
Int J App Pharm, Vol 18, Issue 2, 2026, 9-20Review Article
EMERGING NOSE-TO-BRAIN DRUG DELIVERY STRATEGIES FOR AMYOTROPHIC LATERAL SCLEROSIS: A PROMISING FRONTIER IN NEUROTHERAPEUTICS
AKANSHA TAMBOLI, KANDUKURI SUSHMA*, KHUSHBOO KATHAROTIYA
Department of Pharmaceutics, School of Pharmacy, Faculty of Pharmacy, Parul University, Vadodara, Gujarat-391760, India
*Corresponding author: Kandukuri Sushma; *Email: sushma.pharmaceutics@gmail.com
Received: 25 Sep 2025, Revised and Accepted: 20 Jan 2026
ABSTRACT
The progressive neurodegenerative illness known as amyotrophic lateral sclerosis is characterized by the death of motor neurons, which causes paralysis, muscle atrophy, and ultimately fatal respiratory failure. Despite advances in understanding its molecular pathology, currently approved therapies such as Riluzole and Edaravone offer only modest clinical benefits, largely due to limited central nervous system exposure. The blood–brain barrier remains a major challenge for effective drug delivery to neural targets. In this context, intranasal nose-to-brain delivery has emerged as a promising non-invasive strategy to bypass the blood–brain barrier via olfactory and trigeminal neural pathways, enabling direct drug transport to the brain while reducing systemic exposure. Recent preclinical studies have explored a range of nanocarrier-based systems, including polymeric nanoparticles, lipid-based carriers, nanoemulsions, and hybrid formulations, to enhance nasal residence time, epithelial uptake, and neuronal transport. Functionalization with targeting ligands and mucoadhesive components has further improved brain targeting efficiency and therapeutic retention. However, challenges related to formulation complexity, long-term safety, scalability, and clinical translation remain unresolved. This review critically evaluates recent advances in nanocarrier-mediated nose-to-brain delivery for amyotrophic lateral sclerosis, highlighting current limitations and future directions required for successful clinical application.
Keywords: ALS (amyotrophic lateral sclerosis), Neurotherapeutics, Nose-to-brain delivery, Olfactory pathway, Nanocarriers
© 2026 The Authors.Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i2.56993 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
The disease of the central nervous system is a major health concern attracting the researcher’s interest, and it also poses a notable challenge to humans. Neurological illness is the main cause of mortality and infirmity worldwide. “In 2016, neurological diseases were the second greatest cause of mortality [9.0 million (8.8–9.4)] and the main cause of disability-adjusted life-years (DALYs) [276 million (95% UI 247–308)] worldwide.” This indicates that CNS illnesses are contributing more and more to the worldwide health challenge [1-3]. There is no well-established diagnosis and treatment for neurological diseases.
Neuroprotective agents should enter the brain to show their efficacy. The brain is the organ that controls various physiological processes of the body. It is encased in the endothelium fluid barrier, which is densely packed, and is protected by the skull. The cells of the endothelium of the microvascular barrier protect the brain from external stimuli; these barriers comprise the brain cerebrospinal fluid barrier, meningeal barrier, blood-brain barrier (BBB), and circumventricular organ barrier. The complicated mechanism of the ciliary endothelial cell controls permeability through the brain barrier [4-7].
Limited blood–brain barrier (BBB) permeability, which limits effective brain accumulation, and inadequate access to CNS-resident immune cells, is a significant obstacle in the development of neurotherapeutics [8–10]. To enhance BBB penetration, of drugs with tiny molecules that target CNS illnesses are typically made to be somewhat lipophilic and to have molecular weights of less than 400–500 Da [11]. Despite this, insufficient pharmacologically relevant BBB transport causes many candidates to fail early screening. Endothelial transporters further regulate and limit drug entrance, and only tiny lipophilic substances, such as alcohol and steroid hormones, easily cross the blood-brain barrier through transcellular processes [10, 12].
“Neurotherapeutics treats the CNS disorders-like Parkinson’s, Alzheimer’s, and ALS by targeting the drug directly to the brain or spine [13, 14].” The blood-brain barrier significantly impedes the direct targeting of a substance to the brain [15]. To bypass the BBB, researchers explore options such as intranasal (nose-to-brain) delivery, which uses the olfactory nerve or trigeminal nerve pathway [16]. This non-invasive route enables fast, targeted CNS delivery while reducing systemic exposure [17-19].
“Amyotrophic lateral sclerosis (ALS) is a deadly neurodegenerative disorder that leads to the sequential decline of motor neurons' function in the cerebrum, brainstem, and spinal cord [20].” Unfortunately, there are few or limited potential treatments that give fair clinical benefits. Potential treatment approaches are few and limited to modest clinical gains. “The first drug approved by the FDA for ALS was Riluzole.” Riluzole reduces glutamate-driven excitotoxicity. It improves patients' survival by about 2-3 mo. P-glycoprotein and high hepatic first-pass metabolism affect the advantage of Riluzole [21].
“The second approved therapy, Edaravone, is applied for ALS as a free radical scavenger, countering oxidative stress, and is proposed as another hallmark of ALS pathology.” However, herculean dosing regimens associated with long-term uncertainty, coupled with intravenous-only use, limit its clinical application [22, 23]. “Tofersen, an antisense oligonucleotide against SOD1 mutations, and AMX0035, a combination of sodium phenylbutyrate and Taurursodiol, are some newer therapies.” These treatments offer promise for a specific genetic form of ALS but have challenges in permeating the blood-brain barrier [24].
To cope with the trouble of the restricted bioavailability of the brain for certain drugs, N2B drug delivery systems have become an alternative. N2B drug delivery provides a non-intrusive route through the trigeminal or olfactory nerve pathway. This route provides direct access to the brain by bypassing the BBB [25]. Parikh and Patel et al. (2014) formulated a nanoemulsion for the direct targeting of Riluzole to the brain, which showed faster action and better brain uptake compared to the oral formulation [26]. Banerjee et al. (2023) formulated Edaravone nanoparticles to target them directly to the CNS via the intranasal route; the second drug approved for ALS treatment also showed better brain targeting and reduced oxidative damage in the ALS model [27, 28].
To overcome nasal enzyme-mediated and mucus drainage degradation in the nasal passage while enhancing drug solubility, permeability, and sustained release, advanced nanocarriers are currently being developed, including polymeric nanoparticles (e. g., PLGA, chitosan), solid lipid nanoparticles (SLNs), and thermo-responsive in situ gels [29]. Advanced neural therapies like gene therapies, neurotrophic factors, and antioxidants, which are being investigated for the treatment of ALS, are well-delivered by these platforms.
By addressing oxidative stress, neuroinflammation, mitochondrial dysfunction, and excitotoxicity all at once, these approaches offer a potent new frontier in neurotherapeutics that offers painless, targeted, and patient-compatible drug delivery that closely matches the pathophysiology of ALS [30, 31].
Literature search strategy
A comprehensive literature search was conducted in PubMed, Scopus, ScienceDirect, Google Scholar, and ClinicalTrials. gov for the period 2010–2025 using the keywords and Boolean combinations: ‘amyotrophic lateral sclerosis’, ‘ALS’, ‘nose-to-brain delivery’, ‘intranasal’, ‘nanocarrier’, ‘nanoparticle’, ‘liposome’, and ‘ALS therapy’. Only English language, peer-reviewed original research and review articles were included. Non-peer-reviewed sources, conference abstracts, and duplicate records were excluded.
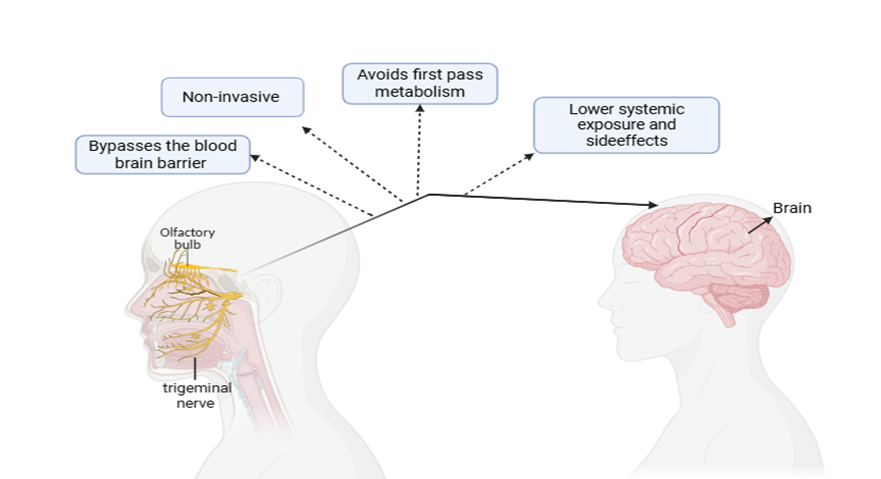
Fig. 2: Advantages of nose-to-brain drug delivery (made with BioRender)
Nose-to-brain drug delivery system
The cure of CNS disorders like amyotrophic lateral sclerosis continues to be limited by the restrictive nature of the BBB, which selectively hinders the entry of nearly 98% of small-molecule drugs and almost all biologics into the brain [55, 56]. This poses a serious problem for the efficient treatment of neurological conditions where direct drug access to the brain is necessary for therapeutic efficiency, such as amyotrophic lateral sclerosis, Alzheimer's disease, brain tumors, Parkinson's disease, and epilepsy [57, 58]. To overcome this obstacle, the nose-to-brain (N2B) targeting is a possible alternative, bypassing the BBB entirely [59, 60]. This route leverages the different anatomical and physiological characteristics of the nasal cavity, providing a straight pathway to the brain via olfactory and trigeminal neural pathways [61, 15, 62].
Despite these advantages, we need to address other physiological and formulation challenges. These include the restricted surface of the olfactory epithelium, restricted drug absorption volume, enzymatic degradation, and rapid mucosal clearance [63, 64]. Additionally, variability in nasal anatomy and physiology among patients leads to inconsistent drug delivery outcomes [65].
Advanced drug delivery technologies, including liposomes, in situ gels, mucoadhesive polymers, and nanoparticles, have been developed by researchers [66, 67]. These formulations improve nasal residence time, protect labile drugs, and improve transport across the nasal epithelium, thereby increasing CNS drug delivery efficiency [68].
With the growing prevalence of brain-related disorders and the limitations of traditional delivery systems, N2B delivery is garnering more attention as a viable, innovative platform for both biological and small molecules targeted at brain diseases [69].
Anatomy of the nose
The nostril has been split into two symmetrical halves by the nasal septum, each extending from the external nostrils to the choanae, which open into the nasopharynx [70]. “Each side is further subdivided into three turbinates (inferior, middle, and superior), which increase surface area and create turbulence for better air filtration and drug deposition [71, 72].” It is bordered superiorly by the cribriform plate, which plays a key role in the olfactory transport [68].
Each nasal passage consists of three regions
Vestibular region-It is located interior to the nostrils, lined by stratified squamous epithelium and vibrissae (coarse hairs). It provides mechanical filtration of large particles and is not suitable for drug absorption due to limited vascularization [65, 73].
Respiratory region-It engages most of the nasal epithelial surface (~90%) and borders with pseudostratified ciliated columnar epithelium and goblet cells, making it extensively vascularized due to its rich supply of capillaries, making it ideal for systemic absorption [63, 74].
Olfactory region-The olfactory region, positioned at the top of the nostril, is directly connected to the olfactory bulb via sensory neurons that traverse the cribriform plate [75]. This facilitates drug transport through intracellular (neuronal) and paracellular (and perineural) (extracellular) routes, enabling rapid and effective CNS delivery [57].
Likewise, trigeminal nerve branches in the respiratory epithelium offer a direct pathway for delivery of medication to the brain [76, 77]. These pathways collectively allow drug molecules to penetrate the brain within minutes of administration, offering a key benefit compared to conventional systemic methods [78].
Vascular and neural supply
The rich vascular network of the respiratory mucosa allows for rapid systemic absorption and avoidance of presystemic metabolism [68, 79]. Blood supply is acquired from branches of both the ophthalmic artery and the sphenopalatine, facial, and maxillary arteries, contributing to a dense capillary network [80].
Neural pathway for N2B transport
The nose connects to the brain via two major neural pathways: Olfactory nerve (cranial nerve I)-Axons in the olfactory surface, which are extended from the cribriform plate into the olfactory bulb by OSNs in the olfactory nerve. It uses neural, transcellular, and paracellular transport systems to allow medication delivery straight to the forebrain [81].
Trigeminal nerve (cranial nerve V)-Innervates both the olfactory and respiratory systems. It offers a different route to the brain stem and spinal cord, specifically for drugs targeting extensive brain structures [82].
The nostril offers an absorptive surface area of 150-200 cm², facilitated by the nasal turbinates, which increase surface folding [83].
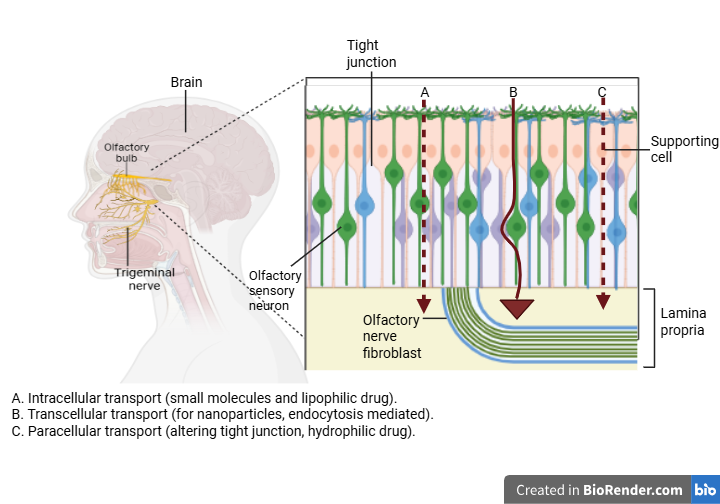
Fig. 3: Transport of a drug from nose-to-brain (made with BioRender)
Mechanism of nose-to-brain drug delivery
“Once the drug is administered intranasally, it gets deposited in the nasal cavity, particularly in the olfactory region, which is located at the top of the nasal cavity.” It begins here, then goes through the cribriform plate to join the olfactory bulb of the brain. The medication travels across the olfactory epithelium by paracellular transport. From here, the chemicals are carried intracellularly by axonal transport into the CNS [57]. This transport allows molecules to bypass liver processing and systemic circulation, leading to rapid CNS delivery. The trigeminal nerve pathway serves as a different way for drug transport; it penetrates deeper into the brain [17, 75].
Bulk flow movement of molecules is facilitated by the perineural and perivascular channels that surround the nerve, enhancing drug penetration into the brain parenchyma. Small molecules and macromolecules reach the CNS compartment by intracellular axonal transport facilitated by extracellular diffusion. Apart from neural pathways, the rich vasculature of the nasal mucosa can absorb drugs into systemic circulation, but does not guarantee efficient brain targeting and may result in side effects. So, to enhance the drug's stay time, protect the drug from enzymatic breakdown, and enhance transport efficiency through neural pathways, formulations are designed with mucoadhesive properties and nanoparticle carriers [29, 84].
This multimodal transport system offers a powerful platform for delivering neurotherapeutics directly to the brain, especially for medicines that are otherwise restricted from penetrating the BBB effectively [59].
Barriers to nasal drug delivery
Although the nasal cavity offers an appealing route for drug delivery due to its high vascularization, large surface area, and direct connection to the brain, several physiological, enzymatic, and anatomical barriers often hinder efficiency, particularly for nose-to-brain (N2B) applications.
Mucociliary clearance-There is a built-in cleaning system in our nose that acts like a moving walkway. Cilia, tiny hair-like structures, push mucus backwards through the nose and down into the throat. This mucus catches the dust, germs, and medication before clearing them out. When medicine is sprayed into the nose, this system starts working immediately. It gives the drug just about 20 min to be absorbed before it's swept away. That short window makes it harder for some medicines to fully take effect. Certain substances can change how this cleaning system works. Some help speed things up (like acetylcholine and salmeterol), while others can slow it down or stop it altogether, especially some preservatives and chemicals used in nasal products [85].
Enzymatic degradation-When drugs enter the nose, they come in contact with enzymes that break them down, especially protein-based ones like peptides. These are similar to proteases and aminopeptidases found in the nasal lining. Another challenge is that some peptides may bind to immune proteins (immunoglobulins), creating larger complexes. That makes them harder to absorb through the nasal membrane because their size reduces permeability [86].
Low membrane permeability-Low membrane permeability in the nasal cavity makes it hard for drugs, especially large and water-soluble ones, to get absorbed. Small molecules under 1000 Daltons can sometimes pass through tight junctions between cells, but larger one’s face restrictions. Enhancers may improve absorption but can also cause irritation or damage to the nasal lining, especially in humans [61].
The respiratory and olfactory epithelium-The olfactory and respiratory tissues that help access the brain’s olfactory and trigeminal pathways are situated deep in the top-back part of the nasal fossa. That area is hard to reach, even during regular breathing. The respiratory zone, which lines much of the nostrils, doesn’t directly connect to the brain and usually clears substances out through the nostrils. Because of that, drugs delivered here often have little to no chance of entering the bloodstream or reaching the central nervous system [87, 12, 88].
Low volume capacity-The nasal cavity can only hold 100–200 μl of administered volume per nostril. Larger volumes may drain out or be swallowed, affecting dosing accuracy and reducing local/systemic bioavailability [89].
Epidemiology and clinical features of ALS
“Amyotrophic lateral sclerosis is also known as Lou Gehrig’s disease.” The degeneration of motor neuronal tissue, which is crucial for voluntary motion, occurs in this disease. When they degenerate, it leads to immobility and weakness of muscles and eventually respiratory insufficiency [32, 33]. Adults between the ages of 40 and 70 are primarily affected. Males are affected more commonly than females. Normal survival of a patient after getting diagnosed with ALS is 3-5 years, which may be longer in a few patients [34].
Early clinical symptoms of ALS include fasciculations, limb weakness, and dysarthria, which further lead to loss of all voluntary function. Significantly, between 35 and 50 percent of patients experience cognitive impairment, and some go on to develop frontotemporal dementia (FTD), demonstrating that ALS is a condition that affects more than just motor degeneration [35].
Types of ALS
Sporadic ALS (sALS)-It contributes to about 90-95% of ALS cases. It is caused by complex gene-environment interactions, oxidative stress, and excitotoxicity [32].
Familial ALS (fALS)-It contributes to about 5-10% of ALS cases. It is associated with a mutation in genes such as SOD1 (superoxide dismutase 1), C9orf72 (hexanucleotide repeat expansion), FUS, and TARDBP [36, 37].
Fig. 1: Normal motor nerve, slightly degenerated motor nerve, severely degenerated motor nerve. (made with BioRender)
Table 1: Types of ALS based on site
| Type | Symptoms | Reference |
| Bulbar | Hindered Speech and swallowing ability | [38] |
| Classic | Affect limbs | |
| Predominantly upper motor neuron | Paresis | |
| Flail arm | Weakness of the upper arm | |
| Flail leg | Weakness of the lower leg | |
| Respiratory | Difficulty breathing |
Etiology (causes of ALS)
ALS is thought to result from multiple interacting factors rather than a single cause. Genetic mutations in genes such as SOD1, C9orf72, TARDBP, and FUS can impair motor neuron survival, even in individuals without a clear family history. Abnormal protein misfolding and aggregation, particularly of TDP-43, disrupt normal cellular processes. Excessive glutamate activity leads to excitotoxic neuronal damage, while oxidative stress and mitochondrial dysfunction reduce the ability of neurons to cope with metabolic stress. Persistent neuroinflammation further accelerates motor neuron loss [36]. In addition, environmental factors such as exposure to toxins, pesticides, heavy metals, smoking, and repeated physical trauma may act as triggers in genetically susceptible individuals [38, 39, 43].
Table 2: Causes of ALS
| Category | Causes |
| Genetic mutation | SOD1, C9orf72, TDP-43, FUS (Taylor et al., 2016) [36] |
| Environmental |
|
| Lifestyle-related |
|
| Cellular mechanisms |
|
Pathophysiology
Rather than having a single cause, ALS has a multifactorial pathophysiology that involves multiple overlapping cellular and molecular functions. This includes-
Glutamate excitotoxicity: Glutamate-induced excitotoxicity is the earliest and most widely accepted hypothesis in ALS pathogenesis. Glutamate gets accumulated excessively in the synaptic cleft due to a decrease in the activity of EAAT2. This excessive accumulation of glutamate triggers abnormal calcium influx, mitochondrial dysfunction, and eventually neuronal death [50, 51].
Oxidative stress: When the generation of reactive oxygen and nitrogen species surpasses the power of the organism's antioxidant defense systems, oxidative stress takes place, leading to damage to proteins, lipids, and nucleic acids. The result of a mutation in the gene SOD1 [46, 52].
Protein aggregation: Proteins such as TDP-43, FUS, or C9orf72 aggregate and interfere with RNA metabolism, proteasome function, and cell survival [48].
RNA metabolism dysfunction: Proteins like FUS and TDP-43 are RNA-binding proteins that play a role in the replication, folding, mobility, and processing of microRNA. In ALS, these proteins get mislocalized to the cytoplasm and get aggregated, disrupting mRNA splicing, axonal mRNA transport, stress granule dynamics, and nuclear cytoplasmic transport [53, 54].
Neuroinflammation: In ALS, microglia and astrocytes get activated and shift from protective to toxic phenotypes, resulting in the secretion of pro-inflammatory cytokines, nitric oxide, and ROS, which amplify neurotrauma [49].
Nanocarrier-enabled intranasal formulation strategies to overcome delivery barriers
Nanocarrier-based formulations are essential for overcoming these barriers by protecting therapeutic agents, enhancing residence time, and actively targeting the neural pathways.
Nanocarriers explored for ALS treatment
Targeted Polymeric Nanoparticles: Kurano et al. (2022) utilized the PEG-PCL-TAT polymeric nanocarrier ( To deliver N-acetyl-L-cysteine (NAC) in ALS mice. This approach leverages the TAT peptide for enhanced neuronal targeting and cellular uptake, while the polymeric core provides a sustained-release profile. The significant survival extension in mice validates the use of peptide-functionalized polymeric nanocarriers for delivering antioxidant neurotherapeutics to treat chronic ALS progression [90].
To deliver N-acetyl-L-cysteine (NAC) in ALS mice. This approach leverages the TAT peptide for enhanced neuronal targeting and cellular uptake, while the polymeric core provides a sustained-release profile. The significant survival extension in mice validates the use of peptide-functionalized polymeric nanocarriers for delivering antioxidant neurotherapeutics to treat chronic ALS progression [90].
Nanoemulsions: Parikh and Patel et al. (2016) formulated a riluzole-loaded nanoemulsion with ultra-small droplets (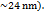 The ultra-small size and lipidic nature of nanoemulsions are a core strategy for enhancing the nasal delivery of lipophilic drugs like riluzole. In vivo studies confirmed higher brain uptake compared to oral dosing, demonstrating that this platform is promising for boosting the bioavailability of currently approved ALS drugs and circumventing their high first-pass metabolism [91].
The ultra-small size and lipidic nature of nanoemulsions are a core strategy for enhancing the nasal delivery of lipophilic drugs like riluzole. In vivo studies confirmed higher brain uptake compared to oral dosing, demonstrating that this platform is promising for boosting the bioavailability of currently approved ALS drugs and circumventing their high first-pass metabolism [91].
PLGA Nanoparticles: Lu et al. (2023) utilized poly(lactic-co-glycolic acid) (PLGA) nanoparticles (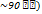 and nanocapsules of edaravone for intranasal administration. PLGA, a widely used biodegradable polymeric nanocarrier, successfully enhanced the stability and sustained release of the free radical scavenger edaravone. The nanoparticles showed superior antioxidant ability in vitro by protecting BV-2 cells against H2O induced ROS generation. This demonstrated that PLGA carriers can protect and deliver agents targeting oxidative stress, a core ALS pathology, for more efficient brain delivery than the current intravenous formulation [92].
and nanocapsules of edaravone for intranasal administration. PLGA, a widely used biodegradable polymeric nanocarrier, successfully enhanced the stability and sustained release of the free radical scavenger edaravone. The nanoparticles showed superior antioxidant ability in vitro by protecting BV-2 cells against H2O induced ROS generation. This demonstrated that PLGA carriers can protect and deliver agents targeting oxidative stress, a core ALS pathology, for more efficient brain delivery than the current intravenous formulation [92].
Dantrolene Nanoparticles: Bhuiyan et al. (2025) delivered dantrolene via nanoparticles to SOD1-G93A transgenic ALS mice. This highlights the potential of nanocarriers to deliver compounds that modulate motor neuron function, achieving therapeutic levels in the spinal cord and slowing disease degeneration, which is the primary site of motor neuron loss in ALS [93].
Extracellular Vesicles (sEVs): Zhou et al. (2024) used stem cell-derived small extracellular vesicles (sEVs) via intranasal administration. sEVs represent a highly biocompatible and naturally targeted nanocarrier capable of modulating complex disease pathways. They successfully reached the spinal cord, lowering inflammatory responses and overactive immune signaling (NF-ĸB signaling) linked to ALS progression. This is a crucial platform for delivering complex biological signaling molecules (e. g., microRNAs, proteins) that target neuroinflammation and glial dysfunction [94].
Targeted Lipid Nanoparticles (LNPs): Teixeria et al. (2022) created lactoferrin-modified LNPs loaded with riluzole (high encapsulation efficiency: 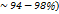 . Functionalization with lactoferrin helps the LNPs cross the BBB by targeting highly expressed receptors. This LNP strategy is essential for maximizing the delivery of drugs by implementing active targeting at the BBB-CNS interface [95].
. Functionalization with lactoferrin helps the LNPs cross the BBB by targeting highly expressed receptors. This LNP strategy is essential for maximizing the delivery of drugs by implementing active targeting at the BBB-CNS interface [95].
Silica-based Nanoparticles for Combination Therapy: Diaz-Gracia et al. (2022) used silica-based particles to deliver a combination of leptin and pioglitazone. Silica nanoparticles offer high stability and controlled release for combination therapy. This approach validates the use of nanocarriers for delivering multiple agents simultaneously to address the multifactorial nature of ALS [96].
Mucoadhesive Protein Nanohybrids: Gupta et al. (2025) developed a protein nanohybrid system using carboxylated fullerene attached to BSA protein nanoparticles. This nanohybrid strategy focuses on overcoming the mucociliary clearance barrier. The BSA component enhances drug retention, providing a sustained release profile. Which is critical for efficient neural uptake by prolonging exposure in the nasal cavity [97].
Which is critical for efficient neural uptake by prolonging exposure in the nasal cavity [97].
Table 3: Limitations of previous studies in ALS
| Study | Limitation |
| Kurano et al. (2022) | Conducted only in mice; human translation uncertain Limited data on the long-term safety of PEG-PCL-TAT nanoparticles [90]. |
| Parikh and Patel et al.(2016) | Nanoemulsion stability was tested for the short term; the long-term effects are unclear. In vivo studies are limited; there are no human trials [91]. |
| Lu et al. (2023) | Moderate drug loading (~3%) may limit the therapeutic dose. Stability was shown only for 30 days; it needs longer evaluation [92]. |
| Bhuiyan et al. (2025) | Used animal models; results may not fully predict human outcomes. Functional improvements need confirmation with larger sample sizes [93]. |
| Zhou et al. (2024) | The complexity of sEVs makes standardization and large-scale production challenging. Mechanisms of sEV action in ALS require further elucidation [94]. |
| Teixeria et al. (2022) | Lactoferrin targeting is promising but may vary in efficacy in humans. Biocompatibility tested in vitro, lacking in vivo safety data [95]. |
| Diaz-Gracia et al. (2022) | Silica nanoparticles’ long-term toxicity and clearance need assessment Combination drug effects require more extensive behavioural and survival studies [96]. |
| Gupta et al. (2025) | Nanohybrid complexity could pose manufacturing challenges. Nasal retention benefits need validation in human subjects [97]. |
Collective analysis of the limitations reported in previous ALS studies (table 3) reveals several recurring translational bottlenecks that extend beyond individual formulations or carrier systems. A predominant limitation across studies is the heavy reliance on small-animal models, with limited predictive value for human ALS pathology and virtually no validation in clinical settings. Short-duration stability assessments and modest drug-loading capacities further constrain the ability to extrapolate sustained therapeutic efficacy, particularly for chronic neurodegenerative conditions such as ALS. In addition, many advanced nanocarrier systems-including extracellular vesicles, hybrid nanostructures, and inorganic platforms-exhibit increasing formulation complexity, raising concerns related to scalability, batch-to-batch reproducibility, and regulatory feasibility. Importantly, long-term neurotoxicity, biodistribution, and clearance profiles remain insufficiently characterized across most platforms. Together, these shared limitations highlight that the primary barrier to clinical translation in ALS is not proof-of-concept efficacy, but rather the absence of long-term safety data, standardized manufacturing strategies, and human-relevant validation models.
Critical comparison of nanocarrier system for N2b delivery in ALS
Note-Several studies included in this table were conducted in non-ALS disease models. These studies are discussed to highlight transferable nanocarrier design principles relevant to ALS rather than direct therapeutic evidence.
Although diverse nanocarrier platforms demonstrate improved drug solubility, protection, and nasal uptake, the limitations summarized in table 4 reveal several overarching translational challenges for nose-to-brain delivery in ALS. First, polymeric and lipid-based nanoparticles, while biodegradable and scalable, frequently suffer from rapid mucociliary clearance and limited residence time, which restrict sustained CNS exposure unless repeated dosing is employed. Second, surface-modified systems that enhance targeting efficiency often introduce formulation complexity, increased manufacturing cost, and batch-to-batch variability, raising concerns regarding clinical scalability. Third, biologically derived carriers such as exosomes offer superior biocompatibility and intrinsic targeting potential, yet their clinical translation is hindered by stability issues, low production yield, and insufficient standardization. Finally, hybrid and inorganic systems, despite improved drug loading and synergistic therapeutic effects, raise unresolved safety concerns related to long-term accumulation and neurotoxicity. Collectively, these limitations indicate that no single nanocarrier system currently fulfills all the requirements for effective ALS therapy, underscoring the need for rational carrier selection based on therapeutic class, dosing frequency, and disease stage rather than universal platform adoption.
Future nanocarrier development for ALS should therefore prioritize disease-specific validation in ALS model, long-term safety assessment, and manufacturability alongside delivery efficiency.
Table 4: Nanocarrier strategies for nose-to-brain delivery: evidence from ALS and related CNS model with potential applicability to ALS
| Platform category | Specific studies (Ref.) | Unique features/Drug type | Observed CNS effect and relevance to ALS pathology | Major limitation |
| Polymeric Nanoparticles | Kurano et al. [90] (PEG-PCL-TAT+NAC); Lu et al. [92] (PLGA+edaravone); Bhuiyan et al. [93] (Dantrolene NPs) |
Sustained Release; Active Targeting (TAT peptide); Biodegradable polymers (PLGA). | Chronic management; targeted delivery of antioxidants (NAC) and motor-function modulators (Dantrolene) to address oxidative stress. | Potential long-term toxicity/clearance issues; moderate drug loading (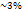 for PLGA [92]); high synthesis cost for targeted ligands. for PLGA [92]); high synthesis cost for targeted ligands. |
| Lipid Nanoparticles (LNPs)/Nanoemulsions | Parikh and Patel [91] (riluzole Nanoemulsion); Teixeria et al. [95] (Lactoferrin-LNP+riluzole) | Enhanced solubility for lipophilic drugs; receptor-mediated targeting (lactoferrin), and high encapsulation efficiency. | Maximized bioavailability of approved drugs (riluzole) by bypassing first-pass metabolism; direct targeting of the BBB interface. | Shorter stability/shelf life (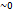 days for some nanoemulsions [91]); reliance on small droplet size. days for some nanoemulsions [91]); reliance on small droplet size. |
| Biological Carriers (Extracellular Vesicles) | Zhou et al. [94] (Stem cell-derived sEVs) | Innate Homing mechanism; superior biocompatibility; ideal for delivering complex biological cargo (miRNA/proteins). | Targets neuroinflammation and glial dysfunction; offers a systems-level approach to disease modulation. | Challenging Scalability and Standardization for clinical production [94]; risk of variable cargo and yield. |
| Hybrid/Inorganic Carriers | Diaz-Gracia et al. [96] (Silica NPs+Combination); Gupta et al. [97] (BSA/Fullerene Nanohybrids) | Mucoadhesion (BSA protein); high stability; capacity for combination therapy (Silica NPs). | Overcomes mucociliary clearance (BSA/Fullerene); synergistic drug action (Leptin/Pioglitazone) for multifactorial disease. | High manufacturing complexity (Nanohybrids) [97]; unknown long-term toxicity of inorganic components (Silica NPs) [96]. |
Current therapeutic landscape and biomarkers in ALS
Analysis of approved ALS therapeutics (table 5) reveals overarching limitations that rationalize the growing interest in alternative delivery strategies, including intranasal and nanocarrier-based approaches. All currently approved small-molecule drugs-riluzole and edaravone-exhibit modest clinical benefit and require chronic systemic administration, which is associated with limited CNS bioavailability and significant interpatient variability. Although AMX0035 targets multiple pathological pathways, its oral route remains constrained by gastrointestinal absorption and first-pass metabolism, potentially limiting consistent CNS exposure. In contrast, tofersen represents a paradigm shift toward disease-modifying therapy through direct gene targeting; however, its intrathecal administration necessitates invasive dosing and restricts patient accessibility. Collectively, these limitations highlight that current ALS therapies are constrained not only by pharmacology but also by delivery route, reinforcing the need for non-invasive, CNS-targeted delivery platforms capable of improving brain exposure while reducing systemic burden.
Table 5: Approved drugs used in ALS
| Drug name | Mechanism of action | Route | Approval | Patent no. |
| Riluzole | Glutamase release inhibitor (reduces excitotoxicity) | Oral (tablet and suspension) | FDA (1995), approved outside US. | US 5,527,814: ALS use (expired Jun 18, 2013) US8,763,150: riluzole suspension (expires Mar 12, 2029) |
| Edaravone | Free radical scavenger (reduces oxidative stress) | IV and oral | FDA, and approved outside US. | Oral-US 6,933,310: ALS treatment (expired Nov 13, 2020) US 10,987,341 and US 11,241,416 and others: oral suspension formulations (expires Nov 01, 2039) |
| AMX0035 (sodium phenylbutyrate+taurursodiol/Relyvrio) | Reduces mitochondrial and ER stress | oral | FDA (2022); marketing subsequently discontinued (2024) | Patent status complex; key composition patents expired/render commercially inactive. |
| Tofersen (Qalsody) | SOD1-targeting antisense oligonucleotide | Intrathecal | FDA | US 10,385,341: SOD1 expression modulating (Apr 1, 2035) US 10,669,546 and US 10,968,453: family patents (Apr 1, 2035) |
Table 6: Other experimental agents under approval to treat ALS. (clinicaltrials. gov)
| Drug name | Development status | Note |
| Masitinib | EMA | Tyrosine kinase inhibitor |
| Methylcobalamin | Japan | High dose vitamin B12; some benefit in early ALS |
| ION541/BIIB105 | Phase I | ASO for ATXN2 (sporadic ALS) |
| Antisense for C9orf72 | Phase I | Target repeat expansion |
| IL-2+Riluzole (MIROCALS) | Phase II | Enhances T-regs, reduces inflammation |
| SOD1-targeted+anti-inflammatory drugs | Early trials | - |
Neurofilament light chain-NfL has gained recognition as a powerful prognostic biomarker in amyotrophic lateral sclerosis (ALS). According to Lu et al. (2015), elevated levels of NfL increase the disease progression in ALS patients. This early study highlighted how measuring NfL can help predict the course of the disease, enabling better patient stratification [98]. Building on this, Benatar et al. (2024) discuss the evolving role of NfL in both research and clinical settings, emphasizing its qualification as a biomarker for ALS. This study explores how NfL monitors the effectiveness of treatment. According to this, NfL can speed up therapeutic development by providing a sensitive marker of neurodegeneration. Together, these studies establish NfL as a vital tool in ALS diagnosis, prognosis, and treatment monitoring, moving the field closer to personalized and effective interventions [99].
TDP-43-It is a protein that participates in RNA processing and has become a key focus in ALS research due to its abnormal accumulation in neurons of patients with ALS and frontotemporal lobar degeneration (FTLD). According to Feneberg et al. (2018), TDP-43 detection in cerebrospinal fluid provides a biomarker to improve diagnosis and track disease progression [100, 101]. Building on this, Gambino et al. (2023) conducted a systematic review, in which they found TDP-43’s promise as a diagnostic biomarker. Their analysis supports that altered levels of TDP-43 in patients distinguish ALS from healthy individuals, although challenges remain in standardizing measurement techniques and confirming its clinical utility [102]. Overall, TDP-43 has a strong potential as a biomarker to enhance early diagnosis and monitor ALS progression, but further research is essential to know its application in clinical practice.
Inflammatory cytokines-Inflammatory cytokines in the blood are increasingly studied as one of the major biological markers for amyotrophic lateral sclerosis (ALS) [103]. Hu Y et al. (2017) and Xu CZ et al. (2024) conducted a meta-study revealing pro-inflammatory cytokines are significantly higher in the peripheral fluid of ALS patients compared to healthy patients. This finding highlights the contribution of systemic inflammation in ALS pathology and cytokines as a potential indicator for disease activity [104, 105]. Hence, the need for a standard method to improve accuracy [106]. Together, these studies highlight that inflammatory cytokines could help track ALS progression and response to treatment, but more work is needed before they become routine clinical tools.
Metabolic biomarker-Metabolic disturbances are increasingly recognized in ALS, and Li et al. (2022) explored metabolic disruption and biomarkers that are altered in ALS patients. Their study also revealed significant changes in lipid, amino acid, and energy metabolism, suggesting that ALS involves systemic metabolic dysfunction beyond just motor neuron degeneration. Some metabolites were even associated with disease progression, highlighting their possible use in evaluating treatment response. In short, metabolic biomarkers may offer a new window into ALS pathology and therapeutic targeting, though further validation is needed for clinical use [107].
Genetic biomarker-Genetic biomarkers play a crucial role in understanding and diagnosing ALS. Calvo et al. (2012) studied transgenic SOD1G93A mice, a common ALS model, and identified several genes whose altered expression correlates with disease progression. These genes are linked to inflammation, cell death, and muscle function, providing insight into the disease mechanisms and potential early biomarkers to track ALS development [108]. Expanding on this, Pampalakis et al. (2019) reviewed the latest molecular diagnostic approaches, emphasizing advances in identifying genetic mutations and biomarkers associated with ALS. Their work highlights that mutations in genes like SOD1, C9orf72, and TARDBP (which encodes TDP-43) are key diagnostic markers. They also discuss emerging trends, such as genetic screening and multi-omics approaches, which promise earlier diagnosis and personalized treatment strategies [109]. Together, these studies underscore the importance of genetic biomarkers in improving ALS diagnosis, understanding disease biology, and guiding therapeutic development.
Challenges, research gap, and future perspectives in intranasal delivery for ALS
The clinical translation of the nose-to-brain (N2B) route for ALS is hindered by several physiological and formulation challenges inherent to the nasal cavity. Effective nanocarrier design must directly address these hurdles and fill existing research gaps to achieve consistent therapeutic drug concentrations in the central nervous system (CNS).
Table 7: Formulation and physiological challenges
| Challenge | Impact on delivery | Nanocarrier solution | Supporting studies |
| Poor Nasal Retention (Mucociliary Clearance) | Rapid clearance (within 15–20 min) reduces the absorption window, leading to insufficient drug transfer. | Mucoadhesive Strategies: Incorporate mucoadhesive polymers (e. g., chitosan, Carbopol) or use protein nanohybrids to dramatically increase residency time on the nasal mucosa, enhancing absorption [110, 111]. | Riluzole-loaded protein nanohybrid (PNH) systems, for instance, utilise Bovine Serum Albumin (BSA) and Fullerene for improved mucoadhesion and sustained release [97]. |
| Low Drug Loading/Stability | Low concentrations of therapeutic agents, particularly large biologics or small molecules with poor solubility, limit the final brain dose. | Advanced Carrier Systems: Utilise high-capacity carriers like Nanostructured Lipid Carriers (NLCs) or Solid Lipid Nanoparticles (SLNs), which offer high loading efficiency and protection against enzymatic degradation [112, 113]. | Edaravone-loaded PLGA nanocapsules demonstrated superior stability and sustained release compared to solution formulations [92]. |
| Epithelial Toxicity/Irritation | Components (e. g., permeation enhancers, solvents) can damage the sensitive nasal epithelium or cilia, leading to mucosal inflammation. | Biocompatible Components: Prioritise biodegradable, non-irritating materials (e. g., silk fibroin, PLGA, or silica) [96]and precisely control the zeta potential and particle size (ideally<200 nm) to minimize tissue interaction. | Nanocarriers utilising silica were shown to be effective for the delivery of leptin and pioglitazone without significant cytotoxicity [96]. |
| Scalability and Sterility | Complex, multi-step synthesis of highly customised nanocarriers makes large-scale manufacturing difficult and costly. | Simplified Methods: Transition from solvent-intensive methods (e. g., double emulsification) to scalable, non-toxic techniques such as spray-drying or microfluidic continuous flow for industrial production. | - |
Research gap and future perspective
The future trajectory of N2B delivery for ALS must shift from preclinical proof-of-concept to clinical translation by addressing formulation realism and regulatory requirements.
1. Translational ASO and biologic delivery
The primary translational goal is to develop a non-invasive alternative to intrathecal injections, such as that required for the ASO Tofersen (Qalsody)[24]. This necessitates engineering nanocarriers (e. g., lipid nanoparticles or polymer conjugates) with surface modifications, such as blood-brain barrier-penetrating peptides (BBBpp), to maximise CNS bioavailability [114]. The research gap here is the lack of clinical validation showing that nasally-administered ASOs achieve therapeutic concentrations in the spinal cord, a critical target for ALS. Success in this area will allow for non-invasive targeting of key genetic drivers like SOD1 or C9orf72 in the brainstem and spinal cord without the risks associated with systemic administration.
2. Device-driven precision and scale
Moving forward, research must prioritize the development of specialised metered-dose nasal devices capable of delivering precise nanocarrier doses to the high-absorption olfactory region, bypassing the respiratory region. Simultaneously, focus must be placed on developing robust, scalable manufacturing methods for these complex nanocarriers that meet Good Manufacturing Practice (GMP) standards, ensuring rapid translation from lab bench to clinical trial [115]. The research gap involves optimizing device design parameters (e. g., spray angle, velocity) to consistently target the superior turbinate/olfactory area in human subjects, as current devices primarily deposit in the lower respiratory region.
3. Validation of specific N2B pathways
A critical research gap is the lack of rigorous, quantitative comparative studies needed to map the exact proportion of drug reaching the CNS via the olfactory versus trigeminal routes in large animal models. This mechanistic clarity will allow formulators to design tailored carriers to achieve targeted drug release to specific motor neuron pools, ultimately improving the therapeutic index for ALS patients [116].
ABBREVIATIONS
| Abbreviations | Definitions |
| ALS | Amyotrophic lateral sclerosis |
| BBB | Blood-brain barrier |
| CNS | Central nervous system |
| DALYs | Disability adjusted life years |
| Da | Dalton |
| FDA | Food and Drug Administration |
| SOD1 | Superoxidase dismutase 1 |
| PLGA | Poly(lactic-co-glycolic acid) |
| SLNs | Solid-lipid nanoparticles |
| FTD | Frontotemporal dementia |
| sALS | Sporadic amyotrophic lateral sclerosis |
| fALS | Familial amyotrophic lateral sclerosis |
| C9orf72 | Chromosome 9 open reading frame 72 |
| FUS | Fused in sarcoma |
| TARDBP | Transactive response DNA-binding protein |
| EAAT2 | Excitatory amino acid transporter 2 |
| EMA | European medical agency |
| PMDA | Pharmaceutical and medical devices agency |
| IV | Intravenous |
| ER | Endoplasmic reticulum |
| NfL | Neurofilament light chain |
| FTLD | Frontotemporal lobar degeneration |
| N2B | nose to brain |
| Nm | nanometer |
| AgNPs | silver nanoparticles |
CONCLUSION
The exploration of nose-to-brain (N2B) pathways marks a significant paradigm shift in the therapeutic landscape of Amyotrophic Lateral Sclerosis (ALS). For decades, the management of this neurodegenerative pathology has been hampered by the physiological constraints of the blood-brain barrier (BBB), which effectively isolates the central nervous system from the majority of circulating pharmacological agents [117]. By circumventing this barrier through the olfactory and trigeminal nerve clusters, N2B delivery offers a strategic bypass that not only enhances drug concentration within the brain parenchyma but also mitigates the systemic side effects typically associated with high-dose oral or intravenous regimens [118].
Current evidence suggests that the success of these emerging strategies hinges on the synergy between innovative delivery routes and advanced material science. The integration of mucoadhesive nano systems and in situ gel technology represents a vital step forward in overcoming the natural limitations of nasal anatomy, such as rapid mucociliary clearance and enzymatic degradation [119]. While these neurotherapeutic frontiers offer a promising outlook for extending the functional lifespan of motor neurons, the clinical translation of these systems remains the final hurdle. Moving forward, rigorous human trials focusing on the pharmacokinetics of nasal-to-CNS transport will be essential to validate these localized therapies as a standard of care for ALS patients [120].
ACKNOWLEDGEMENT
The Authors would like to thank Parul University, Vadodara, Gujarat, India, for providing all resources and time for review. Only the authors listed in the manuscript have contributed to this review.
FUNDING
The authors declare that no funding was received from any organization or agency in support of this review.
AUTHORS CONTRIBUTIONS
Dr. Kandukuri Sushma guided this review and supported completing this review, and Dr. Khushboo Katharotiya edited some sections of the manuscript. Ms. Akansha Tamboli contributed to writing the original draft, reviewed it, and edited various sections of the manuscript.
CONFLICT OF INTERESTS
Declared none
REFERENCES
GBD 2016 Neurology Collaborators. Global regional and national burden of neurological disorders, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019 May;18(5):459-80. doi: 10.1016/S1474-4422(18)30499-X, PMID 30879893.
Wu S, Yin Y, Du L. FUS aggregation following ischemic stroke favors brain astrocyte activation through inducing excessive autophagy. Exp Neurol. 2022 Sep;355:114144. doi: 10.1016/j.expneurol.2022.114144, PMID 35718207.
Nguyen L, Hindiyeh N, Ray S, Vann RE, Aurora SK. The gut-brain connection and episodic migraine: an update. Curr Pain Headache Rep. 2023 Nov;27(11):765-74. doi: 10.1007/s11916-023-01175-6, PMID 37792173.
Patel V, Chavda V, Shah J. Nanotherapeutics in neuropathologies: obstacles challenges and recent advancements in CNS targeted drug delivery systems. Curr Neuropharmacol. 2021;19(5):693-710. doi: 10.2174/1570159X18666200807143526, PMID 32851949.
Teixido M, Giralt E. The role of peptides in blood-brain barrier nanotechnology. J Pept Sci. 2008 Feb;14(2):163-73. doi: 10.1002/psc.983, PMID 18085720.
Huang MW. The molecular pathogenesis of neuropsychiatric lupus: potential mechanisms and therapeutic targets. New York: Albert Einstein College of Medicine; 2022. doi: 10.53608/dissertation.1022.
Rakotoarisoa M, Angelov B, Garamus VM, Angelova A. Curcumin and fish oil-loaded spongosome and cubosome nanoparticles with neuroprotective potential against H2O2 induced oxidative stress in differentiated human SH-SY5Y cells. ACS Omega. 2019 Feb 12;4(2):3061-73. doi: 10.1021/acsomega.8b03101.
Nagpal K, Singh SK, Mishra DN. Drug targeting to brain: a systematic approach to study the factors parameters and approaches for prediction of permeability of drugs across BBB. Expert Opin Drug Deliv. 2013 Jul;10(7):927-55. doi: 10.1517/17425247.2013.762354, PMID 23330786.
Misra A, Ganesh S, Shahiwala A, Shah SP. Drug delivery to the central nervous system: a review. J Pharm Pharm Sci. 2003;6(2):252-73. PMID 12935438.
Naqvi S, Panghal A, Flora SJ. Nanotechnology: a promising approach for delivery of neuroprotective drugs. Front Neurosci. 2020 Jun 9;14:494. doi: 10.3389/fnins.2020.00494, PMID 32581676.
Kumar P, Dwivedi A, Plaimas K, Sagar K, Chauhan L. Nanoengineering and nanotechnology for diagnosis and treatment of CNS and neurological diseases. In: Emerging nanotechnologies for medical applications. Amsterdam: Elsevier; 2023. p. 55-94. doi: 10.1016/B978-0-323-91182-5.00006-1.
Gomes MJ, Neves JD, Sarmento B. Nanoparticle-based drug delivery to improve the efficacy of antiretroviral therapy in the central nervous system. Int J Nanomedicine. 2014 Apr 7;9:1757-69. doi: 10.2147/IJN.S45886, PMID 24741312.
Nicholls AJ. Negotiating masculinity and identity as a Jewish British Male. Cham: Springer Nature Switzerland; 2023. doi: 10.1007/978-3-031-38107-2.
Kozlovskaya L, Abou-Kaoud M, Stepensky D. Quantitative analysis of drug delivery to the brain via nasal route. J Control Release. 2014 Sep 10;189:133-40. doi: 10.1016/j.jconrel.2014.06.053, PMID 24997277.
Crowe TP, Greenlee MH, Kanthasamy AG, Hsu WH. Mechanism of intranasal drug delivery directly to the brain. Life Sci. 2018 Feb 15;195:44-52. doi: 10.1016/j.lfs.2017.12.025, PMID 29277310.
Mahajan HS, Patil PH. Gel-based delivery of neurotherapeutics via naso-brain pathways. In: Direct nose-to-brain drug delivery. London: Elsevier; 2021. p. 225-45. doi: 10.1016/B978-0-12-822522-6.00026-6.
Wang Z, Xiong G, Tsang WC, Schatzlein AG, Uchegbu IF. Nose-to-brain delivery. J Pharmacol Exp Ther. 2019 Sep;370(3):593-601. doi: 10.1124/jpet.119.258152, PMID 31126978.
Chatterjee B, Gorain B, Mohananaidu K, Sengupta P, Mandal UK, Choudhury H. Targeted drug delivery to the brain via intranasal nanoemulsion: available proof of concept and existing challenges. Int J Pharm. 2019;565:258-68. doi: 10.1016/j.ijpharm.2019.05.032, PMID 31095983.
Saccon RA, Bunton Stasyshyn RK, Fisher EM, Fratta P. Is SOD1 loss of function involved in amyotrophic lateral sclerosis? Brain. 2013 Aug;136(8):2342-58. doi: 10.1093/brain/awt097, PMID 23687121.
Zhang M, Xiang C, Niu R, He X, Luo W, Liu W. Liposomes as versatile agents for the management of traumatic and nontraumatic central nervous system disorders: drug stability targeting efficiency and safety. Neural Regen Res. 2025 Jul;20(7):1883-99. doi: 10.4103/NRR.NRR-D-24-00048, PMID 39254548.
Miller RG, Mitchell JD, Moore DH. Riluzole for amyotrophic lateral sclerosis (ALS)/motor neuron disease (MND). Cochrane Database Syst Rev. 2012;2012(3):CD001447. doi: 10.1002/14651858.CD001447.pub3, PMID 22419278.
Writing Group; Edaravone (MCI-186) ALS 19 Study Group. Safety and efficacy of edaravone in well-defined patients with amyotrophic lateral sclerosis: a randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2017 Jul;16(7):505-12. doi: 10.1016/S1474-4422(17)30115-1, PMID 28522181.
Bucklan JN, Morren JA, Shook SJ. Ultrasound in the diagnosis and management of fibular mononeuropathy. Muscle Nerve. 2019;60(5):544-8. doi: 10.1002/mus.26652, PMID 31361339.
Miller TM, Cudkowicz ME, Genge A, Shaw PJ, Sobue G, Bucelli RC. Trial of antisense oligonucleotide tofersen for SOD1 ALS. N Engl J Med. 2022 Sep 22;387(12):1099-110. doi: 10.1056/NEJMoa2204705, PMID 36129998.
Rai G, Gauba P, Dang S. Recent advances in nanotechnology for intra-nasal drug delivery and clinical applications. J Drug Deliv Sci Technol. 2023 Sep;86:104726. doi: 10.1016/j.jddst.2023.104726.
Parikh RH, Patel RJ. Nose-to-brain delivery of riluzole via nanoemulsions. Curr Drug Deliv. 2016;13(7):903-15. doi: 10.2174/1567201813666160104130026.
Banerjee A, Bandyopadhyay S, Santra S, Bose S. Edaravone-loaded nanocarriers for nasal CNS delivery. Drug Deliv Transl Res. 2023. doi: 10.1007/s13346-023-01452-9.
Lai F, Caddeo C, Manca ML, Manconi M, Sinico C, Fadda AM. What’s new in the field of phospholipid vesicular nanocarriers for skin drug delivery. Int J Pharm. 2020;583:119398. doi: 10.1016/j.ijpharm.2020.119398, PMID 32376441.
Maher R, Moreno Borrallo A, Jindal D, Mai BT, Ruiz Hernandez E, Harkin A. Intranasal polymeric and lipid-based nanocarriers for CNS drug delivery. Pharmaceutics. 2023 Mar 10;15(3):746. doi: 10.3390/pharmaceutics15030746, PMID 36986607.
Patharapankal EJ, Ajiboye AL, Mattern C, Trivedi V. Nose-to-brain (N2B) delivery: an alternative route for the delivery of biologics in the management and treatment of central nervous system disorders. Pharmaceutics. 2023 Dec 31;16(1):66. doi: 10.3390/pharmaceutics16010066, PMID 38258077.
Pawlyk AC, Cassel JA, Reitz AB. Current nervous system-related drug targets for the treatment of amyotrophic lateral sclerosis. Curr Pharm Des. 2010;16(18):2053-73. doi: 10.2174/138161210791293024, PMID 20370663.
Hardiman O, Al-Chalabi A, Chio A, Corr EM, Logroscino G, Robberecht W. Amyotrophic lateral sclerosis. Nat Rev Dis Primers. 2017 Oct 5;3:17071. doi: 10.1038/nrdp.2017.71, PMID 28980624.
Enge TG. Looking for the missing link: application of copper isotope metallomics to amyotrophic lateral sclerosis. Wollongong (AU): University of Wollongong; 2023.
Brown RH, Al-Chalabi A. Amyotrophic lateral sclerosis. N Engl J Med. 2017 Jul 13;377(2):162-72. doi: 10.1056/NEJMra1603471, PMID 28700839.
Phukan J, Elamin M, Bede P, Jordan N, Gallagher L, Byrne S. The syndrome of cognitive impairment in amyotrophic lateral sclerosis: a population-based study. J Neurol Neurosurg Psychiatry. 2012 Jan;83(1):102-8. doi: 10.1136/jnnp-2011-300188, PMID 21836033.
Taylor JP, Brown RH Jr, Cleveland DW. Decoding ALS: from genes to mechanism. Nature. 2016 Nov 10;539(7628):197-206. doi: 10.1038/nature20413, PMID 27830784.
Yamashita T, Nakano Y, Sasaki R, Tadokoro K, Omote Y, Yunoki T. Safety and clinical effects of a Muse cell-based product in patients with amyotrophic lateral sclerosis: results of a phase 2 clinical trial. Cell Transplant. 2023;32:1-9. doi: 10.1177/09636897231214370, PMID 38014622.
Kamel F, Umbach DM, Bedlack RS, Richards M, Watson M, Alavanja MC. Pesticide exposure and amyotrophic lateral sclerosis. Neurotoxicology. 2012 Jun;33(3):457-62. doi: 10.1016/j.neuro.2012.04.001, PMID 22521219.
Fang F, Kwee LC, Allen KD, Umbach DM, Ye W, Watson M. Association between blood lead and the risk of amyotrophic lateral sclerosis. Am J Epidemiol. 2010 May 15;171(10):1126-33. doi: 10.1093/aje/kwq063, PMID 20406759.
Horner RD, Grambow SC, Coffman CJ, Lindquist JH, Oddone EZ, Allen KD. Amyotrophic lateral sclerosis among 1991 Gulf War veterans: evidence for a time-limited outbreak. Neuroepidemiology. 2008;31(1):28-32. doi: 10.1159/000136648, PMID 18535397.
Berger JR, Espinosa PS, Kissel J. Brachial amyotrophic diplegia in a patient with human immunodeficiency virus infection: widening the spectrum of motor neuron diseases occurring with the human immunodeficiency virus. Arch Neurol. 2005 May;62(5):817-23. doi: 10.1001/archneur.62.5.817, PMID 15883271.
Pupillo E, Messina P, Giussani G, Logroscino G, Zoccolella S, Chio A. Physical activity and amyotrophic lateral sclerosis: a European population-based case-control study. Ann Neurol. 2014 May;75(5):708-16. doi: 10.1002/ana.24150, PMID 24706338.
Alonso A, Logroscino G, Jick SS, Hernan MA. Association of smoking with amyotrophic lateral sclerosis risk and survival in men and women: a prospective study. BMC Neurol. 2010 Jan 14;10:6. doi: 10.1186/1471-2377-10-6, PMID 20074360.
Okamoto K, Kihira T, Kondo T, Kobashi G, Washio M, Sasaki S. Lifestyle factors and risk of amyotrophic lateral sclerosis: a case-control study in Japan. Ann Epidemiol. 2009 Jun;19(6):359-64. doi: 10.1016/j.annepidem.2009.01.015, PMID 19342254.
Van Damme P, Braeken D, Callewaert G, Robberecht W, Van Den Bosch L. GluR2 deficiency accelerates motor neuron degeneration in a mouse model of amyotrophic lateral sclerosis. J Neuropathol Exp Neurol. 2005 Jul;64(7):605-12. doi: 10.1097/01.jnen.0000171647.09589.07, PMID 16042312.
Barber SC, Shaw PJ. Oxidative stress in ALS: key role in motor neuron injury and therapeutic target. Free Radic Biol Med. 2010 Mar 1;48(5):629-41. doi: 10.1016/j.freeradbiomed.2009.11.018, PMID 19969067.
Webster CP, Smith EF, Shaw PJ, De Vos KJ. Protein homeostasis in amyotrophic lateral sclerosis: therapeutic opportunities? Front Mol Neurosci. 2017 May 2;10:123. doi: 10.3389/fnmol.2017.00123, PMID 28512398.
Blokhuis AM, Groen EJ, Koppers M, Van Den Berg LH, Pasterkamp RJ. Protein aggregation in amyotrophic lateral sclerosis. Acta Neuropathol. 2013 Jun;125(6):777-94. doi: 10.1007/s00401-013-1125-6, PMID 23673820.
Philips T, Robberecht W. Neuroinflammation in amyotrophic lateral sclerosis: role of glial activation in motor neuron disease. Lancet Neurol. 2011 Mar;10(3):253-63. doi: 10.1016/S1474-4422(11)70015-1, PMID 21349440.
Rothstein TL, Wang JK, Panka DJ, Foote LC, Wang Z, Stanger B. Protection against Fas-dependent Th1-mediated apoptosis by antigen receptor engagement in B cells. Nature. 1995 Mar 9;374(6518):163-5. doi: 10.1038/374163a0, PMID 7533263.
Xie M, Pallegar PN, Parusel S, Nguyen AT, Wu LJ. Regulation of cortical hyperexcitability in amyotrophic lateral sclerosis: focusing on glial mechanisms. Mol Neurodegener. 2023 Oct 19;18(1):75. doi: 10.1186/s13024-023-00665-w, PMID 37858176.
Lu H, Zuo Y, Meng X, Ding X, Jian T, Liu Y. Phenolic profiles, antioxidant activity and inhibition of digestive enzymes of water caltrop pericarps. J Sci Food Agric. 2022 Apr;102(6):2342-51. doi: 10.1002/jsfa.11572, PMID 34625980.
Strong MJ. The evidence for altered RNA metabolism in amyotrophic lateral sclerosis (ALS). J Neurol Sci. 2010 Jan 15;288(1-2):1-12. doi: 10.1016/j.jns.2009.09.029, PMID 19840884.
Goutman SA, Hardiman O, Al-Chalabi A, Chio A, Savelieff MG, Kiernan MC. Emerging insights into the complex genetics and pathophysiology of amyotrophic lateral sclerosis. Lancet Neurol. 2022 May;21(5):465-79. doi: 10.1016/S1474-4422(21)00414-2, PMID 35334234.
Pardridge WM. Drug transport across the blood-brain barrier. J Cereb Blood Flow Metab. 2012 Nov;32(11):1959-72. doi: 10.1038/jcbfm.2012.126, PMID 22929442.
Zhang F, Wen L, Wang K, Huang Z, Jin X, Xiong R. Effect of axitinib regulating the pathological blood-brain barrier functional recovery for glioblastoma therapeutics. CNS Neurosci Ther. 2022 Mar;28(3):411-21. doi: 10.1111/cns.13788, PMID 34967104.
Lochhead JJ, Thorne RG. Intranasal delivery of biologics to the central nervous system. Adv Drug Deliv Rev. 2012 May 15;64(7):614-28. doi: 10.1016/j.addr.2011.11.002, PMID 22119441.
Moosavi SG, Rahiman N, Jaafari MR, Arabi L. Lipid nanoparticle (LNP) mediated mRNA delivery in neurodegenerative diseases. J Control Release. 2025;381:113641. doi: 10.1016/j.jconrel.2025.113641, PMID 40120689.
Hammarlund Udenaes M, De Lange EC, Thorne RG. Drug delivery to the brain: physiological concepts methodologies and approaches. Springer; 2014. doi: 10.1007/978-1-4614-9105-7.
Nguyen TT, Duong VA. Advancements in nanocarrier systems for nose-to-brain drug delivery. Pharmaceuticals (Basel). 2025 Apr 23;18(5):615. doi: 10.3390/ph18050615, PMID 40430435.
Illum L. Nasal drug delivery possibilities problems and solutions. J Control Release. 2003 Feb 21;87(1-3):187-98. doi: 10.1016/S0168-3659(02)00363-2, PMID 12618035.
Shevalkar GB, Prajapati MK, Pai RV, Mali KD. Applications of direct nose-to-brain drug delivery in medicine and pharmacy. In: Direct nose-to-brain drug delivery. London, UK: Elsevier; 2021. p. 249-66. doi: 10.1016/B978-0-12-822522-6.00011-4.
Djupesland PG. Nasal drug delivery devices: characteristics and performance in a clinical perspective-a review. Drug Deliv Transl Res. 2013 Feb;3(1):42-62. doi: 10.1007/s13346-012-0108-9, PMID 23316447.
Nadim N, Khan AA, Khan S, Parveen R, Ali J. A narrative review on potential applications of spanlastics for nose-to-brain delivery of therapeutically active agents. Adv Colloid Interface Sci. 2025 Jan;335:103341. doi: 10.1016/j.cis.2024.103341, PMID 39566150.
Gizurarson S. Anatomical and histological factors affecting intranasal drug and vaccine delivery. Curr Drug Deliv. 2012 Nov;9(6):566-82. doi: 10.2174/156720112803529828, PMID 22788696.
Ugwoke MI, Agu RU, Verbeke N, Kinget R. Nasal mucoadhesive drug delivery: background applications trends and future perspectives. Adv Drug Deliv Rev. 2005 Oct 31;57(11):1640-65. doi: 10.1016/j.addr.2005.07.009, PMID 16182408.
Costa C, Moreira JN, Amaral MH, Sousa Lobo JM, Silva AC. Nose-to-brain delivery of lipid-based nanosystems for epileptic seizures and anxiety crisis. J Control Release. 2019 Mar 10;295:187-200. doi: 10.1016/j.jconrel.2018.12.049, PMID 30610952.
Illum L. Transport of drugs from the nasal cavity to the central nervous system. Eur J Pharm Sci. 2000 Sep;11(1):1-18. doi: 10.1016/S0928-0987(00)00087-7, PMID 10913748.
Mahajan HS, Deshmukh SR. Nose to brain drug delivery system: a novel approach for brain targeting. Curr Drug Discov Technol. 2013 Sep;10(3):167-79. doi: 10.2174/1568009611310030002.
Maity KR. Targeting the trigeminal nerve system for orofacial pain treatment. Iowa City, IA: University of Iowa; 2022. doi: 10.17077/etd.006456.
Keller LA, Merkel O, Popp A. Intranasal drug delivery: opportunities and toxicologic challenges during drug development. Drug Deliv Transl Res. 2022 Apr;12(4):735-57. doi: 10.1007/s13346-020-00891-5, PMID 33491126.
Alnasser S. A review on nasal drug delivery system and its contribution in therapeutic management. Asian J Pharm Clin Res. 2019 Jan 7;12(1):40-5. doi: 10.22159/ajpcr.2019.v12i1.29443.
Mundlia J, Kumar M. Nasal drug delivery-an overview. Int J Pharm Sci Res. 2015 Mar;6(3):951-8. doi: 10.13040/IJPSR.0975-8232.6(3).951-58.
Chonkar A, Nayak U, Udupa N. Smart polymers in nasal drug delivery. Indian J Pharm Sci. 2015 Jul-Aug;77(4):367-75. doi: 10.4103/0250-474X.164770, PMID 26664051.
Zhang Y, Liu M, Wang Y, Hu D, Wu S, Zhao B. Nasal nanotherapeutics for central nervous system disorders: bridging the translational gap in central nervous system drug delivery. Eur J Pharmacol. 2025;1003:177958. doi: 10.1016/j.ejphar.2025.177958, PMID 40680978.
Chung S, Peters JM, Detyniecki K, Tatum W, Rabinowicz AL, Carrazana E. The nose has it: opportunities and challenges for intranasal drug administration for neurologic conditions, including seizure clusters. Epilepsy Behav Rep. 2023 Jan 1;21:100581. doi: 10.1016/j.ebr.2022.100581, PMID 36636458.
Johnson FO, Atchison WD. The role of environmental mercury lead and pesticide exposure in development of amyotrophic lateral sclerosis. Neurotoxicology. 2009 Sep;30(5):761-5. doi: 10.1016/j.neuro.2009.07.010, PMID 19632272.
Hanson LR, Frey WH. Intranasal delivery bypasses the blood-brain barrier to target therapeutic agents to the central nervous system and treat neurodegenerative disease. BMC Neurosci. 2008 Dec 10;9(Suppl 3):S5. doi: 10.1186/1471-2202-9-S3-S5, PMID 19091002.
Boyuklieva R, Pilicheva B. Micro and nanosized carriers for nose-to-brain drug delivery in neurodegenerative disorders. Biomedicines. 2022 Jul 14;10(7):1706. doi: 10.3390/biomedicines10071706, PMID 35885011.
Crowe TP, Hsu WH. Evaluation of recent intranasal drug delivery systems to the central nervous system. Pharmaceutics. 2022 Mar 12;14(3):629. doi: 10.3390/pharmaceutics14030629, PMID 35336004.
Sharma G, Wadhwa K, Kumar S, Singh G, Pahwa R. Revolutionizing Parkinson’s treatment: harnessing the potential of intranasal nanoemulsions for targeted therapy. Drug Deliv Transl Res. 2025;15(8):2589-607. doi: 10.1007/s13346-024-01770-z, PMID 39777646.
Johnson NJ, Hanson LR, Frey WH. Trigeminal pathways deliver a low molecular weight drug from the nose to the brain and orofacial structures. Mol Pharm. 2010 Jun 7;7(3):884-93. doi: 10.1021/mp100029t, PMID 20420446.
Kundoor V, Dalby RN. Assessment of nasal spray deposition pattern in a silicone human nose model using a color-based method. Pharm Res. 2010 Jan;27(1):30-6. doi: 10.1007/s11095-009-0002-4, PMID 19902337.
Bolon M, Fieux M, Monge C, Richard S. Polymeric nanoparticles for efficient nose-to-brain delivery. Nanoscale. 2025;17(31):17947-79. doi: 10.1039/D5NR00870K, PMID 40693481.
Dehghan MH, Gaikwad VM, Dandge B. Nasal absorption of drugs–barriers and solutions. Res J Pharm Technol. 2009;2(4):634-41. doi: 10.52711/0974-360X.
Dhakar RC, Maurya SD, Tilak VK, Gupta AK. A review on factors affecting the design of nasal drug delivery system. Int J Drug Deliv. 2011 Apr 1;3(2):194-202.
D’Souza AA, Kutlehria S, Huang D, Bleier BS, Amiji MM. Nasal delivery of nanotherapeutics for CNS diseases: challenges and opportunities. Nanomedicine (Lond). 2021 Dec;16(30):2651-5. doi: 10.2217/nnm-2021-0311, PMID 34802292.
Bharadwaj VN, Tzabazis AZ, Klukinov M, Manering NA, Yeomans DC. Intranasal administration for pain: oxytocin and other polypeptides. Pharmaceutics. 2021 Jul 16;13(7):1088. doi: 10.3390/pharmaceutics13071088, PMID 34371778.
Suman JD, Laube BL, Dalby R. Comparison of nasal deposition and clearance of aerosol generated by nebulizer and an aqueous spray pump. Pharm Res. 1999 Oct;16(10):1648-52. doi: 10.1023/A:1011933410898, PMID 10554112.
Kurano T, Kanazawa T, Iioka S, Kondo H, Kosuge Y, Suzuki T. Intranasal administration of N-acetyl-L-cysteine combined with cell-penetrating peptide-modified polymer nanomicelles as a potential therapeutic approach for amyotrophic lateral sclerosis. Pharmaceutics. 2022 Nov 24;14(12):2590. doi: 10.3390/pharmaceutics14122590, PMID 36559085.
Parikh RH, Patel RJ. Nanoemulsions for intranasal delivery of riluzole to improve brain bioavailability: formulation development and pharmacokinetic studies. Curr Drug Deliv. 2016;13(7):1130-43. doi: 10.2174/1567201813666151202195729, PMID 26638977.
Lu Y, Wang JT, Li N, Zhu X, Li Y, Bansal S. Intranasal administration of edaravone nanoparticles improves its stability and brain bioavailability. J Control Release. 2023 Jul;359:257-67. doi: 10.1016/j.jconrel.2023.06.001, PMID 37290723.
Bhuiyan P, Yi Y, Wei B, Yan A, Dong L, Wei H. Intranasal dantrolene nanoparticles for treatment of amyotrophic lateral sclerosis as a disease-modifying drug. bioRxiv. 2025:2025.05.21.655232. doi: 10.1101/2025.05.21.655232, PMID 40501612.
Zhou J, Li F, Jia B, Wu Z, Huang Z, He M. Intranasal delivery of small extracellular vesicles reduces the progress of amyotrophic lateral sclerosis and the overactivation of complement-coagulation cascade and NF-κB signaling in SOD1G93A mice. J Nanobiotechnology. 2024 Aug 22;22(1):503. doi: 10.1186/s12951-024-02764-2, PMID 39174972.
Teixeira MI, Lopes CM, Gonçalves H, Catita J, Silva AM, Rodrigues F. Formulation characterization and cytotoxicity evaluation of lactoferrin functionalized lipid nanoparticles for riluzole delivery to the brain. Pharmaceutics. 2022 Jan 13;14(1):185. doi: 10.3390/pharmaceutics14010185, PMID 35057079.
Diaz Garcia D, Ferrer Donato A, Mendez Arriaga JM, Cabrera Pinto M, Diaz-Sanchez M, Prashar S. Design of mesoporous silica nanoparticles for the treatment of amyotrophic lateral sclerosis (ALS) with a therapeutic cocktail based on leptin and pioglitazone. ACS Biomater Sci Eng. 2022 Oct 14;8(11):4838-49. doi: 10.1021/acsbiomaterials.2c00865, PMID 36240025.
Gupta U, Kumar A, Alam MI, Balaji PG, Sharma A, Yadav AK. Synthesis and characterization of protein nanohybrid systems for the brain delivery of riluzole. Ther Deliv. 2025 Jun;16(6):569-79. doi: 10.1080/20415990.2025.2478805, PMID 40125959.
Lu CH, Macdonald Wallis C, Gray E, Pearce N, Petzold A, Norgren N. Neurofilament light chain: a prognostic biomarker in amyotrophic lateral sclerosis. Neurology. 2015 Jun 2;84(22):2247-57. doi: 10.1212/WNL.0000000000001642, PMID 25934855.
Benatar M, Ostrow LW, Lewcock JW, Bennett F, Shefner J, Bowser R. Biomarker qualification for neurofilament light chain in amyotrophic lateral sclerosis: theory and practice. Ann Neurol. 2024 Feb;95(2):211-6. doi: 10.1002/ana.26860, PMID 38110839.
Feneberg E, Gray E, Ansorge O, Talbot K, Turner MR. Towards a TDP-43-based biomarker for ALS and FTLD. Mol Neurobiol. 2018 Oct;55(10):7789-801. doi: 10.1007/s12035-018-0947-6, PMID 29460270.
Jaiswal MK. TDP-43 and neurodegenerative diseases: past present and future. In: Jaiswal MK, editor. TDP-43 and neurodegeneration. London, UK: Academic Press; 2022. p. 1-26. doi: 10.1016/B978-0-323-90956-3.00010-3.
Gambino CM, Ciaccio AM, Lo Sasso B, Giglio RV, Vidali M, Agnello L. The role of TAR DNA binding protein 43 (TDP-43) as a candidate biomarker of amyotrophic lateral sclerosis: a systematic review and meta-analysis. Diagnostics (Basel). 2023 Jan 23;13(3):416. doi: 10.3390/diagnostics13030416, PMID 36766521.
Nguyen TT, Duong VA. Advancements in nanocarrier systems for nose-to-brain drug delivery. Pharmaceuticals (Basel). 2025 Apr 23;18(5):615. doi: 10.3390/ph18050615, PMID 40430435.
Hu Y, Cao C, Qin XY, Yu Y, Yuan J, Zhao Y. Increased peripheral blood inflammatory cytokine levels in amyotrophic lateral sclerosis: a meta-analysis study. Sci Rep. 2017 Aug 22;7(1):9094. doi: 10.1038/s41598-017-09097-1, PMID 28831083.
Xu CZ, Huan X, Luo SS, Zhong HH, Zhao CB, Chen Y. Serum cytokines profile changes in amyotrophic lateral sclerosis. Heliyon. 2024;10(7):e28553. doi: 10.1016/j.heliyon.2024.e28553, PMID 38596011.
Moreno Martinez L, Calvo AC, Munoz MJ, Osta R. Are circulating cytokines reliable biomarkers for amyotrophic lateral sclerosis? Int J Mol Sci. 2019 Jun 5;20(11):2759. doi: 10.3390/ijms20112759, PMID 31195629.
Li JY, Cui LY, Sun XH, Shen DC, Yang XZ, Liu Q. Alterations in metabolic biomarkers and their potential role in amyotrophic lateral sclerosis. Ann Clin Transl Neurol. 2022 Jul;9(7):1027-38. doi: 10.1002/acn3.51580, PMID 35584112.
Calvo AC, Manzano R, Atencia Cibreiro G, Olivan S, Munoz MJ, Zaragoza P. Genetic biomarkers for ALS disease in transgenic SOD1(G93A) mice. PLOS One. 2012;7(3):e32632. doi: 10.1371/journal.pone.0032632, PMID 22412900.
Pampalakis G, Mitropoulos K, Xiromerisiou G, Dardiotis E, Deretzi G, Anagnostouli M. New molecular diagnostic trends and biomarkers for amyotrophic lateral sclerosis. Hum Mutat. 2019 Apr;40(4):361-73. doi: 10.1002/humu.23697, PMID 30556231.
Kumar A, Singh S. Chitosan-coated liposomes: a mucoadhesive strategy for enhanced nose-to-brain delivery. Int J Curr Pharm Res. 2024;16(2):10-5. doi: 10.22159/ijcpr.2024v16i2.4012.
Kumar P, Dhiman A, Kumar A. Mucoadhesive nanosuspension for enhanced nose to brain delivery: a review. Int J Curr Pharm Res. 2024;16(2):1-6. doi: 10.22159/ijcpr.2024v16i2.4010.
Patel P, Shah J. Nanostructured lipid carriers: a next-generation approach for nose-to-brain targeting. Asian J Pharm Clin Res. 2023;16(5):1-7. doi: 10.22159/ajpcr.2023.v16i5.47211.
Garg D, Sharma N, Jain M, Dube A. Solid lipid nanoparticles: a promising carrier for nose-to-brain delivery. Asian J Pharm Clin Res. 2024;17(3):12-8. doi: 10.22159/ajpcr.2024.v17i3.50114.
Wang J, Lee Y, Chen M. Efficient systemic CNS delivery of a therapeutic antisense oligonucleotide with a blood-brain barrier-penetrating peptide. Mol Ther. 2023 Jan 4;31(1):164-75. doi: 10.1016/j.ymthe.2022.08.016.
Schinkel AH, Jonker JW. Mammalian drug efflux transporters of the ATP-binding cassette (ABC) family: an overview. Adv Drug Deliv Rev. 2003;55(1):3-29. doi: 10.1016/S0169-409X(02)00169-2, PMID 12535572.
Kunda NK, Vaka SR, Boddapati N. Recent advances in nose-to-brain drug delivery: mechanistic understanding and device development. Drug Discov Today. 2022 Jan;27(1):153-65. doi: 10.1016/j.drudis.2021.09.004.
Kumar R, Singh A. Clinical variations and diagnostic challenges in motor neuron diseases. Int J Pharm Pharm Sci. 2021;13(4):15-22. doi: 10.22159/ijpps.2021v13i4.40821.
Pandey V, Agnihotri A, Vishwakarma N. Nose-to-brain delivery of microcarriers in the treatment of neurodegenerative diseases: a review. J Appl Pharm Sci. 2024;14(1):1-8. doi: 10.7324/JAPS.2024.148112.
Butani S. Fabrication of an ion-sensitive in situ gel loaded with nanostructured lipid carrier for nose to brain delivery of donepezil. Asian J Pharm. 2018;12(4):293-9. doi: 10.22377/ajp.v12i04.2811.
Sharma P, Patel B. Role of oxidative stress and advanced delivery systems in neurodegenerative disorders. Asian J Pharm Clin Res. 2022;15(2):45-51. doi: 10.22159/ajpcr.2022.v15i2.43561.