
Int J Curr Pharm Res, Vol 18, Issue 2, 9-15Review Article
RAFT-FORMING DRUG DELIVERY SYSTEM: A FLOATATION-BASED STRATEGY FOR GASTRORETENTION
GOMATHI J.*, SAMA SULAIKA M.
Department of Pharmaceutics, C. L. Baid Metha College of Pharmacy, Affiliated to the Tamil Nadu Dr. M. G. R. Medical University, Chennai-600097, Tamil Nadu, India
*Corresponding author: Gomathi J.; *Email: gomathidinesh1@gmail.com
Received: 17 Nov 2025, Revised and Accepted: 07 Jan 2026
ABSTRACT
Gastroretentive drug delivery systems (GRDDS) have emerged as a promising strategy to enhance the therapeutic efficacy and bioavailability of orally administered drugs with a narrow absorption window or that are poorly soluble in higher pH environments. Among various approaches, the raft-forming system has garnered significant attention for its simplicity, efficacy, and patient compliance. These systems form a low-density, gel-like "raft" upon contact with gastric fluids, allowing the formulation to float and remain in the stomach for an extended duration. This enhances localized drug action and prevents gastric reflux by acting as a barrier between the stomach and esophagus, particularly benefiting conditions such as GERD, heartburn, and esophagitis. The design involves gel-forming agents like alginic acid and gas-generating components such as sodium bicarbonate, which facilitate in situ gelation and buoyancy. Despite formulation challenges related to pH sensitivity, polymer stability, and gastric motility variations, raft-forming systems offer several advantages, including rapid onset, prolonged activity, ease of administration, and reduced dosing frequency. This review highlights the anatomy and physiology relevant to gastric retention, mechanisms of raft formation, formulation strategies, and potential candidates, emphasizing the system’s growing role in advanced oral drug delivery.
Keywords: Gastroretentive drug delivery system (GRDDS), Raft-forming system, In situ gelation, Alginic aci, Sodium bicarbonate
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i2.8029 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
The primary objective of designing any drug delivery system is to administer the drug directly to the target site, ensuring and maintaining the desired drug concentration in the body. In recent years, there has been a growing focus on developing controlled and sustained drug delivery systems. Extensive research has been conducted to design such systems to enhance therapeutic efficacy while minimizing side effects [1]. Among the various drug delivery methods, the oral route has emerged as the most preferred, effective, and promising option. This is due to its numerous advantages, including cost-effectiveness, ease of administration, formulation flexibility, prolonged drug release, and improved bioavailability, collectively enhancing patient compliance. Currently, approximately 50% of marketed drug delivery systems utilize the oral route [2].
However, this approach poses several physiological challenges, including an unpredictable gastric emptying rate that differs among individuals, a short gastrointestinal transit time (8–12 h), and the presence of an absorption window in the upper small intestine for many drugs. These limitations have led researchers to develop drug delivery systems that can remain in the stomach for an extended and controlled duration. Gastroretentive dosage forms are designed to stay in the gastric region for longer, thereby significantly enhancing drug retention time (GRT) [3].
Gastroretentive drug delivery systems (GRDDS) aim to enhance a medication's bioavailability by prolonging its retention in the stomach. By steadily releasing the drug over an extended period before it reaches the absorption site, GRDDS facilitates controlled drug delivery for medications with an absorption window, ensuring optimal bioavailability [4].
Drugs suitable for gastroretentive systems are those that exert local effects in the stomach, are predominantly absorbed there, have limited solubility in alkaline pH, are rapidly absorbed in the gastrointestinal tract, or degrade in the colon [5].
Various approaches are employed to develop effective stomach-specific or gastroretentive drug delivery systems (DDS), such as hydro dynamically balanced systems (HBS) or floating drug delivery systems, low-density systems [6], raft forming system, mucoadhesive or bio adhesive systems, high-density systems, super porous hydro-gels, and magnetic systems [7].
Among the various systems, the raft-forming system is the most commonly used, as it is a feasible and preferred approach for achieving prolonged and predictable drug delivery in the gastrointestinal (GI) tract. This system allows the sustained release of a drug, ensuring relatively stable plasma levels [8]. At room temperature, these hydrogels remain in a liquid state. However, upon contact with body fluids or a shift in pH, they undergo gelation, forming a viscous layer known as a Raft. This Raft floats on gastric fluid due to its low bulk density, which results from CO₂ generation. The system already contains gel-forming alkaline bicarbonates or carbonates that release CO₂, and the gel formation further reduces the system's density, allowing it to remain buoyant on the gastric fluid's surface [9]. Antacids and prescription drugs are administered with this technique to treat gastrointestinal disorders and infections [10].
The primary aim of this system is to reduce dosing frequency or enhance drug efficacy by localizing it at the site of action, lowering the required dose, or ensuring uniform drug delivery. Additionally, the raft-forming system offers advantages such as simple manufacturing, improved patient compliance, and ease of administration [11].
Anatomy and physiology of the stomach
Structure and function of the stomach
 The gastrointestinal tract can be divided into three main regions, namely
The gastrointestinal tract can be divided into three main regions, namely
Stomach
Small intestine-Duodenum, Jejunum and Ileum
Large intestine [12].
The gastrointestinal (GI) tract is a continuous, approximately nine-meter-long tube that stretches from the mouth to the anus. It includes the throat (pharynx), oesophagus, stomach, small intestine (comprising the duodenum, jejunum, and ileum), and large intestine (comprising the cecum, appendix, colon, and rectum) [13].
The stomach functions as an organ for both storage and mixing. The antrum region specifically handles the mixing and grinding of gastric contents [14].
Anatomically, the stomach is divided into three parts: The fundus, the body, and the antrum (pylorus) [15].
The proximal part of the stomach, consisting of the fundus and body, serves as a reservoir for undigested material, while the antrum functions as a pump for gastric emptying through propulsive actions. The antrum is also the primary site for mixing. Due to its limited surface area, minimal absorption occurs in the stomach. Additionally, it acts as a barrier to the delivery of drugs to the small intestine (fig. 1) [16].
Gastric motility and emptying
Gastric emptying takes place in both fasting and fed states. During fasting, a cyclic series of interdigestive electrical events occurs, passing through the stomach and intestine every 2 to 3 h. This electrical activity is known as the interdigestive myoelectric cycle or migrating myoelectric complex (MMC). The MMC is further categorized into four distinct phases (fig. 2) [17].
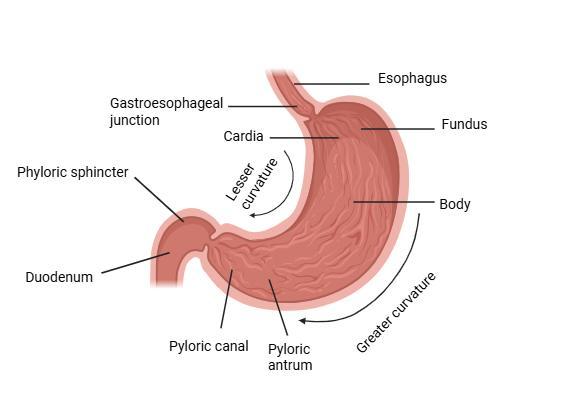
Fig. 1: Anatomy of the stomach, created in https://BioRender.com
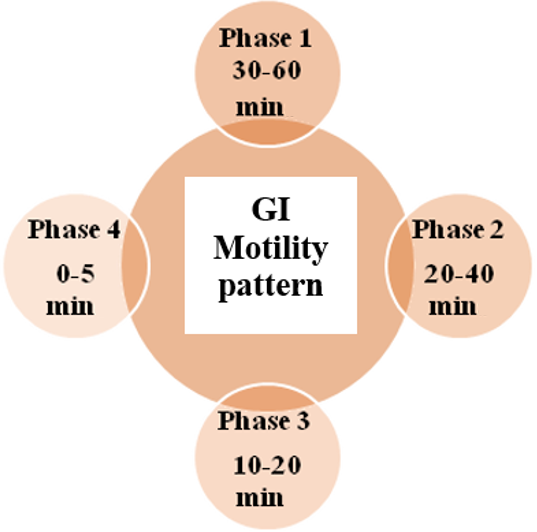
Fig. 2: GI Motility pattern
Phase I (Basal Phase)
This quiescent phase lasts between 40 to 60 min and is marked by minimal or absent secretory, electrical, and contractile activity.
Phase II (Preburst phase)
This phase lasts for 40 to 60 min; this phase features intermittent action potentials and contractions. As it progresses, both the intensity and frequency of these activities gradually increase [18].
Phase III (Housekeeper waves)
Known as the housekeeper phase, this stage is marked by strong, high-amplitude contractions that promote maximum pyloric opening and effective clearance of stomach contents. It typically lasts 10 to 20 min, occurring at a frequency of 4 to 5 contractions per minute [19]
Phase IV (Transitional phase)
Acting as an intermediate stage between Phase III and the following Phase I, this phase is short in duration, typically lasting less than 5 min. [20]
Gastro-reentive drug delivery system
Overview of GRDDS
Gastroretentive drug delivery systems (GRDDS) are designed to remain in the stomach for extended periods, thereby increasing the duration a drug stays in the gastric environment. This prolonged retention enhances the solubility of drugs that are otherwise poorly soluble in higher pH conditions, such as those found in the intestines. As a result, drug wastage is minimized, and bioavailability is improved. Additionally, GRDDS can be used for targeted drug delivery to the stomach and upper part of the small intestine, offering new therapeutic possibilities and significant advantages for patients.
Controlled gastric retention of solid dosage forms can be achieved through various mechanisms such as the use of pharmacological agents that delay gastric emptying, as well as systems that utilize mucoadhesion, flotation, sedimentation (via high-density formulations), swelling or expansion, and shape modification. Recent studies have demonstrated the success of these approaches in improving the bioavailability of certain medications, especially those with poor absorption characteristics in non-gastric environments.
The main objective in developing oral sustained-release formulations, particularly those intended for once-daily administration, is to prolong the residence time of the drug in the stomach or upper small intestine and to maintain therapeutic drug levels for 24 h. However, factors such as age, gender, ethnicity, dietary habits, and medical conditions can significantly influence the performance of controlled-release systems. Therefore, it is essential to design GRDDS that offer extended gastrointestinal residence time and consistent drug release, regardless of these patient-specific variables.
Ultimately, the goal of GRDDS is to enable site-specific drug release in the upper gastrointestinal tract for both local and systemic therapeutic effects by maximizing gastric retention time [21].
Advantages of GRDDS
Gastroretentive systems provide several therapeutic and pharmacological benefits, particularly for drugs that are absorbed predominantly in the stomach or upper small intestine. Medications such as antacids and ferrous salts show improved bioavailability when retained in the gastric region for extended periods. Furthermore, GRDDS are especially useful for drugs that exert localized effects in the stomach, including antacids used for gastric acidity and reflux disorders.
By prolonging gastric residence time, GRDDS enhance drug absorption even in conditions where rapid intestinal transit would otherwise reduce therapeutic outcomes, such as in certain diarrheal disorders. Another important advantage is the ability of these systems to improve patient compliance by reducing the frequency of dosing. Controlled release from GRDDS also stabilizes plasma drug concentrations, minimizes fluctuations, and maintains the desired therapeutic level over extended durations. Drugs with a short biological half-life can particularly benefit from this approach, as sustained release ensures prolonged activity without the need for frequent administration. Additionally, GRDDS offer a suitable platform for delivering drugs that are unstable in the alkaline conditions of the intestine, thereby preserving their therapeutic potential [22-24].
Disadvantages of GRDDS
Despite their advantages, GRDDS are associated with certain limitations. Their effectiveness is highly dependent on the presence of gastric fluids, as a sufficient volume is required to enable flotation and drug release. In patients who remain in a supine position after administration, such as those who take medication before bedtime, floating systems may be displaced by gastric motility, reducing their retention time. Moreover, these systems are unsuitable for drugs that are unstable or poorly soluble in the acidic environment of the stomach, or for compounds that cause gastric irritation.
Another challenge is the variability in gastric physiology, including differences in pH, motility, and the presence of food, all of which can significantly influence drug release and buoyancy. Since these physiological conditions are not consistent across patients, predicting the performance of GRDDS can be difficult. For this reason, the selection of suitable drug candidates and optimization of formulation parameters are critical for ensuring the success of these systems [25, 26].
Approaches to gastric retention
To enhance the efficiency of oral drug delivery, especially for drugs with a narrow absorption window or those that are poorly absorbed in the lower gastrointestinal tract, various gastric retention strategies have been developed. These approaches are designed to prolong the residence time of a dosage form in the stomach, ensuring sustained drug release and improved bioavailability. Depending on the mechanism of action, gastric retention systems are classified below:
Non floating drug delivery systems high density system
Superporous hydrogels
Mucoadhesive or bioadhesive system
Ion exchange system
Expandable system
Magnetic system
Floating drug delivery systems
Effervescent system
Gas generating systems
Volatile liquid containing system
Non effervescent system
Single layer\bilayer floating tablet
Alginate beads
Hollow microsperes
Raft forming system
Among these technologies, the raft-forming technique is the most widely used, as it is the most practical and preferred method for achieving continuous and prolonged drug delivery in the gastrointestinal tract. By enabling sustained release of drug molecules, this approach helps maintain a relatively stable plasma concentration profile [2].
Raft forming system
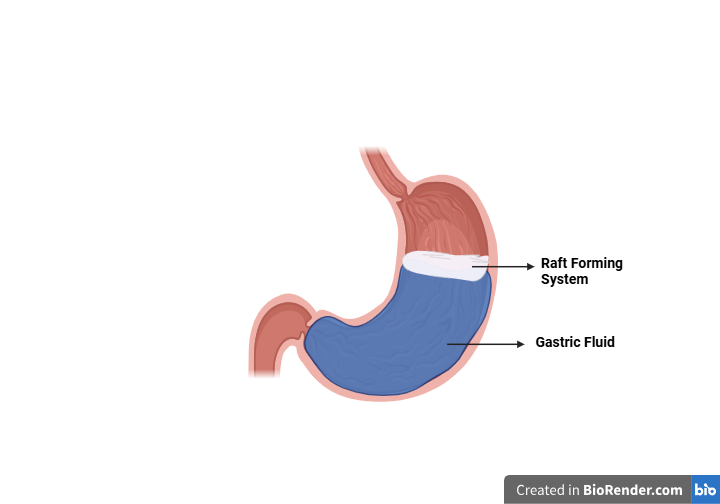
Fig. 3: Schematic illustration of the barrier formed by the raft forming system, created in https://BioRender.com
Several strategies have been developed to prolong gastric retention, with raft-forming systems emerging as a promising approach for sustained drug delivery, especially in treating gastrointestinal conditions like GERD.
Mechanism of raft formation
The mechanism involved in the raft formation includes the formation of a viscous, cohesive gel upon contact with gastric fluids. As the liquid swells, it forms a continuous floating layer-referred to as a "raft"-due to the low bulk density created by carbon dioxide release. These systems typically contain a gel-forming agent such as alginic acid, sodium bicarbonate, and an acid neutralizer. When exposed to gastric fluids, they produce a floating sodium alginate gel. The antacid ingredients help maintain a relatively neutral pH barrier. Calcium carbonate serves both as an antacid and a raft-strengthening agent by releasing calcium ions that interact with alginate to form an insoluble gel. Various polymers, especially different polysaccharides, have been used in various research works. Alginic acid, alginates, and pectin are the most widely used raft-forming agents. Other polysaccharides are also being used, which include guar gum, locust bean gum, carrageenan, pectin, and isapgol [27].
The raft thus formed floats on the gastric fluids and prevents the reflux of the gastric contents (i. e., gastric acid) into the esophagus by acting as a barrier between the stomach and esophagus [28]. While floating on the stomach contents, the medication is gradually released at a controlled rate. Once the drug has been fully released, any remaining system is cleared from the stomach. This approach helps to better regulate fluctuations in plasma drug concentration and enhances gastric retention time [29].
Raft-forming anti-reflux systems are widely used to treat stomach acid-related conditions such as GERD, heartburn, and esophagitis [30]. These systems form a thick, gel-like neutral layer that floats on the surface of the stomach contents. This floating barrier acts like a raft, helping to maintain the position of the lower esophageal sphincter (LES) and preventing acidic contents from flowing back into the esophagus. This mechanism provides relief from GERD symptoms. Unlike traditional antacids and other GERD treatments, raft-forming preparations offer symptom relief by forming a physical floating barrier rather than simply neutralizing stomach acid [31].
As a result, this leads to the retention of the dosage form and extends its residence time in the stomach, thereby prolonging the duration over which the drug is delivered to the gastrointestinal tract [32].
Raft-forming anti-reflux preparations are a new and emerging approach designed to combat severe acidity, peptic ulcers, and gastritis. These formulations are mainly used to treat gastric acid-related conditions, particularly GERD, heartburn, and esophagitis [33].
Design criteria
The design of a raft-forming system is influenced by several factors, including the physicochemical properties of the drug, the specific medical condition being treated, the target patient population, and market considerations. Key physicochemical factors include molecular weight, lipophilicity, and molecular charge. Anatomical and physiological factors, such as membrane transport and tissue fluid pH, also play a role. Additionally, formulation-related parameters like pH, gelation temperature, viscosity, osmolarity, and spreadability are critical in determining the effectiveness of the system [34].
To achieve effective gastric retention, the dosage form should be capable of providing sustained and controlled drug release while withstanding the mechanical stress of peristaltic movements and the continuous contractions, grinding, and churning actions within the stomach. Additionally, the system must possess a specific gravity lower than that of gastric fluids (1.004–1.01 g/cm³) to ensure buoyancy, remain in the stomach for an extended duration, and promote improved patient compliance. Moreover, it should be designed for ease of administration to enhance convenience for the patient [35].
Types of raft formation mechanisms
Raft-forming drug delivery systems represent a significant advancement in oral drug administration. These formulations are liquid at room temperature but transform into a gel upon contact with body fluids or changes in pH. This transformation is driven by their unique temperature-sensitive and cation-triggered gelation properties.
The gelation process begins with the formation of double helical junction zones, which then aggregate to create a three-dimensional network. This structure is stabilized cation interactions and hydrogen bonding through [36].
Several strategies are employed to initiate raft formation within the gastrointestinal tract, depending on the specific mechanism involved.
Raft formation based on physical mechanism
Swelling
Gel formation takes place when a liquid effervescent system comes into contact with gastric fluids. In situ gelation occurs as materials absorb water from the surrounding environment, swell, and expand to occupy the targeted site. This polymer swelling, caused by water absorption, leads to the formation of the gel matrix.
A notable example is Myverol 18-99 (glycerol mono-oleate), a biodegradable polar lipid that swells in water to form lyotropic liquid crystalline phase structures. It exhibits bioadhesive properties and can be enzymatically degraded in vivo [37].
Diffusion
Diffusion-based gel formation involves the movement of a solvent from a polymer solution into the surrounding tissue. This diffusion leads to the precipitation or solidification of the polymer matrix. One commonly used solvent for this mechanism is N-methyl pyrrolidone (NMP), which facilitates the formation of the gel structure through solvent exchange [38].
Ionic cross-linking
Certain polysaccharides exhibit phase transitions when exposed to specific ions, making them ideal for ion-sensitive drug delivery systems. These polymers undergo in situ gelation in the presence of monovalent and divalent cations such as K⁺, Ca²⁺, Mg²⁺, and Na⁺.
For instance
κ-Carrageenan forms rigid, brittle gels with small amounts of K⁺
ι-Carrageenan produces flexible, elastic gels mainly in the presence of Ca²⁺
Gellan gum (commercially known as Gelrite®) is an anionic polysaccharide that gels in the presence of both mono-and divalent cations,
Low-methoxy pectin undergoes gelation primarily due to Ca²⁺,
Alginic acid forms gels upon exposure to divalent or polyvalent cations, particularly Ca²⁺, through interactions with guluronic acid blocks in the alginate chain [39].
Raft formation based on physiological stimuli mechanism
pH-dependent gelling
In situ gel formation can also be induced by changes in the pH of the surrounding environment. This mechanism utilizes pH-sensitive polymers that undergo a sol-to-gel transition in response to shifts in pH levels. Commonly used polymers include poly (acrylic acid) and its derivatives such as Carbopol® (carbomer), polyvinylacetal diethylaminoacetate (AEA), and mixtures of poly (methacrylic acid) (PMA) with poly (ethylene glycol) (PEG). These polymers, often referred to as polyelectrolytes due to their abundance of ionizable groups, react differently depending on their ionic nature. In the case of anionic (weakly acidic) polymers, swelling increases as the external pH rises, leading to gel formation. On the other hand, swelling decreases in cationic (weakly basic) polymers when pH increases. pH-sensitive polymers may be anionic, cationic, or neutral. Anionic polymers, which contain negatively charged groups such as carboxylic or sulfonic acids, undergo ionization when the surrounding pH exceeds the pKa of these moieties. This ionization promotes swelling and contributes to the formation of the gel matrix within the targeted area of the gastrointestinal tract.
Temperature-dependent gelling
These hydrogels remain in a liquid state at room temperature (20 °C–25 °C) and undergo gelation upon exposure to body temperature (35 °C–37 °C), triggered by a temperature rise. This process takes advantage of temperature-induced phase transitions. Certain polymers exhibit a sudden change in solubility when the temperature increases, a behavior defined by the Lower Critical Solution Temperature (LCST). At the LCST, hydrogen bonding between the polymer and water becomes less favorable than interactions among polymer chains and between water molecules. As a result, the hydrated polymer rapidly dehydrates and adopts a more hydrophobic structure, leading to gel formation. Additionally, some amphiphilic polymers self-assemble into micelles in solution and form gels upon heating due to intensified polymer–polymer interactions. Temperature-sensitive hydrogels are among the most widely explored environmentally responsive polymer systems in drug delivery research. Commonly used polymers include Pluronics (PEO–PPO–PEO triblock copolymers), poly (acrylic acid) (PAA), polyacrylamide (PAAm), and poly (acrylamide-co-butyl methacrylate). Some hydrogels, known as positive temperature-sensitive hydrogels, possess an Upper Critical Solution Temperature (UCST) and shrink when cooled below this temperature. Polymers like PAA, PAAm, and their copolymers demonstrate positive temperature-dependent swelling, making them ideal for controlled drug delivery applications [36, 40].
Advantages of raft forming system
Raft-forming systems provide several therapeutic advantages. They are commonly used for the symptomatic treatment of heartburn, gastroesophageal reflux disease (GERD), and laryngopharyngeal reflux (LPR), a condition characterized by the backflow of gastric contents into the laryngeal and pharyngeal regions. They do not interfere with the activity of promotility agents or antisecretory drugs such as cimetidine, making them suitable for combination therapy. Another key advantage is their ability to provide both rapid onset of action-often within seconds-and a long duration of therapeutic effect due to raft formation. Importantly, these systems do not interfere with the physiological function of the pyloric sphincter. In addition, they are generally well tolerated by patients and promote better compliance with treatment [30].
Limitations of the raft forming system
Raft-forming systems also face certain stability challenges. They are inherently more susceptible to degradation due to microbial contamination or chemical processes such as oxidation and hydrolysis. Proper storage is therefore essential, as unsuitable conditions can gradually alter the pH of the system, resulting in instability, particularly when exposed to inappropriate temperature ranges. In addition, some polymers used in these systems may undergo gel formation within the packaging when subjected to radiation such as ultraviolet, visible light, or electromagnetic waves [2].
Evaluation parameters
In vitro evaluation parameters of raft-forming systems
The in vitro evaluation of raft-forming systems involves several important tests to determine their mechanical, physicochemical, and drug-release properties. Texture analysis is performed to assess the consistency and cohesiveness of the formulation. This test ensures that the preparation can be easily administered, such as through a syringe, and that it possesses sufficient adhesiveness to maintain intimate contact with gastric tissues.
The sol–gel transition temperature and gelling time are determined to evaluate the formulation’s ability to transform from a liquid (sol) to a semi-solid (gel). The sol–gel transition temperature represents the point at which the sol first converts into a gel, while the gelling time indicates the duration required for this transformation to occur [41].
The floating or buoyancy test measures the time taken by the dosage form to float on simulated gastric fluid (floating lag time) and the total duration it remains buoyant [42]. Gel strength is another important parameter, determined using a rheometer to measure the resistance of the gel to penetration by a probe, which reflects the firmness of the formed gel [43].
Viscosity and rheology studies are carried out using instruments such as a Brookfield or Ostwald viscometer to evaluate the flow characteristics of the formulation, ensuring ease of administration and consistent gel formation [44]. Drug–excipient interaction studies are performed using Fourier-Transform Infrared Spectroscopy (FTIR) and Differential Scanning Calorimetry (DSC) to confirm compatibility and detect any possible interactions during gelation [45].
Finally, in vitro drug-release studies are conducted in 0.1 N HCl using a USP Type II dissolution apparatus at 37±0.2 °C. Samples are collected at predetermined time intervals, and the concentration of the released drug is measured spectrophotometrically to determine the rate and extent of drug release from the raft-forming system [46].
In vivo evaluation parameters of raft-forming systems
In vivo evaluation provides insight into the performance of raft-forming systems within the gastrointestinal tract. Radiology and scintigraphy techniques using radio-opaque markers such as barium sulfate or γ-emitting radionuclides like technetium-99 (⁹⁹Tc) are commonly employed to visualize the position of the dosage form and determine its gastric-retention time [47].
Gastroscopy, which involves per-oral endoscopy using fiber-optic or video systems, allows direct visual inspection of the formulation inside the stomach and helps assess its gastroretentive behavior [48]. Another non-radiative technique, magnetic marker monitoring, utilizes formulations labeled with iron powder. The movement and location of these magnetically marked dosage forms can then be tracked in real time using sensitive biomagnetic detection systems.
In addition, the ¹³C octanoic-acid breath test is a non-invasive method used to estimate gastric retention. A raft-forming formulation containing ¹³C-labeled octanoic acid releases ¹³CO₂ in the stomach, and the duration for which ¹³CO₂ is detected in exhaled breath corresponds to the gastric residence time of the system [49].
Marketed formulations
Several marketed raft-forming preparations are currently available for the management of gastroesophageal reflux and related disorders. Algicon® (Manufacturer: Sanofi, formerly Rorer) is a suspension containing alginic acid, aluminum hydroxide, and magnesium carbonate, which forms a floating raft gel upon contact with gastric acid, thereby neutralizing acid and preventing reflux. Gastrocote® (Manufacturer: Roche, formerly Boehringer) is another suspension composed of alginates, aluminum/magnesium hydroxide, and simethicone, providing both antacid activity and a protective barrier, and is commonly used in GERD and reflux management. In contrast, Bisodol® (Manufacturer: Pfizer, formerly Whitehall) is a chewable tablet that acts as a non-raft antacid through the combined effects of calcium carbonate, magnesium carbonate, and simethicone, offering potent acid neutralization and relief from bloating. Gastron® (Manufacturer: Sanofi Winthrop) is available as a suspension or tablet, containing alginic acid, magnesium trisilicate, and aluminum hydroxide, which function both as antacids and as a protective barrier via raft or mucosal coating. Among the most popular preparations, Liquid Gaviscon® (Manufacturer: Reckitt) consists of sodium alginate, calcium carbonate, and sodium bicarbonate, which together form a strong floating raft layer on gastric contents, providing effective anti-reflux action. Similarly, Gaviscon Advance® tablets (Manufacturer: Reckitt) are chewable formulations with higher doses of sodium alginate and calcium carbonate or potassium bicarbonate, offering the same mechanism as the liquid formulation but in a portable, solid dosage form.
CONCLUSION
Controlled release gastroretentive dosage forms (CR-GRDF) are designed to prolong drug retention in the upper gastrointestinal (GI) tract, enhancing the bioavailability of drugs with a narrow absorption window. These systems are especially beneficial for medications with poor bioavailability due to limited gastric contact. By forming a floating raft on gastric fluids, they help prevent reflux by acting as a physical barrier between the stomach and esophagus, offering therapeutic benefits in conditions like heartburn and esophagitis.
Drug absorption in the GI tract can be highly variable; thus, extending gastric retention improves the absorption window and overall bioavailability, particularly for drugs absorbed primarily in the upper GI tract or those with a short half-life. Raft-forming systems typically include a gel-forming agent (such as alginic acid), a bicarbonate (e. g., sodium bicarbonate), and an acid neutralizer. Upon contact with gastric acid, the bicarbonate releases CO₂, causing the raft to float and prolong contact with gastric fluids.
Despite some formulation challenges, many pharmaceutical companies are actively exploring and developing these systems due to their potential in treating gastrointestinal diseases, GERD, and related conditions.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
CONFLICT OF INTERESTS
Declared none
REFERENCES
Prajapati VD, Jani GK, Khutliwala TA, Zala BS. Raft forming system-an upcoming approach of gastroretentive drug delivery system. J Control Release. 2013;168(2):151-65. doi: 10.1016/j.jconrel.2013.02.028, PMID 23500062.
Devi KR. English language teaching in India: a critical review. Int J Pure App Biosci. 2015;3(5):178-83. doi: 10.18782/2320-7051.2119.
Nayak A, Maji R, Das B. Gastro retentive drug delivery systems: a review. Asian J Pharm Clin Res. 2010;3(1):2-10.
Aparna M, Anju P, Aswini PV. Raft forming system an innovative approach of gastro retentive drug delivery system. Int J Creat Res Thoughts. 2024;12:1-14.
Shukla S, Patidar A, Agrawal S, Choukse R. A review on: recent advancement of stomach specific floating drug delivery system. Int J Pharm Biol Arch. 2011;2(6):1561-8.
Rathod HJ, Mehta DP, Yadav JS. A review on gastro-resistant drug delivery systems. Pharma Tutor. 2016;4(2):29-40.
Nayak AK, Malakar J, Sen KK. Gastroretentive drug delivery technologies: current approaches and future potential. J Pharm Educ Res. 2010;1(2):1-12.
Gaur A, Saraswat R. Formulation and evaluation of sodium alginate-based in-situ gel drug delivery system of famotidine. Int J Pharm Innov. 2011;1(1):99-109.
Gananarajan G, Pal R. Raft forming gastro retentive drug delivery system a novel approach: a review. World J Pharm Pharm Sci. 2021;10(6):650-9.
Andrew A. A review on raft forming drug delivery system mechanism and its significance. AMJ 2022;15(2):336-7. doi: 10.21767/AMJ.2022.3868.
Singh BN, Kim KH. Floating drug delivery systems: an approach to oral controlled drug delivery via gastric retention. J Control Release. 2000;63(3):235-59. doi: 10.1016/S0168-3659(99)00204-7, PMID 10601721.
Gurnany E, Singhai P, Soni A, Jain R, Jain SK, Jain A. Gastro retentive drug delivery system a review. J Pharm Res. 2011;4(6):1899-908.
Chanda R, Roy A, Bahadur S, Saha S, Das S, Choudhury A. Floating drug delivery: a potential alternative to conventional therapy. Int J Pharm Tech Res. 2010;2(1):49-59.
Chaturvedi S, Kumari P, Singh S, Agrawal V. Approaches to increase the gastric residence time: floating drug delivery systems a review. Asian J Pharm Clin Res. 2013;6(3):1-9.
Gupta G, Singh AA. A short review on stomach-specific drug delivery system. Int J PharmTech Res. 2012;4(4):1527-45.
Kumar A, Verma R. Overview of gastroretentive drug delivery system. J Nat Conscientia. 2011;2(2):423-36.
Helliwell M. The use of bioadhesives in targeted delivery within the gastrointestinal tract. Adv Drug Deliv Rev. 1993;11(3):221-51. doi: 10.1016/0169-409X(93)90011-R.
Jadi RK, Chinnala KM. A comprehensive review on gastroretentive drug delivery systems. Indo Am J Pharm Sci. 2016;3(1):115-28.
Aute SM, Payghan SA, D’Souza AI, Navhkar S, Lad D, Jirole U. Novel approach in gastroretentive drug delivery system: floating microspheres. Int J Pharm Biol Sci Arch. 2014;2(5):9-22.
Patel A, Patel V, Patel H, Patel J, Patel H. A review on mucoadhesive microcapsules: novel approach for stomach specific drug delivery. Int J Pharm Res Bio-sci. 2014;3(1):449-65.
Vignesh R, Kamaleshwari B, Kaviya G, Kovarthanan M, Raj UM. Innovations in oral drug administration: exploring gastro retentive raft forming system. Int J Pharm Sci. 2024;2(3):289-302. doi: 10.5281/zenodo.10805754.
Reddy BV, Navaneetha K, Sandeep P, Deepthi A. Gastroretentive drug delivery system a review. J Glob Trends Pharm Sci. 2013;4(1):1018-33.
Singh N, Pandurangan A, Rana K, Anand P, Ahamad A, Tiwari AK. Benzimidazole: a short review of their antimicrobial activities. Int Curr Pharm J. 2012;1(5):110-8. doi: 10.3329/icpj.v1i5.10284.
Gunda RK, Vijayalakshmi A. Formulation development and evaluation of gastro retentive drug delivery systems a review. J Pharm Res. 2017;11(2):167-78.
Sharma S, Pawar A. Low density multiparticulate system for pulsatile release of meloxicam. Int J Pharm. 2006;313(1-2):150-8. doi: 10.1016/j.ijpharm.2006.02.001, PMID 16540268.
Rajinikanth PS, Balasubramaniam J, Mishra B. Development and evaluation of a novel floating in situ gelling system of amoxicillin for eradication of Helicobacter pylori. Int J Pharm. 2007;335(1-2):114-22. doi: 10.1016/j.ijpharm.2006.11.008, PMID 17141986.
Bhavsar DN. Advances in GRDDS: raft forming system a review. J Drug Delivery Ther. 2012;2(5):123-8. doi: 10.22270/jddt.v2i5.228.
Ibrahim HK. A novel liquid effervescent floating delivery system for sustained drug delivery. Drug Discov Ther. 2009;3(4):168-75. PMID 22495603.
Prajapati ST, Mehta AP, Modhia IP, Patel CN. Formulation and optimisation of raft-forming chewable tablets containing H2 antagonist. Int J Pharm Investig. 2012;2(4):176-82. doi: 10.4103/2230-973X.106988, PMID 23580933.
Kapadia CJ, Mane VB. Raft-forming agents: antireflux formulations. Drug Dev Ind Pharm. 2007;33(12):1350-61. doi: 10.1080/03639040701385691, PMID 18097809.
Shah S, Upadhyay P, Shah J. In situ gel: a novel approach of gastroretentive drug delivery. Asian J Biomed Pharm Sci. 2012;2(8):1-8.
Ali J, Ahuja A, Baboota S, Qureshi M. Formulation strategy for low absorption window antihypertensive agent. Indian J Pharm Sci. 2007;69(3):360-4. doi: 10.4103/0250-474X.34543.
Suresh S, Bhaskaran S. Nasal drug delivery: an overview. Indian J Pharm Sci. 2005;67(1):19-25.
Gananarajan G, Pal R. Raft forming gastroretentive drug delivery system a novel approach. World J Pharm Pharm Sci. 2021;10(6):650-9.
Kubo W, Konno Y, Miyazaki S, Attwood D. In situ gelling pectin formulations for oral sustained delivery of paracetamol. Drug Dev Ind Pharm. 2004;30(6):593-9. doi: 10.1081/DDC-120037490, PMID 15285332.
Schild HG. Poly(N-isopropylacrylamide): experiment, theory and application. Prog Polym Sci. 1992;17(2):163-249. doi: 10.1016/0079-6700(92)90023-R.
Sawhney AS, Pathak CP, Hubbell JA, Hill JL, Desai NP. Photopolymerizable biodegradable hydrogels as tissue contacting materials and controlled release carriers. US Patent 5410016A; 1995.
Rathod H, Patel V, Modasia M. In situ a novel approach of gastroretentive drug delivery. Int J Pharm Life Sci. 2010;1(6):440-5.
Teaima M, Abdelmonem R, Saady M, El-nabarawi M, Shoman NA. Comprehensive overview on recent updates gastroretentive of raft-forming systems: a review. Int J App Pharm. 2022;14(3):40-8. doi: 10.22159/ijap.2022v14i3.44098.
Bineesha KB, Dharan S, Abraham S, Panicker JT, James JE. Gastroretentive raft forming drug delivery: a novel expansion: a review. Int J Pharm Pharm Res. 2019;15(2):200-12.
Miyazaki S, Aoyama H, Kawasaki N, Kubo W, Attwood D. In situ-gelling gellan formulations as vehicles for oral drug delivery. J Control Release. 1999;60(2-3):287-95. doi: 10.1016/S0168-3659(99)00084-X, PMID 10425334.
Gulkari VD, Bakhle SS, Yelane LS. Development and evaluation of ofloxacin floating tablets using natural polymer: Sterculia foetida Linn. gum. Int J Pharm Pharm Sci. 2016;8(5):356-60.
Maheswaran A, Padmavathy J, Nandhini V, Saravanan D, Angel P. Formulation and evaluation of floating oral in situ gel of diltiazem hydrochloride. Int J App Pharm. 2016;9(1):50. doi: 10.22159/ijap.2017v9i1.15914.
Kashyap N, Viswanad B, Sharma G, Bhardwaj V, Ramarao P, Ravi Kumar MN. Design and evaluation of biodegradable biosensitive in situ gelling system for pulsatile delivery of insulin. Biomaterials. 2007;28(11):2051-60. doi: 10.1016/j.biomaterials.2007.01.007, PMID 17240443.
Chandrashekar G, Udupa N. Biodegradable injectable implant systems for long term drug delivery using poly (lactic-co-glycolic) acid copolymers. J Pharm Pharmacol. 1996;48(7):669-74. doi: 10.1111/j.2042-7158.1996.tb03948.x, PMID 8866326.
Teaima MH, Abdel Hamid MM, Shoman NA, Jasti BR, El-Nabarawi MA, Yasser M. Formulation characterization and comparative pharmacokinetic study of bupropion floating raft system as a promising approach for treating depression. J Pharm Sci. 2020;109(11):3451-61. doi: 10.1016/j.xphs.2020.08.011, PMID 32835701.
Boddupalli BM, Mohammed ZN, Nath RA, Banji D. Mucoadhesive drug delivery system: an overview. J Adv Pharm Technol Res. 2010;1(4):381-7. doi: 10.4103/0110-5558.76436, PMID 22247877.
More S, Gavali K, Doke O, Kasgawade P. Gastroretentive drug delivery system. J Drug Delivery Ther. 2018;8(4):4. doi: 10.22270/jddt.v8i4.1788.
Prajapati VD, Jani GK, Khutliwala TA, Zala BS. Raft forming system-an upcoming approach of gastroretentive drug delivery system. J Control Release. 2013;168(2):151-65. doi: 10.1016/j.jconrel.2013.02.028, PMID 23500062.