
Int J Curr Pharm Res, Vol 17, Issue 2, 79-85Original Article
ANTIBIOTIC RESISTANCE PATTERN IN ISOLATED PATHOGENS OF URINARY TRACT INFECTIONS IN PEDIATRIC PATIENTS AT A TERTIARY CARE HOSPITAL
NILANJAN BISWAS1, DOLLY ROY2, MUKUNDAM BORAH3, DEBADATTA DHAR CHANDA4, PRANAB DAS5*, KAUSHIK DATTA6
1,2,5Department of Pharmacology, Silchar Medical College and Hospital, Ghungoor, Silchar-788014, Assam, India, 3Department of Pharmacology, Gauhati Medical College and Hospital, Gauhati, Assam, India, 4,6Department of Microbiology, Silchar Medical College and Hospital, Ghungoor, Silchar-788014, Assam, India
*Corresponding author: Pranab Das; *Email: pranabdas2580123@gmail.com
Received: 15 Dec 2024, Revised and Accepted: 24 Feb 2025
ABSTRACT
Objective: To identify the types of uro-pathogens present in the collected urine samples and to determine the antibiotic resistance pattern in urine cultures of pediatric patients attending or admitted to the hospital.
Methods: This was a Prospective, Observational and Cross sectional which were conducted for a period of six (6) months in the Department of Pharmacology in collaboration with the Department of Microbiology, Silchar Medical College and Hospital, Silchar, Assam. Antibiotic sensitivity testing was done by Kirby Bauer Disc Diffusion Method in Mueller Hinton Agar. Zone diameters of various antibiotics was measured with the help of Vernier Calipers and interpreted as per Clinical and Laboratory Standards Institute 2024 guidelines. Pediatric age group patients who were diagnosed with UTI and those who were admitted or attending the hospital during the study period were included.
Results: Among 386 samples, 154 have showed microbial growth. The most common organism isolated were Escherichia Coli (36%), followed by Methicillin resistant Staphylococcus aureus (13%), Klebsiella oxytoca (13%) and Methicillin sensitive Staphylococcus aureus (12%). Incidence of UTI was more in females (55.8%). Resistance among E. coli was higher for Cefixime (75%), Ceftriaxone (55%) and Ciprofloxacin (51%).
Conclusion: Frequent and prolonged use of antimicrobial agents can promote emergence of resistance so, antibiotic stewardship is recommended as a means of reducing antimicrobial confrontation, along with lowering risk of adverse drug events, treatment complications and institutional costs.
Keywords: Urinary tract infections, Uropathogens, Antibiotic resistance, Antibiotic sensitivity, Antibiotic stewardship, Pediatric UTI
© 2025 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2025v17i2.6083 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
One of the most prevalent bacterial illnesses in children and a leading reason for hospitalisation are urinary tract infections (UTIs) [1]. Numerous factors, including alterations in the intestinal microbes, a still developing immune response, and urinary tract abnormalities—of which vesicoureteral reflux (VUR) is particularly common—have been linked to young children's susceptibility to UTIs [2]. Prior to the development of antibiotics, up to 20% of deaths were caused by UTIs [3]. Antimicrobial medicine may be empirically offered in most situations, as early treatment of UTIs reduces the morbidity rate [4]. 90% of primary UTI cases in girls and 80% in boys are caused by Escherichia coli, making it the most prevalent pathogen in children [5]. Due to structural differences and the normal occupancy of the preputial region by non-E. coli Gram-negative microbes in uncircumcised males, the bacterial spectrum of UTIs in boys and girls differs. E. coli's capacity to adhere to the urothelium of the urinary system is a significant susceptibility factor that accounts for its predominance. Non- E. coli species including Enterobacteriaceae, Enterococci, Klebsiella, and Proteus are more commonly linked to recurrent UTIs. Additionally, infections brought on by invasive operations or indwelling catheters are more likely to contain atypical species. Because repeated antibiotic medication selects and encourages atypical bacteria, non-E. coli species are additionally susceptible to cause UTIs after receiving antibacterial therapy for other illnesses [6]. Additionally, the Infectious Diseases Society of America has recognised a class of bacteria known as "the ESKAPE pathogens," which include Pseudomonas aeruginosa, Staphylococcus aureus, Acinetobacter, Enterococcus fecium, Klebsiella pneumoniae and Enterobacter spp., as having the ability to "escape" the bactericidal effects of antibiotics [7, 8]. When treating complex UTIs empirically with antibiotics, this group of bacteria is problematic [7-9]. UTI treatment begins as soon as a urine sample is collected for culture; however, before the culture results are available, a specific therapy is modified based on the uropathogens' sensitivity [9, 10]. The goals of treatment are to eliminate the infection, prevent bacteremia, alleviate symptoms, improve clinical status, minimise sequelae, and prevent renal scarring [10].
Regional surveillance is urgently required to inform empirical treatment regimens and antimicrobial stewardship in light of the increasing resistance patterns among paediatric UTI bacteria. This study aims to characterise antibiotic resistance trends in uropathogens isolated from paediatric UTI cases in a tertiary care hospital. The findings will allow empirical therapy practices to be updated based on data, lowering treatment failure, recurrent infections, as well as the proliferation of resistant strains. Furthermore, there is a lack of study on antibiotic resistance trends in paediatric UTI patients in northeastern India, thus this information is required. The study's aims were to determine the types of uro-pathogens discovered in the collected urine samples and to investigate the pattern of antibiotic resistance in paediatric patients' urine cultures at a tertiary care hospital.
MATERIALS AND METHODS
Study design: Observational, Prospective and Cross-Sectional Study
Study setting and procedure
It was carried out in the Department of Pharmacology in partnership with the Department of Microbiology at SMCH. Using Mueller Hinton Agar and the Kirby Bauer Disc Diffusion Method, urine samples were examined for antibiotic sensitivity. The zone diameters of various antibiotics were measured using Vernier Callipers and interpreted according to the Clinical and Laboratory Standards Institute 2024 criteria.
Sample size: 386 samples.
Ethical clearance
Upon receiving approval and clearance from the Institutional Ethics Committee (vide no. SMC/18,846), the research was started.
Study duration
Six (6) months with effect from 1stJul, 2024 to 31st Dec, 2024.
Study population
All pediatric age group patients admitted/attending SMCH between 1st Jul 2024 to 31st Dec 2024.
Inclusion criteria
1. Microbiological data of both the genders of pediatric patients that are aged below 18 y.
2. Pediatric patients admitted/attending SMCH during the study period.
Exclusion criteria
1. Microbiological data of patients aged more than18 y.
2. Data of pediatric patients attending the hospital before or after the evaluation/study period.
Data management and analysis
Data was collected on case record forms and entered in Microsoft Office Excel (Version: 2007).
RESULTS
The average age of the 386 patients in our study was 4.56±5.48 y. Age groupings (less than 1, one to five, six to ten, and eleven to eighteen) were created for the study group. Patients under one year old accounted for 35.745% of the total, with the age group of 6–10 y old coming in second (26.89%). Females made up the majority of the study participants (55.8%). E. Coli (36%) was the most primarily recovered bacterium from urine cultures (fig. 1). E. Coli showed a notable degree of resistance to Cefixime (75%), Ceftriaxone (55%), and expensive antibiotics such as Meropenem (5%), and Piperacillin-Tazobactam combination (13%), as illustrated in fig. 2. Ninety percent of Methicillin-resistant Staphylococcus aureus (MRSA) organisms were resistant to Ciprofloxacin (fig. 3). The highest level of resistance (60%) to Cefixime, Ceftriaxone, and Ciprofloxacin was demonstrated by Klebsiella oxytoca (fig. 4). Cefixime resistance was highest in Methicillin-sensitive Staphylococcus aureus (MSSA) at 72% (fig. 5). A high level of Cefixime resistance (45%) was demonstrated by Klebsiella pneumoniae (fig. 6). Azithromycin resistance was highest in Enterococcus (75%), as seen in fig. 7. The highest resistance to Cefixime (67%) was displayed by Proteus mirabilis (fig. 8). The highest Cefixime resistance was exhibited by Pseudomonas (83%) (fig. 9). Proteus vulgaris flaunted a substantial degree of resistance (60%) to Ofloxacin, Doxycycline, Cefoperazone, and Ceftriaxone (fig. 10). Among all antibiotics, Cefixime resistance was highest in Acinetobacter (80%) (fig. 11).
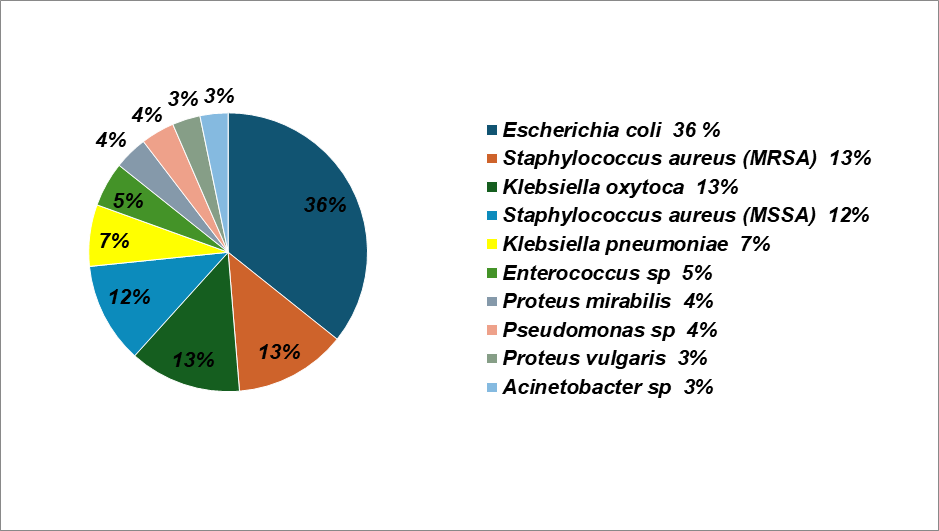
Fig. 1: Isolated organisms from urine culture
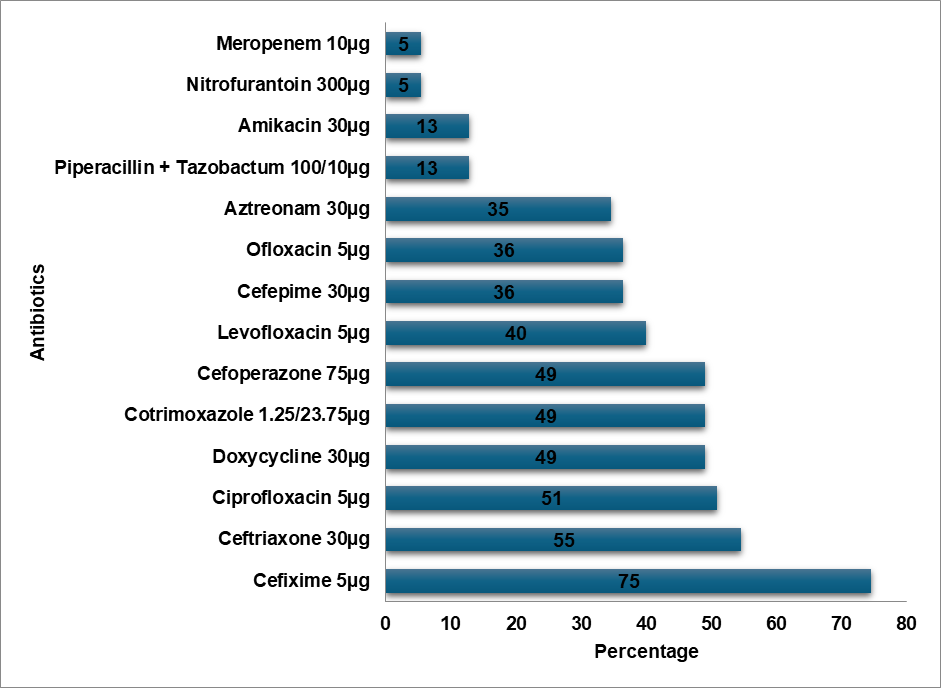
Fig. 2: Escherecia coil resistance pattern
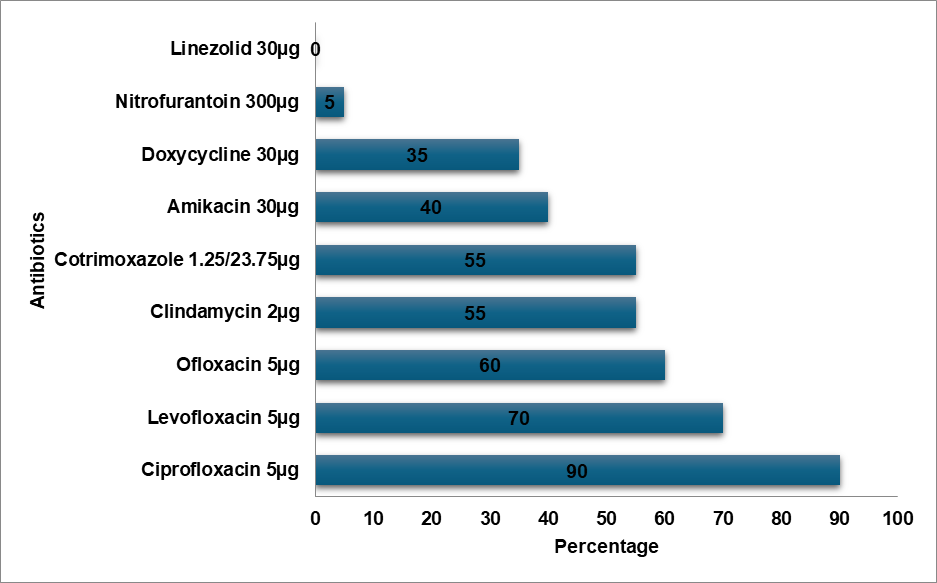
Fig. 3: MRSA resistance pattern
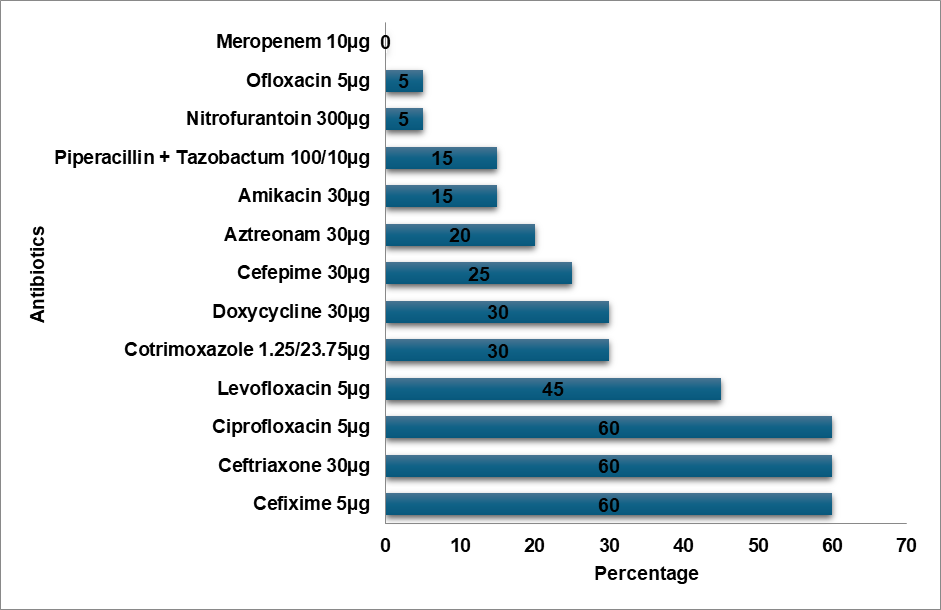
Fig. 4: Klebsiella oxytoca resistance pattern
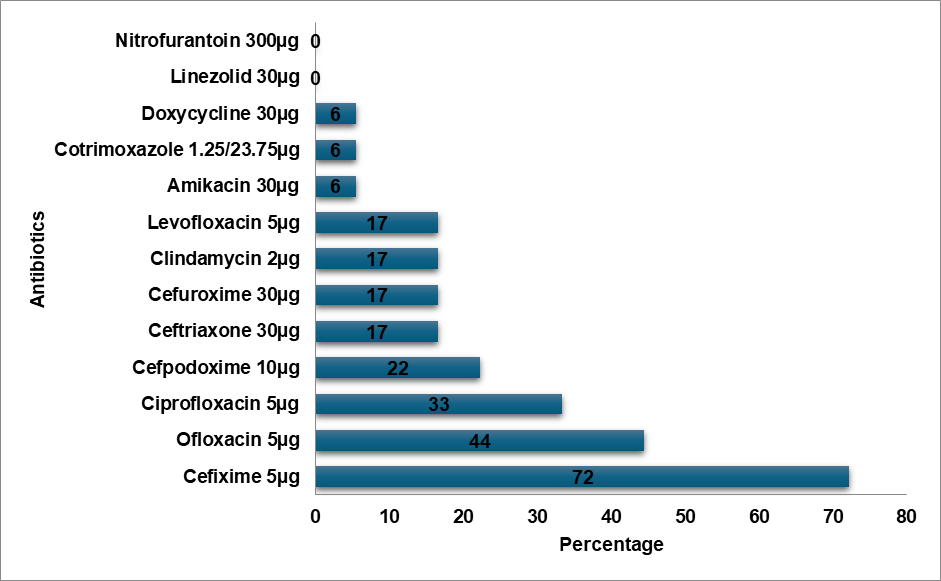
Fig. 5: MSSA resistance pattern
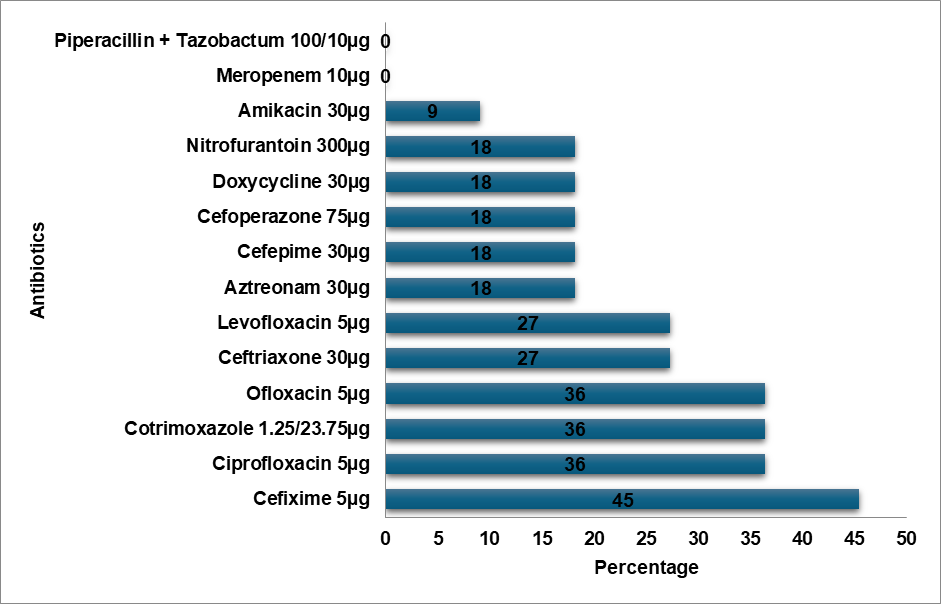
Fig. 6: Klebsiella pneumoniae resistance pattern
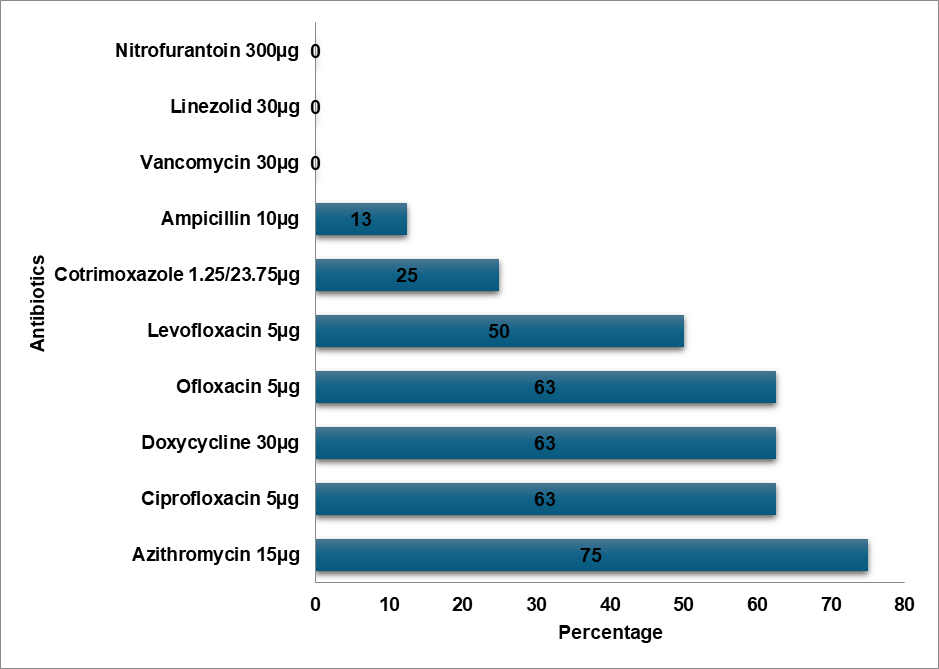
Fig. 7: Enterococcus resistance pattern
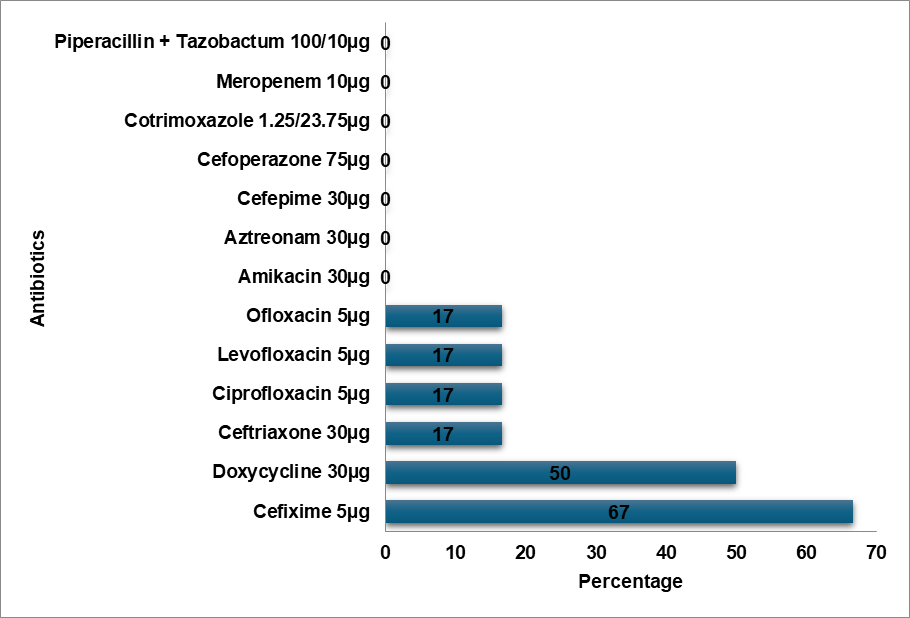
Fig. 8: Proteus mirabilis resistance pattern
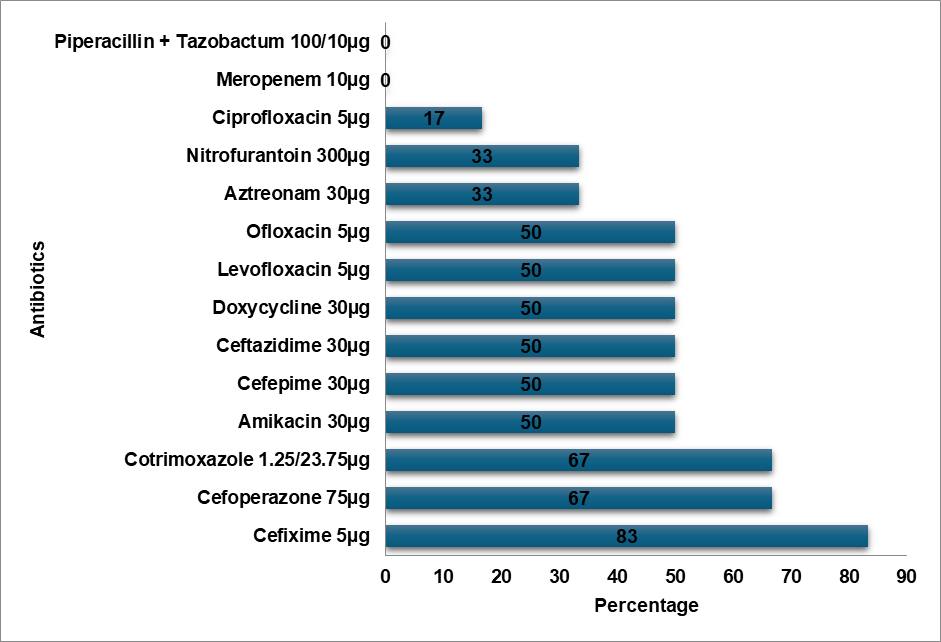
Fig. 9: Pseudomonas resistance pattern
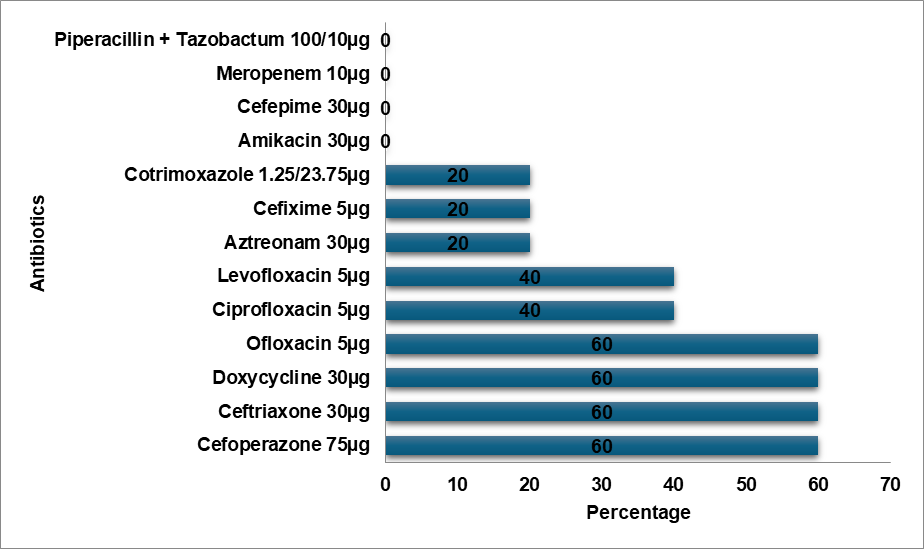
Fig. 10: Proteus vulgaris resistance pattern
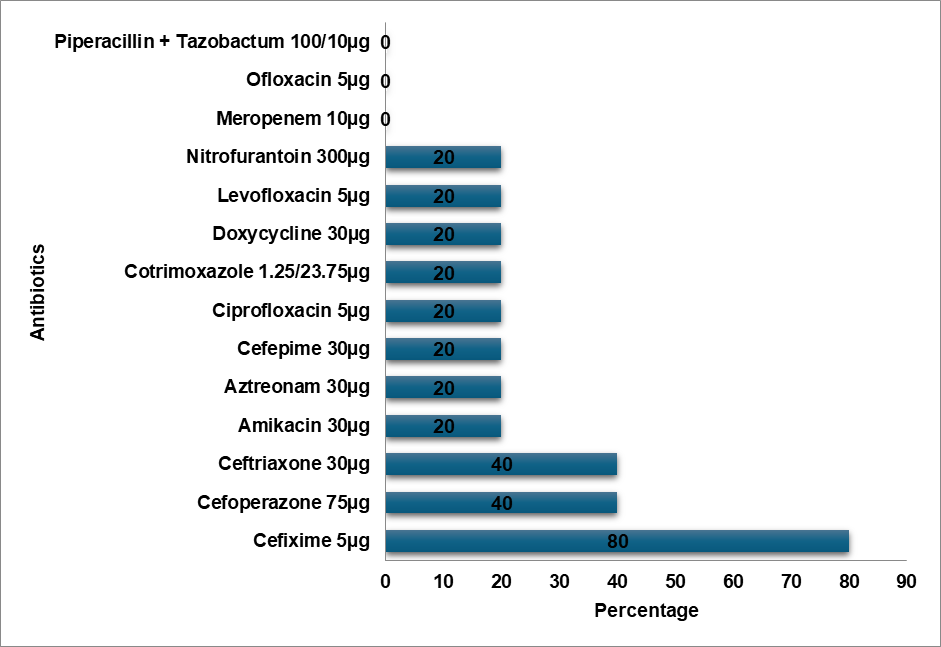
Fig. 11: Acinetobacter resistance pattern
DISCUSSION
As far as we are aware, this is among the first comprehensive studies to evaluate the resistance to antibiotics in paediatric urinary tract infections in Northeast India. The average age among 386 patients in our study was 4.56±5.48 y. Age groupings (less than 1, one to five, six to ten, and eleven to eighteen) were created for the study group. According to the outcomes of an investigation by Duicu et al. [11], 35.745% of the patients were under a year old, with the age group of 6–10 y old coming in second with 26.79%. Females made up the majority of the study participants (55.8%), which is consistent with a study’s conclusion steered by Vazouras K et al. [12]. E. Coli was identified as the most prevailing uro-pathogen in paediatric UTI cases, which is consistent with research conducted by Shaaban OA et al. and Raya GB et al. [13, 14].
E. Coli demonstrated a high level of resistance to Cefixime (75%), Ceftriaxone (55%), and high-end antibiotics such as Meropenem (5%) and Piperacillin-Tazobactam combination (13%). In contrast, E. Coli showed the topmost level of resistance to Cefazolin in a study by Shaaban OA et al. [14]. According to a study by Mitiku A et al., Nitrofurantoin was the most prevalent resistant antibiotic in UTI cases, whereas the Methicillin-resistant Staphylococcus aureus (MRSA) bacteria exhibited a high degree of resistance to Ciprofloxacin (90%) [15]. In contrast to Tarnagda H et al.'s study, which found that Amoxicillin and Amoxicillin/Clavulanic acid had the highest resistance to Klebsiella species, Klebsiella oxytoca exhibited the highest resistance (60%) to Cefixime, Ceftriaxone, and Ciprofloxacin [16]. Methicillin-sensitive Staphylococcus aureus (MSSA) exhibited the highest level of Cefixime resistance (72%), while Ceftizole was the most often resistant antibiotic against MSSA infections in an Issa FA study [17]. Klebsiella pneumoniae exhibited a high level of Cefixime resistance (45%) in our study, while the bacteria exhibited the highest level of Ampicillin resistance in a study by Shaaban OA et al. [14], while Klebsiella pneumoniae was more resistant to Ceftriaxone, Cefuroxime, and Ampicillin in a study by Huang l et al. [18]. In a research by Huang l et al., Enterococcus was more resistant to Rifampicin than to Erythromycin, while Enterococcus exhibited the highest resistance to Azithromycin (75%) [18]. Proteus mirabilis had the highest level of Cefixime resistance (67%) in contrast to the study conducted by Shaaban OA et al. Cefazolin resistance was highest in Proteus mirabilis [14]. Pseudomonas exhibited the highest level of resistance to Cefixime (83%) in contrast to the highest levels of resistance to Cefazolin, Ampicillin, Cefuroxime,Co-amoxiclav, and Ceftriaxone in the study conducted by Shaaban OA et al. [14]. Proteus vulgaris had a high level of resistance (60%) to Ofloxacin, Doxycycline, Cefoperazone, and Ceftriaxone. Cefixime resistance was highest in Acinetobacter (80%). Cefixime was determined to be the most often resistant antibiotic in our investigation.
LIMITATIONS
This study has certain drawbacks. Evaluation of data pertaining to positive urine cultures conducted outside the hospital was not possible. The sample size was modest due to the short study period and one study location. The study's cross-sectional design made it impossible to evaluate potential shifts in medication prescriptions in response to antibiotics. The study's findings could not be applied to adult or senior populations because it only encompassed paediatric age groups.
CONCLUSION
Antibiotic stewardship is advised as a way to reduce antimicrobial resistance, as well as the potential for treatment problems, lengthy hospital stays, adverse medication events, and institutional costs, in addition to frequent and extensive use of antibiotics may promote the development of resistance.
ACKNOWLEDGMENT
The authors expressed their appreciation to the Silchar Medical College and Hospital's Department of Microbiology for their significant contribution.
FUNDING
This study was entirely self-funded.
AUTHORS CONTRIBUTIONS
All authors have contributed equally
CONFLICT OF INTERESTS
There are no conflict of interests.
REFERENCES
Kocak M, Buyukkaragoz B, Celebi Tayfur A, Caltik A, Koksoy AY, Cizmeci Z. Causative pathogens and antibiotic resistance in children hospitalized for urinary tract infection. Pediatr Int. 2016 Jun;58(6):467-71. doi: 10.1111/ped.12842, PMID 26513232.
Swerkersson S, Jodal U, Ahren C, Hansson S. Urinary tract infection in small outpatient children: the influence of age and gender on resistance to oral antimicrobials. Eur J Pediatr. 2014;173(8):1075-81. doi: 10.1007/s00431-014-2289-3, PMID 24623269.
Zorc JJ, Kiddoo DA, Shaw KN. Diagnosis and management of pediatric urinary tract infections. Clin Microbiol Rev. 2005;18(2):417-22. doi: 10.1128/CMR.18.2.417-422.2005, PMID 15831830.
Linhares I, Raposo T, Rodrigues A, Almeida A. Frequency and antimicrobial resistance patterns of bacteria implicated in community urinary tract infections: a ten year surveillance study (2000-2009). BMC Infect Dis. 2013 Jan 18;13:19. doi: 10.1186/1471-2334-13-19, PMID 23327474.
Brandstrom P, Hansson S. Long term low dose prophylaxis against urinary tract infections in young children. Pediatr Nephrol. 2015;30(3):425-32. doi: 10.1007/s00467-014-2854-z, PMID 24906665.
Kutasy B, Coyle D, Fossum M. Urinary tract infection in children: management in the era of antibiotic resistance a pediatric urologists view. Eur Urol Focus. 2017 Apr;3(2-3):207-11. doi: 10.1016/j.euf.2017.09.013, PMID 28965960.
Pendleton JN, Gorman SP, Gilmore BF. Clinical relevance of the ESKAPE pathogens. Expert Rev Anti Infect Ther. 2013;11(3):297-308. doi: 10.1586/eri.13.12, PMID 23458769.
Benko R, Gajacs M, Matuz M, Bodo G, Lazar A, Hajdu E. Prevalence and antibiotic resistance of eskape pathogens isolated in the emergency department of a tertiary care teaching hospital in hungary: a 5 y retrospective survey. Antibiotics (Basel). 2020;9(9):624. doi: 10.3390/antibiotics9090624, PMID 32961770.
T Hoen LA, Bogaert G, Radmayr C, Dogan HS, Nijman RJ, Quaedackers J. Corrigendum to update of the EAU/ESPU guidelines on urinary tract infections in children [J Pediatr Urol 17 (2021) 200-207]. J Pediatr UrolJ Pediatr Urol. 2021;17(4):598. doi: 10.1016/j.jpurol.2021.06.022, PMID 34294583.
Ammenti A, Alberici I, Brugnara M, Chimenz R, Guarino S, LA Manna A. Updated Italian recommendations for the diagnosis treatment and follow up of the first febrile urinary tract infection in young children. Acta Paediatr. 2020 Feb;109(2):236-47. doi: 10.1111/apa.14988, PMID 31454101, PMCID PMC7004047.
Duicu C, Cozea I, Delean D, Aldea AA, Aldea C. Antibiotic resistance patterns of urinary tract pathogens in children from Central Romania. Exp Ther Med. 2021;22(1):748. doi: 10.3892/etm.2021.10180, PMID 34055063.
Vazouras K, Velali K, Tassiou I, Anastasiou Katsiardani A, Athanasopoulou K, Barbouni A. Antibiotic treatment and antimicrobial resistance in children with urinary tract infections. J Glob Antimicrob Resist. 2020;20:4-10. doi: 10.1016/j.jgar.2019.06.016, PMID 31252156.
Raya GB, Dhoubhadel BG, Shrestha D, Raya S, Laghu U, Shah A. Multidrug-resistant and extended spectrum beta-lactamase producing uropathogens in children in Bhaktapur Nepal. Trop Med Health. 2020;48(65):65. doi: 10.1186/s41182-020-00251-6, PMID 32774128.
Shaaban OA, Mahmoud NA, Zeidan AA, Kumar N, Finan AC. Prevalence and resistance patterns of pediatric urinary tract infections in Bahrain. Cureus. 2021 Dec 31;13(12):e20859. doi: 10.7759/cureus.20859, PMID 35141098, PMCID PMC8802020.
Mitiku A, Aklilu A, Biresaw G, Gize A. Prevalence and associated factors of methicillin resistance staphylococcus aureus (MRSA) among urinary tract infection suspected patients attending at Arba Minch General Hospital, Southern Ethiopia. Infect Drug Resist. 2021 Jun 9;14:2133-42. doi: 10.2147/IDR.S306648, PMID 34135603.
Tarnagda H, Ouermi D, Sagna T, Nadembega WM, Ouattara AK, Traore L. Prevalence and antibiotic resistance of urinary tract pathogens with molecular identification of Klebsiella pneumoniae Klebsiella oxytoca and acinetobacter SPP using multiplex real time PCR. Am J Mol Biol. 2024 Aug 27;14(4):245-60. doi: 10.4236/ajmb.2024.144018.
Issa FA. Antibiotic resistance patterns of common uropathogens isolated from females at Zakho city Kurdistan region Iraq. Sci J Univ Zakho. 2024 Nov 18;12(4):490-6. doi: 10.25271/sjuoz.2024.12.4.1395.
Huang L, Huang C, Yan Y, Sun L, LI H. Urinary tract infection etiological profiles and antibiotic resistance patterns varied among different age categories: a retrospective study from a tertiary general hospital during a 12 y period. Front Microbiol. 2021;12:813145. doi: 10.3389/fmicb.2021.813145, PMID 35154037.