
Int J App Pharm, Vol 18, Issue 2, 2026, 387-397Original Article
INNOVATION OF GREEN ESSENTIAL OIL NANOEMULSION (EO-NES) COMBINING NUTMEG (MYRISTICAFRAGRANS) AND GINGERGRASS (CYMBOPOGONMARTINIIVAR. SOFIA): POTENTIAL AS A HALAL COSMETIC PRESERVATIVE
NANIEK WIDYANINGRUM1*, EKA WULANSARI1, NADIA MIFTAHUL JANNAH1, RINA WIJAYANTI1, THENDI ABDUL ARIEF2
1Professional Pharmacist Study Program, Faculty of Pharmacy, Sultan Agung Islamic University, Semarang-50112, Indonesia. 2Master of Clinical Pharmacy, Faculty of Pharmacy, Universitas Gadjah Mada, Yogyakarta, Indonesia
*Corresponding author: Naniek Widyaningrum; *Email: naniek@unissula.ac.id
Received: 14 Sep 2025, Revised and Accepted: 19 Jan 2026
ABSTRACT
Objective: Nutmeg (Myristica fragrans) and gingergrass (Cymbopogon martinii var. sofia) essential oils in nanoemulsions promising antimicrobial and antioxidant activities. This study aimed to optimize the formulation of essential oil nanoemulsions (EO-NEs) combining Nutmeg and gingergrass using the Simplex Lattice Design (SLD) and antimicrobial effectiveness as a halal cosmetic preservative.
Methods: The nanoemulsions were prepared using a high-energy homogenization method with Tween 80 as the surfactant. Optimization was conducted using SLD to determine the optimal combination of essential oils and surfactant. Physical characterizations, including particle size, viscosity, pH, and stability, were performed. Antimicrobial activity was assessed using the disc diffusion method against bacteria and fungi.
Results: The optimized EO-NEs showed that the ratio of 0.591% Nutmeg and 9.409% ginger grass is the optimum mixture that produces physical properties of viscosity 9.39; pH 3.95; particle size 89.44; zeta potential-10.95; transmittance 88.04 with good stability and a desirability value of 0.915. Thermal cycle stability testing showed only minor changes in physical properties. Antimicrobial evaluation revealed that the EO-NEs demonstrated significant inhibitory effects, withno bacterial growth<10 CFU/ml for 28 days, excellent microbial reduction with a reduction of at least 99.0% (2 log) on day 2 and 99.9% (3 log) on day 7, and excellent microbial reduction with a reduction of at least 99.0% (2 log) on day 2 and 99.9% (3 log) on day 7.
Conclusion: The nutmeg and gingergrass EO-Nes formulated using the SLD method showed good physical stability and significant antimicrobial activity, indicating its potential as an effective natural preservative for halal cosmetic products.
Keywords: Antimicrobial activity, Gingergrass, Nutmeg, Nanoemulsion, Natural preservative, Halal, Simplex lattice design
© 2026 The Authors.Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i2.56868 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Nutmeg essential oil (EO) (Myristica fragrans) contains various bioactive compounds, including myristicin, elemicin, safrole, and eugenol, which are important as antioxidants and anti-inflammatory agents [1]. Gingergrass EO (Cymbopogon martinii var. sofia) is rich in compounds such as geraniol, citronellol, and farnesol, which exhibit strong antifungal and antioxidant activities [2]. In addition, both essential oils possess potent antimicrobial properties [3, 4]. Prior investigations have demonstrated that nanoemulsion formulations of Cymbopogon martinii EO achieved minimum inhibitory concentration (MIC) values as low as0.37-1.97 mg ml⁻¹ when tested against Enterococcus faecalis [5]. Furthermore, Nutmeg EO has exhibited significant antibacterial activity against Shigella sp. and Escherichia coli, characterized by inhibition zones ranging from 12.11 mm to 10.11 mm [6]. These findings underscore the significant promise of essential oils as antimicrobial constituents. Therefore, it is anticipated that combining Nutmeg EO and Gingergrass EO will result in a synergistic effect in inhibiting bacterial growth and promoting bacterial cell death. This research is situated within the pretext consumer rising demand for natural-based cosmetics and simultaneously escalating global necessity for halal-certified formulations, which must comply with Islamic jurisprudence and avoid ingredients, such as alcohol of non-halal origin. This ethical and regulatory aspect constitutes a significant driver in the global cosmetic industry.
Nutmeg EO and Gingergrass EO possess highly hydrophobic characteristics, resulting in low water-phase solubility, high volatility, and poor long-term stability. These inherent limitations subsequently restrict their roles as direct, natural preservatives within various ranges of food and cosmetic products [7]. Consequently, the development of innovative strategies is essential to modify the formulation of these essential oils in order to enhance their physicochemical properties, stability, and bioactivity. One of the most promising approaches is the development of nanoparticles in the form of nanoemulsions [8]. Encapsulating essential oils into nanoemulsions has been demonstrated to effectively increase water solubility, mitigate volatility, reduce overall toxicity, and mask potent flavors. Thus, formulating Nutmeg EO and Gingergrass EO into nanoemulsions with optimized composition is imperative to maximizing their efficacy [9].
In cosmetics, nanoemulsions function as drug delivery systems, facilitating the enhanced penetration of active compounds into the skin, while also serving as a matrix for encapsulating desirable bioactive compounds within alcohol-free formulations [10]. The inclusion of ethanol as a solvent or preservative, however, entails challenges for halal compliance. As a consequence, developing alcohol-free formulations offers a crucial advantage, ensuring suitability for Muslim consumers and aligning with the principles of clean beauty and sustainable formulation. The physicochemical characteristics of nanoemulsions are responsible for their robust long-term stability, typically exhibiting droplet sizes ranging from 20 to 100 nm, which can be achieved through both high-and low-energy processes. Further, their capacity to improve interactions with target sites within products contributes to antibacterial and antioxidant properties, positioning them asa more environmentally friendly alternative. As a result, EO-based nanoemulsions hold significant potential for utilization as natural preservatives in various formulations [11].
In alignment with the current global trends toward natural ingredients and halal-certified products, the development of Nutmeg EO and Gingergrass EO in nanoemulsion form is deemed highly relevant. Nanoemulsion technology provides an innovative solution to enhance the bioavailability, stability, and efficacy of essential oils for industrial applications, particularly as active ingredients in natural cosmetics [12]. Therefore, research on the formulation of Nutmeg EO and Gingergrass EO nanoemulsions holds not only scientific value but also extensive application potential in the cosmetic industry. This study further emphasizes the development of a halal-compliant, alcohol-free preservative system utilizing a nanoemulsion approach, contributing to the expansion global halal cosmetics market. Consequently, this study is expected to contribute to the advancement of nanoemulsion-based delivery systems, enhancing the utilization of essential oils as natural preservatives while supporting halal biotechnology-driven product innovation.
MATERIALS AND METHODS
Tools and materials
Nutmeg EO and Gingergrass EO were sourced by PT. Rumah Atsiri and procuredon June 4, 2025. PT. Rumah Atsiri’s current operations are rooted in the legacy of a former Citronella distillation facility initially established in 1963. In 1986, the distillation was taken over by PT. Intan Purnama Sejati, and after a series of ownership transitions, PT. Rumah Atsiri Indonesia eventually acquired the company in 2015. Additionally, the equipment and materials used in this study included Tween from Merck, GC-FID and GC-MS from Shimadzu, Ethanol pro analysis from Merck, and PSA from Malvern.
Identification test of nutmeg EO and gingergrass EO compounds using GC-MS
The chemical components of Nutmeg and Gingergrass EOs were identified using gas chromatography–mass spectrometry (GC-MS, Shimadzu GC-MS-QP2010) equipped with an SH-RTX-5MS capillary column (60 m × 0.32 mm × 0.25 µm; 5% diphenyl/95% dimethylpolysiloxane as the stationary phase) [13]. The chromatographic analysis commenced with an initial oven temperature of 60 °C (held for 1 min), followed by a ramp to 200 °C. Operating parameters included an injector temperature of 200 °C, an ion source temperature of 200 °C, an interface temperature of 250 °C, a split ratio of 1:30, a helium carrier gas pressure of 36.2 kPa, a column flow rate of 0.75 ml/min, and a total gas flow of 101.3 ml/min. Detection was accomplished via electron ionization (EI) within a mass range of 40–400 m/z. The total duration for each sample analysis was 14 min. The constituents of the essential oils were identified by comparing the obtained mass spectra with reference data from the MS Library Index [14, 15].
Identification test of ethanol content of nutmeg EO and gingergrass EO using GC-FID
A series of methanol standard solutions were systematically prepared via the dilution of the methanol stock solution to yield concentrations of 0.1, 0.3, 0.5, 1.0, 1.5, 2.0, 2.5, and 3.5%. Nutmeg and Gingergrass essential oils, as research samples, were each supplemented with 2% methanol. To each standard solution and sample, 2% n-propanol was incorporated to serve as an internal standard. The analysis was executed using a GC-FID instrument with an injector temperature of 250 °C and a detector temperature of 300 °C, utilizing split ratio of 1:50. The column temperature program was commenced at an initial temperature of 50 °C for 2 min, followed by a programmed ramp of 10 °C/min until reaching 200 °C, which was maintained for 5 min. The column flow rate was set at 0.8 ml/min. Meanwhile, the flow rates for the respective gases were as follows: carrier gas (helium) at 1.2 ml/min, hydrogen at 35 ml/min, nitrogen at 30 ml/min, and the airflow as oxidant at350 ml/min [16, 17].
Production of EO-NEs combination of nutmeg and gingergrass
The nanoemulsions were prepared using a modified high-energy homogenization method. The formulation comprised essential oil (EO) at concentrations ranging from 2.5–10%, Tween 80 surfactant at a fixed 10%, and distilled water (aqua destillata) added to reach a total volume of 100%. Tween 80 was first dissolved in distilled water under magnetic stirring (500 rpm, 25 °C). Upon achieving homogeneity, EO was gradually introduced while continuously stirring for 20 min. The nanoemulsions were prepared using a high-energy ultrasonication technique to achieve fine droplet dispersion. The emulsification process was performed using a QSonica Q500 ultrasonic processor (500 W, 20 kHz) equipped with a 13 mm titanium probe. The sonication was performed at 10% amplitude, corresponding to an approximate acoustic power output of 50 W, with a 50% duty cycle (5 seconds on/5 seconds off) to minimize heat buildup. The total sonication duration was 15 min for each 50 ml batch, which was simultaneously immersed in an ice bath to strictly control temperature (<30 °C) and thereby prevent oil volatilization or thermal degradation [10].
Evaluation of EO-NEs characterization of nutmeg and ginger grass combination
The characterization of nanoemulsions was conducted using a Zetasizer (Malvern Instruments) to determine the average particle size (droplet size), polydispersity index (PDI), and zeta potential. Prior to measurement, nanoemulsion samples were diluted 100-fold with distilled water and analyzed at 25 °C after reaching equilibrium for 120 sec. Physical quality evaluation of the nanoemulsions included measurements of pH, viscosity, and homogeneity. Before pH measurement, the pH meter (Aqua Horiba) was calibrated using standard buffer solutions at pH 4 (acidic), pH 7 (neutral), and pH 10 (alkaline) to ensure accuracy. pH values were determined by immersing the electrode into the sample until a stable reading was achieved. Viscosity was measured using a Brookfield DV-2 viscometer with a sample volume of 100 ml, spindle No. 63, and a rotational speed of 30 rpm. Homogeneity was evaluated visually by observing the presence or absence of phase separation or particle dispersion irregularities within the samples. The morphology of the nanoemulsions was examined using Transmission Electron Microscopy (TEM, JEOL) to obtain visual images of particle structures. Turbidity was measured using a UV-Vis spectrophotometer at a wavelength of 600 nm and a temperature of 25 °C, employing samples that were diluted 10-fold [18-22].
Stability test of EO-NEs combination of nutmeg and ginger grass
Stability testing was first conducted using a cycling test method over a duration of 16 days (8 cycles). The EO-NEs formulations were stored at 4 °C for 24 h and thereafter transferred to 40 °C for an additional 24 h [22]. Cycling, droplet size, polydispersity index, and zeta potential were evaluated. To assess pH stability, 5 ml of nanoemulsion was adjusted to the specified pH (4–8) using 0.1 mol/l NaOH or HCl. The samples were then diluted 100-fold with distilled water, after which droplet size and zeta potential were measured [23]. Although cosmetic products are typically formulated within a narrow pH range suited to skin compatibility (approximately pH 4.5-6.5), this comprehensive assessment was conducted to determine the formulation’s robustness and physicochemical behaviour under stress conditions such as contamination, temperature fluctuations, or contact with other formulation components. The objective was to observe the system’s resilience to pH-induced destabilization, particularly in terms of phase separation, particle size variation, and visual appearance, rather than to propose pH adjustment as a component of production. Future research should involve complementing accelerated stability tests to provide a more comprehensive evaluation of formulation stability under standard cosmetic storage conditions.
RESULTS AND DISCUSSION
Preservative effectiveness test on multiple emulsion cream
The multiple emulsion cream formulation was developed from the study conducted by [23], which served as the reference base composition. This research involved the modification of the base formula by incorporating 0.8% essential oil nanoemulsion (EO–NEs) to boost the formulation’s antimicrobial and antioxidant potential, as well as evaluating its physical compatibility within the emulsion matrix. Each sample container was inoculated with 1 ml of microbial suspension, each containing 1.1 x 105cfu/ml for Aspergillus niger, 1.6 x 105cfu/ml for Candida albicans, 4.6 x 106cfu/ml for Enterobacter aerogenes, 4.2 x 106cfu/ml for Pseudomonas aeruginosa, and 7.6 x 106cfu/ml for Staphylococcus aureus. Homogenization was performed using a vortex mixer. Next, 20 g of each inoculated sample was transferred into three distinct containers for evaluation at 7, 14, and 28 d intervals. Microorganism quantification at each time point involved serial dilutions from 10-1 to 10-4 using 1% peptone solution. 1 ml of each dilution was then inoculated into petri dishes containing the appropriate media (Tryptic Soy Agar (TSA) for bacteria and Sabouraud Dextrose Agar (SDA) for fungi). The bacterial cultures were incubated at 30 °C for 2 d, while the fungal cultures were incubated at 25 °C for 5 d. Microorganism quantification was then conducted using a colony counter. The log reduction of these microorganisms was then calculated using the formula:
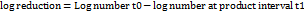
Log number t0 represents the quantity of microorganisms on day 0, while log number t1 indicates the quantity of microorganisms on day 7, 14, or 28. According to the British Pharmacopoeia, a preservative is considered effective in inhibiting the growth of microorganisms if it demonstrates a decrease in bacteria of at least 99.90% (3 logs) on day 7. The quantity does not increase until day 28. The preservative can reduce the numbers of mold or yeast by at least 99.00% (2 logs) on day 14, with no detection of mold or yeast by day 28 of the challenge test [24].
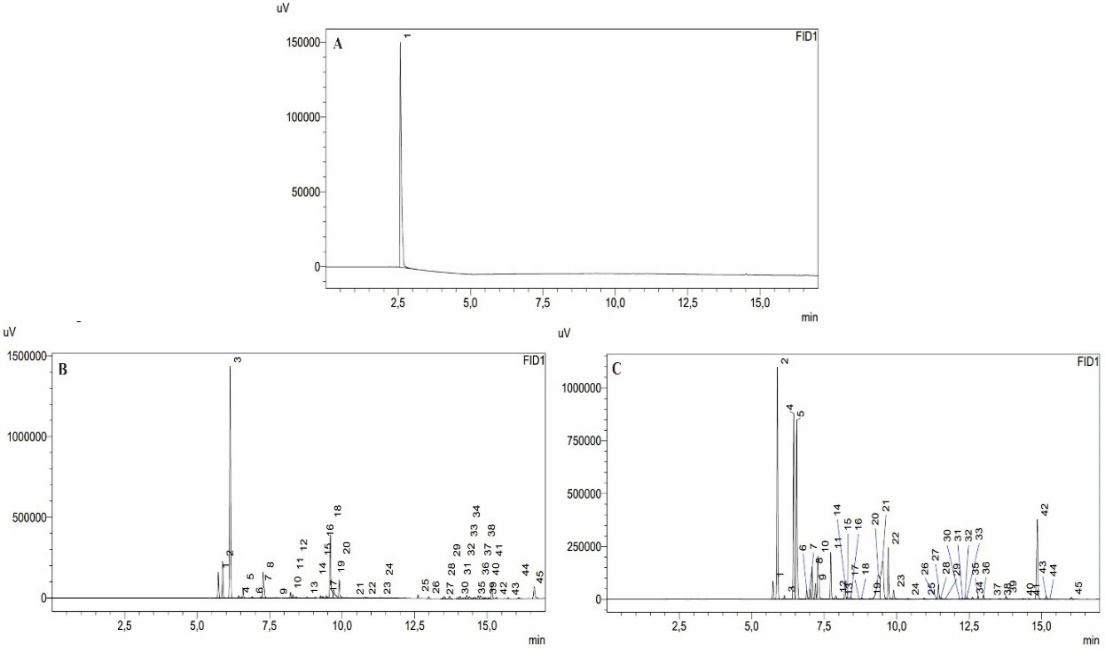
Fig. 1: Chromatogram of GC-FID analysis results. (A) Ethanol standard. (B) Ginger Grass essential oil. (C) Nutmeg essential oil
Table 1: Components of EO ginger grass and EO nutmeg compounds
| Essential oil | Peak | Retention time | Contents | % |
| Ginger grass | 1 | 2,87 | Tricyclene | 3,70 |
| 2 | 2,98 | α-Pinene | 5,19 | |
| 3 | 3,15 | β-Phellandrene | 23,63 | |
| 4 | 3,39 | β-Myrcene | 0,29 | |
| 6 | 3,55 | α-Phellandrene | 1,54 | |
| 8 | 3,98 | p-Cymene | 0,20 | |
| 9 | 4,05 | Limonene | 5,11 | |
| 11 | 4,94 | Linalool | 0,72 | |
| Nutmeg | 1 | 2,90 | α-Thujene | 1,86 |
| 2 | 2,98 | α-Pinene | 16,83 | |
| 4 | 3,14 | Camphene | 0,40 | |
| 5 | 3,40 | Sabinene | 16,23 | |
| 6 | 3,45 | β-Pinene | 14,91 | |
| 7 | 3,55 | β-Myrcene | 2,38 | |
| 8 | 3,75 | l-Phellandrene | 0,86 | |
| 9 | 3,82 | Δ-3-Carene | 0,95 | |
| 10 | 3,89 | α-Terpinene | 3,38 | |
| 12 | 4,05 | Terpinolene | 6,64 | |
| 13 | 4,42 | p-Cymene | 4,79 |
Table 2: Components and responses in EO-Nes optimization
| Run | Component 1 A: Nutmeg (%) |
Component 1 B: Ginger grass (%) |
Response 1 Viscosity (Cp) |
Response 2 pH |
Response 3 Particle size (nm) |
Response 4 PDI |
Response 5 zeta potential (mV) |
Response 6 Transmitter (%) |
| 1 | 5 | 5 | 8.64±0.12 | 3.92±0.09 | 86.72±20.85 | 0.44±0.15 | -15.82±0.74 | 89.459±0,29 |
| 2 | 0 | 10 | 9.13±0.13 | 4.04±0.02 | 104.08±27.21 | 0.57±0.15 | -14.69±1.02 | 90.512±0,18 |
| 3 | 10 | 0 | 8.4±0.06 | 3.72±0.03 | 204.9±43.18 | 0.57±0.05 | -11.34±1.18 | 63.927±0,37 |
| 4 | 7.5 | 2.5 | 9.44±0.24 | 3.77±0.01 | 123.9±14.42 | 0.66±0.08 | -15.61±4.65 | 83.392±0,31 |
| 5 | 10 | 0 | 8.4±0.32 | 3.68±0.035 | 185.7±31.75 | 0.55±0.08 | -11.12±2.21 | 61.435±1,10 |
| 6 | 0 | 10 | 8.91±0.1 | 3.96±0.02 | 103.53±0.31 | 0.62±0.13 | -14.96±1.08 | 82.924±0,31 |
| 7 | 5 | 5 | 8.46±0.24 | 3.7±0.02 | 177.43±6.16 | 0.48±0.07 | -15.84±9.153 | 89.877±0,44 |
| 8 | 2.5 | 7.5 | 8.96±0.09 | 3.85±0.06 | 63.45±2.71 | 0.57±0.08 | -9.72±8.97 | 88.966±0,17 |
Data is expressed as mean and standard deviation. The sample size was 8 formula with 3 replications.
Table 3: Results of ANOVA analysis of EO-Nes formulation with a combination of ginger grass and nutmeg using the simplex lattice design method
| Test | Adjusted R² | Predicted R² | Adeq Precision |
| Viscosity | 0.9105 | NA | 11.5443 |
| pH | 0.6851 | 0.5986 | 7.5969 |
| Particle Size | 0.4649 | 0.2865 | 5.0175 |
| PDI | 0.8840 | NA | 10.5203 |
| Zeta Potential | 0.9968 | NA | 55.199 |
| %Transmitter | 0.9284 | 0.8496 | 14.5153 |
PDI: Polydispersity index
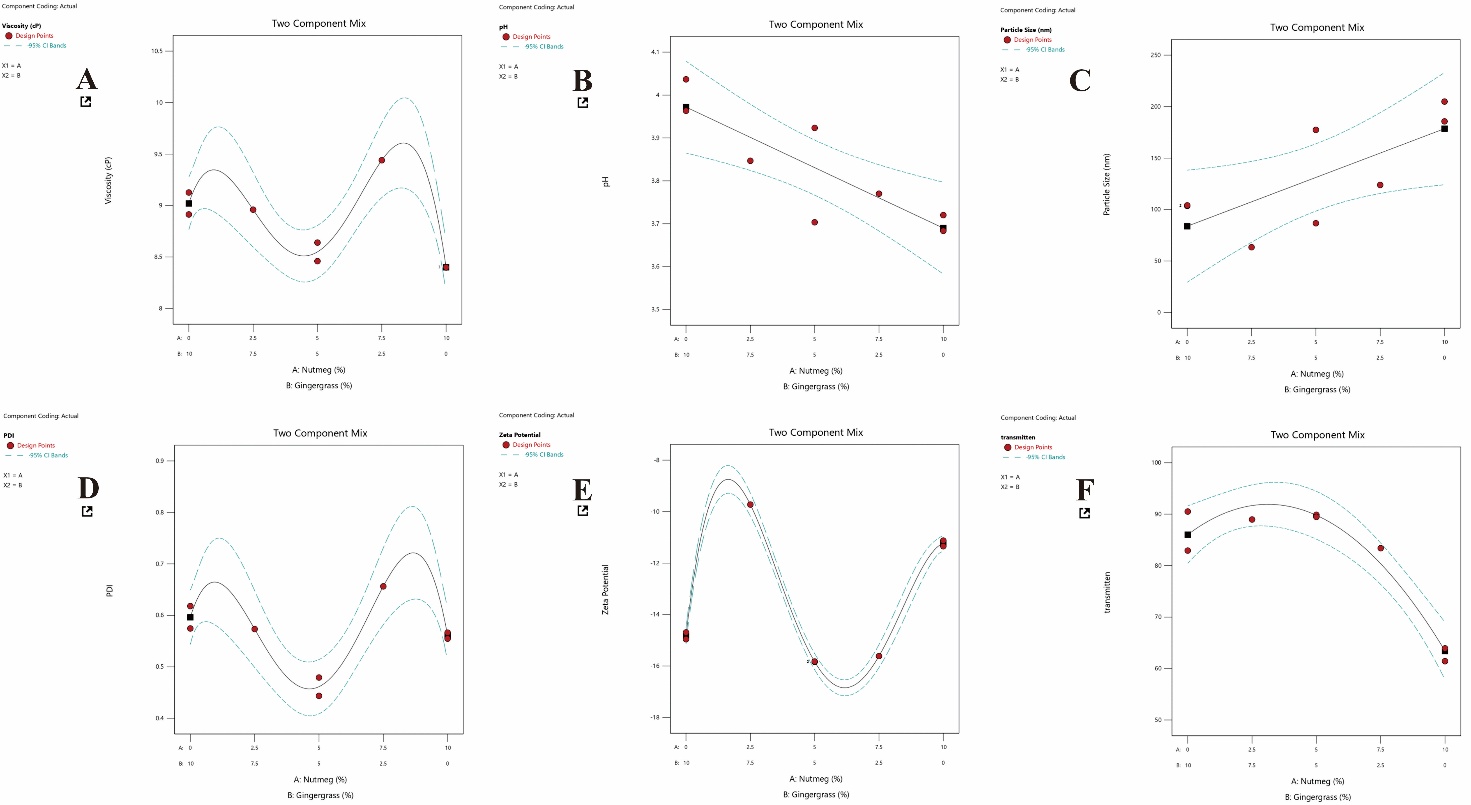

Fig. 2: Response graph generated based on component coding for EO-Nes formulation with a combination of ginger grass and nutmeg using the Simplex Lattice Design. (A) Viscosity value. (B) pH value. (C) Particle size. (D) Polydispersity Index Values. (E) Zeta Potential Values. (F) Transmittance percent values
Table 4: EO-Nes desirability value based on the optimization of the resulting solution
| Number | Nutmeg | Ginger grass | Viscosity | pH | Particle size | PDI | Zeta potential | Transmitter | Desirability | |
| 1 | 0.591 | 9.409 | 9.304 | 3.955 | 89.437 | 0.656 | -10.954 | 88.038 | 0.915 | Selected |
Table 5: Physical characteristics of the selected EO-Nes formulation
| Parameter | Mean±SDa | Lower-upper limitb | Optimum formula valuec | P-valued |
| Viscosity (cP) | 8.59 ±1.44 | 8.4-9.44 | 8.59 | 0.404 |
| pH | 3.90±0.87 | 3.68-4.03 | 3.9 | 0.389 |
| Particle Size (nm) | 161.10±22.36 | 63.45-100 | 161.1 | 0.031 |
| PDI | 0.53 ±0.39 | 0.44-0.65 | 0.5357 | 0.033 |
| Zeta Potential (mV) | -7.59 ±5.80 | -15.84--9.72 | -7.5907 | 0.421 |
| Transmitter (%) | 91.65 ±1.01 | 61.43-90.5 | 91.6523 | 0,025 |
a. Data are presented as mean±standard deviation (mean ±SD) from 3 replications. b. The Lower–Upper Limit represents the predicted range from the SLD model. c. The Optimum Formula Value is obtained from the optimized point of the SLD design. d. P-values indicate the comparison between predicted and experimental values.
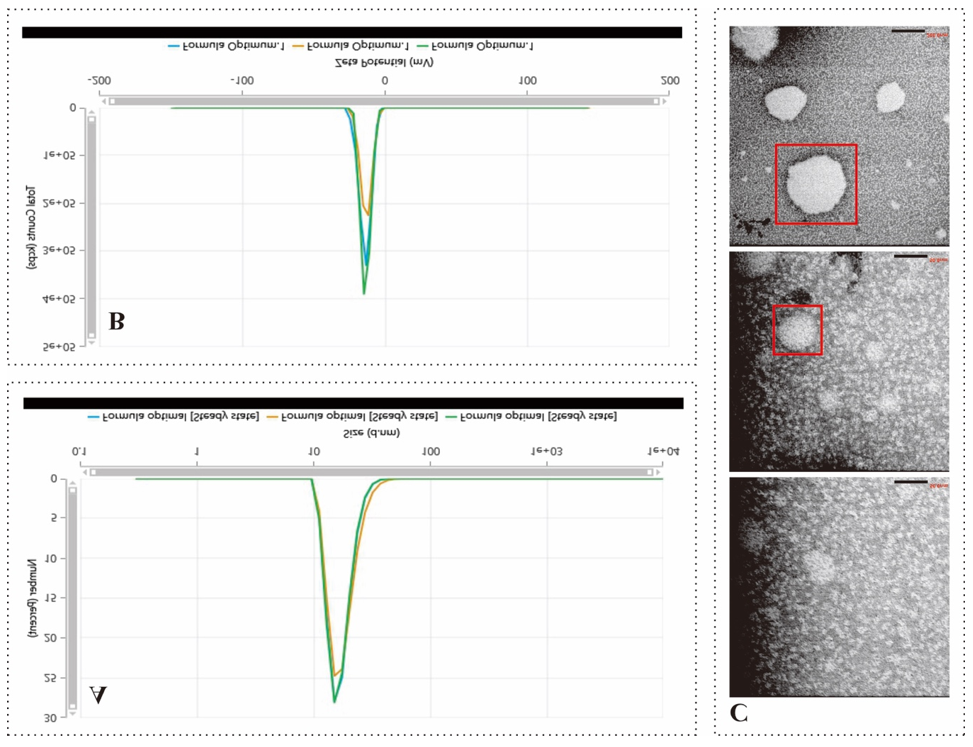
Fig. 3: (A) Graph of EO-Nes particle size distribution. (B) Graph of EO-Nes zeta potential values. (C) Morphology of EO-Nes
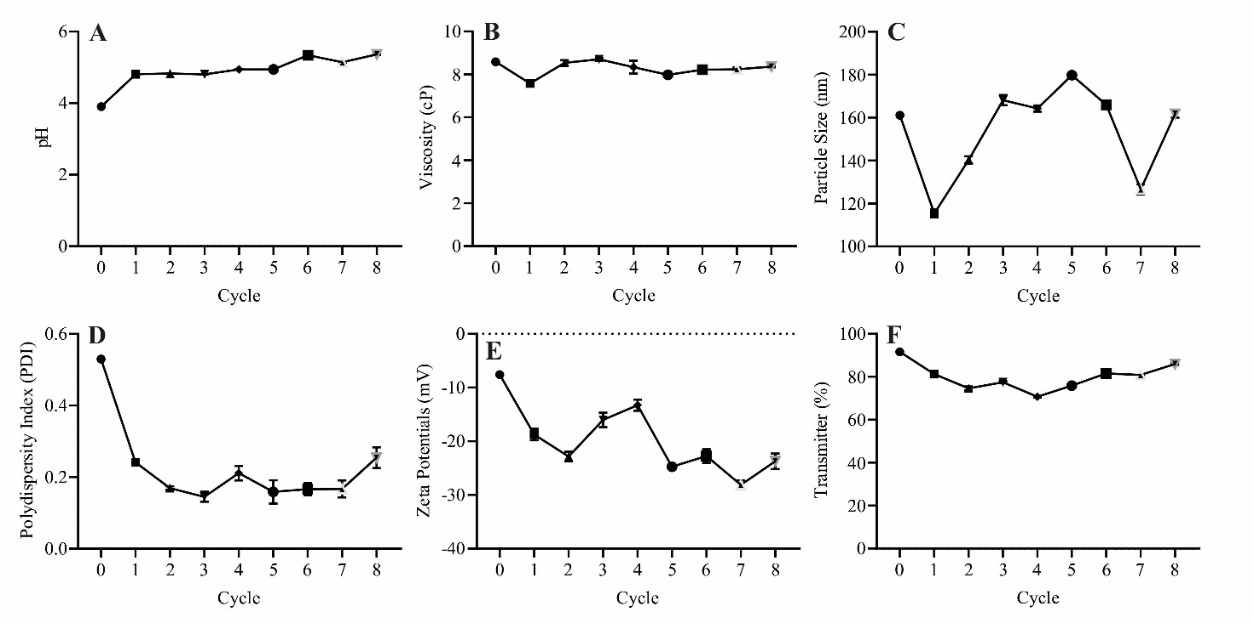
Fig. 4: Results of the stability test of eight heat-cold cycles over 16 d. (A) pH value graph. (B) Viscosity value graph. (C) Particle size graph. (D) Polydispersity index value graph. (E) Zeta potential value graph. (F) Transmittance percentage graph. The samples were analyzed using One-Way ANOVA with 95% confidence, followed by post hoc testing to determine significant differences between each group
Table 6: Preservative effectiveness test on multiple emulsion cream
| Microorganisms | Day-1 (CFU/ml) | Day-2 (CFU/ml) | Day-7 (CFU/ml) | Day-14 (CFU/ml) | Day-28 (CFU/ml) |
| Aspergillus brasiliensis ex A. niger | 1.1 × 105 | <10 | <10 | <10 | <10 |
| Candida albicans | 1.6 × 105 | <10 | <10 | <10 | <10 |
| Enterobacter aerogenes | 4.6 × 106 | <10 | <10 | <10 | <10 |
| Pseudomonas aeruginosa | 4.2 x106 | <10 | <10 | <10 | <10 |
| Staphylococcus aureus | 7.6 × 106 | <10 | <10 | <10 | <10 |
Table 7: Log reduction of preservative effectiveness on multiple emulsion cream
| Microorganisms | Log (t0) | Log (t2) | Reduction log (t7) | Reduction log (t14) | Reduction log (t28) |
| Aspergillus brasiliensis ex A. niger | 5.04 | >4 | >4 | >4 | >4 |
| Candida albicans | 5.02 | >4 | >4 | NI>4 | NI>5 |
| Enterobacter aerogenes | 6.66 | >5 | >5 | NI>5 | NI>5 |
| Pseudomonas aeruginosa | 6.62 | >5 | >5 | NI>5 | NI>5 |
| Staphylococcus aureus | 6.88 | >5 | >5 | NI>5 | NI>5 |
NI: No increase
The GC–MS analysis identified a total of 43 components in ginger grass essential oil and 39 components in nutmeg essential oil (table 1). However, a significant proportion of these compounds demonstrated low similarity indices (35 out of 43 for ginger grass and 28 out of 39 for nutmeg), which hindered accurate identification. This limitation indicates potential issues related to the analytical parameters or the spectral library used for compound matching. The specific and updated MS database, such as the NIST or Wiley library, should be explicitly identified, as variations in database versions can significantly affect the reliability of compound identification [25].
The presence of a major unidentified peak at a retention time (RT) of 12.93, comprising25.85% of the total composition of ginger grass essential oil, represents a critical limitation in the chemical characterization process. To address this issue, further studies are advised to optimize the analytical method. Despite this limitation, several essential components were successfully identified with high similarity index (SI) values (SI>97). A higher SI value indicates a stronger correlation with reference compounds [14].
In ginger grass essential oil, β-Phellandrene (23.63%), α-Pinene (5.19%), and limonene (5.11%) were identified as the dominant components, which is consistent with previous findings. Meanwhile, in nutmeg essential oil, α-pinene (16.83%), sabinene (16.23%), and β-pinene (14.91%) were identified as the primary components, consistent with previous studies. α-Pinene and β-pinene are recognized for their anti-inflammatory, antioxidant, and antimicrobial effects [26, 27]. Whereas sabinene exhibits strong antioxidant and antifungal activity. These findings highlight the potential use of nutmeg essential oil as a natural preservative in cosmetic formulations due to its antimicrobial activity [28].
Qualitative analysis using GC-FID was conducted by identifying the retention times (RT) of the sample readings (fig. 1). The results confirmed that ginger grass and nutmeg essential oils did not contain alcohol, as no peaks indicating alcohol were detected at the expected RT. The chromatograms shown in fig. 1 clearly identified ethanol, with no interfering peaks observed near the ethanol peak, which appeared at RT 2.58 min.
In Indonesia, the use of alcohol or ethanol in cosmetic products is not strictly regulated. Although no specific limits are set for the ethanol concentration in cosmetics, a fundamental principle must be observed: ethanol used in cosmetic formulations must not originate from khamr industries or fermentation processes involving prohibited (haram) materials. Ethanol in cosmetics must comply with safety and quality standards to ensure that products do not pose harmful side effects to consumers [29, 30]. Thus, despite the absence of explicit concentration limits, cosmetic manufacturers must verify that the ethanol used passes safety testing and does not endanger consumers. This analysis indicates that ginger grass and nutmeg essential oils can be considered safe, as they have been confirmed to be alcohol-free. This finding also supports their compliance with halal product requirements, since the absence of ethanol eliminates potential violations of halal standards.
Using the Simplex Lattice Design (SLD), formulations are optimized to determine the optimal ratio of the two essential oils while keeping a constant total oil concentration. The independent variables were defined as A = % nutmeg oil and B = % ginger grass oil, with the constraint A+B = 10% (w/w). Tween 80 was maintained as a fixed surfactant level (10% w/w), while distilled water was added q. s. to 100% of the total formulation weight. Thus, the SLD focused on the relative proportions of the two essential oils as the variables influencing the physicochemical responses. Nutmeg (A) and ginger grass (B) mixtures (table 2) demonstrated a significant influence of compositional variations on product characteristics. The viscosity of the formulations varied between 8.64 and 9.44 cP, with the combination of 5% nutmeg and 5% ginger grass showing the most stable viscosity. The pH values ranged from 3.72 to 4.04, which may influence both product stability and user comfort. Particle sizes varied from 63.45 nm to 204.94 nm, with smaller particles indicating better potential for skin absorption. The polydispersity index (PDI) reflected a relatively homogeneous particle size distribution, with the lowest values recorded in runs 6 and 8. Zeta potential values were between-11.34 and-15.82 mV, which means that the suspension or emulsion was stable. Transmission values of the formulations varied between 63.93% and 89.88%, with the highest transparency observed in runs 1 and 6 [31].
The results of the ANOVA analysis indicated that the EO-NEs formulations combining ginger grass and nutmeg exerted different effects on each test parameter (table 3). The plots in fig. 2 illustrate that the combination of ginger grass and nutmeg influenced various physicochemical response parameters of the EO-NEs formulations. In the graphs, the black line represents the model’s predicted values (fitted line), while the dashed blue lines above and below denote the 95% confidence intervals of the predictions. The red dots correspond to the actual experimental data. When the red data dots are closely aligned with the black line and fall within the range of the blue confidence interval lines, the model can be considered valid and demonstrates good correlation with the experimental results.
For viscosity, the adjusted R² value of 0.9105 and Adeq Precision of 11.5443 indicated that the model was able to explain more than 91% of the data variation and was sufficiently reliable, even though the predicted R² value was not available (table 3). This suggests that formulation plays an important role in determining viscosity; however, the validity of external prediction requires further investigation. In fig. 2A, the viscosity of the ginger grass and nutmeg combination produced a curved pattern, with viscosity values increasing at certain proportions. The red dots, which are relatively close to the black line, indicate that the model aligns completely with the experimental data. A higher viscosity generally reflects a more stable emulsion structure, although excessively high values may reduce the flow ability of the formulation.
For the pH parameter, the adjusted R² value of 0.6851 and predicted R² of 0.5986 indicated that the model could explain the data variation at a moderate level. The Adeq Precision value of 7.5969 confirmed that the model was significant and valid, although the effect of formulation on pH was not as strong as on other parameters (table 3). This finding is understandable since pH is often influenced by additional factors such as excipient composition or storage conditions. Fig. 2B shows a decreasing trend in pH with increasing proportions of ginger grass, while higher proportions of nutmeg tended to maintain more stable pH values. The red dots closely aligned with the black fitted line and stayed within the blue confidence interval. This means that the model can be considered reliable for predicting formulation pH.
For particle size, the adjusted R² value of 0.4649 and predicted R² of 0.2865 indicated that the model explained only a small portion of the variation. Although Adeq Precision (5.0175) remained above the threshold for significance, the relatively low R² suggested that particle size was influenced more strongly by external factors such as homogenization method or processing conditions rather than solely by formulation variations (table 3). As shown in fig. 2C, particle size tended to increase with higher proportions of ginger grass, whereas nutmeg contributed to smaller and more homogeneous particles. While the red data dots followed the general predicted trend, the wider spread of the blue confidence interval indicates greater variability, further emphasizing the influence of processing factors on particle size.
The polydispersity index (PDI) showed favorable results, with an adjusted R² of 0.8840 and Adeq Precision of 10.5203, indicating that the EO-Nes formulations consistently affected particle size distribution (table 3). Fig. 2D PDI exhibits fluctuations across different mixture proportions, but the red dots predominantly followed the predicted line. Lower PDI values reflected more consistent particle size distribution, while higher values indicated heterogeneity. Specific formulation ratios produced low PDI values, which are essential for achieving nanoemulsion stability. In contrast, exceptionally high results were observed for zeta potential, with an adjusted R² of 0.9968 and adeq precision of 55.199 (table 3). These values indicated that the model nearly completely explained the variation in colloidal stability, highlighting zeta potential as a critical parameter for nanoemulsion physical stability. Although predicted R² was not available, the strong relationship between the experimental data and model predictions highlights the importance of formulation in this parameter. Fig. 2E shows the zeta potential exhibiting distinct peak patterns at specific dots, reflecting optimal electrostatic stability. Nearly all red data dots aligned closely with the fitted black line and stayed within the confidence interval, confirming that the model was highly robust. High zeta potential values are particularly important for preventing particle aggregation.
For percent transmittance, the results provided strong conclusions about the clarity of the system. The adjusted R² value of 0.9284 and predicted R² of 0.8496 in table 3 indicated high predictive accuracy. Adeq precision of 14.5153 further supported the validity and reliability of the model in explaining nanoemulsion clarity. As shown in fig. 2F, transmittance values decreased as ginger grass proportions increased. High transmittance values indicated greater clarity, while lower values reflected turbidity resulting from increased particle size. The red dots aligned well with the fitted line, and the narrow confidence intervals suggested strong reliability for the model in explaining clarity variations.
Finally, the desirability value in table 4 was 0.915, indicating that the EO-NEs formulation with 0.591 nutmeg and 9.409 ginger grass approached optimal conditions across the evaluated parameters. Desirability is a composite measure used to evaluate how well a formulation meets the target criteria for each response, such as viscosity, pH, particle size, PDI, zeta potential, and percent transmittance. A value closer to 1.0 reflects better overall balance among the desired responses, confirming that this formulation effectively achieved optimal performance. The stability test results (fig. 4E) showed that the zeta potential values tended to become less negative and increasingly fluctuated during the experimental cycles, indicating a decline in the electrostatic stability of the system. The shift of zeta potential values toward zero shows that the repulsive forces between droplets are getting weaker and the droplets are more likely to come together. However, the low PDI values still indicate a homogeneous particle distribution. This finding is consistent with previous reports showing that a decrease in absolute zeta potential values correlates with reduced colloidal stability and an increased risk of aggregation in nanoemulsion systems [32]. The observed zeta potential fluctuations suggest that the effectiveness of this mechanism in maintaining long-term stability still requires further investigation. These findings may serve as a foundation for formulation optimization in future studies to achieve more consistent physical stability.
Overall, these results confirm that the optimization method based on Response Surface Methodology (RSM) using the Simplex Lattice Design (SLD) approach successfully identified the optimal combination of nutmeg and ginger grass compositions capable of balancing multiple physicochemical responses simultaneously. The high desirability value (0.915) indicates that the formulation can be considered optimal, as it integrates physical stability parameters (viscosity, particle size, PDI, and zeta potential) with favorable product characteristics such as clarity and appropriate pH. Therefore, the optimized formulation has strong potential to be applied as an effective, stable, and safe nanoemulsion-based delivery system [33, 34].
The physical characteristics of the EO-NEs nanoemulsion can be explained by the interactions between raw material composition, the concentration of each component, and the high-energy preparation method using an ultrasonic probe (table 5). Nutmeg oil (5%) and ginger grass oil (5%) served as the oil phase, acting as the core in the formation of nanoemulsion droplets. Both essential oils possess distinct chemical properties, including lipophilic compounds (such as sabinene and myristicin in nutmeg and geraniol in ginger grass), which are relatively insoluble in the aqueous phase without the aid of surfactants. Tween 80, used at 10%, functioned as a nonionic surfactant that reduced the interfacial tension between oil and water, thereby facilitating the formation of smaller oil globules [35].
The low viscosity value obtained (8.59±1.44 cP) was influenced by the relatively low concentration of the oil phase (a total of 10%) and the high proportion of the aqueous phase. Such low viscosity is characteristic of oil-in-water (O/W) nanoemulsions, where the continuous phase is dominated by water. The 10% surfactant concentration was sufficient to stabilize the dispersion, although it was insufficient to significantly increase the system’s thickness. This indicates that the oil–surfactant ratio used produced a system with favorable flow properties.
The relatively acidic pH value of the nanoemulsion formulation (3.90±0.87) was primarily attributed to the natural properties of nutmeg and ginger grass essential oils, which contain phenolic and terpenoid compounds. Constituents such as myristicin, sabinene, and terpinene-4-ol in nutmeg, as well as geraniol and linalool in ginger grass, possess weakly acidic characteristics that tend to lower the formulation’s pH [36, 37]. In addition, the absence of buffering agents in the formulation further contributed to the reduction in pH [38]. Without a buffer system, the acidic properties of the active compounds in the essential oils could not be neutralized, resulting in a lower overall pH. Although the pH of the formulation was slightly below the physiological skin range (4.5–5.5), the value of 3.90±0.87 remains within an acceptable range for short-term topical application [39]. However, long-term use may still pose a risk of irritation. Therefore, irritation testing and skin stability assessments are necessary to ensure the safety of the formulation.
The particle size of the nanoemulsion produced in this study was 161.10±22.36 nm. This size falls within the range generally considered optimal for nanoemulsions, namely 50–200 nm. Particle sizes within this range offer several advantages, including increased surface area for solubility and bioavailability of active compounds, as well as enhanced penetration through semipermeable membranes such as the skin [40]. The use of probe ultrasonication as a high-energy technique played a critical role in achieving this particle size. Ultrasonication generates high-frequency sound waves that induce cavitation, characterized by the formation and collapse of gas bubbles within the liquid. This process produces shear forces and turbulence that break oil droplets into nanometer-sized particles. Although the particle size obtained was within the optimal range, it was slightly larger than the ideal value, which is approximately 89 nm. This discrepancy may be attributed to the limited surfactant-to-oil ratio in the formulation. The 10% concentration of Tween 80 may not have been sufficient to fully cover the oil droplet surfaces, leading to the formation of larger droplets or partial aggregation. Increasing the surfactant concentration could potentially reduce particle size further and improve the overall stability of the nanoemulsion [41, 42].
The PDI value of 0.53±0.39 indicated a heterogeneous particle size distribution. This result is closely related to the ultrasonic homogenization process. Although probe ultrasonication is effective in producing nanosized particles, parameters such as sonication duration, amplitude, and duty cycle strongly influence the final outcome. Insufficient sonication time or inadequate amplitude may lead to non-uniform particle formation. In addition, the complex nature of essential oils, comprising numerous compounds with varying polarities, can compromise system homogeneity, thereby broadening the particle size distribution [43, 44]. The polydispersity index (PDI) reflects the uniformity of particle size distribution, ranging from 0 (indicating perfectly uniform particles) to 1 (indicating highly polydisperse samples with a wide particle size distribution) [45]. The particle size distribution of the nanoemulsion, as shown in fig. 3A, exhibited a monomodal pattern with a narrow peak in the tens-to-hundreds nanometer range, accompanied by a right-hand tail indicating the presence of a fraction of larger droplets. This pattern suggests that the probe ultrasonication process successfully fragmented the oil phase into nanosized globules through cavitation mechanisms; however, some droplets underwent recoalescence once the sonication process ceased. This phenomenon may be influenced by the chemical properties of nutmeg and ginger grass essential oils, which are rich in monoterpenes and volatile compounds, making them more susceptible to ostwald ripening. In addition, the surfactant-to-oil ratio (10% Tween 80 to 10% oil) may not have been sufficient to fully coat the droplet surfaces, causing some globules to merge and resulting in a more heterogeneous particle size distribution. This condition is consistent with the relatively high PDI values, which further indicate that particle size distribution was not yet optimally uniform [46, 47].
The relatively low zeta potential value (07.59 mV±5.80) can be explained by the nature of the materials used. Tween 80 is a nonionic surfactant that stabilizes emulsions primarily through steric stabilization rather than electrostatic stabilization. As a result, the formed particles do not carry a high surface charge; instead, their short-term stability is maintained by the steric barrier provided by the polyoxyethylene chains of Tween 80. The finding explains why the zeta potential values were relatively low, despite the system remaining physically stable in the short term. For strong electrostatic stability, a zeta potential below –30 mV or above+30 mV is generally considered good stability [48]. In this study, the measured zeta potential (fig. 2B) displayed a narrow curve with a peak around –10 mV, indicating that the nanoemulsion droplets carried only a low surface charge, thus providing weak electrostatic stability. This outcome is consistent with the use of Tween 80 as the main surfactant, since its non-ionic nature means stabilization occurs mainly via steric hindrance rather than electrostatic repulsion. Additionally, the relatively acidic pH of the formulation (±3.9) may have contributed to the low zeta potential values. Terpenoid compounds found in nutmeg and ginger grass essential oils are generally neutral, so they do not impart significant charges to the droplet surface. Consequently, the low zeta potential in this system is best explained by the dominance of steric stabilization mechanisms over electrostatic effects [41, 49].
The high transmittance value (91.65±1.01%) indicated that most particles were within the nanometer range and were homogeneously dispersed in the continuous phase. This level of clarity was achieved by combining probe ultrasonication to reduce particle size and Tween 80 to make the dispersion clear. However, since the particle size remained slightly above 100 nm and the distribution was not completely narrow, the transmittance was less than 100% [40].
The morphological analysis of EO-NEs using TEM demonstrated that the droplets were generally spherical in shape, with particle sizes varying within the nanometer range. Fig. 3C shows a relatively dense particle distribution, with most droplets appearing small and homogeneous; however, larger droplets (aggregates) were also observed, as indicated in the red-marked area. This phenomenon aligns with the Dynamic Light Scattering (DLS) measurements, which produced a PDI value of 0.53, indicating a still-heterogeneous particle size distribution [50, 51]. The formation of spherical droplets can be attributed to the amphiphilic nature of Tween 80, a nonionic surfactant that reduces the oil–water interfacial tension and enables the stabilization of oil globules. Tween 80 stabilizes the system primarily through steric mechanisms, in which its polyoxyethylene chains form a physical barrier to prevent coalescence between droplets. However, the 10% surfactant concentration used in this formulation may not have been sufficient to completely coat the entire surface of the oil globules, particularly under conditions of intensive homogenization. This limitation explains the presence of larger droplets, which likely formed due to recoalescence, where smaller droplets merged into larger ones after the ultrasonication process [52].
The stability test results for the EO-Nes formulation, shown in fig. 4, revealed significant fluctuations in several parameters after undergoing eight heat-cold cycles. Based on the analysis using one-way ANOVA with a 95% confidence level, followed by post-hoc testing, significant differences were observed in pH values between cycle 0 and cycles 1 to 5 (p<0.0001), indicating a drastic pH change after the formulation underwent heat-cold cycles. This change may be due to the interaction between the active ingredients, nutmeg and ginger grass oils, and Tween 80 as the surfactant in the nanoemulsion system, which affects pH stability during high-and low-temperature cycles. For the viscosity parameter, significant differences were found between cycle 0 and cycle 1 (p<0.0001), cycle 5 (p<0.001), and cycles 6 to 7 (p<0.05). However, no significant differences were observed between cycle 0 and cycles 2, 3, or 4. These viscosity fluctuations are likely influenced by changes in water content and thickening agents in the formulation, which undergo high and low-temperature cycles [32,39]. A similar trend was observed for particle size (p<0.0001 for cycle 0 compared to cycles 1 and 2, p<0.0001 for cycle 0 compared to cycle 5, p<0.0001 for cycle 0 compared to cycle 7, but not significant for cycle 0 compared to cycles 4 and 8). Significant changes in particle size during several cycles suggest possible coalescence or droplet aggregation due to temperature fluctuations, which could affect the stability and quality of the nanoemulsion [53]. However, the particle size remained under 200 nm, thus meeting the criteria for nanoemulsions and within the acceptable range for pharmaceutical and cosmetic applications.
The Polydispersity Index (PDI) and zeta potential also showed significant results (p<0.0001 for all cycles), indicating the particle size distribution and electrostatic charge stability on the particle surface due to the temperature cycles. A decrease in zeta potential may indicate reduced stability in the nanoemulsion system due to lower repulsion forces between droplets, increasing the likelihood of coalescence. The PDI remained low, ranging from 0.10 to 0.24, which suggests that the particle size distribution is sufficiently homogeneous and complies with nanoemulsion standards, which set PDI<0.3 as an indicator of stability [54]. The zeta potential value theoretically falls below the electrostatic stability limit (±30 mV) [19]. However, such a result does not necessarily indicate instability, as the system uses Tween 80, a nonionic surfactant that operates via steric mechanisms. Thus, despite the zeta potential falling outside the ideal range, the system can remain practically stable.
Furthermore, the transmittance values measured in each cycle showed significant changes (p<0.0001 for all cycles), indicating changes in transparency or turbidity of the formulation, which are related to physical instability caused by fluctuations in particle size and distribution. This indicates the formation of larger droplets or temporary aggregation due to extreme temperature cycles. However, the transmittance increased again in subsequent cycles and remained above 80% for most cycles, indicating that the formulation remained within the acceptable range for nanoemulsions with transparent or semi-transparent characteristics [55].
Overall, the stability test results indicated that the EO-NEs nanoemulsion formulation, consisting of 0.591% nutmeg oil, 9.409% ginger grass oil, 10% Tween 80, and distilled water up to 100 ml, can still be categorized as physiochemically stable, despite showing a tendency toward particle size enlargement and fluctuations in zeta potential during storage. The main factors influencing these parameter changes were the chemical properties of the essential oils, which are prone to Ostwald ripening; the borderline surfactant-to-oil ratio, which may have been insufficient to fully cover the droplet surfaces; and the extreme storage conditions applied during the cycling test. Nevertheless, all evaluated parameters except for zeta potential in some cycles remained within the acceptable range for nanoemulsion systems. These findings confirm that the developed formulation has strong potential as a nanoemulsion-based delivery system with adequate stability, although further optimization of surfactant concentration and strategies to prevent ripening are required to ensure long-term stability.
The preservative effectiveness test in this study uses a method based on BPOM regulations [24]; the method refers to the challenge test method. One of the advantages of the challenge test method compared to other methods in assessing the effectiveness of preservatives is that the challenge test can provide strong empirical data on the effectiveness of preservatives in preventing contamination of pathogenic microorganisms. Challenge tests can also provide accurate and objective data on the effectiveness of a preservative under controlled conditions. Consumers use multiple emulsion cream products; the risk of contamination does not come from just one type of bacteria. Instead, it may come from several types, both g-negative and g-positive. Therefore, the study used a mixed test technique, or multispecies inoculation, to simulate the practicalities that may occur when consumers use the product. The types of bacteria to be tested are five microorganisms: Aspergillus brasiliensis ex A. niger, Candida albicans, Enterobacter aerogenes, Pseudomonas aeruginosa, and Staphylococcus aureus.
On day 1, the sample was exposed to a certain number of microorganisms. Table 6 showed no bacterial growth<10 CFU/ml for 28 d, indicating that the preservative was effective in the multiple emulsion cream preparation. In line with the data, table 7 showed excellent microbial reduction with a reduction of at least 99.0% (2 log) on day 2 and 99.9% (3 log) on day 7 and no further increase during subsequent tests within normal data variations, and for molds showed a reduction of 90.0% (1 log) on day 14 and 99.0% (2 log) on day 28. This fig. shows that the mixed bacterial population was reduced by more than 2 log units, indicating very high preservative effectiveness. The result indicates that EO-NEs have a potent bactericidal effect on bacteria so that they can kill bacteria very quickly. The success of all formulas in achieving log reductions of>4 and>5 at the end of the challenge test period indicates that the developed formulations have a strong and stable preservative system, enabling them to overcome potential microbial growth problems. The ability to maintain this effectiveness until day 28 is an important indicator of the product's long-term stability against bacterial contamination during its shelf life.
In general, several factors influence the antimicrobial activity of EO-NEs. First, the composition of active compounds: nutmeg oil is rich in sabinene, myristicin, and elemicin, which possess antibacterial properties, while ginger grass oil contains geraniol, citronellol, and borneol, known for their antibacterial and antifungal activities. Second, the nanoemulsion formulation: nanosized particles (<200 nm) increase the surface contact area with microorganisms and improve the solubility of hydrophobic compounds in aqueous media. Third, synergistic mechanisms among active compounds: the combination of both essential oils produces stronger effects compared to their individual use. Fourth, the structural characteristics of the target microorganisms: Gram-positive bacteria are generally more susceptible than Gram-negative bacteria due to the latter’s more complex cell wall, while fungi such as Candida albicans are highly sensitive to lipophilic compounds that disrupt ergosterol biosynthesis [56].
CONCLUSION
The successful optimization of Nutmeg and Gingergrass Essential Oil Nanoemulsions (EO-NEs) using the Simplex Lattice Design (SLD) method demonstrated a significant improvement in the stability and effectiveness of the active compounds. Optimization of the formulation with varying compositions of nutmeg and ginger grass resulted in an optimal particle size (161.10±22.36 nm), which enhanced the solubility, penetration, and bioavailability of the active constituents. The nanoemulsion formulation also exhibited stable viscosity, pH, and particle size distribution, meeting the required standards for cosmetic applications. The SLD method proved effective in determining the optimal combination of essential oils and surfactant, yielding a nanoemulsion system with good stability and a desirability value of 0.915, indicating strong potential for contributing to the development of halal cosmetic products based on essential oils. Stability testing under temperature cycling (4 °C and 40 °C) revealed only minimal changes in physical properties, with the formulation remaining stable over time. Antimicrobial testing further confirmed that EO-NEs displayed significant inhibitory activity against Gram-positive bacteria, Gram-negative bacteria, and fungi, with inhibition zones comparable to those of the positive control, demonstrating enhanced antimicrobial effectiveness of the essential oils. In addition, the formulation exhibited excellent spreadability and optimal stability without any significant phase separation.
ACKNOWLEDGEMENT
We would like to extend our sincere gratitude to Hibah Dikti for providing the financial support that made this research possible. We also express our appreciation to LPPM Unissula for their invaluable assistance throughout the process of securing the research grant. Additionally, we are deeply grateful to the Faculty of Pharmacy at Unissula for their unwavering support, particularly in providing the necessary laboratory facilities that greatly contributed to the successful execution of this study. Their continuous encouragement and resources have played a significant role in the progress and completion of this research. We declare that there is no conflict of interest in the conduct and publication of this research.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
Naniek Widyaningrum contributed to the conceptualization, supervision, and overall direction of the study, as well as the writing, review, and editing of the manuscript. Eka Wulansari was responsible for the investigation process, methodology development, and formal data analysis. Nadia Miftahul Jannah contributed to the investigation and data curation. Rina Wijayanti performed validation activities and supported the writing and editing of the manuscript. Thendi Abdul Arief contributed to the methodology, data interpretation, and the writing of the discussion section.
CONFLICT OF INTERESTS
Declared none
REFERENCES
Ashokkumar K, Simal Gandara J, Murugan M, Dhanya MK, Pandian A. Nutmeg (Myristica fragrans Houtt.) essential oil: a review on its composition, biological and pharmacological activities. Phytother Res. 2022 Jul;36(7):2839-51. doi: 10.1002/ptr.7491, PMID 35567294.
Dangol S, Poudel DK, Ojha PK, Maharjan S, Poudel A, Satyal R. Essential oil composition analysis of Cymbopogon species from Eastern Nepal by GC-MS and chiral GC-MS, and antimicrobial activity of some major compounds. Molecules. 2023 Jan 1;28(2):543. doi: 10.3390/molecules28020543, PMID 36677603.
Moirangthem S, Patra G, Biswas S, Das A, Nath S, Verma AK. Effect of nutmeg (Myristica fragrans) and tea tree (Melaleuca alternifolia) essential oils on the oxidative and microbial stability of chicken fillets during refrigerated storage. Foods. 2024 Dec 1;13(24):4139. doi: 10.3390/foods13244139, PMID 39767083.
Sezener Kabay MG, Inal S, Gokmen S, Erguden VE, Fındık A, Guvenc T. Antibacterial effects of essential oils on P. aeruginosa, methicillin-resistant S. aureus, and Staphylococcus spp. isolated from dog wounds. Pharmaceuticals (Basel). 2024 Nov 1;17(11):1494. doi: 10.3390/ph17111494, PMID 39598405.
Marinkovic J, Boskovic M, Tasic G, Vasilijevic B, Markovic D, Markovic T. Cymbopogon martinii essential oil nanoemulsions: physico-chemical characterization antibacterial and antibiofilm potential against Enterococcus faecalis. Ind Crops Prod. 2022 Nov;187:115478. doi: 10.1016/j.indcrop.2022.115478.
Ansory HM, Fitriani IN, Nilawatii A. Chemical separation and antibacterial activity of nutmeg seed essential oil against Shigella sp. and Escherichia coli ATCC 25922. IOP Conf Ser: Mater Sci Eng. 2020 May;846(1):012005. doi: 10.1088/1757-899X/846/1/012005.
Guzman E, Lucia A. Essential oils and their individual components in cosmetic products. Cosmetics. 2021;8(4):114. doi: 10.3390/cosmetics8040114.
Chabib L, Hidayat AM, Trianloka AM, Pangestu MI, Suryani A, Yulianto. Therapeutic potential of Cymbopogon schoenanthus (L.) developed into nanoparticle technology. Pharm Educ. 2021;21(2):210-4. doi: 10.46542/pe.2021.212.210214.
Chabib L, Suryani A, Hakim SN, Rizki MI, Firmansyah F, Yulianto. Stevia rebaudiana as a nutraceutical for COVID-19 patients with no sugar diet during recovery and its nanoparticle application. Pharm Educ. 2022;22(2):174-9. doi: 10.46542/pe.2022.222.174179.
Chabib L, Suryani A, Dewi LS, Noviani H, Maharani WH, Indraswari AA. Pineapple fruit extract (Ananas comosus L. merr) as an antioxidant and anti-acne agent made with the nano-emulsion gel delivery system. Pharm Educ. 2023 May 15;23(2):126-32. doi: 10.46542/pe.2023.232.126132.
Kumar A, Kanwar R, Mehta SK. Nanoemulsion as an effective delivery vehicle for essential oils: properties, formulation methods destabilizing mechanisms and applications in agri-food sector. Next Nanotechnol. 2025;7:100096. doi: 10.1016/j.nxnano.2024.100096.
Chabib L, Suryani A, Pangestu MI, Hidayat AM, Trianloka AM. The development of Origanum vulgare L. into nanoparticles in dosage forms. Pharm Educ. 2021;21(2):205-9. doi: 10.46542/pe.2021.212.205209.
Ibibia ET, Bankole TA, Ogunjimi DV, Mmuo AI, Amuda MO. GC-MS profile synthesis and spectral characterization of flavonoid-metal (cadmium, cobalt, copper, nickel) complexes of Jatropha curcus leaves. Int J Chem Res. 2023 Apr 1;7(2):4-12. doi: 10.22159/ijcr.2023v7i2.217.
Guntarti A, Gandjar IG, Jannah NM. Authentication of wistar rat fats with gas chromatography mass spectometry combined by chemometrics. Potravinarstvo Slovak J Food Sci. 2020;14:52-7. doi: 10.5219/1229.
Gamal El Din MI, Youssef FS, Altyar AE, Ashour ML. GC/MS analyses of the essential oils obtained from different Jatropha species, their discrimination using chemometric analysis and assessment of their antibacterial and anti-biofilm activities. Plants (Basel). 2022 May 1;11(9):1268. doi: 10.3390/plants11091268, PMID 35567269.
Monteiro C, Franco JM, Proenca P, Castanera A, Claro A, Vieira DN. Qualitative and quantitative analysis of a group of volatile organic compounds in biological samples by HS-GC/FID: application in practical cases. Forensic Sci Int. 2014 Oct 1;243:137-43. doi: 10.1016/j.forsciint.2014.07.016, PMID 25124884.
Silva Flores PG, Perez Lopez LA, Rivas Galindo VM, Paniagua Vega D, Galindo Rodriguez SA, Alvarez Roman R. Simultaneous GC-FID quantification of main components of Rosmarinus officinalis L. and Lavandula dentata essential oils in polymeric nanocapsules for antioxidant application. J Anal Methods Chem. 2019;2019:2837406. doi: 10.1155/2019/2837406, PMID 30881726.
Widyaningrum N, Bhakti ADA, Maulana AH, Safitri A, Ramadhani AF, Febriani A. Optimization of formula stability testing and antioxidant activity of clay masks as skin moisturizers. Pharm Sci Res. 2025;12(1):33-40. doi: 10.7454/psr.v12i1.1410.
Suryani A, Chabib L, Fitria A, Nurlina S, Kartika ER. Peppermint essential oil nanoliposomes: innovative formulation for effective hair growth. J Appl Pharm Sci. 2025;15(6):178-89. doi: 10.7324/JAPS.2025.207281.
Chabib L, Suryani A, Munawiroh SZ, Mariyam S, Nafiah Z, Laksitorini MD. Enhancing the physical characteristics and shelf life of rice water (Oryza sativa L.) gel shampoo: the role of propylene glycol concentration. Int J Appl Pharm. 2024 Mar 1;16(2):364-70. doi: 10.22159/ijap.2024v16i2.49766.
Widyaningrum N, Arief TA, Darma Y, Ningrum A. Optimization characterization and primary irritation test of serum based on simplex lattice design. 2024;14(2):176-84. doi: 10.22435/jki.v14i1.6650.
Widyaningrum N, Septiana FN, Wijayanti R, Arief TA. Formulasi dan uji antibakteri formula optimum gel kitosan kulit udang vannamei (Litopenaeus vannaemei) sebagai antiakne. JPSCR J Pharm Sci Clin Res. 2024;9(2):268–81. doi: 10.20961/jpscr.v9i2.78870.
Widyaningrum N. Development of anti-acne cream (W/O/W multiple emulsion) containing green tea leaf waste. Sains Medika: Jurnal Kedokteran dan Kesehatan. 2017;8(2):74–8. doi: 10.30659/sainsmed.v8i2.2030.
BPOM. Peraturan kepala badan pengawas obat dan makanan RI nomor HK.03.1.23.08.11.07331 Tahun 2011 Tentang Metode Analisis Kosmetika; 2011.
Wallace WE, Moorthy AS. NIST mass spectrometry data center standard reference libraries and software tools: application to seized drug analysis. J Forensic Sci. 2023 Sep 1;68(5):1484-93. doi: 10.1111/1556-4029.15284, PMID 37203286.
Carolina A, Maman M. Larvicidal activity of essential oils from the leaves and fruits of nutmeg (Myristica fragrans Houtt) against Aedes aegypti (Diptera: Culicidae). Turk J Agric-Food Sci Technol. 2016;4(7):552. doi: 10.24925/turjaf.v4i7.552-556.705.
Shafiq MI, Ahmed M, Rasul A, Samra ZQ, Qadir MA, Mazhar S. Chemical composition of the essential oils of nutmeg and mace by GC-FID/MS indigenous to Pakistan and evaluation of their biological activities. Lat Am J Pharm. 2016;35(10):2176–84. doi: 10.24327/ajpps.2016.2176.
Judzentiene A, Peciulyte D, Nedveckyte I. In situ antimicrobial properties of sabinene hydrate a secondary plant metabolite. Molecules. 2024 Sep 1;29(17):4252. doi: 10.3390/molecules29174252, PMID 39275100.
Herdiana Y. Alcohol in daily products: health risks cultural considerations and economic impacts. Risk Manag Healthc Policy. 2025;18:217-37. doi: 10.2147/RMHP.S495493, PMID 39845405.
Lachenmeier DW. Safety evaluation of topical applications of ethanol on the skin and inside the oral cavity. J Occup Med Toxicol. 2008;3:26. doi: 10.1186/1745-6673-3-26, PMID 19014531.
Suryani A, Cahyaningsih T. Formulation optimization of tomato seed oil (Solanum lycopersicum) cleansing balm by simplex lattice design: physical characteristics and anti-aging effects. J Pharm Sci Electron. 2020;8(1):391-401. doi: 10.36490/journal-jps.com.
Rachman ES, Widji Soeratri, Tristiana Erawati M. Characteristics and physical stability of nanoemulsion as a vehicle for anti-aging cosmetics: a systematic review. JFIKI. 2023;10(1):62-85. doi: 10.20473/jfiki.v10i12023.62-85.
Chakraborty P, Dey S, Parcha V, Bhattacharya SS, Ghosh A. Design expert supported mathematical optimization and predictability study of buccoadhesive pharmaceutical wafers of loratadine. BioMed Res Int. 2013;2013:197398. doi: 10.1155/2013/197398, PMID 23781498.
Nahdhia N, Agus Syamsur Rijal M, Hendradi E, Widodo RT. Application of the simplex lattice design method to determine the optimal formula of diclofenac sodium nanoemulsion. JFIKI. 2024 Aug 31;11(2):137-46. doi: 10.20473/jfiki.v11i22024.137-146.
Pavoni L, Perinelli DR, Bonacucina G, Cespi M, Palmieri GF. An overview of micro and nanoemulsions as vehicles for essential oils: formulation preparation and stability. Nanomaterials (Basel). 2020;10(1):135. doi: 10.3390/nano10010135, PMID 31940900.
Balta I, Brinzan L, Stratakos AC, Linton M, Kelly C, Pinkerton L. Geraniol and linalool loaded nanoemulsions and their antimicrobial activity. BUASVMCN-ASB. 2017;74(2):157. doi: 10.15835/buasvmcn-asb:0025.
Gupta AD, Bansal VK, Babu V, Maithil N. Chemistry antioxidant and antimicrobial potential of nutmeg (Myristica fragrans Houtt). J Genet Eng Biotechnol. 2013 Jun 1;11(1):25-31. doi: 10.1016/j.jgeb.2012.12.001.
Vardan V. Influence of pH on the stability of pharmaceutical compounds in Japan. J Chem. 2024 Aug 31;3(2):21-30. doi: 10.47672/jchem.2404.
Zdrada Nowak J, Aniolkowska S, Deska M. The evaluating skin acid–base balance after application of cold-processed and hot-processed natural soaps: a double-blind pH monitoring study. Cosmetics. 2025 Jun 1;12(3):120. doi: 10.3390/cosmetics12030120.
Preeti SS, Sambhakar S, Malik R, Bhatia S, Al Harrasi A, Rani C. Nanoemulsion: an emerging novel technology for improving the bioavailability of drugs. Scientifica. 2023;2023:6640103. doi: 10.1155/2023/6640103, PMID 37928749.
Mushtaq A, Mohd Wani S, Malik AR, Gull A, Ramniwas S, Ahmad Nayik G. Recent insights into nanoemulsions: their preparation, properties and applications. Food Chem X. 2023;18:100684. doi: 10.1016/j.fochx.2023.100684, PMID 37131847.
Modarres Gheisari SM, Gavagsaz Ghoachani R, Malaki M, Safarpour P, Zandi M. Ultrasonic nano-emulsification a review. Ultrason Sonochem. 2019;52:88-105. doi: 10.1016/j.ultsonch.2018.11.005, PMID 30482437.
Elhabal SF, Faheem AM, Hababeh S, Nelson J, Elzohairy NA, Ibrahim YF. Augmented marshmallow extract lipid nanoparticles with clove oil embedded in collagen sponge for ultimate antimicrobial healing of diabetic mouth ulcer. Pharmaceutics. 2025 May 1;17(5):611. doi: 10.3390/pharmaceutics17050611, PMID 40430902.
Sompech S, Srion A, Nuntiya A. The effect of ultrasonic treatment on the particle size and specific surface area of LaCoO3. Procedia Eng. 2012;32:1012-8. doi: 10.1016/j.proeng.2012.02.047.
Suryani A, Laksitorini MD, Sulaiman TN. Ferrous fumarate nanoliposomes: formulation characterization and diffusion profiles. J Appl Pharm Sci. 2024 May 1;14(5):157-65. doi: 10.7324/JAPS.2024.154580.
Silva AC, Sousa Lobo JM. Preclinical evaluation of lipid-based nanosystems. Pharmaceutics. 2021;13(5):708. doi: 10.3390/pharmaceutics13050708, PMID 34066100.
Zhu Y, Li Y, Wu C, Teng F, Qi B, Zhang X. Stability mechanism of two soybean protein-phosphatidylcholine nanoemulsion preparation methods from a structural perspective: a Raman spectroscopy analysis. Sci Rep. 2019 Dec 1;9(1):6985. doi: 10.1038/s41598-019-43439-5, PMID 31061497.
Chabib L, Rodli FH, Nugroho BH, Suryani A, Firmansyah F. Development of nanoliposome formulation of betacarotene using high-speed homogeniser method. Pharm Educ. 2024;24(2):1-8. doi: 10.46542/pe.2024.242.18.
Zhao S, Wang Z, Wang X, Kong B, Liu Q, Xia X. Characterization of nanoemulsions stabilized with different emulsifiers and their encapsulation efficiency for oregano essential oil: Tween 80, soybean protein isolate tea saponin and soy lecithin. Foods. 2023 Sep 1;12(17):3183. doi: 10.3390/foods12173183, PMID 37685117.
Rachmawati H, Novel MA, Nisa RM, Berlian G, Tandrasasmita OM, Rahma A. Co-delivery of curcumin-loaded nanoemulsion and Phaleria macrocarpa extract to NIH 3T3 cell for antifibrosis. J Drug Deliv Sci Technol. 2017 Jun 1;39:123-30. doi: 10.1016/j.jddst.2017.03.015.
Kumari S, Kumaraswamy RV, Choudhary RC, Sharma SS, Pal A, Raliya R. Thymol nanoemulsion exhibits potential antibacterial activity against bacterial pustule disease and growth-promotory effect on soybean. Sci Rep. 2018 Dec 1;8(1):6650. doi: 10.1038/s41598-018-24871-5, PMID 29703904.
Abdulredha MM, Siti Aslina H, Luqman CA. Overview on petroleum emulsions formation influence and demulsification treatment techniques. Arab J Chem. 2020;13(1):3403-28. doi: 10.1016/j.arabjc.2018.11.014.
Aswathanarayan JB, Vittal RR. Nanoemulsions and their potential applications in food industry. Front Sustain Food Syst. 2019;3:95. doi: 10.3389/fsufs.2019.00095.
Putra PS, Adhika DR, Genecya G, Al Madanie MS, Asri LA. Evaluation of chitosan-encapsulated lemongrass (Cymbopogon citratus) essential oil nanoemulsion for fruit edible coating. OpenNano. 2025 Jul 1;24:100246. doi: 10.1016/j.onano.2025.100246.
Algahtani MS, Ahmad MZ, Ahmad J. Investigation of factors influencing formation of nanoemulsion by spontaneous emulsification: impact on droplet size, polydispersity index and stability. Bioengineering (Basel). 2022 Aug 1;9(8):384. doi: 10.3390/bioengineering9080384, PMID 36004909.
Liu J, Fauconnier ML, Richel A, Jin Y. Preparation of active films with enhanced antioxidant and antibacterial properties by incorporating ginger essential oil nanoemulsions with xylan and PVA. Int J Biol Macromol. 2024;281:135780. doi: 10.1016/j.ijbiomac.2024.135780.