
Int J Curr Pharm Res, Vol 18, Issue 2, 38-42Original Article
EVALUATION OF DIURETIC ACTIVITY OF ALCOHOLIC EXTRACT OF FLOWER BUDS OF NEOLAMARCKIA CADAMBA IN ALBINO RATS
AKASH*, AMAR PAL SINGH, AJEET PAL SINGH
Department of Pharmacology, St. Soldier Institute of Pharmacy, Lidhran Campus, Behind NIT (R. E. C.), Jalandhar –Amritsar by pass, NH-1, Jalandhar-144011, Punjab, India
*Corresponding author: Akash; *Email: akashkaushal5360@gmail.com
Received: 16 Nov 2025, Revised and Accepted: 03 Jan 2026
ABSTRACT
Objective: To study the diuretic activity of Alcoholic Extract of Flower buds of Neolamarckia cadamba by Lipschitz method in albino Wistar rats.
Methods: Metabolic cages were utilised to test the diuretic effect of Alcoholic Extract of Flower Buds of Neolamarckia cadamba in four Albino Wistar rats. Group I, the normal control, got vehicle (10 ml/kg or 1 ml of 0.9% NaCl/100 gm), group II received Frusemide (10 mg/Kg, p. o), and groups III and IV received low (200 mg/kg) and high (400 mg/kg) dosages of Alcoholic Extract of Neolamarckia cadamba flower buds Following Neolamarckia cadamba Alcoholic Extract treatment, rats were hydrated with saline (15 ml/kg, p. o) and put in metabolic cages at 25 °C±0.5 °C. Animals went 5 h without food or water. Each metabolic cage's urine volume was assessed after 5 and 24 h. Total urine volume, ion concentration (Sodium, Potassium, Chloride), and pH were measured.
Results: In this model when compared to control group the Alcoholic Extract of Flower buds of Neolamarckia cadamba treated groups at different dose levels (200 and 400 mg/kg) have noted with significant increase in the urine volume and also significantly enhanced the excretion of Sodium, Potassium and Chloride ions in urine. In the urine pH no difference has been found.
Conclusion: Results showed that single dose administration of standard Furosemide and Alcoholic Extract of Flower buds of Neolamarckia cadamba significantly increased the urine output along with an increase in elimination of Sodium, Potassium, and Chloride ions. Alcoholic Extract of Flower buds of Neolamarckia cadamba, 400 mg/Kg produced a comparable diuretic activity with standard Frusemide.
Keywords: Flower buds of Neolamarckia cadamba, Alcoholic extract, Hydrated rats, Diuretic activity, Metabolic cage
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i2.8018 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Drugs known as diuretics cause an increase in the excretion of sodium and water ions. Drugs called diuretics change how the kidneys work. They treat renal and cardiovascular conditions. One Diuretics, often known as water tablets, aid in the body's removal of water and salt (sodium) [1]. The majority of these medications aid in the kidneys' increased salt excretion in urine. By removing water from the circulation, salt helps reduce the volume of fluid that passes through arteries and veins [2]. Blood pressure is lowered as a result. Diuretics are used to help the body get rid of excess salt or fluid. Diuretics are often used to treat renal illness, heart failure, excessive blood pressure, and swollen tissues. An excess of fluid in the body may cause breathing difficulties and interfere with the heart's ability to function [3]. The initial impact of prescription diuretics, sometimes known as water pills, is to increase urination. Here are some important diuretic facts [4].
Doctors may prescribe diuretic medicine; diuretics can be pharmaceuticals; diuretics can also be foods and beverages that serve as natural diuretics; and diuretics can alter the body's levels of certain minerals [5]. The Cadamba is a significant medicinal plant that is a member of the Rubiaceae family. Because it contains the greatest number of phytochemicals and secondary metabolites with pharmacological and biological qualities (such as cadambagenic acid, cadamine, quinovic acid, β-sitosterol, cadambine, etc.), it is very important [6]. It may be used to prevent and cure a number of incurable illnesses in place of other synthetic chemical chemicals. Numerous phytochemicals and their effects have been discovered during more than a century of study [7]. Because of the absence of an appropriate model system and other contentious concerns, only a small percentage of them-roughly 2%-have been commercialised. Another big worry is the solubility of phytochemicals; the solvent employed will potentially cause unpredicted additional reactions [8]. Additionally, the Cadamba is a religiously significant decorative plant. In order to generate interest that would aid in their commercialisation, we have attempted to provide a summary of all phytochemicals and their importance here [9]. It is a decorative plant that is also used to make paper and lumber. In Indian religion and mythology, it is very important. A Cadamba tree's immense importance to humanity has led many Indian faiths to firmly believe that God resides within it.
The discovery of many phytochemicals and secondary metabolites (saponins, indole and quinoline alkaloids, secoiridoids, and triterpenes) having pharmacological importance has been the subject of investigation by several scientists worldwide [10]. The importance of the Cadamba and its derivative products to humanity serves as the foundation for this study [11].
MATERIALS AND METHODS
Experimental animals
Because of its small size, adaptability, docility, cheap husbandry expenses, fertility, well-defined health and genetic backgrounds, and relative simplicity of genetic modification, the domestic mouse (Mus musculus and related subspecies) is a popular choice for mammalian research. The use of mice as study subjects was significantly expanded with the advent of genetic engineering methods that allowed for the insertion of foreign genes (transgenes) into the mouse genome and the deletion of genes, creating what are known as "knockout" mice. Since these developments, many mouse mutant genotypes have been created, ranging from mild immune function abnormalities to serious hereditary illnesses that are almost identical to those of higher animals [12].
The domestic Norway rat (Rattus norvegicus) is the second most popular rodent for study, behind the mouse. Rats are appealing for study because they have many of the same characteristics as mice, but since they are bigger, they may be subjected to a wider range of treatments. A wide range of research topics, including ageing, cancer, reproductive physiology, drug effects, behaviour, addiction, alcoholism/cirrhosis, arthritis, brain and nerve injury, hypertension, embryology, teratology, endocrine diseases, neurophysiology, infectious disease, stroke, organ transplantation, and surgically induced disease, use a variety of mutant and inbred strains and outbred stocks of rats. However, compared to mice, the genetic engineering of mutant rat types has trailed behind and is just now starting to show results [13].
The most often utilised animals in research, apart from mice and rats, are rabbits and guinea pigs, however their populations have decreased since the 1980s. Despite being one of the earliest species used in medical research, the guinea pig (Cavia porcellus) has lost some of its appeal in comparison to mice and rats due to its lengthy gestation period (59–72 d), small litter size (2–5), inadequate vascular access, and anaesthesia challenges. There are still noteworthy use for guinea pigs in immunology, vaccination and infectious disease research, and as models for hearing. The most common uses for rabbits are in the manufacturing of polyclonal antibodies, the assessment of product safety, and research in the fields of cardiology, orthopaedics, and eyesight [14].
Other hamster species employed in study include the Armenian, Siberian (Djungarian), Chinese, European, and Turkish species, in addition to the Syrian hamster (Mesocricetus auratus). Hamsters are widely accessible, have an easy time reproducing, and are comparatively free from spontaneous infections, however they are vulnerable to several viral diseases. Obesity, induced carcinogenesis, prostate illness, toxicity, infectious disorders (including slow viruses), dental caries, chronic bronchitis, and teratogenesis are among the topics they are utilised to study.
Gerbils, deer mice, chinchillas, cotton rats, rice rats, multi-mammate rats, Egyptian spiny mice, degus, voles, and woodchucks are some of the additional rodent species that are used in research [15].
Larger animal models provide special prospects for biomedical research, even if rodent models are unquestionably the most often used for scientific purposes. Cardiology, endocrinology, orthopaedics, prosthetics, surgery, pharmacokinetics, and product safety are among the fields that utilise dogs. Livestock has been employed more often since the usage of dogs started to decline in 1984. This has resulted from public and regulatory pressure over the use of dogs, but it is also because of characteristics of comparative anatomy and physiology that make cattle better suitable for specific research. For instance, pigs are employed as surgical models, in studies of gut physiology, in cardiovascular research (especially atherosclerosis), and in xenotransplantation. Sheep are utilised for research on human vaccine development, the pathophysiology and treatment of asthma, medication delivery, circadian rhythms, newborn development, and surgical methods [16].
Nonhuman primates continue to be essential for research on infectious illnesses, neurology, vision, vaccinations, and product safety. Their value as models of immunodeficiency virus infection and aging-related neurodegenerative disorders has grown in recent years. Cats continue to be valuable models in the study of infectious illnesses and the neurosciences, despite a gradual drop in the total number of cats employed in research since 1980. The African clawed frog and the zebra fish are the two most significant aquatic species used in research. Goats, calves, horses, ferrets, armadillos, opossums, domestic and wild birds, amphibians, reptiles, other fish species, and invertebrates are among the various animals that are employed in scientific study [17]. In the current study, albino rats (150–200 g) of either sex were kept in various polypropylene cages with husk bedding in the central animal house facility of St. Soldier Institute of Pharmacy, Jalandhar, Punjab, under standard light and dark cycle conditions. They were fed a standard laboratory pellet chow diet and given unlimited access to water. Before the experimental trial began, the animals were acclimated to the laboratory environment. Every trial took place in a semi-soundproof laboratory setting between 8:00 and 16:00. The Institutional Animal Ethics Committee (IAEC) approved the experimental protocol, which was conducted in accordance with the Committee for the purpose of Control and Supervision of Experimental Animals (CPCSEA) guidelines for the use and care of experimental animals, Ministry of Environment and Forests, Government of India (Reg. No. 2011/PO/Re/S/18/CPCSEA, with a registration date of 1/5/2018). Sufficient precautions were made to reduce any pain or discomfort experienced by the animals throughout the experiment. IAEC/CPCSEA has formally accepted the research protocol (IAEC/SSIP/2023/PR-029).
Drugs and reagents/Chemical
Every chemical and biological reagent utilised in this investigation was of analytical quality and was made just before use. All analytical-grade chemicals were purchased from S. D. Fine Chem. Ltd. in India and Sigma Chemical in the United States.
RESULTS
Acute oral toxicity study
Using five rats that were fasted overnight prior to being given an oral dose of ENC extract at a maximum dose level of up to 1000 mg/kg, starting with doses of 5, 50, and 300 mg/kg, the median lethal dose (LD50) of ENC was calculated in compliance with the Organisation for Economic Co-operation and Development (OECD, 425) guidelines. After giving one rat its first dosage, the meal was further withheld for four hours. Signs of toxicity (alterations in mucous membranes, skin, hair, and eyes, as well as circulatory, respiratory, somato-motor, and behavioural patterns) and death were monitored for the first 24 h and subsequently for 14 d. No behavioural reactions have changed, and there is no evidence of acute oral toxicity. For two weeks, the other four rats were similarly given doses and monitored. The LD50 was then calculated.
The lipschitz test and estimation of urine electrolytes
The Lipschitz Test was used to examine the diuretic effects of an ethanolic extract of Neolamarckia cadamba flower buds in albino rats. Rats were given 1 ml per 100 g of body weight of distilled water in the first experiments. Urine was then collected from each of them in metabolic cages, and after six hours, the volume was assessed. Test animals were chosen based on their ability to excrete at least 40% of the dosing solution volume. Those with excretion rates below 40% were not included. Four groups of six rats each were created from the chosen animals. After that, each animal was kept separately in metabolic cages for seven days to acclimatise. Rats were kept in typical humidity and temperature conditions. The diuretic activity was measured using the Lipschits et al. technique. The Institutional Animal Ethical Committee has authorised the experimental procedures. The rats in each group who had fasted overnight were given the test medication and vehicles. Since rats typically produce just 1-2 ml of urine per day, distilled water (2 ml per 100 g) was given to all groups of rats 30 min after the test medication was delivered in order to get a quantifiable amount of pee.
Prior to the experiment, four groups of six rats each were fasted and denied water for eighteen hours. The animals in the first group were used as the control group and were given either normal saline (10 ml/kg, p. o. or 1 ml of 0.9% NaCl/100 g body weight); Frusemide (10 mg/kg, i. p.) in saline; and the ethanolic extract (200 mg/kg and 400 mg/kg, respectively) in normal saline. Conical flasks were placed beneath the polythene funnel of the metabolic cages, which were maintained at room temperature 25±0.5° throughout the experiment. The animals were immediately placed in metabolic cages (one animal per metabolic cage), which had a wire mesh bottom and a funnel to collect urine and faeces. The funnel was specially made to separate urine and faeces (faecal matter is retained and urine is allowed to pass through stainless steel sieves). Every precaution was made to prevent faecal particles from contaminating the urine. All control and drug-treated groups had their urine collected in measuring cylinders for up to 24 h, and the volume was measured 1, 2, 3, 4, 5, and 24 h after the extract was administered. Animals were not given access to food or water during this time. The Lipschitz value is used to calculate this index for both the 5-hour and 24-hour excretion periods, which show how long the diuretic impact lasts. It has been shown that the Lipschitz test is a reliable and effective technique for identifying possible diuretics.
By increasing the excretion of sodium, potassium, and chloride, we can show in this research that an ethanolic extract of Neolamarckia cadamba flower buds may have a diuretic impact. Controlling plasma sodium is crucial for controlling blood pressure and volume. For the heart and skeletal muscles to continue to operate properly, plasma potassium levels must be controlled. Renal control of acid-base balance is closely linked to the regulation of sodium and potassium balance. Hypokalaemia may result from the potassium loss that many diuretics cause. Potassium-sparing diuretics are thus often advised. The potassium-sparing ability of ethanolic extracts has to be studied since in the current investigation, their use led to increased potassium levels in the urine, which may raise the risk of hypokalaemia. Diuretic action is known to be caused by active ingredients such flavanoids and glycosides. The table's findings unequivocally shown that Neolamarckia cadamba flower buds' ethanolic extract has a dose-dependent diuretic effect. The existence of these secondary metabolites may have been inferred by the first screening of the recommended plant extract.
Estimation of urinary electrolytes/Volume/pH
According to the user manual for the biochemical kits (Roche, Roche Diagnostics Pvt. Ltd, Gurgaon, Haryana), the Ion Selective Electrode technique was used to determine the electrolytes (sodium, potassium, and chloride) in urine. Standard pH paper was used to measure the urine's volume, electrolytes (Na+, K+(cations), and Cl–(anions), and pH. Urine volume was estimated in order to evaluate diuretic activity.
Statistical analysis
The mean+SEM (n=6) was used to represent the experimental findings and values. The data obtained throughout the research was analysed using the Student's t test for unpaired data, with a significance level of statistical significance and a P-value (probability) threshold of ≤0.05. The significance level was observed and interpreted appropriately.
Phenolic compounds, proteins, tannins, glycosides, carbohydrates, starches, vitamins, and minerals were among the phytoconstituents identified by the ethanolic extract of Neolamarckia cadamba flower buds.
Even after 14 d, every animal in the acute toxicity investigation survived. This suggests that the extract was deemed safe up to the 1000 mg/kg maximum dosage level that was examined. No significant changes in behaviour were seen over this research period.
Table 1: Details of experimental protocols for lipschitz test
| Group name | Number of animals required |
| Naïve Animal, Control, Normal saline (10 ml/Kg) p. o. | 1x6=6 |
| Group treated with Frusemide 10 mg/kg p. o. | 1x6=6 |
| Ethanolic extract of flower buds of Neolamarckia cadambalow dose (200 mg/kg b. wt) (D1) p. o. | 1 X 6=6 |
| Ethanolic extract of flower buds of Neolamarckia cadamba High dose (400 mg/kg b. wt) (D2) p. o. | 1 X 6=6 |
| Total Number of Animals required: n=6; N=4; = 24 rats |
Total No. of animals required: No. of the animal in each group (n) = 6, No. of groups (N) = 4, Total no. of animals required = 24
Table 2: Percentage yield and preliminary phyto-profile of Neolamarckia cadamba extract
| S. No. | Groups | Total urine vol. (ml/5 h) | Na+μmol/l | K+μmol/l | Cl– μmol/l |
| 1 | Control (10 ml/Kg b. wt) p. o. | 11.45±0.06 | 102.04±2.18 | 53.08±1.52 | 72.96±1.44 |
| 2 | Standard (Frusemide 10 mg/kg b. wt) p. o. | 20.23±0.04*** | 181.06±2.08*** | 88.22±1.64*** | 112.06±1.68*** |
| 3 | Alcoholic extract of flower buds of Neolamarckia cadamba low dose (200 mg/kg b. wt) p. o. | 14.41±0.06* | 144.44±2.06* | 66.44±2.68* | 82.66±1.24* |
| 4 | Alcoholic extract of flower buds of Neolamarckia cadamba High dose (400 mg/kg b. wt) p. o. | 17.46±0.08** | 165.54±2.34** | 72.22±1.78** | 98.38±2.44** |
Effect of alcoholic extract of flower buds of Neolamarckia cadamba low dose on urine volume up to 5 h and electrolyte concentration in hydrated rat model in albino rats
Value expressed as mean±SEM, n=6, Statistical analysis was performed with one-way ANOVA followed by Dunnett’s‘t’ test using Graph Pad Prism software. Significance at p<0.05*, p<0.01**, p<0.001***, Compared with control group.
Effects of ethanolic effect of Neolamarckia cadamba on pH in hydrated rats
The table displays the findings from the assessment of the alcoholic extract of Neolamarckia cadamba's diuretic activity. According to the results, when compared to the control, the alcoholic extract of Neolamarckia cadamba had a substantial diuretic effect by increasing urine production and excreting more salt, potassium, and chloride. It was discovered that the alcoholic extract of Neolamarckia cadamba had a dosage-dependent impact, meaning that the larger the dose, the greater the effect. The alcoholic extract of Neolamarckia cadamba was shown to have a considerable diuretic impact on urine output, salt, potassium, and chloride concentrations when compared to the common diuretic medication, frusemide. Urinary electrolyte concentration measurements showed that Neolamarckia cadamba alcoholic extract was successful in raising the levels of all three measured ions (Na+, K+, and Cl–). The test medication caused a dose-dependent rise in urine volume. Urine volume increased statistically significantly, particularly at higher dosage levels. When compared to normal control rats, the test drug's administration at both dosages had no discernible effect on the pH of the urine.
The current study's findings indicate that Neolamarckia cadamba's alcoholic extract has the potential to operate as an oral diuretic. The reported impact was likely intrinsic, based on the observed dose-dependent activity. Urine volume is dependent upon two elements. Two factors are the degree of tube re-absorption and the rate of glomerular filtration. Mechanisms such as increasing renal blood flow and the resulting rise in glomerular filtration rate might be responsible for the observed impact. Given that polyurea results from the suppression of antidiuretic hormone (ADH), it may also have an inhibitory impact on ADH production. Stimulating the release of endogenous natriuretic peptides, which encourages the secretion of water and salt, is another potential route. Since the amount of Na+in the urine increased significantly, such a method of action seems implausible. In conclusion, Neolamarckia cadamba's alcoholic extract has strong diuretic effects. Its bioactive components may act alone or in combination to provide the reported activity. Additional pharmacodynamic and phytochemical research is needed to identify the active ingredient causing the diuretic effect.
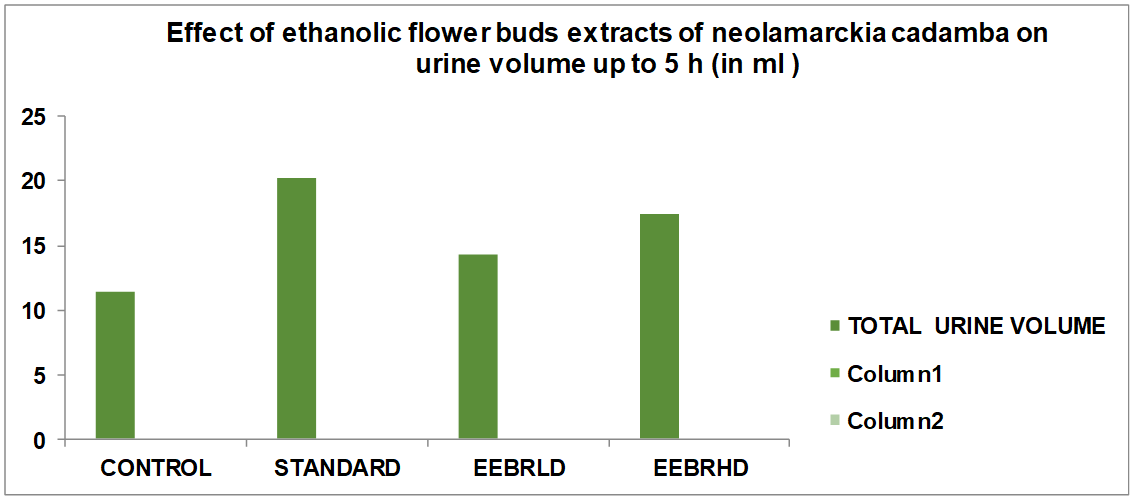
Fig. 1: Effect of Alcoholic extract of flower buds of Neolamarckia cadamba total urine volume (in 5 h)
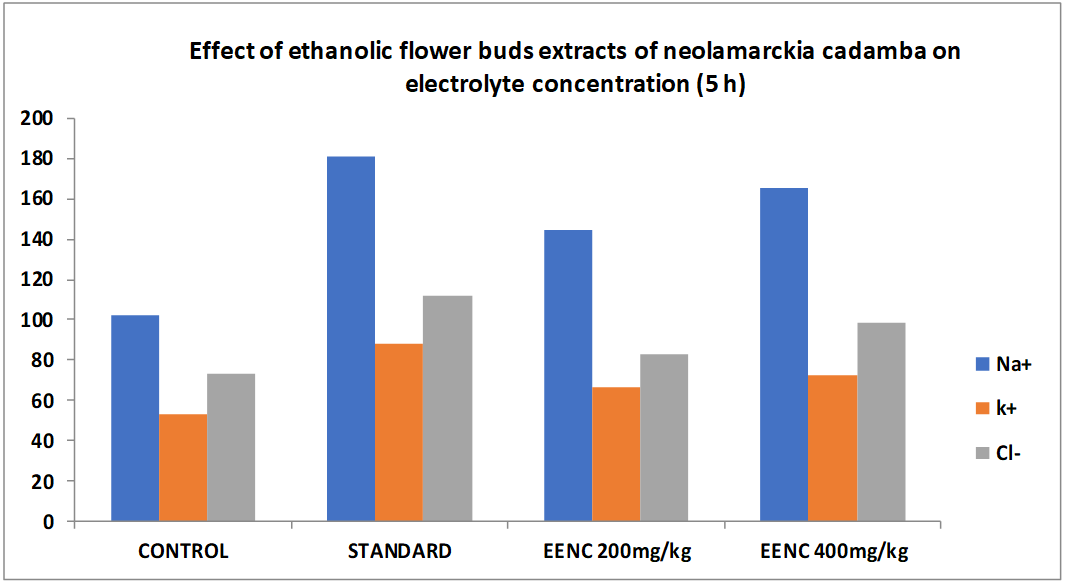
Fig. 2: Effect of ethanolic extracts of Neolamarckia cadamba on urinary electrolyte concentration (in 5 h)
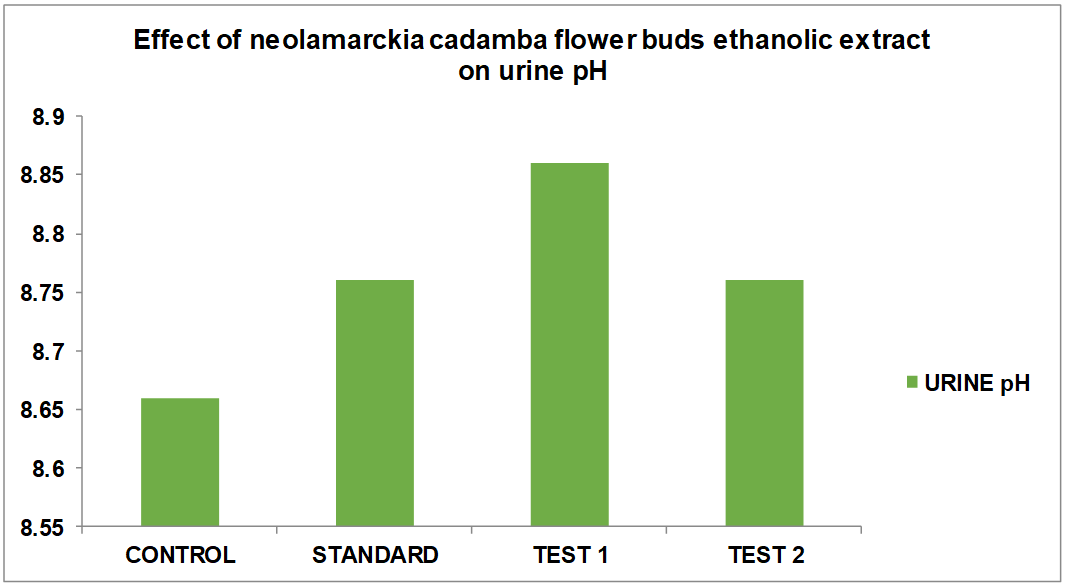
Fig. 3: Showing effects of Ethanolic effect of Neolamarckia cadamba on pH in hydrated rats
DISCUSSION
The present study highlights the pharmacological potential of Neolamarckia cadamba as an important medicinal plant with multiple therapeutic benefits. Reactive oxygen species and free radicals generated during metabolism often lead to oxidative stress, which causes membrane damage and DNA mutations. Studies revealed that Neolamarckia cadamba possesses strong antioxidant activity, primarily attributed to the phytoconstituents present in its leaves, such as flavonoids, terpenoids, and phenolic compounds, which were confirmed through UPLC-ESI-QTOF/MS profiling. Apart from its antioxidant properties, the plant demonstrated activity on the central nervous system and provided protective effects against convulsions in animal models [18].
Acute toxicity evaluation of ethanolic extracts indicated their safety at a wide range of doses, supporting their use in pharmacological studies at 200 and 400 mg/kg. Phytochemical screenings from different parts of the plant identified a wide variety of bioactive molecules, including flavonoids, terpenoids, essential oils, alkaloids, steroids, and glycosides, each contributing to different biological effects. Importantly, the leaves and stem bark contained constituents linked to both antioxidant and diuretic properties [19].
The diuretic activity of Neolamarckia cadamba was demonstrated effectively at doses of 200 and 400 mg/kg, with the 400 mg/kg dose showing remarkable results comparable to conventional diuretics like furosemide. The extract significantly increased urinary output and enhanced excretion of sodium, potassium, and chloride ions, suggesting a saluretic mechanism. These observations align with the ethnomedicinal use of the plant as a diuretic and support its role in managing conditions associated with edema, hypertension, and congestive heart failure. However, variations in activity among extracts may be linked to solvent polarity, phytochemical concentration, and extraction conditions.
CONCLUSION
The alcoholic extract of Neolamarckia cadamba demonstrated significant diuretic activity in a dose-dependent manner, with the strongest effect observed at 400 mg/kg. This effect was comparable to standard diuretics and is attributed to the presence of diverse phytochemicals such as flavonoids, terpenoids, and phenolic compounds. In addition, the plant also showed potent antioxidant and neuroprotective properties, further establishing its pharmacological importance. The absence of acute toxicity supports its safety profile for therapeutic use. Thus, Neolamarckia cadamba justifies its traditional use in treating disorders linked with oxidative stress and fluid retention, and it should be further explored for isolation of active compounds and development into safe and effective herbal formulations.
ACKNOWLEDGMENT
It’s our privilege to express the profound sense of gratitude and cordial thanks to our respected chairman Mr. Anil Chopra and Vice Chairperson Ms. Sangeeta Chopra, St. Soldier Educational Society, Jalandhar for providing the necessary facilities to complete this research work.
AUTHORS CONTRIBUTIONS
All the authors have contributed equally.
CONFLICT OF INTERESTS
Declared none
REFERENCES
Dehkordi FR, Kamkhah AF. Antihypertensive effect of Nigella sativa seed extract in patients with mild hypertension. Fundam Clin Pharmacol. 2008;22(4):447-52. doi: 10.1111/j.1472-8206.2008.00607.x, PMID 18705755.
Hobson RM, Maughan RJ. Hydration status and the diuretic action of a small dose of alcohol. Alcohol Alcohol. 2010;45(4):366-73. doi: 10.1093/alcalc/agq029, PMID 20497950.
Jimenez Ferrer E, Alarcon Alonso J, Aguilar Rojas A, Zamilpa A, Jimenez Ferrer C I, Tortoriello J. Diuretic effect of compounds from Hibiscus sabdariffa by modulation of the aldosterone activity. Planta Med. 2012;78(18):1893-8. doi: 10.1055/s-0032-1327864, PMID 23150077.
Kreydiyyeh SI, Usta J. Diuretic effect and mechanism of action of parsley. J Ethnopharmacol. 2002;79(3):353-7. doi: 10.1016/S0378-8741(01)00408-1, PMID 11849841.
Leong XF, Rais Mustafa MR, Jaarin K. Nigella sativa and its protective role in oxidative stress and hypertension. Evid Based Complement Alternat Med. 2013;2013:120732. doi: 10.1155/2013/120732, PMID 23533459.
Zeratsky K. Weight loss can natural diuretics reduce fluid retention and help with weight loss? Mayo Clinic Publications. 2015 Mar 4. Available from: http://www.mayoclinic.org/healthy-lifestyle/weight-loss/expert-answers/water-retention/faq-20058063. [Last accessed on 31 Jan 2026].
DaSilva EJ, Baydoun E, Badran A. Biotechnology and the developing world. Electron J Biotechnol. 2002;5(1):5-6. doi: 10.2225/vol5-issue1-fulltext-1.
Cravotto G, Boffa L, Genzini L, Garella D. Phytotherapeutics: an evaluation of the potential of 1000 plants. J Clin Pharm Ther. 2010;35(1):11-48. doi: 10.1111/j.1365-2710.2009.01096.x, PMID 20175810.
Gilani AH, Shah AJ, Zubair A, Khalid S, Kiani J, Ahmed A. Chemical composition and mechanisms underlying the spasmolytic and bronchodilatory properties of the essential oil of Nepeta cataria L. J Ethnopharmacol. 2009;121(3):405-11. doi: 10.1016/j.jep.2008.11.004, PMID 19041706.
Talalay P, Talalay P. The importance of using scientific principles in the development of medicinal agents from plants. Acad Med. 2001;76(3):238-47. doi: 10.1097/00001888-200103000-00010, PMID 11242573.
Elvin Lewis M. Should we be concerned about herbal remedies? J Ethnopharmacol. 2001;75(2-3):141-64. doi: 10.1016/S0378-8741(00)00394-9, PMID 11297844.
Muller JL. Love potions and the ointment of witches: historical aspects of the nightshade alkaloids. J Toxicol Clin Toxicol. 1998;36(6):617-27. doi: 10.3109/15563659809028060, PMID 9776969.
Dwevedi A, Sharma K, Sharma YK. Cadamba: a miraculous tree having enormous pharmacological implications. Pharmacogn Rev. 2015;9(18):107-13. doi: 10.4103/0973-7847.162110, PMID 26392707.
Banerji N, Siddhanta AK, Dutta NL. New saponin from stem bark of Anthocephalus cadamba Miq. Indian J Chem B. 1977;15:654-5.
Nadkarni KM. Indian materia medica. Bombay: Popular Press Prakashan; 2002. p. 118-9.
Kirtikar KR, Basu BD. Indian medicinal plants. 2nd ed. Allahabad: Lalit Mohan Basu Publishers; 1999. p. 1250-2.
Kapil A, Koul IB, Suri OP. Antihepatotoxic effects of chlorogenic acid from Anthocephalus cadamba. Phytother Res. 1995;9(3):189-93. doi: 10.1002/ptr.2650090307.
Nadkarni KM. Indian materia medica. 3rd ed. Bombay: Popular Press Prakashan; 1999. p. 118-9.
Sahu NP, Chakraborty B, Banerjee S. A new triterpenic acid from Anthocephalus cadamba Miq. Indian J Pharm Sci. 1974;12:284-6.