
Int J App Pharm, Vol 18, Issue 2, 2026, 221-234Original Article
ANTI-INFLAMMATORY AND ANTI-HEMORRHOIDAL ACTIVITY OF POLYHERBAL GEL WITH THEIR CHARACTERIZATION USING CROTON OIL-INDUCED MODEL AND IN SILICO ACTIVITY AGAINST INFLAMMATORY MEDIATORS
ISHAN AGGARWAL1,2*, SUKIRTI UPADHYAY1, ARVIND KUMAR2
1Faculty of Pharmacy, IFTM University, Moradabad-244102, U. P., India. 2Department of Pharmacy, S. D. College of Pharmacy and Vocational Studies, Muzaffarnagar-251001, U. P., India
*Corresponding author: Ishan Aggarwal; *Email: ishanagl@gmail.com
Received: 27 Oct 2025, Revised and Accepted: 22 Dec 2025
ABSTRACT
Objective: To formulate and optimize a polyherbal gel containing Azadirachta indica, Melia azedarach, Piper longum and Piper nigrum extracts and evaluate its anti-hemorrhoidal effect in rat model of croton oil-induced haemorrhoids and anti-diabetic effect by molecular docking.
Methods: Using fruit extracts, HPLC and HPTLC analysis were used to analyse phyto-chemicals (azadirachtin, gallic acid, quercetin, rutin, ellagic acid, etc.). The gels were prepared using mixture of plant extract and optimized for anti-inflammatory activity. Evans blue extravasation, recto-anal coefficient, TNF-α, IL-6, neutrophil count and histopathological examination in wistar rats was done for anti-haemorrhoidal activity. In Silico activity of the bioactive compounds were attached to the inflammation and metabolism-related proteins, that is COX-2, 5-LOX, MMP-9 and α-glucosidase.
Results: The extraction yield of A. indica fruits ranged from 1.6% to 4.8%, P. longum from 2.5% to 5.0%, P. nigrum from 2.1% to 4.4% and Melia azedarach was 5.1% in petroleum ether, 3.8% in chloroform, 3.4% in ethyl acetate and 5.3% in ethyl alcohol, with extraction times between 8 and 14 h. Among the solvents used, ethanol (95%) produced the highest extractive yield for all fruits. HPTLC of the combined mixture fraction showed peaks for rutin (Rf 0.16), gallic acid (Rf 0.47), quercetin (Rf 0.58), and ellagic acid (Rf 0.81), with additional peaks at Rf 0.34–0.38. Semi-quantitative analysis indicated rutin (2.8 mg/g), quercetin (2.5 mg/g), gallic acid (2.4 mg/g), and ellagic acid (1.3 mg/g). In the hemorrhoidal rat model, CMF (1:1:1:1) at 400 mg/kg reduced Evans blue to 0.38±0.02 µg/g, neutrophils to 16.5±0.6%, recto-anal coefficient to 0.91±0.01, serum TNF-α to 173.5±1.3 pg/ml, serum IL-6 to 91.2±0.6 pg/ml, protein TNF-α to 135.3±8.5 pg/mg, protein IL-6 to 248±12.0 pg/mg, and restored stool weight to 17.3±0.18 g. CMF (1:1:2:2) showed comparable reductions (Evans blue 0.37±0.02 µg/g; neutrophils 15.5±0.6%; recto-anal coefficient 0.89±0.01; serum TNF-α 166.8±1.2 pg/ml). Based on anti-hemorrhoidal activity CMF (1:1:1:1) and CMF (1:1:2:2) were selected for gel formulation. In the carrageenan-induced paw edema model, CMF-G1 (1111) reduced paw volume to 1.13±0.05 ml (2 h) and 0.95±0.04 ml (4 h), producing 48.2±0.31% edema inhibition, while CMF-G2 (1122) reduced paw volume to 1.20±0.02 ml (2 h) and 1.10±0.03 ml (4 h), with 35.4±0.42% inhibition, compared to 56.0±0.31% for diclofenac gel.
Conclusion: The herbal gels formulated were effective and synergistic, exhibiting biphasic anti-hemorrhoidal and strong α-glucosidase inhibitory activities suggesting development of a novel treatment for hemorrhoids with simultaneous metabolic dysfunctions which is phytotherapeutic, safe and effective.
Keywords: Veno protective, Synergy, Evans blue extravasation, Dual-acting phytotherapy
© 2026 The Authors.Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i2.57289 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
One of the most commonly seen anorectal disorders are hemorrhoids, and are medically and surgically most neglected [1, 2]. They are characterized by abnormal dilatation and inflammation of the rectal venous plexuses and presence of pain, bleeding, swelling, and discomfort. This condition greatly interferes with day-to-day living and the overall quality of life. The prevalence of hemorrhoids as estimated in the literature is anywhere between 4% to 35% with the noted prevalence being higher for the adults aged 40 and above [3, 4]. Even though haemorrhoids are quite common, there is a scarcity of treatment modalities. Conventional drugs such as corticosteroids and local anaesthetic agents offer temporary relief, but their side effects of thinning the mucosa, irritation, and high recurrence of the condition are even more problematic [5]. These factors indicate a need for something more useful and safer that acts on the inflammation and the vascular pathology associated with the haemorrhoids. Diabetes mellitus continues to be one of the most significant public health concerns, with the most recent estimates showing over 90% of cases worldwide attributed to type 2 diabetes mellitus [6, 7], consequently, it is the most prevalent type of diabetes. The illness comes along with the health consequences of hyperglycemia, including microvascular and macrovascular damage, impaired wound healing, and susceptibility to infections. One of the main focuses of diabetes treatment and management is the inhibition of α-glucosidase, which delays the digestion of carbohydrates and post-hyperglycemia [8-10].
Synthetic α-glucosidase inhibitors such as acarbose do work effectively [11], however, their sales are hindered due to the associated side effects of flatulence [12]. The fact that haemorrhoids and diabetes have in common pathological mechanisms such as oxidative stress, inflammation, and impaired vascular and tissue repair as well as functionality. The diabetes condition with the symptom of hyperglycaemia is also associated with the phenomenon of endothelial dysfunction, contributing to the fragility of the venous system and therefore worsening the symptoms of haemorrhoids [13, 14].
As a result, the present study’s focus was the formulation and evaluation of the polyherbal gel for its anti-hemorrhoidal effect using croton oil as well as its anti-diabetic activity through molecular docking studies against α-glucosidase. These two health concerns highlight the growing global interest in natural, more effective therapeutic approaches, particularly polyherbal medicines, for managing underlying diseases. Moreover, deeper insights gained from previous studies contribute to the development of improved treatment strategies for patients [15-17]. In this particular study, polyherbal gel formulated using the fruits of Azadirachta indica, Melia azedarach, Piper nigrum, and Piper longum was developed. The formulation was tested in the croton oil–induced hemorrhoid model using the following parameters: recto-anal coefficient, evansblue dye leakage, cytokine levels (TNF-α, IL-6), neutrophil count, stool count, and histopathology. These provided an in-depth value of anti-hemorrhoidal activity. Also, in silico molecular docking studies of the phytoconstituents were performed on α-glucosidase to study the possible anti-diabetic activity. In diabetes, chronic hyperglycemia promotes excessive production of reactive oxygen species, resulting in pronounced oxidative stress that damages endothelial and surrounding tissues [18-21]. This oxidative imbalance triggers chronic inflammation, characterized by elevated pro-inflammatory cytokines such as TNF-α and IL-6, which impair normal tissue repair and promote further vascular injury [22]. Additionally, diabetes is strongly associated with vascular dysfunction, where endothelial cells exhibit reduced nitric oxide availability, increased permeability, and diminished structural integrity [23-25]. These mechanisms, central to diabetic complications, parallel and exacerbate the pathological processes observed in hemorrhoids, including venous fragility, inflammation, and impaired healing.
MATERIALS AND METHODS
Materials
Fruits of Azadirachta indicaajuss (neem), Melia azedarach l. (bakayan), Piper longum l. (long pepper), and Piper nigrum l. (black pepper) were purchased from a local market in Muzaffarnagar, Uttar pradesh, India. Plant materials were taxonomically verified by Dr. Sunita garg, former chief scientist and then head of the RHMD division of CSIR-NIScPR. The authentication no.-NIScPR/RHMD/Consult/2021/3839-40-4, was provided for Melia azedarach, (bakayan), fruit, NIScPR/RHMD/Consult/2021/3839-40-1, was provided for Piper nigrum, (black pepper), fruit, NIScPR/RHMD/Consult/2021/3839-40-2, was provided for Piper longum, (pipapali), fruit and NIScPR/RHMD/Consult/2021/3839-40-3 was provided for Azadirachta indica, (neem), fruit.
Chemicals and reagents
The study used the following chemical and reagent ethanol (Merck), n-hexane (Qualigens), chloroform (Merck), benzene (Rankem), petroleum ether (Qualigens), ethyl acetate (Merck), carrageenan (CDH laboratory reagents, Mumbai), diclofenac gel (Himalaya drug company), carbopol (Lubrizol advanced materials, USA), Pilex gel (Himalaya drug company, Bengaluru), croton oil (Sigma-aldrich, USA), evans blue dye (HiMedia laboratories, Mumbai) and HPTLC plates silica gel 60 F 254.
Instruments
The study uses the following instrument CAMAG visualizer: 201398 (Visualizer_201398), winCATSplanar chromatography manager, CAMAG TLC scanner "scanner_201368" S/N 201368 (2.01.02), twin trough chamber 10x10 cm glass tank, CAMAG automatic TLC sampler 4 (ATS4) "ATS4_201152" S/N 201152 (1.02.18), UV-visible double beam spectrophotometer (LabtronicsLT-2700).
Extraction of selected plants
The dried fruits were powdered and each powder used (100 g) was extracted successively with petroleum ether, chloroform, ethyl acetate, and 95% ethanol followed by chloroform-water maceration for 36 h (2-3 cycles) [26]. Each extract was filtered using whatman No. 1 filter paper and concentration of extracts was done by water bath. The water bath was set at 40 °C. The % w/w yield of the extractive values were determined and preserved at 4ºC for further use. In this study, A refers to extract of black pepper (Piper nigrum) whereas B refers to long pepper (Piper longum) whereas C refers to bakayan (Melia azedarach) and D refers to nimboli/neem (Azadirachta indica). After completion of extraction, qualitative tests (phytochemical tests) were performed to confirm the presence of plant metabolite.
The evaluation was carried out with five experimental combinations. The combinations included A+B (each 200 mg/kg), C+D (each 200 mg/kg), CMF (1: 1: 1: 1, each 100 mg/kg), CMF (2:2: 1: 1, piperaceae enriched, A+B 150 mg/kg each and C+D 50 mg/kg each) and CMF (1: 1: 2: 2, meliaceae enriched, A+B 50 mg/kg each and C+D 150 mg/kg each). They comprise the combined mixtures of fractions derived from the fruits of Piper nigrum, Piper longum, Melia azedarach, and Azadirachta indica.
Chromatography
High-performance liquid chromatography
Fruits of Piper nigrum, Piper longum, Melia azedarach and Azadirachta indica have specific profiles. Utilizing HPLC on a C₁₈ divided column (4.6×250 mm, 5 µm) by means of a binary gradient of water (A) and acetonitrile (B) at 1.0 ml min⁻¹ the gradient was from 95% A – 5% B to 5% A – 95% B over 55 min and re-equilibration afterwards. The sample was filtered (0.45 µm) using methanol HPLC-grade and injected 20 µl**. The compounds used for the comparison to identify the peaks with respect to retention time and UV spectra are piperine, gallic acid, quercetin, rutin and ellagic acid. The small spikes in the chromatogram after 50 min were found to be derivatives of nimbolide and azadirachtin, which are dominated by limonoids.
High-performance thin-layer chromatography
HPTLC was executed on the composite fractions mixture generated from the fruit matrix of Piper nigrum l., Piper longum l., Melia azedarach l., and Azadirachta indica ajuss. Analysis was performed on commercially pre-coated silica gel 60 F254 aluminum sheets (dimensions: 10 × 10 cm, Merck, Darmstadt, Germany) utilizing the CAMAG Automatic TLC sampler (ATS 4) for depositing both test and reference compounds—namely, gallic acid, quercetin, rutin, and ellagic acid.
Animal activity
All experimental animals were housed under standardized laboratory conditions and acclimatized for five days prior to experimentation in a temperature-controlled environment maintained between 20–25 °C with a 12-hour light and 12-hour dark cycle. Standard pellet feed and water were provided ad libitum, following the CPCSEA “no-starvation” guideline. All procedures were performed in accordance with IAEC regulations and were approved under protocol number SDCOP and VS/AH/CPCSEA/02/19. After completing the extraction procedures, qualitative phytochemical tests were conducted to confirm the presence of characteristic plant metabolites in each extract. For clarity, the extracts were designated as follows: A representing Piper nigrum (black pepper), B representing Piper longum (long pepper), C representing Melia azedarach (bakayan), and D representing Azadirachta indica (nimboli/neem). All treatments were administered intrarectally at a uniform total dose of 400 mg/kg. Animals were randomly assigned to eight groups including normal and disease controls, a standard group receiving croton oil preparation followed by pilex ointment, and five treatment groups prepared from different combinations of extracts. The evaluated combinations consisted of A+B in a 1:1 ratio (200 mg/kg each), C+D in a 1:1 ratio (200 mg/kg each), and a composite mixed formulation (CMF) of all four extracts in varying proportions. The CMF was prepared in three forms: an equal-ratio combination A+B+C+D (1:1:1:1; 100 mg/kg each), a piperaceae-enriched formulation (2:2:1:1) containing A and B at 150 mg/kg each and C and D at 50 mg/kg each, and a meliaceae-enriched formulation (1:1:2:2) containing A and B at 50 mg/kg each and C and D at 150 mg/kg each. These extract combinations, derived from the fruits of Piper nigrum, Piper longum, Melia azedarach, and Azadirachta indica, were selected to investigate their potential synergistic activity in croton oil–induced hemorrhoidal inflammation.
Anti-hemorrhoidal activity
Hemorrhoidal disease was induced in wistar albino rats using a sterile cotton swab pre-soaked in 10% (v/v) croton oil emulsion, inserted 10 mm into the anus for 10 seconds, leading to anorectal inflammation and edema within 7–8 h[27]. Rats of both sexes were randomly assigned to groups (n = 6) as mentioned in table 1 [28]. Twenty-four hours post-induction, standard and test groups treated rectally with respective ethanolic plant extracts applied using sterile swabs. A bolus of evans blue dye was administered intravenously 30 min prior to induction. The six-day study included blood and recto anal tissue collection for analysis of polymorphonuclear leukocytes and TNF-α and IL-6 levels. Additional parameters assessed were evans blue dye extravasation, recto-anal coefficient (RAC), average stool count, and histopathological alterations. Evans blue quantification involved drying recto anal tissues at 56 °C for 48 h, followed by extraction in formamide for another 48 hr, absorbance of the filtrate was measured at 625 nm and concentrations were extrapolated from a standard evans blue curve [29].
Table 1: Experimental design for anti-hemorrhoidal activity
| Group | Dose | Treatment |
| Normal Control | – | Saline |
| Disease Control | – | Croton oil preparation (ir)+Saline |
| Standard | 400 mg/kg (ir) | Croton oil preparation (ir)+Pilex ointment |
| Treatment Group 1 (A+B, 1:1) | 400 mg/kg (ir) | Croton oil preparation (ir)+Combination of A (P. nigrum) and B (P. longum), 200 mg/kg each |
| Treatment Group 2 (C+D, 1:1) | 400 mg/kg (ir) | Croton oil preparation (ir)+Combination of C (M. azedarach) and D (A. indica), 200 mg/kg each |
| Treatment Group 3 (CMF 1:1:1:1) | 400 mg/kg (ir) | Croton oil preparation (ir)+A+B+C+D, 100 mg/kg each |
| Treatment Group 4 (CMF 2:2:1:1) | 400 mg/kg (ir) | Croton oil preparation (ir)+Piperaceae-enriched CMF (A+B = 150 mg/kg each; C+D = 50 mg/kg each) |
| Treatment Group 5 (CMF 1:1:2:2) | 400 mg/kg (ir) | Croton oil preparation (ir)+Meliaceae-enriched CMF (A+B = 50 mg/kg each; C+D = 150 mg/kg each) |
Where, A = black pepper (Piper nigrum), B = long pepper (Piper longum), C = bakayan (Melia azedarach), D = nimboli/neem (Azadirachta indica), CMF = Combined mixed fraction, ir = intra rectal, mg = milligram, kg = kilogram.
Formulation and optimization of herbal gel
To obtain the best gels possible, variable concentration of different gelling agents was optimized to achieve best viscosity, spreadability and homogeneity. Carbopol 934, HPMC, and Na-CMC at a concentration range of (1.0–2.5) % were separately taken for evaluation. On behalf of anti-hemodialysis activity CMF (1:1:1:1) and CMF (1:1:2:2) is selected for formulation of gel. The pH, viscosity, spreadability, extrudability, and homogeneity of all prepared gel formulations were assessed. The six gel formulations made from carbopol with different concentration which are CMF-1111 – G1, G2, G3 and CMF-1122 – G1, G2, G3 were selected as optimal ones. Each polymer were dispersed singly in distilled water and stirred. After that methyl paraben (0.5 g) was added as the preservative and propylene glycol 400 (5 g) used as the cosolvent and the humectant. The polymer base was hydrated and then a CMF-1111 or CMF-1122 extract of 2 gs was added. Triethanolamine was added to the mixture so that the pH was adjusted towards the gel formation range of 6.8-7.0. The control gels were made the same way as other gels, except that no extract was added.
Anti-inflammatory activity (carrageenan-induced rat paw edema model)
The anti-inflammatory property of the polyherbal gel formulations were studied using the carrageenan-induced paw edema method in Wistar rats. The right back paw of the mouse was injected subcutaneously with 0.1 ml of freshly prepared 1% carrageenan suspension to induce edema. This model can be used to test the application of anti-inflammatory agents and shows a 2-phase response. The first phase, which is 0–2-hour duration, protects from or limits an injury. This phase gets positively influenced as formation of serotonin and histamine occurs directly. The next phase is 2–4-hour duration, which brings about injury. In this phase, bradykinin and prostaglandin form an influence. Maximum edema is seen 4 h post the injection. Rats were randomly divided the rats into groups of 6, creating a total of 4 groups, as shown in table 2 [30-32].
Table 2: Groups and dose in carrageenan-induced rat paw edema method
| S. No. | Group | Gel composition and dose received |
| 1 | Control group | Received 2 g gel base only (sodium CMC) |
| 2 | CMF-G1 (1111, Carbopol base) | Received 2 g CMF-1111 gel |
| 3 | CMF-G2 (1122, Carbopol base) | Received 2 g CMF-1122 gel |
| 4 | Standard drug group | Received 2 g Diclofenac sodium gel (CMC base) |
Where, CMF = Combined mixed fraction, CMC = carboxy methyl cellulose, g = g.
In silico study
Molecular docking was performed using MOE 2022 to investigate the binding affinity of selected phytochemicals azadirachtin, ellagic acid, gallic acid, nimbolide, piperine, quercetin, and rutoside along with two reference standards, diclofenac and mimosine. All ligand structures were retrieved from pubChem in sdf format and prepared using the quickprep function of MOE, which applies 3D protonation, structure correction, and energy minimization using the Amber10:EHT forcefield. The inflammatory protein targets COX-2 (PDB: 5KIR), 5-LOX (PDB: 3O8Y), and MMP-9 (PDB: 1GKC) were chosen due to their known roles in prostaglandin biosynthesis, leukotriene formation, and extracellular-matrix degradation, which are central mechanisms contributing to hemorrhoidal inflammation, and an additional diabetes-related target, α-glucosidase (PDB: 5NN5), was included to evaluate the glucose-regulatory potential of the selected phytochemicals [33]. Protein structures were refined using MOE quickprep, and active binding pockets were identified through MOE site finder tool. Docking simulations were performed using the triangle matcher placement method and londond G scoring function, followed by force field refinement. For each ligand, multiple conformations were generated, and the top-ranked poses were analyzed using the MOE ligand interactions and MOE report modules to characterize hydrogen bonding, hydrophobic contacts, pi-stacking, and electrostatic interactions relevant to their potential anti-inflammatory and anti-hemorrhoidal activities as well as their possible contribution to anti-diabetic mechanisms through α-glucosidase inhibition [34-36].
RESULTS AND DISCUSSION
The extraction times of Azadirachta indica fruit ranged from nine to thirteen hours and gave a yield of 4.5% with petroleum ether, 1.6% with chloroform, 3.1% with ethyl acetate, 4.8% with ethanol (95%) and 2.2% with chloroform water. The yield of the P. longum fruits was 4.7% in petroleum ether; 3.2% in chloroform; 3.6% in ethyl acetate; 5% in ethanol; and 2.5% in chloroform-water, with extraction periods of 9–14 h. The fruits of P. nigrum give 3.9%, 2.7%, 3.0%, 4.4%, and 2.1% using respective solvents in 8–13 h. The yield of Melia azedarach fruits was 5.1% in petroleum ether, 3.8% in chloroform, 3.4% in ethyl acetate and 5.3% in ethyl alcohol. In general, ethanol (95%) gave the highest extractive yield of all plant species tested, followed by petroleum ether and ethyl acetate while chloroform and chloroform water extraction gave lower yields.
High-performance liquid chromatography
The HPLC results indicate that each fruit extract showed different phytochemical profiles thus distinguishing one from the other. The major peaks obtained from the HPLC chromatogram of Azadirachta indica are shown in fig. 2 indicate the presence of gallic acid together with quercetin as well as other derivative molecules along with contributions from the nimbolide and azadirachtin. The presence of ellagic acid, as techniques beyond doubt have shown, eludes to the phenolic acid as well as the phenolic based acid contribution along with the limonoids found paralleling the neem as documented from findings [37]. The chromatogram as shown from fig. 3 of Melia azedarach mark the importance of ellagic acid all through the quercetin along with the gallic acid in the phytochemical analysis documented [38]. Based on the study in the paper, the chromatogram as shown in fig. 4 demonstrates how Piper longum manages to peak with the presence of a highly concentrated piperine with other minor peaks of amide that traces across the other noted alkaloids at 37.8 along with 39.6 min. The methods of piperine along with the notes of the relation amides confirm the evidence shown by means of standard retention profile. Fig. 5 chromatogram illustrates the existence of Piper nigrum as indicated by the peak, which has lower concentrations of the phenolic drugs. Consistent with previous reports, the presence of characteristic compounds, particularly nitrogen-containing phenolic alkaloids, was verified by a distinct chromatographic peak at 33.3 min [39]. From the chromatography presentations, it can be concluded that the extracts studied contain traces of defence compounds such as the flavonoids and bioactive compounds as well as the limonoids, thus proving that the extracts produced are antioxidant and anti-inflammatory.

Fig. 1: Hot extraction method
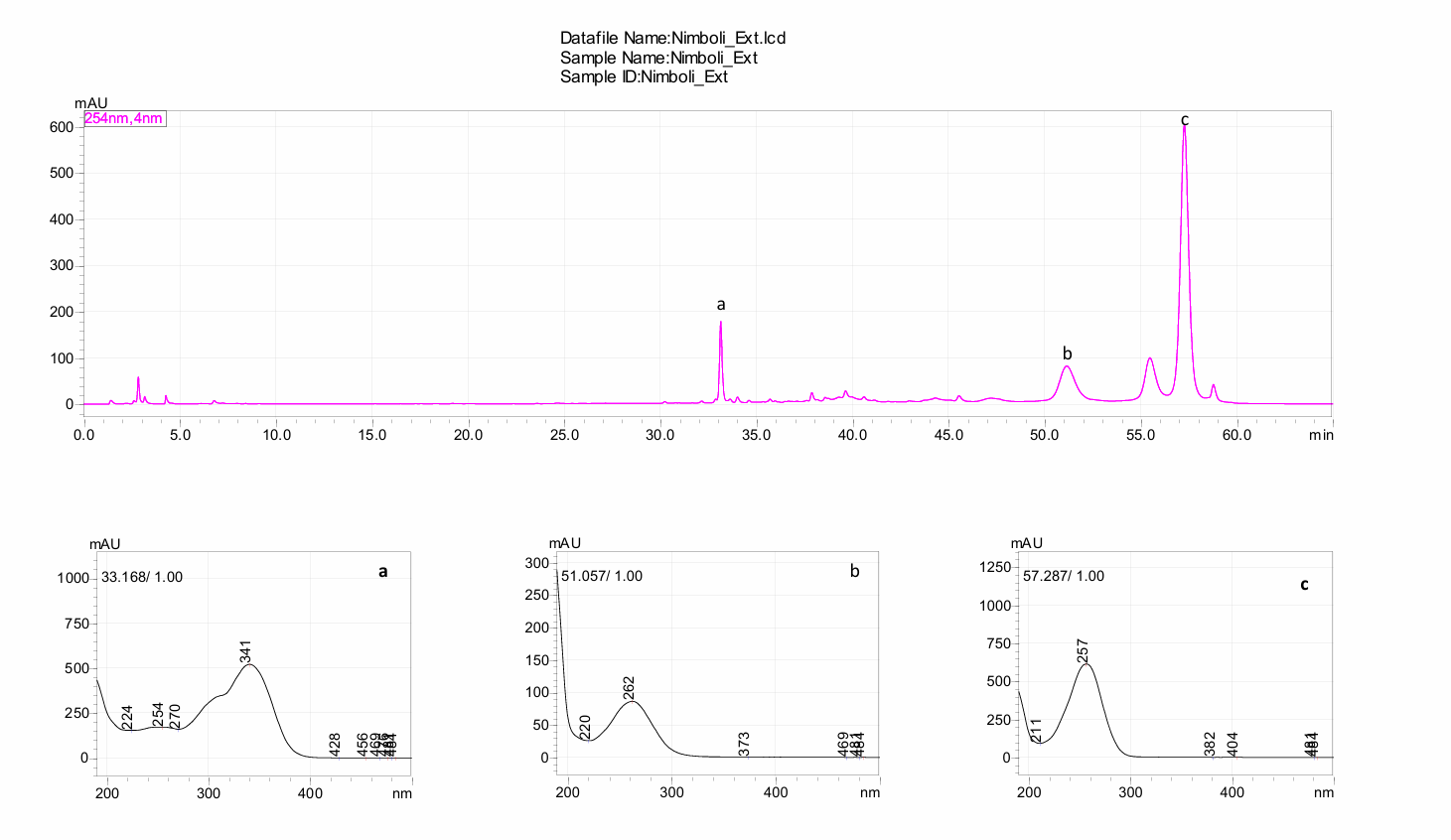
Fig. 2: HPLC chromatogram of Azadirachta indica fruit extract
High-performance thin-layer chromatography
The key phytoconstituents that are involved in anti-haemorrhoidal actions through alteration of inflammatory and oxidative pathways are gallic acid (GA), quercetin (Q), and ellagic acid (EA), which are present in Azadirachta indica, Melia azedarach, Piper longum and Piper nigrum whose structures are drawn in fig. 6, fig. 7 and fig. 8. These compounds belong to the class of phenolics and flavonoids, which are reported for their anti-inflammatory and free radical scavenging activity and their structures are reported in fig. 9. Piper longum contains piperine, which possibly inhibits pro-inflammatory signalling as well as providing analgesic effect which may provide some relief from the pain and swelling. There are reports on possible synergism between alkaloids and phenolic compounds with potential vascular protective and anti-inflammatory activity occurring in Piper nigrum with the presence of piperine and gallic acid. We have carried out extraction of phytoconstituents by HPTLC from some tropical plants with possible pharmacological activity. The chromatogram of combined mixture fraction in fig. 10 shows that rutin had a peak corresponding to a Rf of 0.16 (fig. 8) and gallic acid a peak with Rf of 0.47 (fig. 6). Similarly, quercetin gave a peak with Rf of 0.58 (fig. 7) and ellagic acid a peak corresponding to Rf of 0.81 (fig. 9), after derivatizations at 254 nm and 366 nm of the TLC plates. The combined mixture fraction is likely to possess significant pharmacological relevance, as the constituent herbs have been reported to exhibit therapeutic activity. These herbs contain phenolic and flavonoid compounds with established pharmacological properties, including gallic acid, quercetin, rutin, and ellagic acid, which are known for their anti-inflammatory and antioxidant effects. In addition, piperine, the major bioactive compound, has been reported to exhibit anti-inflammatory and analgesic effects, as well as to improve blood circulation.
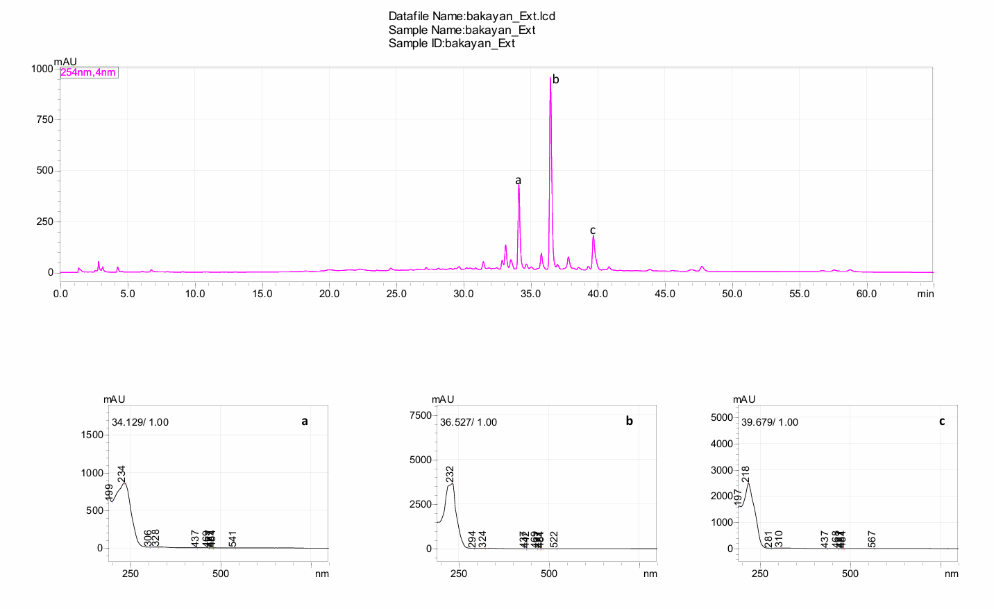
Fig. 3: HPLC chromatogram of Melia azedarach fruit extract
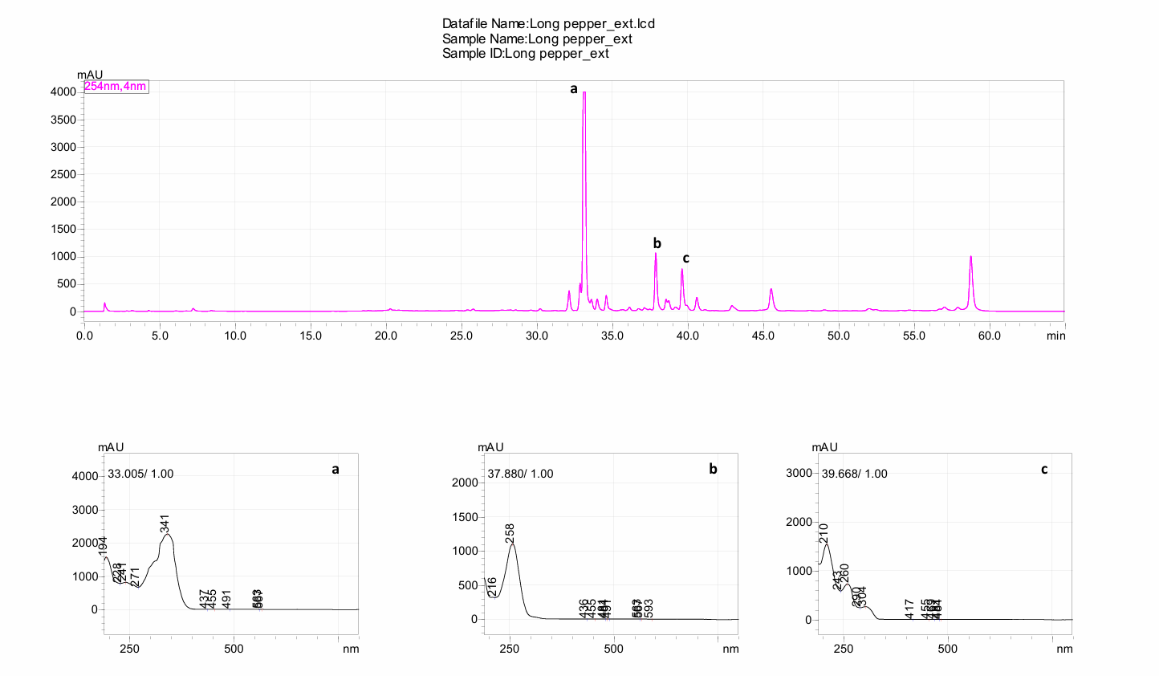
Fig. 4: HPLC chromatogram of Piper longum fruit extract
Table 3: Semi-quantitative content of key phytochemical markers (mg/g of extract)
| Phytochemical | Peak area (AU) | Relative % | Semi-quantitative content (mg/g extract) |
| Rutin | 9,106 | 28% | 2.8 mg/g |
| Quercetin | 8,140 | 25% | 2.5 mg/g |
| Gallic acid | 7,749 | 24% | 2.4 mg/g |
| Ellagic acid | 4,088 | 13% | 1.3 mg/g |
| Piperine | HPLC dominant peak | 40% | 4.0 mg/g |
Where, AU = Absorbance units, mg = milligram, g = g, HPLC = high performance liquid chromatography
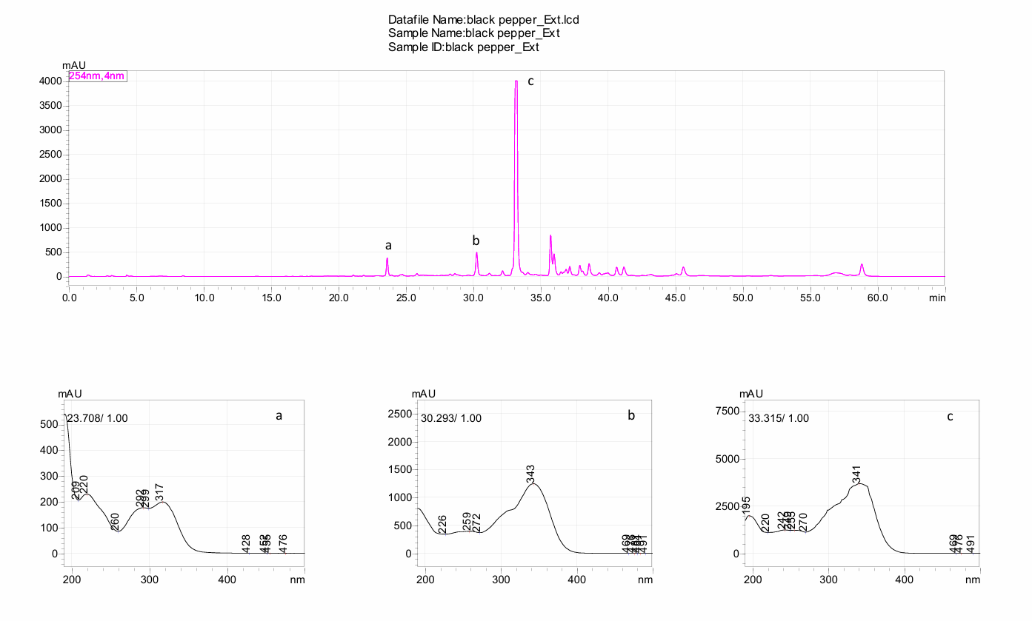
Fig. 5: HPLC chromatogram of Piper nigrum fruit extract
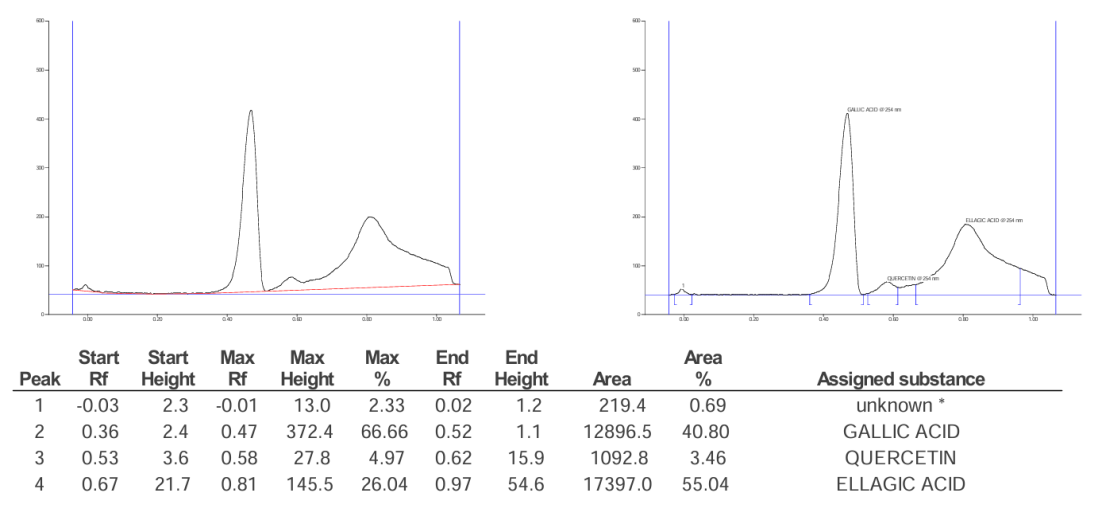
Fig. 6: Chromatogram of reference standard gallic acid
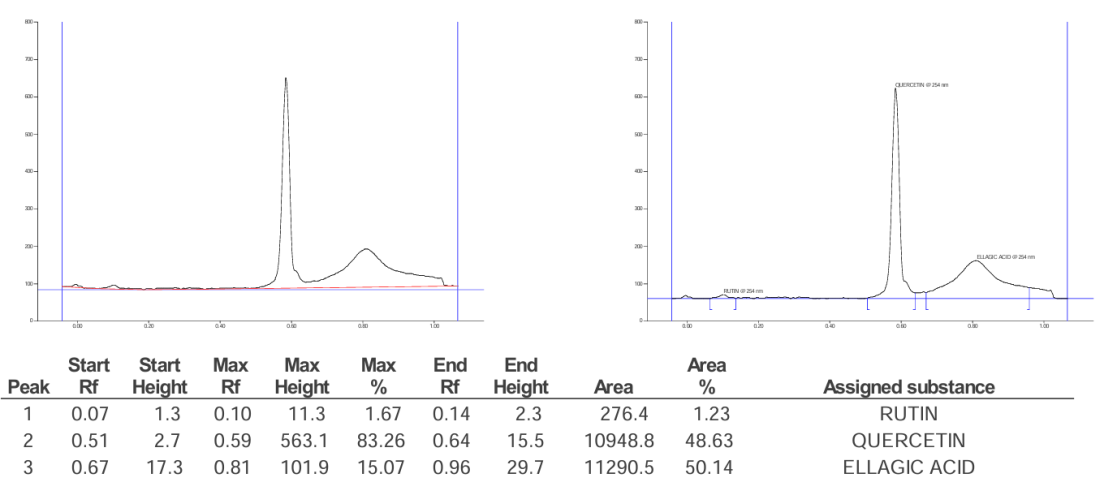
Fig. 7: Chromatogram of reference standard quercetin
Anti-hemorrhoidal activity
It is evident that evans blue concentration, neutrophil counts, recto anal coefficient, serum and protien levels of TNF-α and IL-6, and rats stool weight were significantly (p<0.001) lower than the control after hemorrhoids were induced, as presented in table 4. Treatment at a dosage of 400 mg/kg (p<0.001) has normalized most of the parameters. The test formulations, unknown to us that have maximum values for all the parameters are CMF (1:1:1:1) formulation suspended in 400 mg/kg that has maximum evans blue (0.38±0.02 µg/g), neutrophils (16.5±0.6 %), recto-anal coefficient (0.91±0.01), serum TNF-α (173.5±1.3 pg/ml), IL-6 (91.2±0.6 pg/ml), protein TNF-α (135.3±8.5 pg/mg), IL-6 (248±12.0 pg/mg) protein. CMF (2:2:1:1) enriched with piperaceae and CMF (1:1:2:2) enriched with meliaceae caused anti-inflammatory effects; combinations A+B and C+D also created moderate activity. All of the treated groups were in balance in stool weight, unlike the diseased control that was significantly (p<0.001) different. The present study evaluated the anti-hemorrhoidal activity using a single dose level (400 mg/kg). Dose–response relationships were not explored, which is acknowledged as a limitation of the study.
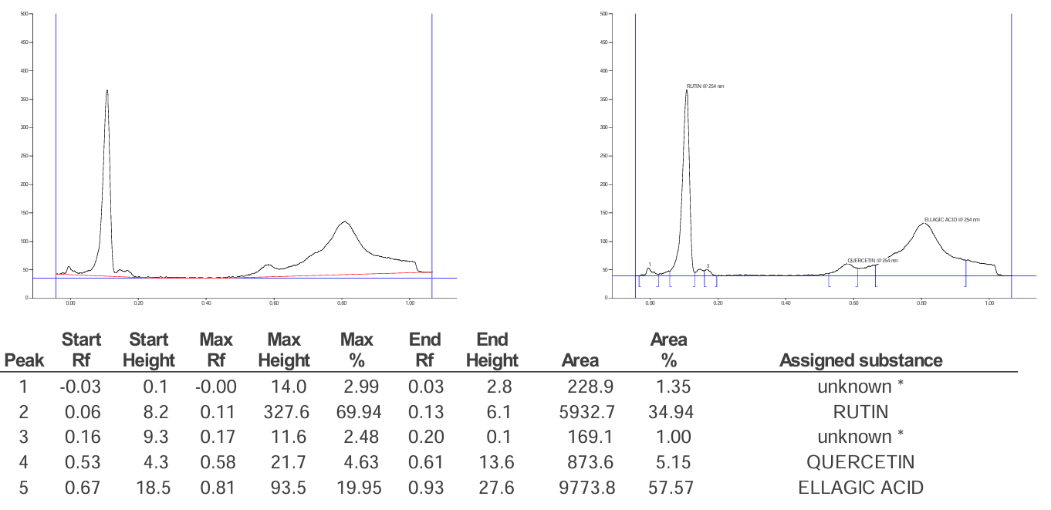
Fig. 8: Chromatogram of reference standard rutin
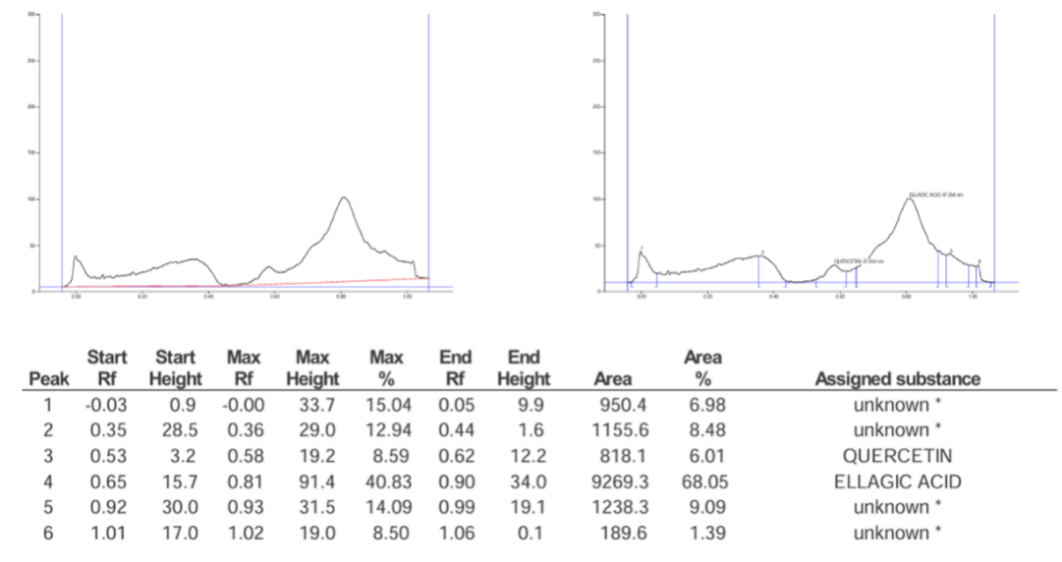
Fig. 9: Chromatogram of reference standard ellagic acid
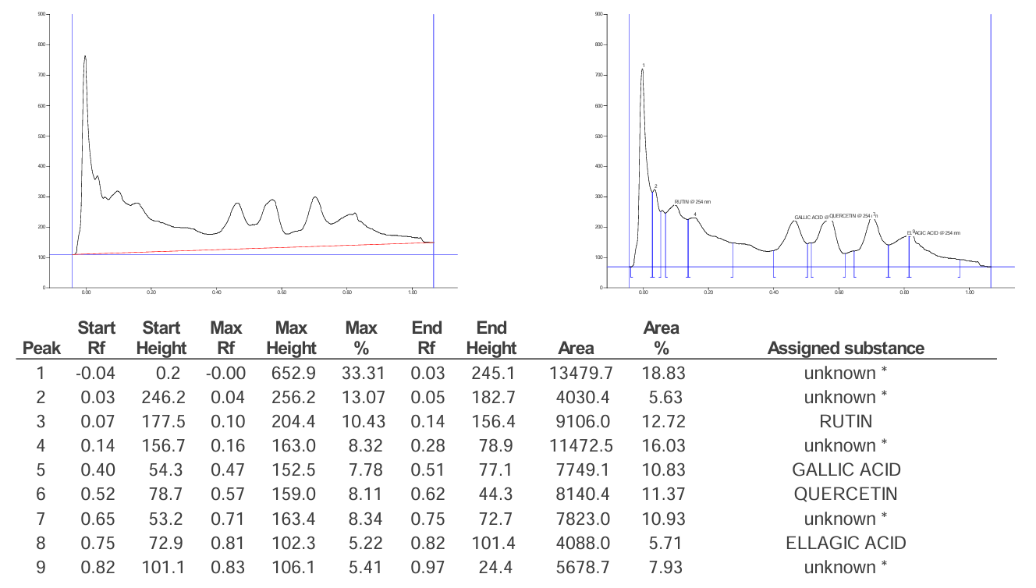
Fig. 10: Chromatogram of reference standard combined mixture fraction

Fig. 11: TLC plates after derivatization at 254 nm and 366 nm
Table 4: Effect of plant combinations on various parameters in hemorrhoidal rats
| Parameters | Normal Control | Disease Control | Standard 400 mg/kg | A+B 400 mg/kg | C+D 400 mg/kg | CMF 400 mg/kg (1:1:1:1) | CMF 400 mg/kg (2:2:1:1) | CMF 400 mg/kg (1:1:2:2) |
| Evans blue (µg/g) | 0.09±0.02 | 0.73±0.03### | 0.43±0.05*** | 0.54±0.03*† | 0.47±0.02** | 0.38±0.02*†† | 0.41±0.02** | 0.37±0.02*†† |
| % Neutrophil count | 12.7±0.7 | 27.4±1.2### | 19.1±0.9*** | 21.5±0.8*† | 18.9±0.7** | 16.5±0.6*†† | 18.3±0.7** | 15.5±0.6*††† |
| Recto-anal coefficient | 0.77±0.03 | 1.55±0.02### | 1.12±0.02*** | 1.22±0.01*† | 1.14±0.01** | 0.91±0.01*††† | 1.00±0.01*†† | 0.89±0.01*††† |
| Serum TNF-α (pg/ml) | 34.3±1.2 | 246.7±2.2### | 128.7±1.7*** | 223.8±1.7*††† | 188.3±1.1**†† | 173.5±1.3*††† | 179.5±1.1*†† | 166.8±1.2*††† |
| Serum IL-6 (pg/ml) | 78.3±2.2 | 114.8±2.6### | 94.2±1.3*** | 101.5±1.5**† | 95.2±1.0** | 91.2±0.6*† | 94.5±0.8** | 90.5±0.7*†† |
| Protein TNF-α (pg/mg) | 99.0±7.5 | 492.0±24.3### | 272.0±29.4** | 196.5±24.8*† | 147.0±14.2**†† | 135.3±8.5*††† | 152.2±4.7**† | 129.2±3.1*††† |
| Protein IL-6 (pg/mg) | 140±12.5 | 595±42.0### | 375±32.8** | 304±21.6**† | 268±18.2***† | 248±12.0*†† | 269.7±7.1**† | 237.0±4.6*††† |
| Stool weight (g) | 19.1±0.46 | 9.42±0.22### | 16.0±0.45** | 14.8±0.23*† | 16.6±0.24** | 17.3±0.18*† | 16.9±0.20** | 17.7±0.15*†† |
Where, n = 6. The observations are mean±SEM. TNF-α = Tumor necrosis factor-alpha; IL-6 = Interleukin-6; RAC = Recto-anal coefficient; CMF = Combined mixed formulation. *P<0.05, **P<0.01, *P<0.001 as compared to disease control; †P<0.05, ††P<0.01, †††P<0.001 as compared to standard; ###P<0.001 as compared to normal control. (ANOVA followed by dunnett’s test). n = 6. The observations are mean±SEM *P<0.05, **P<0.01, *P<0.001 as compared to disease control; †P<0.05, ††P<0.01, †††P<0.001 as compared to standard; ###P<0.001 as compared to normal control (ANOVA followed by Dunnett’s test).
Histopathological studies
It’s clear from the results of the study that the different experimental groups have recto-anal tissues that undergo different histomorphological changes as shown in fig. 12. The control group (A) kept the vertical configuration of the overlying mucosa with an epithelial layer as well as a sub mucosal layer. On the other hand, the diseased control (B) had serious pathology like tissue bleeding and damage. Moreover, the rectal blood vessels were stretched and over-filled. Similarly, the tissue was severely decaying. The mucosal structure of the rats treated with the standard drug (C) was almost normal and had only a slight inflammatory infiltration. The group which was treated with the extracts of Piper nigrum and Piper longum (D) showed some mild inflammation and scant hemorrhage on the other hand, group (E) showed mild to moderate inflammation, necrosis, and necrosis of the vessels due to the extracts of Melia azedarach and Azadirachta indica. When the ratio of CMF was set at 1:1:1:1 and treatment of the rat is done with 400 mg/kg (F), CMF heals these tissues as shown by the slight inflammation and lesser necrosis. The CMF (2:2:1:1) group with piperine (G) showed tissues that looked normal but had a few dilated red vessels. The CMF (1:1:2:2) group that had meliaceae-enriched (H) contained a mild inflammatory infiltrate but showed preservation structural features, which indicates retention of mucosal protective restorative function.
Where, T. S. = Transverse section, A)-Group-I: represents normal cell arrangement, architecture, and intact mucosal lining; B) Disease control Group II: ◊Tissue hemorrhage, Δ with elongation of rectal blood vessel necrosis, ☆degenerative changes were identified in disease control animals; C) Group III-Nonsignificant inflammatory changes with minimal structural alterations in standard drug treated animals; D) Group IV: Mild pathological changes like inflammation and hemorrhage in rats treated with black pepper+long pepper extract; E) Group V-Mild to moderate inflammation, hemorrhage, necrosis appeared in rats treated with bakayan+nimboli extract; F) Group VI-Minimal inflammatory phase, blood marks, necrosis, blood vessel damage in rats applied with CMF (1:1:1:1) extract; G) Group-VI: Inflammation, hemorrhage, blood vessel dilation is minimal in rats rectal tissue treated with Piperine-rich CMF (2:2:1:1); H) Last Group VII: inflammation and non-significant changes noticed in cellular architecture of rectal tissue of rats treated with meliaceae-enriched CMF 400 mg/kg/Ir.
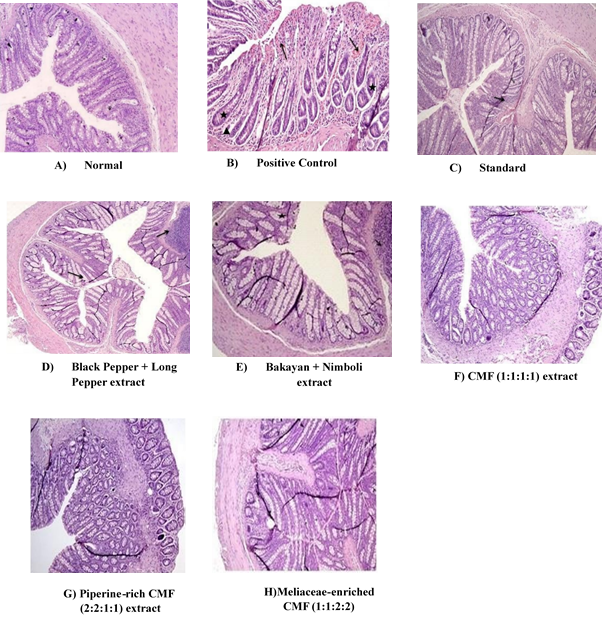
Fig. 12: Effect of gel on histology T. S. of recto anal tissue of rats in croton oil-induced haemorrhoids
Formulation of herbal gel
On behalf of anti-hemorrhoidal activity, CMF (1:1:1:1) and CMF (1:1:2:2) is selected for formulation of gel. The pH, viscosity, spreadability, extrudability, and homogeneity of all prepared gel formulations were assessed. The six formulations made from carbopol which are CMF-1111 – G1, G2, G3 and CMF-1122 – G1, G2, G3 were selected as optimal ones, having their pH in the range of 6.2 to 6.5, having high homogeneous and having spreadability (32 to 35 g·cm/s), having a high degree of extrudability and having an appropriate viscosity. The formulations CMF-1111 and CMF-1122 were examined further for in vivo activity. Time taken for drug release was 240 min. Cumulative drug release increased progressively in CMF-1111 formulations. The cumulative drug release, which has increased during G1 was from 14.20% at 15 min to 76.60% at 240 min, G2-15% at 15 min to 78.30% at 240 min, G3-13.60% at 15 min and 74.60% at 240 min. Similarly, the G1 of CMF-1122 was from 13.20% at 15 min to 74.50% at 240 min, G2-13.90% at 15 min to 76.30% at 240 min and G3-14.10% at 15 min to 77.00% at 240 min. Among these CMF-1111–G2, 78.30 % and CMF-1122–G3, 77.00 %, at 240 min showed maximum release with optimized physicochemical parameter and were selected for optimum formulation for further in vivo evaluation.

Fig. 13: Representation of combined mixture fraction polyherbal gel
Table 5: In vitro release study of carbopol gel formulations
| Time (min) | G1 (Carbopol 1.0 g) % | G2 (Carbopol 1.5 g) % | G3 (Carbopol 2.5 g) % |
| 15 | 14.20 | 15.00 | 13.60 |
| 30 | 28.15 | 28.90 | 26.30 |
| 45 | 34.00 | 35.60 | 32.40 |
| 60 | 45.00 | 46.50 | 43.10 |
| 90 | 54.70 | 56.20 | 52.00 |
| 120 | 63.60 | 65.10 | 61.20 |
| 180 | 71.30 | 72.90 | 69.00 |
| 240 | 76.60 | 78.30 | 74.60 |
Where, g = g, min = minutes, % = percentage release (A fixed quantity of 1 g gel was used per test).
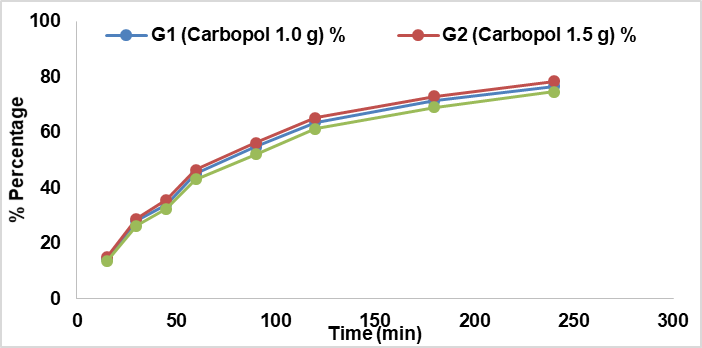
Fig. 14: Comparative in vitro release study of gel containing carbopol-based formulations
Carrageenan induced rat paw edema method
Carrageenan-induced paw edema is a sensitive and reproducible model to evaluate acute anti-inflammatory activity. In this study, the control group (carrageenan+gel base) showed the maximum increase in paw edema volume (1.68±0.05 ml at 4 h). Treatment with optimized herbal gel formulations significantly reduced paw edema. The CMF-G1 gel (1111, carbopol base) decreased paw edema to 0.95±0.04 ml, showing 48.2% reduction at 4 h, while CMF-G2 gel (1122, carbopol base) reduced paw volume to 1.10±0.03 ml, corresponding to 35.4% reduction. The standard diclofenac gel showed the strongest inhibition with a final paw volume of 0.74±0.02 ml and 56.0% reduction at 4 h. These results demonstrate that both CMF gels possess significant local anti-inflammatory activity, with CMF-G1 (1111, carbopol base) being the most effective, approaching the efficacy of the standard drug.
Table 6: Mean paw edema volume in carrageenan-induced rat paw edema method
| S. No. | Groups | Mean paw volume (1 h) | Mean paw volume (2 h) | Mean paw volume (3 h) | Mean paw volume (4 h) |
| 1 | Control group | 1.72±0.03 | 1.82±0.05 | 1.73±0.05 | 1.68±0.05 |
| 2 | CMF-G1 (1111, Carbopol base) | 1.20±0.03* | 1.13±0.05* | 1.08±0.04* | 0.95±0.04* |
| 3 | CMF-G2 (1122, Carbopol base) | 1.31±0.04* | 1.20±0.02* | 1.15±0.03* | 1.10±0.03* |
| 4 | Standard drug (Diclofenac gel) | 1.20±0.05* | 0.83±0.04* | 0.78±0.03* | 0.74±0.02* |
Where, n = 6. The observations are mean±SEM. *P<0.001 as compared to control (ANOVA followed by dunnett’s test). CMF = Combined mixed fraction, h = h
Table 7: Effect of formulated gel preparation in carrageenan-induced rat model
| S. No. | Groups | % Increase in paw weight (0 h) | % Reduction of edema (1 h) | % Reduction of edema (2 h) | % Reduction of edema (3 h) | % Reduction of edema (4 h) |
| 1 | Control group | 33.5±0.45 | 0 | 0 | 0 | 0 |
| 2 | CMF-G1 (1111, Carbopol base) | 18.3±0.42* | 22.2±0.43* | 38.3±0.39* | 41.0±0.36* | 48.2±0.31* |
| 3 | CMF-G2 (1122, Carbopol base) | 25.3±0.29* | 18.6±0.33* | 27.3±0.34* | 29.4±0.29* | 35.4±0.42* |
| 4 | Standard drug (Diclofenac gel) | 17.6±0.28* | 21.3±0.38* | 35.4±0.31* | 40.6±0.22* | 56.0±0.31* |
Where, n = 6. The observations are mean±SEM. *P<0.001 as compared to control. (ANOVA followed by dunnett’s test). CMF = Combined Mixed Formulation.
Fig. 15: Comparative X-ray image of the gel and the standard gel
In silico study
Docking analysis demonstrated that azadirachtin, rutoside, piperine, and quercetin exhibited the strongest and most consistent multi-target interactions across all four key therapeutic targets—COX-2, 5-LOX, MMP-9, and the diabetes-related transporter GLUT-1. The reference drugs diclofenac (anti-inflammatory) and mimosine (standard herbal constituent) displayed favourable binding poses comparable to previous crystallographic reports, validating the reliability of the docking workflow. Further methodological accuracy was confirmed through re-docking of native ligands, which produced RMSD values<2.0 Å, meeting the accepted criteria for a robust in silico protocol. Overall, the combined docking outcomes highlight that azadirachtin and rutoside show superior multi-target inhibition potential, outperforming standards across inflammation and diabetes-linked pathways, while piperine and quercetin contribute additional stabilizing interactions.
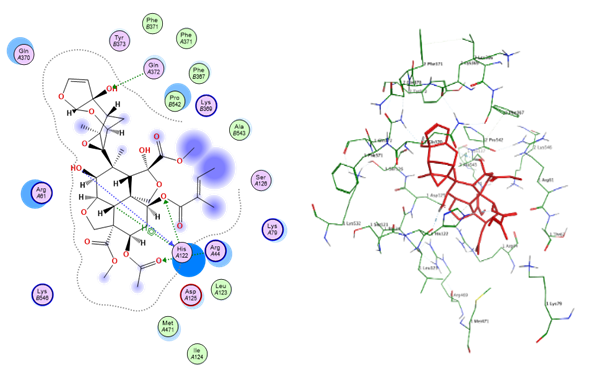
Fig. 16: Docking conformation of azadirachtin with cyclooxygenase-2
Table 8: Docking interaction profile of ligands against COX-2 (PDB: 5KIR)
| S. No. | Compound | Ligand atoms | Receptor atoms (Residue, chain) | Interaction type | Distance (Å) | Energy (kcal/mol) | Docking score (kcal/mol) | RMSD (Å) |
| 1 | Azadirachtin | O5 O6 O7 O16 C24 |
O–His122(A) NE2–His122(A) NE2–Gln372(A) NH1–Arg44(A) 5-ring–His122(A) |
H-donor H-acceptor H-acceptor H-acceptor H–pi |
2.78 2.91 3.10 2.99 4.47 |
–2.4 –1.6 –0.5 –0.7 –0.5 |
–8.4094667 | 1.4745684 |
| 2 | Ellagic acid | O5 O4 O7 |
OE1–Glu465(A) NE–Arg44(A) N–Cys47(A) |
H-donor H-acceptor H-acceptor |
2.67 2.86 3.05 |
–3.9 –0.8 –1.1 |
–5.8269463 | 0.94446844 |
| 3 | Gallic acid | O4 | O–Asp125(A) | H-donor | 3.51 | –0.6 | –5.5328655 | 1.3100287 |
| 4 | Nimbolide | O3 5-ring |
NZ–Lys546 (B) NH1–Arg44 (A) |
H-acceptor pi–cation |
3.07 4.53 |
–1.5 –0.7 |
–6.9973025 | 1.3593959 |
| 5 | Piperine | O1 | NE2–Gln372(A) | H-acceptor | 3.13 | –1.2 | –6.6139102 | 1.3149517 |
| 6 | Quercetin | O6 6-ring |
O–Ala151(A) NE–Arg44(A) |
H-donor pi–cation |
2.94 3.57 |
–2.6 –0.5 |
–6.9146142 | 1.9714825 |
| 7 | Rutoside | O5 O7 O10 O3 O4 C20 6-ring 6-ring |
O–His122(A) O–His122(A) O–Phe367(B) NE2–Gln372(A) N–Ser126(A) 5-ring–His122(A) CB–Arg61(A) CD–Arg61(A) |
H-donor H-donor H-donor H-acceptor H-acceptor, H–pi pi–H pi–H |
3.15 3.17 2.94 3.15 3.30 3.75 4.49 4.17 |
–0.7 –0.9 –1.2 –1.5 –0.9 –0.6 –0.6 –0.5 |
–8.32988 | 1.523569 |
| 8 | Diclofenac (Standard) | Cl1 O4 |
NZ–Lys532(A) NE2–Gln372(A) |
H-acceptor H-acceptor |
3.88 2.89 |
–0.8 –0.8 |
–6.1826372 | 1.3860111 |
| 9 | Mimosine (Standard) | O1 O3 O3 |
O–Ile124(A) N–Gln372(A) NZ–Lys532(A) |
H-donor H-acceptor H-acceptor |
3.12 3.14 3.06 |
–1.9 –2.5 –5.6 |
–5.0199652 | 1.4055604 |
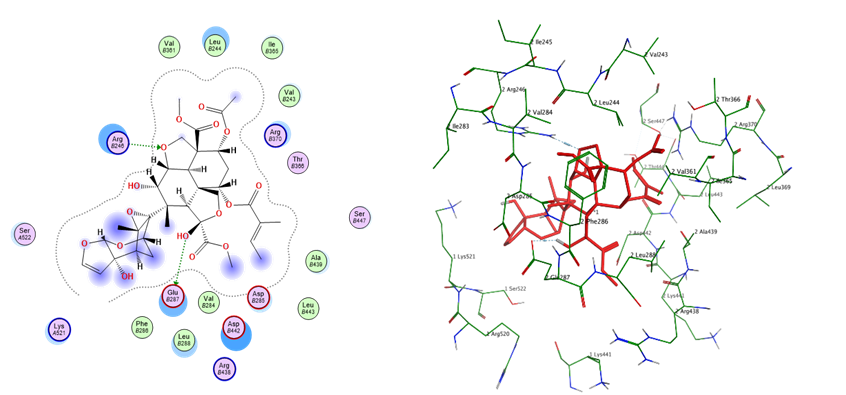
Fig. 17: Docking conformation of azadirachtin with 5-lipoxygenase
Table 9: Docking interaction profile of ligands against 5-lipoxygenase (PDB: 3O8Y)
| S. No. | Compound | Ligand atoms | Receptor atoms (Residue, Chain) | Interaction type | Distance (Å) | Energy (kcal/mol) | Docking score (kcal/mol) | RMSD (Å) |
| 1 | Azadirachtin | O8 O3 |
OE2–Glu287(B) NH2–Arg246(B) |
H-donor H-acceptor |
3.04 2.79 |
–1.1 –2.8 |
–8.4511747 | 1.7659231 |
| 2 | Ellagic acid | O7 6-ring |
NH1–Arg457(A) NH2–Arg370(A) |
H-acceptor pi–cation |
3.27 4.04 |
–0.7 –0.5 |
–6.6019568 | 1.460695 |
| 3 | Gallic acid | O2 O1 |
O–Cys99(B) NH1–Arg101(B) |
H-donor H-acceptor |
3.07 3.10 |
–2.3 –0.5 |
–5.2542105 | 0.45928627 |
| 4 | Nimbolide | O1 | NH2–Arg246(B) | H-acceptor | 3.18 | –1.0 | –7.3243484 | 1.4519179 |
| 5 | Piperine | O1 | NH2–Arg246(B) | H-acceptor | 3.14 | –1.7 | –6.2346239 | 1.6768535 |
| 6 | Quercetin | O7 | OD1–Asp442(B) | H-donor | 3.18 | –1.0 | –6.1792221 | 1.2508181 |
| 7 | Rutoside | O9 O14 O16 |
OE1–Glu287(B) O–Val243(B) O–Asp285(B) |
H-donor H-donor H-donor |
2.79 3.26 2.99 |
–1.7 –1.0 –1.7 |
–7.7909441 | 1.6830847 |
| 8 | Diclofenac (Standard) | N5 6-ring |
OD2–Asp442(B) CD–Lys441(A) |
H-donor pi–H |
2.79 3.60 |
–4.6 –1.2 |
–5.7130647 | 1.748126 |
| 9 | Mimosine (Standard) | O1 N6 N6 |
O–Tyr100 (B) O–Tyr100 (B) OE1–Glu622 (B) |
H-donor H-donor H-donor |
2.89 3.13 3.25 |
–2.3 –0.9 –0.5 |
–5.4374838 | 1.5344635 |
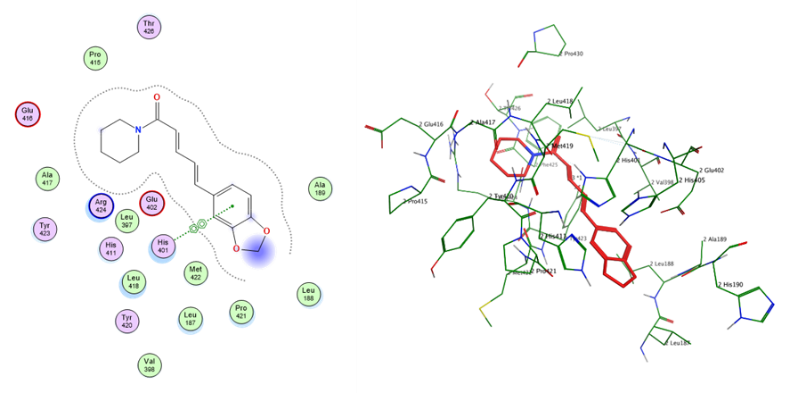
Fig. 19: Docking conformation of Piperine with matrix metalloproteinase-9
Table 10: Docking interaction profile of ligands against matrix metalloproteinase-9 (PDB: 1GKC)
| S. No. | Compound | Ligand atoms | Receptor atoms (Residue, chain) | Interaction type | Distance (Å) | Energy (kcal/mol) | Docking score (kcal/mol) | RMSD (Å) |
| 1 | Azadirachtin | O16 C35 |
N–Ala191(B) 5-ring–His405(B) |
H-acceptor H–pi |
3.31 3.95 |
–1.3 –2.9 |
–6.7632527 | 1.7569052 |
| 2 | Ellagic acid | O3 O6 6-ring 6-ring |
O–Tyr420(A) O–Glu416(A) CA–Tyr423(A) N–Arg424(A) |
H-donor H-donor pi–H pi–H |
2.58 3.17 3.80 4.17 |
–4.5 –1.4 –0.7 –0.6 |
–6.3106909 | 1.3809241 |
| 3 | Gallic acid | O1 O2 O4 6-ring |
O–Pro415(B) O–Met422(B) O–Arg424(B) CA–Leu418(B) |
H-donor H-donor H-donor pi–H |
2.98 2.81 2.97 4.23 |
–1.1 –4.1 –1.7 –0.5 |
–5.245779 | 1.1521168 |
| 4 | Nimbolide | C12 | 5-ring–His411(B) | H–pi | 4.50 | –0.6 | –6.7196736 | 1.6098577 |
| 5 | Piperine | 6-ring | 5-ring–His401(B) | pi–pi | 4.17 | -0.8 | –7.6294742 | 0.99619162 |
| 6 | Quercetin | O6 C21 6-ring 6-ring |
O–Pro421(B) OE2–Glu402(B) CA–Leu418(B) CB–Arg424(B) |
H-donor H-donor pi–H pi–H |
2.74 3.22 4.19 4.47 |
–1.3 –0.5 –0.7 –0.6 |
–7.1494236 | 1.120424 |
| 7 | Rutoside | O8 O9 |
N–Ala189(B) N–Leu188(B) |
H-acceptor H-acceptor |
3.00 3.02 |
–0.6 –1.2 |
–7.1987834 | 1.6020147 |
| 8 | Diclofenac (Standard) | 6-ring | CA–Tyr423(B) | pi–H | 4.22 | –0.7 | –5.7445488 | 0.71560484 |
| 9 | Mimosine (Standard) | O1 | O–Arg424(B) | H-donor | 2.88 | –0.6 | –5.8737283 | 0.52034706 |
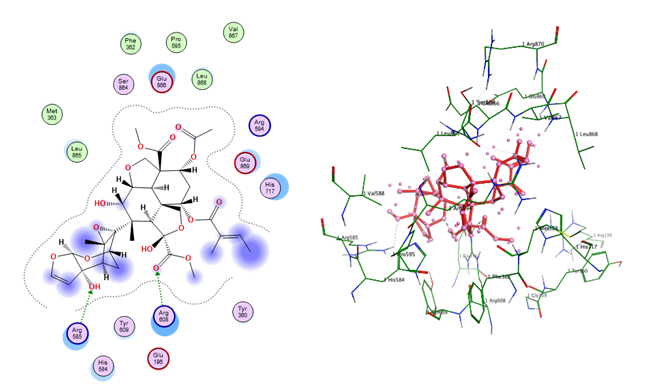
Fig. 20: Docking conformation of azadirachtin with glucose transporter 1
Table 11: Docking interaction profile of ligands against glucose transporter 1 (PDB: 5NN5)
| S. No. | Compound | Ligand atoms | Receptor Atoms (Residue, chain) | Interaction type | Distance (Å) | Energy (kcal/mol) | Docking score (kcal/mol) | RMSD (Å) |
| 1 | Azadirachtin | O7 O14 |
NH2–Arg585 (A) NH2–Arg608 (A) |
H-acceptor H-acceptor |
2.95 2.98 |
−0.5 −1.7 |
−7.3163295 | 1.8130846 |
| 2 | Ellagic acid | 6-ring | CB–Pro194 (A) | π–H | 3.74 | −0.7 | −5.4468966 | 0.72785813 |
| 3 | Gallic acid | O1 O3 |
O–Ile823 (A) O–Glu856 (A) |
H-donor H-donor |
2.90 3.24 |
−1.7 −1.3 |
−4.8737097 | 0.4188526 |
| 4 | Nimbolide | 5-ring | N–Val867 (A) | π–H | 3.91 | −2.6 | −6.7458315 | 1.4956416 |
| 5 | Piperine | O1 6-ring |
NH1–Arg199 (A) CA–Leu195 (A) |
H-acceptor π–H |
3.04 4.18 |
−3.4 −0.8 |
−6.0474176 | 1.5308647 |
| 6 | Quercetin | O5 6-ring |
O–Arg608 (A) N–Glu866 (A) |
H-donor π–H |
2.93 3.78 |
−2.0 −0.5 |
−5.5102310 | 1.6910031 |
| 7 | Rutoside | O8 O14 O15 O7 O9 |
NE2–His717 (A) O–Val718 (A) O–Tyr360 (A) NH2–Arg608 (A) N–Val867 (A) |
H-donor H-donor H-donor H-acceptor H-acceptor |
3.13 3.20 3.36 3.16 3.43 |
−0.7 −1.0 −0.5 −2.3 −0.5 |
−6.8043351 | 1.7932277 |
| 8 | Diclofenac | 6-ring 6-ring |
N–Val867 (A) N–Leu868 (A) |
π–H π–H |
3.89 4.62 |
−1.2 −1.4 |
−5.7565293 | 1.4774476 |
| 9 | Mimosine | O1 O3 6-ring |
O–Gln776 (A) N–Ile780 (A) CG–Gln776 (A) |
H-donor H-acceptor π–H |
3.07 3.16 3.83 |
−3.1 −0.9 −0.5 |
−5.2098951 | 1.4636953 |
| 10 | Metformin (Standard) | N5 C7 |
OD2–Asp518 (A) 6-ring–Phe649 (A) |
H-donor H–π |
2.97 3.74 |
−4.6 −0.5 |
−4.4575167 | 1.5689560 |
CONCLUSION
The present study indicates that the formulated polyherbal gel from Azadirachta indica, Melia azedarach, Piper nigrum and Piper longum possesses good anti-hemorrhoidal and anti-inflammatory activity. The croton oil-induced haemorrhoids model showed a significant improvement in evan blue leakage, neutrophil count, recto-anal coefficient, cytokine levels and tissue healing. This shows that the combined extract mixtures show a synergistic action to control vascular congestion, inflammation and mucosal injury. The optimized polyherbal gel showed excellent topical anti-inflammatory action in the carrageenan paw edema model, confirming that it could reach deeper sites to relieve skin inflammation and pain. All these findings suggest that the gel is effective and safe for site-specific management of irritation and swelling due to haemorrhoids. The polyherbal gel lower TNF-α and IL-6 levels and improved the histology of the tissues indicates that it could be a useful phytotherapeutic substitute for haemorrhoid ointments. Future insights have been added for in silico molecular docking. Several phytochemicals show glucose transporter-1 properties such as azadirachtin, quercetin, rutin, piperine etc. Other than that, they also have strong affinity towards anti-inflammatory targets such as COX-2, 5-LOX, MMP-9. The strong binding interactions with GLUT-1 may suggest a role for these plants in glucose regulation. This indicates that the polyherbal formulation may be useful in future diabetes-related studies, especially for patients in whom inflammation and metabolic imbalance occur together.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
The authors contributed equally to this manuscript. Ishan Aggarwal developed and wrote the manuscript. Sukirti Upadhyay supervised and validated the manuscript. Arvind Kumar assisted with data analysis and writing the manuscript.
CONFLICT OF INTERESTS
Declared none
REFERENCES
Lohsiriwat V, Sheikh P, Bandolon R, Ren DL, Roslani AC, Schaible K. Recurrence rates and pharmacological treatment for hemorrhoidal disease: a systematic review. Adv Ther. 2023;40(1):117-32. doi: 10.1007/s12325-022-02351-7, PMID 36331754, PMCID PMC9859842.
Gallo G, Martellucci J, Sturiale A, Clerico G, Milito G, Marino F. Consensus statement of the Italian society of colorectal surgery (SICCR): management and treatment of hemorrhoidal disease. Tech Coloproctol. 2020;24(2):145-64. doi: 10.1007/s10151-020-02149-1, PMID 31993837, PMCID PMC7005095.
Van Tol RR, Van Zwietering E, Kleijnen J, Melenhorst J, Stassen LP, Dirksen CD. Towards a core outcome set for hemorrhoidal disease-a systematic review of outcomes reported in literature. Int J Colorectal Dis. 2018;33(7):849-56. doi: 10.1007/s00384-018-3046-2, PMID 29680897, PMCID PMC6002447.
Davis BR, Lee Kong SA, Migaly J, Feingold DL, Steele SR. The american society of colon and rectal surgeons clinical practice guidelines for the management of hemorrhoids. Dis Colon Rectum. 2018;61(3):284-92. doi: 10.1097/DCR.0000000000001030, PMID 29420423.
Sheikh P, Regnier C, Goron F, Salmat G. The prevalence characteristics and treatment of hemorrhoidal disease: results of an international web-based survey. J Comp Eff Res. 2020;9(17):1219-32. doi: 10.2217/cer-2020-0159, PMID 33079605.
Sandler RS, Peery AF. Rethinking what we know about hemorrhoids. Clin Gastroenterol Hepatol. 2019;17(1):8-15. doi: 10.1016/j.cgh.2018.03.020, PMID 29601902.
Ergul A, Li W, Elgebaly MM, Bruno A, Fagan SC. Hyperglycemia diabetes and stroke: focus on the cerebrovasculature. Vascul Pharmacol. 2009;51(1):44-9. doi: 10.1016/j.vph.2009.02.004, PMID 19258053, PMCID PMC3732462.
Marcovecchio ML, Lucantoni M, Chiarelli F. Role of chronic and acute hyperglycemia in the development of diabetes complications. Diabetes Technol Ther. 2011;13(3):389-94. doi: 10.1089/dia.2010.0146, PMID 21299400.
Hu WS, Lin CL. Hemorrhoid risk among people with diabetes mellitus using glucose-lowering agents. Naunyn Schmiedebergs Arch Pharmacol. 2025;398(12):17479-85. doi: 10.1007/s00210-025-04272-y, PMID 40504374.
Levy L, Smiley A, Latifi R. Independent predictors of in-hospital mortality in elderly and non-elderly adult patients undergoing emergency admission for hemorrhoids. Am Surg. 2022;88(5):936-42. doi: 10.1177/00031348211060420, PMID 34978210.
Dirir AM, Daou M, Yousef AF, Yousef LF. A review of alpha-glucosidase inhibitors from plants as potential candidates for the treatment of type-2 diabetes. Phytochem Rev. 2022;21(4):1049-79. doi: 10.1007/s11101-021-09773-1, PMID 34421444.
Chen Y, Xiao Y, Lian G, Yi J, Liu X. Pneumatosis intestinalis associated with alpha-glucosidase inhibitors: a pharmacovigilance study of the FDA adverse event reporting system from 2004 to 2022. Expert Opin Drug Saf. 2023. p. 1-10. doi: 10.1080/14740338.2023.2278708, PMID 37929311.
Maidadi B, Ntchapda F, Miaffo D, Kamgue Guessom O. Efficacy of Rytigynia senegalensis Blume on free radical scavenging inhibition of alpha-amylase and alpha-glucosidase activity and blood glucose level. Evid Based Complement Alternat Med. 2022;2022(1):9519743. doi: 10.1155/2022/9519743, PMID 36204130, PMCID PMC9532085.
Agrawal J, Kar A. Chronic treatment of diabecon in the regulation of alloxan-induced hyperglycemia and oxidative stress in different tissues of adolescent diabetic rats. Int J Pharm Pharm Sci. 2014;6(4):83-7.
Atawodi SE, Atawodi JC. Azadirachta indica (neem): a plant of multiple biological and pharmacological activities. Phytochem Rev. 2009;8(3):601-20. doi: 10.1007/s11101-009-9144-6.
Kumar S, Kamboj J, Suman S, Sharma S. Overview for various aspects of the health benefits of Piper longum linn. fruit. J Acupunct Meridian Stud. 2011;4(2):134-40. doi: 10.1016/S2005-2901(11)60020-4, PMID 21704957.
Neharkar PS, Matre YB, Lad AG, Zanwar PR, Khandare RY. Occurrence of giant African snail, Achatina fulica Bowdich in soybean growing areas of Maharashtra and its management strategies: a review. J Entomol Res. 2025;49(2):537-41. doi: 10.5958/0974-4576.2025.00090.5.
Brownlee M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes. 2005;54(6):1615-25. doi: 10.2337/diabetes.54.6.1615, PMID 15919781.
Giacco F, Brownlee M. Oxidative stress and diabetic complications. Circ Res. 2010;107(9):1058-70. doi: 10.1161/CIRCRESAHA.110.223545, PMID 21030723.
Anush N, Sukhdeep K, Riju M, Pranav Kumar P. Prevalence of small dense ldl in type 2 diabetes mellitus and its correlation with inflammatory markers: a comprehensive study. Asian J Pharm Clin Res. 2025;18(4):161-6. doi: 10.22159/ajpcr.2025v18i4.54592.
Phongpradist R, Chaiyana W, Anuchapreeda S. Curcumin-loaded multi-valent ligand conjugated nanoparticles for anti-inflammatory activity. Int J Pharm Pharm Sci. 2015;7(4):203-8.
Donath MY, Shoelson SE. Type 2 diabetes as an inflammatory disease. Nat Rev Immunol. 2011;11(2):98-107. doi: 10.1038/nri2925, PMID 21233852.
Kaur R, Kaur M, Singh J. Endothelial dysfunction and platelet hyperactivity in type 2 diabetes mellitus: molecular insights and therapeutic strategies. Cardiovasc Diabetol. 2018;17(1):121. doi: 10.1186/s12933-018-0763-3, PMID 30170601.
Koteshwar K, Rakesh KT, Koushik NV. Comparative assessment of ultrasound-guided bilateral erector spinae plane block and local anaesthetic infiltration for perioperative pain management in lumbar spine fusion surgeries. Int J Curr Pharm Res. 2025;17(6):25-9. doi: 10.22159/ijcpr.2025v17i6.7063.
Pamecha S, Sharma R, Khan J, Barya S. A comparative study to analyse the effect of exercise vs standard antenatal care in back pain during the third trimester in primigravida females. Int J Curr Pharm Res. 2024;16(5):37-40. doi: 10.22159/ijcpr.2024v16i5.5055.
Alara OR, Abdurahman NH, Ukaegbu CI. Soxhlet extraction of phenolic compounds from Vernonia cinerea leaves and its antioxidant activity. J Appl Res Med Aromat Plants. 2018;11:12-7. doi: 10.1016/j.jarmap.2018.07.003.
Nallajerla SK, Ganta S. Croton oil-induced hemorrhoidal rats: evaluation of anti-inflammatory mediated anti-hemorrhoidal activity of Tridax procumbens whole plant. Curr Bioact Compd. 2023;19(7):17-28. doi: 10.2174/1573407219666230207105946.
Tripathi RK, Bolegave SS, Shetty PA, Uchil DA, Rege NN, Chawda MB. Efficacy and safety of a polyherbal formulation in hemorrhoids. J Ayurveda Integr Med. 2015;6(4):225-32. doi: 10.4103/0975-9476.172382, PMID 26834421.
Waithe OlY, Peng X, Childs EW, Tharakan B. Measurement of blood-brain barrier hyperpermeability using Evans blue extravasation assay. Vascular hyperpermeability: methods and protocols: springer; 2023. p. 177-84.
Amdekar S, Roy P, Singh V, Kumar A, Singh R, Sharma P. Anti‐inflammatory activity of Lactobacillus on carrageenan-induced paw edema in male wistar rats. Int J Inflam. 2012;2012(1):752015. doi: 10.1155/2012/752015, PMID 22518342.
Sadeghi H, Hajhashemi V, Minaiyan M, Movahedian A, Talebi A. Further studies on the anti-inflammatory activity of maprotiline in carrageenan-induced paw edema in rat. Int Immunopharmacol. 2013;15(3):505-10. doi: 10.1016/j.intimp.2013.01.018, PMID 23415748.
Smrutiranjan D, Rajasekaran S. Phytochemical characterization of Urochloa distachya (l.) through gas chromatography-mass spectrometry and liquid chromatography-mass spectrometry analysis with in silico and in vivo anti-inflammatory assessment in carrageenan-induced paw edema. Asian J Pharm Clin Res. 2025;18(4):199-222. doi: 10.22159/ajpcr.2025v18i4.53861.
Baba H, Bunu SJ. Spectroscopic and molecular docking analysis of phytoconstituent isolated from Solenostemon monostachyus as potential cyclooxygenase enzymes inhibitor. Int J Chem Res. 2025;9(1):1-6. doi: 10.22159/ijcr.2025v9i1.241.
Smith WL, DeWitt DL, Garavito RM. Cyclooxygenases: structural cellular and molecular biology. Annu Rev Biochem. 2000;69(1):145-82. doi: 10.1146/annurev.biochem.69.1.145, PMID 10966456.
Haeggstrom JZ, Funk CD. Lipoxygenase and leukotriene pathways: biochemistry biology and roles in disease. Chem Rev. 2011;111(10):5866-98. doi: 10.1021/cr200246d, PMID 21936577.
Lohsiriwat V. Hemorrhoids: from basic pathophysiology to clinical management. World J Gastroenterol. 2012;18(17):2009-17. doi: 10.3748/wjg.v18.i17.2009, PMID 22563187.
Jain S, Arora A, Paliwal V. Bark of woody plants of India as a source of phenolics and their promising biological activities. Natural products: phytochemistry botany metabolism of alkaloids phenolics and terpenes: springer; 2025. p. 1-55.
Jeba Malar TR, Antonyswamy J, Vijayaraghavan P, Ock Kim Y, Al-Ghamdi AA, Elshikh MS. In vitro phytochemical and pharmacological bio-efficacy studies on Azadirachta indica A. Juss. and melia azedarach linn for anticancer activity. Saudi J Biol Sci. 2020;27(2):682-8. doi: 10.1016/j.sjbs.2019.11.024, PMID 32210688.
Pandey KB, Rizvi SI. Plant polyphenols as dietary antioxidants in human health and disease. Oxid Med Cell Longev. 2009;2(5):270-8. doi: 10.4161/oxim.2.5.9498, PMID 20716914.