
Int J Curr Pharm Res, Vol 18, Issue 2, 1-8Review Article
ADVANCEMENTS AND CHALLENGES IN PSORIASIS MANAGEMENT: EXPLORING INNOVATIVE THERAPIES, DRUG DELIVERY SYSTEMS, AND HOLISTIC APPROACHES
TANVINDER KOUR1*, AJEETPAL SINGH2, AMAR PAL SINGH2
¹Department of Pharmaceutics, St. Soldier Institute of Pharmacy, Lidhran Campus, Behind NIT (R. E. C), Jalandhar–Amritsar Bypass, NH-1, Jalandhar–144011, Punjab, India. ²Department of Pharmacology, St. Soldier Institute of Pharmacy, Lidhran Campus, Behind NIT (R. E. C), Jalandhar–Amritsar Bypass, NH-1, Jalandhar–144011, Punjab, India
*Corresponding author: Tanvinder Kour; *Email: tanvinderkour123@gmail.com
Received: 15 Nov 2025, Revised and Accepted: 02 Jan 2026
ABSTRACT
The fast proliferation of skin cells that results in the production of red, scaly plaques-most frequently on the scalp, elbows, knees, and lower back-is the hallmark of psoriasis, a chronic, non-contagious skin illness. Psoriasis is generally acknowledged to be a complex illness involving immune system dysregulation and genetic predisposition; even if it’s precise origin is still unknown. Its development and exacerbation are also significantly influenced by environmental triggers, trauma, stress, and climatic circumstances. Itching, discomfort, dry skin, joint inflammation (psoriatic arthritis), and irregular nail growth are some of the clinical manifestations of psoriasis. Patients' physical, mental, social, and financial quality of life is greatly impacted by the illness, which frequently results in stigmatization, depression, and diminished capacity for day-to-day functioning. Although there are many therapeutic options available, there is no permanent cure, hence the main goals of illness management are symptom control and quality of life enhancement. Given the mounting evidence that psoriasis is a systemic inflammatory disease, understanding its comorbidities-such as diabetes and cardiovascular disease-is essential to provide patients with complete care. Biological therapies, phototherapy, non-biological systemic medications, and topical medicines are all used in modern treatment plans. Systemic and biologic medicines are essential for treating severe psoriasis, but topical therapies are the main choice for mild to moderate instances. These traditional treatments, however, are constrained by side effects, exorbitant expenses, and inconsistent patient reactions. Promising substitutes to improve therapeutic results are provided by the development of nano-carrier drug delivery systems, such as liposomes, phytosomes, niosomes, ethosomes, and transferosomes. These vesicular systems provide targeted, sustained drug release, improved bioavailability, and reduced systemic toxicity. Continued exploration and clinical validation of these innovative delivery platforms are crucial to overcoming the shortcomings of existing therapies and improving patient quality of life in psoriasis care.
Keywords: Psoriasis management, Innovative therapies, Drug delivery systems, Holistic approaches, Treatment advancements, Clinical challenges
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i2.7057 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCION
Psoriasis is skin disorder in which cells of the skin proliferate ten times more rapidly than normal due to which red bumpy patches with whitish-colored scales emerge on the scalp, lower back, elbows and knees but can appear on other parts as well. Psoriasis is not transferred from one person to another person but sometimes people from the same family can have it [1-3]. The word "psoriasis" comes from the Greek word "psora," meaning "itch," "plaque," or "rash," although most patients who suffer from the condition do not complain of itching. The disease has been known since ancient times and was initially thought to be a type of leprosy [4]. Psoriasis usually affects only a few areas of the body, but in severe cases, the major area is affected. Over the course of a person's life, spots tend to disappear and reappear [5]. Psoriasis affects approximately 2% of the population worldwide [6]. Increasing evidences supports the recognition of psoriasis as a multisystem chronic inflammatory disorder with multiple associated co morbidities, including myocardial infarction, metabolic syndrome, diabetes (Type 2 diabetes) and/or Corhn’s disease [7-10]. Overall, psoriasis treatment is inadequate because there are no proven cures for the condition, however there are numerous treatments that can result in a brief and partial remission [11]. Usually, a skin cell grows for a month before falling off. In contrast, skin cells in psoriasis proliferate over the course of three to four days and accumulate on the skin in the form of plaques instead of dropping off. Although psoriasis can appear at any age, including youth, its two peak onset age groups are 15–25 and 50–60 years old [12]. Two clinical manifestations of psoriasis, type I and type II, were identified by Henseler and Christophers after analyzing a group of 2147 patients. They were differentiated by a bimodal age of onset. Type I starts at or before age 40, while Type II starts after age 40. Over 75% of cases are caused by type I illness [13]. Compared to people with type II psoriasis, which develops later, those with early onset, or type I psoriasis, typically had more afflicted relatives and more severe illness [14]. The outer layer and nails are affected by psoriasis, which is linked to several co-morbidities. Localized or widespread, symmetrical, well-defined, red papules and plaques, and typically covered in white or silver scales are all characteristics of skin lesions. Itching, stinging, and pain are all symptoms of lesions. Psoriatic arthritis, a chronic inflammatory arthritis that causes joint deformities and impairment, affects 1.3% to 34.7% of people with psoriasis. Changes to the nails occur in 4.2% to 69% of all psoriasis patients. According to reports, those who have psoriasis are more likely to develop other severe clinical illnesses like cardiovascular disease and other non-communicable diseases [15].
Causes of psoriasis
Although the reason is unknown, scientists believe it to be caused by a number of circumstances. When the immune system is disrupted, new cells proliferate quickly. New skin cells typically take the place of old ones with contrast, with psoriasis, new cells begin to grow within 3–4 days, rather than 10–30 d. Silvery grey scales grow on the old cells when the new ones take their place [16, 1]. Researchers now know that psoriasis is influenced by a person's genes and immune system. Here are the findings from research on each of them.
Immune system
T-cells, another name for white blood cells, are a component of the immune system. By combating potentially harmful substances like bacteria and viruses, these cells aid in preventing illness. When a person has psoriasis, their immune system malfunctions, causing T-cells to target their skin cells as well. The body produces more skin cells as a result of this onslaught. Psoriasis results from the accumulation of excess skin cells on the skin's surface. T-cells often assault skin cells throughout the remainder of a person's life after they begin. One exception exists. There are some kids who acquire guttate (gut-tate) psoriasis and never get it again.
Genes
Psoriasis is known to run in families. Researchers have discovered that psoriasis is more common in those with specific genes. What scientists have discovered further complicates the situation. It appears that some psoriasis sufferers do not possess the genes that make them more susceptible to the condition. It's also possible to have genes that make you more likely to get psoriasis yet never get it. Because of this finding, experts now think that a trigger must be encountered before psoriasis manifests [17-19]. A number of risk factors that contributed to the genesis of psoriasis are listed below:
Trauma
The condition known as the Koebner phenomenon is psoriasis at the site of damage. It is known that a variety of harmful local stressors, such as physical, chemical, electrical, surgical, infectious, and inflammatory assaults, can cause psoriatic lesions.
Environmental factors
A number of studies have confirmed the significance of the interplay between genes and environment in psoriasis manifestation. Psoriasis has been connected to numerous environmental elements, which have also been linked to the disease's development and the aggravation of pre-existing conditions.
Weather
For those who have psoriasis, winter is typically the most difficult time of year. Numerous studies show that hot, sunny climates seem to cleanse the skin, while chilly weather is a common trigger for many people. Wintertime is dry, and the heat inside deprives the skin of moisture. Usually, this makes psoriasis worse. The combination of winter diseases and holiday stress can exacerbate psoriasis by weakening the immune system. Air conditioning can dry up the skin and exacerbate psoriasis, even if hot and sunny weather may help clear it up [20-25].
Symptoms of psoriasis
Burning feelings, itching, or discomfort, especially when plaques are extensive or situated in sensitive regions of skin [26].
Intense dryness of skin, which can lead to fissures and, in severe instances, bleeding [27].
The main symptom of psoriasis is the emergence of red, irritated areas of skin topped with silvery-white scales, referred to as plaques [27].
Many individuals with psoriasis experience psoriatic arthritis, leading to swelling, joint pain, and stiffness, particularly in the toes and fingers [28].
Nail issues, including color changes, thickening, pitting, and detachment of the nail from the nail bed [28, 29].
Impact of psoriasis on patient’s quality of life
Psoriasis essentially impacts different aspects of patient’s lives, counting physical consolation, mental wellbeing, and social intelligent. Severe itching, soreness, and skin scaling are some of the symptoms that can disrupt sleep, everyday activities, and general physical health [30]. Psoriasis sufferers have had trouble getting the same therapy or care in a variety of settings, including public swimming pools, fitness clubs, and hair salons and barbershops [31, 32]. Patients with psoriasis might experience embarrassment when they have to reveal their skin while swimming, during intimate moments, in public showers, or in situations lacking adequate privacy. A lot of patients with psoriasis frequently feel compelled to conceal their condition, which greatly impacts their self-esteem. Individuals frequently encounter feelings of depression, anxiety, and diminished self-worth [33]. The cost of care, the time required to treat psoriasis, disruptions to work, and a decline in quality of life in both financial and professional affairs are all factors that contribute to the financial burden on the individual patient. As anticipated, people with more severe psoriasis and lower family incomes experience a higher detrimental effect on their financial quality of life [34, 35].
Current treatment approaches
Many traditional methods, including topical, oral, biological and parenteral therapies, can be used to treat psoriasis. Additionally, phototherapy has been used to alleviate symptoms.
Topical therapy
Topical therapy for psoriasis typically results in mediocre to unsatisfactory results and is frequently linked to irritation. The primary treatment for mild to moderate psoriasis is thought to be topical. Systemic administration combined with topical treatment, however, may be helpful in extreme circumstances. The classifications of topical agents are as follows:
Anthralin
Anthralin has proven to be one of the best topical therapies for stable plaque psoriasis over time. It results in mitochondrial malfunction, restored cell differentiation, and decreased keratinocyte proliferation [36].
Side effects
Skin irritation is the most frequent adverse effect of anthralin, and it is dose-related. Additionally, anthralin discolors the patient's skin, hair, nails, clothes, and other items that they come into touch with [37].
Coal tar
A common daily dressing or inpatient procedure involves the application of coal tar. It is widely known to be used in conjunction with UVB, or the Goeckerman regimen. Coal tar most likely inhibits DNA synthesis, which lowers keratinocyte hyper proliferation.
Side effect
Adverse effects of coal tar include odor, staining, irritant contact dermatitis and erythema, stinging [37].
Corticosteroids
Topical corticosteriod is used worldwide to treat psoriasis of all severity. Vasoconstricting, antiproliferative, anti-inflammatory, and immunosuppressive properties are all exhibited by corticosteroids. They control the transcription of many genes, including those that code for proinflammatory cytokines, by binding to the intracellular corticosteroid receptor.
Side effect
Long-term usage of strong topical corticosteroids may cause considerable systemic absorption, which can result in hyperglycemia, skin atrophy along with adrenal axis suppression [38].
Calcineurininhibitors (e. g. Tacrolimus, Pimecrolimus): Calcineurin inhibitors works by preventing the cytoplasmic enzyme calcineurin phosphatase from being active. The translocation of NFAT (nuclear factor of activated T cells) within T cells is inhibited by calcineurinphosphatase.
Side effects
When this medication is first applied, irritation or burning may occur. Rare side effects include headaches, burning eyes, and flu-like symptoms [39].
Vitamin D Analogues: Vitamin D analogs bind to the intracellular Vitamin D receptor which then binds to and regulates the genes involved in epidermal proliferation, inflammation, and keratinization. There are three Vitamin D analog preparations available to treat psoriasis, namely, calcipotriene (calcipotriol in Europe), calcitriol, and tacalcitol. Side effect: The most common adverse effect is skin irritation on or around the psoriasis plaques [37, 40]
Non-biological systemic therapy
When topical therapy is ineffective for moderate to severe psoriasis, systemic medicines are employed. These drugs reduce inflammation throughout the body.
Methotrexate
It inhibits the enzyme dihydrofolate reductase competitively by acting as an antagonist of folic acid, which is necessary for the synthesis of purines and pyrimidines in DNA production. Methotrexate stops dihydrofolic acid from being converted to tetrahydrofolic acid because methotrexate has a higher affinity for dihydrofolatereductase than dihydrofolic acid does. Side effects: It causes liver toxicity, bone marrow depression, renal damage, necrosis of soft tissue and bone with long term use [41, 42].
Cyclosporine
For many years, moderate to severe psoriasis has been treated with this possible calcineurin inhibitor. This inhibition results in a reduction in IL2 production. Additionally, it prevents gamma interferon from being produced. It has a very rapid onset of action [43].
Side effect
When treating a patient on cyclosporine, the main concerns are hypertension, renal failure, and various drug interactions [44].
Acitretin
It has been utilized as a therapeutic since the 1970s and is an acid metabolite of etretinate. It is a retinoid that works by preventing keratinocytes from proliferating. It derived from vitamin A and doesn't suppress the immune system [45].
Side effect
It has a negative impact on the integumentary system by making the skin and mucosal membranes more dry, which can worsen pre-existing xerosis, which is frequent in the elderly [44]
Apremilast
Its works by inhibiting phosphodiesterase-4 (PDE-4), which lowers the thickness of the epidermal layer and decreases the generation of pro-inflammatory cytokines.
Side effect
Headache, nausea, and vomiting are frequent side symptoms that may be associated with apermilast [46].
Fumaric acid esters
In 1959, they were first used to treat psoriasis. Monoethylfumarate and dimethyl fumarate combine to form this substance. These have antiproliferative, antioxidative, and anti-inflammatory properties. It has demonstrated a strong safety and effectiveness profile.
Side effect
A number of gastrointestinal adverse effects are recognized to be connected to Ester of fumaric acid [47].
Mycophenolate mofetil
This prodrug is converted into mycophenolic acid, an inhibitor of purine synthesis that aids in the development of B and T lymphocytes. According to studies, mycophenolate mofetil is more effective than methotrexate and cyclosporine for psoriasis treatment.
Side effect
Treatment is frequently linked to reversible hematologic abnormalities, diarrhea, and nausea [48].
Biological therapy
Biologics for psoriasis treatment are classified into various categories, which is given as follows:
Anti-TNF-α therapy: It involve four drugs as described below:
Etanercept
It is used for the treatment of plaque psoriasis in adults and chronic plaque psoriasis in children. It is contraindicated in heart failure.
Adalimumab
This antibody is monoclonal. It works by attaching itself to host ligands and blocking the interaction of the TNF receptor, which is involved in nail psoriasis, psoriatic arthritis, and plaque psoriasis.
Infliximab
Another monoclonal antibody used to treat psoriatic arthritis and persistent plaque psoriasis is infliximab. It is linked to an increased risk of infection and cancer.
Certolizumabpegol
This PEGylated TNF-α antibody is used to treat psoriatic arthritis and moderate to severe plaque psoriasis [36].
Limitations of current treatments
The following problems make managing psoriasis difficult even with the variety of therapies available:
Lack of efficient carrier for delivery of anti-psoriatic drug
Efficient delivery of antipsoriatic drug is stalled by the inherent side effects of drug moiety or their improper incorporation in the conventional vehicles. Major issues are variation in the physicochemical characteristics of the carrier and of the active moiety used leading to variation in drug absorption and the drug efficacy.
Adverse effect
Psoriasis treatments have many adverse effects like skin thinning, hepatotoxicity, gastrointestinal issues and severe immune suppression and so on
High cost
Many therapies are effective but costly, making them inaccessible for many patients.
Variability in treatment response: treatment response varies among individual to individual [50].
Nano-carrier drug delivery system
Drug targeting and intended administration with sustained or controlled release are crucial elements for enhancing therapeutic efficacy in the current situation. Because vesicular nanocarrier systems like phytosomes, liposomes, niosomes, and transferosomes offer significant potential to overcome the drawbacks of traditional medications, researchers have concentrated on creating them [51].
Liposomes
Both hydrophilic and lipophilic compounds can be encapsulated in liposomes, making them flexible drug delivery vehicles. They range in size from 50 nm to more than 1 μm and self-assemble into a spherical shape using phospholipids and cholesterol. New-generation liposomes have been created to offer enhanced sustained/controlled release, stability, drug loading, target selectivity, longer action, and greater drug entrapment efficiency. However, liposomes still have drawbacks, such as leakage and short half-life [52].
Phytosomes
This cutting-edge herbal medication delivery method enhances the active plant ingredients' stability, bioavailability, and target specificity. In addition to phospholipid, phytosomes are vesicular drug delivery vehicles that may transport lipid-and water-soluble substances. There are certain stoichiometric proportions of 2:1 or 1:2 between the phospholipid and phytoconstituent [53]. Therapeutic compounds, a solvent, and a carrier material make up phytosomes. While the carrier phospholipid is selected based on its chemical stability, the hydrophilicity or lipophilicity of the active phytoconstituent or standardized herbal extract determines which one is used. Commonly utilized phospholipids include phosphatidylserine, phosphatidylcholine, and phosphatidyl-ethanolamine [54].
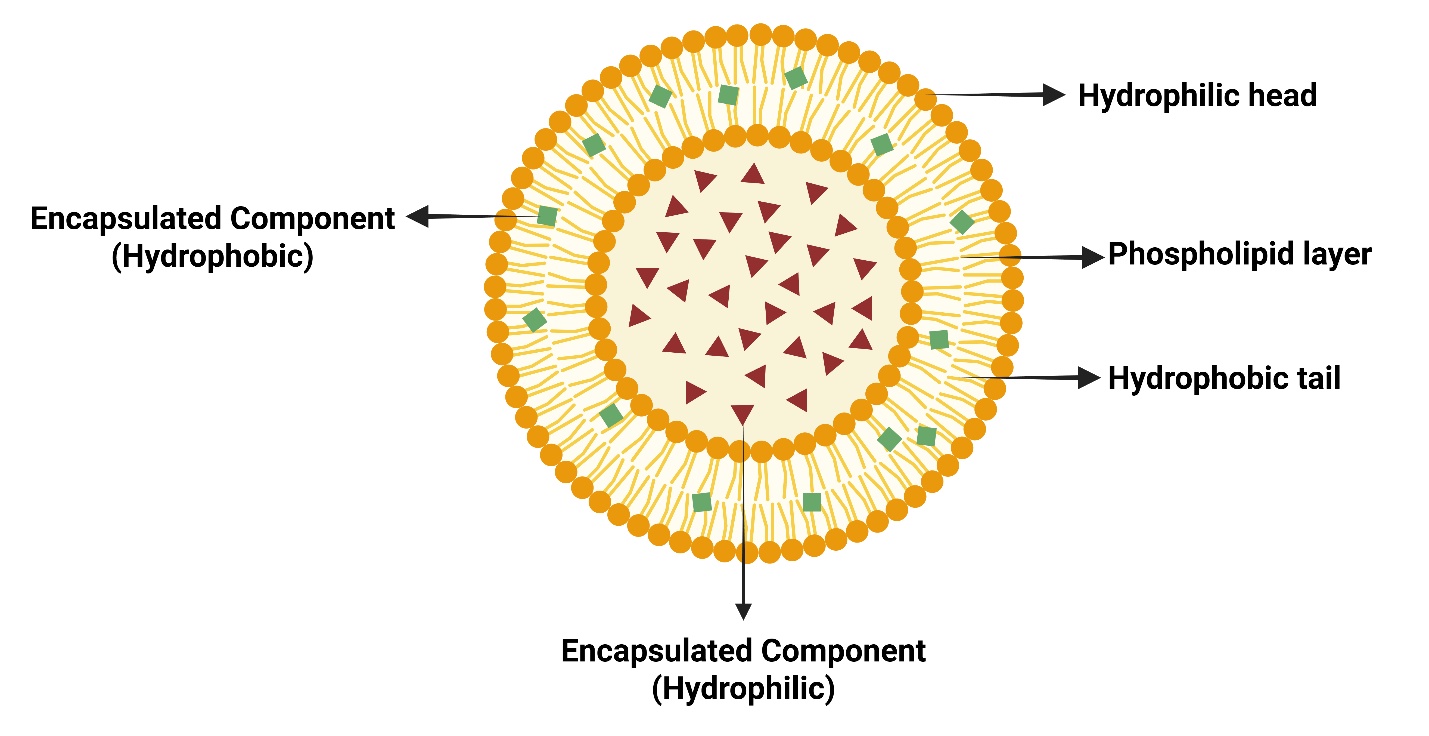
Fig. 1: Detailed compositional structure of liposome
Niosomes
Bi-layered lipid-based vesicular carriers known as niosomes have longer circulation durations than liposomes and are capable of encasing a variety of compounds. Compared to liposomes, they have the following benefits: improved stability, less adverse effects, and focused drug delivery, improved effectiveness and lower dosage needs [55]. Reverse phase evaporation and thin-film hydration are two techniques that can be used to create niosomes, which are made of cholesterol, di-ethyl ether, and nonionic surfactant. Niosomes have become widely accepted as a viable drug delivery method that can get over the drawbacks of liposomes [56].
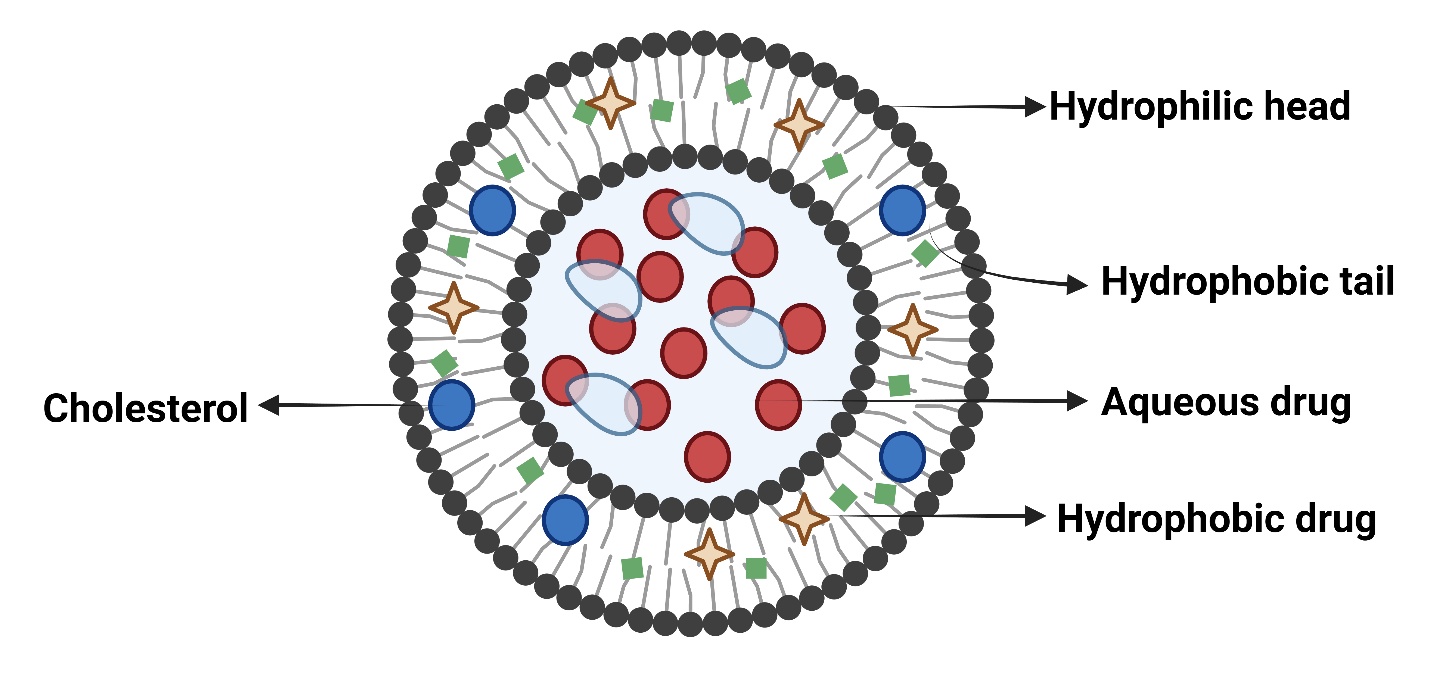
Fig. 2: Detailed compositional structure of niosomes
Ethosomes
To improve skin penetration for medication delivery, ethosomal systems with high ethanol, phospholipid, and water concentrations have been created. For transdermal drug delivery, classical ethosomes are better than liposomes due to their negative ζ-potential, increased entrapment efficiency, and improved stability profiles. By combining another alcohol with classical ethosomes, binary ethosomes are produced [57].
Sphingosomes
These are liposomal formulations that form one or more membranes and include cholesterol and sphingolipids in different proportions. With their polar headgroups facing the assembly's exterior, these lipids interact with nearby molecules. Ordered membranes are created when sphingolipids tend to form ordered domains [58].
Menthosomes
Phospholipids, menthol, and surfactants are found in vesicular carrier systems called mentosomes, which make lipid bilayers more elastic. They improve drug dispersion and partitioning, which increases medication penetration through the skin. Menthosome reduces the hexagonal/orthogonal hydrocarbon packing ratio via changing the stratum corneum's lipid packing pattern [59].
Cubosomes
A particular kind of nanoparticle known as a cubosome is created when molecules that resemble surfactants self-assemble in liquid crystalline phases with cubic symmetry. They are 100–500 nm in size and have a solid lipid crystalline structure resembling honeycomb. Cubosomes have a transparent, gel-like appearance and are viscous and optically isotropic. Amphiphilic molecules form bi-continuous water and oil channels, with a bilayer separating two independent hydrophilic areas [59].
Archaeosomes
Vesicles containing bilayers of Total Polar Lipids (TPL) that are isolated from microbes belonging to the Archaea domain are called archaeosomes. These microorganisms, which include obligate anaerobes, extreme halophiles, hyperthermophiles, and thermoacidophiles, need harsh environments to develop well, in contrast to those found in the Eukarya and Bacteria domains. Unlike eukaryotic cells, archaea are single-celled creatures with unique cell and cell-membrane adaptations for survival in hostile settings [60].
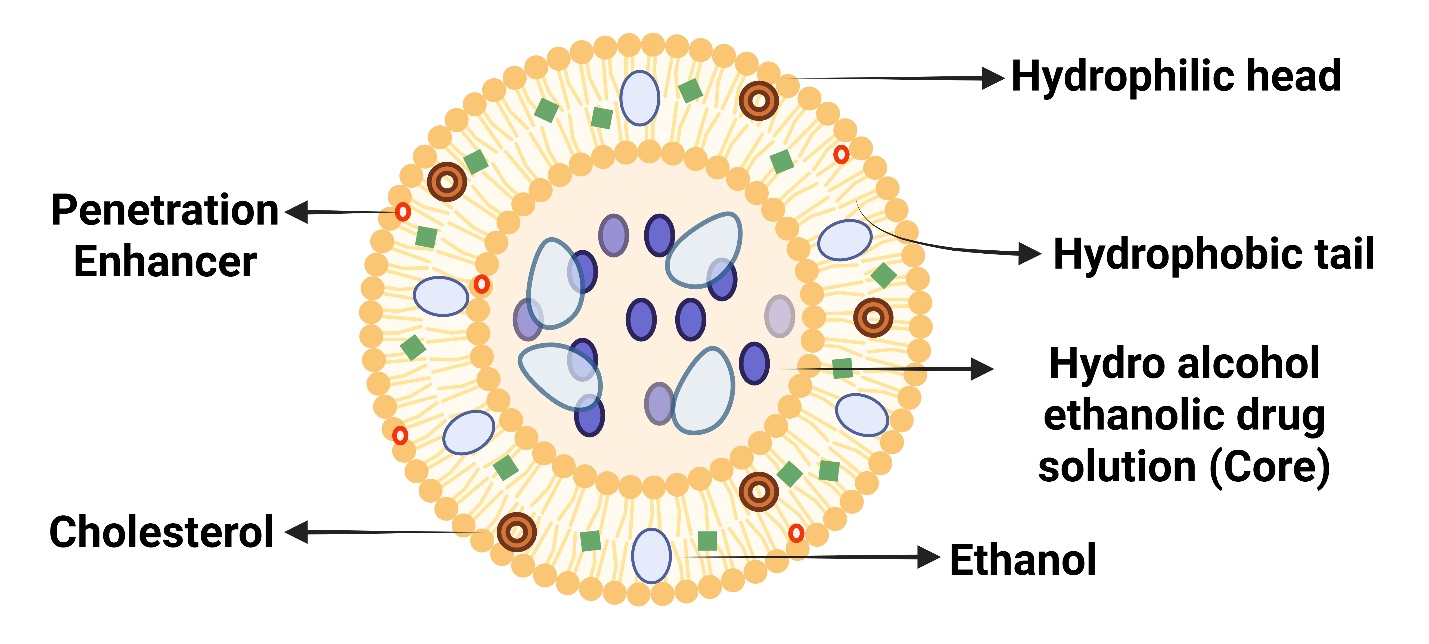
Fig. 3: Detailed compositional structure of ethosomes
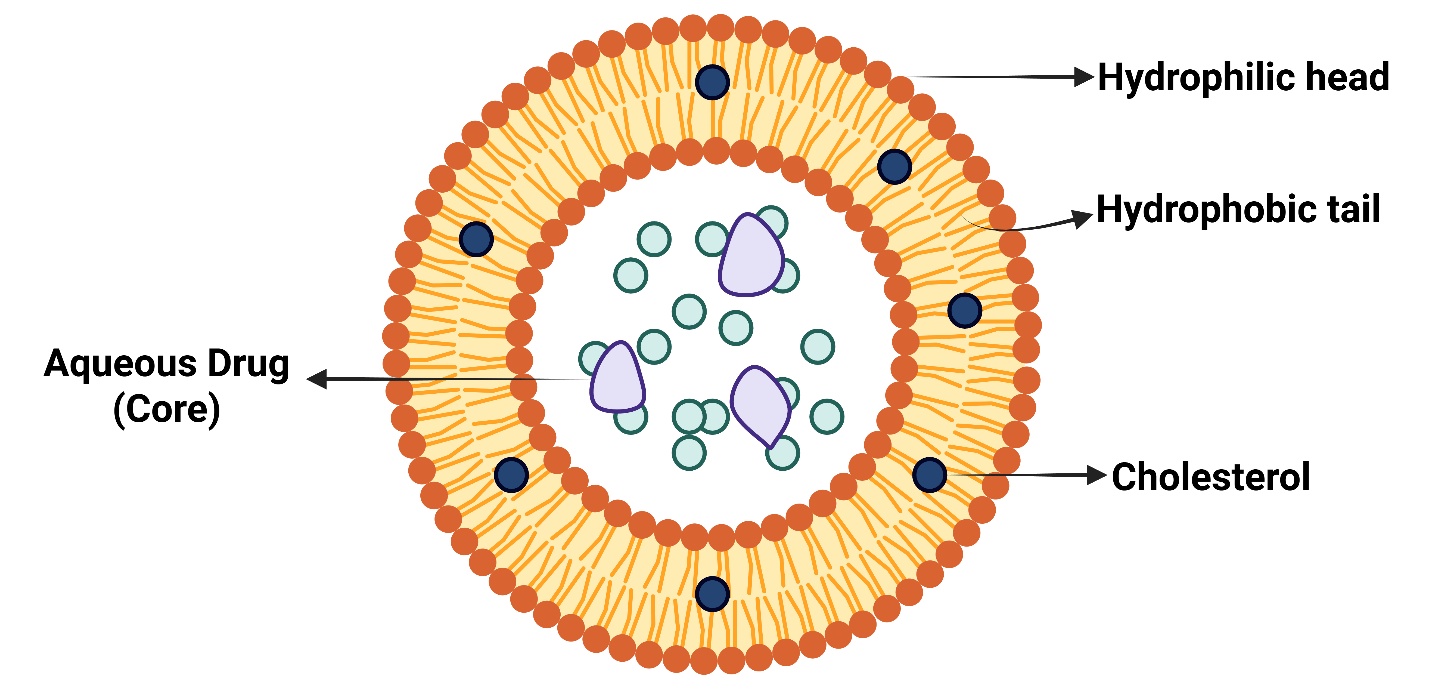
Fig. 4: Detailed compositional structure of sphingosomes
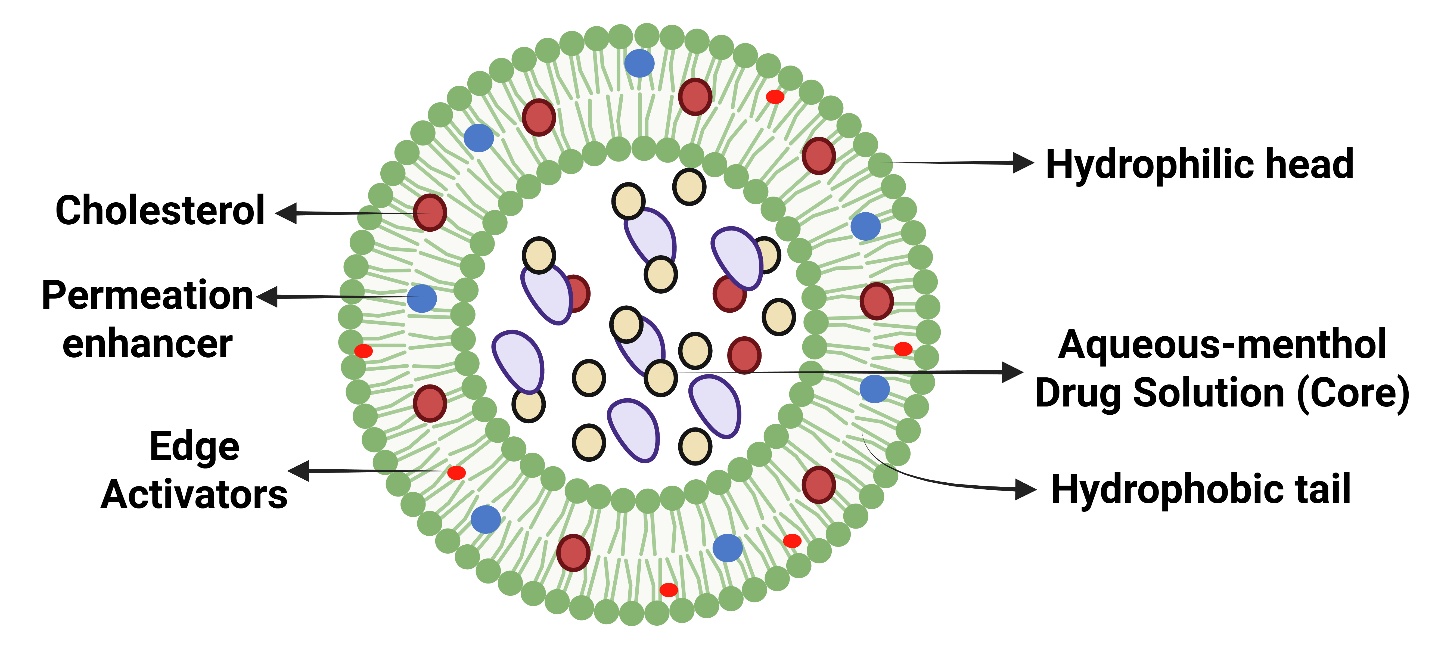
Fig. 5: Detailed compositional structure of menthosome
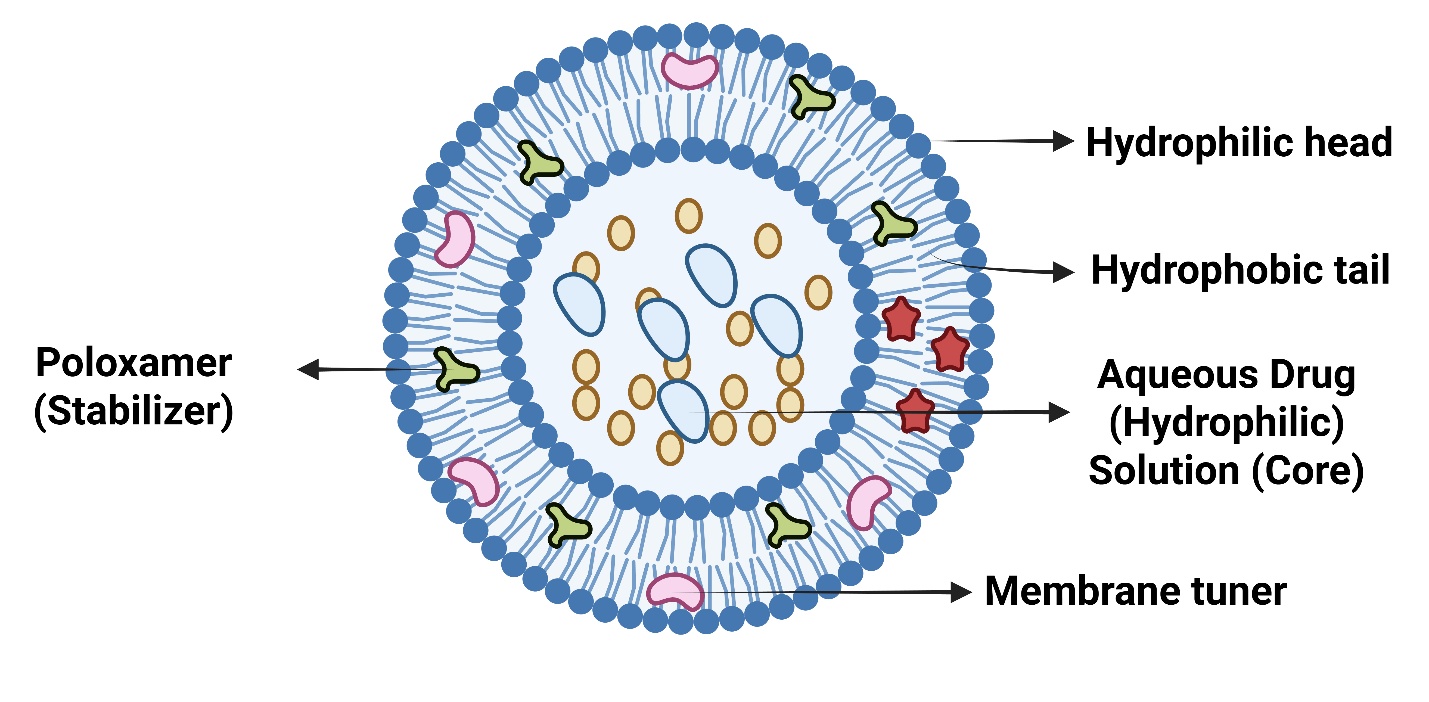
Fig. 6: Detailed compositional structure of cubosomes
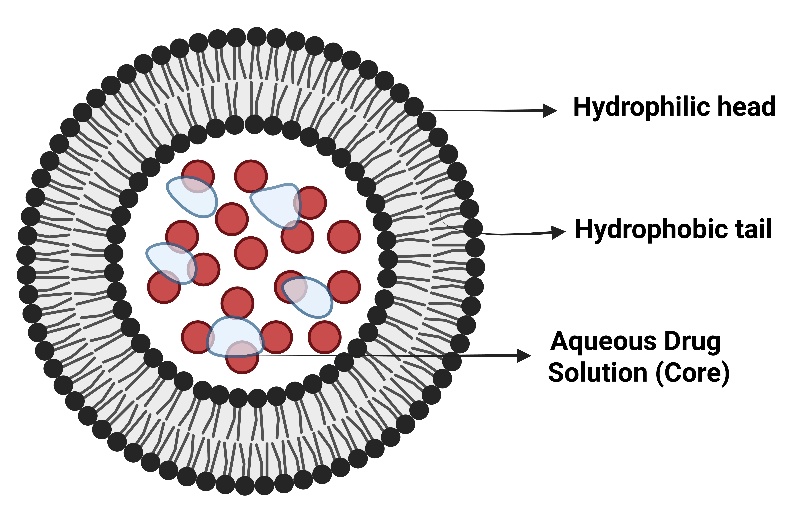
Fig. 7: Detailed compositional structure of archaeosomes
Invasosomes
Lipid-based, deformable vesicles called invasomes are used to administer drugs non-invasively by penetrating deeper into the skin or bloodstream. These nanoscales, soft, elastic vesicles are made of water, ethanol, terpene(s), and different phospholipids in trace amounts. Ethanol fluidizes the skin's intercellular lipoidal layer and lowers the density of the bilayer lipid cell membrane, allowing invasomes to penetrate through terpene, ethanol, and invasome actions [61].
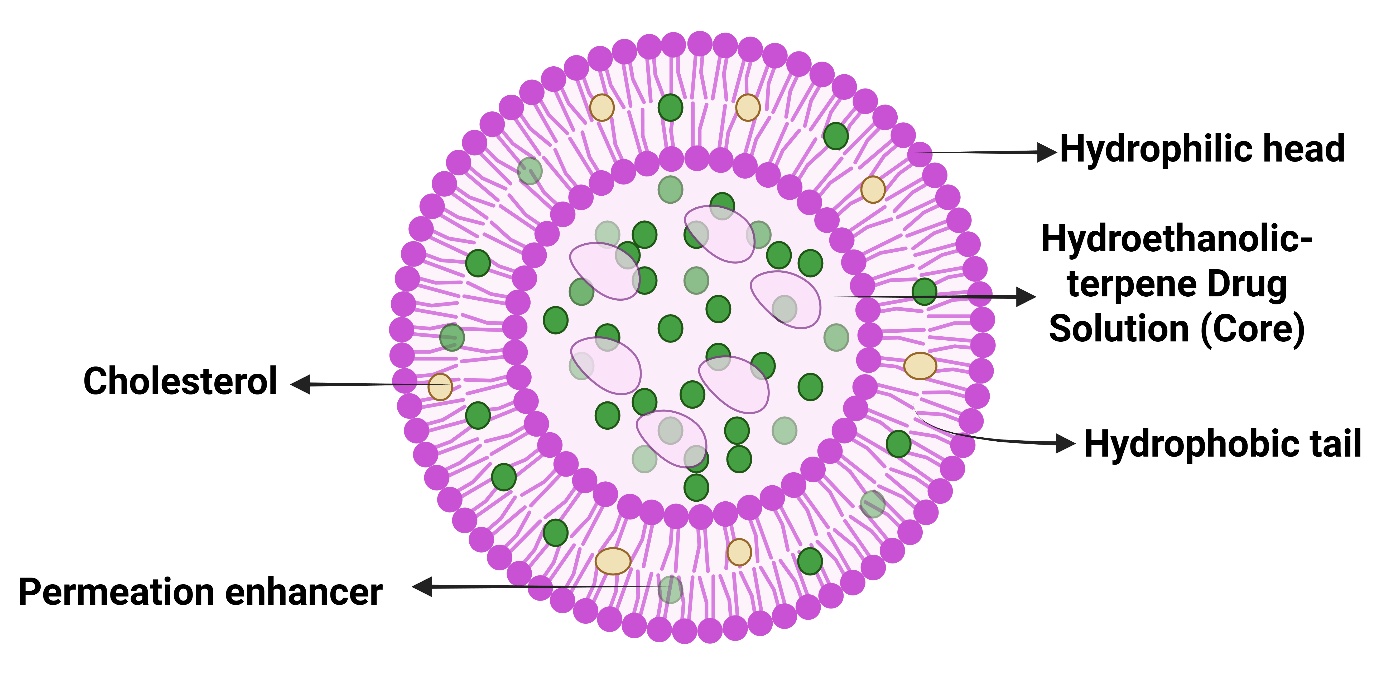
Fig. 8: Detailed compositional structure of invasomes
Transferosomes
Specially made, extremely deformable vesicles with at least one interior water compartment surrounded by lipid vesicles are known as transferosomes. Although they resemble liposomes in appearance, they can be sufficiently deformed to pass through membrane pores [62]. It is made up of the phrases "some" and "transferred," where "some" denotes body and "transferred" means carrying. Water, surfactant, and phospholipid make up transferosomes. Their ability to be elastic and highly versatile aggregates helps them quickly pass through the subcutaneous tissue's intercellular lipid route. They are about 100 nm in diameter [63].
CONCLUSION
Psoriasis is a chronic inflammatory skin condition that has many different causes, such as environmental triggers, immune system dysfunction, and genetic predisposition. Millions of people worldwide suffer from it, and it is frequently linked to systemic comorbidities such psoriatic arthritis, metabolic syndrome, and cardiovascular disease. There is presently no cure for the illness, despite advances in our understanding of it; the only treatments available concentrate on managing and remitting its symptoms. Further investigation into the immunological and genetic causes of psoriasis could lead to more precise, potent treatments. Improving patient outcomes and quality of life requires raising awareness and detecting problems early. Patients' physical health, social interactions, mental health, and financial stability are all negatively impacted by psoriasis, which has a substantial negative influence on their quality of life. In addition to causing discomfort through symptoms like itching and scaling, the illness also causes emotional distress, social stigma, and financial hardships because of treatment expenses and missed work. There are several therapeutic possibilities, such as phototherapy, systemic, biological, and topical therapies. Though they may have adverse effects, systemic and biological therapy offer relief for more severe cases of psoriasis, while topical treatments are useful for moderate cases. Another strategy is phototherapy, which uses regulated UV exposure to lessen symptoms. Psoriasis is still a chronic disorder that needs to be managed over the long term to improve patients' quality of life, even with advances in treatment. Overall, the limitations of current treatments-such as ineffective medication delivery, side effects, exorbitant prices, and inconsistent treatment responses-make managing psoriasis difficult. In order to overcome these obstacles, scientists are concentrating on sophisticated drug delivery systems, especially nano-carriers that provide improved drug stability, bioavailability, and targeted administration, such as liposomes, phytosomes, niosomes, ethosomes, and transferosomes. Natural remedies and herbal remedies, such as neem, turmeric, aloe vera, and olive oil, also offer supplementary advantages with immune-stimulating and anti-inflammatory qualities. Although these developments have the potential to improve the treatment of psoriasis, more investigation and clinical testing are required to maximize their efficacy and safety for general application.
ACKNOWLEDGMENT
It’s our privilege to express the profound sense of gratitude and cordial thanks to our respected chairman Mr. Anil Chopra and Vice Chairperson, Ms. Sangeeta Chopra, St. Soldier Educational Society, Jalandhar for providing the necessary facilities to complete this review work
AUTHORS CONTRIBUTIONS
All authors have contributed equally
CONFLICT OF INTERESTS
Declared none
REFERENCES
Mahbubur Rahman Y, Manveen Kaur A, Omveer Singh. A review on psoriasis. Neuro Quanology. 2022;20(16):5786-93. doi: 10.48047/NQ.2022.20.16.NQ880588.
Raharja A, Mahil SK, Barker JN. Psoriasis: a brief overview. Clinical Medicine (London, England). 2021 May;21(3):170–3. doi: 10.7861/clinmed.2021-0257.
Dand N, Mahil SK, Capon F, Smith CH, Simpson MA, Barker JN. Psoriasis and genetics. Acta Derm Venereol. 2020;100(3):adv00030. doi: 10.2340/00015555-3384, PMID 31971603.
Singh S, Prasad R, Tripathi JS, Rai NP. Psoriasis-an overview. World Journal of Pharmaceutical Sciences. 2015;3(8):1732–40.
Augustin M, Langenbruch A, Gutknecht M, Reich K, Korber A, Maaßen D. Definition of psoriasis severity in routine clinical care: current guidelines fail to capture the complexity of long-term psoriasis management. Br J Dermatol. 2018;179(6):1385-91. doi: 10.1111/bjd.17128, PMID 30334253.
Menaa F. Recent highlights and expert opinion on psoriasis management. Journal of Dermatology Research. 2020;1(1):1–4. doi: 10.46889/JDR.2020.1102.
Shahwan KT, Kimball AB. Psoriasis and cardiovascular disease. Medical Clinics of North America. 2015 Nov;99(6):1227-42. doi: 10.1016/j.mcna.2015.08.001.
Owczarczyk Saczonek AB, Nowicki RJ. Prevalence of cardiovascular disease risk factors and metabolic syndrome and its components in patients with psoriasis aged 30 to 49 y. Postepy Dermatologii i Alergologii. 2015;32(4):290–5. doi: 10.5114/pdia.2014.40966.
Boehncke S, Thaci D, Beschmann H, Ludwig RJ, Ackermann H, Badenhoop K. Psoriasis patients show signs of insulin resistance. Br J Dermatol. 2007 Dec;157(6):1249-51. doi: 10.1111/j.1365-2133.2007.08190.x, PMID 17916217.
Perk J, De Backer G, Gohlke H, Graham I, Reiner Z, Verschuren WM. European guidelines on cardiovascular disease prevention in clinical practice (version 2012) the fifth joint task force of the European society of cardiology and other societies on cardiovascular disease prevention in clinical practice (constituted by representatives of nine societies and by invited experts). Eur Heart J. 2012 Jul;33(13):1635-701. doi: 10.1093/eurheartj/ehs092.
Nazeer M, Ravindran S, Gangadharan G, Criton S. A survey of treatment practices in management of psoriasis patients among dermatologists of Kerala. Indian Dermatol Online J. 2019;10(4):437-40. doi: 10.4103/idoj.IDOJ_306_18, PMID 31334065.
Medically reviewed by Joan Paul. MD: MPH, DTMH, Dermatology-Written By Adam Felman Updated on; 2024 Jan 8.
Langley RG, Krueger GG, Griffiths CE. 3Psoriasis: epidemiology clinical features and quality of life. Ann Rheum Dis. 2005;64(Suppl 2):ii18-23. doi: 10.1136/ard.2004.033217, PMID 15708928.
Farber EM, Nall LM. The natural history of psoriasis in 5600 patients. Dermatological. 1974;148(1):1-18. doi: 10.1159/000251595.
World Health Organization. Global report on psoriasis. Geneva: World Health Organization; 2016. Available from: https://www.who.int/publications/i/item/9789241565189.
Boehncke WH. Systemic inflammation and cardiovascular comorbidity in psoriasis patients: causes and consequences. Front Immunol. 2018 Apr;9(9):579. doi: 10.3389/fimmu.2018.00579, PMID 29675020.
Alexis AF, Blackcloud P. Psoriasis in skin of color: epidemiology, genetics, clinical presentation and treatment nuances. J Clin Aesthet Dermatol. 2014;7(11):16-24. PMID 25489378.
Gottlieb A, Korman NJ, Gordon KB, Feldman SR, Lebwohl M, Koo JY. Guidelines of care for the management of psoriasis and psoriatic arthritis: section 2 psoriatic arthritis: overview and guidelines of care for treatment with an emphasis on the biologics. J Am Acad Dermatol. 2008 May;58(5):851-64. doi: 10.1016/j.jaad.2008.02.040, PMID 18423261.
Van De Kerkhof PC, Schalkwijk J. Psoriasis. In: Bolognia JL, Jorizzo JL, Rapini RP, editors. Dermatology. 2nd ed. St Louis: Mosby/Elsevier; 2008. p. 115.
Naldi L, Peli L, Parazzini F, Carrel CF. Family history of psoriasis stressful life events and recent infectious disease are risk factors for a first episode of acute guttate psoriasis: results of a case-control study. J Am Acad Dermatol. 2001 Mar;44(3):433-8. doi: 10.1067/mjd.2001.110876, PMID 11209111.
Abel EA, Di Cicco LM, Orenberg EK, Fraki JE, Farber EM. Drugs in exacerbation of psoriasis. J Am Acad Dermatol. 1986 Nov;15(5 Pt 1):1007-22. doi: 10.1016/S0190-9622(86)70265-X, PMID 2878015.
Gupta MA, Gupta AK, Kirkby S, Schork NJ, Gorr SK, Ellis CN. A psychocutaneous profile of psoriasis patients who are stress reactors: a study of 127 patients. Gen Hosp Psychiatry. 1989 May;11(3):166-73. doi: 10.1016/0163-8343(89)90036-4, PMID 2721939.
Higgins E. Alcohol smoking and psoriasis. Clin Exp Dermatol. 2000;25(2):107-10. doi: 10.1046/j.1365-2230.2000.00588.x, PMID 10733631.
Naldi L, Peli L, Parazzini F. Association of early-stage psoriasis with smoking and male alcohol consumption: evidence from an Italian case-control study. Arch Dermatol. 1999;135(12):1479-84. doi: 10.1001/archderm.135.12.1479, PMID 10606053.
Chaudhury S, Das AL, John RT, Ramadasan P. Psychological factors in psoriasis. Indian J Psychiatry. 1998 Jul;40(3):295-9. PMID 21494487.
Armstrong AW, Read C. Pathophysiology, clinical presentation and treatment of psoriasis: a review. JAMA. 2020;323(19):1945-60. doi: 10.1001/jama.2020.4006, PMID 32427307.
Menter A, Korman NJ, Elmets CA, Feldman SR, Gelfand JM, Gordon KB. Guidelines of care for the management of psoriasis and psoriatic arthritis: section 4. Guidelines of care for the management and treatment of psoriasis with traditional systemic agents. J Am Acad Dermatol. 2009 Sep;61(3):451-85. doi: 10.1016/j.jaad.2009.03.027, PMID 19493586.
National Psoriasis Foundation. About psoriasis: clinical presentation and symptoms. National Psoriasis Foundation; 2023. Available from: https://www.psoriasis.org.
Mease PJ. Psoriatic arthritis: update on pathophysiology assessment and management. J Rheumatol. 2009;36(Suppl 83):3-8.
Finlay AY, Coles EC. The effect of severe psoriasis on the quality of life of 369 patients. Br J Dermatol. 1995;132(2):236-44. doi: 10.1111/j.1365-2133.1995.tb05019.x, PMID 7888360.
Krueger G, Koo J, Lebwohl M, Menter A, Stern RS, Rolstad T. The impact of psoriasis on quality of life: results of a 1998 national psoriasis foundation patient-membership survey. Arch Dermatol. 2001 Mar;137(3):280-4. PMID 11255325.
Fortune DG, Richards HL, Griffiths CE, Main CJ. Psychological stress, distress and disability in patients with psoriasis: consensus and variation in the contribution of illness perceptions, coping and alexithymia. Br J Clin Psychol. 2002;41(2):157-74. doi: 10.1348/014466502163949, PMID 12034003.
Bhosle MJ, Kulkarni A, Feldman SR, Balkrishnan R. Quality of life in patients with psoriasis. Health Qual Life Outcomes. 2006;4:35. doi: 10.1186/1477-7525-4-35.
Choi J, Koo JY. Quality of life issues in psoriasis. J Am Acad Dermatol. 2003 Aug;49(2 Suppl):S57-61. doi: 10.1016/S0190-9622(03)01136-8, PMID 12894127.
Horn EJ, Fox KM, Patel V, Chiou CF, Dann F, Lebwohl M. Association of patient-reported psoriasis severity with income and employment. J Am Acad Dermatol. 2007;57(6):963-71. doi: 10.1016/j.jaad.2007.07.023, PMID 17761358.
Bakshi H, Nagpal M, Singh M, Dhingra GA, Aggarwal G. Treatment of psoriasis: a comprehensive review of entire therapies. Curr Drug Saf. 2020;15(2):82-104. doi: 10.2174/1574886315666200128095958, PMID 31994468.
Torsekar R, Gautam MM. Topical therapies in psoriasis. Indian Dermatol Online J. 2017 Jul-Aug;8(4):235-45. doi: 10.4103/2229-5178.209622, PMID 28761838.
Bernhard J, Whitmore C, Guzzo C, Kantor I, Kalb RE, Ellis C. Evaluation of halobetasol propionate ointment in the treatment of plaque psoriasis: report on two double-blind, vehicle-controlled studies. J Am Acad Dermatol. 1991 Dec;25(6 Pt 2):1170-4. doi: 10.1016/0190-9622(91)70320-2, PMID 1757612.
Cather JC, Abramovits W, Menter A. Cyclosporine and tacrolimus in dermatology. Dermatol Clin. 2001;19(1):119-37. doi: 10.1016/S0733-8635(05)70234-5, PMID 11155577.
Binderup L, Bramm E. Effects of a novel vitamin D analogue MC903 on cell proliferation and differentiation in vitro and on calcium metabolism in vivo. Biochem Pharmacol. 1988;37(5):889-95. doi: 10.1016/0006-2952(88)90177-3, PMID 2830885.
Kalb RE, Strober B, Weinstein G, Lebwohl M. Methotrexate and psoriasis: 2009 national psoriasis foundation consensus conference. J Am Acad Dermatol. 2009;60(5):824-37. doi: 10.1016/j.jaad.2008.11.906, PMID 19389524.
Collins P, Rogers S. The efficacy of methotrexate in psoriasis a review of 40 cases. Clin Exp Dermatol. 1992;17(4):257-60. doi: 10.1111/j.1365-2230.1992.tb02161.x, PMID 1451315.
Salvarani C, Boiardi L, Macchioni P, Pipitone N, Catanoso M, Pigatto P. Multidisciplinary focus on cyclosporin a. J Rheumatol Suppl. 2009;83:52-5. doi: 10.3899/jrheum.090225, PMID 19661542.
Di Caprio R, Caiazzo G, Cacciapuoti S, Fabbrocini G, Scala E, Balato A. Safety concerns with current treatments for psoriasis in the elderly. Expert Opin Drug Saf. 2020;19(4):523-31. doi: 10.1080/14740338.2020.1728253, PMID 32056449.
Dogra S, Yadav S. Acitretin in psoriasis: an evolving scenario. Int J Dermatol. 2014;53(5):525-38. doi: 10.1111/ijd.12365, PMID 24601982.
Papp K, Reich K, Leonardi CL, Kircik L, Chimenti S, Langley RG. Apremilast an oral phosphodiesterase 4(PDE4) inhibitor, in patients with moderate to severe plaque psoriasis: results of a phase III, randomized controlled trial (efficacy and safety trial evaluating the effects of apremilast in psoriasis [esteem] 1). J Am Acad Dermatol. 2015 Jul;73(1):37-49. doi: 10.1016/j.jaad.2015.03.049, PMID 26089047.
Balak DM. Fumaric acid esters in the management of psoriasis. Psoriasis (Auckl). 2015;5:9-23. doi: 10.2147/PTT.S51490, PMID 29387578.
Pedraz J, Dauden E, Delgado Jimenez Y, Garcia Rio I, Garcia Diez A. Sequential study on the treatment of moderate-to-severe chronic plaque psoriasis with mycophenolate mofetil and cyclosporin. J Eur Acad Dermatol Venereol. 2006;20(6):702-6. doi: 10.1111/j.1468-3083.2006.01577.x, PMID 16836499.
Wong T, Hsu L, Liao W. Phototherapy in psoriasis: a review of mechanisms of action. J Cutan Med Surg. 2013;17(1):6-12. doi: 10.2310/7750.2012.11124, PMID 23364144.
Feldman SR, Goffe B, Rice G, Mitchell M, Kaur M, Robertson D. The challenge of managing psoriasis: unmet medical needs and stakeholder perspectives. Am Health Drug Benefits. 2016 Dec;9(9):504-13. PMID 28465778.
Pandey R, Bhairam M, Shukla SS, Gidwani B. Colloidal and vesicular delivery system for herbal bioactive constituents. Daru. 2021;29(2):415-38. doi: 10.1007/s40199-021-00403-x, PMID 34327650.
Nakhaei P, Margiana R, Bokov DO, Abdelbasset WK, Jadidi Kouhbanani MA, Varma RS. Liposomes: structure, biomedical applications and stability parameters with emphasis on cholesterol. Front Bioeng Biotechnol. 2021 Sep 9;9:705886. doi: 10.3389/fbioe.2021.705886, PMID 34568298.
Alharbi WS, Almughem FA, Almehmady AM, Jarallah SJ, Alsharif WK, Alzahrani NM. Phytosomes as an emerging nanotechnology platform for the topical delivery of bioactive phytochemicals. Pharmaceutics. 2021 Sep 15;13(9):1475. doi: 10.3390/pharmaceutics13091475, PMID 34575551.
Lu M, Qiu Q, Luo X, Liu X, Sun J, Wang C. Phyto-phospholipid complexes (phytosomes): a novel strategy to improve the bioavailability of active constituents. Asian J Pharm Sci. 2019 May;14(3):265-74. doi: 10.1016/j.ajps.2018.05.011, PMID 32104457, PMCID PMC7032241.
Kazi KM, Mandal AS, Biswas N, Guha A, Chatterjee S, Behera M. Niosome: a future of targeted drug delivery systems. J Adv Pharm Technol Res. 2010 Oct;1(4):374-80. doi: 10.4103/0110-5558.76435, PMID 22247876.
Ge X, Wei M, He S, Yuan WE. Advances of non-ionic surfactant vesicles (niosomes) and their application in drug delivery. Pharmaceutics. 2019;11(2):55. doi: 10.3390/pharmaceutics11020055, PMID 30700021.
Abdulbaqi IM, Darwis Y, Khan NA, Assi RA, Khan AA. Ethosomal nanocarriers: the impact of constituents and formulation techniques on ethosomal properties in vivo studies and clinical trials. Int J Nanomedicine. 2016;11:2279-304. doi: 10.2147/IJN.S105016, PMID 27307730.
Van Meer G, Voelker DR, Feigenson GW. Membrane lipids: where they are and how they behave. Nat Rev Mol Cell Biol. 2008;9(2):112-24. doi: 10.1038/nrm2330, PMID 18216768.
Umar H, Wahab HA, Gazzali AM, Tahir H, Ahmad W. Cubosomes: design, development and tumor-targeted drug delivery applications. Polymers (Basel). 2022;14(15):3118. doi: 10.3390/polym14153118, PMID 35956633.
Gonzalez RO, Higa LH, Cutrullis RA, Bilen M, Morelli I, Roncaglia DI. Archaeosomes made of halorubrum tebenquichense total polar lipids: a new source of adjuvancy. BMC Biotechnol. 2009 Aug 13;9:71. doi: 10.1186/1472-6750-9-71, PMID 19678953.
Babaie S, Bakhshayesh AR, Ha JW, Hamishehkar H, Kim KH. Invasome: a novel nanocarrier for transdermal drug delivery. Nanomaterials (Basel). 2020;10(2):341. doi: 10.3390/nano10020341, PMID 32079276.
Cevc G. Material transport across permeability barriers by means of lipid vesicles. In: Lipowsky R, editor. Handbook of Physics of Biological Systems. Biological Physics. Amsterdam: Elsevier Science; 1994. p. 441–66.
Cevc G, Blume G, Schatzlein A, Gebauer D, Paul A. The skin: a pathway for systemic treatment with patches and lipid-based agent carriers. Adv Drug Deliv Rev. 1996;18(3):349-78. doi: 10.1016/0169-409X(95)00091-K.