
Int J Curr Pharm Res, Vol 18, Issue 2, 43-47Original Article
EVALUATION OF DIURETIC ACTIVITY OF ETHANOLIC EXTRACT OF LEAVES OF PIPER BETEL IN ALBINO RATS
NISHANT YADAV*, AJEET PAL SINGH, AMAR PAL SINGH
Department of Pharmacology, St. Soldier Institute of Pharmacy, Lidhran Campus, Behind NIT (R. E. C.), Jalandhar –Amritsar by pass, NH-1, Jalandhar-144011, Punjab, India
*Corresponding author: Nishant Yadav; *Email: niishantyadav@gmail.com
Received: 16 Nov 2025, Revised and Accepted: 04 Jan 2026
ABSTRACT
Objective: To assess the diuretic capability of the alcoholic extract of Piper betel leaves, the current research was planned to make use of the Lipschitz technique on albino rats.
Methods: Four groups of albino rats were used for the experiment, and diuretic activity was assessed in metabolic cages. Group I served as the control and received normal saline (10 ml/kg, p. o. or 1 ml of 0.9% NaCl/100 g). Group II received the standard diuretic drug frusemide (10 mg/kg, p. o). Groups III and IV were treated with the alcoholic extract of Piper betel leaves at doses of 200 mg/kg and 400 mg/kg, respectively. Following drug or extract administration, all animals were hydrated with normal saline (15 ml/kg, p. o.) and placed individually in metabolic cages maintained at 25 °C±0.5 °C. Food and water were withheld during the 5 h study period. At the end of 5 h, urine was collected and analyzed for total volume, pH, and electrolyte content (sodium, potassium, and chloride ions).
Results: Compared to the control group, both doses of the Piper betel leaf extract significantly increased urine output and promoted greater excretion of sodium, potassium, and chloride ions. No remarkable differences in urinary pH were observed between the groups.
Conclusion: A single administration of frusemide as well as the alcoholic extract of Piper betel leaves enhanced urine production and electrolyte excretion. At the higher dose (400 mg/kg), the extract demonstrated a diuretic effect comparable to that of the standard drug frusemide.
Keywords: Piper betel, Leaves, Alcoholic extract, Hydrated rats, Diuretic activity, Metabolic cage
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i2.8023 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Diuretics are essential for removing excess water from the body, particularly in conditions like edema. They induce fluid loss by increasing urinary excretion and are widely used in managing hypertension, heart failure, and electrolyte imbalances. Acetazolamide inhibits carbonic anhydrase, affecting Na, HCO3, and water reabsorption. Loop diuretics like furosemide inhibit the Na-K-2Cl transporter, enhancing Na delivery to the distal tubule and promoting K secretion. Thiazides block the NaCl cotransporter, increasing Na delivery to collecting ducts and promoting K wasting. Potassium-sparing diuretics impair Na reabsorption at the cortical collecting duct. Mannitol disrupts tubular water reabsorption, while vasopressin receptor antagonists prevent free water reabsorption at the collecting ducts [1-3]. Diuretics, crucial for fluid loss from the body, primarily target sodium reabsorption in kidney tubules, alongside water, facilitating their elimination [4, 5]. This process, augmenting urine volume and urinary sodium excretion, contributes to reduced blood volume, aiding cardiovascular function. Plant-based diuretics harbour a diverse array of phytoconstituents like alkaloids, glycosides, tannins, phenolics, coumarins, and triterpenoids, which underpin their natural diuretic properties. Notable compounds from various plants, including phenolics from Tribulus terrestris and Urtica dioica, alkaloids from Aerva lanata and Foeniculum vulgare, triterpenes from Taraxacum officinale and Abutilon indicum, saponins from Asparagus racemosus and Tribulus terrestris, contribute to diuretic mechanisms [6, 7]. These natural diuretics, such as Lepidium sativum, Costus speciosus, and Phyllanthus fraternus, elevate urine output and urinary electrolyte concentration, while others like Spilanthes acmella and Tribulus alatus exhibit loop diuretic properties. However, caution is warranted with Rungia repens due to potential risk of hypokalemia from increased urinary potassium levels. Numerous Indian medicinal plants stand out for their potent diuretic effects, offering potential avenues for therapeutic exploration [8, 9]. For millennia, Indian medicine has appreciated the leaves of the Piper betel Linn plant. They were considered holy in antiquity and still have ceremonial importance in many Asian societies today. The ideal growing conditions for the plant are warm, humid areas. Piper betel has been used extensively in Indonesia as a natural treatment for ailments including vaginal and oral candidiasis. According to recent research, bacterial endophytes found in therapeutic plants such piper betel may provide an alternate supply of antibacterial chemicals, negating the requirement for direct plant extraction. With its heart-shaped leaves, this perennial, evergreen vine is sometimes referred to as a miraculous creation. Additionally, archaeological discoveries show that betel was used at Spirit Cave in Northwest Thailand as early as 5500–7000 BC, before organised agriculture was developed [10, 13].
Nutritional composition
The proximate analysis of Piper betel leaves revealed the presence of phytochemicals and macro and micronutrients [14, 15].
MATERIALS AND METHODS
Experimental animals
For this study, albino rats of either sex weighing 150–200 g were chosen. They were kept in polypropylene cages coated with husk bedding in the central animal facility of the St. Soldier Institute of Pharmacy in Jalandhar, Punjab, under carefully regulated light-dark cycles. Before the tests started, the animals were given a regular pellet meal, unrestricted access to water, and time to become used to the lab setting. Every technique was carried out in a semi-soundproof laboratory environment between the hours of 8:00 and 16:00. According to the guidelines of the Committee for the Purpose of Control and Supervision of Experimental Animals (CPCSEA), Ministry of Environment and Forests, Government of India, the study protocol was previously approved by the Institutional Animal Ethics Committee (IAEC) (Registration No. 2011/PO/Re/S/18/CPCSEA; registration date 01/05/2018). Every effort was made to reduce any pain or discomfort throughout the experimental procedures. IAEC/SSIP/2020/PR-010 is the IAEC/CPCSEA reference number that officially authorised the study protocol.
Chemicals and reagents
All of the biochemical reagents and chemicals utilized in this investigation were of analytical quality and were made fresh before use. All analytical-grade chemicals were purchased from S. D. Fine Chem. Ltd. in India and Sigma Chemical in the United States.
Piper betel leaves ethanolic extract was collected from the dealer of Shreedha Phyto extracts Jaipur-302019. The same group also provided a certification of the Plant’s identify and quality.
Phytochemical tests
The phytochemical test are performed as for the preliminary test of plants like Molisch’s Test (Carbohydrates), Barfoed’s Test (Monosaccharides),Fehling’s Test (Reducing Sugars),Fehling’s Test (Combined Reducing Sugars), Test for Tannins, Borntrager’s Test (Anthraquinones), Liebermann–Burchard Test (Steroids),Test for Terpenoids, Test for Saponins, Shinoda’s Test (Flavonoids),Ferric Chloride Test (Flavonoids),Lead Ethanoate Test (Flavonoids),Hydroxide Test (Flavonoids), Test for Alkaloids, Test for Soluble Starch [16-19].
Animals
The research will utilise adult albino rats of either sex that weigh between 150 and 200 g. The animals will be housed in a controlled laboratory environment with a 12 h light and 12 h dark cycle, at a temperature of 25±2 °C. Throughout the trial, they will have unfettered access to normal food and water.
Method
EBR will be administered orally by gavage to five rats who had fasted over the entire night at varying doses (5, 50, 300, and 1000 mg/kg). In order to record any behavioural abnormalities, including tremors, convulsions, salivation, diarrhoea, lethargy, sleep, or coma, the animals will be thoroughly observed for the first two hours after injection and again after twenty-four hours. Up to 14 d will be spent under observation in order to look for any indications of toxicity or death. The remaining rats will be rehabilitated at the conclusion of the 14 d period and then used for further experimental research.
Rat as a model for experimentation [20-23]
Because of their strong genetic, physiological, and anatomical similarities to humans, rats are often employed as experimental models. They are especially useful for researching human illnesses since, in reality, more than 90% of the rat genome and over 95% of the mouse genome are similar to those of humans. Rats and mice, in particular, are widely used in preclinical screening for novel therapeutic drugs, mental diseases including anxiety and depression, and central nervous system studies. Because of their neurological significance and genetic resemblance, scientists may study mechanisms-especially those related to stress-that are unethical to study in people. The cost-effectiveness, shorter reproductive cycles, simplicity of handling, and need for reduced pharmacological dosages in relation to body weight are other advantages of employing mice and rats as models. These characteristics make them excellent and controllable study subjects. Furthermore, rodent models are essential for diuretic research and are used to establish treatment plans for diseases including congestive heart failure, nephritis, hypertension, and kidney problems.
Experimental parameters
Diuretic medications, which encourage the body to rid itself of extra water, can treat a number of edema-related illnesses, such as hypertension, nephritis, congestive heart failure, premenstrual syndrome, and pregnancy-related toxaemia. However, adverse effects include electrolyte imbalances and metabolic abnormalities are often associated with synthetic diuretics, such as thiazides and loop diuretics, which are frequently administered. It's interesting to note that many diuretics have plant-based roots, and many of the plants used in Ayurveda are known to have diuretic properties. Cissampelos pareira, Barbara vulgaris, Bacopa monnieri, and Abelmoschus esculentus are a few examples.
Body weight analysis
Weekly body weight analysis will be documented throughout the research period.
Statistical analysis
The T-test or one-way ANOVA (Graph Pad Prism version 5.00 software) will be used to examine the collected data, and an appropriate post-test will be conducted thereafter.
Table 1: Preliminary tests for Piper betel linn
| Phytochemical group | Test/Method applied | Observation (+/-) |
| Alkaloids | Mayer’s reagent | – |
| Dragendorff’s reagent | – | |
| Hager’s reagent | – | |
| Wagner’s reagent | – | |
| Purine group | Murexide test | – |
| Carbohydrates | Molisch’s test | + |
| Fehling’s test | + | |
| Benedict’s test | + | |
| Glycosides | Anthraquinone glycosides | – |
| Borntrager’s test | – | |
| Modified Borntrager’s test | – | |
| Keller–Killiani test (Cardiac) | – | |
| Raymond test (Cardiac) | – | |
| Legal test (Cardiac) | – | |
| Cyanogenic glycosides | – | |
| Coumarin glycosides | – | |
| Sterols | Salkowski test | + |
| Liebermann–Burchard’s test | + | |
| Saponins | Froth test | + |
| Tannins | Ferric chloride test | + |
| Goldbeater’s skin test | + | |
| Proteins and Amino acids | Millon’s test | + |
| Biuret test | + | |
| Ninhydrin test | + | |
| Mucilage | Ruthenium red/swelling test | + |
| Terpenoids | Color reaction test | + |
| Flavonoids | Shinoda test | + |
| Alkali reagent test | + | |
| Acid reagent test | + | |
| Zinc/HCl reduction test | + | |
| Volatile oils | Odor test/staining test | + |
RESULTS
Acute oral toxicity study
The median lethal dosage (LD₅₀) of EPB was assessed using OECD guideline 425. Five overnight-fasted rats received oral doses of the extract at 5, 50, 300, and 1000 mg/kg. Food was withheld for 4 h after one rat received the test dosage. After 24 h, the animal was evaluated daily for 14 d for clinical symptoms of toxicity, including skin, hair, mucous membranes, eyes, respiratory and circulatory functions, motor activity, behaviour, and death. No acute toxicity or aberrant behaviour was observed. After that, the other four rats received the extract under identical settings and were examined for two weeks. The EPB LD₅₀ was determined from the findings.
The lipschitz test and estimation of urine electrolytes
The diuretic effect of ethanolic extract of Piper betel leaves was studied in albino rats using the Lipschitz test. Preliminary screening was done by giving rats distilled water (1 ml/100 g), and only those excreting at least 40% of the dose volume within 6 h were selected. Four groups of six rats each were used: control (saline), standard (frusemide 2 mg/kg), and two test groups treated with the extract at 200 and 400 mg/kg. After drug administration, rats were placed in metabolic cages and urine was collected at different intervals up to 24 h, with total output measured.
The ethanolic extract significantly increased urine production in a dose-related manner. Analysis of electrolytes showed higher excretion of sodium, potassium, and chloride. Since sodium balance affects blood volume and pressure, and potassium is essential for heart and muscle function, excessive potassium loss may cause hypokalemia. This suggests the extract has notable diuretic potential, though its potassium-sparing effect remains to be studied. The activity may be linked to phytoconstituents like flavonoids and glycosides
Estimation of urinary electrolytes/volume/pH
The biochemical kits' user instructions (Roche, Roche Diagnostics Pvt. Ltd, Gurgaon, Haryana) explained the Ion Selective Electrode technique for determining urine electrolytes (sodium, potassium, and chloride). Using standard pH paper, urine volume, electrolytes (Na+, K+, and Cl–) and pH were determined. To determine diuretic action, urine volume was assessed.
Statistical analysis
Mean+SEM (n=6) were used to represent experimental findings. Statistical significance and P-values (probability) were established at ≤0.05 for the study's unpaired data analysis using Student's t test. The significance level was assessed. The alcoholic extract of Piper betel leaves exhibited phytoconstituents as phenolic compound, proteins, tannins, glycosides, carbohydrates, starch, vitamins, and minerals. After 14 d, all acute toxicity study animals survived. The extract was safe up to 1000 mg/kg. No notable behavioral changes were noticed throughout the trial.
Biochemical parameters
The biochemical kits' user instructions (Roche, Roche Diagnostics Pvt. Ltd, Gurgaon, Haryana) explained the Ion Selective Electrode technique for determining urine electrolytes (sodium, potassium, and chloride). Using standard pH paper, urine volume, electrolytes (Na+, K+, and Cl–) and pH were determined and assessed was shown in fig. 1 to 5.
The alcoholic extract of Piper betel leaves showed a marked diuretic effect, as indicated by increased urine output and higher excretion of sodium, potassium, and chloride compared to controls. The activity was dose-dependent, with the higher dose producing stronger effects, and results were comparable to the standard diuretic frusemide. Urine analysis confirmed significant elevation of electrolyte levels without major changes in pH.
The findings indicate that the extract acts as an effective oral diuretic, likely through mechanisms such as enhanced renal blood flow, increased glomerular filtration, or possible inhibition of antidiuretic hormone. Increased sodium excretion further supports its natriuretic action. The activity may be attributed to bioactive compounds like flavonoids or glycosides present in the extract.
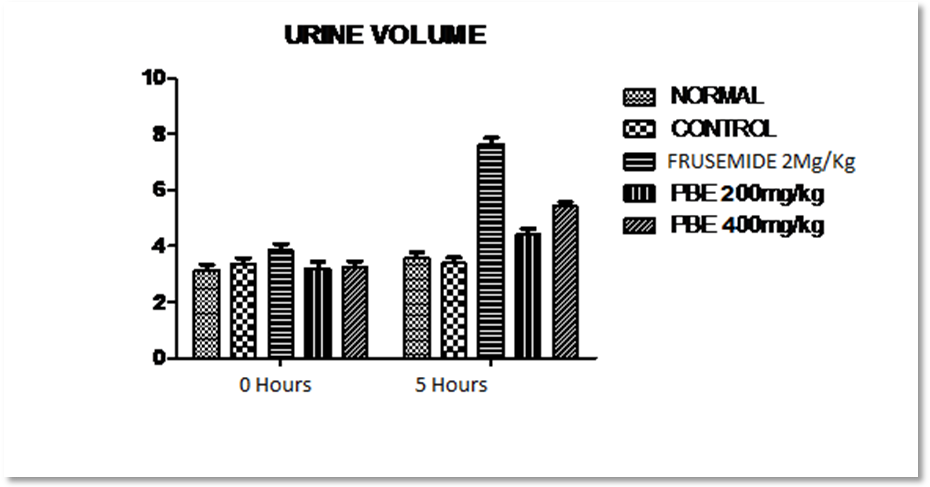
Fig. 1: Showing effect of frusemide and piper betel extract on urine volume
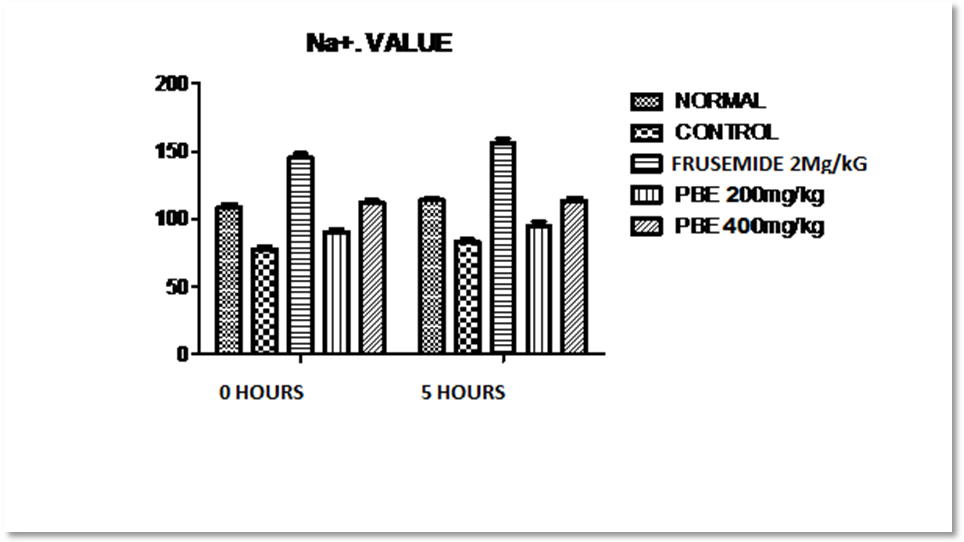
Fig. 2: Showing effect of frusemide and piper betel extract on sodium content NA+ in urine
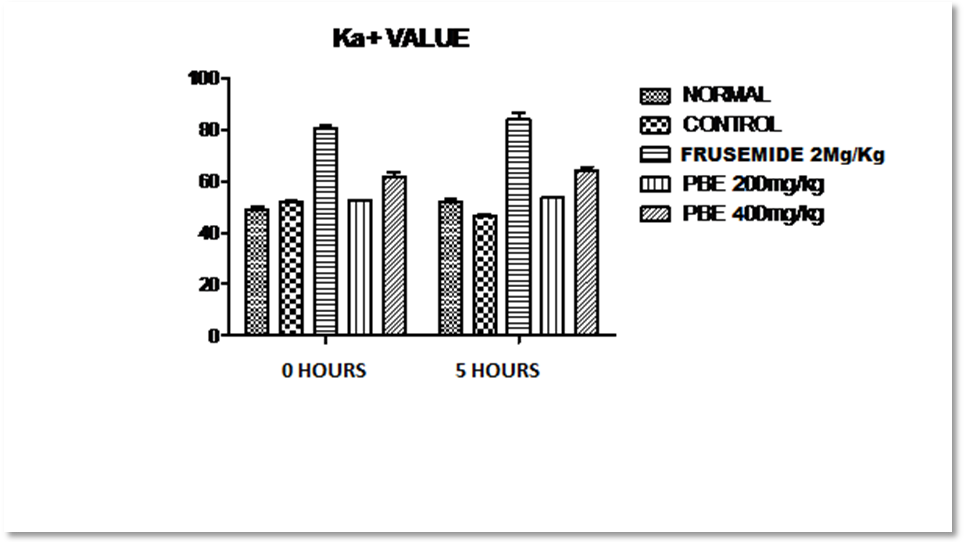
Fig. 3: Showing effect of frusemide and piper betel extract on potassium content in urine
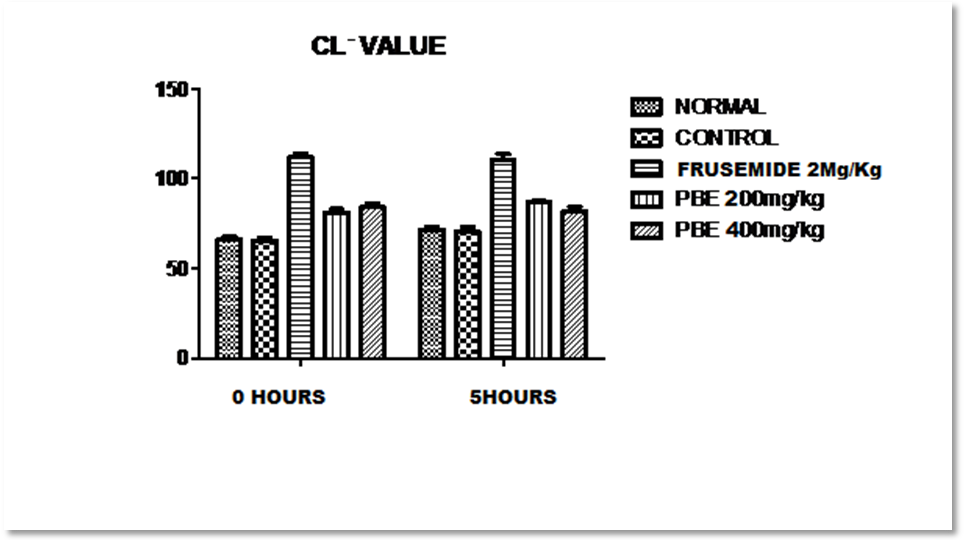
Fig. 4: Showing effect of frusemide and piper betel extract on chloride content in urine
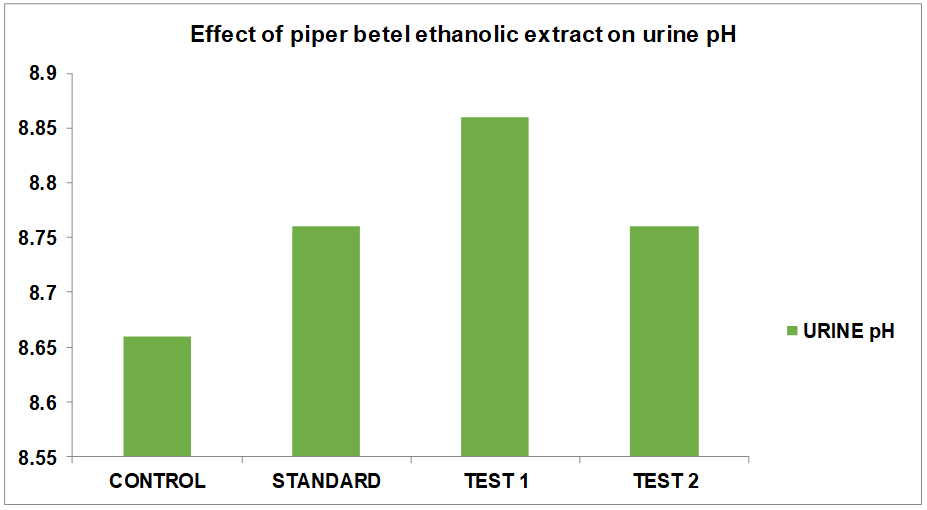
Fig. 5: Graph showing effects of Ethanolic effect of Piper betel on pH in hydrated rats
DISCUSSION
The ethanolic extract of Piper betel leaves demonstrated significant diuretic activity in albino rats. The extract, particularly at the higher dose (400 mg/kg), consistently increased urine output and promoted enhanced excretion of sodium, potassium, and chloride ions, showing effects similar to frusemide, the standard pharmaceutical diuretic. These findings suggest that the diuretic effect may be attributed to bioactive compounds like flavonoids and glycosides present in the Piper betel extract. The activity observed appears dose-dependent and could be mediated by mechanisms such as increased renal blood flow, greater glomerular filtration, or possibly inhibition of antidiuretic hormone function. No significant changes in urinary pH were noted, and acute toxicity studies established the extract as safe up to 1000 mg/kg with no observable adverse effects.
CONCLUSION
A single administration of the ethanolic extract of Piper betel leaves produces notable diuretic effects in rats, comparable to the standard drug frusemide at higher doses. The extract enhances both urine production and urinary electrolyte excretion, indicating its potential as a natural diuretic agent. These properties support further investigation into Piper betel leaves as a source for therapeutic diuretic compounds, especially considering the safety observed at experimental doses.
ACKNOWLEDGMENT
It’s our privilege to express the profound sense of gratitude and cordial thanks to our respected chairman Mr. Anil Chopra and Vice Chairperson Ms. Sangeeta Chopra, St. Soldier Educational Society, Jalandhar for providing the necessary facilities to complete this research work.
AUTHORS CONTRIBUTIONS
All authors have contributed equally
CONFLICT OF INTERESTS
Declared none
REFERENCES
Roush GC, Kaur R, Ernst ME. Diuretics: a review and update. J Cardiovasc Pharmacol Ther. 2014;19(1):5-13. doi: 10.1177/1074248413497257, PMID 24243991.
Klabunde RE. Cardiovascular pharmacology concepts. Available from: https://www.cvpharmacology.com/diuretic/diuretics. [Last accessed 17 Nov 2017].
Klabunde RE. Cardiovascular pharmacology concepts. Available from: https://www.cvpharmacology.com/diuretic/diuretics. [Last accessed on 07 Feb 2026].
Wilcox CS, Testani JM, Pitt B. Pathophysiology of diuretic resistance and its implications for the management of chronic heart failure. Hypertension. 2020;76(4):1045-54. doi: 10.1161/hypertensionaha.120.15205, PMID 32829662.
Ellison DH. Clinical pharmacology in diuretic use. Clin J Am Soc Nephrol. 2019;14(8):1248-57. doi: 10.2215/CJN.09630818, PMID 30936153.
Mishra G. Ethnobotany and diuretic activity of some selected Indian medicinal plants: a scientific review. The Pharma Innovation Journal. 2013;2(3):109-21.
Chandrasekar R, Sivagami B. Indian medicinal plants with diuretic activity. Indo Am J Pharm Res. 2017;7(1):7360.
Shah SK, Garg G, Jhade D, Patel N. Piper betle: phytochemical pharmacological and nutritional value in health management. J Harmonized Res. 2016;38(2):181-9.
Patra S, Das MT, Dey SK. A review on Piper betle L. J Med Plants Stud. 2016;4(6):185-92.
Silalahi M. Piper betle L. piperaceae local names. Universitas Kristen Indonesia; 2020.
Shah SK, Garg G, Jhade D, Patel N. Piper betle: phytochemical pharmacological and nutritional value in health management. J Harmonized Res. 2016;38(2):181-9.
Shah SK, Garg G, Jhade D, Patel N. Piper betle: phytochemical pharmacological and nutritional value in health management. J Harmonized Res. 2016;38(2):181-9.
Pakrashi SC, Pakrashi A. Ginger: a versatile healing herb. New Delhi: Vedams eBooks (P) Ltd; 2003.
Patel NM, Jain DD, Suryawanshi HP, Pawar SP. Phytopharmacological study of Piper betle leaf. SJMPS. 2019;5(11):964-71. doi: 10.36348/sjmps.2019.v05i11.008.
Verma S, Gupta ML, Dutta A, Sankhwar S, Shukla SK, Flora SJ. Modulation of ionizing radiation induced oxidative imbalance by semi-fractionated extract of Piper betle: an in vitro and in vivo assessment. Oxid Med Cell Longev. 2010;3(1):44-52. doi: 10.4161/oxim.3.1.10349, PMID 20716927.
Guha P. Betel leaf: the neglected green gold of India. J Hum Ecol. 2006;19(2):87-93. doi: 10.1080/09709274.2006.11905861.
Pradhan D, Suri KA, Pradhan DK, Biswasroy P. Golden heart of the nature: Piper betle L. J Pharmacogn Phytochem. 2013;1(6):147-67.
Bajpai V, Sharma D, Kumar B, Madhusudanan KP. Profiling of Piper betle Linn. cultivars by direct analysis in real time mass spectrometric technique. Biomed Chromatogr. 2010;24(12):1283-6. doi: 10.1002/bmc.1437, PMID 21077247.
Pradhan D, Suri KA, Pradhan DK, Biswasroy P. Golden heart of the nature: Piper betle L. J Pharmacogn Phytochem. 2013;1(6):147-61.
Pradhan D, Suri KA, Pradhan DK, Biswasroy P. Golden heart of the nature: Piper betle L. J Pharmacogn Phytochem. 2013;1(6):1-6.
Patel NM, Jain DD, Suryawanshi HP, Pawar SP. Phytopharmacological study of Piper betle leaf. SJMPS. 2019;5(11):964-71. doi: 10.36348/sjmps.2019.v05i11.008.
Betalvine KN. Piper betle L. cultivation: a unique case of plant establishment under anthropogenically regulated microclimatic conditions. Indian J Hist Sci. 1999;34(1):19-32.
Chowdhury I, Amin R, Binzaid S. Optimal control on environments for improving the piper betle (paan). Life Sciences Leaflets. 2011;60(17):5-15.