
Int J App Pharm, Vol 18, Issue 2, 2026, 424-435Original Article
POLOXAMER-188 BASED CURCUMIN NANOEMULSION: FORMULATION, CHARACTERIZATION, AND STABILITY FOR ENTRAPMENT EFFICIENCY
ZULKIFLI B. POMALANGO1,2, MOHAMAD APRIANTO PANEO3*, MUCHTAR NORA ISMAIL SIREGAR4, ZULFIAYU SAPIUN5, KARMILA LIHAWA3, NURAIN THOMAS3
1Department of Nursing, Faculty of Sports and Health, Universitas Negeri Gorontalo, Gorontalo-96128, Indonesia. 2Doctoral Program in Nursing Science, Faculty of Nursing, Universitas Indonesia, Indonesia. 3Department of Pharmacy, Faculty of Sports and Health, Universitas Negeri Gorontalo, Gorontalo-96128, Indonesia. 4Faculty of Medicine, Universitas Negeri Gorontalo, Gorontalo-96128, Indonesia. 5Department of Pharmacy, Health Polytechnic of Gorontalo, Indonesia
*Corresponding author: Mohamad Aprianto Paneo; *Email: apriyanto07@ung.ac.id
Received: 28 Oct 2025, Revised and Accepted: 09 Feb 2026
ABSTRACT
Objective: This study aimed to develop and characterize a curcumin-loaded nanoemulsion formulation to improve curcumin's solubility and bioavailability using poloxamer 188, dimethyl sulfoxide (DMSO), tween-80, and polyethylene glycol 400 (PEG-400) as excipients.
Methods: Ten curcumin-loaded nanoemulsion formulations were optimized based on excipient combinations. These formulations were subjected to freeze-thaw cycles, particle size analysis, polydispersity index (PDI), zeta potential, differential scanning calorimetry (DSC), Fourier transform infrared spectroscopy (FTIR), transmission electron microscopy (TEM), high-performance liquid chromatography (HPLC), and entrapment efficiency analysis to evaluate stability, solubility, and curcumin encapsulation efficiency.
Results: The study identified the optimal formulations (F5 to F8) based on their superior physical stability, as evidenced by minimal changes in viscosity and pH during freeze-thaw cycles. The entrapment efficiency of curcumin in these formulations ranged from 87.55% to 77.98%, indicating effective encapsulation. Characterization techniques, including DSC, HPLC, and FTIR, confirmed the stable incorporation of curcumin into the nanoemulsion with only minor structural modifications. The best formulation was demonstrated by Formula 5 with an average particle size of 195.3 nm, PDI of 0.36, and zeta potential of-16.2 mV. This formula showed uniform particle distribution and moderate colloidal stability among the other formulas. The TEM image showed spherical nanoparticles with smooth surfaces and consistent size distribution. Additionally, HPLC analysis of pure curcumin and nanoemulsion showed similar chromatographic profiles, confirming the stability and preservation of curcumin during the formulation process.
Conclusion: The optimized curcumin-loaded nanoemulsion demonstrated enhanced solubility and stability, with entrapment efficiency (EE) values ranging from 87.55% to 77.98%, indicating effective encapsulation of curcumin within the nanoemulsion system.
Keywords: Curcumin, Nanoemulsion, Poloxamer-188, Particle size, Entrapment efficiency
© 2026 The Authors.Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i2.57293 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Curcumin, the principal bioactive component of Curcuma longa, has attracted significant scientific attention owing to its diverse pharmacological activities, including antioxidant, anti-inflammatory, antimicrobial, anticancer, and neuroprotective effects. These therapeutic potentials highlight curcumin as a promising candidate for various clinical applications, ranging from chronic inflammatory disorders to degenerative diseases [1-3].
Despite its broad pharmacological relevance, the clinical application of curcumin remains limited due to its inherently poor aqueous solubility, rapid metabolism, and low systemic bioavailability. Orally administered curcumin typically undergoes extensive first-pass metabolism, resulting in minimal plasma concentrations and restricted therapeutic outcomes. Consequently, strategies to improve its solubility and bioavailability are urgently needed to enable its translation into effective pharmaceutical formulations [4-6].
Among various drug delivery approaches, Nanoemulsion systems, due to their unique properties such as large surface area, kinetic stability, and improved permeability, have emerged as a promising platform for enhancing the solubility and bioavailability of hydrophobic bioactives like curcumin. Nanoemulsions consist of oil, surfactant, cosurfactant, and aqueous phases organized into droplets at the nanometer scale. Their unique characteristics, such as large surface area, kinetic stability, and improved permeability, make them suitable for delivering poorly soluble bioactives such as curcumin [7-9].
Several studies have demonstrated that curcumin nanoemulsions significantly enhance drug solubility and gastrointestinal absorption compared to conventional formulations. Additionally, nanoemulsions protect the active compound from environmental degradation, thereby improving stability during storage and administration. However, challenges remain in developing a robust formulation with optimized droplet size distribution, surface charge, and long-term stability [10, 11].
Excipient selection is a critical determinant in the success of nanoemulsion systems. Poloxamer-188, a non-ionic block copolymer, has been widely used in nanoemulsion systems for its stabilizing properties. This excipient helps to reduce interfacial tension and provides steric hindrance, preventing droplet aggregation. Specifically, its use in curcumin nanoemulsions helps to improve both the physical stability and the uniformity of the emulsions, critical for maintaining curcumin's solubility and bioavailability [12, 13].
Dimethyl sulfoxide (DMSO) has been employed as a cosolvent to enhance the solubility of hydrophobic drugs. Its amphiphilic nature allows curcumin to be solubilized effectively before incorporation into the nanoemulsion system. Furthermore, DMSO contributes to maintaining drug integrity during the formulation process, reducing the risk of precipitation and phase separation [14].
Tween-80, a non-ionic surfactant, is another essential excipient in nanoemulsion preparation due to its high emulsification efficiency and biocompatibility. Its hydrophilic-lipophilic balance (HLB) value enables effective stabilization of oil-in-water dispersions, which is particularly advantageous in improving curcumin solubilization and uniform distribution within the aqueous medium [15, 16].
Polyethylene glycol 400 (PEG-400) serves as a cosolvent and solubilizer, contributing to the hydrophilic environment necessary for nanoemulsion stability. Its low toxicity and compatibility with pharmaceutical formulations make PEG-400 a valuable component in enhancing dispersion and maintaining nanoemulsion integrity under storage conditions [17, 18].
Although previous studies have demonstrated curcumin nanoemulsions' effectiveness, our study aims to address the gap by combining Poloxamer-188, DMSO, Tween-80, and PEG-400, hypothesizing that this specific combination will optimize curcumin’s solubilization, stability, and emulsification, offering a more efficient formulation for improving curcumin’s bioavailability and therapeutic efficacy [11, 19, 20].
Based on this rationale, the present study aims to develop and characterize a curcumin nanoemulsion formulated with Poloxamer-188, DMSO, Tween-80, and PEG-400. Comprehensive physicochemical evaluation, including particle size, polydispersity index, zeta potential, morphology, encapsulation efficiency, IR spectrum, and stability analysis, was performed. The findings of this study are expected to provide a strong scientific basis for advancing curcumin delivery strategies and contribute to the broader development of nanotechnology-based drug delivery systems.
MATERIALS AND METHODS
Materials
Curcumin (≥95% purity) used in this study was sourced from Xi'an Prius Biological Engineering Co., Ltd., Xi'an, China. Poloxamer-188, was also supplied by Xi'an Prius Biological Engineering Co., Ltd., Xi'an, China. Dimethyl sulfoxide (DMSO), Sigma Aldrich, St. Louis, USA. Olive oil, Alfa Laval, Gothenburg, Sweden. Tween 80, Sigma Aldrich, St. Louis, USA. Polyethylene glycol 400 (PEG-400), Sigma Aldrich, St. Louis, USA. Distilled water (Aquadest) Damhil Medical Store, Gorontalo, Indonesia. Ethanol 95%, Gorontalo, Indonesia.
Equipment
The curcumin nanoemulsion was prepared and characterized using a range of analytical and laboratory instruments. Particle size and polydispersity index (PDI) were measured using a particle size analyzer (Horiba), and zeta potential was determined using the same instrument to evaluate colloidal stability. Molecular interactions between curcumin and the excipients were assessed by Fourier-transform infrared (FTIR) spectroscopy using an FTIR spectrometer (Thermo Scientific). Curcumin content and encapsulation efficiency were quantified using an ultraviolet-visible (UV-Vis) spectrophotometer (Thermo Scientific). Phase separation and purification were performed using a Mascot centrifuge, and homogenization of the aqueous and organic phases was carried out using a Mag HS magnetic stirrer combined with a Mag HS Ultra-Turrax high-shear homogenizer. Stability studies were conducted under controlled temperature conditions using a thermal climatic chamber. The rheological properties of the nanoemulsion were measured using a Brookfield DV2T viscometer. Thermal behavior and crystallinity were evaluated by differential scanning calorimetry (DSC) using a Hitachi NEXTA DSC instrument (Japan). Quantification by high-performance liquid chromatography (HPLC) was performed using an HPLC system (Thermo Scientific). Morphology was examined by transmission electron microscopy (TEM) using a Hitachi HT7700 (Japan).
Optimization of nanoemulsion base
The preparation of the nanoemulsion base began by formulating 10 different variations of the formulation with distinct combinations of excipients. The purpose of these different combinations was to determine which formulation yielded a transparent and stable physical structure. Stability was assessed using centrifugation at 3000 RPM, 7000 RPM, 8000 RPM, and 12000 RPM to evaluate the effect of shear force on the formulations. Furthermore, the formulations were subjected to freeze-thaw stability tests at 4 °C and 40 °C with a relative humidity (RH) of 75%, conducted over 7 cycles with 48 h intervals. The stability tests were performed in a controlled temperature chamber (Climatic Chamber) to ensure consistent conditions throughout the process. The best qualifying formula will be supplemented with 10 mg of curcumin [21, 34, 35]. The details of the 10 formulation variations are presented in table 1 below.
Table 1: Optimization of nanoemulsion base variations
| Material | Formula (%) |
| F1 | |
| Olive oil | 5 |
| Tween-80 | 10 |
| PEG-400 | 7.5 |
| Poloxamer-188 | 2 |
| Dimetilsulfoksida | 1.5 |
| Aquadest | Ad 100 |
Preparation of curcumin nanoemulsion formula
The preparation of the curcumin nanoemulsion formula began with the preparation of the nanoemulsion base. Olive oil, curcumin, and Tween-80 were mixed using a magnetic stirrer at 100 RPM for one hour. Subsequently, a mixture of PEG-400 and Dimethyl Sulfoxide (DMSO) was slowly added to the previous mixture. Poloxamer-188 was then dissolved in an appropriate amount of water and introduced into the mixture. The resulting mixture was titrated with distilled water at a rate of one drop every 20 seconds. Once a transparent nanoemulsion was formed, the mixture was subjected to sonication at 40 kHz for 1 h, followed by centrifugation at 12,000 RPM [38].
Physical stability testing of curcumin nanoemulsion
The physical stability of the curcumin nanoemulsion was evaluated using a freeze-thaw protocol, where the samples were alternated repeatedly between 4 °C and 40 °C. The freeze-thaw test was performed by alternating storage at 4 °C and 40 °C. One cycle was defined as 24 h at 4 °C followed by 24 h at 40 °C (48 h per cycle). The relative humidity (RH) was maintained at 75% during the freeze-thaw cycles [26].
Wavelength analysis of curcumin nanoemulsion
The λ max of curcumin in the nano emulsion was found to be 430 nm, consistent with the known absorption characteristics of curcumin. A series of standard concentrations ranging from 2 ppm to 8 ppm were prepared to construct a calibration curve. The absorbance values were recorded for each concentration, and the corresponding wavelength of the nanoemulsion was analyzed based on the absorption peak within the specified wavelength range [20-23, 28].
High-performance liquid chromatography (HPLC) analysis of curcumin stability
To evaluate the stability of curcumin in the nanoemulsion over time, the curcumin concentration was monitored using high-performance liquid chromatography (HPLC). A reverse-phase HPLC system was used for the analysis. The HPLC method employed a C18 column (4.6 mm × 150 mm, 5 µm) with a mobile phase composed of acetonitrile and water (80:20, v/v) at a flow rate of 1.0 ml/min.
Particle size, polydispersity index and zeta potential analysis
The particle size, polydispersity index (PDI), and zeta potential of the curcumin nanoemulsion were determined using a particle size analyzer (PSA). The particle size distribution was measured in the range of 20-500 nm. A PDI value of less than 0.5 was considered indicative of a narrow particle size distribution, suggesting uniformity of the emulsion. The zeta potential was measured to assess the colloidal stability of the nanoemulsion, with values approximately close to ±30 mV indicating good stability [21, 24, 25].
Thermal characterization using differential scanning calorimetry (DSC)
The thermal properties of curcumin, the nanoemulsion base, and the curcumin nanoemulsion were analyzed by differential scanning calorimetry (DSC) over a temperature range of 50 °C to 410 °C. This analysis was performed to evaluate the heat capacity and to assess any thermal transitions such as melting, crystallization, or other phase changes in the materials. The DSC thermograms were recorded to provide insights into the thermal behavior and stability of both the curcumin and the formulated nanoemulsion.
Functional group analysis using fourier transform infrared spectroscopy (FTIR)
The prepared grids were then analyzed using a Transmission Electron Microscope (TEM) (TEM HT7700) at magnifications of 10000x, 15000x, 50000x and 70000x to observe the morphology and size distribution of the curcumin nanoemulsion droplets. Images were captured at each magnification to examine the nanoscale structure of the emulsion droplets and confirm their uniformity
Entrapment efficiency determination using dialysis bag
Entrapment efficiency (EE) of curcumin-loaded nanoemulsions was evaluated using a dialysis bag method, which indirectly reflects EE by measuring curcumin released through the membrane. A 12,000 Da MWCO dialysis bag containing 1 ml nanoemulsion was immersed in 100 ml PBS (pH 7.4) and incubated at 37 °C with agitation (50 rpm). Samples were collected at 10, 20, 40, 60, 120, and 180 min, and curcumin in the external medium was quantified by UV-Vis at 425 nm. This approach may overestimate EE because it cannot fully differentiate free from encapsulated curcumin [30, 31].
Data analysis
Data analysis was performed using the statistical software SPSS (Statistical Package for the Social Sciences) version 30. To test the differences between groups on the pH and viscosity variables, One-Way ANOVA was used. This method allows for the comparison of means between more than two independent groups. In this study, One-Way ANOVA was applied to evaluate the differences in pH and viscosity across various nanoemulsion formulations tested during multiple freeze-thaw cycles. If the One-Way ANOVA results indicated a significant difference between groups (p<0.05), the analysis was followed by post-hoc Tukey HSD (Honest Significant Difference) testing. The Tukey HSD test was used to determine which specific groups differed significantly in terms of pH and viscosity. All statistical tests were conducted at a significance level of 5% (α = 0.05). The data used in the analysis included the mean pH and viscosity values for each formulation, which were presented in tables and graphs to illustrate the differences between groups and trends observed during the freeze-thaw cycles.
RESULTS AND DISCUSSION
Optimization of nanoemulsion base variations
The optimization of the nanoemulsion base variations was carried out by preparing 10 different formulations with varying combinations of excipients. Upon visual inspection, Formulas 1 to 4 exhibited a cloudy appearance, indicating instability or improper emulsification. In contrast, Formulas 5 through 8 displayed a clear, transparent appearance, suggesting successful emulsification and improved stability, However, formulas 9 to 10 are very thick. These results highlight the importance of optimizing the composition of excipients to achieve the desired physical properties and stability of the nanoemulsion. Fig. 1. Physical appearance of formulations F1-F10, showing the visual clarity and homogeneity of each formulation. Formulations F1-F4 exhibit cloudiness, indicating instability, while formulations F5-F8 are clear, fig. 2. Phase separation and stability of formulations F1-F10 observed after freeze-thaw cycles. Formulations F5-F8 demonstrate minimal phase separation, suggesting good physical stability, while F1-F4 show visible phase separation, indicating poor stability.

Fig. 1: Nanoemulsion base variations

Fig. 2: Nanoemulsion with curcumin 10 mg
Ten formulations were prepared to optimize curcumin-loaded nanoemulsions using different excipient combinations. Formulations F1-F4 were cloudy (unstable), whereas F5-F8 were transparent and remained stable during centrifugation and freeze-thaw testing, with minimal changes in appearance, viscosity, and pH. F9-F10 were overly viscous. After curcumin loading, F5-F8 stayed clear, supported by the roles of Poloxamer 188, Tween 80, PEG 400, and DMSO in emulsification and solubilization. Thus, F5-F8 were selected for further characterization; future studies should assess long-term stability, release, and in vivo performance [19, 36, 37, 39].
Physical stability testing of curcumin nanoemulsion
The physical stability of the four selected curcumin nanoemulsion formulations (Formulas 5, 6, 7 and 8) was evaluated over seven freeze-thaw cycles, with each cycle lasting 48 h. The samples were subjected to temperatures of 4 °C and 40 °C with a relative humidity (RH) of 75% using a Claimitic Chamber. During each cycle, parameters including viscosity and pH were monitored. The detailed results for color, viscosity, emulsion type, and pH across all cycles are summarized in fig. 3, fig. 4 and table 2.
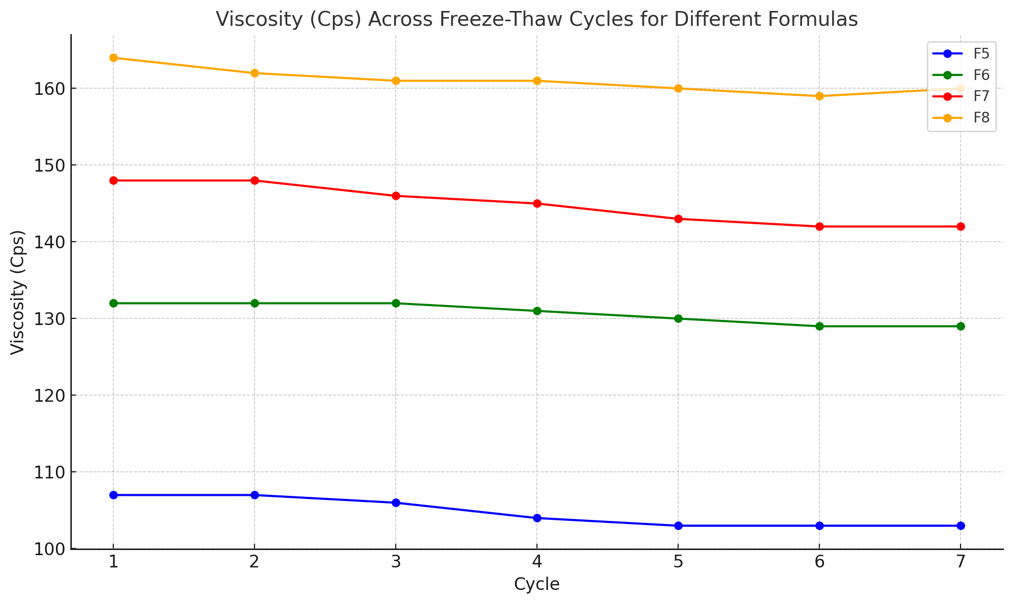
Fig. 3: Freeze-thaw stability testing of curcumin nanoemulsions, showing changes in viscosity after seven cycles of alternating between 4 °C and 40 °C
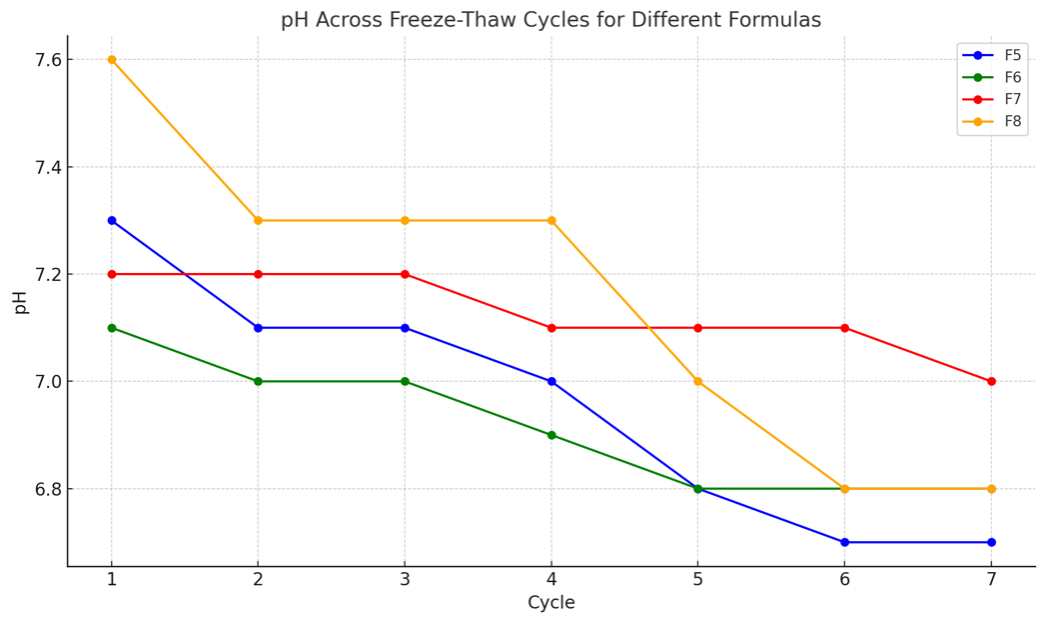
Fig. 4: Freeze-thaw stability testing of curcumin nanoemulsions, showing changes in pH after seven cycles of alternating between 4 °C and 40 °C
Table 2: Physical stability testing of curcumin nanoemulsion
| Cycle | Parameters | Results | |||
| F5 | F6 | F7 | F8 | ||
| 1 | Viscosity (Cps) pH |
107 7.3 |
132 7.1 |
148 7.2 |
164 7.6 |
| 2 | Viscosity (Cps) pH |
107 7.1 |
132 7.0 |
148 7.2 |
162 7.3 |
| 3 | Viscosity (Cps) pH |
106 7.1 |
132 7.0 |
146 7.2 |
161 7.3 |
| 4 | Viscosity (Cps) pH |
104 7.0 |
131 6.9 |
145 7.1 |
161 7.3 |
| 5 | Viscosity (Cps) pH |
103 6.8 |
130 6.8 |
143 7.1 |
160 7.0 |
| 6 | Viscosity (Cps) pH |
103 6.7 |
129 6.8 |
142 7.1 |
160 6.8 |
| 7 | Viscosity (Cps) pH |
103 6.7 |
129 6.8 |
142 7.0 |
159 6.8 |
| SD Viscosity | ±1.75 | ±1.28 | ±2.42 | ±1.51 | |
| SD pH | ±0.21 | ±0.11 | ±0.07 | ±0.28 | |
Note: Cps: centipoise, SD: standard deviation, Sample size: n = 3
Freeze-thaw testing showed a slight, gradual decrease in viscosity for formulations F5-F8 (F5: 107→103 Cps; F6: 132→129 Cps; F7: 148→142 Cps; F8: 164→160 Cps) and only minor pH fluctuations (F5: 7.3-6.7; F6: 7.1-6.8; F7: 7.2-7.0; F8: 7.6-6.8). Overall, the formulations remained physically stable across cycles, supported by low standard deviations for viscosity and pH. The results are presented in fig. 3-4 and table 2.
Table 3: Data analysis ANOVA of viscosity
| ANOVAof viscosity | |||||
| Sum of squares | df | Mean square | F | Sig. | |
| Between Groups | 11958.393 | 3 | 3986.131 | 1066.354 | <.001 |
| Within Groups | 89.714 | 24 | 3.738 | ||
| Total | 12048.107 | 27 | |||
Table 4: Multiple comparisons (Post-hoc) of viscosity
| Multiple comparisons of viscosity | ||||||
| Tukey HSD | ||||||
| (I) Formula | (J) Formula | Mean difference (I-J) | Std. Error | Sig. | 95% confidence interval | |
| Lower bound | Upper bound | |||||
| Formula 5 | Formula 6 | -26.0000* | 1.0335 | <.001 | -28.851 | -23.149 |
| Formula 7 | -40.1429* | 1.0335 | <.001 | -42.994 | -37.292 | |
| Formula 8 | -56.2857* | 1.0335 | <.001 | -59.137 | -53.435 | |
| Formula 6 | Formula 5 | 26.0000* | 1.0335 | <.001 | 23.149 | 28.851 |
| Formula 7 | -14.1429* | 1.0335 | <.001 | -16.994 | -11.292 | |
| Formula 8 | -30.2857* | 1.0335 | <.001 | -33.137 | -27.435 | |
| Formula 7 | Formula 5 | 40.1429* | 1.0335 | <.001 | 37.292 | 42.994 |
| Formula 6 | 14.1429* | 1.0335 | <.001 | 11.292 | 16.994 | |
| Formula 8 | -16.1429* | 1.0335 | <.001 | -18.994 | -13.292 | |
| Formula 8 | Formula 5 | 56.2857* | 1.0335 | <.001 | 53.435 | 59.137 |
| Formula 6 | 30.2857* | 1.0335 | <.001 | 27.435 | 33.137 | |
| Formula 7 | 16.1429* | 1.0335 | <.001 | 13.292 | 18.994 | |
Table 5: ANOVA of viscosity
| ANOVA of pH | |||||
| Sum of squares | df | mean square | F | Sig. | |
| Between Groups | .310 | 3 | .103 | 2.535 | .081 |
| Within Groups | .977 | 24 | .041 | ||
| Total | 1.287 | 27 | |||
Table 6: Multiple comparisons (Post-hoc) of pH
| Multiple comparisons of pH | ||||||
| Tukey HSD | ||||||
| (I) Formula | (J) Formula | Mean difference (I-J) | Std. error | Sig. | 95% Confidence Interval | |
| Lower bound | Upper bound | |||||
| Formula 5 | Formula 6 | .0429 | .1079 | .978 | -.255 | .340 |
| Formula 7 | -.1714 | .1079 | .403 | -.469 | .126 | |
| Formula 8 | -.2000 | .1079 | .274 | -.498 | .098 | |
| Formula 6 | Formula 5 | -.0429 | .1079 | .978 | -.340 | .255 |
| Formula 7 | -.2143 | .1079 | .221 | -.512 | .083 | |
| Formula 8 | -.2429 | .1079 | .138 | -.540 | .055 | |
| Formula 7 | Formula 5 | .1714 | .1079 | .403 | -.126 | .469 |
| Formula 6 | .2143 | .1079 | .221 | -.083 | .512 | |
| Formula 8 | -.0286 | .1079 | .993 | -.326 | .269 | |
| Formula 8 | Formula 5 | .2000 | .1079 | .274 | -.098 | .498 |
| Formula 6 | .2429 | .1079 | .138 | -.055 | .540 | |
| Formula 7 | .0286 | .1079 | .993 | -.269 | .326 | |
ANOVA showed a significant effect of formulation on viscosity (F = 1066.354, p<0.001), and Tukey HSD indicated several significant pairwise differences (p<0.05), with Formula 4 having the highest viscosity and differing most from the others. In contrast, pH did not differ significantly among formulations (ANOVA: F = 2.535, p = 0.081; Tukey: non-significant). Overall, viscosity distinguishes formulations, whereas pH remains relatively stable. Results are presented in tables 3-6.
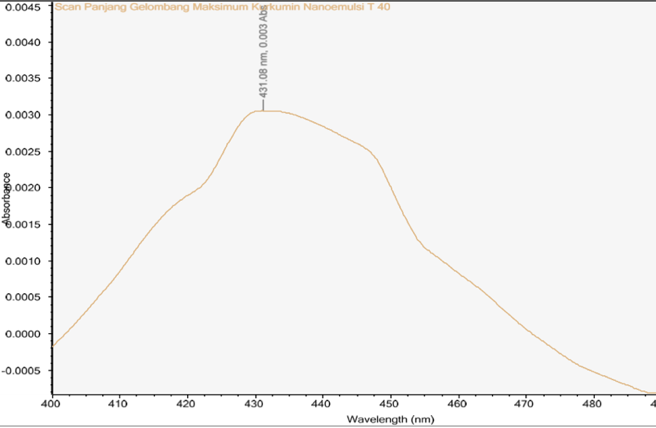
Fig. 5: UV-Vis spectrum of the curcumin nanoemulsion used for curcumin quantification
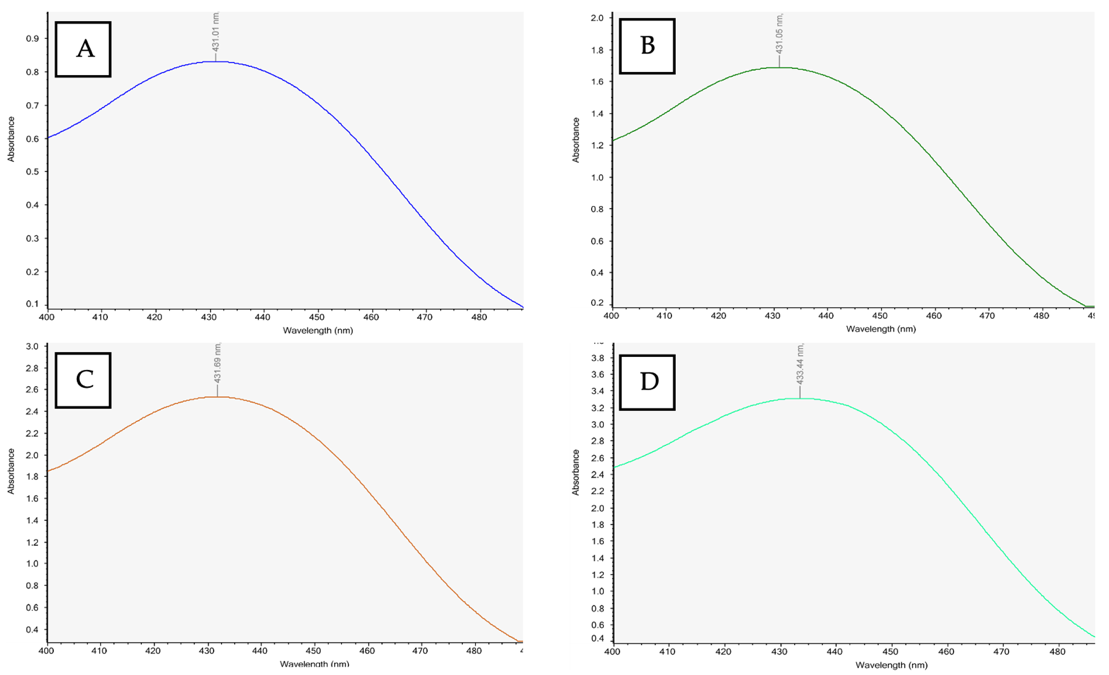
Fig. 6: UV-Vis spectra of standard curcumin solutions: (A) 2 ppm, (B) 4 ppm, (C) 6 ppm, and (D) 8 ppm
Qualitative test with spectrofotometer uv-vis
UV-Vis scanning showed a maximum absorbance at 431 nm for the curcumin nanoemulsion, while standard curcumin solutions (2-8 ppm) exhibited peaks at 430-433 nm (Fig. 3-4). Physical stability was evaluated by freeze-thaw testing for seven cycles alternating 4 °C and 40 °C (75% RH), assessing color, viscosity, emulsion type, and pH. Formulations F5-F8 remained transparent and maintained an O/W emulsion type throughout the cycles, with only a slight, gradual decrease in viscosity, indicating no major aggregation or phase separation [29]. Formulation F5 showed only a slight viscosity decrease (107 to 103 cps) from cycle 1 to cycle 7, with similar trends for F6-F8, indicating stable flow properties. The pH remained relatively constant across cycles (approximately 6.7-7.3), suggesting no meaningful chemical change. Overall, the freeze-thaw results indicate that formulations F5-F8 were physically stable under simulated storage conditions, with minimal changes in viscosity, pH, and emulsion type.
Particle size analysis of formulations F5-F8 measured particle size, polydispersity index (PDI), and zeta potential. All formulations were within the nano-size range with PDI<0.5, indicating a relatively uniform distribution. Zeta potential values (−16.2 to −25.6 mV) suggest moderate colloidal stability, likely supported by steric stabilization from non-ionic surfactants (Poloxamer 188 and Tween 80) rather than electrostatic repulsion alone. Results are shown in fig. 7-8 and table 7 [30, 31].
Particle size analysis of nanoemulsion formulations
Formulations F5-F8 were evaluated by particle size analysis for particle size, polydispersity index (PDI), and zeta potential. All were in the nano-size range with PDI<0.5, indicating a relatively uniform distribution. Zeta potential values (−16.2 to −25.6 mV) suggest moderate colloidal stability, likely aided by steric stabilization from non-ionic surfactants (Poloxamer 188 and Tween 80). Results are shown in fig. 7-8 and table 7.
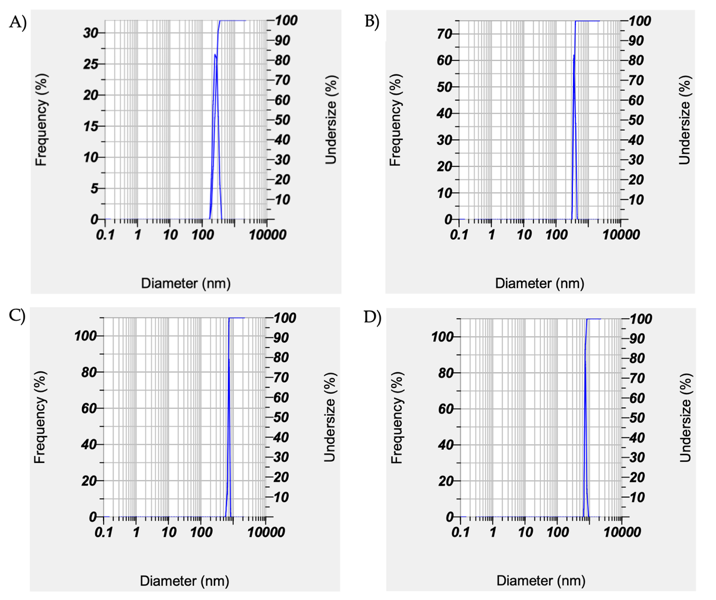
Fig. 7: Graph of diameter dan frequency of curcumin nanoemulsion using particle size analysis. A. F5 (195.3 nm), B. F6 (304.8 nm), C. F7 (514.1 nm) and D. F8 (703.7 nm)
Table 7: Analysis nanoemulsion particle size, polydispersity index (PDI), and zeta potential
| Formula | Z-Average (nm) | (PDI) | Zeta potential (mV) |
| F5 | 195.3±2.1 | 0.366± 0.05 | -16.2± 0.5 |
| F6 | 304.8± 2.0 | 0.702± 0.02 | -25.6± 0.1 |
| F7 | 514.1± 2.3 | 0.715± 0.03 | -6.7± 0.7 |
| F8 | 703.7± 2.4 | 0.738± 0.07 | -22.0± 0.3 |
Note: PDI = Polydispersity Index, Z-average = Z-average diameter, mV = Millivolts
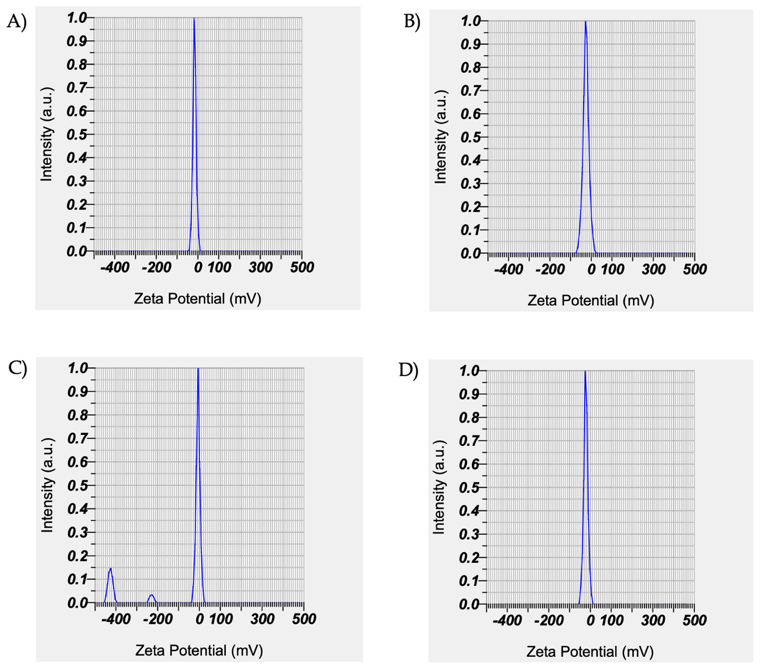
Fig. 8: Graph of intensity and zeta potential of curcumin nanoemulsion using particle size analysis. A. F5 (-16.2 mV), B. F6 (-25.6 mV), C. F7 (-6.7 mV) and D. F8 (-22.0 mV)
Particle size analysis of formulations F5-F8 (DLS, n = 3; mean±SD) showed sizes of 195.3±2.1 to 703.7±8.5 nm, with PDI 0.36±0.05 to 0.738±0.04 and zeta potential −16.2±0.5 to −25.6±0.8 mV, indicating moderate stability. Droplet size increased in F6-F8 with higher surfactant/cosurfactant ratios. Based on particle size, distribution, and zeta potential, F5 was selected for further characterization (DSC, FTIR, and entrapment efficiency).
Thermal characterization using differential scanning calorimetry (DSC)
DSC of pure curcumin (fig. 9A) showed a melting peak at 177.69 °C (onset 176.32 °C; endset 179.76 °C) with a heat change of −39.71 J/g, consistent with a crystalline structure. In the curcumin nanoemulsion (F5; fig. 9B), the main peak occurred at 177.34 °C with a markedly lower heat change (−11.87 J/g), suggesting partial conversion to an amorphous or molecularly dispersed state. A secondary transition near 87.05 °C was also observed, likely related to excipient thermal events (e. g., Poloxamer 188). F5 was selected for further analysis based on its favorable particle size, PDI, and zeta potential [27].
Functional group analysis using Fourier transform infrared spectroscopy (FTIR)
Fourier Transform Infrared (FTIR) spectroscopy was performed to evaluate the chemical structure and functional groups of curcumin, the nanoemulsion base (Formula 5), and the curcumin-loaded nanoemulsion (Formula 5). The FTIR spectra enabled the identification of characteristic absorption bands of curcumin and the assessment of potential interactions between curcumin and the excipients within the nanoemulsion system. The results provide insight into the chemical integrity of curcumin after incorporation into the nanoemulsion. The FTIR spectra of all samples are presented in fig. 10, and the detailed peak assignments are summarized in table 8.
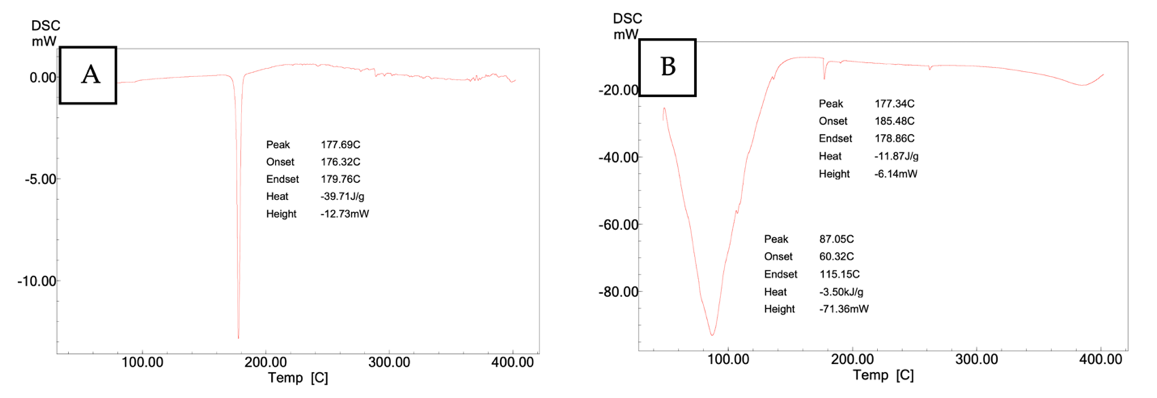
Fig. 9: Peak temperature using differential scanning calorimetry (DSC). A. curcumin, B. formula 5 (F5) nanoemulsion curcumin
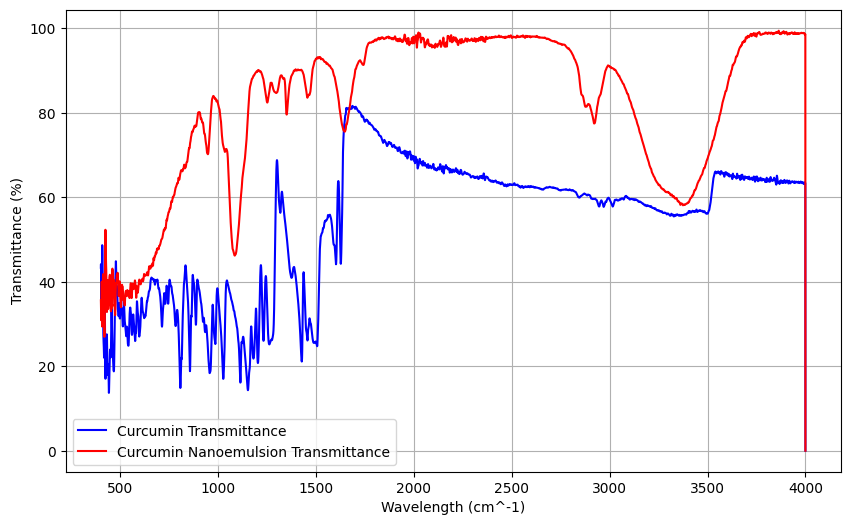
Fig. 10: Spectrum using fourier transform infrared spectroscopy FTIR curcumin (blue line) and formula 5 (F5) nanoemulsion curcumin (red line)
Table 8: Interpretation of FTIR spectrum of curcumin and nanoemulsion curcumin
| No | Wavelength (cm⁻¹) | Band | Interpretation |
| 1 | 944.92 | C-O bond | Characteristic of phenolic groups; slight intensity variation between pure curcumin and nanoemulsion. |
| 2 | 1033.37 | C-O vibration | Indicates interaction with emulsion components. |
| 3 | 1083.71 | C-O absorption | Present in both curcumin and nanoemulsion, confirming consistency in structure |
| 4 | 1249.02 | C-O-C vibration | Stable functional groups present in both curcumin and nanoemulsion. |
| 5 | 1296.74 | C-H vibration in phenyl group | Consistent structure in both curcumin and nanoemulsion, confirming aromatic integrity. |
| 6 | 1349.06 | CH₂ vibration | Maintained in both curcumin and nanoemulsion, indicating the preservation of molecular structure. |
| 7 | 1456.22 | C-H vibration in phenyl ring | Small changes suggesting slight structural modification after nanoemulsion formulation. |
| 8 | 1646.53 | C=C vibration in aromatic ring | Core structure remains stable in both curcumin and nanoemulsion. |
| 9 | 2920.92 | C-H vibration in aliphatic chains | Indicates the presence of emulsifier components in the nanoemulsion. |
| 10 | 3375.40 | O-H vibration | Retained hydroxyl groups, consistent in both curcumin and nanoemulsion, suggesting stability. |
FTIR spectra of pure curcumin and the curcumin-loaded nanoemulsion showed the characteristic curcumin bands (e. g., C-O at 944.92 cm⁻¹ and aromatic C-H at 1296.74 cm⁻¹), indicating preserved structural features. In the nanoemulsion, slight shifts and intensity changes (e. g., C-O to 969.77 cm⁻¹) suggest interactions with excipients, likely hydrogen bonding, consistent with molecular dispersion. Additional bands around 2920.92 cm⁻¹ reflect contributions from the emulsifier components, supporting curcumin incorporation into the nanoemulsion [21].
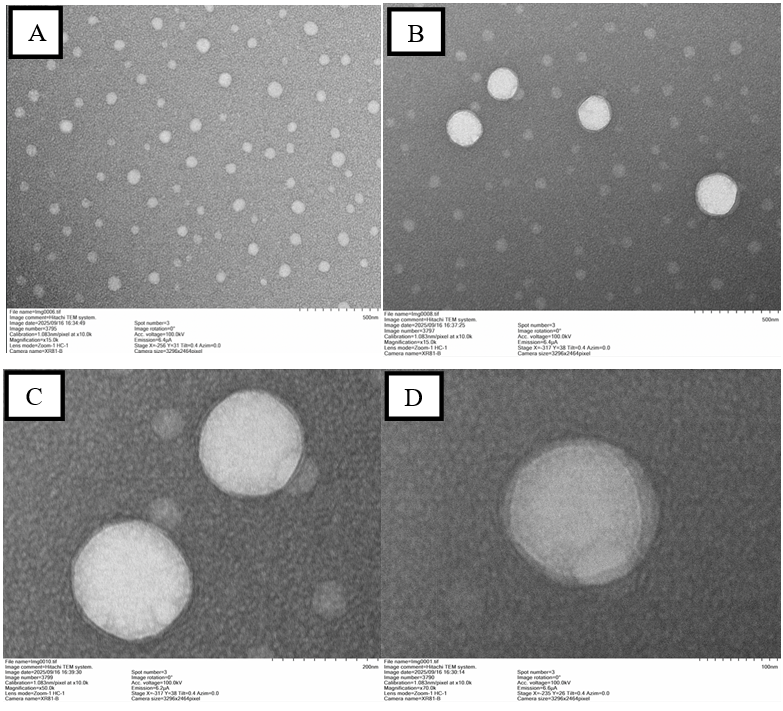
Fig. 11: TEM image of curcumin-loaded nanoemulsion (F5 formulation) showing spherical nanoparticles with a smooth surface and uniform size distribution. The particle size is approximately 195.3 nm as observed at 50,000x magnification. Scale bar represents 500 nm (Panel A and B), 200 nm (Panel C) and 100 nm (Panel D)
Analysis of transmission electron microscopy (TEM) imaging of curcumin nanoemulsion
TEM images showed well-dispersed, spherical nanoparticles with minimal aggregation across all magnifications (10,000×-70,000×), indicating a uniform and stable nanoemulsion. Higher magnifications confirmed smooth particle surfaces and consistent morphology, supporting successful formulation for potential pharmaceutical applications. Can be seen in fig. 11.
Table 9: Calibration standard of curcumin
| No | Injection (ppm) | Ret. time* min | Area mAU*min | Height mAU | Amount |
| 1 | 10 | 1.333 | 26.721 | 49.297 | 15.996 |
| 2 | 25 | 1.367 | 27.561 | 48.743 | 16.249 |
| 3 | 50 | 1.280 | 26.571 | 47.354 | 15.950 |
| 4 | 75 | 1.323 | 56.189 | 106.14 | 24.868 |
| 5 | 100 | 1.292 | 120.68 | 235.82 | 44.288 |
| Average | 1.319 | ||||
| Standard deviation 0.031 | |||||
| % RSD 2.34 | |||||
| R-Square 0.99 |
Note: RSD = Relative standard deviation
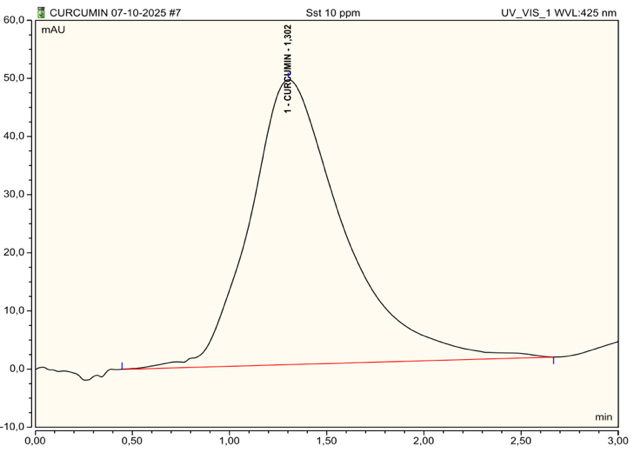
Fig. 12: High-performance liquid chromatography (HPLC) chromatograms of curcumin confirming the stability and integrity of curcumin in the nanoemulsion
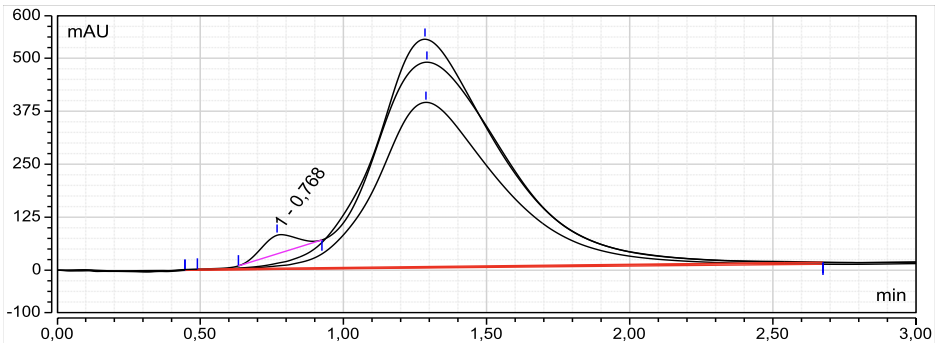
Fig. 13: High-performance liquid chromatography (HPLC) chromatograms of curcumin-loaded nanoemulsion, confirming the stability and integrity of curcumin in the nanoemulsion
Table 10: Retention time poloxamer 188-based curcumin nanoemulsion
| No | Replication | Ret. Time*min | Rel Area % |
| 1 | Replication 1 | 1.288 | 100 |
| 2 | Replication 2 | 1.285 | 100 |
| 3 | Replication 3 | 1.292 | 97.87 |
| Average | 1.29 | 99.29 | |
| Standard deviation 0.0029 1.004 | |||
| % RSD 0.26% 1.24 |
Note: n = 3, RSD = Relative standard deviation
High-performance liquid chromatography (HPLC) analysis of curcumin stability
HPLC chromatograms showed a well-defined curcumin peak at ~1.3 min in the pure standard (fig. 12) and a comparable peak in the nanoemulsion (fig. 13) across three injections, with no meaningful shift in retention time or peak loss, suggesting curcumin remains chemically intact after formulation with Poloxamer 188. The HPLC method was validated with good linearity (R²>0.99) and acceptable accuracy/precision, with RSD 2.34% (standard) and 0.26% (nanoemulsion) (<5%). To strengthen the stability claim, we added chromatograms of stressed samples (heat, light, and pH challenge) to demonstrate the method’s ability to detect degradation products and monitor formulation changes over time.
Furthermore, the consistency observed across the three tests highlights the reproducibility and stability of the nanoemulsion formulation. The absence of degradation or significant peak area fluctuation suggests that the curcumin encapsulated in the nanoemulsion retains its integrity even after being subjected to the formulation process. This finding supports the hypothesis that nanoemulsions, specifically those stabilized with Poloxamer-188, can effectively preserve the bioactive properties of curcumin, making it a promising carrier system for improving the solubility and bioavailability of curcumin in therapeutic applications. Calibration Standard of Curcumin can be seen in table 9, Chromatograms of curcumin can be seen in fig. 12, and Chromatograms of curcumin-loaded nanoemulsion can be seen in fig. 13.
Entrapment efficiency analysis of curcumin nanoemulsion
The entrapment efficiency of curcumin nanoemulsion was evaluated at different time intervals. As shown in table 10 and fig. 14, the entrapment efficiency was observed to be high across all time points, indicating the successful encapsulation of curcumin in the nanoemulsion.
Table 10: Entrapment efficiency (EE) of curcumin-loaded nanoemulsions determined by the dialysis bag method (mean±SD, n = 3)
| No | Time (Min) | Entrapment efficiency (%) |
| 1 | 10 | 87.55 |
| 2 | 20 | 85.22 |
| 3 | 40 | 83.12 |
| 4 | 60 | 80.45 |
| 5 | 80 | 79.23 |
| 6 | 120 | 77.98 |
The entrapment efficiency of curcumin in the nanoemulsion formulations was evaluated over time (mean±SD, n = 3), with values ranging from 87.55% to 77.98% across the sampling points (10, 20, 40, 60, 80, and 120 min). At the early time points (10 and 20 min), high entrapment efficiencies (87.55% and 85.22%) indicate effective encapsulation of curcumin within the nanoemulsion droplets, which supports stability and minimizes premature release. The gradual decrease in entrapment efficiency, particularly after 80 min, may reflect curcumin leakage or partitioning from the nanoemulsion matrix due to diffusion and/or progressive destabilization over the test duration. Can be seen ini table 10 and fig. 14 [33, 41].
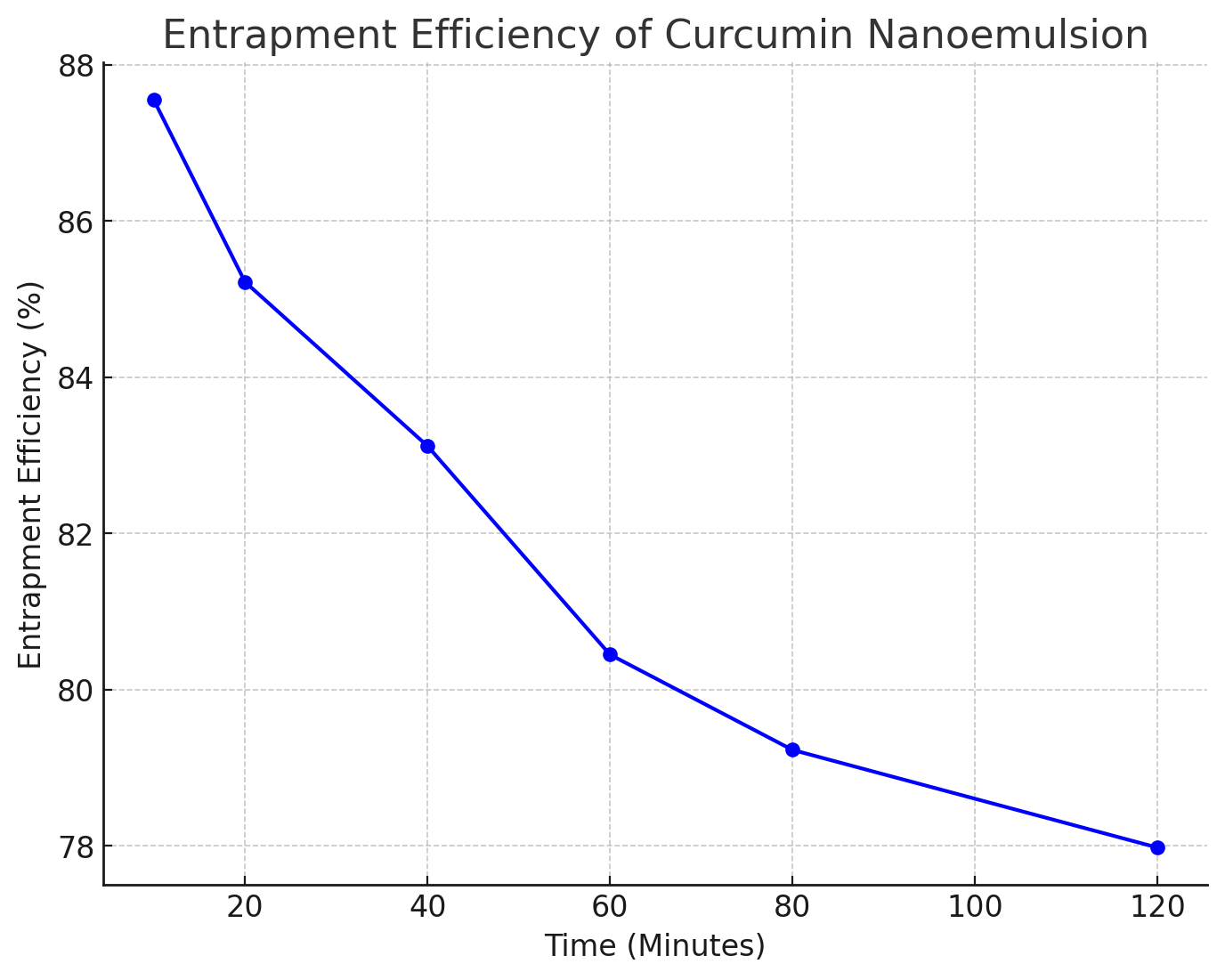
Fig. 14: Entrapment efficiency analysis of curcumin nanoemulsion
As the time progressed (from 10 to 120 min), a gradual decline in entrapment efficiency was observed, ranging from 87.55% to 77.98%. This reduction can be attributed to the natural tendency of nanoemulsions to release encapsulated substances over time, especially when exposed to conditions such as temperature fluctuations or physical stress. Although dialysis is a common method for measuring entrapment efficiency (EE), it is not without limitations. The method does not fully separate encapsulated curcumin from free curcumin, which could lead to overestimation of EE. To ensure more accurate and validated results, future studies will implement ultrafiltration or centrifugation with a membrane cut-off for direct separation of encapsulated and free curcumin. The results also highlight the dynamic nature of nanoemulsions, where, although the encapsulation efficiency remains relatively high, it may not remain constant. These findings emphasize the need for further optimization of formulation parameters, such as surfactant concentration and emulsion preparation techniques, to ensure that the nanoemulsion remains stable and retains high encapsulation efficiency over extended periods. Further investigations into the stability under different storage conditions could provide more insights into the long-term release characteristics and potential applications in therapeutic delivery systems [34, 42].
CONCLUSION
This study successfully developed and characterized a curcumin-loaded nanoemulsion, with optimized formulations (F5-F8) demonstrating excellent physicochemical properties. The optimized formulations exhibited uniform droplet size (195.3 nm) and a low polydispersity index (PDI of 0.36), indicating moderate emulsion stability. Entrapment efficiency was high, ranging from 87.55% to 77.98%, though the method used for quantification could benefit from further validation. The current study demonstrates that curcumin can be effectively encapsulated within the nanoemulsion system, with high EE values observed. However, to further improve the reliability of these results, we plan to validate the entrapment efficiency method by incorporating more accurate techniques, such as ultrafiltration and centrifugation with membrane cut-off, in future research. The formulations showed acceptable physical stability across freeze-thaw cycles, with minimal changes in viscosity and pH. Differential scanning calorimetry (DSC) analysis indicated that curcumin may be molecularly dispersed within the nanoemulsion, suggesting enhanced solubility and stability. These findings provide a strong basis for further development of curcumin nanoemulsions in pharmaceutical applications.
ACKNOWLEGMENT
The authors would like to express their sincere gratitude to the Ministry of Research, Technology, and Higher Education (Kemendikti Saintek) of the Republic of Indonesia for funding this research through the BIMA program. Special thanks are extended to LPPM Universitas Negeri Gorontalo DPPM with Contract Number 700/UN47. D1/PT.01.03/2025, particularly the Department of Pharmacy at the Faculty of Sports and Health, as well as other laboratories that contributed to the success of this study. We also wish to acknowledge all parties involved in this research for their invaluable support and assistance.
AUTHORS CONTRIBUTIONS
Zulkifli B. Pomalango and Mohamad Aprianto Paneo contributed to the conceptualization and design of the study, as well as the overall manuscript writing and data interpretation.
Mohamad Aprianto Paneo, Nurain Thomas and Zulfiayu Sapiun was responsible for the formulation and preparation of the curcumin-loaded nanoemulsion, including laboratory experiments and data analysis.
Multiani S Latif and Zulfiayu Sapiun performed the stability testing and physicochemical characterization of the nanoemulsions and contributed to the analysis and discussion of the results.
Muchtar Nora Siregar and Karmila Lihawa provided critical revisions to the manuscript, particularly in the statistical analysis and validation of the results.
CONFLICTS OF INTERESTS
The authors declare no conflict of interest regarding the publication of this paper.
REFERENCES
Koser N, Abbas R, Rizwan B, Sultan H, Islam Z, Jawad M. Pharmacological effects of Curcuma longa and its bioactive constituent curcumin. Pak Biomed J. 2022;5(6):441. doi: 10.54393/pbmj.v5i6.441.
Chaudhary S, Chaudhary G, Bana S, Singh J, Dubey S, Sharma L. Pharmacological facet of Curcuma longa: a review. IJPSRR. 2022;76(1):119-25. doi: 10.47583/ijpsrr.2022.v76i01.022.
Pomalango ZB, Paneo MA, Pakaya N, Aman LO, Hutuba AH. Cholesterol-lowering effects of turmeric effervescent dosage in preventing atherosclerosis. Sci Technol Indones. 2025;10(2):482-92. doi: 10.26554/sti.2025.10.2.482-492.
Stohs SJ, Chen O, Ray SD, Ji J, Bucci LR, Preuss HG. Highly bioavailable forms of curcumin and promising avenues for curcumin-based research and application: a review. Molecules. 2020;25(6):1397. doi: 10.3390/molecules25061397, PMID 32204372.
Chen Y, Lu Y, Lee RJ, Xiang G. Nano-encapsulated curcumin: and its potential for biomedical applications. Int J Nanomedicine. 2020;15:3099-120. doi: 10.2147/IJN.S210320, PMID 32431504.
Thomas N, Paneo MA, Mo’o FRC, Puluhulawa LE, Latif MS. Formulation of natural hydrogel from bromelain enzyme and alginate-chitosan and the effectiveness test by in vivo in healing burns. J Med Pharm Chem Res. 2025;7:944-59. doi: 10.48309/jmpcr.2025.472155.1374.
Boby JG. A review on self emulsifying nanoemulsion. OAJPR. 2017;1(4):1-17. doi: 10.23880/oajpr-16000123.
Chime SA, Kenechukwu FC, Attama AA. Nanoemulsions advances in formulation characterization and applications in drug delivery. In: Sezer AD, editor. Application of nanotechnology in drug delivery. InTech; 2014. p. 77-126. doi: 10.5772/58673.
Jusuf H, Hadjarati H, Mustapa MA, Abdulkadir WS, Suryadi AM, Taupik M. Characterization and formulation of anti-aging cream using collagen derived from sardine (Sardinella spp.) fish scales. Tropical Journal of Natural Product Research. 2025;7(9):3013-20. doi: 10.26538/tjnpr/v9i7.8.
Zhang G, Zhang Q, Wang L, Ji L, Han P, Zhao F. Preparation and optimization of O/W emulsions stabilized by triglycerol monolaurate for curcumin encapsulation. Molecules. 2022;27(24):8861. doi: 10.3390/molecules27248861, PMID 36557994.
Ye Q, Kwon S, Gu Z, Selomulya C. Stable nanoemulsions for poorly soluble curcumin: from production to digestion response in vitro. J Mol Liq. 2024 Jan 15;394:123720. doi: 10.1016/j.molliq.2023.123720.
Kontogiannis O, Selianitis D, Perinelli DR, Bonacucina G, Pippa N, Gazouli M. Non-ionic surfactant effects on innate pluronic 188 behavior: interactions and physicochemical and biocompatibility studies. Int J Mol Sci. 2022;23(22):13814. doi: 10.3390/ijms232213814, PMID 36430294.
Azeem A, Rizwan M, Ahmad FJ, Iqbal Z, Khar RK, Aqil M. Nanoemulsion components screening and selection: a technical note. AAPS PharmSciTech. 2009;10(1):69-76. doi: 10.1208/s12249-008-9178-x, PMID 19148761.
Tinjaca DA, Martinez F, Almanza OA, Pena MA, Jouyban A, Acree WE. Increasing the equilibrium solubility of meloxicam in aqueous media by using dimethyl sulfoxide as a cosolvent: correlation dissolution thermodynamics and preferential solvation. Liquids. 2022;2(3):161-82. doi: 10.3390/liquids2030011.
Ma P, Zeng Q, Tai K, He X, Yao Y, Hong X. Development of stable curcumin nanoemulsions: effects of emulsifier type and surfactant-to-oil ratios. J Food Sci Technol. 2018;55(9):3485-97. doi: 10.1007/s13197-018-3273-0, PMID 30150807.
Kanlayavattanakul M, Lourith N. Preparation of stable tea seed oil nano-particle emulsions by a low energy method with non-ionic surfactants. Grasas Aceites. 2017;68(2):e196. doi: 10.3989/gya.1271162.
Syukri Y, Fitriani H, Pandapotan H, Nugroho BH. Formulation characterization and stability of ibuprofen-loaded self-nanoemulsifying drug delivery system. Indonesian J Pharm. 2019;30(2):105-13. doi: 10.14499/indonesianjpharm30iss2pp105-113.
Chidi E. Development and evaluation of nanoemulsion formulations for improved oral delivery of carvedilol. UJPR. 2017;2(1):5-11. doi: 10.22270/ujpr.v2i1.R2.
Naz Z, Ahmad FJ. Curcumin-loaded colloidal carrier system: formulation optimization mechanistic insight ex vivo and in vivo evaluation. Int J Nanomedicine. 2015;10:4293-307. doi: 10.2147/IJN.S82788, PMID 26170665.
Okonogi S, Naksuriya O, Charumanee S, Sirithunyalug J. Effect of aromatic substitution of curcumin nanoformulations on their stability. Sci Pharm. 2016;84(4):625-33. doi: 10.3390/scipharm84040625, PMID 28656941.
Putri EF, Indahyanti E, Mardiana D, Lestari ML, Ningsih Z. Okra mucilage extract as a co-surfactant increased the curcumin nanoemulsion stability and encapsulation efficiency. Sci Technol Indones. 2023;8(3):509-15. doi: 10.26554/sti.2023.8.3.509-515.
Rahman MA. Development of UV spectrophotometric method for estimation of curcumin in bulk drug and nanogel formulation: a hydrolytic degradation studies. Int J Pharm Chem Anal. 2022;9(2):87-92. doi: 10.18231/j.ijpca.2022.014.
Srinivas B, Ashraf S, Vivek N, Rajashree H. Development and validation of bivariate UV-visible spectroscopic method for simultaneous estimation of curcumin and piperine in their combined nanoparticulate system. J Appl Pharm Sci. 2021;11(3):64-70. doi: 10.7324/JAPS.2021.110509.
Lee BN, Hong SJ, Yu MH, Shin GH, Kim JT. Enhancement of storage stability and masking effect of curcumin by turmeric extract-loaded nanoemulsion and water-soluble chitosan coating. Pharmaceutics. 2022;14(8):1547. doi: 10.3390/pharmaceutics14081547, PMID 35893803.
Kim SW, Garcia CV, Lee BN, Kwon HJ, Kim JT. Development of turmeric extract nanoemulsions and their incorporation into canned ham. Korean J Food Sci Anim Resour. 2017;37(6):889-97. doi: 10.5851/kosfa.2017.37.6.889, PMID 29725211.
Putri EF, Indahyanti E, Mardiana D, Lestari ML, Ningsih Z. Okra mucilage extract as a co-surfactant increased the curcumin nanoemulsion stability and encapsulation efficiency. Sci Technol Indones. 2023;8(3):509-15. doi: 10.26554/sti.2023.8.3.509-515.
Fatahi H, Claverie J, Poncet S. Thermal characterization of phase change materials by differential scanning calorimetry: a review. Appl Sci. 2022;12(23):12019. doi: 10.3390/app122312019.
Sheikhzadeh S, Alizadeh M, Rezazad M, Hamishehkar H. Application of response surface methodology and spectroscopic approach for investigating of curcumin nanoencapsulation using natural biopolymers and nonionic surfactants. J Food Sci Technol. 2016;53(11):3904-15. doi: 10.1007/s13197-016-2377-7, PMID 28035146.
Kadhim ZJ, Rajab NA. Formulation and characterization of glibenclamide nanoparticles as an oral film. Int J Drug Deliv Technol. 2022;12(1):387-94. doi: 10.25258/ijddt.12.1.70.
Anuchapreeda S, Fukumori Y, Okonogi S, Ichikawa H. Preparation of lipid nanoemulsions incorporating curcumin for cancer therapy. J Nanotechnol. 2012;2012:1-11. doi: 10.1155/2012/270383.
Chiang TC, Chang JY, Chou TH. Formulation and characteristics of edible oil nanoemulsions modified with polymeric surfactant for encapsulating curcumin. Polymers (Basel). 2023;15(13):2864. doi: 10.3390/polym15132864, PMID 37447509.
Onodera T, Kuriyama I, Andoh T, Ichikawa H, Sakamoto Y, Lee-Hiraiwa E. Influence of particle size on the in vitro and in vivo anti-inflammatory and anti-allergic activities of a curcumin lipid nanoemulsion. Int J Mol Med. 2015;35(6):1720-8. doi: 10.3892/ijmm.2015.2186, PMID 25891482.
Araiza Calahorra A, Akhtar M, Sarkar A. Recent advances in emulsion-based delivery approaches for curcumin: from encapsulation to bioaccessibility. Trends Food Sci Technol. 2018;71:155-69. doi: 10.1016/j.tifs.2017.11.009.
Lashkari D, Tejpal Yadav, Shiv Kumar Garg, Kumar V. Impact of carbopol-loaded curcumin nanoemulsion on psoriasis: investigating treatment efficacy and histopathological changes in an animal model. Asian J Pharm Clin Res. 2025;18(5):105-10. doi: 10.22159/ajpcr.2025v18i5.54295.
Maity S, Banerjee I. Nanoemulsion as a drug delivery system of anticancer drug. Asian J Pharm Clin Res. 2025;18(4):82-94. doi: 10.22159/ajpcr.2025v18i4.53759.
Bodke V, Kumbhar P, Belwalkar S, Mali AS, Waghmare K. Design and development of nanoemulsion of Smilax china for anti-psoriasis activity. Int J Pharm Pharm Sci. 2024;16(5):54-66. doi: 10.22159/ijpps.2024v16i5.50327.
Hashim DM, Sheta NM, Elwazzan VS, Sakran WS. Enhancing the sunscreen efficacy of Bemotrizinol micropigment by using O/W nanoemulsion topical preparations. Int J Pharm Pharm Sci. 2019;11(7):47-56. doi: 10.22159/ijpps.2019v11i7.32652.
Bagade PV, Kulkarni NS, Chaudhari PD, Dhole SN, Desai US. Exploring modern techniques for solubility enhancement in drug formulations. Int J Curr Pharm Sci. 2025;17(5):31-41. doi: 10.22159/ijcpr.2025v17i5.7044.
Shah J, Patel S, Bhairy S, Hirlekar R. Formulation optimization, characterization and in vitro anti-cancer activity of curcumin-loaded nanostructured lipid carriers. Int J Curr Pharm Sci. 2022;14(1):31-43. doi: 10.22159/ijcpr.2022v14i1.44110.
Gokul M, Umarani G, Esakki A. Green synthesis and characterization of isolated flavonoid mediated copper nanoparticles by using Thespesia populnea leaf extract and its evaluation of anti-oxidant and anti-cancer activity. Int J Chem Res. 2022;6(1):15-32. doi: 10.22159/ijcr.2022v6i1.197.
Yallapu MM, Nagesh PK, Jaggi M, Chauhan SC. Therapeutic applications of curcumin nanoformulations. AAPS J. 2015;17(6):1341-56. doi: 10.1208/S12248-015-9811-Z, PMID 26335307.
Mahadev M, Nandini HS, Ramu R, Gowda DV, Almarhoon ZM, Al-Ghorbani M. Fabrication and evaluation of quercetin nanoemulsion: a delivery system with improved bioavailability and therapeutic efficacy in diabetes mellitus. Pharmaceuticals (Basel). 2022;15(1):70. doi: 10.3390/ph15010070, PMID 35056127.