
Int J Curr Pharm Res, Vol 18, Issue 2, 77-81Original Article
ANTIMICROBIAL PRESCRIPTION PRACTICES AMONG THE INPATIENTS OF E. N. T DEPARTMENT USING WHO AWARE CLASSIFICATION: A CROSS-SECTIONAL STUDY
K. G. SATHEESH KUMAR, UPPU BHARATHI*, L. MEENAKSHI, M. ASHA LATHA
Department of Pharmacology, Sri Venkateswara Medical College (SVMC), Tirupati, India
*Corresponding author: Uppu Bharathi; *Email: ubharathi1212@gmail.com
Received: 10 Nov 2025, Revised and Accepted: 03 Jan 2026
ABSTRACT
Objective: To collect antimicrobial prescriptions of patients admitted to the E. N. T ward. To analyze the collected antimicrobial prescriptions of E. N. T inpatients based on the WHO AWaRe classification.
Methods: An observational cross-sectional study was conducted in the Department of E. N. T at a tertiary care hospital in Tirupati, Andhra Pradesh, from January to June 2025, after obtaining approval from the Institutional Ethics Committee. All inpatient prescriptions containing at least one antibiotic, irrespective of age, gender, or indication, were included. Written informed consent was obtained from adult participants, and assent from parents for minors. The original prescriptions were returned after documentation, and photocopies or photographs were analyzed. Antibiotics were classified according to the WHO AWaRe classification (2023), and WHO core prescribing indicators were applied to assess rational drug use.
Results: Out of 310 prescriptions analyzed, Amoxyclav (82.58%) from the Access group was most frequently prescribed, followed by Watch group antibiotics-Ceftriaxone (13.55%) and Cefixime (3.87%). No Reserve antibiotics were used. The average number of drugs per encounter was 5.13, and 100% of prescriptions contained antibiotics. Injections were used in 86.4%, 97% of drugs were from the Essential Medicine List, and 100% were prescribed by generic name.
Conclusion: The predominance of Access group antibiotics indicates partial adherence to WHO guidelines; however, polypharmacy and high antibiotic use highlight the need to strengthen antimicrobial stewardship in the E. N. T department.
Keywords: Antimicrobial resistance, AWaRe classification, Prescription audit, E. N. T inpatients, Rational drug use
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i2.8069 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
In the recent years, irrational prescription and increased use of anti-biotics has resulted in worldwide problem of antimicrobial resistance (AMR) [1]. AMR is a serious danger sign for general public health [2]. In 2019 alone, antibiotic-resistant infections were the direct cause of 1.27 million deaths and a contributing factor in nearly 5 million additional deaths. If this growing threat isn't addressed soon, experts warn that drug-resistant infections could claim up to 10 million lives every year by 2050 [3].
Rising rates of antimicrobial resistance can result in longer hospital stay and increased treatment duration, as well as higher treatment expenses and mortality [4]. To manage these problems World Health Organization (WHO) introduced the “Access”, “Watch”, and “Reserve” (AWaRe) classification of antibiotics [5]. The revised 2023 AWaRe framework encompasses over 298 antibiotics, including 41 classified as essential, and provides clear, practical guidance to support clinicians in selecting appropriate treatments in both primary care and hospital settings [6]. Access category contains the narrow spectrum antibiotics and it will be considered for the first choice of treatment, mostly this category antibiotic shows less resistance. Watch category contains the broad-spectrum antibiotics which are all having a more resistance pattern compared to the Access category. Careful selection of the anti-biotic is needed while choosing the watch category anti-biotics due to their increased resistance property. Reserve category antibiotics are used as last resort treatment option in which Access and Watch category antibiotics are not at all useful or not indicated. Reserve category anti-biotics mostly used for high priority cases after assessing the risk-benefit ratio [7].
Studies have shown that raising awareness and adherence to AWaRe guidelines leads to higher Access antibiotic use and significant reductions in Watch and Reserve prescriptions [8]. To effectively combat AMR, it is crucial to assess prescribing patterns, which provide the foundation for targeted improvements [9]. Implementation of antimicrobial stewardship programs, including education on AWaRe, has been proven to reduce the emergence of multidrug-resistant organisms and optimize antibiotic use [10].
The otorhinolaryngology (ENT) department frequently encounters infections of the ear, nose, throat, and related structures, many of which are treated empirically with antibiotics. Despite its importance, limited data are available on antibiotic prescribing trends within ENT department, particularly among in-patients where infection burden and antibiotic exposure are high. Therefore, this study was planned to understand current antimicrobial prescription practices among in-patients in the ENT department, assess their appropriateness as per the WHO AWaRe classification, and provide evidence-based recommendations to promote rational antimicrobial use.
MATERIALS AND METHODS
An observational study was conducted in Department of E. N. T at tertiary care hospital in Tirupati, Andra Pradesh, from January to June 2025 after obtaining clearance from Institutional Ethics Committee.
Inclusion criteria
All prescriptions containing at least one antibiotic, prescribed to both genders, irrespective of age, regardless of the indication admitted in the ENT ward were considered as eligibility criteria to get participated in the study.
Before start of the study, purpose of the study was explained to eligible participants and written informed consent was obtained from the patients enrolled in the study, and assent was taken from parents if the patient was under 18 y of age. The original prescriptions were returned to the patients, and photocopies or clear photographs of the prescriptions were taken and analysed using the WHO AWaRe classification of antibiotics.
RESULTS
In the Department of ENT during the study period, 310 patient prescriptions were taken for the analysis in that almost all the patients are received antibiotics and the results were as follows
Demographic details of study participants:
Table 1: Demographic data
| Age group (in years) | Number (%) |
| <18 y | 34 (11%) |
| 18-59 y | 214 (69%) |
| >59 y | 62 (20%) |
| mean age | 41.48±18.79 |
As represented in table 1, majority of patients were in the adult age group 18–59 y.
Gender distribution of study participants
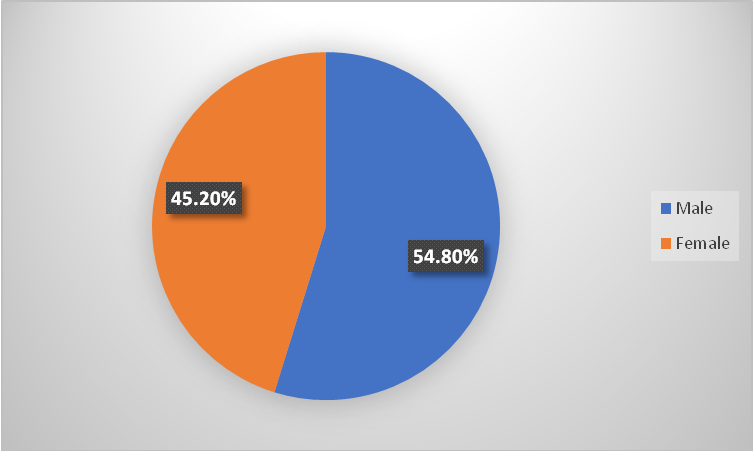
Fig. 1: Gender distribution
Fig. 1 illustrates the gender distribution shows higher proportion of males (54.8%) were prescribed antimicrobials compared to females.
Age wise distribution of antimicrobials
Table 2: Age-wise distribution of antimicrobials
| Age in years | Amoxiclav | Cefixime | Ceftriaxone |
| <18 y | 32 (10.3%) | 02(0.64%) | 0 |
| 18-59 y | 170 (54.8%) | 08 (2.5%) | 36 (11.6%) |
| >59 y | 54 (17.4%) | 02 (0.64%) | 06 (1.9%) |
As represented in table 2, Amoxyclav was the most frequently prescribed antibiotic across all age groups, especially in adults (54.8%).
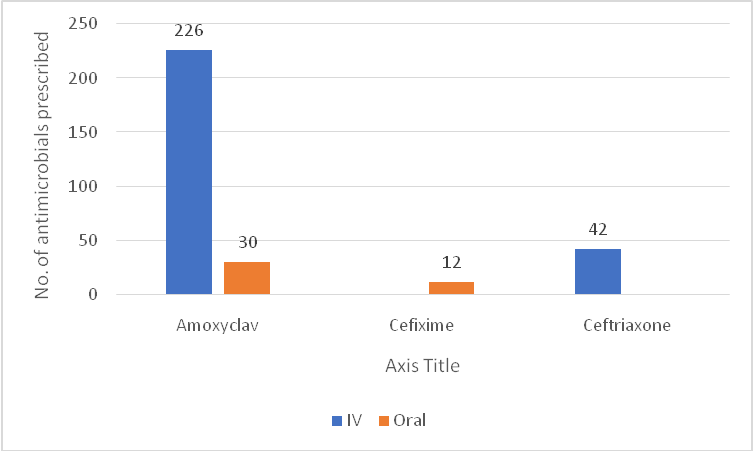
Fig. 2: Route of administration, as represented in fig. 2, Amoxyclav was predominantly administered intravenously (IV)
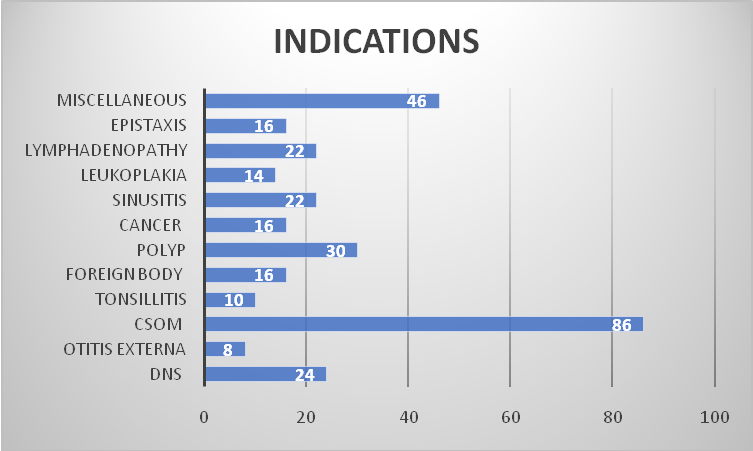
Fig. 3: Indications of antimicrobials
As represented in fig. 3, the most frequent indication for prescription of antimicrobials was Chronic Suppurative Otitis Media (CSOM)/Chronic Otitis Media (COM), accounting for 86 cases (27.7%), followed by Miscellaneous ENT disorders (46 cases, 14.8%) and Deviated Nasal Septum (DNS) with 24 cases (7.7%).
Table 3: Prescribing patterns of antibiotics according to WHO AWaRe classification
| Antibiotic name | Access, n (%) | Watch, n (%) | Reserve, n (%) |
| Amoxyclav | 256 (82.58%) | - | - |
| Cefixime | - | 12 (3.87%) | - |
| Ceftriaxone | - | 42 (13.55%) | - |
As represented in table 3, Amoxyclav, which falls under the Access group, accounted for majority of prescriptions (82.58%).
Table 4: “WHO” core prescribing indicators used to assess study prescriptions
| Prescribing indicators assessed | N (%) | Standard value (%) |
| Average number of drugs per encounter | 5.135 | 1.6-1.8 |
| Percent of prescriptions with antibiotics | 100 | 20-26.8 |
| Percent of prescriptions with injections | 86.4 | 13.4-24.1 |
| Percent of drugs prescribed in generic name(n=1592) | 100 | 100 |
| Percent of drugs prescribed from essential medicine list(n=1544) | 97 | 100 |
The average number of drugs per prescription was 5.135, which is significantly above the WHO recommended range of 1.6–1.8%. The percentage of prescriptions with antibiotics was 100%, far exceeding the WHO standard of 20-26.8. Injection use was high (86.4%), far above the WHO recommendation of 13.4-24. On a positive note, 100% of drugs were prescribed by generic name, and 97% were from the Essential Medicines List (EML), indicating good adherence to rational prescribing practices in terms of drug selection.
DISCUSSION
The present study analysed 310 patients’ prescriptions, amongst which most of the prescriptions were adults (18–59 y) suggests that working-age individuals are more commonly affected by ENT infections. A similar trend was seen in Onay O et al., from a tertiary care hospital in North Kashmir, where the majority of patients (33.65%) were between 41 and 60 y old. This could be due to their greater exposure to environmental pollutants, occupational risks, and lifestyle-related factors that are more common in this age group [12, 13].
In our study, a greater proportion of male patients (54.8%) were prescribed antimicrobials compared to female patients. A similar trend was observed in Manouchehr Khanipour et al., from a tertiary care hospital in India, where 56.87% of patients who received antibiotics were male [14]. This pattern could be due to gender-related differences in healthcare-seeking behavior, higher occupational exposure, and a greater incidence of certain ENT conditions among men. On the other hand, findings from Abraham JP et al., showed that women were prescribed a higher proportion of generic drugs and received fewer antibiotics than men (22.22% vs. 33.46%). This highlights how prescription practices can vary significantly depending on regional settings, institutional policies, and patient profiles [15].
In the present study, chronic suppurative otitis media (CSOM) emerged as the most common ENT disorder, aligning with earlier Indian studies that attribute its persistence to poor ear hygiene and delayed medical care. Deviated nasal septum (DNS) and sinonasal polyps were also frequently observed, likely influenced by allergic reactions and environmental pollutants. Similar trends were reported by Gour et al. (2019) and Rai et al. (2020), where CSOM and nasal pathologies constituted the major disease load in ENT outpatient departments [16, 17]. The notable incidence of lymphadenopathy and oral cavity cancers in our study further underscores the rising burden of tobacco-related conditions. Overall, these findings highlight the importance of early screening, community awareness, and preventive strategies to reduce the long-term complications of chronic ENT diseases.
In our study, Amoxicillin-Clavulanate (Amoxyclav) was mainly given intravenously, even though it’s also available as an oral medication. This likely reflects a clinical preference for IV administration in hospitalized patients, possibly because their infections were more severe or because IV delivery provides faster and more reliable drug levels. Similarly, Ceftriaxone, a third-generation cephalosporin, was used exclusively via the IV route, which is consistent with standard hospital practices. Supporting this, Ain MR et al. reported that Amoxicillin-Clavulanate was the most commonly prescribed antibiotic in their study, making up 21.74% of all prescriptions, highlighting its prominent use in hospital settings [18]. This highlights a broader trend of responsible antibiotic use in ENT departments [19]. Similarly, studies by Priyadharsini RP et al. and Mandal P et al. from tertiary care hospitals in South India also found that Access group antibiotics, particularly Amoxicillin, were the most frequently prescribed. These consistent patterns across multiple institutions reinforce the growing adherence to the AWaRe classification framework and emphasize the importance of prioritizing Access group antibiotics in clinical practice to mitigate the development of resistance [20, 21].
In our study, we found that the average number of drugs prescribed per patient encounter was 5.135-well above the WHO-recommended range of 1.6 to 1.8. This indicates a clear tendency toward polypharmacy in the ENT inpatient setting. One possible reason could be the need to address multiple symptoms or coexisting health conditions in these patients. A similar observation was made by Varma JN et al., who analyzed 200 prescriptions in a tertiary care hospital in India [22]. Their findings revealed an even higher average of 6.94 drugs per prescription, with 83% of prescriptions containing two or more medications. This supports the notion that polypharmacy is a common practice in inpatient care across various settings. These findings highlight the importance of regularly reviewing prescribing habits to encourage more rational drug use. Overprescribing not only increases the risk of adverse drug interactions but also contributes to broader concerns like antimicrobial resistance. Careful monitoring can help strike a better balance between therapeutic necessity and medication safety [23].
In our study, antibiotics were prescribed in every single prescription-100%-which is significantly above the WHO-recommended range of 20% to 26.8%. This high rate of antibiotic use may reflect the genuine need to manage frequent or severe infections commonly seen in ENT inpatients. However, it could also indicate a tendency toward defensive prescribing, where antibiotics are used pre empirically to avoid potential complications. Our findings are supported by Bhattacharjee S et al., who reported an overall antibiotic prescription rate of 78.9%. In certain high-priority clinical areas, this fig. rise even further to 92.2%, highlighting a broader trend of extensive antibiotic use in inpatient settings. These observations raise important concerns about the rational use of antibiotics. Overprescribing not only increases the risk of drug resistance but also places patients at greater risk of side effects and unnecessary costs. This underscores the need for stricter antibiotic stewardship and regular prescription audits to ensure that antibiotics are used appropriately and judiciously [24].
Our study revealed an alarmingly high rate of injection use at 86.4%, which far exceeds the WHO-recommended range of 13.4% to 24%. This finding is consistent with Raghute LB et al., found that 51% of prescriptions included injectable drugs, with 41% containing one injectable and 8% containing two injectables. This rate significantly exceeds the WHO-recommended range of 13.4% to 24.1% for injectable drug prescriptions [25]. Our study showed strong adherence to rational prescribing practices, with all drugs (100%) written by their generic names and 97% selected from the Essential Medicines List (EML). This reflects a commendable standard of prescribing that supports cost-effective, accessible, and consistent treatment for patients.
The high level of adherence seen in our study may be the result of several positive factors, such as stricter institutional policies, focused training for prescribers, and improved availability of essential medicines. These are encouraging signs, as prescribing by generic name helps reduce treatment costs for both patients and the healthcare system. Similarly, choosing drugs from the Essential Medicines List ensures the use of medications that are proven to be safe, effective, and affordable. Overall, our findings highlight the value of ongoing education and regular monitoring to uphold and further strengthen rational prescribing practices. Maintaining these standards is crucial not only for achieving better treatment outcomes but also for reducing the risks and costs associated with irrational medication use in clinical settings.
CONCLUSION
Our study provides valuable insights into how antibiotics are prescribed in the ENT ward. Most patients were adults, and slightly more men received antimicrobial therapy, likely reflecting lifestyle and occupational exposures. Amoxicillin-Clavulanate and Ceftriaxone were the most commonly used antibiotics, often given intravenously, suggesting treatment of more severe infections. On a positive note, prescribers followed rational practices, with nearly all drugs written by their generic names and most selected from the Essential Medicines List. However, we observed some areas of concern: every patient received antibiotics, polypharmacy was common, and injectable use was high. These patterns highlight the ongoing need for antimicrobial stewardship, careful prescription reviews, and prescriber awareness to ensure antibiotics are used wisely and safely. Overall, the study demonstrates encouraging adherence to the AWaRe classification but also points to opportunities for improvement in aligning practice with WHO recommendations.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
All authors have contributed equally
CONFLICT OF INTERESTS
Declared none
REFERENCES
Appaneal HJ, Caffrey AR, Lopes V, Dosa D, La Plante KL. Antibiotic prescribing in outpatient settings: rural patients are more likely to receive fluoroquinolones and longer antibiotic courses. Antibiotics (Basel). 2023;12(2):224. doi: 10.3390/antibiotics12020224, PMID 36830137.
Mugada V, Mahato V, Andhavaram D, Vajhala SM. Evaluation of prescribing patterns of antibiotics using selected indicators for antimicrobial use in hospitals and the access watch reserve (AWaRe) classification by the world health organization. Turk J Pharm Sci. 2021 Jun 18;18(3):282-8. doi: 10.4274/tjps.galenos.2020.11456, PMID 34157817.
Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet. 2022 Feb 12;399(10325):629-55. doi: 10.1016/S0140-6736(21)02724-0, PMID 35065702.
Naylor NR, Silva S, Kulasabanathan K, Atun R, Zhu N, Knight GM. Methods for estimating the burden of antimicrobial resistance: a systematic literature review protocol. Syst Rev. 2016;5(1):187. doi: 10.1186/s13643-016-0364-8, PMID 27821153.
Mudenda S, Chilimboyi R, Matafwali SK, Daka V, Mfune RL, Kemgne LA. Hospital prescribing patterns of antibiotics in Zambia using the WHO prescribing indicators post-COVID-19 pandemic: findings and implications. JAC Antimicrob Resist. 2024 Feb 22;6(1):dlae023. doi: 10.1093/jacamr/dlae023, PMID 38389802.
Moja L, Zanichelli V, Mertz D, Gandra S, Cappello B, Cooke GS. WHO’s essential medicines and AWaRe: recommendations on first and second-choice antibiotics for empiric treatment of clinical infections. Clin Microbiol Infect. 2024 Apr;30(Suppl 2):S1-51. doi: 10.1016/j.cmi.2024.02.003, PMID 38342438.
World Health Organization. AWaRe classification of antibiotics. Geneva: World Health Organization; 2023. Available from: https://www.who.int/publications/i/item/9789240069182.
Abu Ajaleh S, Darwish Elhajji F, Al-Bsoul S, Abu Farha R, Al-Hammouri F, Amer A. An evaluation of the impact of increasing the awareness of the WHO access watch and reserve (AWaRe) antibiotics classification on knowledge, attitudes, and hospital antibiotic prescribing practices. Antibiotics (Basel). 2023 May 23;12(6):951. doi: 10.3390/antibiotics12060951, PMID 37370270.
Kalungia AC, Mukosha M, Mwila C, Banda D, Mwale M, Kagulura S. Antibiotic use and stewardship indicators in the first and second-level hospitals in Zambia: findings and implications for the future. Antibiotics (Basel). 2022 Nov 15;11(11):1626. doi: 10.3390/antibiotics11111626, PMID 36421270.
Khadse SN, Ugemuge S, Singh C. Impact of antimicrobial stewardship on reducing antimicrobial resistance. Cureus. 2023 Dec 4;15(12):e49935. doi: 10.7759/cureus.49935, PMID 38179391.
McEwen SA, Collignon PJ. Antimicrobial resistance: a one health perspective. Microbiol Spectr. 2018 Mar;6(2):ARBA‑0009‑2017. doi: 10.1128/microbiolspec.ARBA-0009-2017, PMID 29600770.
Aalasyam N, Potnuru S, Sileveru CM, Pokala U, Naveen P. Antimicrobial prescription pattern in ENT outpatient department in a Tertiary Care Teaching Hospital, Telangana, India. Int J Recent Surg Med Sci. 2024;10(Suppl 1):S28-34. doi: 10.1055/s-0043-1761502.
Onay O, Aydin C. The distribution of geriatric problems in otolaryngology and their alteration from young adults. Niger J Clin Pract. 2021 May;24(5):640-6. doi: 10.4103/njcp.njcp_509_19, PMID 34018971.
Khanipour M, Mohammadi MS, Ramaiah B. Evaluation of the antibiotic prescribing pattern and related adverse drug reaction in an India tertiary care hospital: a prospective observational study. Int J Pharm Investigation. 2023;13(3):625-31. doi: 10.5530/ijpi.13.3.077.
Abraham JP, Giri RR, Jaiswal KM. Assessing prescribing practices in a rural hospital: a cross-sectional study using WHO prescribing indicators. Indian J Pharm Pharmacol. 2024 May 15;11(1):38-43. doi: 10.18231/j.ijpp.2024.007.
Gour N, Kumar A, Sinha S, Agarwal S, Yadav N. Pattern of ENT diseases at a tertiary care hospital: a retrospective study. Indian J Otolaryngol Head Neck Surg. 2019;71(Suppl 3):1817-23. doi: 10.1007/s12070‑019‑01790‑z.
Rai A, Thapa N, Sharma A. Spectrum of ear nose and throat diseases in a tertiary care hospital: a descriptive study. J Nepal Health Res Counc. 2020;18(1):36-40. doi: 10.33314/jnhrc.v18i1.2782.
Ain MR, Shahzad N, Aqil M, Alam MS, Khanam R. Drug utilization pattern of antibacterials used in ear nose and throat outpatient and inpatient departments of a university hospital at New Delhi, India. J Pharm Bioallied Sci. 2010 Jan;2(1):8-12. doi: 10.4103/0975-7406.62695, PMID 21814423.
Dar MA, Kalsi S, Rehman SU. To evaluate drug utilization pattern among ENT patients in a Tertiary Care Hospital of North Kashmir. World J Pharm Pharm Sci. 2022 Aug;11(8):1622-38. doi: 10.20959/wjpps20228-22899.
Priyadharsini RP, Ramasamy K, Amarendar S. Antibiotic-prescribing pattern in the outpatient departments using the WHO prescribing indicators and AWaRe assessment tool in a tertiary-care hospital in South India. J Family Med Prim Care. 2022 Jan;11(1):74-8. doi: 10.4103/jfmpc.jfmpc_527_21, PMID 35309648.
Mandal P, Asad M, Kayal A, Biswas M. Assessment of use of World Health Organization access, watch, reserve antibiotics and core prescribing indicators in pediatric outpatients in a Tertiary Care Teaching Hospital in Eastern India. Perspect Clin Res. 2023 Apr-Jun;14(2):61-7. doi: 10.4103/picr.picr_22_22, PMID 37325582.
Varma JN, Kumar ES, Subair A, Sunil G, Swamy VB, Patil S. A cross-sectional study on drug utilization and its cost analysis in urological disorders and other health issues in patients of a tertiary care hospital. Indian J Pharm Pharmacol. 2023;10(4):309-18. doi: 10.18231/j.ijpp.2023.052.
Bhattacharjee S, Mothsara C, Shafiq N, Panda PK, Rohilla R, Kaore SN. Antimicrobial prescription patterns in tertiary care centres in India: a multicentric point prevalence survey. E Clinical Medicine. 2025 Mar 28;82:103175. doi: 10.1016/j.eclinm.2025.103175, PMID 40224675.
Raghute LB, Jaiswal KM, Dudhgoankar S, Turkar A, Jawade A, Vaishnao LS. A cross-sectional study assessing prescriptions of a tertiary care teaching institute of central India using the WHO core drug indicators. JMSH. 2019 Jun 10;5(1):1-6. doi: 10.46347/JMSH.2019.v05i01.001.
Meena DK, Mathaiyan J, Thulasingam M, Ramasamy K. Assessment of medicine use based on WHO drug-use indicators in public health facilities of the South Indian Union Territory. Br J Clin Pharmacol. 2022 May;88(5):2315-26. doi: 10.1111/bcp.15165, PMID 34859476.