
Int J Curr Pharm Res, Vol 17, Issue 2, 1-5Original Article
A STUDY TO EVALUATE THE EFFICACY OF ORAL TERBINAFINE AND A COMBINATION OF ORAL TERBINAFINE AND TOPICAL SERTACONAZOLE IN TINEA CORPORIS PATIENTS ATTENDING DERMATOLOGY OUTPATIENT DEPARTMENT IN A TERTIARY CARE HOSPITAL, HYDERABAD
SWATHI MIRYALA1, M. SNEHA MANJU2*, PURAM GIRI RAJA3, MAHESH KUMAR K.4
1Department of Pharmacology, GMC Vikarabad, Telangana, India. 2*Department of Pharmacology, MediCiti Institute of Medical Sciences, Hyderabad, India. 3Department of Pharmacology, GMC Nagarkurnool, Telangana, India. 4Department of Pharmacology, Osmania Medical College, Hyderabad, India
*Corresponding author: M. Sneha Manju; *Email: sneha.vims@gmail.com
Received: 10 Dec 2024, Revised and Accepted: 22 Feb 2025
ABSTRACT
Objective: To evaluate the efficacy of Oral Terbinafine (250 mg) and a combination of oral terbinafine (250 mg) and topical sertaconazole (2%) in patients with Tinea corporis attending Dermatology Out Patient Department (OPD), in a tertiary care centre.
Methods: An open-labeled comparative study was conducted in Dermatology OPD at Osmania General Hospital for 12 mo. Patients diagnosed with tinea corporis were randomly assigned into two groups: Group A: 50 Patients received Oral Terbinafine (250 mg) and Group B: 50 Patients received combination of Oral Terbinafine (250 mg) and Topical Sertaconazole (2%) for 2 w. The patients were advised to come after 1st and 2nd w for follow-up visits.
Results: The study involved 100 patients with tinea corporis, with a mean age of 32.42±8.48 years in Group A and 33.38±7.76 years in Group B. Most participants were males (Group A – 62%, Group B – 80%). Clinical features (itching, burning, scaling, crusting, vesicles, pustules) showed a significant reduction in Group B compared to Group A after 2 w of treatment, with a statistically significant difference (P<0.000001). Lesion size decreased more in Group B (from 26.1±14.9 to 2.0±1.2) than in Group A (from 24.5±14.4 to 6.01±4.2), with a significant difference (P<0.0000001). All specimens in Group B were free of fungal growth.
Conclusion: Clinical cure and mycological cure were better with a Combination of Oral Terbinafine (250 mg) and Topical Sertaconazole (2%) than with Oral Terbinafine (250 mg) alone. Both drugs were well tolerated and no major adverse drug reactions were noted during the study period.
Keywords: Tinea corporis, Oral terbinafine, Sertaconazole, 4-point rating scale, Clinical cure, Mycological cure
© 2025 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2025v17i2.54212 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Dermatophytosis (Tinea) refers to a superficial fungal infection characterized by scaly lesions on keratinized tissues, primarily caused by dermatophytes such as Microsporum, Trichophyton, and Epidermophyton. These fungi can infect the skin, hair, and nails of the host, with tinea corporis being the most prevalent form, affecting the trunk and limbs [1-3].
According to the World Health Organization (WHO), the global prevalence of dermatophytosis is estimated to be between 20 and 25%, with higher rates observed in tropical and subtropical regions like India, where elevated temperatures and humidity prevail for most of the year [4]. Recent studies indicate that the prevalence of dermatophytosis in India ranges from 36.6% to 78.4%, with tinea corporis accounting for 39.1% of cases [5]. This condition affects individuals of all ages and both sexes but is more common in males, with a male-to-female ratio of approximately 5.7:1, particularly among those aged 21 to 50 y [2, 6].
While tinea infections are typically painless and superficial, neglect or misdiagnosis can lead to invasive fungal infections that may become resistant and result in widespread lesions. Such complications can have significant negative impacts on social, psychological, and occupational well-being, ultimately compromising the quality of life [2, 6].
Localized and mild cases of tinea corporis generally respond well to topical treatments alone; however, more severe, extensive, and inflammatory lesions often require systemic therapy. Various antifungal medications, both topical (e.g., azoles and allylamines) and systemic, are available for the treatment of tinea corporis. Topical treatments tend to demonstrate superior pharmacokinetics compared to their systemic counterparts [4]. Thus, a combination of therapies is anticipated to yield better clinical outcomes and mycological clearance than either systemic or topical treatments alone. To ensure broad coverage and minimize the risk of resistance development, the combination should consist of agents from different drug classes.
Despite the potential benefits of combination therapy, there is a lack of comparative studies in Indian literature examining the efficacy of combined systemic and topical treatments versus monotherapy with systemic antifungal agents [4]. Terbinafine [7-9] and Sertaconazole [10, 11] are reported to have higher efficacy among antifungals. Consequently, this study aims to evaluate the efficacy of oral Terbinafine alone and in combination with topical Sertaconazole for the treatment of tinea corporis.
MATERIALS AND METHODS
An open-labeled comparative study was conducted in Dermatology outpatient department at Osmania General Hospital for 12 mo. After obtaining ethical clearance from the institutional ethics committee of Osmania Medical College (with approval number ECR/300/Inst/AP/2013/RR-16), patients diagnosed with tinea corporis aged between 21 and 50 y, of both genders, not undergoing any other antifungal therapy and willing to provide informed consent for participation were included in this study. Patients diagnosed with systemic mycosis or mycosis affecting the hands, face, scalp, systemic diseases and pregnant or lactating women were excluded.
After obtaining written informed consent from the eligible participants, thorough general physical examination and local examination of the lesion (size and border) was done to assess the extent, severity of the skin lesions. Clinical assessment of the lesions, considering symptoms and signs like itching, burning, scaling, crusts, vesicles and pustules, were done by a 4-point rating scale (0-no symptoms/signs,1-mild, 2-moderate,3-severe). Direct microscopy of the Potassium hydroxide (KOH) wet mount of the skin scrapings was done to confirm the diagnosis. The demographic data and clinical findings of the participants were recorded in case report forms.
Diagnosed patients were randomly assigned into two groups: Group A: 50 Patients received Oral Terbinafine (250 mg) once daily for 2 w and Group B: 50 Patients received combination of Oral Terbinafine (250 mg) once daily for 2 w and Topical Sertaconazole (2%) was applied twice daily for 2 w. The patients were advised to apply the ointment and take the drugs at the same time every day. The patients were advised to come after 1st and 2nd week for follow-up visits.
The efficacy/treatment response was assessed by comparing the clinical improvement (by local examination and 4-point rating scale of signs and symptoms) of the lesions before and after 2 w of treatment. The safety was assessed by monitoring and recording any adverse effects that occurred during the period of study.
Statistical methods
The data was entered in the Microsoft Excel 2019 version. The data was analyzed using Microsoft Excel 2019 and Epi Info 7.2.0. Descriptive and inferential statistical analyses were used in the present study. Results on continuous measurements were presented as mean±SD and results on categorical measurements were presented in percentages. Significance was assessed at a 5% level of significance. Student t-test was used to compare inter-group variation for continuous variables. The Chi-square test was used to compare categorical variables.
RESULTS
In this study, 100 patients diagnosed with tinea corporis both clinically (based on clinical presentation) and mycologically (based on the presence of fungal growth on KOH wet mount) were recruited over 12 mo and divided into Group A and Group B, 50 patients each. The results obtained from this study are as follows; among Group A, 40% belonged to the age group of 21-29 y, followed by 31-39 y (36%), and in Group B, 40% belonged to 31-39 y, followed by 21-29 y (38%), as shown in table 1.
Table 1: Shows the age-wise distribution
| Age group | Group A | Percentage | Group B | Percentage |
| 21-29 y | 20 | 40 | 19 | 38 |
| 31-39 y | 18 | 36 | 20 | 40 |
| 41-49y | 12 | 24 | 11 | 22 |
| Total | 50 | 100 | 50 | 100 |
| Mean±SD | 32.42 ± 8.48 y | 33.38 ± 7.76 y |
In this study, 62% were males and 38% were females in Group A and 80% were males and 20% were females in Group B, as shown in table 2. There was no statistically significant difference observed between the mean heights, weights and BMI of both the groups.
Table 2: Shows the gender distribution
| Gender | Group A | Percentage | Group B | Percentage |
| Males | 31 | 62 | 40 | 80 |
| Females | 19 | 38 | 10 | 20 |
| Total | 50 | 100 | 50 | 100 |
In Group A – 82% had itching, 64% had burning, 98% had scaling, 80% had crusting, and 66% had vesicles and pustules. The mean size of the lesion was 24.5±14.4 cm, with 72% having well-defined borders. In Group B – 84% had itching, 64% had burning, 96% had scaling, 80% had crusting, 44% had vesicles and pustules. The mean size of the lesion was 26.1±14.9 cm, with 82% having well-defined borders, as shown in fig. 1.
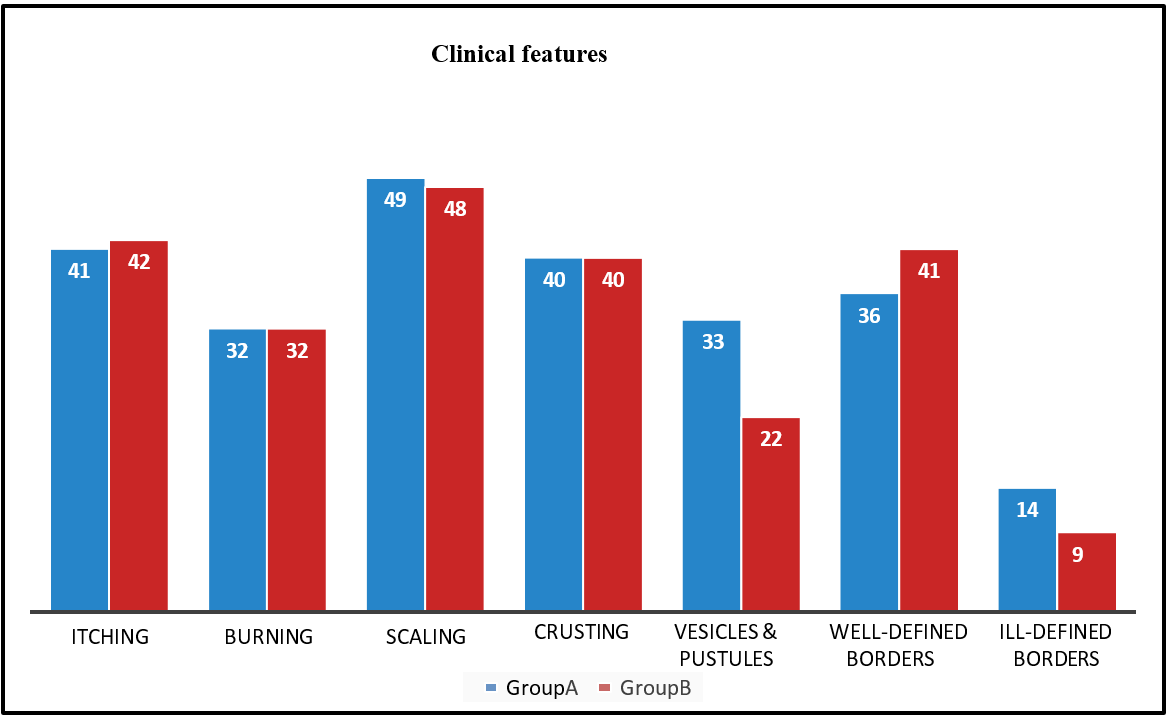
Fig. 1: Shows the clinical features in both study groups A (n = 50) and B (n = 50)
In Group A, 4% had family history of tinea infections and none of them had family history of tinea infections in Group B. All the specimens had fungal growth on KOH mount in both the groups.
Clinical features were assessed using a 4-point rating scale at baseline, after Week 1 and Week 2 in both groups A and B, and are summarized as shown in Tables 3,4,5,6,7, and fig. 2. As observed, there was a statistically significant clinical cure in Group B as compared with Group A (**: p<0.01).
After treatment for two weeks in Group A fungal growth was reported in 16% of the cases and among Group B there was no fungal growth reported in all specimens i. e. 100% mycological cure as shown in fig. 3.
Table 3: Showing means of itching using a 4-point rating scale at various visits:
| Itching | Group A | Group B | t-test (p-value) |
| Baseline | 2.4±0.4 | 2.3±0.5 | 0.2 |
| Week 1 | 1.9±0.3 | 1.7±0.4 | 0.005** |
| Week 2 | 1.4±0.2 | 0.8±0.3 | 0.0003** |
Table 4: Showing means of burning using a 4-point rating scale at various visits
| Burning | Group A | Group B | t-test (p-value) |
| Baseline | 1.9±0.5 | 1.8±0.6 | 0.3 |
| Week 1 | 1.6±0.4 | 1.4±0.5 | 0.02** |
| Week 2 | 1.1±0.4 | 0.7±0.3 | 0.0000001** |
Table 5: Showing means of scaling using a 4-point rating scale at various visits
| Scaling | Group A | Group B | t-test (p-value) |
| Baseline | 2.9±0.3 | 2.8±0.2 | 0.05 |
| Week 1 | 2.4±0.6 | 2.1±0.7 | 0.02** |
| Week 2 | 1.6±0.6 | 0.8±0.1 | <0.0000001** |
Table 6: Showing means of crusts using a 4-point rating scale at various visits
| Crusting | Group A | Group B | t-test (p-value) |
| Baseline | 2.1±0.6 | 2.3±0.7 | 0.12 |
| Week 1 | 1.7±0.8 | 1.3±0.9 | 0.02** |
| Week 2 | 1.1±0.5 | 0.5±0.4 | <0.0000001** |
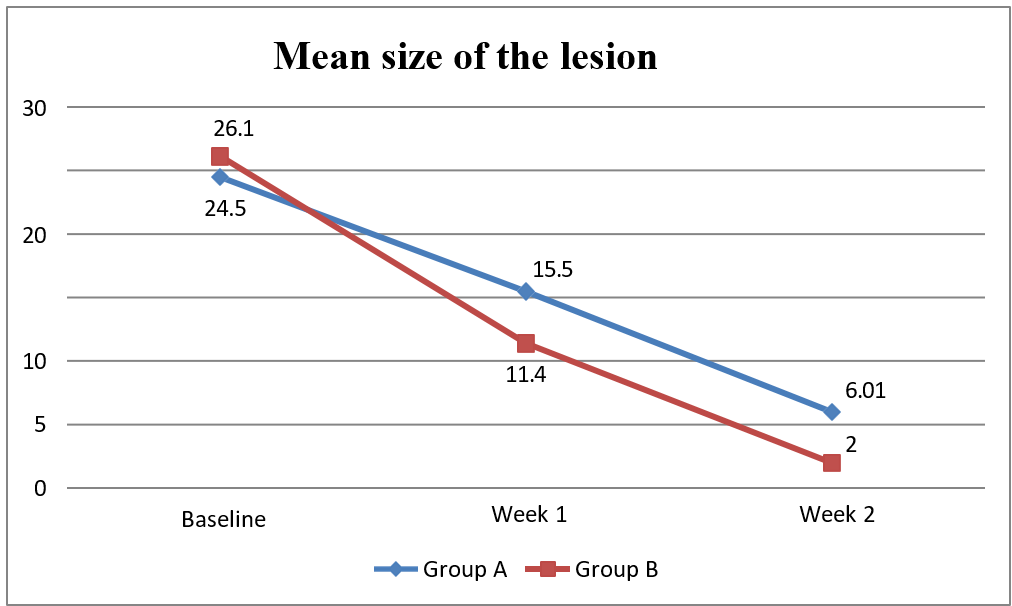
Fig. 2: Showing the mean size of the lesion (in centimeters) at various visits
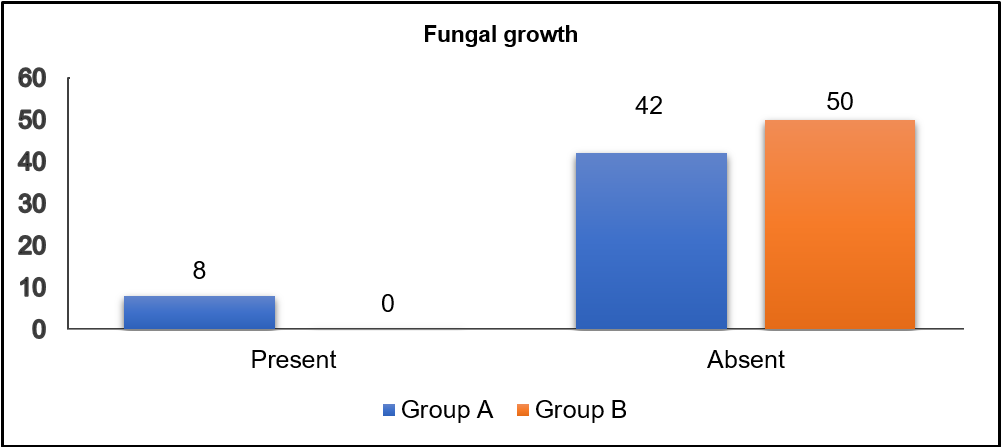
Fig. 3: Showing KOH wet mount details after two weeks of treatment
Table 7: Showing means: vesicles and pustules using a 4-point rating scale at various visits
| Vesicles and pustules | Group A | Group B | t-test (p-value) |
| Baseline | 1.2±0.8 | 1.3±0.7 | 0.5 |
| Week 1 | 1.0±0.7 | 0.7±0.5 | 0.01** |
| Week 2 | 0.7±0.4 | 0.4±0.2 | 0.000001** |
In the study population: in Group A-Adverse drug reactions were reported in 10% of the cases and in Group B-Adverse drug reactions were reported in 8% of the cases. Liver function tests and renal function tests were normal both before and after the completion of treatment.
DISCUSSION
In the present study, the majority of the patients were in age group of 21-29 y in Group A (32.42±8.48 y) and in Group B, the majority belonged to 31-39 y (33.38±7.76 y), the results were similar to other studies conducted by Sumitha A et al. [12], Choudhary S et al. [13], Chatterjee D et al. [14], Tamilselvan A et al. [15], Jerajani H R et al. [16], Siddiqui et al. [17]. The higher incidence in this age group may be attributed to increased outdoor activities and exposure. In both study groups, more than two-thirds were males, with the male-to-female ratio in Group A-31:19 and Group B-40:10, these findings are in congruence with other studies conducted by Sumitha A et al. [12], Chatterjee D et al. [14], Tamilselvan A et al. [15], Jerajani H R et al. [16], Siddiqui et al. [17] where there was higher occurrence in males. The greater occurrence in males may be because of their occupation or because of under-reporting in females.
In this study, at baseline, 82% of Group A and 84% of Group B had itching. After 2 w of treatment, there was a 48% cure in Group A and complete cure (100%) in Group B. The clinical cure rates were consistent with the study done by Shekhar et al. [18] and were better as compared with other studies done by Choudhary S et al. [13], Chatterjee D et al. [14], Tamilselvan A et al. [15], Jerajani H R et al. [16]. In Group A and Group B, 64% had burning, which had cure rates of 60% in Group A and 90% in Group B on follow-up. In Group A 98% and 96 % in Group B had scaling, and after 2 w of treatment, there was a complete cure in both groups A and B. This study has higher cure rates in comparison with other studies done by Sumitha A et al. [12], Choudhary S et al. [13]. In Group A, 80% had crusting, 66% had vesicles and pustules and among Group B, 80% had crusting, 44% had vesicles and pustules. On follow-up, in Group A there was a reduction of 30 % in crusting and 35% in vesicles and pustules and Group B had a complete cure. The results were better compared to other studies, Sumitha A. et al. [12], Jerajani H R et al. [16]. The baseline mean size of the lesion (in centimeters) was 24.5±14.4 which decreased to 6.01±4.2 in Group A and the mean size decreased from 26.1±14.9 to 2.0±1.2 in Group B, after 2nd week. The results were better than the study done by Tamilselvan A. et al. [15].
These differences in overall clinical cure rates may be due to the use of only topical antifungals (Terbinafine, Sertaconazole) in these studies, which is in contrast to our study treatment, which combines both systemic (oral Terbinafine) and topical (Sertaconazole) antifungal therapies. The betterment in the clinical parameters in Group B can be due to the additional effect of Sertaconozole on the lesions. Hence, the combination was proven to be better, both clinically and also statistically. Complete mycological cure at the end of treatment was obtained in Group B and results were consistent with other studies, Sumitha A et al. [12], Choudhary S et al. [13], Chatterjee D et al. [14], Jerajani H R et al. [16]. The treatment was well tolerated in both groups, the results were in congruence with other studies, Sumitha A et al. [12], Choudhary S et al. [13], Chatterjee D et al. [14], Jerajani H R et al. [16]. No major adverse drug reactions were noted during the study period.
LIMITATIONS OF STUDY
It is an open-labeled comparative study, and hence results cannot be generalized to the entire population. The sample size is 100; with a larger sample size, the results would have been more accurate, and a long-term follow-up may show the drug's long-term benefits and adverse effects.
CONCLUSION
Clinical cure and mycological cure were better with the combination of Oral Terbinafine (250 mg) and Topical Sertaconazole (2%) than Oral Terbinafine (250 mg) alone. The improvement in the clinical parameters can be due to the additional effect of Sertaconozole on the lesions. Hence, combination was proven to be better both clinically and statistically. Both drugs were well tolerated and no major adverse drug reactions were noted during the study period.
ACKNOWLEDGMENT
The authors thank the Professor and Head of the Department of Pharmacology for expert advice. We are thankful to the Department of Dermatology, Osmania General Hospital, and the Department of Pharmacology, Osmania Medical College, for approving this study; special thanks to all the authors for their help in data collection, statistics, putting zeal into the study and all the patients who participated in the study.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
Data collection, writing, and revising the manuscript was done by Dr Swathi Miryala. Study conceptualization, designing the study methodology, result analysis, writing, and revising the manuscript were done by Dr M. Sneha Manju. Dr Puram Giri Raja and Dr Mahesh Kumar K helped with data collection, reviewing, and editing the manuscript.
CONFLICTS OF INTERESTS
Declared none
REFERENCES
Jartarkar SR, Patil A, Goldust Y, Cockerell CJ, Schwartz RA, Grabbe S. Pathogenesis immunology and management of dermatophytosis. J Fungi (Basel). 2021 Dec 31;8(1):39. doi: 10.3390/jof8010039, PMID 35049979, PMCID PMC8781719.
Rajagopalan M, Inamadar A, Mittal A, Miskeen AK, Srinivas CR, Sardana K. Expert consensus on the management of dermatophytosis in India (ectoderm India). BMC Dermatol. 2018 Jul 24;18(1):6. doi: 10.1186/s12895-018-0073-1, PMID 30041646, PMCID PMC6057051.
Gupta AK, Chaudhry M, Elewski B. Tinea corporis tinea cruris tinea nigra and piedra. Dermatol Clin. 2003 Jul;21(3):395-400. doi: 10.1016/s0733-8635(03)00031-7, PMID 12956194.
Sahoo AK, Mahajan R. Management of tinea corporis tinea cruris and tinea pedis: a comprehensive review. Indian Dermatol Online J. 2016 Mar-Apr;7(2):77-86. doi: 10.4103/2229-5178.178099, PMID 27057486, PMCID PMC4804599.
Naglot A, Shrimali S, Nath B, Gogoi H, Veer V, Chander J. Recent trends of dermatophytosis in Northeast India (Assam) and interpretation with published studies. Int J Curr Microbiol Appl Sci. 2015;4(11):111-20.
Weitzman I, Summerbell RC. The dermatophytes. Clin Microbiol Rev. 1995 Apr;8(2):240-59. doi: 10.1128/CMR.8.2.240, PMID 7621400, PMCID PMC172857.
Faergemann J, Zehender H, Denouel J, Millerioux L. Levels of terbinafine in plasma stratum corneum dermis epidermis (without stratum corneum) sebum hair and nails during and after 250 mg terbinafine orally once per day for four weeks. Acta Derm Venereol. 1993 Aug;73(4):305-9. doi: 10.2340/000155557300304, PMID 7904107.
Van Der Schroeff JG, Cirkel PK, Crijns MB, Van Dijk TJ, Govaert FJ, Groeneweg DA. A randomized treatment duration finding study of terbinafine in onychomycosis. Br J Dermatol. 1992 Feb;126 Suppl 39:36-9. doi: 10.1111/j.1365-2133.1992.tb00008.x, PMID 1531927.
Goodfield MJ, Rowell NR, Forster RA, Evans EG, Raven A. Treatment of dermatophyte infection of the finger and toe nails with terbinafine (SF 86-327 Lamisil) an orally active fungicidal agent. Br J Dermatol. 1989 Dec;121(6):753-7. doi: 10.1111/j.1365-2133.1989.tb08217.x, PMID 2532927.
Poojary SA. Topical antifungals: a review and their role in current management of dermatophytoses. Clin Dermatol Rev. 2017;1(3) Suppl 1:S24-9. doi: 10.4103/CDR.CDR_34_17.
Croxtall JD, Plosker GL. Sertaconazole: a review of its use in the management of superficial mycoses in dermatology and gynaecology. Drugs. 2009;69(3):339-59. doi: 10.2165/00003495-200969030-00009, PMID 19275277.
Sumitha A, Geetha M, Shashikala GH, Somashekar HS, Chandan KS. Efficacy and safety of 1% terbinafine hydrochloride versus 2% sertaconazole cream in the treatment of tinea corporis. Int J Basic Clin Pharmacol. 2017;4(3):474-8. doi: 10.18203/2319-2003.
Choudhary S, Bisati S, Singh A, Koley S. Efficacy and safety of terbinafine hydrochloride 1% cream vs. sertaconazole nitrate 2% cream in tinea corporis and tinea cruris: a comparative therapeutic trial. Indian J Dermatol. 2013 Nov;58(6):457-60. doi: 10.4103/0019-5154.119958, PMID 24249898, PMCID PMC3827518.
Chatterjee D, Ghosh SK, Sen S, Sarkar S, Hazra A, DE R. Efficacy and tolerability of topical sertaconazole versus topical terbinafine in localized dermatophytosis: a randomized observer-blind parallel-group study. Indian J Pharmacol. 2016 Nov-Dec;48(6):659-64. doi: 10.4103/0253-7613.194850, PMID 28066103, PMCID PMC5155466.
Selvan AT, Gutha G, Vijaybhaskar SR, Suthakaran R. Comparative evaluation of newer topical antifungal agents in the treatment of superficial fungal infections (tinea or dermatophytic). Int Res J Pharm. 2013;4(6):224-8. doi: 10.7897/2230-8407.04651.
Jerajani H, Janaki C, Kumar S, Phiske M. Comparative assessment of the efficacy and safety of sertaconazole (2%) cream versus terbinafine cream (1%) versus luliconazole (1%) cream in patients with dermatophytoses: a pilot study. Indian J Dermatol. 2013 Jan;58(1):34-8. doi: 10.4103/0019-5154.105284, PMID 23372210, PMCID PMC3555370.
Siddiqui SM, Phulari YJ, Hiremath RN, Patil C, Patel P. A clinic mycological evaluation of dermatophytic infections a single centre prospective observational study. Asian J Pharm Clin Res. 2022;15(4):122-6. doi: 10.22159/ajpcr.2022.v15i4.44454.
Shekar A, Mittal G, Chandane RD, Suranagi UD. A prospective randomized open-labeled parallel-group study to compare the efficacy of oral terbinafine versus oral terbinafine plus oral fluconazole in dermatophyte infections. Asian J Pharm Clin Res. 2023;16(5):104-8. doi: 10.22159/ajpcr.2023.v16i5.47224.