
Int J Curr Pharm Res, Vol 18, Issue 2, 87-91Original Article
TREATMENT OPTIMIZATION IN RECURRENT DERMATOPHYTOSIS: A PROSPECTIVE, RANDOMIZED, COMPARATIVE STUDY OF EXTENDED-DURATION VERSUS HIGH-DOSE TERBINAFINE AND ITRACONAZOLE
RASHI SINGH1*, SAVITA VERMA2, SURABHI DAYAL3, SAPNA VASHISTH4
1,2,4Department of Pharmacology, Pt. B. D. Sharma, PGIMS, Rohtak, Haryana, India. 3Department of Dermatology, Venerology and Leprology, Pt. B. D. Sharma, PGIMS, Rohtak, Haryana
*Corresponding author: Rashi Sigh; *Email: rashinain@gmail.com
Received: 15 Nov 2025, Revised and Accepted: 03 Jan 2026
ABSTRACT
Objective: To compare efficacy and safety of terbinafine versus itraconazole administered in double dose and prolonged duration in patients with recurrent tinea corporis and cruris.
Methods: This prospective, randomized study included 166 patients in four groups: A (terbinafine 250 mg once daily (OD), 4 w), B (terbinafine 500 mg OD, 2 w), C (itraconazole 100 mg twice daily (BD), 4 w), and D (itraconazole 200 mg BD, 2 w). Primary outcomes were clinical/mycological cure, global assessment, safety; secondary outcomes included quality of life, escape treatment, adherence. Significance was set at p<0.05.
Results: Group C had highest efficacy (92.7% clinical cure and 100% mycological cure) with lowest recurrence. Liver Function Tests (LFTs) showed no significant abnormalities across all groups. No therapy discontinuations/serious Adverse Drug Reactions (ADRs) were reported. Dermatology Life Quality Index (DLQI) improved significantly from baseline to treatment end and 6 w post-treatment, indicating substantial and sustained improvement. Treatment compliance was high across all groups (≥90%), with no significant difference.
Conclusion: Itraconazole 100 mg BD for 4 w was most effective, well-tolerated regimen, highlighting benefit of extended duration over increased dose in recurrent dermatophytosis.
Keywords: Dermatophytosis, Recurrent, Terbinafine, Itraconazole, Mycological cure, Double dose, Extended duration, Efficacy, Safety, Quality of life, Adherence
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i2.8071 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Dermatophytosis affects more than 1 billion people worldwide, frequently failing to respond to standard antifungal therapies, including commonly used agents like terbinafine and itraconazole [1, 2]. The prevalence of chronic and recurrent dermatophytosis has significantly increased, with as many as 6.1-9.3% cases in community-based surveys to 61.5% cases in hospital-based studies experiencing relapses despite adherence to prescribed treatment regimens [3-5]. Dermatophytosis is considered to be recurrent when there is a re-occurrence of the disease (lesions) within a few weeks (<6 w) after completion of the treatment [6]. Recurrent dermatophytic infections are often attributed to inadequate treatment, environmental exposure, host susceptibility, etc [6]. Oral antimycotics such as terbinafine, itraconazole, and fluconazole are commonly employed for managing these infections [7]. There are no consensus guidelines for managing recurrent tinea corporis and cruris, leading dermatologists to rely on varied, trial-and-error approaches. This study aimed to generate evidence for an effective treatment regimen for patients with recurrent tinea corporis and cruris. This study provides comparative data on modified terbinafine and itraconazole regimens in recurrent dermatophytosis, highlighting the benefit of extended duration over increased dose. The findings offer practical guidance for optimizing antifungal therapy in routine clinical settings.
MATERIALS AND METHODS
Study design
This was a prospective, randomized, parallel comparative study conducted in a tertiary care hospital. Approval was taken from the Institutional Ethics Committee before the commencement of the study. The study was done in accordance with the principles of the Declaration of Helsinki and Good Clinical Practice (Indian-GCP). The study was registered with the Clinical Trials Registry-India before the enrolment of patients.
Study participants
Patients with recurrent tinea corporis and tinea cruris who attended the Outpatient Department of Dermatology, Venereology, and Leprology, were screened and enrolled in the study based on predefined inclusion and exclusion criteria. Written informed consent was obtained from all the patients before enrolment in the study. Eligible participants included individuals of either sex, aged between 18 and 65 y, who presented with a recurrence of tinea corporis or tinea cruris. Patients were excluded if they had dermatophytosis involving sites other than the body or groin, a history of hypersensitivity or intolerance to the study drugs, or any comorbidities affecting participation or outcomes, including other skin conditions (e. g., contact dermatitis, atopic dermatitis, psoriasis), pregnancy/lactation, and hepatic, renal, or cardiac disease.
Randomisation
The eligible patients were randomly divided into four study groups, i. e. Group A, Group B, Group C, and Group D, via randomizer. org.
Patients were assessed at baseline, treatment completion, and six weeks post-treatment.
Outcome measures
Primary outcomes
Clinical cure assessed by Four-Point Scale7 at baseline, end of treatment, and six weeks post-completion of treatment, and Composite Score7 at end of treatment.
Mycological cure assessed by Direct Microscopic Examination (Potassium hydroxide/KOH mount)6 at baseline, end of treatment, and six weeks post-completion of treatment.
Global scoring9 at the end of treatment.
Safety assessed by adverse drug reactions (ADRs) monitoring and liver function tests (LFTs) at baseline and end of treatment.
Secondary outcomes
Dermatology Life Quality Index (DLQI) at baseline, end of treatment, and six weeks post-completion of treatment.
Treatment adherence by pill count method at end of treatment.
Number of patients requiring escape treatment.
Clinical cure was assessed using a Four-Point Scale7 evaluating itching/pruritus, erythema, and scaling, with each parameter graded from 0 (none) to 3 (severe). Treatment efficacy was primarily defined as clinical cure, indicated by complete resolution (Grade 0) of erythema and scaling, and either complete or mild residual itching (Grade 0 or 1). A composite score7 was also calculated to quantify overall improvement: Grade I (1–25%), Grade II (26–50%), Grade III (51–75%), and Grade IV (76–100%). Patients with less than 25% improvement (Grade I) were classified as treatment failures, while Grade IV was considered a clinical cure. Grades II and III were categorized as partial responders.
Direct microscopic examination using a 10% potassium hydroxide (KOH) wet mount6 was performed to confirm dermatophyte infection. At end of treatment, global scoring9 was accomplished by combining clinical cure (resolution of erythema, scaling, and itching) with mycological cure (negative KOH), providing a comprehensive measure of treatment efficacy through both symptom relief and fungal eradication.
ADRs were recorded throughout study using the Central Drugs Standard Control Organisation (CDSCO) Suspected Adverse Reaction Reporting Form through both spontaneous and solicited methods. Severity was graded using the Division of AIDS (DAIDS) table for Grading the Severity of Adult and Pediatric Adverse Events, Version 2.1 (Grade 1 to 5), and causality was assessed using the World Health Organization-Uppsala Monitoring Centre (WHO-UMC) Scale. All ADRs were uploaded to the WHO-UMC database via VigiFlow.
LFTs were conducted at baseline and treatment end to monitor for hepatotoxicity, a known risk with terbinafine and itraconazole therapy.
Quality of life was assessed using DLQI9 questionnaire at baseline, end of treatment, and six weeks post-treatment across all groups, with a total score ranging from 0 to 30. Higher scores indicate greater impairment. The questionnaire was provided in the patient's vernacular language, and official permission for its use was obtained.
Adherence was objectively evaluated using pill count at the end of treatment. The adherence ratio was calculated as:
Pill Count 10 = (Pills dispensed − Pills remaining)/(Prescribed pills/day × Days between visits)
A ratio of 1 indicates perfect adherence; values below 1 suggest underuse, while values above 1 may reflect overuse or counting errors.
Sample size
Assuming a cure rate of 50% with standard therapy and an expected improvement to 80% with increased dose or duration, the calculated sample size was 38 per group. Considering a 10% loss to follow-up, the final sample size was adjusted to 42 participants per group, i. e. 168 subjects across four groups.
Statistical analysis
Data were compiled using Microsoft Excel. Descriptive results were expressed as mean±standard deviation (±SD). Paired t-test was used for within-group comparisons of normally distributed continuous variables (Four-Point Scale scores, DLQI, LFTs). Kruskal-Wallis test assessed differences in non-normally distributed clinical parameters across treatment groups. Chi-square test evaluated categorical variables including mycological cure, demographics, and ADRs. One-way ANOVA was used for intergroup comparisons of composite scores and global outcomes. A p-value<0.05 was considered statistically significant.
RESULTS
Patient enrolment
176 patients were screened, with 168 meeting the inclusion criteria and 2 lost to follow-up. Patients were randomized into four groups:
Group A: Terbinafine 250 mg Once Daily (OD) for 4 w (n=42)
Group B: Terbinafine 500 mg Once Daily (OD) for 2 w (n=41)
Group C: Itraconazole 100 mg Twice Daily (BD) for 4 w (n=41)
Group D: Itraconazole 200 mg Twice Daily (BD) for 2 w (n=42)
Topical antifungal-Luliconazole was advised to all the patients in each group.
The study included 166 patients (72 males, 94 females; mean age 37.2±12.4 y, range 18–65), with most being married (77.1%), literate (91.6%), and from rural areas (56%). No significant demographic differences were found among groups (p>0.05). While Group A had the most mixed infections (42.9%), Group D had the highest isolated tinea corporis (47.6%) and Group C the most tinea cruris (43.9%), these differences were not statistically significant (p = 0.111).
Itching severity improved significantly in all groups from baseline to end of treatment and at 6-week follow-up (p<0.001). Initially, all patients had moderate to severe itching. By treatment end, most improved to none/mild—highest in Group C (95.12%), with complete relief in 21.95% (p = 0.0003). At 6 w post-treatment, itching was absent in 35.7% (A), 24.4% (B), 29.3% (C), and 19% (D), with no severe cases. Recurrence of moderate itching was lowest in Group C (12.2%) and highest in Group D (31%). Intergroup differences were not significant (p>0.05), but Group C showed the best long-term control. Fig. 1 illustrates itching grades across time points.
Erythema significantly improved in all groups from baseline to end of treatment and at 6-week follow-up (p<0.001). At baseline,>92% in each group had mild to severe erythema. Complete resolution at treatment end was highest in Group C (100%, p = 0.0037), followed by Groups A and D (85.7%), and B (75.6%). At 6 w post-treatment, erythema was absent in 95.1% (C), 85.7% (A), 80.5% (B), and 69% (D), with mild recurrence lowest in Group C (4.9%) and highest in D (28.6%). No severe cases recurred. While baseline intergroup differences were significant (p = 0.028), they were not at treatment end or follow-up, indicating all regimens were effective, with Group C showing the most sustained benefit. Fig. 2 illustrates erythema grades across time points.
Scaling severity improved significantly in all groups from baseline to treatment end and 6-week follow-up (p<0.001). At baseline, nearly all patients had mild to severe scaling. By treatment end, resolution was highest in Group C (100%), followed by D (95.2%), A (90.5%), and B (82.9%). At 6 w post-treatment, absence of scaling persisted in Group C (100%), with mild recurrence in A (4.8%), B (12.2%), and D (7.1%); no moderate/severe recurrence occurred. While baseline intergroup differences were significant (p = 0.024), they were not at treatment end (p = 0.583) or follow-up (p = 0.814), confirming comparable efficacy. Group C (Itraconazole 100 mg BD for 4 w) showed the most sustained improvement (p = 0.028). Fig. 3 illustrates scaling grades across time points.
Clinical improvement, based on Sharma et al.7, criteria, showed no patients in Grade I. Partial responders (Grades II and III) were seen in 44 patients, while 122 (73.5%) achieved complete response (Grade IV), with Group C having the highest (92.7%, p = 0.0013). Intragroup analysis showed significant improvement in all groups (p<0.001), while intergroup comparison revealed significant differences (p = 0.0106), with Group C (Itraconazole 100 mg BD for 4 w) being most effective (p = 0.0068 vs. Group A). No significant difference was noted between Groups B and D (p = 0.9347).
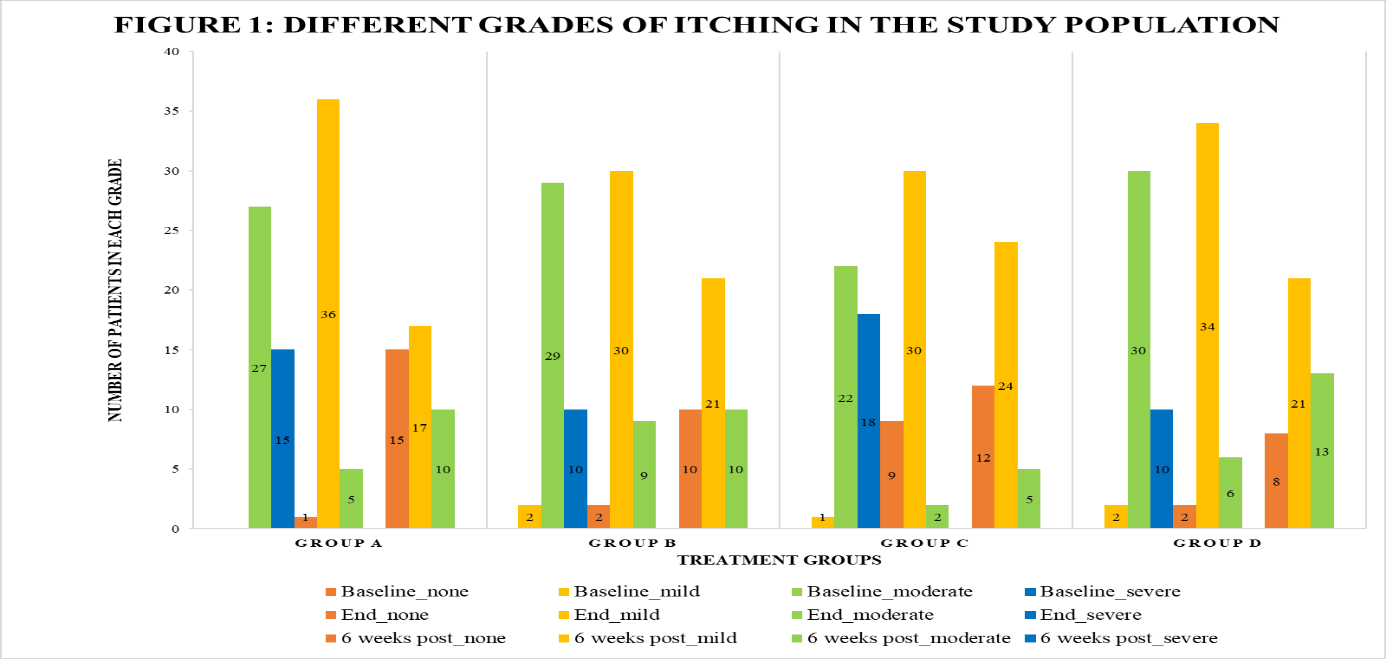
Fig. 1: Different grades of itching in the study population
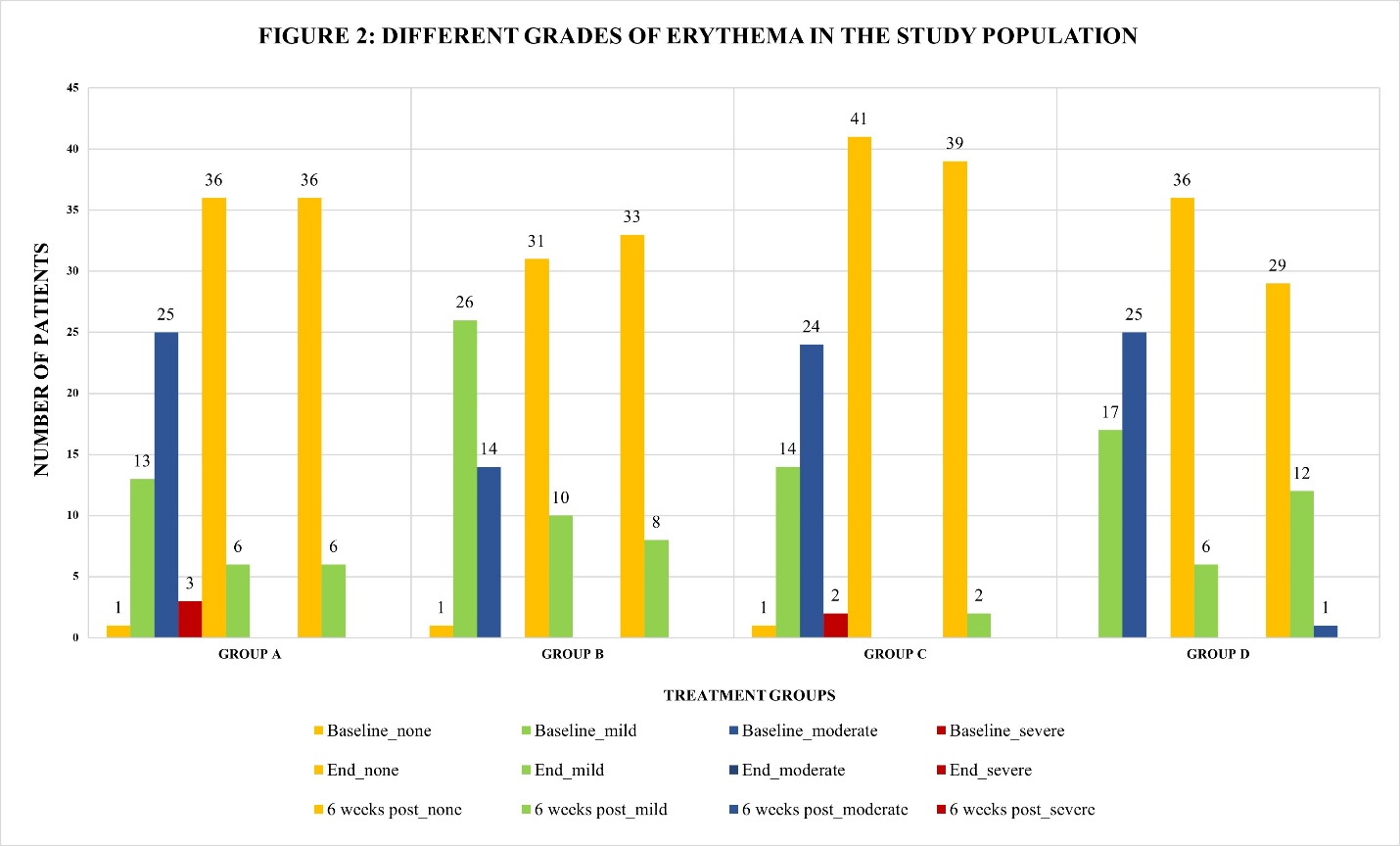
Fig. 2: Different grades of erythema in the study population
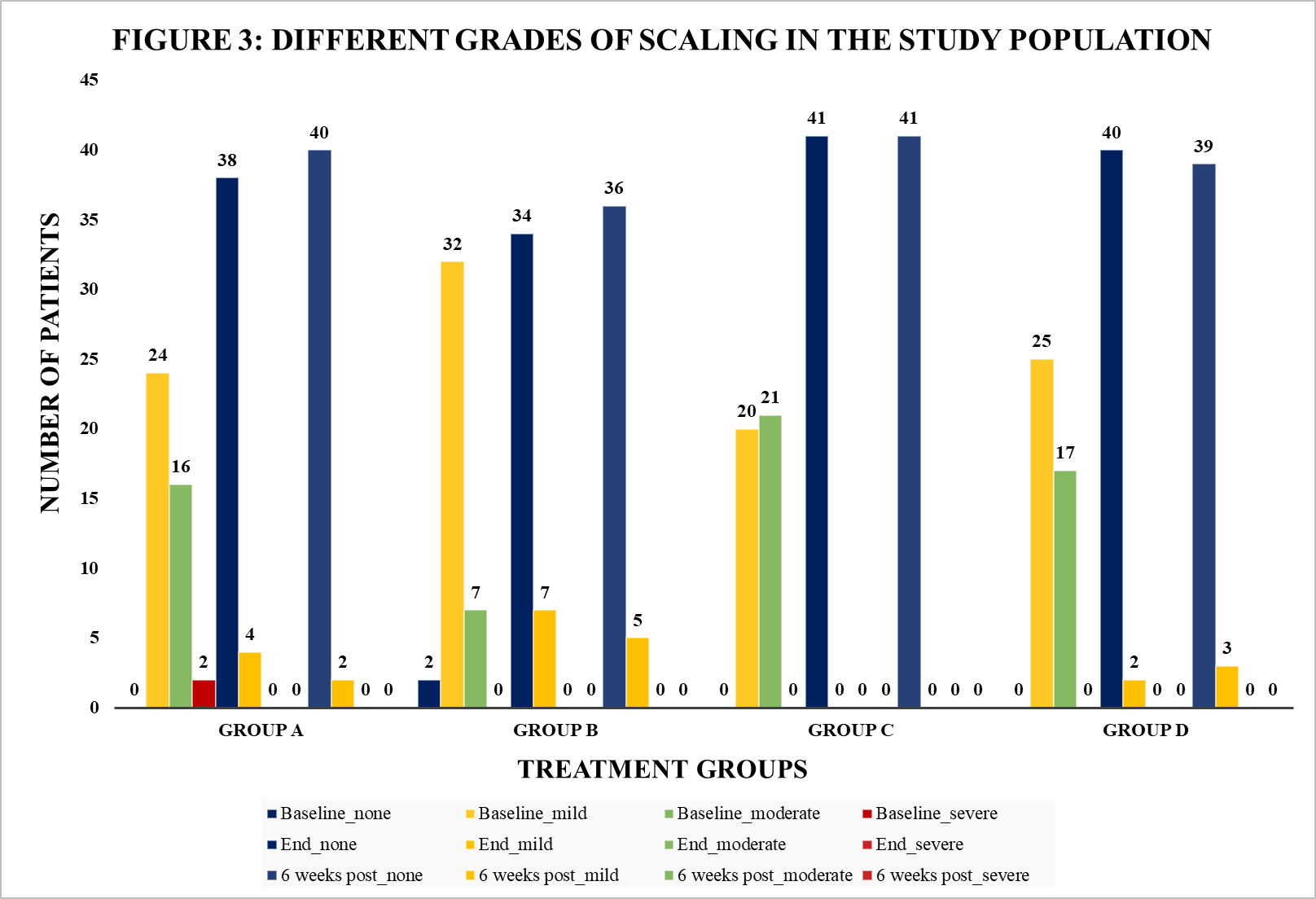
Fig. 3: Different grades of scaling in the study population
All groups showed significant mycological improvement (p<0.001) from baseline to end of treatment and at 6 w post-treatment. Group C (Itraconazole 100 mg BD, 4 w) had the highest KOH negativity (100%), followed by Group A (97.62%), Group D (88.10%), and Group B (87.80%), with Group B significantly lower than Group D (p = 0.038). At 6 w, high cure rates persisted across all groups (p = 0.361), with Group C leading (97.6%), though differences were not statistically significant. Itraconazole 100 mg BD for 4 w showed the most rapid and effective clearance.
Global scoring9 showed significant efficacy differences (p<0.001), with itraconazole 100 mg BD for 4 w (Group C) being the most effective, achieving 100% KOH negativity and 92.7% Grade IV improvement at the end of treatment, as shown in table 1. Group D also had 100% KOH negativity but a lower Grade IV response (66.7%). Groups A and B had 97.6% and 90.2% KOH negativity, respectively, with fewer patients reaching Grade IV (73.2% and 70.3%, p = 0.042). These findings confirm itraconazole 100 mg BD for 4 w as the most effective regimen.
Table 1: Global scoring9 AT end OF treatment in study population
| Group | KOH | Variables | Composite score (GRADE) | |||
| I | II | III | IV | |||
| A | Negative | % within Group | 0% | 07.14% | 19.04% | 71.42% |
| Positive | % within Group | 0% | 02.40% | 0% | 0% | |
| B | Negative | % within Group | 0% | 02.43% | 24.40% | 63.41% |
| Positive | % within Group | 0% | 07.31% | 02.40% | 0.0% | |
| C | Negative | % within Group | 0% | 0% | 07.31% | 92.70% |
| Positive | % within Group | 0% | 0% | 0% | 0% | |
| D | Negative | % within Group | 0% | 14.30% | 19.04% | 66.67% |
| Positive | % within Group | 0% | 0% | 0% | 0% | |
All groups were monitored for adverse effects, with mild nausea and diarrhoea being most common. Group A had 3 cases of nausea, 5 of diarrhoea; Group B: 1 and 4; Group C: 3 and 5; Group D: 2 and 2, respectively. No other adverse events occurred. WHO-UMC causality assessment deemed these possibly treatment-related but not requiring drug withdrawal. Adverse reaction rates were similar across groups (p = 0.456), indicating all regimens were well tolerated.
Mild Alkaline Phosphatase (ALP) elevation was noted in Group C (Itraconazole 100 mg BD, 4 w), with significant intra-group changes in Aspartate Aminotransferase (AST), Alanine Aminotransferase (ALT), and bilirubin, but only ALP showed significant inter-group difference (p<0.001). No clinically relevant LFT abnormalities or escape treatments were observed, indicating all regimens were effective, safe, and well-tolerated without treatment failures or serious adverse effects.
At baseline, 81.3% of patients reported a very large quality-of-life impact, highest in Groups D (88.1%) and A (85.7%). By treatment end, 69.3% had a small impact and 10.8% no impact, notably Group C (22%). At 6 w post-treatment, 45.2% reported no impact, led by Group C (61%). All groups showed significant improvement (p<0.05), with Group C achieving the greatest and most sustained quality-of-life gains.
DISCUSSION
Dermatophytosis has surged in India over the past decade, with rising resistance to terbinafine and itraconazole. Recurrent infections, defined as lesion recurrence within six weeks post-treatment, are common due to suboptimal treatment, environmental factors, and host susceptibility. Management remains empirical, lacking standardized, evidence-based guidelines.
This study compared different regimens of terbinafine and itraconazole in recurrent tinea corporis and tinea cruris patients, assessing efficacy and safety. Among 166 participants, the mean age was 37.20±12.38 y, aligning with studies by Pathania et al. [5], (mean age 32.5 y) and Singh et al. [11], (mean age 28.6±12.0 y). Unlike previous reports suggesting a male predominance (Sharma et al. [12], reported 64.58% males), this study had a higher female representation (56.62%), consistent with Hassaan et al. [13], (82.2% female).
Clinical patterns were comparable to Tigga et al. [14], where tinea corporis et cruris was most common (53.1%).
Efficacy was assessed via a four-point scale7, composite7, KOH mount6, and global scoring8. Itraconazole 100 mg BD for 4 w showed the highest clinical cure (92.7%) and lowest recurrence (12.2%). Singh et al.[15], found itraconazole superior to terbinafine over 8 w. Mycological cure was highest (100%) in Groups C and D (Itraconazole regimens), with minimal recurrence in Group C. Urmila et al.[16],reported 77.33% KOH positivity, with Trichophyton mentagrophytes as the most common isolate (66.66%). Global Scoring8 confirmed that prolonged itraconazole therapy gave the most sustained cure, aligning with findings bySharma et al. [17]
Safety analysis revealed that diarrhoea was the most common adverse event (9.6% of patients), consistent with Hassaan et al. [18], who observed gastrointestinal disturbances in all treatment groups. Liver function monitoring showed no significant concerns.
DLQI scores significantly improved across all groups, dropping from a mean of 12.13 ± 2.20 at baseline to 2.9 ± 3.27 post-treatment. Adherence was highest in shorter-duration regimens, though Tsunemi et al. [19], reported poor adherence in tinea pedis patients using Morisky Medication Adherence Scale-8 (MMAS-8).
This study underscores the importance of extended-duration itraconazole therapy in recurrent dermatophytosis, particularly given the rising terbinafine resistance.
LIMITATION
This being an academic trial with limited resources like time, no external funding and manpower. The study's sample size, comprising 166 patients, may not fully represent the broader population affected by recurrent dermatophytosis.
CONCLUSION
All treatment regimens were effective and safe for recurrent tinea corporis and cruris; however, doubling the duration of Itraconazole 100 mg twice daily to 4 w resulted in superior clinical and mycological cure rates with minimal recurrence, supporting its use as the optimal treatment strategy.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
CONFLICT OF INTERESTS
Declared none
REFERENCES
Khan SS, Hay R, Saunte DM. An international survey of recalcitrant and recurrent tinea of the glabrous skin-a potential indicator of antifungal resistance. J Eur Acad Dermatol Venereol. 2024;39(6):1185-91. doi: 10.1111/jdv.20146, PMID 39001674.
Bongomin F, Gago S, Oladele RO, Denning DW. Global and multi-national prevalence of fungal diseases-estimate precision. J Fungi (Basel). 2017;3(4):57. doi: 10.3390/jof3040057, PMID 29371573.
Verma SB, Panda S, Nenoff P, Singal A, Rudramurthy SM, Uhrlass S. The unprecedented epidemic-like scenario of dermatophytosis in India: I. epidemiology, risk factors and clinical features. Indian J Dermatol Venereol Leprol. 2021 Mar 23;87(2):154-75. doi: 10.25259/IJDVL_301_20, PMID 33769736.
Kaur R, Panda PS, Sardana K, Khan S. Mycological pattern of dermatomycoses in a tertiary care hospital. J Trop Med. 2015;2015(1):157828. doi: 10.1155/2015/157828, PMID 26491453.
Pathania S, Rudramurthy SM, Narang T, Saikia UN, Dogra S. A prospective study of the epidemiological and clinical patterns of recurrent dermatophytosis at a tertiary care hospital in India. Indian J Dermatol Venereol Leprol. 2018;84(6):678-84. doi: 10.4103/ijdvl.IJDVL_645_17, PMID 30058568.
Rajagopalan M, Inamadar A, Mittal A, Miskeen AK, Srinivas CR, Sardana K. Expert consensus on the management of dermatophytosis in India (Ectoderm India). BMC Dermatol. 2018;18(1):6. doi: 10.1186/s12895-018-0073-1, PMID 30041646.
Sharma P, Bhalla M, Thami GP, Chander J. Evaluation of efficacy and safety of oral terbinafine and itraconazole combination therapy in the management of dermatophytosis. J Dermatolog Treat. 2020;31(7):749-53. doi: 10.1080/09546634.2019.1612835, PMID 31030594.
Pereda J, Noguera X, Boncompte E, Alguero M, Izquierdo I. Efficacy of flutrimazole 1% powder in the treatment of tinea pedis. Mycoses. 2003;46(3-4):126-31. doi: 10.1046/j.1439-0507.2003.00849.x, PMID 12870201.
Finlay AY, Khan GK. Dermatology Life Quality Index (DLQI) a simple, practical measure for routine clinical use. Clin Exp Dermatol. 1994;19(3):210-6. doi: 10.1111/j.1365-2230.1994.tb01167.x, PMID 8033378.
Lam WY, Fresco P. Medication adherence measures: an overview. BioMed Res Int. 2015 Oct 11;2015:217047. doi: 10.1155/2015/217047, PMID 26539470.
Singh S, Verma P, Chandra U, Tiwary NK. Risk factors for chronic and chronic-relapsing tinea corporis tinea cruris and tinea faciei: results of a case-control study. Indian J Dermatol Venereol Leprol. 2019 Mar 1;85(2):197-200. doi: 10.4103/ijdvl.IJDVL_807_17, PMID 30719987.
Sharma R, Adhikari L, Sharma RL. Recurrent dermatophytosis: a rising problem in Sikkim, a Himalayan state of India. Indian J Pathol Microbiol. 2017;60(4):541-5. doi: 10.4103/IJPM.IJPM_831_16, PMID 29323069.
Hassaan ZR, Mohamed HA, Eldahshan RM, Elsaie ML. Comparison between the efficacy of terbinafine and itraconazole orally vs. the combination of the two drugs in treating recalcitrant dermatophytosis. Sci Rep. 2023;13(1):19037. doi: 10.1038/s41598-023-46361-z, PMID 37923859.
Tigga RA, Das S, Bhattacharya SN, Saha R, Pandhi D, Datt S. Burden of chronic dermatophytosis in a tertiary care hospital: interaction of fungal virulence and host immunity. Mycopathologia. 2018;183(6):951-9. doi: 10.1007/s11046-018-0303-4, PMID 30386967.
Singh SK, Subba N, Tilak R. Efficacy of terbinafine and itraconazole in different doses and in combination in the treatment of tinea infection: a randomized controlled parallel group open labeled trial with clinico-mycological correlation. Indian J Dermatol. 2020;65(4):284-9. doi: 10.4103/ijd.IJD_548_19, PMID 32831369.
Urmila Y, Gopal KV, Turpati NR, Karri SB, Raju PV. A clinico-mycological and histopathological study of recurrent dermatophytosis. Indian Dermatol Online J. 2023;14(6):799-806. doi: 10.4103/idoj.idoj_670_22, PMID 38099009.
Sharma P, Bhalla M, Thami GP, Chander J. Evaluation of efficacy and safety of oral terbinafine and itraconazole combination therapy in the management of dermatophytosis. J Dermatolog Treat. 2020;31(7):749-53. doi: 10.1080/09546634.2019.1612835, PMID 31030594.
Hassaan ZR, Mohamed HA, Eldahshan RM, Elsaie ML. Comparison between the efficacy of terbinafine and itraconazole orally vs. the combination of the two drugs in treating recalcitrant dermatophytosis. Sci Rep. 2023;13(1):19037. doi: 10.1038/s41598-023-46361-z, PMID 37923859.
Tsunemi Y, Abe S, Kobayashi M, Kitami Y, Onozuka D, Hagihara A. Adherence to oral and topical medication in 445 patients with tinea pedis as assessed by the morisky medication adherence scale-8. Eur J Dermatol. 2015;25(6):570-7. doi: 10.1684/ejd.2015.2650, PMID 26574949.