
Int J App Pharm, Vol 18, Issue 2, 2026, 377-386Original Article
A COMBINED EXPERIMENTAL AND NETWORK PHARMACOLOGY APPROACH TO ELUCIDATE THE ANTI‑ULCER POTENTIAL OF PERSEA MACRANTHA
FAISAL MAMDOUH ALHAJRI1, RAVI SHANKAR N.2*, PRAMOD S.2, SIBGHATULLAH MUHAMMAD ALI SANGI3, GIRISH MERAVANIGE4, SREEHARSHA NAGARAJA1
1Department of Pharmaceutical Sciences, College of Clinical Pharmacy, King Faisal University, Al-Ahsa-31982, Saudi Arabia. 2Department of Pharmacology, Visveswarapura Institute of Pharmaceutical Sciences, Bengaluru, Karnataka, India. 3Basic Medical Sciences Department, College of Medicine, Dar Al Uloom University, Riyadh, Saudi Arabia. 4Department of Biomedical Sciences, College of Medicine, King Faisal University, Al-Ahsa-31982, Saudi Arabia
*Corresponding author: Ravi Shankar N. and Girish Meravanige; *Email: ravi.vips05@gmail.com and gmeravanige@kfu.edu.sa
Received: 15 Dec 2025, Revised and Accepted: 02 Feb 2026
ABSTRACT
Objective: The main objective of the present study is to evaluate the anti‑ulcer potential of Persea macrantha ethanolic and aqueous extracts, by using both network pharmacology approaches and in vivo animal models.
Methods: Network pharmacology was employed to identify potential ulcer-related targets and pathways, while molecular docking validated interactions between phytochemicals and these targets. The anti-ulcer activity of Persea macrantha ethanolic and aqueous leaf extracts was assessed in Albino Wistar rats at doses of 200 and 400 mg/kg (p. o.), with omeprazole (20 mg/kg) and sucralfate (100 mg/kg) as standards. Ulcers were induced using ethanol (8 ml/kg, p. o.) and aspirin (400 mg/kg, p. o.), followed by microscopic examination of gastric lesions. In vitro methods were performed by H+/K+ATPase inhibition and acid‑neutralizing capacity (ANC) tests.
Results: Network pharmacology and molecular docking identified key targets (PTPN11, NFKB1, CYP1B1) interacting with phytoconstituents such as β‑phellandrene, machiline, and sesamin, regulating pathways including Ras, T‑cell receptor, MAPK signaling, and tryptophan metabolism. In vivo studies in ulcer‑induced Albino Wistar rats showed that Persea macrantha (P. Macrantha) leaf extracts significantly (p<0.001) reduced ulcer index in a dose‑dependent manner and improved protection. In vitro assays confirmed dose‑dependent H+/K+ATPase inhibition, while both extracts exhibited higher acid‑neutralizing capacity than aluminum‑magnesium hydroxide.
Conclusion: Overall, Persea macrantha exerts a multitargeted gastroprotective effect by modulating ulcer-healing regulators, strengthening protective mechanisms, and supporting its potential as a natural therapeutic agent for gastric ulcers.
Keywords: Persea macrantha, Peptic ulcer, Aspirin-induced, Network pharmacology, Molecular docking
© 2026 The Authors.Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijap.2026v18i2.57804 Journal homepage: https://innovareacademics.in/journals/index.php/ijap
INTRODUCTION
Gastric ulcer or peptic ulcer disease (PUD) is a prevalent gastrointestinal condition globally, encompassing two main types: gastric ulcers and duodenal ulcers. Its development stems from an imbalance between protective and harmful factors affecting the gastroduodenal mucosa and causes ulcer toxicity [1]. Gastric ulcer is linked with complications like Gastrointestinal bleeding, Perforation, gastric outflow obstruction [2]. These complications, much like those seen in cancer, place significant strain on healthcare infrastructure and increase the burden on medical systems worldwide [3]. Plant-derived therapeutics used in traditional medicine contain a diverse array of secondary metabolites and phytochemicals that target multiple proteins involved in disease pathogenesis, while exhibiting minimal side effects [4]. Persea macrantha is a promising medicinal plant with significant potential for therapeutic intervention in ulcer based on their traditional claims [5]. Additionally, Persea macrantha has proven for its antiinflammatory and cytoprotective activities [6.] Previous studies on Persea macrantha have primarily highlighted its Ethnomedical relevance and broad pharmacological effects. Unlike conventional ethnopharmacological studies that are largely descriptive, the present work integrates network pharmacology–based target and pathway prediction with in vitro assays (acid neutralizing capacity and H⁺/K⁺-ATPase inhibition) and in vivo experimental validation. This combined approach provides mechanistic insight into the Gastroprotective potential of Persea macrantha, thereby strengthening the scientific basis of its traditional use.
MATERIALS AND METHODS
Aspirin was procured form the United States Vitamins Private Limited, India. Omeprazole was procured from max life sciences, India. Sucralfate was obtained from MODASA India. Ethanol was procured from witham. Aluminum and magnesium Antacids (Wellona Pharma, Surat). Chemicals used in in vitro, H+/K+ATPase-Magnesium chloride and potassium chloride (Thermo fisher PVT. LTD, Mumbai), Tris hydrochloride (Lobachemie, Mumbai). HCL from nutan chemicals pune. Phenolphthalein (mubychem, India). NaOH (vizag chemical, India)
In silico pharmacology
Retrieval of phytoconstituents
The phytoconstituents of Persea macrantha reported in the literature were compiled through a comprehensive search of standard reference books, peer-reviewed journals, and specialized databases such as IMPATT and PCIDB
Prediction of drug-likeness
Canonical SMILES of the phytoconstituents were retrieved from the PubChem web service (PubChem, nih. gov), while their drug-likeness profiles were predicted using the Molsoft server (Molsoft L. L. C.).
Target prediction of phytoconstituent
The selected phytoconstituents were analyzed for their potential interactions with biological targets using Superpred (prediction. charite. de). Molecules showing a probability score greater than 70% were considered for further evaluation. Gene IDs corresponding to all predicted protein targets were then retrieved from the UniProt database (https://www.uniprot.org/).
Retrieval of disease gene target, gene enrichment, and pathway elucidation
The disease targets for gastric or peptic ulcers were first obtained from the GeneCards Database (GeneCards – Human Genes Gene Database Gene Search). Disease-related targets for gastric ulcer/peptic ulcer disease were retrieved from the GeneCards database using the keywords “gastric ulcer” and “peptic ulcer disease.” To ensure relevance, targets were filtered based on the GeneCards relevance score, and only genes with a relevance score ≥ 5 were selected for subsequent network pharmacology analysis. These target names were then cross-verified using the KEGG pathway database, and only those targets present in KEGG were selected for further analysis along with their associated pathways. Next, a set of proteins corresponding to these targets, identifiable by their Gene IDs, were submitted to the STRING 11.0v database to investigate protein–protein interactions. This analysis was used to determine which gastric ulcer pathology‑related pathways could be influenced by the phytoconstituents identified in the KEGG pathway database.
Network construction
The interaction network between phytoconstituents and protein targets was constructed using Cytoscape 3.6.1 software [7]. Enriched pathways were analyzed, with particular attention to node size and edge count, which emphasize their significance in disease targeting.
Molecular docking
Virtual screening was employed to examine the interactions between phytoconstituents and protein targets, with particular phytoconstituents influencing obesity therapeutic targets chosen based on network analysis. The 3D structures of compounds/ligands were retrieved from the PubChem database (PubChem, nih. gov) and converted into. pdb format using Discovery Studio Visualizer, while protein structures along with their PDB IDs were acquired from the RCSB PDB database. Molecular docking was then performed using PyRx version 0.8 to analyze the interactions between the phytoconstituents and targets, and the conformation with the lowest binding energy was subjected to interaction analysis [8]. Docking was performed using default parameters. The grid box was defined to cover the active site region of each target protein. Protein structures were prepared by removing water molecules and adding polar hydrogens, while ligand structures were energy minimized and appropriately protonated within PyRx prior to docking. Finally, the results were visualized using Discovery Studio Visualizer version 2021.
Collection, authentication and extraction of Persea macrantha
Collection and authentication of plant material
In the month of December, the Persea macrantha leaves were collected from FRLHT (Foundation for Revitalisation of Health Traditions Ethno Medicinal Garden and Nursery) yalahanka, Bengaluru. The collected leaves were identified and authenticated by Dr. V. Rama Rao (Research officer, Botany, Central Ayurveda Research Institute) as Persea macrantha (Nees) Kosterm belong to the family Lauraceae (RRCBI-112295).
Preparation of extract
Leaves of Persea macrantha leaves were collected from yalahanka, Bengaluru. The leaves were separated, washed and shade dried. The dried leaves were subjected for size reduction to coarse powder (355µm). The Soxhlet apparatus (Garg Process glass India private limited, Mumbai), Extraction method was employed using 100 g of Persea macrantha powder and 500 ml of 95% ethanol. The extraction was carried out at 50 °C for 12 h. The resulting extract was dried in a water bath, transferred into an air-tight glass container, and stored at 4 °C for further use. The percentage yield was found to be 12.1% and 10.8% for aqueous and ethanolic extract respectively.
Maintenance of experimental animals and ethics approval for the study
The animal experimental protocols were reviewed and approved by IAEC under the protocol number VIPS/IAEC/03/08/24/33-RN, and the experiment was performed after receiving ethical clearance. Adult Albino Wistar rats (8-12 w, weighing 150–200 g) maintained at the institute’s animal facility were housed in polypropylene cages under a 12:12 h light/dark cycle. The ambient temperature was maintained at 22±2 °C with relative humidity of 60-70%. Rats were provided with standard pellet diet and water ad libitum. To maintain hygiene, bedding material was changed every alternate day, and cages were cleaned regularly.
In vivo pharmacological investigations
Acute oral toxicity studies
The acute oral toxicity of Persea macrantha, was evaluated as per the ‘Acute Oral Toxicity Study’’ of OECD (organization for economic co-operation and development) Guideline No. 423. Six female’s Albino wistar rats (3 animals per step) were used for the limit test of 2000 mg/kg body weight. The drugs were suspended in demineralized water and administered at a single oral dose of 2000 mg/kg body weight to overnight fasted rats. Animals were observed initially after dosing at least once during the first 30 min, periodically during the first 30 min, periodically during the first 24 h. The additional observations like change in skin and fur, eyes, clinical signs (tremors, convulsions) and behavioral patterns were also observed. Body weight was monitored for 14 days (once every week) [9].
Aspirin-induced gastric ulcer
Animals were randomly divided into 7 groups (n=6). Group I Rats are normal control and were given vehicle alone; Group II Rats were given aspirin 400 mg/kg, on 14th day. Group III Rats were given Omeprazole (20 mg/kg). Group IV Rats were given Ethanolic extract of Persea macrantha (200 mg/kg) for 2 w. Group V Rats were given Ethanolic extract Persea macrantha (400 mg/kg) for 2 w. Group VI and VII rats were given aqueous extract of Persea macrantha 200 and 400 mg/kg, respectively. On the 14th day (last day) the rats were sacrificed (Ketamine+xylazine 75+10 mg/kg b. w. i. p.,) and stomach was dissected out and severity of ulcer is examined by calculating the ulcer index [10, 11].
Ethanol-induced gastric ulcer
Animals were randomly divided into 7 groups (n=6). Group I rats are normal control and were given vehicle alone. Group II rats were treated with the ethanol 8 ml/kg body weight, group III rats were treated with the sucralfate+ethanol (100 mg/kg and 8 ml/kg b, w. respectively). Group IV rats were treated with the ethanolic extract of Persea macrantha+ethanol (200 mg and 8 ml per kg body weight). Group V rats were treated with the ethanolic extract of Persea macrantha+ethanol (400 mg and 8 ml/kg body weight). Group VI rats were treated with the aqueous extracts of Persea macrantha+ethanol (200 mg and 8 ml/kg body weight, respectively), and group VII rats were treated with the aqueous extract of Persea macrantha+ethanol (400 mg and 8 ml/kg body weight). On the day of experiment, 1 hour after the administration of extracts and standard, absolute ethanol at the dose of 8 ml/kg was administered per oral to each animal in the groups except normal control. Sacrifice (Ketamine+xylazine 75+10 mg/kg b. w. i. p.,) the animals 6 hour after ethanol administration, isolate the stomachs and cut open along the greater curvature and pin on a soft board. The ulcer index was calculated by examining the lesions [12-14].
Parameters were evaluated
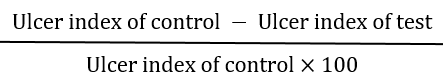
Ulcer Index
An ulcer index UI was calculated:
% Protection
Total acidity
Gastric volume
Determination of pH
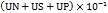
UN= average of number of ulcers per animal
US = average of severity score
UP = percentage of animals with ulcers
An aliquot of 1 ml of the centrifuged gastric juice was diluted with 1 ml of distilled water and transferred to a 50 ml conical flask. Two drops of phenolphthalein indicator were added to the mixture, which was titrated with 0.01N NaOH until a permanent pink color was achieved. The volume of 0.01 N NaOH consumed was recorded.
The total acidity is expressed as mEq/l, and was calculated using the following formula:
Total acidity = 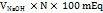
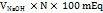
Where V is volume and N is normality.
The gastric juice was centrifuged, decanted, and squeezed into graduated glass tubes. The amount of gastric juice in the stomach was measured.
The pH of the solution was calculated using a digital pH meter (model: 1 TDS EC, by aptechdeals) after an aliquot of 1 ml gastric juice was diluted with 1 ml of distilled water.
In vitro investigation
Determination of H+K+ATPase
The H+K+ATPase activity in the presence of different concentrations of test extracts and omeprazole was assayed. The enzyme source was preincubated with different concentration of the test material (10-70μg) for 30 min. Extract treated enzyme in 20 mmol tris-HCl, pH 7.4, 2 mmol magnesium chloride (MgCl2) and 2 mmol potassium chloride (KCl). The reaction was started with the addition of 2 mmol adenosine-5'-triphosphate (ATP) and incubated for 30 min at 30 °C and terminated by the addition of 10% Trichloroacitic acid followed by centrifugation at 2000xg. The amount of inorganic phosphorous released from adenosine-5'-triphosphate (ATP) was determined spectrophotometrically using spectrophotometer (by REMI model UV-1900) at 640 nm. The enzyme source was also treated similarly with the standard drug omeprazole and the enzyme activity was measured.
Acid neutralization capacity test
The acid-neutralizing capacity values of the extract are 100 mg; 500 mg, 1000 mg, and 1500 mg. Standard antacids aluminum hydroxide magnesium hydroxide (AHMH) have been compared. Mixture of plant extract, 70 ml of water and 30 ml of 1N HCL was added and swirled for 15 min. The surplus HCL was titrated with 0.5N NAOH, using phenolphthalein as an indicator until the Appearance of pink Colour. Calculating the moles of acid neutralized involves dividing the volume of HCl by its normality and the volume of NaOH by its normality. Moles of HCl neutralized divided by grams of antacid/extract equals acid neutralizing capability (ANC) per g of antacids.
Statistical analysis
Statistical analysis was carried out using Graph Pad Prism 10 software. The analysis was performed using one-way ANOVA followed by Dunnett’s test, P<0.05, P<0.01, P<0.001, were considered statistically significant
RESULTS
Findings from insilco studies
A total of 7 phytochemicals were identified from previous review of literature. Out of the 7 phytoconstituents, all the constituents were followed Lipinski rule of five or positive DLS.
Table 1: List of phytochemicals following either lipinski rule or positive drug likeness score
| S. No. | Phytoconstituents | Lipinski rule | DLS | ||||
| MW | HBA | HBD | Log P | PSA | |||
| 1 | Micranthine | 548.23 | 7 | 2 | 4.73 | 59.94A2 | 0.97 |
| 2 | Beta-phellandrene | 136.13 | 0 | 0 | 3.7 | 0.00 A2 | -1.35 |
| 3 | Machiline | 285.14 | 4 | 3 | 2 | 54.06A2 | 0.89 |
| 4 | Sesamin | 354.11 | 6 | 0 | 3.4 | 52.71A2 | -0.91 |
| 5 | Homoegonol | 342.15 | 5 | 1 | 3.7 | 48.04A2 | -0.25 |
| 6 | Beta-sitosterol-glucoside | 576.44 | 6 | 4 | 6.3 | 79.93A2 | 0.50 |
| 7 | Ellagic acid | 302.01 | 8 | 4 | 1.5 | 105.23A2 | -1.11 |
Note: MW-molecular weight, HBA-hydrogen bond acceptor, HBD-hydrogen bond donor, DLS-Drug Likeness Score. Both positive and negative values were included in the study to comprehensively assess potential bioactivity.
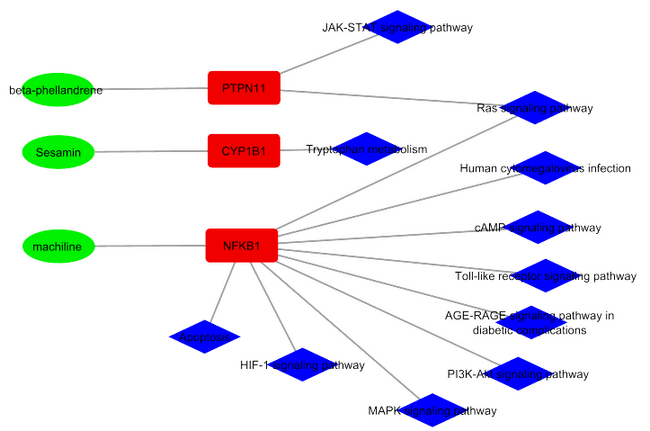
Fig. 1: Interaction network of phytoconstituents of Persea macrantha with their targets and regulated pathways. In the fig. 1, green oval nodes represent the phytochemicals which are present in the leaves of Persea macrantha and that were sharing bond with targets, which are represented by the red square nodes and the blue diamond nodes represents the pathways that are associated with the target and their binding phytochemicals
Table 2: List of phytochemicals with their targets and regulated pathways
| Phytochemicals | Targets (protein) | Regulated pathways |
| Beta-phellandrene | PTPN11(Tyrosine-protein phosphatase non-receptor type 11) | Ras signaling pathway, T cell receptor signaling pathway, |
| Machiline | NFKB1 (Nuclear Factor NF-kappa-B) | cAMP signaling pathway, Toll-like receptor signaling pathway, MAPK Pathway, HIF1 signaling pathway. |
| Sesamin | CYP1B1(Cytochrome P450 family 1 subfamily B member 1) | Tryptophan metabolism pathway |
Note: Regulated pathways are selected from the 24 identified pathways based on their direct involvement in gastric mucosal protection, inflammation, and repair mechanisms relevant to ulcer pathophysiology.
Network construction
A total of 24 targets were obtained from the Superpred database, interacting with the phytoconstituents having a prediction probability greater than 70% and from the GeneCards database 24 disease gene targets were obtained with relevance greater or equal to 5. The pathway enrichment analysis was employed for the 24 targets using String and KEGG databases. Consequently, these 24 targets were found to be associated with the distinct pathways and a network was constructed to visualize the interaction between pathways, targets and the phytoconstituents.
Molecular docking
Docking was performed for beta-phellandrene, machiline, and sesamin with targets PTPN11, NFKB11, CYP1B1, respectively. Sesamin formed hydrogen bond with CYP1B1, exhibiting a binding energy of-9.7 Kcal/mol and Machiline exhibited a binding energy of-5.8 Kcal/mol when docked with NFKB1, by forming two hydrogen bond. Whereas Beta-phellandrene formed alkyl bond with the PTPN11 and exhibited the binding energy of-5.2 kcal/mol.
Findings from in vitro studies
Acid neutralizing capacity test
The acid neutralizing capacity (ANC) assay was conducted using standard aluminum-magnesium hydroxide and both ethanolic and aqueous extracts of Persea macrantha at concentrations of 100, 500, 1000, and 1500 mg. The ANC of the standard was recorded at 13.5 mEq. In comparison, the ethanolic extract exhibited ANC values of 137.5, 26, 11.5, and 6.6 mEq, while the aqueous extract showed 132, 24.1, 10.45, and 5.36 mEq, respectively. Notably, at the highest tested dose of 1500 mg, both extracts demonstrated significantly greater acid neutralizing capacity than the standard, indicating potent antacid-like activity, here at lower doses of extracts we could observe that more ANC value because at lower doses more acid is required to neutralize the base, when compare to the higher doses therefore low ANC values indicates the better neutralizing action of test. Compared to the standard, which is already proven, at the dose of 1000 mg and 1500 mg of extracts are near to the value of standard drug values, hence we can say that extract (both aqueous and ethanolic) may have better neutralizing capacity.
Table 3: Binding energy and hydrogen bond interaction of phytoconstituents with targets
| Target | Phytoconstituents | Binding energy (Kcal/mol) | Hydrogen bond interaction | Van der waals interaction | Alkyl interaction |
| PTPN11 | Beta-phellandrene | -5.2 | - | SER A: 499 GLN A: 257 ARG A: 498 GLN A: 256 THR A: 253 |
TYR A: 263 LYS A: 260 |
| NFKB1 | Machiline | -5.8 | TRP A: 292 THR A: 261 |
GLU A: 293 GLY A: 294 LYS A: 316 LYS A: 317 GLY A: 262 and 263 |
LYS A: 315 |
| CYP1B1 | Sesamin | -9.7 | LYS A: 512 | PHE A: 231 LEU A: 509 ASP A: 333 ASN A: 228 ALA A: 330 SER A: 336 |
VAL A: 198 PHE A: 231(pi-pi interaction) |
Note: Non-hydrogen bond interactions are included such as, van der Waals, alkyl interactions, and pi-alkyl interactions.
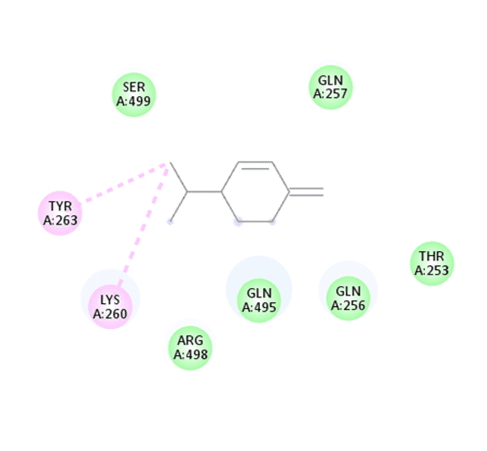
Fig. 2: 2D Binding representation of beta-phellandrene with PTPN11, (Tyrosine-protein phosphatase non-receptor type 11), Here the molecular docking was performed by using BIOVIA Discovery studio software and the hydrogen bond interaction is absent, other interaction such as alkyl (pink Colour) and van der waals (green Colour) was present
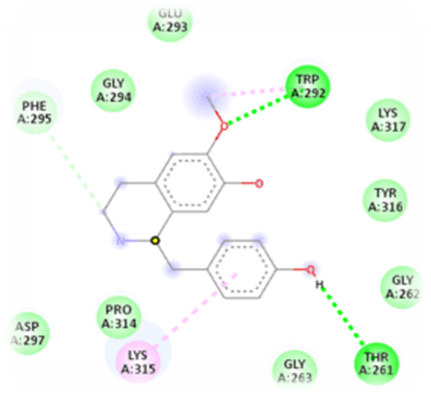
Fig. 3: 2D Binding representation of machiline with NFKB1 (Nuclear Factor NF-kappa-B), Here the molecular docking was performed by using BIOVIA Discovery studio software and the dark green Colour indicates (TRP A: 292) and THR A: 261) represents the hydrogen bond interaction between the phytochemical and target and pink Colour indicates the alkyl interaction.
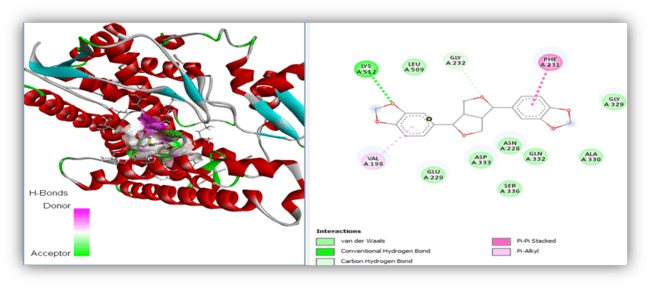
Fig. 4: 2D Binding representation of sesamin with CYP1B1 (Cytochrome P450 family 1 subfamily B member 1), Here the molecular docking was performed by using BIOVIA Discovery studio software and the green Colour (A: 512) indicates the hydrogen bond interaction between the phytochemical and target other than this dark pink Colour indicates the pi-pi standard, light pink indicates the alkyl and green colour indicates the van der waals interactions
Table 4: Acid neutralizing capacity test of Persea macrantha ethanolic extracts compared with standard aluminum and magnesium antacids
| S. No. | Concentration of drug samples (mg) | ANC (mEq) |
| 1 | 100 mg (PMEE) | 137.5 |
| 2 | 500 mg (PMEE) | 26 |
| 3 | 1000 mg (PMEE) | 11.5 |
| 4 | 1500 mg (PMEE) | 6.6 |
| 5 | 500 mg Al(OH)3+Mg(OH)2 | 13.5 |
Note: PMEE: Persea macrantha ethanolic extract.
Table 5: Acid neutralizing capacity test of Persea macrantha aqueous (PMAE) extracts compared with standard aluminum and magnesium antacids
| S. No. | Concentration of drug samples (mg) | ANC (mEq) |
| 1 | 100 mg (PMEE) | 132 |
| 2 | 500 mg (PMEE) | 24.1 |
| 3 | 1000 mg (PMEE) | 10.45 |
| 4 | 1500 mg (PMEE) | 5.36 |
| 5 | 500 mg Al(OH)3+Mg(OH)2 | 13.5 |
Note: PMAE: Persea macrantha aqueous extract.
H+/K+ATPase enzyme inhibition assay
The alcoholic extract of Persea macrantha demonstrated notable inhibitory activity, reaching 42.56% inhibition at a concentration of 70 µg. In comparison, the reference standard exhibited 55.06% inhibition at the same dose. A progressive enhancement in inhibitory potential was observed with increasing concentrations of the extract, indicating a clear dose-dependent relationship.
Table 6: H+/K+ATPase inhibitory activity of ethanolic leaves extracts of Persea macrantha (%inhibition)
| S. No. | Concentration (µg) | % Inhibition of ethanolic extract of Persea macrantha leaves | % Inhibition of omeprazole |
| 1 | 10 | 7.56 | 17.56 |
| 2 | 20 | 12.56 | 23.81 |
| 3 | 30 | 17.56 | 30.08 |
| 4 | 40 | 23.82 | 36.7 |
| 5 | 50 | 30.1 | 42.78 |
| 6 | 60 | 36.31 | 48.81 |
| 7 | 70 | 42.56 | 55.06 |
In comparison with standard anti-ulcer drugs suggests that the extract exhibits significant gastroprotective activity. The observed efficacy may be attributed to the combined effects of multiple phytoconstituents, which collectively contribute to acid neutralization and proton pump inhibition. While the activity is slightly lower than that of reference drugs, the results support the extract’s potential as a natural anti-ulcer agent.
Findings from acute toxicity studies
The acute toxicity was determined on female Albino wistar rats by acute toxicity class method of OECD Guideline No.423. The Ethanolic and Aqueous extracts of Persea macrantha Nees was given orally a dose of 2000 mg/kg body weight. There were no signs of toxicity or a death noted in the animals, so it is found to be safe. Based on the results it can be concluded that the product belongs to GHS (Globally Harmonized System) category 5. In the present study 1/10th and 1/5th of the NOAEL (No observed adverse effect level) dose of the test drug was chosen as 200 mg/kg and 400 mg/kg body weight.
Findings from in vivo studies
Aspirin-induced ulcer
The disease control group treated with aspirin showed a very high ulcer index (18.66), confirming successful ulcer induction. In contrast, the normal control group had a negligible ulcer index (0.05) with nearly complete protection (98.3%). The standard drug omeprazole markedly reduced ulcer severity, yielding an ulcer index of 1.13 and 95.4% protection, demonstrating its strong antiulcer efficacy. Among the test extracts, the ethanol extract (PMEE) was more effective than the aqueous fraction (PMAE). At 200 mg/kg, PME reduced the ulcer index to 3.25 with 61.4% protection, while at 400 mg/kg it achieved 6.98 with 81.84% protection, approaching the standard drug’s efficacy. Conversely, PMAE showed weaker activity, with 200 mg/kg giving 7.70 ulcer index and 40% protection, and 400 mg/kg yielding 10.44 ulcer indexes with 58.19% protection. Overall, the ethanol extract demonstrated dose-dependent Gastroprotective effects, with the higher dose showing substantial protection, whereas the aqueous extract was comparatively less effective.
Table 7: Observations
| Day/time | Clinical signs | Behavioral changes | Body weight in g | Mortality |
| 1hr | 0 | 1 | 180g | 0 |
| 2hr | 0 | 0 | 180g | 0 |
| 4hr | 0 | 0 | 178g | 0 |
| 24hr | 0 | 0 | 177g | 0 |
| Day (2-7) | 0 | 0 | 177g | 0 |
| Days (8-14) | 0 | 0 | 181g | 0 |
Note: 0, indicates the normal skin/fur, no tremors and alive, 1-mild lethargy and death
Table 8: Antiulcer activity of Persea macrantha leaves extracts on parameters of aspirin-induced gastric ulcer model
| Treatment (n=6) | Ulcer index | % protection |
| Normal control | 0.05+0.12*** | 98.3% |
| Disease control(aspirin) | 18.66+0.81 | _ |
| Standard (omeprazole) | 1.13+1.76*** | 95.4% |
| PMELD-200 mg/kg | 6.98+0.04*** | 61.4% |
| PMEHD-400 mg/kg | 3.25 6+3.56*** | 81.84% |
| PMALD-200 mg/kg | 10.44+0.11*** | 40% |
| PMAHD-400 mg/kg | 7.70+4.75*** | 58.19% |
Note: PMELD-Persea macrantha ethanolic low dose, PMEHD-Persea macrantha ethanolic high dose, PMALD-Persea macrantha aqueous low dose, PMAHD-Persea macrantha aqueous high dose and n=6, all the values are expressed as mean+SD, one-way ANOVA, followed by Dunnett’s test, ***P<0.001, **P<0.01 and *P<0.05 compared to disease control (Aspirin induced).
Table 9: Gastric volume, gastric pH and total acidity of aspirin-induced gastric toxicity
| Treatment (n=6) | Gastric volume | Gastric pH | Total acidity (mEq/l) |
| Normal control | 0.05+0.08*** | 4.51+0.6*** | 0.01+0.04*** |
| Aspirin induced | 3.06+0.64 | 2.2+0.46 | 132.8+20.72 |
| Omeprazole 20 mg/kg | 0.21+0.48*** | 6.35+0.54*** | 1.83+1.83*** |
| PMELD-200 mg/kg | 1.36+0.29*** | 5.6+0.37*** | 30.17+8.79*** |
| PMEHD-400 mg/kg | 0.31+0.27*** | 5.8+0.45*** | 14.17+2.63*** |
| PMALD-200 mg/kg | 2.06+0.10** | 4.13+0.22*** | 82.83+6.04* |
| PMAHD-400 mg/kg | 1.83+0.16*** | 4.95+0.16*** | 60.83+5.41** |
Note: PMELD-Persea macrantha ethanolic low dose, PMEHD-Persea macrantha ethanolic high dose, PMALD-Persea macrantha aqueous low dose, PMAHD-Persea macrantha aqueous high dose and n=6, all the values are expressed as mean+SD, one-way ANOVA, followed by Dunnett’s test, ***P<0.001, **P<0.01 and *P<0.05 compared to Disease control (Aspirin induced).
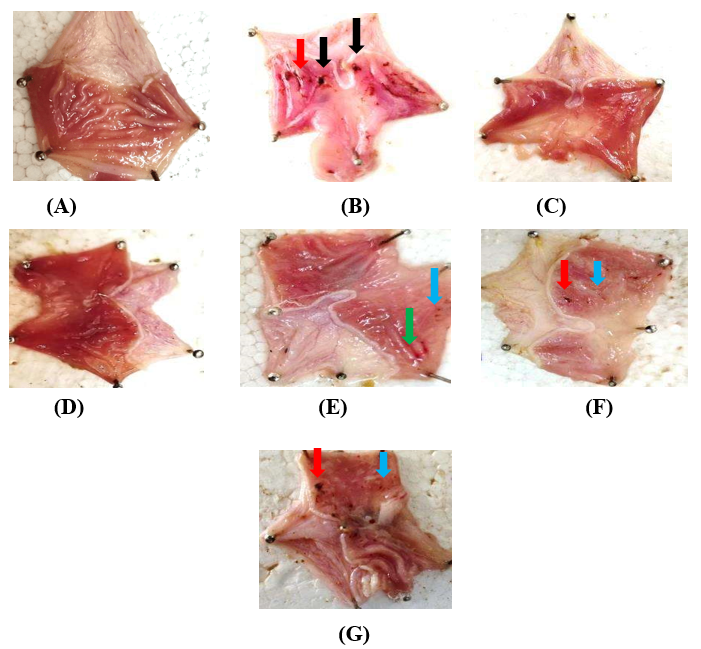
Fig. 5: ulcerated rat stomach (induced by aspirin) in fig. 5: A-normal control, B-aspirin induced, C-omeprazole treated, D-ethanolic high dose, E-ethanolic low dose, F-aqueous high dose, G-aqueous low dose. Black Colour indicates the presence of deep ulcer, red Colour indicates the presence of hemorrhagic streaks blue and green colour indicate the presence of spot ulcers and redness respectively
Ethanol-induced gastric toxicity
In the ethanol-induced ulcer, the disease control group exhibited a markedly high ulcer index (15.23), confirming severe gastric damage. The normal control group maintained a very low ulcer index (0.30) with nearly complete protection (98%), while the standard drug sucralfate at 100 mg/kg significantly reduced ulcer severity to 0.60, corresponding to 91.86% protection. Among the test extracts, the ethanol extract (PMEE) demonstrated stronger gastroprotective activity compared to the aqueous extract (PMAE). At 200 mg/kg, PME lowered the ulcer index to 7.78 with 48% protection, and at 400 mg/kg it further reduced the index to 3.26 with 78.6% protection, showing a clear dose-dependent effect. In contrast, PMAE was less effective, with 200 mg/kg yielding an ulcer index of 11.03 and 26% protection, while 400 mg/kg produced 11.40 with only 24% protection. Overall, PMEE at higher doses showed substantial antiulcer efficacy approaching that of sucralfate, whereas PMAE offered minimal protection, highlighting the superior role of the ethanol extract in mitigating ethanol-induced gastric injury.
Table 10: Ulcer index and % protection of ethanol-induced ulcer model
| Treatment (n=6) | % Ulcer index | % Protection |
| Normal control | 0.30+0.73*** | 98% |
| Ethanol induced | 15.23+0.75 | _ |
| Sucralfate 100 mg/kg | 0.60+1.47*** | 91.86% |
| PMELD-200 mg/kg | 7.78+3.82*** | 48% |
| PMEHD-400 mg/kg | 3.26+3.66*** | 78.6% |
| PMALD-200 mg/kg | 11.03+0.52** | 26% |
| PMAHD-400 mg/kg | 11.40+0.60* | 24% |
Note: PMELD-Persea macrantha ethanolic low dose, PMEHD-Persea macrantha ethanolic high dose, PMALD-Persea macrantha aqueous low dose, PMAHD-Persea macrantha aqueous high dose and n=6, all the values are expressed as mean+SD, one-way ANOVA, followed by Dunnett’s test, ***P<0.001, **P<0.01 and *P<0.05 compared to disease control (Ethanol induced).
Table 11: Effect of gastric volume, gastric pH, and total acidity of ethanol-induced ulcer model
| Treatment (n=6) | Gastric volume (ml) | Gastric pH | Total acidity (mEq/l) |
| Normal control | 0.05+0.08*** | 4.33+0.42*** | 0.50+1.22*** |
| Ethanol induced | 3.20+0.50 | 2.23+0.38 | 146.2+18.9 |
| Sucralfate 100 mg/kg | 0.45+0.49*** | 6.88+0.17*** | 2.66+2.42*** |
| PMELD-200 mg/kg | 1.53+0.25*** | 5.58+0.60*** | 40.00+8.98*** |
| PMEHD-400 mg/kg | 0.70+0.37*** | 5.91+0.14*** | 16.33+2.58*** |
| PMALD-200 mg/kg | 2.08+2.23** | 4.81+0.38*** | 87.33+2.08*** |
| PMAHD-400 mg/kg | 1.93+0.12*** | 4.91+0.11*** | 69.33+4.73*** |
Note: PMELD-Persea macrantha ethanolic low dose, PMEHD-Persea macrantha ethanolic high dose, PMALD-Persea macrantha aqueous low dose, PMAHD-Persea macrantha aqueous high dose and n=6, all the values are expressed as mean+SD, one-way ANOVA, followed by Dunnett’s test, ***P<0.001, **P<0.01 and *P<0.05 compared to Disease control (Ethanol induced).
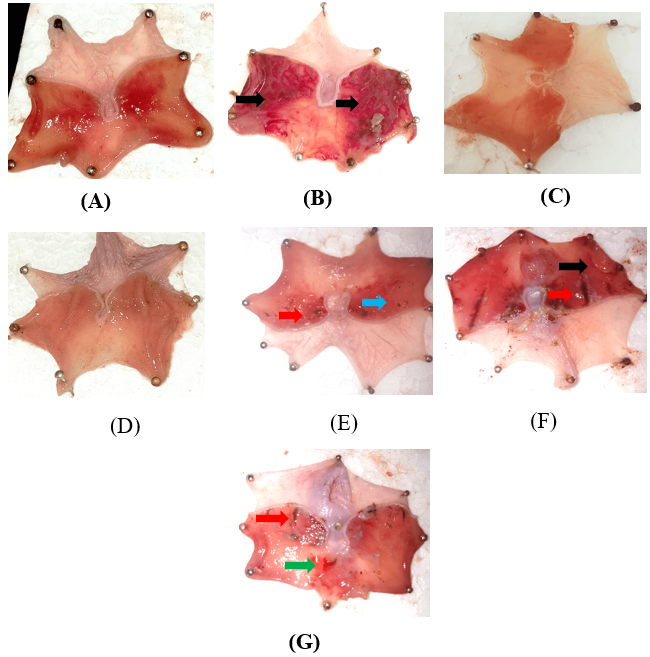
Fig. 6: Ulcerated rat stomach (induced by ethanol) in fig. no 6 A-normal control, B-aspirin induced, C-omeprazole treated D-ethanolic high dose, E-ethanolic low dose, F-aqueous high dose, G-aqueous low dose. Black colour indicates the presence of deep ulcer red colour indicates the presence of hemorrhagic streaks blue and green colour indicates the presence of spot ulcers and redness respectively.
DISCUSSION
Gastric ulcers represent a major global health burden, with their prevalence continuing to rise due to lifestyle habits, pharmacological influences, and microbial infections. Among these risk factors, psychological stress emerges as a particularly critical contributor, capable of disturbing the body both mentally and physiologically. Prolonged stress disrupts neuroendocrine regulation, weakens mucosal defense mechanisms, and enhances gastric acid secretion, thereby creating a favorable environment for ulcer development [15]. Another major risk factor is the widespread use of non-steroidal anti-inflammatory drugs (NSAIDs), particularly among cardiac patients. These medications are commonly prescribed to prevent thrombotic events such as myocardial infarction by inhibiting platelet aggregation. However, NSAIDs also suppress prostaglandin synthesis, which compromises gastric mucosal integrity and leads to ulceration. The present study investigated the effect of ethanolic and aqueous leaves extracts of Persea macrantha plant on rats by performing various antiulcer models. The observation of present study revealed the Gastroprotective potential effect of Persea macrantha. These findings are supported by in silico prediction of interaction of reported phytochemicals of Persea macrantha with therapeutic targets.
Mainly, there are 3 Targets were identified to play crucial roles in metabolic process of gastric ulcer, through peer literature review 3 key targets (PTPN11, NFKB1, CYP1B1), majorly related to ulcer, were identified for in silico docking studies. PTPN11 (Tyrosine-protein phosphatase non-receptor type 11 involved in cell signaling pathway that is Ras signaling pathway in gastric ulcer it may interact with the H. Pylori CagA protein leads to inflammation and epithelial damage [16], which means this gene has a role in causing gastric ulcer blocking this gene by interaction with the beta-phellandrene phytoconstituents may reduce the causing of ulcer or may involve in ulcer healing process by acting through the Ras signaling pathway, this interaction suggests that beta-phellandrene may modulate PTPN11 activity, potentially affecting antiulcer property. NFKB1 (Nuclear Factor NF-kappa B) plays a significant role in managing gastric ulcer, through the Toll-like receptor 4 signaling pathway, which is involved in managing or ulcer healing process induced by the stress by regulating the glucocorticoid production, which helps in suppress inflammation and epithelial. Tryptophan metabolism influences gastric mucosal protection and modulates oxidative stress. Also, activation of this gene may trigger ulcer formation through H. pylori and may heal ulcer by reverse mechanism [17, 18]. Machiline exhibited strong binding affinity with NFKB1, which subsequently may have effect on ulcer healing.
CYP1B1 (Cytochrome P450 family 1 subfamily B member 1) as a contributor to oxidative stress and inflammatory signaling by mediating metabolism of xenobiotics and endogenous substrates, can generate reactive oxygen species, which exacerbate epithelial damage and delaying of ulcer healing [19]. Docking studies showed that effective binding of sesamin phytoconstituents with the CYP1B1 through tryptophan metabolism pathway may involve in gastric ulcer healing by reducing formation of Reactive oxygen species. Aspirin, non-steroidal anti-inflammatory drugs (NSAIDS) can induce gastric ulcer by inhibiting the production of protective prostaglandins in the stomach through the blockade of cyclooxygenase (COX-1) enzyme [20, 21]. This inhibition decreases the secretion of protective mucus and bicarbonate, rendering the stomach lining more susceptible to damage. Moreover, NSAIDs can provoke an inflammatory response in the gastric mucosa, leading to elevated levels of reactive oxygen species (ROS), which further contribute to mucosal injury and the development of ulcers. Aspirin-induced ulcer genesis is associated with several factors, including elevated gastric acid secretion, depletion of gastric wall mucin, and vascular injury. In the aspirin-induced ulcer model, rats were pretreated with plant leaf extracts and omeprazole (20 mg/kg), a standard anti-ulcer drug. Both treatments resulted in a significant reduction (P<0.001) in ulcer index compared to the disease control group. The plant extracts demonstrated a dose-dependent and statistically significant (P<0.001) decrease in ulcer index as well the percentage of ulcer protection observed was 95.4% for omeprazole, 81.84% for PMEHD, 58.19% for PMELD, and 40% for both PMAHD and PMALD. Additionally, other evaluated parameters also showed significant differences (P<0.001) when compared to the disease control group, indicating the potential involvement of the prostaglandin pathway in the protective effects observed. In the ethanol-induced ulcer model, rats were pretreated with ethanolic and aqueous extracts of Persea macrantha leaves at doses of 200 mg/kg and 400 mg/kg, along with sucralfate at a standard dose of 100 mg/kg. Both plant extracts produced a significant reduction in ulcer index compared to the disease control group (P<0.001) and (P<0.01), respectively. The ethanolic extract exhibited a dose-dependent reduction in ulcer index, indicating a stronger protective effect at the higher dose. Interestingly, the aqueous extract at low dose (200 mg/kg) showed a more significant effect (P<0.01) compared to the high dose (400 mg/kg), which was less significant (P<0.05) when compared to the disease control group. Additionally, other evaluated gastric parameters, such as gastric pH, volume of gastric secretions, and total acidity, further supported the antiulcer activity of the extracts, suggesting that Persea macrantha possesses promising Gastroprotective properties. Both ethanolic and aqueous extracts of Persea macrantha were assessed and compared with the standard antacid formulation, aluminum magnesium hydroxide. The superior ANC observed with Persea macrantha extracts, particularly at the 1500 mg dose, may be responsible for its enhanced gastric protection, potentially due to a synergistic interplay of alkaline phytoconstituents, antioxidant properties, and mucosal protective effects. The ethanolic extract, being richer in non-polar bioactive, may concentrate these compounds more effectively, which could explain its slightly higher ANC compared to the aqueous extract. In the H⁺/K⁺ ATPase enzyme inhibition assay, standard omeprazole was used as a reference to evaluate the ethanolic leaf extract of Persea macrantha. The extract demonstrated a clear dose-dependent inhibitory effect, with increasing concentrations from 10 to 70 µg resulting in progressively higher enzyme inhibition. At the maximum tested dose of 70 µg, the extract exhibited inhibitory activity that closely approached the efficacy of omeprazole, indicating its potential as a natural proton pump inhibitor. In all the tested extracts, the ethanolic preparations demonstrated a more potent, dose-dependent anti-ulcer effect compared to the aqueous extracts, as reflected in the ulcer index values. This enhanced activity may be attributed to the broader solubility spectrum of ethanol, which facilitates the extraction of semi-polar bioactive compounds. These constituents possess superior membrane permeability and bioavailability, thereby increasing the yield of pharmacologically active phytochemicals. Consequently, ethanolic extracts exhibit greater therapeutic efficacy than their aqueous counterparts [22].
CONCLUSION
The results of in vitro, in vivo animal studies and the in silico studies revealed the multitargeting interactions with important, relative targets involved in Gastroprotective activity and hence highlight the potential use of Persea macrantha for the management of ulcer. In vivo studies have demonstrated that both ethanolic and aqueous extracts of Persea macrantha possess significant Gastroprotective properties, as evidenced by reductions in ulcer index, gastric volume, and increases in gastric pH. Among the tested doses, the high-dose ethanolic extract exhibited the most pronounced protective effect on gastric tissue, outperforming its aqueous counterpart. These findings suggest that the ethanolic extract, particularly at higher concentrations, confers superior antiulcer efficacy, likely due to enhanced bioavailability of active phytoconstituents. The Insilco analysis, supported by the review of literature data suggests that this observed effects may be mediated through interaction of phytochemicals such as beta-phellandrene, machiline and sesamin with critical ulcer-related targets such as PTPN11, NFKB1, and CYP1B1, respectively.
ACKNOWLEDGMENT
The authors wish to thank the following institutions for their support: the Visveswarapura Institute of Pharmaceutical Sciences, Bengaluru, India; the college of College of Medicine and the Clinical Pharmacy at King Faisal University, Saudi Arabia; and the College of Medicine at Dar Al Uloom University, Saudi Arabia.
FUNDING
This work was supported by the Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research, King Faisal University, Saudi Arabia [Grant No. KFU260696].
AUTHORS CONTRIBUTIONS
Faisal Mamdouh Alhajri: Conceptualization, Investigation, Data curation, Writing – original draft. Ravi Shankar N: Methodology, Software, Formal analysis, Validation, Writing – review and editing. Pramod S: Software, Data curation, Visualization, Investigation. Sibghatullah Muhammad Ali Sangi: Formal analysis, Validation, Writing – review and editing. Girish Meravanige: Supervision, Resources, Project administration, Writing – review and editing. Sreeharsha Nagaraja: Conceptualization, Supervision, Funding acquisition, Writing – review and editing.
CONFLICT OF INTERESTS
Declared none
REFERENCES
Danole VM, Raut YB, Bais SK. A review of herbal remedies for peptic ulcer. Int J Pharm Herb Technol. 2024;2(4):2533-48.
Kumar A, Kumar D, Kumar R, Prasad J, Kumar M, Joshi P. Peptic ulcers and their complications. J Drug Delivery Ther. 2020;10(3-s):256-61. doi: 10.22270/jddt.v10i3-s.4082.
Reyes VE. Helicobacter pylori and its role in gastric cancer. Microorganisms. 2023 May 17;11(5):1312. doi: 10.3390/microorganisms11051312, PMID 37317287.
Kanagali SN, Patil BM, Khanal P, Unger BS. Cyperus rotundus L. reverses the olanzapine-induced weight gain and metabolic changes-outcomes from network and experimental pharmacology. Comput Biol Med. 2022;141:105035. doi: 10.1016/j.compbiomed.2021.105035, PMID 34802711.
Tatiya AU, Beldar VG, Surana SJ. Pharmacognostic, phytochemical and pharmacological review on Machilus macrantha Nees. Eur J Pharm Med Res. 2017;4(3):174-8.
Shrikanth P, Naik TI. Comparative antioxidant effect of leaf and bark of Persea macrantha (Nees.) Kosterm an in vitro study. Int J Res Anal Rev. 2024;11(2):3397-402.
Japti VP, Patil MB, Unger BS, Mallapur SP, Shamnewadi A, Patil VS, Patil S, Desai AV. Network pharmacology-based toxicity molecular docking and molecular dynamics analysis of phytoconstituents from roots of Nerium indicum L. Pharmacol Res Mod Chin Med. 2025;16:100640. doi: 10.1016/j.prmcm.2025.100640.
Shamnewadi A, Unger BS, Palit P, Mallapur SP, Patil VS, Darasaguppe Ramachandra HD. In silico and in vivo pharmacological study of Acmella paniculata flowers for anti-inflammatory and antiarthritic potential. Chem Biodivers. 2025;22(10):e00428. doi: 10.1002/cbdv.202500428, PMID 40387128.
Organisation for Economic Co-operation and Development (OECD). Test No. 423: Acute oral toxicity-acute toxic class method. Paris: OECD Publishing; 2002. doi: 10.1787/9789264071001-en.
Choi JI, Raghavendran HR, Sung NY, Kim JH, Chun BS, Ahn DH. Effect of fucoidan on aspirin-induced stomach ulceration in rats. Chem Biol Interact. 2010;183(1):249-54. doi: 10.1016/j.cbi.2009.09.015, PMID 19788892.
Mohammed MA, El-Hadi AE, Mohammed EA. Gastro-protective effects of green banana (Musa cavendishii Lamb.) pulp powder on aspirin-induced gastric ulcer in albino rats. Med Aromat Plants. 2021;10(1):367. doi: 10.35248/2167-0412.21.10.367.
Bodhankar S, Raygude K, Kandhare A, Ghosh P. The ameliorative effect of fisetin a bioflavonoid on ethanol-induced and pylorus ligation-induced gastric ulcer in rats. Int J Green Pharm. 2011;5(3):236-43. doi: 10.4103/0973-8258.91233.
Bharathi KN, Ankitha M. Evaluation of anti-ulcer activity of aqueous and ethanolic extracts of whole plant of Byttneria herbaceae in rats. World J Pharm Pharm Sci. 2024;13(4):1075-89. doi: 10.20959/wjpps20244-26993.
Adinortey MB, Ansah C, Galyuon I, Nyarko A. In vivo models used for evaluation of potential antigastroduodenal ulcer agents. Ulcers. 2013;2013:1-12. doi: 10.1155/2013/796405.
Kumar A, Prasad A, Kumar A, Kumar A, Kumar A. In silico exploration of piperine targeting PTPN11: a novel approach for gastric ulcer management. J Med Pharm Allied Sci. 2022;11(6):5385-9. doi: 10.55522/jmpas.V11I6.1865.
Li WS, Lin SC, Chu CH, Chang YK, Zhang X, Lin CC. The gastroprotective effect of naringenin against ethanol-induced gastric ulcers in mice through inhibiting oxidative and inflammatory responses. Int J Mol Sci. 2021;22(21):11985. doi: 10.3390/ijms222111985, PMID 34769415.
Zebrowska Nawrocka M, Wosiak A, Pietrzak J. NFKB2 gene expression in patients with peptic ulcer diseases and gastric cancer. Mol Biol Rep. 2020;47(10):7835-42. doi: 10.1007/s11033-020-05299-5.
Joshi DC, Joshi N, Kumar A, Maheshwari S. Recent advances in molecular pathways and therapeutic implications for peptic ulcer management: a comprehensive review. Horm Metab Res. 2024;56(9):615-24. doi: 10.1055/a-2256-6592, PMID 38467155.
Umamaheswari M, Asokkumar K, Rathidevi R, Sivashanmugam AT, Subhadradevi V, Ravi TK. Antiulcer and in vitro antioxidant activities of Jasminum grandiflorum L. J Ethnopharmacol. 2007;110(3):464-70. doi: 10.1016/j.jep.2006.10.017, PMID 17125945.
Bhajoni PS, Meshram GG, Lahkar M. Evaluation of the antiulcer activity of the leaves of Azadirachta indica: an experimental study. Integr Med Int. 2016;3(1-2):10-6. doi: 10.1159/000442750.
Paricharak SP, Hugar S. Evaluation of antiulcer activity of Moringa oleifera pods extract. Eur J Pharm Med Res. 2020;7(6):702-8.
Chaabani E, Abert Vian M, Bettaieb Rebey I, Bourgou S, Zar Kalai F, Chemat F. Ethanol–water binary solvent affects phenolic composition and antioxidant ability of Pistacia lentiscus L. fruit extracts: a theoretical versus experimental solubility study. Food Measure. 2023;17(5):4705-14. doi: 10.1007/s11694-023-01983-9.