
Int J Curr Pharm Res, Vol 17, Issue 2, 66-69Original Article
STUDY OF DIAGNOSTIC ACCURACY OF XPERT MTB/RIF IN PULMONARY TUBERCULOSIS IN A TERTIARY CARE TEACHING HOSPITAL, SVIMS, TIRUPATI, ANDHRA PRADESH
K. VINEELA, N. RAMAKRISHNA, C. TIRUMALA, R. JAYAPRADA*, B. VENKATA RAMANA
1Department of Microbiology, Sri Venkateswara Medical College, Tirupathi-517507, Andhra Pradesh, India. 2Department of Microbiology, Sri Venkateswara Institute of Medical Sciences, Tirupathi-517507, Andhra Pradesh, India. 3Department of Pulmonology, Sri Balaji Medical College and Research Institute, Tirupathi-517507, Andhra Pradesh, India. 4,5Department of Microbiology, S. V. Institute of Medical Sciences, Tirupathi-517507, Andhra Pradesh, India
*Corresponding author: R. Jayaprada; *Email: pradarangineni@yahoo.com
Received: 11 Dec 2024, Revised and Accepted: 20 Feb 2025
ABSTRACT
Objective: Tuberculosis is one of the major global health problems, while India contributing to a major chunk of the disease burden. A molecular method, the Xpert MTB/RIF (Cepheid, USA) can detect the presence of Mycobacterium tuberculosis DNA and the diagnostic accuracy of Xpert MTB/RIF in pulmonary tuberculosis was evaluated.
Methods: This prospective study was conducted from September 2019 to September 2020. Sputum specimens were collected and processed for Ziehl Neelsen staining technique, culture on solid Lowenstein Jensen medium and Xpert MTB/RIF. Differentiation of NTM’s from MTB by was done by performing Rapid test method using TB antigen MPT64 immunoprotein antigen. Drug susceptibility testing by proportion method was performed. The data was compiled and analyzed statistically. The sensitivity, specificity, positive predictive value, negative predictive value and diagnostic accuracy of Xpert MTB/RIF was calculated using sputum culture for Mycobacterium tuberculosis as a gold standard test.
Results: A total of 435 samples satisfying the inclusion criteria were processed and analyzed. Considering Mycobacterial culture as a “gold standard” the sensitivity, specificity and accuracy of Xpert MTB/RIF in the diagnosis of pulmonary tuberculosis was noted as 96.4% (95% CI 87.4%-99.5%), 93.3% (95%CI 93.8%-97.9%) and 96.3% (95% CI 94%-97.8%) respectively. Among 9 rifampicin resistance samples detected by Xpert MTB/RIF, only 7 (77.8%) were rifampicin resistant by DST; additional 2 samples were detected by Xpert MTB/RIF alone.
Conclusion: The current study concluded that Xpert MTB/RIF is a first-line diagnostic tool in pulmonary tuberculosis which can be used in field settings as a point-of-care test and to be compared with clinical aspect of the patient as a part of National Tuberculosis Elimination Programme.
Keywords: Diagnostic accuracy, Mycobacterium tuberculosis, Pulmonary tuberculosis, Rifampicin resistance, Xpert MTB/EIF
© 2025 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2025v17i2.6077 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Tuberculosis (TB) is a slowly progressive communicable disease and a global epidemic. It is one of the major global health problem while India contributing to a major chunk of the disease burden. TB affects all age groups and both males and females are affected, but the highest burden observed in adult men who accounted for 56% of all TB cases [1]. The most important aspect in control of TB remains early diagnosis and treatment, which has been made difficult with the emergence of multi-drug resistant tuberculosis (MDR-TB) and Extensively Drug-Resistant (XDR) strains of Mycobacterium tuberculosis (MTB). The advent of newer diagnostic tests including use of liquid culture media and nucleic acid amplification tests such as Xpert MTB/RIF and Line Probe Assay (LPA) has assisted in diagnosing the disease. By rapidly diagnosing TB, we can initiate treatment more quickly which ultimately helps in reducing the disease transmission rate and prevents complications [2]. A molecular method, Xpert MTB/RIF assay (Cepheid, USA), was endorsed in 2010 by World Health Organization (WHO) for rapid identification of MTB and antibiotic resistance in the clinical specimens [3]. Ministry of Health and Family Welfare has engaged with international partners to start Xpert MTB/RIF for the diagnosis of pulmonary TB as a part of National Tuberculosis Elimination Programme (NTEP) [4]. Xpert MTB/RIF (Cepheid, USA) can detect the presence of Mycobacterium tuberculosis (MTB) DNA and mutations associated with rifampicin (RIF) resistance directly from sputum samples in less than 2 h, and it minimizes staff manipulation and biosafety risk [5]. There are meager studies on the accuracy of Xpert MTB/RIF, especially from India, which prompted the present study at our tertiary care teaching hospital.
MATERIALS AND METHODS
This prospective study was carried out in the Mycobacteriology laboratory in the department of Microbiology (NTEP accredited), SVIMS, Tirupati, from September 2019 to September 2020.
The following laboratory investigations were done on each sputum sample
Smear microscopy by Ziehl –Neelsen smear (ZN smear) technique to detect Acid Fast Bacilli (AFB)
Conventional solid culture using Lowenstein-Jensen medium (L-J medium) to detect Mycobacterium species in duplicate.
Xpert MTB/RIF to detect Mycobacterium tuberculosis (MTB) and Rifampicin Resistance (RR)
Differentiation of NTMs from MTB by Rapid test method using TB antigen MPT64 immunoprotein antigen.
Drug susceptibility testing by proportion method for all positive culture samples after excluding Nontuberculous Mycobacteria (NTM) in duplicate.
A total of 435 sputum samples from patients suspected of having pulmonary tuberculosis was collected and processed by all the above-mentioned methods simultaneously.
All the sputum samples were processed as per the diagnostic algorithm described in the fig. 1.
Statistical analysis
Data was recorded on a predesigned proforma and descriptive statistics were represented as mean±standard deviation, median [interquartile range (IQR)]. Categorical variables were reported as percentages. Accuracy, sensitivity, specificity and negative and positive predictive values of Xpert MTB/RIF were calculated considering mycobacterial culture as gold standard. A two by two contingency table was constructed and the true positives (TP), true negatives (TN), false positives (FP) and false negatives (FN) were listed. Then, the sensitivity, specificity, positive and negative predictive values and accuracy were calculated [6].
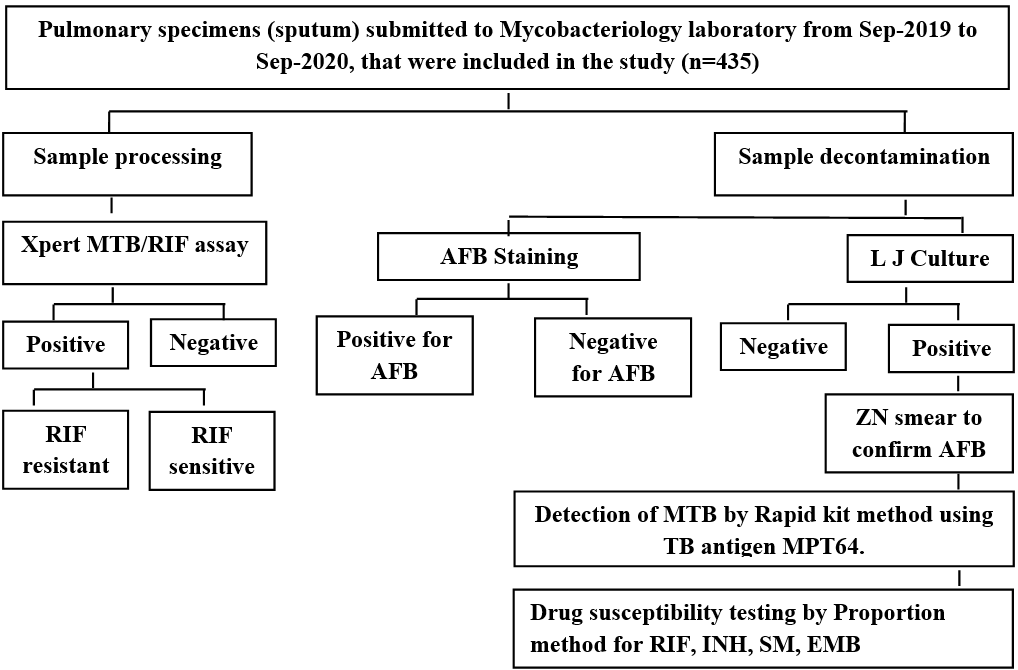
Fig. 1: Algorithm showing study design, **(TB: Tuberculosis, MTB: Mycobacterium tuberculosis, AFB: Acid Fast Bacilli, ZN smear: Ziehl –Neelsen smear, RIF: Rifampicin, INH: Isoniazid, SM: Streptomycin, EMB: Ethambutol, LJ MEDIUM: Lowenstein-Jensen medium)
RESULTS
A total of 435 samples satisfying the inclusion criteria were processed and analyzed. Among them 6 samples were NTM’s, which were detected by MPT64 rapid test method and sample size numbered to 429.
All the samples (n=429) were tested for Mycobacterium tuberculosis by various methods like sputum smear examination by Ziehl-Neelsen (ZN) technique, sputum culture by solid culture method on Lowenstein-Jensen medium and molecular diagnosis to detect Mycobacterium tuberculosis by Xpert MTB/RIF. All the positive sputum cultures were confirmed by ZN smear technique to confirm Acid Fast Bacilli (AFB) and were differentiated from Non Tuberculous Mycobacteria (NTM) by rapid test method that detect Mycobacterial protein tuberculosis 64 (MPT64) immunoprotein which is present only in the culture fluid of Mycobacterium tuberculosis (MTB). The remaining positive samples were evaluated for drug susceptibility method (DST) by Proportion method and the results were compiled and analyzed.
Among 429, sixty-nine samples which came positive by sputum culture and/or smear and/or Xpert MTB/RIF were analyzed. All the positive samples were analyzed to know the sensitivity, specificity, PPV, NPV and accuracy of Xpert MTB/RIF in pulmonary tuberculosis considering mycobacterial culture as ‘gold standard’.
Demographic characteristics
The majority of the patients were in their fifth and sixth decades of life, followed by fourth decade. Their mean age of presentation was 50.68±17 y.
In the present study, males (n=286, 66%) outnumbered females (n=149, 39%) with a male to female ratio of 1.9:1.
Clinical presentation among patients suspected of pulmonary tuberculosis (PTB) at the time of sample collection
Majority of patients who are positive for PTB presented with productive cough since 30 days (n=310, 71.2%) followed by fever since 20 days (n=80, 18.3% breathlessness (n-=28, 6.4%), fatigue and exertional dyspnea (n=17, 3.9%).
Diagnostic accuracy of Xpert MTB in pulmonary tuberculosis
Among 75 positive sputum samples, 6 were NTM’s that were detected by Rapid Test Method using MPT64 antigen which is present in the culture fluid of MTB. A total of 69 MTB isolates were detected by various methods.
Among 69 positive samples, 67 were tested positive for MTB by Xpert MTB/RIF and 2 were tested negative by Xpert MTB/RIF. These 2 samples were tested positive for Mycobacterium tuberculosis with LJ culture alone. Fifty-five samples were tested positive for MTB by LJ culture method. In 22 samples, AFB were detected by ZN smear technique and among them 18 were positive by LJ culture also. Remaining 4 were tested negative by LJ culture method and were positive by Xpert MTB/RIF.
In 67 Xpert MTB/RIF positive samples, fourteen were negative by LJ culture. All the positive culture samples (n=55) were subjected to drug susceptibility testing (DST) to Isoniazid (0.2µg/ml), Rifampicin (40µg/ml), Ethambutol (2µg/ml) and Streptomycin (4µg/ml) by proportion method as per RNTCP guidelines [7]. From DST results, Rifampicin resistance was detected in 7 samples, whereas RIF Resistance was identified in 9 samples by Xpert MTB/RIF.
Based on the above results the sensitivity, specificity, PPV, NPV and accuracy of Xpert MTB/RIF was calculated using sputum for Mycobacterial culture as gold standard.
Fig. 2: Recovery rate of mycobacteria by culture and Xpert MTB/RIF (n=435)
| Species identified | Total no. of samples positive by LJ culture | Total no samples positive by Xpert MTB/RIF | No. of samples positive by LJ culture and Xpert MTB/RIF |
| MTB | 55 | 67 | 53 |
| NTM | 6 | - | - |
| Total | 61 | 67 | 53 |
Diagnostic accuracy of Xpert MTB/RIF in pulmonary tuberculosis with sputum culture as the gold standard
Out of 435 sputum samples processed in this study, 75 samples were tested positive by different methods. Out of 75 samples, 6 were NTM’s and, which were differentiated by rapid test method using MPT64 antigen. Remaining 69 samples were identified as MTB positive. As sputum for mycobacterial culture is the gold standard, sensitivity, specificity, PPV, NPV, accuracy of Xpert MTB/RIF were calculated as 96.4%, 96.3%, 79.1%, 99.45% and 96.3%, respectively using data depicted in fig. 3.
Identification of rifampicin resistance by xpert MTB/RIF and phenotypic DST by proportion method
A total of 53 positive sputum samples tested by proportion method and Xpert MTB/RIF to detect Rifampicin resistance. Among them, 7 were tested resistance to rifampicin by DST using proportion method and 9 were identified with Rifampicin resistance by Xpert MTB/RIF. As phenotypic DST is the gold standard in identifying Rifampicin resistance, the sensitivity, specificity, PPV, NPV and accuracy of Xpert MTB/RIF is calculated as 100%, 95%, 77.7%, 100% and 96.23% respectively.
Fig. 3: Diagnostic accuracy of Xpert MTB/RIF in PTB with sputum culture as gold standard
| Xpert MTB/RIF | Sputum culture | ||
| Positive | Negative | Total | |
| Positive | 53 | 14 | 67 |
| Negative | 2 | 360 | 362 |
| Total | 55 | 374 | 429 |
Fig. 4: Rifampicin susceptibility testing by Xpert MTB/RIF and phenotypic DST by Proportion method
| Rifampicin | Rifampicin susceptibility testing | ||
| Rifampicin resistance by DST | Rifampicin sensitive by DST | Total | |
| Rifampicin resistance by Xpert MTB/RIF | 7 | 2 | 9 |
| Rifampicin sensitive by Xpert MTB/RIF | 0 | 44 | 44 |
| Total | 7 | 46 | 53 |
DISCUSSION
In the current study, the incidence of culture-confirmed pulmonary tuberculosis was found to be 12.8% (55/429) whereas the conventional diagnostic tool, smear microscopy detected only 5.1% (n=22 out of 429) of cases among the study participants. Xpert MTB/RIF diagnosed more number of patients 15.6% (67/429) than smear microscopy and LJ culture in our study.
Demographic characteristics
Forty-five percent of study populations were under 50 y of age and among them, 48% have been diagnosed with pulmonary tuberculosis. While the remaining 55% of study population were above 50 y of age. In studies conducted in Mumbai by Vadwai V et al. [8] the mean age of presentation was 37 y. In another studies conducted in Delhi by Sharma S K et al. [9] the mean age of presentation was 37±18 y. This reinforces the fact that tuberculosis affects persons in their economically most productive years of life. Hence, is therefore not only a medical disease but also poses a major economic problem.
In the present study, male to female ratio was: 1.9:1. Among the positivity rates males exceeds females by 2 times (males: n=55; 79.7%, females: n= 14, 20.2%) male to female ratio was 2.04:1. A similar demographic trend was noted in a study conducted in by Sharma S K et al. [9] where the male to female ratio being 1.8:1. The increased incidence of PTB in males when compared with females can be attributed to the prevalence of risk factors for TB in males such as smoking and alcoholism and other substance abuse than females. There is a higher odds ratio of acute and chronic diseases like COPD (chronic obstructive pulmonary disease), silicosis in males than females.
In our study, majority of the patients presented with chief complaint of productive cough since 30 days (71.2%) followed by low-grade fever since 20 days (18.3%). Similar results were noted in a study conducted by T K Barman et al. [10] from Bangladesh where 73.3% of patients presented with chronic cough for more than one month followed by low-grade fever.
Sensitivity and specificity of Xpert MTB/RIF in pulmonary tuberculosis
In our study, a total of 429 samples were processed by Xpert MTB/RIF, LJ culture method and ZN smear method. Among 429 samples, 69 were tested positive for MTB by different methods. Among 69 positives, Xpert MTB/RIF detected 67 samples and two were negative by Xpert MTB/RIF which are tested positive by LJ culture alone. Fifty-five samples were positive by LJ culture method. Among 55 culture positives, 53 were shown positive by Xpert MTB/RIF. Twenty-two samples were positive by ZN smear method and among them only 18 were positive by LJ culture and all 22 were positive by Xpert MTB/RIF. Seven samples were shown to have resistance for Rifampicin by phenotypic DST by Proportion method, whereas 9 were positive for Rifampicin resistance gene by Xpert MTB. By considering mycobacterial culture as the ‘gold standard’ for PTB diagnosis, the sensitivity, specificity, PPV, NPV and accuracy of sputum smear was calculated as 33.7%, 98.9%, 81.8%, 90.9% and 90.4% respectively. Similarly considering LJ culture as gold standard the sensitivity, specificity, PPV, NPV and accuracy of Xpert MTB/RIF in PTB was calculated as 96.4%, 96.3%, 79.1%, 99.45% and 96.3% respectively. The above results showed that Xpert MTB/RIF has higher sensitivity than AFB smear microscopy in sputum samples.
A similar study conducted in New Delhi by Agrawal et al. [11] over 170 pulmonary specimens including Broncho alveolar lavage samples, the overall sensitivity, specificity, PPV, NPV of Xpert MTB/RIF in sputum samples were 86.6%, 93.1%, 78.5% and 96% respectively considering Mycobacterial growth indicator tube BACTEC 320 (MGIT BACTEC 320) liquid culture systems as gold standard. The sensitivity specificity of AFB microscopy in sputum samples were 22.2% and 78.5%, respectively regarding liquid culture as gold standard.
Results obtained from a study conducted in New Delhi by Sharma S K et al. [9] showed sensitivity and specificity of 95.7% and 99.3%. In smear-negative culture-positive samples it had a sensitivity of 77.7%(56/77) and for detecting smear-positive samples was 99.2%(374/377) whereas in our study, the sensitivity of Xpert MTB/RIF in smear negative-culture positive samples was identified as 94.6% and smear positive-culture positive samples was noted as 88.89% In the same study for the sputum samples, the Xpert MTB/RIF has a sensitivity and specificity in detecting the rifampicin resistance when taking the drug susceptibility testing (DST) on solid culture media by Proportion method as gold standard was 99% and 99.3% respectively. In our study, the sensitivity and specificity of Xpert MTB/RIF in detecting rifampicin resistance when taken DST by Proportion method on solid media as gold standard was calculated as 100% and 95%.
A study conducted in South Korea by Lee H S et al. [12] the cumulative sensitivity and specificity were 74.1% and 97.5% for Xpert MTB/RIF, whereas only 38.8% and 97.7% for smear microscopy, respectively. Out of 82 positive NTM’s, 81(98.8%) were not detected by Xpert MTB/RIF in their study, where as in our study all 6 NTM’s (100%) were not detected by Xpert MTB/RIF.
A much similar study conducted in Tanzania by Rachow et al. [13] where the Xpert MTB/RIF had an overall sensitivity of 88.4% among patients with positive culture and 99% specificity. Seven additional cases (9.1% of 77) were detected by Xpert MTB/RIF assay among the patient with suspected TB based on clinical findings and were culture negative. In our study, 14 additional cases (20.2%, 14/69) were detected positive by Xpert MTB/RIF and were culture negative having clinical symptoms related to PTB. Further in 4 samples which were tested negative by LJ culture, but were positive by ZN smear and Xpert MTB/RIF. This can be cleared by the fact that the patient may be already on treatment and hence, the dead bacilli may be present in the sputum sample due to which no growth was observed in culture but are stained with ZN smear and nonviable MTB was detected by Xpert MTB/RIF.
CONCLUSION
By the above-stated facts, this study concluded Xpert MTB/RIF is first-line diagnostic tool in PTB which can be used in field setting to perform at the point of care and to be compared with clinical aspect of the patient as a part of NTEP. Xpert MTB/RIF also requires minimal training and less technical expertise to perform the test. Further, Xpert MTB/RIF is less laborious and requires shorter turnaround time. Simultaneous detection of MTB and RIF resistance by Xpert MTB/RIF would considered as a salient diagnostic tool to roll-out in national TB elimination programme.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
All authors have contributed equally
CONFLICT OF INTERESTS
There are no conflicts of interest
REFERENCES
Global tuberculosis report 2020. Geneva: World Health Organization; 2020. Available from: www.who.int/tb/publications/global_report/en/. [Last accessed on 27 Oct 2020].
Shenoi SV, Escombe AR, Friedland G. Transmission of drug-susceptible and drug-resistant tuberculosis and the critical importance of airborne infection control in the era of HIV infection and highly active antiretroviral therapy rollouts. Clin Infect Dis. 2010;50 Suppl 3:S231-7. doi: 10.1086/651496, PMID 20397953.
Lawn SD, Nicol MP. Xpert® MTB/RIF assay: development evaluation and implementation of a new rapid molecular diagnostic for tuberculosis and rifampicin resistance. Future Microbiol. 2011;6(9):1067-82. doi: 10.2217/fmb.11.84, PMID 21958145.
Index TB Guidelines Guidelines on Extra Pulmonary Tuberculosis for India. Initiative of Central TB Division. Ministry of Health and Family welfare Government of India; 2016. Available from: https://tbcindia.gov.in/showfile.php?lid=3245. [Last accessed on 14 Dec 2018].
Piatek AS, Van Cleeff M, Alexander H, Coggin WL, Rehr M, Van Kampen S. GeneXpert for TB diagnosis: planned and purposeful implementation. Glob Health Sci Pract. 2013;1(1):18-23. doi: 10.9745/GHSP-D-12-00004, PMID 25276513.
Baratloo A, Hosseini M, Negida A, El Ashal G. Part 1: simple definition and calculation of accuracy sensitivity and specificity-part 1: simple definition and calculation of accuracy sensitivity and specificity. Emerg (Tehran). 2015;3(2):48-9. PMID 26495380.
Tb Cindia. Revised national TB control programme for mycobacterium tuberculosis culture and drug susceptibility testing; 2009.
Vadwai V, Boehme C, Nabeta P, Shetty A, Alland D, Rodrigues C. Xpert MTB/RIF: a new pillar in diagnosis of extrapulmonary tuberculosis? J Clin Microbiol. 2011;49(7):2540-5. doi: 10.1128/JCM.02319-10, PMID 21593262.
Sharma SK, Kohli M, Yadav RN, Chaubey J, Bhasin D, Sreenivas V. Evaluating the diagnostic accuracy of Xpert MTB/RIF assay in pulmonary tuberculosis. Plos One. 2015;10(10):e0141011. doi: 10.1371/journal.pone.0141011, PMID 26496123.
Barman TK, Roy S, Hossain MA, Bhuiyan GR, Abedin S. Clinical presentation of adult pulmonary tuberculosis (PTB): a study of 103 cases from a tertiary care hospital. Mymensingh Med J. 2017;26(2):235-40. PMID 28588156.
Agrawal M, Bajaj A, Bhatia V, Dutt S. Comparative study of genexpert with zn stain and culture in samples of suspected pulmonary tuberculosis. J Clin Diagn Res. 2016;10(5):DC09-12. doi: 10.7860/JCDR/2016/18837.7755, PMID 27437212.
Lee HS, Kee SJ, Shin JH, Kwon YS, Chun S, Lee JH. Xpert MTB/RIF assay as a substitute for smear microscopy in an intermediate burden setting. Am J Respir Crit Care Med. 2019;199(6):784-94. doi: 10.1164/rccm.201804-0654OC, PMID 30252496.
Rachow A, Zumla A, Heinrich N, Rojas Ponce G, Mtafya B, Reither K. Rapid and accurate detection of mycobacterium tuberculosis in sputum samples by cepheidXpert MTB/RIF assay a clinical validation study. Plos One. 2011;6(6):e20458. doi: 10.1371/journal.pone.0020458, PMID 21738575.