
Int J Curr Pharm Res, Vol 17, Issue 2, 53-58Original Article
EVALUATION OF ANTIDEPRESSANT ACTIVITY OF FICUS THONNINGII ROOT IN ALBINO MICE
ANU BALA*, MEENAKSHI MALHOTRA, AJEET PAL SINGH, AMAR PAL SINGH
Department of Pharmacology, St. Soldier Institute of Pharmacy, Lidhran Campus, Behind NIT (R. E. C.), Jalandhar –Amritsar by pass, NH-1, Jalandhar-144011, Punjab, India
*Corresponding author: Anu Bala; *Email: anubadsar2811@gmail.com
Received: 10 Dec 2024, Revised and Accepted: 22 Feb 2025
ABSTRACT
Objective: The present study aimed to evaluate the antidepressant effects of the ethanolic extract of Ficus thonningii (EERFT) in albino Swiss mice and to assess its potential as atherapeutic agent for depression.
Methods: Preliminary phytochemical analysis of Ficus thonningii revealed the presence of phenolic compounds, proteins, tannins, glycosides, carbohydrates, starch, vitamins, and minerals. Acute oral toxicity testing was performed using EERFT at doses of 5, 50, 300, and 1000 mg/kg via the oral route, with no observed behavioral changes or toxicity. Based on these results, doses of 200 mg/kg and 400 mg/kg were selected for further evaluation. The antidepressant activity of EERFT was assessed using the Forced Swim Test (FST) and Tail Suspension Test (TST), with fluoxetine (10 mg/kg) as the standard drug.
Results: In TST, the control group (0.9% w/v sodium chloride, 10 ml/kg) showed insignificant immobility, whereas EERFT at 200 mg/kg and 400 mg/kg significantly reduced immobility. Fluoxetine also demonstrated a significant reduction in immobility. In FST, the control group exhibited insignificant immobility and number of jumps, while EERFT at 200 mg/kg and 400 mg/kg significantly reduced immobility and increased the number of jumps. Fluoxetine produced similar significant effects.
Conclusion: The results support the traditional use of Ficus thonningii for managing depression. The ethanolic extract demonstrated significant antidepressant activity in both FST and TST models, comparable to fluoxetine. These findings suggest that Ficus thonningii contains bioactive compounds with antidepressant properties, offering potential for the development of novel neuropsychiatric therapies.
Keywords: Ficusthonningii, Antidepressant, Forced swim test, Tail suspension test
© 2025 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2025v17i2.6069 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Depression is a widespread mood disorder that profoundly affects an individual’s emotional state, thoughts, and behaviors. It is characterized by persistent sadness, loss of interest or pleasure in previously enjoyable activities, and a range of physical and psychological symptoms, including changes in appetite, sleep disturbances, difficulty concentrating, and feelings of hopelessness. Severe cases may even lead to suicidal ideation. According to the World Health Organization (WHO), depression currently impacts approximately 350 million people worldwide, with an increasing prevalence. Projections indicate that depression will become the second-largest contributor to global disease burden within the next decade. This condition is not limited to adults; it also affects children and adolescents, often going undiagnosed, further complicating the need for timely and effective treatment [1-4].
While synthetic antidepressants, such as tricyclic antidepressants (TCAs) and monoamine oxidase inhibitors (MAOIs), have been used effectively for decades, their application is often hindered by undesirable side effects, including sedation, dry mouth, blurred vision, cardiovascular risks, and severe drug-drug interactions. These limitations have spurred the search for alternative treatment options, particularly those derived from natural sources. Medicinal plants, with their wide array of bioactive compounds, have shown promise in offering safer and more effective antidepressant therapies. Flavonoids, alkaloids, and other plant-derived compounds have been extensively studied for their antidepressant-like effects in animal models, highlighting their potential in modulating key neurotransmitter systems such as serotonin and norepinephrine, which are central to the pathophysiology of depression [5-8].
One such plant of interest is Ficus thonningii, a multistemmed tree belonging to the Moraceae family. This evergreen or briefly deciduous tree is native to tropical and subtropical regions of Africa, thriving in light, deep, and well-drained soils [9, 10]. Traditionally, various parts of Ficus thonningii, including its leaves, bark, and latex, have been used to treat a range of ailments such as diabetes, arthritis, asthma, gastric discomfort, and mental health conditions. The tree’s phytochemical profile reveals the presence of alkaloids, flavonoids, terpenoids, tannins, and saponins compounds known for their pharmacological properties, including antioxidant, anti-inflammatory, and neuroprotective activities [11-13].
Despite its traditional use in treating mental health disorders, limited scientific evidence is available to substantiate the antidepressant properties of Ficus thonningii. Research on the therapeutic potential of its ethanolic extract could provide valuable insights into its pharmacological effects and mechanisms of action. Studies on other medicinal plants, such as Curcuma longa and Piper tuberculatum, have demonstrated significant antidepressant-like effects in animal models, often comparable to conventional antidepressants like fluoxetine and imipramine. These findings underscore the potential of plant-derived compounds as alternative therapies for depression, particularly for individuals seeking treatments with fewer side effects and lower risks of drug interactions [14-17].
The objective of this study is to evaluate the antidepressant activity of the ethanolic extract of Ficus thonningii in validated animal models, such as the forced swim test (FST) and tail suspension test (TST). This investigation aims to provide scientific validation for the traditional use of Ficus thonningii in managing depression and to explore its potential as a source of novel therapeutic agents for mood disorders.
Experimental design
MATERIALS AND METHODS
Healthy, adult albino Swiss mice of either sex weighing (25-40 g), maintained under standard laboratory conditions at temperature 25±2 °C for 12 h light and 12 h dark period will be employed for the experimentation. Food and water will be provided ad libitum. All the chemicals and biochemical reagents used in this study were of analytical grade and were freshly prepared before use. All chemicals of analytical grade were procured from Sigma chemical, USA and S. D. Fine Chem. Ltd., India.
Antidepressant animal model
Twenty-Fouradults either sex mice weighing between 25-30 g were obtained from the animal house of the department of pharmacology St. Soldier Institute of Pharmacy, Jalandhar, Punjab, India–144011. Healthy, adult Swiss albino mice of either sex weighing (25-30 g), maintained under standard laboratory conditions at temperature 25±2 °C and a 12 h light-12 h dark period will be employed for the experimentation. Food and water will be provided ad libitum.
No. of groups (N) = 04
No. of the animal in each group (n) = 06
Total no. of animals required = 24
a) Forced swimming test: In this model, the animal is placed in a glass cylinder (30 cm in diameter and 50-cm height) containing 40 cm of water at 23±1 °C for 5 min, forcing the mice to either swim or float.
b) Tail suspension test in mice: In this model, Groups of 5 animals are treated with the test compounds or the vehicle by intraperitoneal injection 30 min prior to testing. For the test the mice are suspended on the edge of a shelf 58 cm above a tabletop by adhesive tape placed approximately 1 cm from the tip of the tail. The duration of immobility is recorded for a period of 5 min. Mice is considered immobile when they hang passively and completely motionless for at least 1 min.
Procurement of Ficusthonningii root extract
Ficusthonningiirootethanolic extract was collected from the ShreedhaPhyto extracts Jaipur – 302019. The same group also provided a certification of the Plant’s identify and quality.
Acute oral toxicity study [1]
Acute toxicity study for the ethanolic extract of “Ficusthonningii” done according to the OECD guidelines No: 423 and low, medium and high dose will be selected for treatment. The overnight fasted mice will be divided into 04 groups, each group consisting of 06 animals. The EFT will be given in various doses (0, 10, 200 and 400 mg/Kg) by oral route with a gavage. After administration of the extract, the animal will be observed continuously for the first 2 h and at 24 h to detect changes in behavioral responses and also for tremors, convulsion, salivation, diarrhea, lethargy, sleep, and coma and also will be monitored up to 14 d for the toxic symptoms and mortality [18, 19].
Mouse as a model for depression and anxiety
Major advantages of using mouse as a model, is their remarkable similarity to human ingenetics, anatomy and physiology. More than 95% of the mouse genome is similar to human beings, making mouse genetic research specifically appropriate to human disease. Anxiety and depression Studies related to the Central Nervous System and brain is accomplished using animals as experimental models. Animal models form the backbone of preclinical research on the neurobiology of psychiatric disorders, and are employed as screening tools in the search for novel therapeutic agents. Rodents, especially mice have proven to be helpful in research as mice and humans share more than 90% of their genes in common. Furthermore, animal models are particularly helpful in situations when the impact of stress cannot be studied in humans because of ethical and other reasons Other advantages of selecting mice as a model are, cost-effectiveness, small dose in relation to body weight, easy to handle and an accelerated breeding time; these points will definitely make the research easily manageable for the researcher. In addition, animal models of depression and anxiety are crucial for identifying novel therapies for depression and anxiety [6-8, 19].
Phytochemical test
Molisch’s test for carbohydrates
Few drops of Molisch’s reagent were added to each of the portion dissolved in distilled water, this was then followed by addition of 1 ml of conc. H2SO4 by the side of the test tube. The mixture was then allowed to stand for two minutes and then diluted with 5 ml of distilled water. Formation of a red or dull violet color at the interphase of the two layers was a positive test [20-22].
Barfoed’s test monosaccharides
About 0.5 g each portion was dissolved in distilled water and filtered. 1 ml of the filtrate was then mixed with 1 ml of Barfoed’s reagent in a test tube and then heated on a water bath for a period of 2 min. Reddish precipitate of cuprous oxide was considered as a positive test [2].
Fehling’s test for free reducing sugar
About 0.5 g each portion was dissolved in distilled water and filtered. The filtrate was heated with 5 ml of equal volumes of Fehling’s solution A and B. Formation of a red precipitate of cuprous oxide was an indication of the presence of reducing sugars.
Fehling’s test for combined reducing sugars
About 0.5 g each portion was hydrolysed by boiling with 5 ml of dilute hydrochloric acid and the resulting solution neutralised with sodium hydroxide solution. To this, few drops of Fehling’s solution was added and then heated on a water bath for 2 min. Appearance of a reddish-brown precipitate of cuprous oxide indicates the presence of combined reducing sugars.
Test for tannins
About 0.5 g each portion was stirred with about 10 ml of distilled water and then filtered. Few drops of 1% ferric chloride solution were added to 2 ml of the filtrate occurrence of a blue-black, green or blue-green precipitate indicates the presence of tannins.
Borntrager’s test
About 0.2 g of each portion to be tested was shaken with 10 ml of benzene and then filtered. Five millilitres of the 10% ammonia solution was then added to the filtrate and thereafter the shaken. Appearance of a pink, red or violet colour in the ammoniacal (lower) phase was taken as the presence of free anthraquinones [2].
Liebermann-burchard test for steroids
To 0.2 g of each portion, 2 ml of acetic acid was added, the solution was cooled well in ice followed by the addition of conc. H2SO4 carefully. Color development from violet to blue or bluish-green indicated the presence of a steroidal ring i. e. aglycone portion of cardiac glycoside [2].
Test for terpenoids
A little of each portion was dissolved in ethanol. To it 1 ml of acetic anhydride was added followed by the addition of conc. H2SO4. A change in colour from pink to violet showed the presence of terpenoids.
Test for saponins
One g of each portion was boiled with 5 ml of distilled water filtered. To the filtrate, about 3 ml of distilled water was further added and shaken vigorously for about 5 min. Frothing which persisted on warming was taken as evidence for the presence of saponins.
Shinoda’s test for flavonoids
About 0.5 of each portion was dissolved in ethanol, warmed and then filtered. Three pieces of magnesium chips was then added to the filtrate followed by few drops of conc. HCl. A pink, orange, or red to purple colouration indicates the presence of flavonoids.
Ferric chloride test for flavonoids
About 0.5 of each portion was boiled with distilled water and then filtered. To 2 ml of the filtrate, few drops of 10% ferric chloride solution were then added. A green-blue or violet colouration indicated the presence of a phenolic hydroxyl group.
Leadethanoate test for flavonoids
Few quantity of the each portion was dissolved in water and filtered. To 5 ml of each of the filtrate, 3 ml of lead ethanoate solution was added. Appearance of a buff-coloured precipitate indicates the presence of flavonoids.
Hydroxide test for flavonoid
Few quantity of the each portion was dissolved in water and filtered; to this 2 ml of the 10% aqueous sodium hydroxide was later added to produce a yellow colouration. A change in colour from yellow to colourless on addition of dilute hydrochloric acid was an indication for the presence of flavonoids.
Test for alkaloids
Few quantity of the each portion was stirred with 5 ml of 1% aqueous HCl on water bath and then filtered. Of the filtrate, 1 ml was taken individually into 2 test tubes. To the first portion, few drops of Dragendorff’s reagent were added; occurrence of orange-red precipitate was taken as positive. To the second 1 ml, Mayer’s reagent was added and appearance of buff-coloured precipitate will be an indication for the presence of alkaloids.
Test for soluble starch
Few quantity of each portion was boiled with 1 ml of 5% KOH, cooled and acidified with H2SO4. A yellow colouration was taken as the presence of soluble starch.
Treatment
Standard dose of 10 mg/Kg Fluoxetine and ethanolic extract of Ficus Thonningii root of 200 and 400 mg/Kg is given orally daily to the animal as shown in (table 1) [23-26].
Parameters for depression in mice
The behavioral effects of an acute or sub acute (14 d course) will be orally administered. Ficusthonningiiethanolic extract (200, 400 mg/Kg) will be evaluated in male and female Swiss mice in forced swim test (FST) and Tail Suspension test (TST). The animals were housed in plastic cages in groups of ten per cage, at room temperature about 21±1 ℃, and with free access to water and food. They were kept on an artificial 12 h/12 h day/night cycle [23-25].
Table 1: Standard and test group for the treatment of mice by FicusThonningii
| Groups (where N=4) | Treatment |
| Group I | Naive animal, received standard pellet diet and tap water ad libitum daily. |
| Group II | Standard group received 10 mg/Kg Fluoxetine orally daily. |
| Group III | Test group-I received 200 mg/Kg ethanolic extract ofFicusThonningiiroot orally daily. |
| Group IV | Test group –II received 400 mg/Kg ethanolic extract ofFicusThonningiiroot orally daily. |
Forced swimming test
A 2L glass cylinder was filled with water at room temperature (approximately 25±2 ℃). The cylinder is 19 cm tall, 10 cm in diameter and filled to 13 cm (the 1600 ml point) with tap water and allowed to sit overnight to achieve room temperature.
Mice should not be able to touch the bottom of the cylinder with their tails, as this may alter their behavior (fig. 1a). Mice are picked up from their home cages in a plastic container with holes in the bottom to let out water, and are individually dropped (placed in, head downward, trying to ensure that the mouse’s head does not go underwater) into the glass cylinder and observed for immobility for one 6-min trial. Immobility is defined as the cessation of all movements except those necessary to stay afloat, such as paddling lightly with one. A stopwatch was used to record immobility [18, 20, 26].


Fig. 1a, b: Pictures (a) Forced swimmimng test and (b) Tail suspension test
Tail suspension test
This method is based on the observation that a mouse suspended by the tail shows alternate periods of agitation and immobility. For these experiments, the recording device was as follows: metallic gallows were connected to a nylon catheter (d = 1.5 ram, length = 350 ram) with a hook attached to its extremity. The distance between the floor of the device and the hook was 350 mm (fig. 1b). The mouse was hung on the hook by an adhesive tape placed 20 mm from the extremity of its tail.
The mouse was 150 mm away from the nearest object and was both acoustically and visually isolated. The articulated stylus of the gallows was connected to a Mareycapsula that transmitted any pressure difference to another capsula by a pneumatic connection. The receiver capsula was connected to a drawing stylus, marking on a cylinder covered with black smoke. The cylinder rotated at 2 cm/min regulated by an electric motor. This device provided an analogue record of the movements of the mouse. The device was set in order to ignore respiratory movements and recorded only body movements.
RESULTS
All the parameters were performed with suitable time interval to prevent unwanted stress in animals.
Evaluation of antidepressant effect of Ficusthonningiiroote thanolic extracts in TST and FST models [10, 12, 18, 22]
Tail suspension test (TST)
EERFT: ethanolic extract of root of Ficusthonningii
*P<0.05, #P<0.01, ##P<0.001, When compared with the control group. All values represent = mean±SEM, n= 6 in each group
Table 2: Effects of different doses of Ficusthonningiiroot ethanolic extract in antidepressant activity as compared to standard (Fluoxetine)
| S. No. | Group and dose | Immobility (sec.) |
| 1. | Control 0.9% w/v sodium chloride Normal saline (10 ml/Kg) or (1 ml/100 gm, p. o.) | 266±3.608 |
| 2. | EERFT 200 mg/kg (Test I) | 45.4±1.885* |
| 3. | EERFT400 mg/kg (Test II) | 38.8±2.666# |
| 4. | Standard dose (Fluoxetine-10 mg/kg) | 18.2±1.626## |
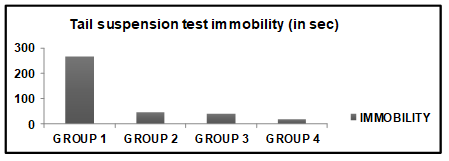
Fig. 2: Immobility result of tail suspension test
Graphical representation of effects of different doses of Ficusthonningiiroot ethanolicextracton antidepressant effect and compared to standard (Fluoxetine) (fig. 2).
Group 1: In this group animals were treated with normal saline solution in which mice Immobility period was 266±3.608 sec.
Group 2: In this group animals were treated with lower dose of ethanolic extract (200 mg/kg) solution in which mice Immobility period was 45.4±1.885 sec.
Group 3: In this group animals were treated with higher dose of ethanolic extract (400 mg/Kg) solution in which mice Immobility period was 38.8±2.666 sec.
Group 4: In this group animals were treated with fluoxetine (10 mg/Kg) solution in which mice Immobility period was 18.2±1.626 sec.
Treatment with fluoxetine significantly decreased the duration of immobility time (P<0.001) in TST (table 2). ethanolic and aqueous extract of Ficusthonningii treated mice also exhibited dose dependent significant decreased the duration of immobility time. The duration of immobility was also significantly reduced as compared to the vehicle treated group. But there is no significant difference between Ficusthonningii extracts treated animals and Fluoxetine treated animal. The above observation suggests that Ficusthonningii has antidepressant activity.
Table 3: Effects of different doses of Ficusthonningiiroot ethanolic extract in antidepressant activity as compared to standard (Fluoxetine)
| S. No. | Group | Forced swimming test |
| Immobility No. of jumps | ||
| 1. | Control 0.9% w/v sodium chloride Normal saline (10 ml/Kg) or (1 ml/100 gm, p. o.) | 258±2.986 |
| 2. | EERFT 200 mg/kg (Test I) | 58±2.384** |
| 3. | EERFT400 mg/kg (Test II) | 48.8±1.816** |
| 4. | Standard dose (Fluoxetine-10 mg/kg) | 38.8±1.706** |
EERFT: ethanolic extract of root of Ficusthonningii
#P<0.005, *P<0.01, **P<0.001 when compared with the control group. All values represent Mean±SEM,n= 6 in each group
Graphical representation of effects of different doses of Ficusthonningiiroot ethanolicextracton antidepressant effect and compared to standard (Fluoxetine) (fig. 3, table 3).
Group 1: In this group animals were treated with Normal saline solution in which mice Immobility period was 258±2.986 sec. and numbers of jumps were 2.6±0.378.
Group 2: In this group animals were treated with lower dose of Ethanolic extract (200 mg/Kg) solution in which mice Immobility period was 58±2.384 sec. and number of jumps were 12.8±0.374.
Group 3: In this group animals were treated with higher dose of Ethanolic extract (400 mg/Kg) solution in which mice Immobility period was 48.8±1.816 sec. and number of jumps were 14.6±0.928.
Group 4: In this group animals were treated with Fluoxetine (10 mg/Kg) solution in which mice Immobility period was 38.8±1.706 sec. and numbers of jumps were 18.8±0.816.
Treatment with Fluoxetine significantly decreased the duration of immobility time (P<0.001) in FST. Ethanolic and aqueous extract of Ficusthonningii treated mice also exhibited dose dependent significant decreased the duration of immobility time. The duration of immobility was also significantly reduced as compared to the vehicle treated group.
But there is no significant difference between Ficusthonningii extracts treated animals and Fluoxetine treated animal. The above observation suggests that KalanchoePinnata has antidepressant activity.
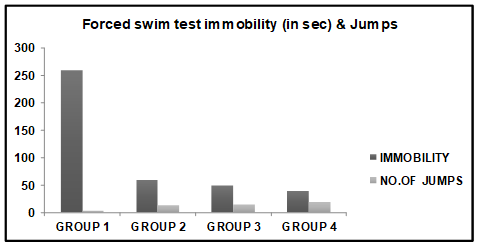
Fig. 3: Immobility result of tail suspension test
DISCUSSION
The common wild fig., Ficusthonningii, is extensively used in African ethnomedicine for treating a number of disease conditions which include diarrhoea, urinary tract infections, diabetes mellitus, gonorrhoea, respiratory infections and mental illnesses. This work aims to present a logical analysis of the nutritional, phytochemical and pharmacological properties of F. thonningii in relation to its therapeutic applications. Ficusthonningii contains various bioactive compounds which include alkaloids, terpenoids, flavonoids, tannins and active proteins, all of which contribute to its curative properties. In vitro and in vivo pharmacological studies revealed that F. thonningii possesses antimicrobial, antidiarrhoeal, antihelmintic, antioxidant, anti-inflammatory and analgesic properties. Acute and sub-chronic toxicity studies have shown that Ficusthonningii is non-toxic if administered orally in low doses. Scientific research has validated the ethnomedicinal claims that Ficusthonningii is useful in disease management. However, there is need to continue identifying, isolating and quantifying the active principles and possibly determine the mechanisms underlying its curative properties [17, 18].
F. thonningii contains various biologically active compounds that exhibit physiological effects which could be responsible for its curative potential in a broad range of disease conditions. These non-nutritive chemicals, commonly known as phytochemicals are naturally produced by the tree as protection against biotic and abiotic stresses. Most researchers used standard qualitative methods for screening for phytochemicals in F. thonningii. The main groups of phytochemicals isolated from F. thonningii are: alkaloids, terpenoids, flavonoids, tannins and essential oils [18, 23, 25].
Other phytochemicals with antinutritional properties present in F. thonningii include phytate (130 mg/100 g DM) and oxalate (230 mg/100 g DM). Despite the presence of these antinutritional factors, the leaves of F. thonningii still possess high feed value evidenced by their high palability and digestibility when used as fodder. F. thonningii leaves were shown to contain other secondary metabolites such as lignins, lignans, active carbohydrates and proteins which might also add to its remarkable pharmacological and biological activity.
Preliminary phytochemical analysis of Ficusthonningii revealed the presence of phenolic compound, Proteins, tannins, glycosides, carbohydrate, starch, vitamins and minerals etc.
Acute oral toxicity of Ficusthonningii was detected with ethanolic extracts (EERFT) having dose (5, 50, 300, 1000 mg/Kg) via oral route, shows no change in behavioral responses and observation shows no acute oral toxicity. Hence depending upon it, 200 mg/Kg and 400 mg/Kg dose was selected for the present experimental work. Successive isolation of botanical compounds from plant material is largely dependent on the type of solvent used in the extraction procedure. The traditional healers use primarily water as the solvent but we found in this study the plant extracts by ethanol provided more consistent activity compared to those extracted by water. The results of antidepressant activity of plant Ficusthonningii against the investigated FST and TST parameters are shown in table 2 and 3. The higher dose extract produced more effect as compared to lower extract in both the parameters. This might be due to the fact that they have more active constituents in the higher dose or ethanol extract showed some degree of antidepressant activity. Further trials using solvents of various polarities will explore the effects of solvent composition on extract efficacy [18-24].
Preliminary phytochemical analysis of Ficusthonningii revealed the presence of phenolic compound, Proteins, tannins, glycosides, carbohydrate, starch, vitamins and minerals etc. It is not surprising that there are differences in the antideprassant effects of plant species, due to the phytochemical properties and differences among species.
It is possible that the active chemical constituents were not soluble in ethanol or water. The drying process may have caused conformational changes to occur in some of the chemical constituents found in these plants. Active compounds may be present in insufficient quantities in the crude extracts to show activity with the dose levels employed. Lack of activity can thus only be proven by using large doses. Alternatively, if the active principle is present in high enough quantities, there could be other constituents exerting antagonistic effects or negating the positive effects of the bioactive agents [10]. With no significant activity, extracts may be active against other biological activity which were not tested [11]. Ficusthonningii (200 and 400 mg/Kg) ethanol extract was evaluated in albino mice in Tail Suspension test (TST) and forced swim test (FST)The effects of fluoxetine (FXT; 10 mg/Kg) were also being assessed.
In TST, group control 0.9% w/v sodium chloride normal saline (10 ml/Kg) or (1 ml/100 gm, p. o.) administration have shown their immobility is insignificant. When ethanolic extract of Ficusthonningii (200 mg/Kg) orally administered, have shown immobility is significant. When ethanolic extract of Ficusthonningii (400 mg/Kg) orally administered, have shown immobility is significant. When standard dose of fluoxetine (10 mg/kg) was orally administered, have shown immobility is significant.
In FST,group control 0.9% w/v sodium chloride normal saline (10 ml/Kg) or (1 ml/100 gm, p. o.) administration have shown their immobility and number of jumps is insignificant.
When ethanolic extract of Ficusthonningii (200 mg/Kg) orally administered, have shown immobility and number of jumps is significant. When Ethanolic extract of Ficusthonningii (400 mg/kg) orally administered, have shown immobility and number of jumps is significant. When standard dose of fluoxetine (10 mg/kg) was orally administered, have shown immobility and number of jumps is significant.
The Ethanolic extract of Ficusthonningii 400 mg/kg showed the most remarkable activity. This plant can be further subjected to isolation of the therapeutic Antidepressant compound and carry out further pharmacological evaluation [24-26].
CONCLUSION
Foregone discussion showed that F. thonningii root extract is very efficient antidepressant. In the present work albino Swiss mice was used to study the effect of F. thonningii forth treatment of depression and for this mice was orally administrated with 200 and 400 mg/Kg of the ethanolic extract of “F. thonningii root. Results clearly showed that both the doses have shown significant results as an antidepressant effect using forced swim test (FST) and Tail Suspension test (TST) parameters. The results of the present study support the folkloric usage of the studied plants and suggest that some of the plant extracts possess compounds with antidepressant properties that can be used as antidepressant agents in new drugs for the therapy of neuro-psycho diseases caused by various factors. The most active extracts can be subjected to isolation of the therapeutic antidepressant and undergo further pharmacological evaluation.
ACKNOWLEDGEMENT
It’s our privilege to express the profound sense of gratitude and cordial thanks to our respected chairman Mr. Anil Chopra and Vice Chairperson Ms. Sangeeta Chopra, St. Soldier Educational Society, Jalandhar for providing the necessary facilities to complete this research work.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
All authors have contributed equally
CONFLICT OF INTERESTS
There is no conflict of interest regarding this manuscript.
REFERENCES
World Health Organization. World suicide prevention day; 2012. Available from: http://www.who.int/mediacentre/events/annual/world.suicidepreventionday/en/accessed. [Last accessed on 07 Mar 2025].
DE Zwart PL, Jeronimus BF, DE Jonge P. Empirical evidence for definitions of episode remission recovery relapse and recurrence in depression: a systematic review. Epidemiol Psychiatr Sci. 2019;28(5):544-62. doi: 10.1017/S2045796018000227, PMID 29769159.
Gilbert P. Psychotherapy and counselling for depression. 3rd ed. Los Angeles: SAGE Publications; 2007. doi: 10.4135/9781446279830.
Kessler RC, MC Gonagle KA, Zhao S. Lifetime and 12 mo prevalence of DSM-IIIR psychiatric disorders in the United States results from the national comorbidity survey. Arch Gen Psychiatry. 1994;51(1):8-19. doi: 10.1001/archpsyc.1994.03950010008002.
Robins L, Regier D. Psychiatric disorders in America. New York: Free Press; 2004.
Depression Guideline Panel. Depression in primary care: detection and diagnosis clinical practice guideline number 5. Rockville MD: United States Department of Health and Human Services; 2001.
Katon W. The epidemiology of depression in medical care. Int J Psychiatry Med. 1987;17(1):93-112. doi: 10.2190/xe8w-glcj-kem6-39fh, PMID 3294691.
Judd LL. The clinical course of unipolar major depressive disorders. Arch Gen Psychiatry. 1997;54(11):989-91. doi: 10.1001/archpsyc.1997.01830230015002, PMID 9366654.
Hasler G. Pathophysiology of depression: do we have any solid evidence of interest to clinicians? World Psychiatry. 2010;9(3):155-61. doi: 10.1002/j.2051-5545.2010.tb00298.x, PMID 20975857.
Zhao Z, Wang W, Guo H, Zhou D. Antidepressant like effect of liquiritin from Glycyrrhiza uralensis in chronic variable stress induced depression model rats. Behav Brain Res. 2008;194(1):108-13. doi: 10.1016/j.bbr.2008.06.030, PMID 18655806.
Machado DG, Bettio LE, Cunha MP, Santos AR, Pizzolatti MG, Brighente IM. Antidepressant like effect of rutin isolated from the ethanolic extract from Schinus molle L. in mice: evidence for the involvement of the serotonergic and noradrenergic systems. Eur J Pharmacol. 2008;587(1-3):163-8. doi: 10.1016/j.ejphar.2008.03.021, PMID 18457827.
YU ZF, Kong LD, Chen Y. Antidepressant activity of aqueous extracts of Curcuma longa in mice. J Ethnopharmacol. 2002;83(1-2):161-5. doi: 10.1016/s0378-8741(02)00211-8, PMID 12413724.
Yao CY, Wang J, Dong D, Qian FG, Xie J, Pan SL. Laetispicine an amide alkaloid from Piper laetispicum presents antidepressant and antinociceptive effects in mice. Phytomedicine. 2009;16(9):823-9. doi: 10.1016/j.phymed.2009.02.008, PMID 19447013.
Lee S, Kim DH, Lee CH, Jung JW, Seo YT, Jang YP. Antidepressant like activity of the aqueous extract of Allium Macrostemon in mice. J Ethnopharmacol. 2010;131(2):386-95. doi: 10.1016/j.jep.2010.07.015, PMID 20637276.
Cicero Bezerra Felipe F, Trajano Sousa Filho J, DE Oliveira Souza LE, Alexandre Silveira J, Esdras DE Andrade Uchoa D, Rocha Silveira E. Piplartine an amide alkaloid from Piper tuberculatum presents anxiolytic and antidepressant effects in mice. Phytomedicine. 2007;14(9):605-12. doi: 10.1016/j.phymed.2006.12.015, PMID 17399971.
Schmidt E, Lutter M, MC Cleland W. Trees and shrubs of Mpumalanga and Kruger National Park. South Africa: Jacana; 2002. p. 80.
Usman H, Abdulrahman FI, Usman A. Qualitative phytochemical screening and in vitro antimicrobial effects of methanol stem bark extract of Ficus thonningii (Moraceae). Afr J Tradit Complement Altern Med. 2009;6(3):289-95. doi: 10.4314/ajtcam.v6i3.57178, PMID 20448855.
Dangarembizi R. Effects of crude leaf extracts of ficusthonningiion growth gastrointestinal morphometry and clinical biochemistry of suckling spraquedawley rats: a thesis submitted to the faculty of health sciences. Johannesburg: University of the Witwatersrand; 2012.
The Organization of Economic Co-Operation and Development (OECD). The OECD guideline for testing of chemicals: 423 acute oral toxicity. Paris: OECD; 2001. p. 1-14.
Sofowora A. Screening plants for bioactive agents. In: medicinal plants and traditional medicinal in Africa. Spectrum. Books Ltd sunshine house Ibadan Nigeria: 2nd ed; 1993;8:134-56.
Trease GE, Evans WC. Pharmacognosy. 15th ed. Philadelphia: Saunders Publishers; 2002. p. 42-4, 221-9, 246-9, 304-6, 331-2, 391-3.
Vishnoi NR. Advanced practical chemistry. Ghaziabad India: Yikas Publication House, PVT. Ltd; 1979. p. 447-9.
Roy M, David N, Cueva M, Giorgetti M. A study of the involvement of melanin-concentrating hormone receptor 1 (MCHR1) in murine models of depression. Biol Psychiatry. 2007;61(2):174-80. doi: 10.1016/j.biopsych.2006.03.076, PMID 16934771.
Stdru L, Thierry B, Chermat R, Simon P. Animal modeling of depression: an ethological approach and its relevance to psychopharmacology. Presented at the 13th CINP Congress Jerusalem Israel Jun 20-25 (abstract book p 694); 1982.
Lin J, Opoku AR, Geheeb Keller M, Hutchings AD, Terblanche SE, Jager AK. Preliminary screening of some traditional zulu medicinal plants for anti-inflammatory and anti-microbial activities. J Ethnopharmacol. 1999;68(1-3):267-74. doi: 10.1016/s0378-8741(99)00130-0, PMID 10624887.
Yao J, Moellering R. Antibacterial agents. In: Murray P, Baron E, Pfaller M, Tenover F, Yolken R, editors. Manual of clinical microbiology. Washington, DC: ASM; 1995. p. 1281-90.