
Int J App Pharm, Vol 18, Issue 2, 2026, 30-42Original Article
BIOPOLYMER-BASED EMULGEL OF KASTURI (CITRUS MICROCARPA BUNGE) ESSENTIAL OIL: STABILITY, SPF, ANTIOXIDANT, AND ANTIBACTERIAL PROPERTIES
YAHDIAN RASYADI1* , WIDA NINGSIH2
, WIDA NINGSIH2 , DHONA AFRIZA3
, DHONA AFRIZA3 , VARIZA PUJAAN4
, VARIZA PUJAAN4
1,2,4Pharmaceutical Technology Department, Clinical Pharmacy Bachelor Program, Faculty of Health Sciences, Universitas Baiturrahmah, West Sumatra, Indonesia. 3Department of Oral Biology, Faculty of Dentistry, Baiturrahmah University, Padang, West Sumatra, Indonesia
*Corresponding author: Yahdian Rasyadi; *Email: yahdian_rasyadi@fk.unbrah.ac.id
Received: 30 Sep 2025, Revised and Accepted: 31 Dec 2025
ABSTRACT
Objective: To develop a biopolymer-based emulgel of kasturi essential oil and evaluate its physical stability, photoprotection, antioxidant, and antibacterial performance.
Methods: Oil of kasturi was obtained by steam distillation and profiled by gas chromatography–mass spectrometry (GC–MS). Twelve formulations were prepared and subjected to stability, pH, viscosity, spreadability, and phase separation tests. Bioactivity assays included antioxidant activity using the 2,2-diphenyl-1-picrylhydrazyl (DPPH) method, in vitro sun protection factor (SPF) with the Mansur method, and antibacterial activity against Staphylococcus aureus using the disc diffusion method.
Results: Essential oil yield from fresh Citrus microcarpa peels obtained by non-optimised steam distillation was 0.34% w/w, which lies at the lower end of values reported for this species and other citrus peels under similar conditions; notably, optimized hydro distillation of Citrus microcarpa can afford higher yields; density 0.84 g/ml; 41 compounds were identified (dominant D-limonene). Formula 2 (F2) (sodium carboxymethyl cellulose (Na-CMC)), formula 4 (xanthan gum), and formula 12 (sodium alginate) passed accelerated tests and remained stable under freeze–thaw. Antioxidant activity (inhibitory concentration 50% (IC₅₀), ppm): kasturi oil (95.905), F2, F4, and F12 (99.255-171.834). SPF values for F2, F4, and F12 were 19.38–44.13, and kasturi oil (12.20). Antibacterial testing against Staphylococcus aureus produced small inhibition zones; F2, F4, and F12 were 2.00–4.54 mm, far below the positive control chloramphenicol (31.50 ± 0.00 mm).
Conclusion: F2, F4, and F12 were physically stable. F2 showed the highest in vitro SPF estimate, but this value is likely overestimated and requires in vivo confirmation before firm photoprotective claims can be made. All emulgel formulations exhibited a weak antibacterial effect, and the overall antioxidant activity of the oil was also very weak.
Keywords: Emulgel, Citrus microcarpa Bunge, SPF, Staphylococcus aureus, Antioxidant
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i2.57058 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Volatile essential oils offer antioxidant and antibacterial benefits for topical skincare but remain challenging to formulate due to their rapid evaporation, susceptibility to oxidation, and potential skin irritation, resulting in a short residence time and inconsistent performance [1–5]. Kasturi (Citrus microcarpa Bunge) is rich in bioactive constituents, particularly D-limonene [6–8], and shows antioxidant [9–11] and antibacterial activity [12, 13]. However, its high volatility and potential irritancy hinder direct application and necessitate an appropriate delivery strategy to maintain stability and user comfort.
Emulgels, hybrids of emulsions dispersed within a gel network, are attractive carriers because the structured matrix can immobilize oil droplets, enhance physical stability, and modulate release at the skin surface [14]. Within such systems, the choice of gelling/thickening polymers is critical to counteract evaporation, stabilise the dispersed oil phase, and preserve spreadability and sensory properties. Natural biopolymers are widely used as thickening and gelling agents in topical emulgels. Beyond building viscosity and structural integrity, they offer biocompatibility and biodegradability; families commonly explored include cellulose derivatives, alginates, and microbial gums [15].
Guided by the delivery challenges of Citrus microcarpa oil, this study employs a biopolymer network of sodium carboxymethyl cellulose (Na-CMC), xanthan gum, and sodium alginate. Na-CMC contributes film-forming and viscosity that may thicken the interfacial layer and limit evaporation; xanthan forms a shear-thinning viscoelastic network that suppresses droplet mobility and coalescence while maintaining spreadability; and alginate provides ionic-gelation–mediated reinforcement that strengthens the matrix and can help regulate release. Together, these complementary functions directly target volatility-driven loss, instability, and potential discomfort. The application of natural biopolymers as gelling agents, including sodium carboxymethyl cellulose (Na-CMC), xanthan gum, chitosan, carrageenan, tragacanth, and sodium alginate, has demonstrated effectiveness in enhancing viscosity and formulation stability [16]. Na-CMC at approximately 1–2% is commonly used in topical film-forming gels; the film-forming and UV-blocking properties of the Na-CMC matrix support selecting a level that is not too high to maintain spreadability. Xanthan at 1–1.5% is used to build viscosity and emulgel stability. Chitosan at 0.5–1% is used for topical gels because it requires protonation to dissolve/gel; this range helps maintain controlled viscosity. Carrageenan at 0.5–1% together with divalent cations (Ca²⁺) promotes helix strengthening and can enhance thermogelation (particularly for ι/κ types). Tragacanth at 1–2% serves as a natural thickener/emulsifier in topical and cosmetic formulations. Alginate at 1–2% combined with CaCl₂ initiates ionotropic gelation, reinforcing the hydrogel network [17–27]. Leveraging such biopolymers in an emulgel is expected to enhance the topical usability of volatile citrus oils by retaining the oil within a structured matrix, mitigating irritation, while maintaining practical spreadability and sensorial attributes. From a formulation development perspective, alignment with sustainability principles is also relevant; natural polymer-based systems are consistent with the current direction toward greener skincare, as long as efficacy and safety are preserved [28, 29]. Natural polymers used in topical semisolids, such as cellulose derivatives, alginates, microbial gums, and selected polysaccharides, are generally biocompatible, biodegradable, and capable of forming stable viscoelastic networks that support droplet stabilisation and controlled release in emulgel matrices [15].
Accordingly, this study specifically addresses the delivery gap for volatile Citrus microcarpa Bunge essential oil by developing and evaluating an emulgel based on Na-CMC/xanthan/alginate to test whether this polymer network can reduce evaporation, improve physical stability, and enhance topical tolerability and usability.
MATERIALS AND METHODS
Materials
The materials utilized in this research were kasturi orange essential oil (Citrus microcarpa Bunge); liquid paraffin (Brataco); tween 80 (Micromaster); span 80 (Nantong Feng Yuan Chemical); sodium carboxymethyl cellulose (Na-CMC; Arbe Chemindo); distilled water (Brataco); xanthan gum (Making Cosmetics); chitosan (CDH); glacial acetic acid (Smart Lab); carrageenan (Indo Food Chem); calcium chloride dihydrate (Smart Lab); tragacanth; sodium alginate (Himedia); 70% ethanol; ethanol pro analysi (p. a) (Merck); ethanol analytical reagent (AR) (Smart Lab); methanol (p. a.) (Merck); dimethyl sulfoxide (DMSO); glycerin (Brataco); butylated hydroxytoluene (BHT) (Advent Chembio); methyl paraben (Med Chem Express); propyl paraben (Alpha Chemica); 2,2-diphenyl-1-picrylhydrazyl (DPPH) (Smart Lab); ascorbic acid (Smart Lab); physiological natrium chloride (NaCl); McFarland 0.5 standard; mueller hinton agar (MHA); nutrient agar (NA)(Merck); Staphylococcus aureus American type culture collection (ATCC) BAA-976 PK/5; chloramphenicol; antimicrobial susceptibility test discs (Oxoid CT0998B); cotton buds; sterile cotton swabs (Onemed); sterile blue tips; tissues; cotton; aluminum foil; dish washing soap; sponge; cleaning brush; headloop mask; gloves, petri dishes; sterile paper discs; emulgel tube containers; amber glass bottles; glass plates; microscope slides; cover glasses; label paper; and stickers.
Tools
The tools utilized in this research comprised a distillation apparatus; homogenizer; oven (Memmert); ultrasonic bath (Taffware Ultrasonic 40 kHz); spectrophotometer ultraviolet-visible (UV-Vis) (Agilent Technologies Cary 8454 UV–Vis); gas chromatography–mass spectrometry (Agilent 7890B GC and 5977B MSD); viscometer (ATAGO Visco); pH meter (Orion Star A211®); centrifuge (Zenith LC-04S); digital stopwatch; biological safety cabinet (Biobase 11231BBC86); autoclave (Tomy SX-500); incubator (Thermo Scientific); micropipettes (Eppendorf; Rainin); test tubes (Iwaki Pyrex); Erlenmeyer flasks (Iwaki); volumetric flasks (Iwaki); funnels (Iwaki); graduated cylinders; beakers; filter paper (Whatman No. 42); analytical balance (KERN); watch glass; spatula; dropper pipettes; sterile inoculating loop; glass spirit lamp; and sterile forceps.
Plant species Identification
The identification of the Citrus microcarpa Bunge (kasturi) plant species utilised as research material was conducted by submitting a plant sample to Herbarium ANDA (Universitas Andalas) for species confirmation [30, 31].
Extraction of essential oil from Citrus microcarpa Bunge
Fresh Citrus microcarpa Bunge samples were obtained from Padang. The peels were separated after juice pressing, then cut into small pieces to increase the contact surface area and subsequently extracted using steam distillation. In this method, hot steam is passed through a still containing the citrus peels, volatilizing the essential oil, which is carried with the steam to the condenser. The vapour-oil mixture is condensed to a liquid and then separated into an essential oil layer and an aqueous hydrosol layer, with the essential oil collected as the final product. Steam distillation is a commonly used technique to extract essential oils from citrus peels due to its simplicity and effectiveness [8].
Calculation of essential oil yield
The isolated Citrus microcarpa Bunge essential oil was placed into vials, and subsequently, the percentage yield was determined. Yield is defined as the ratio of the mass of essential oil extracted to the mass of citrus peel utilised [32]. The yield is calculated using Equation (1).
% Yield= 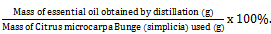 . . . .(1)
. . . .(1)
Determination of specific gravity of Citrus microcarpa bunge essential oil
The isolated Citrus microcarpa Bunge essential oil was placed into bottles, and subsequently, the percentage yield was determined. Yield is defined as the ratio of the mass of essential oil extracted to the mass of citrus peel utilised [33]. The density (specific gravity) of the peel essential oil was determined following Equation (2).
Density= 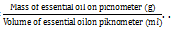 . . . (2)
. . . (2)
Analysis of chemical constituents in Citrus microcarpa bunge essential oil
The chemical composition of Citrus microcarpa Bunge peel essential oil was analysed using gas GC–MS (Agilent 7890B GC and 5977B MSD). A 1 µl** sample was injected in split mode at an injector temperature of 250 °C, with helium as the carrier gas at a constant flow rate. Separation was achieved on an Agilent capillary column, type 19091S-433 (30 m × 0.25 mm × 0.25 µm) with a 5% phenyl methyl siloxane stationary phase. The GC oven program started at 40 °C (held for 1 min), then was ramped stepwise to a final temperature of 325 °C, followed by a post-run at 300 °C. Mass detection was performed with an ion source at 220 °C, an interface/transfer line at 270 °C, and a scan range of m/z 35–500. Compound identification was carried out by matching the sample mass spectra with standard spectral libraries available in the GC–MS software [8].
Table 1: Emulgel formulation of Citrus microcarpa bunge essential oil
| Ingridients | Percentage of ingredients (%) | |||||||||||
| F1 | F2 | F3 | F4 | F5 | F6 | F7 | F8 | F9 | F10 | F11 | F12 | |
| Citrus microcarpa Bunge essential oil | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 |
| paraffin liquid | 7.5 | 7.5 | 7.5 | 7.5 | 7.5 | 7.5 | 7.5 | 7.5 | 7.5 | 7.5 | 7.5 | 7.5 |
| tween 80 | 1.2 | 1.2 | 1.2 | 1.2 | 1.2 | 1.2 | 1.2 | 1.2 | 1.2 | 1.2 | 1.2 | 1.2 |
| span 80 | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 |
| propylene glycol | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 |
| methyl paraben | 0.18 | 0.18 | 0.18 | 0.18 | 0.18 | 0.18 | 0.18 | 0.18 | 0.18 | 0.18 | 0.18 | 0.18 |
| propyl paraben | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 |
| BHT | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 |
| Na-CMC | 1 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| xanthan Gum | 0 | 0 | 0.3 | 0.6 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| chitosan | 0 | 0 | 0 | 0 | 0.5 | 1 | 0 | 0 | 0 | 0 | 0 | 0 |
| acetic acid | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 |
| carrageenan | 0 | 0 | 0 | 0 | 0 | 0 | 0.5 | 1 | 0 | 0 | 0 | 0 |
| CaCl₂ | 0 | 0 | 0 | 0 | 0 | 0 | 0.1 | 0.1 | 0 | 0 | 0.1 | 0.1 |
| tragacanth | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 2 | 0 | 0 |
| natrium alginate | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 2 |
| aquadest | ad 100 | ad 100 | ad 100 | ad 100 | ad 100 | ad 100 | ad 100 | ad 100 | ad 100 | ad 100 | ad 100 | ad 100 |
Preparation of Citrus microcarpa bunge essential oil emulgel formulations
All ingredients were weighed according to the formula specified in table 1. Biopolymer gelling agents were dissolved or dispersed in distilled water in accordance with their characteristics. Once the gel formed, the remaining aqueous phase excipients, such as tween 80, propylene glycol, methyl paraben, propyl paraben, and BHT, were added. The aqueous phase was heated to approximately 70 °C. The oil phase was developed by combining Citrus microcarpa Bunge essential oil with liquid paraffin, followed by the addition of Span 80 as the lipophilic surfactant. The oil phase was slowly incorporated into the aqueous (gel) phase with continuous stirring until a homogeneous mixture was achieved [34, 35].
Characterisation and evaluation of Citrus microcarpa bunge essential oil emulgel
Organoleptic examination
Testing was performed by observing the physical appearance of the preparation, including its form, color, and odor, as well as the resulting Citrus microcarpa Bunge essential oil emulgel [36].
Physical stability test by centrifugation
Each emulgel formula sample was placed into balanced centrifuge tubes (Zenith LC-04S) and centrifuged at 4000 rpm for 30 min at room temperature. The samples were then examined for any phase separation or changes in the physical appearance of the emulgel [37].
Physical stability test by heating
Each emulgel formula sample was placed in a loosely closed container and heated at 60 °C for 4 h using an oven (Memmert). After cooling to room temperature, the physical appearance of the emulgel was re-examined for any changes or phase separation [38].
Freeze–thaw stability test
Emulgel samples that passed the preliminary tests (F2, F4, and F12), centrifugation at 4000 rpm/30 min, and heating at 60 °C/4 h underwent a freeze–thaw stability study for six cycles, with each cycle consisting of 24 h at 4±2 °C followed by 24 h at 40±2 °C, without agitation and using the same container. At the end of each cycle, the physical appearance (presence/absence of phase separation), color, odor, and texture were documented, and pH, viscosity, and spreadability were measured at 25 °C with consistency across cycles [39]. A commercial emulgel was included as an external comparator (P′). This commercial emulgel was subjected to the same six freeze-thaw cycles, and its pH, viscosity, spreadability, and phase separation profiles were evaluated.
pH test
The pH value of each emulgel (F2, F4, F12) was determined at each stability cycle throughout a six-cycle period by utilizing a pH meter (Orion Star A211). Before usage, the pH meter was calibrated using buffer solutions at pH 4, pH 7, and pH 10. The prepared material was placed in a glass beaker, and the pH electrode was placed in the sample. The reading was captured once the measurement was stable. The reading was recorded once the measurement was stabilized [40].
Viscosity test
Viscosity (F2, F4, F12) was measured at each stability cycle over six cycles using a Brookfield viscometer (Atago®). For each formula, 15 ml of the sample was transferred into a glass beaker. Spindle A3S was attached and dragged until placed in the sample. The spindle speed was set between 20 and 100 rpm, and the viscosity value was measured using the viscometer [41].
Spreadability test
Spreadability (F2, F4, F12) was evaluated at each stability cycle over six cycles by weighed 0.5 g of emulgel and placed it at the center of a 20 × 20 cm glass plate, then covering it with a top glass plate without additional load. Spreading was allowed to occur solely due to the weight of the top plate. After 1 minute, the spread diameter was measured in two perpendicular directions, averaged, and measured in triplicate; results were reported as mean±SD [41, 42].
Antioxidant activity of the Citrus microcarpa bunge essential oil emulgel
Antioxidant testing of the emulgel (F2, F4, F12) was conducted by the DPPH method using a UV–Vis spectrophotometer (Agilent Technologies Cary 8454 UV–Vis). A 5 g sample was extracted with 10 ml methanol p. a., sonicated for 30 min, and filtered. The filtrate was diluted with methanol to obtain a range of concentrations. Then, 2 ml of each sample was mixed with 1 ml of a 200 µM DPPH solution. Mixtures were incubated for 30 min in the dark at room temperature, and absorbance was measured at 517 nm by UV–Vis spectrophotometry. Antioxidant activity was expressed as % inhibition, and the IC₅₀ value was determined by linear regression of concentration versus % inhibition [43, 44].
SPF testing
In vitro SPF testing of the emulgel (F2, F4, F12) was performed using a UV–Vis spectrophotometer (Agilent Technologies Cary 8454 UV–Vis). A 1 g sample was dissolved in 10 ml ethanol p. a., sonicated for 30 min, filtered, and diluted as needed. Absorbance was measured at 290–320 nm (5 nm intervals) with ethanol as the blank. SPF values were calculated using the Mansur equation with a correction factor (CF = 10) and the EE × I constants at each wavelength and reported as mean±SD [45].
Antibacterial activity testing
The antibacterial activity of the emulgel (F2, F4, F12) was evaluated using the disc diffusion method. Nutrient agar (NA) medium was prepared by dissolving NA powder in distilled water, heating it to boiling, and then sterilizing it. A sterile physiological solution was prepared in test tubes as the diluent for the test bacterium. Staphylococcus aureus inoculum was suspended in sterile physiological saline and adjusted to a 0.5 McFarland standard (1.5 × 10^8 CFU/ml), verified against the McFarland 0.5 reference. Sterile NA was aseptically poured into petri dishes and allowed to solidify. The test bacterial suspension was spread evenly over the agar surface with a sterile swab and left to dry for 10 min. Sterile paper discs were immersed in the emulgel samples, the positive control (chloramphenicol), and the negative control (sterile distilled water), and then placed onto the inoculated medium using sterile forceps. Plates were incubated at 37 °C for 24 h, after which inhibition zones around the discs were observed. The clear zone diameters were measured in mm using a calliper. Antibacterial activity was determined by comparing the size of the inhibition zones with those of the control [42, 46].
RESULTS
Species identification of kasturi orange
Based on the identification and verification carried out at Herbarium ANDA, Universitas Andalas, the sample used was identified as Citrus microcarpa Bunge, as stated in Identification No. 434/K-IDE/Anda/VII/2025.
Essential oil yield of kasturi
The yield of essential oil from kasturi peel obtained from the extraction process is shown in table 2.
Specific gravity of kasturi essential oil
The results of the specific gravity test of kasturi essential oil are shown in table 3.
Analysis of chemical compounds in kasturi peel essential oil
The chromatogram of the essential oil components from kasturi peel is shown in fig. 1. A total of 20 major chemical compounds were identified in the essential oil of the peel (table 4).
Table 2: Yield of kasturi peel essential oil
| Sample weight (g) | Oil weight (g) | % yield |
| 50000 | 169 | 0.34% w/w |
Table 3: Specific gravity of kasturi essential oil
| Properties | Nilai |
| Mass of kasturi oil | 4.2 g |
| Volume of kasturi oil | 5 ml |
| Density (specific gravity) of kasturi oil | 0.84 g/ml |
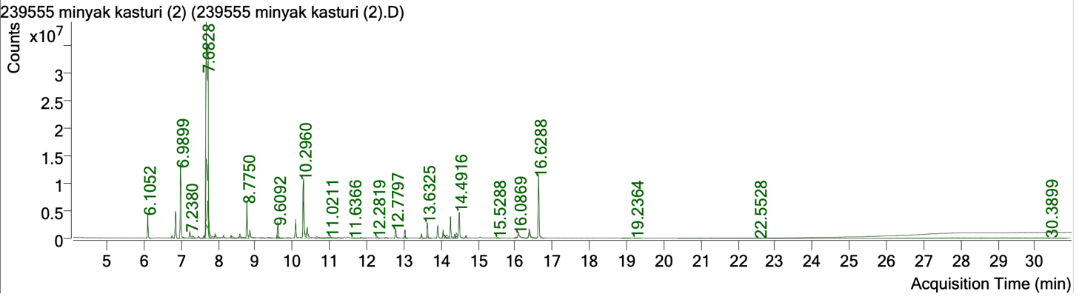
Fig. 1: GC–MS chromatogram of the chemical constituents present in Citrus microcarpa bunge essential oil
Table 4: Top 20 chemical compounds in Citrus microcarpa bunge peel essential oil identified by GC–MS
| No | Chemical compounds | RT (min) | % Area |
| 1 | D-Limonene | 7.6909 | 49.208 |
| 2 | α-Terpineol | 10.3009 | 6.443 |
| 3 | β-Myrcene | 6.996 | 6.246 |
| 4 | Patchouli alcohol | 16.6347 | 6.062 |
| 5 | Linalool | 8.781 | 3.018 |
| 6 | Bicyclo [3.1.1] heptane derivative | 6.8588 | 2.353 |
| 7 | Azulene derivative | 14.4967 | 2.720 |
| 8 | Germacrene D | 14.2627 | 1.880 |
| 9 | (1S)-2,6,6-Trimethyl bicyclo [3.1.1] hept-2-ene | 6.1097 | 1.844 |
| 10 | Bicyclo [2.2.1] heptan-2-one derivative | 9.6132 | 1.699 |
| 11 | α-Guaiene | 13.6378 | 1.421 |
| 12 | 2-(4a,8-Dimethyl-hexahydro-naphthalenyl) propan-2-ol | 16.3858 | 1.499 |
| 13 | Decanal | 10.4036 | 1.505 |
| 14 | Seychellene | 13.9187 | 1.222 |
| 15 | 2-Naphthalene methanol derivative | 16.0922 | 0.991 |
| 16 | Butanoic acid ester | 12.7846 | 0.991 |
| 17 | 4,7-Methanoazulene derivative | 13.0343 | 0.865 |
| 18 | Nonanal | 8.8597 | 0.864 |
| 19 | Cyclohexene derivative | 8.5906 | 0.458 |
| 20 | Octanal | 7.2429 | 0.644 |
Table 5: Organoleptic results of Citrus microcarpa bunge essential oil emulgel with various polymers
| No | Formula | Color | Odor | Texture | Phase separation |
| 1 | F1 | White | Characteristic scent of Citrus microcarpa Bunge essential oil | Semisolid | None |
| 2 | F2 | White | Characteristic scent of Citrus microcarpa Bunge essential oil | Semisolid | None |
| 3 | F3 | White | Characteristic scent of Citrus microcarpa Bunge essential oil | Semisolid | Separated |
| 4 | F4 | White | Characteristic scent of Citrus microcarpa Bunge essential oil | Semisolid | None |
| 5 | F5 | White | Characteristic scent of Citrus microcarpa Bunge essential oil | Semisolid | Separated |
| 6 | F6 | White | Characteristic scent of Citrus microcarpa Bunge essential oil | Semisolid | Separated |
| 7 | F7 | White | Characteristic scent of Citrus microcarpa Bunge essential oil | Semisolid, slightly thickened | None |
| 8 | F8 | White | Characteristic scent of Citrus microcarpa Bunge essential oil | Semisolid, slightly thickened | None |
| 9 | F9 | White | Characteristic scent of Citrus microcarpa Bunge essential oil | Semisolid | Separated |
| 10 | F10 | White | Characteristic scent of Citrus microcarpa Bunge essential oil | Semisolid | Separated |
| 11 | F11 | Whitish-yellow | Characteristic scent of Citrus microcarpa Bunge essential oil | Semisolid | Separated |
| 12 | F12 | Whitish-yellow | Characteristic scent of Citrus microcarpa Bunge essential oil | Semisolid | None |
Results of characterization and evaluation of the Citrus microcarpa bunge essential oil emulgel
Organoleptic examination results
Based on organoleptic observations, all formulations produced a semisolid preparation, as shown in table 5 and fig. 2.
Results of physical stability by centrifugation
From the centrifugation stability test, stable formulations were obtained for F2, F4, F8, and F12. The physical observations following centrifugation are presented in table 6 and illustrated in fig. 3 below.
Physical stability test by heating
From the heating stability test at 60 °C for 4 h, stable formulations were obtained for F1, F2, F4, F8, and F12. The stability outcomes for all formulas are shown in table 7 and fig. 4 below.
Results of freeze–thaw stability testing for the Citrus microcarpa bunge essential oil emulgel
The Citrus microcarpa Bunge essential oil emulgel formulations F2, F4, and F12 were subjected to a freeze–thaw stability test for six cycles. Organoleptic appearance, pH, viscosity, spreadability, and phase separation were observed with the following outcomes:
Organoleptic results
Organoleptic observations of the Citrus microcarpa Bunge essential oil emulgels F2, F4, and F12 are presented in fig. 5 and table 8 below.
Summary of pH, Viscosity, and spreadability test results for the Citrus microcarpa bunge emulgel
In addition to the three selected formulations (F2, F4, and F12), a commercial emulgel base (P′) was evaluated as an external comparator. The comparator underwent identical freeze–thaw procedures to allow direct comparison of changes in pH, viscosity, spreadability, and physical stability. Test Results are shown in table 9 and fig. 6-7 below.

Fig. 2: Citrus microcarpa bunge essential oil emulgel formulations with various polymers
Table 6: Centrifugation test results of the emulgel
| No | Formula | Result |
| 1 | F1 | Phase separation |
| 2 | F2 | No phase separation |
| 3 | F3 | Phase separation |
| 4 | F4 | No phase separation |
| 5 | F5 | Phase separation |
| 6 | F6 | Phase separation |
| 7 | F7 | Phase separation |
| 8 | F8 | No phase separation |
| 9 | F9 | Phase separation |
| 10 | F10 | Phase separation |
| 11 | F11 | Phase separation |
| 12 | F12 | No phase separation |
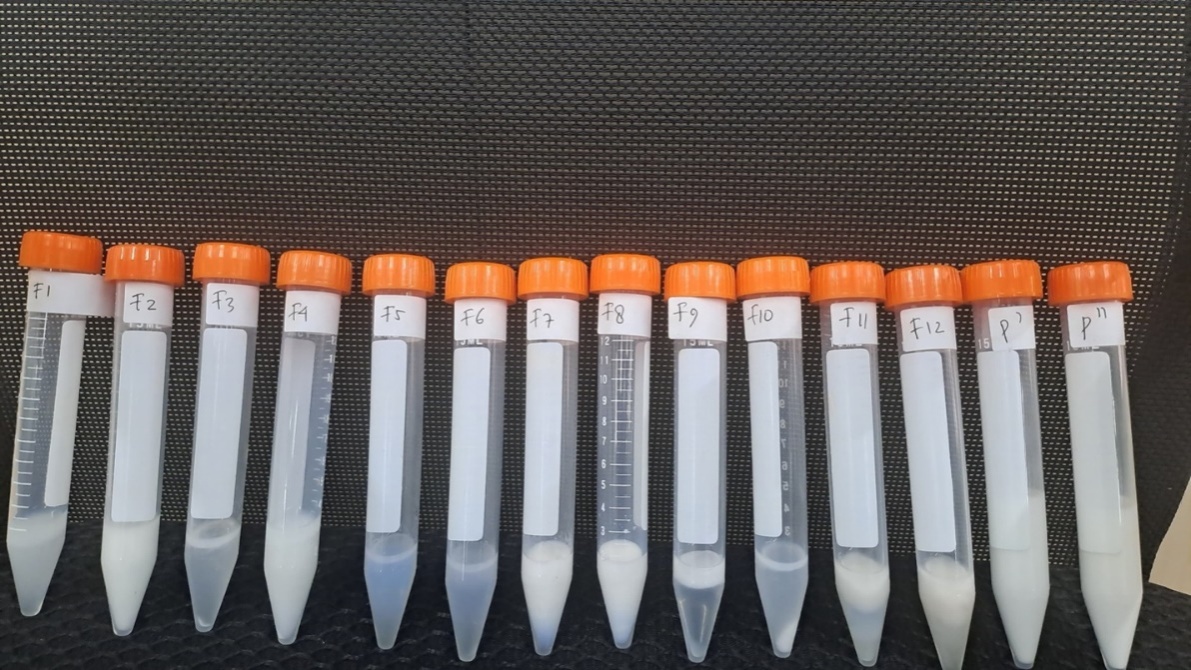
Fig. 3: Citrus microcarpa bunge essential oil emulgel formulations after centrifugation
Table 7: Heating method, physical stability results of the emulgel
| No | Formula | Result |
| 1 | F1 | No phase separation |
| 2 | F2 | No phase separation |
| 3 | F3 | Unstable |
| 4 | F4 | No phase separation |
| 5 | F5 | Unstable |
| 6 | F6 | Unstable |
| 7 | F7 | Unstable |
| 8 | F8 | No phase separation |
| 9 | F9 | Unstable |
| 10 | F10 | Phase separation |
| 11 | F11 | Phase separation |
| 12 | F12 | No phase separation |

Fig. 4: Citrus microcarpa bunge essential oil emulgel formulations after heating at 60 °C for 4 h

Fig. 5: Citrus microcarpa bunge essential oil emulgel formulations F2, F4, and F12 after six freeze–thaw cycles
Table 8: Organoleptic observations after 6-cycle stability test
| No | Cycle(s) | Formula | Color | Odor | Texture |
| 1 | 1–6 | F2 | White | Characteristic Citrus microcarpa Bunge essential oil odor | Semisolid |
| 2 | 1–6 | F4 | White | Characteristic Citrus microcarpa Bunge essential oil odor | Semisolid |
| 3 | 1–6 | F12 | Whitish-yellow | Characteristic Citrus microcarpa Bunge essential oil odor | Semisolid |
Table 9: pH, viscosity, and spreadability of emulgel after 6-cycle stability test
| Parameter | Formula | Cycle 1 (Mean±SD) | Cycle 6 (Mean±SD) | % Change (C6 vs C1) |
| pH | F2 | 7.14±0.06 | 7.09±0.03 | 0.70% |
| F4 | 6.95±0.03 | 6.83±0.03 | 1.73% | |
| F12 | 6.85±0.05 | 6.74±0.04 | 0.61% | |
| P′ | 6.50±0.02 | 6.46±0.01 | 0.62% | |
| Viscosity (cps) | F2 | 5577±25 | 5500±20 | 1.38% |
| F4 | 4810±35 | 4700±20 | 2.29% | |
| F12 | 4277±25 | 4180±20 | 2.27% | |
| P′ | 6000±20 | 5940±5 | 1.00% | |
| Spreadability (cm) | F2 | 5.50±0.10 | 5.23±0.06 | 4.91% |
| F4 | 5.03±0.06 | 4.87±0.06 | 3.18% | |
| F12 | 4.80±0.10 | 4.53±0.06 | 5.62% | |
| P′ | 6.03±0.06 | 5.77±0.06 | 4.31% |
Data expressed as mean±SD, (n=3)
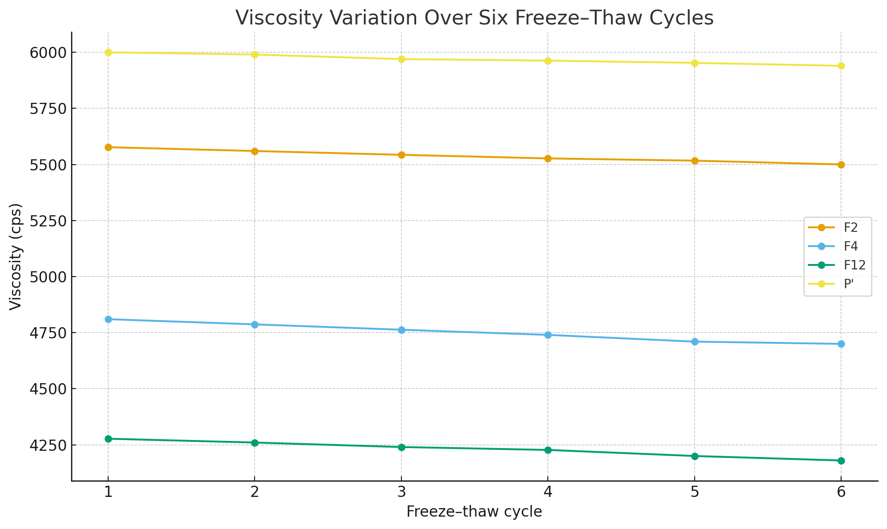
Fig. 6: Chart of emulgels viscosity variation over six freeze-thaw cycles
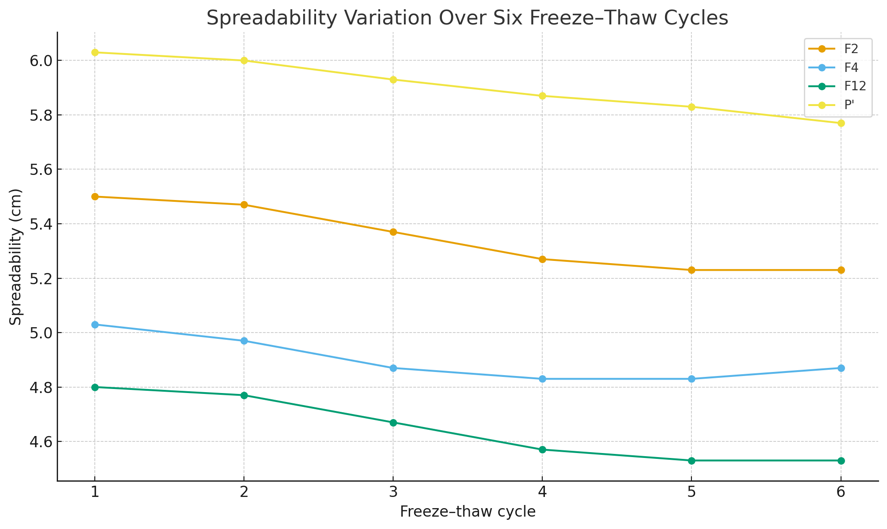
Fig. 7: Chart of emulsified spreadability variation over six freeze-thaw cycles
Paired t-tests with α = 0.05 revealed statistically significant differences between Cycle 1 and Cycle 6 for some formulations. For pH, significant changes were observed in F4 and F12 (p<0.05), whereas F2 and P′ did not differ significantly. For viscosity, all formulations (F2, F4, F12, and P′) showed statistically significant differences (p<0.05). For spreadability, F4 and P′ presented significant reductions (p<0.05), while F2 and F12 did not.
Phase-separation observations of the Citrus microcarpa bunge essential oil emulgel phase-separation observations of the emulgel are shown in table 10 below.
Table 10: Phase-separation observation results
| Cycle | F2 | F4 | F12 | P’ |
| 1 | No separation | No separation | No separation | No separation |
| 2 | No separation | No separation | No separation | No separation |
| 3 | No separation | No separation | No separation | No separation |
| 4 | No separation | No separation | No separation | No separation |
| 5 | No separation | No separation | No separation | No separation |
| 6 | No separation | No separation | No separation | No separation |
Antioxidant testing of the Citrus microcarpa bunge essential oil emulgel
Antioxidant testing of the Citrus microcarpa Bunge essential oil emulgel F2, F4, and F12 by the DPPH method using a UV–Vis spectrophotometer (Agilent Technologies Cary 8454 UV–Vis) yielded IC₅₀ values as shown in table 11 below.
SPF testing
In vitro SPF testing of the Citrus microcarpa Bunge essential oil emulgel F2, F4, and F12 was performed using a UV–Vis spectrophotometer (Agilent Technologies Cary 8454 UV–Vis) with an SPF 50 reference preparation. The SPF values obtained are presented in table 12 below.
Table 11: Results of phase-separation observations of the Citrus microcarpa bunge essential oil emulgel
| No | Sample/Formula | IC₅₀ (ppm) |
| 1 | Vitamin C | 3.89 |
| 2 | Citrus microcarpa Bunge essential oil | 95.905 |
| 3 | F2 | 171.834 |
| 4 | F4 | 105.132 |
| 5 | F12 | 99.255 |
Data expressed as mean, (n=3)
Table 12: Calculated SPF values of the Citrus microcarpa bunge essential oil emulgel
| No | Formula/Sample | SPF±SD |
| 1 | F2 | 44.13±0.21 |
| 2 | F4 | 19.38±0.10 |
| 3 | F12 | 19.58±0.08 |
| 4 | Citrus microcarpa Bunge essential oil | 12.20±0.10 |
Data are expressed as mean±SD, (n=3)
Antibacterial activity testing
Disc‑diffusion assays against Staphylococcus aureus showed small zones of inhibition for all emulgels (F2 4.54 ± 2.79 mm; F4 3.06 ± 2.86 mm; F12 2.00 ± 0.00 mm) and for the essential oil (4.33 ± 1.04 mm), while the positive control (chloramphenicol) yielded 31.50 ± 0.00 mm. Accordingly, the antibacterial activity of our samples should be interpreted as weak under the present test conditions. Antibacterial testing of the Citrus microcarpa Bunge essential oil emulgel F2, F4, and F12 against Staphylococcus aureus produced inhibition zones as presented in table 13 below.
Table 13: Inhibition-zone diameters of the Citrus microcarpa bunge essential oil emulgel against Staphylococcus aureus
| No | Formula/Sample | mean Inhibition Zone±SD (mm) |
| 1 | F2 | 4.54±2.79 |
| 2 | F4 | 3.06±2.86 |
| 3 | F12 | 2.00±0.00 |
| 4 | Citrus microcarpa Bunge essential oil | 4.33±1.04 |
| 5 | Chloramphenicol | 31.50±0.00 |
| 6 | Distilled water | 0.00±0.00 |
Data are expressed as mean±SD, (n=3)
DISCUSSION
Based on the determination conducted at Herbarium ANDA, Universitas Andalas, the kasturi plant sample used was identified as Citrus microcarpa Bunge according to Identification No. 434/K-IDE/Anda/VII/2025. The extraction of kasturi peel essential oil yielded 0.34% w/w. A yield of 0.34% w/w can be categorized as good for extraction from fresh peel without specific process optimization. Another study reported a yield of 2.45%, with higher values generally achieved after optimizing the water-to-material ratio, time, and distillation temperature. Yield can be further improved by optimizing particle size, water-to-material ratio, and distillation time–temperature parameters [47–49].
Testing showed a specific gravity of 0.84 g/ml. This value is suitable for Citrus microcarpa Bunge peel essential oil and is consistent with reports for Citrus microcarpa Bunge, which range around 0.85 g/ml [8]. In general, citrus peel oils are lighter than water (1.00 g/ml), so they float during hydrodistillation separation. Thus, the result of 0.84 g/ml is acceptable and supports the validity of the extraction process [50].
The GC–MS chromatogram (fig. 1) and the 41 identified compounds in table 4 indicate that the peel essential oil is primarily composed of hydrocarbon terpenes and oxygenated terpenes, including limonene (D-limonene), linalool/trans-linalool/cis-linalool oxide, α-pinene/β-myrcene, terpinen-4-ol/α-terpineol, as well as various sesquiterpenes (bergamotene, germacrene, seychellene) and additional oxygenated constituents such as camphor and carvone. Citrus peel components enhance antibacterial activity (via membrane disruption), antioxidant activity (by oxygenated/aromatic ring compounds), and photoprotective/SPF potential. Terpinen-4-ol exhibits activity against Staphylococcus aureus by inhibiting and destroying biofilms (MIC around 0.08–0.32% v/v) through interfering with purine/pyrimidine production and membrane permeability, thus underscoring the significance of terpinen-4-ol/α-terpineol in antibacterial efficacy. Linalool demonstrates efficacy against MRSA, as validated by metabolomics techniques, while D-limonene exhibits effectiveness that is enhanced when encapsulated or nano-emulsified. The presence of linalool, terpinen-4-ol, α-terpineol, and D-limonene in the current kasturi oil certainly contributes to the inhibitory zones against Staphylococcus aureus, suggesting that emulgels containing this oil may possess antibacterial properties [51–53]. Kasturi peel oil, containing α-pinene, α-terpineol, and 1,8-cineole, shows compounds with antioxidant and antibacterial effects [54, 55]. Essential oils (especially those rich in phenolics/coumarins) can display in vitro UV absorption and may serve as photoprotective agents [56–58]. In the present study, D-limonene eluted at 7.69 min under our GC–MS conditions. This retention time falls within the range reported in other chromatographic studies, although absolute RT values differ substantially depending on the temperature program, column characteristics, and carrier gas settings. For instance, limonen was eluted at approximately 7.5 min in a chromatographic assay [59]. Microbial volatile oil content profiling reported limonene at 12.19 min in a 20-minute GC–MS run [60]. Such variation is expected because RT is not a fixed physicochemical constant; rather, it is highly dependent on the instrumental conditions used.
All formulas (F1–F12) showed semisolid consistency and the characteristic kasturi odor; F1–F10 were white, while F11–F12 were whitish-yellow (use of sodium alginate, which is naturally off-white/yellowish). Organoleptic parameters, such as color, odor, homogeneity, and consistency, are recommended as initial quality tests for emulgels [35, 61]. Of the resulting preparations, F1, F2, F4, and F12 showed good physical appearance, whereas F3, F5, F6, F9, F10, and F11 exhibited phase separation. F7 and F8 were slightly gelled/solidified like agar, making them difficult to pour and spread. An ideal emulgel is pseudoplastic, remaining stable at rest but exhibiting shear-thinning properties upon application, which can be achieved by adjusting the level of gelling agents or adding cosolvents and humectants [62].
Centrifugation is used as an accelerated stress test to predict the tendency for phase separation during storage. Centrifugal force magnifies gravitational effects so that creaming/coalescence appears sooner in unstable systems [63, 64]. Many emulgel studies assess stability by the presence/absence of separation after centrifugation cycles [65–68]. Here, stable formulas were F2 (Na-CMC base), F4 (xanthan gum base), F8 (carrageenan base), and F12 (sodium alginate base). Increased matrix viscosity and the formation of a hydrogel network that traps emulsion droplets reduce droplet mobility and delay coalescence/creaming during centrifugation [66].
Heating at 60 °C for 4 h induces thermal stress to evaluate resistance to coalescence, flocculation, and creaming, which are typically triggered by increased thermal energy. Under these conditions, a higher temperature reduces the viscosity of the continuous phase and increases the frequency of droplet collisions. Formulas that remain stable typically have a sufficiently strong, viscoelastic gel network, which limits droplet mobility and better protects the interfacial film. The results show that F1–F2 (Na-CMC base), F4 (xanthan gum base), and F12 (sodium alginate base) remained stable, indicating that these three anionic polysaccharides successfully increased continuous-phase viscosity and provided adequate stabilization to maintain the emulgel under short-term heat exposure, consistent with the role of gelling agents in raising continuous-phase viscosity and directly improving emulsion-gel physical stability [69, 70].
Following stable centrifugation and heating, formulas F2, F4, and F12 underwent freeze–thaw stability testing, with observations made on organoleptics, pH, viscosity, spreadability, and phase separation. Selecting only three formulas, F2 (Na-CMC base), F4 (xanthan gum base), and F12 (sodium alginate base) for organoleptic testing indicates they had adequate physical robustness in prior stability tests and were suitable for further evaluation. Across cycles 1–6, no changes in color, odor, or texture were detected: F2 and F4 remained white; F12 remained whitish-yellow; all retained the characteristic essential-oil odor and semisolid consistency [71].
The instability of chitosan-based formulations (F5–F6) can be attributed to the physicochemical behavior of protonated chitosan in acidic media. Chitosan becomes strongly cationic in 1% acetic acid, and its chains can adsorb onto oppositely charged or weakly charged droplet surfaces, inducing polymer bridging flocculation. This well-known mechanism accelerates coalescence under thermal or mechanical stress [27]. In addition, chitosan hydrogels at 0.5–1% exhibit insufficient yield stress to immobilize high internal-phase emulsions containing more than 10% oil, especially [25]. These mechanisms explain the rapid phase separation observed in F5–F6 under centrifugation and heating.
Similarly, formulations based on tragacanth (F9–F10) exhibited early-phase separation. Although tragacanth is anionic, its gel network is relatively loose and weakly viscoelastic in the absence of co-gelling ions or other structuring agents, resulting in only a low to medium viscosity and inadequate droplet immobilization. The presence of 5% propyleneglycol, which can partially depress the hydration and viscosity of polysaccharide networks, may further reduce the continuous-phase elasticity necessary to stabilize emulsions [72]. These properties are consistent with the phase separation observed in F9 and F10.
The excessive thickening in carrageenan formulations (F7–F8) is expected because carrageenan gelation is strongly promoted by divalent cations such as Ca²⁺, which induce helix-to-helix aggregation and strong 3-D network formation [20]. With 0.1% CaCl₂ added, even 0.5–1% carrageenan forms rigid, high-modulus gels, explaining the “agar-like” texture and poor spreadability observed. This phenomenon is widely documented, particularly for κ-carrageenan, which becomes markedly stiffer in the presence of Ca²⁺ [19, 21]. In contrast, Na-CMC (F2), xanthan gum (F4), and sodium alginate (F12) produced stable systems because these anionic polysaccharides form highly hydrated, viscoelastic networks that maintain adequate yield stress and continuous-phase viscosity, thereby suppressing droplet mobility [35,69]. Xanthan gum exhibits strong shear-thinning behavior and forms stable gel structures, whereas alginate forms cohesive hydrogels even in the absence of divalent cations. These mechanisms explain the absence of phase separation during centrifugation, heating, and freeze–thaw cycling.
Across six cycles, pH decreased only slightly and not meaningfully for all three formulas: F2 from 7.14±0.06 (cycle 1) to 7.09±0.03 (cycle 6); F4 from 6.95±0.03 to 6.83±0.03; F12 from 6.85±0.05 to 6.74±0.04. The control/comparator (P′) also decreased marginally from 6.50 (cycle 1) to 6.46 (cycle 6), reinforcing that pH changes were uniform across preparations. Such negligible changes indicate the preparations remained stable after six cycles [73, 74]. After six cycles, viscosity showed only minor, non-meaningful decreases: F2 from 5577 cps (cycle 1) to 5500 cps (cycle 6); F4 from 4810 cps to 4700 cps; F12 from 4277 cps to 4180 cps, while control P′ decreased from 6000 cps to 5940 cps. These small changes through cycle 6 reflect good rheological stability under stress cycling, as the emulgels did not exhibit sharp viscosity drops indicative of structural damage during temperature cycling [73]. Over six cycles, spreadability decreased slightly and not meaningfully for all three formulas: F2 from 5.50±0.10 cm (cycle 1) to 5.23±0.06 cm (cycle 6); F4 from 5.03±0.06 to 4.87±0.06 cm; F12 from 4.80±0.10 to 4.53±0.06 cm; control P′ from 6.03 cm to 5.77 cm. This indicates good physical endurance under stress cycling with no sign of rheological failure [75]. Across all six cycles, there was no phase separation in any formula F2 (Na-CMC base), F4 (xanthan gum base), F12 (Na-alginate base), or in the comparator control. Rheologically, the absence of phase separation indicates good stability [76, 77].
The freeze–thaw stability evaluation of the developed emulgel formulations revealed only minimal changes in pH, viscosity, and spreadability after six cycles, supporting their physical robustness. The importance of pH, viscosity, and spreadability in topical emulgel systems has been well-documented. Stable pH values of emulgel contributed to maintaining formulation integrity during storage [78]. Emulgel systems should be characterized for their rheology and spreadability, as well as pH, to ensure stability and performance [79].
In our study, although paired t-tests revealed statistically significant differences for certain formulations (e. g., viscosity for all formulations, pH for specific formulas). However, despite statistical significance, the absolute percentage changes remained small (≤ 1.73% for pH, ≤ 2.29% for viscosity, and ≤ 6.48% for spreadability), indicating that the formulations maintained acceptable physical characteristics throughout the six freeze–thaw cycles.
This size of change is consistent with the acceptable variability found in other topical formulations; for example, in a comparative study of polymer-based emulgels, slight changes in spreadability and viscosity did not compromise product performance [80]. Additionally, the study on emulgel formulation emphasized that while topical systems may undergo minor rheological changes during storage, as long as macroscopic stability (i. e., no phase separation, no syneresis) is maintained and key parameters remain within acceptable limits, the formulation can be considered stable. Moreover, the inverse relationship between viscosity and spreadability is well recognized: as viscosity decreases slightly, spreadability may increase slightly, enhancing application ease without impairing structural integrity. Our data reflect this trend: viscosity is reduced by up to 2%, whereas spreadability varies by up to 6%. This behaviour is consistent with formulations designed to achieve optimal topical delivery and good patient acceptability. Taken together, although statistical significance was detected, the practical pharmaceutical relevance of the changes is negligible. Therefore, the emulgel formulations investigated can be regarded as physically stable under freeze-thaw conditions, supporting their suitability for topical application and further development [81].
Monoterpenes (linalool, α-terpineol, terpinen-4-ol) contribute to radical scavenging in the DPPH assay [55]. Antioxidant activity correlates with the content of active compounds in the emulgel. Although the oil content in F2, F4, and F12 is the same, the polymer matrix affects the availability of reactive antioxidants toward DPPH. Xanthan gum (F4) and alginate (F12) form hydrogel networks that are relatively more open/porous for diffusion of small molecules; thus, lipophilic antioxidants (terpinen-4-ol, α-terpineol, some linalool) can more readily diffuse from droplets and interact with DPPH, giving lower IC₅₀ (better activity) [82–85].
The DPPH results showed the following order of antioxidant strength (smaller IC₅₀ = stronger): Vitamin C (control) = 3.89 (very strong); kasturi essential oil 95.905 ppm (very weak); F12 99.255 ppm (very weak); F4 105.133 ppm (very weak); F2 171.834 ppm (very weak). Relative to Vitamin C, IC₅₀ values were higher by 24.654× (oil), 25.515× (F12), 27.026× (F4), and 44.173× (F2). Thus, absolute antioxidant activity is negligible [86]. Therefore, interpretation of the results was focused primarily on the relative performance among the three stable emulgels, where F12 exhibited the lowest IC₅₀, followed by F4 and F2. Any potential explanation relating these differences to polymer-matrix effects, such as variations in polymer network structure, viscosity, or antioxidant availability, should be regarded as hypothetical, since no release or diffusion studies were conducted. Such mechanisms may influence the accessibility of antioxidants to the DPPH medium; however, this cannot be confirmed from the present data. Accordingly, causative claims have been avoided, and these interpretations are presented only as tentative considerations that warrant further investigation through dedicated release-profile studies.
In vitro SPF (UV–Vis, spectrophotometric) of the kasturi emulgels F2, F4, and F12 showed the following order from highest SPF: F2, SPF 44.13±0.21; F12, SPF 19.58±0.08; F4, SPF 19.38±0.10; neat essential oil, SPF 12.20±0.10. In general, spectrophotometric in vitro SPF reflects total UVB absorption at 290–320 nm [87]. Citrus essential oils (including kasturi) are dominated by hydrocarbon monoterpenes like D-limonene, with oxygenated fractions (linalool, α-terpineol, terpinen-4-ol). Chromophorically, monoterpenes lack strong UVB-absorbing groups, so the SPF of neat oil tends to be low–moderate; photoprotective contribution comes more from oxygenated minor components and antioxidant activity (limiting ROS/photodegradation). This is also reflected by the neat oil SPF of 12.2 and the increase after formulation into an emulgel matrix (F2, F4, F12) [58, 88]. CMC is known to form films that increase surface layer thickness, thereby enhancing optical path length and scattering, which effectively increases absorbance in the UVB range without the addition of chromophores. Several studies have demonstrated that CMC-based films possess UV-blocking properties. When combined with particulates or other actives, UV transmittance decreases, and the SPF of CMC-containing[89]. Although xanthan and alginate provide good rheological stability, hydrophilic thickeners do not always raise SPF if their film-forming function is weak or the formed layer is insufficient; conversely, polymers with stronger film-forming ability (or combined with film formers) tend to boost SPF. The SPF increase for emulgels compared with neat oil (SPF 12.2) a rises from gel matrix effects (enhanced optical interaction+stabilization of actives). F2 (Na-CMC base) yielded the highest SPF (44) due to a more effective film in absorbing/scattering UV. Polymer choice thus affects not only rheology, but also photoprotection via film forming and microlayer formation [90, 91]. The Mansur equation is widely used for rapid in vitro SPF screening, but it has known limitations. The method assumes a clear, homogeneous solution, whereas emulgels are turbid and heterogeneous, generating light scattering that artificially increases absorbance and inflates SPF values. Additionally, the formulations in this study do not contain recognized UV filters; thus, the unusually high SPF estimate for F2 likely reflects optical scattering and film-forming effects, rather than true UVB absorption. Because of these limitations, the SPF values reported here, especially for F2, should be regarded as tentative pending confirmation using in vivo methods.
Antibacterial testing yielded inhibition zones as follows: chloramphenicol (positive control), 31.5 mm; essential oil, 4.33±1.04 mm; F2, 4.54±2.79 mm; F4, 3.06±2.86 mm; F12, 2.00±0 mm; aquadest (negative control), 0 mm. The oil composition, dominated by D-limonene with contributions from monoterpenes such as terpinen-4-ol, α-terpineol, and linalool, can inhibit the growth of Staphylococcus aureus [92]. Among the emulgels, antibacterial activity was greatest for F2 (Na-CMC base), followed by F4 (xanthan base), and lowest for F12 (Na-alginate base). The highly hydrophilic, swellable CMC matrix tends to wet the agar surface, facilitating the early release of essential oil components and producing relatively larger inhibition zones. In contrast, the more cohesive/compact xanthan and alginate networks may retain actives longer, reducing the released amount and thus the inhibition zone [93–95]. The small inhibition zones relative to chloramphenicol indicate weak antibacterial activity in the disc diffusion format. The relatively large standard deviations (e. g., F2±2.79 mm) suggest limited reproducibility, which is plausibly influenced by viscosity-limited diffusion of actives from the emulgel matrix and technical variability during disc impregnation/drying. We have therefore refrained from over-interpreting the antibacterial data and emphasize the need for a quantitative broth microdilution MIC of the neat essential oil to establish intrinsic activity independent of matrix effects [96].
CONCLUSION
This study successfully developed a stable Citrus microcarpa Bunge essential oil emulsion gel with potential applications in skin care. From 12 formula candidates, F2, F4, and F12 were found to be physically stable. F2 showed the highest in vitro SPF estimate; however, this value is likely overestimated and requires in vivo confirmation before firm photoprotective claims can be made. All emulgel formulations indicated a weak antibacterial effect, and the antioxidant activity of the oil and all emulgel formulations was very weak overall (DPPH IC₅₀ = 95.905–171.834 ppm). Further formulation optimization is warranted to enhance bioactivity while preserving stability.
ACKNOWLEDGMENT
The authors thank the Ministry of Higher Education, Science, and Technology of the Indonesian Republic for funding support through the Regular Fundamental Research scheme under contract number 006/lL10/Dt. 05.00/PL/2025, dated 02 June 2025, and 023/UM/Kontrak-Unbrah/PL/VI/2025, dated 03 June 2025. This research was made possible by the facilities and scientific/technical support provided by Universitas Baiturrahmah and the Advanced Chemical Characterisation Laboratory at the National Research and Innovation Agency (BRIN).
AUTHORS CONTRIBUTIONS
Conceptualisation: Y. R., D. A., W. N.; Methodology: Y. R., D. A., W. N., V. P.; Investigation: Y. R., D. A., W. N., V. P.; Formal analysis: Y. R., D. A.; Writing original draft: Y. R., D. A., W. N.; Writing review and editing: Y. R.; Supervision: D. A. All authors have read and approved the final version of the manuscript.
CONFLICT OF INTERESTS
Declare none
REFERENCES
Rasyadi Y. Aktivitas antioksidan handbody lotion ekstrak etanol daun sirsak (Annona muricata Linn.) dengan metode DPPH [antioxidant activity of handbody lotion from ethanol extract of soursop leaves (Annona muricata Linn.) using the DPPH method]. Parapemikir. 2022 Apr 27;11(2):169. doi: 10.30591/pjif.v11i2.3442.
Vijayakumar R, Abd Gani SS, Mohd Mokhtar NF. Anti-elastase, anti-collagenase and antimicrobial activities of the underutilized red pitaya peel: an in vitro study for anti-aging applications. Asian J Pharm Clin Res. 2017 Aug 1;10(8):251-5. doi: 10.22159/ajpcr.2017.v10i8.19048.
Ahmed AA, Nath B. Formulation and in vitro evaluation of poly-herbal anti-aging face cream of Coriandrum sativum and rose hip oil. Int J Curr Pharm Sci. 2017 Jul 14;9(4):75-8. doi: 10.22159/ijcpr.2017v9i4.20961.
Maru AD, Lahoti SR. Formulation and evaluation of moisturizing cream containing sunflower wax. Int J Pharm Pharm Sci. 2018 Nov 1;10(11):54-9. doi: 10.22159/ijpps.2018v10i11.28645.
Mukul S, Surabhi K, Atul N. Cosmeceuticals for the skin: an overview. Asian J Pharm Clin Res. 2011;4(2):1-6.
Palma CE, Cruz PS, Cruz DT, Bugayong AM, Castillo AL. Chemical composition and cytotoxicity of Philippine calamansi essential oil. Ind Crops Prod. 2019 Feb 1;128:108-14. doi: 10.1016/j.indcrop.2018.11.010.
Husni E, Yeni F, Dachriyanus. Chemical contents profile of essential oil from calamansi (Citrus microcarpa Bunge) peels and leaves and its antibacterial activities. Advances in Health Sciences Research. 2021;40:314-22. doi: 10.2991/ahsr.k.211105.046.
Rasyadi Y, Ningsih W, Mulya WP, Hanifa D. Kasturi orange peel (Citrus microcarpa Bunge) essential oil: chemical profile formulation as roll-on aromatherapy and its evaluation. Int J App Pharm. 2024 Feb 15;16(1):126-31. doi: 10.22159/ijap.2024.v16s1.28.
Husni E, Hamidi D, Pavvellin D, Hidayah H, Syafri S. Metabolite profiling antioxidant and in vitro wound healing activities of Citrus medica L. and Citrus x microcarpa Bunge peels and leaves essential oils. Prosp Pharm Sci. 2024 Dec 26;22(4):122-30. doi: 10.56782/pps.250.
Venkatachalam K, Charoenphun N, Srean P, Yuvanatemiya V, Pipatpanukul C, Pakeechai K. Phytochemicals bioactive properties and commercial potential of calamondin (Citrofortunella microcarpa) fruits: a review. Molecules. 2023 Apr 12;28(8):3401. doi: 10.3390/molecules28083401, PMID 37110643.
Othman HI, Alkatib HH, Zaid A, Sasidharan S, Rahiman SS, Lee TP. Phytochemical composition antioxidant and antiproliferative activities of Citrus hystrix Citrus limon Citrus pyriformis and Citrus microcarpa leaf essential oils against human cervical cancer cell line. Plants (Basel). 2022 Dec 27;12(1):134. doi: 10.3390/plants12010134, PMID 36616263.
Septama AW, Yuandani Y, Khairunnisa NA, Nasution HR, Utami DS, Kristiana R. Antibacterial bacteriolytic antibiofilm and synergistic effects of the peel oils of Citrus microcarpa and Citrus x amblycarpa with tetracycline against foodborne Escherichia coli. Lett Appl Microbiol. 2023 Nov 1;76(11):ovad126. doi: 10.1093/lambio/ovad126, PMID 37898554.
Roanisca O, Mahardika RG. Citrus x microcarpa bunge fruit extract as antibacterial against Staphylococcus aureus. IOP Conf Ser: Earth Environ Sci. 2020 Nov 1;599(1):012043. doi: 10.1088/1755-1315/599/1/012043.
Sharma K, Bhatti S, Bhateja P, Goyal A, Malik N, Piplani M. Emulgel: new potential for enhanced topical medication delivery. Int J Pharm Sci. 2025;3(4):9312–35. doi: 10.5281/zenodo.15227917.
Shergujri DA, Khanday MA, Noor A, Adnan M, Arif I, Raza SN. Next-generation biopolymer gels: innovations in drug delivery and theranostics. J Mater Chem B. 2025;13(10):3222-44. doi: 10.1039/D4TB02068E, PMID 39903271.
Jimenez Gomez CP, Cecilia JA. Chitosan: a natural biopolymer with a wide and varied range of applications. Molecules. 2020 Sep 1;25(17):3981. doi: 10.3390/molecules25173981, PMID 32882899.
Adipramana I, Bakar A, Afriza D. Organoleptic and antifungal test of Euchema spinosum carrageenan hydrogel mouthwash against Candida albicans. B-Dent. 2024 Jul 15;11(1):37-48. doi: 10.33854/jbd.v11i1.1543.
Cheng Z, Xiang M, Qiao D, Zhao S, Pi X, Zhang B. Incorporating κ-carrageenan regulates the gel properties and structural characteristics of corn starch-soy protein isolate based ternary system. Food Hydrocoll. 2025 Feb;159:110654. doi: 10.1016/j.foodhyd.2024.110654.
Joy R, Vigneshkumar PN, John F, George J. Hydrogels based on carrageenan. In: Plant and algal hydrogels for drug delivery and regenerative medicine. Elsevier; 2021. p. 293-325. doi: 10.1016/B978-0-12-821649-1.00003-9.
Pacheco Quito EM, Ruiz Caro R, Veiga MD. Carrageenan: drug delivery systems and other biomedical applications. Mar Drugs. 2020 Nov 23;18(11):583. doi: 10.3390/md18110583, PMID 33238488.
Anggraini J, Lo D. Health impact of carrageenan and its application in food industry: a review. IOP Conf Ser: Earth Environ Sci. 2023 Apr 1;1169(1):012098. doi: 10.1088/1755-1315/1169/1/012098.
Nannar AR, Tamboli AA, Shendge RS. Review on pharmaceutical gelling agents. PSBJ. 2024;8(1):1-11. doi: 10.23880/psbj-16000266.
Geonzon LC, Descallar FB, Du L, Bacabac RG, Matsukawa S. Gelation mechanism and network structure in gels of carrageenans and their mixtures viewed at different length scales a review. Food Hydrocoll. 2020 Nov;108:106039. doi: 10.1016/j.foodhyd.2020.106039.
Umar S, Ningsih W, Meliana M. Formulation of mucoadhesive ketoprofen granule using chitosan polymer. J Sains Far Klin. 2015 Feb 6;1(1):48. doi: 10.29208/jsfk.2014.1.1.11.
Das A, Ghosh S, Pramanik N. Chitosan biopolymer and its composites: processing properties and applications a comprehensive review. Hybrid Adv. 2024 Aug;6:100265. doi: 10.1016/j.hybadv.2024.100265.
Blebea NM, Puscasu C, Vlad RA, Hancu G. Chitosan-based gel development: extraction gelation mechanisms and biomedical applications. Gels. 2025 Apr 6;11(4):275. doi: 10.3390/gels11040275, PMID 40277711.
Hong F, Qiu P, Wang Y, Ren P, Liu J, Zhao J. Chitosan-based hydrogels: from preparation to applications a review. Food Chem X. 2024 Mar;21:101095. doi: 10.1016/j.fochx.2023.101095, PMID 38268840.
Yihan W, Jinjin D, Yingqi W, Guanai M, Xiwu Z. Advances in plant essential oils and drug delivery systems for skincare. Front Pharmacol. 2025 Apr 17;16:1578280. doi: 10.3389/fphar.2025.1578280, PMID 40313613.
Nguyen TN, Huynh TN, Tran VT, Dang CH, Hoang TK, Nguyen TD. Physicochemical characterization and bioactivity evaluation of essential oils from Citrus microcarpa Bunge leaf and flower. J Essent Oil Res. 2018 Jul 4;30(4):285-92. doi: 10.1080/10412905.2018.1435428.
Husni E, Ismed F, Awaliana A AA. Standardization extracts and simplicia of limau Sundai peel (Citrus x aurantiifolia sundai) determine content of nobiletin and antibacterial activity test. Pharmacogn J. 2021 Nov 13;13(6):1323-31. doi: 10.5530/pj.2021.13.167.
Efendi MR, Bakhtiar A, Rusdi MS, Putra DP. Comparative study of antibacterial activity of Elephantopus scaber Linn. and Elephantopus mollis Kunth. extract. Curr Appl Sci Technol. 2023 Nov 9:e0258350. doi: 10.55003/cast.2023.258350.
Samadi M, Zainal Abidin Z, Yoshida H, Yunus R, Awang Biak DR. Towards higher oil yield and quality of essential oil extracted from Aquilaria malaccensis wood via the subcritical technique. Molecules. 2020 Sep 1;25(17):3872. doi: 10.3390/molecules25173872, PMID 32858782.
Ifeanyichukwu E. Determining the physicochemical properties of lemongrass oil extract for suitability in various applications. JNFP. 2023;6(2):1-6. doi: 10.31579/2637-8914/117.
Akram MA, Khan BA, Khan MK, Alqahtani A, Alshahrani SM, Hosny KM. Fabrication and characterization of polymeric pharmaceutical emulgel co-loaded with eugenol and linalool for the treatment of Trichophyton rubrum infections. Polymers (Basel). 2021 Nov 11;13(22):3904. doi: 10.3390/polym13223904, PMID 34833203.
Malavi S, Kumbhar P, Manjappa A, Chopade S, Patil O, Kataria U. Topical emulgel: basic considerations in development and advanced research. Indian J Pharm Sci. 2022;84(5)1105-15. doi: 10.36468/pharmaceutical-sciences.1005.
Agapin JS. Physico-chemical characterization of essential oil from peel and leaf of dalanghita (Citrus nobilis). SSRN Journal. 2017 May 1;4(2):1-13. doi: 10.2139/ssrn.3624873.
Arianto A, Cindy C. Preparation and evaluation of sunflower oil nanoemulsion as a sunscreen. Open Access Maced J Med Sci. 2019 Nov 14;7(22):3757-61. doi: 10.3889/oamjms.2019.497, PMID 32127969.
Xie W, Tang C, Zhang Y, Fan W, Qin J, Xiao H. Effect of stigmasterol and polyglycerol polyricinoleate concentrations on the preparation and properties of rapeseed oil-based gel emulsions. Food Chem X. 2024 Oct;23:101636. doi: 10.1016/j.fochx.2024.101636, PMID 39113734.
Subbalakshmi AR, Sahoo S, Manjunatha P, Goyal S, Kasiviswanathan VA, Mahesh Y. The ELF3 transcription factor is associated with an epithelial phenotype and represses epithelial-mesenchymal transition. J Biol Eng. 2023;17(1):17. doi: 10.1186/s13036-023-00333-z, PMID 36864480.
Sedlarikova J, Dolezalova M, Egner P, Pavlackova J, Krejci J, Rudolf O. Effect of oregano and marjoram essential oils on the physical and antimicrobial properties of chitosan based systems. Int J Polym Sci. 2017 Jun 20;2017:1-12. doi: 10.1155/2017/2593863.
Farouk A, Hathout AS, Amer MM, Hussain OA, Fouzy AS. The impact of nanoencapsulation on volatile constituents of Citrus sinesis L. essential oil and their antifungal activity. Egypt J Chem. 2022 Mar 1;65(3):519-30.
Pagano C, Ceccarini MR, Marinelli A, Imbriano A, Beccari T, Primavilla S. Development and characterization of an emulgel based on a snail slime useful for dermatological applications. Int J Pharm. 2024 Jul;660:124337. doi: 10.1016/j.ijpharm.2024.124337, PMID 38885774.
Refilda R, Gustira D, Yefrida Y, Hanifa D. Edible coating effect on physicochemical properties of banana (Musa sapientum L.) during storage. IOP Conf Ser: Earth Environ Sci. 2024 Jun 1;1356(1):012017. doi: 10.1088/1755-1315/1356/1/012017.
Suena NM, Rahayu LR, Wijaya SC, Antari NP, Adrianta KA. Formulation and antioxidant activity test of Centella asiatica herba extract and Moringa oleifera leaves extract as an anti-aging emulgel. Trop J Nat Prod Res. 2024 Mar 30;8(3):6525-36.
Almeida WA, Antunes AS, Penido RG, Correa HS, Nascimento AM, Andrade AL. Photoprotective activity and increase of SPF in sunscreen formulation using lyophilized red propolis extracts from Alagoas. Rev Bras Farmacogn. 2019 May;29(3):373-80. doi: 10.1016/j.bjp.2019.02.003.
Fitriyasti B, Ferilda S, Sari W, Saputra MR, Khong HY. In vitro antibacterial activity of seribu kuman leaf (Rhinacanthus nasutus (I.) Kurz) extracts against Staphylococcus aureus and Pseudomonas aeruginosa. Integr Biomed Res. 2024;8(1):1-7. doi: 10.25163/angiotherapy.819433.
Tran KN, Ngo CQ, Tran BL, To PM, Huynh PX, Pham TV. Hydrodistillation of essential oil from peels of orange (Citrus sinensis) in the mekong delta vietnam: process optimization and chemical profiling. Food Res. 2023 Dec 27;7(6):272-7. doi: 10.26656/fr.2017.7(6).816.
Quyen NT, Ngan TT, Dao TP, Quynh Anh PN, Anh NQ, Nguyen Thi NT. Essential oil hydrodistillation process from vietnamese calamondin (Citrus microcarpa) peels and GC/MS analysis of essential oils components. Asian J Chem. 2019 Oct 12;31(11):2585-8. doi: 10.14233/ajchem.2019.22148.
Andrade MA, Barbosa CH, Shah MA, Ahmad N, Vilarinho F, Khwaldia K. Citrus by-products: valuable source of bioactive compounds for food applications. Antioxidants (Basel). 2022 Dec 25;12(1):38. doi: 10.3390/antiox12010038, PMID 36670900.
Bozova B, Golukcu M, Tokgoz H, Turgut DY, Cınar O, Turgutoglu E. The physico-chemical characteristics of peel essential oils of sweet orange with respect to cultivars harvesting times and isolation methods. AIMSAGRI. 2025;10(1):40-57. doi: 10.3934/agrfood.2025003.
Long N, Qiu M, Zuo Y, Deng H. Antimicrobial activity and metabolomic analysis of linalool against pathogenic bacteria methicillin-resistant Staphylococcus aureus. Infect Drug Resist. 2025;18:731-44. doi: 10.2147/IDR.S491358, PMID 39936039.
Cordeiro L, Figueiredo P, Souza H, Sousa A, Andrade Junior F, Medeiros D. Terpinen-4-ol as an antibacterial and antibiofilm agent against Staphylococcus aureus. Int J Mol Sci. 2020 Jun 25;21(12):4531. doi: 10.3390/ijms21124531, PMID 32630600.
Cheng F, Mo Y, Chen K, Shang X, Yang Z, Hao B. Integration of metabolomics and transcriptomics indicates changes in MRSA exposed to terpinen-4-ol. BMC Microbiol. 2021 Dec 4;21(1):305. doi: 10.1186/s12866-021-02348-2, PMID 34736405.
Ben Akacha B, Michalak M, Generalic Mekinic I, Kacaniova M, Chaari M, Brini F. Mixture design of α-pinene α-terpineol and 1,8-cineole: a multiobjective response followed by chemometric approaches to optimize the antibacterial effect against various bacteria and antioxidant activity. Food Sci Nutr. 2024 Jan;12(1):574-89. doi: 10.1002/fsn3.3780, PMID 38268912.
Yang J, Park MJ. Antioxidant effects of essential oils from the peels of citrus cultivars. Molecules. 2025 Feb 11;30(4):833. doi: 10.3390/molecules30040833, PMID 40005143.
Wardiyah SU, Cartika H, Puspita N. Physical stability photoprotective effect and primer irritation test of cream O/W lime peel extract (Citrus aurantifolia) as a sunscreen. Int J Appl Pharm. 2022 Jan 1;14(1):12-20. doi: 10.22159/ijap.2022.v14s1.04.
Chavda VP, Acharya D, Hala V, Daware S, Vora LK. Sunscreens: a comprehensive review with the application of nanotechnology. J Drug Deliv Sci Technol. 2023 Sep;86:104720. doi: 10.1016/j.jddst.2023.104720.
Gallardo KC, Rincon PQ, Stashenko EE, Verbel JO. Photoprotective agents obtained from aromatic plants grown in Colombia: total phenolic content antioxidant activity and assessment of cytotoxic potential in cancer cell lines of Cymbopogon flexuosus L. and Tagetes lucida Cav. essential oils. Plants. 2022 Jun 27;11(13):1-17. doi: 10.3390/plants11131693.
Zhu Y, Xu W, Zhang J, Liao Y, Firempong CK, Adu-Frimpong MA. Self-microemulsifying drug delivery system for improved oral delivery of limonene: preparation characterization in vitro and in vivo evaluation. AAPS PharmSciTech. 2019 May 26;20(4):153. doi: 10.1208/s12249-019-1361-8, PMID 30915610.
Ayed A, Essid R, Jallouli S, Ben Zaied A, Ben Fdhila S, Limam F. Antifungal activity of volatile organic compounds (VOCs) produced by Streptomyces olivochromogenes S103 against Candida albicans. Euro-Mediterr J Environ Integr. 2022 Jun 21;7(2):251-5. doi: 10.1007/s41207-022-00302-w.
Kumar D, Rajni SR, Rani S, Kumari R. Formulation and evaluation of emulgel of an antifungal drug for topical drug delivery. J Pharm Negat Results. 2023 Jan 3;13(8):4087-100. doi: 10.47750/pnr.2022.13.S08.517.
Khan BA, Ali A, Hosny KM, Halwani AA, Almehmady AM, Iqbal M. Carbopol emulgel loaded with ebastine for urticaria: development characterization in vitro and in vivo evaluation. Drug Deliv. 2022 Dec 31;29(1):52-61. doi: 10.1080/10717544.2021.2015483, PMID 34962186.
Da Costa SD, Basri M, Shamsudin N, Basri H. Stability of positively charged nanoemulsion formulation containing steroidal drug for effective transdermal application. J Chem. 2014;2014:1-8. doi: 10.1155/2014/748680.
Esposito T, Sansone F, Russo P, Picerno P, Aquino RP, Gasparri F. A water-soluble microencapsulated milk thistle extract as active ingredient for dermal formulations. Molecules. 2019 Apr 19;24(8):1547. doi: 10.3390/molecules24081547, PMID 31010144.
Wu Y, Wang L, Ding X, Wang W, Zhao L. Preparation of high internal phase emulsions based on pumpkin seed oil body and its physicochemical properties. LWT. 2023 Nov;189:115533. doi: 10.1016/j.lwt.2023.115533.
Grossi Bovi Karatay G, Medeiros Theophilo Galvao AM, Dupas Hubinger M. Storage stability of conventional and high internal phase emulsions stabilized solely by Chickpea aquafaba. Foods. 2022 May 28;11(11):1588. doi: 10.3390/foods11111588, PMID 35681338.
Sun A, Dong W, Li S, Huang A, Wang X. Preparation and characterization of pickering emulsion gel stabilized by WPI/SPI composite particles and encapsulation of milk-derived peptide FDRPFL. LWT. 2024 May;199:116093. doi: 10.1016/j.lwt.2024.116093.
Bhardwaj S, Bhatia S, Gupta PS, Singh S. Thiazole derivative based topical nanoemulgel for inhibition of bacterial virulence in surface infections. Iran J Basic Med Sci. 2022 Mar;25(3):352-63. doi: 10.22038/IJBMS.2022.59419.13192, PMID 35656177.
Milutinov J, Krstonosic V, Cirin D, Pavlovic N. Emulgels: promising carrier systems for food ingredients and drugs. Polymers (Basel). 2023 May 13;15(10):2302. doi: 10.3390/polym15102302, PMID 37242878.
Khan BA, Ahmad S, Khan MK, Hosny KM, Bukhary DM, Iqbal H. Fabrication and characterizations of pharmaceutical emulgel co-loaded with naproxen-eugenol for improved analgesic and anti-inflammatory effects. Gels. 2022 Sep 22;8(10):608. doi: 10.3390/gels8100608, PMID 36286109.
Drais HK. Development and evaluation essential oils nanoemulgel as human skin sanitizer using novel method. Turk J Pharm Sci. 2024;21(5):456-62. doi: 10.4274/tjps.galenos.2023.78006, PMID 39569700.
Pires PC, Damiri F, Zare EN, Hasan A, Neisiany RE, Veiga F. A review on natural biopolymers in external drug delivery systems for wound healing and atopic dermatitis. Int J Biol Macromol. 2024 Apr;263(1):130296. doi: 10.1016/j.ijbiomac.2024.130296, PMID 38382792.
Maded ZK, Sfar S, Taqa GA, Lassoued MA, Ben Hadj Ayed OB, Fawzi HA. Development and optimization of dipyridamole and roflumilast-loaded nanoemulsion and nanoemulgel for enhanced skin permeation: formulation characterization and in vitro assessment. Pharmaceuticals (Basel). 2024 Jun 19;17(6):803. doi: 10.3390/ph17060803, PMID 38931470.
Paneo MA, Thomas NA, Latif MS, Tuloli TS, Nurkamiden SD. Formulation and physical characterization of emulgel facial wash containing a combination of almond oil (Prunus amygdalus Dulcis) and sodium lauryl sulfate surfactant. J Pharm Sci. 2025 Jun 27;8(2):1318-27. doi: 10.36490/journal-jps.com.v8i2.640.
Yılmaz Usta D, Teksin ZS, Tugcu Demiroz F. Evaluation of emulgel and nanostructured lipid carrier-based gel formulations for transdermal administration of ibuprofen: characterization mechanical properties and ex-vivo skin permeation. AAPS PharmSciTech. 2024 May 31;25(5):124. doi: 10.1208/s12249-024-02831-9, PMID 38822143.
Donthi MR, Saha RN, Singhvi G, Dubey SK. Dasatinib-loaded topical nano-emulgel for rheumatoid arthritis: formulation design and optimization by QbD, in vitro, ex vivo, and in vivo evaluation. Pharmaceutics. 2023 Feb 22;15(3):736. doi: 10.3390/pharmaceutics15030736, PMID 36986597.
Xiao T, Ma X, Hu H, Xiang F, Zhang X, Zheng Y. Advances in emulsion stability: a review on mechanisms role of emulsifiers and applications in food. Food Chem X. 2025 Jul;29:102792. doi: 10.1016/j.fochx.2025.102792, PMID 40747487.
Kamble V, Fegade B, Bhaskar VH. Formulation development and evaluation of an emulgel. IJJPR. 2023 Jun;27(3):222-57.
Sarella PN, Godavari LR. The expanding scope of emulgels: formulation evaluation and medical uses. Int J Curr Sci Res Rev. 2023 May 29;6(5):3030-41. doi: 10.47191/ijcsrr/V6‑i5‑42.
Kriti S, Rishu Y, Pankaj B, Anjana D, Mona P. Comparative study of different polymer based emulgel. Int J Pharm Sci. 2024;2(3):129-36. doi: 10.5281/zenodo.10781299.
Patel BM, Kuchekar AB, Pawar SR. Emulgel approach to formulation development: a review. Biosci Biotech Res Asia. 2021 Oct 27;18(3):459-65. doi: 10.13005/bbra/2931.
Kabalan Y, Bernarda H, Montane X, La Flor SD, Tylkowski B, Trojanowska A. Preparation and characterization of alginate-based capsules containing lavender essential oil for acne therapy. Carbohydr Polym Technol Appl. 2025;10:100766. doi: 10.1016/j.carpta.2025.100766.
El-Rhouttais C, Elfazazi K, Kettabi ZE, Laaraj S, Elgoulli M, Al-Zharani M. Effect of xanthan gum-based edible coating enriched with cloves and cinnamon for extending the shelf-life of pomegranate fruit during cold storage. Sci Rep. 2025 Aug 27;15(1):31518. doi: 10.1038/s41598-025-15467-x, PMID 40858726.
Haqiqi MT, Bankeeree W, Lotrakul P, Pattananuwat P, Punnapayak H, Ramadhan R. Antioxidant and UV-blocking properties of a carboxymethyl cellulose–lignin composite film produced from oil palm empty fruit bunch. ACS Omega. 2021 Apr 13;6(14):9653-66. doi: 10.1021/acsomega.1c00249, PMID 33869945.
Lopes AI, Melo A, Afonso TB, Silva S, Barros L, Tavaria FK. Alginate edible films containing essential oils: characterization and bioactive potential. Polymers (Basel). 2025 Apr 27;17(9):1188. doi: 10.3390/polym17091188, PMID 40362972.
Gulcin I, Alwasel SH. DPPH radical scavenging assay. Processes. 2023 Jul 26;11(8):2248. doi: 10.3390/pr11082248.
Javed S, Mangla B, Salawi A, Sultan MH, Almoshari Y, Ahsan W. Essential oils as dermocosmetic agents their mechanism of action and nanolipidic formulations for maximized skincare. Cosmetics. 2024 Dec 2;11(6):210. doi: 10.3390/cosmetics11060210.
Permadi N, Nurzaman M, Doni F, Julaeha E. Elucidation of the composition antioxidant and antimicrobial properties of essential oil and extract from Citrus aurantifolia (Christm.) Swingle peel. Saudi J Biol Sci. 2024 Jun;31(6):103987. doi: 10.1016/j.sjbs.2024.103987, PMID 38617568.
Badry R, El-Nahass MM, Nada N, Elhaes H, Ibrahim MA. Structural and UV-blocking properties of carboxymethyl cellulose sodium/CuO nanocomposite films. Sci Rep. 2023 Dec 1;13(1):1123. doi: 10.1038/s41598-023-28032-1, PMID 36670212.
Nurlaila PS, Makiyatuzahro DA, Kuncoro A, Mardianingrum R. Uji penentuan nilai spf (Sun Protection Factor) pada sediaan emulgel ekstrak kulit buah manggis (Garcinia mangostana L.) sebagai tabir surya. JIFB. 2024;15(2):163-72. doi: 10.52434/jifb.v15i2.3187.
Farhamzah F, Kusumawati AH, Alkandahri MY, Hidayah H, Sujana D, Gunarti NS. Sun protection factor activity of black glutinous rice emulgel extract (Oryza sativa var glutinosa). Indian J Pharm Educ Res. 2022 Jan 12;56(1):302-10. doi: 10.5530/ijper.56.1.36.
Brandes A, Dunning M, Langland J. Antimicrobial activity of individual volatile compounds from various essential oils. Molecules. 2024 Apr 16;29(8):1811. doi: 10.3390/molecules29081811, PMID 38675631.
Ersanli C, Skoufos I, Fotou K, Tzora A, Bayon Y, Mari D. Release profile and antibacterial activity of Thymus sibthorpii essential oil-incorporated optimally stabilized type I collagen hydrogels. Bioengineering (Basel). 2025 Jan 19;12(1):89. doi: 10.3390/bioengineering12010089, PMID 39851363.
Rusu AG, Nita LE, Roșca I, Croitoriu A, Ghilan A, Mititelu Tartau LM. Alginate-based hydrogels enriched with lavender essential oil: evaluation of physicochemical properties antimicrobial activity and in vivo biocompatibility. Pharmaceutics. 2023 Nov 9;15(11):2608. doi: 10.3390/pharmaceutics15112608, PMID 38004586.
Bayraktar S, Copur B, Gunes E, Uysal A, Okudan A. Antimicrobial activity and characterization of PVA/CMC hydrogels prepared with essential oil. J Thermoplast Compos Mater. 2025 May 19;38(11):4301-28. doi: 10.1177/08927057251344282.
Raissa TH, Pratiwi ET, Hidayati AR. Formulation and evaluation of moringa leaf ethanol extract emulgel and its effectiveness test against Staphylococcus aureus. JBT. 2024 Dec 30;24(1b):736-45. doi: 10.29303/jbt.v24i1b.8218.