 1 | 40 | 75.5 | 71 |
| F12 | 10 | 3 | 40 | 1132.3 | 65 |
| F13-F17 | 10 | 2 | 30 | 386.8± 28.9* | 88± 4.8* |
*The (Mean±SD) of the 5 replicate batches. PS: 386.8±28.9, EE: 88±4.8, PS-Particle size, EE-Entrapment efficiency
Response surface methodology was used to optimize the CMAs and achieve desirable CQAs. The polynomial equations generated by the software modelled the experiment. It can be observed that increasing total lipid and liquid lipid contents increased the particle size, while increasing surfactant concentrations helped to achieve a lower particle size. The increased surfactants stabilized the emulsion and reduced interfacial tensions to reduce the particle size. However, a few outliers were observed during the study, showing widely varying particle sizes in some of the batches (F3, F7, F12). This can be plausibly due to the inherent instability of nanoemulsions at certain lipid/surfactant ratios, leading to coalescence or incomplete solidification. These outliers were not replicated and the future work can encompass replicating these batches to confirm the status of outliers.
In case of entrapment efficiency, similar pattern was observed wherein as the lipid content was raised the entrapment efficiency increased and increasing surfactants reduced the drug entrapment. The higher lipids in the system enhanced the drug dissolution into the lipid matrix to raise the entrapment. The system was optimized with a perspective to minimize the particle size keeping 200 nm as upper limit and maximize the entrapment efficiency with a lower limit of 80% [19, 20, 30, 31]. Several previous studies have utilized the QbD-based approaches for the optimization of the nano formulations and achieved low particle size and high entrapment efficiency and the surfactant and lipid contents have been key determiners for the observed results. Thus, QbD-driven optimization yields robust NLC formulations with consistent size and drug loading for further applications [30-35].
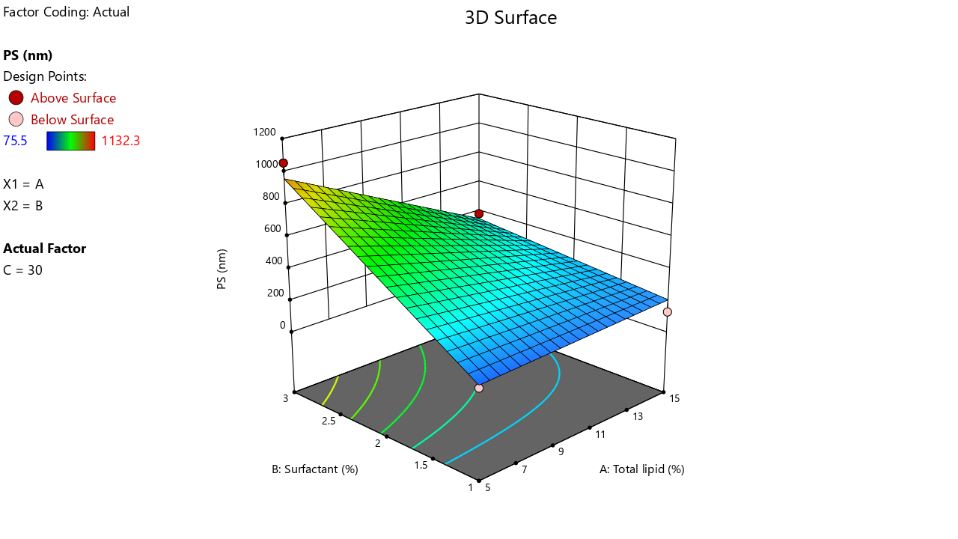
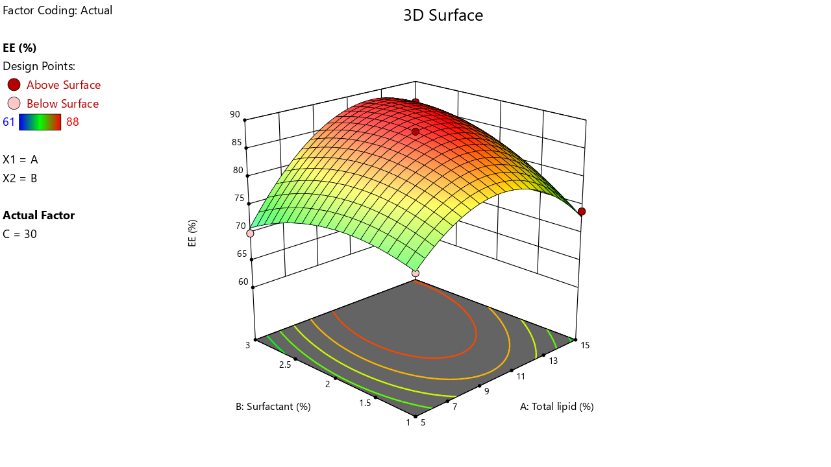
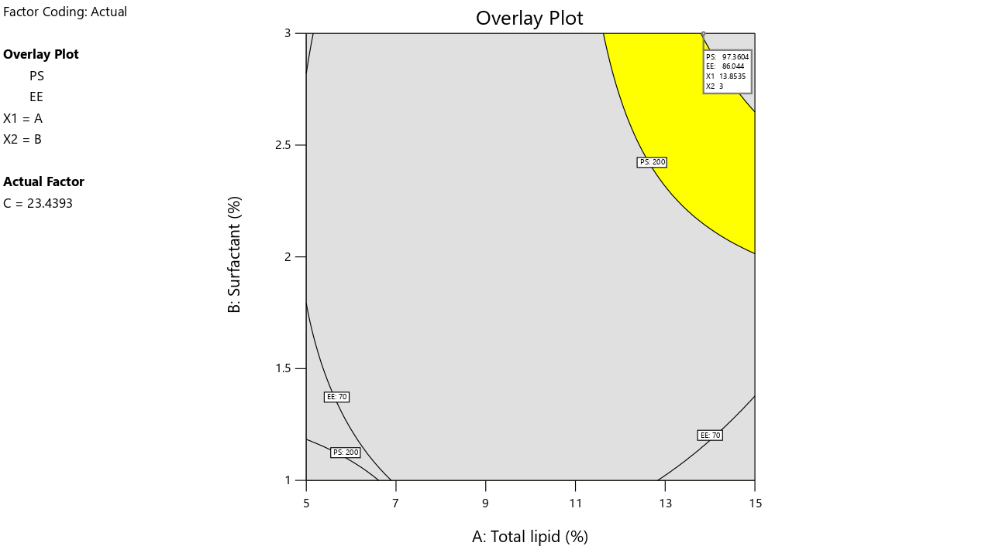
Fig. 1: a) 3D response surface graph for particle size b) 3D response surface graph for entrapment efficiency c) overlay plot for graphical optimization
PS =+200.20+59.88 X1-38.38 X2+125.50 X3-30.50 X1X2+33.75 X1 X3+23.40 X12+71.65 X32………(6)
EE =+85.20+4.50X1-4.00X2+9.00X3-5.60X22-5.10X32……… (7)
The polynomial equations for the PS and EE model the CMAs for the respective CQA. NLC formulation was optimized using numerical and graphical optimization technique using the constraints as discussed above. The software-generated solutions were screened for maximal desirability. The graphical optimization identified the yellow region in the overlay plot, which was the design space. Despite the outliers in the study, the model could identify a robust design space which consistently gave the desirable outcomes. This can be evidenced from the optimized batch with a low particle size below 150 nm. The solution with maximum global desirability was selected as the optimized formulation (table 3).
Table 3: Composition of optimized formulation
| Ingredient | Quantity (%) |
| Quercetin NLC | |
| Quercetin | 1 |
| Glyceryl Monostearate | 9.21 |
| Capmul MCM | 2.1703 |
| Transcutol HP | 2.1703 |
| Soya lecithin | 1 |
| Poloxamer 188 | 1 |
| Ethanol | 5 |
| Water (q. s.) | 100 |
| Hybrid film | |
| Ethyl cellulose (backing layer) | 5 |
| PEG 400 | 1.5 |
| Hydroxy Propyl Methyl Cellulose K15 | 1 |
| Carbopol 934P | 0.5 |
The desirability optimized formulation (GMS 9.21%, Capmul MCM 2.17%, Transcutol 2.17%) was prepared following the procedure outlined previously. The optimized formulation of NLC provided PS of 142.3 nm and EE of 90.21%. These results are in agreement with another study where NLCs of quercetin, along with another phytconstitutent showed low particle size and high entrapment efficiency [33]. HR‑TEM conducted on the optimized formulation showed spherical NLCs and in close agreement of the particle size obtained by DLS. The optimized batch showed a negative zeta potential of-13.3±2.1 mV and showed drug loading of approximately 1 % (0.9±0.04). Thus, it can be said that the chosen excipients at the levels used could successfully prepare a low particle size stable NLC dispersion with good entrapment efficiency. QbD facilitated efficient optimization and defined a design space for robust manufacture [32, 36].
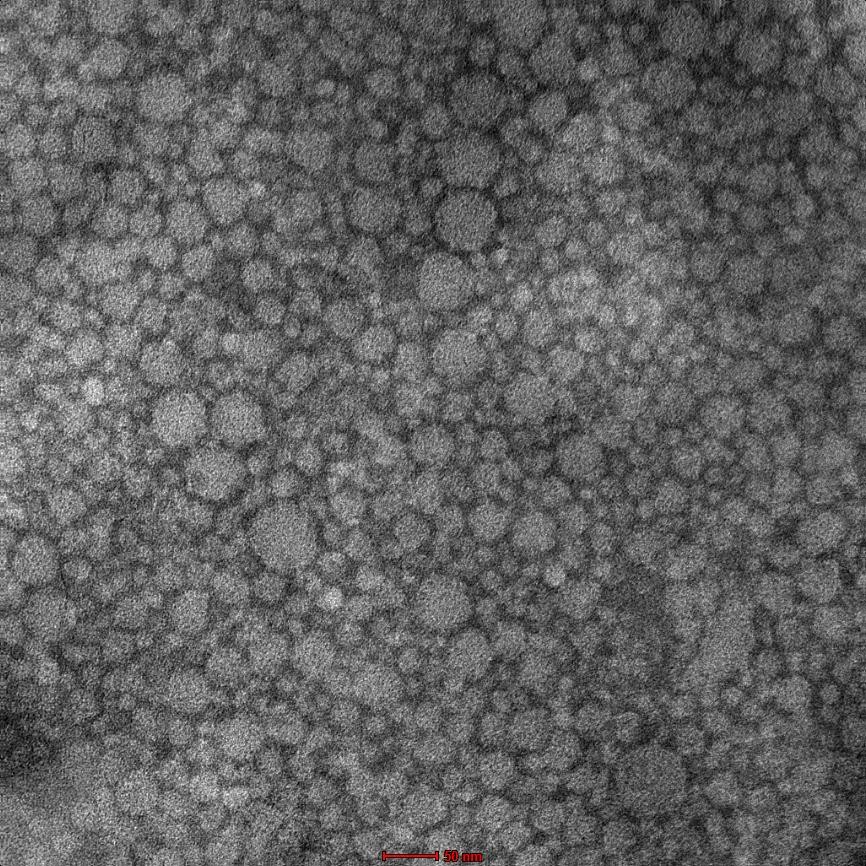
Fig. 2: HR-TEM of optimized NLC formulation (Magnification 150,000X)
Table 4: Summary of characterization parameters of NLC and hybrid film
| Characterization parameter (for NLC and hybrid film) | Observation* |
| Particle Size (nm) | 144±8.3 |
| Entrapment Efficiency (%) | 90.21±4.02 |
| Zeta Potential (mV) | -13.3±2.1 |
| Drug Loading (%) | 0.9±0.04 |
| Thickness (mm) | 0.97±0.02 |
| Tensile Strength (MPa) | 3.93±1.05 |
| Surface pH | 6.8±0.5 |
| Folding Endurance | 121±4 |
| Elongation (%) | 24.47±0.4 |
| Drug Content (%) | 95.49±1.9 |
| Moisture Uptake (%) | 16.48±0.158 |
| Swelling Index (%) | 35.9±0.41 |
| In vitro drug release at 8 h (%) | 80.78±4.8 |
Data are expressed mean±SD, n=3
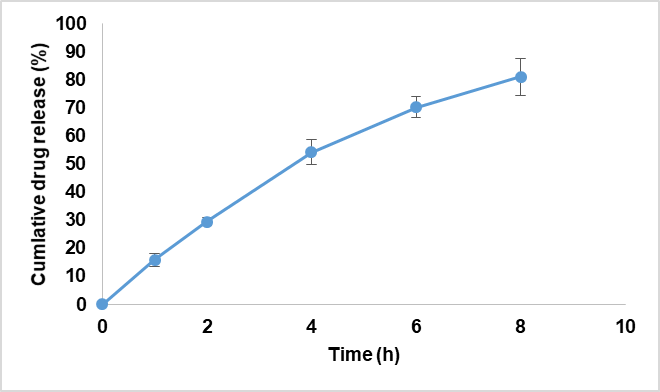
Fig. 3: In vitro drug release study (n=3)
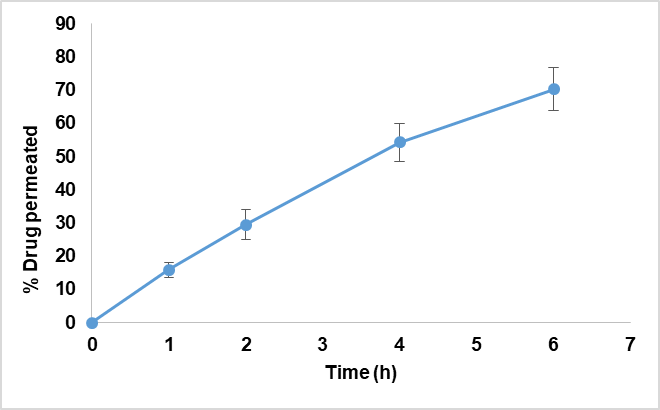
Fig. 4: Buccal permeability of QCT NLC-based hybrid film (n=3)
The bilayered hybrid films were casted using solvent cast method. The prepared films were thin with less than 1 mm thickness. Films displayed tensile strength 3.93 MPa and elongation 24.47%, with folding endurance of 121 cycles. The mechanical properties indicated that the prepared film has sufficient mechanical strength and flexibility to perform effectively in vivo. Surface pH was near neutral, thus chances of mucosal irritation would be minimal. The film showed moderate moisture uptake of 16.48±0.158 % and swelling index of 35.9±0.41 %. The film has a moderate ability to absorb moisture from the environment. This suggests that the film is not overly hygroscopic, which is beneficial for maintaining its stability during storage and reducing the risk of microbial contamination or degradation. The moderate swelling index is advantageous for mucoadhesive drug delivery, as swelling helps the film to adhere better to the mucosal surface and potentially enhances drug release and retention time. In‑vitro release reached ~80% by 8 h (fig. 3). This demonstrates that the drug is steadily released in 8h, facilitating prolonged and sustained delivery. Ex‑vivo porcine buccal permeation was 72.8% of the drug loading at 6 h (fig. 4), which includes the drug released from the dosage form, permeated across mucosa. The data is indicative of good permeability of the drug from the lipidic nanoformulation. The observed data indicated the high buccal permeation and controlled release of the drug from NLC hybrid films in congruence with other reported studies. The NLCs provide sustenance of release as well as facilitating higher permeability across mucosa by virtue of lipophilicity of the formulation. The combination of NLCs and bilayer film offers localized, sustained delivery with minimal systemic exposure, appropriate for chemopreventive applications [37, 38].
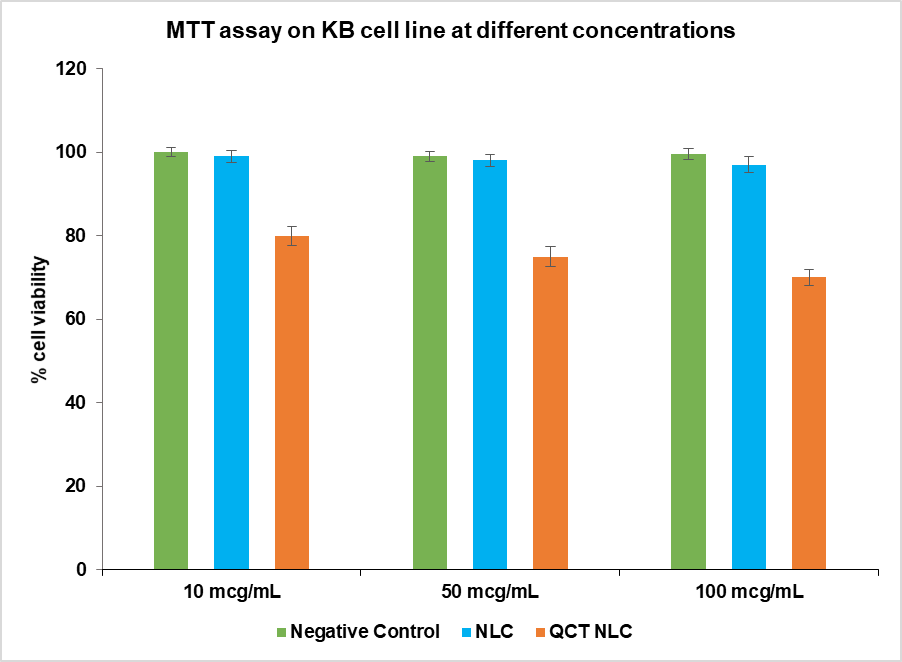
Fig. 5: Cell viability assay of optimized QCT NLCs. Data are expressed mean±SD, n=3
Cytotoxicity studies (fig. 5) confirmed QCT’s ROS-mediated apoptosis in OSCC cells, consistent with recent systematic reviews showing reduced proliferation and metastasis [1, 4, 39, 40]. It can be observed that the blank formulation shows minimal toxicity, while the drug-loaded formulation exhibits reduced cancer cell viability with increasing concentration. IC50 values for QCT-NLC was observed to be 280 µg/ml. Similar observations were seen in other studies where drug encapsulated in NLCs showed higher cytotoxicity and hence the anticancer potential of QCT [33-35]. Future studies can include a comparative assessment of the developed formulation with free QCT also. Overall, this system advances localized chemoprevention, potentially reducing oral cancer incidence in high-risk populations. The small-sized buccal film can be placed in the cheek pouch and provide localized risk prevention to oral cancer.
The formulation stability was investigated for a period of 30 days for NLC as well as the buccal film. It was observed in the short-term studies that the NLC as well as the film could retain their key performance parameters in limits and did not vary significantly from the initial values. The positive outcomes from the short term studies prompt long-term real-time studies in future.
Table 5: Short-term stability studies data
| Formulation/Characterization | 4 ºC | 25 °C±2 °C and 60%±5% RH |
| QCT-NLC | At 0 Day | At 30 Days |
| PS (nm) | 144±8.3 | 175±12.3 |
| EE (%) | 90.21±4.02 | 88.6±3.2 |
| QCT-NLC-Film | At 0 Day | At 30 Days |
| Drug content (%) | 95.49±1.904 | 94.1±3.4 |
| Folding endurance | 121±4 | 114±6 |
| In vitro drug release at 8 h (%) | 80.78±4.8 | 78.3±3.4 |
Data are expressed Mean±SD, n=3, PS-Particle size, EE-Entrapment efficiency
CONCLUSION
The studies demonstrated that a QbD-guided nanostructured lipid carrier (NLC) integrated into a bilayer mucoadhesive buccal film can overcome key development barriers of quercetin and enable localized, sustained chemoprevention at the oral mucosa. The optimized system achieved nanoscale size with high entrapment, mechanical robustness suitable for handling, near-neutral surface pH, sustained in vitro release to ~8 h, high ex-vivo buccal permeation within 6–8 h, and a favorable cytotoxicity profile, collectively supporting translational potential for high-risk populations. Future work should validate mucosal residence and tissue deposition, delineate pharmacokinetics and safety under repeat dosing, and confirm long-term chemopreventive efficacy in relevant carcinogen-induced OSCC models, alongside scale-up and stability studies under ICH conditions. Overall, the NLC-buccal hybrid platform offers a pragmatic pathway to clinically meaningful chemoprevention with a phytoconstituent that has otherwise been limited by solubility and bioavailability constraints.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
HP-Investigation, Methodology, formal analysis, ML-Conceptualization, writing – review and editing
CONFLICT OF INTERESTS
The author declares no conflicts of interest
REFERENCES
Shahbaz M, Naeem H, Momal U, Imran M, Alsagaby SA, Al Abdulmonem W. Anticancer and apoptosis-inducing potential of quercetin against a wide range of human malignancies. Int J Food Prop. 2023;26(1):2590-626. doi: 10.1080/10942912.2023.2252619.
Chen XJ, Zhang XQ, Liu Q, Zhang J, Zhou G. Nanotechnology: a promising method for oral cancer detection and diagnosis. J Nanobiotechnology. 2018 Jun 11;16(1):52. doi: 10.1186/s12951-018-0378-6, PMID 29890977.
Tomou EM, Papakyriakopoulou P, Saitani EM, Valsami G, Pippa N, Skaltsa H. Recent advances in nanoformulations for quercetin delivery. Pharmaceutics. 2023 Jun 5;15(6):1656. doi: 10.3390/pharmaceutics15061656, PMID 37376104.
Li X, Guo S, Xiong XK, Peng BY, Huang JM, Chen MF. Combination of quercetin and cisplatin enhances apoptosis in OSCC cells by downregulating xIAP through the NF-κB pathway. J Cancer. 2019 Jul 25;10(19):4509-21. doi: 10.7150/jca.31045, PMID 31528215.
Wang M, Chen X, Yu F, Zhang L, Zhang Y, Chang W. The targeting of noncoding RNAs by quercetin in cancer prevention and therapy. Oxid Med Cell Longev. 2022 May 24;2022:4330681. doi: 10.1155/2022/4330681, PMID 35656022.
Tomou EM, Papakyriakopoulou P, Saitani EM, Valsami G, Pippa N, Skaltsa H. Recent advances in nanoformulations for quercetin delivery. Pharmaceutics. 2023;15(6):1656. doi: 10.3390/pharmaceutics15061656, PMID 37376104.
Sakagami H, Kobayashi M, Chien CH, Kanegae H, Kawase M. Selective toxicity and type of cell death induced by various natural and synthetic compounds in oral squamous cell carcinoma. In vivo. 2007 Mar-Apr;21(2):311-20. PMID 17436582.
Son HK, Kim D. Quercetin induces cell cycle arrest and apoptosis in YD10B and YD38 oral squamous cell carcinoma cells. Asian Pac J Cancer Prev. 2023 Jan 1;24(1):283-9. doi: 10.31557/APJCP.2023.24.1.283, PMID 36708578.
Poonia N, Kharb R, Lather V, Pandita D. Nanostructured lipid carriers: versatile oral delivery vehicle. Future Sci OA. 2016 Jul 15;2(3):FSO135. doi: 10.4155/fsoa-2016-0030, PMID 28031979.
Beloqui A, Solinis MA, Rodriguez Gascon A, Almeida AJ, Preat V. Nanostructured lipid carriers: promising drug delivery systems for future clinics. Nanomedicine. 2016 Jan;12(1):143-61. doi: 10.1016/j.nano.2015.09.004, PMID 26410277.
Salvi VR, Pawar P. Nanostructured lipid carriers (NLC) system: a novel drug targeting carrier. J Drug Deliv Sci Technol. 2019;51:255-67. doi: 10.1016/j.jddst.2019.02.017.
Muller RH, Radtke M, Wissing SA. Solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) in cosmetic and dermatological preparations. Adv Drug Deliv Rev. 2002 Nov 1;54 (Suppl 1):S131-55. doi: 10.1016/s0169-409x(02)00118-7, PMID 12460720.
Pardeike J, Hommoss A, Muller RH. Lipid nanoparticles (SLN, NLC) in cosmetic and pharmaceutical dermal products. Int J Pharm. 2009 Jan 21;366(1-2):170-84. doi: 10.1016/j.ijpharm.2008.10.003, PMID 18992314.
Shojaei AH. Buccal mucosa as a route for systemic drug delivery: a review. J Pharm Pharm Sci. 1998 Jan-Apr;1(1):15-30. PMID 10942969.
Nafee NA, Ismail FA, Boraie NA, Mortada LM. Mucoadhesive buccal patches of miconazole nitrate: in vitro/in vivo performance and effect of ageing. Int J Pharm. 2003 Oct 2;264(1-2):1-14. doi: 10.1016/s0378-5173(03)00371-5, PMID 12972331.
Gilhotra RM, Ikram M, Srivastava S, Gilhotra N. A clinical perspective on mucoadhesive buccal drug delivery systems. J Biomed Res. 2014 Mar;28(2):81-97. doi: 10.7555/JBR.27.20120136, PMID 24683406.
Perioli L, Ambrogi V, Rubini D, Giovagnoli S, Ricci M, Blasi P. Novel mucoadhesive buccal formulation containing metronidazole for the treatment of periodontal disease. J Control Release. 2004 Mar 24;95(3):521-33. doi: 10.1016/j.jconrel.2003.12.018, PMID 15023463.
Tavares Luiz M, Santos Rosa Viegas J, Palma Abriata J, Viegas F, Testa Moura De Carvalho Vicentini F, Lopes Badra Bentley MV. Design of experiments (DoE) to develop and to optimize nanoparticles as drug delivery systems. Eur J Pharm Biopharm. 2021;165:127-48. doi: 10.1016/j.ejpb.2021.05.011, PMID 33992754.
Lalan M, Shah P, Shah K, Prasad A. Developmental studies of curcumin NLCs as safe alternative in management of infectious childhood dermatitis. NANOASIA. 2020 Aug 1;10(4):390-403. doi: 10.2174/2210681209666181226153741.
Kharwade RS, Mahajan NM. Formulation and evaluation of nanostructured lipid carriers based anti-inflammatory gel for topical drug delivery system. Asian J Pharm Clin Res. 2019;12(4):286-91. doi: 10.22159/ajpcr.2019.v12i4.32000.
Semalty M, Semalty A, Kumar G. Formulation and characterization of mucoadhesive buccal films of glipizide. Indian J Pharm Sci. 2008 Jan;70(1):43-8. doi: 10.4103/0250-474X.40330, PMID 20390079.
Patel VN, Patel HV, Agrawal K, Soni I, Shah P, Mangrulkar SV. Comprehensive developmental investigation on simvastatin enriched bioactive film forming spray using the quality by design paradigm: a prospective strategy for improved wound healing. J Drug Target. 2024;32(9):1139-53. doi: 10.1080/1061186X.2024.2382405, PMID 39042496.
Ammanage A, Rodriques P, Kempwade A, Hiremath R. Formulation and evaluation of buccal films of piroxicam co-crystals. Futur J Pharm Sci. 2020;6(1):16. doi: 10.1186/s43094-020-00033-1.
Meher JG, Tarai M, Patnaik A, Mishra P, Yadav NP. Cellulose buccoadhesive film bearing glimepiride: physicomechanical characterization and biophysics of buccoadhesion. AAPS PharmSciTech. 2016 Aug;17(4):940-50. doi: 10.1208/s12249-015-0419-5, PMID 26831445.
Mukherjee D, Bharath S. Design and characterization of double layered mucoadhesive system containing bisphosphonate derivative. ISRN Pharm. 2013;2013:604690. doi: 10.1155/2013/604690, PMID 24455313.
Caon T, Simoes CM. Effect of freezing and type of mucosa on ex vivo drug permeability parameters. AAPS PharmSciTech. 2011 Jun;12(2):587-92. doi: 10.1208/s12249-011-9621-2, PMID 21541829.
Suhasini SJ, Roy A, Sosa G, Lakshmi T. The cytotoxic effect of Caralluma fimbriata on Kb cell lines. Res J Pharm Technol. 2019;12(10):4995-8. doi: 10.5958/0974-360X.2019.00865.5.
Pinheiro M, Ribeiro R, Vieira A, Andrade F, Reis S. Design of a nanostructured lipid carrier intended to improve the treatment of tuberculosis. Drug Des Devel Ther. 2016 Aug 2;10:2467-75. doi: 10.2147/DDDT.S104395, PMID 27536067.
Jaiswal P, Gidwani B, Vyas A. Nanostructured lipid carriers and their current application in targeted drug delivery. Artif Cells Nanomed Biotechnol. 2016;44(1):27-40. doi: 10.3109/21691401.2014.909822, PMID 24813223.
Alam T. Quality by design based development of nanostructured lipid carrier: a risk based approach. Exploration of Medicine. 2022;3:617-38. doi: 10.37349/emed.2022.00118.
Vakilinezhad MA, Tanha S, Montaseri H, Dinarvand R, Azadi A, Akbari Javar H. Application of response surface method for preparation optimization and characterization of nicotinamide loaded solid lipid nanoparticles. Adv Pharm Bull. 2018 Jun;8(2):245-56. doi: 10.15171/apb.2018.029, PMID 30023326.
Bajwa N, Mahal S, Naryal S, Singh PA, Baldi A. Development of novel solid nanostructured lipid carriers for bioavailability enhancement using a quality by design approach. AAPS PharmSciTech. 2022;23(7):253. doi: 10.1208/s12249-022-02386-7, PMID 36109467.
Chaudhari VS, Gawali B, Saha P, Naidu VG, Murty US, Banerjee S. Quercetin and piperine enriched nanostructured lipid carriers (NLCs) to improve apoptosis in oral squamous cellular carcinoma (FaDu cells) with improved biodistribution profile. Eur J Pharmacol. 2021 Oct 15;909:174400. doi: 10.1016/j.ejphar.2021.174400, PMID 34332920.
Imran M, Iqubal MK, Imtiyaz K, Saleem S, Mittal S, Rizvi MM. Topical nanostructured lipid carrier gel of quercetin and resveratrol: formulation optimization in vitro and ex vivo study for the treatment of skin cancer. Int J Pharm. 2020 Sep 25;587:119705. doi: 10.1016/j.ijpharm.2020.119705, PMID 32738456.
AlMulhim FM, Nair AB, Aldhubiab B, Shah H, Shah J, Mewada V. Design development evaluation and in vivo performance of buccal films embedded with paliperidone-loaded nanostructured lipid carriers. Pharmaceutics. 2023;15(11):2530. doi: 10.3390/pharmaceutics15112530, PMID 38004510.
Muller RH, Mader K, Gohla S. Solid lipid nanoparticles (SLN) for controlled drug delivery a review of the state of the art. Eur J Pharm Biopharm. 2000 Jul;50(1):161-77. doi: 10.1016/s0939-6411(00)00087-4, PMID 10840199.
Sodata P, Duangjit S, Sarisuta N, Kraisit P. Optimization of mucoadhesive film reinforced with functionalized nanostructured lipid carriers (NLCs) for enhanced triamcinolone acetonide delivery via buccal administration: a box-behnken design approach. Sci. 2025;7(1):22. doi: 10.3390/sci7010022.
Koland M, Charyulu RN, Vijayanarayana K, Prabhu P. In vitro and in vivo evaluation of chitosan buccal films of ondansetron hydrochloride. Int J Pharm Investig. 2011 Jul;1(3):164-71. doi: 10.4103/2230-973X.85967, PMID 23071939.
Lee YK, Hwang JT, Kwon DY, Surh YJ, Park OJ. Induction of apoptosis by quercetin is mediated through AMPKalpha1/ASK1/p38 pathway. Cancer Lett. 2010 Jun 28;292(2):228-36. doi: 10.1016/j.canlet.2009.12.005, PMID 20083342.
Farah A, Omran R. Cytotoxic activity of alkaloid extracts of different plants against breast cancer cell line. Asian J Pharm Clin Res. 2017;10(7):181-9. doi: 10.22159/ajpcr.2017.v10i7.18189.