
Int J Curr Pharm Res, Vol 18, Issue 2, 82-86Original Article
COMPARATIVE EFFICACY OF SACUBITRIL/VALSARTAN VERSUS RENIN–ANGIOTENSIN SYSTEM INHIBITORS IN ADULT HEART FAILURE: A META-ANALYSIS OF RANDOMIZED CONTROLLED TRIALS
PRANAB DAS1*, BODHISATYA DAS2
1Department of Pharmacology, Pragjyotishpur Medical College and Hospital, Guwahati, Assam, India. 2Department of Paediatrics, Burdwan Medical College and Hospital, Burdwan, West Bengal, India
*Corresponding author: Pranab Das; *Email: pranabdas2580123@gmail.com
Received: 12 Nov 2025, Revised and Accepted: 05 Jan 2026
ABSTRACT
Objective: To evaluate the comparative effect of sacubitril/valsartan versus angiotensin-converting enzyme inhibitors or angiotensin receptor blockers on all-cause mortality in adults with heart failure using evidence from randomized controlled trials.
Methods: A systematic review and meta-analysis of randomized controlled trials was conducted in accordance with PRISMA 2020 guidelines. Electronic databases including PubMed, Scopus, Google Scholar, and the Cochrane Library were searched to identify eligible studies. Trials enrolling adults with heart failure and directly comparing sacubitril/valsartan with an ACE inhibitor or ARB were included. The primary outcome was all-cause mortality. Risk ratios with 95% confidence intervals were pooled using an inverse-variance fixed-effect model.
Results: Three randomized controlled trials comprising more than 18,000 participants met the inclusion criteria. Sacubitril/valsartan was associated with a statistically significant reduction in all-cause mortality compared with ACE inhibitors or ARBs (pooled RR = 0.89; 95% CI: 0.83–0.95). Statistical heterogeneity was low (I² = 12.7%). The observed mortality benefit was largely driven by trials enrolling patients with reduced ejection fraction.
Conclusion: This meta-analysis demonstrates that sacubitril/valsartan confers a modest but significant reduction in all-cause mortality compared with conventional renin–angiotensin system inhibitors in adults with heart failure. These findings support the preferential use of angiotensin receptor–neprilysin inhibition in appropriate patients, particularly those with reduced ejection fraction, while highlighting the need for further randomized evidence in other heart-failure phenotypes.
Keywords: Heart failure, Sacubitril/valsartan, Angiotensin-converting enzyme inhibitors, Angiotensin receptor blockers, All-cause mortality, Meta-analysis
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/)
DOI: https://dx.doi.org/10.22159/ijcpr.2026v18i2.8070 Journal homepage: https://innovareacademics.in/journals/index.php/ijcpr
INTRODUCTION
Cardiovascular diseases remain the leading cause of mortality worldwide, accounting for a substantial proportion of global deaths and disability, with heart failure representing a major and growing contributor to this burden [1]. Heart failure affects millions of individuals globally and is associated with high rates of hospitalization, impaired quality of life, and premature mortality despite advances in contemporary medical therapy [2]. The rising prevalence of heart failure is driven by population ageing and improved survival from acute cardiovascular conditions, resulting in an expanding population living with chronic disease [1, 3].
Pharmacological modulation of neurohormonal pathways has long formed the cornerstone of heart failure management, particularly through inhibition of the renin–angiotensin system using angiotensin-converting enzyme inhibitors (ACE inhibitors) or angiotensin receptor blockers (ARBs) [4, 5]. Robust evidence from randomized trials and systematic reviews has demonstrated that ACE inhibitors reduce mortality and morbidity in patients with heart failure with reduced ejection fraction, establishing them as first-line disease-modifying therapy [5, 6]. ARBs have been shown to provide an alternative in patients intolerant to ACE inhibitors, although their mortality benefit compared with ACE inhibitors has remained less consistent across studies [6].
Sacubitril/valsartan, an angiotensin receptor–neprilysin inhibitor, was developed to enhance neurohormonal modulation by combining neprilysin inhibition with angiotensin II receptor blockade, thereby augmenting natriuretic peptide activity while suppressing maladaptive renin–angiotensin signaling [7]. Large randomized controlled trials have evaluated sacubitril/valsartan across diverse clinical settings, including heart failure with reduced ejection fraction, heart failure with preserved ejection fraction, and post–myocardial infarction populations [8–10]. While some trials demonstrated significant reductions in mortality and morbidity compared with conventional renin–angiotensin system inhibition, others reported neutral effects, particularly in populations without reduced ejection fraction [9, 10].
All-cause mortality represents the most objective and clinically meaningful endpoint in cardiovascular outcomes research, as it avoids misclassification and adjudication bias inherent in cause-specific mortality outcomes [6, 11]. Despite the widespread adoption of sacubitril/valsartan in clinical practice and guideline recommendations supporting its use in selected patient populations, uncertainty remains regarding its overall effect on all-cause mortality when compared directly with ACE inhibitors or ARBs across the spectrum of adult heart failure [7, 11, 12]. Prior reviews have often combined heterogeneous study designs, included observational data, or focused on composite outcomes rather than mortality alone, limiting definitive conclusions [12–14].
Given the central role of mortality reduction in guiding therapeutic decision-making and the availability of few yet large randomized controlled trials directly comparing sacubitril/valsartan with renin–angiotensin system inhibitors, a focused synthesis of randomized evidence is warranted [8–10]. Clarifying the comparative effect of sacubitril/valsartan on all-cause mortality may help inform clinicians, guideline developers, and policymakers regarding its overall survival benefit relative to established therapies [2, 7].
Therefore, the objective of this meta-analysis was to evaluate whether sacubitril/valsartan reduces all-cause mortality compared with ACE inhibitors or angiotensin receptor blockers in adults with heart failure using evidence from randomized controlled trials.
MATERIALS AND METHODS
Research question
The present meta-analysis was conducted to address the following research question: Among adult patients with heart failure, does treatment with sacubitril/valsartan, compared with angiotensin-converting enzyme inhibitors or angiotensin receptor blockers, reduce all-cause mortality?
Study design and reporting standards
This study employed a quantitative meta-analytic approach using data derived exclusively from eligible published studies. The conduct and reporting of the meta-analysis were guided by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines to ensure methodological transparency, reproducibility, and scientific rigor [15]. As this analysis was based solely on previously published data and did not involve the collection of new data from human or animal subjects, formal ethical approval or institutional review board clearance was not required.
Eligibility criteria
Studies were considered eligible for inclusion if they were randomized controlled trials involving adult participants aged 18 years or older and enrolled patients diagnosed with heart failure, regardless of ejection fraction phenotype. Eligible trials were required to directly compare sacubitril/valsartan with an angiotensin-converting enzyme inhibitor or an angiotensin receptor blocker and to report outcomes related to all-cause mortality. Only studies published as full-text articles in peer-reviewed journals in English language were included to ensure methodological quality and data completeness. Studies were excluded if they were observational in design, non-randomized, or based on registry data. Trials involving paediatric populations were not considered. Additionally, studies were excluded if they did not include a direct comparator arm using an ACE inhibitor or ARB, or if sacubitril/valsartan was evaluated as part of combination therapy without the ability to isolate its independent effect. Conference abstracts, editorials, review articles, and studies lacking sufficient outcome data were also excluded. In cases of duplicate publications, the most comprehensive and recent report was retained for analysis.
Search strategy
After conducting a comprehensive search of electronic databases, the reviewers were able to discover research that met the eligibility criteria. Several databases, such as Google Scholar, Scopus, PubMed, and the Cochrane Library, were searched thoroughly by the reviewers.
Boolean operators along with the keywords, such as the following, were incorporated into the search method:
(“sacubitril/valsartan” or “angiotensin receptor–neprilysin inhibitor” or “arni ”and“ heart failure ”and“ randomized controlled trial” or “randomised trial ”and“ mortality” or “all-cause mortality”)
Search results were restricted to studies published in English and involving human participants. Reference lists of relevant articles and reviews were also manually screened to identify additional eligible studies.
Study selection process
All records retrieved from the literature search were initially screened based on titles and abstracts to exclude clearly irrelevant studies. Full-text articles were then assessed for eligibility according to the predefined inclusion and exclusion criteria. The study selection process was conducted systematically by two independent reviewers, and only trials meeting all eligibility requirements were included in the final analysis. Discrepancies during study selection were resolved through discussion and consensus by a third independent reviewer.
Data extraction process
Data were extracted from eligible studies using a standardized data collection approach. Extracted information included study characteristics, patient population, intervention and comparator details, dosage regimens, duration of follow-up, and the number of all-cause mortality events in each treatment group. Outcome data were extracted directly from the primary trial reports to ensure accuracy. When necessary, calculations were performed using raw event counts to derive effect estimates.
Statistical analysis
The primary outcome of interest was all-cause mortality. For each included trial, risk ratios (RRs) and corresponding standard errors were calculated using raw event counts from the intervention and comparator groups. Pooled effect estimates were generated using an inverse-variance fixed-effect model, assuming a common underlying treatment effect across studies. Results are presented as pooled RRs with 95% confidence intervals. Statistical heterogeneity was assessed using Cochran’s Q statistic and the I² statistic; however, heterogeneity estimates were interpreted cautiously due to the small number of included studies. Formal assessment of publication bias using funnel plots or statistical tests such as Egger’s regression was not performed because fewer than ten studies were included, rendering such methods unreliable and potentially misleading. Subgroup analyses and leave-one-out sensitivity analyses were not conducted as the heterogeneity was low, as only three randomized controlled trials met the inclusion criteria.
RESULTS
Study selection and sample characteristics
The systematic literature search identified multiple records relevant to the comparative efficacy of sacubitril/valsartan in heart failure. After removal of duplicate records, titles and abstracts were screened for relevance. Studies that were clearly observational, non-randomized, or did not include a direct comparison with angiotensin-converting enzyme inhibitors or angiotensin receptor blockers were excluded. Full-text articles were then assessed for eligibility, resulting in the inclusion of three randomized controlled trials that metal predefined criteria. These trials collectively enrolled over 18,000 adult participants with heart failure across diverse clinical settings, including heart failure with reduced ejection fraction, heart failure with preserved ejection fraction, and patients with recent myocardial infarction and left ventricular dysfunction. All included studies reported all-cause mortality and were published as full-text articles in peer-reviewed journals. A PRISMA flow diagram summarizing the study selection process is presented in table 1.
Table 1: PRISMA summary table
| Stage | Frequency (n) |
| Records identified through database searching | 114 |
| Duplicates removed | 68 |
| Records screened | 46 |
| Records excluded | 38 |
| Full-text articles assessed for eligibility | 8 |
| Full-text articles excluded | 5 |
| Studies included in qualitative and quantitative synthesis | 3 |
Study characteristics
Across all trials, sacubitril/valsartan was administered at guideline-recommended target doses and compared against standard-of-care renin–angiotensin system inhibitors. Follow-up durations ranged from 96 to 152 w, ensuring adequate capture of mortality outcomes. Details of study attributes which were included in this meta-analysis are depicted in table 2.
Meta-analysis findings
Using an inverse-variance fixed-effect model, sacubitril/valsartan was associated with a statistically significant reduction in all-cause mortality compared with ACE inhibitors or ARBs. The pooled risk ratio was 0.89, corresponding to a 11% relative reduction in mortality, with a 95% confidence interval ranging from 0.83 to 0.95 (statistically significant). The effect estimates of the meta-analysis are summarised below in table 3.
Table 2: Attributes of included randomized controlled trials (RCTs)
| Study ID | First Author (Year) | Study Design | Population | Intervention | Comparator | Intervention dose | Comparator dose | Primary outcome | Proportion of patients achieving target outcome (Intervention)* | Proportion of patients achieving target outcome (Comparator)** | Duration (weeks) |
| PARADIGM-HF | McMurray (2014) [8] | Double-blind RCT | Chronic HFrEF | Sacubitril/valsartan | Enalapril | 200 mg BID | 10 mg BID | All-cause mortality | 0.1698 | 0.1982 | 117 |
| PARAGON-HF | Solomon (2019) [9] | Double-blind RCT | HFpEF | Sacubitril/valsartan | Valsartan | 97/103 mg BID | 160 mg BID | All-cause mortality | 0.1421 | 0.1461 | 152 |
| PARADISE-MI | Pfeffer (2021) [10] | Double-blind RCT | Post-MI, EF ≤40% | Sacubitril/valsartan | Ramipril | 97/103 mg BID | 5 mg BID | All-cause mortality | 0.0753 | 0.0855 | 96 |
*Proportion of patients achieving target outcome in intervention group = Sample size of intervention group showing the desired outcome/Total sample size of intervention group. **Proportion of patients achieving target outcome in Comparator group = Sample size of comparator group showing the desired outcome/Total sample size of comparator group.
Table 3: Summary of effect estimates for included randomized controlled trials
| Study ID | RR | Log(RR) | SE | Lower 95% CI (Individual study) | Upper 95% CI (Individual study) | Weight | Weighted Log (RR) | Pooled RR | SE of pooled Log(RR) | Lower 95% CI (Meta-analysis) | Upper 95% CI (Meta-analysis) |
| PARADIGM-HF | 0.857 | −0.155 | 0.046 | 0.783 | 0.938 | 469.96 | −72.75 | 0.89 | 0.035 | 0.83 | 0.95 |
| PARAGON-HF | 0.973 | −0.028 | 0.070 | 0.847 | 1.117 | 201.81 | −5.60 | ||||
| PARADISE-MI | 0.880 | −0.127 | 0.090 | 0.738 | 1.051 | 123.15 | −15.68 |
RR: Risk Ratio; SE: Standard Error; CI: Confidence Interval.
Forest plot
A forest plot illustrating individual study effect estimates and the pooled fixed-effect result is presented in (fig. 1). The plot demonstrates that the overall effect favours sacubitril/valsartan, with the pooled estimate lying entirely to the left of the line of no effect (RR = 1.0). The PARADIGM-HF trial contributed the greatest statistical weight, while the remaining trials showed neutral but directionally consistent effects.
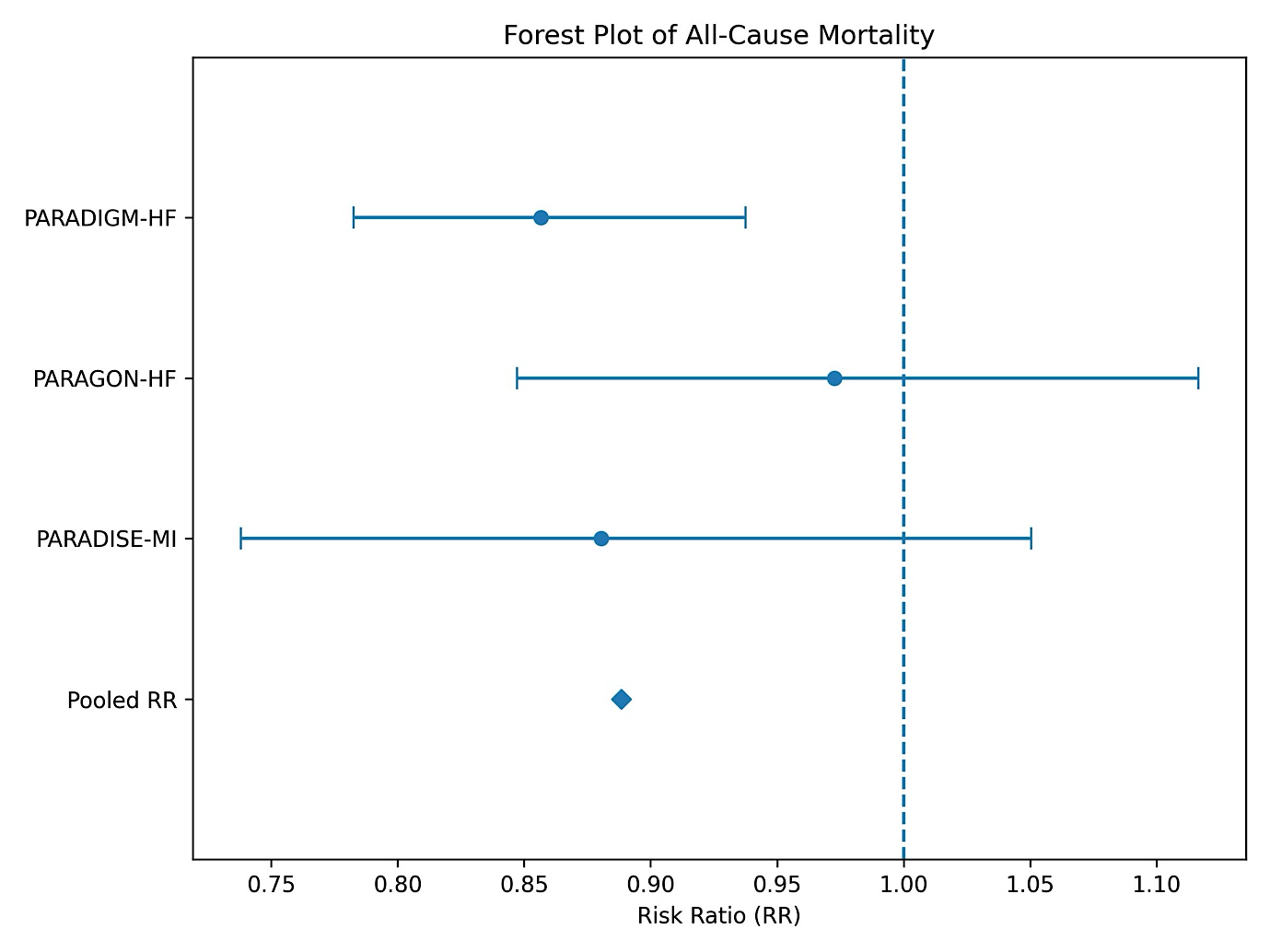
Fig. 1: Forest plot
Risk of bias assessment
All included trials demonstrated a low risk of bias across all domains, reflecting high methodological quality, appropriate randomization, adequate blinding, and complete outcome reporting. Table 4 shows the Risk of Bias (RoB) assessment of the meta-analysis as per the Cochrane RoB 2 guidance tool [16].
Heterogeneity assessment
Statistical heterogeneity was low, with an I² value of 12.7%, indicating minimal inconsistency across studies. However, given the inclusion of only three trials, heterogeneity estimates were interpreted cautiously. Table 5 shows Cochrane’s Q statistic and I2statistic calculation for the heterogeneity assessment of the meta-analysis.
Table 4: Risk of bias (RoB) assessment for included randomized controlled trials
| Study ID | Randomization process | Allocation concealment | Blinding of participants and personnel | Blinding of outcome assessment | Incomplete outcome data | Selective reporting | Overall risk of bias |
| PARADIGM-HF | Low | Low | Low | Low | Low | Low | Low |
| PARAGON-HF | Low | Low | Low | Low | Low | Low | Low |
| PARADISE-MI | Low | Low | Low | Low | Low | Low | Low |
Table 5: Heterogeneity assessment of included studies
| Study ID | ((Log RR-Pooled log RR)^2)*weight | Cochran's Q statistic | Degrees of freedom (df) | I2 statistic |
| PARADIGM-HF | 0.627 | 2.29 | 2 | 12.7% |
| PARAGON-HF | 1.654 | |||
| PARADISE-MI | 0.010 |
DISCUSSION
In this fixed-effect meta-analysis of randomized controlled trials, treatment with sacubitril/valsartan was associated with a statistically significant reduction in all-cause mortality compared with angiotensin-converting enzyme inhibitors or angiotensin receptor blockers among adults with heart failure. The pooled risk ratio indicated an approximate 11% relative reduction in mortality, underscoring a clinically meaningful survival benefit with angiotensin receptor–neprilysin inhibition in selected heart-failure populations [17].
The observed mortality benefit is largely consistent with prior systematic reviews and meta-analyses that have demonstrated the superiority of sacubitril/valsartan over conventional renin–angiotensin system inhibition, particularly in patients with heart failure and reduced ejection fraction [17, 18]. Earlier pooled analysis have reported reductions in cardiovascular mortality and heart-failure hospitalization with angiotensin receptor–neprilysin inhibitors, although the magnitude of benefit has varied depending on study design, population characteristics, and outcome definitions [17]. By focusing exclusively on randomized controlled trials and all-cause mortality, the present analysis minimizes bias associated with composite endpoints and observational data.
The findings of this meta-analysis are strongly influenced by evidence from the PARADIGM-HF trial, which demonstrated a significant reduction in all-cause and cardiovascular mortality with sacubitril/valsartan compared with enalapril in patients with chronic heart failure and reduced ejection fraction [8]. In contrast, trials enrolling patients with preserved ejection fraction or those with recent myocardial infarction have reported more neutral effects on mortality, highlighting the heterogeneity of treatment response across heart-failure phenotypes [9, 10]. This differential effect likely reflects underlying pathophysiological differences, with neurohormonal modulation playing a more central role in systolic heart failure than in preserved ejection fraction syndromes.
Real-world observational studies have further supported the mortality benefit of sacubitril/valsartan when compared with ACE inhibitor or ARB therapy in routine clinical practice, reporting reductions in all-cause mortality ranging from 10% to 25% [18, 19]. Although such studies are subject to residual confounding, their consistency with randomized evidence strengthens the external validity of the present findings and suggests that the benefits of sacubitril/valsartan extend beyond highly selected trial populations [18]. Importantly, real-world data have also highlighted disparities in treatment response across demographic subgroups, emphasizing the need for individualized therapeutic decision-making [19].
The biological plausibility of the observed mortality reduction is supported by the dual mechanism of sacubitril/valsartan, which simultaneously suppresses the deleterious effects of the renin–angiotensin system while enhancing natriuretic peptide activity [20]. This combined neurohormonal modulation has been shown to improve ventricular remodelling, reduce myocardial stress, and favourably influence hemodynamic and metabolic pathways implicated in heart-failure progression [21]. Such mechanisms may explain the sustained survival benefit observed in long-term follow-up of patients with reduced ejection fraction.
This meta-analysis has several important strengths. First, it is restricted exclusively to randomized controlled trials, thereby minimizing confounding and selection bias and strengthening the internal validity of the pooled estimates. Second, the analysis focuses solely on all-cause mortality, a hard and objective clinical endpoint that avoids misclassification and adjudication bias associated with cause-specific or composite outcomes. Third, all included trials were large, multicenter studies with rigorous methodology, standardized outcome reporting, and adequate follow-up duration, enhancing the reliability of the findings.
In addition, the use of a fixed-effect inverse-variance model is appropriate given the limited number of included studies and the low observed statistical heterogeneity, allowing for precise estimation of the pooled effect. Data extraction was based on raw event counts, ensuring transparency and reproducibility of calculations. Finally, by avoiding inclusion of observational studies and short-term surrogate endpoints, this meta-analysis provides a focused and clinically meaningful synthesis of the highest level of available evidence.
Despite these strengths, several limitations merit consideration. First, only three randomized controlled trials met the inclusion criteria, limiting the statistical power to explore heterogeneity, conduct subgroup analysis, or formally assess publication bias. Second, the pooled effect estimate was predominantly driven by a single large trial, and the mortality benefit may not be uniformly applicable across all heart-failure phenotypes. Third, differences in trial populations, background therapy, and follow-up duration may have contributed to variability in individual study effects, although overall heterogeneity was low. Fourth, Microsoft Excel was used to perform the meta-analytic calculations; it has inherent limitations when compared with dedicated meta-analysis software. Excel lacks built-in functions for advanced meta-analytic modelling, automated error checking, and reproducible workflows, increasing the potential for manual calculation or referencing errors. Nevertheless, transparent use of raw event data and standardized formulas allowed accurate estimation of effect sizes in the present analysis.
CONCLUSION
In summary, this meta-analysis provides robust randomized evidence supporting a modest but significant reduction in all-cause mortality with sacubitril/valsartan compared with ACE inhibitors or ARBs in adults with heart failure. The findings reinforce current guideline recommendations favouring angiotensin receptor–neprilysin inhibition in appropriate patients, while also highlighting the need for further randomized studies to clarify its role in populations beyond heart failure with reduced ejection fraction. At a broader level, the results may inform health policy and formulary decisions by providing randomized evidence of a survival advantage with sacubitril/valsartan. Future research should aim to clarify its role in populations with preserved ejection fraction and in post–myocardial infarction settings, as well as to explore long-term outcomes and cost-effectiveness in real-world practice.
ACKNOWLEDGEMENT
The authors sincerely acknowledge the contributions of all investigators whose original studies informed this meta-analysis and appreciate the valuable comments and suggestions provided by the peer reviewers.
FUNDING
This study did not receive any external financial support from public, commercial, or non-profit funding agencies.
AUTHORS CONTRIBUTIONS
Pranab Das conceptualized and designed the study, performed the literature search, extracted and analysed the data, and drafted the manuscript. Bodhisatya Das assisted with data verification, interpretation of results, and critical revision of the manuscript. Both authors reviewed and approved the final version and accept full responsibility for the accuracy and integrity of the work.
CONFLICT OF INTERESTS
The authors declare no conflicts of interest.
REFERENCES
World Health Organization. Cardiovascular diseases (CVDs). Geneva: WHO. Available from: https://www.who.int/health-topics/cardiovascular-diseases. [Last accessed on 23 Feb 2026].
Sindone AP, De Pasquale C, Amerena J, Burdeniuk C, Chan A, Coats A. Consensus statement on the current pharmacological prevention and management of heart failure. Med J Aust. 2022;217(4):212-7. doi: 10.5694/mja2.51656, PMID 35908234.
Abdin A, Schulz M, Riemer U, Haderi B, Wachter R, Laufs U. Sacubitril/valsartan in heart failure: efficacy and safety in and outside clinical trials. ESC Heart Fail. 2022;9(6):3737-50. doi: 10.1002/ehf2.14097, PMID 35921043.
Heran BS, Musini VM, Bassett K, Taylor RS, Wright JM. Angiotensin receptor blockers for heart failure. Cochrane Database Syst Rev. 2012;2012(4):CD003040. doi: 10.1002/14651858.CD003040.pub2, PMID 22513909.
Sohani ZN, Behlouli H, De Moura CS, Abrahamowicz M, Pilote L. Sex differences in the effectiveness of angiotensin-converting enzyme inhibitors angiotensin II receptor blockers and sacubitril-valsartan for the treatment of heart failure. J Am Heart Assoc. 2023;12(14):e028865. doi: 10.1161/JAHA.122.028865, PMID 37421275.
Zhang H, Huang T, Shen W, Xu X, Yang P, Zhu D. Efficacy and safety of sacubitril-valsartan in heart failure: a meta-analysis of randomized controlled trials. ESC Heart Fail. 2020;7(6):3841-50. doi: 10.1002/ehf2.12974, PMID 32977362.
McMurray JJ, Packer M, Desai AS, Gong J, Lefkowitz MP, Rizkala AR. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med. 2014;371(11):993-1004. doi: 10.1056/NEJMoa1409077, PMID 25176015.
Solomon SD, McMurray JJ, Anand IS, Ge J, Lam CS, Maggioni AP. Angiotensin-neprilysin inhibition in heart failure with preserved ejection fraction. N Engl J Med. 2019;381(17):1609-20. doi: 10.1056/NEJMoa1908655, PMID 31475794.
Pfeffer MA, Claggett B, Lewis EF, Granger CB, Kober L, Maggioni AP. Angiotensin receptor-neprilysin inhibition in acute myocardial infarction. N Engl J Med. 2021 Nov 11;385(20):1845-55. doi: 10.1056/NEJMoa2104508, PMID 34758252.
Ambrosy AP, Butler J, Gheorghiade M. Clinical trials in acute heart failure: beginning of the end or end of the beginning? Eur J Heart Fail. 2017;19(11):1358-60. doi: 10.1002/ejhf.925, PMID 28656635.
Gao J, Zhang X, Xu M, Deng S, Chen X. The efficacy and safety of sacubitril/valsartan compared with ACEI/ARB in the treatment of heart failure following acute myocardial infarction: a systematic review and meta-analysis of randomized controlled trials. Front Pharmacol. 2023;14:1237210. doi: 10.3389/fphar.2023.1237210, PMID 37601056.
Luedde M, Agewall S, Ambrosio G, Bayes Genis A, Borghi C, Cerbai E. European journal of heart failure consensus statement heart failure pharmacotherapy for patients with heart failure with reduced ejection fraction and concomitant atrial fibrillation: review of evidence and call to action. Eur J Heart Fail. 2025 Nov;27(11):2198-210. doi: 10.1002/ejhf.70069, PMID 41123042.
Cleland JG. Contemporary management of heart failure in clinical practice. Heart. 2002;88(Suppl 2):ii5-8. doi: 10.1136/heart.88.suppl_2.ii5, PMID 12213792.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021 Mar 29;372:n71. doi: 10.1136/bmj.n71, PMID 33782057.
Sterne JA, Higgins JP, Elbers RG, Reeves BC. Risk of bias 2: a tool to assess risk of bias in randomised trials. Version 22. In: Cochrane; Aug 2019. Available from: https://www.riskofbias.info/welcome/rob-2-0-tool/current-version-of-rob-2. [Last accessed on 23 Feb 2026].
Virk GS, Hameed B, Baloch Z, Mahmood A, Zubair H, Aravind Rongali DA. Comparative efficacy of angiotensin receptor-neprilysin inhibitors (arnis) vs. ace inhibitors in heart failure systemic review and meta analysis. Cardiol Cardio Vasc Med. 2025;9(3):177-86. doi: 10.26502/fccm.92920438.
Tan NY, Sangaralingham LR, Sangaralingham SJ, Yao X, Shah ND, Dunlay SM. Comparative effectiveness of sacubitril-valsartan versus ACE/ARB therapy in heart failure with reduced ejection fraction. JACC Heart Fail. 2020;8(1):43-54. doi: 10.1016/j.jchf.2019.08.003, PMID 31838035.
Fu M, Pivodic A, Kack O, Costa Scharplatz M, Dahlstrom U, Lund LH. Real-world comparative effectiveness of ARNI versus ACEi/ARB in HF with reduced or mildly reduced ejection fraction. Clin Res Cardiol. 2023;112(1):167-74. doi: 10.1007/s00392-022-02124-w, PMID 36443599.
She J, Lou B, Liu H, Zhou B, Jiang GT, Luo Y. ARNI versus ACEI/ARB in reducing cardiovascular outcomes after myocardial infarction. ESC Heart Fail. 2021;8(6):4607-16. doi: 10.1002/ehf2.13644, PMID 34664407.
Jering KS, Claggett B, Pfeffer MA, Granger C, Kober L, Lewis EF. Prospective arni vs. ace inhibitor trial to determine superiority in reducing heart failure events after myocardial infarction (paradise-mi): design and baseline characteristics. Eur J Heart Fail. 2021;23(6):1040-8. doi: 10.1002/ejhf.2191, PMID 33847047.