
Int J Curr Pharm Res, Vol 18, Issue 2, 16-23Review Article
POLYMERIC SILVER NANOCARRIERS: INNOVATIVE DRUG DELIVERY STRATEGIES FOR ALZHEIMER’S DISEASE AND NEURODEGENERATIVE DISORDERS
HIMANSHU KINKAR, DIVYANSH DHARPURE, DHANASHRI TUMME*
School of Pharmacy, GH Raisoni University Saikheda Madhya Pradesh, India
*Corresponding author: Dhanashri Tumme; *Email: dhanashri.tumme@ghru.edu.in
Received: 17 Nov 2025, Revised and Accepted: 07 Jan 2026
ABSTRACT
Alzheimer’s disease (AD) is a multifactorial neurodegenerative disorder marked by amyloid-β plaque accumulation, tau hyperphosphorylation, oxidative stress, neuroinflammation, and progressive cognitive impairment. The clinical effectiveness of conventional therapies is limited due to poor brain bioavailability, rapid systemic clearance, and the restrictive nature of the blood–brain barrier (BBB), resulting mainly in symptomatic relief rather than disease modification. In this context, polymeric silver nanocarriers (PSNs) have emerged as a promising nanotherapeutic approach for targeted and controlled drug delivery to the central nervous system. By integrating the antioxidant and anti-inflammatory properties of silver nanoparticles with the biocompatibility, stability, and controlled release characteristics of polymeric matrices such as PLGA, PEG, and chitosan, PSNs enable enhanced drug encapsulation, sustained release, and efficient BBB penetration through surface functionalization and receptor-mediated transport mechanisms. Preclinical in vitro and in vivo studies demonstrate that PSNs can effectively reduce amyloid-β aggregation, inhibit tau pathology, attenuate oxidative stress, and suppress neuroinflammatory responses, leading to improved neuronal survival and cognitive outcomes. Alternative delivery strategies, including intranasal and nose-to-brain routes, further improve therapeutic efficiency while minimizing systemic toxicity. Although challenges related to long-term safety, neurotoxicity, scalability, and regulatory approval remain, polymeric silver nanocarriers represent a versatile and multifunctional platform with significant potential for advancing disease-modifying therapies in Alzheimer’s disease.
Keywords: Alzheimer's disease, Blood-brain barrier, Polymeric silver nanocarriers, Neurodegeneration, Nanomedicine, Targeted drug delivery
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i2.8049 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Neurodegenerative diseases are characterized by progressive neuronal dysfunction and loss, leading to cognitive and functional impairment. These disorders share common pathological mechanisms such as protein aggregation, oxidative stress, mitochondrial dysfunction, and neuroinflammation, which collectively contribute to disease progression and therapeutic complexity [1, 2].
Alzheimer’s diseases (AD) is the most common cause of dementia worldwide and represents a major public health challenge, particularly in the aging population [3]. The pathological hallmarks of AD include extracellular amyloid-beta (Aβ) plaque deposition and intracellular neurofibrillary tangles formed by hyperphosphorylated tau protein, which causes specific damage to synapses, causes brain cells to die, and accelerates impaired thinking [4]. These pathological mechanisms, particularly oxidative stress and protein aggregation, are central to Alzheimer’s diseases and form the basis for targeted therapeutic strategies [5].
Despite significant research progress, effective treatment of AD remains limited due to the restrictive nature of the blood-brain barrier (BBB), which severely limits the entry of therapeutic agents into the central nervous system [6]. This barrier allows only a small fraction of small molecules and virtually no large biologics to reach the brain, thereby reducing therapeutic efficacy [7].
Consequently, conventional systemic therapies often require high doses to achieve therapeutic concentration in the brain, increasing the risk of peripheral toxicity [8, 9]. Alternative strategies such as intranasal delivery have been explored to bypass the BBB; however, limited dosing capacity and rapid mucociliary clearance restrict their clinical utility [10]. Invasive approaches such as intracerebral injection can enhance drug delivery to the brain but are associated with significant risks, including tissue damage and infection [11].
Nanotechnology-based drug delivery systems, particularly polymeric silver nanocarriers (PSNs), have emerged as promising strategies to overcome BBB-related limitations. These nanoscale systems enable drug encapsulation, protection from degradation, controlled release, and enhanced brain targeting through modulation of size, surface charge, and functionalization [12]. Silver nanoparticles (AgNPs), when integrated with polymeric systems, exhibit antimicrobial, anti-inflammatory, and antioxidant properties that may provide neuroprotective benefits. Polymeric encapsulation improves their stability, biocompatibility, and controlled release, thereby reducing cytotoxicity [13, 14].
Polymeric silver nanocarriers can cross the BBB via receptor-mediated transcytosis by exploiting endogenous transport pathways, enabling enhanced brain delivery without permanent barrier disruption [8, 15].
Recent preclinical studies have demonstrated that polymeric silver nanocarriers enable brain-targeted delivery of anti-Alzheimer agents, resulting in reduced amyloid burden and improved cognitive performance in animal models [16]. Additionally, polymer-based silver nanocarriers have shown improved pharmacokinetic profiles, including prolonged circulation time and reduced off-target accumulation [17]. Despite these encouraging findings, challenges related to large-scale production, reproducibility, and long-term safety assessment remain critical barriers to clinical translation [9].
The complex pathophysiology of Alzheimer’s disease, combined with the restrictive nature of the blood–brain barrier, underscores the urgent need for advanced drug delivery strategies. Polymeric silver nanocarriers offer a promising nanotechnological platform by enhancing brain targeting, reducing systemic toxicity, and improving therapeutic efficacy. This review provides a focused overview of the design principles, BBB-crossing mechanisms, therapeutic potential, and clinical relevance of polymeric silver nanocarrier-based drug delivery systems for Alzheimer’s disease and related neurodegenerative disorders.
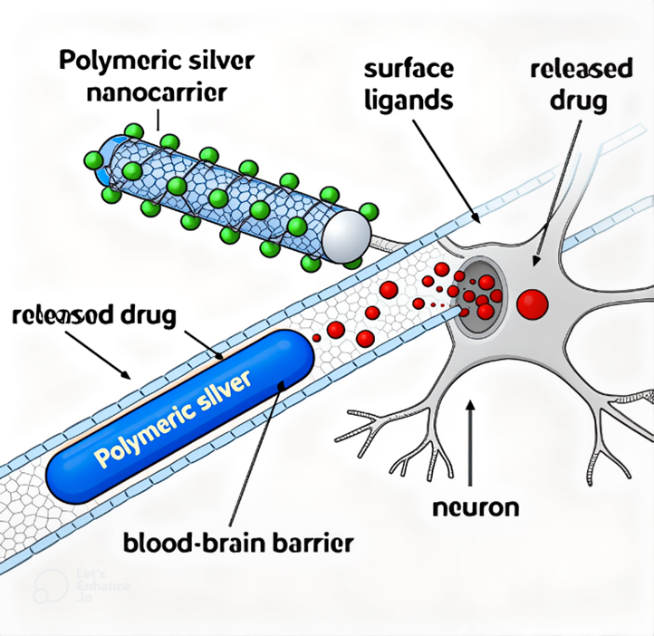
Fig. 1: Graphical representation of the generalised mode of action of PSNs
Pathology of alzheimer’s disease
Alzheimer’s disease is characterized by a multifactorial pathological cascade involving amyloid-β accumulation, tau protein alterations, neuroinflammation, oxidative stress, and vascular dysfunction. These interacting mechanisms explain why single-target therapeutic strategies have largely failed, necessitating a multi-targeted approach [18, 19].
The amyloid cascade hypothesis proposes that abnormal accumulation of amyloid-β peptides leads to extracellular plaque formation, triggering synaptic dysfunction, neuroinflammation, and neuronal toxicity [20]. Although extensive experimental evidence supports this hypothesis, clinical trials targeting amyloid-β have demonstrated limited success, indicating that amyloid pathology alone may not fully account for disease progression [21, 22]. Tau pathology represents another major hallmark of Alzheimer’s disease. Hyperphosphorylation and aggregation of tau protein result in neurofibrillary tangles, leading to impaired axonal transport, synaptic dysfunction, and neuronal death [23, 24]. Importantly, tau burden shows a stronger correlation with cognitive decline than amyloid plaque load, highlighting tau as a critical therapeutic target [25].
Calcium dyshomeostasis plays a critical role in Alzheimer’s disease by impairing synaptic plasticity, reducing neuronal excitability, and promoting neuronal dysfunction [26]. Abnormal calcium signaling is closely associated with amyloid-β and tau toxicity, contributing to disease progression [27]. Mitochondrial dysfunction is another key pathological feature of Alzheimer’s disease, characterized by impaired energy metabolism, increased reactive oxygen species (ROS] production, and activation of apoptotic pathways [28, 29]. Defective autophagy further exacerbates neuronal damage by allowing accumulation of misfolded proteins and dysfunctional organelles, creating a neurotoxic cellular environment [30].
Progressive synaptic loss represents the final pathological outcome of Alzheimer’s disease and underlies cognitive impairment and dementia [19]. Oxidative stress and chronic neuroinflammation amplify neuronal injury, while excessive glutamate-mediated excitotoxicity and calcium overload further accelerate neurodegeneration [23, 27].
Rather than arising from a single dominant process, Alzheimer’s disease emerges from the cumulative interaction of amyloid-β accumulation, tau dysfunction, oxidative injury, mitochondrial abnormalities, neuroinflammation, synaptic loss. The multifactorial character of these pathological events underscores the limitation of conventional therapies and supports the exploration of advanced treatment strategies designed to overcome biological barriers and complex disease mechanisms.
Drug delivery challenges: blood-brain barrier (BBB)
The blood–brain barrier (BBB) is a highly selective physiological interface that maintains central nervous system homeostasis by regulating molecular exchange between the bloodstream and brain tissue [31]. While essential for neural protection, this restrictive barrier significantly limits the effective delivery of therapeutic agents in neurodegenerative disorders such as Alzheimer’s disease [32].
Structurally, the BBB is formed by specialized endothelial cells interconnected by tight junction complexes and supported by pericytes and astrocytic end-feet, collectively known as the neurovascular unit. This organization creates a highly regulated barrier that restricts paracellular transport and tightly controls molecular trafficking into the brain [33, 34].
BBB permeability is governed by molecular size, lipophilicity, charge, and conformational flexibility [35]. While small lipophilic molecules may cross via passive diffusion, most therapeutic agents rely on specialized transport mechanisms such as carrier-mediated and receptor-mediated transcytosis, which remain limited for efficient drug delivery [32].
In Alzheimer’s disease, BBB integrity is further compromised by neuroinflammation, amyloid accumulation, and endothelial dysfunction, resulting in altered permeability and transporter expression [9]. These dynamic and heterogeneous changes complicate drug delivery and reduce the effectiveness of conventional therapeutic strategies, particularly in elderly patients [36].
Conventional pharmacological approaches remain largely ineffective for central nervous system drug delivery due to limited BBB permeability, active efflux mechanisms, and the need for high systemic doses that increase toxicity [37]. Invasive strategies such as direct intracerebral delivery have shown limited clinical utility because of safety concerns, underscoring the need for alternative, non-invasive delivery approaches [38]. Nanotechnology-based delivery systems, particularly polymeric silver nanocarriers, offer a promising strategy to overcome BBB-associated limitations. Their nanoscale size, surface modifiability, and controlled release properties enable efficient encapsulation of both hydrophilic and hydrophobic drugs, improving brain bioavailability and neuroprotective outcomes [39, 40].
Nanocarriers primarily exploit endogenous BBB transport mechanisms, particularly receptor-mediated transcytosis, to facilitate brain entry. Surface modification with appropriate ligands enhances nanoparticle uptake and enables controlled transport across endothelial cells without permanent barrier disruption. Intranasal delivery has also emerged as a complementary non-invasive approach for selected nanocarrier systems [41, 42].
Surface functionalization plays a critical role in enhancing BBB targeting and therapeutic specificity. Conjugation with ligands, peptides, or antibodies targeting BBB receptors improves cellular uptake, prolongs circulation time, and reduces immune clearance [43]. Optimization of nanoparticle size, shape, and surface charge further supports efficient brain penetration and intracellular trafficking [44].
Receptor-mediated targeting remains the most effective strategy for nanocarrier-based BBB transport. Successful delivery depends on careful ligand selection, efficient endosomal escape, and controlled intracellular release to achieve therapeutic concentrations within the brain parenchyma while minimizing off-target effects [45, 46].
These dynamic changes in BBB integrity and transport regulation significantly complicate effective drug delivery to the central nervous system, highlighting the need for advanced delivery strategies in Alzheimer’s disease.
Synthesis and characterization of polymeric silver nanocarriers
Polymeric silver nanocarriers represent an emerging class of nanomedicine platforms that integrate the therapeutic potential of silver nanoparticles with the structural versatility of polymers. These systems are designed to enhance drug loading, stability, controlled release, and targeted delivery across biological barriers such as the blood–brain barrier, making them promising candidates for central nervous system applications [41, 47].
A variety of natural and synthetic polymers have been employed in the fabrication of polymeric silver nanocarriers. Natural polymers such as chitosan and cellulose offer biocompatibility and functional groups for drug conjugation, while synthetic polymers, including PLGA and PEG provide controlled degradation, prolonged circulation, and tunable release profiles. Hybrid polymer systems further enhance structural stability and delivery performance [48].
Polymeric silver nanocarriers can be synthesized using chemical, physical, and green biological approaches. Chemical reduction methods allow control over particle size and shape, while physical techniques such as laser ablation enable surfactant-free nanoparticle generation. Green synthesis using plant-derived phytochemicals and biopolymers offers an eco-friendly alternative, providing inherent stabilization and improved biocompatibility. Composite nanocarriers are often formed through in situ nucleation of silver nanoparticles within polymeric matrices, improving dispersion stability and controlled drug release. Core–shell and micellar architectures further protect therapeutic payloads from premature degradation and enhance in vivo performance [14, 15].
Physicochemical characterization is essential to evaluate nanocarrier performance. Particle size, morphology, and dispersion are commonly assessed using TEM, SEM, and dynamic light scattering. Surface charge and colloidal stability are determined by zeta potential measurements, while UV–Vis spectroscopy and X-ray diffraction confirm nanoparticle formation and crystallinity [49].
Polymer selection, molecular weight, and ligand density significantly influence nanocarrier stability, release kinetics, and safety. In vitro cytotoxicity and hemocompatibility studies demonstrate that biopolymer-based matrices reduce silver ion leaching and maintain cellular viability, supporting their suitability for neurological applications [50].
In summary, polymeric silver nanocarriers can be fabricated using diverse polymer systems and synthesis approaches that allow precise control over size, stability, and drug release behavior. Advances in green synthesis, polymer engineering, and characterization techniques continue to support the development of safe and reproducible nanocarrier platforms suitable for brain-targeted drug delivery.
Mechanisms of action in alzheimer’s therapy
One of the major challenges in achieving effective AD therapy is the restricted transport of therapeutic agents across the blood–brain barrier (BBB). Conventional drug delivery approaches often fail to maintain adequate drug concentrations at pathological sites due to rapid systemic clearance and poor brain bioavailability. These limitations have driven the development of nanocarrier-based systems capable of enhancing CNS delivery and therapeutic efficacy [51, 52].
Polymeric silver nanocarriers (PSNs) exert therapeutic effects in AD through multiple complementary mechanisms, including enhanced brain targeting, protection of loaded drugs from enzymatic degradation, and controlled intracellular release [53]. The polymeric matrix enables sustained drug availability, while silver nanoparticles contribute intrinsic anti-inflammatory and antioxidant effects that modulate neuroinflammation and oxidative stress. Surface functionalization of PSNs further enables interaction with disease-specific pathways, thereby improving therapeutic outcomes in Alzheimer’s disease [54].
Drug loading capabilities of polymeric silver nanocarriers
The success of polymeric silver nanocarriers in Alzheimer’s therapy largely depends on their drug loading capacity and physicochemical stability. The nanoscale size and high surface-to-volume ratio of PSNs allow efficient encapsulation of both hydrophobic and hydrophilic therapeutic agents. Biocompatible polymers such as poly (lactic-co-glycolic acid) (PLGA) and polyethylene glycol (PEG) enhance drug stability, prolong systemic circulation, and enable sustained release within the CNS environment [51, 55].
Various formulation approaches, including ionic gelation and antisolvent precipitation techniques, have been employed to achieve high encapsulation efficiency in polymeric silver nanocarriers while maintaining particle stability and uniform size distribution [56].
Silver nanoparticles provide additional advantages in drug loading systems due to their inherent antimicrobial, antioxidant, and anti-inflammatory properties. When incorporated within polymeric nanocarriers, silver enhances payload stability, reduces premature drug degradation, and synergistically contributes to neuroprotection by attenuating oxidative stress and inflammatory responses associated with Alzheimer’s pathology [57. 58].
Profiles of controlled release for polymeric silver nanocarriers
Controlled drug release is an essential feature of polymeric silver nanocarriers, enabling sustained and localized delivery of therapeutic agents to diseased brain regions. Drug release primarily occurs through diffusion across the polymeric matrix and gradual degradation of the polymer backbone, allowing maintenance of therapeutic drug levels over extended periods. Additionally, stimuli-responsive systems sensitive to pathological microenvironmental cues such as pH, enzymatic activity, and oxidative stress further enhance site-specific release and therapeutic precision [59].
Precise control over release kinetics improves therapeutic efficacy by maintaining effective drug concentrations within brain tissue while minimizing systemic exposure and toxicity. This is particularly beneficial for compounds such as curcumin, which exhibit poor bioavailability and rapid systemic clearance in their free form. Encapsulation within polymeric silver nanocarriers enhances drug retention in the brain, allowing concentrations to remain above the minimum inhibitory threshold required to limit amyloid and tau aggregation [60, 61].
Surface functionalization plays a critical role in optimizing controlled release and targeting efficiency. Polyethylene glycol (PEG) is widely employed to improve nanocarrier biocompatibility and prolong systemic circulation by reducing opsonization and reticuloendothelial clearance. Additionally, conjugation with targeting ligands such as the B6 peptide enables selective interaction with CNS receptors, improving localization at diseased sites and modulating release kinetics for enhanced therapeutic outcomes [62].
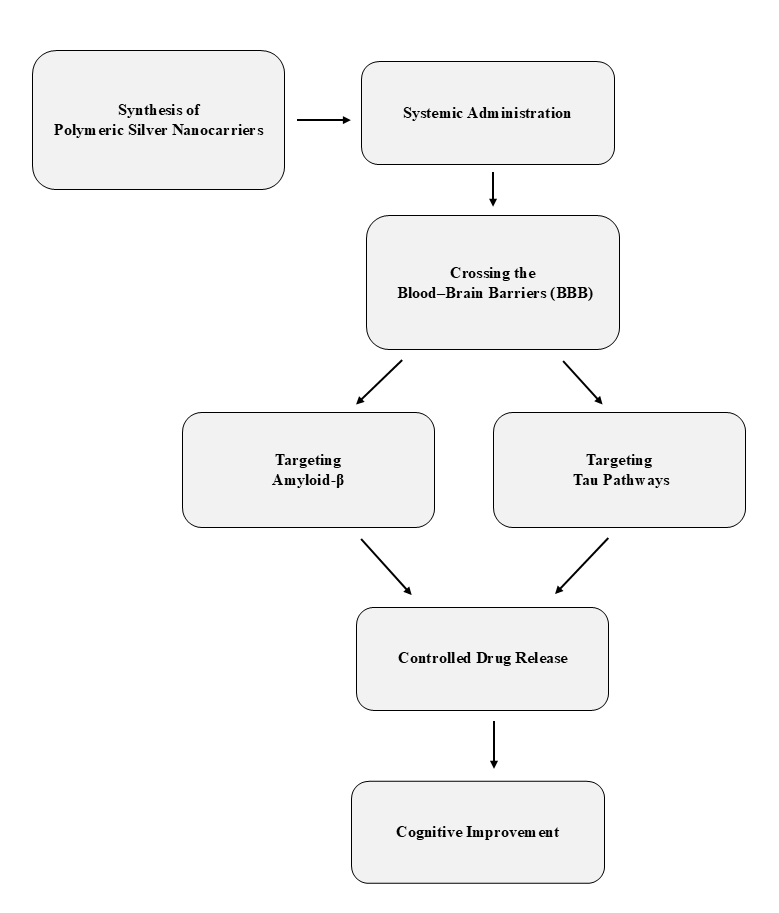
Fig. 2: Flowchart illustrating the process of polymeric silver nanocarrier-mediated drug delivery for alzheimer's disease
Targeting amyloid plaques using polymeric silver nanocarriers
One of the primary treatment objectives of the Alzheimer Disease (AD) is the reduction of the amyloid-beta plaque burden, of which polymeric silver nanocarriers are thoroughly designed to enhance binding and recognition to amyloid aggregates. Surface ligands, targeting peptides, including β6 peptide, have been shown to strongly affiliate with amyloid-beta deposition, activating nanocarriers at precise localization spots at plaque locations [63]. Such specific binding assists in the process of deliver therapeutic payloads specifically in pathogenicand this has made the drug perform better and minimizes the side effects in the body. Upon reaching amyloid plaques, therapeutic payloads such as curcumin exert anti-amyloidogenic effects by inhibiting fibril formation and promoting plaque destabilization. The polyphenolic structure of curcumin enables effective binding to amyloid fibrils, reducing oligomer formation and neurotoxicity. Nanocarrier-mediated delivery enhances curcumin stability and bioavailability, overcoming limitations associated with its hydrophobic nature and rapid clearance [64].
In addition to acting as delivery vehicles, silver nanoparticles contribute directly to amyloid clearance through modulation of oxidative stress pathways. By regulating reactive oxygen species (ROS) levels and attenuating neuroinflammatory responses, polymeric silver nanocarriers indirectly destabilize amyloid aggregates and promote neuronal protection. This synergistic interaction between nanocarrier-mediated drug delivery and the intrinsic biological activity of silver enhances overall therapeutic efficacy [65].
Addressing tau pathology via nanocarrier-mediated delivery
Tau pathology represents a major therapeutic target in Alzheimer’s disease, characterized by excessive hyperphosphorylation of tau protein leading to neurofibrillary tangle formation and neuronal dysfunction. Polymeric silver nanocarriers enable selective delivery of tau-modulating agents through receptor-mediated endocytosis, particularly in vulnerable hippocampal neurons. This targeted approach improves intracellular drug bioavailability and facilitates modulation of tau-related pathways critical for maintaining cognitive function [23, 25].
Nanocarriers assist in the transport of small molecules capable of modulating tau signaling pathways and preventing tau aggregation. Experimental evidence supports the role of curcumin and related polyphenols in regulating key intracellular pathways, including PI3K/Akt/mTOR and ROS/JNK, which are involved in tau phosphorylation and degradation. Sustained modulation of these signaling cascades through nanocarrier delivery contributes to the inhibition of neurofibrillary tangle formation [60, 66].
Sustained drug delivery via polymeric silver nanocarriers is essential for maintaining effective intracellular concentrations required to counter chronic tau-mediated neurodegeneration. Nanocarrier designs incorporating mitochondrial-targeted strategies have demonstrated improved neuronal energy metabolism and reduced oxidative damage, further supporting their potential in tau-focused therapeutic interventions [67].
Multifunctional therapeutic properties of polymeric silver nanocarriers
Polymeric silver nanocarriers exhibit multifunctional therapeutic properties that extend beyond targeted drug delivery, enabling simultaneous modulation of multiple pathological pathways involved in Alzheimer’s disease. In addition to facilitating the transport of therapeutic payloads across the blood–brain barrier, these nanocarriers demonstrate intrinsic anti-inflammatory, antioxidant, and anti-amyloid activities. Silver nanoparticles suppress microglial overactivation and reduce pro-inflammatory cytokine release, thereby attenuating chronic neuroinflammation associated with disease progression. Furthermore, their redox-active nature allows effective scavenging of reactive oxygen and nitrogen species, leading to reduced oxidative stress, mitochondrial dysfunction, and neuronal apoptosis [68]. When combined with pharmacological agents such as curcumin or other neuroprotective compounds, polymeric silver nanocarriers enable synergistic inhibition of amyloid-β aggregation and tau hyperphosphorylation, resulting in enhanced neuroprotection and preservation of synaptic function [61]. This multifunctionality positions polymeric silver nanocarriers as a promising therapeutic platform for addressing the complex and multifactorial pathogenesis of Alzheimer’s disease [67].
Preclinical in vitro evidence of therapeutic efficacy
Cellular and in vitro studies play a crucial role in evaluating the therapeutic efficacy and safety of polymeric silver nanocarriers for Alzheimer’s disease. Neuronal cell lines such as HT22 have demonstrated efficient internalization of polymeric silver nanocarriers due to their optimized nanoscale size and surface properties [69]. Techniques, including flow cytometry and dynamic light scattering confirm uniform particle size distribution and cellular uptake efficiency. Curcumin-loaded polymeric silver nanocarriers exhibit significant neuroprotective effects by reducing oxidative stress and amyloid-β–induced toxicity, thereby preserving synaptic function [61]. Furthermore, treatment with these nanocarriers results in decreased levels of pro-inflammatory cytokines and reduced tau hyperphosphorylation in neuronal cultures, highlighting their combined anti-inflammatory and tau-targeting potential. Collectively, these findings confirm the suitability of polymeric silver nanocarriers as multifunctional in vitro therapeutic platforms for Alzheimer’s disease [67].
In vivo preclinical results indicating therapeutic possibilities
In vivo studies using Alzheimer’s disease animal models further support the therapeutic potential of polymeric silver nanocarriers. In APP/PS1 transgenic mouse models, administration of curcumin-loaded polymeric silver nanocarriers significantly improved cognitive performance, as assessed by behavioural tests such as the Morris water maze. These improvements indicate enhanced learning and memory functions due to efficient drug delivery to the brain [70]. Histopathological analyses reveal a marked reduction in amyloid plaque burden and tau hyperphosphorylation in hippocampal regions following nanocarrier treatment. Immunostaining and western blot studies further confirm decreased pathological protein aggregation at the molecular level. Additionally, biocompatibility and hemocompatibility studies demonstrate an acceptable safety profile with minimal systemic toxicity, supporting the feasibility of polymeric silver nanocarriers for long-term therapeutic application [71].
Comparison between PSNs and other nanotherapeutics in AD
Compared to other nanotherapeutic systems, such as lipid-based or metallic nanoparticles, polymeric silver nanocarriers offer distinct advantages in the treatment of Alzheimer’s disease. The polymeric matrix enables controlled and sustained drug release, improving CNS bioavailability and prolonging circulation time. Unlike lipid carriers that may undergo rapid degradation, polymeric silver nanocarriers provide enhanced structural stability along with intrinsic antioxidant and antimicrobial properties derived from silver [58]. This multifunctionality allows simultaneous modulation of oxidative stress, neuroinflammation, and protein aggregation pathways. Consequently, polymeric silver nanocarriers present a superior therapeutic platform by combining controlled drug delivery with inherent neuroprotective effects not commonly observed in other nanotherapeutic systems [70].
Despite promising therapeutic outcomes, the clinical translation of polymeric silver nanocarriers remains challenging. Issues related to pharmacokinetics, long-term safety, potential immunogenicity, and large-scale reproducibility require careful evaluation. Addressing these limitations through optimized formulation design and targeted delivery strategies is essential for successful clinical application in Alzheimer’s disease [72].
Recent advances: intranasal and nose-to-brain delivery
Intranasal and nose-to-brain delivery has emerged as a promising non-invasive strategy to bypass the blood–brain barrier and enhance central nervous system drug delivery in Alzheimer’s disease [73]. This route exploits direct connections between the nasal cavity and the brain through the olfactory and trigeminal nerve pathways, enabling rapid drug transport while avoiding systemic circulation and first-pass metabolism [74, 75].
Polymeric nanocarriers have gained significant attention for intranasal delivery due to their biocompatibility, biodegradability, and ability to provide controlled and sustained drug release. Polymers such as PLGA, chitosan, polyethylene glycol (PEG), and poly-L-lysine dendrimers improve nasal residence time, protect drugs from enzymatic degradation, and enhance mucosal permeability [76]. Surface modifications, including mucoadhesive polymers and ligand conjugation, further improve retention, penetration, and targeting efficiency within the nasal mucosa [77].
Despite these advantages, challenges such as mucociliary clearance, enzymatic degradation, limited dosing volume, and inter-individual variability remain significant barriers to clinical translation. Recent formulation strategies incorporating mucoadhesive systems, surface charge optimization, and stimuli-responsive coatings have shown potential to overcome these limitations [78]. Overall, intranasal polymeric nanocarrier-based delivery represents a promising advancement for targeted brain drug delivery in Alzheimer’s disease, although further optimization and clinical validation are required [79].
Safety, neurotoxicity, and regulatory factors
Safety, neurotoxicity, and regulatory considerations remain critical challenges in the clinical translation of polymeric silver nanocarriers for Alzheimer’s disease therapy. While silver nanoparticles exhibit promising antimicrobial, anti-inflammatory, and drug delivery properties, their small size and surface reactivity raise concerns regarding oxidative stress generation, neuroinflammation, and long-term accumulation in the central nervous system [80].
Polymeric encapsulation of silver nanoparticles using biocompatible polymers such as PLGA, PEG, polydopamine, and hyaluronic acid has been shown to significantly reduce direct cytotoxicity, suppress reactive oxygen species production, and improve stability by minimizing uncontrolled ion release. Surface functionalization further enhances biocompatibility, reduces immunogenicity, and improves pharmacokinetic profiles, making polymeric silver nanocarriers safer for CNS applications [81].
Despite these advancements, comprehensive in vivo toxicological evaluation remains essential to assess biodistribution, clearance, chronic exposure risks, and neuroimmune interactions. Regulatory approval of CNS-targeted nanomedicines is challenging due to the lack of standardized testing protocols specific to nanomaterials. Agencies such as the FDA and EMA emphasize rigorous physicochemical characterization, reproducible manufacturing, neurotoxicity assessment, immunotoxicity evaluation, and long-term safety profiling prior to clinical translation [82].
Overall, polymeric silver nanocarriers represent a promising yet cautiously advancing strategy for neurodegenerative disease treatment, where safety-by-design approaches and regulatory alignment will play a decisive role in their future clinical success [83].
These safety and regulatory considerations underline the need for optimized polymeric design, standardized evaluation frameworks, and interdisciplinary collaboration to enable the safe clinical translation of polymeric silver nanocarriers for Alzheimer’s disease.
CONCLUSION
Future research on polymeric silver nanocarriers should focus on optimizing polymer composition, surface functionalization, and release kinetics to enhance safety, targeting precision, and therapeutic efficacy in Alzheimer’s disease. Long-term in vivo studies are essential to evaluate chronic neurotoxicity, biodistribution, clearance mechanisms, and immunological responses, particularly under repeated dosing conditions.
Advancements in stimuli-responsive and ligand-mediated targeting systems offer opportunities to improve selective delivery across the blood–brain barrier while minimizing off-target effects. Integration of nanocarriers into theranostic platforms may further enable real-time tracking of drug distribution and treatment response, supporting personalized therapeutic strategies.
From a translational perspective, scalable and GMP-compliant manufacturing processes, batch-to-batch reproducibility, and standardized regulatory evaluation frameworks remain critical challenges. Addressing these issues through interdisciplinary collaboration among pharmaceutical scientists, neuroscientists, clinicians, and regulatory bodies will be key to advancing polymeric silver nanocarriers from experimental systems to clinically viable treatments.
In conclusion, polymeric silver nanocarriers hold substantial promise as next-generation neurotherapeutic platforms. With continued refinement in safety, targeting efficiency, and regulatory alignment, these systems may contribute meaningfully to disease-modifying strategies for Alzheimer’s disease and other neurodegenerative disorders.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
All authors have contributed equally
CONFLICT OF INTERESTS
Declared none
REFERENCES
Lamptey RN, Chaulagain B, Trivedi R, Gothwal A, Layek B, Singh J. A review of the common neurodegenerative disorders: current therapeutic approaches and the potential role of nanotherapeutics. Int J Mol Sci. 2022 Feb 6;23(3):1851. doi: 10.3390/ijms23031851, PMID 35163773.
Erkkinen MG, Kim MO, Geschwind MD. Clinical neurology and epidemiology of the major neurodegenerative diseases. Cold Spring Harb Perspect Biol. 2018 Apr;10(4):a033118. doi: 10.1101/cshperspect.a033118, PMID 28716886.
Lane CA, Hardy J, Schott JM. Alzheimer’s disease. Eur J Neurol. 2018 Jan 19;25(1):59-70. doi: 10.1111/ene.13439, PMID 28872215.
DeTure MA, Dickson DW. The neuropathological diagnosis of Alzheimer’s disease. Mol Neurodegener. 2019 Dec 2;14(1):32. doi: 10.1186/s13024-019-0333-5, PMID 31375134.
Simonian NA, Coyle JT. Oxidative stress in neurodegenerative diseases. Annu Rev Pharmacol Toxicol. 1996 Apr;36(1):83-106. doi: 10.1146/annurev.pa.36.040196.000503, PMID 8725383.
Wu D, Chen Q, Chen X, Han F, Chen Z, Wang Y. The blood–brain barrier: structure regulation and drug delivery. Signal Transduct Target Ther. 2023 May 25;8(1):217. doi: 10.1038/s41392-023-01481-w, PMID 37231000.
Appelboom G, Detappe A, Lo Presti M, Kunjachan S, Mitrasinovic S, Goldman S. Stereotactic modulation of blood–brain barrier permeability to enhance drug delivery. Neuro Oncol. 2016 Dec;18(12):1601-9. doi: 10.1093/neuonc/now137, PMID 27407134.
Ahlawat J, Guillama Barroso G, Masoudi Asil S, Alvarado M, Armendariz I, Bernal J. Nanocarriers as potential drug delivery candidates for overcoming the blood–brain barrier: challenges and possibilities. ACS Omega. 2020 Jun 9;5(22):12583-95. doi: 10.1021/acsomega.0c01592, PMID 32548442.
Saraiva C, Praca C, Ferreira R, Santos T, Ferreira L, Bernardino L. Nanoparticle-mediated brain drug delivery: overcoming blood–brain barrier to treat neurodegenerative diseases. J Control Release. 2016 Aug;235:34-47. doi: 10.1016/j.jconrel.2016.05.044, PMID 27208862.
Lee D, Minko T. Nanotherapeutics for nose-to-brain drug delivery: an approach to bypass the blood brain barrier. Pharmaceutics. 2021 Nov 30;13(12):2049. doi: 10.3390/pharmaceutics13122049, PMID 34959331.
Awad R, Avital A, Sosnik A. Polymeric nanocarriers for nose-to-brain drug delivery in neurodegenerative diseases and neurodevelopmental disorders. Acta Pharm Sin B. 2023 May;13(5):1866-86. doi: 10.1016/j.apsb.2022.07.003, PMID 37250152.
Mulvihill JJ, Cunnane EM, Ross AM, Duskey JT, Tosi G, Grabrucker AM. Drug delivery across the blood–brain barrier: recent advances in the use of nanocarriers. Nanomedicine (Lond). 2020 Jan 9;15(2):205-14. doi: 10.2217/nnm-2019-0367, PMID 31916480.
Tsou YH, Zhang XQ, Zhu H, Syed S, Xu X. Drug delivery to the brain across the blood–brain barrier using nanomaterials. Small. 2017 Nov 16;13(43):1701921. doi: 10.1002/smll.201701921, PMID 29045030.
Youssif KA, Haggag EG, Elshamy AM, Rabeh MA, Gabr NM, Seleem A. Anti-alzheimer potential metabolomic profiling and molecular docking of green synthesized silver nanoparticles of Lampranthus coccineus and Malephora lutea aqueous extracts. PLOS One. 2019 Nov 6;14(11):e0223781. doi: 10.1371/journal.pone.0223781, PMID 31693694.
Ramirez Acosta CM, Cifuentes J, Castellanos MC, Moreno RJ, Munoz Camargo C, Cruz JC. PH-responsive cell-penetrating core/shell magnetite/silver nanoparticles for the delivery of plasmids: preparation characterization and preliminary in vitro evaluation. Pharmaceutics. 2020 Jun 17;12(6):561. doi: 10.3390/pharmaceutics12060561, PMID 32560390.
Afzal O, Altamimi AS, Ather H, Sultana S, Almalki WH, Bharti P, Mir Najib Ullah SN. Nanomedicine in the management of Alzheimer’s disease: state-of-the-art. Biomedicines. 2023;11(6):1752. doi: 10.3390/biomedicines11061752.
Zhang QL, Fu BM, Zhang ZJ. Borneol a novel agent that improves central nervous system drug delivery by enhancing blood–brain barrier permeability. Drug Deliv. 2017 Jan 1;24(1):1037-44. doi: 10.1080/10717544.2017.1346002, PMID 28687052.
Reitz C. Alzheimer’s disease and the amyloid cascade hypothesis: a critical review. Int J Alzheimers Dis. 2012;2012:369808. doi: 10.1155/2012/369808, PMID 22506132.
Ju Y, Tam KY. Pathological mechanisms and therapeutic strategies for Alzheimer’s disease. Neural Regen Res. 2022;17(3):543-9. doi: 10.4103/1673-5374.320970, PMID 34380884.
Monteiro AR, Barbosa DJ, Remiao F, Silva R. Alzheimer’s disease: insights and new prospects in disease pathophysiology biomarkers and disease-modifying drugs. Biochem Pharmacol. 2023 May;211:115522. doi: 10.1016/j.bcp.2023.115522, PMID 36996971.
Tiwari S, Atluri V, Kaushik A, Yndart A, Nair M. Alzheimer’s disease: pathogenesis diagnostics and therapeutics. Int J Nanomedicine. 2019 Jul;14:5541-54. doi: 10.2147/IJN.S200490, PMID 31410002.
Wechsler ME, Ramirez JE, Peppas NA. 110th anniversary: nanoparticle mediated drug delivery for the treatment of Alzheimer’s disease: crossing the blood–brain barrier. Ind Eng Chem Res. 2019 Aug 21;58(33):15079-87. doi: 10.1021/acs.iecr.9b02196, PMID 32982041.
Bakota L, Brandt R. Tau biology and tau-directed therapies for Alzheimer’s disease. Drugs. 2016 Mar 4;76(3):301-13. doi: 10.1007/s40265-015-0529-0, PMID 26729186.
Chun W, Johnson GV. The role of tau phosphorylation and cleavage in neuronal cell death. Front Biosci. 2007;12(1):733-56. doi: 10.2741/2097, PMID 17127334.
Boutajangout A, Wisniewski T. Tau-based therapeutic approaches for Alzheimer’s disease a mini-review. Gerontology. 2014;60(5):381-5. doi: 10.1159/000358875, PMID 24732638.
Calvo Rodriguez M, Kharitonova EK, Bacskai BJ. Therapeutic strategies to target calcium dysregulation in Alzheimer’s disease. Cells. 2020 Nov 20;9(11):2513. doi: 10.3390/cells9112513, PMID 33233678.
Lafay Chebassier C, Paccalin M, Page G, Barc Pain S, Perault Pochat MC, Gil R. mTOR/p70S6k signalling alteration by Abeta exposure as well as in APP-PS1 transgenic models and in patients with Alzheimer’s disease. J Neurochem. 2005 Jul 10;94(1):215-25. doi: 10.1111/j.1471-4159.2005.03187.x, PMID 15953364.
Zhang Z, Yang X, Song YQ, Tu J. Autophagy in Alzheimer’s disease pathogenesis: therapeutic potential and future perspectives. Ageing Res Rev. 2021 Dec;72:101464. doi: 10.1016/j.arr.2021.101464, PMID 34551326.
Liu J, Li L. Targeting autophagy for the treatment of Alzheimer’s disease: challenges and opportunities. Front Mol Neurosci. 2019 Aug 22;12:203. doi: 10.3389/fnmol.2019.00203, PMID 31507373.
Jantas D, Lason W. Preclinical evidence for the interplay between oxidative stress and RIP1-dependent cell death in neurodegeneration: state of the art and possible therapeutic implications. Antioxidants (Basel). 2021 Sep 24;10(10):1518. doi: 10.3390/antiox10101518, PMID 34679652.
Luissint AC, Artus C, Glacial F, Ganeshamoorthy K, Couraud PO. Tight junctions at the blood brain barrier: physiological architecture and disease-associated dysregulation. Fluids Barriers CNS. 2012 Dec 9;9(1):23. doi: 10.1186/2045-8118-9-23, PMID 23140302.
Kutuzov N, Flyvbjerg H, Lauritzen M. Contributions of the glycocalyx endothelium and extravascular compartment to the blood–brain barrier. Proc Natl Acad Sci USA. 2018 Oct 2;115(40):E9429-38. doi: 10.1073/pnas.1802155115, PMID 30217895.
Singh SA, Vellapandian C. Structure of the blood brain barrier and its role in the transporters for the movement of substrates across the barriers. Curr Drug Metab. 2023 Aug 4;24(4):250-69. doi: 10.2174/1389200224666230608110349, PMID 37291784.
Ruck T, Bittner S, Meuth SG. Blood–brain barrier modeling: challenges and perspectives. Neural Regen Res. 2015;10(6):889-91. doi: 10.4103/1673-5374.158342, PMID 26199600.
Dong X. Current strategies for brain drug delivery. Theranostics. 2018;8(6):1481-93. doi: 10.7150/thno.21254, PMID 29556336.
Haumann R, Videira JC, Kaspers GJ, Van Vuurden DG, Hulleman E. Overview of current drug delivery methods across the blood–brain barrier for the treatment of primary brain tumors. CNS Drugs. 2020 Nov 23;34(11):1121-31. doi: 10.1007/s40263-020-00766-w, PMID 32965590.
Naqvi S, Panghal A, Flora SJ. Nanotechnology: a promising approach for delivery of neuroprotective drugs. Front Neurosci. 2020 Jun 9;14:494. doi: 10.3389/fnins.2020.00494, PMID 32581676.
Satapathy MK, Yen TL, Jan JS, Tang RD, Wang JY, Taliyan R. Solid lipid nanoparticles (SLNs): an advanced drug delivery system targeting brain through BBB. Pharmaceutics. 2021 Jul 31;13(8):1183. doi: 10.3390/pharmaceutics13081183, PMID 34452143.
Masserini M. Nanoparticles for brain drug delivery. ISRN Biochem. 2013 May 21;2013:238428. doi: 10.1155/2013/238428, PMID 25937958.
Pulgar VM. Transcytosis to cross the blood brain barrier new advancements and challenges. Front Neurosci. 2019 Jan 11;12:1019. doi: 10.3389/fnins.2018.01019, PMID 30686985.
Busquets MA, Espargaro A, Sabate R, Estelrich J. Magnetic nanoparticles cross the blood–brain barrier: when physics rises to a challenge. Nanomaterials (Basel). 2015 Dec 11;5(4):2231-48. doi: 10.3390/nano5042231, PMID 28347118.
Moura RP, Martins C, Pinto S, Sousa F, Sarmento B. Blood–brain barrier receptors and transporters: an insight on their function and how to exploit them through nanotechnology. Expert Opin Drug Deliv. 2019 Mar 4;16(3):271-85. doi: 10.1080/17425247.2019.1583205, PMID 30767695.
Roda F, Caraffi R, Picciolini S, Tosi G, Vandelli MA, Ruozi B. Recent advances on surface-modified GBM targeted nanoparticles: targeting strategies and surface characterization. Int J Mol Sci. 2023 Jan 27;24(3):2496. doi: 10.3390/ijms24032496, PMID 36768820.
Barar J, Rafi MA, Pourseif MM, Omidi Y. Blood–brain barrier transport machineries and targeted therapy of brain diseases. BioImpacts. 2016 Dec 5;6(4):225-48. doi: 10.15171/bi.2016.30, PMID 28265539.
Lajoie JM, Shusta EV. Targeting receptor-mediated transport for delivery of biologics across the blood–brain barrier. Annu Rev Pharmacol Toxicol. 2015 Jan 6;55(1):613-31. doi: 10.1146/annurev-pharmtox-010814-124852, PMID 25340933.
Van Schaik PE, Zuhorn IS, Baron W. Targeting fibronectin to overcome remyelination failure in multiple sclerosis: the need for brain- and lesion-targeted drug delivery. Int J Mol Sci. 2022 Jul 29;23(15):8418. doi: 10.3390/ijms23158418, PMID 35955549.
Sim TM, Tarini D, Dheen ST, Bay BH, Srinivasan DK. Nanoparticle-based technology approaches to the management of neurological disorders. Int J Mol Sci. 2020 Aug 23;21(17):6070. doi: 10.3390/ijms21176070, PMID 32842530.
Huang CL, Hsiao IL, Lin HC, Wang CF, Huang YJ, Chuang CY. Silver nanoparticles affect on gene expression of inflammatory and neurodegenerative responses in mouse brain neural cells. Environ Res. 2015 Jan;136:253-63. doi: 10.1016/j.envres.2014.11.006, PMID 25460644.
De Jong WH, Borm PJ. Drug delivery and nanoparticles:applications and hazards. Int J Nanomedicine. 2008 Jun;3(2):133-49. doi: 10.2147/ijn.s596, PMID 18686775.
Lin HC, Ho MY, Tsen CM, Huang CC, Wu CC, Huang YJ. From the cover: comparative proteomics reveals silver nanoparticles alter fatty acid metabolism and amyloid beta clearance for neuronal apoptosis in a triple cell coculture model of the blood–brain barrier. Toxicol Sci. 2017 Jul;158(1):151-63. doi: 10.1093/toxsci/kfx079, PMID 28460142.
Wong KH, Riaz MK, Xie Y, Zhang X, Liu Q, Chen H. Review of current strategies for delivering Alzheimer’s disease drugs across the blood–brain barrier. Int J Mol Sci. 2019 Jan 17;20(2):381. doi: 10.3390/ijms20020381, PMID 30658419.
Wong XY, Sena Torralba A, Alvarez Diduk R, Muthoosamy K, Merkoci A. Nanomaterials for nanotheranostics: tuning their properties according to disease needs. ACS Nano. 2020 Mar 24;14(3):2585-627. doi: 10.1021/acsnano.9b08133, PMID 32031781.
Hu L, Tao Y, Jiang Y, Qin F. Recent progress of nanomedicine in the treatment of Alzheimer’s disease. Front Cell Dev Biol. 2023 Jun 29;11:1228679. doi: 10.3389/fcell.2023.1228679, PMID 37457297.
Karimi M, Ghasemi A, Sahandi Zangabad P, Rahighi R, Moosavi Basri SM, Mirshekari H. Smart micro/nanoparticles in stimulus-responsive drug/gene delivery systems. Chem Soc Rev. 2016;45(5):1457-501. doi: 10.1039/C5CS00798D, PMID 26776487.
Kumar N, Desagani D, Chandran G, Ghosh NN, Karthikeyan G, Waigaonkar S. Biocompatible agarose-chitosan coated silver nanoparticle composite for soft tissue engineering applications. Artif Cells Nanomed Biotechnol. 2018 Apr 3;46(3):637-49. doi: 10.1080/21691401.2017.1337021, PMID 28639839.
Rocha CV, Goncalves V, Da Silva MC, Banobre Lopez M, Gallo J. PLGA-based composites for various biomedical applications. Int J Mol Sci. 2022 Feb 12;23(4):2034. doi: 10.3390/ijms23042034, PMID 35216149.
Karthivashan G, Ganesan P, Park SY, Kim JS, Choi DK. Therapeutic strategies and nano-drug delivery applications in management of ageing Alzheimer’s disease. Drug Deliv. 2018 Jan 1;25(1):307-20. doi: 10.1080/10717544.2018.1428243, PMID 29350055.
Mathur P, Jha S, Ramteke S, Jain NK. Pharmaceutical aspects of silver nanoparticles. Artif Cells Nanomed Biotechnol. 2018 Oct 31;46(Suppl 1):115-26. doi: 10.1080/21691401.2017.1414825, PMID 29231755.
Huang KS, Wang LS, Wang CY, Yang CH, Hsieh CL, Chen SY. Synthesis and anti-fungal effect of silver nanoparticlesandamp; ndash; chitosan composite particles. Int J Nanomedicine. 2015 Apr 1;10:2685-96. doi: 10.2147/IJN.S77410.
Bhattacharya T, Soares GA, Chopra H, Rahman MM, Hasan Z, Swain SS. Applications of phyto-nanotechnology for the treatment of neurodegenerative disorders. Materials (Basel). 2022 Jan 21;15(3):804. doi: 10.3390/ma15030804, PMID 35160749.
Ege D. Action mechanisms of curcumin in Alzheimer’s disease and its brain targeted delivery. Materials (Basel). 2021 Jun 16;14(12):3332. doi: 10.3390/ma14123332, PMID 34208692.
Fan S, Zheng Y, Liu X, Fang W, Chen X, Liao W. Curcumin-loaded PLGA-PEG nanoparticles conjugated with B6 peptide for potential use in Alzheimer’s disease. Drug Deliv. 2018 Jan 1;25(1):1091-102. doi: 10.1080/10717544.2018.1461955, PMID 30107760.
Song C, Shi J, Zhang P, Zhang Y, Xu J, Zhao L. Immunotherapy for Alzheimer’s disease: targeting β-amyloid and beyond. Transl Neurodegener. 2022 Dec 18;11(1):18. doi: 10.1186/s40035-022-00292-3, PMID 35300725.
Maiti P, Dunbar GL. Use of curcumin a natural polyphenol for targeting molecular pathways in treating age-related neurodegenerative diseases. Int J Mol Sci. 2018 May 31;19(6):1637. doi: 10.3390/ijms19061637, PMID 29857538.
Chung IM, Park I, Seung Hyun K, Thiruvengadam M, Rajakumar G. Plant-mediated synthesis of silver nanoparticles: their characteristic properties and therapeutic applications. Nanoscale Res Lett. 2016 Dec 28;11(1):40. doi: 10.1186/s11671-016-1257-4, PMID 26821160.
Karthikeyan A, Senthil N, Min T. Nanocurcumin: a promising candidate for therapeutic applications. Front Pharmacol. 2020 May 1;11:487. doi: 10.3389/fphar.2020.00487, PMID 32425772.
Bhaskar S, Tian F, Stoeger T, Kreyling W, De La Fuente JM, Grazu V. Multifunctional nanocarriers for diagnostics drug delivery and targeted treatment across blood–brain barrier: perspectives on tracking and neuroimaging. Part Fibre Toxicol. 2010 Dec 3;7(1):3. doi: 10.1186/1743-8977-7-3, PMID 20199661.
Cano A, Turowski P, Ettcheto M, Duskey JT, Tosi G, Sanchez Lopez E. Nanomedicine-based technologies and novel biomarkers for the diagnosis and treatment of Alzheimer’s disease: from current to future challenges. J Nanobiotechnology. 2021 Apr 29;19(1):122. doi: 10.1186/s12951-021-00864-x, PMID 33926475.
Sukhanova A, Bozrova S, Sokolov P, Berestovoy M, Karaulov A, Nabiev I. Dependence of nanoparticle toxicity on their physical and chemical properties. Nanoscale Res Lett. 2018 Dec 7;13(1):44. doi: 10.1186/s11671-018-2457-x, PMID 29417375.
Begines B, Ortiz T, Perez Aranda M, Martinez G, Merinero M, Arguelles Arias F. Polymeric nanoparticles for drug delivery: recent developments and future prospects. Nanomaterials (Basel). 2020 Jul 19;10(7):1403. doi: 10.3390/nano10071403, PMID 32707641.
Shumbula NP, Nkabinde SS, Ndala ZB, Mpelane S, Shumbula MP, Mdluli PS. Evaluating the antimicrobial activity and cytotoxicity of polydopamine capped silver and silver/polydopamine core-shell nanocomposites. Arab J Chem. 2022 Jun;15(6):103798. doi: 10.1016/j.arabjc.2022.103798.
Simko M, Mattsson MO. Risks from accidental exposures to engineered nanoparticles and neurological health effects: a critical review. Part Fibre Toxicol. 2010;7(1):42. doi: 10.1186/1743-8977-7-42, PMID 21176150.
Gandhi S, Shastri DH, Shah J, Nair AB, Jacob S. Nasal delivery to the brain: harnessing nanoparticles for effective drug transport. Pharmaceutics. 2024 Apr 1;16(4):481. doi: 10.3390/pharmaceutics16040481, PMID 38675142.
Dighe S, Jog S, Momin M, Sawarkar S, Omri A. Intranasal drug delivery by nanotechnology: advances in and challenges for Alzheimer’s disease management. Pharmaceutics. 2023 Dec 29;16(1):58. doi: 10.3390/pharmaceutics16010058, PMID 38258068.
Alexander A, Saraf S. Nose-to-brain drug delivery approach: a key to easily accessing the brain for the treatment of Alzheimer’s disease. Neural Regen Res. 2018;13(12):2102-4. doi: 10.4103/1673-5374.241458, PMID 30323136.
Al-azzawi S, Masheta D, Guildford AL, Phillips G, Santin M. Dendrimeric poly(epsilon-lysine) delivery systems for the enhanced permeability of flurbiprofen across the blood-brain barrier in Alzheimer’s disease. Int J Mol Sci. 2018 Oct 18;19(10):3224. doi: 10.3390/ijms19103224, PMID 30340406.
Sonvico F, Clementino A, Buttini F, Colombo G, Pescina S, Staniscuaski Guterres S. Surface-modified nanocarriers for nose-to-brain delivery: from bioadhesion to targeting. Pharmaceutics. 2018 Mar 15;10(1):34. doi: 10.3390/pharmaceutics10010034, PMID 29543755.
Mittal D, Ali A, Md S, Baboota S, Sahni JK, Ali J. Insights into direct nose to brain delivery: current status and future perspective. Drug Deliv. 2014 Mar 9;21(2):75-86. doi: 10.3109/10717544.2013.838713, PMID 24102636.
Islam SU, Shehzad A, Ahmed MB, Lee YS. Intranasal delivery of nanoformulations: a potential way of treatment for neurological disorders. Molecules. 2020;25(8):1929. doi: 10.3390/molecules25081929, PMID 32326318.
Prabhu S, Poulose EK. Silver nanoparticles: mechanism of antimicrobial action synthesis medical applications and toxicity effects. Int Nano Lett. 2012 Dec 29;2(1):32. doi: 10.1186/2228-5326-2-32.
Del Amo L, Cano A, Ettcheto M, Souto EB, Espina M, Camins A. Surface functionalization of PLGA nanoparticles to increase transport across the BBB for Alzheimer’s disease. Appl Sci. 2021 May 10;11(9):4305. doi: 10.3390/app11094305.
Desai N. Challenges in development of nanoparticle-based therapeutics. AAPS J. 2012 Jun 10;14(2):282-95. doi: 10.1208/s12248-012-9339-4, PMID 22407288.
Oberdorster G. Safety assessment for nanotechnology and nanomedicine: concepts of nanotoxicology. J Intern Med. 2010 Jan 11;267(1):89-105. doi: 10.1111/j.1365-2796.2009.02187.x, PMID 20059646.