
Int J App Pharm, Vol 18, Issue 1, 2026, 84-94Reviewl Article
DENDRIMERS IN OCULAR DRUG DELIVERY: A COMPREHENSIVE REVIEW
SHALU VERMA1*, VIVEK SILORI1, PRASHANT KUMAR1, MUNEESH KANAUJAYA1
1Department of Pharmaceutics, Uttaranchal Institute of Pharmaceutical Sciences, Uttaranchal University, Premnagar, Dehradun-248007, India
*Corresponding author: Shalu Verma; *Email: vermashalu339@gmail.com
Received: 10 Sep 2025, Revised and Accepted: 20 Nov 2025
ABSTRACT
The anatomical and physiological complexity of the eye significantly hinders drug delivery to both the anterior and posterior segments, complicating the treatment of various ocular diseases. Although conventional ophthalmic formulations, primarily topical eye drops, are widely used, their bioavailability remains low. Recent advancements in nanotechnology have introduced dendrimers as promising nanoscale drug carriers for ocular therapy. Dendrimers can enhance drug solubility, permeability, stability, and retention owing to their unique branched architecture, multivalent surface functionality, and capacity for precise drug targeting. This review discusses the anatomical barriers to ocular drug delivery, innovative strategies to overcome them, and highlights various dendrimer generations, types, synthesis methods, and their applications in treating eye disorders such as glaucoma, age-related macular degeneration, diabetic retinopathy, and uveitis. Furthermore, it explores clinical studies, patents and their ocular applications/rationale, and regulatory hurdles associated with the clinical translation of dendrimer-based therapeutics. Despite, promising preclinical data, manufacturing and regulatory challenges remain major hurdles for their widespread clinical use. Dendrimers hold great promise as advanced ocular drug delivery systems, with the potential to improve treatment efficacy for a wide range of vision-impairing disorders.
Keywords: Ocular drug delivery, Dendrimers, Nanocarriers, PAMAM dendrimers, Ocular barriers
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i1.56820 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Among the sensory organs, the eyes stand out for its intricate structure and distinct physiological mechanisms [1]. Structurally, the eye can be divided into two major regions: the anterior and posterior segments. The anterior portion, occupying roughly one-third of the eye, includes the cornea, lens, pupil, ciliary body and the tear film. The posterior segment, forming the remaining two-thirds, contains the sclera, choroid, retina, vitreous body, optic nerve and associated tissues [2]. Both the anterior and posterior regions of the eye are susceptible to a wide range of acute and chronic conditions. Posterior segment disorders, often grouped under posterior segment eye diseases (PSEDs), primarily involve the retina, optic nerve and choroid with common examples including glaucoma, age-related macular degeneration (AMD), diabetic retinopathy (DR), and retinitis. In contrast, the anterior segment diseases encompass conditions such as cataracts, dry-eye syndrome, conjunctivitis, keratoconus and various refractive errors [3]. A recent global survey by the World Health Organization (WHO) reported 196 million cases of age-related macular degeneration (AMD), 146 million cases of diabetic retinopathy (DR), 76 million cases of glaucoma, and 65 million cases of cataracts worldwide [4]. The major causes of these ocular diseases are eye trauma, infections, long-term eye fatigue and adverse reactions to high-dose medications [5]. The structural and physiological features of eyes act as a barrier to prevent ocular compartment from mechanical, chemical, metabolic, pathogen damage and systemic circulation [6]. Topical eye drops remain the most widely used dosage form in ophthalmology, representing more than 90% of marketed products due to their ease of administration. Despite this, their bioavailability is typically below 5% due to tear turnover, blinking, nasolacrimal drainage, and limited residence time, necessitating frequent dosing [7]. With advancements in nanotechnology, novel therapeutic interventions have been developed to treat ocular disorders. Compared to conventional methods, these nanocarriers offer various advantages, like overcoming ocular barriers, enhance trans corneal permeability, increase drug residence time, reduce drug degradation, reduce dosing frequency, achieve sustained/controlled release, drug targeting, gene targeting and improving patient compliance [8]. These advantages were observed when particles ranging from nanometers to microns were used [9]. These new methods include “dendrimers,” “nanoparticles,” “liposomes,” “nanosuspensions,” “implants,” “microneedles,” and “in situ gels” [10]. Despite the promising nature of these novel drug delivery systems (NDDS), most of them are in the early stages and are not widely used for ocular therapy [11]. Among these, dendrimers have emerged as the most important in recent times due to its structural advantage to treat various ophthalmic diseases [12]. This review focuses on recent advancements in dendrimer formulation for ophthalmic use, clinical studies and patents related to dendrimers and their ocular applications.
Search criteria
The literature for this review was retrieved from electronic databases including PubMed, Scopus and Web of Science. The search strategy employed combinations of keywords like ocular drug delivery, dendrimers, nanocarriers, PAMAM dendrimers, ocular barriers. Publications from 2000 to 2025 were considered. Only included studies which were original research, reviews and data relevant to dendrimers and ocular drug delivery. Publications not in English, conference abstracts without full-texts, and studies unrelated to the topic were excluded.
Ocular drug delivery barriers and novel solutions
These barriers and their respective novel solutions are illustrated in fig. 1.
Barriers to the ocular drug delivery can be broadly classified as static barriers, dynamic barriers, metabolic barriers and intraocular barriers with each acting as obstacle in ocular drug delivery as shown in fig. 1 [13]. Static barriers include stratified corneal epithelium, corneal stroma, sclera and conjunctiva. Dynamic barriers include tear turnover, reflex blinking and nasolacrimal drainage. Metabolic barriers include phase 1, phase 2 enzymes and efflux pumps. Intraocular barriers include blood-aqueous barrier (BAB) and the blood-retinal barrier (BRB).
Overcoming ocular barriers
Recent advancements in this field have led to novel solutions for the conventional problems like use of nanosized carriers due to their size [14], use of mucoadhesive polymers [15], use of enzyme inhibitors/efflux pump inhibitors [16], and using surface modifications with Polyethylene glycol (PEG) also known as PEGylation and with ligands for sustained drug release [17] and controlled drug release [18] to overcome the above problems respectively. Due to the restrictive nature of the eyes, conventional formulations often fail to deliver adequate amounts of drugs to the target site. Therefore, scientists have developed innovative approaches to overcome these barriers. Physical methods include iontophoresis, electroporation, sonophoresis can temporarily disrupt ocular barriers enhancing drug penetration without causing significant ocular tissue damage [19]. In a research study, it was observed that chitosan-coated nano systems enhance ocular drug penetration by improving interaction with the corneal epithelium and enabling transcellular transport without causing any ocular irritation [20]. Prodrugs utilizing transport targeting and nanomicelles encapsulating hydrophobic drugs offer strategies to improve drug solubility, permeability, retention and bioavailability [16]. Lipid-based nanoparticles such as solid-lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) also help bypass physiological limitations of the eye and achieve targeted, prolonged drug action [21]. Nanocarrier-based strategies like PHP-DPS@INS (A Liposome based nanocarrier composed of a PEG200-Hyd-PEI (PHP) polymer coating, a DSPE-PEG2k-SS31 (DPS) mitochondria-targeted antioxidant layer and encapsulated insulin (INS) for anti-inflammation) offer enhanced surface adhesion, combat oxidative stress and inflammation in dry eye disease [22]. L-valine-modified nanomicelles enhance ocular drug uptake by targeting peptide transporters and loosening tight junction barriers in the cornea which means temporarily reducing the integrity and connectivity of connected corneal epithelium cells that form a physical barrier and prevent the entry of foreign substances and drug molecules in the ocular compartment [23] and by using fluorescent quantum dots the integrity and disruptions in the ocular compartments can be studied by monitoring nanoparticle penetration [24]. Nanocrystals are another approach to increasing drug solubility and improving the bioavailability of poorly soluble drugs by reducing the particle size [25]. Vesicular proniosomes, like dendrimer hydrogels, enable sustained release and reduced dosing frequency [26]. Additionally, microneedle technology offers a minimally invasive way for accurate ocular therapy as compared to conventional hypodermic injections for anterior and posterior eye diseases [27]. Novel carriers, such as nanoparticles and liposomes, have been developed. Hydrogel and implants are used to tackle ocular barriers and achieve sustained delivery of anti-vascular endothelial growth factor (anti-VEGF) proteins to the eye segment. Anti-VEGF refers to anti-vascular endothelial growth factor agents, which are drugs that block VEGF, a protein responsible for angiogenesis (formation of new blood vessels). In eye diseases like-Age-related macular degeneration (AMD) and diabetic retinopathy (DR) abnormal blood vessels grow in the retina causing vision damage [28]. Thus, overcoming ocular barriers using innovative delivery platforms, such as bio adhesive polymers, cell penetrating peptides and nano-particles based carriers is crucial for enhancing the therapeutic efficacy of ophthalmic drugs. These approaches continue to pave the way for improved treatment outcomes in ocular diseases [29]. One of the novel approaches for improving ophthalmic drug delivery are stimuli-responsive systems which are being developed to enable spatiotemporally controlled delivery of drugs. They release drugs in response to microenvironmental cues-such as pH variation, ionic strength and reactive oxygen species. For instance, temperature-responsive gels like poloxamer undergo sol to gel transition at ocular temperature prolonging residence time and enhancing bioavailability. pH responsive gels and ion-sensitive formulations, such as Carbopol use sol to gel transition in response to ocular microenvironmental changes. Enzyme-responsive systems, triggered by ocular enzymes like lysozyme, allow site-specific release and minimize off-target toxicity exemplified by chitosan nanogels embedded in contact lenses for sustained timolol release [30]. These advancements have significantly improved therapeutic efficacy, reduced the need for frequent dosing, improved patient compliance and enhanced drug penetration in the eye. A summary of common ocular diseases and their respective novel treatments is presented in “Table 1”.
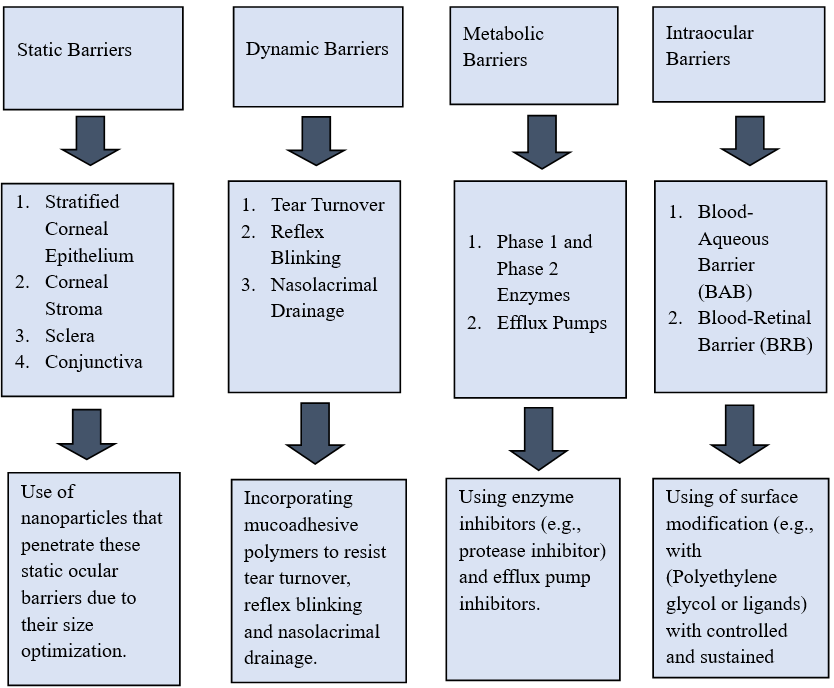
Fig. 1: Fundamental ocular barriers as a major challenge for ocular delivery of drugs and novel solutions
Dendrimers
The term dendrimers derived from Greek words; “dendros” means “tree” or “branch” and “meros” means “part.” These were first synthesized by Fritz Vogtle in 1978 and Donald Tomalia and co-workers in the 1980s. During the same time, Newkome and his group independently synthesized the similar macromolecules and called as “arborols.” The word “arbor” means a tree in Latin [50]. Dendrimers are nanoscale, spherical macromolecules characterized by a unique structure as shown in “Fig.2” comprising three components: (i) A central core, consisting of a single atom or a group of atoms with at least two identical chemical functionalities; (ii) branches extending from the core, made up of repeating units with at least one branching point arranged in geometrical progression to form concentric layers known as generations; (iii) numerous terminal functional groups, typically situated on the macromolecules outer surface which significantly influence its properties [51]. Each of these groups is critical to drug delivery. The core defines molecular weight and architectural structure influencing the number of branching generations that can be built. The branched interior creates internal cavities for drug encapsulation protecting them from premature degradation and enabling sustained drug release. The surface groups provide abundant functional sites for covalent drug conjugation, solubility enhancement, and the attachment of targeting ligands, thereby improving bioavailability and specificity. This design allows dendrimers to act as multifunctional nanocarriers with the ability to improve solubility, prolong ocular residence time and enhance ophthalmic drug delivery [11].
Table 1: Common ocular diseases and their novel treatment
| Disease | Cause | Description | Novel treatment | Reference |
| Glaucoma | Degeneration of retinal ganglion cells and their axons. | Glaucoma is a common eye disease that can cause irreversible blindness if left undiagnosed and untreated. Glaucoma is caused by elevated intraocular pressure (IOP)* that leads to optic nerve damage and visual field loss. | Laser therapies like selective laser trabeculoplasty (SLT)*, minimally invasive glaucoma surgeries (MIGS)* and sustained release implants. | [31, 32] |
| Cataract | Ageing, UV-radiations, genetics, diabetes, smoking, alcohol and chemical toxicity. | A cataract is a lens abnormality characterized by decreased transparency. Cataract is the leading cause of reversible visual impairment and blindness globally. |
Antioxidants, calpain inhibitors, aldose reductase inhibitors, protein stabilizers, calories restriction, hormonal therapy. | [33] |
| Age-related macular degeneration (AMD) | Degeneration of retinal pigment epithelium and choriocapillaris, ageing and genetics. | AMD* is the chief cause of severe and irreversible loss of vision. It is more common in people with European ancestry. | Laser photocoagulation therapy, photodynamic therapy and Anti-VEGF* (anti-vascular endothelial growth factor) therapy. | [34, 35] |
| Diabetic Retinopathy | Chronic hyperglycemia induced damage to retinal microvasculature unit. | Diabetic retinopathy is the most common microvascular complication of diabetes. | Laser photocoagulation, intravitreal anti-VEGF (anti-vascular endothelial growth factor) drugs and intravitreal corticosteroids. | [36, 37] |
| Dry Eye Syndrome | Tear film instability and ocular surface inflammation which causes decreased tear production/increased tear evaporation. | Dry eye disease is caused by extrinsic or intrinsic factors that trigger tear film instability which causes blurred vision, irritation, burning and fluctuating vision. | Artificial tear solutions, artificial tear gels or ointments, cyclosporine ophthalmic, | [38, 39] |
| Conjunctivitis | Infection caused by bacteria, viruses, allergens, irritants or auto-immune inflammation. | Inflammation or infection of the conjunctiva is known as conjunctivitis and is characterized by dilatation of the conjunctival vessels, resulting in hyperemia and edema. | Immunomodulators, mast cell stabilizers, corticoids, antihistamines, NSAID’s* and dual acting/combination therapy (combining two or more therapies for symptom relief) | [40, 41] |
| Uveitis | Auto-immune disorder, infection, trauma, medications, toxins, environmental and genetic factors. | Uveitis is a general term used to describe inflammation of the uveal tract, which is the middle layer of the eye, between the sclera, conjunctiva and the anterior chamber on the outside and the retina on the inside. | Targeted immunotherapy, anti-adalimumab antibodies (AAA)* and extended-release biologics with longer half-lives for extended treatment. | [42, 43] |
| Retinoblastoma | Mutation in the RB1 gene. | A rare malignant tumor of the retina that typically affects young children. | Intra-arterial chemotherapy (IAC)*, intravitreal chemotherapy, transpupillary thermotherapy (TTT)* and targeted molecular therapy. | [44, 45] |
| Optic Neuritis | Immune mediated inflammation; caused by immune system attacking the optic nerve and often associated with multiple sclerosis and autoimmune/infectious triggers. | Inflammation of the optic nerve causing sudden vision loss with eye pain. | Neuroprotective therapies such as: phenytoin, erythropoietin (EPO)*, memantine, interferon-beta and atacicept. | [46, 47] |
| Ocular Surface Squamous Neoplasia (OSSN) | Ultraviolet-B radiation (UVB)*, (HPV)*, (HIV)* infection and other immunosuppressive conditions. | OSSN* is the most common ocular surface tumor. It appears as a pearly, raised lesion on the conjunctiva or cornea and may cause redness, irritation or vision loss. | Topical therapies like: 5-fluorouracil (5FU), mitomycin-C (MMC)*. cryotherapy and adjuvant radiotherapy. | [48, 49] |
SLT*= Selective Laser Trabeculoplasty, MIGS* = Minimally Invasive Glaucoma Surgeries, IOP* = Intraocular Pressure, AMD* = Age-Related Macular Degeneration, Anti-VEGF* = Anti-Vascular Endothelial Growth Factor, NSAID* = Non-Steroidal Anti-Inflammatory Drugs, AAA* = Anti-Adalimumab Antibodies, IAC* = Intra-Arterial Chemotherapy, TTT* = Transpupillary Thermotherapy, EPO* = Erythropoietin, UVB* = Ultraviolet-B radiation, HPV = Human papillomavirus, HIV* = Human immunodeficiency virus, OSSN* = Ocular Surface Squamous Neoplasia, MMC* = Mitomycin-C.
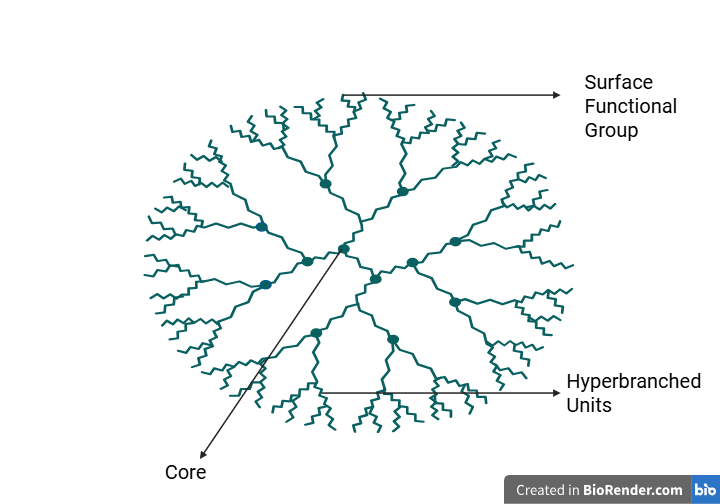
Fig. 2: Structure of dendrimer created in https://BioRender.com
Synthesis of dendrimers
Two primary methods have been developed for dendrimer synthesis. The first is known as the Divergent Method,” which initiates growth from a central core, building outwards by adding monomeric units in a radial, branch upon branch pattern [52]. This method is often through Micheal addition reactions and enables the preparation of very high-generation dendrimers; however, it is difficult to control due to side reactions, requires large quantity of reagents and often results in imperfect symmetry [53]. The second method “Convergent Method’’ it starts from the periphery (dendrons) and converges inward to a focal point, which is then attached to a core. The method is limited by stearic hindrance which results in reduced size of dendrimers but it offers better control over purity, symmetry and overall structural uniformity [54]. Click chemistry is a novel method for synthesis of dendrimers which relies on azide-alkyne cycloaddition reactions. This approach allows to join the molecular building blocks in a highly specific and predictable manner, producing dendrimers with well-defined structures, minimal defects and high purity. This method can be applied to either convergent or divergent strategies and is valued for its modularity, high yield and defect free structures [55].
Generations of dendrimers
The core consists of a single atom or an atomic group that possesses at least two identical chemical functions. Branches extend from the core and contain repeating units with at least one branch junction. These units repeat in a geometrically controlled manner, resulting in a series of radially concentric layers known as generation. With each repetition, a new generation forms, leading to an exponential increase in the number of end groups. As dendrimer generations increase, the structure expands radially, adopting a globular shape. The number of functional surface groups plays a key role in determining the dendrimer properties. These, surface functional groups can be modified, enabling them to be specifically tailored for various biomedical applications in the future. For this to be widely accepted in a clinical setting, Dendrimers’ nomenclature needs to be studied. The nomenclature of Poly amidoamine (PAMAM) dendrimer generations typically begins at generation zero (G0), which has four amino groups. The next level, with eight amino groups, is termed the first generation (G1), and subsequent generations are named similarly [56]. The number of terminal groups and number of atoms in PAMAM Dendrimers G0 to G11 increases with each generation, as shown in table 2.
Table 2: Number of terminal groups and atoms in PAMAM dendrimers [57]
| Generation | Terminal groups | Number of atoms |
| G0 | 4 | 84 |
| G1 | 8 | 228 |
| G2 | 16 | 516 |
| G3 | 32 | 1092 |
| G4 | 64 | 2244 |
| G5 | 128 | 4548 |
| G6 | 256 | 9156 |
| G7 | 512 | 18372 |
| G8 | 1024 | 36804 |
| G9 | 2048 | 73668 |
| G10 | 4096 | 147396 |
| G11 | 8192 | 294852 |
For ocular applications, the choice of dendrimers generation is particularly important, as each tier exhibits distinct physiochemical and biological profiles. Low generations (G2 and G3) are small (1-3 nm), biocompatible, and less immunogenic making them suitable for small molecule delivery. Mid generations (G4-G5) provide a balance between manageable size and increased surface functionality, allowing conjugation of multiple ligands or drugs for tasks such as gene delivery and multimodal imaging. By contrast, higher generations (G6-G7), although offering greater loading capacity, often suffer from incomplete branching, charge heterogeneity and aggregation which limit their reproducibility and in vivo utility. Accordingly, there is a trend favoring low to mid generations (particularly G3-G4) of dendrimers for ocular use. These generations combine favorable size with sufficient functional groups for drug/ligand attachment, while maintaining better safety and stability than higher generations. Thus, G3 and G4 dendrimers are emerging as most practical and reliable choices for ocular delivery systems [58].
Types of dendrimers
Dendrimers are classified based on their shape, structure, branching, chirality, etc. Among the different classes of dendrimers, poly(amidoamine) (PAMAM), poly (propylene imine) (PPI) and poly (propyl ether imine) (PETIM) are the most widely explored dendrimers for pharmaceutical applications because of their commercial availability, high drug loading capacity and ease of synthesis [59]. A detailed overview of these dendrimers types, along with their structural features and distinguished character is provided in table 3.
Table 3: Types of dendrimers
| Type | Description | Reference |
| Poly amidoamine (PAMAM) Dendrimers | In PAMAM dendrimers*, tertiary amine has been used as focal point or core which produces the suitable generation of dendrimers with poly amide branches. | [60] |
| Polypropylene imine (PPI) Dendrimers | PPI dendrimers* also consist of tertiary amines in the interior and propylene imine as branching units. | [61] |
| Poly propyl ether amine (PETIM) Dendrimers | PETIM dendrimers* are synthesized from divergent method having ether linkage and tertiary amine branching points. They have higher water solubility and less cytotoxicity as compared to other types of dendrimers. | [62] |
| Peptide Dendrimers | Peptide dendrimers are radially branched macromolecules consisting of a peptidyl branching core and covalently attached surface functional units. | [63] |
| Hybrid Dendrimers | Hybrid dendrimers are nano systems formed by combining dendrimers with other nanocarriers like liposomes, carbon nanotubes, quantum dots, etc. | [64] |
| Chiral Dendrimers | Chiral dendrimers refer to dendrimers possessing chirality either in their core, branching units or terminal groups. (Chirality is an optical property of a molecule that makes it non-superimposable on its mirror image, much like left and right hands) | [65] |
| Amphiphilic Dendrimers | Amphiphilic dendrimers contain both hydrophilic and hydrophobic component. They are self-assembling structures in both aqueous and polar environments into nanostructures like micelles, vesicles, etc. | [66] |
PAMAM* = Poly amidoamine Dendrimers, PPI* = Polypropylene imine Dendrimers, PETIM* =Poly propyl ether amine Dendrimers
Dendrimer based drug delivery systems for ocular diseases
Recent advancements in dendrimers-based ocular drug delivery systems have demonstrated significant promise in enhancing drug solubility, permeation, bioavailability and targeted delivery to ocular tissues. Various generations and surface modified dendrimers, particularly PAMAM and PPI types have been explored for treatment of anterior and posterior segment eye diseases, including glaucoma, diabetic retinopathy, keratitis and intraocular tumors. These nanocarriers offer advantages such as sustained drug release, improved pharmacokinetics and reduced systemic toxicity. For example, PAMAM Dendrimers have been used to increases the ocular bioavailability of ruboxistaurin, a drug that inhibits protein kinase C beta and reduces vascular endothelial growth factor release, thereby targeting diabetic retinopathy. This approach reduces the need for repeated intravitreal injections, potentially increasing patient compliance. Similarly, dendrimer hydrogel nanoparticles have been explored for anti-glaucoma therapy, carrying drugs such as brimonidine tartrate and timolol maleate. These formulations exhibited low toxicity, minimal ocular irritation, stronger corneal permeation and more effective reduction of intraocular pressure [55]. Although some dendrimer-based systems have not been successful in clinical translation, several new nanoparticles are currently in development and exhibit great promise, thus providing hope for novel therapy options for ocular diseases in upcoming times [67]. A comprehensive summary of recent dendrimer-based formulations, their method of preparation, target applications and therapeutic outcomes is provided in table 4.
Table 4: Novel formulation of dendrimers for the treatment of ocular diseases
| Drug | Excipients | Methods | Application | Key findings | Reference |
| Gatifloxacin | Dendrimeric-polyguanidilyated-translocators (DPT) | Complexation of gatifloxacin with DPT* to make topical eye drops | Conjunctivitis | The complex enhanced solubility, permeability and thus enhances delivery to the cornea | [68] |
| Acetazolamide (ACZ) | G3 cationic carbosilane dendrimers | Synthesis to form mucoadhesive drops | Glaucoma | G3 cationic carbosilane dendrimers improved ACZ* solubility and ocular penetration, reducing onset time and enhancing hypotensive effect duration | [69] |
| Brominidine, Timolol Maleate | PAMAM G3.0 and PEG* acrylate conjugates | Synthesis of PAMAM G.0-PEG Acrylate conjugates via amide bond formation to form Dendrimer Hydrogel | Glaucoma | PAMAM dendrimer hydrogel enhanced solubility, mucoadhesion, precorneal retention,transcorneal penetration and biocompatibility for ocular use of hydrophobic drugs | [70] |
| Puerarin | PAMAM Dendrimers | Topical eye drop formulation | Ocular hypertension | Enhanced corneal permeation and bioavailability with low toxicity | [71] |
| Triamcinolone acetonide | PAMAM Dendrimers | Conjugation to form dendrimer-triamcinolone acetonide conjugates | Age-related macular degeneration (AMD)* | Selective uptake by activated microglia/macrophages and retinal pigmented epithelium, improving therapeutic efficacy | [72] |
| Dexamethasone | PAMAM Dendrimers G3.5 and G4.5 | Complexation; Dex-PAMAM Dendrimer Complex | Diabetic Retinopathy | Enhanced permeability and increased concentration of drug in the retina | [73] |
| Fluocinolone Acetonide | Hydroxyl Terminated PAMAM Dendrimers | Conjugation | Retinal neuroinflammation | Enhanced neuroprotection of photoreceptor against retinal neuroinflammation | [74] |
| Levofloxacin | PPI-G3-DS-Mal (Dense Maltose Shelled PPI* Dendrimer) and Anionic Phosphorus Dendrimer (AN G4) | Non-Covalent Co-Mixing (Physical Mixing) | Bacterial keratitis | Synergistic Antibacterial activity; reduced bacterial growth even with low doses of levofloxacin | [75] |
| Ketorolac | PAMAM Dendrimers (G3.0-NH2 and G4.0-NH2) | Electrostatic complexation | Post-operative inflammation | G4.0-NH2dendrimer enhanced aqueous solubility, showed sustained release and showed improved post-operative anti-inflammation effects. | [76] |
| Carboplatin | PEGylated PAMAM Dendrimers(G4-PEG) | Physical encapsulation | Intraocular tumors (Retinoblastoma) | PEGylated PAMAM G4 Dendrimers enabled higher drug loading capacity, decreased cytotoxicity and enhanced anti-cancer activity against tumor cells. | [77] |
| Benzene Sulfonamide | PAMAM Dendrimers (G0-G3) functionalized with sulfonamide groups. | Covalent conjugation | Glaucoma | G2 and G3 generation dendrimers exhibited strong inhibition of carbonic anhydrase (CA isoforms II and XII) significantly lowering IOP* (Intra ocular pressure) and showed enhanced efficacy and sustained action. | [78] |
| Curcumin | Quaternary ammonium modified PAMAM G3 Dendrimer | Encapsulation using emulsion solvent evaporation | Cataract | Improved solubility, sustained release and reduced cataract formation. | [79] |
| Acyclovir | Thiolated PAMAM G3.5 Dendrimer | Physical encapsulation and covalent conjugation | Herpes Simplex Keratitis/Ocular Viral Infections | Thiolated dendrimers enhanced mucoadhesion, showed sustained drug release and improved drug retention. | [80] |
| Glucosamine-6-sulphate | PAMAM G3.5 Dendrimer | Conjugation | Corneal Scarring/ Post-Surgical Inflammation | Reduced scar formation and inflammation | [81] |
| Pilocarpine Nitrate and Tropicamide | PAMAM Dendrimers (G1.5, G4, G4-OH) | Complexation | Ocular Diagnostics (Glaucoma and Retinal Examination) | PAMAM Dendrimers increased corneal residence time and enhanced pharmacological response. | [82] |
| Ruboxistuarin (RBX) | PAMAM Dendrimers (G4.5 and G5) | Complexation (25:1) ratio (RBX-PAMAM), followed by dialysis and lyophilization. | Diabetic Retinopathy | G4.5 and G5 dendrimer-RBX* complexes showed higher drug loading efficiency, sustained release and safety in retinal cells. | [83] |
| Sulfonamide based MMP* (Matrix Metalloproteinases) inhibitor (Compound 5) | PAMAM Dendrimers conjugated with hydroxamate sulfonamide | Covalent Conjugation | Dry Eye Syndrome (DES)* | Compound 5 showed improved inhibition, increased residence time and restored corneal hydration. | [84] |
| Methotrexate | PAMAM Dendrimers | Conjugation | Ocular Lymphoma | Increased bioavailability and reduced systemic toxicity | [85] |
| Cisplatin | Anionic PAMAM Dendrimer (G3.5-G6.5) | Complexation | Intraocular Tumors | Greater in vivo antitumor efficacy, sustained release and reduced systemic toxicity | [86] |
| 5-Fluorouracil | PEGylated PAMAM-Dendrimer(G4.0) | Complexation | Intraocular Tumors | Increased drug entrapment, sustained release, reduced hemolytic toxicity and increased mean residence time (MRT)* | [87] |
DPT* = Dendrimericpolyguanidilyated translocators, ACZ * = Acetazolamide, PEG* = Polyethylene glycol, PPI* = Poly(propylene imine), AMD* = Age-related macular degeneration, IOP* = Intra ocular pressure, RBX* = Ruboxistuarin, MMP* = Matrix Metalloproteinases, DES* = Dry eye syndrome, MRT* = mean residence time.
Clinical studies of dendrimers for ocular applications
Dendrimers have emerged as promising nanocarriers for ocular drug delivery due to their highly branched architecture, multivalency and modifiable surface functionalities. Before advancing to human trials, pre-clinical evaluation using in vitro and in vivo models is essential to assess safety, efficacy, and pharmacokinetic profiles. Safe and efficient dendrimer drug delivery therapeutics will emerge with rationale chemical design, understanding of biological rationale and predict potential adverse reactions and underlying mechanism and principles [88]. Dendrimers offer several pharmaceutical advantages due to the modification strategies used that make them highly suitable for drug delivery applications. Their aqueous solubility can be tailored through functionalization terminal groups, allowing for improved compatibility with biological fluids. Additionally, dendrimers exhibit enhanced in vivo stability as the hyperbranched architecture provides steric shielding of labile bonds, protecting the molecule from enzymatic degradation or chemical breakdown. This means the dense branching acts as like a protective shield around sensitive chemical bonds, preventing them from being broken down too quickly in the body. Furthermore, their ability to present multiple ligands simultaneously enables bioactivity modulation, facilitating targeted interactions with specific cells or receptors and enhancing the therapeutic activity of the delivered drug [89].
Common modification strategies
Various modification strategies have been used to enhance dendrimer biocompatibility, reduce toxicity and improve targetability. Such modifications not only diminish the undesired effect of dendrimers but also optimize their pharmacokinetics, biodistribution and therapeutic efficacy. Functionalization and Conjugation are two major methods deployed. Functionalization refers to modifying the dendrimer’s core, branches, or surface to improve its physical, chemical or biological properties, such as solubility, stability or biocompatibility. Common strategies include-PEGylation, involves attaching polyethylene glycol (PEG) to mask positive charges, enhancing solubility, drug entrapment, biodistribution and membrane penetration. Longer PEG chains and higher generation dendrimers further improve drug encapsulation and stability. Acetylation neutralizes cationic surface charges, increases water solubility, reducing toxicity and enabling controlled drug delivery with improved pharmacokinetics. Compared to PEGylation, acetylation offers higher cellular uptake and less steric hindrance. Conjugation on the other hand involves attaching specific molecules-like drugs, peptides, anti-bodies or genes directly to the dendrimer. Conjugation strategies further enhance ocular targeting and therapeutic efficacy. Amino acid or peptide conjugation (e. g., arginine, lysine or cell penetrating peptides) improve corneal penetration, retinal uptake and reduces toxicity. Drug, gene or DNA conjugation allows delivery of ocular therapeutics, including anti-inflammatory drugs, anti-glaucoma drugs, improving solubility, stability and cellular internalization. Targeted delivery can be achieved through ligand-conjugated dendrimers, such as folate or sugar derivatives for retinal cells, and RGD (tripeptide sequence containing Arginine-Glycine-aspartic acid) or tuftsin (tetrapeptide sequence containing Threonine-Lysine-Proline-Arginine) peptides for retinal pigment epithelium or macrophages in inflammatory eye diseases. Overall, functionalization and conjugation together provide safer, more efficient, and site-specific dendrimer based ocular drug delivery systems [90].
In vitro studies
Cytotoxicity studies of dendrimers were carried out and it was found that PAMAM Dendrimers having terminal amine groups are cationic in nature and display major difference when compared with anionic and neutral dendrimers. Lower generation of dendrimers with anionic or neutral surface groups had lower toxicity as compared to higher generation dendrimers with neutral and cationic surface groups [74]. In vitro studies using microglial (BV-2) and retinal pigment epithelial (ARPE-19) cells showed that Dendrimer-Triamcinolone Acetonide (D-TA) conjugates were significantly less toxic than Free Triamcinolone Acetonide (TA). At higher concentrations, free TA reduced cell viability by approximately 55-58% due to crystal formation and precipitation, which damaged the cells. While D-TA showed no precipitation and showed over 90% cell viability in both cell lines [73]. The safety of PAMAM Dendrimers was tested on three types of eye cells: human corneal epithelial cells (HCECs), human lens epithelial cells (HLEC’s) and retinal pigment epithelial cells (RPE’s), representing surface, middle and back part of the eye. PAMAM G5.0 was found to be safe at concentrations below 50 mg/ml, but higher concentrations, such as 100 mg/ml, caused cell damage. The cytotoxicity of dendrimers depends on their generation (G3.0, G4.0, G5.0). Higher generation and doses led to more cell damage and apoptosis (programmed cell death) with RPE’s being most sensitive [5]. In transcellular transport studies, PAMAM Dendrimers showed variable permeability based on their generation. Lower generation dendrimers (G0-G2.0) showed higher permeability across epithelial cells while higher generations (G3.0-G4.0) showed lower permeability [91]. Modifications of dendrimers like PEGylation or conjugation with therapeutic agents like – triamcinolone acetonide and DNA help increase solubility and minimize toxicity [92]. Dendrimers are tested for their hemolytic activity to assess how they interact with cell membranes and whether they damage blood cells. Studies have shown that cationic dendrimers can cause generation-and concentration-dependent hemolysis after 1 mg/ml after 1 h of exposure; these tests are called red blood cell (RBC) lysis tests. However, anionic or PEGylated dendrimers exhibited negligible or minimal hemolytic activity, even at elevated concentrations. Changes in the RBC shape, such as cell rounding and aggregation, were observed with some dendrimers, even at very low concentrations. Thus, surface charge, generation number, chemical structure play key role in dendrimer safety and surface modification can greatly reduce their toxicity to blood cells [89].
In vivo studies
Preclinical evaluation of dendrimer based ocular therapeutics commonly employs animal models that mimic human eye diseases. Rodent models including rats and mice, are frequently used due to their well characterized ocular anatomy and suitability for intravitreal and sub conjunctival injections. Rabbit models are preferred for studying dry eye disease (DES) or for lager volume injections due to their larger ocular surface areas and similarity in tear dynamics. Disease-specific models such as the retinal-ischemia-reperfusion (RIR) for glaucoma or acute retinal injury, and autoimmune dry eye models, are routinely employed to assess therapeutic efficacy, tissue distribution and dendrimer formulation [93]. A Rabbit Model having autoimmune dry eye disease (DES) was taken and a single subconjunctival injection of dendrimer-dexamethasone (D-Dex) was administered and it significantly reduced inflammation and tissue damage. Compared to free dexamethasone, the D-Dex conjugate showed better clinical results due to targeted and sustained drug release [92]. In vivo studies on rat models and fluorescent tracking of dexamethasone PAMAM Dendrimers were performed to analyze ocular tissue distribution. In subconjunctival route, Fluorescence level in retina and vitreous were low, indicating limited drug penetration and higher levels were found in sclera and conjunctiva close to injection site. Most of the conjugate was cleared within 2 h due to multiple clearance pathway and less than 10% of administered dose remained after 2 h. In intravitreal route, both G3.5 and conjugates showed presence in vitreous for more than 24 h. G3.5 dendrimers showed higher fluorescence suggesting better retention than G4.5 dendrimers. In G3.5 dendrimers 33% conjugate remained in ocular tissue after 24 h and 15% in G4.5 dendrimers after 24 h. Hence, dexamethasone PAMAM dendrimers improved ocular distribution and extended drug retention especially in intravitreal injection [94]. In another study conducted on Albino Rats similar results were seen with enhanced retention and controlled release via intravitreal route and limited penetration and residence via subconjunctival route due to rapid clearance through blood and lymphatic vessels. Dendrimer formulations when PEGylated and surface modifications are non-irritating to ocular tissues and show minimal toxicity in animal models [95]. In vivo ocular studies using BALB/c mice demonstrated that intravitreal injection of dexamethasone conjugated PAMAM Dendrimer led to sustained drug release for up to 21 days. The formulation showed targeted localization to inflamed retinal cells with no detectable toxicity indicating its potential for long-acting targeted therapy in ocular diseases[88]In a retinal ischemia-reperfusion (RIR) Model, a common model to mimic glaucoma or acute retinal injury in mice, G4 PAMAM Dendrimer-N-Acetyl cysteine (D-NAC) conjugate were administered via sub-conjunctival route and it was observed that D-NAC localized specifically to microglia and muller glia in retina-cells which are responsible for inflammation and degeneration. D-NAC also reduced oxidative stress by inhibiting the expression of iNOS (inducible nitric oxide synthase) and 4-HNE (oxidative stress marker). D-NAC also preserved retinal ganglion cells (RGC’s) after ischemic injury better than free NAC. Moreover, no adverse structural effects were observed indicating good biocompatibility and its potential for non-invasive treatment of retinal diseases [96].
Clinical trials
Clinical trials play an important role in bridging the gap between preclinical research and real-world therapeutic application, ensuring safety, efficacy and regulatory approval of novel drug delivery systems. ARVO (Association for Research in vision and ophthalmology) identified some barriers in clinically translating basic research, these include-Development of a safe and effective product, identifying best mode of delivery and right delivery systems, assessment of product in animal models and recognizing differences in human equivalence, appropriate design of clinical trials and commercialization by finding a suitable commercial partner [97]. Ashvattha Therapeutic’s hydroxyl dendrimer-based PET (Positron emission tomography) imaging agent, 18FOP-801, is undergoing clinical evaluations to assess its efficacy in detecting early stage neuroinflammation, which could have implications for various ocular and neurological conditions [98]. One of the earliest dendrimer candidates, OP-101 (D-NAC), a G4 PAMAM-OH conjugated to N-acetylcysteine, was initially developed for neuro and ocular inflammation. Although clinical trials for OP-101 have primarily targeted COVID-19, demonstrating reduced systemic inflammation and neurological injury in a Phase 2a study, its mechanism – scavenging reactive oxygen species in activated glia, holds direct relevance for ocular inflammatory conditions like – uveitis and diabetic retinopathy [99]. More directly addressing posterior segment diseases, D4517.2 (migaldendranib), a G4 PAMAM-OH conjugated to a VEGFR tyrosine kinase inhibitor has progressed significantly. Following a complete Phase-1 trial establishing safety and pharmacokinetics in healthy volunteers via subcutaneous dosing [100], the ongoing phase 2 trial evaluates chronic dosing in patients with neovascular (wet) AMD and DME(diabetic macular edema) who were previously reliant on intravitreal anti-VEGF injections [101]. As ongoing trials yield further data and new studies emerge, dendrimers are poised to redefine precision medicine in ophthalmology, addressing unmet needs in inflammatory and vascular ocular diseases while paving the way for broader nanomedicine applications.
Regulatory barriers and challenges for approval of novel ocular formulations
The regulatory approval of novel ocular drug delivery systems (ODDS) is substantially difficult due to complexity of the anatomy of the eye and the absence of harmonized, product-specific regulatory frameworks. Advanced nanocarriers based systems like-Dendrimers, micelles, liposomes, etc. have shown potential for enhancing drug retention, trans-corneal permeation and targeted delivery to posterior and anterior ocular tissues. However, their physiochemical complexity causes challenges in Chemistry, Manufacture and Control (CMC) evaluations, regarding reproducibility, critical quality attributes (CQA’s) and scale-up under cGMP conditions [6] Regulatory agencies including Food and Drug Administration (FDA) and European Medicines Agency (EMA) lack definitive guidance regarding ophthalmic medications which often leads case to case assessment of pharmacokinetics, ocular biodistribution and toxicity [102, 103]. Scaling dendrimer production for clinical use presents distinct hurdles depending on the synthetic approach. Divergent synthesis often suffers from incomplete reactions at higher generations, resulting in structural defects, heterogeneous products, and significant batch-to-batch variability complicates CMC compliance and reproducibility. In contrast, convergent synthesis, though structurally more precise, is labor-intensive, requires extensive purification and becomes increasingly impractical for large scale production due to limited yields and high costs [104]. Toxicology concerns further complicate clinical translations. Nanoparticles are known to exhibit size and surface dependent interactions with intraocular tissues, leading to potential accumulation in sensitive sites such as vitreous cavity and retina requiring in vivo and in vitro cytotoxicity profiling using ocular cell lines and animal models [105]. The challenge is made difficult by limited permeability of static barriers such as corneal epithelium, sclera and blood-retinal barrier (BRB) which requires the formulation to be therapeutically effective and also have minimal immunogenicity and off-target effects [8]. Regulatory authorities focus on several key toxicological endpoints when evaluating dendrimer-based ocular formulations. Complement activation is a major concern, as cationic dendrimers can trigger the complementation system, potentially causing inflammation and retinal damage. Long term vitreous retention is another critical factor, since prolonged presence of dendrimers in the vitreous humor may lead to sustained exposure to retinal tissues, increasing the risk of toxicity. Additionally, ocular inflammation and retinal toxicity have been observed with certain dendrimer formulations, highlighting the need for thorough in vivo biocompatibility assessments [106]. Sterilization of complex delivery systems also poses a significant challenge. Conventional methods like autoclaving may compromise the structural integrity or functionality of nanoscale carriers thus requiring the development of aseptic manufacturing protocols and sterile formulations [107]. The lack of in vivo-in vitro correlation (IVIVC) models specific to ocular delivery prolongs the clinical development timeline and increases reliance on Phase 2 and Phase 3 trials [102]. The regulatory landscape is divided across various jurisdictions; for example, The Central Drugs Standard Control Organization (CDSCO) (India), EMA (Europe) and FDA (USA) differ in their emphasis on biocompatibility standards, toxicity limits and requirement for ocular specific pharmacological studies, leading to delays in global harmonization and commercialization[6]. Polydispersity, referring to the variability in size and molecular weight of dendrimer batches, poses significant challenges in CMC evaluations. This variability can affect the reproducibility of critical quality attributes (CQA’s) such as drug loading efficiency, release profiles and surface functionalization. Regulatory agencies require consistent and reproducible manufacturing processes to ensure product quality and patient safety. Study have highlighted the batch-to-batch variations can lead to discrepancies in pharmacokinetics and therapeutical efficacy, complicating the regulatory approval process [104]. Harmonizing global regulatory expectations and adopting a multidisciplinary approach involving formulation scientists, regulatory agencies, and clinical stakeholders to enable safe, effective and timely approval of novel ocular therapeutics.
Advancements and future prospects
Recent advancements in dendrimer-based nanocarriers have demonstrated potential in overcoming ocular barriers enabling more effective and targeted drug delivery. Innovation in small molecules and topical anti-VEGF therapies supported by dendrimer nanocarriers are transforming retinal diseases management through non-invasive and patient-centric approach [108]. Emerging innovations such as 3-D printed personalized devices, smart contact lenses with drug-eluting and real-time monitoring features and AI-assisted treatment strategies promise highly precise therapies [109]. Advanced Thera gnostic approach which refers to a medical approach that combines therapeutics and diagnostic processes into one system allowing for precise and personalized treatment is also being developed to for visual drug tracking during ocular treatment plans. Gene therapy has also emerged as a new scientific approach in ocular therapeutics offering the potential to address genetic and degenerative eye diseases at the molecular level. More than 1500 Clinical trials are ongoing focusing on drug and gene therapy for treating various ocular diseases [110]. Emerging ocular nanocarriers like dendrimers and lipid-based systems enhance tissue penetration, prolonged drug release improving pharmacodynamic precision. Future innovations will focus on non-invasive, site-specific delivery to posterior ocular tissues using surface modified biodegradable vectors reducing systemic toxicity and dosing frequency while improving therapeutic outcomes [111]. Micro-robotic systems guided by magnetic fields are also being explored for targeted drug delivery offering a non-invasive method that could help in ocular diseases treatments [112]. Biodegradable nanocarriers offer controlled, site-specific ocular drug delivery but the clinical progress is constrained by limited human compatible models and safety data [113]. Emerging nanotechnologies in ocular delivery aim to enhance drug targeting, extend residence time and improve residence time. Future innovations include stimuli-responsive nanocarriers, implantable sustained release devices and gene loaded nano-scaffolds for retinal and optic nerve regeneration. However clinical translation will require extensive safety profiling, long term pharmacodynamic evaluation and scalability manufacturing strategies. Sustained interdisciplinary efforts are required for advancements that will translate into real world solutions for complex ocular diseases. The surge of patents in these areas highlights the global interest in translating these innovations into clinical solutions. An overview of key patents involving dendrimers that were granted in recent years is provided in “Table 5,” illustrating the global interest and ongoing development in this field. Building upon these patented innovations several nanotechnology-based delivery systems have advanced to commercialization. Many of these patented technologies are now available as commercially available products addressing clinical needs and providing real world therapies.
Table 5: Patents related to dendrimers
| Patent description | Patent No. | Year of patent | Ocular application/Rationale |
| Dendrimers for sustained release of compounds | US11684569B2 | 2023 | Targeted delivery for suppressing neuroinflammation in retinal diseases such as macular degeneration |
| Compositions comprising a dendrimer-resveratrol complex and methods for making and using the same | US11931321B2 | 2024 | Potential use in treating cataracts and ocular inflammatory diseases; dendrimer-resveratrol complexes may be applied topically to ocular mucosa for targeted therapy. |
| Dendrimer compositions for use in angiography | US11160881B2 | 2021 | Dendrimers can carry imaging agents to the eye, helping detect or treat abnormal blood vessels and inflammation in diseases like diabetic retinopathy and AMD. |
| Injectable dendrimer hydrogel nanoparticles | US11446238B2 | 2022 | Injectable PAMAM dendrimer-based hydrogel nanoparticles (25 nm-10μm) can deliver drugs or imaging agents to the eye, enabling targeted therapy and improved imaging for ocular diseases. |
| Dendrimer compositions and methods for drug delivery to the eye | US12121585B2 | 2024 | Hydroxyl-terminated PAMAM Dendrimers (G4-G10) deliver drugs like VEGFR (vascular endothelial growth factor receptor) inhibitor to retinal/choroidal microglia, enabling targeted therapy and diagnosis of inflammatory or angiogenic eye diseases. |
| Method of delivering genes and drugs to a posterior segment of an eye | US11110183B2 | 2021 | Multifunctional PAMAM Dendrimer nanoparticles deliver drugs and genes (shRNA, small molecules) to the posterior eye enabling targeted and non-invasive treatment of posterior segment diseases. |
CONCLUSION
Dendrimers have moved from being chemical curiosities to some of the most adaptable nanoscale platforms for ocular therapy. Yet, their transition into routine clinical practice is being is being slowed not by lack of promise, but by gaps in manufacturing reproducibility, standardized safety evaluation, and regulatory clarity. The next decade must therefore be shaped less by demonstrating what dendrimers can do, and more by solving how they can be reliably made, tested, and approved. Future progress will depend on four coordinated priorities. First, synthetic scalability must be reimagined: continuous-flow and click-chemistry–based assembly offer the most credible route to reproducible G4–G5 PAMAM dendrimers with minimized polydispersity. Second, ocular-specific toxicology frameworks are needed, including standardized models for complement activation, vitreous retention, and retinal cell compatibility. Third, predictive translational tools—particularly robust IVIVC and computational biodistribution models—should be established to shorten the gap between preclinical and clinical outcomes. Finally, regulatory harmonization across FDA, EMA, and CDSCO must progress from case-by-case judgments to explicit dendrimer guidance, with clearly defined CMC and biocompatibility expectations. By embracing these priorities, dendrimer research can move beyond incremental laboratory studies to clinically transformative solutions. With strategic focus on synthesis, safety, predictability, and policy alignment, dendrimers have the potential not only to overcome the barriers of ocular drug delivery but to set new standards for precision nanomedicine in ophthalmology.
ACKNOWLEDGEMENT
The authors conveyed special thanks to Mr. Jitender Joshi, president, and Prof. (Dr.) Dharam Buddhi, Vice-Chancellor of Uttaranchal University, for their encouragement of research.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
Shalu Verma: Investigation, Conceptualization, drafting, Supervision and visualization. Vivek Silori: Writing review and editing. Prashant Kumar and Muneesh Kanaujaya: writing and analysis.
CONFLICT OF INTERESTS
Declared none
REFERENCES
Singh M, Bharadwaj S, Lee KE, Kang SG. Therapeutic nanoemulsions in ophthalmic drug administration: concept in formulations and characterization techniques for ocular drug delivery. J Control Release. 2020;328:895-916. doi: 10.1016/j.jconrel.2020.10.025, PMID 33069743.
Ahmed S, Amin MM, Sayed S. Ocular drug delivery: a comprehensive review. AAPS PharmSciTech. 2023;24(2):66. doi: 10.1208/s12249-023-02516-9, PMID 36788150.
Bairagi RD, Reon RR, Hasan MM, Sarker S, Debnath D, Rahman MT. Ocular drug delivery systems based on nanotechnology: a comprehensive review for the treatment of eye diseases. Discov Nano. 2025;20(1):75. doi: 10.1186/s11671-025-04234-6, PMID 40317427.
Allyn MM, Luo RH, Hellwarth EB, Swindle Reilly KE. Considerations for polymers used in ocular drug delivery. Front Med (Lausanne). 2022;8:787644. doi: 10.3389/fmed.2021.787644, PMID 35155469.
Qin C, Wen S, Zhu S, Liu D, Chen S, Qie J. Are poly (amidoamine) dendrimers safe for ocular applications? Toxicological evaluation in ocular cells and tissues. J Ocul Pharmacol Ther. 2020;36(10):715-24. doi: 10.1089/jop.2020.0078, PMID 33121321.
Gorantla S, Rapalli VK, Waghule T, Singh PP, Dubey SK, Saha RN. Nanocarriers for ocular drug delivery: current status and translational opportunity. RSC Adv. 2020;10(46):27835-55. doi: 10.1039/D0RA04971A, PMID 35516960.
Yetisgin AA, Sivakumar PM, Cetinel S. Current state and potential of polymersomes as ocular drug delivery systems. Nanoscale. 2025;17(24):14458-76. doi: 10.1039/D5NR01273B, PMID 40471086.
Li S, Chen L, Fu Y. Nanotechnology-based ocular drug delivery systems: recent advances and future prospects. J Nanobiotechnology. 2023;21(1):232. doi: 10.1186/s12951-023-01992-2, PMID 37480102.
Wang J, Li B, Huang D, Norat P, Grannonico M, Cooper RC. Nano-in-nano dendrimer gel particles for efficient topical delivery of antiglaucoma drugs into the eye. Chem Eng J. 2021;425:130498. doi: 10.1016/j.cej.2021.130498, PMID 34121919.
Akhter MH, Ahmad I, Alshahrani MY, Al Harbi AI, Khalilullah H, Afzal O. Drug delivery challenges and current progress in nanocarrier-based ocular therapeutic system. Gels. 2022;8(2):82. doi: 10.3390/gels8020082, PMID 35200463.
Dhull A, Yu C, Wilmoth AH, Chen M, Sharma A, Yiu S. Dendrimers in corneal drug delivery: recent developments and translational opportunities. Pharmaceutics. 2023;15(6):1591. doi: 10.3390/pharmaceutics15061591, PMID 37376040.
Wang J, Li B, Qiu L, Qiao X, Yang H. Dendrimer-based drug delivery systems: history challenges and latest developments. J Biol Eng. 2022;16(1):18. doi: 10.1186/s13036-022-00298-5, PMID 35879774.
Suri R, Beg S, Kohli K. Target strategies for drug delivery bypassing ocular barriers. J Drug Deliv Sci Technol. 2020;55:101389. doi: 10.1016/j.jddst.2019.101389.
Bisht R, Mandal A, Jaiswal JK, Rupenthal ID. Nanocarrier mediated retinal drug delivery: overcoming ocular barriers to treat posterior eye diseases. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2018;10(2):e1473. doi: 10.1002/wnan.1473, PMID 28425224.
Khare A, Grover K, Pawar P, Singh I. Mucoadhesive polymers for enhancing retention in ocular drug delivery: a critical review. Rev Adhes Adhesives. 2014;2(4):467-502. doi: 10.7569/RAA.2014.097310.
Gote V, Ansong M, Pal D. Prodrugs and nanomicelles to overcome ocular barriers for drug penetration. Expert Opin Drug Metab Toxicol. 2020;16(10):885-906. doi: 10.1080/17425255.2020.1803278, PMID 32729364.
Al Qaysi ZK, Beadham IG, Schwikkard SL, Bear JC, Al Kinani AA, Alany RG. Sustained release ocular drug delivery systems for glaucoma therapy. Expert Opin Drug Deliv. 2023;20(7):905-19. doi: 10.1080/17425247.2023.2219053, PMID 37249548.
Villanueva J, Villanueva L. Overcoming barriers: controlled-release systems as vectors the posterior segment of the eye approach as a model. Ophthalmology Res. 2018;8(1):1-10. doi: 10.9734/OR/2018/38862.
Huang D, Chen YS, Rupenthal ID. Overcoming ocular drug delivery barriers through the use of physical forces. Adv Drug Deliv Rev. 2018;126:96-112. doi: 10.1016/j.addr.2017.09.008, PMID 28916492.
Alonso MJ. Nanomedicines for overcoming biological barriers. Biomed Pharmacother. 2004;58(3):168-72. doi: 10.1016/j.biopha.2004.01.007, PMID 15082339.
Sanchez Lopez E, Espina M, Doktorovova S, Souto EB, Garcia ML. Lipid nanoparticles (SLN, NLC): overcoming the anatomical and physiological barriers of the eye part I Barriers and determining factors in ocular delivery. Eur J Pharm Biopharm. 2017;110:70-5. doi: 10.1016/j.ejpb.2016.10.009, PMID 27789358.
Xia Y, Zhang Y, Du Y, Wang Z, Cheng L, Du Z. Comprehensive dry eye therapy: overcoming ocular surface barrier and combating inflammation oxidation and mitochondrial damage. J Nanobiotechnology. 2024;22(1):233. doi: 10.1186/s12951-024-02503-7, PMID 38725011.
Wu H, Xu Y, Cai M, You L, Liu J, Dong X. Design of an L-valine-modified nanomicelle based drug delivery system for overcoming ocular surface barriers. Pharmaceutics. 2022;14(6):1277. doi: 10.3390/pharmaceutics14061277, PMID 35745853.
Duncan TJ, Baba K, Oie Y, Nishida K. A novel method using quantum dots for testing the barrier function of cultured epithelial cell sheets. Invest Ophthalmol Vis Sci. 2015;56(4):2215-23. doi: 10.1167/iovs.14-15579, PMID 25736795.
Sharma OP, Patel V, Mehta T. Nanocrystal for ocular drug delivery: hope or hype. Drug Deliv Transl Res. 2016;6(4):399-413. doi: 10.1007/s13346-016-0292-0, PMID 27165145.
Rathore AS, Verma S, Aggarwal K, Singh A. Proniosomes: a novel vesicular carrier for ocular drug targeting. Asian J Pharm Clin Res. 2025;18(7):42-51. doi: 10.22159/ajpcr.2025v18i7.54552.
Thakur Singh RR, Tekko I, McAvoy K, McMillan H, Jones D, Donnelly RF. Minimally invasive microneedles for ocular drug delivery. Expert Opin Drug Deliv. 2017;14(4):525-37. doi: 10.1080/17425247.2016.1218460, PMID 27485251.
Radhakrishnan K, Sonali N, Moreno M, Nirmal J, Fernandez AA, Venkatraman S. Protein delivery to the back of the eye: barriers carriers and stability of anti-VEGF proteins. Drug Discov Today. 2017;22(2):416-23. doi: 10.1016/j.drudis.2016.10.015, PMID 27818255.
Rusciano D. A personal scientific journey in ophthalmology: twenty-five years of translating research into novel therapies. Pharmaceuticals (Basel). 2025;18(6):883. doi: 10.3390/ph18060883, PMID 40573278.
Kundu S, Kumari G, Srinivasarao DA. Emerging drug delivery strategies for glaucoma therapy: focus on nanoparticles and stimuli-responsive systems. RSC Pharm. 2025;2(5):1050-77. doi: 10.1039/d5pm00068h.
Lee DA, Higginbotham EJ. Glaucoma and its treatment: a review. Am J Health Syst Pharm. 2005;62(7):691-9. doi: 10.1093/ajhp/62.7.691, PMID 15790795.
Weinreb RN, Aung T, Medeiros FA. The pathophysiology and treatment of glaucoma: a review. JAMA. 2014;311(18):1901-11. doi: 10.1001/jama.2014.3192, PMID 24825645.
Lam D, Rao SK, Ratra V, Liu Y, Mitchell P, King J. Cataract. Nat Rev Dis Primers. 2015;1:15014. doi: 10.1038/nrdp.2015.14, PMID 27188414.
Jonas JB, Cheung CM, Panda Jonas S. Updates on the epidemiology of age-related macular degeneration. Asia Pac J Ophthalmol (Phila). 2017;6(6):493-7. doi: 10.22608/APO.2017251, PMID 28906084.
Fine SL, Berger JW, Maguire MG, Ho AC. Age-related macular degeneration. N Engl J Med. 2000;342(7):483-92. doi: 10.1056/NEJM200002173420707, PMID 10675430.
Stitt AW, Curtis TM, Chen M, Medina RJ, McKay GJ, Jenkins A. The progress in understanding and treatment of diabetic retinopathy. Prog Retin Eye Res. 2016;51:156-86. doi: 10.1016/j.preteyeres.2015.08.001, PMID 26297071.
Duh EJ, Sun JK, Stitt AW. Diabetic retinopathy: current understanding mechanisms and treatment strategies. JCI Insight. 2017;2(14):e93751. doi: 10.1172/jci.insight.93751, PMID 28724805.
Marshall LL, Roach JM. Treatment of dry eye disease. Consult Pharm. 2016;31(2):96-106. doi: 10.4140/TCP.n.2016.96, PMID 26842687.
Pflugfelder SC, De Paiva CS. The pathophysiology of dry eye disease: what we know and future directions for research. Ophthalmology. 2017;124(11S):S4-13. doi: 10.1016/j.ophtha.2017.07.010, PMID 29055361.
Azari AA, Barney NP. Conjunctivitis: a systematic review of diagnosis and treatment. JAMA. 2013;310(16):1721-9. doi: 10.1001/jama.2013.280318, PMID 24150468.
Singh N, Mazumder R, Monika M, Sakshi S, Khan F, Kumar B. Development in therapeutic strategies for allergic conjunctivitis. Trends Immunother. 2023;7(1):2025. doi: 10.24294/ti.v7.i1.2025.
Neri P, Perez Y, Pichi F. Precision medicine in uveitis: redefining treatment optimization through biomarkers and tailored therapies. Int Ophthalmol. 2025;45(1):41. doi: 10.1007/s10792-025-03413-9, PMID 39856474.
Munoz Fernandez S, Martin Mola E. Uveitis. Best Pract Res Clin Rheumatol. 2006;20(3):487-505. doi: 10.1016/j.berh.2006.03.008, PMID 16777578.
Ancona Lezama D, Dalvin LA, Shields CL. Modern treatment of retinoblastoma: a 2020 review. Indian J Ophthalmol. 2020;68(11):2356-65. doi: 10.4103/ijo.IJO_721_20, PMID 33120616.
Dimaras H, Kimani K, Dimba EA, Gronsdahl P, White A, Chan HS. Retinoblastoma. Lancet. 2012;379(9824):1436-46. doi: 10.1016/S0140-6736(11)61137-9, PMID 22414599.
Bennett JL. Optic neuritis. Continuum. 2019;25(5):1236-64. doi: 10.1212/CON.0000000000000768.
Tsai TH, Lin CW, Chan LW, Tew TB, Chen TC. Neuroprotective effects of novel treatments on acute optic neuritis-a meta-analysis. Biomedicines. 2022;10(1):192. doi: 10.3390/biomedicines10010192, PMID 35052875.
Lee GA, Hirst LW. Ocular surface squamous neoplasia. Surv Ophthalmol. 1995;39(6):429-50. doi: 10.1016/S0039-6257(05)80054-2, PMID 7660300.
Hollhumer R, Williams S, Michelow P. Ocular surface squamous neoplasia: management and outcomes. Eye (Lond). 2021;35(6):1562-73. doi: 10.1038/s41433-021-01422-3, PMID 33564137.
Munavalli BB, Naik SR, Torvi AI, Kariduraganavar MY. Dendrimers. In: Jafar Mazumder MA, Sheardown H, Al Ahmed A. Functional polymers. Berlin: Springer Nature; 2019. p. 289-345. doi: 10.1007/978-3-319-95987-0_9.
Caminade AM. Dendrimers an emerging opportunity in personalized medicine? J Pers Med. 2022;12(8):1334. doi: 10.3390/jpm12081334, PMID 36013283.
Svenson S, Tomalia DA. Dendrimers in biomedical applications reflections on the field. Adv Drug Deliv Rev. 2005;57(15):2106-29. doi: 10.1016/j.addr.2005.09.018, PMID 16305813.
Baig T, Nayak J, Dwivedi V, Singh A, Srivastava A, Tripathi PK. A review about dendrimers: synthesis types characterization and applications. Int J Adv Pharm Biol Chem. 2015;4(1):44-59.
Najafi F, Salami Kalajahi M, Roghani Mamaqani H. A review on synthesis and applications of dendrimers. J Iran Chem Soc. 2021;18(3):503-17. doi: 10.1007/s13738-020-02053-3.
Tung NP, Lim WM, Liew YK, Lim CJ, Then YY, Cheong KW. Recent advances in nanomedicine for ocular drug delivery. Biomater Transl. 2025. doi: 10.12336/bmt.25.00022.
Kesharwani P, Tekade RK, Jain NK. Dendrimer generational nomenclature: the need to harmonize. Drug Discov Today. 2015;20(5):497-9. doi: 10.1016/j.drudis.2014.12.015, PMID 25578746.
Maiti PK, Cagın T, Wang G, Goddard WA. Structure of PAMAM dendrimers: generations 1 through 11. Macromolecules. 2004;37(16):6236-54. doi: 10.1021/ma035629b.
Bharadwaj P, Roullin VG, Chain JL. Crossing the blood brain barrier: advances in dendrimer-based nanocarriers for central nervous system delivery. Nanoscale. 2025;17(40):23202-27. doi: 10.1039/d5nr02548f, PMID 41031431.
Jain V, Maingi V, Maiti PK, Bharatam PV. Molecular dynamics simulations of PPI dendrimer drug complexes. Soft Matter. 2013;9(28):6482-96. doi: 10.1039/c3sm50434d.
Kaur D, Jain K, Mehra NK, Kesharwani P, Jain NK. A review on comparative study of PPI and PAMAM dendrimers. J Nanopart Res. 2016;18(6):146. doi: 10.1007/s11051-016-3423-0.
Wang D, Imae T, Miki M. Reprint of fluorescence emission from PAMAM and PPI dendrimers. J Colloid Interface Sci. 2007;312(1):8-13. doi: 10.1016/S0021-9797(07)00550-4, PMID 17547920.
Jain S, Kaur A, Puri R, Utreja P, Jain A, Bhide M. Poly propyl ether imine (PETIM) dendrimer: a novel non-toxic dendrimer for sustained drug delivery. Eur J Med Chem. 2010;45(11):4997-5005. doi: 10.1016/j.ejmech.2010.08.006, PMID 20805013.
Sadler K, Tam JP. Peptide dendrimers: applications and synthesis. J Biotechnol. 2002;90(3-4):195-229. doi: 10.1016/S1389-0352(01)00061-7, PMID 12071226.
Kesharwani P, Gothwal A, Iyer AK, Jain K, Chourasia MK, Gupta U. Dendrimer nanohybrid carrier systems: an expanding horizon for targeted drug and gene delivery. Drug Discov Today. 2018;23(2):300-14. doi: 10.1016/j.drudis.2017.06.009, PMID 28697371.
Romagnoli B, Hayes W. Chiral dendrimers from architecturally interesting hyperbranched macromolecules to functional materials. J Mater Chem. 2002;12(4):767-99. doi: 10.1039/b110218b.
Rajasekhar Reddy RR, Raghupathi KR, Torres DA, Thayumanavan S. Stimuli sensitive amphiphilic dendrimers. New J Chem. 2012;36(2):340-9. doi: 10.1039/c2nj20879b, PMID 24039387.
Zhu Y, Liu C, Pang Z. Dendrimer-based drug delivery systems for brain targeting. Biomolecules. 2019;9(12):790. doi: 10.3390/biom9120790, PMID 31783573.
Durairaj C, Kadam RS, Chandler JW, Hutcherson SL, Kompella UB. Nanosized dendritic polyguanidilyated translocators for enhanced solubility permeability and delivery of gatifloxacin. Invest Ophthalmol Vis Sci. 2010;51(11):5804-16. doi: 10.1167/iovs.10-5388, PMID 20484584.
Bravo Osuna I, Vicari-de-la-Torre M, Andres Guerrero V, Sanchez Nieves J, Guzman Navarro M, De La Mata FJ. Novel water-soluble mucoadhesive carbosilane dendrimers for ocular administration. Mol Pharm. 2016;13(9):2966-76. doi: 10.1021/acs.molpharmaceut.6b00182, PMID 27149661.
Holden CA, Tyagi P, Thakur A, Kadam R, Jadhav G, Kompella UB. Polyamidoamine dendrimer hydrogel for enhanced delivery of antiglaucoma drugs. Nanomedicine. 2012;8(5):776-83. doi: 10.1016/j.nano.2011.08.018, PMID 21930109.
Yao W, Sun K, Mu H, Liang N, Liu Y, Yao C. Preparation and characterization of puerarin-dendrimer complexes as an ocular drug delivery system. Drug Dev Ind Pharm. 2010;36(9):1027-35. doi: 10.3109/03639041003610799, PMID 20545508.
Kambhampati SP, Mishra MK, Mastorakos P, Oh Y, Lutty GA, Kannan RM. Intracellular delivery of dendrimer triamcinolone acetonide conjugates into microglial and human retinal pigment epithelial cells. Eur J Pharm Biopharm. 2015;95(B):239-49. doi: 10.1016/j.ejpb.2015.02.013, PMID 25701805.
Yavuz B, Pehlivan SB, Vural I, Unlu N. In vitro/in vivo evaluation of dexamethasone PAMAM dendrimer complexes for retinal drug delivery. J Pharm Sci. 2015;104(11):3814-23. doi: 10.1002/jps.24588, PMID 26227825.
Kalomiraki M, Thermos K, Chaniotakis NA. Dendrimers as tunable vectors of drug delivery systems and biomedical and ocular applications. Int J Nanomedicine. 2016;11:1-12. doi: 10.2147/IJN.S93069, PMID 26730187.
Wronska N, Majoral JP, Appelhans D, Bryszewska M, Lisowska K. Synergistic effects of anionic/cationic dendrimers and levofloxacin on antibacterial activities. Molecules. 2019;24(16):2894. doi: 10.3390/molecules24162894, PMID 31395831.
Prajapati RN, Prajapati SK, Singh N, Gupta R. Polyamidoamine dendrimer-mediated formulation development and in vitro-in vivo evaluation of ketorolac. Asian J Pharm. 2018;12(2):S713-21. doi: 10.22377/ajp.v12i02.2419.
Ho MN, Bach LG, Nguyen TH, Ho MH, Nguyen DH, Nguyen CK. Pegylated poly(amidoamine) dendrimers-based drug loading vehicles for delivering carboplatin in treatment of various cancerous cells. J Nanopart Res. 2019;21(2):43. doi: 10.1007/s11051-019-4486-5.
Carta F, Osman SM, Vullo D, Gullotto A, Winum JY, Alothman ZA. Poly(amidoamine) dendrimers with carbonic anhydrase inhibitory activity and antiglaucoma action. J Med Chem. 2015;58(9):4039-45. doi: 10.1021/acs.jmedchem.5b00383, PMID 25849626.
Yogaraj V, Gautham G, Akshata C, Manikandan R, Murugan E, Arumugam M. Quaternary ammonium poly (amidoamine) dendrimeric encapsulated nanocurcumin efficiently prevents cataract of rat pups through regulation of pro-inflammatory gene expression. J Drug Deliv Sci Technol. 2020;58:101785. doi: 10.1016/j.jddst.2020.101785.
Yandrapu SK, Kanujia P, Chalasani KB, Mangamoori L, Kolapalli RV, Chauhan A. Development and optimization of thiolated dendrimer as a viable mucoadhesive excipient for the controlled drug delivery: an acyclovir model formulation. Nanomedicine. 2013;9(4):514-22. doi: 10.1016/j.nano.2012.10.005, PMID 23117047.
Kambhampati SP, Kannan RM. Dendrimer nanoparticles for ocular drug delivery. J Ocul Pharmacol Ther. 2013;29(2):151-65. doi: 10.1089/jop.2012.0232, PMID 23410062.
Vandamme TF, Brobeck L. Poly(amidoamine) dendrimers as ophthalmic vehicles for ocular delivery of pilocarpine nitrate and tropicamide. J Control Release. 2005;102(1):23-38. doi: 10.1016/j.jconrel.2004.09.015, PMID 15653131.
Alshammari RA, Aleanizy FS, Aldarwesh A, Alqahtani FY, Mahdi WA, Alquadeib B. Retinal delivery of the protein kinase C-β inhibitor ruboxistaurin using non-invasive nanoparticles of polyamidoamine dendrimers. Pharmaceutics. 2022;14(7):1444. doi: 10.3390/pharmaceutics14071444, PMID 35890338.
Cerofolini L, Baldoneschi V, Dragoni E, Storai A, Mamusa M, Berti D. Synthesis and binding monitoring of a new nanomolar PAMAM-based matrix metalloproteinases inhibitor (MMPIs). Bioorg Med Chem. 2017;25(2):523-7. doi: 10.1016/j.bmc.2016.11.028, PMID 27914947.
Sherje AP, Jadhav M, Dravyakar BR, Kadam D. Dendrimers: a versatile nanocarrier for drug delivery and targeting. Int J Pharm. 2018;548(1):707-20. doi: 10.1016/j.ijpharm.2018.07.030, PMID 30012508.
Kirkpatrick GJ, Plumb JA, Sutcliffe OB, Flint DJ, Wheate NJ. Evaluation of anionic half generation 3.5-6.5 poly (amidoamine) dendrimers as delivery vehicles for the active component of the anticancer drug cisplatin. J Inorg Biochem. 2011;105(9):1115-22. doi: 10.1016/j.jinorgbio.2011.05.017, PMID 21704583.
Bhadra D, Bhadra S, Jain S, Jain NK. A pegylated dendritic nanoparticulate carrier of fluorouracil. Int J Pharm. 2003;257(1-2):111-24. doi: 10.1016/S0378-5173(03)00132-7, PMID 12711167.
Duncan R, Izzo L. Dendrimer biocompatibility and toxicity. Adv Drug Deliv Rev. 2005;57(15):2215-37. doi: 10.1016/j.addr.2005.09.019, PMID 16297497.
Lin X, Zhou Y, LV K, Wu W, Chen C. Nanomedicine-based ophthalmic drug delivery systems for the treatment of ocular diseases. Int J Nanomedicine. 2025;20:9221-49. doi: 10.2147/IJN.S532074, PMID 40718639.
Wang J, Li B, Qiu L, Qiao X, Yang H. Dendrimer-based drug delivery systems: history challenges and latest developments. J Biol Eng. 2022;16(1):18. doi: 10.1186/s13036-022-00298-5, PMID 35879774.
Boas U, Heegaard PM. Dendrimers in drug research. Chem Soc Rev. 2004;33(1):43-63. doi: 10.1039/b309043b, PMID 14737508.
Lin H, Liu Y, Kambhampati SP, Hsu CC, Kannan RM, Yiu SC. Subconjunctival dendrimer-drug therapy for the treatment of dry eye in a rabbit model of induced autoimmune dacryoadenitis. Ocul Surf. 2018;16(4):415-23. doi: 10.1016/j.jtos.2018.05.004, PMID 29777869.
Loiseau A, Raiche Marcoux G, Maranda C, Bertrand N, Boisselier E. Animal models in eye research: focus on corneal pathologies. Int J Mol Sci. 2023;24(23):16661. doi: 10.3390/ijms242316661, PMID 38068983.
Yavuz B, Bozdag Pehlivan S, Sumer Bolu B, Nomak Sanyal R, Vural I, Unlu N. Dexamethasone PAMAM dendrimer conjugates for retinal delivery: preparation characterization and in vivo evaluation. J Pharm Pharmacol. 2016;68(8):1010-20. doi: 10.1111/jphp.12587, PMID 27283886.
Gupta U, Agashe HB, Asthana A, Jain NK. A review of in vitro-in vivo investigations on dendrimers: the novel nanoscopic drug carriers. Nanomedicine. 2006;2(2):66-73. doi: 10.1016/j.nano.2006.04.002, PMID 17292117.
Mignani S, Shi X, Cena V, Shcharbin D, Bryszewska M, Majoral JP. In vivo therapeutic applications of phosphorus dendrimers: state of the art. Drug Discov Today. 2021;26(3):677-89. doi: 10.1016/j.drudis.2020.11.034, PMID 33285297.
Edelhauser HF, Rowe Rendleman CL, Robinson MR, Dawson DG, Chader GJ, Grossniklaus HE. Ophthalmic drug delivery systems for the treatment of retinal diseases: basic research to clinical applications. Invest Ophthalmol Vis Sci. 2010;51(11):5403-20. doi: 10.1167/iovs.10-5392, PMID 20980702.
Ashvattha Therapeutics. Ashvattha therapeutics announces first patient enrolled in expanded phase 1/2 study of imaging agent 18FOP-801 in additional neurological indications. Redwood City, CA: Ashvattha Therapeutics; 2024 Feb 22. Available from: https://avttx.com/ashvattha-therapeutics-announces-first-patient-enrolled-in-expanded-phase-1-2-study-of-imaging-agent-18f-op-801-in-additional-neurological-indications/. [Last accessed on 04 Oct 2025].
National Library of Medicine (US). A phase 2 study to evaluate the safety and efficacy of OP-101 in patients with COVID-19. Bethesda (MD): National Library of Medicine (US); 2020 Apr 27. Available from: https://clinicaltrials.gov/study/NCT04321980.
National Library of Medicine (US). A study to evaluate the safety tolerability and pharmacokinetics of D-4517.2 after subcutaneous administration in healthy participants. Bethesda (MD): National Library of Medicine (US); 2021 Oct 5. Available from: https://clinicaltrials.gov/study/NCT05105607. [Last accessed on 04 Oct 2025].
National Library of Medicine (US). A phase 2 study of subcutaneous migaldendranib (D-4517.2) in subjects with neovascular age-related macular degeneration or diabetic macular edema. Bethesda (MD): National Library of Medicine (US); 2022 23 May. Available from: https://clinicaltrials.gov/study/NCT05387837. [Last accessed on 04 Oct 2025].
Pignatello R, Almeida H, Santonocito D, Puglia C. New challenges in ocular drug delivery. Pharmaceutics. 2024;16(6):794. doi: 10.3390/pharmaceutics16060794, PMID 38931915.
Chauhan A, Fitzhenry L, Serro AP. Recent advances in ophthalmic drug delivery. Pharmaceutics. 2022;14(10):2075. doi: 10.3390/pharmaceutics14102075, PMID 36297511.
Alamos Musre S, Beltran Chacana D, Moyano J, Marquez Miranda V, Duarte Y, Miranda Rojas S. From structure to function: the promise of PAMAM dendrimers in biomedical applications. Pharmaceutics. 2025;17(7):927. doi: 10.3390/pharmaceutics17070927, PMID 40733135.
Rajan PB, Koilpillai J, Narayanasamy D. Advancing ocular medication delivery with nano-engineered solutions: a comprehensive review of innovations obstacles and clinical impact. Cureus. 2024;16(8):e66476. doi: 10.7759/cureus.66476, PMID 39247042.
Mahaling B, Baruah N, Dinabandhu A. Drug delivery systems for infectious eye diseases: advancements and prospects. JNT. 2024;5(4):133-66. doi: 10.3390/jnt5040010.
Ashique S, Mishra N, Mohanto S, Gowda BH, Kumar S, Raikar AS. Overview of processed excipients in ocular drug delivery: opportunities so far and bottlenecks. Heliyon. 2024;10(1):e23810. doi: 10.1016/j.heliyon.2023.e23810, PMID 38226207.
Wang W, Wang N, Zhao X, Su X, Liu Z. Recent advancements in polymer science for retinal diseases: new frontiers in drug delivery systems. APL Bioeng. 2025;9(2):020902. doi: 10.1063/5.0264382, PMID 40584817.
Rawat V, Dewangan S. Nanotechnology advancements in treating ocular infectious diseases: a paradigm shift in therapeutic strategies. Int J Adv Res Eng Sci Manag. 2023;11(12):231143. doi: 10.56025/IJARESM.2023.1112231143.
Savvidou G, Ellas S, Efstathopoulos EP. Nanomedicine: pioneering a new frontier in neuro-ophthalmology. Recent Prog Mater. 2024;6(3):1-17. doi: 10.21926/rpm.2403022.
Souto EB, Dias Ferreira J, Lopez Machado A, Ettcheto M, Cano A, Camins Espuny AC. Advanced formulation approaches for ocular drug delivery: state of the art and recent patents. Pharmaceutics. 2019;11(9):460. doi: 10.3390/pharmaceutics11090460, PMID 31500106.
Tang Z, Fan X, Chen Y, Gu P. Ocular nanomedicine. Adv Sci (Weinh). 2022;9(15):e2003699. doi: 10.1002/advs.202003699, PMID 35150092.
Wu KY, Tan K, Akbar D, Choulakian MY, Tran SD. A new era in ocular therapeutics: advanced drug delivery systems for uveitis and neuro-ophthalmologic conditions. Pharmaceutics. 2023;15(7):1952. doi: 10.3390/pharmaceutics15071952, PMID 37514137.